Document
Quanterix Releases Financial Results for the First Quarter of 2026
Reports $36.4 million in revenue and approximately $103 million of cash and marketable securities
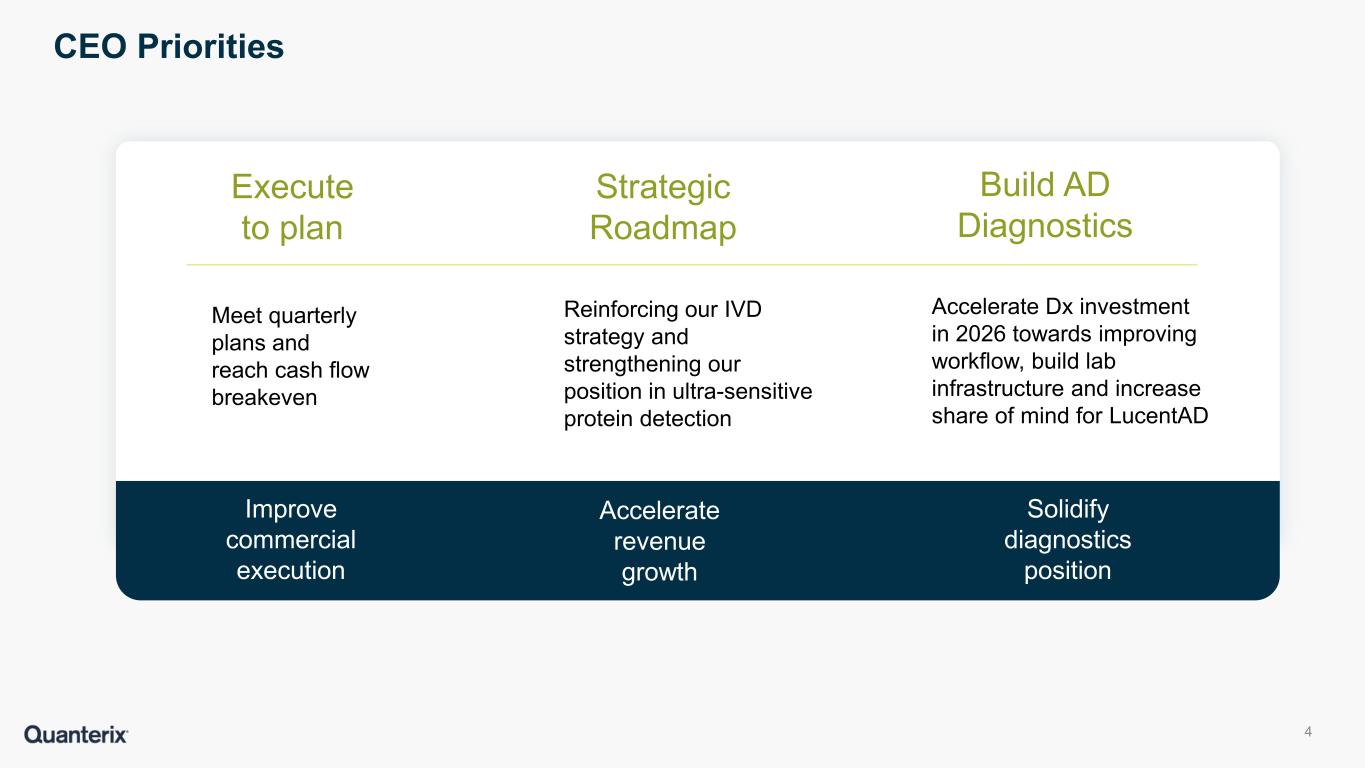
Company prioritizing product roadmap and investing in initiatives to drive commercial effectiveness
BILLERICA, Mass. – May 6, 2026 - Quanterix Corporation (NASDAQ: QTRX), a company transforming healthcare by accelerating biomarker breakthroughs from discovery to diagnostics, today announced financial results for the first quarter ended March 31, 2026.
“We continue to make progress toward achieving cash flow breakeven as we move into a phase of growth now that we have captured the cost synergies from the Akoya acquisition,” said Everett Cunningham, President & CEO of Quanterix. “As part of this process, we are focusing our investment into areas that will benefit our commercial effectiveness and drive improved operating results in 2026 and beyond. Additionally, our Alzheimer’s diagnostics business continues its rapid growth with several key milestones expected in the second half of this year, including the completion of three clinical utility studies and a decision from the FDA on our 510(k) application.”
First Quarter Financial Highlights
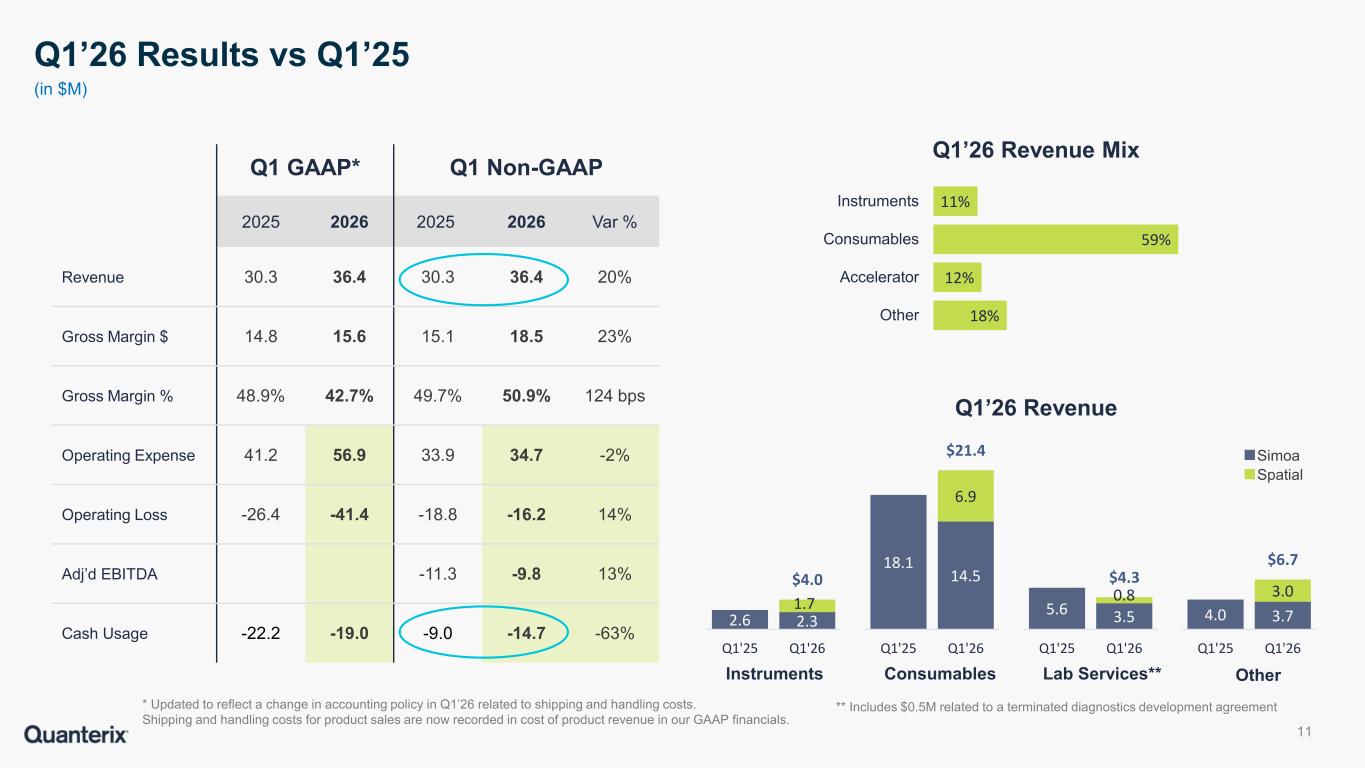
•Revenue of $36.4 million, an increase of 20% compared to $30.3 million in the prior year.
•GAAP gross margin of 42.7%, as compared to 48.9% in the prior year. Adjusted gross margin (non-GAAP) of 50.9% as compared to 49.7% in the prior year. Prior year margins are updated to reflect a change in accounting policy in Q1’26 related to shipping and handling costs. Shipping and handling costs for product sales are now recorded in cost of product revenue in the Company’s GAAP financials.
•Adjusted EBITDA (non-GAAP) loss of $9.8 million, compared to a loss of $11.3 million in the prior year.
•The Company ended the first quarter with $102.6 million of cash, cash equivalents, marketable securities, and restricted cash. Adjusted cash usage, after accounting for one-time deal and employee separation costs of $4.2 million, was $14.7 million in the first quarter, an increase from the fourth quarter of 2025 driven by seasonally higher payments.
Operational and Business Highlights
•Announced a collaboration with Tempus AI to broaden access to a novel blood-based biomarker panel designed to improve detection accuracy for Alzheimer’s disease. Through the agreement, Tempus AI will build a care gap program for Alzheimer’s disease blood-based biomarker testing, with Quanterix’s LucentAD® Complete multi-biomarker blood test becoming available for neurologists to order on the Tempus clinical ordering platform.
•Announced a diagnostics collaboration with Life Line Screening (LLS), a national organization focused on identifying asymptomatic risks for chronic conditions in community health settings. Through the collaboration, Life Line Screening will offer Quanterix’s Lucent Diagnostics non-invasive blood-based biomarker test for p-tau 217 nationally.
•Selected as a Co-Investigator institution in the PD-BUILD program, part of the Aligning Science Across Parkinson’s (ASAP) Collaborative Research Network (CRN) 2026 expansion, supported by The Michael J. Fox Foundation (MJFF). This multi-year grant brings together leading institutions across academia and industry to develop and deploy high-quality biomarker tools aimed at enabling earlier detection, improved patient stratification, and more effective monitoring of Parkinson’s disease in clinical research.
•Quanterix’s newly launched PhenoCode™ Discovery IO60 panel won silver at the Edison Awards. This award-winning product enables simultaneous visualization of 60 key markers across immune cell types, checkpoints, and tumor-specific pathways.
•Simoa® Ultra-Sensitive Immunoassay launched 3 new assays - mammalian GFAP advantage plus, IL12p70 advantage plus and IL17F advantage plus.
•The Accelerator Service Lab announced two new ADC lung cancer panels for Akoya PhenoImager™ HT at the American Association for Cancer Research(AACR) 2026 annual meeting. Building on the ADC breast cancer panel debuted at the AACR 2025 annual meeting, both panels are available today as a fully managed service.
2026 Business Outlook
Quanterix is reaffirming its guidance for 2026. The Company expects revenues of $169 to $174 million, which assumes no underlying improvement in the academic or pharmaceutical end markets. Quanterix anticipates GAAP gross margin of 41% to 45%, and adjusted gross margin (non-GAAP) of 49% to 53%.
In the first quarter, Quanterix changed its accounting policy for classifying shipping and handling costs for product sales to record them within gross margin. Historically, these costs were recorded in selling, general and administrative expenses. This reclassification is reflected in the Company’s GAAP guidance range, but there is no change to the non-GAAP margin expectation.
Quanterix continues to anticipate achieving cash flow breakeven in the second half of the year and expects to end the year with cash in the range of $100M, and no debt.
Conference Call
In conjunction with this announcement, the Company will host a conference call on May 6, 2026, at 4:30 PM ET. The dial-in number for USA & Canada is Toll-Free (800) 715-9871 or (646) 307-1963 and the conference ID is 8523507.
Interested investors can also listen to the live webcast from the Event Details page in the Investors section of the Quanterix website at https://ir.quanterix.com. An archived webcast replay will be available on the Company’s website for one year.
About Quanterix
Quanterix is a global leader in ultra-sensitive biomarker detection, enabling breakthroughs in disease research, diagnostics, and drug development. Its proprietary Simoa® technology delivers industry-leading sensitivity, allowing researchers to detect and quantify biomarkers in blood and other fluids at concentrations far below traditional limits. With approximately 6,500 peer-reviewed publications, Quanterix has been a trusted partner to the scientific community for nearly two decades. In 2025, Quanterix acquired Akoya Biosciences, The Spatial Biology Company®, adding multiplexed tissue imaging with single-cell resolution to its portfolio and 1,450 installed instruments. Together, the combined company offers a uniquely integrated platform that connects biology across blood and tissue—advancing precision medicine from discovery to diagnostics. Learn more at www.quanterix.com.
CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS
Statements included in this press release that are not historical in nature or do not relate to current facts are intended to be, and are hereby identified as, forward-looking statements for purposes of the safe harbor provided by Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. Forward-looking statements include, among other things, statements about Quanterix’s future business outlook, operations, strategy and financial performance, including statements related to our expectations about consistent profitable revenue growth and achieving cash flow breakeven performance, the development and commercialization of our products, the benefits and synergies we may realize from the acquisition of Akoya Biosciences Inc., and under the header “2026 Business Outlook.”. Words and phrases such as “may,” “approximately,” “continue,” “should,” “expects,” “projects,” “anticipates,” “is likely,” “look ahead,” “look forward,” “believes,” “will,” “intends,” “estimates,” “strategy,” “plan,” “could,” “potential,” “possible” and variations of such words and similar expressions are intended to identify such forward-looking statements. Forward-looking statements are subject to certain risks and uncertainties that are difficult to predict with regard to, among other things, timing, extent, likelihood and degree of occurrence, which could cause actual results to differ materially from anticipated results. Such risks and uncertainties include, among others, the following possibilities with respect to Quanterix’s future business, operations, strategy and financial performance: risks related to the impact of changes in U.S. government policies, including impacts of tariffs and reductions in federal research funding; risks associated with the anticipated timing for launch of, and features of, Quanterix’s next-generation instruments to upgrade its existing platforms; risks related to Quanterix’s ability to improve existing diagnostics and develop new diagnostic tests and tools; risks related to Quanterix’s ability to successfully penetrate the diagnostics market; risks related to Quanterix’s ability to retain and expand its customer base and achieve sufficient market acceptance of its products; risks related to the ability of Quanterix’s contract manufacturers and suppliers to reliably and consistently manufacture and supply our instruments; risks that Quanterix may fail to realize the anticipated benefits and synergies of its recent acquisitions of Emission, Inc. and Akoya Biosciences Inc.; risk that integrating Quanterix’s business with that of Akoya could be more difficult, costly or time-consuming than expected; risks that Quanterix’s estimates regarding expenses, future revenues, capital requirements, and needs for additional financing could be incorrect; risks related to Quanterix’s ability to maintain effective internal control over financial reporting and disclosure controls and procedures; and risks related to defects or other quality issues in Quanterix’s products that could lead to unforeseen costs, product recalls, adverse regulatory actions, negative publicity and litigation.
Additional factors that could cause results to differ materially from those described above can be found in the periodic reports filed by Quanterix with the SEC, including the “Risk Factors” sections contained therein, which are available on the SEC’s website at www.sec.gov.
All forward-looking statements, expressed or implied, included in this press release are expressly qualified in their entirety by the cautionary statements contained or referred to herein. If one or more events related to these or other risks or uncertainties materialize, or if Quanterix’s underlying assumptions prove to be incorrect, actual results may differ materially from what Quanterix anticipates. Quanterix cautions readers not to place undue reliance on any such forward-looking statements, which speak only as of the date they are made and are based on information available at that time. Quanterix does not assume any obligation to update or otherwise revise any forward-looking statements to reflect circumstances or events that occur after the date the forward-looking statements were made or to reflect the occurrence of unanticipated events except as required by federal securities laws.
Financial Highlights
QUANTERIX CORPORATION
CONSOLIDATED STATEMENTS OF OPERATIONS
(amounts in thousands, except per share data, unaudited)
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
2026 |
|
2025 |
| Revenues: |
|
|
|
| Product revenue |
$ |
25,479 |
|
|
$ |
20,739 |
|
| Service and other revenue |
10,376 |
|
|
8,823 |
|
| Collaboration and license revenue |
560 |
|
|
771 |
|
| Total revenues |
36,415 |
|
|
30,333 |
|
| Costs of goods sold and services: |
|
|
|
| Cost of product revenue |
15,140 |
|
|
11,341 |
|
| Cost of service and other revenue |
5,709 |
|
|
4,154 |
|
| Total costs of goods sold and services |
20,849 |
|
|
15,495 |
|
| Gross profit |
15,566 |
|
|
14,838 |
|
| Operating expenses: |
|
|
|
| Research and development |
7,323 |
|
|
10,036 |
|
| Selling, general and administrative |
29,770 |
|
|
31,168 |
|
| Impairment |
19,835 |
|
|
— |
|
| Total operating expenses |
56,928 |
|
|
41,204 |
|
| Loss from operations |
(41,362) |
|
|
(26,366) |
|
| Other income (expense), net: |
|
|
|
| Interest income |
892 |
|
|
3,267 |
|
| Change in fair value of contingent liabilities |
1,501 |
|
|
(379) |
|
| Other income, net |
21,421 |
|
|
61 |
|
| Loss before income taxes |
(17,548) |
|
|
(23,417) |
|
| Income tax benefit |
7 |
|
|
2,913 |
|
| Net loss |
$ |
(17,541) |
|
|
$ |
(20,504) |
|
|
|
|
|
| Net loss per common share, basic and diluted |
$ |
(0.37) |
|
|
$ |
(0.53) |
|
|
|
|
|
| Weighted-average common shares outstanding, basic and diluted |
46,979 |
|
38,718 |
QUANTERIX CORPORATION
CONSOLIDATED BALANCE SHEETS
(amounts in thousands, except per share data, unaudited)
|
|
|
|
|
|
|
|
|
|
|
|
|
March 31, 2026 |
|
December 31, 2025 |
| ASSETS |
|
|
|
| Current assets: |
|
|
|
| Cash and cash equivalents |
$ |
36,182 |
|
|
$ |
29,839 |
|
| Marketable securities |
63,083 |
|
|
88,393 |
|
| Accounts receivable, net of allowance for expected credit losses |
26,776 |
|
|
29,972 |
|
| Inventory |
50,959 |
|
|
54,763 |
|
| Prepaid expenses and other current assets |
8,725 |
|
|
9,290 |
|
| Total current assets |
185,725 |
|
|
212,257 |
|
| Restricted cash |
3,344 |
|
|
3,341 |
|
| Property and equipment, net |
21,369 |
|
|
23,672 |
|
| Intangible assets, net |
109,161 |
|
|
131,787 |
|
| Goodwill |
26,710 |
|
|
26,376 |
|
| Operating lease right-of-use assets |
15,861 |
|
|
16,664 |
|
| Other non-current assets |
4,502 |
|
|
4,669 |
|
| Total assets |
$ |
366,672 |
|
|
$ |
418,766 |
|
| LIABILITIES AND STOCKHOLDERS' EQUITY |
|
|
|
| Current liabilities: |
|
|
|
| Accounts payable |
$ |
8,657 |
|
|
$ |
13,568 |
|
| Accrued compensation and benefits |
9,850 |
|
|
14,979 |
|
| Accrued expenses and other current liabilities |
8,275 |
|
|
17,571 |
|
| Deferred revenue |
15,190 |
|
|
20,728 |
|
| Operating lease liabilities |
7,933 |
|
|
7,916 |
|
| Total current liabilities |
49,905 |
|
|
74,762 |
|
| Deferred revenue, net of current portion |
2,795 |
|
|
5,830 |
|
| Operating lease liabilities, net of current portion |
27,403 |
|
|
29,323 |
|
| Non-current portion of contingent liabilities |
3,547 |
|
|
5,024 |
|
| Other non-current liabilities |
883 |
|
|
8,097 |
|
| Total liabilities |
84,533 |
|
|
123,036 |
|
| Total stockholders’ equity |
282,139 |
|
|
295,730 |
|
| Total liabilities and stockholders’ equity |
$ |
366,672 |
|
|
$ |
418,766 |
|
QUANTERIX CORPORATION
CONSOLIDATED STATEMENTS OF CASH FLOWS
(in thousands, unaudited)
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
2026 |
|
2025 |
| Cash flows from operating activities: |
|
|
|
| Net loss |
$ |
(17,541) |
|
|
$ |
(20,504) |
|
| Adjustments to reconcile net loss to net cash used in operating activities: |
|
|
|
| Depreciation and amortization expense |
5,603 |
|
|
2,188 |
|
| Credit losses on accounts receivable |
305 |
|
|
53 |
|
| Accretion of marketable securities |
(150) |
|
|
(979) |
|
| Operating lease right-of-use asset amortization |
797 |
|
|
561 |
|
| Stock-based compensation expense |
4,528 |
|
|
5,462 |
|
| Impairment |
19,835 |
|
|
— |
|
| Change in fair value of contingent liabilities |
(1,501) |
|
|
379 |
|
| Recognition of off-market liability |
(13,975) |
|
|
— |
|
| Other operating activity |
15 |
|
|
(412) |
|
| Changes in assets and liabilities: |
|
|
|
| Accounts receivable |
2,717 |
|
|
4,329 |
|
| Inventory |
3,221 |
|
|
2,085 |
|
| Prepaid expenses and other current assets |
453 |
|
|
421 |
|
| Accounts payable |
(4,846) |
|
|
399 |
|
| Accrued compensation and benefits, accrued expenses, and other current liabilities |
(7,254) |
|
|
(3,517) |
|
| Deferred revenue |
(8,572) |
|
|
299 |
|
| Net change in other operating assets and liabilities |
(1,742) |
|
|
(4,652) |
|
| Net cash used in operating activities |
(18,107) |
|
|
(13,888) |
|
| Cash flows from investing activities: |
|
|
|
| Purchases of marketable securities |
— |
|
|
(30,246) |
|
| Proceeds from sales and maturities of marketable securities |
25,350 |
|
|
73,261 |
|
| Purchases of property and equipment |
(87) |
|
|
(1,256) |
|
| Acquisitions, net of cash acquired |
— |
|
|
(8,997) |
|
| Net cash provided by investing activities |
25,263 |
|
|
32,762 |
|
| Cash flows from financing activities: |
|
|
|
| Deferred acquisition payment |
(1,000) |
|
|
— |
|
| Principal payments on financing leases |
(83) |
|
|
— |
|
| Proceeds from common stock issued under stock plans |
340 |
|
|
668 |
|
| Payments for employee taxes withheld on stock-based compensation awards |
(27) |
|
|
(575) |
|
| Net cash provided by (used in) financing activities |
(770) |
|
|
93 |
|
| Net increase in cash, cash equivalents, and restricted cash |
6,386 |
|
|
18,967 |
|
| Effect of exchange rate changes on cash, cash equivalents, and restricted cash |
(40) |
|
|
861 |
|
| Cash, cash equivalents, and restricted cash at beginning of period |
33,180 |
|
|
59,319 |
|
| Cash, cash equivalents, and restricted cash at end of period |
$ |
39,526 |
|
|
$ |
79,147 |
|
Use of Non-GAAP Financial Measures
To supplement our financial statements presented on a U.S. GAAP basis, we present the following non-GAAP financial measures:
•Adjusted EBITDA and adjusted EBITDA margin: We define adjusted EBITDA as net income (loss) adjusted to exclude interest income, income tax (expense) benefit, depreciation and amortization expense, stock-based compensation expense, acquisition and integration related costs, impairment and restructuring, and certain other items which include other charges or benefits resulting from transactions or events that are unusual or infrequent, significant in size, and that we do not believe are indicative of ongoing or future business operations. These items are discussed in more detail below the tables reconciling the GAAP to non-GAAP measures. Adjusted EBITDA margin is calculated as adjusted EBITDA divided by total revenues.
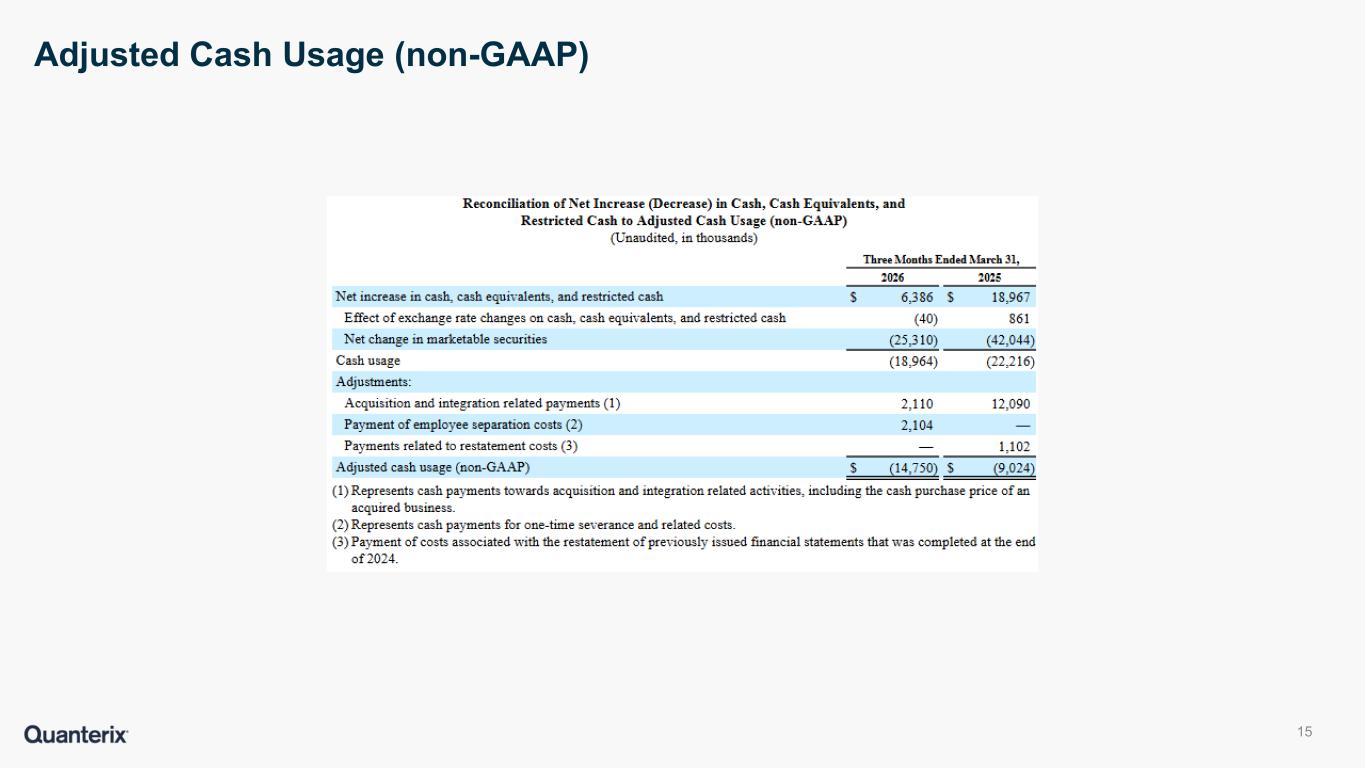
•Adjusted cash usage: We calculate cash usage as the total change in cash, cash equivalents, and restricted cash adjusted to include the net change from purchases, sales, and maturities of marketable securities (excluding any interest receivable). Adjusted cash usage is calculated as cash usage further adjusted to exclude cash payments related to transactions or events that are unusual or infrequent, significant in size, and that we do not believe are indicative of ongoing or future business operations.
•Adjusted gross profit, adjusted gross margin, adjusted total operating expenses, and adjusted loss from operations: We calculate these non-GAAP financial measures by excluding amortization of certain acquired intangible assets, acquisition and integration related costs, and certain other items which include other charges or benefits resulting from transactions or events that are unusual or infrequent, significant in size, and that we do not believe are indicative of ongoing or future business operations. Adjusted gross margin is calculated as adjusted gross profit divided by total revenues.
During the quarter ended March 31, 2026, we changed our accounting policy for classifying shipping and handling costs for product sales and they are now recorded in cost of product revenue. Historically, shipping and handling costs were recorded in selling, general and administrative expenses, and we calculated these non-GAAP financial measures by including shipping and handling costs for product sales within cost of product revenue instead of within selling, general and administrative expenses. We applied the change in accounting policy retrospectively, and no longer reclassify shipping and handling costs in our non-GAAP financial measures.
We believe that presentation of these non-GAAP financial measures provides supplemental information useful to investors in understanding our underlying operating results and trends. We use these non-GAAP financial measures to evaluate our operating performance in a manner that allows for meaningful period-to-period comparison and analysis of trends in our business and our competitors. We believe that presentation of these non-GAAP financial measures provides useful information to investors in assessing our operating performance within our industry and to allow comparability with the presentation of other companies in our industry.
The non-GAAP financial measures presented here should be considered in conjunction with, and not as a substitute for, the financial information presented in accordance with U.S. GAAP. For example, adjusted EBITDA excludes a number of expense items that are included in net loss and adjusted cash usage excludes certain actual cash payments. As a result, positive adjusted EBITDA or positive adjusted cash usage may be achieved even where we record a significant net loss or reduction in our cash and marketable securities balances in accordance with U.S. GAAP.
Investors are encouraged to review the reconciliation of these non-GAAP financial measures to their most directly comparable GAAP financial measures set forth in the tables captioned “Reconciliation of GAAP to Non-GAAP Financial Measures” in the section below.
Additionally, we make certain forward-looking statements about our future financial performance that include non-GAAP financial measures, which are difficult to predict for future periods because the nature of the adjustments pertains to events that have not yet occurred. We do not forecast many of the excluded items for internal use and therefore information reconciling forward-looking non-GAAP financial measures to U.S. GAAP financial measures is not available without unreasonable effort and is not provided. The occurrence, timing, and amount of any of the items excluded from U.S. GAAP to calculate non-GAAP financial measures could significantly impact our U.S. GAAP results.
QUANTERIX CORPORATION
RECONCILIATIONS OF GAAP TO NON-GAAP FINANCIAL MEASURES
Reconciliation of Net Loss to Adjusted EBITDA (non-GAAP) and Adjusted EBITDA Margin (non-GAAP)
(Unaudited, in thousands except percentages)
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
2026 |
|
2025 |
| Net loss |
$ |
(17,541) |
|
|
$ |
(20,504) |
|
| Interest income |
(892) |
|
|
(3,267) |
|
| Income tax expense (benefit) |
(7) |
|
|
(2,913) |
|
| Depreciation and amortization |
5,603 |
|
|
2,188 |
|
| Stock-based compensation expense (1) |
4,177 |
|
|
5,462 |
|
| Acquisition and integration related costs (2) |
1,152 |
|
|
3,578 |
|
| Earnout recorded as compensation expense (3) |
— |
|
|
3,744 |
|
| Changes in contingent liabilities (4) |
(1,501) |
|
|
379 |
|
| Impairment and employee separation costs (5) |
20,787 |
|
|
— |
|
| Income from contract termination (6) |
(21,596) |
|
|
— |
|
| Adjusted EBITDA (non-GAAP) |
$ |
(9,818) |
|
|
$ |
(11,333) |
|
|
|
|
|
| Total revenues |
$ |
36,415 |
|
|
$ |
30,333 |
|
| Adjusted EBITDA margin (non-GAAP) (adjusted EBITDA as a % of revenue) |
(27.0) |
% |
|
(37.4) |
% |
(1)Stock-based compensation expense for certain individuals are included in the caption 'Impairment and employee separation costs'.
(2)Represents acquisition and integration costs directly related to the Company's business combinations. Acquisition costs include professional and consulting fees supporting due diligence, legal, and accounting activities to execute a transaction. Integration costs include third party and internal direct costs to integrate acquired companies, employees, and their customers.
(3)Consists of the earnout recognized as compensation expense related to the Emission acquisition.
(4)Consists of fair value adjustments for contingent consideration liabilities related to acquisitions.
(5)Impairment charges for an intangible asset related to the termination of a diagnostics development agreement assumed in the acquisition of Akoya, as well as one-time severance and related costs.
(6)One-time income related to the impact of terminating a diagnostics development agreement assumed in the acquisition of Akoya.
Reconciliation of Net Increase (Decrease) in Cash, Cash Equivalents, and
Restricted Cash to Adjusted Cash Usage (non-GAAP)
(Unaudited, in thousands)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
|
|
|
|
2026 |
|
2025 |
|
|
|
|
| Net increase in cash, cash equivalents, and restricted cash |
$ |
6,386 |
|
|
$ |
18,967 |
|
|
|
|
|
| Effect of exchange rate changes on cash, cash equivalents, and restricted cash |
(40) |
|
|
861 |
|
|
|
|
|
| Net change in marketable securities |
(25,310) |
|
|
(42,044) |
|
|
|
|
|
| Cash usage |
(18,964) |
|
|
(22,216) |
|
|
|
|
|
| Adjustments: |
|
|
|
|
|
|
|
| Acquisition and integration related payments (1) |
2,110 |
|
|
12,090 |
|
|
|
|
|
| Payment of employee separation costs (2) |
2,104 |
|
|
— |
|
|
|
|
|
| Payments related to restatement costs (3) |
— |
|
|
1,102 |
|
|
|
|
|
| Adjusted cash usage (non-GAAP) |
$ |
(14,750) |
|
|
$ |
(9,024) |
|
|
|
|
|
(1)Represents cash payments towards acquisition and integration related activities, including the cash purchase price of an acquired business.
(2)Represents cash payments for one-time severance and related costs.
(3)Payment of costs associated with the restatement of previously issued financial statements that was completed at the end of 2024.
Reconciliation of Gross Profit, Gross Margin, Total Operating Expenses and Loss from Operations to Non-GAAP Financial Measures
(Unaudited, in thousands, except percentages)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, |
|
|
|
|
|
2026 |
|
2025 |
|
|
|
|
| Gross profit |
$ |
15,566 |
|
$ |
14,838 |
|
|
|
|
| Purchase accounting impact on inventory and property and equipment (1) |
199 |
|
— |
|
|
|
|
| Amortization of acquired intangible assets (2) |
2,772 |
|
227 |
|
|
|
|
| Adjusted gross profit (non-GAAP) |
$ |
18,537 |
|
$ |
15,065 |
|
|
|
|
|
|
|
|
|
|
|
|
| Total revenues |
$ |
36,415 |
|
$ |
30,333 |
|
|
|
|
| Gross margin (gross profit as % of total revenues) |
42.7% |
|
48.9% |
|
|
|
|
| Adjusted gross margin (non-GAAP) (adjusted gross profit as % of total revenues) |
50.9% |
|
49.7% |
|
|
|
|
|
|
|
|
|
|
|
|
| Total operating expenses |
$ |
56,928 |
|
$ |
41,204 |
|
|
|
|
| Purchase accounting impact on property and equipment (1) |
(223) |
|
— |
|
|
|
|
| Amortization of acquired intangible assets (2) |
(77) |
|
— |
|
|
|
|
| Acquisition and integration related costs (3) |
(1,152) |
|
(3,578) |
|
|
|
|
| Earnout recorded as compensation expense (4) |
— |
|
(3,744) |
|
|
|
|
| Impairment and employee separation costs (5) |
(20,787) |
|
— |
|
|
|
|
| Adjusted total operating expenses (non-GAAP) |
$ |
34,689 |
|
$ |
33,882 |
|
|
|
|
|
|
|
|
|
|
|
|
| Loss from operations |
$ |
(41,362) |
|
$ |
(26,366) |
|
|
|
|
| Purchase accounting impact on inventory and property and equipment (1) |
422 |
|
— |
|
|
|
|
| Amortization of acquired intangible assets (2) |
2,849 |
|
227 |
|
|
|
|
| Acquisition and integration related costs (3) |
1,152 |
|
3,578 |
|
|
|
|
| Earnout recorded as compensation expense (4) |
— |
|
3,744 |
|
|
|
|
| Impairment and employee separation costs (5) |
20,787 |
|
— |
|
|
|
|
| Adjusted loss from operations (non-GAAP) |
$ |
(16,152) |
|
$ |
(18,817) |
|
|
|
|
(1)Represents the amortization of the purchase price fair value increase of acquired inventory and property and equipment.
(2)Consists only of the amortization of intangible assets acquired in 2025.
(3)Represents acquisition and integration costs directly related to the Company's business combinations. Acquisition costs include professional and consulting fees supporting due diligence, legal, and accounting activities to execute a transaction. Integration costs include third party and internal direct costs to integrate acquired companies, employees, and their customers.
(4)Consists of the earnout recognized as compensation expense related to the Emission acquisition.
(5)Impairment charges for an intangible asset related to the termination of a diagnostics development agreement assumed in the acquisition of Akoya, as well as one-time severance and related costs.
Contact:
Joshua Young
(508) 846-3327
ir@quanterix.com