UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): January 7, 2026
TANGO THERAPEUTICS, INC.
(Exact name of Registrant as Specified in Its Charter)
| Delaware | 001-39485 | 85-1195036 | ||
| (State or Other Jurisdiction of Incorporation) |
(Commission File Number) |
(IRS Employer Identification No.) |
| 201 Brookline Ave., Suite 901 Boston, MA |
02215 | |
| (Address of Principal Executive Offices) | (Zip Code) |
Registrant’s Telephone Number, Including Area Code: 857-320-4900
(Former Name or Former Address, if Changed Since Last Report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| ☐ | Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ☐ | Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ☐ | Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ☐ | Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class |
Trading |
Name of each exchange on which registered |
||
| Common Stock, $0.001 par value | TNGX | The Nasdaq Global Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
| Item 2.02 | Results of Operations and Financial Condition. |
On January 8, 2026, Tango Therapeutics, Inc. (the “Company”) disclosed that its cash, cash equivalents and marketable securities as of December 31, 2025 totaled $343 million (unaudited). The information in this Item 2.02 has not been audited and has been prepared by, and is the responsibility of, the Company’s management. The data could change as a result of further review and does not present all information necessary for an understanding of the Company’s financial condition as of December 31, 2025.
| Item 5.02 | Departure of Directors or Certain Officers; Election of Directors; Appointment of Certain Officers; Compensatory Arrangements of Certain Officers. |
Resignation of Barbara Weber, M.D. as President and Chief Executive Officer
On January 7, 2026, Barbara Weber, M.D. notified the board of directors (the “Board”) of the Company of her retirement and resignation as President and Chief Executive Officer of the Company, in each case, effective as of January 8, 2026 (the “Effective Date”).
Dr. Weber and the Company entered into an amended and restated employment agreement (the “Amended and Restated Employment Agreement”), effective as of the Effective Date, pursuant to which Dr. Weber has agreed to serve as the Company’s Executive Chair and to provide advisory services to assist in the orderly transition of her roles and responsibilities as President and Chief Executive Officer following the Effective Date through December 31, 2026. Pursuant to the terms of the Amended and Restated Employment Agreement, Dr. Weber will be entitled to:
| • | Annual base salary of $362,000; and |
| • | Target cash bonus opportunity of 60% of Dr. Weber’s current annual base salary, pro-rated for any partial calendar year of service. |
Dr. Weber’s resignation as President and Chief Executive Officer and transition is not the result of any disagreement with respect to the Company’s operations, policies, or practices. Following the Effective Date, Dr. Weber will serve as chairperson of the Board.
The foregoing description of the terms and conditions of the Amended and Restated Employment Agreement does not purport to be complete and is qualified in its entirety by the Amended and Restated Employment Agreement, which is attached hereto as Exhibit 10.1 and incorporated by reference herein.
Appointment of Malte Peters, M.D. as President and Chief Executive Officer
On January 8, 2026, Malte Peters, M.D., a member of the Board, was appointed as the Company’s President and Chief Executive Officer as of the Effective Date. Biographical information for Dr. Peters is set forth in the Company’s definitive proxy statement on Schedule 14A filed with the U.S. Securities and Exchange Commission on April 21, 2025 and is incorporated herein by reference.
In connection with his appointment as President and Chief Executive Officer, Dr. Peters entered into a compensation agreement with the Company (the “Compensation Agreement”), effective January 8, 2026, and an employment agreement with the Company (the “Employment Agreement”), effective upon the date on which Dr. Peters receives work authorization to work in the United States. The Compensation Agreement will expire upon the effective date of the Employment Agreement. Pursuant to the terms of the Compensation Agreement and the Employment Agreement Dr. Peters will be entitled to:
| • | Annual base salary of $720,000; |
| • | Target cash bonus opportunity of 60% of Dr. Peter’s current annual base salary, pro-rated for any partial calendar year of service. |
The foregoing description of the terms and conditions of the Compensation Agreement and the Employment Agreement do not purport to be complete and are qualified in their entirety by the Compensation Agreement and the Employment Agreement, which are attached hereto as Exhibit 10.2 and 10.3, respectively and incorporated by reference herein.
In connection with his appointment as President and Chief Executive Officer, Dr. Peters was also granted (i) an option to purchase 1,650,000 shares of the Company’s common stock under the Company’s 2021 Stock Option and Incentive Plan at an exercise price equal to the closing price of the Company’s common stock as reported on The Nasdaq Global Market on January 8, 2026 , which will vest as to 25% of the shares underlying the option on the one year anniversary of the grant date, and the remainder vesting in 36 monthly thereafter (subject to Dr. Peter’s continued employment with the Company), and (ii) restricted stock units (“RSUs”) to acquire 350,000 shares of the Company’s common stock, which shares will vest in three equal annual installments in accordance with Company’s equity incentive policies, in each case, subject to Dr. Peters’ continued employment with the Company.
There are no understandings or arrangements between Dr. Peters and any other person pursuant to which he was appointed as the Company’s President and Chief Executive Officer. Except as described above, Dr. Peters does not have any material interest in any transaction or proposed transaction in which the Company is or is to be a party. Dr. Peters does not have a family relationship with any other director or executive officer of the Company. Consistent with the Company’s standard non-employee director compensation policy, Dr. Peters will not be eligible to receive compensation for his role on the Board since he is an executive officer.
| Item 7.01 | Regulation FD Disclosures. |
On January 8, 2026, the Company issued a press release announcing the leadership changes described above and updated its corporate presentation to be used from time to time with investors, analysts, and other third parties. Copies of the press release and the updated presentation are being furnished as Exhibits 99.1 and 99.2, respectively, to this Current Report on Form 8-K.
The information in Item 2.02 and 7.01 of this Current Report on Form 8-K and Exhibit 99.1 as well as Exhibit 99.2 attached hereto is being furnished and shall not be deemed “filed” for the purposes of Section 18 of the Exchange Act or otherwise subject to the liability of that section. Nor shall such document be deemed incorporated by reference in any filing under the Securities Act of 1933 or the Exchange Act, regardless of any general incorporation language in the filing, unless specifically stated so therein.
Cautionary Note Regarding Forward Looking Statements
This Current Report on Form 8-K and certain of the materials filed herewith contain forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including, without limitation, statements regarding the Company’s anticipated financial results. The words “may,” “might,” “will,” “could,” “would,” “should,” “expect,” “plan,” “anticipate,” “intend,” “believe,” “expect,” “estimate,” “seek,” “predict,” “future,” “project,” “potential,” “continue,” “target” and similar words or expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. Any forward-looking statements are subject to a number of risks, uncertainties and important factors that may cause actual events or results to differ materially from those expressed or implied by any forward-looking statements contained in this Current Report on Form 8-K or the materials furnished or filed herewith, including (i) the initiation, timing, progress, and cost of our research and development programs and our current and future preclinical studies and clinical trials (including combination clinical trials), enrollment and dosing in our clinical trials (including combination clinical trials), and the timing of initial and interim (and final) safety and efficacy or clinical activity data from our clinical trials may not take place in the timeframe that the Company expects; and (ii) we may be required to utilize our cash resources more quickly than we expect. These and other risks and uncertainties are described in additional detail in the section entitled “Risk Factors” in the Company’s Annual Report on Form 10-K filed February 27, 2025 and its other filings made with the SEC from time to time. Although the Company’s forward-looking statements reflect the good faith judgment of its management, these statements are based only on facts and factors currently known by the Company. As a result, you are cautioned not to rely on these forward-looking statements. Any forward-looking statement made in this Current Report on Form 8-K speaks only as of the date on which it is made. Except as required by applicable law, the Company undertakes no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future developments, or otherwise.
| Item 9.01. | Financial Statements and Exhibits. |
(d) Exhibits.
| Exhibit Number |
Description of Exhibit | |
| 10.1 | Amended and Restated Employment Agreement, dated as of January 8, 2026 by and between the Company and Barbara Weber. | |
| 10.2 | Compensation Agreement, dated as of January 8, 2026 by and between the Company and Malte Peters. | |
| 10.3 | Employment Agreement, dated as of January 8, 2026 by and between the Company and Malte Peters. | |
| 99.1 | Press release, dated January 8, 2026. | |
| 99.2 | Presentation, dated January 8, 2026. | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document). | |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
| Tango Therapeutics, Inc. | ||||||
| Date: January 8, 2026 | /s/ Daniella Beckman |
|||||
| Daniella Beckman | ||||||
| Chief Financial Officer | ||||||
Exhibit 10.1

AMENDED AND RESTATED EMPLOYMENT AGREEMENT
This Amended and Restated Employment Agreement (“Agreement”) is made between Tango Therapeutics, Inc., a Delaware corporation (the “Company”), and Barbara Weber, MD (the “Executive”) and is effective as of January 8, 2026 (the “Effective Date”). Except with respect to the Equity Documents (as defined below) and the Restrictive Covenants Agreement (as defined below), this Agreement supersedes in all respects all prior agreements between the Executive and the Company regarding the subject matter herein, including without limitation (i) the Employment Agreement between the Executive and the Company dated April 13, 2021 (the “Prior Employment Agreement”), and (ii) any other offer letter, employment agreement or severance agreement.
WHEREAS, the Company desires to continue to employ the Executive and the Executive desires to continue to be employed by the Company on the new terms and conditions contained herein as of the Effective Date.
WHEREAS, the Executive acknowledges and agrees that the changes to the Executive’s employment contemplated by this Agreement are voluntary and do not trigger any rights under the Prior Employment Agreement;
NOW, THEREFORE, in consideration of the mutual covenants and agreements herein contained and other good and valuable consideration, the receipt and sufficiency of which is hereby acknowledged, the parties agree as follows:
1. Employment.
(a) Term. The Company shall continue to employ the Executive and the Executive shall continue to be employed by the Company, provided as of the Effective Date this Agreement shall govern and shall continue until the earlier of (i) Executive’s employment is terminated in accordance with the provisions hereof, or (ii) December 31, 2026 (the “Term”). The Executive’s employment with the Company shall continue to be “at will,” meaning that the Executive’s employment may be terminated by the Company or the Executive at any time and for any reason subject to the terms of this Agreement.
(b) Transition; Position and Duties. As the Effective Date, the Executive shall no longer serve as the President and Chief Executive Officer of the Company (the “CEO”) but shall voluntarily transition to the Executive Chair of the Company. As Executive Chair, the Executive shall have such powers and duties as may from time to time be prescribed by the Board of Directors of the Company (the “Board”). In addition, the Executive shall continue to be on the Board as long as the Executive remains the Executive Chair subject to corporate governance rules provided that the Executive shall be deemed to have resigned from the Board and from any related positions upon ceasing to serve as Executive Chair for any reason unless otherwise mutually agreed by the Executive and the Board. The Executive shall devote 50% of her working time and efforts to the business and affairs of the Company. Notwithstanding the foregoing, the Executive may serve on other boards of directors, with the approval of the Board, or engage in religious, charitable or other community activities as long as such services and activities do not materially interfere with the Executive’s performance of the Executive’s duties to the Company.
(c) Location. The Executive’s primary work location will be in the Company’s office, currently located in Boston, Massachusetts; provided that the Executive may be required to travel for business from time to time, consistent with the Company’s business needs.
2. Compensation and Related Matters.
(a) Base Salary. As of the Effective Date, the Executive’s initial base salary shall be paid at the rate of $362,000 per year. The base salary in effect at any given time is referred to herein as “Base Salary.” The Base Salary shall be payable in a manner that is consistent with the Company’s usual payroll practices for its executive officers.
(b) Incentive Compensation. The Executive shall be eligible to receive cash incentive compensation for calendar year 2026 as determined by the Board or the Compensation Committee of the Board. The Executive’s target annual incentive compensation for calendar year 2026 shall be 60 percent of the Executive’s Base Salary, which is referred to as “Target Bonus.” The actual amount of the Executive’s calendar year 2026 incentive compensation, if any, shall be determined in the sole discretion of the Board or the Compensation Committee of the Board, subject to the terms of any applicable incentive compensation plan that may be in effect from time to time. Except as otherwise provided in this Agreement, the Executive must be employed by the Company on December 31, 2026 to earn or receive a 2026 bonus. Any 2026 incentive compensation approved by the Board or the Compensation Committee of the Board shall be payable in a manner that is consistent with the Company’s usual payroll practices for its executive officers but shall in no event be paid later than March 15, 2027. Nothing herein shall be construed to affect the Executive’s right to an annual bonus for calendar year 2025 consistent with the terms of the Prior Employment Agreement.
(c) Expenses. The Executive shall be entitled to receive prompt reimbursement for all reasonable expenses incurred by the Executive during the Term in performing services hereunder, in accordance with the policies and procedures then in effect and established by the Company for its executive officers.
(d) Other Benefits. During the Term, the Executive shall be eligible to participate in or receive benefits under the Company’s employee benefit plans in effect from time to time, subject to the terms of such plans.
(e) Paid Time Off. During the Term, the Executive shall be entitled to take paid time off in accordance with the Company’s applicable paid time off policy for executives, as may be in effect from time to time.
2
(f) Equity.
(i) The equity awards held by the Executive shall continue to be governed by the terms and conditions of the Company’s applicable equity incentive plan(s) and the applicable award agreement(s) governing the terms of such equity awards (collectively, the “Equity Documents”); provided, however, that, subject to approval by the Board or the Compensation Committee of the Board, (i) all of the Executive’s outstanding equity awards shall be amended to provide for continued vesting for as long as the Executive remains employed by the Company or serves on the Board and (ii) subject to the Executive’s continued employment or service on the Board through December 31, 2027, all outstanding equity awards held by the Executive that are subject solely to time-based vesting (the “Time-Based Equity Awards”) shall immediately accelerate and become fully vested and exercisable or nonforfeitable as of December 31, 2027; provided, further, and notwithstanding anything to the contrary in the Equity Documents, Section 6(a)(iii) of this Agreement shall apply in the event of a termination of the Executive’s employment by the Company without Cause or by the Executive for Good Reason in either event within the Change in Control Period (as such terms are defined below).
(ii) Subject to the approval of the Board or a committee thereof, at such time in 2026 as the Company makes its annual grants to employees, the Executive will be granted an equity award in the form of a non-qualified stock option to purchase 222,132 shares of common stock of the Company (the “Option”) at an exercise price equal to the fair market value of the common stock on the date of grant and 37,522 restricted stock units (“RSUs”). The Option will vest over 12 substantially equal monthly installments over one year from the date of grant, subject to the Executive’s continued service relationship (including as director or employee) on such date. The RSUs will vest it their entirety on the first anniversary of the date of grant (or in accordance with Company’s equity grant policies) subject to continued service as an employee or director through the applicable vesting date.
3. Termination. The Executive’s employment hereunder may be terminated without any breach of this Agreement under the following circumstances:
(a) Death. The Executive’s employment hereunder shall terminate upon death.
(b) Disability. The Company may terminate the Executive’s employment if the Executive is disabled and unable to perform or expected to be unable to perform the essential functions of the Executive’s then existing position or positions under this Agreement with or without reasonable accommodation for a period of 180 days (which need not be consecutive) in any 12-month period. If any question shall arise as to whether during any period the Executive is disabled so as to be unable to perform the essential functions of the Executive’s then existing position or positions with or without reasonable accommodation, the Executive may, and at the request of the Company shall, submit to the Company a certification in reasonable detail by a physician selected by the Company to whom the Executive or the Executive’s guardian has no reasonable objection as to whether the Executive is so disabled or how long such disability is expected to continue, and such certification shall for the purposes of this Agreement be conclusive of the issue. The Executive shall cooperate with any reasonable request of the physician in connection with such certification. If such question shall arise and the Executive shall fail to submit such certification, the Company’s determination of such issue shall be binding on the Executive. Nothing in this Section 3(b) shall be construed to waive the Executive’s rights, if any, under existing law including, without limitation, the Family and Medical Leave Act of 1993, 29 U.S.C. §2601 et seq. and the Americans with Disabilities Act, 42 U.S.C. §12101 et seq.
3
(c) Termination by the Company for Cause. The Company may terminate the Executive’s employment hereunder for Cause. For purposes of this Agreement, “Cause” shall mean any of the following:
(i) the Executive’s dishonest statements or acts with respect to the Company or any affiliate of the Company, or any current or prospective customers, suppliers, vendors or other third parties with which such entity does business that results in or is reasonably anticipated to result in material harm to the Company;
(ii) the Executive’s conviction of (A) a felony or (B) any misdemeanor involving moral turpitude, deceit, dishonesty or fraud;
(iii) the Executive’s substantial failure to perform the Executive’s assigned duties and responsibilities which failure continues for 30 days after written notice given to the Executive by the Board describing such failure in reasonable detail in the reasonable good faith judgment of the Board;
(iv) the Executive’s gross negligence, willful misconduct or insubordination with respect to the Company that results in or is reasonably anticipated to result in material harm to the Company;
(v) the Executive’s material violation of this Agreement, the Restrictive Covenants Agreement, or any provision of any agreement(s) between the Executive and the Company relating to noncompetition, nonsolicitation, nondisclosure and/or assignment of inventions; or
(vi) the Executive’s failure to cooperate with a bona fide internal investigation or an investigation by regulatory or law enforcement authorities, after being instructed by the Company to cooperate, or the willful destruction or failure to preserve documents or other materials known to be relevant to such investigation or the inducement of others to fail to cooperate or to produce documents or other materials in connection with such investigation.
(d) Termination by the Company without Cause. The Company may terminate the Executive’s employment hereunder at any time without Cause. Any termination by the Company of the Executive’s employment under this Agreement which does not constitute a termination for Cause under Section 3(c) and does not result from the death or disability of the Executive under Section 3(a) or (b) shall be deemed a termination without Cause.
(e) Termination by the Executive. The Executive may terminate employment hereunder at any time for any reason, including but not limited to, Good Reason. For purposes of this Agreement, “Good Reason” shall mean that the Executive has completed all steps of the Good Reason Process (hereinafter defined) following the occurrence of any of the following events without the Executive’s consent (each, a “Good Reason Condition”):
4
(i) a material diminution in the Executive’s Base Salary or a material reduction in the Executive’s benefits, except for across-the-board salary reductions based on the Company’s financial performance or a general reduction in the benefits, in each case similarly affecting all or substantially all senior management employees of the Company;
(ii) a material change in the geographic location of the principal office of the Company to which the Executive is assigned, such that there is an increase of at least 50 miles of driving distance to such location from the Executive’s principal residence as of such change, not including business travel and short-term assignments;
(iii) a material breach of this Agreement by the Company; or
(iv) a failure by the Company to obtain the assumption of this Agreement by any successor to the Company.
The “Good Reason Process” consists of the following steps:
(i) the Executive reasonably determines in good faith that a Good Reason Condition has occurred;
(ii) the Executive notifies the Company in writing of the first occurrence of the Good Reason Condition within 60 days of the first occurrence of such condition;
(iii) the Executive cooperates in good faith with the Company’s efforts, for a period of not less than 30 days following such notice (the “Cure Period”), to remedy the Good Reason Condition;
(iv) notwithstanding such efforts, the Good Reason Condition continues to exist at the end of the Cure Period; and
(v) the Executive terminates employment within 60 days after the end of the Cure Period.
If the Company cures the Good Reason Condition during the Cure Period, Good Reason shall be deemed not to have occurred.
4. Matters-related to Termination.
(a) Notice of Termination. Except for termination as specified in Section 3(a), any termination of the Executive’s employment by the Company or any such termination by the Executive shall be communicated by written Notice of Termination to the other party hereto. For purposes of this Agreement, a “Notice of Termination” shall mean a notice which shall indicate the specific termination provision in this Agreement relied upon.
5
(b) Date of Termination. “Date of Termination” shall mean: (i) if the Executive’s employment is terminated by death, the date of death; (ii) if the Executive’s employment is terminated on account of disability under Section 3(b) or by the Company for Cause under Section 3(c), the date on which Notice of Termination is given; (iii) if the Executive’s employment is terminated by the Company without Cause under Section 3(d), the date on which a Notice of Termination is given or the date otherwise specified by the Company in the Notice of Termination; (iv) if the Executive’s employment is terminated by the Executive under Section 3(e) other than for Good Reason, 30 days after the date on which a Notice of Termination is given, and (v) if the Executive’s employment is terminated by the Executive under Section 3(e) for Good Reason, the date on which a Notice of Termination is given after the end of the Cure Period. Notwithstanding the foregoing, in the event that the Executive gives a Notice of Termination to the Company, the Company may unilaterally accelerate the Date of Termination and such acceleration shall not result in a termination by the Company for purposes of this Agreement.
(c) Accrued Obligations. If the Executive’s employment with the Company is terminated for any reason, the Company shall pay or provide to the Executive (or to the Executive’s authorized representative or estate) (i) any Base Salary and, if applicable, accrued but unused vacation earned through the Date of Termination; (ii) unpaid expense reimbursements (subject to, and in accordance with, Section 2(c) of this Agreement); and (iii) any vested benefits the Executive may have under any employee benefit plan of the Company through the Date of Termination, which vested benefits shall be paid and/or provided in accordance with the terms of such employee benefit plans (collectively, the “Accrued Obligations”).
(d) Resignation of All Other Positions. To the extent applicable and unless otherwise mutually agreed by the Executive and the Board, the Executive shall be deemed to have resigned from all officer and board member positions that the Executive holds with the Company or any of its respective subsidiaries and affiliates upon the termination of the Executive’s employment for any reason. The Executive shall execute any documents in reasonable form as may be requested to confirm or effectuate any such resignations.
5. Severance Pay and Benefits Upon Termination by the Company without Cause or by the Executive for Good Reason Outside the Change in Control Period. If the Executive’s employment is terminated by the Company without Cause as provided in Section 3(d), or the Executive terminates employment for Good Reason as provided in Section 3(e), in each case outside of the Change in Control Period (as defined below), then, in addition to the Accrued Obligations, and subject to (i) the Executive signing a separation agreement and release in a form and manner satisfactory to the Company, which shall include, without limitation, a general release of claims against the Company and all related persons and entities that shall not release the Executive’s rights under this Agreement, a reaffirmation of all of the Executive’s Continuing Obligations (as defined below), and, in the Company’s sole discretion, a one-year post-employment noncompetition agreement (which shall not expand the scope of proscribed activities under any then-existing noncompetition obligations), and shall provide that if the Executive breaches any of the Continuing Obligations, all payments of the Severance Amount shall immediately cease (the “Separation Agreement”), and (ii) the Separation Agreement becoming irrevocable, all within 60 days after the Date of Termination (or such shorter period as set forth in the Separation Agreement), which shall include a seven business day revocation period:
6
(a) the Company shall pay the Executive an amount equal to the sum of the Base Salary for the lesser of: (i) 6 months or (ii) the remainder of the Term (in either event the “Severance Period”);
(b) the Company shall pay the Executive a pro-rata portion of the annual incentive compensation the Executive would otherwise be entitled to receive based on Company and individual performance for the calendar year in which the Date of Termination occurs, calculated by multiplying such amount by a fraction of which the numerator is the number of days in such calendar year prior to the Date of Termination, and the denominator is 365 (the “Pro-Rated Bonus”); and
(c) subject to the Executive’s copayment of premium amounts at the applicable active employees’ rate and the Executive’s proper election to receive benefits under the Consolidated Omnibus Budget Reconciliation Act of 1985, as amended (“COBRA”), the Company shall pay to the group health plan provider or the COBRA provider a monthly payment equal to the monthly employer contribution that the Company would have made to provide health insurance to the Executive if the Executive had remained employed by the Company until the earliest of (A) the last date of the Severance Period; (B) the date that the Executive becomes eligible for group medical plan benefits under any other employer’s group medical plan; or (C) the cessation of the Executive’s health continuation rights under COBRA; provided, however, that if the Company determines that it cannot pay such amounts to the group health plan provider or the COBRA provider (if applicable) without potentially violating applicable law (including, without limitation, Section 2716 of the Public Health Service Act), then the Company shall convert such payments to payroll payments directly to the Executive for the time period specified above. Such payments to the Executive shall be subject to tax-related deductions and withholdings and paid on the Company’s regular payroll dates; and
(d) notwithstanding anything to the contrary in any applicable option agreement or other stock-based award agreement, the portion of all Time-Based Equity Awards scheduled to vest in the 12 month period following the Date of Termination shall immediately accelerate and become fully vested and exercisable or nonforfeitable as of the later of (A) the Date of Termination or (B) the effective date of the Separation Agreement (the “Accelerated Vesting Date”); provided that in order to effectuate the accelerated vesting contemplated by this subsection, the unvested portion of the Executive’s Time-Based Equity Awards that are subject to acceleration pursuant to this subsection that would otherwise terminate or be forfeited on the Date of Termination will be delayed until the earlier of (A) the effective date of the Separation Agreement (at which time acceleration will occur), or (B) the date that the Separation Agreement can no longer become fully effective (at which time the unvested portion of the Executive’s Time-Based Equity Awards subject to acceleration pursuant to this subsection will terminate or be forfeited). Notwithstanding the foregoing, no additional vesting of the Time-Based Equity Awards shall occur during the period between the Date of Termination and the Accelerated Vesting Date. With respect to any performance-based vesting equity award, such award shall continue to be governed in all respects by the terms of the applicable equity award documents.
7
The amounts payable under Section 5(a) and (c), to the extent taxable, shall be paid out in substantially equal installments in accordance with the Company’s payroll practice over the Severance Period commencing within 60 days after the Date of Termination; provided, however, that if the 60-day period begins in one calendar year and ends in a second calendar year, such payments, to the extent they qualify as “non-qualified deferred compensation” within the meaning of Section 409A of the Internal Revenue Code of 1986, as amended (the “Code”), shall begin to be paid in the second calendar year by the last day of such 60-day period; provided, further, that the initial payment shall include a catch-up payment to cover amounts retroactive to the day immediately following the Date of Termination. The Pro-Rated Bonus will paid at the same time as annual incentive compensation payments are made to active employees for the calendar year in which the Date of Termination occurs, but in no event later than March 15 of the calendar year following the year in which it is earned. Each payment pursuant to this Agreement is intended to constitute a separate payment for purposes of Treasury Regulation Section 1.409A-2(b)(2).
6. Severance Pay and Benefits Upon Termination by the Company without Cause or by the Executive for Good Reason within the Change in Control Period. The provisions of this Section 6 shall apply in lieu of, and expressly supersede, the provisions of Section 5 if (i) the Executive’s employment is terminated either (a) by the Company without Cause as provided in Section 3(d) or (b) by the Executive for Good Reason as provided in Section 3(e), and (ii) the Date of Termination is during the Term and after the occurrence of the first event constituting a Change in Control (such period, the “Change in Control Period”). These provisions shall terminate and be of no further force or effect after the Change in Control Period.
(a) If the Executive’s employment is terminated by the Company without Cause as provided in Section 3(d) or the Executive terminates employment for Good Reason as provided in Section 3(e) and in each case the Date of Termination occurs within the Change in Control Period, then, in addition to the Accrued Obligations, and subject to the signing of a general release of claims against the Company and all related persons and entities that shall not release the Executive’s rights under this Agreement (the “Release”) by the Executive and the Release becoming fully effective, all within the time frame set forth in the Release but in no event more than 60 days after the Date of Termination:
(i) the Company shall pay the Executive a lump sum in cash in an amount equal to the sum of (A) 9 months of the Executive’s then-current Base Salary (or the Executive’s Base Salary in effect immediately prior to the Change in Control, if higher) plus (B) .75 times the Executive’s Target Bonus;
(ii) the Company shall pay the Executive the Pro-Rated Bonus;
(iii) notwithstanding anything to the contrary in any applicable option agreement or other stock-based award agreement, all Time-Based Equity Awards shall immediately accelerate and become fully vested and exercisable or nonforfeitable as of the later of (i) the Date of Termination or (ii) the effective date of the Release (the “Change in Control Accelerated Vesting Date”), provided that in order to effectuate the accelerated vesting contemplated by this subsection, the unvested portion of the Executive’s Time-Based Equity Awards that would otherwise terminate or be forfeited on the Date of Termination will be delayed until the earlier of (A) the effective date of the Release (at which time acceleration will occur), or (B) the date that the Release can no longer become fully effective (at which time the unvested portion of the Executive’s Time-Based Equity Awards will terminate or be forfeited).
8
Notwithstanding the foregoing, no additional vesting of the Time-Based Equity Awards shall occur during the period between the Date of Termination and the Change in Control Accelerated Vesting Date. With respect to any performance-based vesting equity award, such award shall continue to be governed in all respects by the Equity Documents; and
(iv) subject to the Executive’s copayment of premium amounts at the applicable active employees’ rate and the Executive’s proper election to receive benefits under COBRA, the Company shall pay to the group health plan provider or the COBRA provider a monthly payment equal to the monthly employer contribution that the Company would have made to provide health insurance to the Executive if the Executive had remained employed by the Company until the earliest of (A) the 9 month anniversary of the Date of Termination; (B) the date that the Executive becomes eligible for group medical plan benefits under any other employer’s group medical plan; or (C) the cessation of the Executive’s health continuation rights under COBRA; provided, however, that if the Company determines that it cannot pay such amounts to the group health plan provider or the COBRA provider (if applicable) without potentially violating applicable law (including, without limitation, Section 2716 of the Public Health Service Act), then the Company shall convert such payments to payroll payments directly to the Executive for the time period specified above. Such payments to the Executive shall be subject to tax-related deductions and withholdings and paid on the Company’s regular payroll dates.
The amounts payable under Section 6(a)(i) and (iii), to the extent taxable, shall be paid or commence to be paid within 60 days after the Date of Termination; provided, however, that if the 60-day period begins in one calendar year and ends in a second calendar year, such payments to the extent they qualify as “non-qualified deferred compensation” within the meaning of Section 409A of the Code, shall be paid or commence to be paid in the second calendar year by the last day of such 60-day period. The Pro-Rated Bonus will paid at the same time as annual incentive compensation payments are made to active employees for the calendar year in which the Date of Termination occurs, but in no event later than March 15 of the calendar year following the year in which it is earned.
(b) Additional Limitation.
(i) Anything in this Agreement to the contrary notwithstanding, in the event that the amount of any compensation, payment or distribution by the Company to or for the benefit of the Executive, whether paid or payable or distributed or distributable pursuant to the terms of this Agreement or otherwise, calculated in a manner consistent with Section 280G of the Code, and the applicable regulations thereunder (the “Aggregate Payments”), would be subject to the excise tax imposed by Section 4999 of the Code, then the Aggregate Payments shall be reduced (but not below zero) so that the sum of all of the Aggregate Payments shall be $1.00 less than the amount at which the Executive becomes subject to the excise tax imposed by Section 4999 of the Code; provided that such reduction shall only occur if it would result in the Executive receiving a higher After Tax Amount (as defined below) than the Executive would receive if the Aggregate Payments were not subject to such reduction.
9
In such event, the Aggregate Payments shall be reduced in the following order, in each case, in reverse chronological order beginning with the Aggregate Payments that are to be paid the furthest in time from consummation of the transaction that is subject to Section 280G of the Code: (1) cash payments not subject to Section 409A of the Code; (2) cash payments subject to Section 409A of the Code; (3) equity-based payments and acceleration; and (4) non-cash forms of benefits; provided that in the case of all the foregoing Aggregate Payments all amounts or payments that are not subject to calculation under Treas. Reg. §1.280G-1, Q&A-24(b) or (c) shall be reduced before any amounts that are subject to calculation under Treas. Reg. §1.280G-1, Q&A-24(b) or (c).
(ii) For purposes of this Section 6(b), the “After Tax Amount” means the amount of the Aggregate Payments less all federal, state, and local income, excise and employment taxes imposed on the Executive as a result of the Executive’s receipt of the Aggregate Payments. For purposes of determining the After Tax Amount, the Executive shall be deemed to pay federal income taxes at the highest marginal rate of federal income taxation applicable to individuals for the calendar year in which the determination is to be made, and state and local income taxes at the highest marginal rates of individual taxation in each applicable state and locality, net of the maximum reduction in federal income taxes which could be obtained from deduction of such state and local taxes.
(iii) The determination as to whether a reduction in the Aggregate Payments shall be made pursuant to Section 6(b)(i) shall be made by a nationally recognized accounting firm selected by the Company prior to the Change in Control (the “Accounting Firm”), which shall provide detailed supporting calculations both to the Company and the Executive within 15 business days of the Date of Termination, if applicable, or at such earlier time as is reasonably requested by the Company or the Executive. Any determination by the Accounting Firm shall be binding upon the Company and the Executive.
(c) Definitions. For purposes of this Agreement, “Change in Control” shall mean a “Sale Event” as defined in the Company’s 2021 Stock Option and Incentive Plan, as the same may be amended from time to time.
7. Section 409A.
(a) Anything in this Agreement to the contrary notwithstanding, if at the time of the Executive’s separation from service within the meaning of Section 409A of the Code, the Company determines that the Executive is a “specified employee” within the meaning of Section 409A(a)(2)(B)(i) of the Code, then to the extent any payment or benefit that the Executive becomes entitled to under this Agreement or otherwise on account of the Executive’s separation from service would be considered deferred compensation otherwise subject to the 20 percent additional tax imposed pursuant to Section 409A(a) of the Code as a result of the application of Section 409A(a)(2)(B)(i) of the Code, such payment shall not be payable and such benefit shall not be provided until the date that is the earlier of (A) six months and one day after the Executive’s separation from service, or (B) the Executive’s death. If any such delayed cash payment is otherwise payable on an installment basis, the first payment shall include a catch-up payment covering amounts that would otherwise have been paid during the six-month period but for the application of this provision, and the balance of the installments shall be payable in accordance with their original schedule.
10
(b) All in-kind benefits provided and expenses eligible for reimbursement under this Agreement shall be provided by the Company or incurred by the Executive during the time periods set forth in this Agreement. All reimbursements shall be paid as soon as administratively practicable, but in no event shall any reimbursement be paid after the last day of the taxable year following the taxable year in which the expense was incurred. The amount of in-kind benefits provided or reimbursable expenses incurred in one taxable year shall not affect the in-kind benefits to be provided or the expenses eligible for reimbursement in any other taxable year (except for any lifetime or other aggregate limitation applicable to medical expenses). Such right to reimbursement or in-kind benefits is not subject to liquidation or exchange for another benefit.
(c) To the extent that any payment or benefit described in this Agreement constitutes “non-qualified deferred compensation” under Section 409A of the Code, and to the extent that such payment or benefit is payable upon the Executive’s termination of employment, then such payments or benefits shall be payable only upon the Executive’s “separation from service.” The determination of whether and when a separation from service has occurred shall be made in accordance with the presumptions set forth in Treasury Regulation Section 1.409A-1(h).
(d) The parties intend that this Agreement will be administered in accordance with Section 409A of the Code. To the extent that any provision of this Agreement is ambiguous as to its compliance with Section 409A of the Code, the provision shall be read in such a manner so that all payments hereunder comply with Section 409A of the Code. Each payment pursuant to this Agreement or the Restrictive Covenants Agreement is intended to constitute a separate payment for purposes of Treasury Regulation Section 1.409A-2(b)(2). The parties agree that this Agreement may be amended, as reasonably requested by either party, and as may be necessary to fully comply with Section 409A of the Code and all related rules and regulations in order to preserve the payments and benefits provided hereunder without additional cost to either party.
(e) The Company makes no representation or warranty and shall have no liability to the Executive or any other person if any provisions of this Agreement are determined to constitute deferred compensation subject to Section 409A of the Code but do not satisfy an exemption from, or the conditions of, such Section.
8. Continuing Obligations.
(a) Restrictive Covenants Agreement. The Employee Confidentiality, Assignment, Nonsolicitation and Noncompetition Agreement attached hereto as Exhibit A (the “Restrictive Covenants Agreement”) remains in full force and effect. For purposes of this Agreement, the obligations in this Section 8 and those that arise in the Restrictive Covenants Agreement and any other agreement relating to confidentiality, assignment of inventions, or other restrictive covenants shall collectively be referred to as the “Continuing Obligations.” In the event that the Executive is entitled to any payments under the Restrictive Covenants Agreement, then any severance pay that the Executive receives in any calendar year pursuant to this Agreement shall be reduced by the amount of the payments (if any) that the Executive receives in the same calendar year pursuant to the Restrictive Covenants Agreement.
11
(b) Third-Party Agreements and Rights. The Executive hereby confirms that the Executive is not bound by the terms of any agreement with any previous employer or other party which restricts in any way the Executive’s use or disclosure of information, other than confidentiality restrictions (if any), or the Executive’s engagement in any business. The Executive represents to the Company that the Executive’s execution of this Agreement, the Executive’s employment with the Company and the performance of the Executive’s proposed duties for the Company will not violate any obligations the Executive may have to any such previous employer or other party. In the Executive’s work for the Company, the Executive will not disclose or make use of any information in violation of any agreements with or rights of any such previous employer or other party, and the Executive will not bring to the premises of the Company any copies or other tangible embodiments of non-public information belonging to or obtained from any such previous employment or other party.
(c) Litigation and Regulatory Cooperation. During and after the Executive’s employment, the Executive shall cooperate fully with the Company in (i) the defense or prosecution of any claims or actions now in existence or which may be brought in the future against or on behalf of the Company which relate to events or occurrences that transpired while the Executive was employed by the Company, and (ii) the investigation, whether internal or external, of any matters about which the Company believes the Executive may have knowledge or information. The Executive’s full cooperation in connection with such claims, actions or investigations shall include, but not be limited to, being available to meet with counsel to answer questions or to prepare for discovery or trial and to act as a witness on behalf of the Company at mutually convenient times. During and after the Executive’s employment, the Executive also shall cooperate fully with the Company in connection with any investigation or review of any federal, state or local regulatory authority as any such investigation or review relates to events or occurrences that transpired while the Executive was employed by the Company. The Company shall reimburse the Executive for any reasonable out-of-pocket expenses incurred in connection with the Executive’s performance of obligations pursuant to this Section 8(c).
(d) Relief. The Executive agrees that it would be difficult to measure any damages caused to the Company which might result from any breach by the Executive of the Continuing Obligations, and that in any event money damages would be an inadequate remedy for any such breach. Accordingly, the Executive agrees that if the Executive breaches, or proposes to breach, any portion of the Continuing Obligations, the Company shall be entitled, in addition to all other remedies that it may have, to an injunction or other appropriate equitable relief to restrain any such breach without showing or proving any actual damage to the Company.
9. Consent to Jurisdiction. The parties hereby consent to the jurisdiction of the state and federal courts of the Commonwealth of Massachusetts. Accordingly, with respect to any such court action, the Executive (a) submits to the exclusive personal jurisdiction of such courts; (b) consents to service of process; and (c) waives any other requirement (whether imposed by statute, rule of court, or otherwise) with respect to personal jurisdiction or service of process.
12
10. Waiver of Jury Trial. Each of the Executive and the Company irrevocably and unconditionally WAIVES ALL RIGHT TO TRIAL BY JURY IN ANY PROCEEDING (WHETHER BASED ON CONTRACT, TORT OR OTHERWISE) ARISING OUT OF OR RELATING TO THIS AGREEMENT OR THE EXECUTIVE’S EMPLOYMENT BY THE COMPANY OR ANY AFFILIATE OF THE COMPANY, INCLUDING WITHOUT LIMITATION THE EXECUTIVE’S OR THE COMPANY’S PERFORMANCE UNDER, OR THE ENFORCEMENT OF, THIS AGREEMENT.
11. Integration. This Agreement constitutes the entire agreement between the parties with respect to the subject matter hereof and supersedes all prior agreements between the parties concerning such subject matter, including the Prior Agreement, provided that the Equity Documents remain in full force and effect.
12. Withholding; Tax Effect. All payments made by the Company to the Executive under this Agreement shall be net of any tax or other amounts required to be withheld by the Company under applicable law. Nothing in this Agreement shall be construed to require the Company to make any payments to compensate the Executive for any adverse tax effect associated with any payments or benefits or for any deduction or withholding from any payment or benefit.
13. Assignment; Successors and Assigns. Neither the Executive nor the Company may make any assignment of this Agreement or any interest in it, by operation of law or otherwise, without the prior written consent of the other; provided, however, that the Company may assign its rights and obligations under this Agreement (including the Restrictive Covenants Agreement) without the Executive’s consent to any affiliate or to any person or entity with whom the Company shall hereafter effect a reorganization or consolidation, into which the Company merges or to whom it transfers all or substantially all of its properties or assets; provided, further that if the Executive remains employed or becomes employed by the Company, the purchaser or any of their affiliates in connection with any such transaction, then the Executive shall not be entitled to any payments, benefits or vesting pursuant to Section 5 or pursuant to Section 6 of this Agreement solely as a result of such transaction. This Agreement shall inure to the benefit of and be binding upon the Executive and the Company, and each of the Executive’s and the Company’s respective successors, executors, administrators, heirs and permitted assigns. In the event of the Executive’s death after the Executive’s termination of employment but prior to the completion by the Company of all payments due to the Executive under this Agreement, the Company shall continue such payments to the Executive’s beneficiary designated in writing to the Company prior to the Executive’s death (or to the Executive’s estate, if the Executive fails to make such designation).
14. Enforceability. If any portion or provision of this Agreement (including, without limitation, any portion or provision of any section of this Agreement) shall to any extent be declared illegal or unenforceable by a court of competent jurisdiction, then the remainder of this Agreement, or the application of such portion or provision in circumstances other than those as to which it is so declared illegal or unenforceable, shall not be affected thereby, and each portion and provision of this Agreement shall be valid and enforceable to the fullest extent permitted by law.
15. Survival. For the avoidance of doubt, this Agreement shall survive the termination of the Executive’s employment to the extent necessary to effectuate the terms contained herein.
13
16. Waiver. No waiver of any provision hereof shall be effective unless made in writing and signed by the waiving party. The failure of any party to require the performance of any term or obligation of this Agreement, or the waiver by any party of any breach of this Agreement, shall not prevent any subsequent enforcement of such term or obligation or be deemed a waiver of any subsequent breach.
17. Notices. Any notices, requests, demands and other communications provided for by this Agreement shall be sufficient if in writing and delivered in person or sent by a nationally recognized overnight courier service or by registered or certified mail, postage prepaid, return receipt requested, to the Executive at the last address the Executive has filed in writing with the Company or, in the case of the Company, at its main offices, attention of the Board.
18. Amendment. This Agreement may be amended or modified only by a written instrument signed by the Executive and by a duly authorized representative of the Board.
19. Effect on Other Plans and Agreements. An election by the Executive to resign for Good Reason under the provisions of this Agreement shall not be deemed a voluntary termination of employment by the Executive for the purpose of interpreting the provisions of any of the Company’s benefit plans, programs or policies. Nothing in this Agreement shall be construed to limit the rights of the Executive under the Company’s benefit plans, programs or policies except as otherwise provided in Section 8 hereof, and except that the Executive shall have no rights to any severance benefits under any Company severance pay plan, offer letter or otherwise. Except for the Restrictive Covenants Agreement, in the event that the Executive is party to an agreement with the Company providing for payments or benefits under such plan or agreement and under this Agreement, the terms of this Agreement shall govern and the Executive may receive payment under this Agreement only and not both. Further, Section 5 and Section 6 of this Agreement are mutually exclusive and in no event shall the Executive be entitled to payments or benefits pursuant to both Section 5 and Section 6 of this Agreement.
20. Governing Law. This is a Massachusetts contract and shall be construed under and be governed in all respects by the laws of the Commonwealth of Massachusetts, without giving effect to the conflict of laws principles thereof. With respect to any disputes concerning federal law, such disputes shall be determined in accordance with the law as it would be interpreted and applied by the United States Court of Appeals for the First Circuit.
21. Counterparts. This Agreement may be executed in any number of counterparts, each of which when so executed and delivered shall be taken to be an original; but such counterparts shall together constitute one and the same document.
[Signature page follows]
14

IN WITNESS WHEREOF, the parties have executed this Agreement effective on the Effective Date.
| TANGO THERAPEUTICS, INC. | ||
| By: | /s/ Alexis Borisy |
|
| Its: | Chairman | |
| EXECUTIVE | ||
| /s/ Barbara Weber Barbara Weber, MD |
||
Exhibit A
Restrictive Covenants Agreement
Exhibit 10.2
LIMITED TERM EMPLOYMENT AGREEMENT
between
Tango Therapeuitcs, Inc.
201 Brookline Ave., Suite 901
Boston, MA 02215
represented by its
authorized representative
Julie Carretero
– hereinafter referred to as the “Company” –
and
Malte Gerhard Peters
– hereinafter referred to as the “Employee” –
– hereinafter together “Parties“ and each individually “Party” –
Art. 1
Employment / Area of Responsibility
| 1. | This Employment Agreement shall commence on January 8, 2026 and shall have a limited term. |
| 2. | The Employee shall be employed by the Company as Chief Executive Officer and President. |
| 3. | The Employee is obligated to comply with the Company’s policies and directives as amended from time to time as well as the instructions of Employee’s supervisors. |
Art. 2
Place of Work
| 1. | The place of work shall be at the Employee’s residence in Munich, Germany. |
| 2. | The Company reserves the right, at its reasonable discretion, to assign the Employee to a different place of work, either temporarily or permanently. The Employee’s remuneration remains unaffected by such reassignment of the place of work. The Company’s right to direct the Employee’s place of work remains unaffected and is governed by Sec. 106 of the German Trade, Commerce and Industry Regulation Act (Gewerbeordnung – “GewO”). |
| 3. | The Employee’s duties may involve business travel and temporary stays abroad, both within Europe and in non-European countries. |
Art. 3
Working Time
| 1. | The regular weekly working time shall be 40 hours. The beginning, the end and the duration of the daily working time as well as breaks are determined in accordance with operational interests and are subject to the Company’s right to direct and issue instructions. |
| 2. | The Employee shall, in accordance with statutory provisions and upon the Company’s explicit instruction, work overtime (including work on Saturdays, Sundays and public holidays), if operational requirements so demand and if the instruction is made at the Company’s reasonable discretion. |
| 3. | In view of Employee’s higher-level work and his significant above-average fixed salary, the Employee does not expect any compensatory time off or any additional remuneration beyond the fixed annual salary pursuant to Art. 4.1 for any rendered overtime work. |
Seite 2 von 9
Art. 4
Remuneration / Expenses
| 1. | The Employee shall receive a fixed annual gross salary in the amount of EUR 616,086.00 (in words: six hundred and sixteen thousand and eighty-six EURO) for his services, payable in monthly installments, minus statutory taxes and deductions, by bank transfer to a domestic account specified by the Employee. |
| 2. | In addition, the Employee may earn a bonus for the respective fiscal year. The bonus target amount is 60% of the fixed annual salary (“Target”). The relevant targets are determined by the Company at the beginning of each fiscal year at its reasonable discretion. The achievement of these targets is also reviewed and determined by the Company’s Board of Directors after the end of the fiscal year. Further requirements and details are set out in the bonus plan applicable to the respective fiscal year, which the Company may amend and adjust for subsequent fiscal years at its reasonable discretion. |
| 3. | If the employment relationship does not exist for the full twelve months of a calendar year, the fixed annual salary shall be calculated and paid on a pro rata basis. |
| 4. | The Employee acknowledges that any further payments constitute a voluntary benefit, which is determined at the end of the respective reference period and without recognition of any legal obligation. Even if a benefit is granted more than once and unconditionally, this shall not give rise to any entitlement to continued payment of such a benefit, neither in respect of the cause or the amount, nor for the past or for the future. This provision shall not be applicable with respect to any additional payments based on an individual agreement reached by the Parties (Sec. 305b German Civil Code (Bürgerliches Gesetzbuch – “BGB”)), or which constitute consideration for the Employee’s performance. |
| 5. | Upon presentation of corresponding receipts, the Company shall reimburse the Employee for all necessary and reasonable travel costs and further expenses in accordance with the Company’s guidelines, as applicable from time to time and taking into account the applicable tax laws. |
Art. 5
Secondary Employment / Contractual Non-Compete / Conflicts of Interest
| 1. | Any additional employment (“Secondary Employment”) of the Employee, whether paid or typically paid, shall require prior approval of the Company. Such approval is also required for any uncompensated activity that may impair the interests of the Company due to its nature or scope. The Employee shall notify the Company in text form of any intended activity, specifying the location, nature, and duration. |
Seite 3 von 9
| 2. | Excluded from this approval requirement are voluntary, charitable, confessional or political activities, which are not aimed at generating income and do not impair the contractual duties and legitimate interests of the Company. Nevertheless, the Employee shall notify the Company about these activities in text form stating place, type and duration. |
| 3. | The Employee shall immediately notify the Company in text form of any subsequent changes to his Secondary Employment, unless the operational interests of the Company are obviously not affected. |
| 4. | For the duration of this Employment Agreement, the Employee shall not engage in any competition with the Company or with any affiliated company within the meaning of Secs. 15 et seq. of the German Stock Corporation Act (Aktiengesetz – “AktG”) (“Affiliated Company”) (“Contractual Non-Compete Obligation”). In particular, the Employee is prohibited from working for a competitor of the Company as an employee, freelancer, or in any other capacity, as well as from establishing such a competing business or directly or indirectly participating in such a competing company. |
Excluded from this Contractual Non-Compete Obligation is the acquisition of shares or other securities of a company that is publicly listed and whose securities are traded on a stock exchange, for the purpose of private asset management, provided that such participation does not enable the Employee to exert direct or indirect influence over the statutory bodies of the company.
| 5. | In the mutual interest, the Employee shall immediately notify the Company in text form of any potential conflicts of interest arising from his employment. The obligation to report shall not be limited to cases in which a conflict of interest may have a concrete effect on the Employee’s duties; rather, the mere appearance of a conflict of interest is sufficient. |
| 6. | The Employee confirms that he is not bound by any contractual restrictions or obligations to third parties that may limit or preclude the Employee’s ability to perform his duties and obligations under this Employment Agreement. |
Art. 6
Confidentiality Obligations
| 1. | The Employee shall maintain the strictest confidentiality regarding all trade secrets of the Company, any Affiliated Company or any third party maintaining business relations with any of the aforementioned companies. This obligation applies to both vis-à-vis third parties and vis-à-vis employees of the Company or any Affiliated Company who are not authorized to access such information. |
| 2. | Trade secrets are information that are not generally known or easily available, either as a whole or in the precise arrangement and composition of its components, to individuals who normally deal with that type of information, and which is therefore of economic value and subject of confidentiality measures by its rightful owner and where there is a legitimate interest in maintaining confidentiality. The rightful owner is the Company. |
Seite 4 von 9
Trade secrets are, in particular, but not exclusively, all information about clinical data, research, product plans, products, services, equipment, customers, markets, software, inventions, processes, designs, drawings, formulations, specifications, product configuration information, marketing and finance documents, prototypes, samples, data sets.
| 3. | This confidentiality obligation shall apply accordingly to business matters of a confidential nature that are either designated as such in writing by the Company, verbally identified as confidential, or are obviously recognizable as such. |
| 4. | Before disclosing any trade secrets or business matters of a confidential nature – e.g. for the proper performance of the work for the Company – as well as in case of doubt about the scope of this confidentiality obligation, the Employee shall be required to obtain instructions from the Company. |
| 5. | The confidentiality obligation does not apply to information that |
| (i) | was demonstrably already publicly available or otherwise generally known at the time of disclosure, |
| (ii) | was demonstrably already known to the Employee before it was disclosed to Employee by the Company, without breach of any confidentiality obligation, or |
| (iii) | was disclosed to the Employee by a third party who was not bound by a confidentiality obligation vis-à-vis the Company |
and/or its disclosure
| (i) | is made on the basis of statutory provisions or orders of government bodies, whereby the Employee shall immediately notify the Company in text form of any such disclosure, |
| (ii) | is necessary for the legitimate assertion or defence of legal claims in the context of administrative or judicial proceedings, whereby the Employee shall limit the disclosure to the absolute minimum necessary, |
| (iii) | falls under a statutory exemption from confidentiality obligations, such as any under the German Trade Secret Protection Act (Geschäftsgeheimnisgesetz – “GeschGehG”) or the German Whistleblower Protection Act (Hinweisgeberschutzgesetz – “HinSchG”) or |
| (iv) | has been expressly authorized by the Company in text form. |
| 6. | The Employee is obliged to secure all documents, records, data files (regardless of the storage medium) and devices that contain trade secrets or business matters of a confidential nature, keep them properly and protect them against unauthorized access (e.g. by means of pass codes). |
The aforementioned obligations shall also apply, mutatis mutandis, to electronically stored data.
Seite 5 von 9
| 7. | If there is a possibility that at third party has acquired knowledge of a trade secret or a confidential matter, the Employee shall notify the management immediately and at his own initiative. |
| 8. | The aforementioned obligations shall continue to apply beyond the termination of this employment relationship. To the extent that they unreasonably restrict the Employee’s professional advancement, the Employee shall have the right to request a limitation of the confidentiality obligation from the Company. |
| 9. | The Employee is aware that any violation of the above-mentioned duties shall make him liable for damages vis-à-vis the Company and possibly shall render him liable to criminal prosecution pursuant to Sec. 23 GeschGehG. |
| 10. | Further statutory duties of secrecy and confidentiality remain unaffected. |
Art. 7
Data Protection and Information Security
| 1. | The Employee shall comply with the Regulation (EU) 2016/679 (General Data Protection Regulation – “GDPR”), the German Federal Data Protection Act (Bundesdatenschutzgesetz – “BDSG”) and the Company’s internal privacy policy, as amended from time to time. |
| 2. | Any processing of personal data on behalf of the Company in connection with this employment relationship must be authorized by the Company. The Employee shall at all times ensure that unauthorized individuals do not have access to personal data. |
| 3. | The Employee shall maintain strict confidentiality regarding all personal data. In addition, the obligation to maintain confidentiality and to comply with data protection requirements pursuant to the GDPR shall apply, which constitutes an integral part of this Employment Agreement. |
| 4. | The Employee is obligated to immediately report to the Company any data protection incident or any suspected data protection incident. A data protection incident within the meaning of this provision occurs if there is a possibility that personal data, trade secrets or business matters of a confidential nature have been unlawfully disclosed, lost, altered, destroyed, or made accessible to unauthorized third parties. |
| 5. | The foregoing obligations shall continue to apply beyond the termination of the employment relationship. |
Seite 6 von 9
Art. 8
Company Property / Return Obligations
| 1. | The Employee shall, in the case of being terminated, immediately return to the Company all items and business documents, including any copies, that belong to the Company or any Affiliated Company and are in Employee’s possession |
Art. 9
Use and Exploitation Rights / Inventions
| 1. | All work results related to the Employee’s activities shall be the exclusive property of the Company. |
| 2. | Insofar as the work results are protected by copyright, the employee grants the company the exclusive, unlimited, and transferable right to use and exploit such results, including all currently known and future types of use, without any restriction in terms of time, territory, or content. This grant of rights specifically includes the right to modify the work results and to grant sublicenses to third parties. The granted rights shall remain in effect beyond the termination of the employment relationship. |
The Employee expressly waives any other rights he may have as the author or holder of other intellectual property rights in the work results, including, in particular, the right to be credited by name, the right to modify the work, and the right to make the work accessible.
The Employee shall have no claim to separate compensation for the granted rights of use and exploitation. Instead, the rights of use and exploitation are compensated by the remuneration agreed upon in Art. 4.1.
| 3. | Insofar as work results are subject to the provisions of the German Act on Employee Inventions (Arbeitnehmererfindungsgesetz – “ArbEG”), the provisions of this Act shall apply as amended from time to time. |
The Employee shall in particular immediately notify the Company in text form of any inventions made during the term of the employment relationship, provided they result from the work performed for the Company or are primarily based on operational resources, preliminary work, inventions, knowledge, know-how or other experience or work of the Company or its customer base (hereinafter referred to as “Service Inventions”).
Pursuant to the provisions of the ArbEG, the proprietary rights to Service Inventions shall transfer to the Company if it claims them. The same shall apply if the Company does not release a properly reported Service Invention to the Employee in text form within four months after receipt of the notification.
| 4. | The Employee shall immediately notify the Company of any software developments made during the term of the employment relationship, provided they result from work performed for the Company, are created following its instructions, or are based on operational resources, preliminary work, inventions, or suggestions from the Company or its customer base. The provisions of Sections 69a et seq. of the German Copyright Act (Urhebergesetz – “UrhG”) shall apply. |
Seite 7 von 9
Art. 10
Limited Term and Termination of the Employment Relationship
| 1. | This Employment Agreement shall commence on January 8, 2026 and shall expire automatically and without further notice on February 27, 2026. |
| 2. | Further, either Party may terminate this Employment Agreement anytime during its limited term subject to the statutory notice periods. |
| 3. | The right to termination without notice for compelling reason (Sec. 626 BGB) shall remain unaffected thereby. |
| 4. | For a termination to be effective, it must be in written form (Sec. 623 BGB). |
| 5. | If the Employee wants to assert that a dismissal is socially unjustified or legally invalid for other reasons or that the time limitation of the Employment Agreement is legally invalid, he must file an action with the labor court within three weeks after receipt of the written notice of dismissal or as of the agreed expiry of the employment for a declaration that the employment relationship has not been terminated by the dismissal nor the time limitation (Sec. 4 Unfair Dismissal Protection Act (Kündigungsschutzgesetz – “KSchG”), Sec. 17 Part-Time and Limited-Term Act Teilzeit- und Befristungsgesetz – “TzBfG”)). |
| 6. | If the invalidity of a termination or a time limitation is not asserted in due time, the termination or the expiry respectively is deemed to be legally effective from the outset pursuant to Sec. 7 KSchG. A belated claim for protection against dismissal or for invalidity of the time termination may only be accepted subsequently under the conditions set forth in Sec. 5 KSchG. |
Art. 11
Final Provisions
| 1. | This Employment Agreement between the Employee and the Company represents the entire and sole arrangement relating to the Employee’s occupation for the Company and shall replace or result in the termination of any prior arrangements between the Parties, whether made verbally, in writing or electronically. There are no side agreements. |
| 2. | Any amendments or additions to this Employment Agreement must be made in writing to be effective. This also applies to any waiver or modification of this written form requirement. The foregoing written form requirement shall not apply if the parties negotiate an individual agreement after the conclusion of the contract (Sec. 305b BGB). The written form requirement also applies to any claims arising from company practice (betriebliche Übung). |
| 3. | This Agreement is governed by German Law. |
Seite 8 von 9
| 4. | This Employment Agreement is executed in both German and English. Only the German version shall be authoritative and legally binding. The English version is a working translation. |
| 5. | If any provision hereof is or becomes invalid, the validity of the other provisions hereof shall not be affected thereby. In such a case, the invalid provision shall be replaced by the parties with a valid provision, which is as consistent as possible with the economic purpose of the invalid provision intended by the Parties to this Employment Agreement. This shall also apply if a provision is or becomes invalid on account of the scope or extent of an obligation or a time period. In such case, the legally admissible scope or extent of obligation or time period shall apply. The aforesaid shall apply mutatis mutandis to any gap in this Employment Agreement. |
| Tango Therapeutics, Inc. |
| /s/ Julie Carretero |
| Julie Carretero |
| Authorized Representative |
| /s/ Malte Gerhard Peters |
| Malte Gerhard Peters |
Confirmation of Receipt
By this further signature, the Employee confirms that, today, he or she has received a copy of this Employment Agreement originally signed by both Parties.
Seite 9 von 9
Exhibit 10.3

EMPLOYMENT AGREEMENT
This Employment Agreement (“Agreement”) is made between Tango Therapeutics, Inc., a Delaware corporation (the “Company”), and Malte Peters, M.D. (the “Executive”), and is effective as of the Effective Date, as defined in Section 23 herein.
WHEREAS, the Company desires to employ the Executive and the Executive desires to be employed by the Company on the new terms and conditions contained herein.
NOW, THEREFORE, in consideration of the mutual covenants and agreements herein contained and other good and valuable consideration, the receipt and sufficiency of which is hereby acknowledged, the parties agree as follows:
1. Employment.
(a) Term. The Company shall employ the Executive and the Executive shall be employed by the Company pursuant to this Agreement commencing as of the Effective Date and continuing until such employment is terminated in accordance with the provisions hereof (the “Term”). The Executive’s employment with the Company shall be “at will,” meaning that the Executive’s employment may be terminated by the Company or the Executive at any time and for any reason subject to the terms of this Agreement.
(b) Position and Duties. The Executive shall serve as the President and Chief Executive Officer of the Company (the “CEO”) and shall have such powers and duties commensurate with such position as the most senior officer of the Company as may from time to time be prescribed by the Board of Directors of the Company (the “Board”). In addition, the Company shall cause the Executive to be nominated for election to the Board and to be recommended to the stockholders for election to the Board as long as the Executive remains the CEO, provided that, the Executive shall be deemed to have resigned from the Board and from any related positions upon ceasing to serve as CEO for any reason. The Executive shall devote the Executive’s full working time and efforts to the business and affairs of the Company. Notwithstanding the foregoing, the Executive may serve on the boards of directors of other for-profit companies with the prior approval of the Board, or engage in religious, charitable or other community activities as long as such services and activities do not unreasonably interfere with the Executive’s performance of the Executive’s duties to the Company. For the avoidance of doubt, Executive shall be allowed to continue providing services to HOOKIPA Pharma Inc. during the period in which HOOKIPA Pharma Inc. is winding down its business operations.
(c) Location. The Executive’s primary work location will be in the Company’s principal office, currently located in Boston, Massachusetts; provided that the Executive will be (1) allowed to work remotely consistent with Company policy in effect from time to time and (2) required to travel for business from time to time, consistent with the Company’s business needs.
2. Compensation and Related Matters.
(a) Base Salary. The Executive’s initial base salary shall be paid at the rate of $720,000 per year. The Executive’s base salary shall be subject to periodic review and potential increase by the Board or the Compensation Committee of the Board (the “Compensation Committee”). The base salary in effect at any given time is referred to herein as “Base Salary.” The Base Salary shall be payable in a manner that is consistent with the Company’s usual payroll practices for its executive officers.
(b) Incentive Compensation. The Executive shall be eligible to receive cash incentive compensation as determined by the Board or the Compensation Committee from time to time. The Executive’s initial target annual incentive compensation shall be 60% of the Executive’s Base Salary. The target annual incentive compensation in effect at any given time is referred to herein as “Target Bonus.” The actual amount of the Executive’s annual incentive compensation, if any, shall be determined in the sole discretion of the Board or the Compensation Committee in good faith based on Company and/or individual performance objectives established by the Board or the Compensation Committee and in accordance with the terms of any applicable incentive compensation plan that may be in effect from time to time (except as set forth in this Agreement). Any annual incentive compensation will be paid no later than March 15th of the calendar year following the calendar year to which such bonus relates. Except as otherwise provided herein or as may be provided by the Board or the Compensation Committee, the Executive must be employed by the Company on the date such incentive compensation is paid in order to earn or receive any annual incentive compensation.
(c) Expenses. The Executive shall be entitled to receive prompt reimbursement for all reasonable and documented business expenses incurred by the Executive and submitted to the Company during the Term in performing services hereunder, including, notwithstanding any Company policy to the contrary, first-class airfare for any business travel on a domestic commercial flight with a scheduled flight time of two (2) hours or longer and business-class airfare for any business travel on an international commercial flight with a scheduled flight time of two (2) hours or longer, in accordance with the policies and procedures then in effect and established by the Company for its executive officers.
(d) Other Benefits. The Executive shall be eligible to participate in or receive benefits under the Company’s employee benefit plans in effect from time to time, subject to the terms of such plans. In addition, the Company shall pay the Executive (i) during the first two years of the Term once the Executive has obtained the required visa, a monthly housing allowance of $15,000 gross (subject to withholdings) and (ii) so long as the Executive or his dependents are not participating in the Company’s group health plan, at the end of each calendar year during the Term, an annual taxable stipend equal to the amount the Executive paid for his German health insurance premium in that calendar year.
(e) Paid Time Off. The Executive shall be entitled to take paid time off in accordance with the Company’s applicable paid time off policy for executives, as may be in effect from time to time.
(f) Equity.
(i) New Equity Awards. Subject to the approval of the Board or a committee thereof, the Executive will be granted new hire equity awards in the form of a non-qualified stock option to purchase 1,650,000 shares of common stock of the Company (the “Option”) at an exercise price equal to the fair market value of common stock on the date of grant and 350,000 restricted stock units (“RSUs”). The Option will vest over four years, with 25% of the shares underlying the Option vesting on the first anniversary of January 8, 2026 and the remainder vesting in 36 equal monthly installments following the one-year anniversary of the January 8, 2026, subject to the Executive’s continued employment with the Company on each such vesting date. The RSUs will vest in approximately equal annual installments over three years (in accordance with the Company’s standard RSU vesting schedule at the date of grant), subject to the Executive’s continued employment with the Company on each vesting date. The Option and the RSUs will be subject to the terms of and contingent upon the Executive’s execution of equity award agreements issued pursuant to the Company’s equity incentive plan and the applicable equity incentive plan (collectively, the “Equity Documents”). In the event of any inconsistency between this paragraph and the Equity Documents, the Equity Documents shall govern.
2
(ii) The equity awards currently held by the Executive shall continue to be governed by the terms and conditions of the Equity Documents; provided, however, and notwithstanding anything to the contrary in the Equity Documents, Sections 5(d) and 6(a) of this Agreement shall apply in the event of a termination of the Executive’s employment by the Company without Cause or by the Executive for Good Reason.
3. Termination. The Executive’s employment hereunder may be terminated without any breach of this Agreement under the following circumstances:
(a) Death. The Executive’s employment hereunder shall terminate upon death.
(b) Disability. The Company may terminate the Executive’s employment if the Executive is disabled and unable to perform or expected to be unable to perform the essential functions of the Executive’s then existing position or positions under this Agreement with or without reasonable accommodation for a period of 180 days (which need not be consecutive) in any 12-month period. If any question shall arise as to whether during any period the Executive is disabled so as to be unable to perform the essential functions of the Executive’s then existing position or positions with or without reasonable accommodation, the Executive may, and at the request of the Company shall, submit to the Company a certification in reasonable detail by a physician selected by the Company to whom the Executive or the Executive’s guardian has no reasonable objection as to whether the Executive is so disabled or how long such disability is expected to continue, and such certification shall for the purposes of this Agreement be conclusive of the issue. The Executive shall cooperate with any reasonable request of the physician in connection with such certification. If such question shall arise and the Executive shall fail to submit such certification, the Company’s determination of such issue shall be binding on the Executive. Nothing in this Section 3(b) shall be construed to waive the Executive’s rights, if any, under existing law including, without limitation, the Family and Medical Leave Act of 1993, 29 U.S.C. §2601 et seq. and the Americans with Disabilities Act, 42 U.S.C. §12101 et seq.
(c) Termination by the Company for Cause. The Company may terminate the Executive’s employment hereunder for Cause. For the purposes of this Agreement, “Cause” shall mean any of the following:
(i) the Executive’s dishonest statements or acts with respect to the Company or any affiliate of the Company, or any current or prospective customers, suppliers vendors or other third parties with which such entity does business that results in or is reasonably anticipated to result in material harm to the Company;
(ii) the Executive’s commission of (A) a felony or (B) any misdemeanor involving moral turpitude, deceit, dishonesty or fraud;
(iii) the Executive’s substantial failure to perform the Executive’s assigned material duties and responsibilities which failure continues for 30 days after written notice given to the Executive by the Board describing such failure in reasonable detail in the reasonable good faith judgment of the Board; (v) the Executive’s material violation of this Agreement, the Restrictive Covenants Agreement (as defined below), or any provision of any agreement(s) between the Executive and the Company relating to noncompetition, nonsolicitation, nondisclosure and/or assignment of inventions; or
(iv) the Executive’s gross negligence, willful misconduct or insubordination with respect to the Company that results in or is reasonably anticipated to result in material harm to the Company;
3
(vi) the Executive’s failure to cooperate with a bona fide internal investigation or an investigation by regulatory or law enforcement authorities, after being instructed by the Company to cooperate, or the willful destruction or failure to preserve documents or other materials known to be relevant to such investigation or the inducement of others to fail to cooperate or to produce documents or other materials in connection with such investigation.
(d) Termination by the Company without Cause. The Company may terminate the Executive’s employment hereunder at any time without Cause. Any termination by the Company of the Executive’s employment under this Agreement that does not constitute a termination for Cause under Section 3(c) and does not result from the death or disability of the Executive under Section 3(a) or (b) shall be deemed a termination without Cause.
(e) Termination by the Executive. The Executive may terminate employment hereunder at any time for any reason, including but not limited to, Good Reason. For the purposes of this Agreement, “Good Reason” shall mean that the Executive has completed all steps of the Good Reason Process (hereinafter defined) following the occurrence of any of the following events without the Executive’s consent (each, a “Good Reason Condition”):
(i) a material diminution in the Executive’s title, responsibilities, authority or duties;
(ii) a material diminution in the Executive’s Base Salary or a material reduction in the Executive’s benefits, except for across-the-board salary reductions based on the Company’s financial performance or a general reduction in the benefits, in each case similarly affecting all or substantially all senior management employees of the Company;
(iii) a material change in the geographic location of the principal office of the Company to which the Executive is assigned, such that there is an increase of at least 50 miles of driving distance to such location from the Executive’s principal residence as of such change, not including business travel and short-term assignments;
(iv) a material breach of this Agreement by the Company; or
(v) a failure by the Company to obtain the assumption of this Agreement by any successor to the Company.
The “Good Reason Process” consists of the following steps:
(i) the Executive reasonably determines in good faith that a Good Reason Condition has occurred;
(ii) the Executive notifies the Company in writing of the first occurrence of the Good Reason Condition within 60 days of the first occurrence of such condition;
4
(iii) the Executive cooperates in good faith with the Company’s efforts, for a period of not less than 30 days following such notice (the “Cure Period”), to remedy the Good Reason Condition;
(iv) notwithstanding such efforts, the Good Reason Condition continues to exist at the end of the Cure Period; and
(v) the Executive terminates employment within 60 days after the end of the Cure Period.
If the Company cures the Good Reason Condition during the Cure Period, Good Reason shall be deemed not to have occurred.
4. Matters related to Termination.
(a) Notice of Termination. Except for termination as specified in Section 3(a), any termination of the Executive’s employment by the Company or any such termination by the Executive shall be communicated by written Notice of Termination to the other party hereto. For the purposes of this Agreement, a “Notice of Termination” shall mean a notice which shall indicate the specific termination provision in this Agreement relied upon.
(b) Date of Termination. “Date of Termination” shall mean: (i) if the Executive’s employment is terminated by death, the date of death; (ii) if the Executive’s employment is terminated on account of disability under Section 3(b) or by the Company for Cause under Section 3(c), the date on which Notice of Termination is given; (iii) if the Executive’s employment is terminated by the Company without Cause under Section 3(d), the date on which a Notice of Termination is given or the date otherwise specified by the Company in the Notice of Termination; (iv) if the Executive’s employment is terminated by the Executive under Section 3(e) other than for Good Reason, 30 days after the date on which a Notice of Termination is given, and (v) if the Executive’s employment is terminated by the Executive under Section 3(e) for Good Reason, the date on which a Notice of Termination is given after the end of the Cure Period. Notwithstanding the foregoing, in the event that the Executive gives a Notice of Termination to the Company, the Company may unilaterally accelerate the Date of Termination and such acceleration shall not result in a termination by the Company for the purposes of this Agreement.
(c) Accrued Obligations. If the Executive’s employment with the Company is terminated for any reason, the Company shall pay or provide to the Executive (or to the Executive’s authorized representative or estate) (i) any earned but unpaid Base Salary and, if applicable, accrued but unused vacation earned through the Date of Termination; (ii) unpaid expense reimbursements (subject to, and in accordance with, Section 2(c) of this Agreement); and (iii) any vested benefits the Executive may have under any employee benefit plan of the Company through the Date of Termination, which vested benefits shall be paid and/or provided in accordance with the terms of such employee benefit plans (collectively, the “Accrued Obligations”).
(d) Resignation of All Other Positions. To the extent applicable, the Executive shall be deemed to have resigned from all officer and board member positions that the Executive holds with the Company or any of its respective subsidiaries and affiliates upon the termination of the Executive’s employment for any reason. The Executive shall execute any documents in reasonable form as may be requested by the Company to confirm or effectuate any such resignations.
5
5. Severance Pay and Benefits Upon Termination by the Company without Cause or by the Executive for Good Reason Outside the Change in Control Period. If the Executive’s employment is terminated by the Company without Cause as provided in Section 3(d), or the Executive terminates employment for Good Reason as provided in Section 3(e), in each case outside of the Change in Control Period (as defined below), then, in addition to the Accrued Obligations, and subject to (i) the Executive signing a separation agreement and release in a form and manner reasonably satisfactory to the Company (the “Release”), which shall include, without limitation, a general release of claims against the Company and all related persons and entities that shall not release the Executive’s rights under this Agreement or the other agreements referred to in this Agreement, a reaffirmation of all of the Executive’s Continuing Obligations (as defined below), and, in the Company’s sole discretion, a one-year post-employment noncompetition agreement (which shall not expand the scope of proscribed activities under any then-existing noncompetition obligations), and shall provide that if the Executive breaches any of the Continuing Obligations, all payments of the Severance Amount shall immediately cease (the “Separation Agreement”), and (ii) the Separation Agreement becoming irrevocable, all within 60 days after the Date of Termination (or such shorter period as set forth in the Separation Agreement), which shall include a seven business day revocation period:
(a) the Company shall pay the Executive an amount equal to the sum of (i) 12 months of the Executive’s Base Salary plus (ii) a prorated portion of the Target Bonus for the year of termination based on the Date of Termination (the “Prorated Bonus”) (together, the “Severance Amount”); and
(b) if the Date of Termination occurs after the end of a calendar year and the Board or the Compensation Committee, as applicable, has determined and approved annual incentive compensation pursuant to Section 2(b) but has not yet paid such annual incentive compensation, then the Company shall pay the Executive the annual incentive compensation that he otherwise would have received if he had remained employed on the date of payment (the “Prior Year Bonus”); and
(c) if the Executive is covered under the Company’s group health insurance plan as of the Date of Termination and subject to the Executive’s copayment of premium amounts at the applicable active employees’ rate and the Executive’s proper election to receive benefits under the Consolidated Omnibus Budget Reconciliation Act of 1985, as amended (“COBRA”), the Company shall pay to the group health plan provider or the COBRA provider a monthly payment equal to the monthly employer contribution that the Company would have made to provide health insurance to the Executive if the Executive had remained employed by the Company until the earliest of (A) the 12 month anniversary of the Date of Termination; (B) the date that the Executive becomes eligible for group medical plan benefits under any other employer’s group medical plan; or (C) the cessation of the Executive’s health continuation rights under COBRA; provided, however, that if the Company determines that it cannot pay such amounts to the group health plan provider or the COBRA provider (if applicable) without potentially violating applicable law (including, without limitation, Section 2716 of the Public Health Service Act), then the Company shall convert such payments to payroll payments directly to the Executive for the time period specified above. Such payments to the Executive shall be subject to tax-related deductions and withholdings and paid on the Company’s regular payroll dates; and
(d) notwithstanding anything to the contrary in any applicable option agreement or other stock-based award agreement, the portion of all time-based stock options and other stock-based awards subject solely to time-based vesting held by the Executive (the “Time-Based Equity Awards”) scheduled to vest in the 12 month period following the Date of Termination shall immediately accelerate and become fully vested and exercisable or nonforfeitable as of the later of (i) the Date of Termination or (ii) the effective date of the Release (the “Accelerated Vesting Date”), provided that in order to effectuate the accelerated vesting contemplated by this subsection, the unvested portion of the Executive’s Time-Based Equity Awards that would otherwise terminate or be forfeited on the Date of Termination will be delayed until the earlier of (A) the effective date of the Release (at which time acceleration will occur), or (B) the date that the Release can no longer become fully effective (at which time the unvested portion of the Executive’s Time-Based Equity Awards will terminate or be forfeited).
6
Notwithstanding the foregoing, no additional vesting of the Time-Based Equity Awards shall occur during the period between the Date of Termination and the Accelerated Vesting Date. With respect to any performance-based vesting equity award, such award shall continue to be governed in all respects by the terms of the Equity Documents.
(e) Except for the Prior Year Bonus, the cash amounts payable under Section 5, to the extent taxable, shall be paid out in substantially equal installments in accordance with the Company’s payroll practice over 12 months commencing within 60 days after the Date of Termination; provided, however, that if the 60-day period begins in one calendar year and ends in a second calendar year, such payments, to the extent they qualify as “non-qualified deferred compensation” within the meaning of Section 409A of the Internal Revenue Code of 1986, as amended (the “Code”), shall begin to be paid in the second calendar year by the last day of such 60-day period; provided, further, that the initial payment shall include a catch-up payment to cover amounts retroactive to the day immediately following the Date of Termination. The Prior Year Bonus will be paid on the date that the Company’s other executives receive their annual incentive compensation but no later than March 15 of the year following the year to which the Prior Year Bonus relates. Each payment pursuant to this Agreement is intended to constitute a separate payment for the purposes of Treasury Regulation Section 1.409A-2(b)(2).
(f) The Executive acknowledges and agrees that the opportunity to receive severance pay and benefits under this Section 5 or Section 6 below, as applicable, is consideration mutually agreed upon by the Company and the Executive to support the Executive’s noncompetition obligation in the Restrictive Covenants Agreement. The Executive further acknowledges and agrees that absent the Executive’s agreement to the noncompetition obligation in the Restrictive Covenants Agreement, the Company would not provide the Executive with the severance pay and benefits opportunity in Section 5 and Section 6 of this Agreement.
6. Severance Pay and Benefits Upon Termination by the Company without Cause or by the Executive for Good Reason within the Change in Control Period. The provisions of this Section 6 shall apply in lieu of, and expressly supersede, the provisions of Section 5 if (i) the Executive’s employment is terminated either (a) by the Company without Cause as provided in Section 3(d), or (b) by the Executive for Good Reason as provided in Section 3(e), and (ii) the Date of Termination is on or within 12 months after the occurrence of the first event constituting a Change in Control (such period, the “Change in Control Period”). These provisions shall terminate and be of no further force or effect after the Change in Control Period.
(a) If the Executive’s employment is terminated by the Company without Cause as provided in Section 3(d) or the Executive terminates employment for Good Reason as provided in Section 3(e) and in each case the Date of Termination occurs within the Change in Control Period, then, in addition to the Accrued Obligations, and subject to the signing of the Release by the Executive and the Release becoming fully effective, all within the time frame set forth in the Release but in no event more than 60 days after the Date of Termination:
(i) the Company shall pay the Executive a lump sum in cash in an amount equal to the sum of (A) 18 months of the Executive’s then-current Base Salary (or the Executive’s Base Salary in effect immediately prior to the Change in Control, if higher) plus (B) 1.5 times the Executive’s Target Bonus for the then-current year (or the Executive’s Target Bonus in effect immediately prior to the Change in Control, if higher) plus (C) the Prorated Bonus; and (ii) if the Date of Termination occurs after the end of a calendar year and the Board or the Compensation Committee, as applicable, has determined and approved annual incentive compensation pursuant to Section 2(b) but has not yet paid such annual incentive compensation, then the Company shall pay the Executive the Prior Year Bonus; and
7
(iii) notwithstanding anything to the contrary in any applicable option agreement or other stock-based award agreement, the Time-Based Equity Awards shall immediately accelerate and become fully vested and exercisable or nonforfeitable as of the later of (i) the Date of Termination or (ii) the effective date of the Release (the “Change in Control Accelerated Vesting Date”), provided that in order to effectuate the accelerated vesting contemplated by this subsection, the unvested portion of the Executive’s Time-Based Equity Awards that would otherwise terminate or be forfeited on the Date of Termination will be delayed until the earlier of (A) the effective date of the Release (at which time acceleration will occur), or (B) the date that the Release can no longer become fully effective (at which time the unvested portion of the Executive’s Time-Based Equity Awards will terminate or be forfeited). Notwithstanding the foregoing, no additional vesting of the Time-Based Equity Awards shall occur during the period between the Date of Termination and the Change in Control Accelerated Vesting Date. With respect to any performance-based vesting equity award, such award shall continue to be governed in all respects by the terms of the applicable Equity Documents; and
(iv) subject to the Executive’s copayment of premium amounts at the applicable active employees’ rate and the Executive’s proper election to receive benefits under COBRA, the Company shall pay to the group health plan provider or the COBRA provider a monthly payment equal to the monthly employer contribution that the Company would have made to provide health insurance to the Executive if the Executive had remained employed by the Company until the earliest of (A) the 18 month anniversary of the Date of Termination; (B) the date that the Executive becomes eligible for group medical plan benefits under any other employer’s group medical plan; or (C) the cessation of the Executive’s health continuation rights under COBRA; provided, however, that if the Company determines that it cannot pay such amounts to the group health plan provider or the COBRA provider (if applicable) without potentially violating applicable law (including, without limitation, Section 2716 of the Public Health Service Act), then the Company shall convert such payments to payroll payments directly to the Executive for the time period specified above. Such payments to the Executive shall be subject to tax-related deductions and withholdings and paid on the Company’s regular payroll dates.
Except for the Prior Year Bonus, the amounts payable under this Section 6(a), to the extent taxable, shall be paid or commence to be paid within 60 days after the Date of Termination; provided, however, that if the 60-day period begins in one calendar year and ends in a second calendar year, such payments to the extent they qualify as “non-qualified deferred compensation” within the meaning of Section 409A of the Code, shall be paid or commence to be paid in the second calendar year by the last day of such 60-day period. The Prior Year Bonus will be paid on the date that the Company’s other executives receive their annual incentive compensation but no later than March 15 of the year following the year to which the Prior Year Bonus relates.
8
(b) Additional Limitation.
(i) Anything in this Agreement to the contrary notwithstanding, in the event that the amount of any compensation, payment or distribution by the Company or any of its affiliates, or any successor or acquiror to or for the benefit of the Executive, whether paid or payable or distributed or distributable pursuant to the terms of this Agreement or otherwise, calculated in a manner consistent with Section 280G of the Code, and the applicable regulations thereunder (the “Aggregate Payments”), would be subject to the excise tax imposed by Section 4999 of the Code, then the Aggregate Payments shall be reduced (but not below zero) so that the sum of all of the Aggregate Payments shall be $1.00 less than the amount at which the Executive becomes subject to the excise tax imposed by Section 4999 of the Code; provided that such reduction shall only occur if it would result in the Executive receiving a higher After Tax Amount (as defined below) than the Executive would receive if the Aggregate Payments were not subject to such reduction. In such event, the Aggregate Payments shall be reduced in the following order, in each case, in reverse chronological order beginning with the Aggregate Payments that are to be paid the furthest in time from consummation of the transaction that is subject to Section 280G of the Code: (1) cash payments not subject to Section 409A of the Code; (2) cash payments subject to Section 409A of the Code; (3) equity-based payments and acceleration; and (4) non-cash forms of benefits; provided that in the case of all the foregoing Aggregate Payments all amounts or payments that are not subject to calculation under Treas. Reg. §1.280G-1, Q&A-24(b) or (c) shall be reduced before any amounts that are subject to calculation under Treas. Reg. §1.280G-1, Q&A-24(b) or (c).
(ii) For the purposes of this Section 6(b), the “After Tax Amount” means the amount of the Aggregate Payments less all federal, state, and local income, excise and employment taxes imposed on the Executive as a result of the Executive’s receipt of the Aggregate Payments. For the purposes of determining the After Tax Amount, the Executive shall be deemed to pay federal income taxes at the highest marginal rate of federal income taxation applicable to individuals for the calendar year in which the determination is to be made, and state and local income taxes at the highest marginal rates of individual taxation in each applicable state and locality, net of the maximum reduction in federal income taxes which could be obtained from deduction of such state and local taxes.
(iii) The determination as to whether a reduction in the Aggregate Payments shall be made pursuant to Section 6(b)(i) shall be made by a nationally recognized accounting firm selected by the Company prior to the Change in Control (the “Accounting Firm”), which shall provide detailed supporting calculations both to the Company and the Executive within 15 business days of the Date of Termination, if applicable, or at such earlier time as is reasonably requested by the Company or the Executive. Any determination by the Accounting Firm shall be binding upon the Company and the Executive.
(c) Definitions. For the purposes of this Agreement, “Change in Control” shall mean a “Sale Event” as defined in the Company’s 2021 Stock Option and Incentive Plan, as the same may be amended from time to time.
7. Section 409A.
(a) Anything in this Agreement to the contrary notwithstanding, if at the time of the Executive’s separation from service within the meaning of Section 409A of the Code, the Company determines that the Executive is a “specified employee” within the meaning of Section 409A(a)(2)(B)(i) of the Code, then to the extent any payment or benefit that the Executive becomes entitled to under this Agreement or otherwise on account of the Executive’s separation from service would be considered deferred compensation otherwise subject to the 20 percent additional tax imposed pursuant to Section 409A(a) of the Code as a result of the application of Section 409A(a)(2)(B)(i) of the Code, such payment shall not be payable and such benefit shall not be provided until the date that is the earlier of (A) six months and one day after the Executive’s separation from service, or (B) the Executive’s death. If any such delayed cash payment is otherwise payable on an installment basis, the first payment shall include a catch-up payment covering amounts that would otherwise have been paid during the six-month period but for the application of this provision, and the balance of the installments shall be payable in accordance with their original schedule.
9
(b) All in-kind benefits provided and expenses eligible for reimbursement under this Agreement shall be provided by the Company or incurred by the Executive during the time periods set forth in this Agreement. All reimbursements shall be paid as soon as administratively practicable, but in no event shall any reimbursement be paid after the last day of the taxable year following the taxable year in which the expense was incurred. The amount of in-kind benefits provided or reimbursable expenses incurred in one taxable year shall not affect the in-kind benefits to be provided or the expenses eligible for reimbursement in any other taxable year (except for any lifetime or other aggregate limitation applicable to medical expenses). Such right to reimbursement or in-kind benefits is not subject to liquidation or exchange for another benefit.
(c) To the extent that any payment or benefit described in this Agreement constitutes “non-qualified deferred compensation” under Section 409A of the Code, and to the extent that such payment or benefit is payable upon the Executive’s termination of employment, then such payments or benefits shall be payable only upon the Executive’s “separation from service.” The determination of whether and when a separation from service has occurred shall be made in accordance with the presumptions set forth in Treasury Regulation Section 1.409A-1(h).
(d) The parties intend that this Agreement will be administered in accordance with Section 409A of the Code. To the extent that any provision of this Agreement is ambiguous as to its compliance with Section 409A of the Code, the provision shall be read in such a manner so that all payments hereunder comply with Section 409A of the Code. Each payment pursuant to this Agreement or the Restrictive Covenants Agreement is intended to constitute a separate payment for the purposes of Treasury Regulation Section 1.409A-2(b)(2). The parties agree that this Agreement may be amended, as reasonably requested by either party, and as may be necessary to fully comply with Section 409A of the Code and all related rules and regulations in order to preserve the payments and benefits provided hereunder without additional cost to either party.
(e) The Company makes no representation or warranty and shall have no liability to the Executive or any other person if any provisions of this Agreement are determined to constitute deferred compensation subject to Section 409A of the Code but do not satisfy an exemption from, or the conditions of, such Section.
8. Continuing Obligations.
(a) Restrictive Covenants Agreement. As a condition of employment under the terms of this Agreement, the Executive is required to enter into the Employee Confidentiality, Assignment, Nonsolicitation and Noncompetition Agreement attached hereto as Exhibit A (the “Restrictive Covenants Agreement”). The Executive also understands and agrees that the equity, cash incentive, and severance compensation (individually and collectively, the “Noncompete Additional Consideration”) for which the Company has made Executive eligible under this Agreement constitutes (in each case and independent of the other) sufficient, mutually agreed-upon, fair and reasonable consideration for the Restrictive Covenant Agreement (including without limitation the noncompetition and nonsolicitation provisions of the Restrictive Covenant Agreement) that is independent of his employment with the Company. To avoid all doubt, the Executive agrees that his eligibility for any one of the above, or his eligibility for any combination of one or more of the above, constitutes such Additional Consideration. For the purposes of this Agreement, the obligations in this Section 8 and those that arise in the Restrictive Covenants Agreement and any other agreement relating to confidentiality, assignment of inventions, or other restrictive covenants shall collectively be referred to as the “Continuing Obligations.”
10
(b) Third-Party Agreements and Rights. The Executive hereby confirms that the Executive is not bound by the terms of any agreement with any previous employer or other party which restricts in any way the Executive’s use or disclosure of information, other than confidentiality restrictions (if any), or the Executive’s engagement in any business. The Executive represents to the Company that the Executive’s execution of this Agreement, the Executive’s employment with the Company and the performance of the Executive’s proposed duties for the Company will not violate any obligations the Executive may have to any such previous employer or other party. In the Executive’s work for the Company, the Executive will not disclose or make use of any information in violation of any agreements with or rights of any such previous employer or other party, and the Executive will not bring to the premises of the Company any copies or other tangible embodiments of non-public information belonging to or obtained from any such previous employment or other party.
(c) Litigation and Regulatory Cooperation. During and after the Executive’s employment and in all cases subject to the Company’s compliance with its indemnification obligations and with due regard for the Executive’s personal and professional commitments, the Executive shall cooperate fully with the Company in (i) the defense or prosecution of any claims or actions now in existence or which may be brought in the future against or on behalf of the Company which relate to events or occurrences that transpired while the Executive was employed by the Company, and (ii) the investigation, whether internal or external, of any matters about which the Company believes the Executive may have knowledge or information. The Executive’s full cooperation in connection with such claims, actions or investigations shall include, but not be limited to, being available to meet with counsel to answer questions or to prepare for discovery or trial and to act as a witness on behalf of the Company at mutually convenient times. During and after the Executive’s employment and in all cases subject to the Company’s compliance with its indemnification obligations and with due regard for the Executive’s personal and professional commitments, the Executive also shall cooperate fully with the Company in connection with any investigation or review of any federal, state or local regulatory authority as any such investigation or review relates to events or occurrences that transpired while the Executive was employed by the Company. The Company shall reimburse the Executive for any reasonable out-of-pocket expenses incurred in connection with the Executive’s performance of obligations pursuant to this Section 8(c) .
(d) Relief. The Executive agrees that it would be difficult to measure any damages caused to the Company which might result from any breach by the Executive of the Continuing Obligations, and that in any event money damages would be an inadequate remedy for any such breach. Accordingly, the Executive agrees that if the Executive breaches, or proposes to breach, any portion of the Continuing Obligations, the Company shall be entitled, in addition to all other remedies that it may have, to seek an injunction or other appropriate equitable relief to restrain any such breach without showing or proving any actual damage to the Company. If any action, suit, or proceeding arising out of or relating to this Agreement is brought by either the Company or Executive, the prevailing party shall be entitled to recover from the non-prevailing party its reasonable attorneys’ fees, expenses, and court costs incurred in such action (including in any appeals), in addition to any other remedies available at law, in equity, or under contract. For purposes of this Section 8(d), the “prevailing party” means the party that substantially obtains the relief sought.
9. Consent to Jurisdiction. The parties hereby consent to the jurisdiction of the state and federal courts of the Commonwealth of Massachusetts. Accordingly, with respect to any such court action, the Executive (a) submits to the exclusive personal jurisdiction of such courts; (b) consents to service of process; and (c) waives any other requirement (whether imposed by statute, rule of court, or otherwise) with respect to personal jurisdiction or service of process.
11
10. Waiver of Jury Trial. Each of the Executive and the Company irrevocably and unconditionally WAIVES ALL RIGHT TO TRIAL BY JURY IN ANY PROCEEDING (WHETHER BASED ON CONTRACT, TORT OR OTHERWISE) ARISING OUT OF OR RELATING TO THIS AGREEMENT OR THE EXECUTIVE’S EMPLOYMENT BY THE COMPANY OR ANY AFFILIATE OF THE COMPANY, INCLUDING WITHOUT LIMITATION THE EXECUTIVE’S OR THE COMPANY’S PERFORMANCE UNDER, OR THE ENFORCEMENT OF, THIS AGREEMENT.
11. Indemnification Agreement. The Company and the Executive shall enter into the Company’s form of Indemnification Agreement (the “Indemnification Agreement”), which shall also cover his acts and omissions while previously serving as a member of the Board.
12. Integration. This Agreement, together with the Equity Documents, the Restrictive Covenant Agreement and the Indemnification Agreement, constitutes the entire agreement between the parties with respect to the subject matter hereof and supersedes all prior agreements between the parties concerning such subject matter.
13. Withholding; Tax Effect. All payments made by the Company to the Executive under this Agreement shall be net of any tax or other amounts required to be withheld by the Company under applicable law. Nothing in this Agreement shall be construed to require the Company to make any payments to compensate the Executive for any adverse tax effect associated with any payments or benefits or for any deduction or withholding from any payment or benefit.
14. Assignment; Successors and Assigns. Neither the Executive nor the Company may make any assignment of this Agreement or any interest in it, by operation of law or otherwise, without the prior written consent of the other; provided, however, that the Company may assign its rights and obligations under this Agreement (including the Restrictive Covenants Agreement) without the Executive’s consent to any affiliate or to any person or entity with whom the Company shall hereafter effect a reorganization or consolidation, into which the Company merges or to whom it transfers all or substantially all of its properties or assets; provided, further that if the Executive remains employed or becomes employed by the Company, the purchaser or any of their affiliates in connection with any such transaction, then the Executive shall not be entitled to any payments, benefits or vesting pursuant to Section 5 or pursuant to Section 6 of this Agreement solely as a result of such transaction. This Agreement shall inure to the benefit of and be binding upon the Executive and the Company, and each of the Executive’s and the Company’s respective successors, executors, administrators, heirs and permitted assigns. In the event of the Executive’s death after the Executive’s termination of employment but prior to the completion by the Company of all payments due to the Executive under this Agreement, the Company shall continue such payments to the Executive’s beneficiary designated in writing to the Company prior to the Executive’s death (or to the Executive’s estate, if the Executive fails to make such designation).
15. Enforceability. If any portion or provision of this Agreement (including, without limitation, any portion or provision of any section of this Agreement) shall to any extent be declared illegal or unenforceable by a court of competent jurisdiction, then the remainder of this Agreement, or the application of such portion or provision in circumstances other than those as to which it is so declared illegal or unenforceable, shall not be affected thereby, and each portion and provision of this Agreement shall be valid and enforceable to the fullest extent permitted by law.
16. Survival. For the avoidance of doubt, this Agreement shall survive the termination of the Executive’s employment to the extent necessary to effectuate the terms contained herein.
12
17. Waiver. No waiver of any provision hereof shall be effective unless made in writing and signed by the waiving party. The failure of any party to require the performance of any term or obligation of this Agreement, or the waiver by any party of any breach of this Agreement, shall not prevent any subsequent enforcement of such term or obligation or be deemed a waiver of any subsequent breach.
18. Notices. Any notices, requests, demands and other communications provided for by this Agreement shall be sufficient if in writing and delivered in person or sent by a nationally recognized overnight courier service or by registered or certified mail, postage prepaid, return receipt requested, to the Executive at the last address the Executive has filed in writing with the Company or, in the case of the Company, at its main offices, attention of the Board. Notices, requests, demands and other communications provided for by this Agreement shall also be sufficient if sent by email to the Company email address of the Executive or, in the case of the Company, the Company email address of the Company’s Human Resources Department, with confirmation of receipt.
19. Amendment. This Agreement may be amended or modified only by a written instrument signed by the Executive and by a duly authorized representative of the Company.
20. Effect on Other Plans and Agreements. An election by the Executive to resign for Good Reason under the provisions of this Agreement shall not be deemed a voluntary termination of employment by the Executive for the purpose of interpreting the provisions of any of the Company’s benefit plans, programs or policies. Nothing in this Agreement shall be construed to limit the rights of the Executive under the Company’s benefit plans, programs or policies except as otherwise provided in Section 8 hereof, and except that the Executive shall have no rights to any severance benefits under any Company severance pay plan, offer letter or otherwise. Notwithstanding anything to the contrary in this Agreement, all severance pay and benefits provided to the Executive pursuant to Section 5 or Section 6 of this Agreement (as applicable) shall be reduced and/or offset by any amounts or benefits paid to the Executive to satisfy the federal Worker Adjustment and Retraining Notification (WARN) Act, 29 U.S.C. § 2101 et seq., as amended, and any applicable state plant or facility closing or mass layoff law (whether as damages, as payment of salary or other wages during an applicable notice period or otherwise). Except for the Restrictive Covenants Agreement, in the event that the Executive is party to an agreement with the Company providing for payments or benefits under such plan or agreement and under this Agreement, the terms of this Agreement shall govern and the Executive may receive payment under this Agreement only and not both. Further, Section 5 and Section 6 of this Agreement are mutually exclusive and in no event shall the Executive be entitled to payments or benefits pursuant to both Section 5 and Section 6 of this Agreement.
21. Governing Law. This is a Massachusetts contract and, except as set forth in Section 23, shall be construed under and be governed in all respects by the laws of the Commonwealth of Massachusetts, without giving effect to the conflict of laws principles thereof. With respect to any disputes concerning federal law, such disputes shall be determined in accordance with the law as it would be interpreted and applied by the United States Court of Appeals for the First Circuit.
22. Conditions. Notwithstanding anything to the contrary herein, the effectiveness of this Agreement shall be conditioned on the Executive’s satisfactory completion of reference and background checks (if so requested by the Company).
23. Effective Date; Pre-Work Authorization Period. The Agreement shall become effective upon Executive’s receipt of authorization to work in the United States (the “Effective Date”). Notwithstanding anything to the contrary herein, commencing on January 8, 2026 through the Effective Date: (i) the Executive will perform the services for the Company remotely from Germany, (ii) the Executive’s employment shall be governed by the terms of the Limited Term Employment Agreement, between the Executive and the Company dated January 8, 2026 (the “Limited Term Employment Agreement”) (iii) the Limited Term Employment Agreement shall automatically expire on the Effective Date; (iv) the Limited Term Agreement and this Agreement are mutually exclusive and in no event shall the Executive shall be entitled to overlapping or duplicative payments, benefits or other compensation under both the Limited Term Agreement and this Agreement.
13
24. Counterparts. This Agreement may be executed in any number of counterparts, each of which when so executed and delivered shall be taken to be an original; but such counterparts shall together constitute one and the same document.
[Signature page follows]
14

IN WITNESS WHEREOF, the parties have executed this Agreement effective on the Effective Date.
| TANGO THERAPEUTICS, INC. | ||
| By: | /s/ Alexis Borisy |
|
| Its: | Chairman | |
| EXECUTIVE | ||
| /s/ Malte Peters |
||
| Malte Peters, M.D. | ||
Exhibit A
Restrictive Covenants Agreement
Exhibit 99.1

Tango Therapeutics Announces CEO Transition: Barbara Weber to Retire, Malte Peters Appointed Successor
- Founding CEO Barbara Weber, M.D. to become executive chair of the board of directors -
- Malte Peters, M.D., current board member, appointed President and Chief Executive Officer, effective immediately –
BOSTON, January 8, 2026 (GLOBE NEWSWIRE) — Tango Therapeutics, Inc. (NASDAQ: TNGX), a clinical-stage biotechnology company committed to discovering and delivering the next generation of precision cancer medicines today announced the retirement of its Chief Executive Officer, Dr. Barbara Weber, effective today January 8, 2026. Dr. Weber, the company’s founding CEO, will become Executive Chair for 2026 and then serve as non-executive chair starting in 2027. She is succeeded by Dr. Malte Peters, a distinguished leader with extensive clinical development and leadership experience and who has served on the Tango Board of Directors since 2018, who will drive the next phase of company growth.
In her new capacity as Executive Chair, Dr. Weber will remain closely involved with the senior management team of Tango, supporting the company’s strategic priorities and working with Dr. Peters to ensure a seamless transition and continued clinical execution throughout 2026. Alexis Borisy, the current Board Chair, will transition to Lead Independent Director.
“It has been an extraordinary privilege to build and lead Tango from its inception,” said Barbara Weber, M.D., Executive Chair of Tango Therapeutics. “I am incredibly proud of what the team has accomplished in the last eight years as we leveraged rigorous science to advance our mission to improve outcomes for people with cancer. With vopimetostat now advancing into its first registrational trial and actively enrolling potentially transformative combination studies, this is the right moment for Malte’s leadership expertise and strategic vision to propel Tango into the next phase of growth.”
Dr. Weber added, “Having worked closely with Malte for over 15 years, I have seen firsthand his exceptional leadership in late-stage clinical development and his deep commitment to patients. I have the confidence and trust that bringing his extensive late-stage development and regulatory expertise to our promising clinical-stage pipeline will be an important driver of our success as we move vopimetostat into pivotal studies and ultimately commercialization.”
“Following a thorough formal search process, we are thrilled to appoint Dr. Malte Peters as president and CEO,” said Alexis Borisy, Lead Independent Director of Tango Therapeutics. “On behalf of the entire Board, I want to extend our heartfelt gratitude to Barbara for her many years of visionary leadership and unwavering dedication. Her accomplishments, especially in advancing vopimetostat to the threshold of pivotal clinical studies, have been truly transformative for the company. We look forward to her continued guidance as Executive Chair and are confident in the future of Tango under Malte’s capable leadership.”
“I am honored to join the executive team of Tango at such a pivotal time and would like to congratulate Barbara and the team on the outstanding accomplishments,” said Malte Peters, M.D., President and Chief Executive Officer of Tango Therapeutics. “I look forward to continuing to work with Barbara, as she steps into the Executive Chairman role, the Board, and our entire executive team. The progress to date is a testament to Barbara’s remarkable leadership and the dedication of the entire team. I value the trust placed in me and look forward to leading Tango as we build on this strong foundation to pursue our shared vision for the future.”
An accomplished industry veteran, Dr. Peters joins the Tango executive team with a proven track record in both early- and late-stage clinical development, having led global teams and brought innovative therapies to patients. In addition to his service on the Tango board, he was Chief Research and Development Officer at MorphoSys AG, following his tenure as Chief Development Officer and board member since March 2017, where he oversaw a large development organization and the global regulatory approval of Monjuvi in combination with lenalidomide in diffuse large B cell lymphoma. Earlier in his career, he was Global Head of Clinical Development for the Biopharmaceuticals Business Unit at Sandoz International. He also held key leadership positions at Novartis as Clinical Head and Site Head for Basel and East Hanover in Oncology Translational Medicine. Dr. Peters earned his medical degree from the Freie Universität Berlin, Germany, with medical training at the Universities of Padova, Italy, and Bochum and Berlin, Germany.
The company also renewed its previous guidance on anticipated clinical milestones in 2026:
| • | Combination trial with vopimetostat + daraxonrasib, and vopimetostat + zoldonrasib (Revolution Medicines), phase 1/2 initial safety and efficacy data 2026 |
| • | Vopimetostat monotherapy Phase 1/2 clinical data lung cancer update in 2026 |
| • | Vopimetostat monotherapy 2L pancreatic cancer pivotal study start 2026 |
| • | TNG456 monotherapy phase 1/2 trial initial safety and efficacy data 2026 |
Forward-Looking Statements
Certain statements in this press release may be considered forward-looking statements. Forward-looking statements generally relate to future events, Tango’s future operating performance and goals, the anticipated benefits of therapies and combination therapies (that include a Tango pipeline product), as well as the expectations, beliefs and development objectives for Tango’s product pipeline and clinical trials. In some cases, you can identify forward-looking statements by terminology such as “may”, “should”, “expect”, “intend”, “will”, “goal”, “estimate”, “anticipate”, “believe”, “predict”, “designed,” “potential” or “continue”, or the negatives of these terms or variations of them or similar terminology. For example, implicit or explicit statements concerning the following include or constitute forward-looking statements: Dr. Weber’s statements in this press release and statements regarding: (i) the potential of the Company’s PRMT5 molecules, as both standalone treatments and in combination with RAS(ON)-inhibitors, including our belief that vopimetostat has the potential to play a major role in treating patients with MTAP-del cancers; (ii) the preclinical research of the Company’s PRMT5 inhibitors, as a monotherapy and in combination,; (iii) expectations regarding the anticipated benefits of our molecules;; (v) beliefs regarding the ability of the ongoing vopimetostat clinical trial to inform the initiation of a registrational trial in pancreatic cancer next year and our development strategy in lung cancer; (vi) our plans and timing for combination trials, including the ongoing Phase 1/2 clinical trial of vopimetostat with each of two RAS(ON) inhibitors from Revolution Medicines; (vii) the timing of our Phase 1/2 clinical trial in TNG456;; and (ix) the expected timing of: (a) development candidate declaration for certain targets; (b) initiating IND-enabling studies; (c) filing INDs; (d) clinical trial initiation, enrollment, dose escalation and dose expansion (including for combination studies); (e) disclosing initial, interim, updated, additional and final clinical trial results (including for combination studies), including expectations to present clinical updates for vopimetostat, combination, and TNG456 data in 2026; and (f) the expected benefits of the Company’s development candidates and other product candidates. Such forward-looking statements are subject to risks, uncertainties, and other factors which could cause actual results to differ materially from those expressed or implied by such forward-looking statements. These forward-looking statements are based upon estimates and assumptions that, while considered reasonable by Tango and its management, are inherently uncertain. New risks and uncertainties may emerge from time to time, and it is not possible to predict all risks and uncertainties.
Factors that may cause actual results to differ materially from current expectations include, but are not limited to: the benefits of product candidates seen in preclinical tests and analyses may not be evident when tested in later preclinical studies or in clinical trials or when used in broader patient populations (if approved for commercial sale); Tango has limited experience conducting clinical trials (and does and will continue to rely on a third party to operate its clinical trials) and may not be able to commence its clinical trials (including opening clinical trial sites, dosing the first patient, and continued enrollment and dosing of an adequate number of clinical trial participants) when expected, may not be able to continue dosing, initiate dose escalation and/or dose expansion on anticipated timelines, and may not generate or report clinical trial results (including final, initial, interim, updated clinical trial results or additional safety and efficacy data and the establishment of proof-of-mechanism and proof-of-concept) in the anticipated timeframe (or at all); future clinical trial data releases may differ materially from initial or interim data from our current and future clinical trials; Tango’s pipeline products may not be safe and/or effective in humans; Tango has a limited operating history and has not generated any revenue to date from product sales, and may never become profitable; other companies may be able to identify and develop product candidates more quickly than the Company and commercially introduce the product prior to the Company; the Company may not be able to identify development candidates on the schedule it anticipates due to technical, financial or other reasons; the Company may not be able to file INDs for development candidates on time, or at all, due to technical or financial reasons or otherwise; the Company may utilize cash resources more quickly than anticipated; the Company will need to raise capital in the future and if we are unable to raise capital when needed or on attractive terms, we would be forced to delay, scale back or discontinue some of our development programs or future commercialization efforts (which may delay filing of INDs, dosing patients, initiation of dose expansion, reporting clinical trial results and filing new drug applications); the Company may be unable to advance our preclinical development programs into and through the clinic for safety or efficacy reasons or commercialize our product candidates or we may experience significant delays in doing so as a result of factors beyond our control; the Company may not be able to realize the benefits of orphan drug or Fast Track designation (and such designations may not advance any anticipated approval timelines); the expected benefits of our product candidates in patients as single agents and/or in combination may not be realized; the Company may experience delays or difficulties in the initiation, enrollment, or dosing of patients in clinical trials or the announcement of clinical trial results, Tango may not identify or discover additional product candidates or may expend limited resources to pursue a particular product candidate or indication and fail to capitalize on product candidates or indications that may be more profitable or for which there is a greater likelihood of success; the Company’s product candidates may cause adverse or other undesirable side effects (or may not show requisite efficacy) that could, among other things, delay or prevent regulatory approval; our dependence on one or a limited number third parties for conducting clinical trials and producing drug substance and drug product (including drug substance, which is currently sole sourced); government regulation may negatively impact the Company’s business, including the potential approval of the BIOSECURE Act; the impact of trade restrictions such as sanctions or tariffs, legal actions or enforcement and inflation rates on our business, financial condition, and results of operations; inadequate funding for or disruptions at the U.S. Food and Drug Administration or other government agencies may slow the time necessary for new drugs to be reviewed and/or approved or prevent these agencies from performing business functions on which the operation of our business may rely (which could negatively impact our business); uncertainty around the U.S. presidential administration’s approach to governmental agencies and/or product candidate approvals may present challenges for our business or create a more costly environment in which to pursue the development of new therapeutic candidates; our success depends on our ability to obtain and maintain patent and other proprietary protection for our technology and product candidates; and the scope of intellectual property protection obtained may not be sufficiently broad. Additional information concerning risks, uncertainties and assumptions can be found in Tango’s filings with the Securities and Exchange Commission (SEC), including the risk factors referenced in Tango’s Annual Report on Form 10-K for the fiscal year ended December 31, 2024, as supplemented and/or modified by its most recent Quarterly Report on Form 10-Q. You should not place undue reliance on forward-looking statements in this press release, which speak only as of the date they are made and are qualified in their entirety by reference to the cautionary statements herein. Tango specifically disclaims any duty to update these forward-looking statements.
Investors and Media:
Elizabeth Hickin
IR@tangotx.com
media@tangotx.com
Exhibit 99.2

The next wave of targeted therapies in oncology JP Morgan Healthcare Conference January 12, 2026

Disclaimer and safe harbor statement Certain statements in this presentation may be considered forward-looking statements. Forward-looking statements generally relate to future events, Tango’s future financial and operating performance, goals, expectations, beliefs, development plans, as well as development and clinical trial objectives for Tango’s product pipeline (as individual therapies and combination therapies with other party’s drugs). In some cases, you can identify forward-looking statements by terminology such as “may”, “should”, “expect”, “intend”, “will”, “path”, “achievable”, “milestones”, “goal”, “forecast”, “estimate”, “potential”, “anticipate”, “believe”, “predict”, or “continue”, or the negatives of these terms or variations of them or similar terminology. For example, express or implied statements concerning the following include or constitute forward-looking statements: the potential for the Company to have best-in-class oral PRMT5 inhibitors; the Company’s belief that it has a significant opportunity to treat multiple common cancers; the Company’s expected cash runway into 2028; the potential for vopimetostat to have best-in-class tolerability; the Company’s expectations regarding its PRMT5 inhibitors as compared to competitor molecules, including in terms of safety and tolerability; the anticipated milestones and timing for the Company’s drug programs (including registrational studies), including the timing for clinical trial initiation, enrollment, patient dosing, dose escalation, dose expansion, and clinical updates; of initial, interim, and final safety and efficacy or clinical activity data and results from clinical trial(s); the Company’s expectations regarding a registrational trial for vopimetostat; the Company’s plans for and timing of combination trials for vopimetostat and TNG456, including with RAS(ON) inhibitors for vopimetostat and with abemaciclib for TNG456; the Company’s belief that vopimetostat has the potential to transform care in front line pancreatic cancer; the Company’s expectations regarding the combination study with RAS(ON) inhibitors, including future enrollment, study design, and the ability of early signs of activity and tolerability to translate to positive results; the Company’s expectations regarding TNG456’s predicted brain exposure may not be realized; the expected benefits of the Company’s development candidates and other product candidates (including for combination studies); the Company’s expectations around the size and value of the potential patient population for PRMT5 inhibitors (including for lung and pancreatic cancers); potential combination strategies and uses for PRMT5 inhibitors, including vopimetostat and TNG456; the development plans for the PRMT5 franchise (including future single agent and combination clinical trials); future clinical trial designs; TNG260 future clinical trials strategy and implementation; expectations regarding the benefits and success of collaborations and combination clinical trials; and the anticipated benefits of its current and future product candidates; expectations around TNG456’s clinical efficacy, including its potential to treat glioblastoma and expectations around the brain exposure required for clinical efficacy; the development and regulatory pathway for vopimetostat, TNG456, or TNG961; the Company’s belief that TN961 may be a first-in-class HBS1L degrader; and the Company’s belief that there is potential for single agent and vopimetostat combination activity for TNG961. Such forward-looking statements are subject to risks, uncertainties, and other factors which could cause actual results to differ materially from those expressed or implied by such forward looking statements. These forward-looking statements are based upon estimates and assumptions that, while considered reasonable by Tango and its management at the time of this presentation, are inherently uncertain. Drug development, clinical trials and commercialization involve a high degree of risk, and only a small number of research and development programs result in commercialization of a product. New risks and uncertainties may emerge from time to time, and it is not possible to predict all risks and uncertainties. Factors that may cause actual results to differ materially from current expectations include, but are not limited to: Tango has a limited operating history and has not generated any revenue to date from drug sales, and may never become profitable; future clinical trial data releases may differ materially from initial or interim data from our current and future clinical trials; Tango has limited experience with conducting clinical trials (and does and will rely on third parties to operate its clinical trials) and may not be able to commence any clinical trial, enroll and dose patients when expected and may not generate results in the anticipated timeframe (or at all); dosing (including dose expansion) in clinical trials may need be delayed or may be stopped for various reasons, including due to any potential issues at the site, safety issues or supply disruptions; any significant changes required to be made to an applicable IND application or protocol could significantly delay on-going clinical trials); the benefits of Tango pipeline products (stand-alone and as potential combination therapies) that are seen in preclinical experiments may not be present in clinical trials or in use commercially or may not be safe and/or effective in humans (and Tango or a third-party may not be able to obtain approval or commercial sales of any stand-alone or combination therapies); Tango has incurred significant operating losses and anticipates continued losses for the foreseeable future; Tango will need to raise capital in the future and if it is unable to raise capital when needed or on attractive terms, the Company would be forced to delay, reduce, or eliminate or discontinue some development programs or future commercialization efforts; Tango may be unable to advance its preclinical development programs into and through the clinic for safety or efficacy reasons or experience significant delays in doing so as a result of factors beyond Tango’s control; the expected benefits of our product candidates in patients as single agents and/or in combination may not be realized; the Company may experience delays or difficulties in the initiation, enrollment, or dosing of patients in clinical trials or the announcement of initial, interim, or final clinical trial results; Tango’s approach to the discovery and development of product candidates is novel and unproven, which makes it difficult to predict the time, cost of development, and likelihood of successfully developing any products; Tango may not identify or discover development candidates (including next generation products) or may expend a portion of its limited resources to pursue a particular product candidate or indications and fail to capitalize on product candidates or indications that may be more profitable or for which there is a greater likelihood of success; delays or difficulties in the initiation, enrollment or dosing of patients in clinical trials could delay or prevent receipt of regulatory approvals or reporting trial results; our product candidates may cause adverse or other undesirable side effects that could, among other things, delay or prevent regulatory approval; our dependence on third parties for conducting clinical trials and producing drug product and drug substance (including the potential impact of the BIOSECURE Act on our suppliers); the impact of trade restrictions such as sanctions, tariffs, reciprocal and retaliatory tariffs, legal actions or enforcement and inflation rates on our business, financial condition, and results of operations; inadequate funding for or disruptions at the U.S. Food and Drug Administration or other government agencies may slow the time necessary for new drugs to be reviewed and/or approved or prevent these agencies from performing business functions on which the operation of our business may rely (which could negatively impact our business), and uncertainty around the U.S. presidential administration’s approach to governmental agencies and/or product candidate approvals may present challenges for our business or create a more costly environment in which to pursue the development of new therapeutic candidates; our ability to obtain and maintain patent and other intellectual property protection for our technology and product candidates or the scope of intellectual property protection obtained is not sufficiently broad; and delays and other impacts on product development and clinical trials from public health events. Additional information concerning risks, uncertainties and assumptions can be found in Tango’s filings with the SEC, including the risk factors referenced in Tango’s Annual Report on Form 10-K for the year ended December 31, 2024, as may be supplemented and/or modified by its most recent Quarterly Report on Form 10-Q. You should not place undue reliance on forward-looking statements in this presentation, which speak only as of the date they are made and are qualified in their entirety by reference to the cautionary statements herein. Tango specifically disclaims any duty to update these forward-looking statements. Certain information contained in this Presentation relates to or is based on studies, publications, surveys and Tango’s own internal estimates and research. In addition, market data included in this presentation involve assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. Finally, while Tango believes its internal research is reliable, such research has not been verified by any independent source.

Malte Peters, MD President and CEO to lead our next phase of growth Accomplished leader with deep clinical development expertise • Member of Tango Board of Directors since 2018 • Previous roles - Chief Research and Development Officer, MorphoSys AG and member of the Management Board - Global Head of Clinical Development, Biopharmaceuticals Business Unit, Sandoz International - Global Clinical Head, Oncology Translational Medicine, Novartis Oncology • Track record of innovative global regulatory approvals in particular of combination regimens
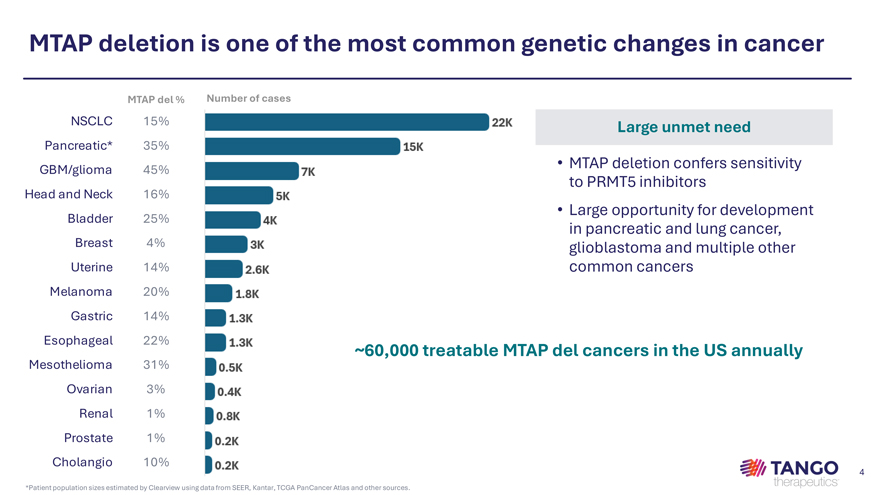
MTAP deletion is one of the most common genetic changes in cancer MTAP del % Number of cases NSCLC 15% Large unmet need Pancreatic* 35% GBM/glioma 45% • MTAP deletion confers sensitivity to PRMT5 inhibitors Head and Neck 16% • Large opportunity for development Bladder 25% in pancreatic and lung cancer, Breast 4% glioblastoma and multiple other Uterine 14% common cancers Melanoma 20% Gastric 14% Esophageal 22% Mesothelioma 31% ~60,000 treatable MTAP del cancers in the US annually Ovarian 3% Renal 1% Prostate 1%
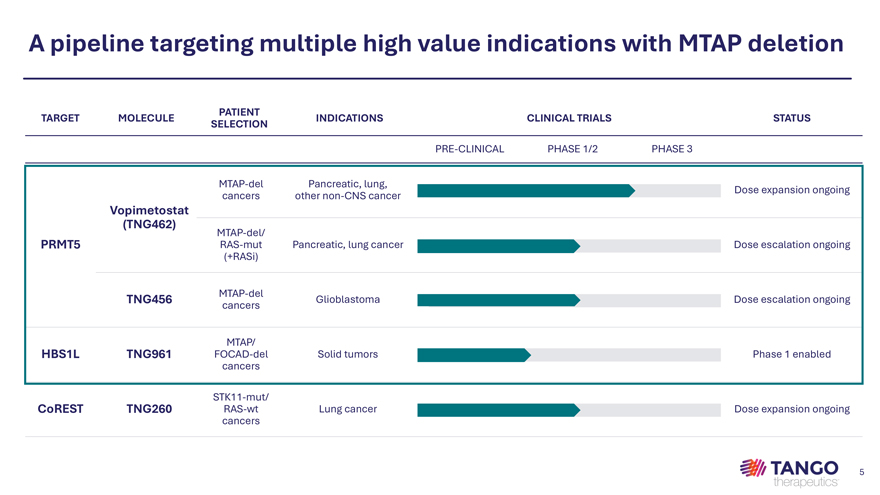
A pipeline targeting multiple high value indications with MTAP deletion PATIENT TARGET MOLECULE INDICATIONS CLINICAL TRIALS STATUS SELECTION PRE-CLINICAL PHASE 1/2 PHASE 3 MTAP-del Pancreatic, lung, Dose expansion ongoing cancers other non-CNS cancer Vopimetostat (TNG462) MTAP-del/ PRMT5 RAS-mut Pancreatic, lung cancer Dose escalation ongoing (+RASi) MTAP-del TNG456 Glioblastoma Dose escalation ongoing cancers MTAP/ HBS1L TNG961 FOCAD-del Solid tumors Phase 1 enabled cancers STK11-mut/ CoREST TNG260 RAS-wt Lung cancer Dose expansion ongoing cancers

Key 2025 achievements Advancing multiple clinical programs Creating value for patients Vopimetostat Pipeline Financial/Corporate • Clinical data supporting • TNG456 (brain penetrant • $225M equity raise vopimetostat as a potentially PRMT5 inhibitor) in phase 1/2 • $343M* cash balance as of best-in-class PRMT5 inhibitor development for MTAP-del December 31, 2025 in multiple MTAP-del cancers glioblastoma • Cash runway into 2028 • FDA supportive of pivotal study • TNG961 advanced to IND-design in 2L MTAP-del ready for multiple MTAP-pancreatic cancer del/FOCAD-del solid tumors • Vopimetostat + RAS(ON) inhibitors (Revolution Medicines) in MTAP-del/RAS-mut pancreatic and lung cancer initiated

Competitive advantage drives 2026 strategic execution Complete Expand Launch pivotal study vopimetostat/RASi vopimetostat data in Evaluate TNG456 in 2L pancreatic study to support 1L lung/other cancers efficacy in cancer pivotal study in to support additional glioblastoma pancreatic cancer pivotal studies • Planned pivotal protocol design including dose selection supported by FDA • MTAP selectivity and potency of vopimetostat provides potentially best-in-class PRMT5 suppression • Potentially best-in-class safety profile supports combinability with other molecules • First PRMT5 inhibitor clinical combination with RAS inhibitors may provide an innovative and fast path to front line approvals

Pipeline poised to deliver meaningful clinical benefit in multiple common cancers with MTAP deletion Potential best-in-class oral PRMT5 inhibitors Vopimetostat TNG456 TNG961 • Key indications* • Brain penetrant PRMT5 inhibitor • HBS1L degrader synthetic lethal being developed for glioblastoma with FOCAD deletion ~20K pancreatic cancer • 45% GBM is MTAP-del (~7K pts/yr • ~30% of MTAP-del cancers also ~22K lung cancer US) have FOCAD deletion ~20K histology selective cluster • CNS penetrance predicted in • Single agent and vopimetostat • 2L MTAP-del pancreatic ca pivotal preclinical studies combination activity in multiple study start planned 2026 preclinical models • Dose escalation ongoing • RAS(ON) inhibitor combo study • Ready for phase 1 in 2026 • Abemaciclib combo planned with ongoing evidence of single agent activity • Potential best-in-class tolerability

Vopimetostat demonstrates clear monotherapy activity across histologies Active doses >6 mo follow-up Key points n=94 • ORR 27% in 16 different histologies • mPFS 6.4 months • DCR 78% • 37/94 patients ongoing • Median follow-up 9.4 months • Good tolerability with no discontinuations for drug related events at 250 mg QD • Patients remaining on treatment

Vopimetostat mPFS in 2L pancreatic cancer more than twice historical SOC trials Key points • ORR in 2L PDAC 25% (2/8 evaluable pts) • Vopimetostat 2L mPFS 7.2 mo • SOC chemotherapy mPFS 2-3.5 mo • Vopimetostat pivotal study control arm may have lower mPFS than historical controls, increasing probability of success Recent studies show MTAP Vopimetostat mPFS 95% CI deletion confers poor prognosis Events Censored due to concurrent CDKN2A (n) (mo) (mo) deletiona 25b 12 13 7.2 2.9—NR Data extract 1 Sept 2025.

Vopimetostat pivotal trial in 2L pancreatic cancer Pivotal study start planned 2026 vopimetostat 250mg QD n~150 2L MTAP-del pancreatic cancer SOC chemotherapy n~150 Planning for 2L registration • FDA supportive of study design • Rapid enrollment of ~300 patients anticipated given high unmet medical need in this patient population

Vopimetostat + zoldonrasib has striking efficacy in preclinical models Vopimetostat + zoldonrasib Key points KP4 • Pancreatic cancer is a RAS-addicted MTAP-null, KRASG12D PDAC CDX cancer • Almost all MTAP-del pancreatic cancers have a RAS mutation • ~40% MTAP del pancreatic ca is RAS G12D mut • Similar preclinical data with daraxonrasib • Clinical collaboration with Revolution Medicines to evaluate vopimetostat + zoldonrasib (RAS G12D-selective inhibitor) and vopimetostat + daraxonrasib (RAS multi-selective inhibitor)
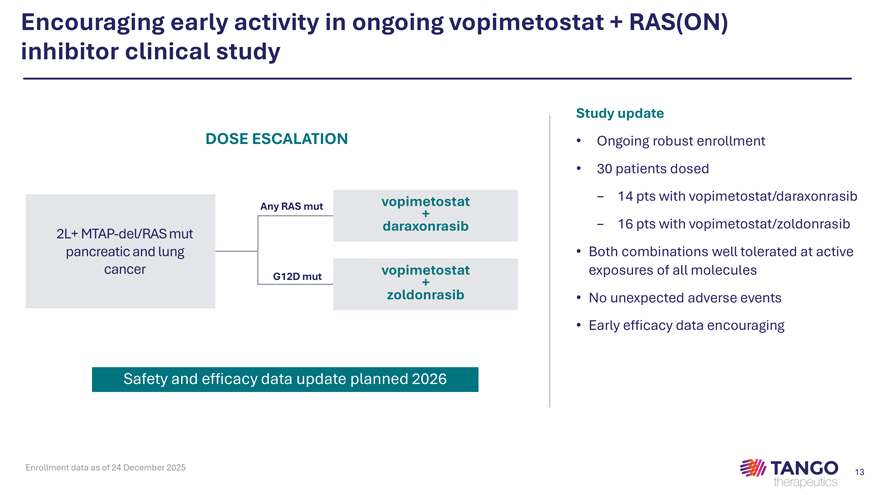
Encouraging early activity in ongoing vopimetostat + RAS(ON) inhibitor clinical study Study update DOSE ESCALATION • Ongoing robust enrollment • 30 patients dosed vopimetostat—14 pts with vopimetostat/daraxonrasib Any RAS mut + daraxonrasib—16 pts with vopimetostat/zoldonrasib 2L+ MTAP-del/RAS mut pancreatic and lung • Both combinations well tolerated at active cancer vopimetostat exposures of all molecules G12D mut + zoldonrasib • No unexpected adverse events • Early efficacy data encouraging Safety and efficacy data update planned 2026

Vopimetostat + RASi strategy for 1L pancreatic cancer RAS inhibitors likely to replace chemotherapy as SOC in 1L pancreatic cancer • More than 90% of pancreatic cancers are RAS-driven1 • Almost all MTAP-del pancreatic cancers have a RAS mutation • ~18k pts with MTAP-del and RAS-mut pancreatic cancer annually in the U.S. Potential path to vopimetostat 1L pivotal study with chemo-sparing RASi combinations • Encouraging early activity and robust enrollment in ongoing study in 2L+ pancreatic and lung cancer • Addition of 1L pancreatic cancer cohort to ongoing study planned 2026 • Phase 1/2 combo data in 2026 could support rapid move into 1L pancreatic cancer pivotal study - Single agent daraxonrasib 2L pancreatic ca ORR 29%, 1L pancreatic ca 47%, 2L+ lung ca 39%2 - Single agent zoldonrasib 2L+ pancreatic ca ORR 30%2 • First PRMT5 inhibitor being clinically evaluated in combination with RAS inhibitors

Vopimetostat 49% ORR in histology selective cohort Tumor evaluable pts at active doses with >6 months follow-up Key points (n=37) • ORR 49% • mPFS 9.1 months • DCR 89% • 21/37 patients ongoing • Median follow-up 9.5 months • Excludes sarcoma, pancreatic and lung cancer • Ongoing Cholangiocarcinoma Mesothelioma Head and neck Carcinoma of unknown primary Esophageal Other (8 histologies)

Vopimetostat 9.1 mo mPFS more than twice historical SOC trials in multiple indications mPFS (95% CI) 9.1 months (6.5 – 12.0) Durable activity in multiple late line, difficult to treat cancers provides: • Further evidence of robust single agent activity • Additional optionality for development in large patient population with high unmet need

TNG456 PRMT5 inhibition being developed for MTAP-deleted glioblastoma

TNG456 is a next-generation CNS-penetrant PRMT5 inhibitor Brain penetrance provides potential to address high unmet need in glioblastoma • Enhanced potency and MTAP selectivity • Predicted brain exposure well above efficacy threshold • In development for MTAP-del glioblastoma (7,000 patients/yr US)1 MTAP Potency CNS exposure selectivity TNG456 20 nM 55X 0.5-1X plasma Vopimetostat 4nM 45X -TNG908 110 nM 15X 0.3X plasma
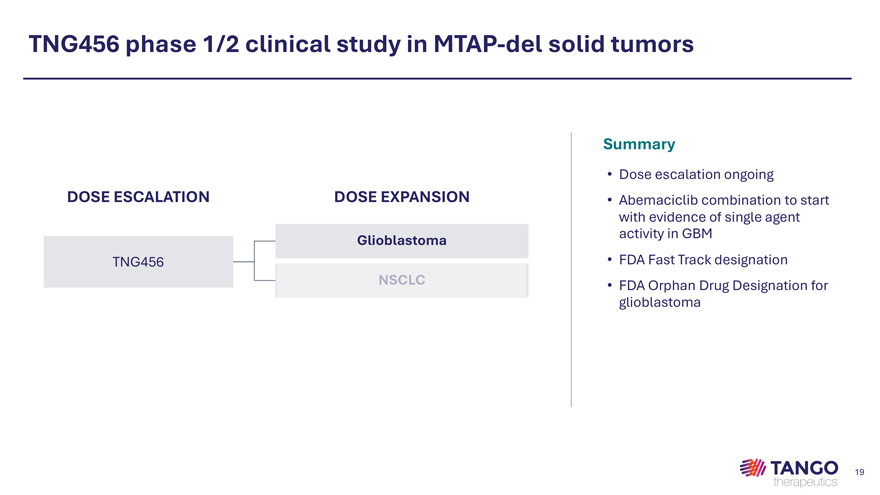
TNG456 phase 1/2 clinical study in MTAP-del solid tumors Summary • Dose escalation ongoing DOSE ESCALATION DOSE EXPANSION • Abemaciclib combination to start with evidence of single agent Glioblastoma activity in GBM TNG456 • FDA Fast Track designation • FDA Orphan Drug Designation for glioblastoma

TNG961 HBS1L degrader for FOCAD-del/MTAP-del cancers
FOCAD deletion and HBS1L are a synthetic lethal pair FOCAD deletion confers HBS1L FOCAD deletion occurs in one third of MTAP-del cancers dependency for RNA stability HBS1L RPS6 ACER2 SLC24A2 MLLT3 FOCAD TNG961 (HBS1L degrader) HACD4 IFNB1 KLHL9 MTAP Vopimetostat CDKN2A Release PELO CDKN2B ribosome DMRTA1 ELAVL2 IZUMO3 TUSC1 CAAP1 PLAA IFT74 TEK SKI Extract and EQTN complex MOB3B degrade mRNA C9orf72 FOCAD LINGO2 ACO1 DDX58 0 10 20 30 40 50 60 70 80 90 100 FOCAD co-deletion frequency with MTAP (%)

TNG961 drives tumor regression in FOCAD-del/MTAP-del preclinical models Strong xenograft activity across lineages TNG961 • Potent and selective HBS1L molecular NCI-H838 MIAPACA2 glue degrader NSCLC PDAC • IC50 110 nM 2500 ) 1500 3 3 ) • 100X selectivity for FOCAD del vs WT m m 2000 m ( ( m e e 1000 cells m 1500 u m l u vo 1000 r vol • IND-enabling studies complete with o r 500 m Tu 500 mo very clean safety profile 0 Tu 0 0 10 20 30 0 10 20 30—Starting dose within the predicted Days of treatment Days of treatment active range supported Vehicle BID Vehicle BID 2 mpk BID 6 mpk BID 6 mpk BID TNG961 TNG961 20 mpk BID 20 mpk BID 60 mpk BID 60 mpk BID

Multiple projected key milestones and strong balance sheet Clinical milestones Cash balance Vopimetostat lung ca data 2026 • $343M cash, cash equivalents and marketable Vopimetostat + daraxonrasib/zoldonrasib data securities as of December 31, 2025* 2026 • Cash runway into 2028 2L PDAC pivotal study start 2026 TNG456 data 2026

TANGO therapeutics