!
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): February 28, 2024
FULGENT GENETICS, INC.
(Exact Name of Registrant as Specified in Charter)
Delaware |
001-37894 |
81-2621304 |
|
(State or other jurisdiction of incorporation) |
(Commission File Number) |
(IRS Employer Identification No.) |
|
4399 Santa Anita Avenue El Monte, California |
91731 |
(Address of Principal Executive Offices) |
(Zip Code) |
(626) 350-0537
(Registrant’s telephone number, including area code)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
☐ |
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
☐ |
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
☐ |
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
☐ |
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
Title of each class |
|
Trading Symbol(s) |
|
Name of each exchange on which registered |
Common Stock, par value $0.0001 per share |
|
FLGT |
|
The Nasdaq Stock Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 2.02 Results of Operations and Financial Condition.
On February 28, 2024, Fulgent Genetics, Inc. (the “Company”) issued a press release announcing its financial results for the fiscal quarter and year ended December 31, 2023. A copy of the Company’s press release containing this information is being furnished as Exhibit 99.1 to this Current Report on Form 8-K.
Item 7.01 Regulation FD Disclosure.
From time to time, the Company presents and/or distributes slides and presentations to the investment community to provide updates and summaries of its business. On February 28, 2024, the Company updated its investor presentation, which is available on the Investor Relations section of the Company’s website at http://ir.fulgentgenetics.com. This presentation is also furnished as Exhibit 99.2 to this Current Report on Form 8-K.
The information in Items 2.02 and 7.01, including Exhibits 99.1 and 99.2, is being furnished and shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that Section, nor shall it be deemed incorporated by reference into any registration statement or other filing under the Securities Act of 1933, as amended, or the Exchange Act, except as shall be expressly set forth by specific reference in such filing.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits.
|
Exhibit No. |
|
Description |
99.1 |
|
Press Release of Fulgent Genetics, Inc., dated February 28, 2024 |
99.2 |
|
|
104 |
|
Cover Page Interactive Data File (embedded within the Inline XBRL document) |
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
Date: February 28, 2024 |
FULGENT GENETICS, INC. |
||
|
|
|
|
|
By: |
|
/s/ Paul Kim |
|
Name: |
|
Paul Kim |
|
Title: |
|
Chief Financial Officer |
Exhibit 99.1
Fulgent Reports Fourth Quarter and Full Year 2023 Financial Results
EL MONTE, CA, February 28, 2024 — Fulgent Genetics, Inc. (NASDAQ: FLGT) (“Fulgent,” or the “Company”), a technology-based company with a well-established laboratory services business and a therapeutic development business, today announced financial results for its fourth quarter and full year ended December 31, 2023.
Fourth Quarter 2023 Results:
Full Year 2023 Results:
Note:
1) Core Revenue excludes revenue from COVID-19 testing products and services including COVID-19 NGS testing revenue.
Non-GAAP income (loss), non-GAAP income (loss) per share, and adjusted EBITDA income (loss) are described below under “Note Regarding Non-GAAP Financial Measures” and are reconciled to the most directly comparable GAAP financial measure, GAAP income (loss), in the accompanying tables.
Ming Hsieh, Chairperson of the Board of Directors and Chief Executive Officer, said, “We are pleased with our results in 2023, a year in which we raised guidance twice and showed year-over-year growth of 44% in our core business, driven by continued momentum in Precision Diagnostics with our Beacon787 Expanded Carrier screen. We have also made good progress with our therapeutic development business, showcasing data for our lead oncology drug candidate, FID-007, at prestigious medical meetings and moving toward Phase 2 clinical testing, while also advancing our preclinical pipeline. We expect to file an Investigational New Drug (IND) application for FID-022 by the end of 2024 and are exploring potential antibody drug conjugates using our nano-drug delivery platform. I’m optimistic that 2024 will bring further progress in these areas as we serve additional patients.”
Paul Kim, Chief Financial Officer, said, “In 2023, we demonstrated efficiency in our business and superb cash management while continuing to advance our pharma pipeline and repurchase shares under our authorized stock repurchase program. We begin 2024 in a strong financial position, with record core revenues, improving core gross margin, and an enviable cash position with which to execute.”
Outlook:
For the full year 2024, Fulgent expects:
*Cash expenditures may be higher or lower than currently estimated due to a variety of facts and circumstances, including as a result of the Company’s ongoing stock repurchase program or other expenditures outside of ordinary course.
Conference Call Information
Fulgent will host a conference call for the investment community today at 8:30 AM ET (5:30 AM PT) to discuss its fourth quarter and full year 2023 results. The call may be accessed through a live audio webcast in the Investor Relations section of the Company’s website, http://ir.fulgentgenetics.com. An audio replay will be available at the same location.
Note Regarding Non-GAAP Financial Measures
Certain information set forth in this press release and/or to be discussed on the Company’s earnings call, including non-GAAP income (loss), non-GAAP income (loss) per share, non-GAAP operating margin and adjusted EBITDA income (loss) are non-GAAP financial measures. Fulgent believes this information is useful to investors because it provides a basis for measuring the performance of the Company’s business, excluding certain income or expense items that management believes are not directly attributable to the Company’s operating results. Fulgent defines non-GAAP income (loss) as net income (loss) calculated in accordance with accounting principles generally accepted in the United States of America, or GAAP, plus amortization of intangible assets, plus goodwill impairment loss, plus restructuring costs, plus acquisition-related costs, including banking fees and legal fees associated with acquisitions, plus equity-based compensation expenses, plus or minus the non-GAAP tax effect, and plus or minus other charges or gains, as identified, that management believes are not representative of the Company’s operations. For the year 2022, the non-GAAP tax effect is calculated by applying the statutory corporate tax rate on the amortization of intangible assets, restructuring costs, acquisition-related costs, and equity-based compensation expenses. For the year 2023, the non-GAAP tax effect is calculated by excluding from the GAAP provision the impact of the amortization of intangible assets, restructuring costs, acquisition-related costs, goodwill impairment loss, and equity-based compensation expenses. Fulgent defines adjusted EBITDA income (loss) as GAAP income (loss) plus or minus interest (expense) income, plus or minus provisions (benefits) for income taxes, plus restructuring costs, plus acquisition-related costs, plus equity-based compensation expenses, plus depreciation and amortization, plus goodwill impairment loss, and plus or minus other charges or gains, as identified, that management believes are not representative of the Company’s operations. Fulgent defines non-GAAP gross profit as gross profit calculated in accordance with GAAP plus equity-based compensation included in cost of revenue as shown in the table below. Fulgent defines non-GAAP gross margin by taking non-GAAP gross profit and dividing it by GAAP revenue. Fulgent defines non-GAAP operating profit (loss) by taking GAAP operating profit (loss) and adding equity-based compensation, acquisition-related costs included in operating expenses, amortization of intangible assets, restructuring costs and goodwill impairment loss. Non-GAAP operating margin is calculated by taking non-GAAP operating profit (loss) and dividing by GAAP revenue. Fulgent may continue to incur expenses similar to the items added to or subtracted from GAAP income (loss) to calculate non-GAAP income (loss) and adjusted EBITDA income (loss); accordingly, the exclusion of these items in the presentation of these non-GAAP financial measures should not be construed as an implication that these items are unusual, infrequent or non-recurring. Management uses these non-GAAP financial measures along with the most directly comparable GAAP financial measure of net income (loss) in evaluating the Company’s operating performance. Non-GAAP financial measures should not be considered in isolation from, or as a substitute for, financial information presented in conformity with GAAP, and non-GAAP financial measures as reported by Fulgent may not be comparable to similarly titled metrics reported by other companies.
About Fulgent
Fulgent is a technology-based company with a well-established laboratory services business and a therapeutic development business. Fulgent’s laboratory services business—to which was formerly referred as the clinical diagnostic business, includes technical laboratory services and professional interpretation of laboratory results by licensed physicians. Fulgent’s therapeutic development business is focused on developing drug candidates for treating a broad range of cancers using a novel nanoencapsulation and targeted therapy platform designed to improve the therapeutic window and pharmacokinetic profile of new and existing cancer drugs.
The Company aims to transform from a genomic diagnostic business into a fully integrated precision medicine company.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Examples of forward-looking statements in this press release include statements about, among other things: future performance; guidance regarding expected quarterly and annual financial results, core revenues, GAAP loss, non-GAAP loss, and cash, cash equivalents and investments in marketable securities; evaluations and judgments regarding the stability of certain revenue sources, the Company’s cash position and sufficiency of its resources, momentum, trajectory, vision, future opportunities and future growth of the Company’s testing services and technologies and expansion, including its Beacon Expanded Carrier screen; the Company’s research and development efforts, including any implications that the results of earlier clinical trials will be representative or consistent with later clinical trials, the expected timing of enrollment for these trials and the availability of data or results of these trials, including any implication that interim or preliminary data will be representative of final data; the Company’s identification and evaluation of opportunities and its ability to capitalize on opportunities, capture market share, or expand its presence in certain markets; and the Company’s ability to continue to grow its business.
Forward-looking statements are statements other than historical facts and relate to future events or circumstances or the Company’s future performance, and they are based on management’s current assumptions, expectations, and beliefs concerning future developments and their potential effect on the Company’s business. These forward-looking statements are subject to a number of risks and uncertainties, which may cause the forward-looking events and circumstances described in this press release to not occur, and actual results to differ materially and adversely from those described in or implied by the forward-looking statements. These risks and uncertainties include, among others: the market potential for, and the rate and degree of market adoption of, the Company’s tests, including its Beacon787 panel; its ability to maintain turnaround times and otherwise keep pace with rapidly changing technology; the Company’s ability to maintain the low internal costs of its business model; the Company’s ability to maintain an acceptable margin; risks related to volatility in the Company’s results, which can fluctuate significantly from period to period; risks associated with the composition of the Company’s customer base, which can fluctuate from period to period and can be comprised of a small number of customers that account for a significant portion of the Company’s revenue; the Company’s level of success in obtaining coverage and adequate reimbursement and collectability levels from third-party payors for its tests and testing services; the Company’s level of success in establishing and obtaining the intended benefits from partnerships, strategic investments, joint ventures, acquisitions, or other relationships; the success of the Company’s development efforts, including the Company’s ability to progress its candidates through clinical trials on the timelines expected; the Company’s compliance with the various evolving and complex laws and regulations applicable to its business and its industry; and the Company’s ability to protect its proprietary technology and intellectual property. As a result of these risks and uncertainties, forward-looking statements should not be relied on or viewed as predictions of future events.
The forward-looking statements made in this press release speak only as of the date of this press release, and the Company assumes no obligation to update publicly any such forward-looking statements to reflect actual results or to changes in expectations, except as otherwise required by law.
The Company’s reports filed with the U.S. Securities and Exchange Commission, or the SEC, including its annual report on Form 10-K for the fiscal year ended December 31, 2022, filed with the SEC on February 28, 2023, and the other reports it files from time to time, including subsequently filed annual, quarterly and current reports, are made available on the Company’s website upon their filing with the SEC. These reports contain more information about the Company, its business and the risks affecting its business, as well as its results of operations for the periods covered by the financial results included in this press release.
Investor Relations Contact:
The Blueshirt Group
Melanie Solomon, melanie@blueshirtgroup.com
FULGENT GENETICS, INC. |
|
|
|
|
|
|
|
Condensed Consolidated Balance Sheet Data |
|
|
|
|
|
|
|
December 31, 2023 and 2022 |
|
|
|
|
|
|
|
(in thousands) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
December 31, |
|
|||||
|
2023 |
|
|
2022 |
|
||
ASSETS: |
|
|
|
|
|
|
|
Cash and cash equivalents |
$ |
97,473 |
|
|
$ |
79,506 |
|
Investments in marketable securities |
|
750,252 |
|
|
|
773,377 |
|
Accounts receivable, net |
|
51,132 |
|
|
|
52,749 |
|
Property, plant, and equipment, net |
|
83,464 |
|
|
|
81,353 |
|
Other assets |
|
253,007 |
|
|
|
399,068 |
|
Total assets |
$ |
1,235,328 |
|
|
$ |
1,386,053 |
|
LIABILITIES & EQUITY: |
|
|
|
|
|
|
|
Accounts payable, accrued liabilities and other liabilities |
$ |
102,042 |
|
|
$ |
116,178 |
|
Total stockholders’ equity |
|
1,133,286 |
|
|
|
1,269,875 |
|
Total liabilities & equity |
$ |
1,235,328 |
|
|
$ |
1,386,053 |
|
FULGENT GENETICS, INC. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Condensed Consolidated Statement of Operations Data |
|
|
|
|
|
|
|
|
|
||||||
Three and Twelve Months Ended December 31, 2023 and 2022 |
|
|
|
|
|
|
|
|
|
|
|
|
|
||
(in thousands, except per share data) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(unaudited) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended December 31, |
|
|
Twelve Months Ended December 31, |
|
||||||||||
|
2023 |
|
|
2022 |
|
|
2023 |
|
|
2022 |
|
||||
Revenue |
$ |
70,505 |
|
|
$ |
67,704 |
|
|
$ |
289,213 |
|
|
$ |
618,968 |
|
Cost of revenue (1) |
|
45,276 |
|
|
|
54,717 |
|
|
|
184,757 |
|
|
|
252,067 |
|
Gross profit |
|
25,229 |
|
|
|
12,987 |
|
|
|
104,456 |
|
|
|
366,901 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Research and development (1) |
|
11,952 |
|
|
|
8,509 |
|
|
|
41,440 |
|
|
|
28,910 |
|
Selling and marketing (1) |
|
10,500 |
|
|
|
10,253 |
|
|
|
41,467 |
|
|
|
38,918 |
|
General and administrative (1) |
|
31,706 |
|
|
|
28,793 |
|
|
|
88,999 |
|
|
|
111,074 |
|
Amortization of intangible assets |
|
1,958 |
|
|
|
2,010 |
|
|
|
7,845 |
|
|
|
6,497 |
|
Goodwill impairment loss |
|
120,234 |
|
|
|
— |
|
|
|
120,234 |
|
|
|
— |
|
Restructuring costs |
|
— |
|
|
|
(26 |
) |
|
|
— |
|
|
|
2,975 |
|
Total operating expenses |
|
176,350 |
|
|
|
49,539 |
|
|
|
299,985 |
|
|
|
188,374 |
|
Operating (loss) income |
|
(151,121 |
) |
|
|
(36,552 |
) |
|
|
(195,529 |
) |
|
|
178,527 |
|
Interest and other income, net |
|
5,925 |
|
|
|
3,090 |
|
|
|
21,444 |
|
|
|
5,498 |
|
(Loss) income before income taxes |
|
(145,196 |
) |
|
|
(33,462 |
) |
|
|
(174,085 |
) |
|
|
184,025 |
|
(Benefit from) provision for income taxes |
|
(10,862 |
) |
|
|
(9,386 |
) |
|
|
1,154 |
|
|
|
42,102 |
|
Net (loss) income from consolidated operations |
|
(134,334 |
) |
|
|
(24,076 |
) |
|
|
(175,239 |
) |
|
|
141,923 |
|
Net loss attributable to noncontrolling interests |
|
6,185 |
|
|
|
244 |
|
|
|
7,414 |
|
|
|
1,480 |
|
Net (loss) income attributable to Fulgent |
$ |
(128,149 |
) |
|
$ |
(23,832 |
) |
|
$ |
(167,825 |
) |
|
$ |
143,403 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net (loss) income per common share attributable to Fulgent: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic |
$ |
(4.30 |
) |
|
$ |
(0.80 |
) |
|
$ |
(5.63 |
) |
|
$ |
4.76 |
|
Diluted |
$ |
(4.30 |
) |
|
$ |
(0.80 |
) |
|
$ |
(5.63 |
) |
|
$ |
4.63 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average common shares: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic |
|
29,771 |
|
|
|
29,625 |
|
|
|
29,784 |
|
|
|
30,097 |
|
Diluted |
|
29,771 |
|
|
|
29,625 |
|
|
|
29,784 |
|
|
|
30,964 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1) Equity-based compensation expense was allocated as follows: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of revenue |
$ |
2,375 |
|
|
$ |
2,521 |
|
|
$ |
9,749 |
|
|
$ |
8,704 |
|
Research and development |
|
3,973 |
|
|
|
3,339 |
|
|
|
14,873 |
|
|
|
10,449 |
|
Selling and marketing |
|
1,320 |
|
|
|
1,225 |
|
|
|
4,964 |
|
|
|
4,373 |
|
General and administrative |
|
3,764 |
|
|
|
2,937 |
|
|
|
13,336 |
|
|
|
9,114 |
|
Total equity-based compensation expense |
$ |
11,432 |
|
|
$ |
10,022 |
|
|
$ |
42,922 |
|
|
$ |
32,640 |
|
FULGENT GENETICS, INC. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Non-GAAP Income (Loss) Reconciliation |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three and Twelve Months Ended December 31, 2023 and 2022 |
|
|
|
|
|
|
|
|
|
||||||
(in thousands, except per share data) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended December 31, |
|
|
Twelve Months Ended December 31, |
|
||||||||||
|
2023 |
|
|
2022 |
|
|
2023 |
|
|
2022 |
|
||||
Net (loss) income attributable to Fulgent |
$ |
(128,149 |
) |
|
$ |
(23,832 |
) |
|
$ |
(167,825 |
) |
|
$ |
143,403 |
|
Amortization of intangible assets |
|
1,958 |
|
|
|
2,010 |
|
|
|
7,845 |
|
|
|
6,497 |
|
Goodwill impairment loss |
|
120,234 |
|
|
|
— |
|
|
|
120,234 |
|
|
|
— |
|
Restructuring costs |
|
— |
|
|
|
(26 |
) |
|
|
— |
|
|
|
2,975 |
|
Acquisition-related costs |
|
— |
|
|
|
1,359 |
|
|
|
— |
|
|
|
7,934 |
|
Equity-based compensation expense |
|
11,432 |
|
|
|
10,022 |
|
|
|
42,922 |
|
|
|
32,640 |
|
Non-GAAP tax effect (1) |
|
2,794 |
|
|
|
(3,742 |
) |
|
|
(15,473 |
) |
|
|
(14,013 |
) |
Non-GAAP (loss) income attributable to Fulgent |
$ |
8,269 |
|
|
$ |
(14,209 |
) |
|
$ |
(12,297 |
) |
|
$ |
179,436 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net (loss) income per common share attributable to Fulgent: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic |
$ |
(4.30 |
) |
|
$ |
(0.80 |
) |
|
$ |
(5.63 |
) |
|
$ |
4.76 |
|
Diluted |
$ |
(4.30 |
) |
|
$ |
(0.80 |
) |
|
$ |
(5.63 |
) |
|
$ |
4.63 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Non-GAAP (loss) income per common share attributable to Fulgent: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic |
$ |
0.28 |
|
|
$ |
(0.48 |
) |
|
$ |
(0.41 |
) |
|
$ |
5.96 |
|
Diluted |
$ |
0.28 |
|
|
$ |
(0.48 |
) |
|
$ |
(0.41 |
) |
|
$ |
5.79 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average common shares: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic |
|
29,771 |
|
|
|
29,625 |
|
|
|
29,784 |
|
|
|
30,097 |
|
Diluted |
|
29,771 |
|
|
|
29,625 |
|
|
|
29,784 |
|
|
|
30,964 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1) Tax rates as follows: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Corporate tax rate of 28% for the three and twelve months ended December 31, 2022. During the three and twelve months ended December 31, 2023, the Company maintained a valuation allowance for deferred tax assets. |
|
||||||||||||||
FULGENT GENETICS, INC. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Non-GAAP Adjusted EBITDA Reconciliation |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three and Twelve Months Ended December 31, 2023 and 2022 |
|
|
|
|
|
|
|
|
|
||||||
(in thousands) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended December 31, |
|
|
Twelve Months Ended December 31, |
|
||||||||||
|
2023 |
|
|
2022 |
|
|
2023 |
|
|
2022 |
|
||||
Net (loss) income attributable to Fulgent |
$ |
(128,149 |
) |
|
$ |
(23,832 |
) |
|
$ |
(167,825 |
) |
|
$ |
143,403 |
|
Interest income, net |
|
(5,947 |
) |
|
|
(3,023 |
) |
|
|
(21,124 |
) |
|
|
(4,610 |
) |
(Benefit from) provision for income taxes |
|
(10,862 |
) |
|
|
(9,386 |
) |
|
|
1,154 |
|
|
|
42,102 |
|
Goodwill impairment loss |
|
120,234 |
|
|
|
— |
|
|
|
120,234 |
|
|
|
— |
|
Restructuring costs |
|
— |
|
|
|
(26 |
) |
|
|
— |
|
|
|
2,975 |
|
Acquisition-related costs |
|
— |
|
|
|
1,359 |
|
|
|
— |
|
|
|
7,934 |
|
Equity-based compensation expense |
|
11,432 |
|
|
|
10,022 |
|
|
|
42,922 |
|
|
|
32,640 |
|
Depreciation and amortization |
|
6,533 |
|
|
|
9,802 |
|
|
|
26,143 |
|
|
|
32,662 |
|
Adjusted EBITDA |
$ |
(6,759 |
) |
|
$ |
(15,084 |
) |
|
$ |
1,504 |
|
|
$ |
257,106 |
|
FULGENT GENETICS, INC. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Non-GAAP Operating Margin |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three and Twelve Months Ended December 31, 2023 and 2022 |
|
|
|
|
|
||||||||||
(in thousands) |
Three Months Ended December 31, |
|
|
Twelve Months Ended December 31, |
|
||||||||||
|
2023 |
|
|
2022 |
|
|
2023 |
|
|
2022 |
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Revenue |
$ |
70,505 |
|
|
$ |
67,704 |
|
|
$ |
289,213 |
|
|
$ |
618,968 |
|
Cost of revenue |
|
45,276 |
|
|
|
54,717 |
|
|
|
184,757 |
|
|
|
252,067 |
|
Gross profit |
|
25,229 |
|
|
|
12,987 |
|
|
|
104,456 |
|
|
|
366,901 |
|
Gross margin |
|
35.8 |
% |
|
|
19.2 |
% |
|
|
36.1 |
% |
|
|
59.3 |
% |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Equity-based compensation included in cost of revenue |
|
2,375 |
|
|
|
2,521 |
|
|
|
9,749 |
|
|
|
8,704 |
|
Non-GAAP gross profit |
|
27,604 |
|
|
|
15,508 |
|
|
|
114,205 |
|
|
|
375,605 |
|
Non-GAAP gross margin |
|
39.2 |
% |
|
|
22.9 |
% |
|
|
39.5 |
% |
|
|
60.7 |
% |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating expenses |
|
176,350 |
|
|
|
49,539 |
|
|
|
299,985 |
|
|
|
188,374 |
|
Equity-based compensation included in operating expenses |
|
9,057 |
|
|
|
7,501 |
|
|
|
33,173 |
|
|
|
23,936 |
|
Acquisition-related costs included in operating expenses |
|
— |
|
|
|
1,359 |
|
|
|
— |
|
|
|
7,934 |
|
Amortization of intangible assets |
|
1,958 |
|
|
|
2,010 |
|
|
|
7,845 |
|
|
|
6,497 |
|
Restructuring costs |
|
— |
|
|
|
(26 |
) |
|
|
— |
|
|
|
2,975 |
|
Goodwill impairment loss |
|
120,234 |
|
|
|
— |
|
|
|
120,234 |
|
|
|
— |
|
Non-GAAP operating expenses |
|
45,101 |
|
|
|
38,695 |
|
|
|
138,733 |
|
|
|
147,032 |
|
Non-GAAP operating (loss) profit |
$ |
(17,497 |
) |
|
$ |
(23,187 |
) |
|
$ |
(24,528 |
) |
|
$ |
228,573 |
|
Non-GAAP operating margin |
|
-24.8 |
% |
|
|
-34.2 |
% |
|
|
-8.5 |
% |
|
|
36.9 |
% |

Founded in 2011 | Located in El Monte, CA | NASDAQ:FLGT Investor Presentation February 28, 2024 Exhibit 99.2

Disclaimer Forward-Looking Statements and Market Data This presentation contains forward-looking statements, which are statements other than those of historical facts and which represent the estimates and expectations of Fulgent Genetics, Inc. (“Fulgent” or the “Company”) about future events based on current views and assumptions. Examples of forward-looking statements made in this presentation include, among others, those related to long-term upside or value, management of risk, anticipated growth and positioning, addressable market estimates, the Company’s mission, vision and strategies, the success of its business model and strategy, anticipated future revenue and guidance, evaluations and judgments regarding the Company’s business, products, technologies, competitive landscape, scalability, plans regarding development and launch of potential future products, and any businesses the Company may seek to acquire or has acquired or has invested in or may seek to invest in, including statements regarding Fulgent Pharma Holdings, Inc. (“Fulgent Pharma”), Inform Diagnostics, CSI Laboratories, and any potential synergies, or transformation of the Company’s business, long-term visions and strategies, including, with respect to Fulgent Pharma, those designated to create a vertically integrated solution for cancer care, the clinical development of Fulgent Pharma’s pipeline and related statements and assumptions regarding development timelines, any potentially accelerated pathway for regulatory approval, the potential safety and efficacy of the nanodrug delivery platform and any related therapeutic candidates, the potential market size for these candidates and platforms and the value of available data, including genomic data, the Company’s research and development efforts, including any implications that the results of earlier clinical trials will be representative or consistent with later clinical trials, the expected timing or timing of enrollment for these clinical trials or that interim or preliminary data will be representative of the final data or results of these trials, and guidance regarding the Company’s future performance and results of operations, including any cash or cash equivalent resource projections. The Company’s views and assumptions on which these forward-looking statements are based may prove to be incorrect. As a result, matters discussed in any forward-looking statements are subject to risks, uncertainties and changes in circumstances that may cause actual results to differ materially from those discussed or implied by any forward-looking statements. Important factors that could cause actual results to differ materially from those implied by forward-looking statements are disclosed under “Risk Factors” and “Management's Discussion and Analysis of Financial Condition and Results of Operations” in the Company’s reports filed with the Securities and Exchange Commission ("SEC"), including its annual report on Form 10-K filed on February 28, 2023, and other reports it files from time to time. Because of these factors, you should not rely upon forward-looking statements as predictions of future events. The forward-looking statements in this presentation are made only as of the date hereof, and, except as required by law, the Company assumes no obligation to update any forward-looking statements in the future. The company’s reports filed with the SEC, including its annual report on Form 10-K for the year ended December 31, 2022, filed with the SEC on February 28, 2023, and the other reports it files from time to time, including subsequently filed annual, quarterly and current reports, are made available on the company’s website upon their filing with the SEC. These reports contain more information about the company, its business and the risks affecting its business, as well as its results of operations for the periods covered by the financial results included in this presentation. This presentation also includes market data and forecasts with respect to the industry in which the Company operates. In some cases, the Company relies upon and refers to market data and certain industry forecasts that have been obtained from third-party surveys, market research, consultant surveys, publicly available information and industry publications that the Company believes to be reliable. These data and estimates involve a number of assumptions and limitations, and you are cautioned not to give undue weight to such estimates. Non-GAAP Financial Measures This presentation contains certain supplemental financial measures that are not calculated pursuant to U.S. generally accepted accounting principles (“GAAP”). These non-GAAP measures are in addition to, not a substitute for or superior to, measures of financial performance prepared in accordance with GAAP. A reconciliation of non-GAAP measures to GAAP measures is contained in this presentation.

Leadership Team Esteemed background in molecular science and pathology Most recently Chief Medical Officer at NeoGenomics Laboratory, Inc.; prior senior role at Clarient, Inc. Chairman Emeritus of Pathology at City of Hope National Medical Center Brandon Perthuis Chief Commercial Officer Dr. Lawrence Weiss Chief Medical Officer Ming Hsieh Chief Executive Officer Dr. Harry Gao Lab Director and Chief Scientific Officer James Xie President and Chief Operating Officer Paul Kim Chief Financial Officer Extensive experience leading genetic testing commercialization programs since 2003 Previously VP of Sales and Marketing of the Medical Genetics Laboratory at Baylor College of Medicine Prior to Baylor, held senior roles at PerkinElmer, Inc. and Spectral Genomics, Inc. B.S. in Biomedical Science Responsible for managing all global operations, product vision and product engineering Served as an SVP of Cogent Systems, Inc. B.A. in Engineering, M.S. in Industrial Engineering and an M.S. in Computer Science Experienced financial leader and Certified Public Accountant Previously CFO of Cogent Systems, Inc.; sold to 3M for $943M in 2010 B.A. in Economics from University of California at Berkeley Previously Lab Director at City of Hope Clinical molecular genetics training fellowship and post-doctoral fellowship at Harvard Medical School M.S. in Immunology, and M.D. and Ph.D. in Microbiology, Immunology, and Medical Genetics Experienced operational leader, entrepreneur and philanthropist Previously CEO, President, and Chairman of Cogent Systems, Inc. Member of the National Academy of Engineering; Fellow of the National Academy of Inventors; Trustee of USC Dr. Ray Yin President, Pharma Founder & CEO, ANP Technologies, Inc. Former Team Leader of Nanobiotechnology for Chem/Bio Defense, U.S. Army Research Laboratory Holder of 46 drug delivery/detection patents

About Fulgent We are a premier global, technology-based genetic testing company focused on transforming patient care in oncology, infectious and rare diseases, and reproductive health. Mission Develop flexible and affordable diagnostics and therapeutics that improve the everyday lives of those around us. Core Values Innovation Customer Service and Commitment Quality and Efficiency Our People Strategy Leverage our proprietary technology platform for broad application Further clinical/regulatory program for Pharma Operational excellence Disciplined M&A

Strategic Vision – A One-Stop Solution for Cancer Care VISION Leading Genetic Testing Company Offering Tech-Enabled Diagnostic Solutions Therapeutic and Diagnostic Entity Providing Comprehensive Solutions Across the Cancer Care Continuum Vertically integrated “one-stop” solution across the healthcare chain following the CSI, Inform Diagnostics, and Pharma acquisitions Proprietary nano drug delivery technology platform serves as an underpinning technology between diagnostic and pharm to help create a more sustainable and profitable business model in precision medicine for years to come Addition of a talented scientific team creates a strong synergy and competitive advantage that may be leveraged across the combined business Potential near-term opportunity includes shortened 505(b)(2) drug development and commercialization timelines and potential long-term opportunity leverages large data insights and novel analytical tools from diagnostics business to enable additional precision medicine pipeline through organic or partnered development strategies Commitment to continue growing diagnostic and therapeutic opportunities through organic investments and M&A Seasoned management team along with strong cash position allow Fulgent to enter therapeutic opportunities while managing risk To build a vertically integrated solution to combat cancer early detection | clinical diagnostics | post treatment monitoring | drug discovery and cancer treatment Exciting Cancer Therapeutic Opportunity Realizing Precision Medicine Potential Nano-Drug Delivery Platform
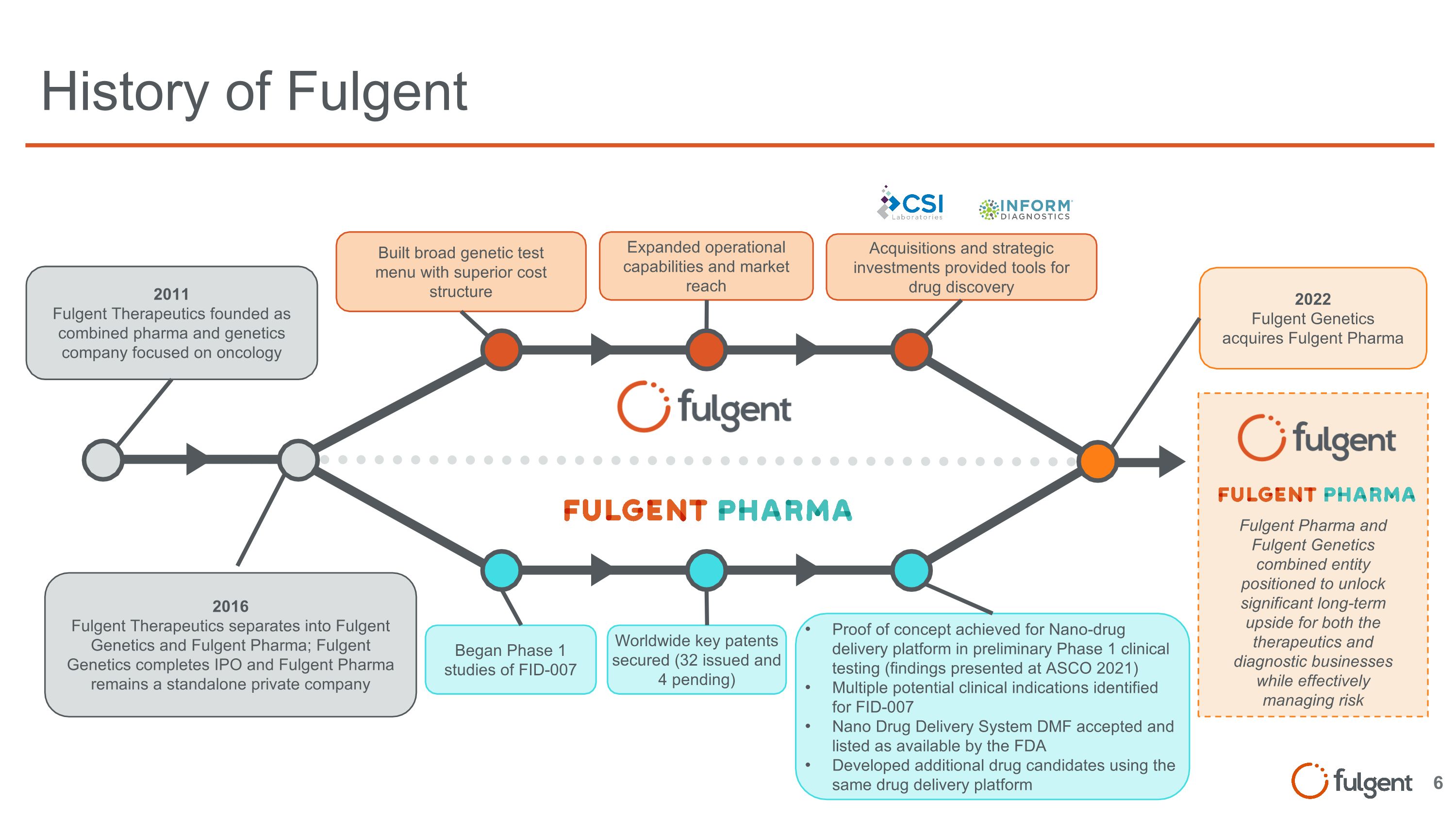
Fulgent Pharma and Fulgent Genetics combined entity positioned to unlock significant long-term upside for both the therapeutics and diagnostic businesses while effectively managing risk History of Fulgent 2011 Fulgent Therapeutics founded as combined pharma and genetics company focused on oncology 2016 Fulgent Therapeutics separates into Fulgent Genetics and Fulgent Pharma; Fulgent Genetics completes IPO and Fulgent Pharma remains a standalone private company 2022 Fulgent Genetics acquires Fulgent Pharma Built broad genetic test menu with superior cost structure Expanded operational capabilities and market reach Acquisitions and strategic investments provided tools for drug discovery Began Phase 1 studies of FID-007 Worldwide key patents secured (32 issued and 4 pending) Proof of concept achieved for Nano-drug delivery platform in preliminary Phase 1 clinical testing (findings presented at ASCO 2021) Multiple potential clinical indications identified for FID-007 Nano Drug Delivery System DMF accepted and listed as available by the FDA Developed additional drug candidates using the same drug delivery platform
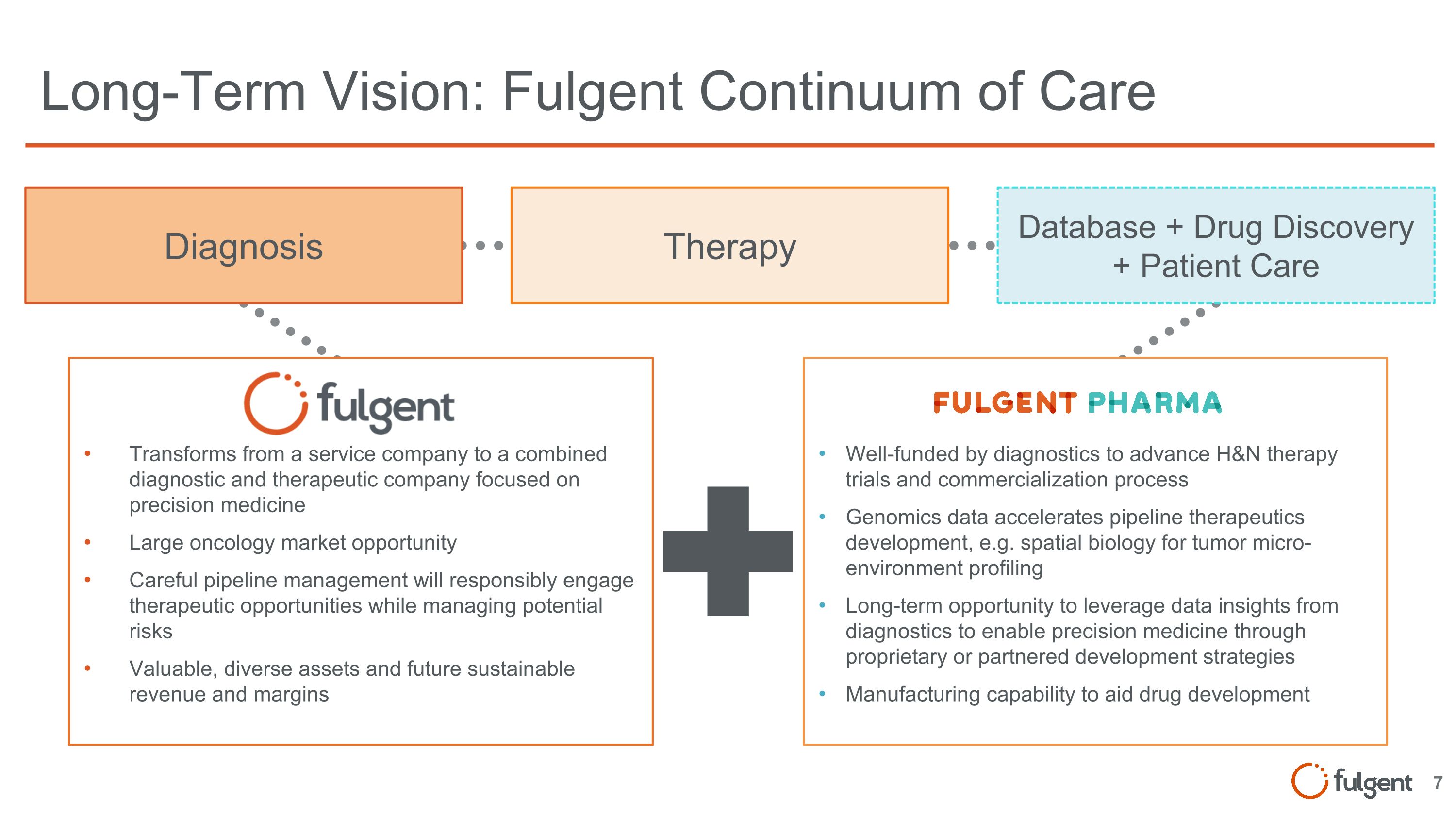
Long-Term Vision: Fulgent Continuum of Care Diagnosis Well-funded by diagnostics to advance H&N therapy trials and commercialization process Genomics data accelerates pipeline therapeutics development, e.g. spatial biology for tumor micro-environment profiling Long-term opportunity to leverage data insights from diagnostics to enable precision medicine through proprietary or partnered development strategies Manufacturing capability to aid drug development Transforms from a service company to a combined diagnostic and therapeutic company focused on precision medicine Large oncology market opportunity Careful pipeline management will responsibly engage therapeutic opportunities while managing potential risks Valuable, diverse assets and future sustainable revenue and margins Therapy Database + Drug Discovery + Patient Care

LABORATORY SERVICES

Well-positioned to execute on a growth strategy that includes organic and inorganic initiatives, including: Transformational acquisition of Inform Diagnostics Ramping of CSI Labs Scaling partnerships Potential future acquisitions with a strategy of short- and long-term ROI, tangible synergies, and efficient capital deployment 1 2 3 $70.5M Q4 Revenue +21% Q4 Year-over-Year Core Revenue Increase 18,400+ GENES | 900+ PANELS CUSTOMIZABLE OFFERINGS Positioned for Growth Proprietary technology platform allows for rapid scaling of a broad, flexible test menu Next-generation sequencing (NGS) platform complemented with growing portfolio of emerging testing technologies with a focus on oncology 9
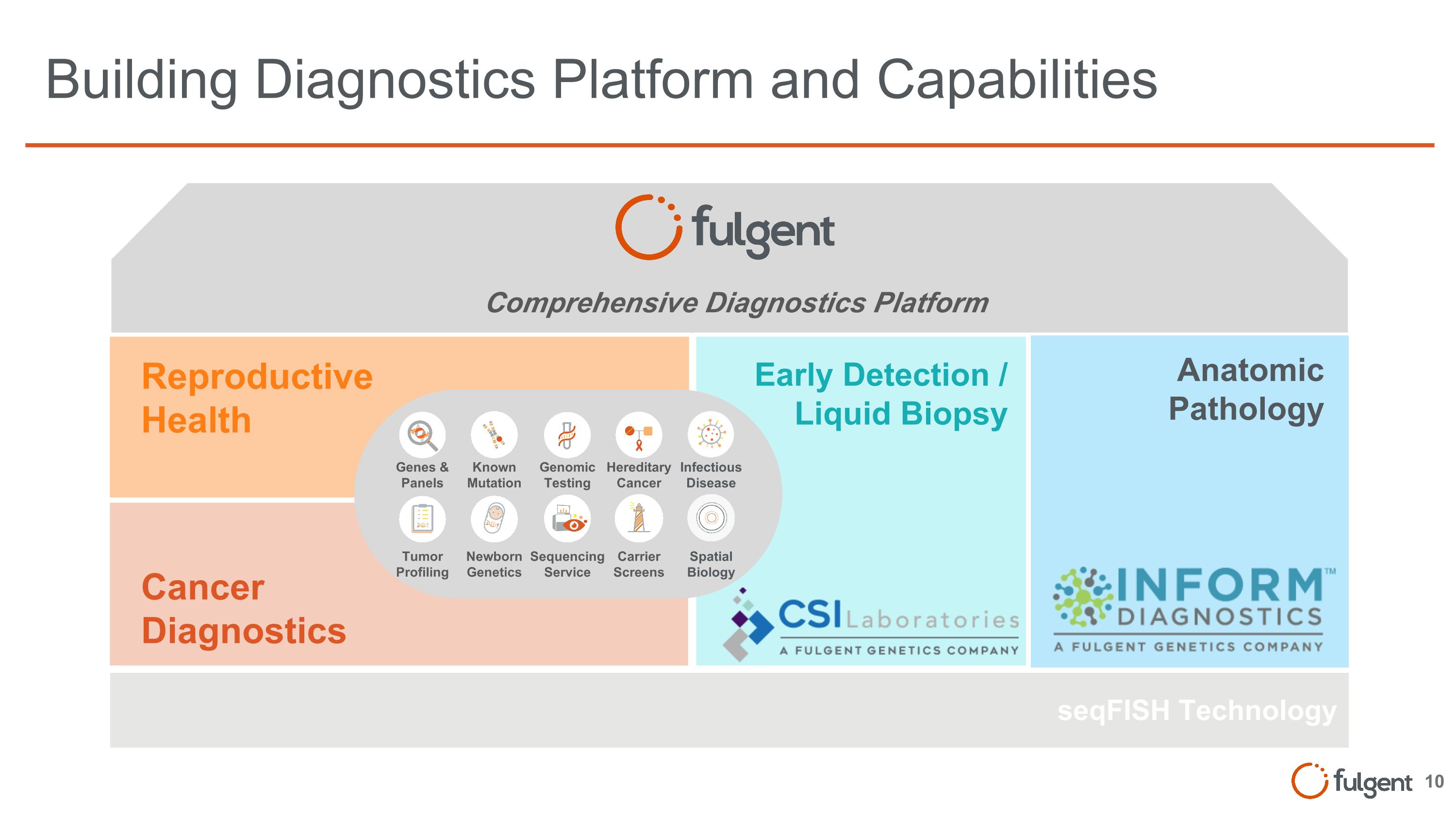
Building Diagnostics Platform and Capabilities Cancer Diagnostics Reproductive Health Early Detection / Liquid Biopsy seqFISH Technology Comprehensive Diagnostics Platform Anatomic Pathology Genes & Panels Tumor Profiling Known Mutation Newborn Genetics Hereditary Cancer Carrier Screens Genomic Testing Sequencing Service Infectious Disease Spatial Biology

Target Market Opportunity Cancer Diagnostics $80B market1 Market sizes sourced from Wall Street equity research Market size sourced from Frost & Sullivan, October 2022 Market size sourced from Research and Markets, April 2022 $18B market1 Early Detection / Liquid Biopsy Reproductive Health $8B market2 BioPharma Services $50B market3 Genes & Panels Tumor Profiling Known Mutation Newborn Genetics Hereditary Cancer Carrier Screens Genomic Testing Sequencing Service Infectious Disease Spatial Biology
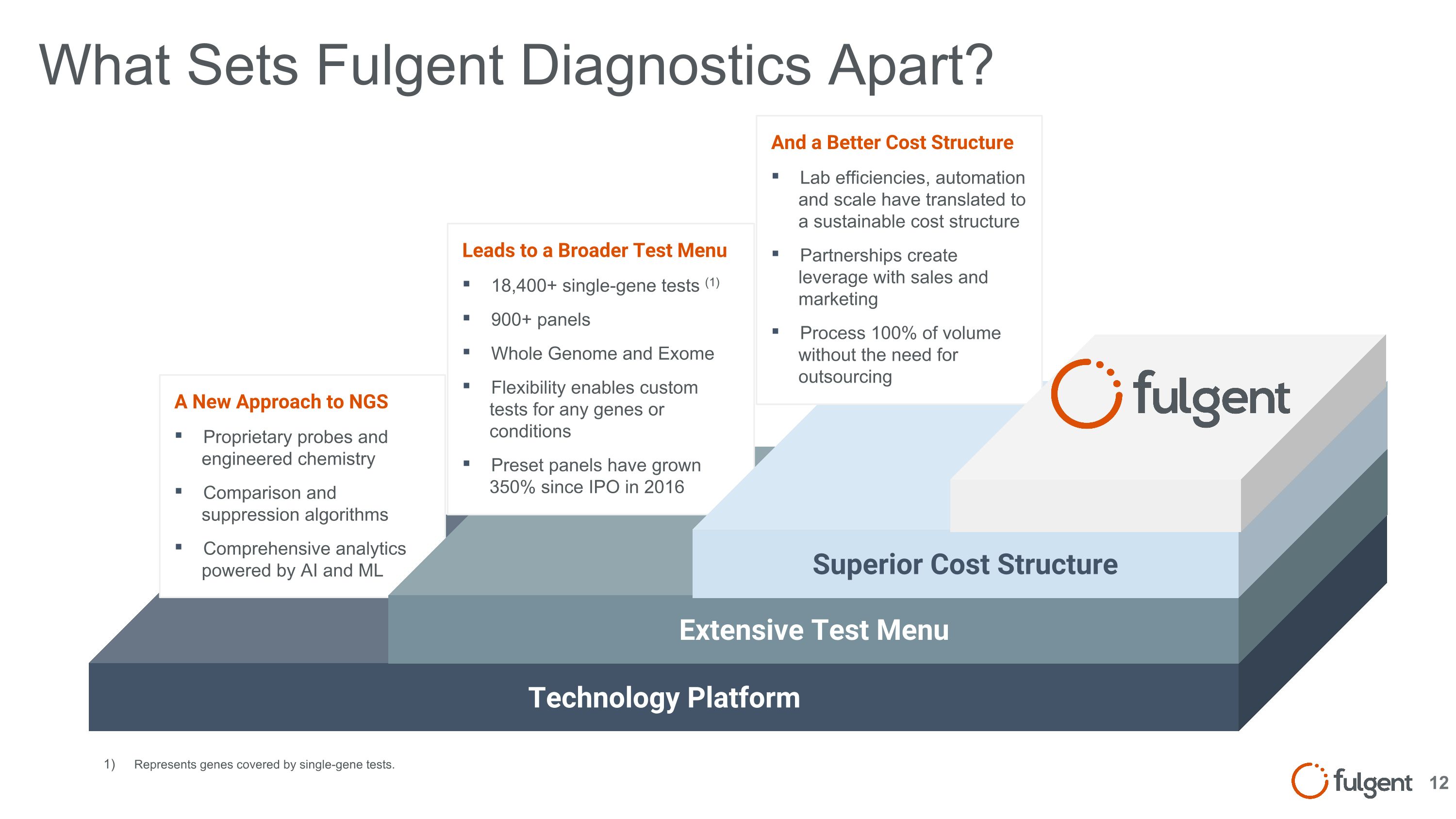
What Sets Fulgent Diagnostics Apart? Technology Platform A New Approach to NGS Proprietary probes and engineered chemistry Comparison and suppression algorithms Comprehensive analytics powered by AI and ML Extensive Test Menu Leads to a Broader Test Menu 18,400+ single-gene tests (1) 900+ panels Whole Genome and Exome Flexibility enables custom tests for any genes or conditions Preset panels have grown 350% since IPO in 2016 Superior Cost Structure And a Better Cost Structure Lab efficiencies, automation and scale have translated to a sustainable cost structure Partnerships create leverage with sales and marketing Process 100% of volume without the need for outsourcing Represents genes covered by single-gene tests. 12
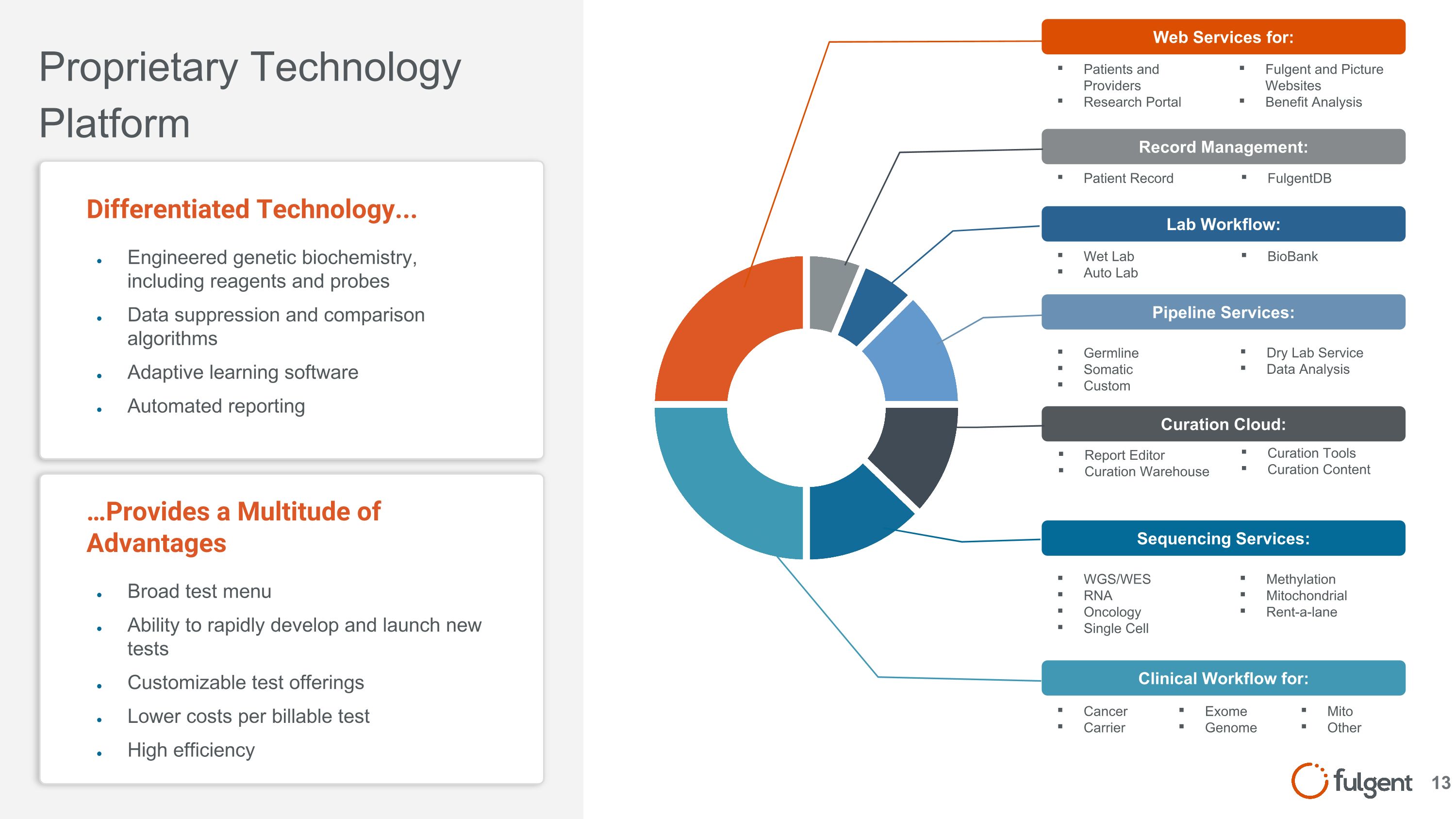
Patients and Providers Research Portal …Provides a Multitude of Advantages Broad test menu Ability to rapidly develop and launch new tests Customizable test offerings Lower costs per billable test High efficiency Proprietary Technology Platform Differentiated Technology... Engineered genetic biochemistry, including reagents and probes Data suppression and comparison algorithms Adaptive learning software Automated reporting Web Services for: Clinical Workflow for: Cancer Carrier Exome Genome Methylation Mitochondrial Rent-a-lane Sequencing Services: WGS/WES RNA Oncology Single Cell Pipeline Services: Curation Tools Curation Content Curation Cloud: Report Editor Curation Warehouse Dry Lab Service Data Analysis Germline Somatic Custom Lab Workflow: Wet Lab Auto Lab Record Management: Patient Record BioBank FulgentDB Fulgent and Picture Websites Benefit Analysis Mito Other 13
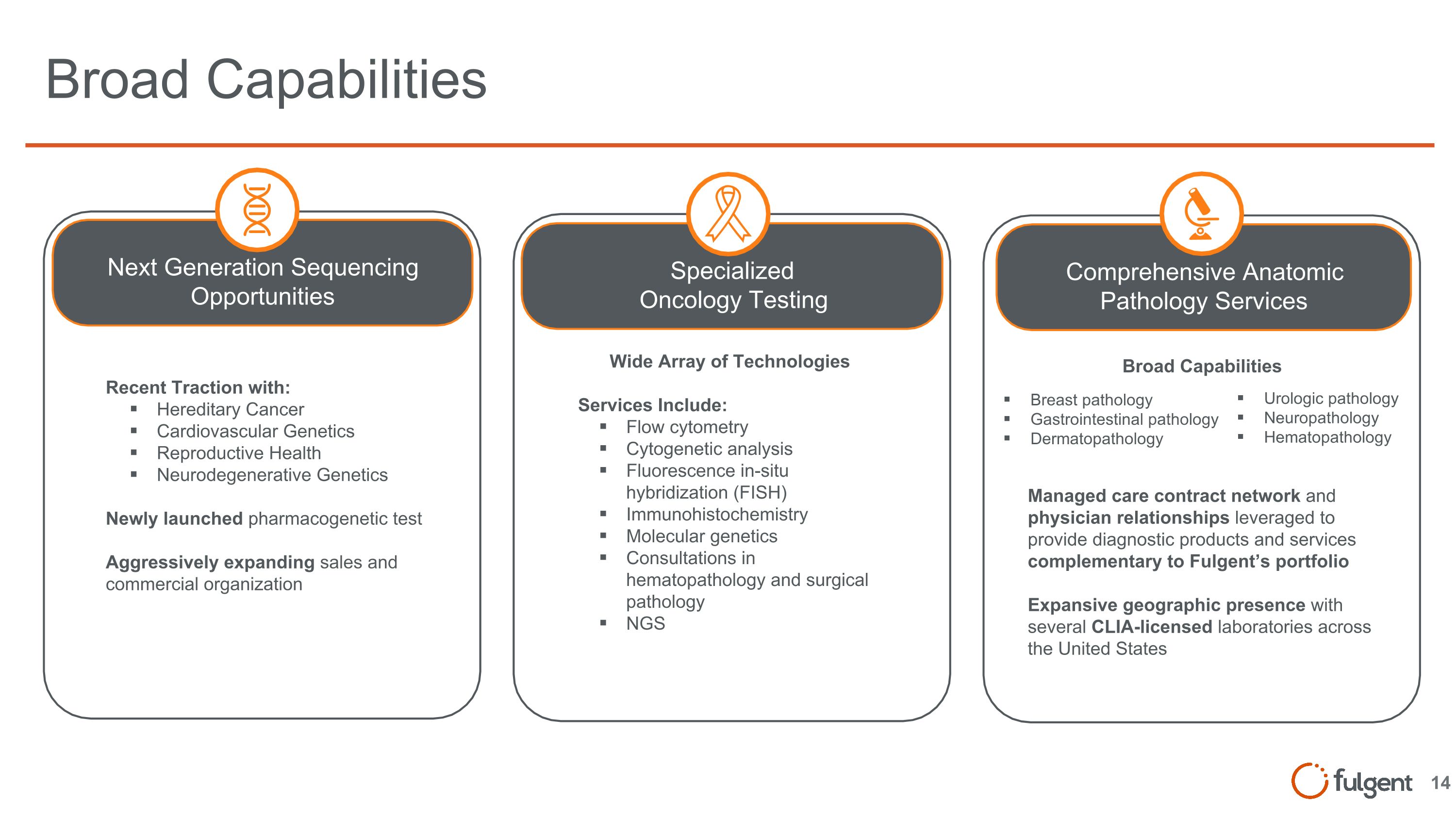
Comprehensive Anatomic Pathology Services Specialized Oncology Testing Recent Traction with: Hereditary Cancer Cardiovascular Genetics Reproductive Health Neurodegenerative Genetics Newly launched pharmacogenetic test Aggressively expanding sales and commercial organization Wide Array of Technologies Services Include: Flow cytometry Cytogenetic analysis Fluorescence in-situ hybridization (FISH) Immunohistochemistry Molecular genetics Consultations in hematopathology and surgical pathology NGS Broad Capabilities Next Generation Sequencing Opportunities Broad Capabilities Breast pathology Gastrointestinal pathology Dermatopathology Urologic pathology Neuropathology Hematopathology Managed care contract network and physician relationships leveraged to provide diagnostic products and services complementary to Fulgent’s portfolio Expansive geographic presence with several CLIA-licensed laboratories across the United States

Fulgent deployed its technology platform to rapidly respond to the COVID-19 Pandemic, scaling operations to provide tests with reliable results and rapid turnaround time Technology Platform Case Study: COVID-19 Research driven platform worked with local and federal government on genomic studies CDC contract awarded to Fulgent, worth up to $47M to study SARS-CoV-2 using Fulgent’s NGS platform Capacity of 10,000 NGS tests per day Used to identify new strains and mutations Next Generation Sequencing for COVID-19 Commercialized COVID-19 Testing Primarily RT-PCR Based Testing Contracts with: School systems Nursing homes Athletic organizations Specialty health clinics Travel organizations Government agencies Offered through: Drive-through sites Picture at-home kits Managed on-site programs Result: Over 19M COVID-19 tests delivered between 2020-2023, generating >$1.7B in revenue for Fulgent
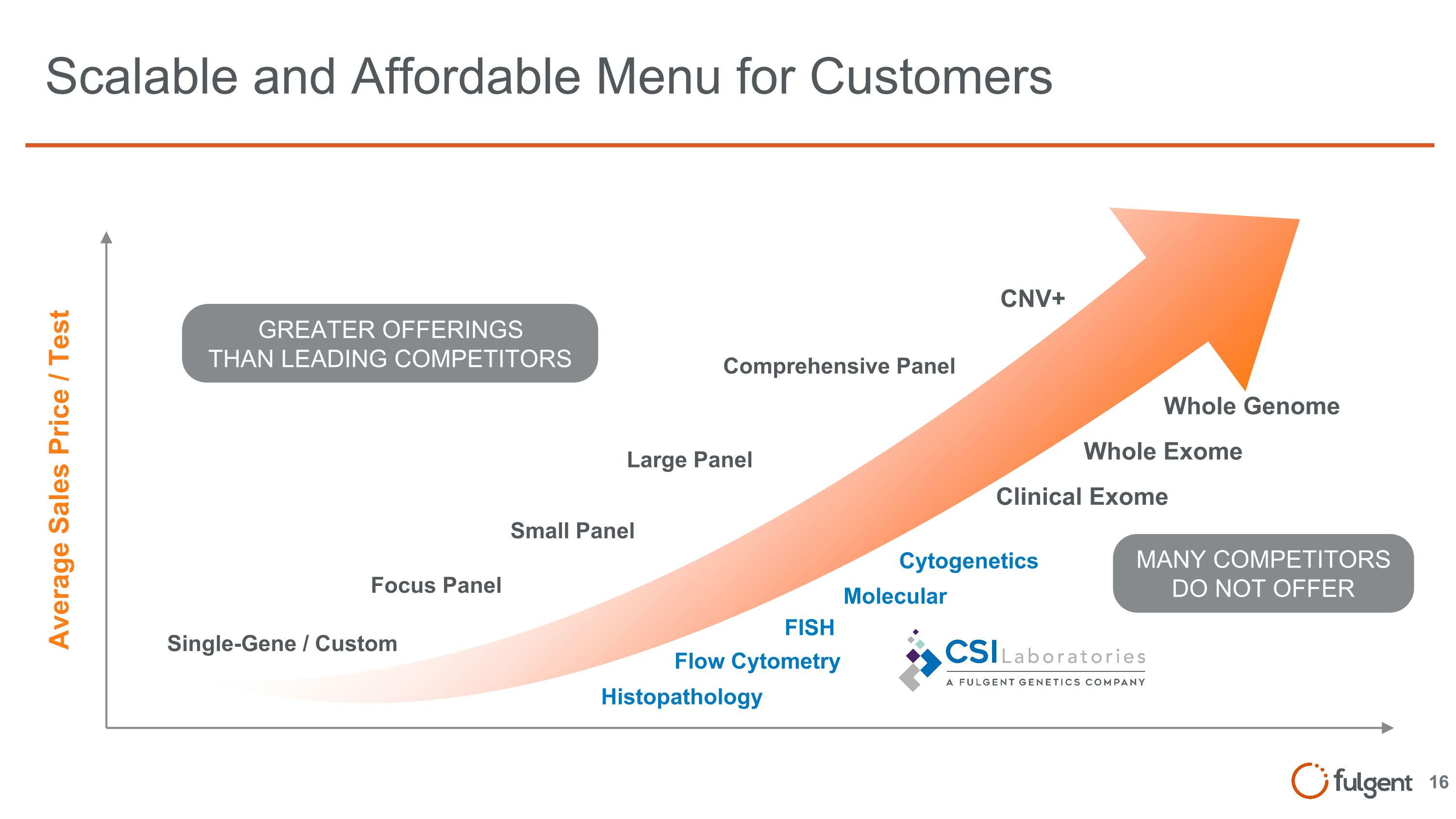
Scalable and Affordable Menu for Customers Large Panel Small Panel Focus Panel Single-Gene / Custom Comprehensive Panel Average Sales Price / Test Whole Genome Whole Exome Clinical Exome GREATER OFFERINGS THAN LEADING COMPETITORS CNV+ MANY COMPETITORS DO NOT OFFER Flow Cytometry FISH Histopathology Cytogenetics Molecular
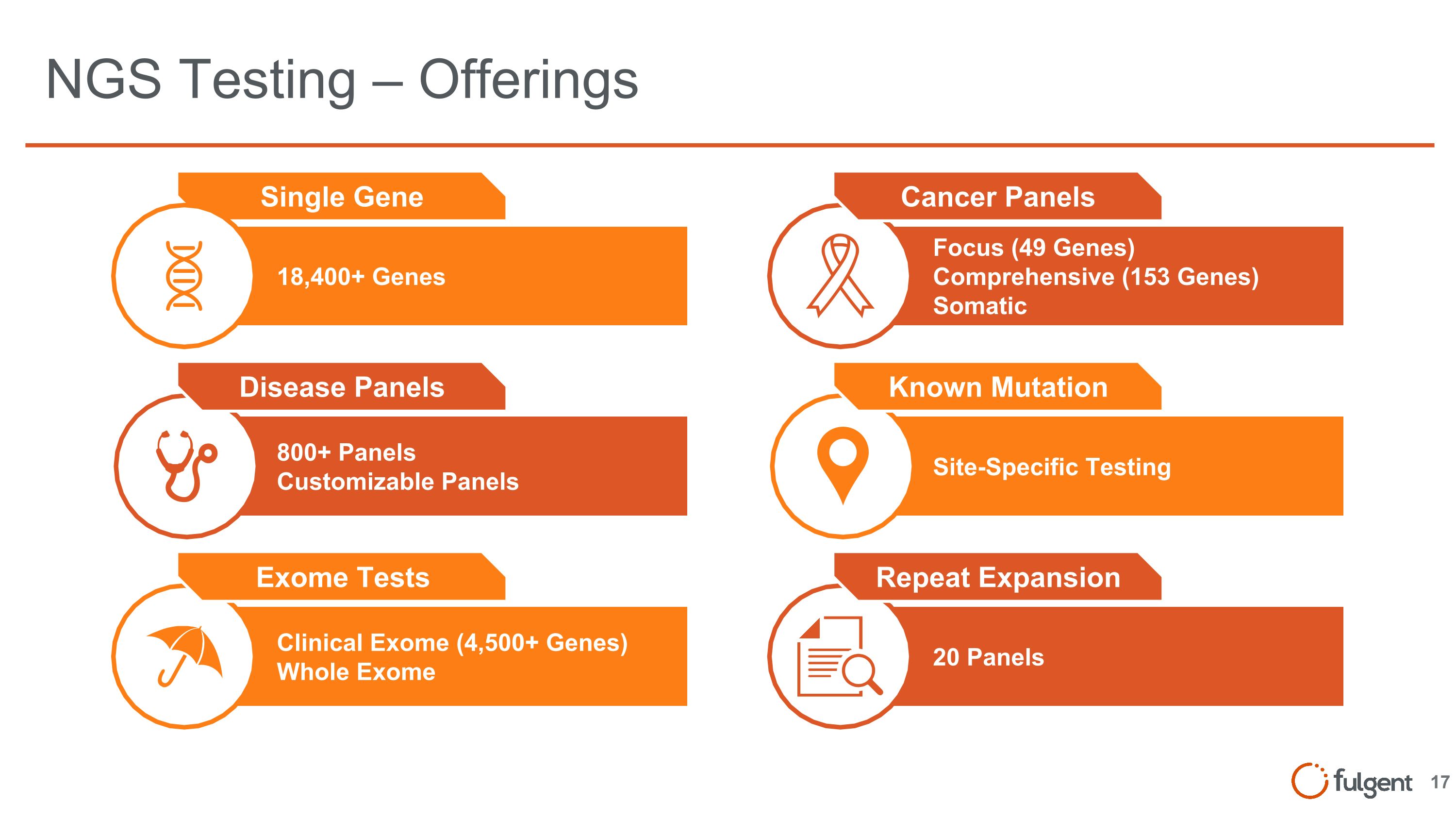
NGS Testing – Offerings Site-Specific Testing Known Mutation Focus (49 Genes) Comprehensive (153 Genes) Somatic Cancer Panels 20 Panels Repeat Expansion 18,400+ Genes Single Gene 800+ Panels Customizable Panels Disease Panels Clinical Exome (4,500+ Genes) Whole Exome Exome Tests
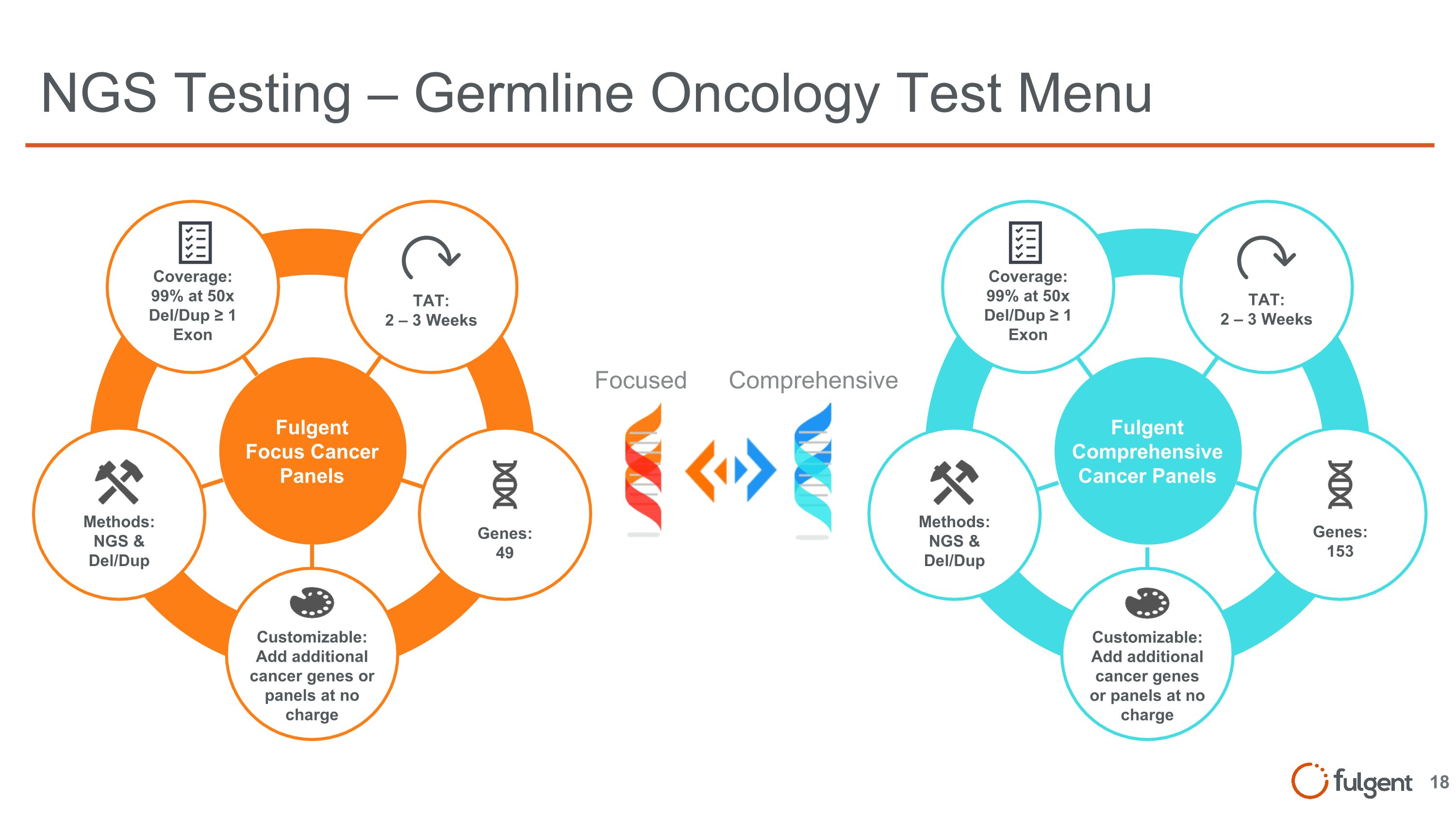
NGS Testing – Germline Oncology Test Menu Fulgent Focus Cancer Panels Fulgent Comprehensive Cancer Panels Customizable: Add additional cancer genes or panels at no charge Genes: 49 Methods: NGS & Del/Dup Coverage: 99% at 50x Del/Dup ≥ 1 Exon TAT: 2 – 3 Weeks Customizable: Add additional cancer genes or panels at no charge Methods: NGS & Del/Dup Genes: 153 Coverage: 99% at 50x Del/Dup ≥ 1 Exon TAT: 2 – 3 Weeks Focused Comprehensive
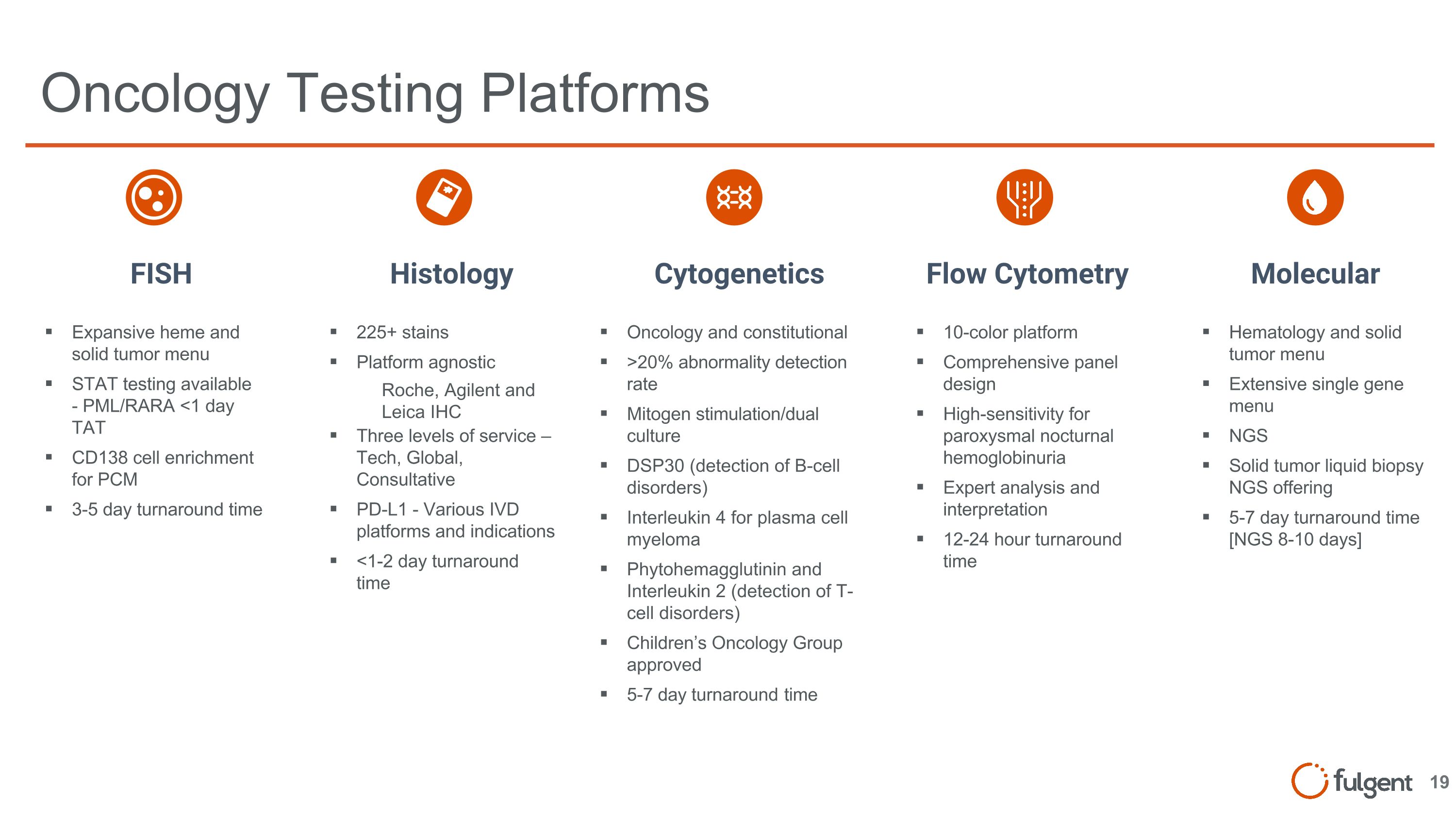
Oncology Testing Platforms Expansive heme and solid tumor menu STAT testing available - PML/RARA <1 day TAT CD138 cell enrichment for PCM 3-5 day turnaround time 225+ stains Platform agnostic Roche, Agilent and Leica IHC Three levels of service – Tech, Global, Consultative PD-L1 - Various IVD platforms and indications <1-2 day turnaround time Hematology and solid tumor menu Extensive single gene menu NGS Solid tumor liquid biopsy NGS offering 5-7 day turnaround time [NGS 8-10 days] Oncology and constitutional >20% abnormality detection rate Mitogen stimulation/dual culture DSP30 (detection of B-cell disorders) Interleukin 4 for plasma cell myeloma Phytohemagglutinin and Interleukin 2 (detection of T-cell disorders) Children’s Oncology Group approved 5-7 day turnaround time FISH Histology Cytogenetics Flow Cytometry Molecular 10-color platform Comprehensive panel design High-sensitivity for paroxysmal nocturnal hemoglobinuria Expert analysis and interpretation 12-24 hour turnaround time
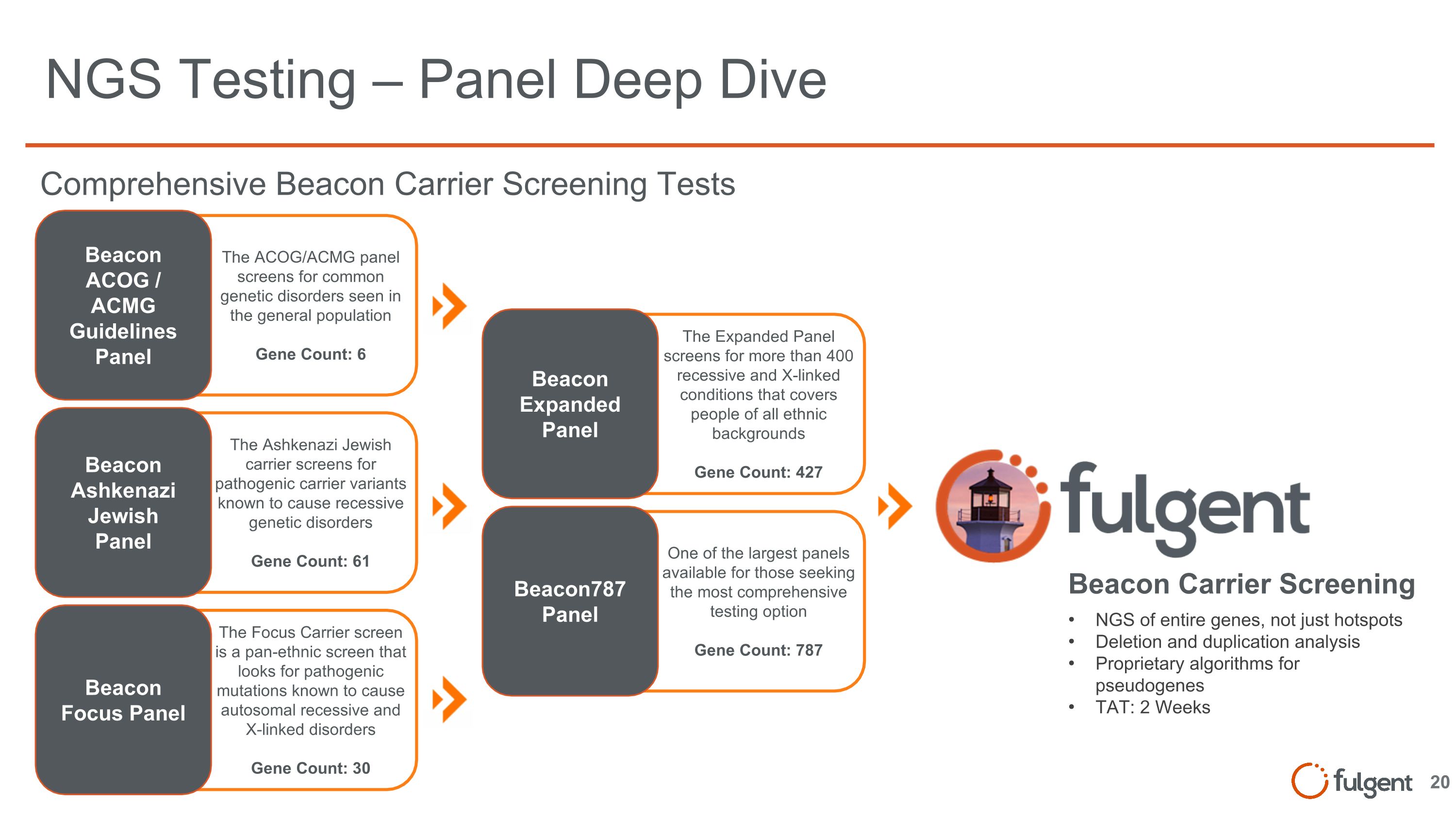
The Focus Carrier screen is a pan-ethnic screen that looks for pathogenic mutations known to cause autosomal recessive and X-linked disorders Gene Count: 30 The Ashkenazi Jewish carrier screens for pathogenic carrier variants known to cause recessive genetic disorders Gene Count: 61 The ACOG/ACMG panel screens for common genetic disorders seen in the general population Gene Count: 6 One of the largest panels available for those seeking the most comprehensive testing option Gene Count: 787 The Expanded Panel screens for more than 400 recessive and X-linked conditions that covers people of all ethnic backgrounds Gene Count: 427 NGS Testing – Panel Deep Dive NGS of entire genes, not just hotspots Deletion and duplication analysis Proprietary algorithms for pseudogenes TAT: 2 Weeks Beacon Carrier Screening Beacon ACOG / ACMG Guidelines Panel Beacon Ashkenazi Jewish Panel Beacon Focus Panel Beacon Expanded Panel Beacon787 Panel Comprehensive Beacon Carrier Screening Tests
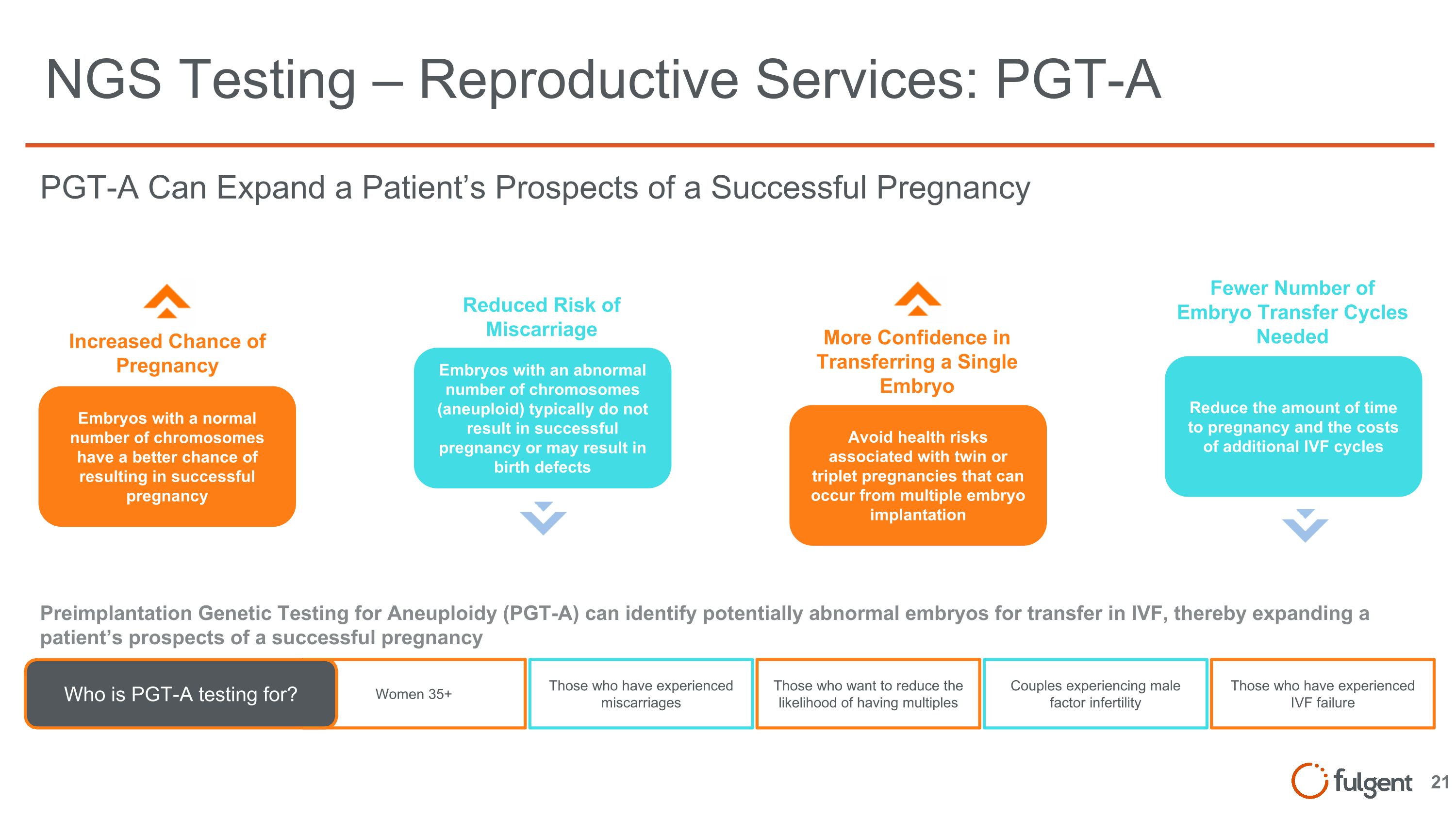
NGS Testing – Reproductive Services: PGT-A Increased Chance of Pregnancy Embryos with a normal number of chromosomes have a better chance of resulting in successful pregnancy More Confidence in Transferring a Single Embryo Avoid health risks associated with twin or triplet pregnancies that can occur from multiple embryo implantation Reduced Risk of Miscarriage Embryos with an abnormal number of chromosomes (aneuploid) typically do not result in successful pregnancy or may result in birth defects Fewer Number of Embryo Transfer Cycles Needed Reduce the amount of time to pregnancy and the costs of additional IVF cycles PGT-A Can Expand a Patient’s Prospects of a Successful Pregnancy Preimplantation Genetic Testing for Aneuploidy (PGT-A) can identify potentially abnormal embryos for transfer in IVF, thereby expanding a patient’s prospects of a successful pregnancy Women 35+ Who is PGT-A testing for? Those who have experienced miscarriages Those who want to reduce the likelihood of having multiples Couples experiencing male factor infertility Those who have experienced IVF failure
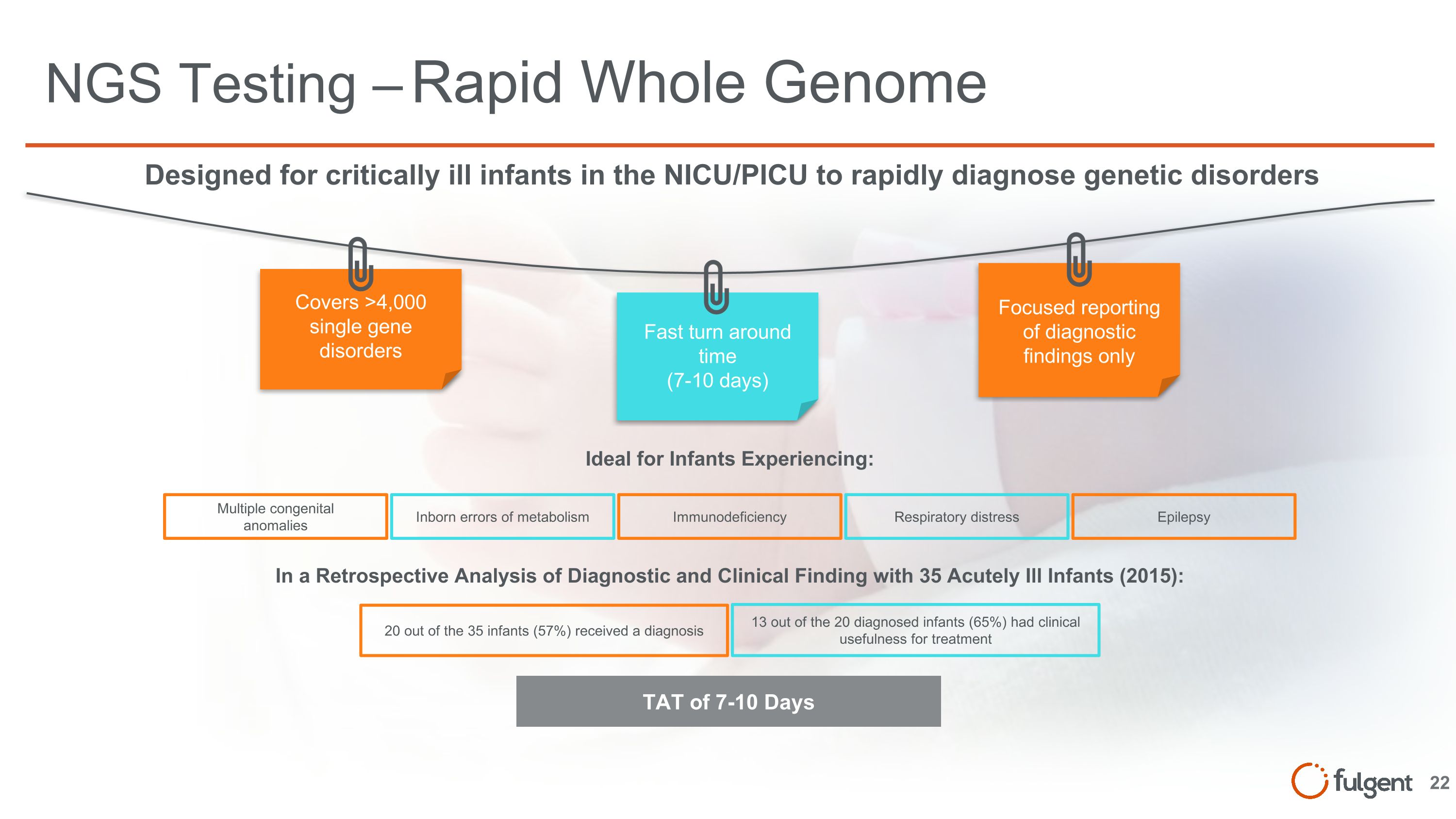
NGS Testing – Rapid Whole Genome Covers >4,000 single gene disorders Fast turn around time (7-10 days) Focused reporting of diagnostic findings only Designed for critically ill infants in the NICU/PICU to rapidly diagnose genetic disorders Multiple congenital anomalies Inborn errors of metabolism Immunodeficiency Respiratory distress Epilepsy Ideal for Infants Experiencing: 20 out of the 35 infants (57%) received a diagnosis 13 out of the 20 diagnosed infants (65%) had clinical usefulness for treatment In a Retrospective Analysis of Diagnostic and Clinical Finding with 35 Acutely Ill Infants (2015): TAT of 7-10 Days

Parenting Screens adults to determine their carrier status for recessive genetic disorders Newborn Screens newborns for genes associated with various severe genetic conditions Wellness Screens adults for genetic variants that indicate disease risk PD Aware Assesses genetic risk for Parkinson's Disease COVID-19 RT-PCR Test At-home collection test for COVID-19 Consumer Initiated Tests – Picture Genetics Targeting the Large Consumer Market with Picture Genetics Launched in 2019 with significant growth amid COVID-19 A consumer-focused offering that merges clinical utility with accuracy of an accredited lab Extends Fulgent’s NGS capabilities to a broader market Validated by successfully scaling to hundreds of thousands of tests performed within months for COVID-19, after receiving an EUA Genetic tests utilizes complete sequencing (vs genotyping) by NGS analysis for better, more accurate results Patient-friendly with easy to use “order from home” model – no doctor office visits or insurance necessary, though many tests are eligible for reimbursement Select full service offering that includes analysis and genetic counseling support PGx Pharmacogenetic test that provides genetic insights on drug response

THERAPEUTIC DEVELOPMENT
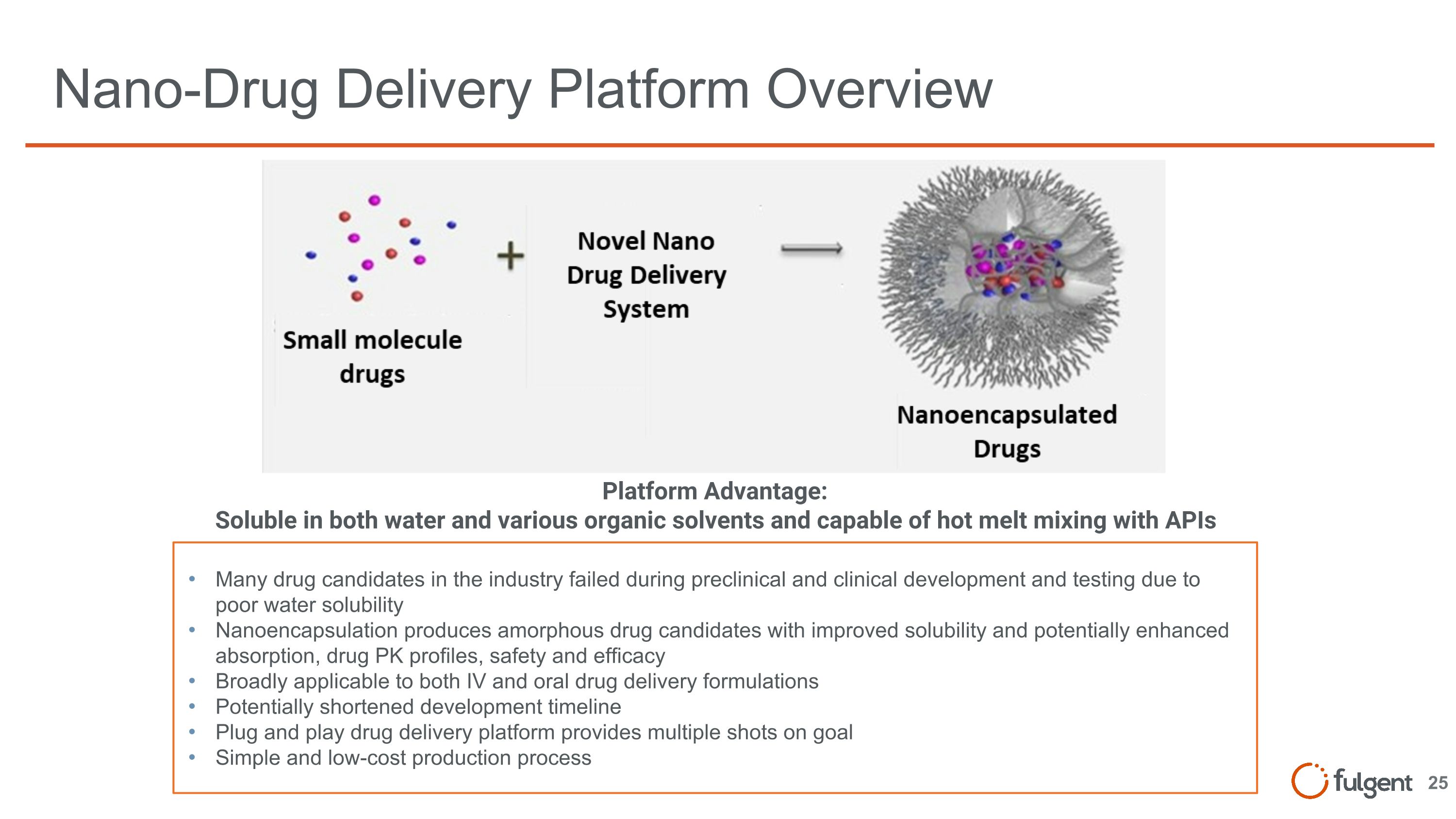
Nano-Drug Delivery Platform Overview Many drug candidates in the industry failed during preclinical and clinical development and testing due to poor water solubility Nanoencapsulation produces amorphous drug candidates with improved solubility and potentially enhanced absorption, drug PK profiles, safety and efficacy Broadly applicable to both IV and oral drug delivery formulations Potentially shortened development timeline Plug and play drug delivery platform provides multiple shots on goal Simple and low-cost production process Platform Advantage: Soluble in both water and various organic solvents and capable of hot melt mixing with APIs

FID-007 Program Overview Note: all findings are preliminary DCR includes Stable Disease (SD), Partial Response (PR), Complete Response (CR) FID-007 Phase I Preliminary Highlights (as of 6/2/23): H&N Cancer 57% ORR and 71% DCR were observed in 7 heavily treated H&N patients. Among them, 6/7 had prior Taxane treatment. Ampullary/Pancreatic 50% ORR and 75% DCR were seen in 4 heavily treated ampullary and pancreatic patients FID-007 Phase I First in Human Clinical Trial – Preliminary Findings (n=40 patients) Dose levels up to 160 mg/m2/week with manageable safety profile RP2D at 125 mg/m2/week There is preliminary evidence of anti-tumor activity in 40 heavily pre-treated patients across different tumor types (ORR = 18%) No high-grade neuropathy often seen in other taxanes Updated clinical data presented at ASCO 2023
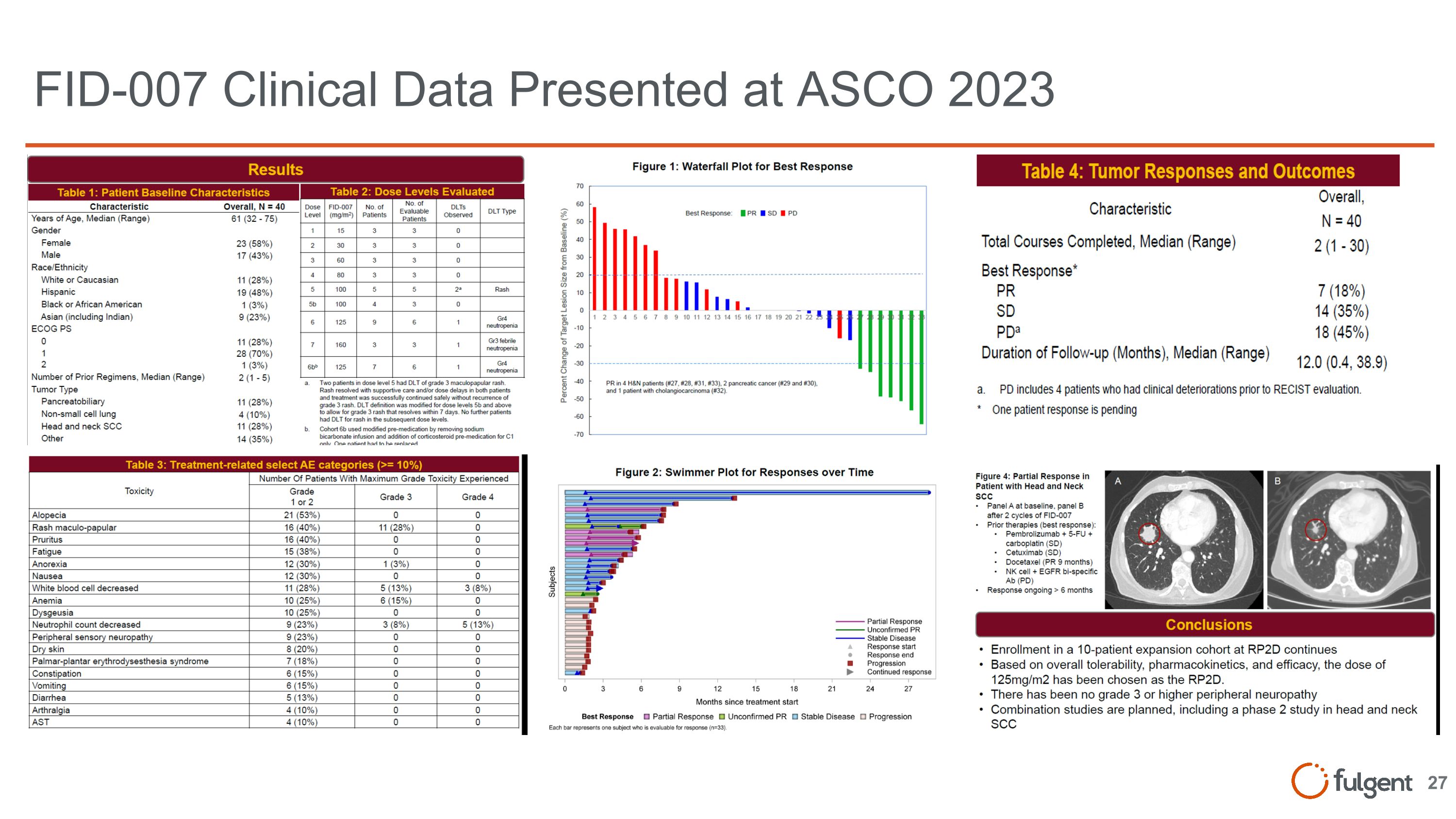
FID-007 Clinical Data Presented at ASCO 2023

Potential Market Opportunity for FID-007 Pancreatic $1.2B in 2023 $1.5B by 2035 NSCLC $123M in 2023 $140M by 2035 Ampullary2 $51M in 2023 $63M by 2035 H&N1 $2.3B in 2023 $2.9B by 2035 Note: U.S. opportunity shown Sources: Evaluate Pharma, Wall Street research, and management pricing expectations H&N market opportunity for both 2nd line and 3rd line therapy Ampullary market opportunity for 2nd line therapy Initial Indication Subsequent Indications Breast $501M in 2023 $521M by 2035
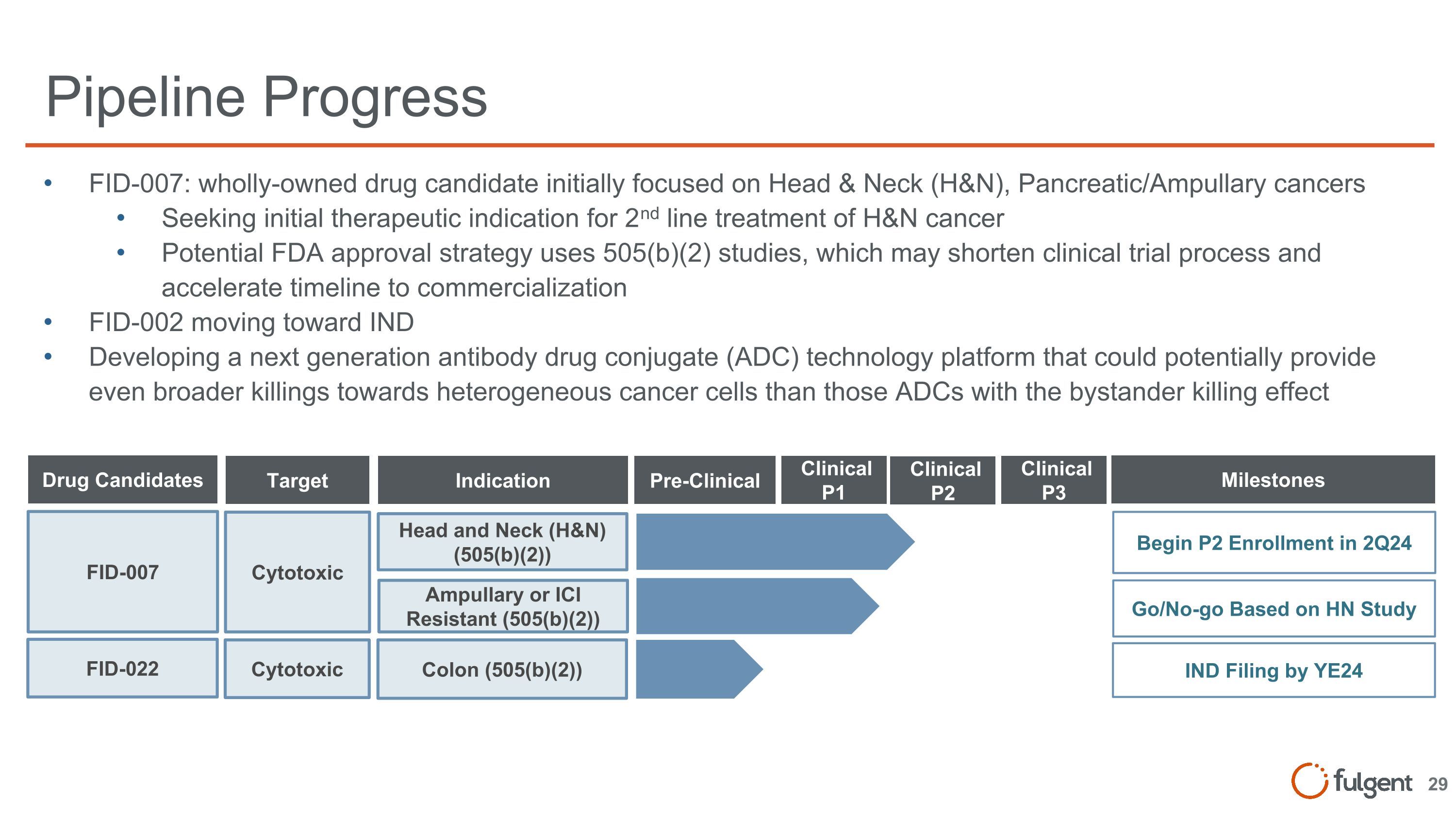
Pipeline Progress FID-007: wholly-owned drug candidate initially focused on Head & Neck (H&N), Pancreatic/Ampullary cancers Seeking initial therapeutic indication for 2nd line treatment of H&N cancer Potential FDA approval strategy uses 505(b)(2) studies, which may shorten clinical trial process and accelerate timeline to commercialization FID-002 moving toward IND Developing a next generation antibody drug conjugate (ADC) technology platform that could potentially provide even broader killings towards heterogeneous cancer cells than those ADCs with the bystander killing effect FID-007 Drug Candidates Pre-Clinical Target Indication Milestones Cytotoxic Head and Neck (H&N) (505(b)(2)) Begin P2 Enrollment in 2Q24 Ampullary or ICI Resistant (505(b)(2)) Go/No-go Based on HN Study Clinical P1 Clinical P2 Clinical P3 FID-022 Cytotoxic Colon (505(b)(2)) IND Filing by YE24

FINANCIALS
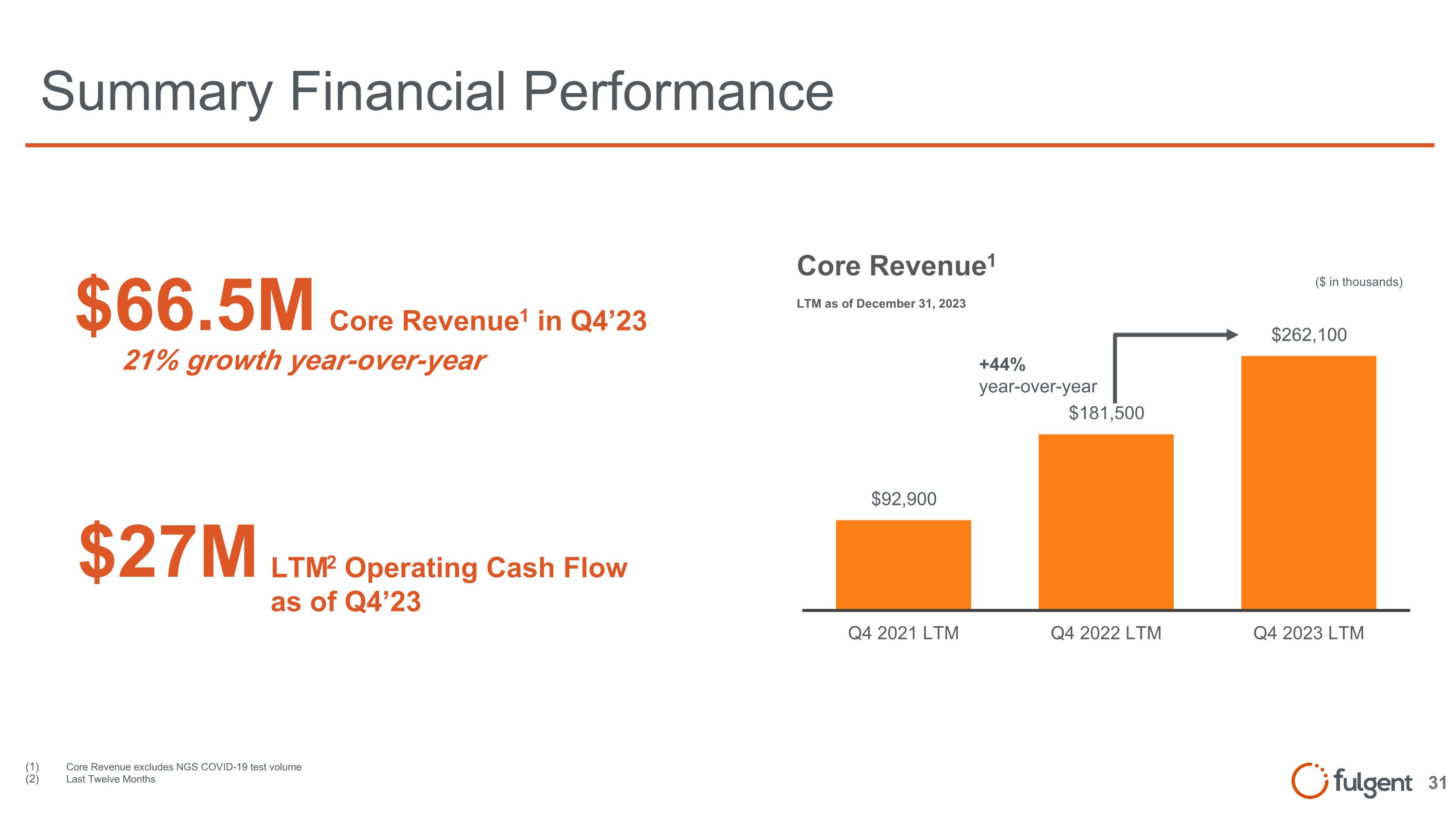
Summary Financial Performance $66.5M Core Revenue1 in Q4’23 $27M LTM2 Operating Cash Flow as of Q4’23 21% growth year-over-year ($ in thousands) +44% year-over-year Core Revenue excludes NGS COVID-19 test volume Last Twelve Months
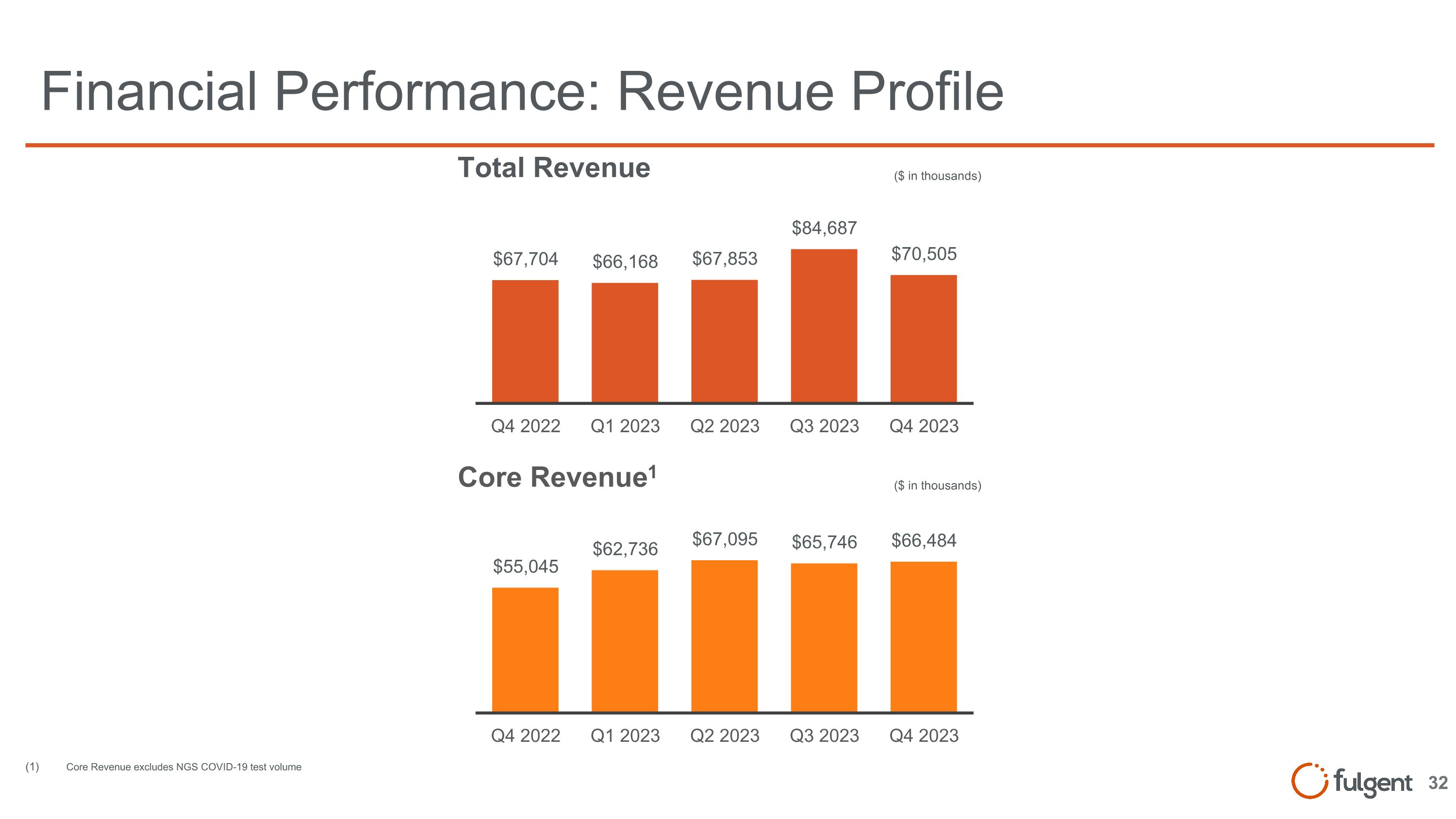
Financial Performance: Revenue Profile ($ in thousands) ($ in thousands) Core Revenue excludes NGS COVID-19 test volume
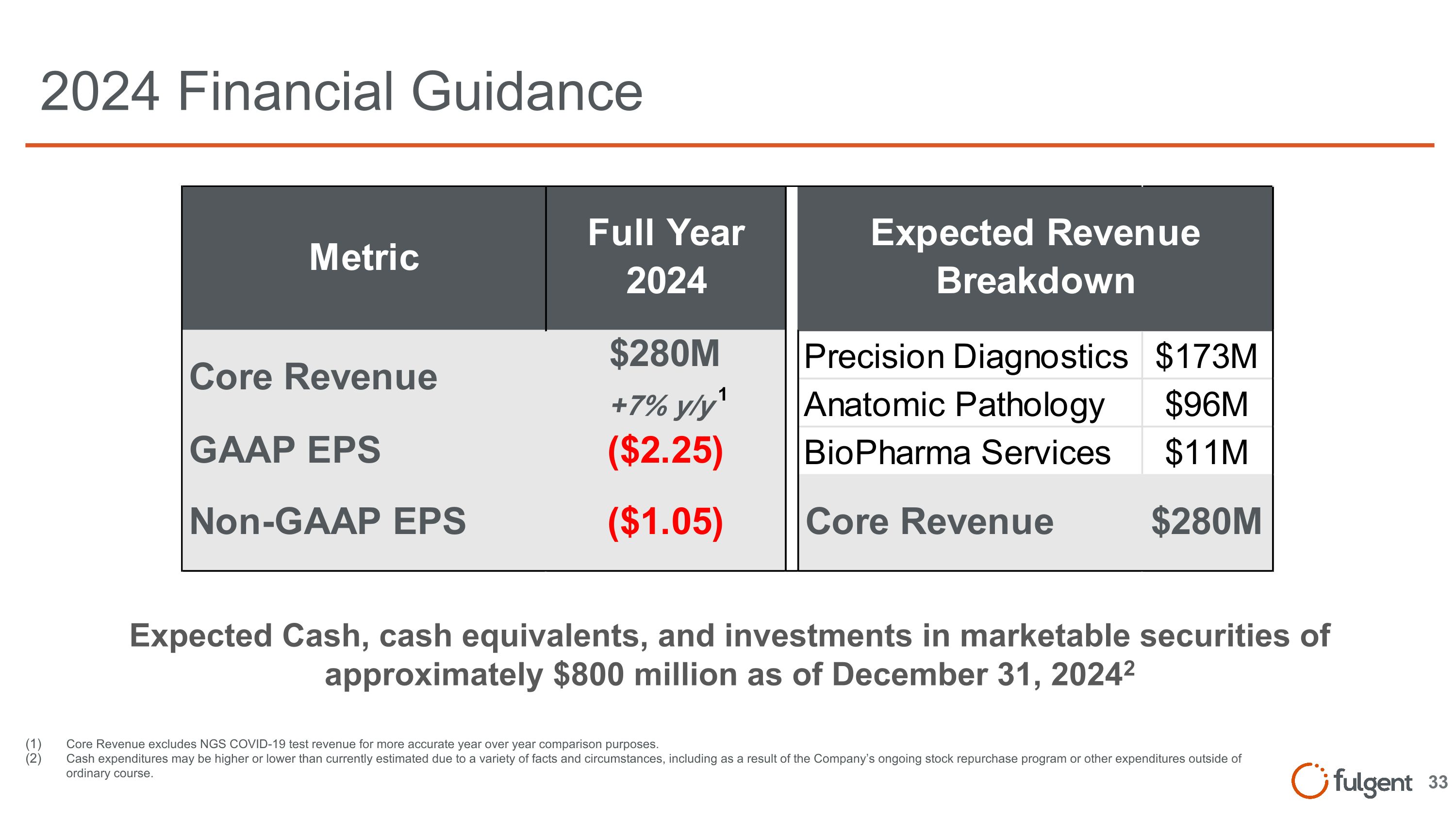
2024 Financial Guidance Core Revenue excludes NGS COVID-19 test revenue for more accurate year over year comparison purposes. Cash expenditures may be higher or lower than currently estimated due to a variety of facts and circumstances, including as a result of the Company’s ongoing stock repurchase program or other expenditures outside of ordinary course. Expected Cash, cash equivalents, and investments in marketable securities of approximately $800 million as of December 31, 20242
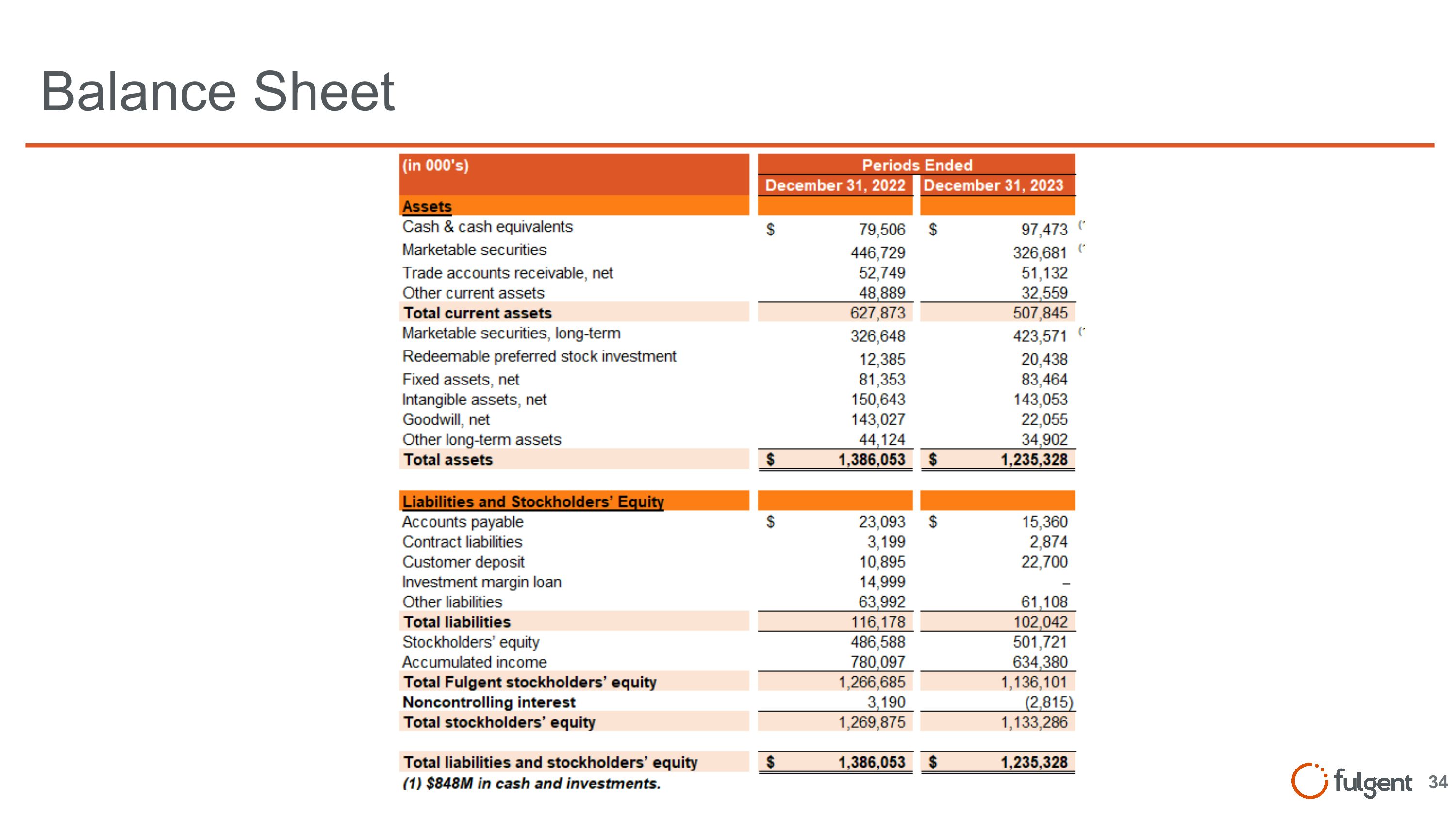
Balance Sheet
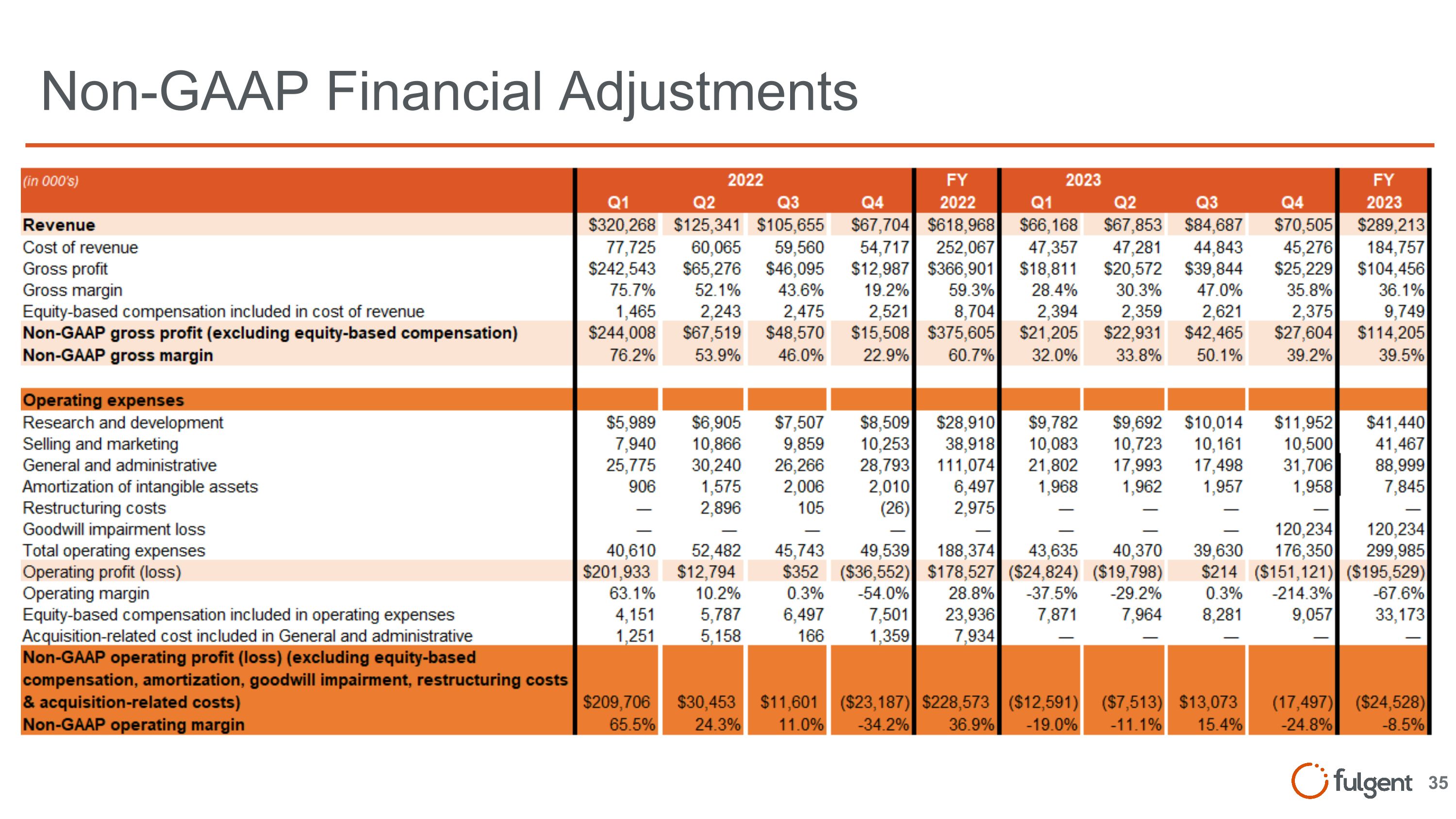
Non-GAAP Financial Adjustments

THANK YOU

Founded in 2011 | Located in El Monte, CA | NASDAQ:FLGT