Document
Bristol Myers Squibb Reports First Quarter Financial Results for 2024
Performance Reflects Execution and Actions to Strengthen the Company's Long-Term Growth Profile
•First Quarter Revenues were $11.9 Billion, increasing 5% (+6% Adjusting for Foreign Exchange)
◦Growth Portfolio Revenues were $4.8 Billion, increasing 8% (+11% Adjusting for Foreign Exchange)
•Strengthened Long-Term Growth Profile Through Completion of Karuna Therapeutics, RayzeBio, Mirati Therapeutics, and SystImmune Transactions
•Including the One-Time Net Impact of Acquired IPRD Charges and Licensing Income of $(6.30) From Recently Closed Transactions, GAAP Loss Per Share was $(5.89); Non-GAAP Loss Per Share was $(4.40)
•Achieved U.S. Approval of Abecma in Earlier-Line Multiple Myeloma and Breyanzi in Chronic Lymphocytic Leukemia and Small Lymphocytic Lymphoma and Positive Proof of Concept for Opdualag in Non-Small Cell Lung Cancer
•Executing a Strategic Productivity Initiative to Deliver ~$1.5 Billion in Cost Savings, the Majority of Which Will be Reinvested to Fund Innovation and Drive Growth
•Updating 2024 Non-GAAP EPS and Line-Item Guidance to Reflect Impact of Recently Completed Transactions
(PRINCETON, N.J., April 25, 2024) – Bristol Myers Squibb (NYSE: BMY) today reports results for the first quarter of 2024, which reflect meaningful progress in the company's growth portfolio and pipeline.
“We had a good start to 2024, with revenue growth, important advances in our pipeline and the closure of several strategically important transactions,” said Christopher Boerner, Ph.D., board chair and chief executive officer, Bristol Myers Squibb. “Our focus remains on strengthening the company's long-term growth profile. As a part of our continued evolution, we're executing a strategic productivity initiative that will allow us to be more agile, drive efficiency across the company, and prioritize investing in opportunities where we see the greatest potential to get the most promising medicines to patients as quickly as possible." * GAAP and Non-GAAP loss per share include the net impact of Acquired IPRD charges and licensing income primarily driven by the Karuna Therapeutics asset acquisition and SystImmune collaboration.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
First Quarter |
|
|
$ in millions, except per share amounts |
2024 |
|
2023 |
|
Change |
|
Change Excl. F/X** |
|
|
|
|
|
|
|
|
| Total Revenues |
$11,865 |
|
|
$11,337 |
|
|
5 |
% |
|
6 |
% |
|
|
|
|
|
|
|
|
(Loss)/Earnings Per Share — GAAP* |
(5.89) |
|
|
1.07 |
|
|
N/A |
|
N/A |
|
|
|
|
|
|
|
|
(Loss)/Earnings Per Share — Non-GAAP* ** |
(4.40) |
|
|
2.05 |
|
|
N/A |
|
N/A |
|
|
|
|
|
|
|
|
Acquired IPRD charge and Licensing Income Net Impact on Earnings Per Share |
(6.30) |
|
|
(0.01) |
|
|
N/A |
|
N/A |
|
|
|
|
|
|
|
|
** See "Use of Non-GAAP Financial Information".
FIRST QUARTER RESULTS
All comparisons are made versus the same period in 2023 unless otherwise stated.
•Bristol Myers Squibb posted first quarter revenues of $11.9 billion, an increase of 5% or 6% when adjusted for foreign exchange impacts, primarily driven by Eliquis, Reblozyl and Opdualag, partially offset by Opdivo and Revlimid.
•U.S. revenues increased 7% to $8.5 billion primarily due to Eliquis, Reblozyl and Opdualag, partially offset by Revlimid. Opdivo revenues were $1.2 billion compared to $1.3 billion, representing a decrease of 10% primarily due to inventory and the timing of orders, partially offset by demand growth.
•International revenues remained relatively flat at $3.4 billion primarily due to lower average net selling prices, offset in part by higher demand for Opdivo, Yervoy and Reblozyl. The negative impact from foreign exchange was 5%.
•On a GAAP basis, gross margin decreased from 77.4% to 75.3%, and on a non-GAAP basis, gross margin decreased from 77.8% to 75.5%, primarily due to product mix.
•On a GAAP basis, marketing, selling and administrative expenses increased 34% to $2.4 billion and on a non-GAAP basis, increased 13% to $2.0 billion, primarily due to the timing of spend and the impact of recent acquisitions.
•On a GAAP basis, research and development expenses increased 16% to $2.7 billion and on a non-GAAP basis, increased 6% to $2.3 billion, primarily due to the impact of recent acquisitions and higher costs to support the overall portfolio.
•On a GAAP and non-GAAP basis, Acquired IPRD increased to $12.9 billion from $75 million, primarily due to the Karuna asset acquisition and SystImmune collaboration. On a GAAP and non-GAAP basis, licensing income was $12 million compared to $43 million during the same period a year ago.
•On a GAAP basis, amortization of acquired intangible assets increased 4% to $2.4 billion, primarily due to the Mirati Therapeutics and RayzeBio acquisitions and approval of Augtyro in the fourth quarter of 2023.
•On a GAAP basis, income tax expense was $392 million on a pre-tax loss of $11.5 billion and on a non-GAAP basis, income tax expense was $732 million on a pre-tax loss of $8.2 billion primarily due to the $12.1 billion one-time, non-tax-deductible charge for the acquisition of Karuna.
•On a GAAP basis, the company reported net loss attributable to Bristol Myers Squibb of $11.9 billion, or ($5.89) per share, during the first quarter of 2024 compared to net earnings of $2.3 billion, or $1.07 per share, for the same period a year ago. In addition to the items above, the decrease was also due to lower litigation and other settlement income. The company reported on a non-GAAP basis net loss attributable to Bristol Myers Squibb of $8.9 billion, or ($4.40) per share, during the first quarter of 2024 compared to net earnings of $4.3 billion, or $2.05 per share, for the same period a year ago. In addition to the items above, the decrease was also due to higher interest expense resulting from new debt issuance to fund recent acquisitions.
FIRST QUARTER PRODUCT REVENUE HIGHLIGHTS
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ($ amounts in millions) |
|
Quarter Ended March 31, 2024 |
|
% Change from Quarter Ended March 31, 2023 |
|
% Change from Quarter Ended March 31, 2023 Ex-F/X** |
| |
|
U.S. |
|
Int'l (c) |
|
WW(d) |
|
U.S. |
|
Int'l(c) |
|
WW(d) |
|
Int'l(c) |
|
WW(d) |
Growth Portfolio |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Opdivo |
|
$ |
1,155 |
|
|
$ |
923 |
|
|
$ |
2,078 |
|
|
(10) |
% |
|
— |
% |
|
(6) |
% |
|
9 |
% |
|
(2) |
% |
| Orencia |
|
572 |
|
|
226 |
|
|
798 |
|
|
4 |
% |
|
6 |
% |
|
4 |
% |
|
13 |
% |
|
6 |
% |
| Yervoy |
|
368 |
|
|
215 |
|
|
583 |
|
|
18 |
% |
|
10 |
% |
|
15 |
% |
|
17 |
% |
|
18 |
% |
| Reblozyl |
|
293 |
|
|
61 |
|
|
354 |
|
|
88 |
% |
|
22 |
% |
|
72 |
% |
|
22 |
% |
|
72 |
% |
| Opdualag |
|
198 |
|
|
8 |
|
|
206 |
|
|
71 |
% |
|
* |
|
76 |
% |
|
* |
|
76 |
% |
| Abecma |
|
52 |
|
|
30 |
|
|
82 |
|
|
(56) |
% |
|
3 |
% |
|
(44) |
% |
|
7 |
% |
|
(44) |
% |
| Zeposia |
|
72 |
|
|
38 |
|
|
110 |
|
|
41 |
% |
|
41 |
% |
|
41 |
% |
|
41 |
% |
|
41 |
% |
| Breyanzi |
|
87 |
|
|
20 |
|
|
107 |
|
|
50 |
% |
|
54 |
% |
|
51 |
% |
|
54 |
% |
|
51 |
% |
| Camzyos |
|
77 |
|
|
7 |
|
|
84 |
|
|
* |
|
N/A |
|
* |
|
N/A |
|
* |
| Sotyktu |
|
34 |
|
|
10 |
|
|
44 |
|
|
* |
|
* |
|
* |
|
* |
|
* |
| Augtyro |
|
6 |
|
|
— |
|
|
6 |
|
|
N/A |
|
N/A |
|
N/A |
|
N/A |
|
N/A |
| Krazati |
|
21 |
|
|
— |
|
|
21 |
|
|
N/A |
|
N/A |
|
N/A |
|
N/A |
|
N/A |
Other Growth Products(a) |
|
148 |
|
|
171 |
|
|
319 |
|
|
3 |
% |
|
26 |
% |
|
14 |
% |
|
30 |
% |
|
16 |
% |
Total Growth Portfolio |
|
3,083 |
|
|
1,709 |
|
|
4,792 |
|
|
9 |
% |
|
8 |
% |
|
8 |
% |
|
15 |
% |
|
11 |
% |
Legacy Portfolio |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Eliquis |
|
2,821 |
|
|
899 |
|
|
3,720 |
|
|
12 |
% |
|
— |
% |
|
9 |
% |
|
1 |
% |
|
9 |
% |
| Revlimid |
|
1,453 |
|
|
216 |
|
|
1,669 |
|
|
(5) |
% |
|
(5) |
% |
|
(5) |
% |
|
(1) |
% |
|
(4) |
% |
| Pomalyst/Imnovid |
|
597 |
|
|
268 |
|
|
865 |
|
|
10 |
% |
|
(8) |
% |
|
4 |
% |
|
(7) |
% |
|
4 |
% |
| Sprycel |
|
282 |
|
|
92 |
|
|
374 |
|
|
(2) |
% |
|
(34) |
% |
|
(13) |
% |
|
(30) |
% |
|
(11) |
% |
| Abraxane |
|
145 |
|
|
72 |
|
|
217 |
|
|
(10) |
% |
|
(8) |
% |
|
(9) |
% |
|
10 |
% |
|
(3) |
% |
Other Legacy Products (b) |
|
95 |
|
|
133 |
|
|
228 |
|
|
19 |
% |
|
(20) |
% |
|
(7) |
% |
|
(17) |
% |
|
(6) |
% |
Total Legacy Portfolio |
|
5,393 |
|
|
1,680 |
|
|
7,073 |
|
|
5 |
% |
|
(7) |
% |
|
2 |
% |
|
(4) |
% |
|
3 |
% |
| Total Revenues |
|
$ |
8,476 |
|
|
$ |
3,389 |
|
|
$ |
11,865 |
|
|
7 |
% |
|
— |
% |
|
5 |
% |
|
5 |
% |
|
6 |
% |
* In excess of +100%
** See "Use of Non-GAAP Financial Information".
(a) Includes Nulojix, Onureg, Inrebic, Empliciti and royalty revenue.
(b) Includes other mature brands.
(c) Beginning in 2024, Puerto Rico revenues are included in International revenues. Prior period amounts have been reclassified to conform to the current presentation.
(d) Worldwide (WW) includes U.S. and International (Int'l).
FIRST QUARTER PRODUCT REVENUE HIGHLIGHTS
Growth Portfolio
Growth Portfolio worldwide revenues increased to $4.8 billion compared to $4.4 billion in the prior year period, representing growth of 8%, or 11% when adjusted for foreign exchange impacts. Growth Portfolio revenues were primarily driven by higher demand for Reblozyl, Opdualag, Yervoy, Camzyos, and Sotyktu, partially offset by Opdivo and Abecma.
Legacy Portfolio
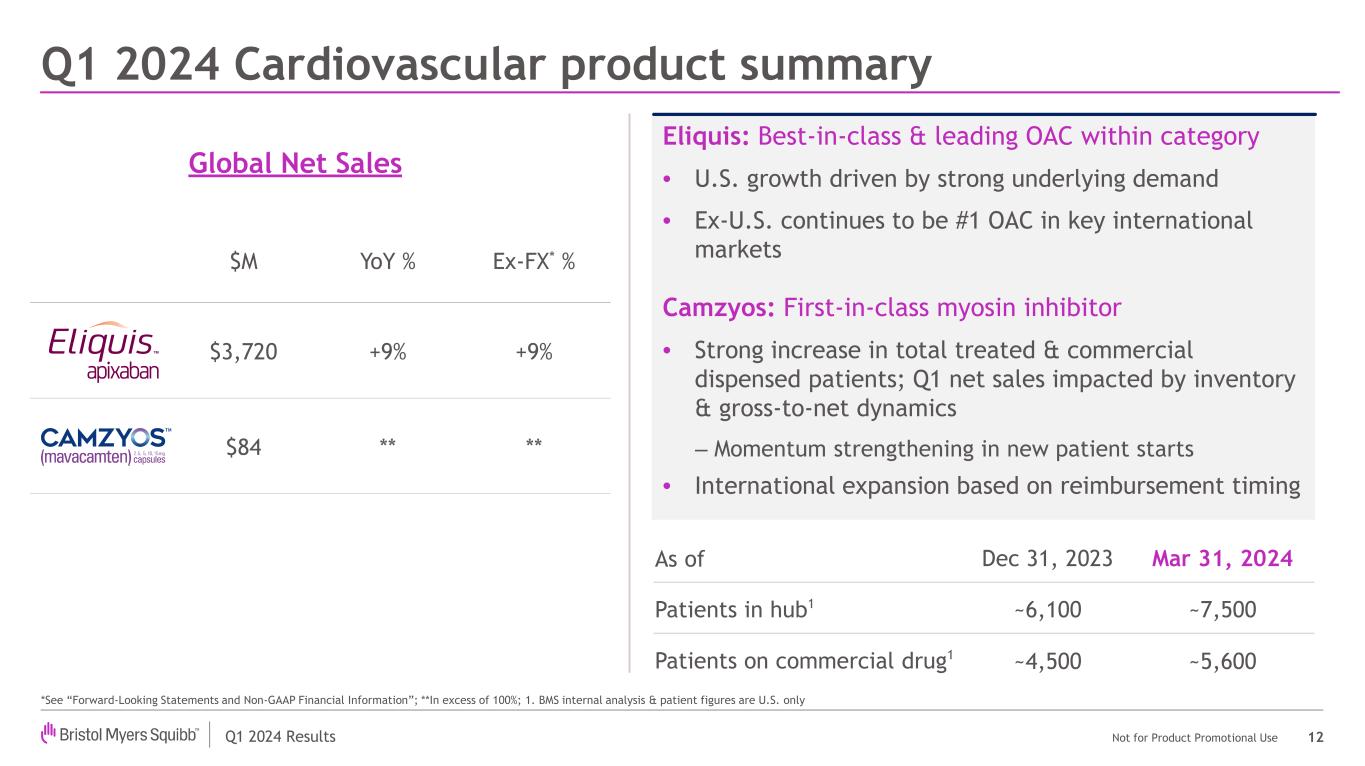
Revenues for the Legacy Portfolio in the first quarter were $7.1 billion compared to $6.9 billion in the prior year period. Legacy Portfolio revenues were largely driven by a 9% increase in Eliquis worldwide revenues on a reported basis and when adjusted for foreign exchange impacts, partially offset by a decline in Revlimid worldwide revenues of 5%, or 4% when adjusted for foreign exchange impacts.
PRODUCT AND PIPELINE UPDATE
Cardiovascular
|
|
|
|
|
|
|
|
|
| Category |
Asset |
Milestone |
| Clinical & Research |
Camzyos® (mavacamten) |
An analysis of results from the 10-month post-launch evaluation of the Camzyos REMS Program in 1,524 patients demonstrated that approximately 1% of patients reported clinical heart failure requiring hospitalization and 2.8% of patients reported a decrease in left ventricular ejection fraction to <50%. |
Oncology
|
|
|
|
|
|
|
|
|
| Category |
Asset |
Milestone |
| Regulatory |
Opdivo® (nivolumab) |
The U.S. Food and Drug Administration (FDA) approved Opdivo, in combination with cisplatin and gemcitabine, for the first-line treatment of adult patients with unresectable or metastatic urothelial carcinoma (UC). The approval is based on results from the Phase 3 CheckMate -901 trial evaluating Opdivo in combination with cisplatin and gemcitabine followed by Opdivo monotherapy, compared to cisplatin-gemcitabine alone, for patients with previously untreated unresectable or metastatic UC. |
|
Krazati® (adagrasib) |
The FDA accepted the supplemental New Drug Application (sNDA) for Krazati in combination with cetuximab for the treatment of patients with previously treated KRASG12C -mutated locally advanced or metastatic colorectal cancer. The acceptance was based on the results of the Phase 1/2 KRYSTAL-1 trial. The FDA granted the application Priority Review and assigned a Prescription Drug User Fee Act (PDUFA) goal date of June 21, 2024. |
|
AugtyroTM (repotrectinib) |
The FDA accepted the sNDA for Augtyro for the treatment of adult and pediatric patients 12 years of age and older with NTRK-positive locally advanced or metastatic solid tumors. The acceptance is based on results from the registrational Phase 1/2 TRIDENT-1 trial and the CARE study. The FDA granted the application Priority Review and assigned a PDUFA goal date of June 15, 2024. |
|
Opdivo |
The FDA accepted the supplemental Biologics Application (sBLA) for neoadjuvant Opdivo for the perioperative treatment of resectable stage IIA to IIIB non-small cell lung cancer (NSCLC). The FDA assigned a PDUFA goal date of October 8, 2024.
In addition, the European Medicines Agency (EMA) validated the type II variation application for neoadjuvant Opdivo with chemotherapy followed by surgery and adjuvant Opdivo for the perioperative treatment of resectable stage IIA to IIIB NSCLC. Application validation confirms the submission is complete and begins the EMA’s centralized review procedure.
The FDA’s sBLA acceptance and the EMA’s application validation are based on results from the Phase 3 CheckMate -77T trial.
|
|
|
|
|
|
|
|
|
|
| Clinical & Research |
Krazati |
The pivotal Phase 3 KRYSTAL-12 study, evaluating Krazati as a monotherapy in patients with pretreated locally advanced or metastatic NSCLC harboring a KRASG12C mutation, met the primary endpoint of progression-free survival (PFS) and the key secondary endpoint of overall response rate as assessed by Blinded Independent Central Review at final analysis for these endpoints. The study remains ongoing to assess the additional key secondary endpoint of overall survival. |
|
Krazati |
Data from the cohorts of the Phase 1/2 KRYSTAL-1 study evaluating Krazati in combination with cetuximab for the treatment of patients with previously treated KRASG12C-mutated locally advanced or metastatic colorectal cancer demonstrated clinically meaningful activity. With a median follow up of 11.9 months in 94 patients, Krazati plus cetuximab demonstrated an objective response rate of 34%, median PFS of 6.9 months, and median overall survival of 15.9 months in pre-treated patients. |
|
OpdualagTM (nivolumab and relatlimab) |
Initial data from a randomized Phase 2 study evaluating Opdualag in NSCLC support the initiation of a Phase 3 trial in 2024 evaluating Opdualag plus chemotherapy versus PD-1/PD-L1 plus chemotherapy in an important segment of the disease. Phase 2 data is expected to be disclosed later this year. |
|
Opdivo+Yervoy |
The Phase 3 CheckMate -9DW trial evaluating Opdivo plus Yervoy as a first-line treatment for patients with advanced hepatocellular carcinoma who have not received a prior systemic therapy met its primary endpoint of improved overall survival compared to investigator's choice of sorafenib or lenvatinib at a pre-specified interim analysis. |
Hematology
|
|
|
|
|
|
|
|
|
| Category |
Asset |
Milestone |
| Regulatory |
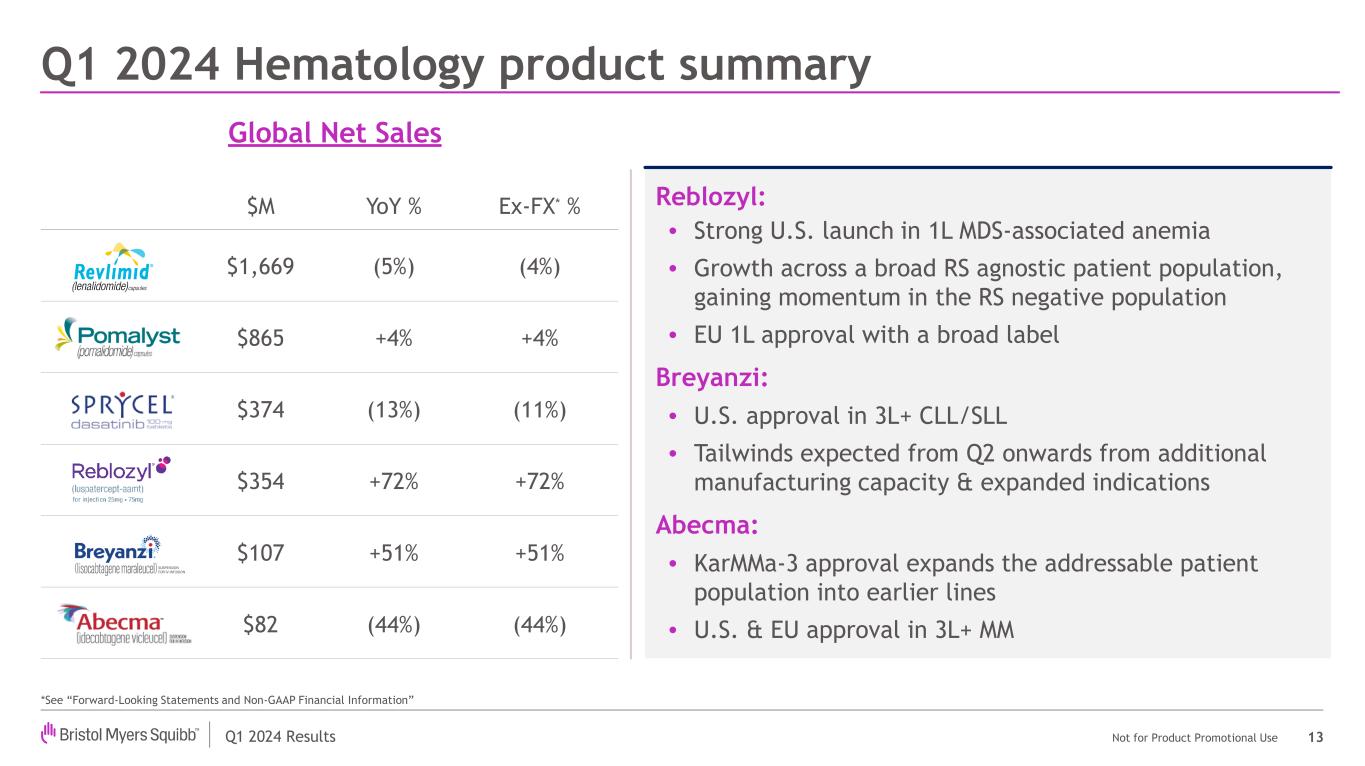
Abecma® (idecabtagene vicleucel) |
The European Commission (EC) approved Abecma for the treatment of adult patients with relapsed and refractory multiple myeloma who have received at least two prior therapies, including an immunomodulatory agent, a proteasome inhibitor, and an anti-CD38 antibody and have demonstrated disease progression on the last therapy. The approval is based on results from the Phase 3 KarMMa-3 trial. Abecma is the first CAR T cell immunotherapy approved in the European Union for use in earlier lines of therapy for relapsed and refractory multiple myeloma. |
|
Abecma |
The FDA approved Abecma for the treatment of adult patients with relapsed or refractory multiple myeloma after two or more prior lines of therapy, including an immunomodulatory agent, a proteasome inhibitor, and an anti-CD38 monoclonal antibody. The approval is based on results from the Phase 3 KarMMa-3 trial.
Abecma is being jointly developed and commercialized in the U.S. by Bristol Myers Squibb and 2seventy bio, Inc.
|
|
Breyanzi® (lisocabtagene maraleucel) |
The FDA granted accelerated approval of Breyanzi for the treatment of adult patients with relapsed or refractory chronic lymphocytic leukemia or small lymphocytic lymphoma who have received at least two prior lines of therapy, including a Bruton tyrosine kinase inhibitor and a B-cell lymphoma 2 inhibitor. The accelerated approval is based on the Phase 1/2 open-label, single-arm TRANSCEND CLL 004 trial. |
|
|
|
|
|
|
|
|
|
|
Reblozyl® (luspatercept-aamt) |
The EC expanded approval of Reblozyl to include the first-line treatment of adult patients with transfusion-dependent anemia due to very low, low and intermediate-risk myelodysplastic syndromes. The approval covers all European Union member states and is based on the pivotal Phase 3 COMMANDS trial. |
Immunology
|
|
|
|
|
|
|
|
|
| Category |
Asset |
Milestone |
| Clinical & Research |
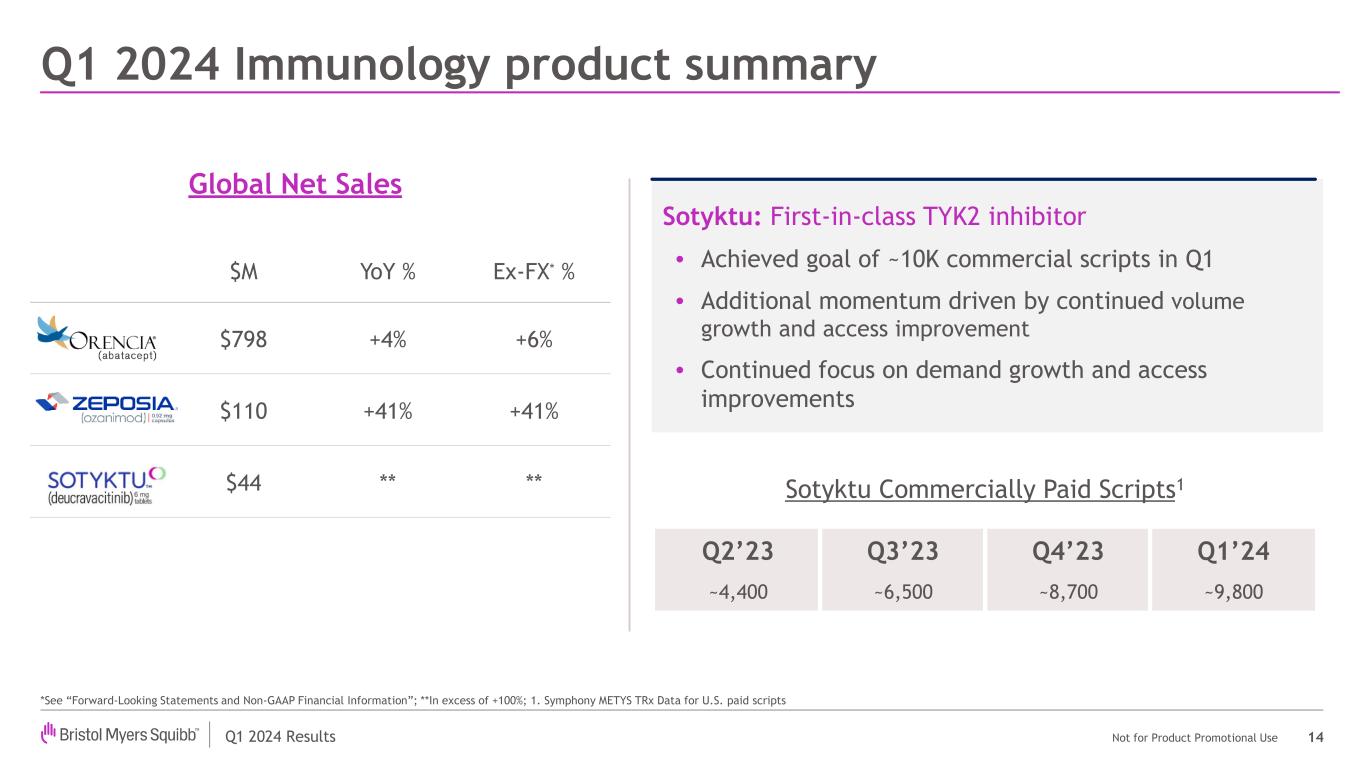
Zeposia® (ozanimod) |
First of two induction Phase 3 YELLOWSTONE trials evaluating Zeposia in adult patients with moderate-to-severe active Crohn's disease did not meet its primary endpoint of clinical remission at Week 12. The safety profile of Zeposia in this study was consistent with that observed in previously reported trials.
|
|
Zeposia |
Data from the Phase 3 DAYBREAK open-label extension trial demonstrated the long-term efficacy and safety profile of Zeposia in patients with relapsing forms of multiple sclerosis. In the DAYBREAK long-term extension study, treatment with Zeposia demonstrated a low annualized relapse rate of 0.098 and 67% of patients were relapse-free at six years. An analysis of DAYBREAK data showed nearly 97% of followed patients were relapse-free at 90 days post Zeposia discontinuation. Patients that did relapse showed no evidence of rebound effect. |
Neuroscience
|
|
|
|
|
|
|
|
|
| Category |
Asset |
Milestone |
| Clinical & Research |
KarXT (xanomeline-trospium) |
Interim long-term efficacy data from the Phase 3 EMERGENT-4 open-label extension trial demonstrated that KarXT was associated with significant improvement in symptoms of schizophrenia across all efficacy measures at 52 weeks.
In addition, pooled interim long-term safety, tolerability and metabolic outcomes data from the Phase 3 EMERGENT-4 and EMERGENT-5 trials evaluating the safety, tolerability and efficacy of KarXT in adults with schizophrenia showed that KarXT demonstrated a favorable weight and long-term metabolic profile where most patients experience stability or improvements on key metabolic parameters over 52 weeks of treatment. KarXT was generally well-tolerated, with a side effect profile consistent with prior trials of KarXT in schizophrenia.
|
Business Development
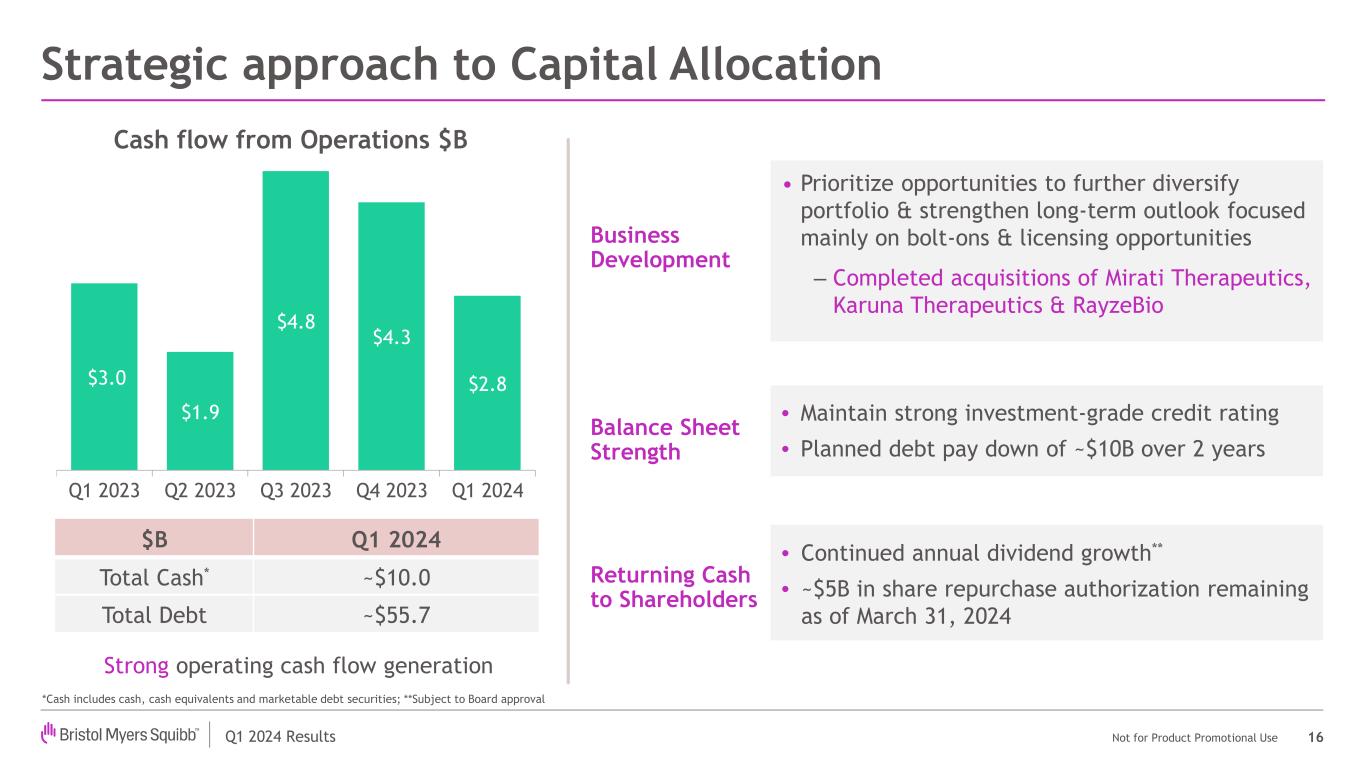
The company recently completed multiple transactions, strengthening its long-term growth profile and enhancing its portfolio and pipeline.
•With the acquisition of Karuna Therapeutics, Inc., Bristol Myers Squibb expanded its position in neuroscience and added important assets, including KarXT, an antipsychotic with a novel mechanism of action and differentiated efficacy and safety. KarXT has a PDUFA goal date of September 26, 2024, for the treatment of schizophrenia in adults.
•By acquiring RayzeBio, Inc., a clinical-stage radiopharmaceutical therapeutics company with a differentiated platform and state-of-the-art manufacturing capabilities, Bristol Myers Squibb further diversified its oncology portfolio and gained a rich pipeline of potentially first-in-class and best-in-class drug development programs currently targeting solid tumors.
•Through the acquisition of Mirati Therapeutics, Inc., Bristol Myers Squibb strengthened its pipeline and added commercialized lung cancer medicine Krazati to its oncology portfolio, as well as several promising clinical assets.
•The company completed an exclusive license and collaboration agreement with SystImmune to develop and commercialize a potentially first-in-class EGFRxHER3 bispecific antibody-drug conjugate with the potential to treat a variety of solid tumors, including lung and breast cancer.
On April 22, Bristol Myers Squibb and Cellares announced a worldwide capacity reservation and supply agreement for the manufacture of CAR T cell therapies. As a part of the agreement, Cellares will optimize, automate, and tech-transfer select Bristol Myers Squibb CAR T cell therapies onto its automated and high-throughput manufacturing platform, the Cell ShuttleTM. This agreement enables Bristol Myers Squibb to expand its manufacturing capacity, meeting the growing demand for its diverse range of cell therapies through a platform that is scalable and has the potential to improve turnaround time, bringing the promise of cell therapies to more patients faster.
Strategically Enhancing Productivity and Efficiency
Bristol Myers Squibb is executing a strategic productivity initiative that will drive approximately $1.5 billion in cost savings by the end of 2025, the majority of which will be reinvested to fund innovation and drive growth.
As a part of this initiative, the company is:
•Focusing resources on R&D programs with the potential to deliver the greatest return on investment;
•Prioritizing investing in key growth brands; and
•Optimizing operations across the organization.
Company executives will provide additional details on these actions during the company's first quarter 2024 earnings conference call.
Financial Guidance
As previously communicated, Bristol Myers Squibb is updating portions of its 2024 line-item guidance, including Non-GAAP EPS, to reflect the impact of recent transactions.
•Non-GAAP EPS was updated to account for the following:
|
|
|
|
|
|
2024 Non-GAAP EPS Guidance |
February Diluted EPS (Prior) |
$7.10 - $7.40 |
Acquired IPRD Impact1 |
($6.30) |
Dilution Impact (RayzeBio) |
($0.13) |
Dilution Impact (Karuna Therapeutics) |
($0.30) |
Total Deals Impact |
($6.73) |
Revised Diluted EPS |
$0.40 - $0.70 |
1 Primarily represents the Acquired IPRD impact from the Karuna Therapeutics asset acquisition and SystImmune
collaboration.
•Non-GAAP other income/(expense) was updated primarily due to the financing of the recent acquisitions.
•Non-GAAP tax rate was updated to approximately 69% to reflect the impact of a $12.1 billion one-time, non-tax-deductible IPRD charge from the Karuna Therapeutics acquisition, which is expected to contribute 51% to the full-year tax rate.
•2024 line-item guidance updates are:
|
|
|
|
|
|
|
|
|
|
Non-GAAP2
|
February
(Prior) |
April
(Revised) |
Total Revenues |
Low single-digit increase |
No Change |
|
Total Revenues
(excl. F/X)
|
Low single-digit increase |
No Change |
| Gross Margin % |
~74% |
No Change |
Operating Expenses1 |
Low single-digit increase |
No Change |
Other income/(expense) |
~$250M |
~($250M) |
Effective tax rate |
~17.5% |
~69% |
| Diluted EPS |
$7.10 to $7.40 |
$0.40 to $0.70 |
1 Operating Expenses = MS&A and R&D, excluding Acquired IPRD and Amortization of acquired intangible assets.
2 See "Use of Non-GAAP Financial Information."
The 2024 financial guidance excludes the impact of any potential future strategic acquisitions, divestitures, specified items that have not yet been identified and quantified, and the impact of future Acquired IPRD charges. To the extent we have quantified the impact of significant R&D charges or other income resulting from upfront or contingent milestone payments in connection with asset acquisitions or licensing of third-party intellectual property rights, we may update this information from time to time on our website www.bms.com, in the "Investors" section. Non-GAAP guidance assumes current exchange rates. The financial guidance is subject to risks and uncertainties applicable to all forward-looking statements as described elsewhere in this press release.
A reconciliation of forward-looking non-GAAP measures, including non-GAAP EPS, to the most directly comparable GAAP measures is not provided because comparable GAAP measures for such measures are not reasonably accessible or reliable due to the inherent difficulty in forecasting and quantifying measures that would be necessary for such reconciliation. Namely, we are not without unreasonable effort, able to reliably predict the impact of accelerated depreciation and impairment charges, legal and other settlements, gains and losses from equity investments and other adjustments. In addition, the company believes such a reconciliation would imply a degree of precision and certainty that could be confusing to investors. These items are uncertain, depend on various factors and may have a material impact on our future GAAP results. See "Cautionary Statement Regarding Forward-Looking Statements" and "Use of Non-GAAP Financial Information."
Environmental, Social & Governance (ESG)
As a leading biopharmaceutical company, Bristol Myers Squibb's passion for making an impact extends beyond the discovery, development and delivery of innovative medicines that help patients prevail over serious diseases.
•On April 2, 2024, the company published its latest ESG report, which details the company's meaningful progress, its evolved ESG strategy, and its long-term aspirational ESG goals. The company's evolved approach further integrates its ESG strategy and its business strategy. The ESG strategy focuses on three core pillars: advancing patient health around the world, expanding the boundaries of science, and fostering a high-performing and inclusive global workforce.
Highlights from the report include:
◦Advancing tailored access programs in low-and middle-income countries;
◦Making clinical trials more accessible to underrepresented groups and ensuring research efforts better reflect patient populations;
◦Sustained workforce representation and increased community engagement by the company's employees;
◦Advancing climate action goals and increasing the company's renewable energy footprint; and ◦Incorporating new ESG metrics into the executive compensation program and advancing data privacy and cybersecurity efforts.
Conference Call Information
Bristol Myers Squibb will host a conference call today, Thursday, April 25, 2024, at 8:00 a.m. ET during which company executives will review quarterly financial results and address inquiries from investors and analysts. Investors and the general public are invited to listen to a live webcast of the call at http://investor.bms.com.
Investors and the public can register for the live conference call here. Those unable to register can access the live conference call by dialing in the U.S. toll-free 1-833-816-1116 or international +1 412-317-0705. Materials related to the call will be available at http://investor.bms.com prior to the start of the conference call.
A replay of the webcast will be available at http://investor.bms.com approximately three hours after the conference call concludes. A replay of the conference call will be available beginning at 11:30 a.m. ET on April 25, 2024, through 11:30 a.m. ET on May 9, 2024, by dialing in the U.S. toll free 1-877-344-7529 or international +1 412-317-0088, confirmation code: 5034750.
About Bristol Myers Squibb
Bristol Myers Squibb is a global biopharmaceutical company whose mission is to discover, develop and deliver innovative medicines that help patients prevail over serious diseases. For more information about Bristol Myers Squibb, visit us at BMS.com or follow us on LinkedIn, Twitter, YouTube, Facebook, and Instagram.
###
corporatefinancial-news
For more information, contact:
Media: media@bms.com
Investor Relations: investor.relations@bms.com
Use of Non-GAAP Financial Information
In discussing financial results and guidance, the company refers to financial measures that are not in accordance with U.S. Generally Accepted Accounting Principles (GAAP).
The non-GAAP financial measures are provided as supplemental information to the financial measures presented in this press release that are calculated and presented in accordance with GAAP and are presented because management has evaluated the company’s financial results both including and excluding the adjusted items or the effects of foreign currency translation, as applicable, and believes that the non-GAAP financial measures presented portray the results of the company's baseline performance, supplement or enhance management's, analysts' and investors' overall understanding of the company’s underlying financial performance and trends and facilitate comparisons among current, past and future periods. In addition, non-GAAP gross margin, which is gross profit excluding certain specified items, as a percentage of revenues, non-GAAP operating margin, which is gross profit less marketing, selling and administrative expenses and research and development expenses excluding certain specified items as a percentage of revenues, non-GAAP operating expenses, which is marketing, selling and administrative and research and development expenses excluding certain specified items, non-GAAP marketing, selling and administrative expenses, which is marketing, selling and administrative expenses excluding certain specified items, and non-GAAP research and development expenses, which is research and development expenses excluding certain specified items, are relevant and useful for investors because they allow investors to view performance in a manner similar to the method used by our management and make it easier for investors, analysts and peers to compare our operating performance to other companies in our industry and to compare our year-over-year results.
This earnings release and the accompanying tables also provide certain revenues and expenses as well as non-GAAP measures excluding the impact of foreign exchange ("Ex-Fx"). We calculate foreign exchange impacts by converting our current-period local currency financial results using the prior period average currency rates and comparing these adjusted amounts to our current-period results. Ex-Fx financial measures are not accounted for according to GAAP because they remove the effects of currency movements from GAAP results.
Non-GAAP financial measures such as non-GAAP earnings and related EPS information are adjusted to exclude certain costs, expenses, gains and losses and other specified items that are evaluated on an individual basis after considering their quantitative and qualitative aspects and typically have one or more of the following characteristics, such as being highly variable, difficult to project, unusual in nature, significant to the results of a particular period or not indicative of past or future operating results. These items are excluded from non-GAAP earnings and related EPS information because the company believes they neither relate to the ordinary course of the company’s business nor reflect the company’s underlying business performance. Similar charges or gains were recognized in prior periods and will likely reoccur in future periods, including amortization of acquired intangible assets, including product rights that generate a significant portion of our ongoing revenue and will recur until the intangible assets are fully amortized, unwind of inventory purchase price adjustments, acquisition and integration expenses, restructuring costs, accelerated depreciation and impairment of property, plant and equipment and intangible assets, costs of acquiring a priority review voucher, stock compensation resulting from acquisition-related equity awards, pension, legal and other contractual settlement charges, equity investment and contingent value rights fair value adjustments (including fair value adjustments attributed to limited partnership equity method investments), income resulting from the change in control of the Nimbus Therapeutics TYK2 Program and amortization of fair value adjustments of debt acquired from Celgene in our 2019 exchange offer, among other items. Deferred and current income taxes attributed to these items are also adjusted for considering their individual impact to the overall tax expense, deductibility and jurisdictional tax rates.
Because the non-GAAP financial measures are not calculated in accordance with GAAP, they should not be considered superior to and are not intended to be considered in isolation or as a substitute for the related financial measures presented in the press release that are prepared in accordance with GAAP and may not be the same as or comparable to similarly titled measures presented by other companies due to possible differences in method and in the items being adjusted.
We encourage investors to review our financial statements and publicly-filed reports in their entirety and not to rely on any single financial measure.
Reconciliations of the non-GAAP financial measures to the most comparable GAAP measures are provided in the accompanying financial tables and will also be available on the company’s website at www.bms.com. Within the accompanying financial tables presented, certain columns and rows may not add due to the use of rounded numbers. Percentages and earnings per share amounts presented are calculated from the underlying amounts.
A reconciliation of forward-looking non-GAAP measures, including non-GAAP EPS, to the most directly comparable GAAP measures is not provided because comparable GAAP measures for such measures are not reasonably accessible or reliable due to the inherent difficulty in forecasting and quantifying measures that would be necessary for such reconciliation. Namely, we are not, without unreasonable effort, able to reliably predict the impact of accelerated depreciation and impairment charges, legal and other settlements, gains and losses from equity investments and other adjustments. In addition, the company believes such a reconciliation would imply a degree of precision and certainty that could be confusing to investors. These items are uncertain, depend on various factors and may have a material impact on our future GAAP results.
Website Information
We routinely post important information for investors on our website, BMS.com, in the “Investors” section. We may use this website as a means of disclosing material, non-public information and for complying with our disclosure obligations under Regulation FD. Accordingly, investors should monitor the Investors section of our website, in addition to following our press releases, Securities and Exchange Commission ("SEC") filings, public conference calls, presentations and webcasts. We may also use social media channels to communicate with our investors and the public about our company, our products and other matters, and those communications could be deemed to be material information. The information contained on, or that may be accessed through, our website or social media channels are not incorporated by reference into, and are not a part of, this document.
Cautionary Statement Regarding Forward-Looking Statements
This earnings release and the related attachments (as well as the oral statements made with respect to information contained in this release and the attachments) contain certain “forward-looking” statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended, regarding, among other things, the company’s 2024 financial guidance, plans and strategy, including its business development and capital allocation strategy, anticipated developments in the company’s pipeline, expectations with respect to the company’s future market position and the projected benefits of the company’s alliances and other business development activities. These statements may be identified by the fact that they use words such as “should,” “could,” “expect,” “anticipate,” “estimate,” “target,” “may,” “project,” “guidance,” “intend,” “plan,” “believe,” “will” and other words and terms of similar meaning and expression in connection with any discussion of future operating or financial performance, although not all forward-looking statements contain such terms. All statements that are not statements of historical facts are, or may be deemed to be, forward-looking statements. No forward-looking statement can be guaranteed and there is no assurance that the company will achieve its financial guidance and long-term targets, that the company’s future clinical studies will support the data described in this release, that the company’s product candidates will receive necessary clinical and manufacturing regulatory approvals, that the company’s pipeline products will prove to be commercially successful, that clinical and manufacturing regulatory approvals will be sought or obtained within currently expected timeframes, or that contractual milestones will be achieved.
Forward-looking statements are based on current expectations and projections about the company’s future financial results, goals, plans and objectives and involve inherent risks, assumptions and uncertainties, including internal or external factors that could delay, divert or change any of them in the next several years, that are difficult to predict, may be beyond the company’s control and could cause the company’s future financial results, goals, plans and objectives to differ materially from those expressed in, or implied by, the statements. Such risks, uncertainties and other matters include, but are not limited to: increasing pricing pressures from market access, pharmaceutical pricing controls and discounting; market actions taken by private and government payers to manage drug utilization and contain costs; the company’s ability to retain patent exclusivity of certain products; regulatory changes that result in lower prices, lower reimbursement rates and smaller populations for whom payers will reimburse; changes under the 340B Drug Pricing Program; the company’s ability to obtain and maintain regulatory approval for its product candidates; the company’s ability to obtain and protect market exclusivity rights and enforce patents and other intellectual property rights; the possibility of difficulties and delays in product introduction and commercialization; increasing industry competition; potential difficulties, delays and disruptions in manufacturing, distribution or sale of products; the company’s ability to identify potential strategic acquisitions, licensing opportunities or other beneficial transactions; failure to complete, or delays in completing, collaborations, acquisitions, divestitures, alliances and other portfolio actions and the failure to achieve anticipated benefits from such transactions and actions; the risk of an adverse patent litigation decision or settlement and exposure to other litigation and/or regulatory actions or investigations; the impact of any healthcare reform and legislation or regulatory action in the United States and international markets; increasing market penetration of lower-priced generic products; the failure of the company’s suppliers, vendors, outsourcing partners, alliance partners and other third parties to meet their contractual, regulatory and other obligations; the impact of counterfeit or unregistered versions of the company’s products and from stolen products; product label changes or other measures that could reduce the product's market acceptance for the company's products and result in declining sales; safety or efficacy concerns regarding the company’s products or any product in the same class as the company’s products; the risk of cyber-attacks on the company’s information systems or products and unauthorized disclosure of trade secrets or other confidential data; the company’s ability to execute its financial, strategic and operational plans; the company’s dependency on several key products; any decline in the company’s future royalty streams; the company’s ability to attract and retain key personnel; the impact of the company’s significant indebtedness; political and financial instability of international economies and sovereign risk; interest rate and currency exchange rate fluctuations, credit and foreign exchange risk management; risks relating to the use of social media platforms; the impact of our exclusive forum provision in our by-laws for certain lawsuits on our stockholders’ ability to obtain a judicial forum that they find favorable for such lawsuits; issuance of new or revised accounting standards; and risks relating to public health outbreaks, epidemics and pandemics.
Forward-looking statements in this earnings release should be evaluated together with the many risks and uncertainties that affect the company’s business and market, particularly those identified in the cautionary statement and risk factors discussion in the company’s Annual Report on Form 10-K for the year ended December 31, 2023, as updated by the company’s subsequent Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and other filings with the SEC.
The forward-looking statements included in this document are made only as of the date of this document and except as otherwise required by applicable law, the company undertakes no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events, changed circumstances or otherwise.
BRISTOL-MYERS SQUIBB COMPANY
CONSOLIDATED STATEMENTS OF EARNINGS
FOR THE THREE MONTHS ENDED MARCH 31, 2024 AND 2023
(Unaudited, dollars and shares in millions except per share data)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
Three Months Ended March 31, |
|
|
| |
2024 |
|
2023 |
|
|
|
|
|
|
|
|
|
|
|
|
| Net product sales |
$ |
11,559 |
|
|
$ |
11,048 |
|
|
|
|
|
| Alliance and other revenues |
306 |
|
|
289 |
|
|
|
|
|
| Total Revenues |
11,865 |
|
|
11,337 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of products sold(a) |
2,932 |
|
|
2,566 |
|
|
|
|
|
| Marketing, selling and administrative |
2,367 |
|
|
1,762 |
|
|
|
|
|
| Research and development |
2,695 |
|
|
2,321 |
|
|
|
|
|
| Acquired IPRD |
12,949 |
|
|
75 |
|
|
|
|
|
| Amortization of acquired intangible assets |
2,357 |
|
|
2,256 |
|
|
|
|
|
| Other (income)/expense, net |
81 |
|
|
(413) |
|
|
|
|
|
| Total Expenses |
23,381 |
|
|
8,567 |
|
|
|
|
|
|
|
|
|
|
|
|
|
| (Loss)/Earnings Before Income Taxes |
(11,516) |
|
|
2,770 |
|
|
|
|
|
| Provision for Income Taxes |
392 |
|
|
503 |
|
|
|
|
|
| Net (Loss)/Earnings |
(11,908) |
|
|
2,267 |
|
|
|
|
|
| Noncontrolling Interest |
3 |
|
|
5 |
|
|
|
|
|
| Net (Loss)/Earnings Attributable to BMS |
$ |
(11,911) |
|
|
$ |
2,262 |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Weighted-Average Common Shares Outstanding: |
|
|
|
|
|
|
|
| Basic |
2,023 |
|
|
2,099 |
|
|
|
|
|
| Diluted |
2,023 |
|
|
2,113 |
|
|
|
|
|
|
|
|
|
|
|
|
|
| (Loss)/Earnings per Common Share: |
|
|
|
|
|
|
|
| Basic |
$ |
(5.89) |
|
|
$ |
1.08 |
|
|
|
|
|
| Diluted |
(5.89) |
|
|
1.07 |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Other (income)/expense, net |
|
|
|
|
|
|
|
Interest expense(b) |
$ |
425 |
|
|
$ |
288 |
|
|
|
|
|
| Royalty and licensing income |
(161) |
|
|
(363) |
|
|
|
|
|
| Royalty income - divestitures |
(271) |
|
|
(188) |
|
|
|
|
|
| Equity investment (gains)/losses |
(102) |
|
|
155 |
|
|
|
|
|
| Integration expenses |
71 |
|
|
67 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Litigation and other settlements |
2 |
|
|
(325) |
|
|
|
|
|
| Investment income |
(183) |
|
|
(102) |
|
|
|
|
|
| Provision for restructuring |
220 |
|
|
67 |
|
|
|
|
|
| Acquisition expense |
49 |
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Other |
31 |
|
|
(12) |
|
|
|
|
|
| Other (income)/expense, net |
$ |
81 |
|
|
$ |
(413) |
|
|
|
|
|
(a) Excludes amortization of acquired intangible assets.
(b) Includes amortization of purchase price adjustments to Celgene debt.
BRISTOL-MYERS SQUIBB COMPANY
PRODUCT REVENUES
FOR THE THREE MONTHS ENDED MARCH 31, 2024 AND 2023
(Unaudited, dollars in millions)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Change vs. 2023 |
| |
|
2024 |
|
2023 |
|
GAAP |
|
Excl. F/X** |
|
|
U.S. |
|
Int'l (c) |
|
WW (d) |
|
U.S. |
|
Int'l (c) |
|
WW (d) |
|
U.S. |
|
Int'l (c) |
|
WW (d) |
|
U.S. |
|
Int'l (c) |
|
WW (d) |
Growth Portfolio |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Opdivo |
|
$ |
1,155 |
|
|
$ |
923 |
|
|
$ |
2,078 |
|
|
$ |
1,281 |
|
|
$ |
921 |
|
|
$ |
2,202 |
|
|
(10) |
% |
|
— |
% |
|
(6) |
% |
|
(10) |
% |
|
9 |
% |
|
(2) |
% |
| Orencia |
|
572 |
|
|
226 |
|
|
798 |
|
|
551 |
|
|
213 |
|
|
764 |
|
|
4 |
% |
|
6 |
% |
|
4 |
% |
|
4 |
% |
|
13 |
% |
|
6 |
% |
| Yervoy |
|
368 |
|
|
215 |
|
|
583 |
|
|
312 |
|
|
196 |
|
|
508 |
|
|
18 |
% |
|
10 |
% |
|
15 |
% |
|
18 |
% |
|
17 |
% |
|
18 |
% |
| Reblozyl |
|
293 |
|
|
61 |
|
|
354 |
|
|
156 |
|
|
50 |
|
|
206 |
|
|
88 |
% |
|
22 |
% |
|
72 |
% |
|
88 |
% |
|
22 |
% |
|
72 |
% |
| Opdualag |
|
198 |
|
|
8 |
|
|
206 |
|
|
116 |
|
|
1 |
|
|
117 |
|
|
71 |
% |
|
* |
|
76 |
% |
|
71 |
% |
|
* |
|
76 |
% |
| Abecma |
|
52 |
|
|
30 |
|
|
82 |
|
|
118 |
|
|
29 |
|
|
147 |
|
|
(56) |
% |
|
3 |
% |
|
(44) |
% |
|
(56) |
% |
|
7 |
% |
|
(44) |
% |
| Zeposia |
|
72 |
|
|
38 |
|
|
110 |
|
|
51 |
|
|
27 |
|
|
78 |
|
|
41 |
% |
|
41 |
% |
|
41 |
% |
|
41 |
% |
|
41 |
% |
|
41 |
% |
| Breyanzi |
|
87 |
|
|
20 |
|
|
107 |
|
|
58 |
|
|
13 |
|
|
71 |
|
|
50 |
% |
|
54 |
% |
|
51 |
% |
|
50 |
% |
|
54 |
% |
|
51 |
% |
| Camzyos |
|
77 |
|
|
7 |
|
|
84 |
|
|
29 |
|
|
— |
|
|
29 |
|
|
* |
|
N/A |
|
* |
|
* |
|
N/A |
|
* |
| Sotyktu |
|
34 |
|
|
10 |
|
|
44 |
|
|
15 |
|
|
1 |
|
|
16 |
|
|
* |
|
* |
|
* |
|
* |
|
* |
|
* |
| Augtyro |
|
6 |
|
|
— |
|
|
6 |
|
|
— |
|
|
— |
|
|
— |
|
|
N/A |
|
N/A |
|
N/A |
|
N/A |
|
N/A |
|
N/A |
| Krazati |
|
21 |
|
|
— |
|
|
21 |
|
|
— |
|
|
— |
|
|
— |
|
|
N/A |
|
N/A |
|
N/A |
|
N/A |
|
N/A |
|
N/A |
Other Growth Products(a) |
|
148 |
|
|
171 |
|
|
319 |
|
|
144 |
|
|
136 |
|
|
280 |
|
|
3 |
% |
|
26 |
% |
|
14 |
% |
|
3 |
% |
|
30 |
% |
|
16 |
% |
Total Growth Portfolio |
|
3,083 |
|
|
1,709 |
|
|
4,792 |
|
|
2,831 |
|
|
1,587 |
|
|
4,418 |
|
|
9 |
% |
|
8 |
% |
|
8 |
% |
|
9 |
% |
|
15 |
% |
|
11 |
% |
Legacy Portfolio |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Eliquis |
|
2,821 |
|
|
899 |
|
|
3,720 |
|
|
2,527 |
|
|
896 |
|
|
3,423 |
|
|
12 |
% |
|
— |
% |
|
9 |
% |
|
12 |
% |
|
1 |
% |
|
9 |
% |
| Revlimid |
|
1,453 |
|
|
216 |
|
|
1,669 |
|
|
1,523 |
|
|
227 |
|
|
1,750 |
|
|
(5) |
% |
|
(5) |
% |
|
(5) |
% |
|
(5) |
% |
|
(1) |
% |
|
(4) |
% |
| Pomalyst/Imnovid |
|
597 |
|
|
268 |
|
|
865 |
|
|
541 |
|
|
291 |
|
|
832 |
|
|
10 |
% |
|
(8) |
% |
|
4 |
% |
|
10 |
% |
|
(7) |
% |
|
4 |
% |
| Sprycel |
|
282 |
|
|
92 |
|
|
374 |
|
|
289 |
|
|
140 |
|
|
429 |
|
|
(2) |
% |
|
(34) |
% |
|
(13) |
% |
|
(2) |
% |
|
(30) |
% |
|
(11) |
% |
| Abraxane |
|
145 |
|
|
72 |
|
|
217 |
|
|
161 |
|
|
78 |
|
|
239 |
|
|
(10) |
% |
|
(8) |
% |
|
(9) |
% |
|
(10) |
% |
|
10 |
% |
|
(3) |
% |
Other Legacy Products(b) |
|
95 |
|
|
133 |
|
|
228 |
|
|
80 |
|
|
166 |
|
|
246 |
|
|
19 |
% |
|
(20) |
% |
|
(7) |
% |
|
19 |
% |
|
(17) |
% |
|
(6) |
% |
Total Legacy Portfolio |
|
5,393 |
|
|
1,680 |
|
|
7,073 |
|
|
5,121 |
|
|
1,798 |
|
|
6,919 |
|
|
5 |
% |
|
(7) |
% |
|
2 |
% |
|
5 |
% |
|
(4) |
% |
|
3 |
% |
| Total Revenues |
|
$ |
8,476 |
|
|
$ |
3,389 |
|
|
$ |
11,865 |
|
|
$ |
7,952 |
|
|
$ |
3,385 |
|
|
$ |
11,337 |
|
|
7 |
% |
|
— |
% |
|
5 |
% |
|
7 |
% |
|
5 |
% |
|
6 |
% |
* In excess of +100%
** See "Use of Non-GAAP Financial Information".
(a) Includes Onureg, Nulojix, Empliciti and royalty revenues.
(b) Includes other mature brands.
(c) Beginning in 2024, Puerto Rico revenues are included in International revenues. Prior period amounts have been reclassified to conform to the current presentation.
(d) Worldwide (WW) includes U.S. and International (Int'l).
BRISTOL-MYERS SQUIBB COMPANY
INTERNATIONAL REVENUES(a)
FOREIGN EXCHANGE IMPACT (%)
FOR THE THREE MONTHS ENDED MARCH 31, 2024
(Unaudited)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, 2024 |
|
|
|
|
|
|
|
|
|
Revenue Change % |
|
F/X % Favorable/ (Unfavorable) ** |
|
Revenue Change % Ex- F/X ** |
|
|
|
|
|
|
|
|
|
|
|
|
Growth Portfolio |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Opdivo |
—% |
|
(9)% |
|
9% |
|
|
|
|
|
|
|
|
|
|
|
|
| Orencia |
6% |
|
(7)% |
|
13% |
|
|
|
|
|
|
|
|
|
|
|
|
| Yervoy |
10% |
|
(7)% |
|
17% |
|
|
|
|
|
|
|
|
|
|
|
|
| Reblozyl |
22% |
|
—% |
|
22% |
|
|
|
|
|
|
|
|
|
|
|
|
| Opdualag |
* |
|
* |
|
* |
|
|
|
|
|
|
|
|
|
|
|
|
| Abecma |
3% |
|
(4)% |
|
7% |
|
|
|
|
|
|
|
|
|
|
|
|
| Zeposia |
41% |
|
—% |
|
41% |
|
|
|
|
|
|
|
|
|
|
|
|
| Breyanzi |
54% |
|
—% |
|
54% |
|
|
|
|
|
|
|
|
|
|
|
|
| Camzyos |
N/A |
|
N/A |
|
N/A |
|
|
|
|
|
|
|
|
|
|
|
|
| Sotyktu |
* |
|
* |
|
* |
|
|
|
|
|
|
|
|
|
|
|
|
| Augtyro |
N/A |
|
N/A |
|
N/A |
|
|
|
|
|
|
|
|
|
|
|
|
| Krazati |
N/A |
|
N/A |
|
N/A |
|
|
|
|
|
|
|
|
|
|
|
|
Other Growth Products(b) |
26% |
|
(4)% |
|
30% |
|
|
|
|
|
|
|
|
|
|
|
|
Total Growth Portfolio |
8% |
|
(7)% |
|
15% |
|
|
|
|
|
|
|
|
|
|
|
|
Legacy Portfolio |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Eliquis |
—% |
|
(1)% |
|
1% |
|
|
|
|
|
|
|
|
|
|
|
|
| Revlimid |
(5)% |
|
(4)% |
|
(1)% |
|
|
|
|
|
|
|
|
|
|
|
|
| Pomalyst/Imnovid |
(8)% |
|
(1)% |
|
(7)% |
|
|
|
|
|
|
|
|
|
|
|
|
| Sprycel |
(34)% |
|
(4)% |
|
(30)% |
|
|
|
|
|
|
|
|
|
|
|
|
| Abraxane |
(8)% |
|
(18)% |
|
10% |
|
|
|
|
|
|
|
|
|
|
|
|
Other Legacy Products(c) |
(20)% |
|
(3)% |
|
(17)% |
|
|
|
|
|
|
|
|
|
|
|
|
Total Legacy Portfolio |
(7)% |
|
(3)% |
|
(4)% |
|
|
|
|
|
|
|
|
|
|
|
|
| Total Revenues |
—% |
|
(5)% |
|
5% |
|
|
|
|
|
|
|
|
|
|
|
|
* In excess of +100%
** See "Use of Non-GAAP Financial Information".
(a) Beginning in 2024, Puerto Rico revenues are included in International revenues. Prior period amounts have been reclassified to conform to the current presentation.
(b) Includes Onureg, Nulojix, Empliciti and royalty revenues.
(c) Includes other mature brands.
BRISTOL-MYERS SQUIBB COMPANY
WORLDWIDE REVENUES(a)
FOREIGN EXCHANGE IMPACT (%)
FOR THE THREE MONTHS ENDED MARCH 31, 2024
(Unaudited)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, 2024 |
|
|
|
|
|
|
|
|
|
Revenue Change % |
|
F/X % Favorable/ (Unfavorable) ** |
|
Revenue Change % Ex- F/X ** |
|
|
|
|
|
|
|
|
|
|
|
|
Growth Portfolio |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Opdivo |
(6)% |
|
(4)% |
|
(2)% |
|
|
|
|
|
|
|
|
|
|
|
|
| Orencia |
4% |
|
(2)% |
|
6% |
|
|
|
|
|
|
|
|
|
|
|
|
| Yervoy |
15% |
|
(3)% |
|
18% |
|
|
|
|
|
|
|
|
|
|
|
|
| Reblozyl |
72% |
|
—% |
|
72% |
|
|
|
|
|
|
|
|
|
|
|
|
| Opdualag |
76% |
|
—% |
|
76% |
|
|
|
|
|
|
|
|
|
|
|
|
| Abecma |
(44)% |
|
—% |
|
(44)% |
|
|
|
|
|
|
|
|
|
|
|
|
| Zeposia |
41% |
|
—% |
|
41% |
|
|
|
|
|
|
|
|
|
|
|
|
| Breyanzi |
51% |
|
—% |
|
51% |
|
|
|
|
|
|
|
|
|
|
|
|
| Camzyos |
* |
|
* |
|
* |
|
|
|
|
|
|
|
|
|
|
|
|
| Sotyktu |
* |
|
* |
|
* |
|
|
|
|
|
|
|
|
|
|
|
|
| Augtyro |
N/A |
|
N/A |
|
N/A |
|
|
|
|
|
|
|
|
|
|
|
|
| Krazati |
N/A |
|
N/A |
|
N/A |
|
|
|
|
|
|
|
|
|
|
|
|
Other Growth Products(b) |
14% |
|
(2)% |
|
16% |
|
|
|
|
|
|
|
|
|
|
|
|
Total Growth Portfolio |
8% |
|
(3)% |
|
11% |
|
|
|
|
|
|
|
|
|
|
|
|
Legacy Portfolio |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Eliquis |
9% |
|
—% |
|
9% |
|
|
|
|
|
|
|
|
|
|
|
|
| Revlimid |
(5)% |
|
(1)% |
|
(4)% |
|
|
|
|
|
|
|
|
|
|
|
|
| Pomalyst/Imnovid |
4% |
|
—% |
|
4% |
|
|
|
|
|
|
|
|
|
|
|
|
| Sprycel |
(13)% |
|
(2)% |
|
(11)% |
|
|
|
|
|
|
|
|
|
|
|
|
| Abraxane |
(9)% |
|
(6)% |
|
(3)% |
|
|
|
|
|
|
|
|
|
|
|
|
Other Legacy Products(c) |
(7)% |
|
(1)% |
|
(6)% |
|
|
|
|
|
|
|
|
|
|
|
|
Total Legacy Portfolio |
2% |
|
(1)% |
|
3% |
|
|
|
|
|
|
|
|
|
|
|
|
| Total Revenues |
5% |
|
(1)% |
|
6% |
|
|
|
|
|
|
|
|
|
|
|
|
* In excess of +100%
** See "Use of Non-GAAP Financial Information".
(a) Worldwide (WW) includes U.S. and International (Int'l).
(b) Includes Onureg, Nulojix, Empliciti and royalty revenues.
(c) Includes other mature brands.
BRISTOL-MYERS SQUIBB COMPANY
RECONCILIATION OF GAAP AND NON-GAAP GROWTH DOLLARS AND PERCENTAGES EXCLUDING FOREIGN EXCHANGE IMPACT *
FOR THE PERIOD ENDED MARCH 31, 2024
(Unaudited, dollars in millions)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2024 |
|
2023 |
|
Change $ |
|
Change % |
|
Favorable / (Unfavorable) F/X $ ** |
|
2024 Excl. F/X ** |
|
Favorable / (Unfavorable) F/X % ** |
|
% Change Excl. F/X ** |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Revenues |
$ |
11,865 |
|
|
$ |
11,337 |
|
|
$ |
528 |
|
|
5 |
% |
|
$ |
(153) |
|
|
$ |
12,018 |
|
|
(1) |
% |
|
6 |
% |
| Gross profit |
8,933 |
|
|
8,771 |
|
|
162 |
|
|
2 |
% |
|
N/A |
|
N/A |
|
N/A |
|
N/A |
Gross profit excluding specified items(a) |
8,955 |
|
|
8,825 |
|
|
130 |
|
|
1 |
% |
|
N/A |
|
N/A |
|
N/A |
|
N/A |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gross margin(b) |
75.3 |
% |
|
77.4 |
% |
|
|
|
|
|
|
|
|
|
|
|
|
| Gross margin excluding specified items |
75.5 |
% |
|
77.8 |
% |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Marketing, selling and administrative |
2,367 |
|
|
1,762 |
|
|
605 |
|
|
34 |
% |
|
21 |
|
|
2,388 |
|
|
2 |
% |
|
36 |
% |
Marketing, selling and administrative excluding specified items(a) |
1,989 |
|
|
1,762 |
|
|
227 |
|
|
13 |
% |
|
21 |
|
|
2,010 |
|
|
1 |
% |
|
14 |
% |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Research and development |
2,695 |
|
|
2,321 |
|
|
374 |
|
|
16 |
% |
|
9 |
|
|
2,704 |
|
|
1 |
% |
|
17 |
% |
Research and development excluding specified items(a) |
2,346 |
|
|
2,206 |
|
|
140 |
|
|
6 |
% |
|
9 |
|
|
2,355 |
|
|
1 |
% |
|
7 |
% |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
* Foreign exchange impacts were derived by converting our current-period local currency financial results using the prior period average currency rates and comparing these adjusted amounts to our current-period results.
** See "Use of Non-GAAP Financial Information".
(a) Refer to the Specified Items schedule below for further details.
(b) Represents gross profit as a percentage of Revenues.
BRISTOL-MYERS SQUIBB COMPANY
SPECIFIED ITEMS
(Unaudited, dollars in millions)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
Three Months Ended March 31, |
|
|
| |
2024 |
|
2023 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Inventory purchase price accounting adjustments |
$ |
8 |
|
|
$ |
53 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Site exit and other costs |
14 |
|
|
1 |
|
|
|
|
|
| Cost of products sold |
22 |
|
|
54 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Acquisition related charges(a) |
372 |
|
|
— |
|
|
|
|
|
| Site exit and other costs |
6 |
|
|
— |
|
|
|
|
|
| Marketing, selling and administrative |
378 |
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
|
|
| IPRD impairments |
— |
|
|
20 |
|
|
|
|
|
| Priority review voucher |
— |
|
|
95 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Acquisition related charges(a) |
348 |
|
|
— |
|
|
|
|
|
| Site exit and other costs |
1 |
|
|
— |
|
|
|
|
|
| Research and development |
349 |
|
|
115 |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Amortization of acquired intangible assets |
2,357 |
|
|
2,256 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest expense(b) |
(13) |
|
|
(14) |
|
|
|
|
|
| Equity investment (gain)/losses |
(102) |
|
|
150 |
|
|
|
|
|
Acquisition expenses |
49 |
|
|
— |
|
|
|
|
|
| Integration expenses |
71 |
|
|
67 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Litigation and other settlements |
— |
|
|
(335) |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Provision for restructuring |
220 |
|
|
67 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Other |
10 |
|
|
(5) |
|
|
|
|
|
| Other (income)/expense, net |
235 |
|
|
(70) |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Increase to pretax income |
3,341 |
|
|
2,355 |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Income taxes on items above |
(340) |
|
|
(293) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Increase to net earnings |
$ |
3,001 |
|
|
$ |
2,062 |
|
|
|
|
|
(a) Includes cash settlement of unvested stock awards, and other related costs incurred in connection with the recent acquisitions.
(b) Includes amortization of purchase price adjustments to Celgene debt.
BRISTOL-MYERS SQUIBB COMPANY
RECONCILIATION OF CERTAIN GAAP LINE ITEMS TO CERTAIN NON-GAAP LINE ITEMS
(Unaudited, dollars and shares in millions except per share data)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, 2024 |
|
|
|
GAAP |
|
Specified Items(a) |
|
Non-GAAP |
|
|
|
|
|
|
| Gross profit |
$ |
8,933 |
|
|
$ |
22 |
|
|
$ |
8,955 |
|
|
|
|
|
|
|
| Marketing, selling and administrative |
2,367 |
|
|
(378) |
|
|
1,989 |
|
|
|
|
|
|
|
| Research and development |
2,695 |
|
|
(349) |
|
|
2,346 |
|
|
|
|
|
|
|
| Amortization of acquired intangible assets |
2,357 |
|
|
(2,357) |
|
|
— |
|
|
|
|
|
|
|
| Other (income)/expense, net |
81 |
|
|
(235) |
|
|
(154) |
|
|
|
|
|
|
|
(Loss)/Earnings before income taxes |
(11,516) |
|
|
3,341 |
|
|
(8,175) |
|
|
|
|
|
|
|
| Provision for income taxes |
392 |
|
|
340 |
|
|
732 |
|
|
|
|
|
|
|
Net (loss)/earnings attributable to BMS used for diluted EPS calculation |
$ |
(11,911) |
|
|
$ |
3,001 |
|
|
$ |
(8,910) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Weighted-average common shares outstanding—diluted |
2,023 |
|
|
2,023 |
|
|
2,023 |
|
|
|
|
|
|
|
| Diluted (loss)/earnings per share |
$ |
(5.89) |
|
|
$ |
1.49 |
|
|
$ |
(4.40) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Effective tax rate |
(3.4) |
% |
|
(5.6) |
% |
|
(9.0) |
% |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended March 31, 2023 |
|
|
|
GAAP |
|
Specified Items(a) |
|
Non-GAAP |
|
|
|
|
|
|
| Gross profit |
$ |
8,771 |
|
|
$ |
54 |
|
|
$ |
8,825 |
|
|
|
|
|
|
|
| Marketing, selling and administrative |
1,762 |
|
|
— |
|
|
1,762 |
|
|
|
|
|
|
|
| Research and development |
2,321 |
|
|
(115) |
|
|
2,206 |
|
|
|
|
|
|
|
| Amortization of acquired intangible assets |
2,256 |
|
|
(2,256) |
|
|
— |
|
|
|
|
|
|
|
| Other (gain)/expense, net |
(413) |
|
|
70 |
|
|
(343) |
|
|
|
|
|
|
|
| Earnings before income taxes |
2,770 |
|
|
2,355 |
|
|
5,125 |
|
|
|
|
|
|
|
| Provision for income taxes |
503 |
|
|
(293) |
|
|
796 |
|
|
|
|
|
|
|
| Net earnings attributable to BMS used for diluted EPS calculation |
$ |
2,262 |
|
|
$ |
2,062 |
|
|
$ |
4,324 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Weighted-average common shares outstanding—diluted |
2,113 |
|
|
2,113 |
|
|
2,113 |
|
|
|
|
|
|
|
| Diluted earnings per share |
$ |
1.07 |
|
|
$ |
0.98 |
|
|
$ |
2.05 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Effective tax rate |
18.2 |
% |
|
(2.7) |
% |
|
15.5 |
% |
|
|
|
|
|
|
(a) Refer to the Specified Items schedule above for further details. Effective tax rate on the Specified Items represents the difference between the GAAP and Non-GAAP effective tax rate.
BRISTOL-MYERS SQUIBB COMPANY
NET DEBT CALCULATION
AS OF MARCH 31, 2024 AND DECEMBER 31, 2023
(Unaudited, dollars in millions)
|
|
|
|
|
|
|
|
|
|
|
|
|
March 31,
2024 |
|
December 31,
2023 |
|
|
|
|
| Cash and cash equivalents |
$ |
9,330 |
|
|
$ |
11,464 |
|
| Marketable debt securities - current |
340 |
|
|
816 |
|
| Marketable debt securities - non-current |
367 |
|
|
364 |
|
| Cash, cash equivalents and marketable debt securities |
$ |
10,037 |
|
|
$ |
12,644 |
|
|
|
|
|
| Short-term debt obligations |
(6,190) |
|
|
(3,119) |
|
| Long-term debt |
(49,487) |
|
|
(36,653) |
|
| Net debt position |
$ |
(45,640) |
|
|
$ |
(27,128) |
|