UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
Date of report (date of earliest event reported): July 10, 2025
TONIX PHARMACEUTICALS HOLDING CORP.
(Exact name of registrant as specified in its charter)
| Nevada | 001-36019 | 26-1434750 |
|
(State or Other Jurisdiction of Incorporation) |
(Commission File Number) |
(IRS Employer Identification No.) |
26 Main Street, Chatham, New Jersey 07928
(Address of principal executive offices) (Zip Code)
Registrant’s telephone number, including area code: (862) 904-8182
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
☐ Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425)
☐ Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12)
☐ Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b))
☐ Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c))
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered |
| Common Stock | TNXP | The NASDAQ Capital Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
| Item 7.01 | Regulation FD Disclosure. |
On July 10, 2025, Tonix Pharmaceuticals Holding Corp. (the “Company”) announced the presentation of new findings on its TNX-801 (recombinant horsepox, live virus vaccine) candidate at the Vaccine Congress 2025. A copy of the press release which discusses this matter is furnished hereto as Exhibit 99.01, and incorporated herein by reference. A copy of the presentation is furnished hereto as Exhibit 99.02, and incorporated herein by reference.
The information in this Item 7.01 of this Current Report on Form 8-K, including Exhibits 99.01 and 99.02 attached hereto, shall not be deemed “filed” for purposes of Section 18 of the United States Securities Exchange Act of 1934 (the “Exchange Act”) or otherwise subject to the liabilities of that section, nor shall they be deemed incorporated by reference in any filing under the United States Securities Act of 1933 or the Exchange Act, except as shall be expressly set forth by specific reference in such a filing.
| Item 8.01. | Other Events. |
On July 10, 2025, the Company announced the presentation of new findings on TNX-801 at the Vaccine Congress 2025. The data demonstrate that TNX-801 can deliver durable immunity while remaining highly attenuated, a balance the Company differentiates TNX-801 from earlier orthopox vaccines. The Company is developing the subcutaneous administration of THNX-201 based on the finding that subcutaneous administration of TNX-801 provides equivalent protection to percutaneous administration. The Company believes this route of administration has the potential benefits of decreased administration-site bacterial superinfection, scarring, and inadvertent transfer to other body sites by itching. The Company believes that TNX-801 has the potential to become a critical tool for containing mpox and preparedness against the deliberate reintroduction of smallpox.
TNX-801 is a recombinant horsepox-derived vaccine candidate designed to prevent orthopox viruses and provide durable humoral and cellular immunity from a single dose. In primary human dermal cells, the virus replicates twenty-seven to one-hundred-nineteen-fold less than licensed vaccinia strains, and in interferon receptor knockout mice it is up to one-hundred-thousand-fold less virulent than those legacy vaccines. Preclinical studies further showed that a single dose of TNX-801 produced strong binding and neutralizing antibody responses across mice, rabbits, hamsters, and cynomolgus macaques, including immunocompromised animals. All vaccinated macaques survived lethal Clade I mpox challenge without lesions, and rabbit models remained fully protected for fourteen months
Forward- Looking Statements
This Current Report on Form 8-K contains certain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934 and Private Securities Litigation Reform Act, as amended, including those relating to the Company’s product development, clinical trials, clinical and regulatory timelines, market opportunity, competitive position, possible or assumed future results of operations, business strategies, potential growth opportunities and other statement that are predictive in nature. These forward-looking statements are based on current expectations, estimates, forecasts and projections about the industry and markets in which we operate and management’s current beliefs and assumptions.
These statements may be identified by the use of forward-looking expressions, including, but not limited to, “expect,” “anticipate,” “intend,” “plan,” “believe,” “estimate,” “potential,” “predict,” “project,” “should,” “would” and similar expressions and the negatives of those terms. These statements relate to future events or our financial performance and involve known and unknown risks, uncertainties, and other factors which may cause actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. Such factors include those set forth in the Company’s filings with the SEC. Prospective investors are cautioned not to place undue reliance on such forward-looking statements, which speak only as of the date of this press release. The Company undertakes no obligation to publicly update any forward-looking statement, whether as a result of new information, future events or otherwise.
| Item 9.01 | Financial Statements and Exhibits. |
| (d) |
Exhibit No. |
Description. | ||
|
99.01 99.02 104 |
Press Release of the Company, dated July 10, 2025 Cover Page Interactive Data File (embedded within the Inline XBRL document) |
SIGNATURE
Pursuant to the requirement of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
| TONIX PHARMACEUTICALS HOLDING CORP. | |||
| Date: July 10, 2025 | By: | /s/ Bradley Saenger | |
| Bradley Saenger | |||
| Chief Financial Officer | |||
TONIX PHARMACEUTICALS HOLDING CORP. 8-K
Exhibit 99.1

Tonix Pharmaceuticals Announces Presentation of New Data on Mpox and Smallpox Vaccine Candidate TNX-801 at the Vaccine Congress 2025
TNX-801 is up to 100,000-fold less virulent than live smallpox vaccine strains and a single dose provides robust immunogenicity and protection against mpox and rabbitpox (more than one year) in animals
Subcutaneous administration of TNX-801 yielded equivalent protection to the traditional percutaneous administration
CHATHAM, N.J., July 10, 2025 (GLOBE NEWSWIRE) – Tonix Pharmaceuticals Holding Corp. (Nasdaq: TNXP), a fully integrated biopharmaceutical company with marketed products and a pipeline of development candidates, today announced the presentation of new findings on TNX-801 (recombinant horsepox, live virus vaccine) at the Vaccine Congress 2025 in Vienna on July 10, 2025, by Sina Bavari, PhD, Executive Vice President, Infectious Disease Research and Development.
“The data show that TNX-801 can deliver durable immunity while remaining highly attenuated, a balance that sets it apart from earlier orthopox vaccines,” said Seth Lederman, MD, Chief Executive Officer of Tonix Pharmaceuticals. “The novel finding that subcutaneous (s.c.) administration of TNX-801 provides equivalent protection to percutaneous administration, is leading us to develop the s.c. product first. Most live-virus vaccines are delivered s.c. and this route of administration has further potential benefits of decreased administration-site bacterial superinfection, scarring, or inadvertent transfer to other body sites by itching. We believe TNX-801 has the potential to become a critical tool for containing mpox and preparedness against the malicious reintroduction of smallpox.We look forward to advancing TNX-801 into the clinic by s.c. delivery.”
TNX-801 is a recombinant horsepox-derived vaccine candidate designed to prevent orthopox viruses and provide durable humoral and cellular immunity from a single dose. In primary human dermal cells, the virus replicates twenty-seven to one-hundred-nineteen-fold less than licensed vaccinia strains, and in interferon receptor knockout mice it is up to one-hundred-thousand-fold less virulent than those legacy vaccines. Preclinical studies further showed that a single dose of TNX-801 produced strong binding and neutralizing antibody responses across mice, rabbits, hamsters, and cynomolgus macaques, including immunocompromised animals. All vaccinated macaques survived lethal Clade I mpox challenge without lesions, and rabbit models remained fully protected for fourteen months.
“Our data show that TNX-801 delivers durable immunity without safety concerns of live virus vaccines,” said Dr Bavari. “TNX-801 dissociates immune protection from some of the side-effects associated with traditional vaccinia-based vaccines and potentially offers a unique risk-benefit profile for mpox and smallpox prevention. We are excited to advance this program toward clinical evaluation in collaboration with public-health partners worldwide.”

The World Health Organization and the Centers for Disease Control and Prevention continue to classify mpox as an ongoing public-health concern. A single-dose vaccine with the attenuation and immunogenicity profile demonstrated by TNX-801 could streamline outbreak response by reducing the need for approved multi-visit immunization schedules.
Tonix Pharmaceuticals Holding Corp.*
Tonix is a fully-integrated biotech company focused on transforming therapies for pain management and vaccines for public health challenges. Tonix’s development portfolio is focused on central nervous system (CNS) disorders. Tonix’s priority is to advance TNX-102 SL, a product candidate for the management of fibromyalgia, for which an NDA was submitted based on two statistically significant Phase 3 studies for the management of fibromyalgia and for which a PDUFA (Prescription Drug User Fee act) goal date of August 15, 2025 has been assigned for a decision on marketing authorization. The FDA has also granted Fast Track designation to TNX-102 SL for the management of fibromyalgia. TNX-102 SL is also being developed to treat acute stress reaction and acute stress disorder under a Physician-Initiated IND at the University of North Carolina in the OASIS study funded by the U.S. Department of Defense (DoD). Tonix’s immunology development portfolio consists of biologics to address organ transplant rejection, autoimmunity and cancer, including TNX-1500, which is an Fc-modified humanized monoclonal antibody targeting CD40-ligand (CD40L or CD154) being developed for the prevention of allograft rejection and for the treatment of autoimmune diseases. Tonix’s infectious disease portfolio includes TNX-801, a vaccine in development for mpox and smallpox, as well as TNX-4200 for which Tonix has a contract with the U.S. DoD’s Defense Threat Reduction Agency (DTRA) for up to $34 million over five years. TNX-4200 is a small molecule broad-spectrum antiviral agent targeting CD45 for the prevention or treatment of infections to improve the medical readiness of military personnel in biological threat environments. Tonix owns and operates a state-of-the art infectious disease research facility in Frederick, Md. Tonix Medicines, our commercial subsidiary, markets Zembrace® SymTouch® (sumatriptan injection) 3 mg and Tosymra® (sumatriptan nasal spray) 10 mg for the treatment of acute migraine with or without aura in adults.
* Tonix’s product development candidates are investigational new drugs or biologics; their efficacy and safety have not been established and have not been approved for any indication.
Zembrace SymTouch and Tosymra are registered trademarks of Tonix Medicines. All other marks are property of their respective owners.
This press release and further information about Tonix can be found at www.tonixpharma.com.

Forward Looking Statements
Certain statements in this press release are forward-looking within the meaning of the Private Securities Litigation Reform Act of 1995. These statements may be identified by the use of forward-looking words such as “anticipate,” “believe,” “forecast,” “estimate,” “expect,” and “intend,” among others. These forward-looking statements are based on Tonix’s current expectations and actual results could differ materially. There are a number of factors that could cause actual events to differ materially from those indicated by such forward-looking statements. These factors include, but are not limited to, risks related to the failure to obtain FDA clearances or approvals and noncompliance with FDA regulations; risks related to the failure to successfully market any of our products; risks related to the timing and progress of clinical development of our product candidates; our need for additional financing; uncertainties of patent protection and litigation; uncertainties of government or third party payor reimbursement; limited research and development efforts and dependence upon third parties; and substantial competition. As with any pharmaceutical under development, there are significant risks in the development, regulatory approval and commercialization of new products. Tonix does not undertake an obligation to update or revise any forward-looking statement. Investors should read the risk factors set forth in the Annual Report on Form 10-K for the year ended December 31, 2024, as filed with the Securities and Exchange Commission (the “SEC”) on March 18, 2025, and periodic reports filed with the SEC on or after the date thereof. All of Tonix’s forward-looking statements are expressly qualified by all such risk factors and other cautionary statements. The information set forth herein speaks only as of the date thereof.
Investor Contact
Jessica Morris
Tonix Pharmaceuticals
investor.relations@tonixpharma.com
(862) 799-8599
Brian Korb
astr partners
(917) 653-5122
brian.korb@astrpartners.com
Media Contact
Ray Jordan Putnam Insights ray@putnaminsights.com Zembrace® SymTouch® (sumatriptan succinate) injection (Zembrace) and Tosymra® (sumatriptan) nasal spray are prescription medicines used to treat acute migraine headaches with or without aura in adults who have been diagnosed with migraine.

Indication and Usage
Zembrace and Tosymra are not used to prevent migraines. It is not known if Zembrace or Tosymra are safe and effective in children under 18 years of age.
Important Safety Information
Zembrace and Tosymra can cause serious side effects, including heart attack and other heart problems, which may lead to death. Stop use and get emergency help if you have any signs of a heart attack:
| ● | discomfort in the center of your chest that lasts for more than a few minutes or goes away and comes back |
| ● | severe tightness, pain, pressure, or heaviness in your chest, throat, neck, or jaw |
| ● | pain or discomfort in your arms, back, neck, jaw or stomach |
| ● | shortness of breath with or without chest discomfort |
| ● | breaking out in a cold sweat |
| ● | nausea or vomiting |
| ● | feeling lightheaded |
Zembrace and Tosymra are not for people with risk factors for heart disease (high blood pressure or cholesterol, smoking, overweight, diabetes, family history of heart disease) unless a heart exam shows no problem.
Do not use Zembrace or Tosymra if you have:
| ● | history of heart problems |
| ● | narrowing of blood vessels to your legs, arms, stomach, or kidney (peripheral vascular disease) |
| ● | uncontrolled high blood pressure |
| ● | hemiplegic or basilar migraines. If you are not sure if you have these, ask your provider. |
| ● | had a stroke, transient ischemic attacks (TIAs), or problems with blood circulation |
| ● | severe liver problems |
| ● | taken any of the following medicines in the last 24 hours: almotriptan, eletriptan, frovatriptan, naratriptan, rizatriptan, ergotamines, or dihydroergotamine. Ask your provider for a list of these medicines if you are not sure. |
| ● | are taking certain antidepressants, known as monoamine oxidase (MAO)-A inhibitors or it has been 2 weeks or less since you stopped taking a MAO-A inhibitor. Ask your provider for a list of these medicines if you are not sure. |
| ● | an allergy to sumatriptan or any of the components of Zembrace or Tosymra |

Tell your provider about all of your medical conditions and medicines you take, including vitamins and supplements.
Zembrace and Tosymra can cause dizziness, weakness, or drowsiness. If so, do not drive a car, use machinery, or do anything where you need to be alert.
Zembrace and Tosymra may cause serious side effects including:
| ● | changes in color or sensation in your fingers and toes |
| ● | sudden or severe stomach pain, stomach pain after meals, weight loss, nausea or vomiting, constipation or diarrhea, bloody diarrhea, fever |
| ● | cramping and pain in your legs or hips; feeling of heaviness or tightness in your leg muscles; burning or aching pain in your feet or toes while resting; numbness, tingling, or weakness in your legs; cold feeling or color changes in one or both legs or feet |
| ● | increased blood pressure including a sudden severe increase even if you have no history of high blood pressure |
| ● | medication overuse headaches from using migraine medicine for 10 or more days each month. If your headaches get worse, call your provider. |
| ● | serotonin syndrome, a rare but serious problem that can happen in people using Zembrace or Tosymra, especially when used with anti-depressant medicines called SSRIs or SNRIs. Call your provider right away if you have: mental changes such as seeing things that are not there (hallucinations), agitation, or coma; fast heartbeat; changes in blood pressure; high body temperature; tight muscles; or trouble walking. |
| ● | hives (itchy bumps); swelling of your tongue, mouth, or throat |
| ● | seizures even in people who have never had seizures before |
The most common side effects of Zembrace and Tosymra include: pain and redness at injection site (Zembrace only); tingling or numbness in your fingers or toes; dizziness; warm, hot, burning feeling to your face (flushing); discomfort or stiffness in your neck; feeling weak, drowsy, or tired; application site (nasal) reactions (Tosymra only) and throat irritation (Tosymra only).
Tell your provider if you have any side effect that bothers you or does not go away. These are not all the possible side effects of Zembrace and Tosymra. For more information, ask your provider.
This is the most important information to know about Zembrace and Tosymra but is not comprehensive. For more information, talk to your provider and read the Patient Information and Instructions for Use. You can also visit https://www.tonixpharma.com or call 1-888-869-7633.
You are encouraged to report adverse effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.
TONIX PHARMACEUTICALS HOLDING CORP. 8-K
Exhibit 99.2

TNX - 801, A SINGLE - DOSE LIVE VACCINE PLATFORM FOR MPOX AND OTHER EMERGING VIRAL DISEASES: SAFETY, IMMUNOGENICITY, AND EFFICACY Sina Bavari, Ph.D. World Congress on Vaccines July 10 th , 2025 © 2025 Tonix Pharmaceuticals Holding Corp.

2 © 2025 Tonix Pharmaceuticals Holding Corp. DISCLAIMER "Certain statements in this presentation regarding strategic plans, expectations and objectives for future operations or results are “forward - looking statements” as defined by the Private Securities Litigation Reform Act of 1995 . These statements may be identified by the use of forward - looking words such as “anticipate,” “believe,” “forecast,” “estimate” and “intend,” among others . These forward - looking statements are based on Tonix’s current expectations and actual results could differ materially . There are a number of factors that could cause actual events to differ materially from those indicated by such forward - looking statements . These factors include, but are not limited to, the risks related to failure to obtain FDA clearances or approvals and noncompliance with FDA regulations ; risks related to the failure to successfully market any of our products ; risks related to the timing and progress of clinical development of our product candidates ; our need for additional financing ; uncertainties of patent protection and litigation ; uncertainties of government or third party payor reimbursement ; limited research and development efforts and dependence upon third parties ; and substantial competition . As with any pharmaceutical under development, there are significant risks in the development, regulatory approval and commercialization of new products . The forward - looking statements in this presentation are made as of the date of this presentation, even if subsequently made available by Tonix on its website or otherwise . Tonix does not undertake an obligation to update or revise any forward - looking statement, except as required by law . Investors should read the risk factors set forth in the Annual Report on Form 10 - K for the year ended December 31 , 2024 , as filed with the Securities and Exchange Commission (the “SEC”) on March 18 , 2025 , and periodic reports and current reports filed with the SEC on or after the date thereof . All of Tonix's forward - looking statements are expressly qualified by all such risk factors and other cautionary statements . " TALK OVERVIEW © 2025 Tonix Pharmaceuticals Holding Corp.

1) Background 2) TNX - 801 attenuation in vitro and in vivo 3) TNX - 801 immunogenicity and efficacy in animal models *TNX - 801 is in the pre - IND stage of development and has not been approved for any indication.

POXVIRUSES » Double stranded DNA, ~128 - 456 kb size » Virions: enveloped, brick - shaped » Size: 220 to 450 nm long î 140 to 260 nm wide î 140 to 260 nm thick » Infect vertebrate or invertebrate hosts » Genus Orthopoxvirus : ▪ Human Pathogens: • VARV: Case fatality rate 30 to 50% • MPXV: Case fatality rate 0.1 to 11% ▪ Vaccines: • Vaccinia, Cowpox, Horsepox • Horsepox virus: TNX - 801 © 2025 Tonix Pharmaceuticals Holding Corp.
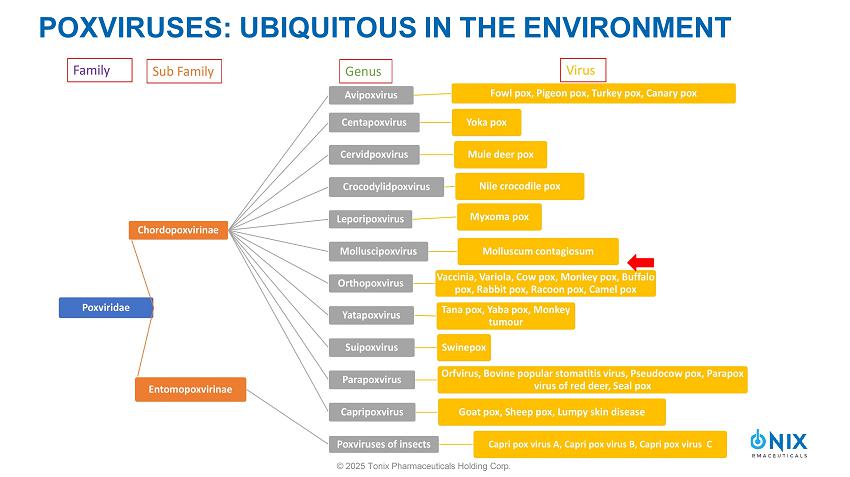
POXVIRUSES: UBIQUITOUS IN THE ENVIRONMENT © 2025 Tonix Pharmaceuticals Holding Corp.

MONKEYPOX VIRUS (MPOX) © 2025 Tonix Pharmaceuticals Holding Corp. » Endemic in Central and West Africa » Two Clades: 1) Clade I (DRC) 2) Clade IIa (West Africa) and IIb (Nigeria) » Human Case Fatality Rate: ▪ Clade I – 11% ▪ Clade IIa – 3% ▪ Clade IIb – <0.1% » Clade IIb – 2022 Outbreak ▪ 122 Countries ▪ 100,000 Confirmed Cases VARIOLA VIRUS (SMALLPOX) © 2025 Tonix Pharmaceuticals Holding Corp.


» Oldest written record – 3,500 years » Oldest sequences – 1,400 years » Human Case Fatality Rate: 30% » 20 th century – 250 to 500 million deaths » Eradication: 1980 EDWARD JENNER - SMALLPOX VACCINE (1796) » Jenner observed milkmaids were protected from smallpox, reasoned that infection with an illness similar to smallpox but less deadly could protect one against smallpox ▪ “Cowpox” was the name of a disease in cows that could transfer to humans and cause sores ▪ Jenner “vaccinated” (from vacca , Latin for “cow”) a patient with pustule matter from “cowpox” sores on a milkmaid’s hands; that patient remained healthy when challenged with smallpox virus » Jenner suspected that the agent causing cowpox, which he called vaccinia originated in horses and had been transferred from horses to cows’ udders by dirty hands The College of Physicians of Philadelphia. Accessed July 15, 2021. https:/ /w w w.historyofvaccines.org © 2025 Tonix Pharmaceuticals Holding Corp.

SMALLPOX VACCINES © 2025 Tonix Pharmaceuticals Holding Corp. » Vaccine: Cowpox origin » Serial passaging: Humans, cows, and horses (143 years) » Vaccine: Vaccinia Virus (1939) closely related to cowpox but serologically distinct 1 » Multiple Vaccinia virus - based vaccines developed » Smallpox eradication 1 Downie AW. 1939. Br J Exp Pathol 20:158 – 176.
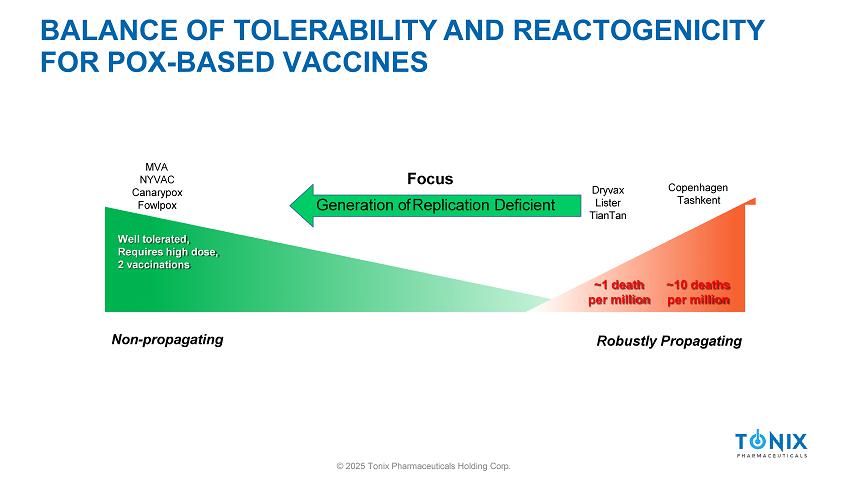
BALANCE OF TOLERABILITY AND REACTOGENICITY FOR POX - BASED VACCINES Well tolerated, Requires high dose, 2 vaccinations ~1 death per million ~10 deaths per million Non - propagating © 2025 Tonix Pharmaceuticals Holding Corp. Robustly Propagating Dryvax Lister TianTan Copenhagen Tashkent MVA NYVAC Canarypox Fowlpox Focus Generation of Replication Deficient BALANCE OF TOLERABILITY AND REACTOGENICITY FOR POX - BASED VACCINES Well tolerated, Requires high dose, 2 vaccinations ~1 death per million ~10 deaths per million Non - propagating © 2025 Tonix Pharmaceuticals Holding Corp.
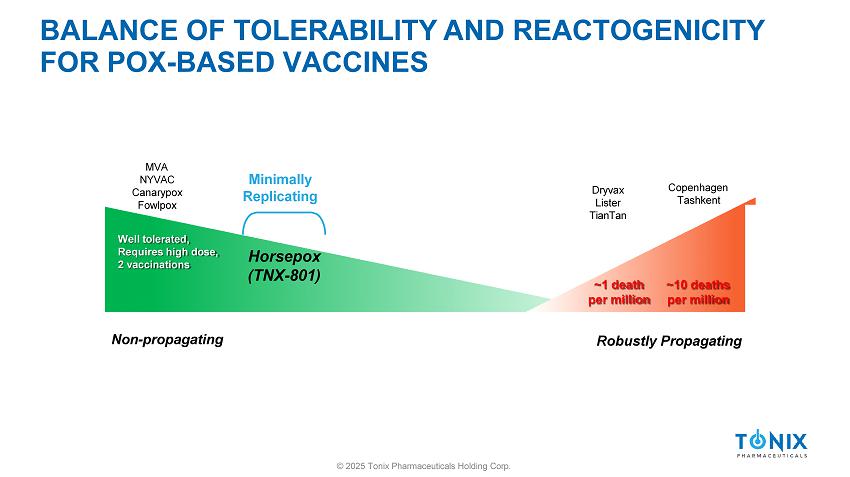
Robustly Propagating Dryvax Lister TianTan Copenhagen Tashkent MVA NYVAC Canarypox Fowlpox Minimally Replicating Horsepox (TNX - 801)

4 PRONG APPROACH TO MPOX/SMALLPOX VACCINE (TNX - 801) © 2025 Tonix Pharmaceuticals Holding Corp. 1) Well - tolerated 2) Single dose 3) Durable 4) Protection against mpox disease (lesions)
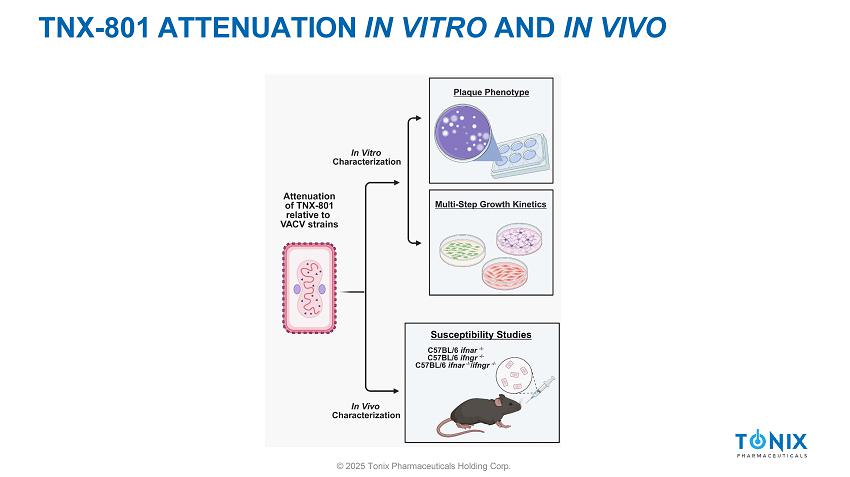
TNX - 801 ATTENUATION IN VITRO AND IN VIVO © 2025 Tonix Pharmaceuticals Holding Corp.

IN VITRO ATTENUATION OF TNX - 801 © 2025 Tonix Pharmaceuticals Holding Corp. » Investigate attenuation of TNX - 801 in vitro relative to VACV strains ▪ Positive Control: VACV - Western Reserve (WR) , VACV - International Health Department (IHD) ▪ Older vaccines used in smallpox eradication: 1) VACV - Lister (Lis) 2) VACV - New York City Board of Health (NYCBH) ▪ New Vaccine: TNX - 801 ▪ Non - replicating control: MVA » In vitro Assays: 1) Plaque phenotype – BSC - 40 and Vero - E6 2) Replication Kinetics ▪ Immortalized non - human primate cell lines ▪ Human primary cells from two main route of poxvirus transmission • Dermal and respiratory tracts TNX - 801 DISPLAYS SMALL PLAQUE PHENOTYPE VACCINA VIRUSES VACV - Western Reserve (WR) VACV - International Health Department (IHD) VACV - Lister (Lis) VACV - New York City Board of Health (NYCBH) TNX - 801 MVA © 2025 Tonix Pharmaceuticals Holding Corp.

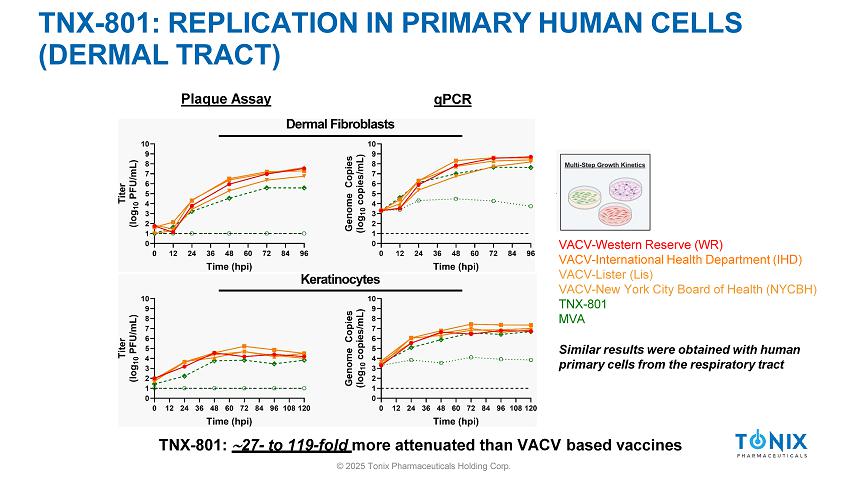
TNX - 801: REPLICATION IN PRIMARY HUMAN CELLS (DERMAL TRACT) Plaque Assay qPCR TNX - 801: 27 - to 119 - fold more attenuated than VACV based vaccines VACV - Western Reserve (WR) VACV - International Health Department (IHD) VACV - Lister (Lis) VACV - New York City Board of Health (NYCBH) TNX - 801 MVA Similar results were obtained with human primary cells from the respiratory tract © 2025 Tonix Pharmaceuticals Holding Corp.

IN VIVO ATTENUATION OF TNX - 801 » Investigate attenuation of TNX - 801 in vivo relative to VACV based vaccines ▪ Immunocompromised Mice (C57BL/6 ifnar - / - , C57BL/6 ifngr - / - , C57BL/6 ifnar - / - /ifngr - / - ) • Interferon receptor knockout model • Sensitive to virus infection ▪ Positive Control: VACV - WR , VACV - IHD ▪ Older vaccines: VACV - Lis, VACV - NYCBH ▪ TNX - 801 ▪ Route: Intranasal » Parameters measured: 1) Disease Score 2) Temperature 3) Weight loss 4) Survival © 2025 Tonix Pharmaceuticals Holding Corp.
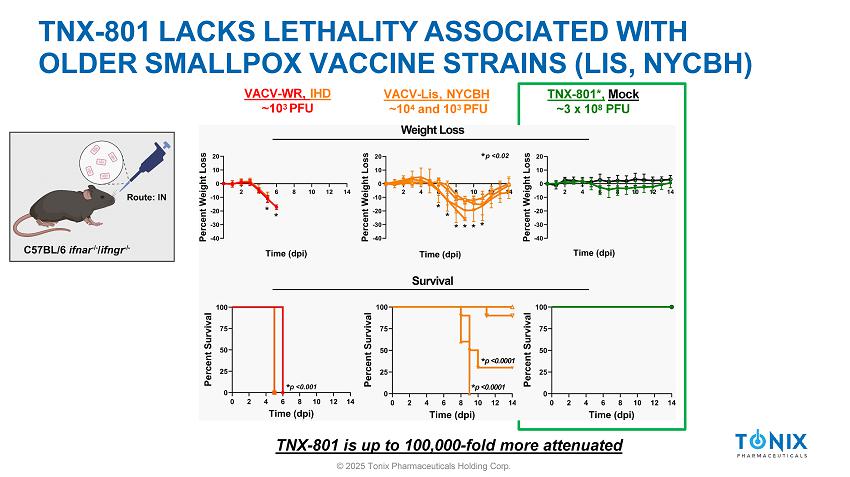
TNX - 801 LACKS LETHALITY ASSOCIATED WITH OLDER SMALLPOX VACCINE STRAINS (LIS, NYCBH) TNX - 801*, Mock ~3 x 10 8 PFU VACV - Lis, NYCBH ~10 4 and 10 3 PFU VACV - WR, IHD ~10 3 PFU TNX - 801 is up to 100,000 - fold more attenuated © 2025 Tonix Pharmaceuticals Holding Corp.

TNX - 801 INFECTION DISPLAYS LIMITED REPLICATION VACV - IHD ~10 6 PFU ( ) VACV - NYCBH ~10 6 PFU ( ) TNX - 801 ~10 8 PFU ( ) © 2025 Tonix Pharmaceuticals Holding Corp.
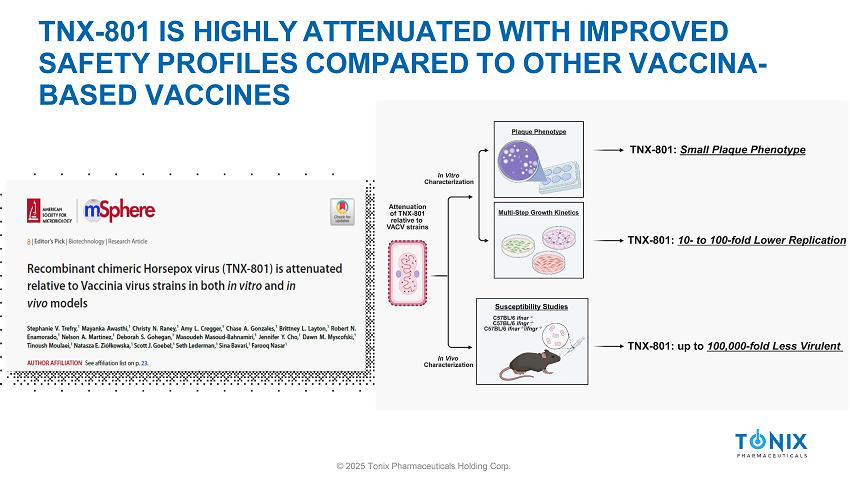
TNX - 801 IS HIGHLY ATTENUATED WITH IMPROVED SAFETY PROFILES COMPARED TO OTHER VACCINA - BASED VACCINES © 2025 Tonix Pharmaceuticals Holding Corp.

TNX - 801 IMMUNOGENICITY AND EFFICACY IN ANIMAL MODELS (SINGLE DOSE) 1) 2) 3) © 2025 Tonix Pharmaceuticals Holding Corp.

NHP IMMUNOGENICITY AND EFFICACY STUDY DESIGN rVACV = Plaque pick from ACAM2000 (Approved Vaccine) © 2025 Tonix Pharmaceuticals Holding Corp.
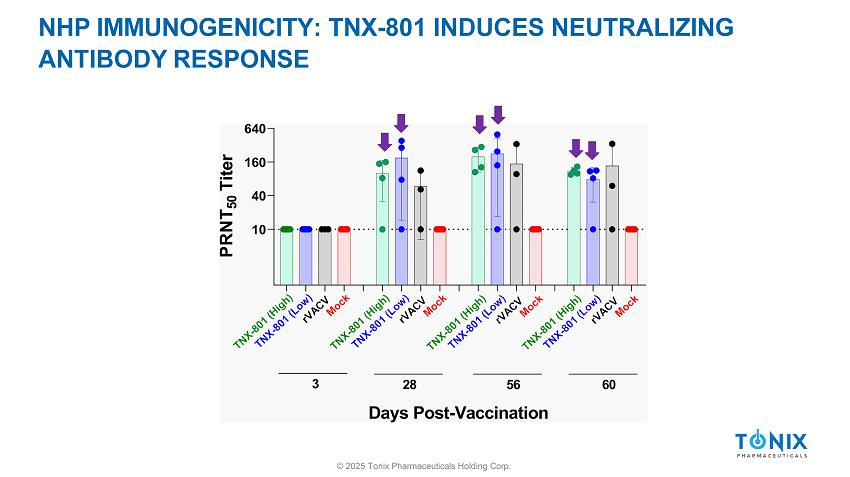
NHP IMMUNOGENICITY: TNX - 801 INDUCES NEUTRALIZING ANTIBODY RESPONSE © 2025 Tonix Pharmaceuticals Holding Corp.

TNX - 801 PROVIDES PROTECTION AGAINST MPOX DISEASE NO LESIONS in TNX - 801 vaccinated groups © 2025 Tonix Pharmaceuticals Holding Corp.

TNX - 801 PROVIDES PROTECTION AGAINST LETHAL MONKEYPOX CLADE I CHALLENGE NO LETHALITY in TNX - 801 vaccinated groups © 2025 Tonix Pharmaceuticals Holding Corp.
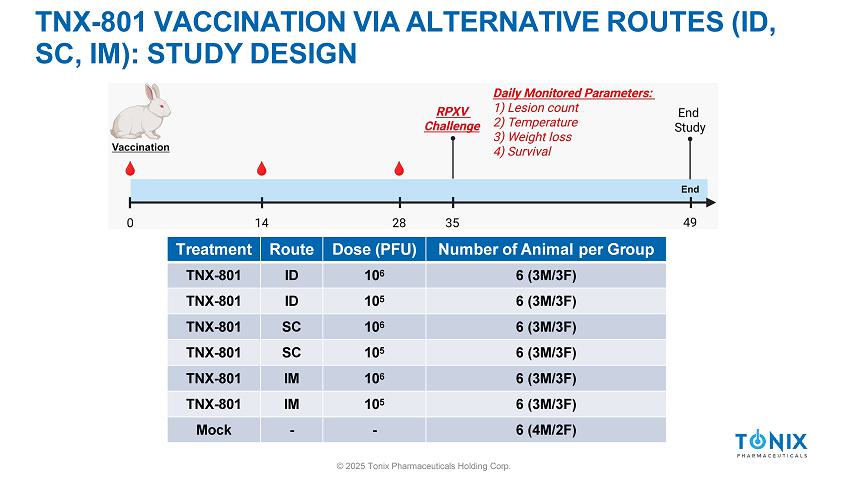
TNX - 801 VACCINATION VIA ALTERNATIVE ROUTES (ID, SC, IM): STUDY DESIGN Number of Animal per Group Dose (PFU) Route Treatment 6 (3M/3F) 10 6 ID TNX - 801 6 (3M/3F) 10 5 ID TNX - 801 6 (3M/3F) 10 6 SC TNX - 801 6 (3M/3F) 10 5 SC TNX - 801 6 (3M/3F) 10 6 IM TNX - 801 6 (3M/3F) 10 5 IM TNX - 801 6 (4M/2F) - - Mock © 2025 Tonix Pharmaceuticals Holding Corp.

IMMUNOGENICITY: DAY 28 ANTI - VACV (MVA) TITERS © 2025 Tonix Pharmaceuticals Holding Corp. LOD Comparable IgG titers regardless of Route or Dose TNX - 801 PROTECTS AGAINST LETHAL RPXV CHALLENGE NO LETHALITY in TNX - 801 vaccinated groups © 2025 Tonix Pharmaceuticals Holding Corp.

TNX - 801 PROTECTS AGAINST CLINICAL DISEASE (LESIONS) NO LESIONS in TNX - 801 vaccinated groups © 2025 Tonix Pharmaceuticals Holding Corp.

TNX - 801 PROTECTS AGAINST CLINICAL DISEASE (LESIONS) NO LESIONS in TNX - 801 vaccinated groups © 2025 Tonix Pharmaceuticals Holding Corp.


TNX - 801 PROVIDES PROTECTION AGAINST LETHAL MONKEYPOX CLADE IIA CHALLENGE: ALTERNATIVE ROUTES © 2025 Tonix Pharmaceuticals Holding Corp.

TNX - 801 PROVIDES PROTECTION AGAINST LETHAL MONKEYPOX CLADE IIA CHALLENGE: ALTERNATIVE ROUTES TNX - 801 (PERCUT) TNX - 801 (SC) TNX - 801 (IM) Mock Weight Loss Survival *p <0.0001 *p <0.003 * * * * © 2025 Tonix Pharmaceuticals Holding Corp.

TNX - 801 PROVIDES DURABLE PROTECTION AGAINST LETHAL RABBITPOX CHALLENGE: 6 MONTHS © 2025 Tonix Pharmaceuticals Holding Corp.
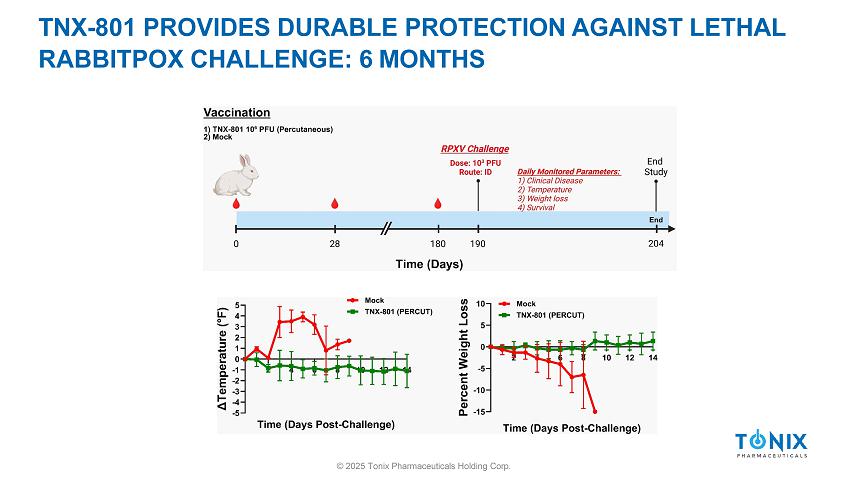
TNX - 801 PROVIDES DURABLE PROTECTION AGAINST LETHAL RABBITPOX CHALLENGE: 6 MONTHS © 2025 Tonix Pharmaceuticals Holding Corp.

TNX - 801 PROVIDES DURABLE PROTECTION AGAINST LETHAL RABBITPOX CHALLENGE: 6 MONTHS © 2025 Tonix Pharmaceuticals Holding Corp.
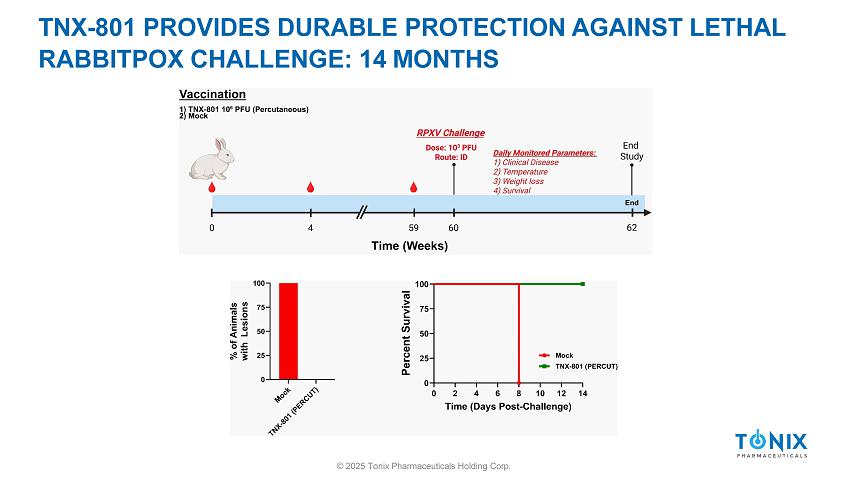
TNX - 801 PROVIDES DURABLE PROTECTION AGAINST LETHAL RABBITPOX CHALLENGE: 14 MONTHS © 2025 Tonix Pharmaceuticals Holding Corp.

TNX - 801 SAFETY © 2025 Tonix Pharmaceuticals Holding Corp. » In vitro : ▪ Small plaque phenotype ▪ Up to 100 - fold lower replication than VACV strains ▪ Primary cells from dermal and respiratory tracts » In vivo : ▪ Well tolerated in mice, rabbits, hamsters, and NHPs ▪ Minimal or no disease in immunocompromised murine models ▪ up to 100,000 - fold more attenuated than VACV - based vaccines ▪ Minimally replicates at site of delivery TNX - 801 IMMUNOGENICITY AND EFFICACY (SINGLE DOSE) © 2025 Tonix Pharmaceuticals Holding Corp.

» Evaluated in multiple animal models ▪ Mouse, Rabbit, and NHP (Cynomolgus) » Elicits IgG and/or neutralizing responses ▪ Various route percutaneous, subcutaneous, intramuscular ▪ Microneedle delivery » Provides 100% protection against lesions ▪ Rabbit and NHP models ▪ Rabbit model: 6 - and 14 - months » Provides 100% protection against lethal challenge ▪ Models: Mouse, Rabbit, and NHP ▪ Viruses: VACV, RPXV, MPXV clade Ia and IIa ACKNOWLEDGEMENTS © 2025 Tonix Pharmaceuticals Holding Corp.

» University of Alberta ▪ Ryan Noyce ▪ David Evans » Southern Research » TNX - 801 Team ▪ Scott Goebel ▪ Tinoush Moulaei ▪ Natasza Ziółkowska ▪ Siobhan Fogarty ▪ Helen Stillwell ▪ Bruce Daugherty ▪ Farooq Nasar ▪ Seth Lederman » TNX - 801 Team ▪ Mayanka Awasthi ▪ Christy Raney ▪ Amy Cregger ▪ Robert Enamorado ▪ Stephanie Trefry ▪ Nelson Martinez ▪ Deborah Gohegan ▪ Zeil Rosenberg