
Developing Next Generation Programmed T Cell Therapies January 2026 Autolus.com For Investor communication only. Not for use in product promotion. EX-99.2

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line Disclaimer These slides contain forward-looking statements within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995. Forward-looking statements are statements that are not historical facts, and in some cases can be identified by terms such as “may,” “will,” “could,” “expects,” “plans,” “anticipates,” and “believes.” These statements include, but are not limited to: statements regarding Autolus’ development and commercialization of its product candidates; Autolus' manufacturing, sales and marketing plans for AUCATZYL, including expectations regarding the commercial launch in the United States and the ability to reach patients in a timely manner; the amount and timing of milestone payments under Autolus' collaboration and license agreements; and future development plans of obe-cel, including the timing or likelihood of expansion into additional markets or geographies and related regulatory approvals. Any forward-looking statements are based on management’s current views and assumptions and involve risks and uncertainties that could cause actual results, performance, or events to differ materially from those expressed or implied in such statements. Actual results and the timing of events could differ materially from those anticipated in such forward-looking statements as a result of various risks and uncertainties, which include, without limitation: Autolus' ability to maintain regulatory approval of AUCATZYL; its ability to execute its commercialization strategy for AUCATZYL; its ability to develop, manufacture and commercialize its other product candidates and the timing or likelihood of expansion of AUCATZYL into additional markets or geographies; Autolus' ability to establish and expand a commercial infrastructure and to successfully launch, market and sell AUCATZYL; actions of regulatory agencies, which may affect the initiation, timing and progress of clinical trials or future regulatory approval; the labelling for AUCATZYL/obe-cel in any future indication or patient population, if approved; the potential for payors to delay, limit or deny coverage for AUCATZYL; Autolus' ability to obtain, maintain and enforce intellectual property protection for AUCATZYL or any product candidates it is developing; the results of clinical trials are not always being predictive of future results; the cost, timing and results of clinical trials; that many product candidates do not become approved drugs on a timely or cost effective basis or at all; the ability to enroll patients in clinical trials; and possible safety and efficacy concerns. For a discussion of other risks and uncertainties, and other important factors, any of which could cause Autolus’ actual results to differ from those contained in the forward-looking statements, see the section titled “Risk Factors” in Autolus' Annual Report on Form 10-K filed with the Securities and Exchange Commission, or the SEC, on March 20, 2025, as well as discussions of potential risks, uncertainties, and other important factors in Autolus’ subsequent filings with the Securities and Exchange Commission. All information in this presentation is as of the date of the presentation, and Autolus undertakes no obligation to publicly update any forward-looking statement, whether as a result of new information, future events, or otherwise, except as required by law. You should, therefore, not rely on these forward-looking statements as representing the Company’s views as of any date subsequent to the date of this presentation. 2 Developing and Delivering a New Generation of T Cell Therapies

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line Autolus is positioned for value creation Obe-cel product franchise supports multiple growth opportunities 3 *Preliminary and unaudited Developing and Delivering a New Generation of T Cell Therapies Initial Indication: Adult r/r B-ALL Strong Execution in First Year of Launch Achieved CAR T market leadership in r/r B-ALL Significant opportunity to grow overall CAR T market in adult r/r B-ALL Physician interest to pursue investigator sponsored trials in 1st line ALL FY 2025 Net Product Revenue Pipeline expansion opportunities grow future commercial potential in new indications Commercial and pipeline opportunities supported by proven manufacturing and product delivery capabilities and established authorized treatment centers Opportunity to establish a pipeline in a product with recent data presentations supporting potentially pivotal Phase 2 clinical trials with obe-cel in lupus nephritis and pediatric ALL ~$75 million*

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line Autolus is a leader in CAR T manufacturing & product delivery Developing and Delivering a New Generation of T Cell Therapies 4 ✓ Manufacturing success rate >90% ✓ Fast, reliable and consistent product delivery ✓ No capacity limitations Executing on manufacturing and product delivery in the first year of launch: Manufacturing Life Cycle Strategy: Opportunities for Innovation to Improve Margins Optimizing the current manufacturing process and operating model Enhancing automation opportunities on our existing process Developing next-generation manufacturing platform with a step change in the cost and capacity profile 1 2 3

AUCATZYL® AUTOLUS’ FIRST APPROVED PRODUCT A potentially best-in-class, standalone CD19 CAR T cell therapy

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line AUCATZYL® now approved in US, UK and EU • Highly active, fast off-rate CD19 CAR T therapy with a well managed safety profile1 – approved in US, UK, EU • First and currently only approved CAR T therapy with customized, tumor-burden guided dosing – no FDA REMS obligation • Established infrastructure for manufacturing and commercialization • Commercial presence in more than 60 US centers • NICE determined AUCATZYL to be cost effective, UK* launch initiated in December 2025 • Approval in EU† in 2025; pricing and reimbursement evaluation ongoing on a country-by-country basis Developing and Delivering a New Generation of T Cell Therapies 6 AUCATZYL indicated for the treatment of adults with relapsed or refractory B-cell precursor acute lymphoblastic leukemia (B-ALL) 1Roddie C, et al "Obecabtagene autoleucel in B-cell acute lymphoblastic leukemia" N Engl J Med 2024; DOI: 10.1056/NEJMoa2406526; *Conditional marketing authorization, †European Commission (EC) conditional approval

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line 7 Strong first year of U.S. AUCATZYL® launch 2026 ExpectationsAUCATZYL Net Product Revenue Q4 2025: ~$24 Million* FY 2025: ~$75 Million* Developing and Delivering a New Generation of T Cell Therapies Strong initial launch based on: • More than 60 treatment centers activated • Positive physician and patient experience • Reliable manufacture and delivery of product FY 2026 Net Product Revenue: $120-$135 million Shift to positive gross margin in 2026 based on increasing volumes and improved manufacturing plant utilization *Preliminary unaudited financials; Autolus will report Q4 & FY 2025 financial results in March 2026 Increase commercial footprint in the US to more than 80 treatment centers and ongoing launch in the UK

AUCATZYL geographic growth opportunities in ALL 8 ✓ Conditional marketing authorization in the UK received April 2025 ✓ Successful NICE pricing and reimbursement process Nov 2025 ✓ AUCATZYL available in routine commissioning in NHS Dec 2025 ✓ European Commission (EC) conditional approval received July 2025 • Ongoing country-by-country evaluation of pricing and reimbursement decisions to assess feasibility of market entry; no anticipated EU sales in 2026 • Exploration of alternate market access mechanisms in 2026 Expansion EU market access - pending Developing and Delivering a New Generation of T Cell Therapies UK Launch Q1 2026

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line Potential for improved potency, reduced toxicity We believe AUCATZYL® has a unique mechanism of action 9 Clinical data show increased activity and reduced toxicity References:1. AUCATZYL. Prescribing Information. Autolus, Inc. 2024. 2. Ghorashian S, Kramer AM, Onuoha S, et al. Enhanced CAR T cell expansion and prolonged persistence in pediatric patients with ALL treated with a low-affinity CD19 CAR. Nat Med. 2019;25:1408- 1414. 3. Data on file. Autolus, Inc. 2024. 4. Zhao X, Yang J, Zhang X, et al. Efficacy and safety of CD28- or 4-1BB-based CD19 CAR-T cells in B cell acute lymphoblastic leukemia. Mol Ther Oncolytics. 2020;18:272-281. Fast off-rate Avoided over-activation of CAR T cells → Reduced toxicities Increased CAR T peak expansion → Improved peak activity and persistence Avoided exhaustion of CAR T-cells → Improved engraftment Improved persistence CAT19 fast off-rate binder² CD8-derived hinge region/ transmembrane domain2 4-1BB co-stimulatory domain1,4 Shown to enhance CAR T-cell expansion and reduce exhaustion compared with CD28 CARs in preclinical studies CD3Ɀ activation domain¹ HD37 FMC63 B4 4G7 CAT Off Rate: Kd [S-1] O n -R at e : K a [M -1 S- 1 ] Other CD19 Binders Obe-cel Binder 10-4 10-3 10-2 10-1 107 106 105 104 Shorter half-life of interaction compared to binders used in other approved products • AUCATZYL® = 9.8 seconds • Kymriah® = 21 minutes Developing and Delivering a New Generation of T Cell Therapies

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line AUCATZYL was approved based on results from the FELIX trial 10 FELIX Phase 1b/2 Cohort IA ≥5% BM blast 1,2 Roddie C, et al "Obecabtagene autoleucel in B-cell acute lymphoblastic leukemia" N Engl J Med 2024; DOI: 10.1056/NEJMoa2406526 Cohort IB <5% BM blast MRD+ Cohort IIA ≥5% BM blast Cohort IIB <5% BM blast MRD+ Cohort IIC Isolated EMD at screening Patients (N) Ph1b/2 pooled1 Enrolled 153 Infused 127 Background • Open-label, multinational, single-arm Phase 1b/2 trial in adult patients with R/R B-ALL1-2; largest CAR T cell therapy trial in R/R B-ALL to date (N=153 enrolled) • Conducted during COVID-19 pandemic with highly immune compromised patients Summary of Trial Experience • High ORR, encouraging EFS/OS and favorable tolerability with low levels of high- grade CRS and ICANS • Timely and reliable clinical product supply and logistics despite COVID-19 pandemic restrictions • Across all Phase 1b/2 cohorts, 40% of responders in ongoing remission without subsequent stem cell transplant/other therapy1 • Survival outcomes suggesting potential of long-term plateau1 Developing and Delivering a New Generation of T Cell Therapies

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line FELIX trial published in New England Journal of Medicine1 High overall response rate with deep molecular responses • Durable responses, particularly in patients with a low-to- intermediate bone marrow burden 11 Favourable response rate and tolerability, despite challenging patient population Excellent tolerability profile • Very low rates of high-grade immunotoxicities • No high-grade events in low disease burden patients 1. Roddie C, et al "Obecabtagene autoleucel in B-cell acute lymphoblastic leukemia" N Engl J Med 2024; DOI: 10.1056/NEJMoa2406526 Response by disease status at lymphodepletion Overall Remission Rate (CR/CRi) All patients (n=127) 77% Morphological disease (n=91) 75% Measurable residual disease (n=29) 96% Isolated extramedullary disease (n=7) 71% Safety by disease burden at lymphodepletion Grade ≥3 CRS Grade ≥3 ICANS All patients (n=127) 2% 7% >75% Blasts (n=40) 2% 12% 5-75% Blasts (n=51) 4% 8% <5% Blasts (n=36) 0% 0% Developing and Delivering a New Generation of T Cell Therapies

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line Data continue to show long term remissions in r/r adult B-ALL At 24 months, overall survival probability was 46.0% Jae H Park et al, EHA 2025, oral presentation #S113 12 Developing and Delivering a New Generation of T Cell Therapies

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line Duration of response: median 42.6 months at last data cut More than half of patients still in remission at 24 months Jae H Park et al, EHA 2025, oral presentation #S113 13 Developing and Delivering a New Generation of T Cell Therapies

Expanding the obe-cel opportunity Deep value program with potentially broad applicability

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line Pipeline supports growth with multiple development opportunities 15 Product Indication Target Preclinical Phase 1 Phase 2/Pivotal Approved Status AUCATZYL® (obe-cel) Adult ALL CD19 FDA, MHRA^ & EC approved† obe-cel Pediatric ALL CD19 Currently enrolling obe-cel Lupus Nephritis CD19 Currently enrolling obe-cel Progressive Multiple Sclerosis CD19 Currently enrolling ^Conditional marketing authorization; †European Commission (EC) conditional approval; *UCL Collaboration Oncology Autoimmune / B-cell mediated Product Indication Target Preclinical Phase 1 Status AUTO8* Multiple Myeloma CD19 & BCMA Currently enrolling AUTO8* Light Chain Amyloidosis CD19 & BCMA Currently enrolling AUTO1/22* Pediatric ALL CD19 & CD22 Currently enrolling Developing and Delivering a New Generation of T Cell Therapies Near-Term Growth Drivers Early Stage / UCL Collaborations

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line Growing the obe-cel franchise commercial opportunity Robust clinical database and demonstrated commercial capabilities position Autolus for efficient path in new indications Developing and Delivering a New Generation of T Cell Therapies 16 Pediatric r/r B-ALL Phase 2 Adult r/r B-ALL Approved Progressive MS Phase 1 Refractory Lupus Nephritis Phase 2 ~1,6001 Estimated U.S. addressable patient population for obe-cel indications approved or in development ~4001 Graphic is illustrative, not to scale AutoimmuneHem Oncology ~25,000 - 35,0002 ~300,0003 Significant Market Opportunity 1. Based on National Cancer Institue SEER estimates and internal analysis; 2. Clarivate/DRG Epidemiology, Arthritis Rheumatol. 2023 Apr;75(4):567-573, Arthritis & Rheumatology. 2017;69(10):2006-2017; 3. GlobalData MS Market Forecast 2020-2030 April 2023;

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line Development Rationale: High Unmet Medical Need Pediatric r/r B-ALL development strategy 17 Regenerative Medicine Advanced Therapy (RMAT) designation supports development pathway Developing and Delivering a New Generation of T Cell Therapies • Kymriah approved for refractory and 2nd relapse patients - excluding first line high-risk relapsed patients • Obe-cel aims to include all patients post-frontline therapy, including first line high-risk relapsed patients Complete CATULUS Phase 1 CATULUS Phase 2 CATULUS trial is currently enrolling; enrollment expected to be complete by first half of 2027 • Data reported at ASH 2025 annual meeting • ORR of 95%, CRR of 91% • Low rates of high-grade CRS and ICANS • 30 patients, SAT • International study (US, UK, SP) • Collaboration with Children's Oncology Group (COG) • Ages 0 - 18 and a minimum weight of 6 kg • Single weight-based infusion of 1 million cells per kg
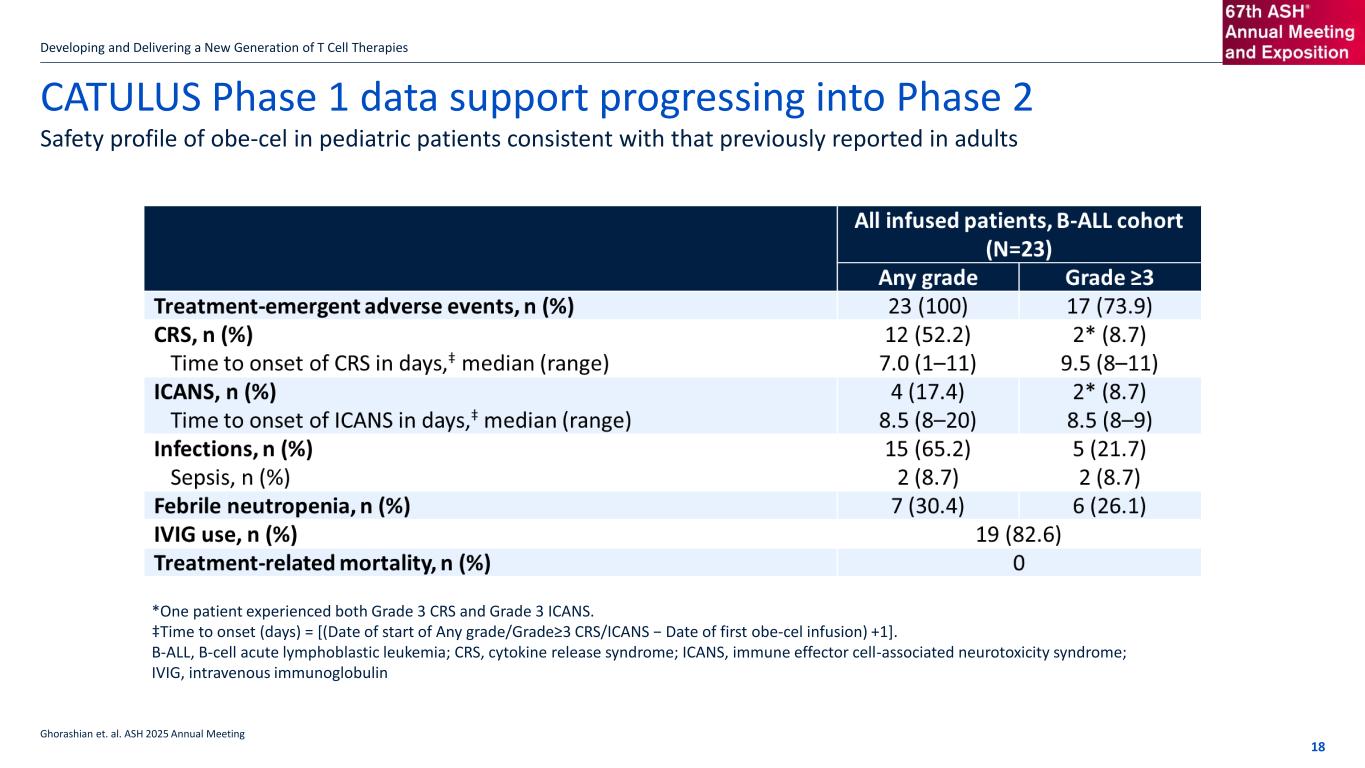
If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line CATULUS Phase 1 data support progressing into Phase 2 Developing and Delivering a New Generation of T Cell Therapies 18 Safety profile of obe-cel in pediatric patients consistent with that previously reported in adults Ghorashian et. al. ASH 2025 Annual Meeting *One patient experienced both Grade 3 CRS and Grade 3 ICANS. ‡Time to onset (days) = [(Date of start of Any grade/Grade≥3 CRS/ICANS − Date of first obe-cel infusion) +1]. B-ALL, B-cell acute lymphoblastic leukemia; CRS, cytokine release syndrome; ICANS, immune effector cell-associated neurotoxicity syndrome; IVIG, intravenous immunoglobulin

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line CATULUS data demonstrate promising initial efficacy in pediatric patients Developing and Delivering a New Generation of T Cell Therapies 19 At median follow-up of 8.8 months in pediatric r/r B-ALL patients: ORR was 95.5%; CR was achieved in 90.9% Ghorashian et. al. ASH 2025 Annual Meeting Swimmer plot showing disease assessments in the B-ALL cohort

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line MOA and commercial capabilities are key differentiators in AID 20 Autolus Potential Advantage Favorable tolerability to drive acceptability in non-oncology indications Deep cut into the CD19+ B cells and plasma blasts Robust, economical and scalable manufacturing and established commercial infrastructure Potential for accelerated clinical program Supports differentiated approach and potential for obe-cel in autoimmune disease areas FDA-approved CAR-T therapy, with existing safety database, now in development for autoimmune indications Obe-cel is the only CD19 CAR approved in other indications that is now being tested for autoimmune disease Developing and Delivering a New Generation of T Cell Therapies

CARSLYLE: Obe-cel shows promise as a new approach for SLE/LN Patient population: • Patients were significantly impaired with their kidney function and had across the board some of the highest SLEDAI-2K disease scores included in current SLE studies. Efficacy: Median follow up of 11.4 months in 50 million cell dose cohort • Achievement of DORIS in 83.3% (n=5/6) of patients • Achievement of renal complete remission in 50% (n=3/6 pts) of patients Safety: Obe-cel was generally well tolerated in all patients with no ICANS, no high-grade CRS PK/Biomarkers: All patients showed deep B-cell depletion shortly after infusion, which was subsequently followed by a predominance of naïve B-cell reconstitution, suggesting an obe-cel-driven immune reset Next Steps: • Completion of adolescent (aged 12–17 years) and higher dose level patient cohorts • Data support progression into a Phase 2 lupus nephritis trial Developing and Delivering a New Generation of T Cell Therapies 50 million cell dose selected as recommended Phase 2 dose Leandro et al., American College of Rheumatology Conference 2025 Abstract #2458 21
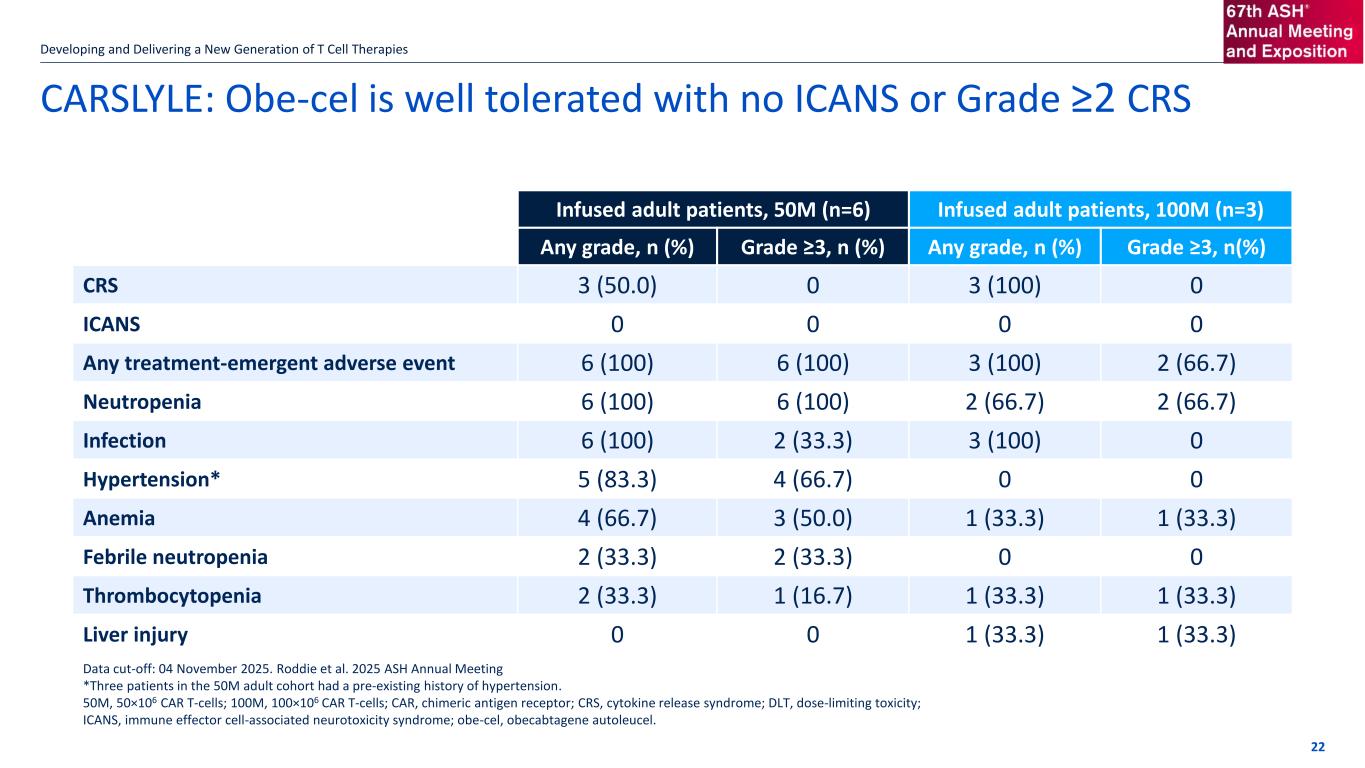
If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line CARSLYLE: Obe-cel is well tolerated with no ICANS or Grade ≥2 CRS Developing and Delivering a New Generation of T Cell Therapies 22 Infused adult patients, 50M (n=6) Infused adult patients, 100M (n=3) Any grade, n (%) Grade ≥3, n (%) Any grade, n (%) Grade ≥3, n(%) CRS 3 (50.0) 0 3 (100) 0 ICANS 0 0 0 0 Any treatment-emergent adverse event 6 (100) 6 (100) 3 (100) 2 (66.7) Neutropenia 6 (100) 6 (100) 2 (66.7) 2 (66.7) Infection 6 (100) 2 (33.3) 3 (100) 0 Hypertension* 5 (83.3) 4 (66.7) 0 0 Anemia 4 (66.7) 3 (50.0) 1 (33.3) 1 (33.3) Febrile neutropenia 2 (33.3) 2 (33.3) 0 0 Thrombocytopenia 2 (33.3) 1 (16.7) 1 (33.3) 1 (33.3) Liver injury 0 0 1 (33.3) 1 (33.3) Data cut-off: 04 November 2025. Roddie et al. 2025 ASH Annual Meeting *Three patients in the 50M adult cohort had a pre-existing history of hypertension. 50M, 50×106 CAR T-cells; 100M, 100×106 CAR T-cells; CAR, chimeric antigen receptor; CRS, cytokine release syndrome; DLT, dose-limiting toxicity; ICANS, immune effector cell-associated neurotoxicity syndrome; obe-cel, obecabtagene autoleucel.

5 of 6 patients achieved DORIS with median onset of 5.1 months Developing and Delivering a New Generation of T Cell Therapies 23 Data cut-off: 04 November 2025. DORIS is defined as: SLEDAI = 0 (irrespective of serology), PGA <0.5, and ≤5 mg/day corticosteroid use. Use of stable antimalarials and immunosuppressives, including biologics, is allowed. 50M, 50×106 CAR T-cells; 100M, 100×106 CAR T-cells; CAR, chimeric antigen receptor; CRR, complete renal response; DORIS, Definition of Remission in systemic lupus erythematosus; PGA, Physician Global Assessment; PRR, partial renal response; SLEDAI-2K, Systemic Lupus Erythematosus Disease Activity Index 2000. • Renal responses reported at last follow-up visit in the 50M adult cohort indicate that three patients (50.0%) achieved CRR with onset at Month 1 and one patient (16.7%) achieved PRR with onset at Month 7 • The length of follow up was insufficient to calculate DORIS response or CRR/PRR for the 100M adult cohort Swimmer plot showing DORIS over time in the 50M adult cohort (n=6) Time since obe-cel infusion (month) Patient 1 Patient 2 Patient 3 Patient 4 Patient 5 Patient 6 5 0 M a d u lt s (n =6 ) DORIS non-responder DORIS responder 1 2 3 4 5 6 7 8 9 10 11 12 23
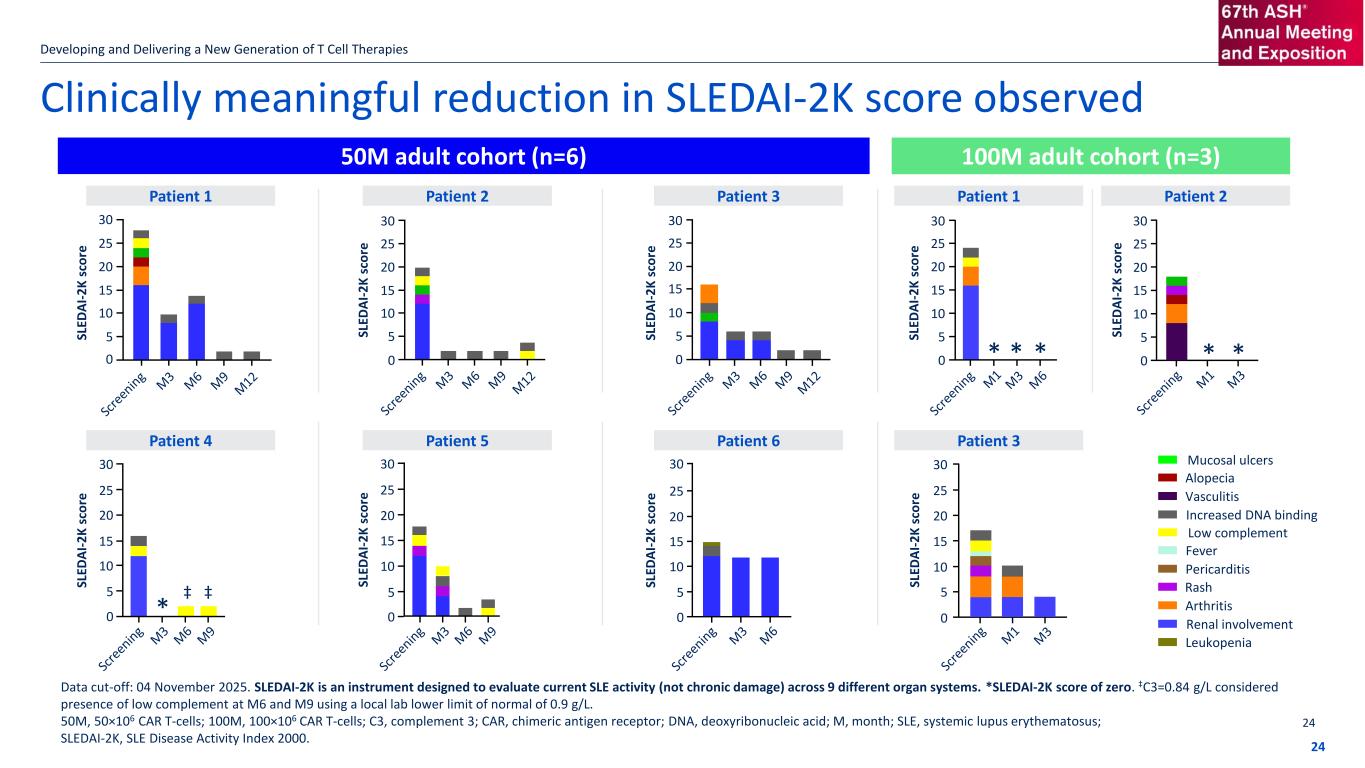
Clinically meaningful reduction in SLEDAI-2K score observed Developing and Delivering a New Generation of T Cell Therapies 24 50M adult cohort (n=6) 100M adult cohort (n=3) Data cut-off: 04 November 2025. SLEDAI-2K is an instrument designed to evaluate current SLE activity (not chronic damage) across 9 different organ systems. *SLEDAI-2K score of zero. ‡C3=0.84 g/L considered presence of low complement at M6 and M9 using a local lab lower limit of normal of 0.9 g/L. 50M, 50×106 CAR T-cells; 100M, 100×106 CAR T-cells; C3, complement 3; CAR, chimeric antigen receptor; DNA, deoxyribonucleic acid; M, month; SLE, systemic lupus erythematosus; SLEDAI-2K, SLE Disease Activity Index 2000. SL ED A I- 2 K s co re Patient 1 Patient 2 Patient 3 Patient 4 Patient 5 Patient 6 SL ED A I- 2 K s co re SL ED A I- 2 K s co re SL ED A I- 2 K s co re SL ED A I- 2 K s co re SL ED A I- 2 K s co re Patient 1 Patient 2 SL ED A I- 2 K s co re Patient 3 * ‡ 30 25 20 15 10 5 0 30 25 20 15 10 5 30 25 20 15 10 5 30 25 20 15 10 5 30 25 20 15 10 5 30 25 20 15 10 5 30 25 20 15 10 5 0 0 0 SL ED A I- 2 K s co re 30 25 20 15 10 5 0 SL ED A I- 2 K s co re 30 25 20 15 10 5 0000 Mucosal ulcers Alopecia Vasculitis Increased DNA binding Low complement Fever Pericarditis Rash Arthritis Renal involvement Leukopenia * * * * * ‡ 24
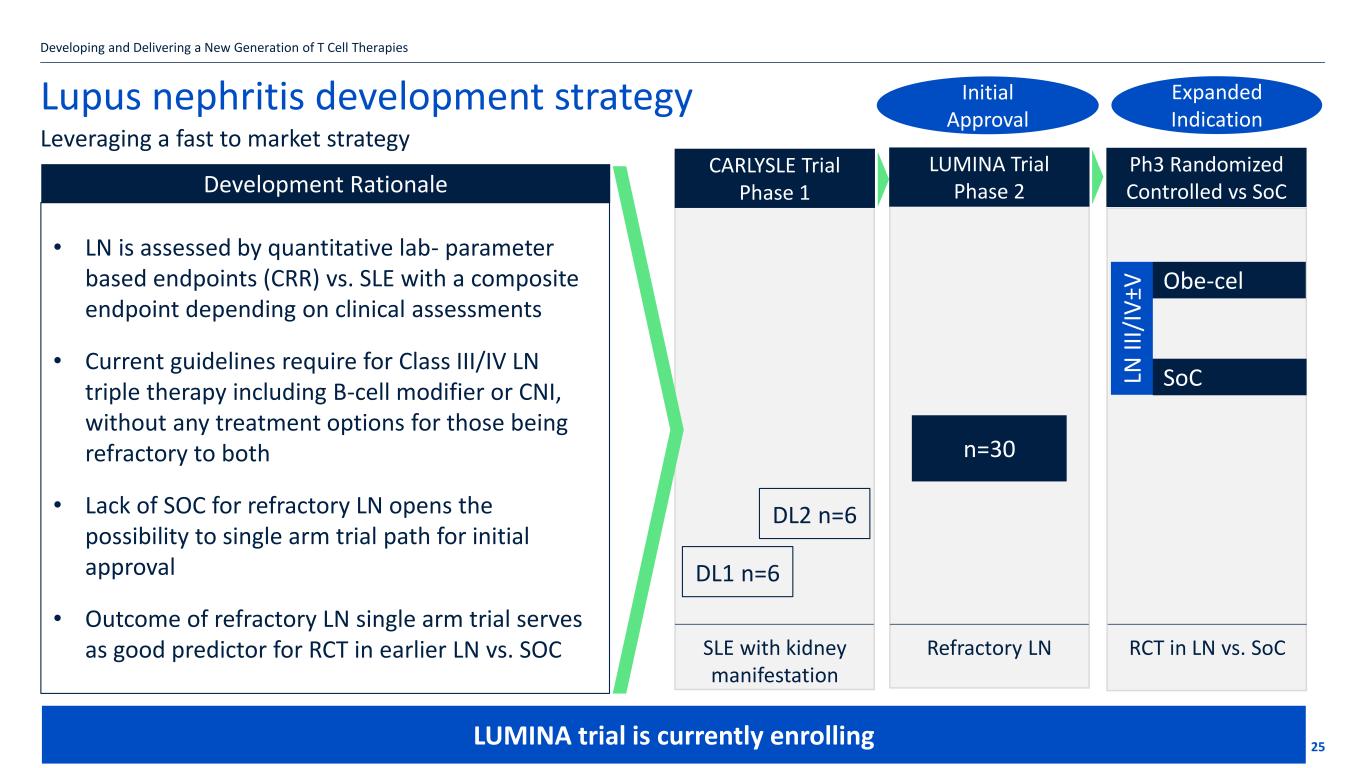
If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line Development Rationale Lupus nephritis development strategy 25 Leveraging a fast to market strategy Developing and Delivering a New Generation of T Cell Therapies • LN is assessed by quantitative lab- parameter based endpoints (CRR) vs. SLE with a composite endpoint depending on clinical assessments • Current guidelines require for Class III/IV LN triple therapy including B-cell modifier or CNI, without any treatment options for those being refractory to both • Lack of SOC for refractory LN opens the possibility to single arm trial path for initial approval • Outcome of refractory LN single arm trial serves as good predictor for RCT in earlier LN vs. SOC DL1 n=6 DL2 n=6 SLE with kidney manifestation Refractory LN n=30 RCT in LN vs. SoC SoC Obe-cel LN II I/ IV ±V CARLYSLE Trial Phase 1 LUMINA Trial Phase 2 Ph3 Randomized Controlled vs SoC Initial Approval Expanded Indication LUMINA trial is currently enrolling

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line Phase 2 LUMINA trial supports efficient path to market 26 Evaluating severe, refractory lupus nephritis (LN) Trial design Single arm, open-label, multi-centre, phase 2 Sample size 30 patients Patient population • 12-65 years of age, body weight ≥ 40kg • Diagnosis of SLE based on (EULAR)/ (ACR) 2019 classification • Positive (ANA) (≥ 1:80), or anti-dsDNA (≥ 30 IU/mL) or anti-Smith (> ULN), anti-histone or anti-chromatin (> ULN) • Severe, refractory LN (ongoing active class III, IV or V (only in combination with III or IV) • Prior immunosuppressive and biologic therapies with inadequate response or intolerance Treatment 50 x 106 CAR positive T-cells following Flu/Cy lymphodepletion Endpoints Primary: Complete Renal Response at 6 months Key Secondary: DORIS at 6 months Timing • LUMINA trial is currently enrolling Developing and Delivering a New Generation of T Cell Therapies Day-5 Day 1 Monthly safety and efficacy follow-upL Leukapheresis Obe-cel Manufacturing Lymphodepletion Obe-cel Infusion Screening Cy 1000 mg/m2 Flu 25 mg/m2

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line • High disease activity is associated with inflammatory processes • Uncontrolled inflammation leads to high chronicity due to accumulated kidney damage • Despite treatment advances including regulatory approvals of belimumab and voclosporin the goal to sufficiently improve short and long-term outcomes in patients with LN remains unmet • There are no treatment options for refractory patients • Kidneys are one of the most common organs involved in SLE - 30% – 40% are lupus nephritis patients Refractory lupus nephritis is a high unmet medical need Developing and Delivering a New Generation of T Cell Therapies 27 Clarivate/DRG Epidemiology, Arthritis Rheumatol. 2023 Apr;75(4):567-573, Arthritis & Rheumatology. 2017;69(10):2006-2017 US Treated SLE ∼350,000 Treated LN (Biopsy Confirmed) ∼110,000 Treated LN Class III-V 70,000 - 90,000 Refractory to SOC 25,000 - 35,000

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line Multiple sclerosis development strategy 28 Establish Phase 1 Clinical Proof of Concept in MS 3 x 6 dose escalation design - a higher dose may be required for CNS effect Biomarker readouts to provide nearer term evidence of biological effect at 6 months + Definitive clinical outcomes based on clinical disability progression at 12 months + Initiate Phase 2/3 study in progressive MS patients exhibiting PIRA • Anticipate a randomised phase 2/3 study design as path to approval • Phase 1 clinical PoC is derisking for initiation of development in other neurology indications Developing and Delivering a New Generation of T Cell Therapies First patient dosed in October 2025

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line BOBCAT study population represents the highest unmet need in MS Developing and Delivering a New Generation of T Cell Therapies 29 Current SOC delays an estimated ~ 6 months but does not halt or reverse disability progression Transformational – halting disease progression Aspirational – improving disability and moving back on EDSS timeline1. Progressive forms of MS including all forms of progressive MS with EDSS scores of 3.5 to 6.5 included 2. Will include both active and non-active patients 3. Have failed high efficacy therapy for at least six months (e.g. CD20 mAb, S1P inhibitor) 1. Graphic based on Kurtzke JF Neurology 1983 Nov;33(11):1444-52.

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line BOBCAT Phase 1 study design Sample size: 12-18 patients infused with obe-cel Dosing: Standard Flu/Cy based preconditioning and a single infusion of 100 or 200 million CART cells and flexibility to adjust dose up or down Primary endpoint: •Safety Secondary endpoints: •12-week confirmed disability progression (CDP) at one-year, composite measure of disability at one year, confirmed disability improvement •Other functional measures: cognition, fatigue, QoL •Imaging: MRI lesion counts (T1 Gd+, T2), MRI – MTR, MRI – regional brain volumes, MRI – cervical spinal cord volume, SEL, PRL) •Biomarkers (blood and CSF): OCBs, IgG index, NfL, GFAP, Kappa chains, PK Interim analysis at 6 months: •Biomarkers including OCBs, IgG index, NfL, MRI lesions, MTR, Kappa chains, PK SEL: slowly expanding lesions, PRL: paramagnetic rim lesions, NfL: Neurofilament light chain, GFAP: glial fibrillary acidic protein Developing Next Generation Programmed T Cell Therapies 30

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line Progressive multiple sclerosis is a high unmet medical need • MS impacts approximately 1,000,000 individuals in the US1 and there is currently no known cure 1: GlobalData MS Market Forecast 2020-2030 April 2023 2:Watson, C., Thirumalai, D., Barlev, A. et al. Treatment Patterns and Unmet Need for Patients with Progressive Multiple Sclerosis in the United States: Survey Results from 2016 to 2021. Neurol Ther 12, 1961–1979 (2023). https://doi.org/10.1007/s40120-023-00532-2 Developing and Delivering a New Generation of T Cell Therapies • Around 30% of patients have progressive disease and more than half of Progressive MS patients experience disability progression despite receiving disease modifying agents2 • Highest unmet need for patients who continue to progress despite being treated with highly effective agents for at least 6 months Relapsing Remitting MS ∼ 700,000 Primary Progressive MS ∼100,000 Secondary Progressive MS ∼ 200,000 Obe-cel focus 31

Upcoming milestones Anticipated Milestone or Catalyst Anticipated Timing Updated real world experience data from ROCCA consortium H1 2026 Longer-term follow up data from CARLYSLE trial ACR Annual Meeting 2026 Initial clinical data from BOBCAT Phase 1 trial in progressive MS By YE 2026 Initial clinical data from ALARIC Phase 1 trial in AL amyloidosis (UCL collaboration) By YE 2026 CATULUS trial in pediatric r/r B-ALL fully enrolled H1 2027 Obe-cel Oncology Obe-cel Autoimmune/B cell Mediated Disease 32 Developing and Delivering a New Generation of T Cell Therapies

If the title goes over more than one line move the subtitle text box down by 1.15cm per additional line 33 Developing and Delivering a New Generation of T Cell Therapies 2026 Focus: Drive market share in ALL – Improve margins – Expand beyond ALL Building value with obe-cel Strong execution in r/r B-ALL: ✓ Market leadership ✓ Broad market access / coverage ✓ Reliable product delivery ✓ Significant opportunity to grow CAR T market in adult B-ALL ✓ Physician interest in ISTs in 1L ALL Leveraging investments: • Optimize manufacturing operating model and technology • Business process efficiencies targeting margin improvement Potential “pipeline in a product" new indications: • Pediatric ALL – Potential pivotal study • Lupus nephritis – Potential pivotal study • Multiple sclerosis – Phase 1 study ExpandLaunch Optimize

Autolus.com Thank you Investor Contact: A.Cray@autolus.com Executive Director, Investor Relations & External Communications





































