FALSE000161985600016198562025-11-032025-11-03
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
________________________________________
FORM 8-K
________________________________________
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): November 3, 2025
________________________________________
Caribou Biosciences, Inc.
(Exact name of Registrant as Specified in Its Charter)
________________________________________
|
|
|
|
|
|
|
|
|
| Delaware |
001-40631 |
45-3728228 |
(State or Other Jurisdiction
of Incorporation) |
(Commission File Number) |
(IRS Employer
Identification No.) |
|
|
|
|
2929 7th Street, Suite 105
Berkeley, California
|
|
94710 |
| (Address of Principal Executive Offices) |
|
(Zip Code) |
Registrant’s Telephone Number, Including Area Code: (510) 982-6030
N/A
(Former Name or Former Address, if Changed Since Last Report)
________________________________________
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
|
|
|
|
|
|
| ☐ |
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
|
|
| ☐ |
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
|
|
| ☐ |
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
|
|
| ☐ |
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Title of each class |
|
Trading
Symbol(s)
|
|
Name of each exchange on which registered |
| Common Stock, $0.0001 par value per share |
|
CRBU |
|
NASDAQ Global Select Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company ☒
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 2.02 Results of Operations and Financial Condition
On November 3, 2025, Caribou Biosciences, Inc. (the “Company”) announced certain preliminary financial results for the quarter ended September 30, 2025. On a preliminary unaudited basis, the Company expects its cash, cash equivalents, and marketable securities as of September 30, 2025 to be approximately $159.2 million. This estimate of cash, cash equivalents, and marketable securities is its preliminary estimate based on currently available information and does not present all necessary information for an understanding of the Company’s financial condition as of September 30, 2025, or its results of operations for the three and nine months ended September 30, 2025. As the Company completes its quarter-end financial close process and finalizes its financial statements for the three and nine months ended September 30, 2025, the Company may be required to make significant adjustments that may result in the estimate provided herein being different than the final cash, cash equivalents, and marketable securities as of September 30, 2025 to be reported in its Form 10-Q.
Item 7.01 Regulation FD Disclosure.
On November 3, 2025, the Company issued a press release announcing results from its ongoing ANTLER phase 1 trial evaluating vispacabtagene regedleucel (“vispa-cel;” formerly CB-010), an allogeneic anti-CD19 chimeric antigen receptor ("CAR")-T (“CAR-T”) cell therapy, in patients with relapsed or refractory B cell non-Hodgkin lymphoma (“r/r B-NHL”). The Company also announced the planned design of its randomized, controlled pivotal phase 3 trial for vispa-cel in second-line (“2L”) large B cell lymphoma (“LBCL”) CD19-naïve patients who are ineligible for transplant and autologous CAR-T cell therapy. A copy of the press release is attached hereto as Exhibit 99.1 and incorporated by reference herein.
Also, on November 3, 2025, the Company issued a press release announcing data from the dose escalation portion of its ongoing CaMMouflage phase 1 trial evaluating CB-011, an allogeneic anti-B cell maturation antigen (“anti-BCMA”) CAR-T cell therapy, in patients with relapsed or refractory multiple myeloma (“r/r MM”). A copy of the press release is attached hereto as Exhibit 99.2 and incorporated by reference herein.
The Company will host a live webcast today, Monday, November 3, 2025, at 8:00 a.m. Eastern Time, to discuss the clinical developments for both vispa-cel and CB-011. A copy of the slide presentation to be used during the Company’s webcast is attached hereto as Exhibit 99.3 and incorporated by reference herein. Details for accessing the webcast are included in both Exhibit 99.1 and Exhibit 99.2.
The information in Item 7.01 of this Current Report on Form 8-K (including Exhibit 99.1, Exhibit 99.2, and Exhibit 99.3) shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be, or be deemed, incorporated by reference in any filings under the Securities Act of 1933, as amended (the “Securities Act”), regardless of any general incorporation language in any such filing or document, unless the Company specifically states that the information is to be considered “filed” under the Exchange Act or incorporates it by reference into a filing under the Securities Act or the Exchange Act.
Item 8.01 Other Matters.
Data Releases
The Company is a clinical-stage Clustered Regularly Interspaced Short Palindromic Repeats (“CRISPR”) genome-editing biopharmaceutical company dedicated to developing transformative therapies for patients with devastating diseases. The Company’s genome-editing platform, including its novel chRDNA (CRISPR hybrid RNA-DNA, pronounced “chardonnay”) technology, enables more precise genome editing of allogeneic cell therapies. The Company’s allogeneic, or off-the-shelf, CAR-T cell therapy product candidates are manufactured in advance with cells from healthy donors, with the goal of enabling broad patient access, rapid patient treatment, and increased manufacturing scale. The Company’s allogeneic CAR-T cell therapy product candidates in clinical development are directed at established cell surface targets against which autologous CAR-T cell therapeutics have already demonstrated clinical proof of concept, CD19 and B cell maturation antigen (“BCMA”). The Company uses its chRDNA technologies to armor its allogeneic CAR-T cell therapies through multiple genome-editing strategies, such as checkpoint disruption and immune cloaking, to enhance activity against hematologic malignancies.
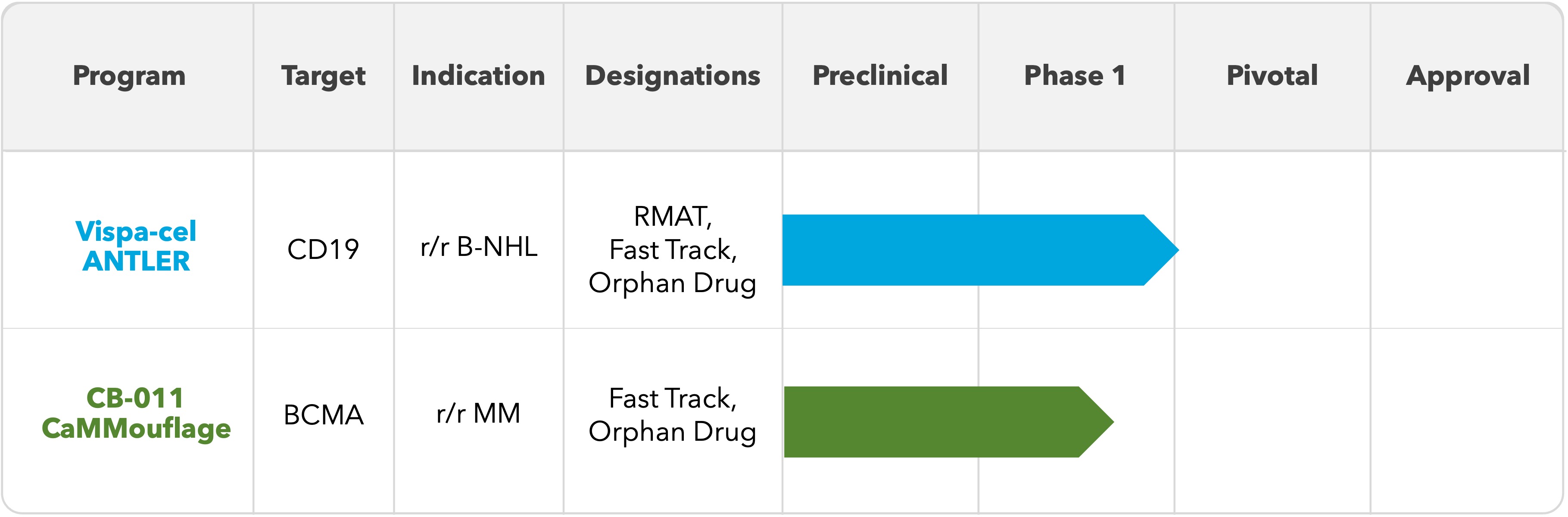
The Company is advancing two clinical-stage allogeneic CAR-T cell therapy product candidates for the treatment of patients with hematologic malignancies:
•Vispacabtagene regedleucel (“vispa-cel”; formerly CB-010): an allogeneic anti-CD19 CAR-T cell therapy that is being evaluated in patients with relapsed or refractory B cell non-Hodgkin lymphoma (“r/r B-NHL”) in the Company’s ongoing ANTLER phase 1 clinical trial. The Company believes vispa-cel will have several advantages over commercially available autologous CAR-T cell therapies, including broad patient access, shorter time to treatment with an off-the-shelf product, significantly lower manufacturing costs, and a substantially smaller footprint for manufacturing.
•CB-011: an allogeneic anti-BCMA CAR-T cell therapy that is being evaluated in patients with r/r MM in the Company’s ongoing CaMMouflage phase 1 clinical trial. The Company believes CB-011 will have several advantages over both commercially available bispecific antibody therapies and autologous CAR-T cell therapies, including a single-dose treatment regimen, potentially lower rates of grade 3 or greater infections, and rapid immune recovery compared to bispecifics, as well as broader patient access, shorter time to treatment with an off-the-shelf product, significantly lower manufacturing costs, and a substantially smaller footprint for manufacturing compared to autologous CAR-T cell therapies.
Vispa-cel has received regenerative medicine advanced therapy (“RMAT”) designation for relapsed or refractory large B cell lymphoma (“r/r LBCL”) as well as fast track designation for r/r B-NHL from the U.S. Food and Drug Administration (“FDA”); CB-011 has received fast track designation for r/r MM from the FDA.
The Company’s CAR-T cell therapy product candidate pipeline is shown below:
ANTLER Clinical Trial Results
On November 3, 2025, the Company announced results from its ongoing ANTLER phase 1 clinical trial evaluating vispa-cel in 2L LBCL patients, including the first data disclosure on 22 patients enrolled in the phase 1 confirmatory cohort to prospectively evaluate patients who received a dose of vispa-cel manufactured from a donor with at least four matching human leukocyte antigen (“HLA”) alleles (of 12 total alleles) with the patient (“partial HLA matching”) as well as on additional LBCL patients from dose escalation and dose expansion.
As of September 2, 2025, the safety data cutoff date, the ANTLER trial had enrolled 84 patients, including the confirmatory cohort of 22 2L LBCL patients designed to prospectively confirm the positive outcomes of partial HLA matching observed in earlier retrospective analyses. Following a lymphodepletion (“LD”) regimen of 60 mg/kg/day of cyclophosphamide for two days and 25 mg/m2/day of fludarabine for five days, each patient in the confirmatory cohort received a dose of vispa-cel at the recommended phase 2 dose (“RP2D”) (80x106 viable CAR-T cells) such that the patient and the T cell donor shared at least four of 12 HLA alleles. As of September 29, 2025, the efficacy data cutoff date, the results for the 22-patient confirmatory cohort were as follows:
• 82% (18/22) overall response rate (“ORR”)
• 64% (14/22) complete response (“CR”) rate
• 51% 12-month progression-free survival (“PFS”)
Median follow-up time for the confirmatory cohort was 6.0 months.
The announced results included an analysis of the 35 CD19-naïve, LBCL patients enrolled in the Company’s ANTLER phase 1 clinical trial who received a dose of vispa-cel that met the criteria for the Company’s optimized product profile, which criteria are a young T cell donor under the age of 30 and a minimum of two of 12 HLA alleles matched between the patient and the T cell donor. Twenty of these 35 patients were enrolled in the confirmatory cohort, and the remaining 15 patients were enrolled in dose escalation or dose expansion. Thirty-two of the 35 patients were 2L patients and three of the 35 patients were third-line or later (“3L+”) patients. As of September 29, 2025, the efficacy data cutoff date, the results for the 35-patient optimized product cohort were as follows:
• 86% (30/35) ORR
• 63% (22/35) CR rate
• 53% 12-month PFS
Median follow-up time for the optimized cohort was 11.8 months.
The following is a summary of results for the confirmatory cohort and the optimized profile for vispa-cel:
12L LBCL 4+ HLA matched, dosed with 80x106 viable CAR-T cells
22L (N=32) and 3L+ (N=3) LBCL patients treated with 40x106 viable CAR-T cells, 80x106 viable CAR-T cells, or 120x106 viable CAR-T cells, optimized for multiple factors, including 2+ HLA matching and young donor-derived
Additional information:
• Efficacy data cutoff date: September 29, 2025
Definitions: “DoR”: duration of response; “NE”: not evaluable; “NR”: not reached; “CI”: confidence interval The results are further summarized in the following swimmer plot for the optimized profile cohort1:
1 2L (N=32) and 3L+ (N=3) LBCL patients treated with 40x106 viable CAR-T cells, 80x106 viable CAR-T cells, or 120x106 viable CAR-T cells, optimized for multiple factors, including 2+ HLA matching and young donor-derived
* Patient diagnosed with lung adenocarcinoma after day 28 scan revealed a non-responsive lung nodule and was enrolled in the Company’s long-term follow-up study. Patient is last known to be in continued response without additional anti-lymphoma therapy at one year post vispa-cel treatment
Additional information:
• Long-term follow-up data reflect the last known response; marked time points indicate confirmation of no disease progression
• One vispa-cel-related grade 5 IEC-HS occurred on day 25 post-infusion
• Median duration of complete response (“DoCR”) was not reached in this cohort
• Certain patients converted from CR or PR to PD at various assessment time points as indicated in the chart above
• Efficacy data cutoff date: September 29, 2025
Definition: “IEC-HS”: immune effector cell-associated hemophagocytic lymphohistiocytosis-like syndrome
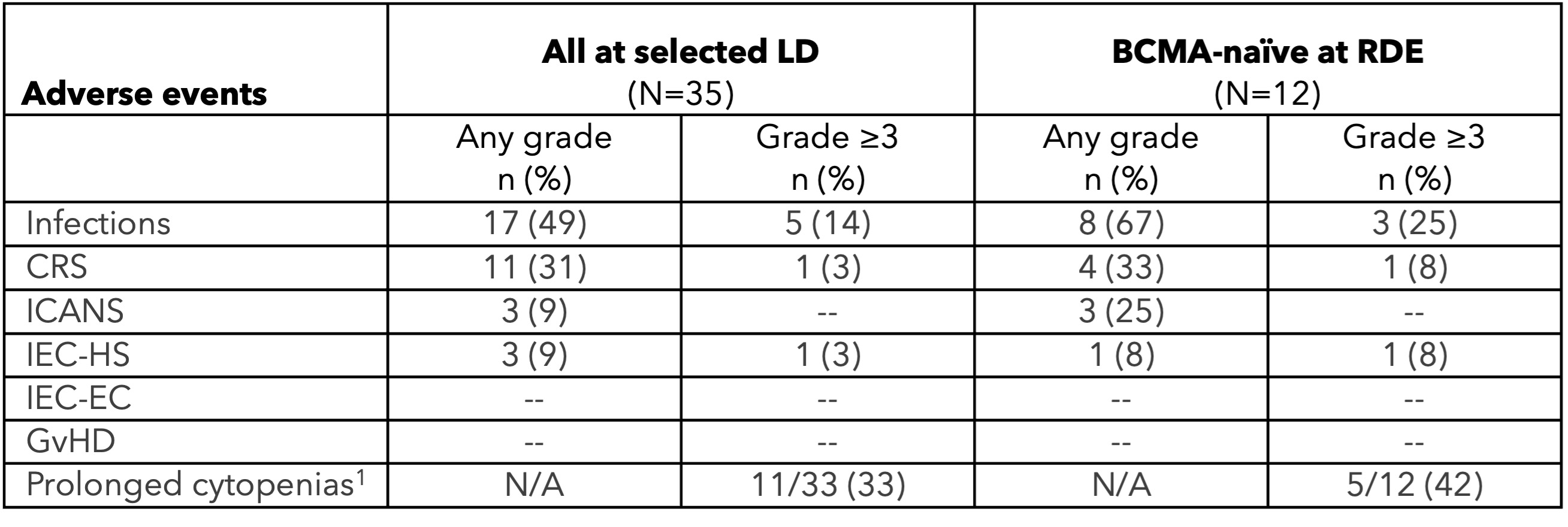
Vispa-cel was generally well tolerated. As of September 2, 2025, the safety data cutoff date, treatment emergent adverse events (“TEAEs”) at any grade in 25% or greater of all 84 patients who received vispa-cel were thrombocytopenia (62%), cytokine release syndrome (“CRS”) (55%), anemia (52%), neutropenia (39%), hypokalemia (26%), and leukopenia (26%). In the confirmatory and optimized profile cohorts, there were no cases of graft-versus-host disease (“GvHD”) or grade 3 or greater immune effector cell-associated neurotoxicity syndrome (“ICANS”), fewer than 5% of patients experienced grade 3 or greater CRS, and 28% (out of 80 patients) experienced prolonged cytopenias.
The following is a summary of notable adverse events for all three cohorts in the ANTLER phase 1 clinical trial (safety data cutoff date: September 2, 2025):
12L LBCL 4+ HLA matched, dosed with 80x106 viable CAR-T cells
22L (N=32) and 3L+ (N=3) LBCL patients treated with 40x106 viable CAR-T cells, 80x106 viable CAR-T cells, or 120x106 viable CAR-T cells, optimized for multiple factors, including 2+ HLA matching and young donor-derived
3Prolonged cytopenias are defined as grade 3 or 4 neutropenia, thrombocytopenia, or anemia ongoing at day 28 (+/- 5 days) post CAR-T cell infusion, based on laboratory data, distinct from investigator-reported clinical adverse events
4Includes one vispa-cel-related grade 5 IEC-HS that occurred day 25 post-infusion
A cohort of 3L+ LBCL patients with prior exposure to CD19-targeted therapy enrolled five patients as of the September 2, 2025, safety data cutoff date. Enrollment in this cohort has been paused to focus on CD19-naïve patients.
Pivotal Phase 3 Clinical Trial in 2L r/r LBCL
In recent interactions, the FDA has recommended that the Company conduct a randomized, controlled trial in 2L LBCL CD19-naïve patients who are ineligible for transplant and autologous CAR-T cell therapy, and the Company intends to follow this approach with its planned pivotal phase 3 trial design and expects to evaluate approximately 250 patients. The Company believes there is a meaningful opportunity within this population, as it anticipates that approximately half of the estimated 10,000 2L DLBCL patients are transplant-ineligible and that a meaningful majority of the transplant-ineligible patients are also autologous CAR-T-ineligible. Patients randomized to the study arm would receive a single dose of vispa-cel at the RP2D of 80x106 viable CAR-T cells following lymphodepletion with 60 mg/kg/day of cyclophosphamide for two days and 25 mg/m2/day of fludarabine for five days. Patients randomized to the comparator arm would be treated with the investigator’s choice of standard of care immunochemotherapy agents. The primary endpoint would be PFS, and an interim analysis is planned. Secondary endpoints would include ORR, CR rate, DoR, DoCR, overall survival, quality of life, and safety. The Company intends to further refine its pivotal phase 3 clinical trial design through continued engagement with the FDA prior to initiation of the pivotal trial. Although the Company believes this approach provides a straightforward regulatory path to full FDA approval of vispa-cel based on FDA interactions to date, the Company cannot assure you that, in future discussions with the FDA, the FDA will not recommend or request additional requirements for the Company’s planned pivotal phase 3 clinical trial design for vispa-cel.
CaMMouflage Clinical Trial Results
On November 3, 2025, the Company announced results from the dose escalation portion of its ongoing CaMMouflage phase 1 trial evaluating CB-011 in r/r MM patients. Forty-eight fourth-line or later (“4L+”) patients were enrolled in the dose escalation portion of the trial; enrollment in dose escalation is now completed. Patients had been treated with three or more prior lines of therapy including a proteasome inhibitor, an immunomodulatory drug, and an anti-CD38 antibody. Prior treatment with an autologous CAR-T cell therapy was not permitted and no BCMA-targeted therapy was permitted within the prior three months.
The Company evaluated two different LD regimens and multiple CAR-T cell dose levels. Thirty-five patients were treated with an LD regimen of 500 mg/m2 cyclophosphamide and 30 mg/m2 fludarabine daily for three days (“the selected LD regimen”). A single dose of CB-011 preceded by the selected LD regimen resulted in responses at all dose levels evaluated (150x106 viable CAR-T cells, N=6; 300x106 viable CAR-T cells, N=13; 450x106 viable CAR-T cells, N=13; and 800x106 viable CAR-T cells, N=3).
Also on November 3, 2025, the Company announced that the 450x106 viable CAR-T cell dose with the selected LD regimen is its recommended dose for expansion (“RDE”).
As of September 24, 2025, the data cutoff date, the results for the 12-patient, BCMA-naïve cohort treated with the selected LD regimen and the 450x106 viable CAR-T cell dose were as follows:
•92% (11/12) ORR
•75% (9/12) CR / stringent CR (“sCR”) rate
•91% (10/11) evaluable patients achieved minimal residual disease (“MRD”) negativity (≤10-5)
•Seven of the 12 patients remain on study as of the data cutoff date in very good partial response (“VGPR”) or better six months or longer following receipt of a single dose of CB-011
The median follow-up for patients dosed with the RDE was 8.3 months, and the longest responding patient in this cohort is in an sCR at 15 months post-infusion.
The results are further summarized in the following swimmer plot for the 12-patient, BCMA-naïve cohort treated with the selected LD regimen and the 450x106 viable CAR-T cell dose:
1MRD negative at ≤10-5
Additional information:
•One patient who had previously withdrawn from the trial died on day 90 of treatment-related ICAHT; one patient died of pneumonia on day 50 (not treatment-related)
•Data shown are from BCMA-naïve patients dosed at the 450x106 viable CAR-T cell dose with the selected LD regimen
•Data cutoff date: September 24, 2025
Definitions: “EMD”: extramedullary disease; “ICAHT”: immune effector cell-associated hematotoxicity; “mo”: months
CB-011 had a manageable safety profile across all dose levels and lymphodepletion regimens (N=48), with no cases of GvHD, immune effector cell-associated enterocolitis (“IEC-EC”), parkinsonism, or cranial nerve palsies. TEAEs in 25% or greater of all patients treated with CB-011 following the selected LD regimen (N=35) were as follows: neutropenia (80%), anemia (60%), thrombocytopenia (49%), infections (49%), dizziness (31%), cytokine release syndrome (31%), fatigue (31%), leukopenia (29%), decreased appetite (29%), constipation (26%), and pyrexia (26%). Notable adverse events in the RDE cohort included one CB-011-related grade 5 ICAHT on day 90, one grade 5 pneumonia not related to CB-011 on day 50, and one grade 4 CB-011-related Guillain-Barré Syndrome on day 129, which is resolving. In the cohort evaluating the 300x106 viable CAR-T cell dose level following the selected LD regimen, there was one grade 5 respiratory syncytial virus not related to CB-011 on day 73. Prophylactic measures for cytopenias and infections and early intervention for IEC-HS have been successfully implemented in the Company’s clinical protocol.
The following is a summary of notable adverse events in the CaMMouflage phase 1 clinical trial (data cutoff date: September 24, 2025):
1Any continued ≥ grade 3 cytopenia based on laboratory data at ≥ day 35; denominator is those evaluable at day 35 (+/- five days)
The Company plans to initiate dose expansion of the CaMMouflage phase 1 clinical trial before the end of 2025 and to report dose expansion data, along with longer follow-up on dose escalation data, in 2026.
Preliminary unaudited cash, cash equivalents, and marketable securities as of September 30, 2025
The information reported under Item 2.02 of this Form 8-K is hereby incorporated by reference herein.
Forward-Looking Statements
The disclosure in this Current Report on Form 8-K contains “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995 relating to the business and operations of the Company. These forward-looking statements are subject to a number of known and unknown risks, assumptions, uncertainties, and other factors that may cause the actual results, levels of activity, performance, or achievements of the Company to be materially different from those expressed or implied by any forward-looking statements. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “expect,” “likely,” “plan,” “anticipate,” “could,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “potential,” “contribute to,” or “continue” or the negative of these terms or other similar expressions, although not all forward-looking statements contain these words. All statements, other than statements of historical facts contained in this presentation, are forward-looking statements, including but not limited to any statements regarding the initiation, timing, progress, strategy, plans, objectives, expectations (including as to the results) with respect to the Company’s CAR-T cell therapy product candidate clinical trials, including the expected design, protocol, and timing of initiation of the pivotal phase 3 clinical trial for vispa-cel in 2L LBCL CD19-naïve patients; its expectations and timing of initiating dose expansion by the end of 2025 and reporting dose expansion data, along with longer follow-up data on dose escalation, in 2026 from its ongoing CaMMouflage Phase 1 clinical trial for CB-011 in patients with r/r MM; the projected manufacturing costs for its CAR-T cell therapy product candidates; the potential commercial opportunities for its CAR-T cell therapy product candidates; the likelihood of its clinical trials demonstrating safety and efficacy of its CAR-T cell therapy product candidates; the beneficial characteristics, safety, efficacy, therapeutic effects, and potential advantages of its CAR-T cell therapy product candidates; the expected timing or likelihood of regulatory filings and approval for its CAR-T cell therapy product candidates; its expected cash, cash equivalents, and marketable securities as of September 30, 2025; and the sufficiency and anticipated use of its existing capital resources to fund its future operating expenses and capital expenditure requirements and needs for additional financing.
As a result of many factors, including but not limited to, risks inherent in the development of allogeneic CAR-T cell therapy products; uncertainties related to the initiation, cost, timing, progress, and results of its current and future clinical trials; the risk that initial, preliminary, or interim clinical trial data will not ultimately be predictive of the safety and efficacy of its CAR-T cell therapy product candidates or that clinical outcomes may differ as patient enrollment continues and as more patient data becomes available; the risk that different conclusions or considerations are reached once additional data have been received and fully evaluated; the ability to obtain key regulatory input and approvals; and risks related to its limited operating history, history of net operating losses, financial position, and its ability to raise additional capital as needed to fund its operations and CAR-T cell therapy product candidate development, including the ability to fully fund its pivotal phase 3 clinical trial for vispa-cel; as well as other risks described in greater detail in its filings with the SEC, including the section titled “Risk Factors” of its Annual Report on Form 10-K for the year ended December 31, 2024, and other filings the Company makes with the SEC, the events and circumstances reflected in its forward-looking statements may not be achieved or may not occur, and actual results could differ materially from those described in or implied by the forward-looking statements contained in this presentation.
As a result of these risks, you should not place undue reliance on these forward-looking statements. The forward-looking statements in this presentation are made only as of the date hereof. Except to the extent required by law, the Company assumes no obligation and does not intend to update any of these forward-looking statements after the date of this presentation or to conform these statements to actual or revised expectations.
The Company has not completed any head-to-head trial that compares the safety and efficacy of vispa-cel or CB-011 with any autologous CAR-T cell therapy or alternative immunotherapy as part of the same investigational setting. Separate clinical trials for autologous CAR-T cell therapy and alternative immunotherapies may differ in trial design and duration, patient population, patient characteristics, clinical trial phase, treatment protocols, investigators, and other important factors, making it difficult to compare data across clinical trials or to draw reliable conclusions from such cross-trial comparisons. As a result of these factors, cross-trial comparisons may have no interpretive value on vispa-cel or CB-011’s existing or future clinical trial results.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits
|
|
|
|
|
|
|
|
|
| Exhibit No. |
|
Description |
| 99.1 |
|
|
| 99.2 |
|
|
| 99.3 |
|
|
| 104 |
|
Cover Page Interactive Data File (embedded within the Inline XBRL document) |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Caribou Biosciences, Inc. |
|
|
|
|
| Date: |
November 3, 2025 |
By: |
/s/ Rachel E. Haurwitz |
|
|
|
Rachel E. Haurwitz
President and Chief Executive Officer |
EX-99.1
2
crbu-20251103xexx991.htm
EX-99.1
Document
Caribou Biosciences Announces Positive Data from ANTLER Phase 1 Trial Demonstrating Efficacy and Durability of Vispa-cel (CB-010), an Allogeneic CAR-T Cell Therapy, on Par with Autologous CAR-T Cell Therapies
•Data demonstrate efficacy and durability of vispa-cel, an allogeneic anti-CD19 CAR-T cell therapy, are on par with autologous CAR-T cell therapies in the confirmatory cohort (N=22) and with longer-term follow-up on patients who received optimized vispa-cel (N=35)
◦82% ORR, 64% CR rate, 51% PFS at 12 months in patients prospectively enrolled in partial HLA matching confirmatory cohort (N=22)
◦86% ORR, 63% CR rate, 53% PFS at 12 months in patients who received vispa-cel with an optimized profile (N=35)
•Vispa-cel is generally well-tolerated, allowing for administration in the outpatient setting
•Data highlight vispa-cel’s potential as best-in-class allogeneic CAR-T cell therapy for LBCL
•Conference call and webcast scheduled for today at 8:00 am ET
BERKELEY, Calif., November 3, 2025 (GLOBE NEWSWIRE) -- Caribou Biosciences, Inc. (Nasdaq: CRBU), a leading clinical-stage CRISPR genome-editing biopharmaceutical company, today announced positive results from its ongoing ANTLER phase 1 clinical trial evaluating vispacabtagene regedleucel (vispa-cel; formerly CB-010), an allogeneic anti-CD19 CAR-T cell therapy, in patients with relapsed or refractory B cell non-Hodgkin lymphoma (r/r B-NHL).
“The ANTLER data mark an exciting advancement for the field of cellular immunotherapy,” said Mehdi Hamadani, MD, professor of medicine and section chief of hematologic malignancies at the Medical College of Wisconsin and an investigator on the ANTLER trial. “This clinical dataset demonstrates vispa-cel’s efficacy and durability are comparable to autologous CAR-T therapies, yet its off-the-shelf availability and favorable tolerability profile make it well suited for outpatient administration at both large academic centers and sophisticated community hospitals. This combination of robust clinical activity and accessibility could significantly broaden patient access to transformative CAR-T cell treatments, particularly for those who cannot wait or are ineligible for transplantation or autologous CAR-T cell therapies.”
As of the September 2, 2025, safety data cutoff date, the ANTLER trial has enrolled 84 patients, including a confirmatory cohort of 22 CD19-naïve second-line (2L) large B cell lymphoma (LBCL) patients. The confirmatory cohort was designed to prospectively confirm the positive outcomes of partial HLA matching (≥4 matched HLA alleles) observed in earlier retrospective analyses. Patients in the confirmatory cohort received vispa-cel at the recommended phase 2 dose (RP2D; 80×10⁶ CAR-T cells), and the data from this cohort, as of an efficacy data cutoff date of September 29, 2025, confirmed that a single dose of partially matched (≥4 matched HLA alleles) vispa-cel results in efficacy that is on par with approved autologous CAR-T cell therapies including an 82% overall response rate (ORR), a 64% complete response (CR) rate, and 51% progression-free survival (PFS) at 12 months (Table 1). Median follow-up for the confirmatory cohort is 6.0 months.
Table 1. ANTLER phase 1 trial endpoints
|
|
|
|
|
|
|
|
|
Endpoints |
Confirmatory cohort1
N=22
|
Optimized profile2
N=35
|
ORR |
82% |
86% |
CR rate |
64% |
63% |
|
Median PFS
(95% CI)
|
NR
(2.0, NE)
|
NR
(2.8, NE)
|
|
12-month PFS
(95% CI)
|
51%
(28, 70)
|
53%
(34, 69)
|
|
Median DoR
(95% CI)
|
NR
(1.7, NE)
|
NR
(2.1, NE)
|
12L LBCL 4+ HLA matched, dosed with 80M vispa-cel CAR-T cells
22L (N=32) and 3L+ (N=3) LBCL patients treated with 40M, 80M, or 120M vispa-cel CAR-T cells optimized for multiple factors, including 2+ HLA matching and young donor-derived
CR: complete response; DoR: duration of response; HLA: human leukocyte antigen; NE: not evaluable; NR: not reached; ORR: overall response rate; PFS: progression-free survival
Data cutoff date for efficacy: September 29, 2025
The Company leveraged its large allogeneic CAR-T cell clinical data set (>140 patients dosed across multiple clinical trials) to identify key factors linked to successful patient outcomes. Two of those factors are donor age (young donors drive enhanced outcomes relative to older donors) and partial HLA matching (matching 2 or more [2+] alleles correlates with outcomes on par with autologous CAR-T cell therapies). Of the 84 patients dosed with vispa-cel, there are 35 CD19-naïve LBCL patients who received vispa-cel with an optimized profile (32 of these patients were 2L and 3 of these patients were 3L+). The optimized profile vispa-cel was manufactured from young donor-derived T cells, and the 35 patients matched a minimum of 2 HLA alleles with the T cell donor. Twenty of the 35 patients in the optimized profile cohort were enrolled in the confirmatory cohort, and the remaining 15 patients were enrolled in dose escalation or expansion.
Data from this 35-patient cohort further confirmed that the efficacy and durability of vispa-cel are on par with the autologous CAR-T cell therapies. Median follow-up for the optimized profile cohort was 11.8 months, and the longest responding patient, who completed the 2-year ANTLER trial and enrolled in the long-term follow-up study, is in complete response at 3 years post infusion. As of the September 29, 2025, efficacy data cutoff date, the results for the 35-patient optimized profile cohort included an 86% ORR, a 63% CR rate, and 53% PFS at 12 months (Table 1).
In all patients treated in ANTLER (N=84), vispa-cel has demonstrated a generally well-tolerated safety profile. As of the September 2, 2025, safety data cutoff date, treatment emergent adverse events at any grade in ≥25% of all patients who received vispa-cel were thrombocytopenia (62%), cytokine release syndrome (CRS; 55%), anemia (52%), neutropenia (39%), hypokalemia (26%), and leukopenia (26%). In the confirmatory and optimized profile cohorts, there were no cases of graft-versus-host disease (GvHD) or ≥grade 3 immune effector cell-associated neurotoxicity syndrome (ICANS), <5% patients experienced ≥grade 3 CRS, and there were manageable rates of infections and prolonged cytopenias (Table 2). The safety profile of vispa-cel allows for use in an outpatient setting.
“We believe that with these results, Caribou has achieved what the field has long sought — strong evidence that an allogeneic CAR-T cell therapy can be on par with the efficacy and durability of autologous treatments and broaden access with a safety profile that allows for outpatient use,” said Rachel Haurwitz, PhD, Caribou’s president and CEO. “This milestone positions vispa-cel as a potentially best-in-class allogeneic CAR-T cell therapy for patients with large B cell lymphoma. We believe we have a straightforward regulatory path toward full registration by following the FDA’s recommendation for a randomized, controlled phase 3 trial in second-line large B cell lymphoma, and we plan to refine our pivotal trial design in the coming months through continued engagement with the FDA.”
Table 2. ANTLER phase 1 trial safety data
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Events, n (%) |
All treated
N=84
|
Confirmatory cohort
N=221
|
Optimized profile
N=352
|
|
All grade |
≥Grade 3 |
All grade |
≥Grade 3 |
All grade |
≥Grade 3 |
ICANS |
12 (14) |
4 (5) |
1 (5) |
0 (0) |
1 (3) |
0 (0) |
CRS |
46 (55) |
1 (1) |
13 (59) |
1 (5) |
19 (54) |
1 (3) |
Infections |
43 (51) |
21 (25) |
9 (41) |
4 (18) |
20 (57) |
6 (17) |
Prolonged cytopenias3 |
NA |
22/80 (28) |
NA |
5/19 (26) |
NA |
7/32 (22) |
IEC-HS4 |
2 (2) |
2 (2) |
1 (5) |
1 (5) |
1 (3) |
1 (3) |
12L LBCL 4+ HLA matched, dosed with 80M vispa-cel CAR-T cells
22L (N=32) and 3L+ (N=3) LBCL patients treated with 40M, 80M, or 120M vispa-cel CAR-T cells optimized for multiple factors, including 2+ HLA matching and young donor-derived
3Prolonged cytopenias are defined as Grade 3 or 4 neutropenia, thrombocytopenia, or anemia ongoing at day 28 (+/- 5 days) post CAR-infusion, based on laboratory data, distinct from investigator-reported clinical adverse events.
4Includes one vispa-cel-related grade 5 IEC-HS that occurred on day 25 post-infusion.
CRS: cytokine release syndrome; IEC-HS: immune effector cell-associated hemophagocytic lymphohistiocytosis-like syndrome; NA: not applicable
Data cutoff date for safety: September 2, 2025
A cohort of third-line or later LBCL patients with prior exposure to CD19-targeted therapy enrolled 5 patients as of the September 2, 2025, safety data cutoff date. Enrollment in this cohort has been paused to focus on CD19-naïve patients.
Pivotal phase 3 trial for 2L LBCL for full approval
In recent interactions, the FDA has recommended the Company conduct a randomized, controlled trial in 2L LBCL CD19-naive patients who are ineligible for transplant and autologous CAR-T cell therapy, and the Company intends to follow this approach with its planned pivotal phase 3 clinical trial design, evaluating approximately 250 patients. Patients randomized to the study arm would receive a single dose of vispa-cel at the recommended phase 2 dose of 80×10⁶ CAR-T cells following lymphodepletion with cyclophosphamide and fludarabine, and patients randomized to the comparator arm would be treated with the investigator’s choice of standard of care immunochemotherapy agents. The primary endpoint is PFS, and an interim analysis of PFS is planned. Secondary endpoints would include ORR, complete response rate (CRR), duration of response (DoR), duration of complete response (DoCR), overall survival (OS), quality of life (QoL), and safety. This expected trial design reflects Caribou’s internal analysis and interactions with the FDA to date. The Company intends to further refine the pivotal trial design through continued engagement with the FDA prior to initiation. Caribou plans to bring vispa-cel closer to where patients live by leveraging community and academic sites within the U.S. and globally.
Webcast conference call today at 8:00 am ET
Caribou will host a live conference call and webcast on Monday, November 3 at 8:00 am ET to discuss the ANTLER trial data and the pivotal phase 3 clinical trial design, as well as the CaMMouflage phase 1 clinical trial for r/r multiple myeloma. The presenters will include:
•Mehdi Hamadani, MD, professor of medicine, section chief of hematologic malignancies at Medical College of Wisconsin, and investigator on the ANTLER trial
•Joseph McGuirk, DO, professor of hematology/oncology and division director for hematologic malignancies and cellular therapeutics at University of Kansas Cancer Center
•Adriana Rossi, MD, director of CAR-T and stem cell transplant clinical program at the center of excellence for multiple myeloma at Mt Sinai, and investigator on the CaMMouflage trial
•Rachel Haurwitz, PhD, president and chief executive officer, Caribou Biosciences
•Tina Albertson, MD, PhD, chief medical officer, Caribou Biosciences
A live webcast of the presentation will be accessible via Caribou’s website on the Events page. The archived webcast will be available on the Caribou website for 30 days after the event.
About vispacabtagene regedleucel
Vispacabtagene regedleucel (vispa-cel; formerly known as CB-010) is an allogeneic anti-CD19 CAR-T cell therapy being evaluated in patients with relapsed or refractory B cell non-Hodgkin lymphoma (r/r B-NHL). To Caribou’s knowledge, vispa-cel is the first allogeneic CAR-T cell therapy in the clinic with a PD-1 knockout, a genome-editing strategy designed to enhance CAR-T cell activity by limiting premature CAR-T cell exhaustion. The FDA granted vispa-cel Regenerative Medicine Advanced Therapy (RMAT), Orphan Drug, and Fast Track designations for B-NHL.
About the ANTLER phase 1 clinical trial
The ANTLER clinical trial is a multicenter, open-label phase 1 trial evaluating vispacabtagene regedleucel (vispa-cel; formerly CB-010) in adults with relapsed or refractory B cell non-Hodgkin lymphoma (B-NHL). Eighty-four patients have been treated in the ANTLER clinical trial as of September 2, 2025. Using a 3+3 enrollment strategy, safety and efficacy were assessed in 16 patients in dose escalation evaluating 40x106, 80x106, and 120x106 CAR-T cell dose levels with a lymphodepletion regimen of cyclophosphamide at 60 mg/kg/day for 2 days followed by fludarabine at 25 mg/m2/day for 5 days. Forty-one patients with 2L LBCL were enrolled in the dose expansion portion, and 80x106 CAR-T cells was selected as the recommended phase 2 dose (RP2D). An additional 22 patients with 2L LBCL were enrolled in the confirmatory cohort, which prospectively evaluated the Company’s partial HLA matching strategy. Five patients were enrolled in a cohort of third-line or later LBCL patients with prior exposure to CD19-targeted therapy. Additional information on the ANTLER trial (NCT04637763) can be found at clinicaltrials.gov.
About Caribou Biosciences, Inc.
Caribou is a clinical-stage CRISPR genome-editing biopharmaceutical company dedicated to developing transformative therapies for patients with devastating diseases. The Company’s genome-editing platform, including its Cas12a chRDNA technology, enables superior precision to develop cell therapies that are armored to potentially improve activity against diseases. Caribou is focused on vispacabtagene regedleucel (vispa-cel) and CB-011 as off-the-shelf CAR-T cell therapies that have the potential to provide broad access and rapid treatment for patients with hematologic malignancies. Follow the Company @CaribouBio and visit www.cariboubio.com.
Forward-looking statements and important information
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “expect,” “plan,” “anticipate,” “could,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “potential,” or “continue,” or the negative of these terms or other similar expressions, although not all forward-looking statements contain these words. These forward-looking statements include, but are not limited to, any statements regarding the initiation, timing, progress, strategy, plans, objectives, expectations (including as to the results) with respect to the Company’s CAR-T cell therapy product candidate clinical trials, including the expected design, protocol, and timing of initiation of the pivotal phase 3 clinical trial for vispa-cel in 2L LBCL CD19-naïve patients; its ability to successfully develop its CAR-T cell therapy product candidates and to obtain and maintain regulatory approval for these product candidates; the likelihood of its clinical trials demonstrating safety and efficacy of its CAR-T cell therapy product candidates; the beneficial characteristics, safety, efficacy, therapeutic effects, and potential advantages of its CAR-T cell therapy product candidates; and the expected timing or likelihood of regulatory filings and approval for its CAR-T cell therapy product candidates. Management believes that these forward-looking statements are reasonable as and when made. However, such forward-looking statements are subject to risks and uncertainties, and actual results may differ materially from any future results expressed or implied by the forward-looking statements. Risks and uncertainties include, without limitation, risks inherent in the development of allogeneic CAR-T cell therapy products; uncertainties related to the initiation, cost, timing, progress, and results of its current and future clinical trials; the risk that initial, preliminary, or interim clinical trial data will not ultimately be predictive of the safety and efficacy of its CAR-T cell therapy product candidates or that clinical outcomes may differ as patient enrollment continues and as more patient data becomes available; the risk that different conclusions or considerations are reached once additional data have been received and fully evaluated; the ability to obtain key regulatory input and approvals; and risks related to its limited operating history, history of net operating losses, financial position, and its ability to raise additional capital as needed to fund its operations and CAR-T cell therapy product candidate development, including the ability to fully fund its pivotal phase 3 clinical trial for vispa-cel; as well as other risk factors described from time to time in Caribou’s filings with the Securities and Exchange Commission (SEC), including its Annual Report on Form 10-K for the year ended December 31, 2024, and subsequent SEC filings. In light of the significant uncertainties in these forward-looking statements, you should not rely upon forward-looking statements as predictions of future events. Except as required by law, Caribou undertakes no obligation to update publicly any forward-looking statements for any reason.
Caution should be exercised when interpreting results from separate trials involving commercially approved autologous CAR-T cell therapies. The results of autologous CAR-T cell therapies referenced in this press release have been derived from publicly available reports of clinical trials not conducted by Caribou, and Caribou has not performed any head-to-head trials comparing any of these autologous CAR-T cell therapies with vispa-cel. As such, the results of these autologous CAR-T cell therapy clinical trials may not be comparable to clinical results for vispa-cel. The autologous CAR-T cell therapy clinical trials vary in material ways from the ANTLER clinical trial for vispa-cel including with respect to trial design and duration, patient population, patient characteristics, clinical trial phase, treatment protocols, investigators, and other important factors. As a result, cross-trial comparisons may have no interpretive value on Caribou’s existing or future clinical results. For further information and to understand these material differences, you should read the reports for the autologous CAR-T cell therapy clinical trials and the sources included in Caribou’s webcast presentation and its corporate presentations on its website.
Caribou Biosciences, Inc. contact:
Peggy Vorwald, PhD
investor.relations@cariboubio.com
media@cariboubio.com
EX-99.2
3
crbu-20251103xexx992.htm
EX-99.2
Document
Caribou Biosciences Announces Positive Data from CaMMouflage Phase 1 Trial of CB-011 in Multiple Myeloma
•First clinical data disclosure for CB-011 highlights its potential as a best-in-class allogeneic CAR-T cell therapy for relapsed or refractory multiple myeloma
•450 million cell dose is the recommended dose for expansion (RDE); dose expansion to initiate by year end and data expected in 2026
•92% ORR, 75% ≥CR rate, 91% MRD negativity in the 12-patient, BCMA-naïve cohort treated at the RDE with the selected lymphodepletion regimen
•Conference call and webcast scheduled for today at 8:00 am ET
BERKELEY, Calif., November 3, 2025 (GLOBE NEWSWIRE) — Caribou Biosciences, Inc. (Nasdaq: CRBU), a leading clinical-stage CRISPR genome-editing biopharmaceutical company, today announced its first clinical data from dose escalation in the ongoing CaMMouflage phase 1 trial evaluating CB-011, an off-the-shelf anti-BCMA CAR-T cell therapy, in relapsed or refractory multiple myeloma (r/r MM), highlighting CB-011 as a potentially best-in-class allogeneic CAR-T cell therapy for this patient population. The Company is advancing the program into dose expansion, which it expects to initiate by the end of this year, and the Company plans to share dose expansion data in 2026.
“Despite a rapidly advancing landscape of treatment options for multiple myeloma, challenges remain with only 10% of eligible patients receiving an autologous CAR-T,” said Adriana Rossi, MD, director of CAR-T and stem cell transplant clinical program at the center of excellence for multiple myeloma at Mount Sinai, and an investigator on the CaMMouflage trial. “I believe the promising responses we are seeing with CB-011 combined with the off-the-shelf nature of this therapy could represent a paradigm shift for patients with relapsed or refractory multiple myeloma. I am excited about the potential this holds for the large number of multiple myeloma patients who simply cannot wait for autologous CAR-T manufacturing and prefer a single dose approach.”
In the dose escalation portion of the CaMMouflage phase 1 clinical trial, safety and efficacy of CB-011 were evaluated in 48 fourth-line and later (4L+) patients at multiple dose levels and following two different lymphodepletion regimens. Thirty-five patients were treated with the selected lymphodepletion (LD) regimen of 500 mg/m2 cyclophosphamide and 30 mg/m2 fludarabine daily for three days. A single dose of CB-011 preceded by the selected LD regimen resulted in responses at all dose levels evaluated (150x106 [N=6], 300x106 [N=13], 450x106 [N=13], and 800x106 [N=3] CAR-T cells). The 450x106 CAR-T cell dose with the selected LD regimen is the recommended dose for expansion (RDE). Twelve BCMA-naïve r/r MM patients were treated with the RDE. The median follow-up for patients dosed with the RDE was 8.3 months, and the longest responding patient is in a stringent complete response (sCR) at 15 months post-infusion. As of the September 24, 2025, data cutoff date, the results for the 12-patient, BCMA-naïve cohort treated with the RDE were as follows:
•92% (11/12) overall response rate (ORR)
•75% (9/12) ≥complete response (CR) rate
•91% (10/11 evaluable patients) achieved minimal residual disease (MRD) negativity (≤10-5)
•7 of 12 patients remained on study as of the data cutoff date in ≥very good partial response (VGPR) 6 months or longer following receipt of a single dose of CB-011
CB-011 had a manageable safety profile across all dose levels and lymphodepletion regimens (N=48), with no cases of graft-versus-host disease (GvHD), immune effector cell-associated enterocolitis (IEC-EC), parkinsonism, or cranial nerve palsies. Treatment emergent adverse events (TEAEs) in ≥25% of all patients treated with CB-011 following the selected LD regimen (N=35) were as follows: neutropenia (80%), anemia (60%), thrombocytopenia (49%), infections (49%), dizziness (31%), cytokine release syndrome (31%), fatigue (31%), leukopenia (29%), decreased appetite (29%), constipation (26%), and pyrexia (26%). Notable adverse events in the RDE cohort included one CB-011-related grade 5 immune effector cell-associated hematotoxicity (ICAHT) on day 90, one grade 5 pneumonia not related to CB-011 on day 50, and one grade 4 CB-011-related Guillain-Barré Syndrome on day 129, which is resolving. In the cohort evaluating the 300x106 CAR-T cell dose level following the selected LD regimen, there was one grade 5 respiratory syncytial virus not related to CB-011 on day 73. Prophylactic measures for cytopenias and infections and early intervention for immune effector cell-associated hemophagocytic lymphohistiocytosis-like syndrome (IEC-HS) have been successfully implemented in the protocol.
“We are very encouraged by the compelling results from the CaMMouflage phase 1 trial, which demonstrate that CB-011 is delivering deep, durable responses in high-risk, heavily pretreated multiple myeloma patients with a manageable safety profile. These data establish CB-011’s potential to be a best-in-class allogeneic CAR-T cell therapy that could expand access and bring meaningful benefit to patients who urgently need a readily available, single-dose option,” said Rachel Haurwitz, PhD, Caribou’s president and chief executive officer. “We plan to initiate dose expansion before year end and to report dose expansion data, along with longer follow up on dose escalation data, in 2026.”
Webcast conference call today at 8:00 am ET
Caribou will host a live conference call and webcast Monday, November 3 at 8:00 am ET to discuss the CaMMouflage trial data, as well as the ANTLER phase 1 clinical trial and anticipated pivotal trial design. The presenters will include:
•Adriana Rossi, MD, director of CAR-T and stem cell transplant clinical program at the center of excellence for multiple myeloma at Mt Sinai, and investigator on the CaMMouflage trial
•Mehdi Hamadani, MD, professor of medicine, section chief of hematologic malignancies at Medical College of Wisconsin, and investigator on the ANTLER trial
•Joseph McGuirk, DO, professor of hematology/oncology and division director for hematologic malignancies and cellular therapeutics at University of Kansas Cancer Center
•Rachel Haurwitz, PhD, president and chief executive officer, Caribou Biosciences
•Tina Albertson, MD, PhD, chief medical officer, Caribou Biosciences
A live webcast of the presentation will be accessible via Caribou’s website on the Events page. The archived webcast will be available on the Caribou website for 30 days after the event.
About CB-011
CB-011 is an allogeneic anti-BCMA CAR-T cell therapy being evaluated in patients with relapsed or refractory multiple myeloma (r/r MM) in the CaMMouflage phase 1 trial. To Caribou’s knowledge, CB-011 is the first allogeneic CAR-T cell therapy in the clinic that is engineered to enable activity through an immune cloaking strategy with a B2M knockout and insertion of a B2M–HLA-E fusion protein to blunt immune-mediated rejection.
CB-011 has been granted Fast Track and Orphan Drug designations by the FDA.
About the CaMMouflage phase 1 clinical trial
The CaMMouflage clinical trial is a multicenter, open-label phase 1 trial evaluating CB-011 in adults with relapsed or refractory multiple myeloma (r/r MM) who have been treated with three or more prior lines of therapy. Using a 3+3 dose escalation design, safety and efficacy of CB-011 were evaluated in 48 patients at multiple dose levels and two different lymphodepletion regimens. Thirteen patients were treated with a single dose of CB-011 (50x106 [N=3], 150x106 [N=7], and 450x106 [N=3] CAR-T cells) with a lymphodepletion (LD) regimen of 300 mg/m2 cyclophosphamide and 30 mg/m2 fludarabine daily for three days, and 35 patients were treated with a single dose of CB-011 (150x106 [N=6], 300x106 [N=13], 450x106 [N=13], and 800x106 [N=3] CAR-T cells) with a LD regimen of 500 mg/m2 cyclophosphamide and 30 mg/m2 fludarabine daily for three days. The dose expansion portion of the trial will evaluate safety and efficacy of CB-011 at 450x106 CAR-T cells with the selected LD of 500 mg/m2 cyclophosphamide and 30 mg/m2 fludarabine daily for three days. Additional information on the CaMMouflage trial (NCT05722418) can be found at clinicaltrials.gov.
About Caribou Biosciences, Inc.
Caribou is a clinical-stage CRISPR genome-editing biopharmaceutical company dedicated to developing transformative therapies for patients with devastating diseases. The Company’s genome-editing platform, including its Cas12a chRDNA technology, enables superior precision to develop cell therapies that are armored to potentially improve activity against diseases. Caribou is focused on vispacabtagene regedleucel (vispa-cel) and CB-011 as off-the-shelf CAR-T cell therapies that have the potential to provide broad access and rapid treatment for patients with hematologic malignancies. Follow the Company @CaribouBio and visit www.cariboubio.com.
Forward-looking statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “expect,” “plan,” “anticipate,” “could,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “potential,” or “continue,” or the negative of these terms or other similar expressions, although not all forward-looking statements contain these words. These forward-looking statements include, but are not limited to, any statements regarding the initiation, timing, progress, strategy, plans, objectives, expectations (including as to the results) with respect to the Company’s CAR-T cell therapy product candidate clinical trials, including its expectations and timing regarding initiating dose expansion by the end of 2025 and reporting dose expansion data, along with longer follow-up data on dose escalation, in 2026 from its ongoing CaMMouflage phase 1 clinical trial for CB-011 in patients with relapsed or refractory multiple myeloma; its ability to successfully develop its CAR-T cell product candidates and to obtain and maintain regulatory approval for these product candidates; the likelihood of its clinical trials demonstrating safety and efficacy of its CAR-T cell therapy product candidates; the beneficial characteristics, safety, efficacy, therapeutic effects, and potential advantages of its CAR-T cell therapy product candidates; and the expected timing or likelihood of regulatory filings and approval for its CAR-T cell therapy product candidates. Management believes that these forward-looking statements are reasonable as and when made. However, such forward-looking statements are subject to risks and uncertainties, and actual results may differ materially from any future results expressed or implied by the forward-looking statements.
Risks and uncertainties include, without limitation, risks inherent in the development of allogeneic CAR-T cell therapy products; uncertainties related to the initiation, cost, timing, progress, and results of its current and future clinical trials; the risk that initial, preliminary, or interim clinical trial data will not ultimately be predictive of the safety and efficacy of its CAR-T cell therapy product candidates or that clinical outcomes may differ as patient enrollment continues and as more patient data becomes available; the risk that different conclusions or considerations are reached once additional data have been received and fully evaluated; the ability to obtain key regulatory input and approvals; and risks related to its limited operating history, history of net operating losses, financial position, and its ability to raise additional capital as needed to fund its operations and CAR-T cell therapy product candidate development; as well as other risk factors described from time to time in Caribou’s filings with the Securities and Exchange Commission (SEC), including its Annual Report on Form 10-K for the year ended December 31, 2024, and subsequent SEC filings. In light of the significant uncertainties in these forward-looking statements, you should not rely upon forward-looking statements as predictions of future events. Except as required by law, Caribou undertakes no obligation to update publicly any forward-looking statements for any reason.
Caribou Biosciences, Inc. contact:
Peggy Vorwald, PhD
investor.relations@cariboubio.com
media@cariboubio.com
EX-99.3
4
crbu-20251103xex993.htm
EX-99.3
crbu-20251103xex993

N O V E M B E R 3 , 2 0 2 5 Positive clinical updates on Caribou’s off-the-shelf CAR-T cell therapy programs: • Vispa-cel for r/r LBCL (vispacabtagene regedleucel; CB-010) • CB-011 for r/r MM 2 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Important information Forward-looking statements This presentation contains forward-looking statements (FLS) within the meaning of the Private Securities Litigation Reform Act of 1995. These forward-looking statements are subject to a number of known and unknown risks, assumptions, uncertainties, and other factors that may cause the actual results, levels of activity, performance, or achievements of Caribou Biosciences, Inc. (the “Company,” “Caribou,” “we,” or “its”) to be materially different from those expressed or implied by any forward-looking statements. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “expect,” “likely,” “plan,” “anticipate,” “could,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “potential,” “contribute to,” or “continue” or the negative of these terms or other similar expressions, although not all forward-looking statements contain these words. All statements, other than statements of historical facts contained in this presentation, are forward-looking statements, including but not limited to any statements regarding the initiation, timing, progress, strategy, plans, objectives, expectations (including as to the results) with respect to the Company’s CAR-T cell therapy product candidate clinical trials, including its expectations regarding the timing of initiating dose expansion by the end of 2025 and reporting dose expansion data, along with longer follow-up data on dose escalation, in 2026 from its ongoing CaMMouflage Phase 1 clinical trial for CB-011 in patients with relapsed or refractory multiple myeloma; the expected design, protocol, and timing of initiation of the pivotal phase 3 clinical trial for vispa-cel in 2L LBCL CD19-naïve patients; its ability to successfully develop its CAR-T cell therapy product candidates and to obtain and maintain regulatory approval for these product candidates; the projected manufacturing costs for its CAR-T cell therapy product candidates; the potential commercial opportunities for its CAR-T cell therapy product candidates; the likelihood of its clinical trials demonstrating safety and efficacy of its CAR-T cell therapy product candidates; the beneficial characteristics, safety, efficacy, therapeutic effects, and potential advantages of its CAR-T cell therapy product candidates; the expected timing or likelihood of regulatory filings and approval for its CAR-T cell therapy product candidates; its expected cash, cash equivalents, and marketable securities as of September 30, 2025; and the sufficiency and anticipated use of its existing capital resources to fund its future operating expenses and capital expenditure requirements and needs for additional financing. As a result of many factors, including but not limited to, risks related to its limited operating history, history of net operating losses, financial position, and its need for and ability to raise substantial additional capital to fund its operations and CAR-T cell therapy product candidate development including the ability to fully fund its pivotal phase 3 trial for vispa-cel, and the potential dilution to its stockholders resulting therefrom; risks associated with the initiation, cost, timing, progress, and results of current and future clinical trials, including risks associated with the manufacturing of its product candidates; the risk that initial, preliminary, or interim clinical trial data will not ultimately be predictive of the safety and efficacy of its CAR-T cell therapy product candidates or that clinical outcomes may differ as patient enrollment continues and as more clinical data becomes available or different conclusions or considerations are reached once additional data have been received and fully evaluated; risks related to its ability to obtain and maintain regulatory approval for its product candidates; risks of it not being ult imately able to commercialize its product candidates; risks that its product candidates, if approved, may not gain market acceptance due to negative public opinion and increased regulatory scrutiny of cell therapies involving genome editing; risks related to its ability to meet future regulatory standards with respect to its products; risks related to the substantial uncertainty regarding the current U.S. Administration’s initiatives and how these might impact the U.S. Food and Drug Administration (the “FDA”) and other government agencies in their implementation of laws, regulations, policies, and guidance; risks related to its ability to establish and/or maintain intellectual property rights covering its product candidates and genome-editing technology; risks of third parties asserting that its product candidates infringe their patents; risks related to developments of its competitors and its industry; risks related to its reliance on third parties to conduct its clinical trials and manufacture its product candidates; risks caused by public health crises or geopolitical events on its business and operations; risks related to the volatili ty of its stock price and its potential failure to meet the continuing listing requirements of Nasdaq; and other risks described in greater detail in its filings with the Securities and Exchange Commission (the “SEC”), including the section titled “Risk Factors” of its Annual Report on Form 10-K for the year ended December 31, 2024, and other filings the Company makes with the SEC, the events and circumstances reflected in its forward-looking statements may not be achieved or may not occur, and actual results could differ materially from those described in or implied by the forward-looking statements contained in this presentation. As a result of these risks, you should not place undue reliance on these forward-looking statements. The forward-looking statements in this presentation are made only as of the date hereof. Except to the extent required by law, the Company assumes no obligation and does not intend to update any of these forward-looking statements after the date of this presentation or to conform these statements to actual or revised expectations. Inherent limitations of comparisons with other immunotherapies Caution should be exercised when interpreting results from separate trials involving other immunotherapies. The clinical trial results of other immunotherapies presented or referenced in these slides have been derived from publicly available reports of clinical trials not conducted by the Company, and the Company has not performed any head-to-head trials comparing any of these other immunotherapies with vispa-cel or CB-011. As such, the results of these other clinical trials may not be comparable to clinical results for vispa-cel or CB-011 and may not accurately reflect the true relative efficacy and safety advantages of vispa-cel or CB-011 in comparison to the other immunotherapies presented. The designs of these other trials vary in material ways from the design of the clinical trial for vispa-cel or CB-011, including with respect to patient populations, follow-up times, the clinical trial phase, and subject characteristics. Most of the other trials presented or referenced in these slides have greater patient populations and patient cohorts and longer follow-up times. Accordingly, it is possible that when vispa-cel or CB-011 is evaluated in equally large patient populations over an equally long time period, their safety and efficacy benefits relative to other immunotherapies may be diminished or eliminated. As a result, cross-trial comparisons may have no interpretive value on vispa-cel or CB-011’s existing or future results. For further information and to understand these material differences, you should read the reports for the other immunotherapies’ clinical trials and the sources included in this presentation. This presentation discusses product candidates that have not yet been approved for marketing by the FDA. No representation is made as to the safety or effectiveness of these product candidates for the therapeutic uses for which they are being evaluated. From time to time, the Company may release additional data from its ANTLER phase 1 clinical trial and its CaMMouflage phase 1 clinical trial. The Company makes no representations regarding such additional clinical data or the timing of its release, or whether any such data will support or contradict the findings of any clinical data reported earlier. This presentation shall not constitute an offer to sell or the solicitation of an offer to buy any securities. 3 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. 1 Initial safety and efficacy data on the confirmatory cohort (20 patients) with partial HLA matching, with a minimum of six months of follow up for the majority of patients, as well as an update on the larger, maturing dataset presented previously. Two exciting clinical readouts to share with you today Opening remarks Rachel Haurwitz, PhD, President and CEO, Caribou Biosciences ANTLER Phase 1 trial data Mehdi Hamadani, MD, Medical College of Wisconsin Pivotal trial study design Tina Albertson, MD, PhD, CMO, Caribou Biosciences CaMMouflage Phase 1 dose escalation data Adriana Rossi, MD, Mount Sinai Discussion with clinicians Tina Albertson, MD, PhD, CMO, Caribou Biosciences Joseph McGuirk, DO, University of Kansas Cancer Center Adriana Rossi, MD, Mount Sinai Concluding remarks Q&A 4 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Caribou has the blueprint for allogeneic CAR-T cell therapies Armoring for functional persistence chRDNA for precision genome editing We leveraged our large clinical data set to identify key attributes Partial HLA matching Donor age 140 patients dosed Vispa-cel (CB-010) efficacy and durability on par with autologous CAR-Ts; safety unlocks outpatient use chRDNA: CRISPR hybrid RNA-DNA; HLA: human leukocyte antigen Vispa-cel CB-011 Anti-CD19 targeting Checkpoint disruption Anti-BCMA targeting Immune cloaking CB-011 drives deep, durable responses with best-in-class allo CAR-T potential for r/r multiple myeloma >

5 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Vispa-cel: delivering on the allogeneic CAR-T cell therapy promise 1Based on previously reported data from approved autologous CAR-T therapies; Caribou has not performed any comparative analysis directly with such therapies (see Important Information) 2Perales, M-A, et al. Poster 549, 2025 Tandem Meetings 3Mikhael, J. et al. JCO Oncology Practice 2022 18:12, 800-807 4Data on file 2L: second-line; CDMO: contract development and manufacturing organization; LBCL: large B cell lymphoma Autologous CAR-Ts1 1 dose per manufacturing batch Weeks to months for treatment3 Vispa-cel ~75% of 2L LBCL patients do not receive auto CAR-Ts2 A cc e ss S p e e d S ca le Sufficient yield for 200-300 doses per manufacturing batch Eligibility to treatment on the same day4 Many more patients could be served with off-the-shelf CAR-T cells M fg Multiple manufacturing plants One 500 ft2 suite at a CDMO Potential for 96% lower COGS than current autologous CAR-Ts 6 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Cohort criteria and breakdown of patient numbers 2L: second-line; 3L+: third line or later; HLA: human leukocyte antigen; LBCL: large B cell lymphoma; yo: years old Confirmatory cohort N=22 • CD19n • 2L LBCL • 80M dose level • 4+ HLA matching 7 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Cohort criteria and breakdown of patient numbers 2L: second-line; 3L+: third line or later; HLA: human leukocyte antigen; LBCL: large B cell lymphoma; yo: years old Confirmatory cohort N=22 • CD19n • 2L LBCL • 80M dose level • 4+ HLA matching Optimized cohort N=35 • CD19n • LBCL o 2L (N=32) o 3L+ (N=3) • 40M, 80M, 120M dose levels • 2+ HLA matching • Young donor, <30 yo 8 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Cohort criteria and breakdown of patient numbers 2L: second-line; 3L+: third line or later; HLA: human leukocyte antigen; LBCL: large B cell lymphoma; yo: years old Confirmatory cohort N=22 • CD19n • 2L LBCL • 80M dose level • 4+ HLA matching Optimized cohort N=35 • CD19n • LBCL o 2L (N=32) o 3L+ (N=3) • 40M, 80M, 120M dose levels • 2+ HLA matching • Young donor, <30 yo 20 pts from confirmatory cohort w/ 4+ HLA and young donor 2 pts old donor

9 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Cohort criteria and breakdown of patient numbers 2L: second-line; 3L+: third line or later; HLA: human leukocyte antigen; LBCL: large B cell lymphoma; yo: years old Confirmatory cohort N=22 • CD19n • 2L LBCL • 80M dose level • 4+ HLA matching Optimized cohort N=35 • CD19n • LBCL o 2L (N=32) o 3L+ (N=3) • 40M, 80M, 120M dose levels • 2+ HLA matching • Young donor, <30 yo 20 pts from confirmatory cohort w/ 4+ HLA and young donor 15 pts from dose escalation/expansion w/ 2+ HLA and young donor 2 pts old donor 10 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Vispa-cel drives deep, durable responses, demonstrating best-in-class allogeneic CAR-T cell therapy potential for r/r LBCL 86% ORR 53% 12-month PFS 63% CR rate No GvHD or Gr 3+ ICANS, <5% grade 3+ CRS, and manageable rates of infections and prolonged cytopenias4 Efficacy and durability on par with autologous CAR-T cells1 Potential best-in-class allogeneic CAR-T cell therapy for safety, efficacy, and durability with optimized vispa-cel3 Pivotal trial in 2L LBCL Expected trial design2: randomized, controlled trial in CD19-naïve, auto CAR-T- and transplant-ineligible patients; control arm to be treated with investigator choice of standard of care immunochemotherapy regimens 1Based on previously reported data from approved autologous CAR-T cell therapies; Caribou has not performed any comparative analysis directly with autologous CAR-T cell therapies (see Important Information) 2Pivotal study approach based on interactions with the FDA to date; the Company intends to further refine the pivotal trial design through continued engagement with the FDA prior to initiation of pivotal trial 3N=35; CD19 naïve, LBCL patients treated with 40M, 80M, or 120M vispa-cel CAR-T cells optimized for multiple factors, including 2+HLA matched and young donor 4Prolonged cytopenias are defined as Grade 3 or 4 neutropenia, thrombocytopenia, or anemia ongoing at day 28 (+/- 5 days) post CAR-T infusion, based on laboratory data, distinct from investigator-reported clinical adverse events. 2L: second-line; CR: complete response; CRS: cytokine release syndrome; GvHD: graft versus host disease; ICANS: immune effector cell-associated neurotoxicity syndrome; ORR: overall response rate; PFS: progression-free survival; r/r: relapsed or refractory Efficacy data cutoff 29Sept2025; safety data cutoff 02Sept2025 11 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Presenting ANTLER Phase 1 clinical trial data Disclosures Research support/funding: ADC Therapeutics, Spectrum Pharmaceuticals, Astellas Pharma Consultancy: Autolus, Forte Biosciences, Byondis, Kite, Daiichi Sankyo, BMS, Caribou Speaker’s bureau: AstraZeneca, ADC Therapeutics, BeiGene, Kite, Sobi DMC: Myeloid Therapeutics (2023), CRISPR (2024) Professor of medicine and section chief of hematologic malignancies and investigator for the ANTLER trial Medical College of Wisconsin Mehdi Hamadani, MD 12 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. 84 patients dosed with vispa-cel in ANTLER Phase 1 trial -9 to -3 DAYS DAY 0 28 DAYS 3 MONTHS 6 MONTHS 9 MONTHS 12 MONTHS Lymphodepletion Safety and tolerability Response assessment Cyclophosphamide (60 mg/kg/d for 2 days) followed by fludarabine (25 mg/m2/d for 5 days)3 SINGLE DOSE of vispa-cel NCT04637763 1B-NHL subtypes include: DLBCL (diffuse large B cell lymphoma ), HGBL (high-grade B cell lymphoma), tFL (transformed DLBCL from follicular lymphoma), PMBCL (primary mediastinal large B cell lymphoma), FL (follicular lymphoma, with POD24 (high risk)), MCL (mantle cell lymphoma), MZL (marginal zone lymphoma) 2LBCL subtypes include: DLBCL NOS (not otherwise specified), HGBL, transformed DLBCL from FL or MZL, and PMBCL 3Clin Cancer Res. 2011 July 1; 17(13): 4550–4557. doi:10.1158/1078-0432.CCR-11-0116 2L: second-line; B-NHL: B cell non-Hodgkin lymphoma: LBCL: large B cell lymphoma; r/r relapsed or refractory ANTLER trial design for all cohorts Eligibility • Dose escalation: aggressive r/r B-NHL1 with ≥2 prior lines of chemoimmunotherapy or primary refractory • Dose expansion: second-line LBCL2 Exclusion • Prior CD19-targeted therapy for CD19 naïve cohorts Part of trial Patient population N CD19 naïve Dose escalation r/r B-NHL 16 Yes Dose expansion 2L LBCL 41 Yes Confirmatory cohort 2L LBCL 4+ HLA match 22 Yes CD19 relapsed LBCL 5 No
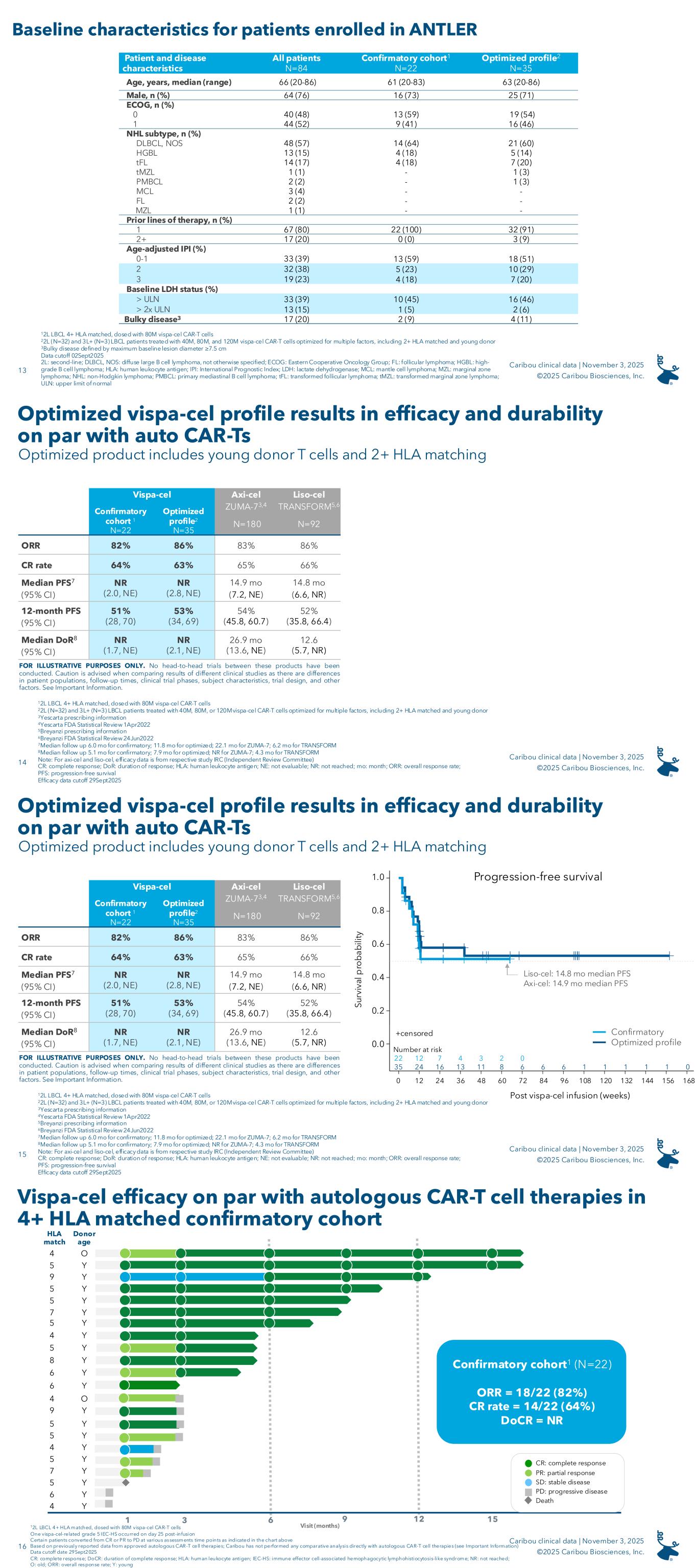
13 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Baseline characteristics for patients enrolled in ANTLER Patient and disease characteristics All patients N=84 Confirmatory cohort1 N=22 Optimized profile2 N=35 Age, years, median (range) 66 (20-86) 61 (20-83) 63 (20-86) Male, n (%) 64 (76) 16 (73) 25 (71) ECOG, n (%) 0 40 (48) 13 (59) 19 (54) 1 44 (52) 9 (41) 16 (46) NHL subtype, n (%) DLBCL, NOS 48 (57) 14 (64) 21 (60) HGBL 13 (15) 4 (18) 5 (14) tFL 14 (17) 4 (18) 7 (20) tMZL 1 (1) - 1 (3) PMBCL 2 (2) - 1 (3) MCL 3 (4) - - FL 2 (2) - - MZL 1 (1) - - Prior lines of therapy, n (%) 1 67 (80) 22 (100) 32 (91) 2+ 17 (20) 0 (0) 3 (9) Age-adjusted IPI (%) 0-1 33 (39) 13 (59) 18 (51) 2 32 (38) 5 (23) 10 (29) 3 19 (23) 4 (18) 7 (20) Baseline LDH status (%) > ULN 33 (39) 10 (45) 16 (46) > 2x ULN 13 (15) 1 (5) 2 (6) Bulky disease3 17 (20) 2 (9) 4 (11) 12L LBCL 4+ HLA matched, dosed with 80M vispa-cel CAR-T cells 22L (N=32) and 3L+ (N=3) LBCL patients treated with 40M, 80M, and 120M vispa-cel CAR-T cells optimized for multiple factors, including 2+ HLA matched and young donor 3Bulky disease defined by maximum baseline lesion diameter ≥7.5 cm Data cutoff 02Sept2025 2L: second-line; DLBCL, NOS: diffuse large B cell lymphoma, not otherwise specified; ECOG: Eastern Cooperative Oncology Group; FL: follicular lymphoma; HGBL: high- grade B cell lymphoma; HLA: human leukocyte antigen; IPI: International Prognostic Index; LDH: lactate dehydrogenase; MCL: mantle cell lymphoma; MZL: marginal zone lymphoma; NHL: non-Hodgkin lymphoma; PMBCL: primary mediastinal B cell lymphoma; tFL: transformed follicular lymphoma; tMZL: transformed marginal zone lymphoma; ULN: upper limit of normal 14 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Optimized vispa-cel profile results in efficacy and durability on par with auto CAR-Ts Optimized product includes young donor T cells and 2+ HLA matching Vispa-cel Axi-cel ZUMA-73,4 N=180 Liso-cel TRANSFORM5,6 N=92 Confirmatory cohort 1 N=22 Optimized profile2 N=35 ORR 82% 86% 83% 86% CR rate 64% 63% 65% 66% Median PFS7 (95% CI) NR (2.0, NE) NR (2.8, NE) 14.9 mo (7.2, NE) 14.8 mo (6.6, NR) 12-month PFS (95% CI) 51% (28, 70) 53% (34, 69) 54% (45.8, 60.7) 52% (35.8, 66.4) Median DoR8 (95% CI) NR (1.7, NE) NR (2.1, NE) 26.9 mo (13.6, NE) 12.6 (5.7, NR) FOR ILLUSTRATIVE PURPOSES ONLY. No head-to-head trials between these products have been conducted. Caution is advised when comparing results of different clinical studies as there are differences in patient populations, follow-up times, clinical trial phases, subject characteristics, trial design, and other factors. See Important Information. 12L LBCL 4+ HLA matched, dosed with 80M vispa-cel CAR-T cells 22L (N=32) and 3L+ (N=3) LBCL patients treated with 40M, 80M, or 120M vispa-cel CAR-T cells optimized for multiple factors, including 2+ HLA matched and young donor 3Yescarta prescribing information 4Yescarta FDA Statistical Review 1Apr2022 5Breyanzi prescribing information 6Breyanzi FDA Statistical Review 24Jun2022 7Median follow up 6.0 mo for confirmatory; 11.8 mo for optimized; 22.1 mo for ZUMA-7; 6.2 mo for TRANSFORM 8Median follow up 5.1 mo for confirmatory; 7.9 mo for optimized; NR for ZUMA-7; 4.3 mo for TRANSFORM Note: For axi-cel and liso-cel, efficacy data is from respective study IRC (Independent Review Committee) CR: complete response; DoR: duration of response; HLA: human leukocyte antigen; NE: not evaluable; NR: not reached; mo: month; ORR: overall response rate; PFS: progression-free survival Efficacy data cutoff 29Sept2025 15 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Optimized vispa-cel profile results in efficacy and durability on par with auto CAR-Ts Optimized product includes young donor T cells and 2+ HLA matching Confirmatory Optimized profile 1.0 0.8 0.6 0.4 0.2 0.0 +censored Number at risk 22 12 7 4 3 2 0 35 24 16 13 11 8 6 6 6 1 1 1 1 1 0 S u rv iv a l p ro b a b il it y 0 12 24 36 48 60 72 84 96 108 120 132 144 156 168 Progression-free survival Post vispa-cel infusion (weeks) Liso-cel: 14.8 mo median PFS Axi-cel: 14.9 mo median PFS FOR ILLUSTRATIVE PURPOSES ONLY. No head-to-head trials between these products have been conducted. Caution is advised when comparing results of different clinical studies as there are differences in patient populations, follow-up times, clinical trial phases, subject characteristics, trial design, and other factors. See Important Information. Vispa-cel Axi-cel ZUMA-73,4 N=180 Liso-cel TRANSFORM5,6 N=92 Confirmatory cohort 1 N=22 Optimized profile2 N=35 ORR 82% 86% 83% 86% CR rate 64% 63% 65% 66% Median PFS7 (95% CI) NR (2.0, NE) NR (2.8, NE) 14.9 mo (7.2, NE) 14.8 mo (6.6, NR) 12-month PFS (95% CI) 51% (28, 70) 53% (34, 69) 54% (45.8, 60.7) 52% (35.8, 66.4) Median DoR8 (95% CI) NR (1.7, NE) NR (2.1, NE) 26.9 mo (13.6, NE) 12.6 (5.7, NR) 12L LBCL 4+ HLA matched, dosed with 80M vispa-cel CAR-T cells 22L (N=32) and 3L+ (N=3) LBCL patients treated with 40M, 80M, or 120M vispa-cel CAR-T cells optimized for multiple factors, including 2+ HLA matched and young donor 3Yescarta prescribing information 4Yescarta FDA Statistical Review 1Apr2022 5Breyanzi prescribing information 6Breyanzi FDA Statistical Review 24Jun2022 7Median follow up 6.0 mo for confirmatory; 11.8 mo for optimized; 22.1 mo for ZUMA-7; 6.2 mo for TRANSFORM 8Median follow up 5.1 mo for confirmatory; 7.9 mo for optimized; NR for ZUMA-7; 4.3 mo for TRANSFORM Note: For axi-cel and liso-cel, efficacy data is from respective study IRC (Independent Review Committee) CR: complete response; DoR: duration of response; HLA: human leukocyte antigen; NE: not evaluable; NR: not reached; mo: month; ORR: overall response rate; PFS: progression-free survival Efficacy data cutoff 29Sept2025 16 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Vispa-cel efficacy on par with autologous CAR-T cell therapies in 4+ HLA matched confirmatory cohort 1 3 6 9 12 15 Confirmatory cohort1 (N=22) ORR = 18/22 (82%) CR rate = 14/22 (64%) DoCR = NR HLA match Donor age 4 O 5 Y 9 Y 5 Y 5 Y 7 Y 5 Y 4 Y 5 Y 8 Y 6 Y 6 Y 4 O 9 Y 5 Y 5 Y 4 Y 5 Y 7 Y 5 Y 6 Y 4 Y CR: complete response PR: partial response SD: stable disease PD: progressive disease Death 12L LBCL 4+ HLA matched, dosed with 80M vispa-cel CAR-T cells One vispa-cel-related grade 5 IEC-HS occurred on day 25 post-infusion Certain patients converted from CR or PR to PD at various assessments time points as indicated in the chart above Based on previously reported data from approved autologous CAR -T cell therapies; Caribou has not performed any comparative analysis directly with autologous CAR-T cell therapies (see Important Information) Data cutoff date 29Sept2025 CR: complete response; DoCR: duration of complete response; HLA: human leukocyte antigen; IEC-HS: immune effector cell-associated hemophagocytic lymphohistiocytosis-like syndrome; NR: not reached; O: old; ORR: overall response rate; Y: young Visit (months)
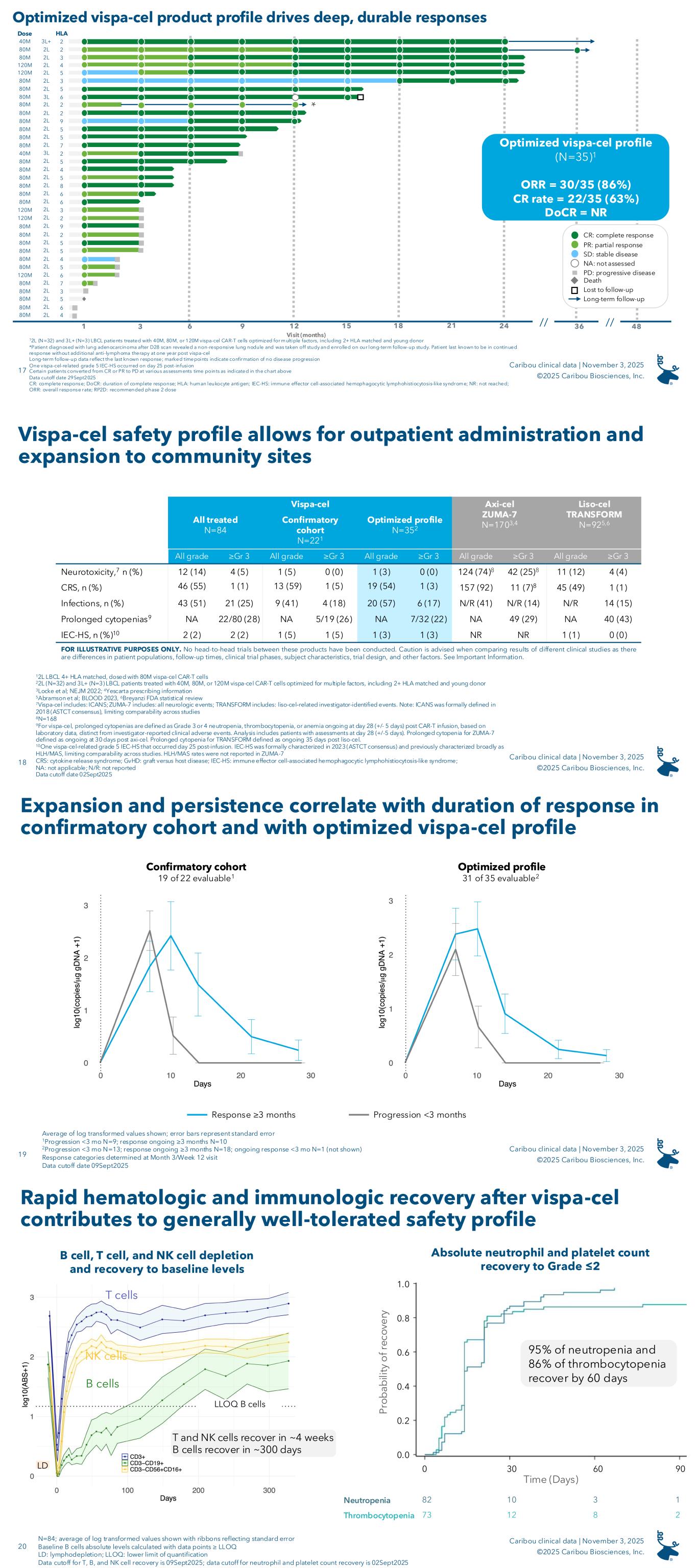
17 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Optimized vispa-cel product profile drives deep, durable responses 1 6 12 18 21 243 9 15 Optimized vispa-cel profile (N=35)1 ORR = 30/35 (86%) CR rate = 22/35 (63%) DoCR = NR Dose HLA 40M 3L+ 2 80M 2L 2 80M 2L 3 120M 2L 4 120M 2L 5 80M 2L 3 80M 2L 5 80M 3L 6 80M 2L 2 80M 2L 2 80M 2L 9 80M 2L 5 80M 2L 5 80M 2L 7 40M 3L 2 80M 2L 5 80M 2L 4 80M 2L 5 80M 2L 8 80M 2L 6 80M 2L 6 120M 2L 3 120M 2L 2 80M 2L 9 80M 2L 2 80M 2L 5 80M 2L 5 80M 2L 4 80M 2L 5 120M 2L 6 80M 2L 7 80M 2L 3 80M 2L 5 80M 2L 6 80M 2L 4 CR: complete response PR: partial response SD: stable disease NA: not assessed PD: progressive disease Death Lost to follow-up Long-term follow-up // 36 48// Visit (months) 12L (N=32) and 3L+ (N=3) LBCL patients treated with 40M, 80M, or 120M vispa-cel CAR-T cells optimized for multiple factors, including 2+ HLA matched and young donor *Patient diagnosed with lung adenocarcinoma after D28 scan revealed a non-responsive lung nodule and was taken off study and enrolled on our long-term follow-up study. Patient last known to be in continued response without additional anti-lymphoma therapy at one year post vispa-cel Long-term follow-up data reflect the last known response; marked timepoints indicate confirmation of no disease progression One vispa-cel-related grade 5 IEC-HS occurred on day 25 post-infusion Certain patients converted from CR or PR to PD at various assessments time points as indicated in the chart above Data cutoff date 29Sept2025 CR: complete response; DoCR: duration of complete response; HLA: human leukocyte antigen; IEC-HS: immune effector cell-associated hemophagocytic lymphohistiocytosis-like syndrome; NR: not reached; ORR: overall response rate; RP2D: recommended phase 2 dose 18 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Vispa-cel safety profile allows for outpatient administration and expansion to community sites Vispa-cel Axi-cel ZUMA-7 N=1703,4 Liso-cel TRANSFORM N=925,6All treated N=84 Confirmatory cohort N=221 Optimized profile N=352 All grade ≥Gr 3 All grade ≥Gr 3 All grade ≥Gr 3 All grade ≥Gr 3 All grade ≥Gr 3 Neurotoxicity,7 n (%) 12 (14) 4 (5) 1 (5) 0 (0) 1 (3) 0 (0) 124 (74)8 42 (25)8 11 (12) 4 (4) CRS, n (%) 46 (55) 1 (1) 13 (59) 1 (5) 19 (54) 1 (3) 157 (92) 11 (7)8 45 (49) 1 (1) Infections, n (%) 43 (51) 21 (25) 9 (41) 4 (18) 20 (57) 6 (17) N/R (41) N/R (14) N/R 14 (15) Prolonged cytopenias9 NA 22/80 (28) NA 5/19 (26) NA 7/32 (22) NA 49 (29) NA 40 (43) IEC-HS, n (%)10 2 (2) 2 (2) 1 (5) 1 (5) 1 (3) 1 (3) NR NR 1 (1) 0 (0) FOR ILLUSTRATIVE PURPOSES ONLY. No head-to-head trials between these products have been conducted. Caution is advised when comparing results of different clinical studies as there are differences in patient populations, follow-up times, clinical trial phases, subject characteristics, trial design, and other factors. See Important Information. 12L LBCL 4+ HLA matched, dosed with 80M vispa-cel CAR-T cells 22L (N=32) and 3L+ (N=3) LBCL patients treated with 40M, 80M, or 120M vispa-cel CAR-T cells optimized for multiple factors, including 2+ HLA matched and young donor 3Locke et al; NEJM 2022; 4Yescarta prescribing information 5Abramson et al; BLOOD 2023, 6Breyanzi FDA statistical review 7Vispa-cel includes: ICANS; ZUMA-7 includes: all neurologic events; TRANSFORM includes: liso-cel–related investigator-identified events. Note: ICANS was formally defined in 2018 (ASTCT consensus), limiting comparability across studies 8N=168 9For vispa-cel, prolonged cytopenias are defined as Grade 3 or 4 neutropenia, thrombocytopenia, or anemia ongoing at day 28 (+/- 5 days) post CAR-T infusion, based on laboratory data, distinct from investigator-reported clinical adverse events. Analysis includes patients with assessments at day 28 (+/-5 days). Prolonged cytopenia for ZUMA-7 defined as ongoing at 30 days post axi-cel. Prolonged cytopenia for TRANSFORM defined as ongoing 35 days post liso-cel. 10One vispa-cel-related grade 5 IEC-HS that occurred day 25 post-infusion. IEC-HS was formally characterized in 2023 (ASTCT consensus) and previously characterized broadly as HLH/MAS, limiting comparability across studies. HLH/MAS rates were not reported in ZUMA-7 CRS: cytokine release syndrome; GvHD: graft versus host disease; IEC-HS: immune effector cell-associated hemophagocytic lymphohistiocytosis-like syndrome; NA: not applicable; N/R: not reported Data cutoff date 02Sept2025 19 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Expansion and persistence correlate with duration of response in confirmatory cohort and with optimized vispa-cel profile Optimized profile 31 of 35 evaluable2 Confirmatory cohort 19 of 22 evaluable1 Response ≥3 months Progression <3 months Average of log transformed values shown; error bars represent standard error 1Progression <3 mo N=9; response ongoing ≥3 months N=10 2Progression <3 mo N=13; response ongoing ≥3 months N=18; ongoing response <3 mo N=1 (not shown) Response categories determined at Month 3/Week 12 visit Data cutoff date 09Sept2025 20 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Rapid hematologic and immunologic recovery after vispa-cel contributes to generally well-tolerated safety profile B cell, T cell, and NK cell depletion and recovery to baseline levels LLOQ B cells LD T cells NK cells B cells Absolute neutrophil and platelet count recovery to Grade ≤2 N=84; average of log transformed values shown with ribbons reflecting standard error Baseline B cells absolute levels calculated with data points ≥ LLOQ LD: lymphodepletion; LLOQ: lower limit of quantification Data cutoff for T, B, and NK cell recovery is 09Sept2025; data cutoff for neutrophil and platelet count recovery is 02Sept2025 T and NK cells recover in ~4 weeks B cells recover in ~300 days 95% of neutropenia and 86% of thrombocytopenia recover by 60 days Pr ob ab ili ty o f r ec ov er y Time (Days) 0 30 60 90 Neutropenia 82 10 3 1 Thrombocytopenia 73 12 8 2 1.0 0.8 0.6 0.4 0.2 0.0
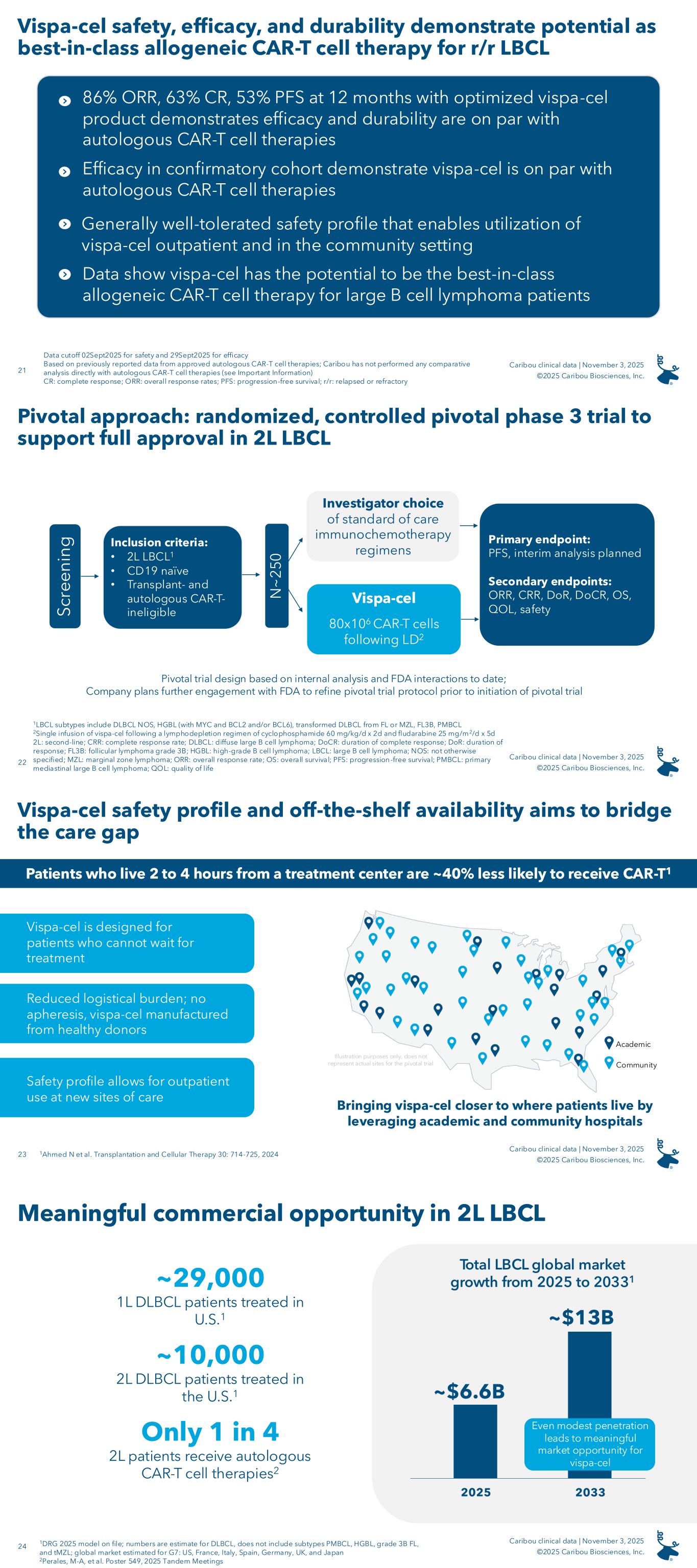
21 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Vispa-cel safety, efficacy, and durability demonstrate potential as best-in-class allogeneic CAR-T cell therapy for r/r LBCL Data cutoff 02Sept2025 for safety and 29Sept2025 for efficacy Based on previously reported data from approved autologous CAR-T cell therapies; Caribou has not performed any comparative analysis directly with autologous CAR-T cell therapies (see Important Information) CR: complete response; ORR: overall response rates; PFS: progression-free survival; r/r: relapsed or refractory Generally well-tolerated safety profile that enables utilization of vispa-cel outpatient and in the community setting Data show vispa-cel has the potential to be the best-in-class allogeneic CAR-T cell therapy for large B cell lymphoma patients Efficacy in confirmatory cohort demonstrate vispa-cel is on par with autologous CAR-T cell therapies 86% ORR, 63% CR, 53% PFS at 12 months with optimized vispa-cel product demonstrates efficacy and durability are on par with autologous CAR-T cell therapies 22 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Pivotal approach: randomized, controlled pivotal phase 3 trial to support full approval in 2L LBCL 1LBCL subtypes include DLBCL NOS, HGBL (with MYC and BCL2 and/or BCL6), transformed DLBCL from FL or MZL, FL3B, PMBCL 2Single infusion of vispa-cel following a lymphodepletion regimen of cyclophosphamide 60 mg/kg/d x 2d and fludarabine 25 mg/m2/d x 5d 2L: second-line; CRR: complete response rate; DLBCL: diffuse large B cell lymphoma; DoCR: duration of complete response; DoR: duration of response; FL3B: follicular lymphoma grade 3B; HGBL: high-grade B cell lymphoma; LBCL: large B cell lymphoma; NOS: not otherwise specified; MZL: marginal zone lymphoma; ORR: overall response rate; OS: overall survival; PFS: progression -free survival; PMBCL: primary mediastinal large B cell lymphoma; QOL: quality of life Vispa-cel Inclusion criteria: • 2L LBCL1 • CD19 naïve • Transplant- and autologous CAR-T- ineligibleSc re en in g Primary endpoint: PFS, interim analysis planned Secondary endpoints: ORR, CRR, DoR, DoCR, OS, QOL, safety 80x106 CAR-T cells following LD2 Investigator choice of standard of care immunochemotherapy regimens N ~2 50 Pivotal trial design based on internal analysis and FDA interactions to date; Company plans further engagement with FDA to refine pivotal trial protocol prior to initiation of pivotal trial 23 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Vispa-cel safety profile and off-the-shelf availability aims to bridge the care gap Vispa-cel is designed for patients who cannot wait for treatment Safety profile allows for outpatient use at new sites of care Patients who live 2 to 4 hours from a treatment center are ~40% less likely to receive CAR-T1 1Ahmed N et al. Transplantation and Cellular Therapy 30: 714-725, 2024 Reduced logistical burden; no apheresis, vispa-cel manufactured from healthy donors Bringing vispa-cel closer to where patients live by leveraging academic and community hospitals Illustration purposes only, does not represent actual sites for the pivotal trial Academic Community 24 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Meaningful commercial opportunity in 2L LBCL ~29,000 1L DLBCL patients treated in U.S.1 Only 1 in 4 2L patients receive autologous CAR-T cell therapies2 ~10,000 2L DLBCL patients treated in the U.S.1 ~$6.6B ~$13B Total LBCL global market growth from 2025 to 20331 1DRG 2025 model on file; numbers are estimate for DLBCL, does not include subtypes PMBCL, HGBL, grade 3B FL, and tMZL; global market estimated for G7: US, France, Italy, Spain, Germany, UK, and Japan 2Perales, M-A, et al. Poster 549, 2025 Tandem Meetings Even modest penetration leads to meaningful market opportunity for vispa-cel 2025 2033

25 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Vispa-cel’s commercial-ready manufacturing enables orders of magnitude lower investment than auto CAR-Ts Potential for 96% lower COGS than current autologous CAR-T cell therapies COGS: cost of goods sold Small footprint at CDMO Easy and fast to expand Single 500 ft2 suite = 9,000 doses/yr Commercial-ready outputs Projected yield for 200-300 doses per batch Efficiency and flexibility Suite usable for any Caribou product On-demand starting materials 26 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Vispa-cel drives deep, durable responses, demonstrating best-in-class allogeneic CAR-T cell therapy potential for r/r LBCL 86% ORR 53% 12-month PFS 63% CR rate No GvHD or Gr 3+ ICANS, <5% grade 3+ CRS, and manageable rates of infections and prolonged cytopenias4 Efficacy and durability on par with autologous CAR-T cells1 Potential best-in-class allogeneic CAR-T cell therapy for safety, efficacy, and durability with optimized vispa-cel3 Pivotal trial in 2L LBCL Expected trial design2: randomized, controlled trial in CD19-naïve, auto CAR-T- and transplant-ineligible patients; control arm to be treated with investigator choice of standard of care immunochemotherapy regimens 1Based on previously reported data from approved autologous CAR-T cell therapies; Caribou has not performed any comparative analysis directly with autologous CAR-T cell therapies (see Important Information) 2Pivotal study approach based on interactions with the FDA to date; the Company intends to further refine the pivotal trial design through continued engagement with the FDA prior to initiation of pivotal trial 3N=35; CD19 naïve, LBCL patients treated with 40M, 80M, or 120M vispa-cel CAR-T cells optimized for multiple factors, including 2+HLA matched and young donor 4Prolonged cytopenias are defined as Grade 3 or 4 neutropenia, thrombocytopenia, or anemia ongoing at day 28 (+/- 5 days) post CAR-T infusion, based on laboratory data, distinct from investigator-reported clinical adverse events. 2L: second-line; CR: complete response; CRS: cytokine release syndrome; GvHD: graft versus host disease; ICANS: immune effector cell-associated neurotoxicity syndrome; ORR: overall response rate; PFS: progression-free survival; r/r: relapsed or refractory Efficacy data cutoff 29Sept2025; safety data cutoff 02Sept2025 27 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. CB-011 Allogeneic anti-BCMA CAR-T cell therapy for r/r multiple myeloma (MM) 28 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. CB-011: delivering on the allogeneic CAR-T cell therapy promise with high response rates and low rates of infection for patients with r/r multiple myeloma Bispecifics CB-011 Tr e a tm e n t b u rd e n E ff ic a c y In fe ct io n High response rates2 with single dose High rates with limited or no B cell recovery1 Repeat dosing until relapse Single-dose treatment Weekly or bi-weekly treatment required for durability Low rates of infection2 and rapid immune recovery 1Frerichs, KA, et al. Blood Adv. 2024 Jan 9;8(1):194-206; Jelinek T, et al. Blood 144 (2024) 1934-1936; Schreiber S, et al. Mol. Therapy 3 (9) 4130-4134; 2025 2Based on Grade 3+ infections, at recommended dose for expansion as seen in results from dose escalation portion of the CaMMouflage clinical trial Based on previously reported data from approved bispecific therapies; Caribou has not performed any comparative analysis directly with bispecifics (see Important Information) r/r: relapsed or refractory

29 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. CB-011: delivering on the allogeneic CAR-T cell therapy promise with broad access, rapid treatment, and scalability for r/r multiple myeloma 1Frerichs, KA, et al. Blood Adv. 2024 Jan 9;8(1):194-206; Jelinek T, et al. Blood 144 (2024) 1934-1936; Schreiber S, et al. Mol. Therapy 3 (9) 4130-4134; 2025 2Based on Grade 3+ infections, at recommended dose for expansion as seen in results from dose escalation portion of the CaMMouflage clinical trial 3Gilead Q3 2024 earnings call transcript; Poseida Therapeutics International Myeloma Society Meeting data call 2024 4Kourelis, T. et al. Transplant Cell Ther 2023 29(4):255-258 Based on previously reported data from approved bispecific and autologous CAR-T therapies; Caribou has not performed any comparative analysis directly with such therapies (see Important Information) MM: multiple myeloma; r/r: relapsed or refractory Bispecifics CB-011 1 dose per manufacturing batch Weeks to months from eligibility to lymphodepletion4 1 of 10 MM patients receive auto CAR-Ts3 E ff ic a c y In fe ct io n Potential for 50-100 doses per manufacturing batch at commercial launch No wait needed between eligibility and lymphodepletion Overcomes access challenges to treat more patients A cc e ss S p e e d S ca le Autologous CAR-T High response rates2 with single dose High rates with limited or no B cell recovery1 Repeat dosing until relapse Single-dose treatment Weekly or bi-weekly treatment required for durability Tr e a tm e n t b u rd e n Low rates of infection2 and rapid immune recovery 30 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. CB-011 drives deep, durable responses, potentially best-in-class allogeneic CAR-T cell therapy for patients with r/r MM CB-011 significantly outperforms bispecific response rates and safety1 1Based on previously reported data from approved bispecific therapies; Caribou has not performed any comparative analysis directly with bispecifics (see Important Information) 2RDE is 450x106 CAR-T cells with selected LD regimen of 500 mg/m2 cy and 30 mg/m2 flu daily x 3 days; N=12 BCMA-naïve patients treated with RDE CR: complete response; GvHD: graft versus host disease; IEC-EC: immune effector cell-associated enterocolitis; MM: multiple myeloma; MRD: minimal residual disease; neg: negative; ORR: overall response rate; PFS: progression-free survival; r/r: relapsed or refractory Data cutoff 24Sept2025 92% ORR (11/12) 91% MRD neg (10/11 evaluable patients) 75% ≥CR rate (9/12) 7 of 12 ≥VGPR at ≥6 months Manageable safety profile with no GvHD, IEC-EC, parkinsonism, or cranial nerve palsies observed at any dose level Recommended dose for expansion (RDE): 450x106 CAR-T cells2 Potentially best-in-class allogeneic CAR-T cell therapy Efficacy in BCMA-naïve patients at the RDE2: 31 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Presenting CaMMouflage dose escalation data Disclosures – consulting or advisor role: Caribou, J&J, BMS, Karyopharm, and Sanofi Director of CAR-T and stem cell transplant clinical program at the center of excellence for multiple myeloma, and investigator for the CaMMouflage trial Mount Sinai Adriana Rossi, MD 32 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. 48 patients dosed with CB-011 in dose escalation for the CaMMouflage Phase 1 trial NCT05722418 1300 mg/m2 cy and 30 mg/m2 flu daily x 3 days 2500 mg/m2 cy and 30 mg/m2 flu daily x 3 days Data cutoff 24Sept2025 cy: cyclophosphamide; flu: fludarabine; HLA: human leukocyte antigen; IMiD: immunomodulatory drug; LD: lymphodepletion; M: million; MM: multiple myeloma; mAb: monoclonal antibody; MTD: maximum tolerated dose; PI: proteasome inhibitor; r/r: relapse or refractory; RP2D: recommended phase 2 dose; RDE: recommended dose for expansion Patients with r/r MM • ≥3 prior lines of therapy, including a PI, an IMiD, and an anti- CD38 antibody • Exclusions: prior CAR-T cell therapy and/or BCMA-targeted therapy within last 3 months Part A: 3+3 dose escalation • Objective: safety, determine MTD, RDE Part B: dose expansion • Objective: antitumor response, RP2D -5 to -3 DAYS DAY 0 28 DAYS 3 MONTHS 6 MONTHS 9 MONTHS 12 MONTHS Lymphodepletion1,2 Safety and tolerability Response assessment SINGLE DOSE of CB-011 Dose levels evaluated at 300 cy LD regimen1 50x106 CAR-T cells 150x106 CAR-T cells 450x106 CAR-T cells Dose levels evaluated at 500 cy LD regimen2 150x106 CAR-T cells 300x106 CAR-T cells 450x106 CAR-T cells 800x106 CAR-T cells Best HLA matching for class II implemented for 500 cy cohort
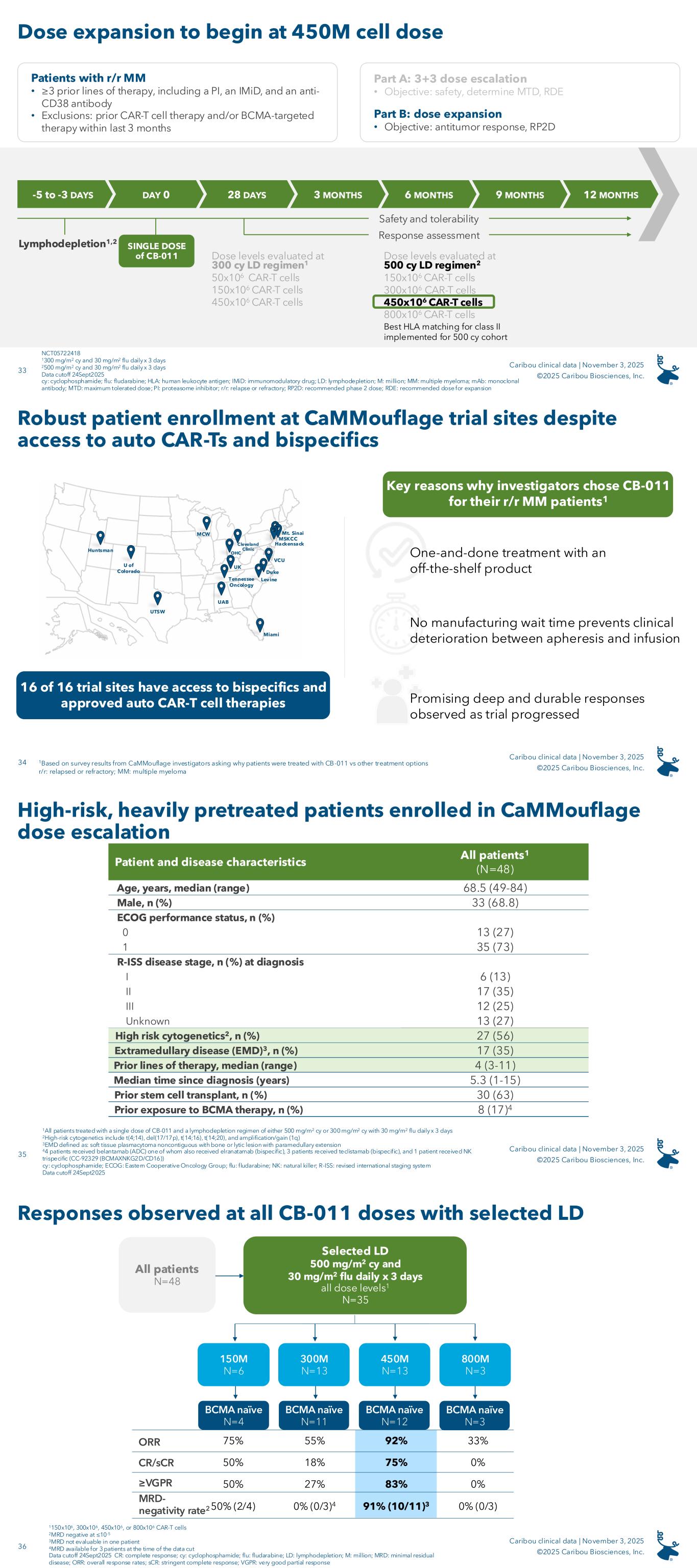
33 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. -5 to -3 DAYS DAY 0 28 DAYS 3 MONTHS 6 MONTHS 9 MONTHS 12 MONTHS Lymphodepletion1,2 Safety and tolerability Response assessment SINGLE DOSE of CB-011 Dose expansion to begin at 450M cell dose Patients with r/r MM • ≥3 prior lines of therapy, including a PI, an IMiD, and an anti- CD38 antibody • Exclusions: prior CAR-T cell therapy and/or BCMA-targeted therapy within last 3 months Part A: 3+3 dose escalation • Objective: safety, determine MTD, RDE Part B: dose expansion • Objective: antitumor response, RP2D Dose levels evaluated at 300 cy LD regimen1 50x106 CAR-T cells 150x106 CAR-T cells 450x106 CAR-T cells Dose levels evaluated at 500 cy LD regimen2 150x106 CAR-T cells 300x106 CAR-T cells 450x106 CAR-T cells 800x106 CAR-T cells Best HLA matching for class II implemented for 500 cy cohort NCT05722418 1300 mg/m2 cy and 30 mg/m2 flu daily x 3 days 2500 mg/m2 cy and 30 mg/m2 flu daily x 3 days Data cutoff 24Sept2025 cy: cyclophosphamide; flu: fludarabine; HLA: human leukocyte antigen; IMiD: immunomodulatory drug; LD: lymphodepletion; M: million; MM: multiple myeloma; mAb: monoclonal antibody; MTD: maximum tolerated dose; PI: proteasome inhibitor; r/r: relapse or refractory; RP2D: recommended phase 2 dose; RDE: recommended dose for expansion 34 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Robust patient enrollment at CaMMouflage trial sites despite access to auto CAR-Ts and bispecifics 1Based on survey results from CaMMouflage investigators asking why patients were treated with CB-011 vs other treatment options r/r: relapsed or refractory; MM: multiple myeloma Key reasons why investigators chose CB-011 for their r/r MM patients1 One-and-done treatment with an off-the-shelf product No manufacturing wait time prevents clinical deterioration between apheresis and infusion Promising deep and durable responses observed as trial progressed LevineTennessee Oncology MCW Huntsman VCU UAB OHC Mt. Sinai Miami MSKCC Cleveland Clinic Hackensack UTSW U of Colorado Duke UK 16 of 16 trial sites have access to bispecifics and approved auto CAR-T cell therapies 35 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. High-risk, heavily pretreated patients enrolled in CaMMouflage dose escalation 1All patients treated with a single dose of CB-011 and a lymphodepletion regimen of either 500 mg/m2 cy or 300 mg/m2 cy with 30 mg/m2 flu daily x 3 days 2High-risk cytogenetics include t(4;14), del(17/17p), t(14;16), t(14;20), and amplification/gain (1q) 3EMD defined as: soft tissue plasmacytoma noncontiguous with bone or lytic lesion with paramedullary extension 44 patients received belantamab (ADC) one of whom also received elranatamab (bispecific), 3 patients received teclistamab (bispecific), and 1 patient received NK trispecific (CC-92329 (BCMAXNKG2D/CD16)) cy: cyclophosphamide; ECOG: Eastern Cooperative Oncology Group; flu: fludarabine; NK: natural killer; R-ISS: revised international staging system Data cutoff 24Sept2025 Patient and disease characteristics All patients1 (N=48) Age, years, median (range) 68.5 (49-84) Male, n (%) 33 (68.8) ECOG performance status, n (%) 0 13 (27) 1 35 (73) R-ISS disease stage, n (%) at diagnosis I 6 (13) II 17 (35) III 12 (25) Unknown 13 (27) High risk cytogenetics2, n (%) 27 (56) Extramedullary disease (EMD)3, n (%) 17 (35) Prior lines of therapy, median (range) 4 (3-11) Median time since diagnosis (years) 5.3 (1-15) Prior stem cell transplant, n (%) 30 (63) Prior exposure to BCMA therapy, n (%) 8 (17)4 36 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Responses observed at all CB-011 doses with selected LD Selected LD 500 mg/m2 cy and 30 mg/m2 flu daily x 3 days all dose levels1 N=35 150M N=6 300M N=13 450M N=13 800M N=3 BCMA naïve N=4 BCMA naïve N=11 BCMA naïve N=12 BCMA naïve N=3 75% 55% 92% 33% 50% 18% 75% 0% 50% 27% 83% 0% 50% (2/4) 0% (0/3)4 91% (10/11)3 0% (0/3) 1150x106, 300x106, 450x106, or 800x106 CAR-T cells 2MRD negative at ≤10-5 3MRD not evaluable in one patient 4MRD available for 3 patients at the time of the data cut Data cutoff 24Sept2025 CR: complete response; cy: cyclophosphamide; flu: fludarabine; LD: lymphodepletion; M: million; MRD: minimal residual disease; ORR: overall response rates; sCR: stringent complete response; VGPR: very good partial response ORR CR/sCR ≥VGPR MRD- negativity rate2 All patients N=48
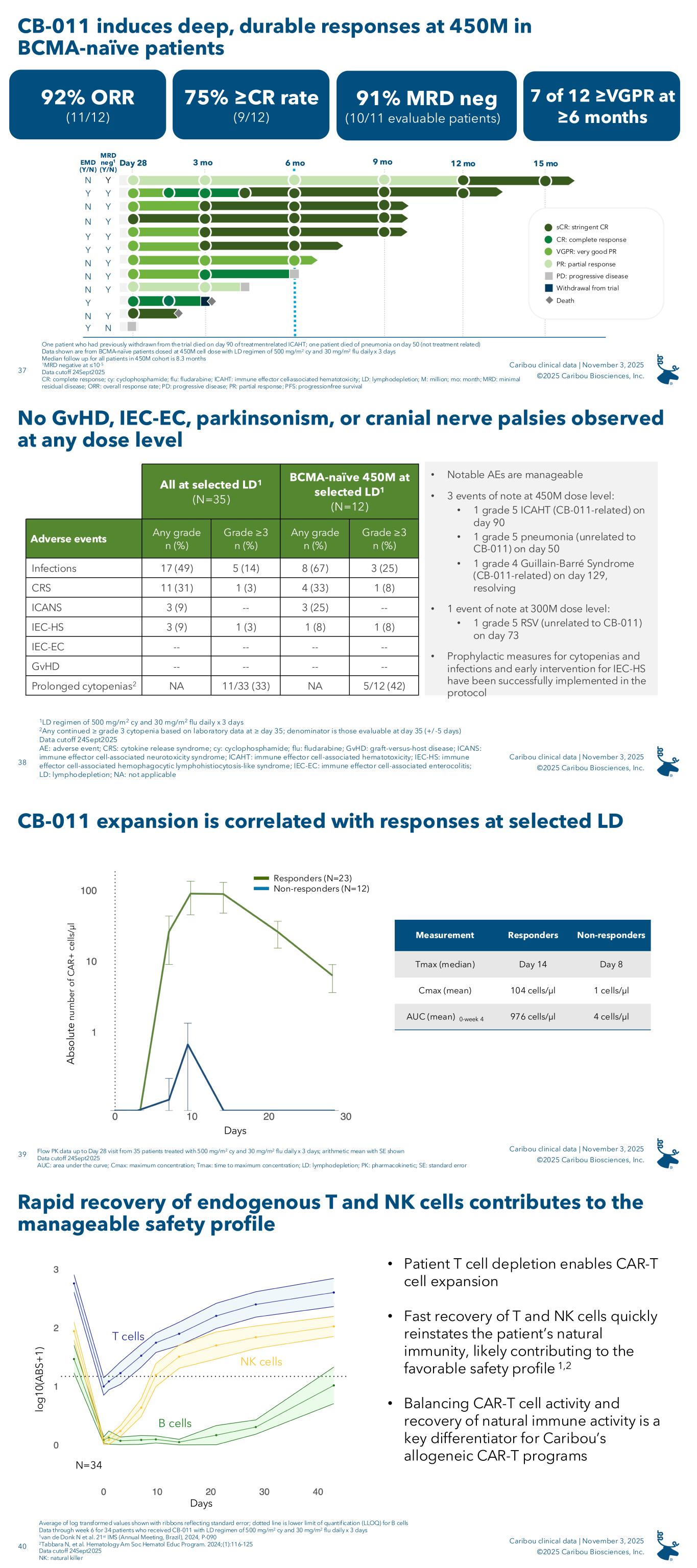
37 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. EMD (Y/N) MRD neg1 (Y/N) N Y Y Y N Y N Y Y Y Y Y N Y N Y N Y Y N Y Y N 12 mo9 mo6 mo3 moDay 28 One patient who had previously withdrawn from the trial died on day 90 of treatment-related ICAHT; one patient died of pneumonia on day 50 (not treatment related) Data shown are from BCMA-naïve patients dosed at 450M cell dose with LD regimen of 500 mg/m2 cy and 30 mg/m2 flu daily x 3 days Median follow up for all patients in 450M cohort is 8.3 months 1MRD negative at ≤10-5 Data cutoff 24Sept2025 CR: complete response; cy: cyclophosphamide; flu: fludarabine; ICAHT: immune effector cell-associated hematotoxicity; LD: lymphodepletion; M: million; mo: month; MRD: minimal residual disease; ORR: overall response rate; PD: progressive disease; PR: partial response; PFS: progression-free survival 92% ORR (11/12) sCR: stringent CR CR: complete response VGPR: very good PR PR: partial response PD: progressive disease Withdrawal from trial Death 91% MRD neg (10/11 evaluable patients) 75% ≥CR rate (9/12) 15 mo 7 of 12 ≥VGPR at ≥6 months CB-011 induces deep, durable responses at 450M in BCMA-naïve patients 38 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. All at selected LD1 (N=35) BCMA-naïve 450M at selected LD1 (N=12) Adverse events Any grade n (%) Grade ≥3 n (%) Any grade n (%) Grade ≥3 n (%) Infections 17 (49) 5 (14) 8 (67) 3 (25) CRS 11 (31) 1 (3) 4 (33) 1 (8) ICANS 3 (9) -- 3 (25) -- IEC-HS 3 (9) 1 (3) 1 (8) 1 (8) IEC-EC -- -- -- -- GvHD -- -- -- -- Prolonged cytopenias2 NA 11/33 (33) NA 5/12 (42) 1LD regimen of 500 mg/m2 cy and 30 mg/m2 flu daily x 3 days 2Any continued ≥ grade 3 cytopenia based on laboratory data at ≥ day 35; denominator is those evaluable at day 35 (+/ -5 days) Data cutoff 24Sept2025 AE: adverse event; CRS: cytokine release syndrome; cy: cyclophosphamide; flu: fludarabine; GvHD: graft-versus-host disease; ICANS: immune effector cell-associated neurotoxicity syndrome; ICAHT: immune effector cell-associated hematotoxicity; IEC-HS: immune effector cell-associated hemophagocytic lymphohistiocytosis-like syndrome; IEC-EC: immune effector cell-associated enterocolitis; LD: lymphodepletion; NA: not applicable • Notable AEs are manageable • 3 events of note at 450M dose level: • 1 grade 5 ICAHT (CB-011-related) on day 90 • 1 grade 5 pneumonia (unrelated to CB-011) on day 50 • 1 grade 4 Guillain-Barré Syndrome (CB-011-related) on day 129, resolving • 1 event of note at 300M dose level: • 1 grade 5 RSV (unrelated to CB-011) on day 73 • Prophylactic measures for cytopenias and infections and early intervention for IEC-HS have been successfully implemented in the protocol No GvHD, IEC-EC, parkinsonism, or cranial nerve palsies observed at any dose level 39 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. CB-011 expansion is correlated with responses at selected LD Flow PK data up to Day 28 visit from 35 patients treated with 500 mg/m2 cy and 30 mg/m2 flu daily x 3 days; arithmetic mean with SE shown Data cutoff 24Sept2025 AUC: area under the curve; Cmax: maximum concentration; Tmax: time to maximum concentration; LD: lymphodepletion; PK: pharmacokinetic; SE: standard error Responders (N=23) Non-responders (N=12) Days A bs ol ut e nu m be r o f C A R+ c el ls /µ l Measurement Responders Non-responders Tmax (median) Day 14 Day 8 Cmax (mean) 104 cells/µl 1 cells/µl AUC (mean) 0-week 4 976 cells/µl 4 cells/µl 40 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Rapid recovery of endogenous T and NK cells contributes to the manageable safety profile Average of log transformed values shown with ribbons reflecting standard error; dotted line is lower limit of quantification (LLOQ) for B cells Data through week 6 for 34 patients who received CB-011 with LD regimen of 500 mg/m2 cy and 30 mg/m2 flu daily x 3 days 1van de Donk N et al. 21st IMS (Annual Meeting, Brazil), 2024, P-090 2Tabbara N, et al. Hematology Am Soc Hematol Educ Program. 2024;(1):116-125 Data cutoff 24Sept2025 NK: natural killer B cells T cells NK cells • Patient T cell depletion enables CAR-T cell expansion • Fast recovery of T and NK cells quickly reinstates the patient’s natural immunity, likely contributing to the favorable safety profile 1,2 • Balancing CAR-T cell activity and recovery of natural immune activity is a key differentiator for Caribou’s allogeneic CAR-T programs N=34 Days lo g1 0( AB S+ 1)

41 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Single-dose treatment with rapid immune reconstitution differentiates CB-011 as an off-the-shelf option CB-011 offers single-dose approach with high response rates and manageable safety profile Allogeneic CAR-T cells Time • Repeat dosing Bispecifics • Limited or no B cell recovery • Single dose • Rapid immune cell reconstitution 42 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. CB-011 has the potential to shift the paradigm for treatment of patients with r/r multiple myeloma Manageable safety profile with no GvHD, colitis, parkinsonism, or cranial nerve palsies observed Potential to expand access and bring meaningful benefit to patients who urgently need a readily available, single-dose option Deep, durable responses observed in heavily pre-treated population 92% ORR, 75% ≥CR rate, 91% MRD negativity, and 7 of 12 ≥VGPR at ≥6 months at recommended dose for expansion MRD negative at ≤10-5 Data cutoff 24Sept2025 CR: complete response; GvHD: graft-versus-host disease; MRD: minimal residual disease; ORR: overall response rate; r/r: relapsed or refractory 43 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Discussion with lymphoma and multiple myeloma clinicians Professor of hematology/oncology and division director for hematologic malignancies and cellular therapeutics University of Kansas Cancer Center Joseph P. McGuirk, DOTina Albertson, MD, PhD Chief medical officer Caribou Biosciences Disclosures for Dr. McGuirk AlloVir: Consultancy, Honoraria, Research Funding, Speakers Bureau; Juno Therapeutics: Consultancy, Honoraria, Research Funding; Magenta Therapeutics: Consultancy, Honoraria, Research Funding; Kite, a Gilead Company: Consultancy, Honoraria, Research Funding, Speakers Bureau; Nektar: Consultancy, Honoraria; BMS: Consultancy, Honoraria, Speakers Bureau; Orca Bio: Research Funding; Sana: Honoraria; CRISPR Therapeutics: Consultancy, Research Funding; In8bio, Inc.: Other: IIT Clinical Trial; Sanofi: Honoraria, Consultancy, Speakers Bureau; Autolus: Consultancy, Honoraria, Research Funding; Astellas Pharma: Consultancy, Research Funding; Gamida Cell: Consultancy, Honoraria, Research Funding; Caribou: Consultancy, Honoraria, Research Funding; Incyte: Consultancy, Honoraria Director of CAR-T and stem cell transplant clinical program at the center of excellence for multiple myeloma Mount Sinai Adriana Rossi, MD 44 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Caribou is defining a new era in CAR-T cell therapy with two leading allogeneic programs ~$159M cash, cash equivalents, and marketable securities1 Continued engagement with FDA to refine pivotal trial prior to initiation Vispa-cel Dose expansion to initiate by EOY 2025 with data in 2026CB-011 1Preliminarily, Caribou expects to report $159.2M in cash, cash equivalents, and marketable securities as of September 30, 2025. Caribou is currently finalizing its financial results for the three and nine months ended September 30, 2025. While complete financial information is not yet available, the results presented above reflect preliminary estimates and the financial closing procedures for the quarter end are not yet complete. As a result, final results may vary from these preliminary estimates. See Important Information.
45 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. 1 Initial safety and efficacy data on the confirmatory cohort (20 patients) with partial HLA matching, with a minimum of six months of follow up for the majority of patients, as well as an update on the larger, maturing dataset presented previously. Q&A 46 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Gratitude for patients, caregivers, and investigators UC Irvine UC San Diego HonorHealth Huntsman Baylor MD Anderson OHC Atlantic HealthUniv of Iowa University of AZ Augusta Univ Arkansas Univ of Kentucky MCW BMTGA Montefiore Advent Health Norton Hackensack USC Univ of Penn NYU Langone Banner MD Anderson Vanderbilt Tufts OHSU VA Commonwealth VA Oncology Assoc. Israel - ANTLER Hadassah University Hospital Rabin Medical Center Tel Aviv Sourasky Medical Center The Sheba Fund for Health Services and Research Australia - ANTLER Epworth Hospital Royal Perth Westmead Hospital Miami MSKCC Cleveland Clinic UTSW U of Colorado Duke UAB Tennessee Oncology ANTLER trial sites CaMMouflage trial sites Appendix Vispa-cel appendix slides

49 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Vispa-cel is generally well tolerated Treatment-emergent adverse events (TEAEs1) in ≥25% of all patients 1TEAEs are defined as adverse events (AEs) with a start date on or after the vispa-cel infusion date 22L LBCL 4+ HLA matched, dosed with 80M vispa-cel CAR-T cells 32L LBCL patients treated with 80M vispa-cel CAR-T cells optimized for multiple factors, including 2+HLA matched and young donor 42L (N=32) and 3L+ (N=3) LBCL patients treated with 40M, 80M, and 120M vispa-cel CAR-T cells optimized for multiple factors, including 2+HLA matched and young donor CRS: cytokine release syndrome; TEAE: treatment-emergent adverse event Data cutoff date 02Sept2025 Event, n (%) All treated (N=84) Confirmatory cohort2 (N=22) Optimized profile at RP2D3 (N=27) Optimized profile4 (N=35) Any grade ≥Grade 3 Any grade ≥Grade 3 Any grade ≥Grade 3 Any grade ≥Grade 3 Any TEAE 83 (99) 73 (87) 21 (95) 16 (73) 26 (96) 20 (74) 34 (97) 26 (74) Thrombocytopenia 52 (62) 50 (60) 10 (45) 10 (45) 14 (52) 14 (52) 19 (54) 19 (54) CRS 46 (55) 1 (1) 13 (59) 1 (5) 17 (63) 1 (4) 19 (54) 1 (3) Anemia 44 (52) 36 (43) 7 (32) 5 (23) 9 (33) 5 (19) 12 (34) 8 (23) Neutropenia 33 (39) 30 (36) 5 (23) 5 (23) 6 (22) 6 (22) 9 (26) 9 (26) Hypokalemia 22 (26) 1 (1) 7 (32) 1 (5) 6 (22) 1 (4) 8 (23) 1 (3) Leukopenia 22 (26) 21 (25) 1 (5) 1 (5) 2 (7) 2 (7) 3 (9) 3 (9) 50 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Median onset and duration for neurotoxicity and CRS in vispa-cel compared to autologous CAR-T cells Vispa-cel Axi-cel ZUMA-7 N=1704,5 Liso-cel TRANSFORM N=926,7All treated N=84 Confirmatory cohort N=221 Optimized profile at 80M N=272 Optimized profile N=353 All grade All grade All grade All grade All grade All grade Neurotoxicity,8 n (%) 12 (14) 1 (5) 1 (4) 1 (3) 124 (74)9 11 (12)10 Median onset, days (range) 8.0 (6-34) 14.0 (14-14) 14.0 (14-14) 14.0 (14-14) 5.0 (1-133) 11.0 (7-17) Median duration, days (range) 2.0 (1-27) 1.0 (1-1) 1.0 (1-1) 1.0 (1-1) 15.0 (N/R) 4.5 (1-30) CRS, n (%) 46 (55) 13 (59) 17 (63) 19 (54) 157 (92) 45 (49) Median onset, days (range) 3.0 (0-22) 3.0 (0-14) 4.0 (0-14) 4.0 (0-14) 3.0 (1-10) 5.0 (1-63) Median duration, days (range) 3.0 (1-20) 3.0 (1-11) 3.0 (1-11) 2.5 (1-11) 7.0 (2-43) 4.0 (1-16) 12L LBCL 4+ HLA matched, dosed with 80M vispa-cel CAR-T cells 22L LBCL patients treated with 80M vispa-cel CAR-T cells optimized for multiple factors, including 2+HLA matched and young donor 32L (N=32) and 3L+ (N=3) LBCL patients treated with 40M, 80M, or 120M vispa-cel CAR-T cells optimized for multiple factors, including 2+HLA matched and young donor 4Locke et al; NEJM 2022; 5Yescarta prescribing information, 6Abramson et al; BLOOD 2023, 7Breyanzi FDA statistical review 8Vispa-cel includes ICANS; ZUMA-7 includes: all neurologic events; TRANSFORM includes: liso-cel–related neuro events. Note: ICANS was formally defined in 2018 (ASTCT consensus criteria), limiting comparability across studies 9N=168 10Investigator-identified neurologic toxicity CRS: cytokine release syndrome; N/R: not reported Data cutoff date for safety 02Sept2025 51 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Vispa-cel ANTLER response duration for 2L patients who received optimized product at RP2D 1 6 12 18 21 243 9 15 HLA 2 3 3 5 2 2 9 5 5 7 5 4 5 8 6 6 9 2 5 5 4 5 7 3 5 6 4 *Patient diagnosed with lung adenocarcinoma after D28 scan revealed a non-responsive lung nodule and was taken off study and enrolled on our long-term follow-up study. Patient last known to be in continued response without additional anti-lymphoma therapy at one year post vispa-cel Long-term follow-up data reflect the last known response; marked timepoints indicate confirmation of no disease progression One vispa-cel-related grade 5 IEC-HS occurred on day 25 post-infusion Certain patients converted from CR or PR to PD at various assessments time points as indicated in the chart above Data cutoff date 29Sept2025 CR: complete response; HLA: human leukocyte antigen; IEC-HS: immune effector cell-associated hemophagocytic lymphohistiocytosis-like syndrome; ORR: overall response rate; RP2D: recommended phase 2 dose CR: complete response PR: partial response SD: stable disease NA: not assessed PD: progressive disease Death Lost to follow-up Long-term follow-up // 36 48// Visit (months) Optimized profile at RP2D (80M) (N=27) ORR = 22/27 (82%) CR rate = 17/27 (63%) 52 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Baseline characteristics for patients enrolled in ANTLER Patient and disease characteristics All patients N=84 Confirmatory cohort1 N=22 Optimized profile at RP2D2 N=27 Optimized profile3 N=35 Age, years, median (range) 66 (20-86) 61 (20-83) 66 (20 -86) 63 (20-86) Male, n (%) 64 (76) 16 (73) 20 (74) 25 (71) ECOG, n (%) 0 40 (48) 13 (59) 14 (52) 19 (54) 1 44 (52) 9 (41) 13 (48) 16 (46) NHL subtype, n (%) DLBCL, NOS 48 (57) 14 (64) 16 (59) 21 (60) HGBL 13 (15) 4 (18) 5 (19) 5 (14) tFL 14 (17) 4 (18) 5 (19) 7 (20) tMZL 1 (1) - 1 (4) 1 (3) PMBCL 2 (2) - - 1 (3) MCL 3 (4) - - FL 2 (2) - - - MZL 1 (1) - - - Age adjusted IPI (%) 0-1 33 (39) 13 (59) 13 (48) 18 (51) 2 32 (38) 5 (23) 7 (26) 10 (29) 3 19 (23) 4 (18) 7 (26) 7 (20) Baseline LDH status (%) > ULN 33 (39) 10 (45) 13 (48) 16 (46) > 2x ULN 13 (15) 1 (5) 2 (7) 2 (6) Bulky disease4 17 (20) 2 (9) 3 (11) 4 (11) 12L LBCL 4+ HLA matched, dosed with 80M vispa-cel CAR-T cells 22L LBCL patients treated with 80M vispa-cel CAR-T cells optimized for multiple factors, including 2+HLA matched and young donor 32L (N=32) and 3L+ (N=3) LBCL 2+HLA matched and young donor, dosed with 40M, 80M, or 120M vispa-cel CAR-T cells 4Bulky disease defined by maximum baseline lesion diameter ≥7.5 cm Data cutoff 02Sept2025 2L: second-line; DLBCL, NOS: diffuse large B cell lymphoma, not otherwise specified; ECOG: Eastern Cooperative Oncology Group; HGBL: high grade B cell lymphoma; HLA: human leukocyte antigen; IPI: International Prognostic Index; LDH: lactate dehydrogenase; MCL: mantle cell lymphoma; MZL: marginal zone lymphoma; NHL: non-Hodgkin lymphoma; PMBCL: primary mediastinal B cell lymphoma; tFL: transformed follicular lymphoma; tMZL: transformed marginal zone lymphoma; ULN: upper limit of normal

53 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Vispa-cel has a PD-1 KO designed to reduce CAR-T cell exhaustion Vispa-cel NHL cell 1 2 3 TCR KO MHC I Anti-CD19 CAR CD19 PD-L1 PD-1 KO Armored with 3 genome edits 1 2 3 TRAC gene knockout (KO) • Eliminates TCR expression, reduces GvHD risk Anti-CD19 CAR site-specific insertion into TRAC locus • Eliminates random integration, targets tumor antigen PD-1 KO for enhanced antitumor activity • Reduces CAR-T cell exhaustion • Potentially contributes to initial tumor debulking CAR: chimeric antigen receptor; KO: knockout; CD: cluster of differentiation; chRDNA: CRISPR hybrid RNA-DNA; CRISPR: clustered regularly interspaced short palindromic repeats; PD-1: programmed cell death protein 1; TCR: T cell receptor; TRAC: T cell receptor alpha constant; scFv: single-chain variable fragment 1 To Caribou’s knowledge. 1st CAR-T in the clinic with checkpoint disruption via PD-1 KO1 Cas9 chRDNA editing for reduced off-target editing and enhanced genomic integrity Anti-CD19 scFv FMC63 with a 4-1BB costimulatory domain CB-011 appendix slides 55 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. 1LD regimen of 500 mg/m2 cy and 30 mg/m2 flu daily x 3 days 2Infections and infestations are by system organ class Data cutoff 24Sept2025 system presented cy: cyclophosphamide; flu: fludarabine; LD: lymphodepletion All at selected LD1 N=35 BCMA-naïve 450M at selected LD1 N=12 Adverse events Any grade n (%) Grade ≥3 n (%) Any grade n (%) Grade ≥3 n (%) Neutropenia/neutrophil count decreased 28 (80) 27 (77) 10 (83) 10 (83) Anemia 21 (60) 16 (46) 10 (83) 8 (67) Thrombocytopenia/platelet count decreased 17 (49) 10 (29) 7 (58) 4 (33) Infections2 17 (49) 5 (14) 8 (67) 3 (25) Dizziness 11 (31) -- 4 (33) -- Cytokine release syndrome 11 (31) 1 (3) 4 (33) 1 (8) Fatigue 11 (31) 4 (11) 3 (25) 2 (17) Leukopenia 10 (29) 10 (29) 1 (8) 1 (8) Decreased appetite 10 (29) 2 (6) 4 (33) -- Constipation 9 (26) -- 1 (8) -- Pyrexia 9 (26) -- 3 (25) -- CB-011 has a manageable safety profile Treatment emergent adverse events (TEAEs) in ≥25% of patients at selected LD 56 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. High-risk, heavily pretreated patients enrolled in CaMMouflage Patient and disease characteristics All patients1 (N=48) All patients at selected LD2 (N=35) BCMA-naïve 450M at selected LD2 (N=12) Age, years, median (range) 68.5 (49-84) 69 (53-82) 71 (53-80) Male, n (%) 33 (68.8) 24 (69) 9 (75) ECOG performance status, n (%) 0 12 (27) 7 (20) 3 (25) 1 35 (73) 28 (80) 9 (75) R-ISS disease stage, n (%) at diagnosis I 6 (13) 4 (11) - II 17 (35) 12 (34) 4 (33) III 12 (25) 8 (23) 5 (42) Unknown 13 (27) 11 (31) 3 (25) High risk cytogenetics3, n (%) 27 (56) 19 (54) 9 (75) Extramedullary disease (EMD), n (%)4 17 (35) 12 (34) 5 (42) Prior lines of therapy, median (range) 4 (3-11) 4 (3-11) 4 (3-5) Median time since diagnosis (years) 5.3 (1-14.9) 5.3 (1-14.9) 4.8 (1.7-14.5) Prior stem cell transplant, n (%) 30 (63) 20 (57) 5 (42) Prior exposure to BCMA therapy, n (%) 8 (17)5 5 (14)6 - 1All patients treated with a single dose of CB-011 and a lymphodepletion regimen of either 500 mg/m2 cy or 300 mg/m2 cy with 30 mg/m2 flu daily x 3 days 2LD regimen of 500 mg/m2 cy and flu 30 mg/m2 daily x 3 days 3High-risk cytogenetics include t(4;14), del(17/17p), t(14;16), t(14;20), and amplification/gain (1q) 4EMD defined as: soft tissue plasmacytoma noncontiguous with bone or lytic lesion with paramedullary extension 54 patients received belantamab (ADC) one of whom also received elranatamb (bispecific), 3 patients received teclistamab (bispecific), and 1 patient received NK trispecific (CC-92329 (BCMAXNKG2D/CD16)) 62 patients received belantamab (ADC) one of whom also received elranatamb (bispecific), 2 patient received teclistamab (bispecific), and 1 patient received NK trispecific (CC-92329 (BCMAXNKG2D/CD16)) Data cutoff 24Sept2025 Cy: cyclophosphamide; ECOG: Eastern Cooperative Oncology Group; flu: fludarabine; LD: lymphodepletion; NK: natural killer; R-ISS: revised international staging system
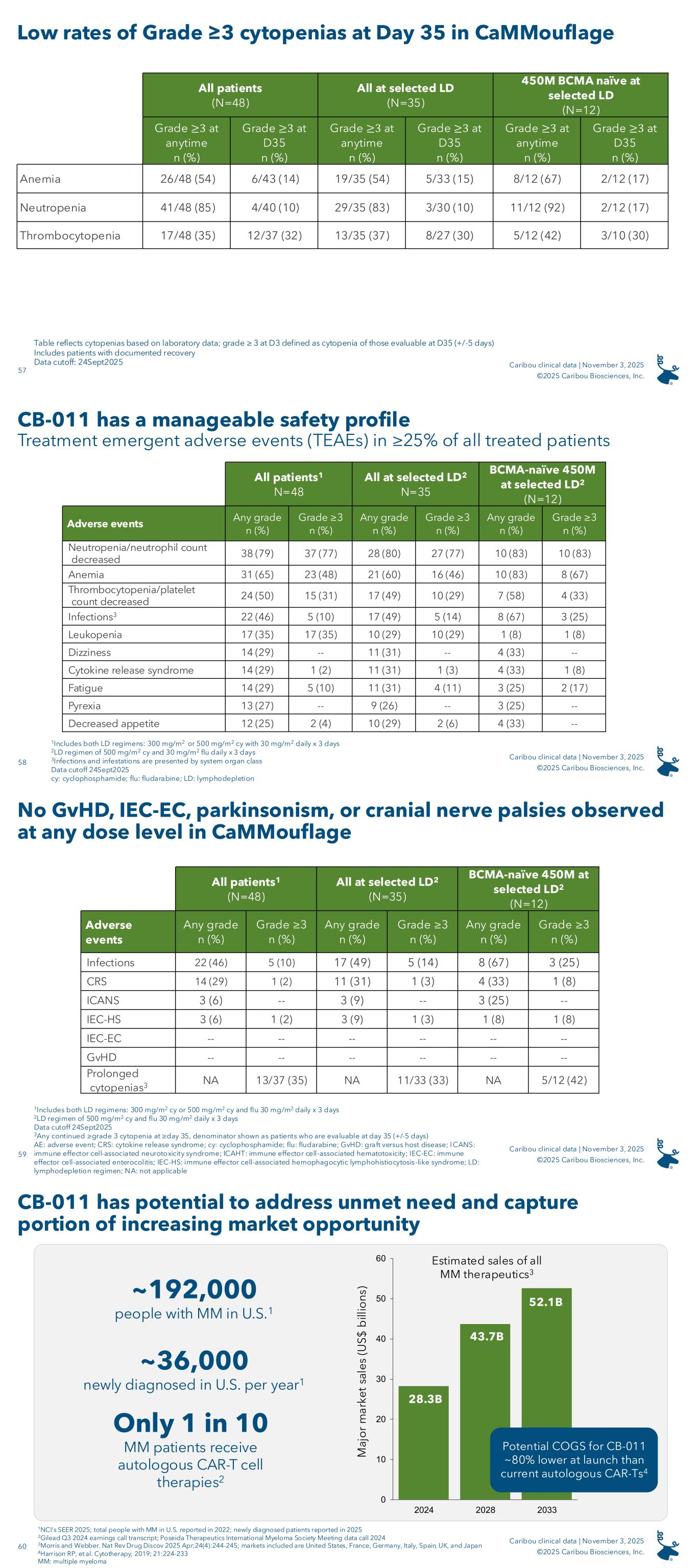
57 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Low rates of Grade ≥3 cytopenias at Day 35 in CaMMouflage All patients (N=48) All at selected LD (N=35) 450M BCMA naïve at selected LD (N=12) Grade ≥3 at anytime n (%) Grade ≥3 at D35 n (%) Grade ≥3 at anytime n (%) Grade ≥3 at D35 n (%) Grade ≥3 at anytime n (%) Grade ≥3 at D35 n (%) Anemia 26/48 (54) 6/43 (14) 19/35 (54) 5/33 (15) 8/12 (67) 2/12 (17) Neutropenia 41/48 (85) 4/40 (10) 29/35 (83) 3/30 (10) 11/12 (92) 2/12 (17) Thrombocytopenia 17/48 (35) 12/37 (32) 13/35 (37) 8/27 (30) 5/12 (42) 3/10 (30) Table reflects cytopenias based on laboratory data; grade ≥ 3 at D3 defined as cytopenia of those evaluable at D35 (+/-5 days) Includes patients with documented recovery Data cutoff: 24Sept2025 58 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. 1Includes both LD regimens: 300 mg/m2 or 500 mg/m2 cy with 30 mg/m2 daily x 3 days 2LD regimen of 500 mg/m2 cy and 30 mg/m2 flu daily x 3 days 3Infections and infestations are presented by system organ class Data cutoff 24Sept2025 cy: cyclophosphamide; flu: fludarabine; LD: lymphodepletion All patients1 N=48 All at selected LD2 N=35 BCMA-naïve 450M at selected LD2 (N=12) Adverse events Any grade n (%) Grade ≥3 n (%) Any grade n (%) Grade ≥3 n (%) Any grade n (%) Grade ≥3 n (%) Neutropenia/neutrophil count decreased 38 (79) 37 (77) 28 (80) 27 (77) 10 (83) 10 (83) Anemia 31 (65) 23 (48) 21 (60) 16 (46) 10 (83) 8 (67) Thrombocytopenia/platelet count decreased 24 (50) 15 (31) 17 (49) 10 (29) 7 (58) 4 (33) Infections3 22 (46) 5 (10) 17 (49) 5 (14) 8 (67) 3 (25) Leukopenia 17 (35) 17 (35) 10 (29) 10 (29) 1 (8) 1 (8) Dizziness 14 (29) -- 11 (31) -- 4 (33) -- Cytokine release syndrome 14 (29) 1 (2) 11 (31) 1 (3) 4 (33) 1 (8) Fatigue 14 (29) 5 (10) 11 (31) 4 (11) 3 (25) 2 (17) Pyrexia 13 (27) -- 9 (26) -- 3 (25) -- Decreased appetite 12 (25) 2 (4) 10 (29) 2 (6) 4 (33) -- CB-011 has a manageable safety profile Treatment emergent adverse events (TEAEs) in ≥25% of all treated patients 59 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. All patients1 (N=48) All at selected LD2 (N=35) BCMA-naïve 450M at selected LD2 (N=12) Adverse events Any grade n (%) Grade ≥3 n (%) Any grade n (%) Grade ≥3 n (%) Any grade n (%) Grade ≥3 n (%) Infections 22 (46) 5 (10) 17 (49) 5 (14) 8 (67) 3 (25) CRS 14 (29) 1 (2) 11 (31) 1 (3) 4 (33) 1 (8) ICANS 3 (6) -- 3 (9) -- 3 (25) -- IEC-HS 3 (6) 1 (2) 3 (9) 1 (3) 1 (8) 1 (8) IEC-EC -- -- -- -- -- -- GvHD -- -- -- -- -- -- Prolonged cytopenias3 NA 13/37 (35) NA 11/33 (33) NA 5/12 (42) 1Includes both LD regimens: 300 mg/m2 cy or 500 mg/m2 cy and flu 30 mg/m2 daily x 3 days 2LD regimen of 500 mg/m2 cy and flu 30 mg/m2 daily x 3 days Data cutoff 24Sept2025 3Any continued ≥grade 3 cytopenia at ≥day 35, denominator shown as patients who are evaluable at day 35 (+/-5 days) AE: adverse event; CRS: cytokine release syndrome; cy: cyclophosphamide; flu: fludarabine; GvHD: graft versus host disease; I CANS: immune effector cell-associated neurotoxicity syndrome; ICAHT: immune effector cell-associated hematotoxicity; IEC-EC: immune effector cell-associated enterocolitis; IEC-HS: immune effector cell-associated hemophagocytic lymphohistiocytosis-like syndrome; LD: lymphodepletion regimen; NA: not applicable No GvHD, IEC-EC, parkinsonism, or cranial nerve palsies observed at any dose level in CaMMouflage 60 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. CB-011 has potential to address unmet need and capture portion of increasing market opportunity ~192,000 people with MM in U.S.1 Only 1 in 10 MM patients receive autologous CAR-T cell therapies2 1NCI’s SEER 2025; total people with MM in U.S. reported in 2022; newly diagnosed patients reported in 2025 2Gilead Q3 2024 earnings call transcript; Poseida Therapeutics International Myeloma Society Meeting data call 2024 3Morris and Webber. Nat Rev Drug Discov 2025 Apr;24(4):244-245; markets included are United States, France, Germany, Italy, Spain, UK, and Japan 4Harrison RP, et al. Cytotherapy, 2019; 21:224-233 MM: multiple myeloma 0 10 20 30 40 50 60 2024 2028 2033 28.3B M aj or m ar ke t s al es (U S$ b ill io ns ) 43.7B 52.1B Estimated sales of all MM therapeutics3 ~36,000 newly diagnosed in U.S. per year1 Potential COGS for CB-011 ~80% lower at launch than current autologous CAR-Ts4
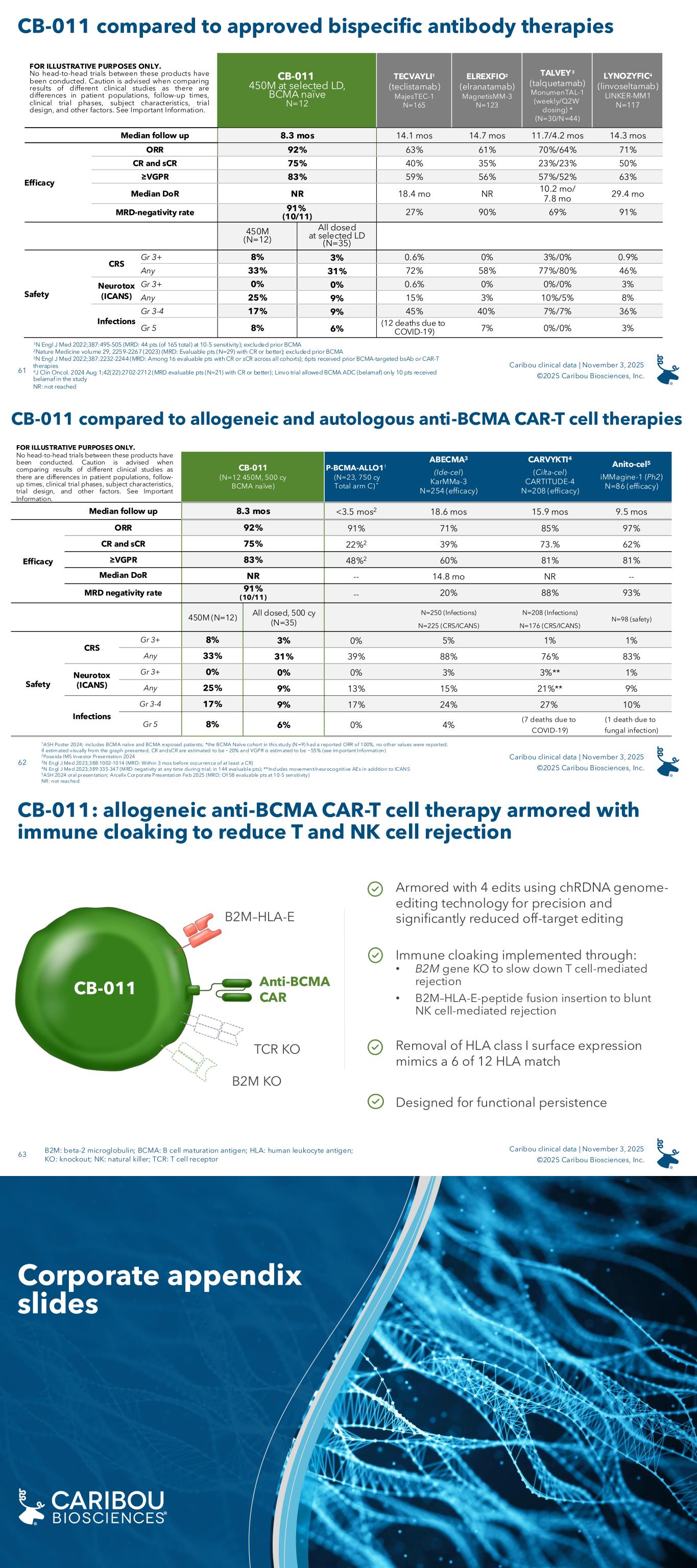
61 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. CB-011 450M at selected LD, BCMA naïve N=12 TECVAYLI1 (teclistamab) MajesTEC-1 N=165 ELREXFIO2 (elranatamab) MagnetisMM-3 N=123 TALVEY 3 (talquetamab) MonumenTAL-1 (weekly/Q2W dosing) * (N=30/N=44) LYNOZYFIC4 (linvoseltamab) LINKER-MM1 N=117 Median follow up 8.3 mos 14.1 mos 14.7 mos 11.7/4.2 mos 14.3 mos Efficacy ORR 92% 63% 61% 70%/64% 71% CR and sCR 75% 40% 35% 23%/23% 50% ≥VGPR 83% 59% 56% 57%/52% 63% Median DoR NR 18.4 mo NR 10.2 mo/ 7.8 mo 29.4 mo MRD-negativity rate 91% (10/11) 27% 90% 69% 91% 450M (N=12) All dosed at selected LD (N=35) Safety CRS Gr 3+ 8% 3% 0.6% 0% 3%/0% 0.9% Any 33% 31% 72% 58% 77%/80% 46% Neurotox (ICANS) Gr 3+ 0% 0% 0.6% 0% 0%/0% 3% Any 25% 9% 15% 3% 10%/5% 8% Infections Gr 3-4 17% 9% 45% 40% 7%/7% 36% Gr 5 8% 6% (12 deaths due to COVID-19) 7% 0%/0% 3% CB-011 compared to approved bispecific antibody therapies 1N Engl J Med 2022;387:495-505 (MRD: 44 pts (of 165 total) at 10-5 sensitivity); excluded prior BCMA 2Nature Medicine volume 29, 2259–2267 (2023) (MRD: Evaluable pts (N=29) with CR or better); excluded prior BCMA 3N Engl J Med 2022;387:2232-2244 (MRD: Among 16 evaluable pts with CR or sCR across all cohorts); 6pts received prior BCMA-targeted bsAb or CAR-T therapies 4J Clin Oncol. 2024 Aug 1;42(22):2702-2712 (MRD evaluable pts (N=21) with CR or better); Linvo trial allowed BCMA ADC (belamaf) only 10 pts received belamaf in the study NR: not reached FOR ILLUSTRATIVE PURPOSES ONLY. No head-to-head trials between these products have been conducted. Caution is advised when comparing results of different clinical studies as there are differences in patient populations, follow-up times, clinical trial phases, subject characteristics, trial design, and other factors. See Important Information. 62 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. CB-011 compared to allogeneic and autologous anti-BCMA CAR-T cell therapies CB-011 (N=12 450M, 500 cy BCMA naïve) P-BCMA-ALLO11 (N=23, 750 cy Total arm C) * ABECMA3 (Ide-cel) KarMMa-3 N=254 (efficacy) CARVYKTI4 (Cilta-cel) CARTITUDE-4 N=208 (efficacy) Anito-cel5 iMMagine-1 (Ph2) N=86 (efficacy) Median follow up 8.3 mos <3.5 mos2 18.6 mos 15.9 mos 9.5 mos Efficacy ORR 92% 91% 71% 85% 97% CR and sCR 75% 22%2 39% 73.% 62% ≥VGPR 83% 48%2 60% 81% 81% Median DoR NR -- 14.8 mo NR -- MRD negativity rate 91% (10/11) -- 20% 88% 93% 450M (N=12) All dosed, 500 cy (N=35) N=250 (Infections) N=225 (CRS/ICANS) N=208 (Infections) N=176 (CRS/ICANS) N=98 (safety) Safety CRS Gr 3+ 8% 3% 0% 5% 1% 1% Any 33% 31% 39% 88% 76% 83% Neurotox (ICANS) Gr 3+ 0% 0% 0% 3% 3%** 1% Any 25% 9% 13% 15% 21%** 9% Infections Gr 3-4 17% 9% 17% 24% 27% 10% Gr 5 8% 6% 0% 4% (7 deaths due to COVID-19) (1 death due to fungal infection) 1ASH Poster 2024; includes BCMA naïve and BCMA exposed patients; *the BCMA Naïve cohort in this study (N=9) had a reported ORR of 100%, no other values were reported; if estimated visually from the graph presented, CR and sCR are estimated to be ~20% and VGPR is estimated to be ~55% (see Important Information) 2Poseida IMS Investor Presentation 2024 3N Engl J Med 2023;388:1002-1014 (MRD: Within 3 mos before occurrence of at least a CR) 4N Engl J Med 2023;389:335-347 (MRD negativity at any time during trial; in 144 evaluable pts); ** Includes movement/neurocognitive AEs in addition to ICANS 5ASH 2024 oral presentation; Arcellx Corporate Presentation Feb 2025 (MRD: Of 58 evaluable pts at 10-5 sensitivity) NR: not reached FOR ILLUSTRATIVE PURPOSES ONLY. No head-to-head trials between these products have been conducted. Caution is advised when comparing results of different clinical studies as there are differences in patient populations, follow- up times, clinical trial phases, subject characteristics, trial design, and other factors. See Important Information. 63 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Armored with 4 edits using chRDNA genome- editing technology for precision and significantly reduced off-target editing Removal of HLA class I surface expression mimics a 6 of 12 HLA match 63 CB-011: allogeneic anti-BCMA CAR-T cell therapy armored with immune cloaking to reduce T and NK cell rejection CB-011 B2M–HLA-E Anti-BCMA CAR TCR KO B2M KO Designed for functional persistence Immune cloaking implemented through: • B2M gene KO to slow down T cell-mediated rejection • B2M–HLA-E-peptide fusion insertion to blunt NK cell-mediated rejection B2M: beta-2 microglobulin; BCMA: B cell maturation antigen; HLA: human leukocyte antigen; KO: knockout; NK: natural killer; TCR: T cell receptor Corporate appendix slides

65 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. chRDNA guides promote on-target and reduce off-target edits First-generation all-RNA CRISPR-Cas chRDNA CRISPR hybrid RNA-DNA 66 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Cas9 Cas12a chRDNA guides significantly improve editing specificity Editing assayed in primary human T cells Insert 1: 80% In se rt 2 : 7 6 % Knockout Knock-in Cas12a chRDNA genome editing + AAV6 transduction leads to >60% of manufacturing-scale engineered T cells with all 4 intended edits % e d it in g chRDNA guide on target chRDNA guide off targetAll-RNA guide on target All-RNA guide off target 0 2 4 6 20 40 60 80 100 all-RNA guide chRDNA PDCD1 ON OFF ON OFF TRAC B2M 0 2 4 6 20 40 60 80 100 chRDNA ON OFFON OFF % e d it in g 13.1 6.9 16.9 63.0 67 Caribou clinical data | November 3, 2025 ©2025 Caribou Biosciences, Inc. Engineering for improved activity against disease is key to unlocking the full potential of allogeneic cell therapies Caribou is implementing multiple armoring strategies Time C A R -T c e ll cy to to x ic a c ti v it y Checkpoint disruption (vispa-cel) PD-1 knockout (KO) sustains initial activity due to exhaustion resistance Immune cloaking (CB-011) B2M KO plus B2M–HLA-E fusion knock-in may delay host immune rejection Pretreatment lymphodepletion “Standard” allo CAR-T Checkpoint disruption Immune cloaking