UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-Q
(Mark One)
☒ QUARTERLY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
For the quarterly period ended January 31, 2026
☐ TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
For the transition period from to
Commission File No. 001-40101
BRIACELL THERAPEUTICS CORP.
(Exact name of registrant as specified in its charter)
| British Columbia, Canada | 47-1099599 | |
|
(State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer Identification No.) |
235 15th Street, Suite 300, West Vancouver, BC, V7T 2X1 |
| (Address of Principal Executive Offices, including zip code) |
| 604-921-1810 |
| (Registrant’s telephone number, including area code) |
| N/A |
| (Former name, former address and former fiscal year, if changed since last report) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol | Name of each exchange on which registered | ||
| Common shares, no par value | BCTX | The Nasdaq Stock Market LLC | ||
| Warrants to purchase common shares, no par value | BCTXW | The Nasdaq Stock Market LLC | ||
| Warrants to purchase common shares, no par value | BCTXZ | The Nasdaq Stock Market LLC | ||
| Warrants to purchase common shares, no par value | BCTXL | The Nasdaq Stock Market LLC |
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| ☐ Large accelerated filer | ☐Accelerated filer | |
| ☒ Non-accelerated filer | ☒ Smaller reporting company | |
| ☒ Emerging growth company |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act): Yes ☒ No ☐
As of March 10, 2026, 7,250,487 common shares, no par value per share, of the Company were issued and outstanding.
BRIACELL THERAPEUTICS CORP.
Form 10-Q
Table of Contents
|
|
PART I-FINANCIAL INFORMATION
Item 1. Financial Statements
BRIACELL THERAPEUTICS CORP.
CONDENSED CONSOLIDATED BALANCE SHEETS
| January 31, 2026 | July 31, 2025 | |||||||
| (Unaudited) | (Audited) | |||||||
| ASSETS | ||||||||
| CURRENT ASSETS: | ||||||||
| Cash and cash equivalents | $ | 29,903,628 |
$ | 10,493,808 | ||||
| Short-term investments | - | 7,372,473 | ||||||
| Amounts receivable and prepaid expenses | 2,275,256 | 2,060,295 | ||||||
| Total current assets | 32,178,884 |
19,926,576 | ||||||
| NON-CURRENT ASSETS: | ||||||||
| Equity investment in BC Therapeutics | 582,455 | 524,278 | ||||||
| Intangible assets, net | 176,889 | 184,525 | ||||||
| Property and equipment, net | 251,141 | 296,819 | ||||||
| Long term prepaid expenses | 405,085 | 717,508 | ||||||
| Total non-current assets | 1,415,570 | 1,723,130 | ||||||
| Total assets | $ | 33,594,454 |
$ | 21,649,706 | ||||
| LIABILITIES AND SHAREHOLDERS’ EQUITY | ||||||||
| CURRENT LIABILITIES: | ||||||||
| Trade payables | $ | 1,879,647 |
$ | 3,283,703 | ||||
| Accrued expenses and other payables | 1,305,903 | 694,285 | ||||||
| Total current liabilities | 3,185,550 | 3,977,988 | ||||||
| NON-CURRENT LIABILITIES: | ||||||||
| Warrant liability | 195,096 | 337,672 | ||||||
| Total non-current liabilities | $ | 195,096 | $ | 337,672 | ||||
| CONTINGENT LIABILITIES AND COMMITMENTS | - | - | ||||||
| SHAREHOLDERS’ EQUITY: | ||||||||
| Share Capital of no par value – Authorized: unlimited at January 31, 2026 and July 31, 2025; Issued and outstanding: 7,250,487 shares at January 31, 2026 and 1,883,906 July 31, 2025, respectively | 117,117,225 | 101,739,923 | ||||||
| Share-based payment reserved | 10,896,682 | 10,316,140 | ||||||
| Warrant reserve | 30,216,165 | 17,719,026 | ||||||
| Accumulated other comprehensive loss | (138,684 | ) | (138,684 | ) | ||||
| Non-controlling interest | (702,631 | ) | (546,795 | ) | ||||
| Accumulated deficit | (127,174,949 | ) | (111,755,564 | ) | ||||
| Total shareholders’ equity | 30,213,808 |
17,334,046 | ||||||
| Total liabilities and shareholders’ equity | $ | 33,594,454 |
$ | 21,649,706 | ||||
The accompanying notes are an integral part of these unaudited condensed consolidated financial statements.
|
|
BRIACELL THERAPEUTICS CORP.
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
(Unaudited)
| Three
months ended January 31, |
Six
months ended January 31, |
|||||||||||||||
| 2026 | 2025 | 2026 | 2025 | |||||||||||||
| (Unaudited) | (Unaudited) | (Unaudited) | (Unaudited) | |||||||||||||
| Operating Expenses: | ||||||||||||||||
| Research, development, and clinical trial expenses, net (note 9) | $ | 6,053,689 | $ | 5,684,777 | $ | 12,737,332 | $ | 9,350,118 | ||||||||
| General and administrative expenses | 1,477,577 | 1,484,666 | 3,116,877 | 2,972,157 | ||||||||||||
| Total operating expenses | 7,531,266 | 7,169,443 | 15,854,209 | 12,322,275 | ||||||||||||
| Operating loss | (7,531,266 | ) | (7,169,443 | ) | (15,854,209 | ) | (12,322,275 | ) | ||||||||
| Financial expenses, net | 84,589 |
67,358 | 243,235 |
79,072 | ||||||||||||
| Change in fair value of the warrant liability | 211,777 | 806,841 | 142,576 | 190,198 | ||||||||||||
| Share of loss on equity investment | (61,993 | ) | (42,584 | ) | (106,823 | ) | (114,099 | ) | ||||||||
| Net loss for the period | $ | (7,296,893 | ) | $ | (6,337,828 | ) | $ | (15,575,221 | ) | (12,167,104 | ) | |||||
| Net loss attributable to non-controlling interest | (75,073 | ) | (46,408 | ) | (155,836 | ) | (73,509 | ) | ||||||||
| Net loss and Comprehensive loss for the period attributable to BriaCell | (7,221,820 | ) | (6,291,420 | ) | (15,419,385 | ) | (12,093,595 | ) | ||||||||
| Net loss per share attributable to BriaCell – basic and diluted | $ | (2.58 | ) | $ | (23.31 | ) | $ | (6.58 | ) | $ | (54.35 | ) | ||||
| Weighted average number of shares used in computing net basic earnings per share of common stock | 2,802,337 | 269,957 | 2,343,122 | 222,498 | ||||||||||||
| Weighted average number of shares used in computing net diluted earnings per share of common stock | 2,802,337 | 269,957 | 2,343,122 | 222,498 | ||||||||||||
The accompanying notes are an integral part of these unaudited condensed consolidated financial statements.
|
|
BRIACELL THERAPEUTICS CORP.
CONDENSED CONSOLIDATED STATEMENTS OF CHANGES IN SHAREHOLDERS’ EQUITY
(Unaudited)
FOR THE THREE AND SIX MONTHS ENDED JANUARY 31, 2026
| Share capital | Additional paid in |
Warrant | Accumulated
other comprehensive |
Accumulated | Non- Controlling |
Total shareholders’ |
||||||||||||||||||||||||||
| Number | Amount | capital | reserve | loss | deficit | Interest | equity | |||||||||||||||||||||||||
| Balance, October 31, 2025 | 1,883,906 | $ | 101,739,923 | $ | 10,610,826 | $ | 17,719,026 | $ | (138,684 | ) | $ | (119,953,129 | ) | $ | (627,558 | ) | $ | 9,350,404 | ||||||||||||||
| Issuance of Options, RSU and PSU | - | - | 285,856 | - | - | - | - | 285,856 | ||||||||||||||||||||||||
| Exercise of warrants | 1,039,051 | - | - | - | - | - | - | - | ||||||||||||||||||||||||
| Issuance of units, net | 4,327,530 | 15,377,302 | - | 12,497,139 | - | - | - | 27,874,441 | ||||||||||||||||||||||||
| Net loss for the period | - | - | - | - | - | (7,221,820 | ) | (75,073 | ) | (7,296,893 | ) | |||||||||||||||||||||
| Balance, January 31, 2026 | 7,250,487 | $ | 117,117,225 | $ | 10,896,682 | $ | 30,216,165 | $ | (138,684 | ) | $ | (127,174,949 | ) | $ | (702,631 | ) | $ | 30,213,808 |
||||||||||||||
| Share capital | Additional paid in |
Warrant | Accumulated
other comprehensive |
Accumulated | Non- Controlling |
Total shareholders’ |
||||||||||||||||||||||||||
| Number | Amount | capital | reserve | loss | deficit | Interest | equity | |||||||||||||||||||||||||
| Balance, July 31, 2025 | 1,883,906 | $ | 101,739,923 | $ | 10,316,140 | $ | 17,719,026 | $ | (138,684 | ) | $ | (111,755,564 | ) | $ | (546,795 | ) | $ | 17,334,046 | ||||||||||||||
| Issuance of Options, RSU and PSU | - | - | 580,542 | - | - | - | - | 580,542 | ||||||||||||||||||||||||
| Exercise of prefunded warrants | 1,039,051 | - | - | - | - | - | - | - | ||||||||||||||||||||||||
| Issuance of units, net | 4,327,530 | 15,377,302 | - | 12,497,139 | - | - | - | 27,874,441 | ||||||||||||||||||||||||
| Net loss for the period | - | - | - | - | - | (15,419,385 | ) | (155,836 | ) | (15,575,221 | ) | |||||||||||||||||||||
| Balance, January 31, 2026 | 7,250,487 | $ | 117,117,225 | $ | 10,896,682 | $ | 30,216,165 | $ | (138,684 | ) | $ | (127,174,949 | ) | $ | (702,631 | ) | $ | 30,213,808 | ||||||||||||||
|
|
| Share capital | Additional paid in |
Warrant | Accumulated
other comprehensive |
Accumulated | Non- Controlling |
Total shareholders’ |
||||||||||||||||||||||||||
| Number | Amount | capital | reserve | loss | deficit | Interest | equity | |||||||||||||||||||||||||
| Balance, October 31, 2024 | 241,229 | $ | 82,040,741 | $ | 9,456,232 | $ | 3,930,221 | $ | (138,684 | ) | $ | (91,245,872 | ) | $ | (329,623 | ) | $ | 3,713,015 | ||||||||||||||
| Issuance of Options | - | - | 635,093 | - | - | - | - | 635,093 | ||||||||||||||||||||||||
| Exercise of prefunded warrants | 4,137 | 796,690 | - | (268,925 | ) | - | - | - | 527,765 | |||||||||||||||||||||||
| Issuance of units | 49,333 | 2,613,688 | - | 2,075,158 | - | - | - | 4,688,846 | ||||||||||||||||||||||||
| Net loss for the period | - | - | - | - | - | (6,291,420 | ) | (46,408 | ) | (6,337,828 | ) | |||||||||||||||||||||
| Balance, January 31, 2025 | 294,699 | $ | 85,451,119 | $ | 10,091,325 | $ | 5,736,454 | $ | (138,684 | ) | $ | (97,537,292 | ) | $ | (376,031 | ) | $ | 3,226,891 | ||||||||||||||
| Share capital | Additional paid in |
Warrant | Accumulated
other comprehensive |
Accumulated | Non- Controlling |
Total shareholders’ |
||||||||||||||||||||||||||
| Number | Amount | capital | reserve | loss | deficit | Interest | equity | |||||||||||||||||||||||||
| Balance, July 31, 2024 | 121,907 | $ | 72,166,414 | $ | 9,189,261 | $ | 1,844,296 | $ | (138,684 | ) | $ | (85,443,697 | ) | $ | (302,522 | ) | $ | (2,684,932 | ) | |||||||||||||
| Issuance of Options | - | - | 902,064 | - | - | - | - | 902,064 | ||||||||||||||||||||||||
| Exercise of prefunded warrants | 666 | - | - | - | - | - | - | - | ||||||||||||||||||||||||
| Exercise of broker warrants | 6,437 | 1,239,367 | - | (418,352 | ) | 821,015 | ||||||||||||||||||||||||||
| Issuance of units | 165,689 | 12,045,338 | - | 4,310,510 | - | - | - | 16,355,848 | ||||||||||||||||||||||||
| Net loss for the period | - | - | - | - | - | (12,093,595 | ) | (73,509 | ) | (12,167,104 | ) | |||||||||||||||||||||
| Balance, January 31, 2025 | 294,699 | $ | 85,451,119 | $ | 10,091,325 | $ | 5,736,454 | $ | (138,684 | ) | $ | (97,537,292 | ) | $ | (376,031 | ) | $ | 3,226,891 | ||||||||||||||
The accompanying notes are an integral part of these unaudited condensed consolidated financial statements.
|
|
BRIACELL THERAPEUTICS CORP.
CONDENSED CONSOLIDATED STATEMENT OF CASH FLOWS
(Unaudited)
| Six months ended January 31, | ||||||||
| 2026 | 2025 | |||||||
| Cash flow from operating activities | ||||||||
| Net loss for the period | $ | (15,575,221 | ) | $ | (12,167,104 | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||
| Amortization | 7,636 | 7,636 | ||||||
| Depreciation | 45,678 | 45,678 | ||||||
| Share-based compensation | 580,542 | 552,064 | ||||||
| Share of loss on equity investment | 106,823 | 114,099 | ||||||
| Change in fair value of warrants | (142,576 | ) | (190,198 | ) | ||||
| Changes in working capital: | ||||||||
| Decrease in amounts receivable and prepaid expenses | 97,462 | 642,846 | ||||||
| Decrease in accounts payable | (1,404,056 | ) | (2,167,542 | ) | ||||
| Increase in accrued expenses and other payables | 611,618 | 287,223 | ||||||
| Total cash flow from operating activities | (15,672,094 | ) | (12,875,298 | ) | ||||
| Cash flows from Investing activities | ||||||||
| Proceeds from short term investments | 7,372,473 | - | ||||||
| Equity investment in BC Therapeutics | (165,000 | ) | (150,000 | ) | ||||
| Total cash flow from investing activities | 7,207,473 | (150,000 | ) | |||||
| Cash flows from financing activities | ||||||||
| Proceeds from exercise of warrants | - | 821,015 | ||||||
| Proceeds from the issuance of shares, net of issuance costs | 27,874,441 | 16,355,848 | ||||||
| Total cash flow from financing activities | 27,874,441 | 17,176,863 | ||||||
| Increase in cash and cash equivalents | 19,409,820 |
4,151,565 | ||||||
| Cash and cash equivalents at beginning of the period | 10,493,808 | 862,089 | ||||||
| Cash and cash equivalents at end of the period | $ | 29,903,628 |
$ | 5,013,654 | ||||
The accompanying notes are an integral part of these unaudited condensed consolidated financial statements.
|
|
BriaCell Therapeutics Corp
Notes to the Condensed Consolidated Financial Statements
(Unaudited, expressed in US Dollars, except share and per share data and unless otherwise indicated)
NOTE 1: GENERAL AND GOING CONCERN
| a. | BriaCell Therapeutics Corp. (“BriaCell” or the “Company”) was incorporated under the Business Corporations Act (British Columbia) on July 26, 2006 and is listed on the Toronto Stock Exchange (“TSX”) under the symbol “BCT”. The Company also trades on the Nasdaq Capital Market (“NASDAQ”) under the symbols “BCTX”, “BCTXW” “BCTXZ”, and “BCTXL”. | |
| b. |
BriaCell is a clinical-stage biotechnology company that is developing novel immunotherapies to transform cancer care. The Company is currently advancing its Bria-IMT™ targeted immunotherapy in combination with an immune check point inhibitor (Retifanlimab) in a pivotal1 Phase 3 study in metastatic breast cancer. Bria-IMT™ is currently under Fast Track Designation by the U.S. Food and Drug Administration (the “FDA”) intended to accelerate the review process of novel treatments that address unmet medical needs. Positive completion of the pivotal study, following review by FDA, could lead to full approval of the Bria-IMT™ immune checkpoint inhibitor combination in metastatic breast cancer. BriaCell Phase 1/2 Study of Bria-OTS™, BriaCell’s personalized off-the-shelf immunotherapy, also known as Bria-BRES™, in metastatic breast cancer is ongoing BriaCell is currently developing Bria-OTS™ and its advanced form, Bria-OTS+™, as a platform technology for personalized off-the-shelf immunotherapies for numerous types of cancer. The Company announced BriaCell had received positive feedback from its Pre-Investigational New Drug Application (Pre-IND) meeting with FDA for Bria-PROS+™ for prostate cancer. |
|
| c. | Basis of presentation of the financial statements: | |
| The accompanying unaudited condensed consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America (“U.S. GAAP”) for interim financial information and in accordance with the instructions to Form 10-Q and Article 8 of Regulation S-X promulgated by the U.S Securities and Exchange Commission (the “SEC”). Certain information or footnote disclosures normally included in financial statements prepared in accordance with U.S. GAAP have been condensed or omitted pursuant to the rules and regulations of the SEC for interim financial reporting. Accordingly, they do not include all the information and footnotes necessary for a complete presentation of financial position, results of operations, or cash flows. In the opinion of management, the accompanying unaudited condensed consolidated financial statements include all adjustments consisting of a normal recurring nature which are necessary for a fair presentation of the financial position, operating results, and cash flows for the periods presented. | ||
| The accompanying unaudited condensed consolidated financial statements should be read in conjunction with the Company’s Annual Report for the year ended July 31, 2025, filed with the SEC on October 16, 2025. The interim period results do not necessarily indicate the results that may be expected for any other interim period or for the full fiscal year. |
| d. |
Going concern |
|
| The Company continues to devote substantially all of its efforts toward research and development activities. In the course of such activities, the Company has sustained operating losses and expects such losses to continue in the foreseeable future. The Company’s accumulated deficit as of January 31, 2026 was $127,174,949 and negative cash flows from operating activities during the six-month period ended January 31, 2026 was $15,672,094. The Company is planning to finance its operations by exploring additional sources of capital and financing, while managing its existing working capital resources. During the year ended July 31, 2025, the Company raised $50.9 million in gross proceeds from equity financings and, in January 2026, completed a public offering generating approximately $30.0 million in gross proceeds. However, the Company’s ability to continue as a going concern is dependent upon its ability to attain future profitable operations and to continue to obtain the necessary financing to meet its obligations arising from normal business operations when they come due. The uncertainty of the Company’s ability to raise such financial capital casts substantial doubt on the Company’s ability to continue as a going concern. These condensed consolidated financial statements do not include any adjustments to the amounts and classification of assets and liabilities that might be necessary should the Company not be able to continue as a going concern. | ||
| e. | The Company has two wholly-owned U.S. subsidiaries: (i) BriaCell Therapeutics Corp. (“BTC”), which was incorporated in April 3, 2014, under the laws of the state of Delaware, and (ii) BTC has a wholly-owned subsidiary, Sapientia Pharmaceuticals, Inc. (“Sapientia”), which was incorporated in September 20, 2012, under the laws of the state of Delaware. The Company also has one Canadian subsidiary: BriaPro Therapeutics Corp, (“BriaPro”) which was incorporated on May 15, 2023, under the Business Corporations Act (British Columbia). BriaPro was established to complete a plan of arrangement spinout transaction in August 2023, pursuant to which certain pipeline assets of the Company were spun-out to BriaPro, including Bria-TILsRx™ and protein kinase C delta (PKCδ) inhibitors for multiple indications including cancer (the “BriaPro Assets”), resulting in a two-third (2/3) owned subsidiary of the Company with the remaining one-third (1/3) held by the Company’s shareholders (the “Amalgamation” and the “Amalgamation Agreement”) – see also note 7(a). (Sapientia and BTC and BriaPro together, the “Subsidiaries”). |
|
|
BriaCell Therapeutics Corp
Notes to the Condensed Consolidated Financial Statements
(Unaudited, expressed in US Dollars, except share and per share data and unless otherwise indicated)
NOTE 2: SIGNIFICANT ACCOUNTING POLICIES
a. Use of estimates:
|
The preparation of financial statements in conformity with U.S. GAAP requires management to make estimates, judgments and assumptions that affect the amounts reported in the condensed consolidated financial statements and accompanying notes. The Company’s management believes that the estimates, judgment and assumptions used are reasonable based upon information available at the time they are made. These estimates, judgments and assumptions can affect the reported amounts of assets and liabilities at the dates of the condensed consolidated financial statements, and the reported amount of expenses during the reporting periods. Actual results could differ from those estimates.
Significant estimates include the determination of the fair value of warrant liabilities, which are measured using valuation models that require assumptions such as share price volatility, expected term, and risk-free interest rates. Changes in these inputs could materially impact the valuation of the warrant liability and the amounts recognized in the condensed consolidated financial statements. |
b. Prepaid expenses
| The Company has prepaid certain expenses in respect of its pivotal phase III trial and estimates the period over which such expenses will be incurred. As of January 31, 2026, the Company revised its estimate of the time to completion in respect of this trial. Amounts estimated to be expenses in more than 12 months have been classified to long-term prepaid expenses. |
c. The useful life of property and equipment
| Property and equipment are depreciated over their useful lives. Useful lives are based on management’s estimates of the period that the assets will be used which are periodically reviewed for continued appropriateness. Changes to estimates can result in significant variations in the amounts charged to the consolidated statement of operations and comprehensive loss in specific periods. |
d. Investment equity method:
| Investments in entities over which the Company does not have a controlling financial interest but has significant influence are accounted for using the equity method, with the Company’s share of losses reported in the loss from equity method investments on the statements of operation and comprehensive loss. The Company has a 67.8% interest in BC Therapeutics. Management evaluates whether it has control over the investee in accordance with the guidance of ASC 810, which requires judgment to assess factors such as power over significant activities of the investee, exposure to variable returns, and the ability to affect those returns. Based on this evaluation, management determines whether control or significant influence is present for accounting purposes. |
e. Segment reporting:
| The Company manages its business activities on a consolidated basis and operates as one reportable segment. The Company’s operations are focused on the research and development of its immunotherapy product candidates and related supporting activities. The Chief Executive Officer is identified as the Company’s Chief Operating Decision Maker (“CODM”). | ||
| The accounting policies of the segment are the same as those used in the condensed consolidated financial statements. The CODM evaluates the Company’s performance and allocates resources using consolidated financial information, including net loss and cash flow forecasts. The Company’s significant expenses, which consist primarily of research and development and general and administrative expenses, are consistent with the captions presented on the consolidated statements of operations and comprehensive loss. |
| The Company accounts for share-based compensation in accordance with ASC No. 718, “Compensation – Stock Compensation”, which requires companies to estimate the fair value of equity-based payment awards on the date of grant using an option-pricing model. The value of the award is recognized as an expense over the requisite service periods, which is the vesting period of the respective award, on a straight-line basis when the only condition to vesting is continued service. | ||
| The Company has selected the Black-Scholes option-pricing model as the most appropriate fair value method for its option awards. The Company recognizes forfeitures of equity-based awards as they occur. Restricted share units use the share price on the grant date to determine the fair value of the restricted share unit award. | ||
| For performance-based stock units (“PSUs”) that do not contain market conditions, the Company measures the grant-date fair value using the closing price of the common stock on the date of grant. Compensation cost for these awards is recognized over the requisite service period based on the number of awards that are expected to vest. Management evaluates the probability of achieving the applicable performance conditions each reporting period and adjusts the expense recognition accordingly. | ||
| As of the date of this report, the Company has issued stock options, RSUs, and PSUs that do not contain market conditions. |
g. Recently issued and adopted accounting standards:
| As an “emerging growth company,” the Jumpstart Our Business Startups Act (“JOBS Act”) allows the Company to delay adoption of new or revised accounting pronouncements applicable to public companies until such pronouncements are made applicable to private companies. The Company has elected to use this extended transition period under the JOBS Act. The adoption dates discussed below reflect this election. The pronouncements below relate to standards that impact the Company. |
|
|
BriaCell Therapeutics Corp
Notes to the Condensed Consolidated Financial Statements
(Unaudited, expressed in US Dollars, except share and per share data and unless otherwise indicated)
NOTE 2: SIGNIFICANT ACCOUNTING POLICIES (Cont.)
| 1. | In January 2025, the FASB issued ASU 2025-01 - Income Statement — Reporting Comprehensive Income — Expense Disaggregation Disclosures (Subtopic 220-40): Clarifying the Effective Date. This standard amends the guidance issued in 2024 to confirm that all public business entities must present the required expense-disaggregation disclosures in annual periods beginning after December 15, 2026, and interim periods within annual periods beginning after December 15, 2027. The ASU is effective for years beginning after those dates, but early adoption is permitted. This ASU should be applied on a prospective basis, although retrospective application is permitted. Because the amendment only affects disclosure timing, the Company does not expect this standard to have a material impact on its financial statements and disclosures. | |
| 2. | In June 2025, the FASB issued ASU 2025-03 - Business Combinations (Topic 805) and Consolidation (Topic 810): Determining the Accounting Acquirer in a Variable-Interest Entity. This standard clarifies that when a business combination is effected primarily by exchanging equity interests and the legal acquiree is a variable-interest entity (“VIE”) that meets the definition of a business, entities must identify the accounting acquirer using the factors in ASC 805-10-55-12 through 55-15, rather than relying solely on the VIE consolidation model. The ASU is effective for years beginning after December 15, 2026, but early adoption is permitted. This ASU should be applied on a prospective basis, although retrospective application is permitted. The Company is currently evaluating the impact of this standard on its financial statements and disclosures. |
NOTE 3: INVESTMENT IN BC THERAPEUTICS INC.
| On December 21, 2021, the Company and BC Therapeutics, Inc. (“BC Therapeutics” or “the Investee”) entered a share purchase agreement (“SPA”), pursuant to which the Company initially provided a loan of $300,000 to BC Therapeutics, with no interest to be paid. Subsequently, in accordance with the SPA, this loan was converted into an equity investment in BC Therapeutics at a rate of $1.25 per share, resulting in a 37.5% ownership interest (“Initial Investment”). | ||
| Pursuant to the SPA (“Initial Investment”), Briacell also received two options to invest an additional $225,000 per option at $1.25 per BC Therapeutics share. The first option expired on February 15, 2024 (“First BC Therapeutics Option”) and the second option expired on June 30, 2024 (“Second BC Therapeutics Options”, together, the “BC Therapeutic Options”). In accordance with ASC 321 and ASC 815, the BC Therapeutics Options were valued at $76,350 in accordance with the Black Scholes Option Price Model, using the following assumptions: Share price: $1.25, Exercise price: $1.25, Dividend yield: 0%, Risk free interest rate: 4.902%, Volatility: 100%. | ||
| BC Therapeutics has a board of four representatives, with two representatives appointed by BriaCell and two representatives appointed by the existing shareholders. All significant decisions related to BC Therapeutics require the approval of at least a majority of the board members. | ||
|
The Company initially acquired a significant interest in BC Therapeutics on February 1, 2024, by exercising the First BC Therapeutics Option, increasing its ownership to 51.2%. On August 7, 2024, following the expiration of the original Second BC Therapeutics Option, the Company and BC Therapeutics amended the SPA to introduce new options, allowing the exercise in tranches of at least 20,000 shares at $1.25 per share. On March 18, 2025, the SPA was amended a second time, such that the Second BC Therapeutics Option is increased to 424,000 shares and expires in June 2026 (a one year extension). During the six-month period ended January 31, 2026, the Company exercised this option in totaling $165,000 and received 132,000 shares.
As of January 31, 2026, the Company holds 816,000 of the 1,204,000 issued and outstanding shares in BC Therapeutics, representing a 67.8% ownership interest. In addition, 28,000 shares remain available for purchase under the Second BC Therapeutics Option at an exercise price of $1.25 per share; these options expire on June 30, 2026. |
||
|
In accordance with ASC 810, the Company continues to account for the investment under the equity method of accounting as the Company does not exercise control over BC Therapeutics. |
|
|
BriaCell Therapeutics Corp
Notes to the Condensed Consolidated Financial Statements
(Unaudited, expressed in US Dollars, except share and per share data and unless otherwise indicated)
NOTE 3: INVESTMENT IN BC THERAPEUTICS INC. (Cont.)
Changes in the Company’s equity investment in BC Therapeutics is summarized as follows:
SCHEDULE OF CHANGES IN INVESTMENT
| Balance – August 1, 2024 | $ | 418,490 | ||
| Funding (including the value of the BC Therapeutics Options) | 330,000 | |||
| Share of losses | (224,212 | ) | ||
| Balance – July 31, 2025 | 524,278 | |||
| Funding (including the value of the BC Therapeutics Options) | 165,000 | |||
| Share of losses | (106,823 | ) | ||
| Balance – January 31, 2026 | $ | 582,455 |
The following amounts represent the Company’s 67.8% share of the assets of BC Therapeutics (July 31, 2025 – 63.1%):
SCHEDULE OF ASSETS AND LIABILITIES OF BC THERAPEUTICS
|
As of January 31, 2026 |
||||
| Current assets: Cash | $ | 1,400 | ||
| Net assets | $ | 1,400 | ||
NOTE 4: CONTINGENT LIABILITIES AND COMMITMENTS
| a. | BriaPro Warrants | |
|
Upon the exercise of certain BriaCell warrants that were outstanding at the time of the Amalgamation Agreement with BriaPro (“Briacell Legacy Warrants”), BriaCell shall, as agent for BriaPro, collect and pay to BriaPro an amount based on an agreed formula. As of January 31, 2026, this amount totaled of up to $241,164 and is eliminated on consolidation.
Pursuant to the Amalgamation Agreement, each BriaCell warrant in issuance at the time of the Amalgamation (“Briacell Legacy Warrant”) shall, in accordance with its terms, entitle the holder thereof to receive, upon the exercise thereof, one BriaCell Share (and post Reverse Splits, as defined below– 150 Briacell Shares) and one BriaPro Share for the original exercise price. Warrants issued by the Company, subsequent to the Arrangement are not subject to the terms above.
Upon the exercise of 150 BriaCell Legacy Warrants (post Reverse Splits), BriaCell shall, as agent for BriaPro, collect and pay to BriaPro an amount for each one (1) BriaPro Share so issued that is equal to the exercise price under the 150 BriaCell Legacy Warrants multiplied by the fair market value of one (1) BriaPro Share at the Effective Date divided by the total fair market value of one (1) BriaCell Share and one (1) BriaPro Share at the Effective Date (“BriaPro Warrant Shares”). On a Reverse Split basis, as of January 31, 2026, 54,075 Briacell Legacy Warrants are exercisable into 54,075 Briacell Shares and 8,111,714 BriaPro Shares. |
||
| b. | Lease | |
| The Company has a month-to-month commitment for office and lab space in Philadelphia, PA, costing approximately $43,500 per month. |
NOTE 5: FAIR VALUE MEASUREMENTS
The following table presents information about our financial instruments that are measured at fair value on a recurring basis as of January 31, 2026 and July 31, 2025:
SCHEDULE OF FINANCIAL INSTRUMENTS MEASURED AT FAIR VALUE ON A RECURRING BASIS
| Fair Value Measurements at | ||||||||||||||||||||||||
| January 31, 2026 | July 31, 2025 | |||||||||||||||||||||||
| Level 1 | Level 2 | Total | Level 1 | Level 2 | Total | |||||||||||||||||||
| Financial Assets: | ||||||||||||||||||||||||
| Cash and cash equivalents | 29,903,628 | - | 29,903,628 | 10,493,808 | - | 10,493,808 | ||||||||||||||||||
| Short-term investments | - | - | - | 7,372,473 | - | 7,372,473 | ||||||||||||||||||
| Total assets measured at fair value | $ | 29,903,628 | $ | - | $ | 29,903,628 | $ | 17,866,281 | $ | - | $ | 17,866,281 | ||||||||||||
| Financial liabilities: | ||||||||||||||||||||||||
| Warrants liability | 38,578 | 156,518 | 195,096 | 151,586 | 186,086 | 337,672 | ||||||||||||||||||
| Total liabilities measured at fair value | $ | 38,578 | $ | 156,518 | $ | 195,096 | $ | 151,586 | $ | 186,086 | $ | 337,672 | ||||||||||||
|
|
BriaCell Therapeutics Corp
Notes to the Condensed Consolidated Financial Statements
(Unaudited, expressed in US Dollars, except share and per share data and unless otherwise indicated)
NOTE 5: FAIR VALUE MEASUREMENTS (Cont.)
We classify our cash and cash equivalents and the liability in respect of publicly traded warrants within Level 1 because we use quoted market prices in active markets.
As of January 31, 2026, the Company did not hold any short-term investments. As of July 31, 2025, the Company held Level 1 short-term investments measured using quoted prices in active markets, with coupon rates ranging from 3% to 5.51%.
The fair value of the warrant liability for non-public warrants is measured using inputs other than quoted prices included in Level 1 that are observable for the liability either directly or indirectly, and thus are classified as Level 2 financial instruments.
NOTE 6: SHAREHOLDERS’ EQUITY
a. Authorized share capital
The authorized share capital consists of an unlimited number of common shares with no par value.
b. Issued share capital
| (i) | Reverse Stock Split: |
| On January 3, 2025, the Company’s board of directors approved a reverse stock split of the Company’s common shares on a 1-for-15 basis, which became effective on January 24, 2025 and on August 25, 2025 (the “Effective Date”), the Company effected a reverse stock split of its common shares on a 1-for-10 basis (the “Reverse Splits”). | ||
| As a result of the Reverse Splits, every one hundred and fifty (150) pre-split common shares issued and outstanding were automatically combined into one (1) new common share. No fractional common shares were issued in connection with the Reverse Splits. Instead, any fractional common shares resulting from the January 2025 Reverse Split were deemed to have been tendered to the Company for cancellation for no consideration. | ||
| Following the January 2025 Reverse Split, the number of common shares outstanding were 294,694. After giving effect to subsequent share issuances and the August 2025 Reverse Split, the number of common shares outstanding was 1,883,906. | ||
| The Reverse Splits also resulted in a proportional adjustment to the number of common shares issuable upon the exercise of the Company’s outstanding warrants, stock options, and other convertible securities, as well as an adjustment to the exercise prices and conversion prices, as applicable. | ||
| All share and per share amounts in the accompanying condensed consolidated financial statements and related notes have been retroactively adjusted to reflect both the January 2025 Reverse Split and the August 2025 Reverse Split for all periods presented. |
| (ii) |
The Company issued the following shares during the six-month period ended January 31, 2026:
1. On January 15, 2026, the Company closed a public offering for the purchase and sale of 5,366,726 units of the Company for aggregate gross proceeds of approximately $30.0 million before deducting placement agent fees and other offering expenses (the “January 2026 Offering”). Each unit consisted of one common share (or one pre-funded warrant (“Pre-Funded Warrants”) in lieu thereof) and one warrant to purchase one common share of the Company at a combined purchase price of $5.59 per unit. The warrants have an exercise price of $6.93 per share, are immediately exercisable, and expire five years from the date of issuance (“January 2026 Warrants”). The common shares (or Pre-Funded Warrants) and January 2026 Warrants were purchased together in the offering but were issued separately. Total issuance costs associated with the offering were approximately $2,125,100, excluding the fair value of placement agent warrants.
In connection with the January 2026 Offering, the Company issued 161,001 placement agent warrants. The placement agent warrants are immediately exercisable at an exercise price of $8.39 per share and expire five years from the date of issuance.
The fair value of the 5,366,726 January 2026 Warrants was determined to be $13,103,125 (gross, before deducting share issuance costs) using the Black-Scholes option pricing model, with the following assumptions: share price – $3.15; exercise price – $6.93; expected life – 5 years; annualized volatility – 123%; dividend yield – 0%; risk-free rate – 3.762%.
The fair value of the 161,001 placement agent warrants was determined to be $572,334 using the Black-Scholes option pricing model, with the following assumptions: share price – $4.49; exercise price – $8.39; expected life – 5 years; annualized volatility – 123%; dividend yield – 0%; risk-free rate – 3.762%. The amounts were credited to the warrant reserve at the date of the January 2026 Offering. |
|
|
BriaCell Therapeutics Corp
Notes to the Condensed Consolidated Financial Statements
(Unaudited, expressed in US Dollars, except share and per share data and unless otherwise indicated)
NOTE 6: SHAREHOLDERS’ EQUITY (Cont.)
c. Share Purchase Warrants
SUMMARY OF CHANGES IN WARRANTS
|
Number of options outstanding |
Weighted average exercise price |
|||||||
| Balance, July 31, 2025 | 1,653,914 | $ | 58.27 | |||||
| Expired | (346 | ) | (579.97 | ) | ||||
| Granted in the January 2026 Offering | 5,366,726 | 6.93 | ||||||
| Balance, January 31, 2026 | 7,020,294 | 19.00 | ||||||
| (ii) | As of January 31, 2026, warrants outstanding were as follows: |
|
Number of Warrants |
Exercise Price |
Exercisable At January 31, 2026 |
Expiry Date | ||||||||
| (*)25,978 | $ | 796.88 | 25,978 | February 26, 2026 – April 26, 2026 | |||||||
| (*)27,820 | $ | 928.50 | 27,820 | December 7, 2026 | |||||||
| 16,019 | $ | 316.50 | 16,019 | November 17, 2029 | |||||||
| 27,753 | $ | 127.50 | 27,753 | October 2, 2029 | |||||||
| 49,333 | $ | 140.63 | 49,333 | December 12, 2029 | |||||||
| 306,665 | $ | 52.50 | 306,665 | April 28, 2030 | |||||||
| 1,200,000 | $ | 15.00 | 1,200,000 | July 15, 2030 | |||||||
| 5,366,726 | $ | 6.93 | 5,366,726 | January 15, 2031 | |||||||
| 7,020,294 | 7,020,294 | ||||||||||
| (*) | Briacell Legacy Warrants – see note 1(e) and note 4(a) |
d. Compensation Warrants
| (i) | A summary of changes in compensation warrants for the six-month period ended January 31, 2026 is presented below: |
|
Number of warrants outstanding |
Weighted average exercise price |
|||||||
| Balance, July 31, 2025 | 28,072 | 93.54 | ||||||
| Expired | (34 | ) | (579.97 | ) | ||||
| Granted in the January 2026 Offering | 161,001 | 8.39 | ||||||
| Balance, January 31, 2026 | 189,039 | $ | 20.93 | |||||
| (ii) | As of January 31, 2026, compensation warrants outstanding were as follows: |
SCHEDULE OF WARRANTS OUTSTANDING
|
Number of Warrants |
Exercise Price |
Exercisable At January 31, 2026 |
Expiry Date | ||||||||
| (*)113 | $ | 796.88 | 113 | February 26, 2026 | |||||||
| (*)164 | $ | 928.50 | 164 | June 7, 2026 | |||||||
| 333 | $ | 348.00 | 333 | May 17, 2029 | |||||||
| 4,108 | $ | 129.38 | 4,108 | September 12, 2029 | |||||||
| 1,709 | $ | 182.81 | 1,709 | October 2, 2029 | |||||||
| 2,466 | $ | 140.63 | 2,466 | December 12, 2029 | |||||||
| 3,812 | $ | 50.00 | 3,812 | February 5, 2030 | |||||||
| 15,333 | $ | 56.50 | 15,333 | April 28, 2030 | |||||||
| 161,001 | $ | 8.39 | 161,001 | January 15, 2031 | |||||||
| 189,039 | 189,039 | ||||||||||
| (*) | Briacell Legacy Warrants – see note 1(e) and note 4(a) |
|
|
BriaCell Therapeutics Corp
Notes to the Condensed Consolidated Financial Statements
(Unaudited, expressed in US Dollars, except share and per share data and unless otherwise indicated)
NOTE 6: SHAREHOLDERS’ EQUITY (Cont.)
e. Warrant liability continuity
The following table presents the summary of the changes in the fair value of the warrants:
SCHEDULE OF CHANGE IN FAIR VALUE OF WARRANTS
| Warrants liability | ||||
| Balance as of August 1, 2025 | $ | 337,672 | ||
| Change in fair value during the period | (142,576 | ) | ||
| Balance as of January 31, 2026 | $ | 195,096 | ||
The key inputs used in the valuation of the non-public warrants as of January 31, 2026 and at July 31, 2025 were as follows:
SCHEDULE OF VALUATION OF NON PUBLIC OFFERING BROKER WARRANTS
| January 31, 2026 | July 31, 2025 | |||||||
| Share price | $ | 4.30 | $ | 7.50 | ||||
| Exercise price | $ | 796.88-928.50 | $ | 796.88-928.50 | ||||
| Expected life (years) | 0.07-0.85 | 0.57-1.35 | ||||||
| Volatility | 177-355 | % | 157-209 | % | ||||
| Dividend yield | 0 | % | 0 | % | ||||
| Risk free rate | 3.48-3.64 | % | 4.10 | % | ||||
The key inputs used in the valuation of the of the BriaPro Warrant Shares as of January 31, 2026 were as follows:
SCHEDULE OF BRIA PRO WARRANTS
|
August 31, 2023 (Effective Date) |
January 31, 2026 | |||||||
| Share price | $ | 0.0365 | $ | 0.0365 | ||||
| Exercise price | $ | 0.0206-0.0308 | $ | 0.0206-0.0308 | ||||
| Expected life (years) | 2.21-3.27 | $ | 0.07-0.85 | |||||
| Volatility | 100 | % | 177-355 | % | ||||
| Dividend yield | 0 | % | 0 | % | ||||
| Risk free rate | 4.40 | % | 2.14-2.33 | % | ||||
|
|
BriaCell Therapeutics Corp
Notes to the Condensed Consolidated Financial Statements
(Unaudited, expressed in US Dollars, except share and per share data and unless otherwise indicated)
| a. |
On August 2, 2022, the Company approved an omnibus equity incentive plan (“Omnibus Plan), which will permit the Company to grant incentive stock options, preferred share units, restricted share units (“RSU’s”), performance-based share units (“PSUs”), and deferred share units (collectively, the “Awards”) for the benefit of any employee, officer, director, or consultant of the Company or any subsidiary of the Company. The maximum number of shares available for issuance under the Omnibus Plan shall not exceed 15% of the issued and outstanding Shares, from time to time, less the number of Shares reserved for issuance under all other security-based compensation arrangements of the Company, including the existing Stock Option Plan. On February 9, 2023, the Omnibus Plan was approved by the shareholders. |
|
| b. | The following table summarizes the number of options granted to directors, officers, employees and consultants under the option plan for three-month period ended January 31, 2026 and related information: |
| Number of options |
Weighted average exercise price |
Weighted average remaining contractual term (in years) |
Aggregate intrinsic value |
|||||||||||||
| Balance as of July 31, 2025 | 13,251 | $ | 896.61 | 1.62 | $ | - | ||||||||||
| Granted (i) | 37,700 | 12.50 | 4.75 | - | ||||||||||||
| Balance as of January 31, 2026 | 50,951 | 242.91 | 3.62 | - | ||||||||||||
| Exercisable as of January 31, 2026 | 22,676 | $ | 530.22 | 2.53 | $ | - | ||||||||||
| (i) | On August 1, 2025, the Company granted 37,700 stock options to employees and members of the scientific advisory board at an exercise price of $12.50 per share. All options vest quarterly over two years. The options expire on August 1, 2030. The grant-date fair value of the award was $218,784. The fair value of options granted during the six-month period ended January 31, 2026 was estimated using the Black-Scholes option-pricing model with the following weighted-average assumptions: expected volatility of 116%, expected term of 5.0 years, risk-free interest rate of 3.98%, dividend yield of 0%, and a stock price of $7.50 on the grant date. |
As of January 31, 2026, there were $1,448,754 of total unrecognized share-based compensation costs related to stock options, restricted share units (RSUs), and performance share units (PSUs) that are expected to be recognized over a period of up to 2.50 years.
|
|
BriaCell Therapeutics Corp
Notes to the Condensed Consolidated Financial Statements
(Unaudited, expressed in US Dollars, except share and per share data and unless otherwise indicated)
NOTE 7: SHARE-BASED COMPENSATION (Cont.)
| c. | The following table summarizes information about the Company’s outstanding and exercisable options granted to employees as of January 31, 2026 |
|
Exercise price |
Options outstanding as of January 31, 2026 |
Weighted average remaining contractual term (years) |
Options exercisable as of January 31, 2026 |
Weighted average remaining contractual term (years) |
Expiry Date | |||||||||||||||
| $ | 12.50 | 37,700 | 4.50 | 9,425 | 4.50 | August 1, 2030 | ||||||||||||||
| $ | 60.00 | 333 | 3.95 | 333 | 3.95 | January 16, 2030 | ||||||||||||||
| $ | 904.50 | 2,663 | 2.39 | 2,663 | 2.39 | June 20, 2028 | ||||||||||||||
| $ | 1,074.00 | 136 | 2.08 | 136 | 2.08 | February 27, 2028 | ||||||||||||||
| $ | 907.97 | 1,195 | 1.51 | 1,195 | 1.51 | August 2, 2027 | ||||||||||||||
| $ | 706.50 | 206 | 1.31 | 206 | 1.31 | May 20, 2027 | ||||||||||||||
| $ | 1,126.50 | 1,000 | 1.04 | 1,000 | 1.04 | February 16, 2027 | ||||||||||||||
| $ | 1,270.50 | 3,160 | 0.95 | 3,160 | 0.95 | January 13, 2027 | ||||||||||||||
| $ | 1,074.83 | 81 | 0.75 | 81 | 0.75 | November 1, 2026 | ||||||||||||||
| $ | 636.00 | 400 | 0.22 | 400 | 0.22 | April 19, 2026 | ||||||||||||||
| $ | 636.00 | 4,077 | 0.16 | 4,077 | 0.16 | March 29, 2026 | ||||||||||||||
| 50,951 | 22,676 | |||||||||||||||||||
| d. | As result of the Arrangement, 2,131,400 BriaPro Options were issued and are outstanding as of January 31, 2026: |
|
Exercise Price |
Options outstanding as of January 31, 2026 |
Options exercisable as of January 31, 2026 |
Expiry Date | |||||||||
| $ | 0.0933 | 440,000 | 440,000 | June 20, 2028 | ||||||||
| $ | 0.1108 | 21,000 | 21,000 | February 27, 2028 | ||||||||
| $ | 0.0984 | 180,100 | 180,100 | August 2, 2027 | ||||||||
| $ | 0.0729 | 31,000 | 31,000 | May 20, 2027 | ||||||||
| $ | 0.1162 | 150,000 | 150,000 | February 16, 2027 | ||||||||
| $ | 0.1310 | 524,700 | 524,700 | January 13, 2027 | ||||||||
| $ | 0.1165 | 12,600 | 12,600 | November 1, 2026 | ||||||||
| $ | 0.0888 | 100,000 | 100,000 | September 1, 2026 | ||||||||
| $ | 0.0656 | 60,000 | 60,000 | April 19, 2026 | ||||||||
| $ | 0.0656 | 612,000 | 612,000 | March 29, 2026 | ||||||||
| 2,131,400 | 2,131,400 | |||||||||||
| e. | Restricted Share Units |
SCHEDULE OF RESTRICTED STOCK UNITS GRANTED
| Number of | ||||||||
| RSU’s | Aggregate | |||||||
| outstanding | intrinsic value | |||||||
| Balance, July 31, 2025 | - | $ | - | |||||
| Granted (i) | 40,000 | 376,000 | ||||||
| Balance, January 31, 2026 | 40,000 | $ | 172,000 | |||||
| (i) | On September 24, 2025, the Company granted 40,000 RSUs to directors under the Omnibus Plan. These RSUs vest in full on the earlier of September 23, 2028 or the occurrence of a change of control, resignation, or dismissal without cause. The grant-date fair value of these RSUs was $376,000. |
|
|
BriaCell Therapeutics Corp
Notes to the Condensed Consolidated Financial Statements
(Unaudited, expressed in US Dollars, except share and per share data and unless otherwise indicated)
NOTE 7: SHARE-BASED COMPENSATION (Cont.)
f. Performance Share Units
SCHEDULE OF PERFORMANCE SHARE UNITS GRANTED
| Number of | ||||||||
| PSU’s | Aggregate | |||||||
| outstanding | intrinsic value | |||||||
| Balance, July 31, 2025 | - | $ | - | |||||
| Granted (i) | 165,935 | 1,434,513 | ||||||
| Balance, January 31, 2026 | 165,935 | $ | 713,521 | |||||
| (i) |
On August 1, 2025, the Company granted 165,935 performance-based stock units (“PSUs”) to the Chief Executive Officer (“CEO”), Chief Financial Officer (“CFO”), Chief Medical Officer (“CMO”), and Chief Scientific Officer (“CSO”) under the Omnibus Plan. These PSUs contain performance conditions tied to the advancement of the Company’s Bria-IMT Phase 3 program, the Bria-OTS program, and certain corporate and governance objectives. The grant-date fair value of the PSUs awarded to these officers totaled $353,228. The PSUs awarded to the CMO and CSO include milestones related to the Bria-OTS program, with grant-date fair values of $70,643 each. The CFO’s PSUs relate to corporate and financial reporting objectives, with a grant-date fair value of $70,643.
On September 24, 2025, the Company granted 100,000 PSUs to a director under the Omnibus Plan. These PSUs contain performance conditions related to the Company’s corporate, strategic, and governance objectives. The grant-date fair value of this award was $940,000, determined using the closing price of the Company’s common stock on the grant date.
The fair value of all PSU awards was determined using the closing price of the Company’s common stock on the respective grant dates. In accordance with ASC 718, management evaluates the probability of achieving each performance condition at each reporting date. As of January 31, 2026, management has determined that achievement of the applicable performance conditions remains probable. Compensation cost is recognized over the requisite service period on a graded vesting (tranche-by-tranche) basis.
During the six-month period ended January 31, 2026, the Company achieved the performance condition related to the CFO’s PSU’s. As a result, 1,177 PSUs granted to the CFO vested and became issuable during the period. |
| g. | The total share-based compensation expense related to all of the Company’s equity-based awards, recognized for the three and six-month period ended January 31, 2026 and 2025 is comprised as follows: |
| Three
months ended January 31, |
Six
months ended January 31, |
|||||||||||||||
| 2026 | 2025 | 2026 | 2025 | |||||||||||||
| (Unaudited) | (Unaudited) | (Unaudited) | (Unaudited) | |||||||||||||
| Research, development, and clinical trial expenses | $ | 79,016 | 50,839 | $ | 162,447 | 83,557 | ||||||||||
| General and administrative expenses | 206,840 | 234,254 | 418,095 | 468,507 | ||||||||||||
| Total share-based compensation | $ | 285,856 | 285,093 | $ | 580,542 | 552,064 | ||||||||||
NOTE 8: FINANCIAL INCOME, NET
SCHEDULE OF FINANCIAL INCOME , NET
|
Three months ended January 31, |
Six
months ended January 31, |
|||||||||||||||
| 2026 | 2025 | 2026 | 2025 | |||||||||||||
| (Unaudited) | (Unaudited) | (Unaudited) | (Unaudited) | |||||||||||||
| Interest income | $ | 105,638 | 46,832 | $ | 243,607 | 59,882 | ||||||||||
| Foreign exchange gain (loss) | (21,049 | ) | 20,526 | (372 | ) | 19,190 | ||||||||||
| Financial income, net | $ | 84,589 | $ | 67,358 | $ | 243,235 | $ | 79,072 | ||||||||
NOTE 9: RESEARCH, DEVELOPMENT, AND CLINICAL TRIAL EXPENSES, NET
| (i) |
On August 25, 2025, the Company announced that it had been awarded a $2.0 million Small Business Innovation Research (SBIR) grant from the National Cancer Institute (“NCI”) to support the advancement of Bria-PROS+™ for the treatment of prostate cancer. The grant provides non-dilutive funding intended to support manufacturing activities and planned clinical evaluation of the program.
The grant reimburses certain qualifying research and development expenditures incurred by the Company in connection with the program. In accordance with the Company’s accounting policy for government assistance, amounts received under the grant are recognized as a reduction of research and development expenses in the period in which the related costs are incurred.
During the three and six months ended January 31, 2026, the Company received $733,815 and $826,158, respectively, under the SBIR grant and recorded this amount as a reduction of research and development expenses in the condensed consolidated statements of operations.
As of January 31, 2026, the Company may receive up to an additional $1.2 million under the grant, subject to the achievement of certain research objectives and compliance with the grant terms. |
NOTE 10: SUBSEQUENT EVENTS
| (i) | On February 1, 2026, the Company granted an aggregate of 291,000 stock options to directors, executive officers, employees and scientific advisory board members under the Omnibus Plan. The stock options have an exercise price of US$5.59, vest in equal quarterly installments beginning May 1, 2026, and expire on February 1, 2031. |
| (ii) |
On February 18, 2026, the Company and BriaPro, announced that they have entered into a definitive purchase agreement (the “Purchase Agreement”) pursuant to which BriaPro has agreed to purchase BriaCell’s exclusive license to develop and commercialize Soluble CD80 (“sCD80”) as a biologic agent for the treatment of cancer and other associated assets (the “Transaction”).
Under the terms of the Purchase Agreement, BriaPro gains the worldwide rights to develop and commercialize sCD80 as a therapeutic agent for the treatment of cancer, while UMBC holds all rights, title and interest in the inventions and the patent, except for certain rights retained by the United States Government. BriaPro will pay 2% royalties to UMBC upon the commercialization of the product plus other development costs.
As part of the Transaction, BriaCell will make available to BriaPro up to $3 million to fund research and development efforts (the “Credit Facility”). Each drawdown under the Credit Facility will be subject to BriaCell’s approval regarding the use of funds.
As consideration for the transfer of the exclusive license and the Credit Facility, BriaPro will issue to BriaCell 23,972,589 Common Shares at an aggregate value of approximately C$1.18M, increasing BriaCell’s interest in BriaPro to approximately 78% post-transaction. The Transaction is expected to close by the end of March 2026, subject to certain conditions including (i) approval of the disinterested shareholders of BriaPro, and (ii) receipt of a third-party valuation confirming that the Transaction is occurring at fair market value.
In accordance with Multilateral Instrument 61-101 – Protection of Minority Security Holders in Special Transactions (“MI 61-101”), the resolution approving the Purchase Agreement must be approved by a simple majority of votes cast by shareholders, present in person or represented by proxy and entitled to vote at the Meeting, excluding the votes cast by any “interested party” (as defined in MI 61-101). As a 10% shareholder with an interest in the Transaction, BriaCell's shareholdings in BriaPro will be excluded from voting.
On March 5, 2026, the disinterested shareholders of BriaPro approved the Transaction. |
|
|
Item 2. Management’s Discussion and Analysis of Financial Condition and Results of Operations.
References to the “Company,” “our,” “us” or “we” refer to BriaCell Therapeutics Corp. The following discussion and analysis of the Company’s financial condition and results of operations should be read in conjunction with the unaudited condensed consolidated financial statements and the notes thereto contained elsewhere in this report. Certain information contained in the discussion and analysis set forth below includes forward-looking statements that involve risks and uncertainties.
Introduction
This Management’s Discussion and Analysis (“MD&A”) should be read together with other information, including our unaudited condensed interim consolidated financial statements and the related notes to those statements included in Part I, Item 1 of this Quarterly Report (the “Condensed Consolidated Financial Statements”), our consolidated financial statements appearing in our Annual Report on Form 10-K for the year ended July 31, 2025 (the “Annual Report”) and Part I, Item 1A, Risk Factors, of the Annual Report. This MD&A provides additional information on our business, recent developments, financial condition, cash flows and results of operations, and is organized as follows:
| ● | Part 1 - Business Overview. This section provides a general description of our business, which we believe is important in understanding the results of our operations, financial condition, and potential future trends. | |
| ● | Part 2 - Results of Operations. This section provides an analysis of our results of operations for the first half and second quarter of fiscal 2026 in comparison to the first half and second quarter of fiscal 2025. | |
| ● | Part 3 - Financial Liquidity and Capital Resources. This section provides an analysis of our cash flows and outstanding debt and commitments. Included in this analysis is a discussion of the amount of financial capacity available to fund our ongoing operations and future commitments. |
We prepare and report our unaudited Condensed Consolidated Financial Statements in accordance with U.S. GAAP. Our unaudited Condensed Consolidated Financial Statements, and the financial information contained herein, are reported in U.S Dollars.
Cautionary Note Regarding Forward-Looking Statements
This Quarterly Report on Form 10-Q includes forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Exchange Act. We have based these forward-looking statements on our current expectations and projections about future events. These forward-looking statements are subject to known and unknown risks, uncertainties and assumptions about us that may cause our actual results, levels of activity, performance or achievements to be materially different from any future results, levels of activity, performance or achievements expressed or implied by such forward-looking statements. In some cases, you can identify forward-looking statements by terminology such as “may,” “should,” “could,” “would,” “expect,” “plan,” “anticipate,” “believe,” “estimate,” “continue,” or the negative of such terms or other similar expressions. Factors that might cause or contribute to such a discrepancy include, but are not limited to, those described in our other SEC filings.
Overview
BriaCell Therapeutics Corp. (“Briacell” or the “Company”) is a clinical-stage biotechnology company that is developing novel immunotherapies to transform cancer care. Immunotherapies have come to the forefront in the fight against cancer as they harness the body’s own immune system to recognize and destroy cancer cells. The Company is currently advancing its Bria-IMT™ targeted immunotherapy in combination with an immune check point inhibitor (Retifanlimab) in a pivotal1 Phase 3 study in metastatic breast cancer (listed on ClinicalTrials.gov as NCT06072612). Bria-IMT™ is currently under Fast Track Designation by the U.S. Food and Drug Administration (the “FDA”) intended to accelerate the review process of novel treatments that address unmet medical needs. Positive completion of the pivotal study, following review by FDA, could lead to full approval of the Bria-IMT™ immune checkpoint inhibitor combination in metastatic breast cancer.
BriaCell has reported benchmark-beating patient survival and clinical benefit in metastatic breast cancer with median overall survival of 13.4 months in BriaCell’s metastatic breast cancer patients vs. 6.7-9.8 months2 for similar patients reported in the literature in its Phase 2 study of Bria-IMT™ combination study with retifanlimab. Additionally, BriaCell reported median overall survival of 16.5 months in Phase 2 Bria-IMT™ study patients treated in combination with immune checkpoint inhibitor in patients treated with the Phase 3 formulation since 2022 (post-COVID). A completed Bria-IMT™ Phase 1/2 combination study with retifanlimab (an anti-PD1 antibody manufactured by Incyte) confirmed tolerability and early-stage efficacy (listed on ClinicalTrials.gov as NCT03328026).
BriaCell Phase 1/2 Study of Bria-OTS™, BriaCell’s personalized off-the-shelf immunotherapy, also known as Bria-BRES™, in metastatic breast cancer is ongoing (listed on ClinicalTrials.gov as NCT06471673). The first patient treated with 4 inoculations of cells (single agent) demonstrated complete resolution of a lung metastasis.
BriaCell is currently developing Bria-OTS™ and its advanced form, Bria-OTS+™, as a platform technology for personalized off-the-shelf immunotherapies for numerous types of cancer. The Company announced BriaCell had received positive feedback from its Pre-Investigational New Drug Application (Pre-IND) meeting with FDA for Bria-PROS+™ for prostate cancer.
|
|
Recent Developments
During the period from August 1, 2025 through to the date of this report, we announced a number of corporate, financing and R&D developments. On August 21, 2025, our board approved a consolidation of the Company’s issued and outstanding common shares on the basis of one post-consolidation common share for every ten pre-consolidation common shares, primarily to help ensure continued compliance with Nasdaq Capital Market listing requirements. The consolidation became effective on August 25, 2025, with the post-consolidation common shares commencing trading on the Toronto Stock Exchange and Nasdaq on that date.
Throughout this time period, there were several important updated on the ongoing pivotal Phase 3 study of Bria-IMT™ in patients with advanced metastatic breast cancer, including accelerating enrollment, adding additional clinical sites, and several positive Data Safety Monitoring Board meetings. Some of these are detailed below.
We also strengthened our non-dilutive funding and external collaborations. On August 13, 2025, we announced acceptance into Memorial Sloan Kettering Cancer Center’s (MSK’s) 2025 Therapeutics Accelerator Cohort program for the Bria-OTS+™ platform, which includes the Bria-BRES+™ product candidate for breast cancer.
On August 25, 2025, we reported that we had been awarded a US$2.0 million Small Business Innovation Research (SBIR) grant from the U.S. National Cancer Institute (NCI) to advance Bria-PROS+™ in prostate cancer, providing non-dilutive funding to support manufacturing and planned clinical evaluation activities for this program
On October 21, 2025, we further announced a collaboration with MSK’s Therapeutics Accelerator program focused on the Bria-OTS+ platform. The collaboration includes support for manufacturing, IND development and clinical protocol work for a planned Phase 1 study of Bria-BRES+ in breast cancer, and is intended to help accelerate clinical development of Bria-OTS+ across multiple cancer indications.
Bria-IMT™
On November 25, 2025, BriaCell highlighted positive Phase 2 & Phase 3 clinical data at SABCS® 2025 with poster presentations at the San Antonio Breast Cancer Symposium (December 10, 2025) will highlight positive Phase 2 safety and efficacy signals and positive biomarker findings in both the Phase 2 and the pivotal Phase 3 studies.On December 9, 2025, BriaCell announced that patient enrollment is on track for 1H2026 topline data readout with over 230 patients screened and over 160 patients enrolled in BriaCell’s pivotal Phase 3 study in metastatic breast cancer (MBC).
Presentation Title: Survival Results of Phase II Bria-IMT Allogenic Whole Cell-Based Cancer Vaccine
Maturing positive Phase 2 data continue to support the potentially meaningful clinical benefit of the Bria-IMT regimen and the ongoing pivotal Phase 3 study is further evaluating this immunotherapy and the role of biomarkers in predicting patient response.
Presentation Title: Th1-biased cytokine signatures as biomarkers of clinical benefit following SV-BR-1-GM cancer vaccination in breast cancer. BriaCell’s data suggests that Th1 biased cytokines and chemokines may serve as potential predictive biomarkers of clinical responses to the Bria-IMT regimen in metastatic breast cancer.
Copies of the posters are available at https://briacell.com/scientific-publications/.
On December 10, 2025, BriaCell presented three posters highlighting robust survival and clinical benefit data in Phase 2, plus positive key biomarker data from the pivotal Phase 3 study of Bria-IMT™ plus immune check point inhibitor (CPI) at the San Antonio Breast Cancer Symposium (SABCS®) 2025. Data from the Phase 3 study continues to support biomarkers to identify patients who benefit from treatment with BriaCell’s regimen. Additionally, maturing Phase 2 study data continues to support meaningful clinical benefit of the Bria-IMT regimen with outstanding long-term survival. Importantly, no toxicity related discontinuations was reported in the Phase 3 study. Summaries of the studies are shown here.
On December 18, 2025, BriaCell announced that its Phase 3 Bria-IMT™ clinical trial in metastatic breast cancer was prominently and independently featured in the Nature Medicine Year In Review’s publication, “ Eleven clinical trials that will shape medicine in 2026”. Nature Medicine asked leading researchers to name their top clinical trial for 2026, from long-awaited vaccines for infectious diseases to new treatments for advanced cancers and long COVID. BriaCell’s Phase 3 clinical trial in metastatic breast cancer featured as “clinical trial to watch in 2026”
On January 27, 2026, BriaCell Highlighted extended >18-47 months survival in Phase 2 metastatic breast cancer patients. 9 of 25 BriaCell patients treated since 2022 remained alive >18-47 months post enrollment, markedly exceeding benchmarks reported for standard of care therapies in similar patient populations. No Bria-IMT™ related discontinuations was reported to that date. Bria-IMT regimen continues under Fast Track Designation from US FDA.
|
|
New positive Phase 2 survival data highlighted multiple patients surpassing expected benchmarks for metastatic breast cancer and survival length over 18 months in 9 cases as of their last assessment.
Table 1: Ongoing Long-Term Survivors
| Patient/Subtype | Months Since Study Start | Age | Number of Prior Regimens | Cycles of Bria-IMT | ||||
| 01-009/ER+/PR+/HER2low | 47 | 74 | 5 | 14 | ||||
| 07-001/ER+/PR+/HER2low | 30 | 55 | 7 | 8 | ||||
| 15-001/ER+/PR-/HER2- | 30 | 62 | 3 | 12 | ||||
| 11-018/ER+/PR+/HER2+ (Highlighted below) |
27 | 66 | 8; including ENHERTU | 35 | ||||
| 15-005/ER+/PR+/HER2- (Highlighted below) |
27 | 44 | 5 | 6 | ||||
| 15-006/ER+/PR-/HER2- (Highlighted below) |
25 | 64 | 8; including TRODELVY | 4 | ||||
| 15-004/ER+/PR+/HER2- | 25 | 50 | 3 | 6 | ||||
| 11-019/ER+/PR+/HER2low | 23 | 63 | 9; including TRODELVY | 6 | ||||
| 07-014/ER+/PR+/HER2low | >18 | 62 | 9; including TRODELVY | 5 |
Note that Trodelvy and Enhertu are antibody-drug conjugates recently approved for late-stage breast cancer.
Table 2: Comparable analysis of 1- and 2-year survival for the BriaCell Phase 2 study using the Phase 3 formulation since 2022 independent of subsequent treatment
|
Breast cancer |
Median prior | Median OS | % Survival at: | |||||||
| Reference | type | lines of therapy | (months) | 1 year | 2 years | |||||
| Bria-IMT ™ plus CPI |
All types 61% HR+ 33% TNBC 6% HER2+ |
6 | 15.6 |
52 % | 32% | |||||
| Cortes et al. 1 | All types 57% HR+ 18-19% TNBC 18-20% HER2+ |
4 | 9.1-9.3 | ~38-40% | 7-14% | |||||
| Kazmi et al. 2 | All types 51-52% HR+ 25-29% TNBC 9-24% HER2+ |
2 | 7.2-9.8 | 30-38% | 11.9-14% | |||||
| Bardia et al. (TPC) 3 | TNBC | 4 | 6.9 | ~23% | 6% | |||||
| Bardia et al. (Trodelvy) 3 | TNBC | 4 | 11.8 | 50% | 21% | |||||
| Rugo et al (TPC) 4 | HR+ HER2- | 4 | 11.2 | 47% | 21% | |||||
| Rugo et al (Trodelvy) 4 | HR+ HER2- | 4 | 14.4 | 60% | 25% | |||||
1. Cortes J, et al. Annals of Oncology 2018 (estimated from Kaplan-Meier curve)
2. Kazmi S, et al. Breast Cancer Res Treat. 2020
3. Bardia A, et al. J Clin Oncol. 2024 (estimated from Kaplan-Meier curve)
4. Rugo HS, et al. The Lancet. 2023 (estimated from Kaplan-Meier curve)
Abbreviations:
HR+: hormone receptor-positive
TNBC: Triple-negative breast cancer (lacks or has low levels of the estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2 (HER2))
HER2+: Human epidermal growth factor receptor 2 positive
HR+ HER2-: hormone receptor-positive and human epidermal growth factor receptor 2 negative
TPC: Treatment of Physicians Choice
On January 28, 2026, BriaCell reported patients’ images showing regression and resolution of metastasized tumors and immune activation in patients with orbital (eye), temporal lobe (brain), liver, and spinal involvement. Survival details on these and other select patients in its Phase 2 study, along with comparable populations, were previously reported.
Images below confirm clinical responses seen in patients with metastatic orbital (eye), temporal (brain), liver, and spine lesions.
|
|
Example 1: Patient 11-018
A 66-year-old woman with ER+/PR+/HER2+ metastatic breast cancer, heavily pretreated with 8 prior lines of therapy, including an antibody-drug conjugate (Enhertu), remains alive 27 months post-enrollment. At baseline, she presented with metastatic involvement of the right orbit (behind the eye), right temporal lobe of the brain and multiple skeletal sites. Following treatment, she achieved complete resolution of the temporal lobe metastasis, substantial improvement in the orbital lesion and stable disease in the bone. She remained on study for 26 months after initiating treatment and receiving 35 cycles of therapy. Images though 20 months for this patient have been previously described (link). Shown here are updated images through 2 years with measurements superimposed (through 18 months for the temporal lobe brain metastases).
Example 1 Images (Patient 11-018): Bria-IMT treatment resulted in complete resolution of the right temporal lobe lesion and continued regression of the right orbital (behind the eye) tumor. Measurements of lesion sizes are shown.
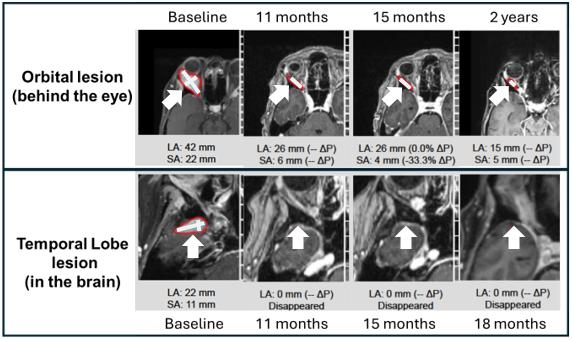
LA = long axis. SA = short axis.
Example 2: Patient 15-005:
A 44-year-old woman with ER+/PR+/HER2- metastatic breast cancer, previously treated with 5 prior lines of therapy. At baseline, she presented with metastases to the spine. She completed 6 cycles of therapy achieving stable disease as her best response and remains in survival follow-up 27 months after study entry.
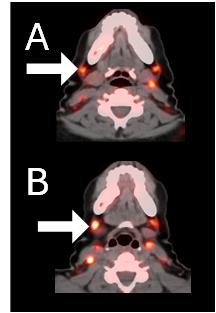
Before BriaCell Treatment Image A: CD8 ImmunoPET image
Pre-treatment imaging of cervical (neck) lymph nodes with moderate uptake indicating presence of some CD8+ cytotoxic (“killer”) T cells.
After BriaCell Treatment Image B: CD8 ImmunoPET image
Post treatment enhancement of cervical (neck) lymph nodes indicating immune system activation and increased presence of CD8+ cytotoxic T cells.
Example 2 Images (Patient 15-005): CD8 ImmunoPET images pre (A) and post (B) Bria-IMT treatment
Example 3: Patient 15-006:
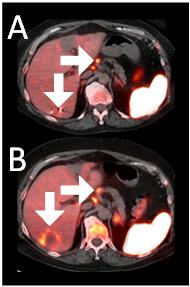
A 64-year-old woman with ER+/PR-/HER2- metastatic breast cancer, heavily pre-treated with 8 prior lines of therapy, including the antibody-drug conjugate Trodelvy, remains alive 25 months post-enrollment. At baseline, presented with hepatic metastasis.
Before BriaCell Treatment Image A: A liver metastasis (lower arrow) is “cold,” indicating minimal to no CD8+ cytotoxic T cells in the tumor while enlarged lymph nodes (upper arrow) show moderate uptake.
After BriaCell Treatment Image B: Swelling (induration) around the metastasis (lower arrow) demonstrates the liver metastasis has become “hot”, indicating marked CD8+ cytotoxic T cell infiltration while further lymph node enlargement is consistent with increased activity (upper arrow) indicating increased CD8+ T cells.
|
|
Example 3 Images (Patient 15-006): Combined MRI and CD8 ImmunoPET images Pre (A) and Post (B) Bria-IMT treatment
The Phase 2 study enrolled 54 heavily pre-treated metastatic breast cancer patients (median six prior therapies) who received the Bria-IMT regimen plus a checkpoint inhibitor. Of these, 37 patients were treated with the same formulation now being evaluated in the pivotal Phase 3 study (NCT06072612). Significantly, no Bria-IMT related discontinuations have been reported to date.No Bria-IMT™ related discontinuations reported to date. Bria-IMT regimen continues under Fast Track Designation from US FDA.
Bria-OTS and Bria-OTS+
On November 7, 2025, BriaCell presented robust anti-cancer activity of Bria-OTS+ in breast and prostate cancer models at the Society for Immunotherapy of Cancer (SITC) 2025. Bria-OTS+ represents a novel, cost-effective, off-the-shelf potent next generation cancer therapy platform. Next generation Bria-OTS+ platform demonstrated rapid, potent and durable anti-cancer activity by activating both innate and adaptive immune responses. Increased tumor cell cytotoxicity induced by both Bria-BRES+™ (breast cancer) and Bria-PROS+™ (prostate cancer) was reported. In summary, preclinical data reinforces broad potential of Bria-OTS+ platform across multiple indications. BriaCell added that lead candidates Bria-BRES+ (breast cancer) and Bria-PROS+ (prostate cancer) are completing GMP manufacturing for planned clinical trials. As a reminder, $2 million National Cancer Institute (NCI) Small Business Innovative Research (SBIR) award supports manufacturing and planned clinical evaluation of Bria-PROS+.
On January 13, 2026, BriaCell reported sustained complete resolution of lung metastasis in Bria-OTS Patient (Figure 1 as shown below). 11-month sustained complete resolution of lung metastasis was observed in Bria-OTS Phase 1/2a metastatic breast cancer study. No treatment limiting toxicities reported. The patient maintained stable disease at all other evaluable sites.
Figure 1: Treatment with Bria-OTS monotherapy resulted in 100% resolution of tumor in the right lung of the metastatic breast cancer (MBC) patient following 2 months of therapy and confirmed at 4, 6, and 11 months of therapy1 (axial and coronal views)
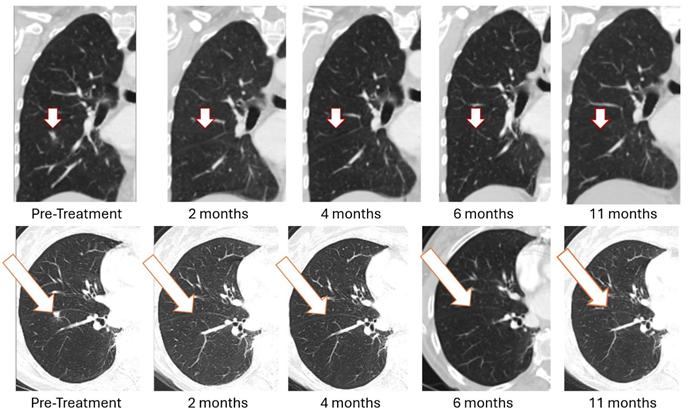
|
|
BriaPro
On February 18, 2026, the Company and BriaPro Therapeutics Corp. (“BriaPro”), a announced that we have entered into a definitive purchase agreement (the “Purchase Agreement”) pursuant to which BriaPro has agreed to purchase BriaCell’s exclusive license to develop and commercialize Soluble CD80 (“sCD80”) as a biologic agent for the treatment of cancer and other associated assets (the “Transaction”).
Background
BriaCell originally secured the exclusive license from the University of Maryland, Baltimore County (“UMBC”) on August 2, 2022. The novel technology, originally developed by Suzanne Ostrand-Rosenberg, Ph.D., Emeritus Faculty at UMBC, and member of BriaCell’s scientific advisory board, is titled “Soluble CD80 as a Therapeutic to Reverse Immune Suppression in Cancer Patients” and is covered under USPN 8,956,619 B2, USPN 9,650,429 B2, and USPN 10,377,810 B2. In animal models, sCD80 was well-tolerated and stopped tumor growth by potentially restoring natural anti-tumor immunity (see Lucas A Horn, et al. and Samuel T Haile et al. in collaboration with Dr. Ostrand-Rosenberg). Additionally, strong anti-tumor activity of sCD80 has been reported in multiple tumor types (see Lucas A Horn, et al.). Importantly, as demonstrated in the same studies, sCD80’s unique actions may involve both awakening and boosting the immune system to recognize and destroy tumor cells.
The Transaction
Under the terms of the Purchase Agreement, BriaPro gains the worldwide rights to develop and commercialize sCD80 as a therapeutic agent for the treatment of cancer, while UMBC holds all rights, title and interest in the inventions and the patent, except for certain rights retained by the United States Government. BriaPro will pay 2% royalties to UMBC upon the commercialization of the product plus other development costs.
As part of the Transaction, BriaCell will make available to BriaPro up to $3 million to fund research and development efforts (the “Credit Facility”). Each drawdown under the Credit Facility will be subject to BriaCell’s approval regarding the use of funds.
As consideration for the transfer of the exclusive license and the Credit Facility, BriaPro will issue to BriaCell 23,972,589 Common Shares at an aggregate value of approximately C$1.18M, increasing BriaCell’s interest in BriaPro to approximately 78% post-transaction. The Transaction is expected to close by the end of March 2026, subject to certain conditions including (i) approval of the disinterested shareholders of BriaPro, and (ii) receipt of a third-party valuation confirming that the Transaction is occurring at fair market value.
Shareholder Approval
In accordance with Multilateral Instrument 61-101 – Protection of Minority Security Holders in Special Transactions (“MI 61-101”), the resolution approving the Purchase Agreement must be approved by a simple majority of votes cast by shareholders, present in person or represented by proxy and entitled to vote at the Meeting, excluding the votes cast by any “interested party” (as defined in MI 61-101). As a 10% shareholder with an interest in the Transaction, BriaCell's shareholdings in BriaPro will be excluded from voting.
Formal Valuation Requirements
In respect of the formal valuation requirement of MI 61-101, BriaPro intends to rely on the specified markets exemptions set forth in subsection 5.5(b) of MI 61-101, as none of its securities are listed or quoted on a specified senior exchange.
Though a formal valuation is not required under applicable securities laws, as a matter of good governance and best corporate practice, BriaPro intends to obtain a valuation from an independent third-party valuator as a condition to closing, verifying and validating that Transaction is occurring at fair market value.
On March 5, 2026, the disinterested shareholders of BriaPro approved the Transaction.
Results of Operations for the Three and Six Months Ended January 31, 2026, and 2025
| Three months ended January 31, | ||||||||
| 2026 | 2025 | |||||||
| (Unaudited) | (Unaudited) | |||||||
| Operating expenses: | ||||||||
| Research and development expenses | $ | 6,053,689 | 5,684,777 | |||||
| General and administrative expenses | 1,477,577 | 1,484,666 | ||||||
| Total operating expenses | 7,531,266 | 7,169,443 | ||||||
| Operating loss | (7,531,266 | ) | (7,169,443 | ) | ||||
| Financial income, net | 84,589 | 67,358 | ||||||
| Change in fair value of the warrant liability | 211,777 | 806,841 | ||||||
| Share of loss on equity investment | (61,993 | ) | (42,584 | ) | ||||
| Net loss for the period | $ | (7,296,893 | ) | $ | (6,337,828 | ) | ||
| Net loss attributable to non-controlling interest | (75,073 | ) | (46,408 | ) | ||||
| Net loss for the period attributable to BriaCell | (7,221,820 | ) | (6,291,420 | ) | ||||
| Net loss per share attributable to BriaCell – basic and diluted | $ | (2.58 | ) | $ | (23.31 | ) | ||
| Weighted average number of shares used in computing net basic and diluted earnings per share of common stock | 2,802,337 | 269,957 | ||||||
|
|
Research and Development Costs
Research costs are comprised primarily of (i) salaries and wages to Company employees at our laboratory and (ii) clinical trials and investigational drug costs, which include the testing and manufacture of our investigational drugs and costs of our clinical trials.
The following is a breakdown of our research and development costs by nature of expenses:
| Three months ended January 31, | ||||||||
| 2026 | 2025 | |||||||
| (Unaudited) | (Unaudited) | |||||||
| Clinical trial sites and investigational drug costs | $ | 3,799,523 | $ | 4,117,576 | ||||
| Wages and salaries | 1,881,893 | 1,292,021 | ||||||
| Laboratory Rent | 130,500 | 114,330 | ||||||
| Supplies | 134,974 | 85,264 | ||||||
| Depreciation | 22,839 | 22,839 | ||||||
| Professional fees | 4,944 | 1,908 | ||||||
| Share-based compensation | 79,016 | 50,839 | ||||||
| $ | 6,053,689 | $ | 5,684,777 | |||||
For the three-month period ended January 31, 2026, total research and development expenses were $6,053,689, compared to $5,684,777 for the three-month period ended January 31, 2025. Clinical trial sites and investigational drug costs decreased modestly year over year, primarily reflecting decreased reliance of third-party vendors in our trials as we expand our in-house headcount to advance our trials, which also explains the increase in wages and salaries as detailed below. The increase in total research and development expenses was primarily driven by wages and salaries, which rose from $1,292,021 in 2025 to $1,881,893 in 2026, reflecting higher headcount and additional personnel required to support clinical operations and ongoing development programs. Laboratory rent increased to $130,500 in 2026, compared to $114,330 in 2025, due to expanded utilization of laboratory space and related facility charges. Supplies increased from $85,264 in 2025 to $134,974 in 2026, reflecting increased consumable usage driven by greater clinical and laboratory activity during the current period. Depreciation expense was consistent year over year at $22,839 for both periods. Professional fees increased from $1,908 in 2025 to $4,944 in 2026, primarily due to higher consulting, regulatory, and scientific support related to advancing clinical development. Share-based compensation increased from $50,839 in 2025 to $79,016 in 2026, reflecting a higher level of equity-based awards outstanding during the period.
Our clinical trial expenses are broken down as follows:
| Three months ended January 31, | ||||||||
| 2026 | 2025 | |||||||
| (Unaudited) | (Unaudited) | |||||||
| Bria-IMT™ Pivotal Phase 3 study | $ | 3,874,242 | $ | 3,642,650 | ||||
| Bria-IMT™ Phase 1/2a | 197,203 | 215,301 | ||||||
| Bria-OTS™ Phase 1/2a | 936,726 | 65,608 | ||||||
| $ | 5,008,171 | $ | 3,923,559 | |||||
Clinical trial expenses for the three months ended January 31, 2026, were $5,008,171, compared to $3,923,559 during the same period in 2025. The increase reflects continued investment in the Bria-IMT™ pivotal Phase 3 program and ongoing advancement of the Bria-OTS™ Phase 1/2a program as it progresses through early clinical development. Together, these programs account for the majority of the year-over-year increase in clinical trial expenses.
For the three-month period ended January 31, 2026, Bria-IMT™ Pivotal Phase 3 Study costs were $3,874,242, compared to $3,642,650 in 2025. The increase reflects the study’s continued progression through an advanced, cost-intensive stage, including expanded patient enrollment, increased clinical site activity, CRO services, central laboratory work, and clinical supply management. The level of spending is consistent with maintaining study momentum as the program advances toward planned interim analyses.
For the three-month period ended January 31, 2026, Bria-IMT™ Phase 1/2a expenses were $197,203, compared to $215,301 in 2025. The decrease reflects the continued wind-down of the program following completion of primary activities in fiscal 2024, with current-period costs primarily related to residual close-out procedures, data management, and limited follow-up activities. Expenses are expected to remain modest as final wrap-up items are completed.
For the three-month period ended January 31, 2026, Bria-OTS™ Phase 1/2a expenses were $936,726, compared to $65,608 in 2025. The substantial increase reflects continued advancement of the OTS program in early clinical development. Current-period costs include expanded development and manufacturing activities, GMP production of Bria-BRES+ and Bria-PROS+, regulatory and analytical support, and operational activities necessary to support ongoing evaluation of the next-generation Bria-OTS+ platform. The increased investment aligns with the program’s progression and the expansion of OTS across multiple solid tumor indications.
|
|
General and Administrative Expenses
For the three-month period ended January 31, 2026, general and administrative expenses were $1,477,577, compared to $1,484,666 for the same period in 2025. The decrease was primarily driven by lower share-based compensation, professional fees, consulting, shareholder communications, and travel expenses, partially offset by higher wages and salaries, regulatory, filing and transfer agent fees, insurance, and other general administrative costs.
Financial income, net
For the three-month period ended January 31, 2026, the Company recorded net financial income of $84,589, compared to $67,358 in the same period of 2025. The increase is mainly attributable to higher foreign exchange losses offset by higher interest income. For the three-month period ended January 31, 2026, financial income was comprised of $105,638 of interest income, and a $21,049 foreign exchange loss.
Profit (loss) for the period
For the three-month period ended January 31, 2026, the Company reported a net loss of $7,296,893, compared to $6,337,828 for the same period in 2025. The higher loss primarily reflects higher operating expenses, including increased research and development spending as the Company continued to advance its pivotal Phase 3 trial, including higher clinical-site activity, investigational product costs, and supporting operational infrastructure.
Results of Operations for the Six Months Ended January 31, 2026 and 2025
| Six months ended January 31, | ||||||||
| 2026 | 2025 | |||||||
| (Unaudited) | (Unaudited) | |||||||
| Operating Expenses: | ||||||||
| Research, development, and clinical trial expenses | $ | 12,737,332 | $ | 9,350,118 | ||||
| General and administrative expenses | 3,116,877 | 2,972,157 | ||||||
| Total operating expenses | 15,854,209 | 12,322,275 | ||||||
| Operating loss | (15,854,209 | ) | (12,322,275 | ) | ||||
| Financial income, net | 243,235 | 79,072 | ||||||
| Change in fair value of the warrant liability | 142,576 | 190,198 | ||||||
| Share of loss on equity investments | (106,823 | ) | (114,099 | ) | ||||
| Net loss for the period | $ | (15,575,221 | ) | (12,167,104 | ) | |||
| Net loss attributable to non-controlling interest | (155,836 | ) | (73,509 | ) | ||||
| Net loss for the period attributable to BriaCell | (15,419,385 | ) | (12,093,595 | ) | ||||
| Net loss per share attributable to BriaCell – basic and diluted | $ | (6.58 | ) | $ | (54.35 | ) | ||
| Weighted average number of shares used in computing net basic earnings per share of common stock | 2,343,122 | 222,498 | ||||||
| Weighted average number of shares used in computing net diluted earnings per share of common stock | 2,343,122 | 222,498 | ||||||
|
|
Research, Development, and Clinical Trial Costs
Research costs are comprised primarily of (i) salaries and wages to Company employees at our laboratory; and (ii) Clinical trials and investigational drug costs, which include the testing and manufacture of our investigational drugs and costs of our clinical trials.
The following is a breakdown of our research, development, and clinical trial costs by nature of expenses:
| Six months ended January 31, | ||||||||
| 2026 | 2025 | |||||||
| (Unaudited) | (Unaudited) | |||||||
| Clinical trial sites and Investigational drug costs | $ | 8,662,722 | $ | 6,557,243 | ||||
| Wages and salaries | 3,185,595 | 2,241,110 | ||||||
| Laboratory Rent | 255,110 | 228,660 | ||||||
| Supplies | 411,631 | 184,694 | ||||||
| Depreciation | 45,678 | 45,678 | ||||||
| Professional fees | 14,149 | 9,176 | ||||||
| Share-based compensation | 162,447 | 83,557 | ||||||
| $ | 12,737,332 | $ | 9,350,118 | |||||
For the six-month period ended January 31, 2026, research, development, and clinical trial expenses were $12,737,332, compared to $9,350,118 for the same period in 2025. The increase was primarily driven by higher clinical trial site and investigational drug costs, which rose from $6,557,243 in 2025 to $8,662,722 in 2026 as the Company advanced its clinical programs. Wages and salaries increased from $2,241,110 in 2025 to $3,185,595 in 2026, reflecting increased headcount and personnel supporting clinical and development activities. Laboratory rent increased to $255,110 in 2026 from $228,660 in 2025 due to expanded use of facilities. Supplies increased to $411,631 in 2026 from $184,694 in 2025, reflecting higher clinical and laboratory activity. Depreciation expense remained consistent year over year at $45,678. Professional fees increased to $14,149 in 2026 from $9,176 in 2025 due to increased consulting and support for clinical operations. Share-based compensation increased to $162,447 in 2026 from $83,557 in 2025, reflecting a higher level of equity-based awards outstanding during the period.
Clinical trial expenses for the period are as follows:
| Six months ended January 31, | ||||||||
| 2026 | 2025 | |||||||
| (Unaudited) | (Unaudited) | |||||||
| Bria-IMT™ Pivotal Phase 3 study | $ | 7,667,192 | $ | 6,089,111 | ||||
| Bria-IMT™ Phase 1/2a | 360,407 | 399,343 | ||||||
| Bria-OTS™ Phase 1/2a | 1,924,764 | 143,195 | ||||||
| $ | 9,952,363 | $ | 6,631,649 | |||||
Clinical trial expenses for the six months ended January 31, 2026, were $9,952,363, compared to $6,631,649 during the same period in 2025. The increase reflects sustained investment in the Bria-IMT™ pivotal Phase 3 program and significant advancement of the Bria-OTS™ Phase 1/2a program as it progresses through early clinical development. These programs continue to be the primary drivers of the year-over-year increase in clinical trial expenses.
For the six-month period ended January 31, 2026, Bria-IMT™ Pivotal Phase 3 Study costs were $7,667,192, compared to $6,089,111 in 2025. The increase reflects continued patient enrollment, expansion of clinical sites, increased clinical site activity, CRO services, central laboratory testing, and clinical supply management associated with the ongoing progression of the Phase 3 study. Spending levels during the period are consistent with maintaining operational momentum as the trial advances toward planned interim analyses.
For the six-month period ended January 31, 2026, Bria-IMT™ Phase 1/2a expenses were $360,407, compared to $399,343 in 2025. The decrease reflects the continued wind-down of the program following completion of primary activities in fiscal 2024. Current-period costs primarily relate to residual close-out procedures, data management, and limited follow-up activities. Expenses are expected to decline further as final wrap-up activities conclude.
For the six-month period ended January 31, 2026, Bria-OTS™ Phase 1/2a expenses were $1,924,764, compared to $143,195 in 2025. The substantial increase reflects continued advancement of the OTS program into early clinical development. Costs incurred during the period include expanded development and manufacturing activities, GMP production of Bria-BRES+ and Bria-PROS+, regulatory and analytical support, and operational activities necessary to support ongoing evaluation of the next-generation Bria-OTS+ platform. The increased investment aligns with the program’s progression and expansion across multiple solid tumor indications.
General and Administrative Expenses
For the six-month period ended January 31, 2026, general and administrative expenses amounted to $3,116,877, compared to $2,972,157 for the same period in 2025. The increase was primarily driven by higher wages and salaries, regulatory and filing fees, insurance, and other administrative costs, partially offset by lower consulting, professional fees, and travel expenses.
Financial income, net
For the six-month period ended January 31, 2026, financial income amounted to $243,235, compared to $79,072 for the six-month period ended January 31, 2025. Financial income for the 2026 period consisted of $243,607 in interest income and a $372 foreign exchange loss. In comparison, financial income for the 2025 period included $59,882 in interest income and a $19,190 foreign exchange gain. The increase in financial income from 2025 to 2026 was primarily due to higher interest income, reflecting increased cash and cash equivalents available for investment in interest-bearing funds.
|
|
Loss for the period
The Company reported a loss of $15,575,221 for the six-month period ended January 31, 2026, compared to $12,167,104 for the same period in 2025. The increase in loss was primarily driven by higher research, development and clinical trial expenses, which rose to $12,737,332 in 2026 from $9,350,118 in 2025 as the Company advanced its clinical programs. This was partially offset by higher financial income in the current period, including increased interest income and a lower foreign exchange loss.
Liquidity, Capital Resources and Going Concern Uncertainty
The financial statements have been prepared on a going concern basis, which assumes that the Company will be able to realize its assets and discharge its liabilities in the normal course of business for the foreseeable future. The continuing operations of the Company are dependent upon its ability to continue to raise adequate financing and to commence profitable operations in the future.
As of January 31, 2026, the company had a positive working capital balance of $28,993,334 (July 31, 2025 positive balance of $15,948,588).
As of January 31, 2026, the Company has total assets of $33,594,454 (July 31, 2025 - $ 21,649,706), a positive working capital of $28,993,334 (July 31, 2025 – positive balance of $15,948,588) and an accumulated deficit of $127,174,949 (July 31, 2025 - negative balance of $ 111,755,564).
As of January 31, 2026, the Company’s capital resources consist primarily of cash and cash equivalents, comprising mostly of cash on deposit with banks, investments in money market funds, investments in U.S. government securities, U.S. government agency securities, and investment grade corporate debt securities. Our investment policy and strategy are focused on preservation of capital and supporting our liquidity requirements.
Historically, the Company has financed its operation through private and public placement of equity securities, as well as debt financing. The Company’s ability to fund its longer-term cash requirements is subject to multiple risks, many of which are beyond its control. The Company intends to raise additional capital, either through debt or equity financings in order to achieve its business plan objectives. Management believes that it can be successful in obtaining additional capital; however, there can be no assurance that the Company will be able to do so. There is no assurance that any funds raised will be sufficient to enable the Company to attain profitable operations or continue as a going concern. To the extent that the Company is unsuccessful, the Company may need to curtail or cease its operations and implement a plan to extend payables or reduce overhead until sufficient additional capital is raised to support further operations. There can be no assurance that such a plan will be successful. To this end, for several months during calendar year 2025, certain directors and officers agreed to defer payment of their directors’ fees/compensation until we completed a financing, after which, these fees were paid in full. Further, certain officers have indicated their willingness to receive a portion of their compensation in equity of the Company, subject to applicable Nasdaq rules. In addition, we continue to reduce expenditure on certain non-core activities whilst maintaining our focus on our Phase 3 Bria-IMT™ pivotal study in advanced metastatic breast cancer.
|
|
During the period ended January 31, 2026, the Company’s overall position of cash and cash equivalents increased by $19,409,820 from the period ended July 31, 2025 (including effects of foreign exchange). This increase in cash can be attributed to the following:
The Company’s net cash used in operating activities during the period ended January 31, 2026, was $15,672,094 as compared to $12,875,298 for the period ended January 31, 2025.
Cash gained in financing activities for the period ended January 31, 2026, was 27,874,441 as compared to 17,176,863 for the period ended January 31, 2025.
Off-Balance Sheet Arrangements
None.
Tabular Disclosure of Contractual Obligations
None.
Critical Accounting Policies and Estimates
There have been no material changes to our critical accounting policies and estimates from the information provided in the MD&A section in our Annual Report.
New Accounting Policies Adopted
The Company did not adopt any new accounting policies during the period ended January 31, 2026.
Item 3. Quantitative and Qualitative Disclosures About Market Risk.
The Company’s financial instruments consist of cash and cash equivalents, investments, warrant liability, short term loans, trade payable, and accrued expenses and other payables. Unless otherwise noted, it is management’s opinion that the Company is not exposed to significant interest or credit risks arising from these financial instruments. The fair value of these financial instruments approximates their carrying values, unless otherwise noted.
Management understands that the Company is exposed to financial risk arising from fluctuations in foreign exchange rates and the degree of volatility of these rates as a portion of the Company’s transactions occur in Canadian Dollars (mainly costs relating to being a public company in Canada), and the Company’s functional and presentation currency is the US dollar. The Company does not use derivative instruments to reduce its exposure to foreign currency risk.
The Company is exposed in varying degrees to a variety of financial instrument related risks. The Board of Directors approves and monitors the risk management process. The overall objectives of the Board are to set policies that seek to reduce risk as far as possible without unduly affecting the Company’s competitiveness and flexibility.
The type of risk exposure and the way in which such exposure is managed is as follows:
Credit risk
The Company has no significant concentration of credit risk arising from operations. Management believes that the credit risk concentration with respect to financial instruments is remote.
Liquidity Risk
The Company’s approach to managing liquidity risk is to ensure that it will have sufficient liquidity to meet liabilities as they come due. As of January 31, 2026, the Company has total assets of $33,594,454 (July 31, 2025 - $21,649,706) and a positive working capital balance of $28,993,334 (July 31, 2025 – positive working capital balance of $15,948,588).
|
|
Market Risk
Interest rate risk
Interest Rate risk is the risk that the fair value of a financial instrument will fluctuate because of changes in market interest rates. Loans payable include both fixed and variable interest rates; however, the Company does not believe it is exposed to material interest rate risk.
Price risk
As the Company has no revenues, price risk is remote.
Exchange risk
The Company is exposed to foreign exchange risk as a portion of the Company’s transactions occur in Canadian Dollars (mainly costs relating to being a public company in Canada) and, therefore, the Company is exposed to foreign currency risk at the end of the reporting period through its Canadian denominated accounts payable and cash. As of January 31, 2026, a 5% depreciation or appreciation of the Canadian dollar against the US dollar would not have a material effect on the in total loss and comprehensive loss.
Fair Values
The carrying values of cash and cash equivalents, trade payable, warrant liability, short term loans, and accrued expenses and other payables approximate their fair values due to their short terms to maturity.
Cash and cash equivalents are valued using quoted market prices in active markets. The fair value of the warrant liability is determined based on the nature of the warrant. For publicly traded warrants we use the quoted market price and for all other warrants we use the Black-Scholes pricing model.
Item 4. Controls and Procedures.
Evaluation of Disclosure Controls and Procedures
We maintain “disclosure controls and procedures,” as defined in Rule 13a-15(e) and Rule 15d-15(e) under the Exchange Act that are designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is recorded, processed, summarized and reported, within the time periods specified in the SEC’s rules and forms. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is accumulated and communicated to our management, including our principal executive and principal financial officers, as appropriate to allow timely decisions regarding required disclosure.
Our management, with the participation of our principal executive officer and principal accounting and financial officer, has evaluated the effectiveness of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934 under the Securities Exchange Act of 1934, as amended, or the Exchange Act), as of the end of the period covered by this Quarterly Report on Form 10-Q. Our management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving their objectives and our management necessarily applies its judgment in evaluating the cost-benefit relationship of possible controls and procedures. Based on such evaluation, our principal executive officer and principal accounting and financial officer have concluded that as of January 31, 2026, our disclosure controls and procedures were effective at the reasonable assurance level.
Changes in Internal Control over Financial Reporting
There have not been material changes in our internal control over financial reporting during the quarter ended January 31, 2026, that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
|
|
PART II - OTHER INFORMATION
Item 1. Legal Proceedings.
None.
Item 1A. Risk Factors.
As of the date of this Quarterly Report on Form 10-Q, there have been no material changes from the risk factors previously disclosed in our Annual Report for the year ended July 31, 2025.
Item 2. Unregistered Sales of Equity Securities and Use of Proceeds.
There were no unregistered sales of equity securities during the six months ended January 31, 2026.
Item 3. Defaults Upon Senior Securities.
None.
Item 4. Mine Safety Disclosures.
Not Applicable.
Item 5. Other Information.
None.
Item 6. Exhibits
The following exhibits are filed as part of, or incorporated by reference into, this Quarterly Report on Form 10-Q.
EXHIBIT INDEX
| * | Filed herewith. |
|
|
SIGNATURES
In accordance with the requirements of the Exchange Act, the registrant caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| BRIACELL THERAPEUTICS CORP. | ||
| March 10, 2026 | By: | /s/ William V. Williams |
| Name: | William V. Williams | |
| Title: | Chief Executive Officer | |
| (Principal Executive Officer) | ||
| March 10, 2026 | By: | /s/ Gadi Levin |
| Name: | Gadi Levin | |
| Title: | Chief Financial Officer | |
| (Principal Financial and Accounting Officer) | ||
|
|
Exhibit 31.1
CERTIFICATIONS
I, William V. Williams, certify that:
| 1. | I have reviewed this Quarterly Report on Form 10-Q of BriaCell Therapeutics Corp.; |
| 2. | Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report; |
| 3. | Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report; |
| 4. | The registrant’s other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have: |
| (a) | Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared; | |
| (b) | Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles; | |
| (c) | Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and | |
| (d) | Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and |
| 5. | The registrant’s other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of the registrant’s board of directors (or persons performing the equivalent functions): |
| (a) | All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information; and | |
| (b) | Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal control over financial reporting. |
| March 10, 2026 | /s/ William V. Williams |
| William V. Williams | |
|
President and Chief Executive Officer (Principal Executive Officer) |
Exhibit 31.2
CERTIFICATIONS
I, Gadi Levin, certify that:
| 1. | I have reviewed this Quarterly Report on Form 10-Q of BriaCell Therapeutics Corp.; |
| 2. | Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report; |
| 3. | Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report; |
| 4. | The registrant’s other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have: |
| (a) | Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared; | |
| (b) | Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles; | |
| (c) | Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and | |
| (d) | Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and |
| 5. | The registrant’s other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of the registrant’s board of directors (or persons performing the equivalent functions): |
| (a) | All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information; and | |
| (b) | Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal control over financial reporting. |
| March 10, 2026 | /s/ Gadi Levin |
| Gadi Levin | |
|
Chief Financial Officer (Principal Financial Officer and Principal Accounting Officer) |
Exhibit 32.1
CERTIFICATION PURSUANT TO
18 U.S.C. SECTION 1350,
AS ADOPTED PURSUANT TO
SECTION 906 OF THE SARBANES-OXLEY ACT OF 2002
I, William V. Williams, President and Chief Executive Officer of BriaCell Therapeutics Corp. (the “Company”), hereby certify, pursuant to 18 U.S.C. §1350, as adopted pursuant to §906 of the Sarbanes-Oxley Act of 2002, that, to the best of my knowledge:
| 1. | The Quarterly Report on Form 10-Q of the Company for the period ended January 31, 2026 (the “Report”), fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934, as amended; and |
| 2. | The information contained in the Report fairly presents, in all material respects, the financial condition and results of operations of the Company. |
| March 10, 2026 | /s/ William V. Williams |
| William V. Williams | |
|
President and Chief Executive Officer (Principal Executive Officer) |
Exhibit 32.2
CERTIFICATION PURSUANT TO
18 U.S.C. SECTION 1350,
AS ADOPTED PURSUANT TO
SECTION 906 OF THE SARBANES-OXLEY ACT OF 2002
I, Gadi Levin, Chief Financial Officer of BriaCell Therapeutics Corp. (the “Company”), hereby certify, pursuant to 18 U.S.C. §1350, as adopted pursuant to §906 of the Sarbanes-Oxley Act of 2002, that, to the best of my knowledge:
| 1. | The Quarterly Report on Form 10-Q of the Company for the period ended January 31, 2026 (the “Report”), fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934, as amended; and |
| 2. | The information contained in the Report fairly presents, in all material respects, the financial condition and results of operations of the Company. |
| March 10, 2026 | /s/ Gadi Levin |
| Gadi Levin | |
|
Chief Financial Officer (Principal Financial Officer and Principal Accounting Officer) |