UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): December 3, 2025
bioAffinity Technologies, Inc.
(Exact name of registrant as specified in its charter)
| Delaware | 001-41463 | 46-5211056 | ||
|
(State or other jurisdiction of incorporation) |
(Commission File Number) |
(I.R.S. Employer Identification Number) |
3300 Nacogdoches Road, Suite 216
San Antonio, Texas 78217
(Address of principal executive offices, including zip code)
(210) 698-5334
(Registrant’s telephone number, including area code)
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
| ☐ | Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ☐ | Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ☐ | Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ☐ | Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e- 4(c)) |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
| Title of each class | Trading Symbols | Name of each exchange on which registered | ||
|
Common Stock, par value $0.007 per share
|
|
BIAF
|
The Nasdaq Stock Market LLC (Nasdaq Capital Market) |
|
| Warrants to purchase Common Stock | BIAFW |
The Nasdaq Stock Market LLC (Nasdaq Capital Market) |
Indicate by check mark whether the registrant is an emerging growth company as defined in in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☒
If an emerging growth company, indicate by checkmark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 8.01. Other Events.
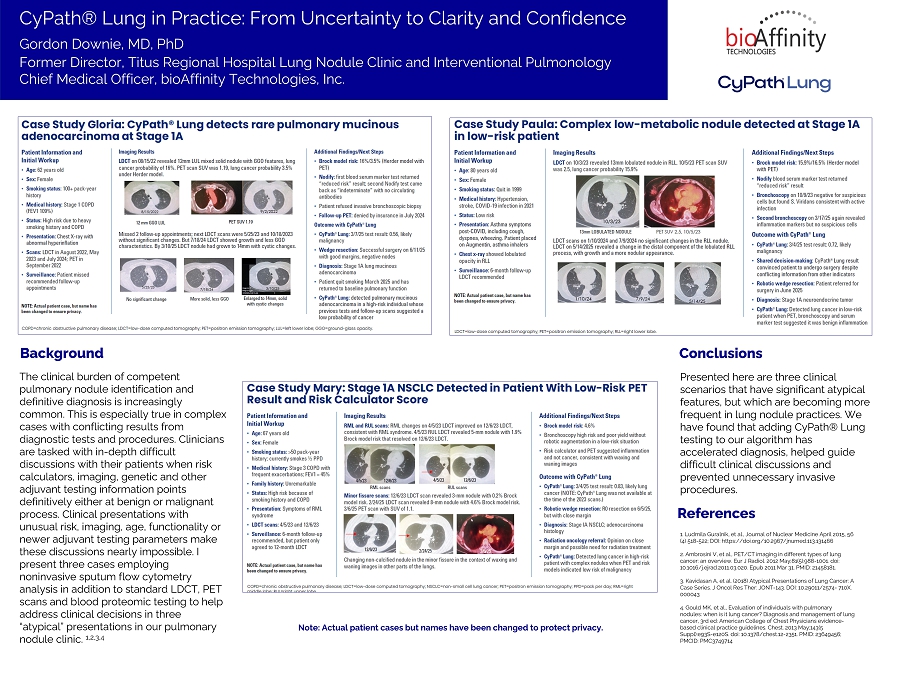
On December 3, 2025, bioAffinity Technologies, Inc., a Delaware corporation, issued a press release announcing that it will present a poster entitled “CyPath® Lung in Practice: From Uncertainty to Clarity and Confidence” (the “Poster”) at the American Cancer Society National Lung Cancer Roundtable on December 8, 2025. The Poster showcases three cases in which CyPath® Lung, a noninvasive sputum-based flow cytometry test, successfully identified Stage 1A lung cancer in patients with atypical and diagnostically challenging presentations.
Copies of the press release and Poster are attached hereto as Exhibit 99.1 and Exhibit 99.2, respectively and are incorporated herein by reference.
Item 9.01. Financial Statements and Exhibits.
| (d) | Exhibits. |
| Exhibit Number |
Description | |
| 99.1 | Press Release issued by bioAffinity Technologies, Inc., dated December 3, 2025 | |
| 99.2 | Poster entitled “CyPath® Lung in Practice: From Uncertainty to Clarity and Confidence” | |
| 104 | Cover Page Interactive Data File (embedded within the XBRL document) |
| - |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this Current Report on Form 8-K to be signed on its behalf by the undersigned hereunto duly authorized.
| Date: December 3, 2025 | BIOAFFINITY TECHNOLOGIES, INC. | |
| By: | /s/ Maria Zannes | |
| Name: | Maria Zannes | |
| Title: | President and Chief Executive Officer | |
| - |
Exhibit 99.1

bioAffinity Technologies’ Noninvasive CyPath® Lung Test to Be Highlighted at American Cancer Society National Lung Cancer Roundtable
SAN ANTONIO, Texas – December 3, 2025 – bioAffinity Technologies, Inc. (Nasdaq: BIAF; BIAFW), a biotechnology company advancing noninvasive diagnostics for lung cancer and other lung diseases, today announced that Chief Medical Officer Gordon Downie, MD, PhD, will present a poster at the American Cancer Society National Lung Cancer Roundtable (NLCRT) showcasing three cases in which CyPath® Lung, a noninvasive sputum-based flow cytometry test, successfully identified Stage 1A lung cancer in patients with atypical and diagnostically challenging presentations.
“Indeterminate pulmonary nodules pose a significant and growing clinical burden, especially when clinicians are confronted with conflicting or inconclusive diagnostic data,” Dr. Downie said. “Risk calculators, imaging, genetic testing, and biomarker tools can at times point in opposing directions, leaving clinicians and patients uncertain about next steps. This challenge is amplified in patients with unusual risk profiles, discordant imaging and advanced age.”
Dr. Downie’s poster, “CyPath® Lung in Practice: From Uncertainty to Clarity and Confidence,” details three complex cases from his tenure as Director of the Titus Regional Hospital Lung Nodule Clinic and Interventional Pulmonology. CyPath® Lung was used alongside other diagnostic tools, including standard low-dose CT (LDCT), PET imaging, risk calculators, bronchoscopy and blood serum marker tests. In each case, CyPath® Lung provided clarity and actionable results which led to confirmed diagnoses at the earliest and most treatable stage.
“These three cases illustrate scenarios that are increasingly common in real-world lung nodule practice,” Dr. Downie said. “Incorporating CyPath® Lung into the diagnostic pathway can accelerate diagnosis, guide difficult conversations with anxious patients, and prevent unnecessary invasive procedures that carry their own risks.”
The NLCRT is a coalition of 194 medical, public health, advocacy, government, and corporate organizations that work together to fight lung cancer by working collectively and collaboratively to reduce lung cancer mortality. This year’s annual meeting is December 8-9, 2025, at the Grand Hyatt Atlanta in Buckhead, Atlanta, Georgia.
Dr. Downie’s poster session is scheduled for Monday, December 8, 2025, from 2:35-3:15 p.m. ET. The poster can be viewed on the bioAffinity website.
| - |
About CyPath® Lung
CyPath® Lung uses proprietary advanced flow cytometry and artificial intelligence (AI) to identify cell populations in patient sputum that indicate malignancy. Automated data analysis helps determine if cancer is present or if the patient is cancer-free. CyPath® Lung incorporates a fluorescent porphyrin that is preferentially taken up by cancer and cancer-related cells. Clinical study results demonstrated that CyPath® Lung had 92% sensitivity, 87% specificity and 88% accuracy in detecting lung cancer in patients at high risk for the disease who had small lung nodules less than 20 millimeters. Diagnosing and treating early-stage lung cancer can improve outcomes and increase patient survival. For more information, visit www.cypathlung.com.
About bioAffinity Technologies, Inc.
bioAffinity Technologies, Inc. addresses the need for noninvasive diagnosis of early-stage cancer and other diseases of the lung and broad-spectrum cancer treatments. The Company’s first product, CyPath® Lung, is a noninvasive test that has shown high sensitivity, specificity and accuracy for the detection of early-stage lung cancer. CyPath® Lung is marketed as a Laboratory Developed Test (LDT) by Precision Pathology Laboratory Services, a subsidiary of bioAffinity Technologies. For more information, visit www.bioaffinitytech.com.
Forward-Looking Statements
Certain statements in this press release constitute “forward-looking statements” within the meaning of the federal securities laws. Words such as “may,” “might,” “will,” “should,” “believe,” “expect,” “anticipate,” “estimate,” “continue,” “predict,” “forecast,” “project,” “plan,” “intend” or similar expressions, or statements regarding intent, belief, or current expectations, are forward-looking statements. These forward-looking statements are subject to various risks and uncertainties, many of which are difficult to predict, that could cause actual results to differ materially from current expectations and assumptions from those set forth or implied by any forward-looking statements. Important factors that could cause actual results to differ materially from current expectations include, among others, the ability of CyPath® Lung to identify lung cancer and the other factors discussed in the Company’s Annual Report on Form 10-K for the year ended December 31, 2024, and its subsequent filings with the SEC, including subsequent periodic reports on Forms 10-Q and 8-K. Such forward-looking statements are based on facts and conditions as they exist at the time such statements are made and predictions as to future facts and conditions. While the Company believes these forward-looking statements are reasonable, readers of this press release are cautioned not to place undue reliance on any forward-looking statements. The information in this release is provided only as of the date of this release, and the Company does not undertake any obligation to update any forward-looking statement relating to matters discussed in this press release, except as may be required by applicable securities laws.
Contacts
bioAffinity Technologies
Julie Anne Overton
Director of Communications
jao@bioaffinitytech.com
| - |
Exhibit 99.2