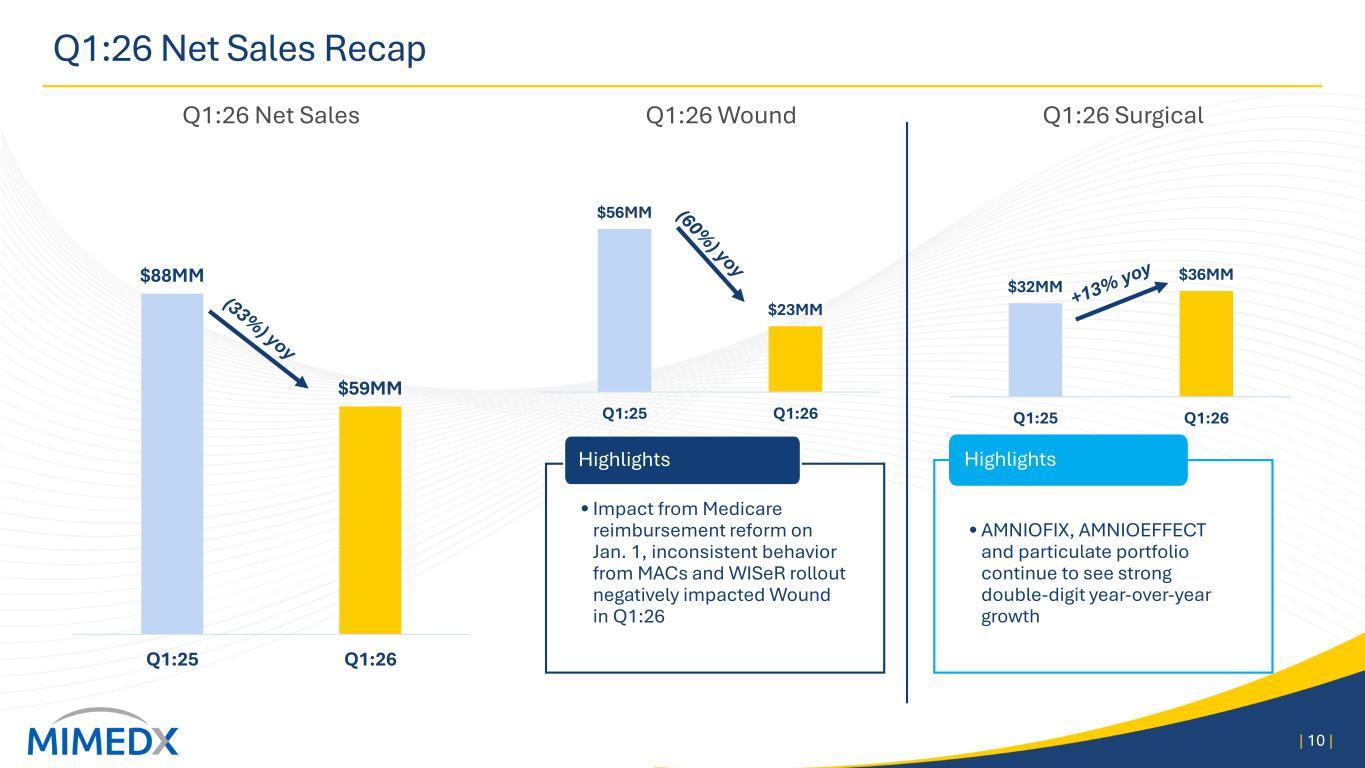
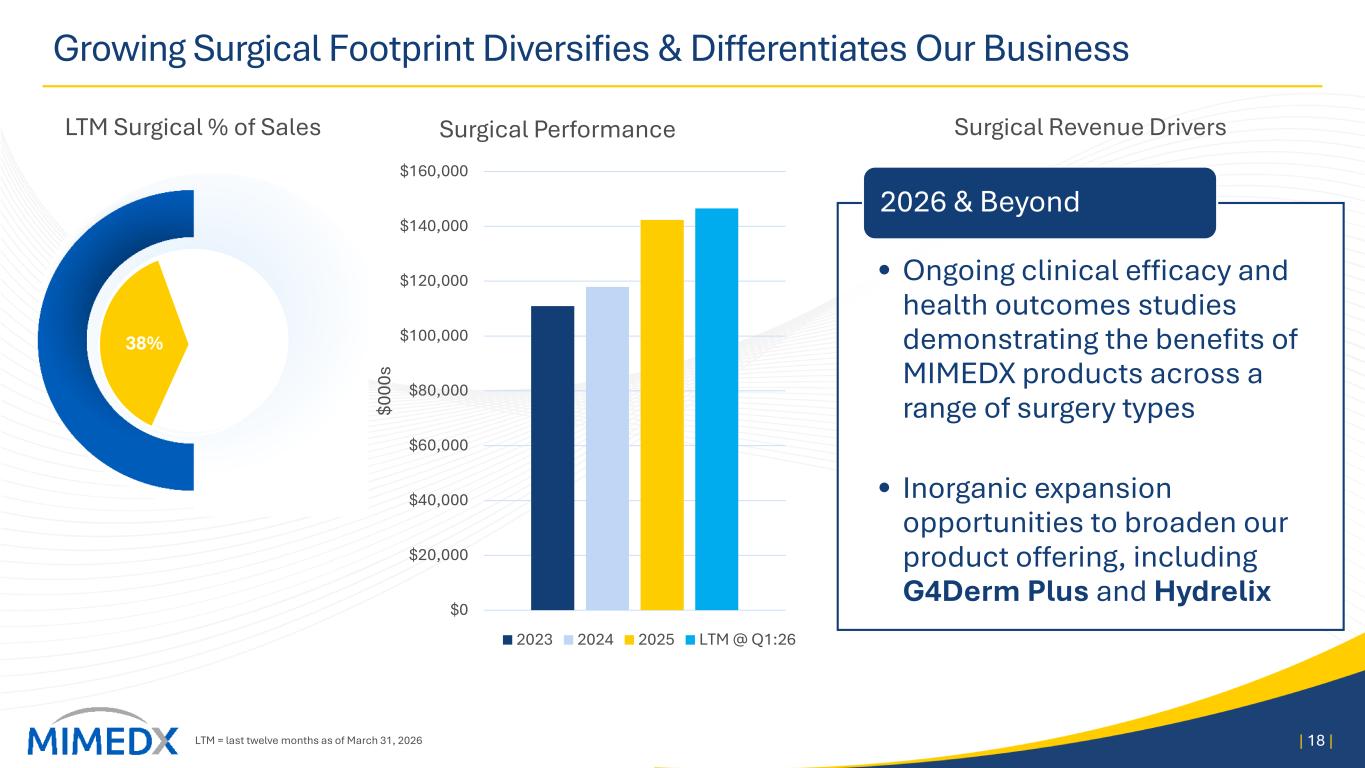
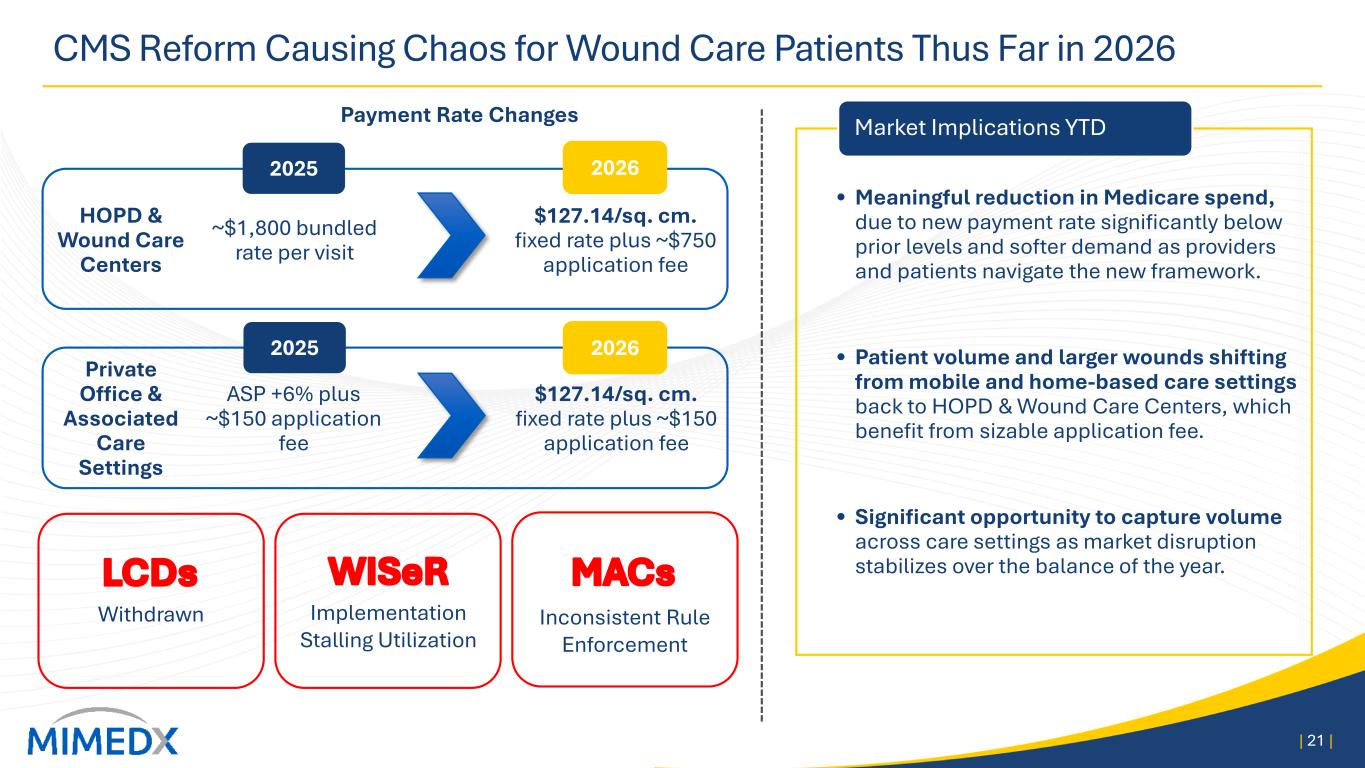
MIMEDX Announces First Quarter 2026 Operating & Financial Results Reports First Quarter Net Sales of $59 Million Revises 2026 Net Sales and Adjusted EBITDA Expectations Management to Host Conference Call Today, April 29, 2026, at 4:30 PM ET MARIETTA, Ga., April 29, 2026 -- MiMedx Group, Inc. (Nasdaq: MDXG) (“MIMEDX” or the “Company”), today announced operating and financial results for the first quarter 2026. Joseph H. Capper, MIMEDX Chief Executive Officer, commented, "The first quarter of 2026 was adversely impacted as new Medicare reimbursement policies in the advanced wound care space went into effect at the start of the year and led to significant confusion across the industry in nearly every care setting. Additionally, inconsistent implementation by the Medicare Administrative Contractors ("MACs") created even greater challenges for providers and their patients." Mr. Capper continued, "We generated $59 million in net sales in the first quarter of 2026. Our Surgical business continued to perform well, increasing 13% year over year and our Wound business declined 60% . As a result of disruption in the wound care market, we are lowering full-year 2026 net sales expectations to a range of $260 to $290 million. Furthermore, as announced a few weeks ago, we have taken steps to adjust the Company's cost structure in response to current market conditions. The $40 million in annualized savings we expect to generate from those actions will position us for a return to profitability over the balance of the year. After the market normalizes, we would anticipate generating double-digit top-line growth in 2027. “While some disruption was expected, the challenges we are now seeing in the market, coupled with irrational behavior by some industry participants, has added complexity that could not have been anticipated. To compound matters, the wound care market is adjusting to the new reimbursement rules at an extremely slow pace. We are working closely with our customers to help them adapt to the changes. Importantly, as we exited the quarter, we saw promising signs of volume recovery in Wound Care Centers and Hospitals. On the other side of this transition, we will be competing in a far more attractive space and believe MIMEDX is uniquely positioned to lead the market. Our Surgical franchise continues to post double-digit top-line growth, with 50% growth over the past three years," concluded Mr. Capper. First Quarter 2026 Results Discussion Net Sales MIMEDX reported net sales for the three months ended March 31, 2026 of $59 million, compared to $88 million for the three months ended March 31, 2025, a decrease of 33%. During the quarter, Surgical

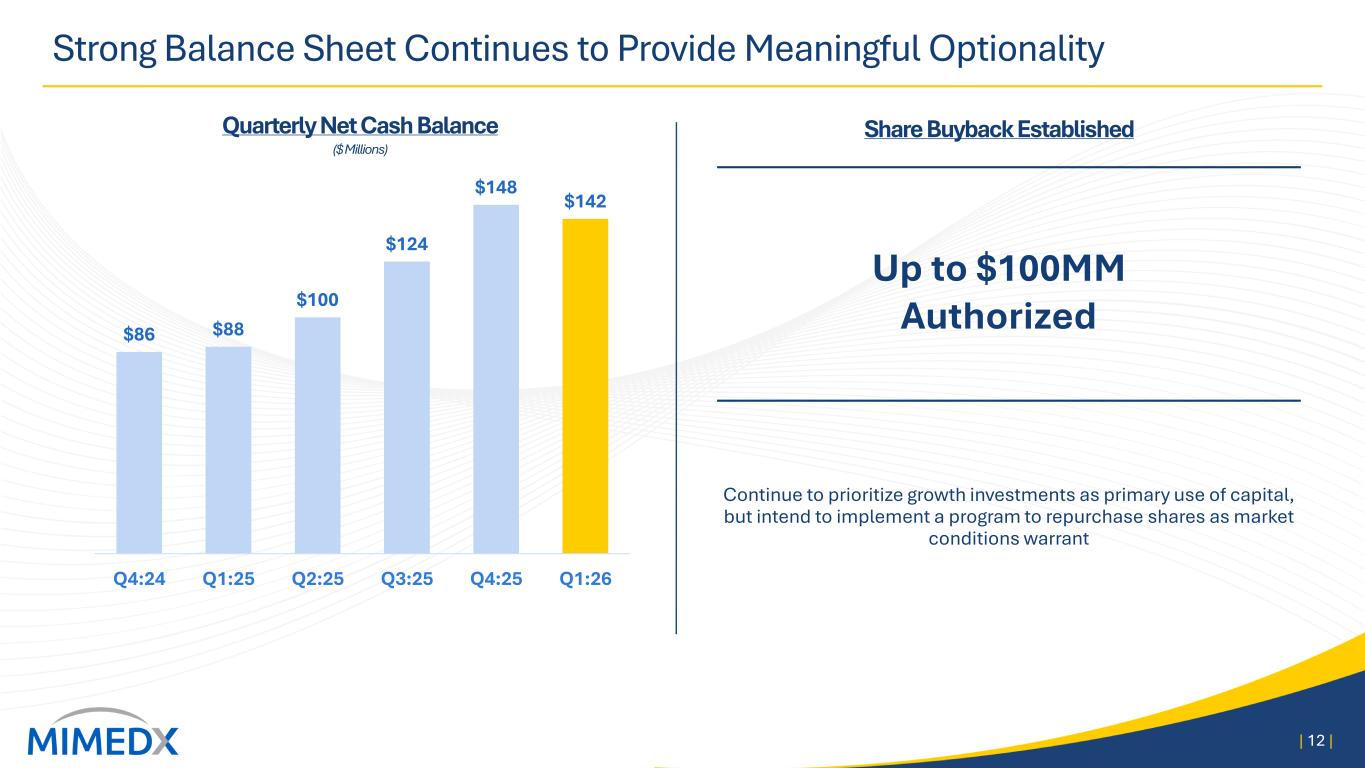
product sales increased 13% compared to the prior year period, led by sales of AMNIOFIX® and AMNIOEFFECT® and another quarter of double-digit growth of our particulate portfolio. These results were offset by a year-over-year decrease in Wound sales of 60%. The performance of the Wound business in the quarter was negatively impacted by several Medicare reimbursement changes that went into effect on January 1, most notably a payment cap on allowable reimbursement for skin substitutes on a per square centimeter basis, coupled with lagging marketplace adoption due to uncertainties around these rule changes. These pressures were primarily seen in the private office and associated care settings, which previously received Medicare reimbursement under the ASP +6% methodology. Sales of Wound products to Wound Care Centers and Hospitals, while also pressured due to the changing rules, showed modest signs of improvement as the quarter progressed. Gross Profit and Margin Gross profit for the three months ended March 31, 2026, was $42 million, compared to $72 million the prior year period. Gross margin for the three months ended March 31, 2026 was 71%, compared to 81% in the prior year period. The quarter-over-quarter decline in gross margin was driven primarily by the Medicare reimbursement rules negatively impacting the prices of our Wound products. Unfavorable product mix and other higher costs also contributed to the decline. Operating Expenses Selling, general and administrative ("SG&A") expenses for the three months ended March 31, 2026, were $53 million compared to $60 million for the three months ended March 31, 2025. The decrease in SG&A was driven primarily by a non-recurring reversal of stock-based compensation expense as well as lower commissions on lower sales. Research and development ("R&D") expenses for the three months ended March 31, 2026 and 2025, were $4 million and $3 million, respectively. R&D spend in the quarter reflects the randomized controlled trial for EPIEFFECT®, which recently completed enrollment, the launch of CHORIOFIX™ and ongoing investments in the development of future products in our pipeline. Net Income Net loss for the three months ended March 31, 2026 was $11 million compared to net income of $7 million for the three months ended March 31, 2025. Cash and Cash Equivalents As of March 31, 2026, the Company had $160 million of cash and cash equivalents compared to $166 million as of December 31, 2025. As of March 31, 2026, our cash position, net of debt on our balance sheet, was $142 million. Financial Outlook For 2026, MIMEDX now expects 2026 net sales to be in a range of $260 to $290 million. 2026 Adjusted EBITDA is expected to be approximately breakeven on a full year basis.

Longer-term, the Company continues to expect to achieve annual net sales growth in the low double-digits as a percentage with an adjusted EBITDA margin above 20%. Conference Call and Webcast MIMEDX will host a conference call and webcast to review its first quarter 2026 results on Wednesday, April 29, 2026, beginning at 4:30 p.m., Eastern Time. The call can be accessed using the following information: Webcast: Click here U.S. Investors: 877-407-6184 International Investors: 201-389-0877 Conference ID: 13759618 A replay of the webcast will be available for approximately 30 days on the Company’s website at www.mimedx.com following the conclusion of the event. Important Cautionary Statement This press release includes forward-looking statements, including statements regarding (i) our 2026 and longer term financial goals and expectations for future financial results, including revenue, net sales growth and Adjusted EBITDA margin; and (ii) any changes to underlying demand in the Wound segment, and (iii) the impact of our restructuring and cost reduction initiatives, including expected cost savings, on our future profitability and growth. Additional forward-looking statements may be identified by words such as "believe," "expect," "may," "plan," “goal,” “outlook,” "potential," "will," "preliminary," and similar expressions, and are based on management's current beliefs and expectations. Forward-looking statements are subject to risks and uncertainties, and the Company cautions investors against placing undue reliance on such statements. Actual results may differ materially from those set forth in the forward-looking statements. Factors that could cause actual results to differ from expectations include: (i) future sales are uncertain and are affected by competition, access to customers, patient access to healthcare providers, the new reimbursement environment, which introduced tighter coverage parameters, lower reimbursement levels in certain categories, and incremental administrative complexity for providers and many other factors; (ii) the Company may change its plans due to unforeseen or evolving circumstances and market response to the reimbursement rules; (iii) the results of scientific research are uncertain and may have little or no value; (iv) our ability to sell our products in other countries depends on a number of factors including adequate levels of reimbursement, market acceptance of novel therapies, and our ability to build and manage a direct sales force or third party distribution relationship; (v) the effectiveness of amniotic tissue as a therapy for particular indications or conditions is the subject of further scientific and clinical studies; (vi) we may alter the timing and amount of planned expenditures for research and development based on regulatory developments; (vii) Medicare spending; and (viii) changes in the size of the addressable market for our products. The Company describes additional risks and uncertainties in the Risk Factors section of its most recent

annual report and quarterly reports filed with the Securities and Exchange Commission. Any forward- looking statements speak only as of the date of this press release and the Company assumes no obligation to update any forward-looking statement. About MIMEDX MIMEDX is a pioneer and leader focused on helping humans heal. With more than a decade of helping clinicians manage chronic and other hard-to-heal wounds, MIMEDX is dedicated to providing a leading portfolio of products for applications in the wound care, burn, and surgical sectors of healthcare. The Company’s vision is to be the leading global provider of healing solutions through relentless innovation to restore quality of life. For additional information, please visit www.mimedx.com. Contact: Matt Notarianni Investor Relations 470.304.7291 mnotarianni@mimedx.com Selected Unaudited Financial Information

MiMedx Group, Inc. Condensed Consolidated Balance Sheets (in thousands) Unaudited March 31, 2026 December 31, 2025 ASSETS Current assets: Cash and cash equivalents $ 159,773 $ 166,121 Accounts receivable, net 46,034 75,707 Inventory 26,228 25,340 Other current assets 8,291 10,303 Total current assets 240,326 277,471 Property and equipment, net 4,756 4,713 Deferred tax asset, net 24,127 19,596 Goodwill 19,441 19,441 Intangible assets, net 13,140 14,158 Other assets 6,886 7,274 Total assets $ 308,676 $ 342,653 LIABILITIES AND STOCKHOLDERS’ EQUITY Current liabilities: Current portion of long term debt $ 1,500 $ 1,500 Accounts payable 11,464 14,528 Accrued compensation 14,524 31,065 Accrued expenses 11,008 11,383 Other current liabilities 6,054 5,790 Total current liabilities 44,550 64,266 Long term debt, net 16,094 16,467 Other liabilities 5,096 5,372 Total liabilities 65,740 86,105 Total stockholders' equity 242,936 256,548 Total liabilities and stockholders’ equity $ 308,676 $ 342,653
MiMedx Group, Inc. Condensed Consolidated Statements of Operations (in thousands, except share and per share amounts) Unaudited Three Months Ended March 31, 2026 2025 Net sales $ 58,991 $ 88,205 Cost of sales 17,368 16,558 Gross profit 41,623 71,647 Operating expenses: Selling, general and administrative 53,231 59,969 Research and development 4,140 3,328 Amortization of intangible assets 301 99 Operating income (16,049) 8,251 Other expense, net Interest income, net 886 506 Other expense, net (168) (145) (Loss) income before income tax (15,331) 8,612 Income tax provision 4,471 (1,589) Net (loss) income $ (10,860) $ 7,023 Basic net (loss) income per common share $ (0.07) $ 0.05 Diluted net (loss) income per common share $ (0.07) $ 0.05 Weighted average common shares outstanding - basic 148,446,017 147,272,324 Weighted average common shares outstanding - diluted 148,446,017 149,677,452 MiMedx Group, Inc. Condensed Consolidated Statements of Cash Flows (in thousands) Unaudited Three Months Ended March 31, 2026 2025 Net cash flows provided by operating activities 1,879 5,299 Net cash flows used in investing activities (5,642) (406) Net cash flows used in financing activities (2,585) (2,878) Net change in cash $ (6,348) $ 2,015 Reconciliation of Non-GAAP Measures In addition to our GAAP results, we provide certain non-GAAP measures including Adjusted EBITDA and related margins, Free Cash Flow, Adjusted Gross Profit, Adjusted Gross Margin, Adjusted Net Income, and Adjusted Earnings Per Share ("Adjusted EPS"). We believe that the presentation of these measures provides important supplemental information to management and investors regarding our performance. These measures are not a substitute for GAAP measures. Company management uses these non-GAAP

measures as aids in monitoring our ongoing financial performance from quarter-to-quarter and year-to- year on a regular basis and for benchmarking against comparable companies. These non-GAAP financial measures reflect the exclusion of the following items: • Share-based compensation - expense recognized related to awards to employees and our board of directors issued pursuant to our share-based compensation plans. This expense is reflected amongst cost of sales, research and development expense, and selling, general, and administrative expense in the unaudited condensed consolidated statements of operations. • Amortization of acquired intangible assets - reflects amortization expense recognized solely related to assets which were acquired as part of a transaction. These expenses are generally reflected in cost of sales in our unaudited condensed consolidated statements of operations. • Transaction-related expenses – reflects expenses incrementally incurred resulting from the consummation of material strategic transactions or the integration of acquired assets or operations into our core business. • Strategic legal and regulatory expenses - relates to litigation and regulatory expenses deemed strategically important to our operations. Litigation expenses primarily relate to legal fees incurred and relate to suits filed against former employees and their employers for violation of non-compete and non-solicitation agreements and certain patent infringement matters. Regulatory expenses relate to legal fees incurred stemming from action taken against the United States Food & Drug Administration ("FDA") surrounding the designation of one of our products. These expenses are generally reflected in selling, general and administrative expense in our unaudited condensed consolidated statements of operations. • Reorganization expense - expenses incurred toward the realignment of our operating strategy. These expenses relate to severance expenses related to certain executive leaders and other employment actions. These expenses are reflected as a component of selling, general, and administrative expense in the unaudited condensed consolidated statements of operations. • Income Tax Adjustment - for purposes of calculating Adjusted Net Income and Adjusted Earnings Per Share, reflects our expectation of a long-term effective tax rate, which is normalized and balance sheet-agnostic. Actual tax expense will be based on GAAP earnings, and may differ from the expected long-term effective tax rate due to a variety of factors, including the tax treatment of various transactions included in GAAP net income and other reconciling items that are excluded in determining Adjusted Net Income and Adjusted EPS. The actual long-term normalized effective tax rate was 25% for each of the quarters ended March 31, 2026 and 2025.

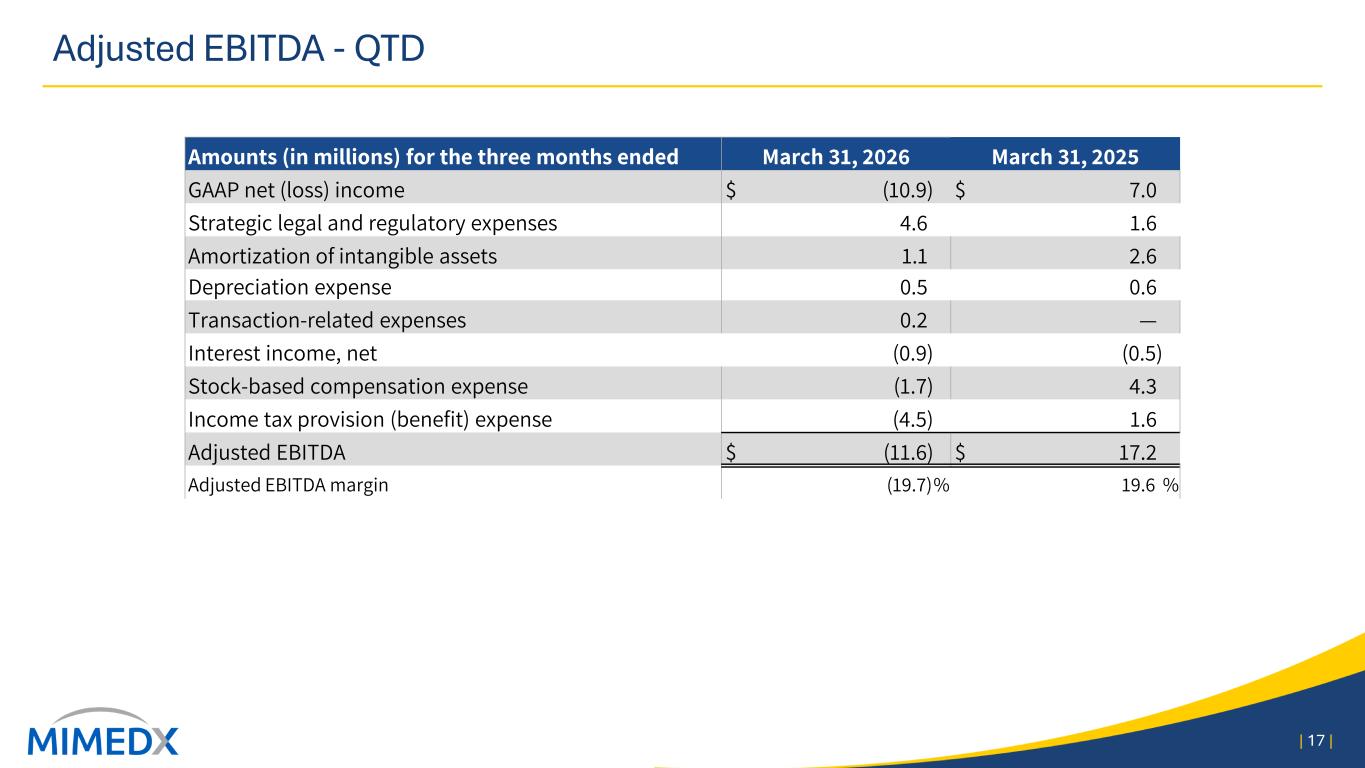
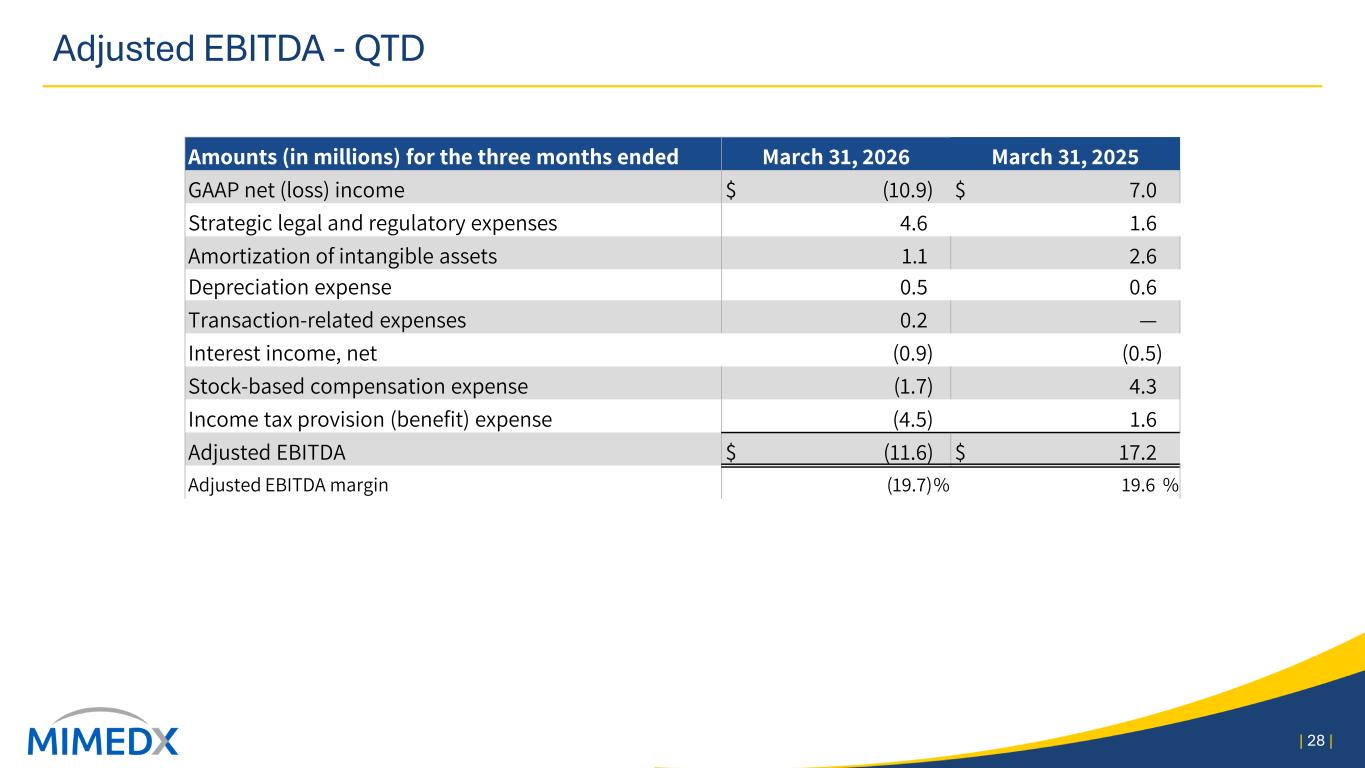
Adjusted EBITDA and Adjusted EBITDA margin Adjusted EBITDA consists of GAAP net income excluding (i) share-based compensation, (ii) income tax provision, (iii) amortization of intangible assets (iv) strategic legal and regulatory expenses, (v) interest (income) expense, net, (vi) depreciation expense, (vii) reorganization expenses, and (viii) transaction- related expenses. Please refer to the tables at the beginning of this press release for reconciliation to GAAP net income. Three Months Ended March 31, 2026 2025 Net (loss) income $ (10,860) $ 7,023 Non-GAAP Adjustments: Strategic legal and regulatory expenses 4,555 1,645 Amortization of intangible assets 1,101 2,646 Depreciation expense 527 558 Transaction related expenses 176 7 Reorganization expenses (7) — Interest (income) expense, net (886) (506) Share-based compensation (1,697) 4,259 Income tax provision (4,471) 1,589 Adjusted EBITDA $ (11,562) $ 17,221 Adjusted EBITDA margin (19.6) % 19.5 % Adjusted Net Income Adjusted Net Income provides a view of our operating performance, exclusive of certain items which are non-recurring or not reflective of our core operations. Adjusted Net Income is defined as GAAP net income plus (i) amortization of acquired intangible assets, (ii) strategic legal and regulatory expenses, (iii) reorganization expenses, (iv) transaction related expenses, and (v) the long-term effective income tax rate adjustment. A reconciliation of GAAP net income to Adjusted Net Income appears in the table below (in thousands): Three Months Ended March 31, 2026 2025 Net (loss) income $ (10,860) $ 7,023 Strategic legal and regulatory expenses 4,555 1,645 Amortization of acquired intangible assets 799 2,547 Transaction related expenses 176 7 Reorganization expenses (7) — Long-term effective income tax rate adjustment (2,019) (1,614) Adjusted net (loss) income $ (7,356) $ 9,608

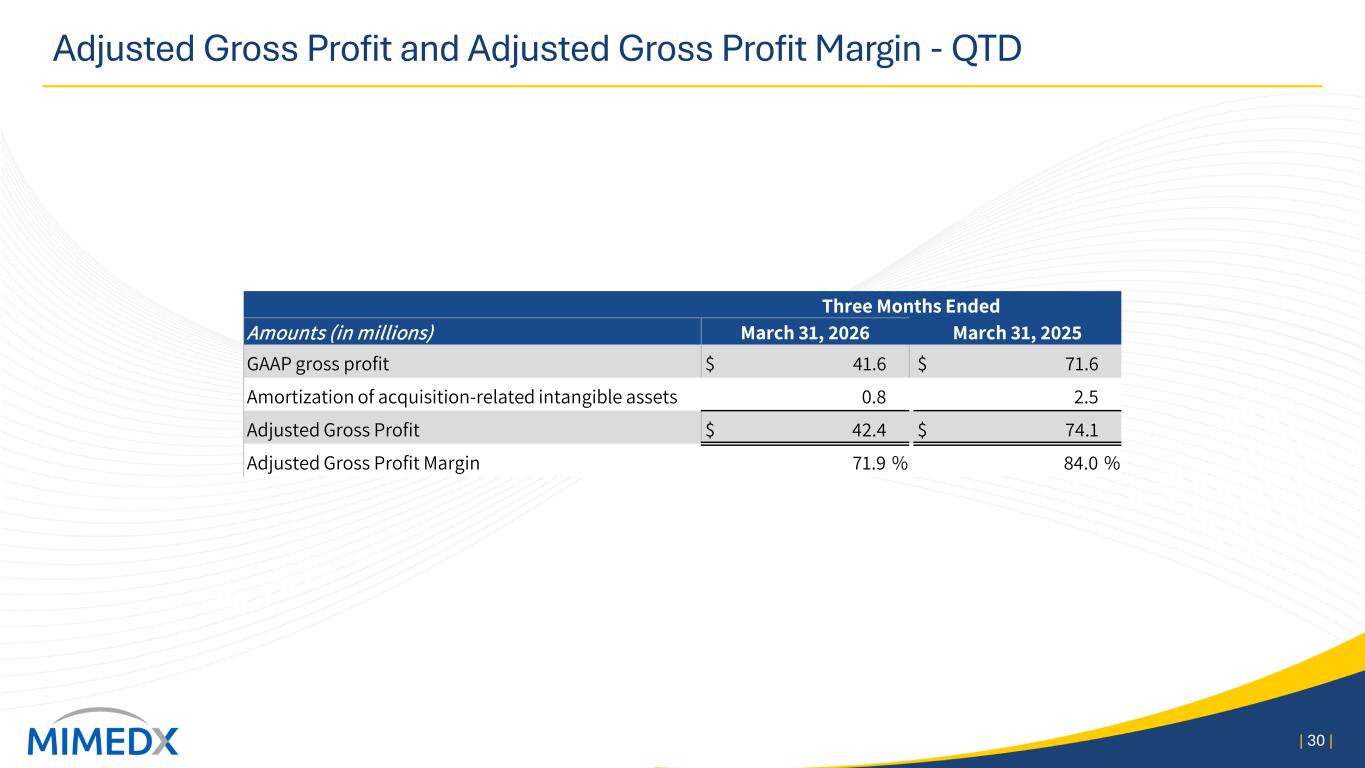
A reconciliation of various line items included in our GAAP unaudited condensed consolidated statements of operations to Adjusted Net Income for the three months ended March 31, 2026 and 2025 are presented in the tables below (in thousands): Three Months Ended March 31, 2026 Gross Profit Selling, General & Administrative Expense Research and Development Expense Net Loss Reported GAAP Measure $ 41,623 $ 53,231 $ 4,140 $ (10,860) Amortization of acquired intangible assets 799 — — 799 Strategic legal and regulatory expenses — (4,555) — 4,555 Reorganization expenses — 7 — (7) Transaction related expenses — (145) — 176 Long-term effective income tax rate adjustment — — — (2,019) Non-GAAP Measure $ 42,422 $ 48,538 $ 4,140 $ (7,356) Gross Profit Margin 70.6 % Gross Profit Margin, as adjusted 71.9 % Three months ended March 31, 2025 Gross Profit Selling, General & Administrative Expense Research and Development Expense Net Income Reported GAAP Measure $ 71,647 $ 59,969 $ 3,328 $ 7,023 Amortization of acquired intangible assets 2,547 — — 2,547 Strategic legal and regulatory expenses — (1,645) — 1,645 Transaction related expenses — — 7 Long-term effective income tax rate adjustment — — — (1,614) Non-GAAP Measure $ 74,194 $ 58,324 $ 3,328 $ 9,608 Gross Profit Margin 81.2 % Gross Profit Margin, as adjusted 84.1 % Adjusted Earnings Per Share Adjusted Earnings Per Share is intended to provide a normalized view of earnings per share by removing items that may be irregular, one-time, or non-recurring from net income. This enables us to identify underlying trends in our business that could otherwise be masked by such items. Adjusted Earnings Per Share consists of GAAP diluted net income per common share including adjustments for (i) amortization of acquired intangible assets, (ii) strategic legal and regulatory expenses, (iii) transaction-related expenses, and (iv) the long-term effective income tax rate adjustment. A reconciliation of GAAP diluted earnings per share to Adjusted Earnings Per Share appears in the table below (per diluted share):

Three Months Ended March 31, 2026 2025 GAAP net (loss) income per common share - diluted $ (0.07) $ 0.05 Amortization of acquired intangible assets 0.01 0.02 Strategic legal and regulatory expenses 0.03 0.00 Reorganization expenses 0.00 0.00 Transaction related expenses 0.00 0.00 Long-term effective income tax rate adjustment (0.02) (0.01) Adjusted Earnings Per Share (0.05) 0.06 Weighted average common shares outstanding - adjusted 148,446,017 149,677,452 Free Cash Flow Free Cash Flow is intended to provide a measure of our ability to generate cash in excess of capital investments. It provides management with a view of cash flows which can be used to finance operational and strategic investments. Free Cash Flow is defined as net cash provided by operating activities less capital expenditures, including purchases of equipment. A reconciliation of GAAP net cash flows provided by operating activities to Free Cash Flow appears in the table below (in thousands): Three Months Ended March 31, 2026 2025 Net cash flows provided by operating activities $ 1,879 $ 5,299 Capital expenditures, including purchases of equipment (570) (377) Free Cash Flow $ 1,309 $ 4,922 Other Information Net Sales by Product Category by Quarter Below is a summary of net sales by product category (in thousands): Three Months Ended March 31, 2026 2025 Surgical $ 36,374 $ 32,132 Wound 22,617 56,073 Net sales $ 58,991 $ 88,205