UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 20-F
(Mark One)
☐ REGISTRATION STATEMENT PURSUANT TO SECTION 12(b) OR 12(g) OF THE SECURITIES EXCHANGE ACT OF 1934
or
☒ ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
For the fiscal year ended December 31, 2025
or
☐ TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
or
☐ SHELL COMPANY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
Date of event requiring this shell company report
For the transition period from ___________________________ to ___________________________
Commission file number 001-42004
NEWGENIVFGROUP LIMITED
(Exact name of Registrant as specified in its charter)
(Translation of Registrant’s name into English)
British Virgin Islands
(Jurisdiction of incorporation or organization)
36/39-36/40, 13th Floor, PS Tower
Sukhumvit 21 Road (Asoke)
Khlong Toei Nuea Sub-district
Watthana District, Bangkok 10110, Thailand
(Address of Principal Executive Offices)
Mr. Wing Fung Alfred Siu, Chief Executive Officer
1/F, Pier 2, Central
Hong Kong, 999077
Tel: +1 (212) 537-4406
Email: Newgenivf.IR@icrinc.com
(Name, Telephone, E-mail and/or Facsimile number and Address of Company Contact Person)
Securities registered or to be registered pursuant to Section 12(b) of the Act.
| Title of each class | Trading Symbol | Name of each exchange on which registered | ||
| Class A ordinary shares, no par value per share | NIVF | The Nasdaq Stock Market LLC (The Nasdaq Capital Market) | ||
| Warrants to purchase Class A ordinary shares | NIVFW | The Nasdaq Stock Market LLC (The Nasdaq Capital Market) |
Securities registered or to be registered pursuant to Section 12(g) of the Act.
(Title of Class)
Securities for which there is a reporting obligation pursuant to Section 15(d) of the Act.
(Title of Class)
Indicate the number of outstanding shares of each of the issuer’s classes of capital or common stock as of the close of the period covered by the annual report.
The number of NewGenIvf Group Limited’s outstanding: (i) Class A Ordinary Shares, no par value, was 240,622 as of December 31, 2025, after retrospective application of the reverse stock splits in February 2025, May 2025, August 2025, December 2025, January 2026 and March 2026; (ii) Class B Ordinary Shares, no par value, was 380 as of December 31, 2025, after retrospective application of the reverse stock splits in February 2025, May 2025, August 2025, December 2025, January 2026 and March 2026; and (iii) Preferred Shares, no par value, was none as of December 31, 2025.
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.
| ☐ Yes ☒ No |
If this report is an annual or transition report, indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934.
| ☐ Yes ☒ No |
Note – Checking the box above will not relieve any registrant required to file reports pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 from their obligations under those Sections.
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
| ☒ Yes ☐ No |
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files).
| ☒ Yes ☐ No |
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or an emerging growth company. See definition of “large accelerated filer, “accelerated filer,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer ☐ | Accelerated filer ☐ | Non-accelerated filer ☒ |
| Emerging growth company ☒ |
If an emerging growth company that prepares its financial statements in accordance with U.S. GAAP, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards† provided pursuant to Section 13(a) of the Exchange Act. ☐
| † | The term “new or revised financial accounting standard” refers to any update issued by the Financial Accounting Standards Board to its Accounting Standards Codification after April 5, 2012. |
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☐
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive- based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate by check mark which basis of accounting the registrant has used to prepare the financial statements included in this filing:
| U.S. GAAP ☒ | International Financial Reporting Standards as issued by the International Accounting Standards Board ☐ | Other ☐ |
If “Other” has been checked in response to the previous question, indicate by check mark which financial statement item the registrant has elected to follow. ☐ Item 17 ☐ Item 18
If this is an annual report, indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). ☐ Yes ☒ No
(APPLICABLE ONLY TO ISSUERS INVOLVED IN BANKRUPTCY PROCEEDINGS DURING THE PAST FIVE YEARS)
Indicate by check mark whether the registrant has filed all documents and reports required to be filed by Sections 12, 13 or 15(d) of the Securities Exchange Act of 1934 subsequent to the distribution of securities under a plan confirmed by a court. ☐ Yes ☐ No In this annual report, except where the context otherwise requires and for purposes of this annual report only:
TABLE OF CONTENTS
INTRODUCTION
“ASCA” means A SPAC I Acquisition Corp., a British Virgin Islands business company.
“A SPAC I Mini Acquisition Corp.” means A SPAC I Mini Acquisition Corp., a British Virgin Islands business company.
“Business Combination” means the transactions contemplated by the Merger Agreement, pursuant to which (i) ASCA reincorporated to the British Virgin Islands by merging with and into the Company; and (ii) Merger Sub merged with and into Legacy NewGenIvf, resulting in Legacy NewGenIvf being a wholly-owned subsidiary of the Company.
“BVI” means British Virgin Islands.
“Class A Ordinary Share” means Class A ordinary shares of the Company, no par value.
“Class B Ordinary Share” means Class B ordinary shares of the Company, no par value.
“Preferred Ordinary Shares” means Preferred ordinary shares of the Company, no par value.
“Closing” means the consummation of the Business Combination, which occurred on April 3, 2024.
“Company” means NewGenIvf Group Limited, a British Virgin Islands business company, the surviving entity of the Business Combination.
“Reverse Stock Splits” means the First Reverse Stock Split, the Second Reverse Stock Split, the Third Reverse Stock Split, the Fourth Reverse Stock Split, the Fifth Reverse Stock Split and the Sixth Reverse Stock Split. In this Form 20F, where we state historical share and per-share numbers, we have, where appropriate, reflected a retroactive adjustment due to the Reverse Stock Splits in parentheses.
“First Reverse Stock Split” means the 1-for-20 reverse stock split effected by the Company on February 11, 2025.
“Second Reverse Stock Split” means the 1-for-10 reverse stock split effected by the Company on May 5, 2025.
“Third Reverse Stock Split” means the 1-for-5 reverse stock split effected by the Company on August 4, 2025.
“Fourth Reverse Stock Split” means the 1-for-5 reverse stock split effected by the Company on December 1, 2025.
“Fifth Reverse Stock Split” means the 1-for-3 reverse stock split effected by the Company on January 26, 2026.
“Sixth Reverse Stock Split” means the 1-for-4 reverse stock split effected by the Company on March 16, 2026.
“Legacy NewGenIvf” means NewGenIvf Limited, a Cayman Islands exempted company, which became a wholly owned subsidiary of ASCA upon the Closing.
“Merger Agreement” means the Merger Agreement entered into on February 15, 2023, and as amended on June 12, 2023 and December 6, 2023, between ASCA, A SPAC I Mini Acquisition Corp., Merger Sub, Legacy NewGenIvf, and certain shareholders of Legacy NewGenIvf, pursuant to which the Reincorporation Merger and Acquisition Merger were consummated.
“Merger Sub” means A SPAC I Mini Sub Acquisition Corp., a Cayman Islands exempted company and former wholly-owned subsidiary of A SPAC I Mini Acquisition Corp.
“NewGenIvf” means NewGenIvf Group Limited, a British Virgin Islands business company, the surviving entity of the Business Combination, unless the context so requires.
“Preferred Shares” means preferred shares of the Company, no par value.
“Reincorporation Merger” means the first step of the Business Combination which occurred pursuant to the Merger Agreement, in which ASCA reincorporated to the British Virgin Islands by merging with and into NewGenIvf.
“US$,” “USD,” “U.S. dollars,” or “dollars” are to the legal currency of the United States.
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 20-F (including information incorporated by reference herein, the “Report”) is being filed by NewGenIvf Group Limited, a British Virgin Islands business company. Unless otherwise indicated, “we,” “us,” “our,” the “Company,” “NewGenIvf” and similar terminology refer to NewGenIvf Group Limited and its subsidiaries. References to “Legacy NewGenIvf” and “NewGenIvf Limited” refer to NewGenIvf Limited, a Cayman Islands exempted company which existed before the Business Combination.
This Report contains or may contain forward-looking statements as defined in Section 27A of the Securities Act of 1933, as amended (the “Securities Act”), and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”) that involve significant risks and uncertainties. All statements other than statements of historical facts are forward-looking statements. These forward-looking statements include information about our possible or assumed future results of operations or our performance.
Words such as “expects,” “intends,” “plans,” “believes,” “anticipates,” “estimates,” and variations of such words and similar expressions are intended to identify the forward-looking statements. The risk factors and cautionary language referred to or incorporated by reference in this Report provide examples of risks, uncertainties and events that may cause actual results to differ materially from the expectations described in our forward-looking statements.
Readers are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date of this Report. Although we believe that the expectations reflected in such forward-looking statements are reasonable, there can be no assurance that such expectations will prove to be correct. These statements involve known and unknown risks and are based upon a number of assumptions and estimates which are inherently subject to significant uncertainties and contingencies, many of which are beyond our control. Actual results may differ materially from those expressed or implied by such forward-looking statements. We undertake no obligation to publicly update or revise any forward-looking statements contained in this Report, or the documents to which we refer readers in this Report, to reflect any change in our expectations with respect to such statements or any change in events, conditions or circumstances upon which any statement is based.
PART I
Item 1. Identity of Directors, Senior Management and Advisers
Not applicable.
Item 2. Offer Statistics and Expected Timetable
Not applicable.
Item 3. Key Information
| A. | [Reserved] |
| B. | Capitalization and indebtedness |
Not applicable.
| C. | Reasons for the offer and use of proceeds |
Not applicable.
| D. | Risk Factors |
Risks Related to NewGenIvf’s Business and Industry
We may not be able to continue operating as a going concern.
As of December 31, 2025, the Company’s cash and cash equivalents stood at approximately US$758,621. While the Company does not have immediate challenges to settle its obligations when payments become due, the Company can make no assurance that it will have sufficient capital to bridge potential financial and liquidity shortfalls.
The Company is always closely monitoring the market for opportunities and has also been carrying out various fundraising projects to improve the Company’s cash flow position. As of March 30, 2026, convertible promissory notes of $5,266,000 remain outstanding. Moreover, the Company has access to an equity line of credit facility of up to US$100,000,000 from White Lion Capital, of which approximately US$15.55 million has been drawn and become equity to date.
The Company can make no assurance that required financing will be available for the amounts needed, or on terms commercially acceptable to the Company, if at all. If one or all of these events does not occur or subsequent capital raises are insufficient to bridge financial and liquidity shortfall, there would likely be a material adverse effect on the Company and its financial statements.
The consolidated financial statements do not reflect adjustments that would be necessary if the going concern basis was not appropriate. If the going concern basis was not appropriate for these consolidated financial statements, then adjustments would be necessary in the carrying value of the assets and liabilities, the reported revenues and expenses, and the balance sheet classifications used. These adjustments could be material.
We may issue additional Ordinary Shares through our equity line of credit facility, upon the exercise of outstanding warrants, the convertible notes, upon the exercise of the options granted pursuant to the Share Incentive Plan, and/or through future issuances of equity or convertible debt securities, all of which would increase the number of shares eligible for future resale in the public market and result in dilution to our shareholders.
On August 7, 2024, the Company entered into a securities purchase agreement with JAK Opportunities VI LLC (“JAK”), pursuant to which, amongst other things: (i) the Company agreed to sell, at an initial closing, (a) a senior convertible note in the aggregate original principal amount not exceeding $1,100,000, convertible into Class A Ordinary Shares pursuant to its terms, (b) a warrant to purchase 1,325,301 Class A Ordinary Shares (22 Shares after adjusted for all the Reverse Stock Splits done as of this Report), and (c) a warrant to purchase 180,722 Class A Ordinary Shares (3 Shares after adjusted for all the Reverse Stock Splits done as of this Report); and (ii) the Company may require JAK (or JAK may require the Company, as applicable) to participate in the sale of (a) one or more additional convertible notes (which aggregate original principal amount for all additional convertible notes shall not exceed $9,500,000) and (b) related warrants.
On November 21, 2024, the Company entered into a Common Shares Purchase Agreement (the “White Lion Purchase Agreement”) with White Lion Capital, LLC (“White Lion”). Pursuant to the White Lion Purchase Agreement, the Company has the right, but not the obligation, to require White Lion to purchase, from time to time, up to $100,000,000 in aggregate gross purchase price of newly issued Class A Ordinary Shares, with an automatic increase to $300,000,000 upon any substantial M&A or Material Transaction (as defined in the White Lion Purchase Agreement) and a further option to increase to $500,000,000 after $250,000,000 has been issued and sold to White Lion under the White Lion Purchase Agreement, subject to certain limitations and conditions set forth in the White Lion Purchase Agreement.
On April 1, 2025, the Company entered into a new securities purchase agreement with JAK, pursuant to which, amongst other things: (i) the Company agreed to sell, at an initial closing, a senior convertible note in the aggregate original principal amount not exceeding $3,200,000, convertible into Class A Ordinary Shares pursuant to its terms; and (ii) the Company may require JAK (or JAK may require the Company, as applicable) to participate in the sale of one or more additional convertible notes (which aggregate original principal amount for all additional convertible notes shall not exceed $25,600,000).
On October 15, 2025, November 4, 2025, and January 22, 2026, the Company entered into three securities purchase agreements (“Vanquish SPAs”) with Vanquish Funding Group Inc. (“Vanquish”). Pursuant to the terms of the Vanquish SPAs, the Company issued three convertible promissory notes to Vanquish in the principal amounts of $257,000, $157,000 and $107,000, respectively.
On November 12, 2025, the Company entered into a securities purchase agreement (“Labrys SPA”) with Labrys Fund II, L.P. (“Labrys”). Pursuant to the terms of the Labrys SPA, the Company issued a convertible promissory note to Labrys in the principal amount of $250,000.
On January 22, 2026, the Company entered into a Securities Purchase Agreement with Boot Capital LLC, pursuant to which, amongst other things, the Company agreed to sell a convertible note in the aggregate principal amount of $50,000, convertible into Class A Ordinary Shares pursuant to its terms.
On March 17, 2026, the Company entered into a Securities Purchase Agreement with CFI Capital LLC, pursuant to which, amongst other things, the Company agreed to sell a convertible note in the aggregate principal amount of $200,000, convertible into Class A Ordinary Shares pursuant to its terms.
On March 25, 2026, the Company entered into a Securities Purchase Agreement with Firstfire Global Opportunities Fund, LLC, pursuant to which, amongst other things, the Company agreed to sell a convertible note in the aggregate principal amount of $220,000, convertible into Class A Ordinary Shares pursuant to its terms.
In addition, the Company also adopted the Share Incentive Plan, under which 113,703 Ordinary Shares of the Company are available for awards.
As of March 30, 2026, the Company has (i) convertible promissory notes of $5,266,000; (ii) 1,224,328 warrants; (iii) 113,703 unallocated shares which may be issuable pursuant to awards under the Share Incentive Plan, remain outstanding. Moreover, under the equity line of credit facility of up to US$100,000,000 from White Lion, approximately US$15.55 million has been drawn and become equity to date.
The potential issuance of additional Ordinary Shares through the equity line of credit facility, upon the exercise of outstanding warrants, the convertible notes, upon the exercise of the options granted pursuant to the Share Incentive Plan will result in dilution to our shareholders and increase the number of Ordinary Shares eligible for resale in the public market. Sales of substantial numbers of such securities in the public market or the fact that such securities may be exercised could adversely affect the market price of our securities. Furthermore, we may issue additional equity or convertible debt securities in the future which may dilute our existing shareholders’ holdings.
Existing shareholders will have no pre-emptive rights in connection with such further issuances. We cannot predict the size or nature of future issuances or the effect that future issuances and sales of our Ordinary Shares will have on the market price of our Ordinary Shares. Issuances of a substantial number of additional Ordinary Shares, or the perception that such issuances could occur, may adversely affect prevailing market prices for our Ordinary Shares. With any additional issuance of our Ordinary Shares, our investors will suffer dilution to their voting power and economic interest.
The fertility market in which NewGenIvf participates is competitive, and if NewGenIvf does not continue to compete effectively, its results of operations could be materially and adversely affected.
The market for NewGenIvf’s solutions is competitive and is likely to attract increased competition, which could make it hard for it to succeed. NewGenIvf faces significant competition from other fertility companies and other players in the fertility market. Some of NewGenIvf’s competitors are more established, have a longer operating history and a larger client base, benefit from greater brand recognition and have substantially greater financial, technical and marketing resources than NewGenIvf does. NewGenIvf’s competitors may compete with NewGenIvf in a variety of ways, including seeking to develop or integrating solutions and services that may become more efficient or appealing to NewGenIvf’s existing and potential clients, achieving superior clinical outcomes, having access to a network of more high-quality fertility specialists, establishing more comprehensive data reporting and sharing systems, conducting brand promotions and other marketing activities, and making investments in and acquisitions of NewGenIvf’s business partners. While NewGenIvf believes that one of its key competitive advantages is its ability to provide a broad range of services, and NewGenIvf does not believe any competitors have developed a similar broad range services in Asia Pacific at this time, current or future competitors may be successful in doing so in the future. If current or future competitors are successful at developing a similar broad range of services, NewGenIvf’s financial performance may be negatively impacted.
In addition, NewGenIvf believes that there is growing awareness of the demand for fertility services. As the fertility services field gains more attention, more competitors may be drawn into the market. NewGenIvf also could be adversely affected if NewGenIvf fails to identify or effectively respond to changes in market dynamics. As a result of any of these factors, NewGenIvf may not be able to continue to compete successfully against its current or future competitors, and this competition could result in the decrease in its clients base and market share and the failure of its platform to continue to maintain market acceptance, which would materially and adversely affect its business, financial condition and results of operations.
NewGenIvf has a limited operating history with its current platform of solutions, which makes it difficult to predict its future prospects, financial performance and results of operations.
The predecessor entity of the Company prior to the Business Combination in April of 2024, NewGenIvf Limited, a Cayman Islands exempted company, was established in 2019, and although its subsidiary First Fertility PGS Center Limited launched fertility services in 2014, has a limited operating history. As a result of its limited operating history with its current platform of solutions, as well as a limited amount of time serving a majority of its client base, its ability to accurately forecast its future results of operations, key operating data, net revenue, cash flows, and operating margins is limited and subject to a number of uncertainties, including its ability to plan for and model future growth. NewGenIvf’s historical revenue growth should not be considered indicative of its future performance. Further, in future periods, its revenue growth could slow or decline for a number of reasons, including risks, challenges and uncertainties that NewGenIvf has encountered and may continue to encounter that are frequently experienced by companies at an early stage, slowing demand for its solutions and fertility services in general, changes in utilization trends by its clients, general economic slowdown, an increase in unemployment, an increase in competition, changes to health care trends and regulations, changes to science relating to the fertility market, a decrease in the growth of the fertility market, or its failure, for any reason, to continue to take advantage of growth opportunities. If NewGenIvf’s assumptions regarding these risks and uncertainties and its future revenue growth are incorrect or change, or if it does not address these risks successfully, its operating and financial results could differ materially from its expectations, and its business could suffer.
NewGenIvf is undergoing a significant and rapid business transformation, which involves substantial risks and may not be successful.
NewGenIvf’s recent and planned strategic initiatives represent a fundamental shift from our historical focus on fertility services to a highly diversified conglomerate with operations in real estate development, digital asset management, and mining. This transformation involves numerous risks, including our inability to manage multiple, unrelated business lines, the failure of these new ventures to generate expected revenues or profits, the diversion of management’s attention from our core operations, and significant unanticipated costs. Our lack of operating history in these new sectors makes it difficult to forecast future performance, and there can be no assurance that this diversification strategy will enhance shareholder value or that we will successfully execute this transformation.
NewGenIvf’s new strategic initiatives may place significant strain on the Company’s management, and operational resources.
The rapid expansion into multiple new and complex business areas will require significant management attention and financial resources. Our senior management has limited experience in the mining, large-scale real estate development, and digital asset tokenization industries. Our ability to manage this growth effectively, recruit and retain qualified personnel with the requisite expertise, and maintain a cohesive corporate culture across diverse business lines is unproven. Any failure to manage this growth could lead to operational inefficiencies, control failures, and a material adverse effect on our business and results of operations.
Our planned Solana strategy may not be successful and exposes us to various risks, including risks associated with the Solana network, SOL token and the blockchain sector.
On June 2, 2025, we announced plans to invest up to US$30 million in staking the digital asset SOL (the native token of the Solana network) and funding such investment using our existing credit facilities. We intend to systematically deploy our available capital to acquire SOL from time to time over a period of 18 months, as further described below. As of the date of this prospectus, we hold 10,000.23 SOL tokens and have not begun staking SOL.
“Staking SOL” refers to the process of delegating our acquired SOL tokens to independent validator nodes on the Solana network to participate in its proof-of-stake consensus mechanism. We are currently evaluating various validators with a view to establishing relationships for staking SOL. In return for this delegation, we earn staking rewards, which are distributed approximately every 2-3 days (each period known as an “epoch”). This strategy is intended to generate returns through (i) these staking rewards, currently yielding approximately 6.78% Annual Percentage Yield, and (ii) potential appreciation in the value of the SOL tokens we hold. Our primary costs include validator commissions (typically 0-10% of rewards), network transaction fees, custodial service fees, and, if credit facilities are used for funding, interest expense on such credit facilities.
We have structured our Solana strategy in phases:
| ● | Phase 1 (completed June-July 2025): We acquired an initial position of 6,703.99 SOL (approximately $1.24 million at acquisition) as an operational pilot. |
| ● | Phase 2 (Q3 2025 – Q4 2026): We plan to systematically deploy the remaining capital (approximately $28.76 million) to acquire SOL using a dollar-cost averaging methodology to mitigate market timing risk. In addition, during this phase, we will evaluate various validators and establishing validator relationships to begin staking SOL. We intend to stake a majority of our SOL holdings. |
| ● | Phase 3 (ongoing): we will actively manage our validator relationships and reward optimization. |
As of the date of this filing, we hold 10,000.23 SOL tokens, with a total value of approximately $0.84 million and an unrealized loss of approximately $1.397 million. As we have not begun staking, we have not earned any staking rewards yet. However, our past performance is not indicative of future results, and we face numerous material risks that could prevent us from realizing our strategy or result in a complete or partial loss of our investment, including:
| ● | Price Volatility of SOL: SOL is a highly volatile cryptocurrency asset that has traded at a low of US$68,04 and a high of $253.38 per SOL in the 12 months preceding the date of this Form 20-F. The trading price of SOL has been highly volatile during prior periods, experiencing multiple previous significant decreases, and such declines may occur again in the future. A decline in the price of SOL could rapidly erase any staking rewards earned and result in a substantial loss of our principal capital. |
| ● | Staking and Liquidity Risks: The Solana network imposes an “unbonding” period of several days when we wish to stop staking and liquidate our tokens. This lock-up period limits our ability to react to adverse market conditions, subjecting us to significant market timing risk and potential illiquidity during periods of volatility. |
| ● | Operational and Validator Risks: Our ability to earn rewards is dependent on the performance and reliability of the third-party validators we delegate to. If a validator fails to perform its functions (e.g., through downtime or malicious activity), our share of staking rewards may be reduced or slashed (penalized). We bear costs for this service, typically validator commissions of 0-10% of rewards earned. |
| ● | Custodial and Security Risks: All our SOL holdings are custodied with OSL Group Limited (“OSL”), a Hong Kong Stock Exchange listed company (0863.HK). We have opened a corporate account with OSL, and our custody arrangements are in accordance with OSL’s general terms and conditions. Under the general terms and conditions, OSL’s associated entity, OSL Custody Services Limited (“OCSL”), a wholly owned subsidiary of OSL with a trust or company service provider licence under the Anti-Money Laundering and Counter-Terrorist Financing Ordinance (Cap. 615 of the Laws of Hong Kong), holds our virtual assets on trust for us in a segregated omnibus account designated as a client or trust account established for the purpose of holding client assets. OCSL has the duty to convey the assets under trust to us as per our instructions, and is required to maintain records which identify our assets, While OSL is a qualified third-party custodian that provides segregated accounts and comprehensive cold storage insurance, the loss of private keys, a security breach, operational failure, or insolvency of the custodian could lead to a partial or total loss of our assets. In addition, although OSL provides institutional-grade proprietary insurance coverage of up to USD 1 billion, in compliance with Hong Kong Securities and Futures Commission requirements, this insurance coverage is shared by its other customers and could result in the coverage being insufficient to compensate us for our losses. |
| ● | Regulatory Uncertainty: The regulatory landscape for digital assets and staking activities, particularly in the U.S., is evolving rapidly. New laws, regulations, or regulatory interpretations could deem our activities non-compliant, subject us to penalties, or force us to liquidate our position at a loss, potentially on short notice. |
| ● | Lack of Experience: Our management team has limited experience in the digital asset and blockchain sector. Our inability to successfully navigate the complexities of this new and rapidly evolving industry could lead to operational errors, poor strategic decisions, and financial losses. |
Our Solana digital asset staking strategy is subject to unique and substantial risks related to the Solana network’s technology, governance, and ecosystem, which could result in a complete or partial loss of our investment.
Our strategy is fundamentally dependent on the continuous and secure operation of the Solana blockchain. Solana is a high-throughput Layer 1 blockchain that utilizes a novel hybrid Proof-of-Stake and Proof-of-History consensus mechanism. While designed for performance, this architecture introduces specific risks, including:
| ● | Network Performance and Outages: The Solana network has a history of performance degradation and full outages due to high transaction volumes, spam attacks, and validator coordination failures. For example, the network experienced significant liveness disruptions in 2021 and 2022. Although upgrades have been implemented to address congestion, there can be no assurance that future issues will not occur. Any disruption, outage, or period of significant congestion could prevent us from staking, unstaking, or transacting our SOL, directly impairing our ability to execute our strategy and potentially eroding the value of our holdings. |
| ● | Concentration and Reliance on Third-Party Software: The Solana validator set is relatively concentrated compared to other networks. Furthermore, a high percentage of validators rely on software provided by Jito Labs, a third party unaffiliated with Solana Labs. If Jito Labs were to discontinue support or if its software contained critical bugs, it could compromise network security and stability, increase the impact of spam transactions, and adversely affect the value of SOL. |
| ● | Technological Obsolescence and Competition: The digital asset ecosystem is characterized by rapid innovation and intense competition. New protocols (e.g., Aptos, Sui, Ethereum Layer 2s like Base) or advancements, including the integration of artificial intelligence, may offer superior technology or attract developer activity away from Solana. If the Solana ecosystem fails to evolve, compete, or maintain its relevance, the utility and value of SOL could decline materially. |
| ● | Lifecycle and Economic Mechanics of SOL: The value of SOL is influenced by its specific tokenomics: |
| ○ | Inflationary and Deflationary Pressures: New SOL is created through an inflationary protocol issuance, which began at 8% annually and decreases by 15% year-over-year to a terminal rate of 1.5%. Counteracting this is a deflationary mechanism where a portion of transaction fees is burned (permanently removed from circulation). The net effect on the SOL supply (inflationary or deflationary) depends on network usage and is uncertain. |
| ○ | Bonding and Unbonding Periods: Staking SOL involves a “warmup” period before rewards are earned and a “cooldown” or unbonding period of several days when unstaking. Typical bonding period requires one full epoch, and unbonding periods requires 2 to 4 days and one full epoch. During this unbonding period, assets are illiquid and earn no rewards. Furthermore, the protocol limits how much stake can unbond per epoch (approximately 25% of the active stake), which could create a queue and further delay access to our capital during a market downturn, exacerbating liquidity risk. |
SOL is a relatively novel digital asset, and is subject to significant legal, commercial, regulatory and technical uncertainty, which could materially adversely affect the Company’s financial position, operations and prospects.
SOL and other digital assets, as well as applications on blockchain networks such as Solana, are relatively novel and are subject to significant uncertainty, which could adversely impact their price. The application of state and federal securities laws and other laws and regulations to digital assets and blockchain-based applications is unclear in certain respects, and it is possible that regulators in the United States or foreign countries may interpret or apply existing laws and regulations in a manner that adversely affects the price of SOL or other digital assets, or the ability of blockchain-based applications to operate.
The U.S. federal government, states, regulatory agencies, and foreign countries may also enact new laws and regulations, or pursue regulatory, legislative, enforcement or judicial actions, that could materially impact the price of SOL or the ability of individuals or institutions to own or transfer SOL and utilize blockchain-based applications on networks such as Solana. For example, the U.S. executive branch, the SEC, the European Union’s Markets in Crypto Assets Regulation, among others, have been active in recent years, and in the United Kingdom, the Financial Services and Markets Act 2023, or FSMA 2023, became law. It is not possible to predict whether, or when, any of these developments will lead to Congress granting additional authorities to the SEC, Commodity Futures Trading Commission (“CFTC”), or other regulators, or whether, or when, any other federal, state or foreign legislative bodies will take any similar actions. It is also not possible to predict the nature of any such additional authorities, how additional legislation or regulatory oversight might impact the ability of digital asset markets to function or the willingness of financial and other institutions to continue to provide services to the digital assets industry, nor how any new regulations or changes to existing regulations might impact the value of digital assets generally and SOL specifically. The consequences of increased regulation of digital assets and digital asset activities could adversely affect the market price of SOL and in turn adversely affect the market price of our common stock.
Moreover, the risks of engaging in a digital asset treasury strategy are relatively novel and have created, and could continue to create, complications due to the lack of experience that third parties have with companies engaging in such a strategy, such as increased costs of director and officer liability insurance or the potential inability to obtain such coverage on acceptable terms in the future.
The growth of the digital assets industry in general, and the use and acceptance of SOL in particular, may also impact the price of SOL and is subject to a high degree of uncertainty. The pace of worldwide growth in the adoption and use of the Solana network and SOL may depend, for instance, on public familiarity with digital assets, ease of buying, accessing or gaining exposure to SOL, institutional demand for SOL as an investment asset, the participation of traditional financial institutions in the digital assets industry, consumer demand for SOL as a means of payment, and the availability and popularity of alternatives to SOL. Even if growth in SOL adoption occurs in the near or medium-term, there is no assurance that SOL and Solana network usage will continue to grow over the long term.
Because SOL have no physical existence beyond the record of transactions on the Solana blockchain, a variety of technical factors related to the Solana blockchain could also impact the price of SOL. For example, malicious attacks by validators, inadequate validation and staking rewards to incentivize validating of Solana transactions, hard “forks” of the Solana blockchain into multiple blockchains, difficulties with upgrades to the Solana network (such as the proposed Alpenglow consensus upgrade or integration of the Firedancer validator client) and advances in digital computing, algebraic geometry, and quantum computing could undercut the integrity of the Solana blockchain and negatively affect the price of SOL. The liquidity of SOL may also be reduced and damage to the public perception of Solana may occur, if financial institutions were to deny or limit banking services to businesses that hold SOL, provide Solana-related services or accept SOL as payment, which could also decrease the price of SOL. Similarly, the open-source nature of the Solana blockchain means the contributors and developers of the Solana blockchain are generally not directly compensated for their contributions in maintaining and developing the blockchain, and any failure to properly monitor and upgrade the Solana blockchain could adversely affect the Solana blockchain and negatively affect the price of SOL.
The liquidity of SOL may also be impacted to the extent that changes in applicable laws and regulatory requirements negatively impact the ability of exchanges and trading venues to provide services for SOL and other digital assets.
A cyberattack or other malicious attack on the Solana blockchain could have a material impact on the value of SOL held by the Company.
Solana and other digital assets and the entities that provide services to participants in blockchain ecosystems have been, and may in the future be, subject to security breaches, cyberattacks, or other malicious activities. A successful security breach or cyberattack could result in:
| ● | a partial or total loss of our digital assets in a manner that may not be covered by insurance or the liability provisions of the custody agreements with the custodian who holds our digital assets; |
| ● | harm to our reputation; or |
| ● | significant regulatory scrutiny, investigations, fines, penalties, and other legal, regulatory, contractual and financial exposure. |
Further, any actual or perceived data security breach or cybersecurity attack directed at other companies with digital assets or companies that operate digital asset networks, regardless of whether we are directly impacted, could lead to a general loss of confidence in the broader Solana ecosystem or in the use of the Solana network to conduct financial transactions, which could materially and adversely affect our financial condition and results of operations.
| NewGenIvf’s tokenization advisory services exposes the Company to a novel, evolving and highly uncertain regulatory and operational landscape. |
On October 20, 2025, NewGenIvf announced its engagement as the worldwide agent for the tokenization of the specific artwork assets owned by World Chinese Museum Co. Ltd. NewGenIvf’s agreements to tokenize assets such as art involve complex legal and regulatory challenges. The legal status of tokenized assets is uncertain in many jurisdictions, and they may be deemed securities, subjecting us and our clients to stringent securities laws. Our ability to execute these services is contingent upon obtaining necessary regulatory approvals from bodies such as the U.S. SEC and UAE regulators, which may not be granted. Furthermore, these initiatives are vulnerable to risks including technical failures of the underlying blockchain, cybersecurity breaches, market volatility in the digital asset space, and a lack of market acceptance for tokenized assets. If we are unable to navigate these risks, our tokenization business may not generate the anticipated commission revenue, and we could be subject to significant legal and financial liabilities.
NewGenIvf’s real estate development projects are subject to numerous risks that could prevent the Company from realizing the projected returns.
NewGenIvf’s real estate joint venture is concentrated in Ras Al Khaimah, UAE, and is dependent on the successful development, marketing, and sale of properties in the specific geographic region. The project is vulnerable to regional economic conditions, regulatory changes, and real estate market fluctuations specific to that emirate. The recent escalation of the Iran-Israel-US conflict has introduced significant immediate risk to the UAE’s property development business, primarily through a severe shock to investor confidence and a corresponding market correction. The direct military strikes on the UAE and explicit threats from Iran against its commercial infrastructure have shattered the perception of the country as a safe haven, triggering a swift and defensive reaction from the market. This is evidenced by a 20% plunge in the Dubai real estate index, which erased all gains for the year, and sharp declines in the share prices of major developers like Emaar and Aldar. The projected net return of up to US$67 million is based on feasibility studies and assumptions, including specific sale prices per square foot and 100% sales efficiency, which may not materialize. These projects are susceptible to risks such as construction delays and cost overruns, fluctuations in the UAE real estate market, changes in regional economic and political conditions, regulatory changes, and reliance on third-party developers and partners. Our financial returns are contingent upon the joint venture’s success, and any failure to achieve the projected financial outcomes could have a material adverse effect on our business and financial condition. Furthermore, the Company’s current commitment is limited to a reservation deposit, as a definitive Sales and Purchase Agreement has yet to be executed. Thus, there is uncertainty over projected final project revenues.
The valuation of acquired assets, such as intellectual property, is complex and subjective, and may not be realized.
On February 28, 2025, the Company acquired the MicroSort® sperm-sorting technology, enhancing its capabilities in the fertility services market. The acquisition also resulted in a prior bargain purchase gain of $2.49 million based on an independent valuation report. On July 29, 2025, the Company acquired advanced cytometry intellectual property and equipment from Nodexus. An independent valuation by a “big-four” accounting firm received in October 2025 set a fair value of $17.9 million for the IP, resulting in a total bargain purchase gain of $19.16 million. This acquisition is central to the Company’s pivot towards a high-margin, technology-licensing business model. However, the gain from IP acquisition is based on estimates, appraisals, and technical reports. These valuations are subject to assumptions and methodologies that may prove to be inaccurate. The actual commercial value of the acquired IP is dependent on our ability to successfully license the technology and generate royalty streams. If the fair value of these assets declines or is not realized, we may be required to recognize impairment charges, which would negatively impact our financial results.
NewGenIvf’s marketing efforts depend significantly on its ability to receive positive references from its existing clients.
NewGenIvf’s marketing efforts depend significantly on its ability to call on its current clients to provide positive references to new, potential clients. Given its limited number of long-term clients, the loss or dissatisfaction of any client could substantially harm its brand and reputation, inhibit the market adoption of its offering and impair its ability to attract new clients and maintain existing clients. Any of these consequences could have an adverse effect on its business, financial condition and results of operations.
As a public reporting company, we are subject to filing deadlines for reports that we file pursuant to the Exchange Act, and our failure to timely file such reports may have material adverse consequences on our business.
In the past, we have not been able to, and may continue to be unable to produce timely financial statements, and file these financial statements as part of a periodic report in a timely manner with the SEC. For example, we failed to timely file with the SEC the requisite Form 20-F for the year ended December 31, 2023. Consequently, we were not compliant with the periodic reporting requirements under the Exchange Act at such time. We cannot guarantee that in the future our reporting will always be timely. Our failure to timely file future periodic reports with the SEC could subject us to enforcement action by the SEC and shareholder lawsuits and could eventually result in the delisting of our Class A Ordinary Shares from Nasdaq, regulatory sanctions from the SEC, and/or the breach of covenants in our credit facilities or of any preferred equity or debt securities we may issue in the future, any of which could have a material adverse impact on our operations and your investment in our Class A Ordinary Shares, and our ability to register with the SEC public offerings of our securities for our benefit or the benefit of our security holders. Additionally, our failure to file our past periodic reports and future periodic reports has resulted in and could result in investors not receiving adequate information regarding us with which to make investment decisions. As a result, investors may not have access to current or timely financial information about our business.
If we are unable to continue to meet the listing requirements of Nasdaq, our Class A Ordinary Shares will be delisted.
On October 8, 2024, the Company received a deficiency letter (“Bid Price Deficiency Letter”) from the Listing Qualifications Department (the “Staff”) of Nasdaq notifying the Company that it is currently not in compliance with the closing bid price requirement under Nasdaq Listing Rule 5450(a)(1) (the “Minimum Bid Price Rule”). The Bid Price Deficiency Letter stated that, for the preceding 30 consecutive business days, the Company’s Class A Ordinary Shares did not meet the minimum closing bid price of $1 per share pursuant to the Minimum Bid Price Rule. The Company has an initial compliance period of 180 calendar days, or until April 7, 2025 to regain compliance with the Minimum Bid Price Rule. The Deficiency letter stated that if at any time the closing bid price of the Company’s Class A Ordinary Shares is at least $1 for a minimum of ten consecutive business days, Nasdaq will provide the Company written confirmation of compliance with this requirement, as applicable. On February 11, 2025, the Company effected a 1-for-20 reverse stock split of its issued and unissued shares. The effect of the reverse stock split was to consolidate every 20 issued and unissued shares into one share. On February 27, 2025, the Company received a notification letter from Nasdaq, indicating that the closing bid price of the Company’s securities had been at $1.00 per share or greater for 10 consecutive business days from February 11, 2025 to February 26, 2025, and the Company had regained compliance with the Minimum Bid Price Rule. Since the First Reverse Stock Split, the Company has carried out 4 additional reverse stock splits on May 5, 2025, August 4, 2025, December 1, 2025, and January 26, 2026, in order to ensure that the Company remains compliant with the Minimum Bid Price Rule. If within one year of the Reverse Stock Splits, the Company’s Class A Ordinary Shares fall below $1.00 per share for 30 consecutive business days, Nasdaq may not provide us with an additional compliance period under its amended Listing Rule 5810(c)(3)(A)(iv) and our common stock could be delisted immediately.
On May 24, 2024, the Company received a MVLS Deficiency Letter from the Listing Qualifications Department (the “Staff”) of Nasdaq notifying the Company that, for the preceding 35 consecutive business days, the Class A Shares did not meet the minimum MVLS Requirement for continued listing on Nasdaq pursuant to Nasdaq Listing Rules 5450(b)(2)(A). In accordance with Nasdaq Rule 5810(c)(3)(C), the Company has been provided an initial period of 180 calendar days, or until November 20, 2024, the Compliance Date, to regain compliance with the MVLS Requirement. If, at any time before the Compliance Date, the MVLS for the Class A Shares is at least $50,000,000 for a minimum of ten consecutive business days, the Staff will provide the Company written confirmation of compliance with the MVLS Requirement. In the event the Company does not regain compliance with the above requirement prior to the expiration of the compliance period, it will receive written notification that its securities are subject to delisting.
On May 24, 2024, the Company received a MVPHS Deficiency Letter from the Staff of Nasdaq notifying the Company that, for the preceding 35 consecutive business days, the Company’s Class A Ordinary Shares did not meet the minimum $15,000,000 MVPHS Requirement for continued listing on Nasdaq pursuant to Nasdaq Listing Rules 5450(b)(2)(C). In accordance with Nasdaq Rule 5810(c)(3)(D), the Company has until the Compliance Date to regain compliance with the MVPHS Requirement. If, at any time before the Compliance Date, the MVPHS for the Class A Shares is at least $15,000,000 for a minimum of ten consecutive business days, the Staff will provide the Company written confirmation of compliance with the MVPHS Requirement. In the event the Company does not regain compliance with the above requirement prior to the expiration of the compliance period, it will receive written notification that its securities are subject to delisting. Alternatively, the Company may apply to transfer the Company’s securities to The Nasdaq Capital Market.
On November 21, 2024, the Company received a notice from the Staff of Nasdaq notifying the Company that its securities are subject to delisting due to the MVPHS Deficiency and MLVS Deficiency. The Company requested a hearing to appeal the delisting determination before the Nasdaq Hearings Panel (the “Panel”) on November 27, 2024. On November 29, 2024, the Company received a formal notice from Nasdaq that the Panel will consider its appeal at an oral hearing on January 28, 2025 (the “Hearing”). On February 19, 2025, the Company received written decision from the Panel, which granted an extension, allowing the Company additional time to regain compliance with the Nasdaq Stock Market’s (“Nasdaq” or the “Exchange”) continued listing requirements, subject to meeting specific compliance criteria within designated timeframes. On February 11, 2025, the Company carried out a 1-for-20 reverse stock split of its issued and unissued shares. The effect of the reverse stock split was to consolidate every 20 issued and unissued share into one share. On February 27, 2025, the Company received a notification letter from Nasdaq, indicating that the closing bid price of the Company’s securities had been at $1.00 per share or greater for 10 consecutive business days from February 11, 2025 to February 26, 2025, and the Company had regained compliance with the Minimum Bid Price Rule. In addition, on February 27, 2025, the Company received a confirmation from Nasdaq that its application to transfer its listing to the Nasdaq Capital Market had been approved and that the Company’s securities would be transferred to the Nasdaq Capital Market at the opening of business on February 28, 2025. On March 10, 2025, the Company received a confirmation letter from Nasdaq confirming that it has demonstrated compliance with all of Nasdaq’s listing requirements, as required in the Panel’s decision letter dated February 19, 2025.
If we are unable to achieve and maintain compliance with such listing standards or other Nasdaq listing requirements in the future, our Class A Ordinary Shares could be delisted from Nasdaq. A delisting of our Class A Ordinary Shares and our inability to list on another national securities market could negatively impact us by: (i) reducing the liquidity and market price of our Class A Ordinary Shares; (ii) reducing the number of investors willing to hold or acquire our Class A Ordinary Shares, which could negatively impact our ability to raise equity financing; (iii) limiting our ability to use certain registration statements to offer and sell freely tradable securities, thereby limiting our ability to access the public capital markets; and (iv) impairing our ability to provide equity incentives to our employees.
If NewGenIvf is unable to attract new clients, its business, financial condition and results of operations would be adversely affected.
To increase its revenue, NewGenIvf must continue to attract new clients. NewGenIvf’s ability to do so depends in large part on the success of its sales and marketing efforts, and the success of references through existing clients. Potential clients may seek out other options; therefore, NewGenIvf must demonstrate that its solutions are valuable and superior to alternatives. If NewGenIvf fails to provide high-quality solutions and convince clients of the benefits of its model and value proposition, NewGenIvf may not be able to attract new clients. If the markets for NewGenIvf’s solutions decline or grow more slowly than it expects, or if the number of clients that contract with it for its solutions declines or fails to increase as it expects, its financial results could be harmed. As the markets in which NewGenIvf participate mature, fertility solutions and services evolve and competitors begin to enter into the market and introduce differentiated solutions or services that are perceived to compete with its solutions, particularly if such competing solutions are adopted by its competitors, its ability to sell its solutions could be impaired. As a result of these and other factors, NewGenIvf may be unable to attract new clients, which would have an adverse effect on its business, financial condition and results of operations.
NewGenIvf’s business depends on its ability to maintain its existing client demographics. Any failure to do so would harm its business, financial condition and results of operations.
As part of its growth strategy, NewGenIvf is focused on maintaining its services within its existing client demographics. NewGenIvf mainly competes with mid-level private clinics and hospitals, which have improved and developed their services and equipment over the years. In addition to private clinics and hospitals already existing, foreign medical companies may also enter the markets where NewGenIvf operates. Such foreign medical companies may be well-placed to compete with NewGenIvf due to their larger network size, reputation as global players and access to more advanced technology and financial resources. The expansion of existing competitors in the industry may erode NewGenIvf’s existing market share or decrease its traditional client pool. There can be no assurance that NewGenIvf will be able to compete effectively and therefore its future business growth may suffer.
A significant reduction in the utilization of NewGenIvf’s solutions could have an adverse effect on its business, financial condition and results of operations.
A significant reduction in the number of clients using NewGenIvf’s solutions could adversely affect its business, financial condition and results of operations. Factors that could contribute to a reduction in the use of its solutions include: general economic downturn that results in adverse financial conditions; regulatory changes; failure to adapt and respond effectively to changing medical landscape, changing regulations, changing client needs, requirements or preferences; negative publicity, through social media or otherwise and news coverage.
If NewGenIvf fails to offer high-quality support, its reputation could suffer.
NewGenIvf relies on its client account management personnel and the patient navigators (the “PNs”) to resolve client issues and help clients realize the full benefits that its solutions and services provide. High-quality support is also important for the renewal and expansion of its services to existing clients. The importance of its support functions will increase as NewGenIvf expands its business and pursue new clients. If NewGenIvf does not help its clients quickly resolve issues and provide effective ongoing supports, its ability to maintain and expand its offerings to existing and new clients could suffer, and its reputation with existing or potential clients could suffer. Further, to the extent that NewGenIvf is unsuccessful in hiring, training and retaining adequate PNs and client account management personnel, its ability to provide adequate and timely support to its clients would be negatively impacted, and its clients’ satisfaction with its solutions and services would be adversely affected.
NewGenIvf’s failure to effectively develop and expand its marketing and sales capabilities could harm its ability to increase its client base and achieve broader market acceptance of solutions NewGenIvf provides.
NewGenIvf’s ability to increase its client base and achieve broader market acceptance of solutions it provides will depend to a significant extent on its ability to expand its marketing and sales capabilities. NewGenIvf plans to continue expanding its direct sales force and to dedicate significant resources to sales and marketing programs, including direct sales, inside sales, targeted direct marketing, advertising, digital marketing, e-newsletter and conference sponsorships. All of these efforts will require it to invest significant financial and other resources. Its business and results of operations could be harmed if its sales and marketing efforts do not generate significant increases in revenue. NewGenIvf may not achieve anticipated revenue growth from expanding its sales and marketing efforts if it is unable to hire, develop, integrate and retain talented and effective sales personnel, if its new and existing sales personnel, on the whole, are unable to achieve desired productivity levels in a reasonable period of time, or if its sales and marketing programs are not effective.
NewGenIvf may experience net losses and may not sustain profitability in the future.
NewGenIvf experienced significant revenue decrease from 2019 to 2020, due to the impact of COVID-19. NewGenIvf is not certain whether it will obtain sufficient levels of sales to sustain its growth or maintain profitability in the future. NewGenIvf also expects its costs and expenses to increase in future periods, which could negatively affect its future results of operations if its revenue does not increase accordingly. In particular, NewGenIvf intends to continue to incrementally expand its sales and client account management teams to educate potential clients and drive new client adoption. NewGenIvf also expects to incur additional costs as it introduces new solutions and services to enhance its comprehensive fertility offering. NewGenIvf will also face increased compliance costs associated with growth, the expansion of its client base and being a public company. NewGenIvf’s efforts to grow its business may be costlier than it expects, and NewGenIvf may not be able to increase its revenue enough to offset its increased operating expenses. NewGenIvf may incur significant losses in the future for a number of reasons, including the other risks described herein, and unforeseen expenses, difficulties, complications and delays, and other unknown events. If NewGenIvf is unable to sustain profitability, the value of its business and common stock may significantly decrease.
NewGenIvf’s future revenue may not grow at the rates it historically has, or at all.
NewGenIvf has experienced growth since its business operations started in 2014. Revenue and NewGenIvf’s client base may not grow at the same rates they historically have, or they may decline in the future. NewGenIvf’s future growth will depend, in part, on its ability to:
| ● | continue to attract new clients and/or maintain existing clients; |
| ● | price its solutions and services effectively so that it is able to attract new clients, expand sales to its existing clients and maintain profitability; |
| ● | provide its clients with client support that meets their needs, including through dedicated PNs; |
| ● | maintain successful collection of applicable receivable balances; |
| ● | retain and maintain relationships with high-quality and respected fertility specialists; |
| ● | attract and retain highly qualified personnel to support all clients; and |
| ● | increase awareness of its brand and successfully compete with other competitors. |
NewGenIvf may not successfully accomplish all or any of these objectives, which may affect its future revenue, and which makes it difficult for it to forecast its future results of operations. In addition, if the assumptions that NewGenIvf uses to plan its business are incorrect or change in reaction to changes in its market, it may be difficult for it to maintain profitability. NewGenIvf’s shareholders should not rely on its revenue for any prior quarterly or annual periods as any indication of its future revenue or revenue growth.
In addition, NewGenIvf expects to continue to expend substantial financial and other resources on:
| ● | sales and marketing; |
| ● | technology infrastructure, including systems architecture, scalability, availability, performance and security; and |
| ● | general administration, including increased legal and accounting expenses associated with being a public company. |
These investments may not result in increased revenue growth in its business. If NewGenIvf is unable to increase its revenue at a rate sufficient to offset the expected increase in its costs, its business, financial position, and results of operations will be harmed, and NewGenIvf may not be able to maintain profitability over the long term. Additionally, NewGenIvf may encounter unforeseen operating expenses, difficulties, complications, delays and other unknown factors that may result in losses in future periods.
If its revenue growth does not meet its expectations in future periods, NewGenIvf may not maintain profitability in the future, its business, financial position and results of operations may be harmed.
NewGenIvf’s interim and annual results may fluctuate significantly and may not fully reflect the underlying performance of NewGenIvf’s business.
NewGenIvf’s interim and annual results of operations, including the levels of NewGenIvf’s revenues, expenses, net (loss)/income and other key metrics, may vary significantly in the future due to a variety of factors, some of which are outside of NewGenIvf’s control, and period-to-period comparisons of NewGenIvf’s operating results may not be meaningful, especially given NewGenIvf’s limited operating history. Accordingly, the results for any one fiscal half year or any one fiscal year are not necessarily an indication of future performance. Fluctuations in interim and/or annual financial results may adversely affect the price of NewGenIvf’s ordinary shares. Factors that may cause fluctuations in NewGenIvf’s interim and annual financial results include:
| ● | NewGenIvf’s ability to attract new customers and maintain relationships with existing customers; |
| ● | changes in NewGenIvf’s products and services offered and introduction of new services and products; |
| ● | the amount and timing of operating expenses related to marketing and the maintenance and expansion of NewGenIvf’s business, operations and infrastructure; |
| ● | changes in the market price of Solana; | |
| ● | changes in the market situation of the residential property in the United Arab Emirates; | |
| ● | general economic, industry and market conditions; and |
| ● | the timing of expenses related to the development or acquisition of technologies or businesses. |
If the estimates and assumptions NewGenIvf uses to determine the size of the target markets for its services are inaccurate, its future growth rate may be impacted and its business would be harmed.
Market opportunity estimates and growth forecasts are subject to significant uncertainty and are based on assumptions and estimates that may not prove to be accurate. Market opportunity estimates and growth forecasts included in this prospectus, including those NewGenIvf has generated itself, are subject to significant uncertainty and are based on assumptions and estimates that may not prove to be accurate, including the risks described in this prospectus. Even if the markets in which NewGenIvf competes achieve the forecasted growth, its business could fail to grow at similar rates, if at all.
NewGenIvf’s estimates of the market opportunity for its services are based on the assumption that the purpose-built, data-driven and disruptive fertility services platform with the plan design NewGenIvf offers will be attractive to clients. Clients may pursue alternatives or may not see the value in providing enhanced fertility-related services. In addition, NewGenIvf believes that it is expanding the size of the fertility market as NewGenIvf enhances demand and increase awareness for fertility services. If these assumptions prove inaccurate, or if the increase in awareness of fertility services attracts potential competitors to the market and results in greater competition, NewGenIvf’s business, financial condition and results of operations could be adversely affected.
It is difficult to predict the demand for NewGenIvf’s solutions, the entry of competitive solutions or the future growth rate and size of the fertility market. The expansion of the fertility market depends on a number of factors, including, but not limited to: the continued trend of individuals starting families later in life, increase in the number of single mothers by choice, adoption of non-traditional paths to parenthood and continued de-stigmatization of infertility.
If there is a reduction in demand caused by a lack of client acceptance, weakening economic conditions, data security or privacy concerns, governmental regulation, competing offerings or otherwise, the market for its solutions and services might not continue to develop or might develop more slowly than NewGenIvf expects, which would adversely affect its business, financial condition and results of operations.
NewGenIvf may not be able to successfully manage its growth, and if NewGenIvf is not able to grow efficiently, its business, financial condition and results of operations could be harmed.
As usage of its solutions grows, NewGenIvf will need to devote additional resources to improving and maintaining its infrastructure. In addition, NewGenIvf will need to appropriately scale its internal business systems and its client account management and services personnel to serve its growing client base. Any failure of or delay in these efforts could result in reduced client satisfaction, resulting in decreased sales to new clients and lower renewal and utilization rates by existing clients, which could hurt its revenue growth and its reputation. Even if NewGenIvf is successful in these efforts, they will require the dedication of management time and attention. NewGenIvf could also face inefficiencies or service disruptions as a result of its efforts to scale its internal infrastructure. NewGenIvf cannot be sure that the expansion and improvements to its internal infrastructure will be effectively implemented on a timely basis, and such failures could harm its business, financial condition and results of operations.
If NewGenIvf’s new solutions and services are not adopted by its clients, or if it fails to innovate and develop new offerings that are adopted by its clients, its revenue and results of operations may be adversely affected.
To date, NewGenIvf has derived a substantial majority of its revenue from sales of its fertility services. As NewGenIvf operates in an evolving industry, its long-term results of operations and continued growth will depend on its ability to successfully develop and market new successful solutions and services to its clients. If its existing clients do not value and/or are not willing to make additional payments for such new solutions or services, it could adversely affect its business, financial condition and results of operations. If NewGenIvf is unable to predict clients’ preferences, if the markets in which NewGenIvf participates change, including in response to government regulation, or if NewGenIvf is unable to modify its solutions and services on a timely basis, NewGenIvf may lose clients. Its results of operations would also suffer if its innovations were not responsive to the needs of the clients, appropriately timed with market opportunity or effectively brought to market.
If NewGenIvf fails to adapt and respond effectively to the changing medical landscape, changing regulations, changing client needs, requirements or preferences, its offerings may become less competitive.
The market in which NewGenIvf competes is subject to a changing medical landscape and changing regulations, as well as changing client needs, requirements and preferences. The success of its business will depend, in part, on its ability to adapt and respond effectively to these changes on a timely basis. NewGenIvf’s business strategy may not effectively respond to these changes, and NewGenIvf may fail to recognize and position itself to capitalize upon market opportunities. NewGenIvf may not have sufficient advance notice and resources to develop and effectively implement an alternative strategy. There may be scientific or clinical changes that require it to change its solutions or that make its solutions less competitive in the marketplace. If there are sensitivities to its model or its existing competitors and new entrants create new disruptive business models and/or develop new solutions that clients prefer to its solutions, NewGenIvf may lose clients, and its results of operations, cash flows and/or prospects may be adversely affected. The future performance of NewGenIvf’s business will depend in large part on its ability to design and implement market appropriate strategic initiatives, some of which will occur over several years in a dynamic industry. If these initiatives of NewGenIvf do not result in met objectives, NewGenIvf’s results of operations could be adversely affected.
If NewGenIvf fails to maintain and enhance its brand, its ability to expand its client base will be impaired and its business, financial condition and results of operations may suffer.
The growth of NewGenIvf’s business partially depends on the recognition of NewGenIvf’s brand and reputation. NewGenIvf believes that maintaining and enhancing its brand is important to support the marketing and sale of its existing and future solutions to new clients and expand sales of its solutions to existing clients. NewGenIvf also believes that the importance of brand recognition will increase as competition in its market increases. Successfully maintaining and enhancing its brand will depend largely on the effectiveness of its marketing efforts, its ability to provide reliable services that continue to meet the needs of its clients at competitive prices, its ability to maintain its clients’ trust, its ability to continue to develop new solutions, and its ability to successfully differentiate its platform from competitive solutions and services. NewGenIvf’s brand promotion activities may not generate client awareness or yield increased revenue, and even if they do, any increased revenue may not offset the expenses NewGenIvf incurs in building its brand. If NewGenIvf fails to successfully promote and maintain its brand, its business, financial condition and results of operations may suffer.
If NewGenIvf fails to retain and motivate members of its management team or other key employees, or fails to attract additional qualified personnel to support its operations, its business and future growth prospects could be harmed.
NewGenIvf’s success and future growth depend largely upon the continued services of its management team and its other key employees. From time to time, there may be changes in its executive management team or other key employees resulting from the hiring or departure of these personnel. Its executive officers and other key employees are employed on an at-will basis, which means that these personnel could terminate their employment with it at any time. The loss of one or more of its executive officers, or the failure by its executive team to effectively work with its employees and lead its company, could harm its business.
In addition, to execute its growth plan, NewGenIvf must attract and retain highly qualified personnel. Competition for these personnel is intense, especially for experienced medical officers and scientific staffs and sales and client account management personnel. There is no guarantee NewGenIvf will be able to attract such personnel or that competition among potential employers will not result in increased salaries or other benefits. From time to time, NewGenIvf has experienced, and NewGenIvf expects to continue to experience, difficulty in hiring and retaining employees with appropriate qualifications. Many of the companies with which NewGenIvf competes for experienced personnel have greater resources than NewGenIvf has. If NewGenIvf hires employees from competitors or other companies, their former employers may attempt to assert that these employees or NewGenIvf has breached their legal obligations, resulting in a diversion of its time and resources. In addition, prospective and existing employees often consider the value of the equity awards they receive in connection with their contribution to the company. If the perceived value of its equity awards declines, experiences significant volatility, or increases such that prospective employees believe there is limited upside to the value of its equity awards, it may adversely affect its ability to recruit and retain key employees. If NewGenIvf fails to attract new personnel or fails to retain and motivate its current personnel, its business and future growth prospects could be harmed.
Furthermore, in order to attract and retain key personnel and employees, the compensation amounts for NewGenIvf’s executive officers may change significantly after consummation of the Business Combination, although there are currently no agreements in place relating to any such post Business Combination compensation arrangements. As a result, NewGenIvf’s expenses associated with the compensation may increase, which may also have an adverse effect on its results of operations.
NewGenIvf’s Share Incentive Plan allows NewGenIvf to enhance its ability to attract and retain exceptionally qualified individuals and agents and to encourage them to acquire a proprietary interest in the company’s growth and performance. Competition for highly skilled personnel and agents is often intense and NewGenIvf may incur significant costs or may not be successful in attracting, integrating, or retaining qualified personnel and agents to fulfill NewGenIvf’s current or future needs. NewGenIvf believes that the granting of share-based awards is of significant importance to NewGenIvf’s ability to attract and retain agents, key personnel and employees, and NewGenIvf will continue to grant share-based awards in the future. In addition, NewGenIvf may, with the approval of its Compensation Committee and the Board, revise the terms of, and increase the size of, its share incentive plan, to ensure that it is able to attract and retain agents, key personnel and employees. On March 31, 2025, NewGenIvf’s Board approved certain amendments to its Share Incentive Plan, including the increase of the size of the share incentive plan to 20% of the outstanding shares of the Company from time to time, and on August 18, 2025, NewGenIvf’s Board approved the replenishment of its award pool to 20% of the outstanding shares of the Company as of August 18, 2025. As a result, NewGenIvf’s expenses associated with share-based compensation may increase, which may have an adverse effect on NewGenIvf’s results of operations. The amended share incentive plan is available as Exhibit 4.32.
To successfully market and sell its services and products in Asia-Pacific markets, NewGenIvf must address many international business risks with which NewGenIvf has limited experience.
NewGenIvf’s business is subject to risks in connection with changes in international, national and local economic and market conditions, including the effects of global financial crises, effects of terrorist acts and war and global pandemics. Such economic changes could negatively impact infertile couples’ abilities to pay for fertility treatments around the world.
NewGenIvf’s strategy is to increase its international presence in Asia-Pacific countries and its international sales are subject to a number of risks, including:
| ● | increased competition as a result of more products and procedures receiving regulatory approval or otherwise free to market in international markets; |
| ● | longer accounts receivable payment cycles and difficulties in collecting accounts receivable; |
| ● | reduced or varied protection for intellectual property rights in some countries; |
| ● | export restrictions, trade regulations, and foreign tax laws; |
| ● | fluctuations in currency exchange rates; |
| ● | foreign certification and regulatory clearance or approval requirements; |
| ● | customs clearance and shipping delays; |
| ● | political, social, and economic instability abroad, terrorist attacks, and security concerns in general; |
| ● | preference for locally provided services; |
| ● | potentially adverse tax consequences, including the complexities of foreign value-added tax systems; |
| ● | the burdens of complying with a wide variety of foreign laws and different legal standards; and |
| ● | increased financial accounting and reporting burdens and complexities. |
If one or more of these risks are realized, its business, financial condition and results of operations could be adversely affected.
Ethical, legal and social concerns related to the use of assisted reproductive technology could reduce demand for the fertility services provided by the medical facilities in NewGenIvf’s network, and thus may adversely affect the business, financial conditions and results of operations of the medical facilities in its network.
Patient sentiment and distrust of the use of assisted reproductive technology may lead to less demand for fertility services. Assisted reproductive technologies, including genetic testing, technologies used for surrogacy and egg donation and gender selection, have raised ethical, legal and social issues regarding privacy and the appropriate uses of the resulting information. Government authorities could, for social or other purposes, limit or regulate the use of assisted reproductive technology to certain conditions. Similarly, these concerns may lead patients to refuse to use, or physicians to be reluctant to order, assisted reproductive services even if permissible. These and other ethical, legal and social concerns may limit market acceptance of fertility services or reduce patient demand for such services, either of which could have a material adverse effect on the business, financial condition and results of operations of the medical facilities in NewGenIvf’s network, and NewGenIvf itself.
NewGenIvf is reliant on revenue from international clients.
Fertility services revenue from international clients are an important part of NewGenIvf’s revenue, though NewGenIvf is expanding rapidly into the local markets. The number of international clients travelling to Thailand, Cambodia and Kyrgyzstan to seek fertility services may, however, be affected by a number of factors, including the economic status of the foreign client’s country of origin, the relative exchange rate of the client’s home currency to the relevant authorities, which may affect the cost of treatment, natural disasters, pandemics like COVID-19, and political tension or acts of terrorism in such countries and the region. For example, the COVID-19 has had resulted in a number of countries declaring a state of emergency and a number of countries, including the countries in Asian Pacific, imposing extensive travel restrictions, which in turn caused a decrease in the numbers of internal clients traveling to Thailand, Cambodia or Kyrgyzstan for treatments.
These events could cause a postponement or a reduction in the number of clients traveling to Thailand, Cambodia or Kyrgyzstan, and could in turn affect revenues from international clients, which is the significant contributor in terms of volume. A decline in the medical tourism industry may have a material adverse effect on NewGenIvf’s financial condition and results of operations.
Fluctuations in exchange rates could have a material and adverse effect on NewGenIvf’s results of operations and the value of your investment.
NewGenIvf’s reporting currency is U.S. dollars. The functional currency of NewGenIvf and its subsidiaries include Hong Kong dollar (“HK$”), Thai baht (“THB”), and United States dollar (“USD”). Accordingly, fluctuations in the value of HK$, and THB relative to the USD could affect its results of operations due to translational remeasurements. As its international operations expand, an increasing portion of its revenue and operating expenses may be denominated in non- HK$, and THB currencies. Accordingly, NewGenIvf’s revenue and operating expenses will become increasingly subject to fluctuations due to changes in foreign currency exchange rates. If NewGenIvf is not able to successfully hedge against the risks associated with currency fluctuations, NewGenIvf’s business, financial condition and results of operations could be materially adversely affected.
Governmental control of currency conversion may limit NewGenIvf’s ability to utilize NewGenIvf’s net revenue effectively and affect the value of your investment.
NewGenIvf’s revenue and expenses for its businesses are substantially denominated in THB, which are currently not freely convertible currencies. A portion of such revenue must be converted into other currencies in order to meet its foreign currency obligations. For example, NewGenIvf’s subsidiaries will need to obtain foreign currency to make payments of declared dividends, if any, on its shares.
Under the existing foreign exchange regulations in Thailand, NewGenIvf will be able to make current account foreign exchange transactions. However, in the future, governments may take measures, at its discretion, to restrict access to foreign currencies for capital account and current account transactions under certain circumstances. If such measures are implemented, NewGenIvf may not be able to pay dividends in foreign currencies to holders of its shares. Foreign exchange transactions under its capital account are subject to significant foreign exchange controls and require certain approvals. These limitations could affect our ability to obtain foreign exchange through offshore financing.
The value of the THB against the U.S. dollar and other currencies fluctuates, and is subject to changes resulting from policies of the Thailand and other governments, and depends to a large extent on domestic and international economic and political developments as well as supply and demand in the local market. For example, the Bank of Thailand, which is the central bank of Thailand, is responsible for formulating and implementing monetary policies in the country to maintain the price stability and promote economic stability and sustainable growth. The Bank of Thailand imposes (four) measures in preventing THB fluctuation. Those are measures to limit THB liquidity, to curb capital inflows, to limit the flows on Non-resident Bank Account and Non-resident Baht for Securities, and to limit the flows on Non-Deliverable Forward transactions. With an increased floating range of the THB’s value against foreign currencies and a more market-oriented mechanism for determining the mid-point exchange rates, the THB may further appreciate or depreciate significantly in value against the U.S. dollar or other foreign currencies in the long-term, depending on the fluctuation of the basket of currencies against which it is currently valued, or it may be permitted to enter into a full float, which may also result in a significant appreciation or depreciation of the THB against the U.S. dollar or other foreign currencies. It cannot be assured that THB will not experience significant appreciation or depreciation against the U.S. dollar or other foreign currencies in the future.
Furthermore, NewGenIvf is also currently required to obtain approvals before converting significant sums of foreign currencies into THB. All of these factors could materially and adversely affect its business, results of operations, financial condition and prospects, and could reduce the value of, and dividends payable on, its shares in foreign currency terms.
Our dual-class voting structure may limit your ability to influence corporate matters and could discourage others from pursuing any change of control transactions that holders of our Class A Ordinary Shares may view as beneficial.
Our authorized and issued ordinary shares are divided into Class A Ordinary Shares and Class B Ordinary Shares. Each Class A Ordinary Share is entitled to one (1) vote, while each Class B Ordinary Share is entitled to one hundred (100) votes with all Ordinary Shares voting together as a single class on most matters. Each Class B Ordinary Share is convertible into one Class A Ordinary Share at any time by the holder thereof, while Class A Ordinary Shares are not convertible into Class B Ordinary Shares under any circumstances. Only Class A Ordinary Shares are listed and traded on NASDAQ, and we intend to maintain the dual-class voting structure. Mr. Wing Fung Alfred Siu and Ms. Hei Yue Tina Fong beneficially own all of the issued Class B Ordinary Shares. As of the date of this prospectus, these Class B Ordinary Shares constitute approximately 0.03% of our total issued and outstanding shares and 2.77% of the aggregate voting power of our total issued and outstanding shares due to the disparate voting powers associated with our dual-class share structure. As a result of the dual-class share structure and the concentration of control, holders of Class B Ordinary Shares have considerable influence over matters such as decisions regarding election of directors and other significant corporate actions. Such holders may take actions that are not in the best interest of us or our other shareholders. This concentration of control may discourage, delay, or prevent a change in control of us, which could have the effect of depriving our other shareholders of the opportunity to receive a premium for their shares as part of a sale of us and may reduce our share price. This concentrated control will limit the ability of holders of Class A Ordinary Shares to influence corporate matters and could discourage others from pursuing any potential merger, takeover, or other change of control transactions that holders of Class A Ordinary Shares may view as beneficial.
Substantially all of NewGenIvf’s assets and operations are located in U.S., United Arabs of Emirates, Thailand, Cambodia and Kyrgyzstan while Chinese clients contributing significant source of revenue and they are subject to economic, legal and regulatory uncertainties in such countries.
Substantially all of NewGenIvf’s operations and assets are based in Thailand, Cambodia and Kyrgyzstan. As a result, its businesses and operations are subject to the changing economic conditions prevailing from time to time in such countries. From 2022 to 2025, Thailand posted the weakest economic performance, with GDP growth averaging just 2.1% to 2.5% annually, constrained by high household debt and sluggish tourism, while Cambodia was the strongest grower with expansion accelerating from 5.1% to a projected 6.3% driven by garment exports and foreign investment. Kyrgyzstan experienced an externally-driven boom with 9.0% growth in 2023-2024 fueled by Russia sanctions re-exports, though this model is now fragile. However, this economic picture was dramatically upended in late 2025 by the outbreak of major armed conflict between Thailand and Cambodia. Beginning with clashes in July and escalating into full-scale warfare in December.
NewGenIvf also derives a substantial portion of its revenue from Chinese clients and as such, its maintenance of PRC-sourced revenues and access to new and existing clients from the PRC are also subject to the economic conditions of China. From 2022 to 2025, China’s economy transitioned to slower but stable growth, successfully meeting official targets of “around 5%” for three consecutive years. According to Focus Economics and GF Securities, real GDP expanded by 3.0% (2022), 5.2% (2023), 5.0% (2024), and 5.0% (2025), with Q4 2025 marking the weakest quarter since Q4 2022 at 4.5%. Domestic demand remained persistently weak due to a prolonged property sector downturn. A prolonged slow growth in the PRC economy generally could materially and adversely affect NewGenIvf’s results of operations.
Factors that may adversely affect the economy and conditions in such countries include:
| ● | political instability; |
| ● | global economic conditions; |
| ● | exchange rate fluctuations and the exchange control policy of the banks; |
| ● | a prolonged period of inflation or increase in regional interest rates; |
| ● | changes in taxation; |
| ● | changes in government policies affecting import and export volumes; |
| ● | decline in tourism; |
| ● | natural disasters, including tsunamis, earthquakes, fires, floods, drought and similar events; |
| ● | a potential recurrence or outbreak of avian influenza, severe acute respiratory syndrome or other infectious or contagious diseases like COVID-19 in Asian countries, and governmental policies to address such outbreak; |
| ● | scarcity of credit or other financing, resulting in lower demand for products and services provided by companies in the region; |
| ● | increases in oil prices and other commodity prices; |
| ● | decreased consumer confidence; |
| ● | other external recessions or potential economic downturns in the United States, Asia or other parts of the world; and |
| ● | other regulatory, political or economic developments in or affecting the countries. |
The economic conditions in Thailand, Cambodia, Kyrgyzstan and China are also affected by global economic conditions. The global credit markets have experienced, and may continue to experience, volatility and liquidity disruptions, which have resulted in the consolidation, failure or near failure of a number of institutions in the banking and insurance industries. There remains a concern that a return of the debt crisis in Europe, the political unrest in the Middle East and Eastern Europe as well as rumors or threats or actual terrorist attacks or conflicts in the Middle East, Southeast Asia, Eastern Europe or other regions will impinge upon the health of the global financial system. These or other such events could adversely affect NewGenIvf’s business, financial condition, results of operations and prospects.
There is no assurance that the economies and social conditions of Thailand, Cambodia, Kyrgyzstan and China will meet current projections or improve in the future. Any instability or economic downturn could have a material adverse effect on NewGenIvf’s business, financial condition, results of operations and prospects.
Failure to comply with the terms of future financing arrangements could result in default, which could have an adverse effect on NewGenIvf’s cash flow and liquidity.
NewGenIvf may from time to time enter into credit facilities and debt financing arrangements containing financial and other covenants that could, among other things, restrict NewGenIvf’s business and operations. If NewGenIvf breaches any of these covenants, including the failure to maintain certain financial ratios, NewGenIvf’s lenders may be entitled to accelerate NewGenIvf’s debt obligations. Any default under the credit facility could result in the repayment of these loans prior to maturity as well as the inability to obtain additional financing, which in turn may have a material adverse effect on NewGenIvf’s cash flow and liquidity.
NewGenIvf requires a significant amount of capital to fund its operations and growth. If NewGenIvf cannot obtain sufficient capital on acceptable terms, its business, financial condition, and prospects may be materially and adversely affected.
NewGenIvf requires a significant amount of capital and resources for its operations and continued growth. NewGenIvf expects to make significant investments to fund operations, laboratory upgrades, new strategic investments in Solana and residential property development in United Arab Emirates, among other things, which may significantly increase NewGenIvf’s net cash used in operating activities. In addition, NewGenIvf will continue to invest in laboratory and facilities which are fundamental to NewGenIvf’s business operation and future growth. However, NewGenIvf cannot assure you that these investments will generate the optimal returns, if at all. To date, NewGenIvf has historically funded its cash requirements primarily through operational, capital contributions from its shareholders and short-term or long-term borrowings. If these resources are insufficient to satisfy NewGenIvf’s cash requirements, NewGenIvf may seek to raise funds through additional equity offering or debt financing or additional bank facilities. NewGenIvf’s ability to obtain additional capital in the future, however, is subject to a number of uncertainties, including those relating to its future business development, financial condition, and results of operations, general market conditions for financing activities by companies in its industry, and macro-economic and other conditions in Thailand, Cambodia, Kyrgyzstan and globally. If NewGenIvf cannot obtain sufficient capital on acceptable terms to meet its capital needs, NewGenIvf may not be able to execute its growth strategies, and NewGenIvf’s business, financial condition, and prospects may be materially and adversely affected.
NewGenIvf’s proposed Solana digital assets purchase arrangement with White Lion is novel and subject to extreme volatility and regulatory scrutiny.
On October 31, 2025, NewGenIvf announced its entry into a binding term sheet with White Lion Capital LLC to potentially receive up to 600,000 Solana tokens in exchange for shares of the Company. This is a novel financing structure that presents unique risks. The value of our compensation is directly tied to the price of SOL, which has been and is likely to continue to be highly volatile. A decline in the price of SOL could significantly reduce the capital we receive. Furthermore, the regulatory treatment of such a transaction is uncertain and could be challenged by the SEC or other regulators, potentially leading to restrictions, fines, or the invalidation of the agreement.
The defects in certain leased property interests and failure to register certain lease agreements may materially and adversely affect NewGenIvf’s business, financial condition, results of operations, and prospects.
NewGenIvf leases premises in Thailand, Cambodia and Kyrgyzstan in various locations. With respect to property leased by First Fertility PGS Center in Thailand, the lessors did not have or provide NewGenIvf with property ownership certificates or other documents evidencing their rights to lease such premises to First Fertility PGS Center. Therefore, NewGenIvf cannot assure that it will not be subject to any challenges, lawsuits, or other actions taken against First Fertility PGS Center with respect to its leased premises for which the relevant lessors do not have valid title or right to lease. If First Fertility PGS Center’s lessors’ right to lease premises is successfully challenged by any third party, First Fertility PGS Center’s lease agreements may not be enforceable and NewGenIvf may be forced to vacate the premises and relocate to a different location. Under such circumstances, NewGenIvf expects to incur relocation costs of up to THB3 million and expects that there would not be material business interruption costs, if any.
In addition, the failure of the lessor to provide sufficient legal evidence of its right to lease the premises has prevented First Fertility PGS Center from registering the clinic with the Bangkok Metropolitan Authority (“BMA”) as required under the Public Health Act B.E. 2535 (1992) (the “PHA”). Under Section 71 of the PHA, First Fertility PGS Center and its directors are subject to imprisonment of up to 6 (six) months and a fine of up to THB50,000, or both. The BMA could also order First Fertility PGS Center to stop operating the clinic which would require relocation of the clinic if First Fertility PGS Center could not make the necessary registration. Under such circumstances, First Fertility PGS Center expects to incur relocation costs of up to THB3 million and expects that there would not be material business interruption costs, if any.
Only one of NewGenIvf’s directors or officers, namely Ms. Fong, Hei Yue Tina, is also a director of First Fertility PGS Center. NewGenIvf believes that if First Fertility PGS Center’s directors, including Ms. Fong, are found guilty of the above offence and subject to imprisonment, the resulting impact on NewGenIvf’s business, results of operations and financial conditions would be limited, as Ms. Fong has limited involvement in the day-to-day management of First Fertility PGS Center’s operations and Mr. Siu, Wing Fung Alfred and the other directors and officers of NewGenIvf and its subsidiaries would be able to keep operating the group’s and First Fertility PGS Center’s activities with limited disruptions. Further, a second clinic has been set up in a separate location which will mitigate the risk of interruptions to operations.
NewGenIvf currently has no insurance coverage for its operations.
The assisted reproductive medical facilities in NewGenIvf’s network are exposed to potential liabilities that are inherent to the provision of services. Medical and other liabilities may not be fully covered by insurance and the medical facilities may face claims in excess of the insurance coverage or claims which are not covered by insurance due to other policy limitations or exclusions or where the medical facilities in NewGenIvf’s network have failed to comply with the terms of the policy. Any uninsured risks may result in substantial costs and the diversion of resources, which could adversely affect its results of operations and financial condition.
The insurance industries in Thailand, Cambodia and Kyrgyzstan are still at early stages of development, and insurance companies in Thailand, Cambodia and Kyrgyzstan currently offer limited business-related insurance products. NewGenIvf does not currently maintain insurance. NewGenIvf cannot assure you that the medical facilities in its network will be able to obtain and/or maintain medical liability insurance on acceptable terms or without substantial premium increases or at all in the future.
In addition, as NewGenIvf’s business expands, the cost for each medical facility in its network and NewGenIvf to maintain an adequate level of insurance may become increasingly high. NewGenIvf cannot ensure that the medical facilities in its network will be able to locate or purchase appropriate insurance to cover the expanding operations in time, on commercially reasonable terms or at all. Any significant uninsured loss could have material and adverse effects on the financial condition and results of operations of the medical facilities in NewGenIvf’s network, and thus may affect its business, results of operations and financial condition.
Moreover, NewGenIvf does not currently maintain professional malpractice liability insurance for its physicians and nurses. As a result, NewGenIvf may be subject to medical disputes and claims arising under relevant laws from time to time, which could cause substantial damage to NewGenIvf if not covered by professional malpractice liability insurance. Any dispute with clients, or any legal proceeding involving the physicians of the medical facilities or medical professionals, regardless of its merit or eventual outcome, could result in significant legal costs and financial and/or reputational damages to the medical facilities and NewGenIvf and materially and adversely affect the business, financial condition and results of operations of the medical facilities in NewGenIvf’s network, and further affect its business, financial condition, results of operations and prospects.
NewGenIvf may not be successful in adapting to technological developments, which may affect its business and results of operations.
It is possible that new technologies could be developed or scientific advances made by NewGenIvf’s competitors, or elsewhere and licensed to NewGenIvf’s competitors, which cannot be replicated by NewGenIvf without significant capital expenditure or at all, or that replace or reduce the requirement for assisted reproductive services, ultrasound or specialized diagnostics. The consequences for NewGenIvf of the development of new technologies could include lower or loss of revenues, loss of market position and reduced prospects of NewGenIvf.
If its computer systems, or those of its providers, specialty pharmacies or other downstream vendors lag, fail or suffer security breaches, NewGenIvf may incur a material disruption of its services, which could materially impact its business and the results of operations.
NewGenIvf’s businesses in Thailand, Cambodia and Kyrgyzstan are increasingly dependent on critical, complex and interdependent information technology systems to support business processes as well as internal and external communications. NewGenIvf’s success is therefore dependent in part on its ability to secure, integrate, develop, redesign and enhance its (or contract with vendors to provide) technology systems that support its business strategy initiatives and processes in a compliant, secure, and cost and resource efficient manner. If NewGenIvf or its providers, specialty pharmacies or other downstream vendors have an issue with its or their respective technology systems, it may result in a disruption to its operations or downstream disruption to its relationships with its clients or its selective network of high-quality fertility specialists. Additionally, if NewGenIvf chooses to insource any of the services currently handled by a third party, it may result in technological or operational disruptions.
In addition, despite the implementation of security measures, its internal computer systems, and those of its provider clinics, specialty pharmacies or other downstream vendors, are potentially vulnerable to damage from malicious intrusion, malware, computer viruses, unauthorized access, natural disasters, terrorism, war and telecommunication and electrical failures. While NewGenIvf is not aware that it has experienced any such system failure, accident or security breach to date, if such an event were to occur and cause interruptions in its operations, it could result in a material disruption to its ability to operate and deliver its solutions. In addition, to the extent that any disruption or security breach were to result in a loss or inappropriate disclosure of confidential information, NewGenIvf could incur liability. See “— Risks Related to Government Regulation — NewGenIvf operates in a highly regulated industry and must comply with a significant number of complex and evolving requirements. Any lack of requisite approvals, licenses, or permits applicable to NewGenIvf’s business may have a material and adverse impact on NewGenIvf’s business, financial condition, and results of operations — Data Protection and Breaches.”
Our share purchase program may not be fully implemented and may not enhance shareholder value.
On November 10, 2025, the Company announced a share repurchase program (“Share Repurchase Program”) under which the Company may repurchase up to US$2 million of its outstanding Class A ordinary shares over the next 24 months. The Share Repurchase Program authorizes the Company to repurchase its shares from time to time through open market purchases, in privately negotiated transactions, or by other means in accordance with applicable securities laws. The timing and actual number of shares repurchased will depend on a variety of factors, including stock price, trading volume, general business and market conditions, and the requirements under Rule 10b-18 and/or Rule 10b5-1 of the Exchange Act. The Board will review the Share Repurchase Program periodically, and may authorize adjustment of its terms and size or suspend or discontinue the program. The repurchase program will be funded using the Company’s existing cash reserves. The execution of this program is discretionary and subject to market conditions, regulatory requirements, and our cash flow needs. We may suspend or terminate the program at any time. There is no guarantee that the program will be fully consummated or that it will lead to an increase in the trading price of our Class A Ordinary Shares. The use of cash for repurchases will also reduce the capital available for other corporate purposes, including funding our new strategic initiatives.
Risks Related to NewGenIvf’s Relationships with Third Parties
NewGenIvf’s business depends on its ability to maintain its network of high-quality fertility specialists and other healthcare providers. If NewGenIvf is unable to do so, its future growth would be limited and its business, financial condition and results of operations would be harmed.
NewGenIvf’s performance and success is dependent upon its continued ability to maintain a credentialed network of high-quality fertility specialists, including its senior management team, other key employees, as well as research and development and operation maintenance personnel, many of whom are difficult to replace. Fertility specialists could refuse to contract, demand higher payments or take other actions that could result in higher medical costs, less attractive service for its clients or difficulty meeting regulatory or accreditation requirements. Identifying high-quality fertility specialists, credentialing and negotiating contracts with them and evaluating, monitoring and maintaining its network, requires significant time and resources. Competition in the healthcare industry for qualified employees is intense. NewGenIvf may need to offer higher compensation and other benefits in order to attract and retain key personnel in the future, which could increase NewGenIvf’s compensation expenses, including stock-based compensation. NewGenIvf’s continued ability to compete effectively depends on NewGenIvf’s ability to attract new employees and to retain and motivate NewGenIvf’s existing employees. If NewGenIvf is not successful in maintaining its relationships with top fertility specialists, these fertility specialists may refuse to renew their contracts with it, and potential competitors may be effective in onboarding these or other high-quality fertility specialists to create a similarly high-quality network. There may be additional shifts in the fertility specialty provider space as the fertility market matures, and high-quality fertility specialists may become more demanding in re-negotiating to remain in its network. Its ability to develop and maintain satisfactory relationships with high-quality fertility specialists also may be negatively impacted by other factors not associated with it, such as regulatory changes impacting providers or consolidation activity among hospitals, physician groups and healthcare providers. In addition, certain organizations of physicians, such as practice management companies (which group together physician practices for administrative efficiency), may change the way in which healthcare providers do business with it and may compete directly with it, which could adversely affect its business, financial condition and results of operations. NewGenIvf intends to grant, and may continue to grant, options and other types of awards, which may result in increased share-based compensation expenses.
NewGenIvf’s Share Incentive Award will allow NewGenIvf to enhance its ability to attract and retain exceptionally qualified individuals and agents and to encourage them to acquire a proprietary interest in the company’s growth and performance. Competition for highly skilled personnel and agents is often intense and NewGenIvf may incur significant costs or may not be successful in attracting, integrating, or retaining qualified personnel and agents to fulfill NewGenIvf’s current or future needs. NewGenIvf believes that the granting of share-based awards is of significant importance to NewGenIvf’s ability to attract and retain agents, key personnel and employees, and NewGenIvf will continue to grant share-based awards in the future. In addition, NewGenIvf may, with the approval of its Compensation Committee and the Board, revise the terms of, and increase the size of, its share incentive plan, to ensure that it is able to attract and retain agents, key personnel and employees. On March 31, 2025, NewGenIvf’s Board approved certain amendments to its Share Incentive Plan, including the increase of the size of the share incentive plan to 20% of the outstanding shares of the Company from time to time. As a result, NewGenIvf’s expenses associated with share-based compensation may increase, which may have an adverse effect on NewGenIvf’s results of operations. The amended share incentive plan is available as Exhibit 4.32.
Meanwhile, the retirement or loss of certain specialists, scientific staff or other key personnel, the activities of competitors, the introduction of a competing service that is perceived to be superior to the services provided by NewGenIvf, or other events which impact NewGenIvf’s reputation could adversely affect NewGenIvf’s relationships with fertility specialists. For example, one specialist who was previously engaged by NewGenIvf brought a lawsuit against NewGenIvf regarding disputed remuneration, which resulted in a settlement for NewGenIvf to compensate the specialist with a sum of approximately US$98,000. Also, fertility specialists’ relationship with NewGenIvf could affect their behaviors in recommending NewGenIvf’s services or referring patients to NewGenIvf, which could in turn adversely impact the number of patients treated by NewGenIvf and adversely impact on its financial performance, market position and prospects.
In addition, the perceived value of NewGenIvf’s solutions and its reputation may be negatively impacted if the services provided by fertility specialists or other healthcare providers are not satisfactory to NewGenIvf’s clients, including as a result of error that could result in litigation. For example, if fertility specialist or other healthcare provider releases sensitive information of its clients, it could incur additional expenses and give rise to litigation against NewGenIvf. Any such issue with one of its providers may expose it to public scrutiny, adversely affect its brand and reputation, expose it to litigation or regulatory action, and otherwise make its operations vulnerable. Further, if its services result in less than favorable outcomes, this could cause it to fail to meet its contractually guaranteed specified service metrics, and NewGenIvf could be obligated to provide the client with a fee reduction or a second chance for free, depending on their contract terms. The failure to maintain its selective network of high-quality fertility specialists or the failure of those specialists to meet and exceed its clients’ expectation, may result in a loss of or inability to grow or maintain its client base, which could adversely affect its business, financial condition and results of operations.
The medical facilities and professionals in NewGenIvf’s network could become the subject of litigation, allegations and other claims, and NewGenIvf is not insured against these liabilities.
NewGenIvf relies on the physicians and other medical professionals of the assisted reproductive medical facilities in its network to make proper clinical decisions regarding the diagnosis and treatment of clients. However, NewGenIvf does not have full and direct control over every step of clinical activities undertaken at each of the medical facilities. In addition, physicians and medical professionals outside NewGenIvf’s network may introduce patients to NewGenIvf and conduct medical treatments and/or procedures for such patients in NewGenIvf’s facilities. NewGenIvf enters into independent contractor agreements with such physicians and medical professionals and treats such patients as NewGenIvf’s own patients. As such, NewGenIvf will have to bear any liabilities arising from their medical treatments and/or procedures conducted in NewGenIvf’s facilities. Any incorrect clinical decision or malpractice on the part of physicians and other medical professionals (including those from outside of its network), or any failure by the medical facilities in its network to properly manage their clinical activities may result in unsatisfactory treatment outcomes, patient injury or even death, which could lead to disputes with patients and/or their families or the medical professionals, including those from outside its network. In its experience, moreover, clients of fertility treatments tend to be more demanding on the medical services received. In addition, the relevant laws governing medical disputes and claims grant claimant liberal rights in bringing claims against physicians and other medical professionals practicing in the jurisdiction. As a result, the medical facilities in its network may be subject to medical disputes and claims arising under relevant laws, from time to time, which could generate substantial damages imposed on such facilities if not covered by professional liability insurance. Any dispute with its patients and/or their families or the medical professionals, including those from outside its network, or any legal proceeding involving the physicians of the medical facilities or medical professionals, including those from outside its network, regardless of its merit or eventual outcome, could result in significant legal costs and reputational damage to the medical facilities and materially and adversely affect the business, financial condition and results of operations of the medical facilities in its network, and further affect its business, financial condition and results of operations.
The assisted reproductive medical facilities in NewGenIvf’s network have limited control over the quality of the pharmaceuticals, medical equipment, medical consumables and other supplies used in its operations, and cannot guarantee that the products in use are not defective or counterfeit. NewGenIvf also has no control over independent sub-contractors and cannot guarantee the services thereof.
The assisted reproductive medical facilities in NewGenIvf’s network procure a variety of pharmaceuticals, medical equipment, consumables and other supplies in NewGenIvf’s operations from third-party suppliers. As the medical facilities in NewGenIvf’s network do not engage in the direct manufacture of such supplies, NewGenIvf cannot assure you that such supplies are free of defects and meet relevant quality standards or, in the case of imported supplies, verify the origin of such products. In addition, there may be counterfeit pharmaceutical products manufactured without proper licenses or approvals or fraudulently mislabeled with respect to their content or manufacturer in the pharmaceutical markets. In some cases these products are very similar in appearance to the authentic products. The quality control checks and processes may not be able to identify all counterfeit pharmaceutical products in the inventory. Any sale of such products by the medical facilities in NewGenIvf’s network, regardless of its knowledge as to their authenticity, may subject the medical facilities to administrative sanctions, civil claims, negative publicity or reputational damage. NewGenIvf cannot assure you that the medical facilities in our network will be able to successfully claim full indemnity from such manufacturers of counterfeit pharmaceutical products.
NewGenIvf also cannot assure you that the medical facilities in our network will not encounter incidents relating to defective products, or that such incidents will not materially and adversely affect our network of medical facilities. If the products provided by NewGenIvf’s suppliers are defective, of poor quality or are otherwise unsafe or ineffective, the medical facilities in NewGenIvf’s network could be subject to liability claims, complaints or adverse publicity, any of which would materially and adversely affect its results of operations and reputation. NewGenIvf cannot assure you that the medical facilities in NewGenIvf’s network will find suitable replacement suppliers on commercially acceptable terms or at all.
The suppliers are also subject to extensive laws, rules and regulations. If any suppliers violate applicable laws, rules and regulations, NewGenIvf’s reputation or procurement may be materially and adversely affected. In addition, the medical facilities in NewGenIvf’s network may be exposed to reputational damages or even liabilities for defective goods provided by the suppliers or negative publicity associated with any suppliers, and the business and results of operations of the medical facilities in NewGenIvf’s network and NewGenIvf could suffer as a result.
Independent sub-contractors and/or agents that work with NewGenIvf are also subject to extensive laws, rules, and regulations. If any sub-contractor and/or agent violates any applicable laws, rules, regulations or breaches any agreements, NewGenIvf’s reputation may be materially and adversely affected and NewGenIvf may be penalized by regulatory or other parties. In addition, NewgenIvf’s clients may engage Newgen’s sub-contractors and/or agents for ongoing services or additional services following the termination of contracts with NewGenIvf. NewGenIvf has no control over the services provided by sub-contractors and cannot assure the quality of such services or ensure compliance with applicable laws, rules and regulations. In addition, the services provided by independent sub-contractors may expose NewGenIvf to public scrutiny, adversely affect its brand and reputation, expose it to litigation or regulatory action, and otherwise make its operations vulnerable if such independent sub-contractors fail to meet their contractual obligations or to comply with applicable laws or regulations.
If NewGenIvf loses its relationship with one or more key pharmaceutical manufacturers, its business and results of operations could be adversely affected.
NewGenIvf maintains contractual relationships with select pharmaceutical manufacturers in Thailand, Cambodia and Kyrgyzstan. The consolidation of pharmaceutical manufacturers, the shortages of drugs provided by such manufacturers, the termination or material alteration of its contractual relationships, or its failure to renew such contracts could have a material adverse effect on its business and results of operations. Adoption of new laws, rules or regulations or changes in, or new interpretations of, existing laws, rules or regulations, relating to any of these programs could materially adversely affect its business and results of operations.
NewGenIvf has engaged in transactions with related parties, and such transactions present potential conflicts of interest that could have an adverse effect on its business and results of operations.
NewGenIvf has entered into a number of transactions with related parties. NewGenIvf may in the future enter into additional transactions with its related parties. Interests of these related parties may not necessarily be aligned with NewGenIvf’s or The Company’s interests and the interests of its other shareholders. For example, conflicts of interest may arise in connection with decisions regarding the transaction arrangements which may be less favorable to NewGenIvf than similar arrangements negotiated with unaffiliated third parties. Conflicts of interest may also arise in connection with the exercise of contractual remedies, such as the treatment of events of default. As a result, those related party transactions, individually or in the aggregate, may have an adverse effect on NewGenIvf’s business and results of operations.
NewGenIvf may be subject to claims and allegations relating to intellectual property and other causes.
NewGenIvf may from time to time receive claims that NewGenIvf infringes on the intellectual property rights of others. Moreover, NewGenIvf may be subject to claims by third parties who maintain that NewGenIvf’s service providers’ technology infringes third-party’s intellectual property rights. If NewGenIvf fails to successfully defend against such claim or does not prevail in such litigation, it could be required to modify, redesign or cease operating, pay monetary amounts as damages or enter into royalty or licensing arrangements with the valid intellectual property holders. Any royalty or licensing arrangements that NewGenIvf may seek in such circumstances may not be available to it on commercially reasonable terms or at all. Also, if NewGenIvf acquires technology licenses from third parties, NewGenIvf’s exposure to infringement actions may increase because NewGenIvf must rely upon these third parties to verify the origin and ownership of such technology. This exposure to liability could result in disruptions in NewGenIvf’s business that could materially and adversely affect NewGenIvf’s results of operations.
Some of NewGenIvf’s employees may previously employed at other companies, including NewGenIvf’s competitors. NewGenIvf may hire additional personnel to expand its development team and technical support team as its business grows. To the extent these employees were involved in the development of content or technology similar to NewGenIvf’s at their former employers, NewGenIvf may become subject to claims that these employees or NewGenIvf has appropriated these employees’ former employers’ proprietary information or intellectual properties. If NewGenIvf fails to successfully defend such claims against itself, NewGenIvf may be exposed to liabilities which could have a material adverse effect on its business.
NewGenIvf is currently not a party to any material legal or administrative proceedings but may subject to legal or administrative actions for defamation, negligence, copyright and trademark infringement, unfair competition, breach of service terms, or other purported injuries resulting from the content NewGenIvf provides or the nature of NewGenIvf’s services. Such legal and administrative actions, with or without merits, may be expensive and time-consuming and may result in significant diversion of resources and management attention from NewGenIvf’s business operations. Furthermore, such legal or administrative actions may adversely affect NewGenIvf’s brand image and reputation.
Certain data and information in this prospectus relied on by NewGenIvf were obtained from third-party data and polls. These metrics were not independently verified by NewGenIvf and may not be accurate.
Certain numbers and information in this prospectus were obtained and provided from numerous sources including management data, third-party data or numbers generally estimated by calculating infertile couples, fertility tourism number, etc. to generally assess potential customer numbers in Asia-Pacific countries.
These metrics were not independently verified. Such databases, third-party information, and calculations may not accurately reflect actual statistics or numbers and NewGenIvf does not have access to specific rating numbers. Similarly, any statistical data in any third-party publications also include projections based on a number of assumptions. If any one or more of the assumptions underlying the market data is later found to be incorrect, actual results may differ from the projections based on these assumptions.
NewGenIvf is dependent on a single joint venture partner for our real estate development project in the UAE and are subject to the legal, regulatory, and economic risks of operating in a new jurisdiction.
Our real estate development project in Ras Al Khaimah, UAE, is conducted through a joint venture with BNW Real Estate Development LLC (“BNW”), on whom we are heavily reliant for the financing, construction, management, and marketing of the project. Any failure by BNW to fulfill its obligations, including covering construction costs and achieving sales targets, could jeopardize the project’s success and our anticipated returns. Furthermore, our lack of an established operating history in the UAE makes us vulnerable to unforeseen complexities in the local legal, regulatory, and business environment. This includes potential challenges in navigating local property laws, tax regulations, and commercial practices, as well as exposure to regional geopolitical tensions or economic downturns that could specifically impact the Emirate of Ras Al Khaimah. Disagreements with our joint venture partner, or the termination of our relationship, could result in disputes, delays, financial losses, and a complete failure to realize the projected US$67 million in net returns, which would have a material adverse effect on our business, financial condition, and results of operations.
Risks Related to Government Regulation
NewGenIvf operates in a highly regulated industry and must comply with a significant number of complex and evolving requirements. Any lack of requisite approvals, licenses, or permits applicable to NewGenIvf’s business may have a material and adverse impact on NewGenIvf’s business, financial condition, and results of operations.
The operations of NewGenIvf are subject to various laws, rules and regulations at the national, regional and local levels in Thailand, Cambodia, Kyrgyzstan, Hong Kong, United Arab Emirates, United States of America and other applicable jurisdictions. Such laws and regulations mainly relate to (i) the licensing of local and foreign medical professionals, nursing professionals, medical technology professionals, pharmaceutical professions and other applicable licensing; (ii) the licensing, registration, and accreditation of medical facilities, laboratories, including but not limited to the licensing, registration, and accreditation of persons performing related activities; (iii) the privacy and security of confidential patient medical records; (iv) the corporate practice of medicine; (v) healthcare fraud and abuse laws; (vi) the donation and transplantation of human cells, tissues and organs; (vii) potential prohibition on surrogacy or providing intermediary assistance in surrogacy; and (viii) licensing and approval of the accommodation provided as parts of the services.
NewGenIvf has attempted to structure its operations to comply with laws, regulations and other requirements applicable to it directly and to its clients and vendors, but there can be no assurance that its operations will not be challenged or impacted by regulatory authorities or enforcement initiatives, or that the relevant authorities in each jurisdiction could impose higher standards or requirements, which NewGenIvf may have difficulty to adhere to, e.g. Medical Facilities Act B.E. 2541 (1998) and Protection of a Child Born by Medically Assisted Reproductive Technology Act B.E. 2558 (2015) for Thailand jurisdiction, Law on Reproduction Rights and on Guarantees of Their Realization of July 4, 2015 No. 148, Law on status of medical worker of May 28, 2013 No. 81 and Temporary Regulation on Procedure of Licensing Private Medical Activity approved by the resolution of government of April 4, 2017 No. 203 for Kyrgyz Republic. NewGenIvf in the future may become involved in governmental investigations, audits, reviews and assessments. Any determination by a court or agency that NewGenIvf’s solutions or services violate, or cause its clients to violate, applicable laws, regulations or other requirements could subject it or its clients to civil, criminal, or administrative penalties. Such a determination also could require it to change or terminate portions of its business, disqualify it from serving clients that do business with government entities, or cause it to refund some or all of its service fees or otherwise compensate its clients. In addition, failure to satisfy laws, regulations or other requirements could adversely affect demand for its solutions and could force it to expend significant capital, research and development and other resources to address the failure. Even an unsuccessful challenge by regulatory and other authorities or parties could be expensive and time-consuming, could result in loss of business, exposure to adverse publicity, and injury to its reputation and could adversely affect its ability to retain and attract clients. If NewGenIvf fails to comply with applicable laws, regulations and other requirements, its business, financial condition and results of operations could be adversely affected. Such non-compliance could also require significant investment to address and may prove costly. There are several additional state statutes, regulations, guidance and contractual provisions related to or impacting the healthcare industry that may apply to its business activities directly or indirectly, including, but not limited to:
| ● | Licensing and Licensed Personnel. Many countries have licensure or registration requirements for entities acting as a medical services provider. The scope of these laws differs from country to country, and the application of such laws to the activities of fertility treatment is often unclear. Given the nature and scope of the solutions and services that NewGenIvf provides, it is required to maintain the License to Operate Medical Facility Business (Sor.Por.7), the License to Manage Medical Facility Business (Sor.Por.19), License to Certify the Standard of Service relating to Medically Assisted Reproductive Technology (KorThorPhor.9), and personnel licenses, i.e., license of medical professionals, nursing professionals, medical technology professionals, pharmaceutical professions and other applicable licenses in Thailand, Approval on Opening of Medical Clinic, Approval on Opening of Pharmacy and relevant approvals to conduct IVF, embryo implant and/or transfer activities issued by the Ministry of Health of Cambodia (“Cambodia MOH”) in Cambodia and licenses to carry out private medical activities (including diagnostics and treatment gynecological diseases, supervision of pregnant women before childbirth, IVF in outpatient and day hospital conditions (for four (4) beds)) in Kyrgyzstan, respectively, and to ensure that such licenses and registrations are in good standing on an annual basis. NewGenIvf is licensed, has licensure applications pending before appropriate regulatory bodies, is exempt from licensure or registration, or is otherwise authorized under such laws in those countries in which it provides its services. These licenses require it to comply with the rules and regulations of the governmental bodies that issued such licenses. NewGenIvf’s failure to comply with such rules and regulations could result in criminal and/ or administrative penalties, the suspension of a license, or the loss of a license, all of which could negatively impact its business. First Fertility PGS had provided arrangements of accommodation without additional charges for its patients without a tourism license in Thailand, all of which was subsequently ceased in early 2023. Pursuant to the Tourism Business and Guide Act 2551 (2008) of Thailand, a maximum fine of THB500,000 may be imposed on First Fertility PGS as a result of the above activity without a tourism license in Thailand. NewGenIvf is unable to predict, however, how its services may be viewed by regulators over time, how these laws and regulations will be interpreted, or the full extent of their applicable. If a regulatory authority in any country determines that the nature of its business requires that NewGenIvf be licensed under applicable laws, it may need to restructure its business or it may need to comply with any related requirements, such as obtaining relevant license, paying additional regulatory fees and/or penalties for previous non-compliance with relevant licensing requirements, which could adversely affect its results of operation. Additionally, in extreme case, NewGenIvf may need to cease operations until it is able to obtain appropriate licensure, which may adversely affect its revenue for a period of time that it cannot estimate. |
| ● | Patients’ Right Protection. Patients’ Right Protection has been significantly reshaped by recent legal developments across Thailand, Cambodia, and Kyrgyzstan, creating an increasingly complex and litigious environment that materially elevates NewGenIvf’s exposure to malpractice claims, regulatory penalties, and reputational harm. In Thailand, a landmark 2025 ruling by the Official Information Board’s Appeal Committee has procedurally strengthened claimants’ ability to pursue medical negligence litigation by confirming that authorized heirs and representatives are entitled to access deceased patients’ medical records for tort lawsuits, explicitly holding that such requests do not constitute impermissible access to “another person’s health information” under Section 7 of the National Health Act B.E. 2550 (2007) and thereby removing a previously available privacy-based defense for healthcare providers. Concurrently, Thailand’s Personal Data Protection Act (PDPA) enforcement has intensified dramatically, with the Personal Data Protection Committee imposing administrative fines totaling approximately 21.5 million baht in August 2025 alone—including a landmark 1.21 million baht penalty against a private hospital following a catastrophic breach wherein over 1,000 patient records were leaked and repurposed as food wrappers by an unsupervised disposal contractor—and the April 2025 clarification of administrative fine procedures now exposes healthcare providers to direct monetary sanctions for data protection failures independent of any underlying malpractice claim. In Cambodia, there are no amendments to the core patients’ rights instruments (Civil Code, Law on Donation and Transplantation 2016, Sub-Decree No. 61 on Medical Ethics 2003), the National Assembly is actively debating a December 2025 draft Resolution on breakthrough healthcare policies for 2026-2035 that, although primarily focused on state sector capacity, signals an elevated policy focus on “improving medical ethics” and “patient satisfaction” that may presage future regulatory tightening. In Kyrgyzstan, a November 2025 Presidential Decree (No. 331) introduces a year-long approbation regime granting managerial and financial autonomy to select state healthcare organizations, while an October 2025 law mandating a five-year public sector service requirement before medical professionals may enter private practice directly restricts the labor market for private providers and may heighten patient expectations regarding service quality standards. Collectively, these developments—Thailand’s pro-disclosure precedent and aggressive data protection enforcement, Cambodia’s embryonic policy shift toward enhanced patient protections, and Kyrgyzstan’s structural healthcare reforms constraining private practice—compound the inherent risks of adverse outcomes in complex medical treatment, exposing NewGenIvf to increased litigation frequency, substantial regulatory fines, and cascading reputational damage across its entire operational footprint. |
Meanwhile, Thailand is considering enacting a Patient Protection Bill (the “Bill”). The Bill, if issued, is intended to alleviate disputes between patients and healthcare providers, which have an impact on the healthcare system in Thailand as a whole. The compensation outlined in the Bill will assist patients in claiming damages, thereby fostering a positive relationship between patients and healthcare providers. Consequently, the rate of disputes is expected to decrease. The provisions under the Bill would require healthcare providers to compensate patients in a timely manner, sometimes without requiring proof of wrongdoing. The Bill also contemplates setting up a patient protection fund for damages to patients pursuant to which healthcare providers have to make mandatory contributions according to the rules determined by a patient protection committee. Failure by it to comply with applicable rules and regulations could result in penalties, the loss of regulatory permits and damage to NewGenIvf’s business reputation, each of which could have a material adverse effect on its financial condition and results of operations.
Furthermore, the Protection of A Child Born By Medically Assisted Reproductive Technology Act B.E. 2558 (2015) of Thailand was promulgated with the intention to appropriately designate the legitimate parenthood status of a child born using medically assisted reproductive technology and regulate any medical scientific research on embryology and medically assisted reproductive technologies to prevent the misuse of medically assisted reproductive technologies. NewGenIvf is therefore under the supervision of a Committee of the Protection for Children Born through Medically Assisted Reproductive Technology, which is a committee established to control, inspect, supervise and formulate various policies relating to such acts. In Cambodia and Kyrgyzstan, all health establishments, including private medical clinics, are under the supervision of the Cambodia MOH and the Ministry of Health of Kyrgyzstan, respectively, which each governs and regulates the operation of medical clinics and activities of medical practitioners in respective countries. In particular, the Medical Council of Cambodia, Cambodian Council of Nurses, Cambodian Midwives Council and The Pharmaceutical Council of Cambodia, all assist the Cambodia MOH to supervise and monitor the practice of health professionals in Cambodia. IVF/embryo implant/transfer activities are subject to an approval by the Cambodia MOH.
| ● | Privacy and Security Requirements. There are numerous laws and regulations related to the privacy and security of health information in each country. In particular, regulations promulgated pursuant to the Personal Data Protection Act B.E. 2562 (2019) of Thailand (“PDPA”), Law on Data of Personal Character of April 14, 2008 No. 58 of Kyrgyzstan (“Data Protection Law”), as well as Regulation of Registration of Personal Data Holders (Owners) approved by the Resolution of the Cabinet of Ministers of KR of November 18, 2022, Offences Code No. 128 of October 28, 2021 of Kyrgyzstan establish privacy and security standards in each country that limit the collection, use, and/ or disclosure of certain individually identifiable health information, whether directly or indirectly (excluding the information of the deceased person) and require the implementation of administrative, physical and technological safeguards to protect the privacy of protected health information and ensure the confidentiality, integrity and availability of electronic protected health information. The privacy regulations established under the PDPA and Data Protection Law also provide patients with rights related to understanding and controlling how their protected health information is collected, used and/ or disclosed. As a provider of services to entities subject to the PDPA and Data Protection Law, NewGenIvf is directly subject to certain provisions of the regulations. To the extent permitted by applicable privacy regulations and contracts with its clients, NewGenIvf is permitted to use and disclose protected health information to perform its services and for other limited purposes, but other uses and disclosures, such as marketing communications, require written authorization from the patient or must meet an exception specified under the privacy regulations. |
NewGenIvf also has downstream entities which provide it with services and are also subject to applicable regulations. If NewGenIvf or any of its downstream entities are unable to properly protect the privacy and security of protected health information entrusted to it, it could be found to have breached its contracts with its clients and be subject to investigation by the relevant supervision institution, i.e., the Office of the Personal Data Protection Committee of Thailand (the Government Authority under the PDPA), the Cambodia MOH and the State Data Protection Agency under the Cabinet of Ministers of Kyrgyzstan (the “Agency”). In the event the Office of the Personal Data Protection Committee or the Agency finds that NewGenIvf has failed to comply with applicable privacy and security standards, it could face civil, criminal, and/ or administrative penalties. In addition, the Office of the Personal Data Protection Committee performs compliance audits in order to proactively enforce the privacy and security standards. The Office of the Personal Data Protection Committee has become an increasingly active regulator and has signaled its intention to continue this trend. The Office of the Personal Data Protection Committee has the discretion to impose penalties and may require companies to enter into resolution agreements and corrective action plans which impose ongoing compliance requirements. The Office of the Personal Data Protection Committee’s enforcement activity, or audit related to incident regarding it or its downstream entity, can result in financial liability and reputational harm, and responses to such enforcement activity can consume significant internal resources. Although NewGenIvf has implemented and maintain policies, processes and compliance program infrastructure to assist it in complying with these laws and regulations and its contractual obligations, NewGenIvf cannot provide assurance regarding how these laws and regulations will be interpreted, enforced or applied to its operations. In associated with enforcement activities and potential contractual liabilities, its ongoing efforts to comply with evolving laws and regulations might also require it to make costly system purchases and/or modifications or otherwise divert significant resources to compliance initiatives from time to time.
| ● | Other Privacy and Security Requirements. In addition, numerous other laws govern the collection, dissemination, use, access to and confidentiality of personal information. For example, the Law on E-Commerce of Cambodia (2019) places an obligation on those who electronically store private information to use all means to ensure that the information is protected by security safeguards in every reasonable circumstance to avoid the loss, access, use, modification, leakage, or disclosure of the information, except with the consent of the data owner or other lawfully authorized party. The Law on E-Commerce also prohibits individuals from dishonestly accessing, downloading, copying, extracting, leaking, deleting, modifying, or otherwise interfering with data stored by other persons. Applicable laws are contributing to increased enforcement activity and may also be subject to interpretation by various courts and other governmental authorities. |
Certain of NewGenIvf’s solutions and services involve the transmission and storage of client data in various jurisdictions, which subjects the operation of those solutions and services to privacy or data protection laws and regulations in those jurisdictions. While NewGenIvf believes those solutions and services comply with current regulatory and security requirements in the jurisdictions in which it provides these solutions and services, there can be no assurance that such requirements will not change or that it will not otherwise be subject to legal or regulatory actions. The laws and regulations are rapidly evolving and changing, and could have an adverse impact on its operations. These laws and regulations are subject to uncertainty in how they may be interpreted and enforced by government authorities and regulators. The costs of compliance with, and the other burdens imposed by, these and other laws or regulatory actions may increase its operational costs, prevent it from providing its solutions, and/or impact its ability to invest in or jointly develop its solutions. NewGenIvf also may face audits or investigations by one or more government agencies relating to its compliance with these laws and regulations.
An adverse outcome under any such investigation or audit could result in fines, penalties, other liability, or could result in adverse publicity or a loss of reputation, and adversely affect NewGenIvf’s business. Any failure or perceived failure by it or by NewGenIvf’s solutions to comply with these laws and regulations may subject it to legal or regulatory actions, damage its reputation or adversely affect its ability to provide its solutions in the jurisdiction that has enacted the applicable law or regulation. Moreover, if these laws and regulations change, or are interpreted and applied in a manner that is inconsistent with its policies and processes or the operation of its solutions NewGenIvf may need to expend resources in order to change its business operations, policies and processes or the manner in which it provides its solutions. This could adversely affect NewGenIvf’s business, financial condition and results of operations.
| ● | Data Protection and Breaches. In recent years, there have been a number of well-publicized data breaches involving the improper dissemination of personal information of individuals both within and outside of the healthcare industry. Pursuant to the applicable data protection law of Thailand, the PDPA requires businesses to notify the data subjects and/or the government authorities upon the occurrence of a data breach. The laws are not consistent, and compliance in the event of a widespread data breach is costly. Each country also constantly amending existing laws, requiring attention to frequently changing regulatory requirements. Most countries require holders of personal information to maintain safeguards and take certain actions in response to a data breach, such as providing prompt notification of the breach to affected individuals. In some countries, these laws are limited to electronic data, but they increasingly are enacting or considering stricter and broader requirements. |
Despite NewGenIvf’s security management efforts with respect to physical and technological safeguards, employee training, vendor (and sub-vendor) controls and contractual relationships, its infrastructure, data or other operation centers and systems used in its business operations, including the internet and related systems of its vendors (including vendors to whom NewGenIvf outsources data hosting, storage and processing functions) are vulnerable to, and may from time to time experience, unauthorized access to data and/or breaches of confidential information due to a variety of causes. Techniques used to obtain unauthorized access to or compromise systems change frequently, are becoming increasingly sophisticated and complex, and are often not detected until after an incident has occurred. As a result, NewGenIvf might not be able to anticipate these techniques, implement adequate preventive measures, or immediately detect a potential compromise. If its security measures, some of which are managed by third parties, or the security measures of its service providers or vendors, are breached or fail, it is possible that unauthorized or illegal access to or acquisition, disclosure, use or processing of personal information, confidential information, or other sensitive client or employee data, including protected health information, may occur. A security breach or failure could result from a variety of circumstances and events, including third-party action, human negligence or error, malfeasance, employee theft or misuse, phishing and other social engineering schemes, computer viruses, attacks by computer hackers, failures during the process of upgrading or replacing software, databases or components thereof, power outages, hardware failures, telecommunication failures, and catastrophic events. If NewGenIvf’s security measures, or those of its service providers or vendors, were to be breached or fail, its reputation could be severely damaged, adversely affecting client or investor confidence. As a result, clients may curtail their use of or stop using its offering and its business may suffer. In addition, NewGenIvf could face litigation, damages for contract breach, penalties and regulatory actions for violation of laws or regulations applicable to data protection and significant costs for remediation and for measures to prevent future occurrences. In addition, any potential security breach could result in increased costs associated with liability for stolen assets or information, repairing system damage that may have been caused by such breaches, incentives offered to clients or other business partners in an effort to maintain the business relationships after a breach and implementing measures to prevent future occurrences, including organizational changes, deploying additional personnel and protection technologies, training employees and engaging third-party experts and consultants. Negative publicity may also result from real, threatened or perceived security breaches affecting it or its industry or clients, which could cause it to lose clients or partners and adversely affect its operations and future prospects. NewGenIvf may not carry insurance or maintain coverage sufficient to compensate for all liability and such insurance may not be available for renewal on acceptable terms or at all, and in any event, insurance coverage would not address the reputational damage that could result from a security incident.
| ● | Fraud and Abuse Laws. NewGenIvf’s operations in Thailand, Cambodia, and Kyrgyzstan are exposed to a complex web of anti-corruption and fraud laws. While our services are not directly reimbursed by government healthcare programs—which often serve as the primary trigger for such laws—we are still subject to general anti-corruption statutes, including the Organic Act on Anti-Corruption B.E. 2561 (2018) (as amended) and the Penal Code of Thailand; the Criminal Code of Cambodia; and the Criminal Code and the Law on the Prevention of Corruption of the Kyrgyz Republic.These laws are often broad and vague, and judicial interpretations can be inconsistent. Recent legislative changes have heightened the risk profile. For instance, Thailand’s Organic Act on Anti-Corruption (No. 2) B.E. 2568 (2025) now provides formal immunity and state protection to whistleblowers, increasing the likelihood that alleged misconduct could be reported to authorities. Similarly, recent amendments in Kyrgyzstan (Law No. 209, effective January 2025) have eliminated the option of fines for corruption offenses, mandating imprisonment and lifetime bans from public office for convicted individuals. Although NewGenIvf might engage regulatory experts to review practices in an effort to comply with all applicable laws, NewGenIvf cannot predict how these laws or their judicial interpretations will evolve. If a regulatory authority determines that NewGenIvf’s activities—or those of NewGenIvf’s clients, vendors, or partners—violate any of these laws, NewGenIvf could face severe civil or criminal penalties and might be required to enter into corporate integrity agreements or similar undertakings, incurring substantial ongoing compliance costs. Such a determination could also disqualify NewGenIvf from providing services to clients, resulting in a material adverse effect on its business, financial condition, and results of operations. Furthermore, even an unsuccessful challenge or investigation by a regulatory authority could result in significant adverse publicity, damage our reputation, and require us to incur substantial costs in responding to such actions. |
| ● | Consumer Protection Laws. Consumer protection laws are being applied increasingly by the Office of the Consumer Protection Board in Thailand and by the Cambodia Ministry of Health to regulate the collection, use, storage and disclosure of personal or health information, through websites or otherwise, and, in Cambodia, by the Consumer Protection Competition and Fraud Repression Directorate-General, to regulate the presentation of website content. Courts may also adopt the standards for fair information practices, which concern consumer notice, choice, security and access. |
| ● | Restrictions on Communication. Communications with NewGenIvf’s clients increasingly may be subject to and restricted by laws and regulations governing communications via telephone, fax, text, and email. NewGenIvf also uses email and social media platforms as marketing tools. For example, NewGenIvf maintains social media accounts. As laws and regulations rapidly evolve to govern the use of these platforms and devices, the failure by it, its employees or third parties acting at its direction to abide by applicable laws and regulations in the use of these platforms and devices could adversely impact its business, financial condition and results of operations or subject it to fines or other penalties. |
| ● | Advertisement Laws. NewGenIvf’s advertisement and announcements, in particular, the messages releasing on the Internet related to medical facilities may subject to the laws and regulations of relevant jurisdictions (and potential prohibition in Cambodia on commercial advertisement of private medical services). |
For example, in Thailand, NewGenIvf shall apply for and obtain the approval and/ or pre-approval from the relevant authority for the images, and text used in advertisements or announcements which shall be in accordance with the Medical Facility Act B.E. 2541 (1998) (and its amendments) and the Notification of the Department of Health Services Support on Rules, Procedures, Conditions, and Costs of Advertisements or Announcements of Healthcare Facilities B.E. 2562 (2019) (and its amendments) and the Operational Manual for Approval of Advertisements or Announcements relating to Healthcare Facilities. If such approval was not obtained by NewGenIvf, it could lead to significant liabilities and consequences, which could adversely impact NewGenIvf’s business, financial condition and results of operations or subject its sales and marketing director to personal liabilities.
For Cambodia, Prakas 028 on Advertisement of Private Medical, Paramedical and Medical Aid Practices dated August 23, 2004 issued by the Cambodia MOH prohibits commercial advertising of private medical services. Advertisement of private health care services is only allowed for any advertisements within the professional framework not affecting the ethics of private medical services and such advertisement requires a permit from the Cambodia MOH. In addition, the Royal Government of Cambodia has recently issued Sub-Decree 232 on the Management of Commercial Advertisements of Goods and Services on November 4, 2022 to provide the legal framework for the management of commercial advertising of goods and services for all types, forms and means in Cambodia. In light of this Sub-Decree, in addition to the permit requirement of the Cambodia MOH, a person wishing to advertise their goods and/or services in Cambodia may also apply for a compliance certificate from the Ministry of Commerce, which certifies that advertising text or content complies with the Law on Consumer Protection or other applicable regulations.
For Kyrgyzstan, the Law on Advertisements of December 24, 1998, No. 155 requires that if the activities of the advertiser subject to licensing, the advertisement of such advertiser must include the license number and the name of the authority that issued the license, except for radio advertising, where it is sufficient to state “licensed activity” on the territory of Kyrgyzstan. In advertising goods (including works and services), and other objects of advertising, cost indicators must be stated in the national currency. There are also other requirements established in relation to size, frequency, cost and other features of advertisements via different types of media.
New laws and regulations relevant to the fertility services may be introduced in the future, or the current applicable regulations may otherwise be amended or replaced requiring the assisted reproductive medical facilities in its network to conduct business with additional oversight and regulatory compliance. If NewGenIvf fails to obtain the necessary licenses, permits and approvals, NewGenIvf may be subject to fines, confiscation of revenues generated from incompliance operations, or the suspension of relevant operations. NewGenIvf may also experience adverse publicity arising from such non-compliance with government regulations that negatively impacts its brand. NewGenIvf may experience difficulties or failures in obtaining the necessary approvals, licenses, and permits for new spaces or new service offerings. If NewGenIvf fails to obtain the material licenses, NewGenIvf’s business activities could be severely delayed. In addition, there can be no assurance that NewGenIvf will be able to obtain, renew, and/or convert all of the approvals, licenses, and permits required for its existing business operations upon their expiration in a timely manner, in a cost-efficient manner or at all, which could adversely affect NewGenIvf’s business operations and financial condition.
In addition, considerable uncertainties exist in relation to the interpretation and implementation of existing and future laws and regulations governing NewGenIvf’s business activities. NewGenIvf could be found not in compliance with any future laws and regulations or of the laws and regulations currently in effect due to changes in the relevant authorities’ interpretation of those laws and regulations. It is possible that different interpretations or enforcement of these regulations could subject the current or past practices to allegations of impropriety or illegality or require the medical facilities in its network to implement changes in the facilities, equipment, personnel or services, or increase capital expenditure and operating expenses. If NewGenIvf fails to complete, obtain, or maintain any of the required licenses or approvals or make the necessary filings, NewGenIvf may be subject to various penalties, such as confiscation of unlawful gains, the imposition of fines, revocation of licenses, and the discontinuation or restriction of NewGenIvf’s operations. Any such penalties or changes in policies, regulations, or enforcement by government authorities may disrupt NewGenIvf’s operations and materially and adversely affect NewGenIvf’s business, financial condition, and results of operations.
Legal or regulatory restriction, government regulation, industry standards and other requirements create risks and challenges with respect to NewGenIvf’s compliance efforts and its business strategies and could adversely impact NewGenIvf’s business and limited the growth of NewGenIvf’s operations.
The healthcare industry is highly regulated and subject to frequently changing laws, regulations, industry standards and other requirements. Many healthcare laws and regulations are complex, and their application to specific solutions, services and relationships may not be clear. In particular, many existing healthcare laws and regulations, when enacted, did not anticipate the solutions and services that NewGenIvf provides, and these laws and regulations may be applied to its solutions and services in ways that NewGenIvf does not anticipate. Efforts to reform or revise aspects of the healthcare industry or to revise or create additional legal or and regulatory requirements could impact its operations, the use of its solutions and services, and its ability to market new solutions and services, or could create unexpected liabilities for it. NewGenIvf also may be impacted by laws, industry standards and other requirements that are not specific to the healthcare industry, such as consumer protection laws and payment card industry standards. These requirements may impact its operations and, if not followed, could result in fines, penalties and other liabilities and adverse publicity and injury to its reputation.
There is a risk that existing or future laws may be interpreted in a manner that is not consistent with the healthcare industry’s current practices and could have an adverse effect on NewGenIvf’s business, financial condition, results of operations and growth prospects.
Advertising or offering of gender selection services in certain jurisdictions in which NewGenIvf operates could expose the Company and its directors and executive officers to significant legal and regulatory risks, including potential penalties.
The marketing or provision of gender selection services in certain jurisdictions in which we operate is subject to stringent legal restrictions. In particular, in Hong Kong, under the Human Reproductive Technology Ordinance (Cap. 561 of the Laws of Hong Kong) (“HRTO”), gender selection for non-medical purposes is prohibited. Violation of these provisions may result in criminal liability. Any person found guilty of advertising or offering gender selection services in Hong Kong may, on a first conviction, face a fine of up to HKD 25,000 and imprisonment for up to 6 months. If we, our representatives, or our sub-contractors inadvertently violate, or are alleged to violate, these laws, we could face significant financial penalties, reputational harm, and potential legal proceedings. Even if we do not directly market our gender selection services in Hong Kong, promotional activities conducted by third parties, affiliates, or other associates on our behalf could also expose us to liability under these laws.
On April 25, 2025, Wing Fung Alfred Siu and Hei Yue Tina Fong, and other individuals, were arrested in Hong Kong for allegedly violating certain sections of the HRTO which prohibit using reproductive technologies for sex selection, advertising sex selection services, commercial surrogacy arrangements, and advertising surrogacy arrangements. All persons were released on a bail amount of HKD 3,000 ($390). No further action has been taken by prosecutors against arrested persons, except as detailed below with respect to Wing Fung Alfred Siu. Under section 26 of the Magistrates Ordinance (Cap. 22 of the Laws of Hong Kong), in any case of an offence, other than an indictable offence, charges must be laid within 6 months from the time when the matter arose. As the alleged violations of the HRTO are triable summarily only, and charges were not laid within 6 months of the arrest, the statutory limitation period for instituting proceedings for the alleged offences has lapsed.
On October 26, 2025, Wing Fung Alfred Siu was served a summons for allegedly violating section 17(2) of the HRTO which prohibits commercial surrogacy arrangements and advertising surrogacy arrangements in his capacity as director of Harcourt Limited. Harcourt Limited is a Hong Kong company that is not part of NIVF. On November 18, 2025, Wing Fung Alfred Siu was further granted bail on a bail amount of HKD 10,000 ($1,285). If convicted with an offence, he could face a fine of up to HKD 25,000 (approximately $3,200), and imprisonment for up to 6 months. Although we do not expect custodial sentences for him, any custodial sentence given could materially impact our business, financial condition, or results of operations. In addition, although under BVI law, such minor first-time offenses would not automatically disqualify any directors or executives from continuing to act in such positions, any director or executive officer found guilty of such offenses could cause reputational harm to the Company. As we do not advertise or promote surrogacy arrangement or gender selection service in Hong Kong, and all such marketing activities are carried out outside of Hong Kong or by agents outside of Hong Kong, we have not been, nor do we expect to be, materially impacted by any such increased enforcement of the HRTO. Moreover, we do not expect any material impact on our ability to generate revenue from Chinese clients, as our PRC-sourced revenues derived from clients in mainland China and Hong Kong are generated through referrals from PRC agents, with payments being made by the clients directly to the local clinics. However, if the regulatory authorities in Hong Kong deem our determination inconsistent from the HRTO, we may face increased scrutiny or enforcement actions by the Hong Kong authorities, which could result in adverse publicity, increase compliance cost, and significant disruption of our business operations in the region.
Additionally, increased scrutiny or enforcement actions by regulatory authorities in other jurisdictions in which we operate could result in adverse publicity, increased compliance costs, and significant disruption of our business operations. As a result, any failure to comply with applicable laws and regulations related to gender selection services could have a material adverse effect on our business, financial condition, and results of operations.
Significant tariffs or other restrictions imposed on imports by the U.S. and related countermeasures taken by impacted countries could have a material adverse effect on our operations and financial results.
If significant tariffs or other restrictions are imposed on imports by the U.S. and related countermeasures are taken by foreign countries, our business, including results of operations, cash flows and financial condition, may be adversely affected. In January 2025, during the initial days of U.S. President Trump’s second term, the U.S. announced the imposition of additional substantial tariffs on imports from various countries, including China, Canada and Mexico, and the subject countries have imposed or indicated their intention to impose counter measures. In February 2025, the U.S. imposed tariffs of 10% on all imported goods from China, followed by an additional 10% tariff in March 2025. The U.S. also imposed a 25% tariff on all steel and aluminum imports, beginning in March 2025. On February 13, 2025, President Trump ordered his trade advisers to come up with “reciprocal” tariffs on U.S. trade partners to retaliate against taxes, tariffs, regulations and subsidies and on April 2, 2025, announced new tariffs on many U.S. trading partners, including a universal baseline tariff of 10% on all imported goods, and country specific tariffs such as an additional 34% tax on imports from China (leading to an effective rate of 54% when combined with existing tariffs) and 20% on products from the E.U. Specific products that are being tariffed, such as automobiles, were to be exempted from the new tariffs, and tariffs on products such as pharmaceutical drugs were to be announced at a later date. Following a period of market turbulence, on April 9, 2025, President Trump announced a 90-day pause to the tariffs announced on April 2, 2025 for most countries. Countries subject to the pause on the tariffs are still to be subject to the baseline 10% tariff. This consequently lowers the tariff rate for the E.U., Japan, and South Korea, among other countries. However, U.S. President Trump announced an increased tariff rate against Chinese imports of a minimum 145%. These and other tariffs and countermeasures could increase the cost of equipment necessary for our operations, disrupt global supply chains and create additional operational challenges. Additionally, ongoing trade tensions and uncertainty regarding future trade policies could negatively impact global economic conditions and consumer confidence, further affecting our business performance.
Any litigation against NewGenIvf could be costly and time-consuming to defend and could harm its business, financial condition and results of operations.
NewGenIvf has in the past and may in the future become subject to regulatory actions, litigation, disputes, or claims of various types, legal proceedings and claims that arise in the ordinary course of business, such as claims brought by its clients or vendors in connection with commercial disputes or employment claims made by its current or former employees, as well as claims brought by relevant regulatory authorities or NewGenIvf’s competitors, patients, employees, or other third parties against NewGenIvf. NewGenIvf is unable to predict the outcome of any of these legal proceedings. Such regulatory actions, disputes, allegations, complaints, or legal proceedings may damage NewGenIvf’s reputation, evolve into litigation, or otherwise have a material adverse impact on NewGenIvf’s reputation and business. Such proceedings might result in substantial costs, regardless of the outcome, and may significantly divert management’s attention and resources from operating NewGenIvf’s business, which might seriously harm its business, financial condition and results of operations. Insurance might not cover such claims, might not provide sufficient payments to cover all the costs to resolve one or more such claims, and might not continue to be available on terms acceptable to it. A claim brought against it that is uninsured or underinsured could result in unanticipated costs, potentially harming its business, financial condition and results of operations. The outcomes of actions NewGenIvf institutes may not be successful or favorable to NewGenIvf. Lawsuits against NewGenIvf may also generate negative publicity that significantly harms NewGenIvf’s reputation, which may adversely affect NewGenIvf’s client base. NewGenIvf may also need to pay damages or settle lawsuits with a substantial amount of cash.
Acquisitions, strategic investments, partnerships, or alliances could be difficult to identify, pose integration challenges, divert the attention of management, disrupt NewGenIvf’s business, dilute stockholder value, and adversely affect its business, financial condition and results of operations.
NewGenIvf may in the future seek to acquire or invest in businesses, joint ventures, products and services, or technologies that it believes could complement or expand its platform, enhance its technical capabilities, or otherwise offer growth opportunities. Any such acquisition or investment may divert the attention of management and cause NewGenIvf to incur various expenses in identifying, investigating and pursuing suitable opportunities, whether or not the transactions are completed, and may result in unforeseen operating difficulties and expenditures. In particular, NewGenIvf may encounter difficulties assimilating or integrating the businesses, technologies, products and services, personnel or operations of the acquired companies, particularly if the key personnel of the acquired company choose not to work for it, they are operationally difficult to integrate, or NewGenIvf has difficulty retaining the clients of any acquired business due to changes in ownership, management or otherwise. These transactions may also disrupt its business, divert its resources, and require significant management attention that would otherwise be available for development of its existing business and may not benefit NewGenIvf’s business strategy, may not generate sufficient revenues to offset the associated acquisition costs or may not otherwise result in the intended benefits. Any such transactions that NewGenIvf is able to complete may not result in any synergies or other benefits it had expected to achieve, which could result in impairment charges that could be substantial. In addition, NewGenIvf may not be able to find and identify desirable acquisition targets or business opportunities or be successful in entering into an agreement with any particular strategic partner. These transactions could also result in dilutive issuances of equity securities or the incurrence of debt, which could adversely affect its results of operations. In addition, if the resulting business from such a transaction fails to meet NewGenIvf’s expectations, or it fails to successfully integrate such businesses into its own, its business, financial condition and results of operations may be adversely affected or it may be exposed to unknown risks or liabilities. Even when NewGenIvf identifies an appropriate acquisition or investment target, it may not be able to negotiate the terms of the acquisition or investment successfully, obtain financing for the proposed transaction, or integrate the relevant businesses into its existing business and operations. Strategic investments or acquisitions will involve risks commonly encountered in business relationships, including:
| ● | difficulties in assimilating and integrating the operations, personnel, systems, data, technologies, products and services of the acquired business; |
| ● | inability of the acquired technologies, products or businesses to achieve expected levels of revenue, profitability, productivity or other benefits; |
| ● | difficulties in retaining, training, motivating and integrating key personnel; |
| ● | diversion of management’s time and resources from NewGenIvf’s normal daily operations; |
| ● | difficulties in maintaining uniform standards, controls, procedures and policies within the combined organizations; |
| ● | difficulties in retaining relationships with customers, employees and suppliers of the acquired business; |
| ● | risks of entering markets in which NewGenIvf have limited or no prior experience; |
| ● | regulatory risks, including remaining in good standing with existing regulatory bodies or receiving any necessary pre-closing or post-closing approvals, as well as being subject to new regulators with oversight over an acquired business; |
| ● | assumption of contractual obligations that contain terms that are not beneficial to NewGenIvf, require it to license or waive intellectual property rights or increase its risk for liability; |
| ● | failure to further successfully develop the acquired technology; |
| ● | liability for activities of the acquired business before the acquisition, including intellectual property infringement claims, violations of laws, commercial disputes, tax liabilities and other known and unknown liabilities; |
| ● | potential disruptions to NewGenIvf’s ongoing businesses; and |
| ● | unexpected costs and unknown risks and liabilities associated with strategic investments or acquisitions. |
Even if the transaction is consummated, NewGenIvf may only have limited control over the companies in which it only has minority stake, it cannot ensure that these companies will always comply with applicable laws and regulations in their business operations. Non-compliance of regulatory requirements by NewGenIvf’s investees may cause substantial harm to NewGenIvf’s reputations and the value of NewGenIvf’s investment. In addition, if the resulting business from such a transaction fails to meet NewGenIvf’s expectations, or it fails to successfully integrate such businesses into its own, its business, financial condition and results of operations may be adversely affected or it may be exposed to unknown risks or liabilities. If NewGenIvf is unable to effectively address these challenges, its ability to execute acquisitions as a component of its long-term strategy will be impaired, which could have an adverse effect on its growth. As a result of the above, NewGenIvf’s strategies may not be successfully implemented beyond the current markets.
Any investment might not achieve the synergies, operational or financial benefits it expects and may adversely impact NewGenIvf’s operating results. In addition, NewGenIvf cannot assure you that any future investment in or acquisition of new businesses or technology will lead to the successful development of new or enhanced products and services or that any new or enhanced products and services, if developed, will achieve market acceptance, or prove to be profitable.
Changes in NewGenIvf’s effective tax rate or tax liability may have an adverse effect on its results of operations.
NewGenIvf’s effective tax rate could increase due to several factors, including, but not limited to:
| ● | changes in the relative amounts of income before taxes in the various jurisdictions in which NewGenIvf operates that have differing statutory tax rates; |
| ● | changes in tax laws, tax treaties, and regulations or the interpretation of them; |
| ● | changes to its assessment about its ability to realize its deferred tax assets that are based on estimates of its future results, the prudence and feasibility of possible tax planning strategies, and the economic and political environments in which NewGenIvf does business; |
| ● | the outcome of future tax audits, examinations, or administrative appeals; and |
| ● | limitations or adverse findings regarding its ability to do business in some jurisdictions. |
Any of these developments could have an adverse effect on its results of operations.
NewGenIvf’s reported financial results may be adversely affected by changes in accounting principles generally accepted in the U.S.
A change in the U.S. accounting principles could have a significant effect on NewGenIvf’s reported results of operations and could affect the reporting of transactions already completed before the announcement of a change. The adoption of new or revised accounting principles may require it to make changes to its systems, processes and control, which could have a significant effect on its reported financial results, cause unexpected financial reporting fluctuations, retroactively affect previously reported results or require it to make costly changes to its operational processes and accounting systems upon or following the adoption of these standards.
NewGenIvf is subject to anti-corruption, anti-bribery, anti-money laundering, and similar laws, and non-compliance with such laws can subject it to criminal or civil liability and harm its business, financial condition and results of operations.
NewGenIvf is subject to the Anti-Money Laundering Act B.E. 2542 (1999) of Thailand, the Act Supplementing the Constitution Relating to the Prevention and Suppression of Corruption B.E. 2561 (2018) of Thailand, and the Penal Code of Thailand, domestic bribery laws, and other anticorruption and anti-money laundering laws in the countries in which it conducts activities. Anti-corruption and anti-bribery laws have been enforced aggressively in recent years and are interpreted broadly to generally prohibit companies, their employees and their third-party intermediaries from authorizing, offering, or providing, directly or indirectly, improper payments or benefits to recipients in the public or private sector. If NewGenIvf expands its business and sales and to the public sector, it may engage with business partners and third-party intermediaries to market its services and to obtain for it the necessary permits, licenses, and other regulatory approvals. In addition, NewGenIvf or its third-party intermediaries may have direct or indirect interactions with officials and employees of government agencies or state-owned or affiliated entities. NewGenIvf can be held liable for the corrupt or other illegal activities of these third-party intermediaries, its employees, representatives, contractors, partners and agents, even if it does not explicitly authorize such activities. Detecting, investigating, and resolving actual or alleged violations of anti-corruption laws can require a significant diversion of time, resources, and attention from senior management. In addition, noncompliance with anti-corruption, anti-bribery, or anti-money laundering laws could subject it to whistleblower complaints, investigations, prosecution, enforcement actions, sanctions, settlements, fines, damages, other civil or criminal penalties or injunctions, suspension or debarment from contracting with certain persons, reputational harm, adverse media coverage, and other collateral consequences. If any subpoenas or investigations are launched, or governmental or other sanctions are imposed, or if NewGenIvf does not prevail in any possible civil or criminal proceeding, its business, financial condition and results of operations could be harmed. In addition, responding to any action will likely result in a materially significant diversion of management’s attention and resources and significant defense costs and other professional fees, which could adversely affect its business, financial condition and results of operations.
NewGenIvf’s digital strategy is subject to financial, operational and regulatory risks on investing Solana.
Acquiring Solana (SOL) exposes the company to financial, operational, and regulatory risks, including extreme price volatility, potential technological failures (such as network outages or security exploits), uncertain and evolving global regulations that may classify SOL as a security or restrict its use, counterparty risks if held through exchanges or custodians, liquidity constraints during market stress, and the inherent challenges of securely managing digital assets, any of which could result in substantial or total loss of the investment.
NewGenIvf’s investment in residential market of United Arab Emirates is exposed to significant market risks.
Operating a residential property development business in the United Arab Emirates carries significant risks, primarily driven by market volatility and potential oversupply, particularly in the area of Ras Al Khaimah’s Beach District. Property values and rental yields are highly sensitive to fluctuations in oil prices, global economic conditions, and regional geopolitics, leading to sharp boom-bust cycles. Intense competition from new developments can rapidly saturate specific submarkets, driving down rents and occupancy rates, while regulatory changes (such as adjustments to rental increase caps, visa rules, or ownership laws) can unexpectedly impact profitability and operational models. Furthermore, reliance on expatriate tenants creates vulnerability to shifts in population demographics, employment trends, and government policies affecting residency, alongside inherent operational challenges including management costs, tenant defaults, and maintaining asset quality in a competitive environment.
NewGenIvf’s businesses and investment is exposed to economic and political risks of the recent US-Isreal-Iran conflict
The recent escalation of the US-Israel-Iran conflict poses a significant threat to the global economy, primarily through the disruption of oil supplies, with cascading effects on the real estate sector in the UAE and a broad economic downturn across Asia, including Hong Kong and China. As the conflict intensifies around the Strait of Hormuz—through which nearly 20% of the world’s oil passes—global oil prices have surged past $100 per barrel, raising fears of an extended energy crisis. This volatility directly undermines the UAE’s real estate market. Across Asia, the shock is equally severe, as most economies are net energy importers. The sustained high oil prices are fueling inflationary pressures, weakening currencies, and depressing stock markets, with Japan’s Nikkei and China’s Shanghai Composite both experiencing declines. As a result, the Company’s fertility business in Asia might also face downturn pressure due to the slowing down of the China and Hong Kong economy and the increasing costs of the company clinics in Thailand, Cambodia and Kyrgyzstan.
For more information about our SEC filings, please see “Where You Can Find More Information” and “Incorporation by Reference.”
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
Some of the statements made under “Prospectus Summary,” “Risk Factors,” “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” “Business” and elsewhere in this prospectus constitute forward-looking statements. In some cases, you can identify forward-looking statements by terminology such as “may,” “will,” “should,” “expects,” “plans,” “anticipates,” “believes,” “estimates,” “predicts,” “potential” “intends” or “continue,” or the negative of these terms or other comparable terminology.
These forward-looking statements may include, but are not limited to, statements relating to our objectives, plans and strategies, statements that contain projections of results of operations or of financial condition, expected capital needs and expenses, statements relating to the research, development, completion and use of our products, and all statements (other than statements of historical facts) that address activities, events or developments that we intend, expect, project, believe or anticipate will or may occur in the future.
Forward-looking statements are not guarantees of future performance and are subject to risks and uncertainties. We have based these forward-looking statements on assumptions and assessments made by our management in light of their experience and their perception of historical trends, current conditions, expected future developments and other factors they believe to be appropriate.
Important factors that could cause actual results, developments and business decisions to differ materially from those anticipated in these forward-looking statements include, among other things:
| ● | our planned level of revenues and capital expenditures; |
| ● | our ability to market and sell our products and services; |
| ● | our plans to continue to invest in research and development to develop technology for both existing and new products; |
| ● | our ability to maintain our relationships with suppliers, manufacturers and other partners; |
| ● | our ability to maintain or protect the validity of our intellectual property and know-how; |
| ● | our ability to retain key executive members; |
| ● | our ability to internally develop and protect new inventions and intellectual property; |
| ● | our ability to expose and educate the industry about the use of our services and products; |
| ● | our expectations regarding our tax classifications; and |
| ● | interpretations of current laws and the passages of future laws. |
These statements are only current predictions and are subject to known and unknown risks, uncertainties and other factors that may cause our or our industry’s actual results, levels of activity, performance or achievements to be materially different from those anticipated by the forward-looking statements. We discuss many of these risks in this prospectus in greater detail under the heading “Risk Factors” and elsewhere in this prospectus. You should not rely upon forward-looking statements as predictions of future events.
Although we believe that the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee future results, levels of activity, performance or achievements. Except as required by law, we are under no duty to update or revise any of the forward-looking statements, whether as a result of new information, future events or otherwise, after the date of this prospectus.
ITEM 4. INFORMATION ON THE COMPANY
| A. | History and Development of the Company |
Prior to the Business Combination, on April 29, 2021, A SPAC I Acquisition Corp. (“ASCA”), was incorporated as a British Virgin Islands business company, specifically a blank check company formed for the purpose of effecting a merger, share exchange, asset acquisition, share purchase, recapitalization, reorganization or similar business combination with one or more target businesses.
The Business Combination
On February 15, 2023, ASCA entered into the Merger Agreement (as amended on June 12, 2023 and December 6, 2023, the “Merger Agreement,” and the transactions contemplated thereunder, the “Business Combination”) with A SPAC I Mini Acquisition Corp., Merger Sub, NewGenIvf Limited, a Cayman Islands exempted company (“Legacy NewGenIvf”) and certain shareholders of Legacy NewGenIvf. Pursuant to the Merger Agreement, the Business Combination was effected in two steps: (i) ASCA was reincorporated to the British Virgin Islands by merging with and into A SPAC I Mini Acquisition Corp. (such transaction, the “Reincorporation Merger”); and (ii) Merger Sub merged with and into Legacy NewGenIvf, resulting in Legacy NewGenIvf being a wholly-owned subsidiary of the Company (such second step in isolation, the “Acquisition Merger”). The surviving entity of the Business Combination, together with its subsidiaries is referred to in this prospectus as “NewGenIvf,” the “Company,” “we,” “our,” or “us,” unless the context otherwise requires.
On June 12, 2023, the parties to the Merger Agreement entered into the First Amendment to Merger Agreement (the “First Amendment”), pursuant to which Legacy NewGenIvf agreed to provide non-interest bearing loans in an aggregate principal amount of up to $560,000 (the “Loan”) to ASCA to fund any amount that would be required in order to further extend the period of time available for ASCA to consummate a business combination and for ASCA’s working capital, payment of professional, administrative and operational fees and expenses, and other purposes as mutually agreed by ASCA and Legacy NewGenIvf. Such loans were to become repayable upon the closing of the Acquisition Merger. In addition, pursuant to the First Amendment, subject to receipt of at least $140,000 as part of the Loan from NewGenIvf, ASCA agreed to waive its termination rights and the right to receive any break-up fee due to Legacy NewGenIvf’s failure to deliver audited financial statements by no later than February 28, 2023.
On December 6, 2023, the parties to the Merger Agreement entered into the Second Amendment to the Merger Agreement (the “Second Amendment”) which amended and modified the Merger Agreement to, among other things, (i) reduce the size of NewGenIvf’s board of directors following the consummation of the Business Combination to five (5) directors, two (2) of whom would be executive directors designated by NewGenIvf and three (3) of whom will be designated by NewGenIvf to serve as independent directors in accordance with Nasdaq requirements, (ii) provide for the conversion of NewGenIvf shares issued by NewGenIvf following the original date of the Merger Agreement into Class A Ordinary Shares in connection with the Acquisition Merger, and (iii) remove the condition that ASCA have in excess of $5,000,000 in net tangible assets immediately after the consummation of the Business Combination.
On April 3, 2024, the Business Combination was consummated with the Company as the surviving entity.
| B. | Business Overview |
With a focus on providing fertility treatments to fulfil the dreams of building families, NewGenIvf mainly offers two services, namely: (i) in vitro fertilization (“IVF”) treatment service, comprising traditional IVF and egg donation; and (ii) fertility referral services. Currently, we have three clinics: one clinic in Thailand, one clinic in Cambodia, and one clinic in Kyrgyzstan.
| ● | IVF treatment service: For the period ended December 31, 2025 and the year ended December 31, 2024, we generated approximately 82.6% and 100% respectively of our revenue from IVF treatments. We primarily provide our clients with conventional IVF/intracytoplasmic sperm injection (“ICSI”) and embryo transfer services. As technology has progressively advanced, we have been able to, through technologies and facilities provided by MicroSort technology, help fulfill the family-balancing dreams of its clients and avoiding certain gender-related hereditary diseases. IVF treatment involves the performance of a series of medical treatment and procedures that are not separately distinct and only brings benefits to clients when embryo is successfully implanted, therefore revenue from IVF treatment is recognized at a point in time when it is completed in clinic. The completion of this treatment is evidenced by a written IVF report indicating successful embryo implantation. |
| ● | Fertility referral services: We also generate revenue from fertility referral services in Kyrgyzstan. For the period ended December 31, 2025 and the year ended December 31, 2024, we generated approximately 17.4% and Nil %, of our revenue from fertility referral services. For customers who were interested in surrogacy services in Kyrgyzstan, we referred the customers to our agents and the surrogacy mother services were provided by our agents independently while we received the referral commission from our agents. We believe that this arrangement enabled us to avoid the risk of surrogacy business while we could enjoy the referral income. |
For the years ended December 31, 2024 and 2025, NewGenIvf’s revenue was US$5,433,375 and US$4,726,433, and its net income/(loss) was US$(474,101) and US$8,763,979, respectively.
Competitive Strengths
NewGenIvf believes that the following competitive strengths have positioned it to meet growing opportunities in the fertility market across Asia-Pacific, and have differentiated it from its competitors:
Broad-range ARS Provider Offering Comprehensive Fertility Treatment Services
With almost a decade of experience in the fertility market, NewGenIvf has built a reputation in the IVF industry in Asia-Pacific. NewGenIvf has reinforced its long-standing position through expanding its service offerings and locations to address the evolving clients’ needs or requests.
NewGenIvf’s comprehensive fertility treatment offerings in Thailand, Cambodia, and Kyrgyzstan, primarily including IVF, egg donation (in Cambodia) and surrogacy referral services (in Kyrgyzstan), make it convenient for clients in Asia-Pacific market to have access to various fertility services but with a relatively low cost, as compared with the US market. Meanwhile, the average cost per IVF cycle by NewGenIvf is around US$7,000 (excluding medication). Each of NewGenIvf’s clinics in Thailand, Cambodia, and Kyrgyzstan has its own specialty, and together, NewGenIvf is able to provide more flexibility and options to its patients. For example, NewGenIvf’s Thailand clinic focuses on IVF and related ancillary services including HIV sperm washing, egg freezing, and chromosome screening. The clinic in Cambodia specializes in providing both IVF services and egg donation services. NewGenIvf refers clients to independent agents for surrogacy services which are legal in Kyrgyzstan.
NewGenIvf attributes its track record of success to its experienced physicians and its ability to provide comprehensive ARS services, allowing it to meet patients’ increasing demand for advanced, high-end, and sophisticated ARS, a higher standard and a wider range of advanced services.
NewGenIvf has extensive experience serving Asia-Pacific patients and a deep understanding of their general profiles. In particular, NewGenIvf has personnel speaking multiple languages, including nurses, facilitators, and translators, who are familiar with the health condition and culture of Asia-Pacific patients from different countries in the region. NewGenIvf believes that it is therefore well-positioned to benefit from market growth driven by Asia-Pacific patients travelling to its clinics for treatment.
Attractive Market with Significant Demand and Fast Growth
NewGenIvf operates in the ARS market in Asia Pacific, positioning it to leverage on an attractive market with compelling underlying growth potential.
Built on years of experience, NewGenIvf has established a strong reputation in its industry, which in turn attracted potential business partners to approach NewGenIvf to negotiate cooperations and referrals. Over the years, NewGenIvf sends representatives to medical expos mostly held in the PRC to approach potential business partners and establish new partnerships by entering into agency agreements with each agent. Agents typically market and promote NewGenIvf’s services by word-to-mouth referrals and other measures and NewGenIvf pays the agents commission upon the completion of client’s treatment. Normally, agents provide potential clients’ contact information to the sales team of NewGenIvf, who then approach potential clients and provide consultation on services. With its partnerships in various countries, NewGenIvf believes it is able to better benefit from the growing market opportunities.
Exclusively Owned and Licensed Technology for Family Planning and Access to Mature Fertility Technologies
NewGenIvf believes that its licenses and/or access to mature technologies contribute to its ability to identify and tailor ARS services to individual patient’s needs. These technologies include:
| ● | MicroSort Technology: NewGenIvf acquired the MicroSort Business from Genetics & IVF Institute, Inc. (“GIVF”) pursuant to a Purchase Agreement dated January 21, 2025 between NewGenIvf and GIVF (“Purchase Agreement”). As such NewGenIvf exclusively owns the MicroSort Technology. Prior to the closing of the MicroSort Acquisition, NewGenIvf held an exclusive license granted by a division of the Genetics & IVF Institute, Inc. to use MicroSort technology in Thailand and Cambodia. MicroSort technology is a form of pre-conception gender selection technology for humans. MicroSort technology aims to separate male sperm cells based on which gender chromosome they contain, which results in separated semen samples that contain a higher percentage of sperm cells that carry the same gender chromosome. The technology ultimately helps couples choose the gender of their future child by choosing semen samples that predominately contain sperm with the X chromosome for a female or Y chromosome for a male. Traditionally and naturally, gender selection occurs after conception, meaning after the eggs are fertilized. As a result, some fertilized eggs will go unused. However, with MicroSort technology, NewGenIvf is able to increase the ratio of male or female embryos, based on the patient’s preference. Eggs are more likely to be fertilized according to the preferences of the parents. Other improvements that MicroSort treatment could help achieve include prevention of certain gender-related hereditary diseases. |
| ● | Preimplantation Genetic Screening (“PGS”): PGS is used in parallel with an IVF treatment cycle. PGS is the practice of determining the presence of aneuploidy (either too many or too few chromosomes) in a developing embryo. PGS improves success rates of in vitro fertilization by ensuring the transfer of euploid embryos that have a higher chance of implantation and resulting in a live birth. PGS has improved clinical outcomes for NewGenIvf by achieving a higher implantation rate of 70.9% and reducing miscarriage rates by 26.6%. |
| ● | Next-Generation Sequencing (“NGS”): NGS is a high-throughput technology for determining the sequence of deoxyribonucleic acid (“DNA”) or ribonucleic acid (“RNA”) to study genetic variation associated with diseases or other biological phenomena. NGS determines the sequence of a sample all at once by using parallel sequencing. Traditional Sanger sequencing determines the sequence of a sample one section at a time. Sequencing thousands of gene fragments simultaneously with NGS reduces time and cost associated with sequencing and increases the coverage quality and data output. |
| ● | Preimplantation Genetic Diagnosis (“PGD”): Similar to PGS, PGD is also used in parallel with an IVF treatment cycle. But PGD is a process more enhanced than PGS since it scans for individual genes. PGD is the practice of evaluating embryos for specific genetic abnormalities, such as sickle cell disease or cystic fibrosis, where carrier status has been documented in each of the parents. By using this technique, physicians are able to check the genes or chromosomes for a specific genetic condition. PGD can decrease the risk of miscarriage and this technology can help women better achieve a healthy pregnancy. Individuals who suspect or know they carry genes for serious medical conditions may opt to screen for healthy embryos ahead of time. | |
| ● | Nodexus Technology: During 2025, NewGenIvf acquired patents and machines from Nodexus Inc, which is a biotechnology company revolutionizing live cell analysis and isolation through its advanced microfluidic technology. The company’s flagship NX One platform is an automated, benchtop system designed to make high-precision cell sorting more accessible and affordable for a wide range of laboratories, from academic institutions to biopharma firms. Unlike traditional, high-pressure sorters that can damage fragile cells, Nodexus utilizes a proprietary technology that operates at incredibly low pressures (under 1 psi), ensuring high cell viability during the sorting process. The system employs single-use, closed cartridges that maintain sterility, prevent cross-contamination, and support the sorting of diverse particle types, from small yeast cells and nuclei to large, delicate organoids up to 200 micrometers in size. This versatile and gentle approach has critical applications in cutting-edge research areas, including CRISPR gene editing, stem cell therapy development, cancer biology, and single-cell genomics. This technology can facilitate the gender selection as well as avoiding genetic diseases in the IVF treatment. |
Well Established Brand with Reliable Reputation
The founders of NewGenIvf entered the fertility market as agents in 2011 by introducing patients in need to a Thailand clinic for fertility treatments. The founders of NewGenIvf started to operate their own clinic in Thailand in 2014 and subsequently added clinics in Cambodia and Kyrgyzstan. Since then, NewGenIvf has attracted clients from countries throughout Asia-Pacific, including Mainland China, Hong Kong, India, Thailand, Australia and Taiwan.
NewGenIvf benefits from the favourable geographic locations of its clinics, especially its clinic in Thailand. Located in central Bangkok and situated in one of the biggest shopping malls of the city, the clinic is located in close proximity to various transportation facilities and popular tourist attractions, such as the Erawan Shrine. In this regard, NewGenIvf believes that its business has benefited from, and will continue to benefit from, the convenience of its locations.
NewGenIvf has developed a relatively replicable and scalable operating model that supports high productivity at its assisted reproductive medical facilities in Asia. Under this model, NewGenIvf’s medical facilities have established standardized operating procedures to select the treatment process according to each patient’s profile. NewGenIvf’s medical and operational personnel are organized into specialized teams according to the different stages of the treatment process and different patient profiles. When patients are initially admitted or would like to seek additional medical services later on, they are assigned to one of the optimal medical teams, which NewGenIvf believes is better suited after taking into account the patient’s diagnosis and preferences. NewGenIvf believes that this model allows each team to improve its efficiency and arrange suitable physicians for patients.
The physicians of NewGenIvf have also developed and employed an operating model that seeks to increase the effectiveness of physicians by utilizing standardized workflows and operating procedures with teams of supporting nurses and medical assistants. This helps to increase the number of IVF treatment cycles that physicians can perform while providing treatment customized based on patient conditions.
With its established client service history, accumulated experience as well as its continuous upgrades and development of treatment models, NewGenIvf believes that it will be able to better monetize its brands through its business.
Experienced Management Team
The NewGenIvf management team has considerable experience in the ARS market and the broader healthcare industry. A considerable number of NewGenIvf’s management are physicians or laboratory technicians who possess extensive experience in the ARS industry and are experts in their respective fields. NewGenIvf’s Chief Executive Officer, Mr. Alfred Siu, has more than 13 years of experience in the fertility service market. Dr. Wiphawee Luangtangvarodom had over 8 years of experience as an obstetrician and gynecologist. NewGenIvf’s two lab supervisors, Ms. Anussara Phinyong, and Ms. Araya Boonchaisitthipong, each had over eight years of experience in the embryologist field. These individuals have extensive experience in managing assisted reproductive medical facilities. NewGenIvf is also led by other members of the professional management team, who are intimately involved in the operational and financial management of NewGenIvf’s Group. Leveraging their experience, NewGenIvf believes that it is well positioned to expand its network and aims to become a leader in the Asia Pacific ARS market.
Strategies
NewGenIvf’s vision is to provide tailored ARS solutions to fulfil patients’ dreams of becoming a parent. To realize this vision, NewGenIvf plans to adopt the following strategies:
Offer Broad Fertility Services for Fertility Tourists across Asia Pacific
NewGenIvf intends to provide broad fertility services for fertility tourists seeking high quality, cost effective and comprehensive fertility solutions. According to CIC, the demand for fertility tourism is driven by a variety of factors including the prevalence of infertility, the introduction of the Three-Child policy in China, the improved understanding of assisted reproductive technology and increased affordability of ARS. To address these needs, NewGenIvf plans to offer its customers a “hassle-free”, seamless and integrated ARS and hospitality arrangement experience. To complement its fertility services, NewGenIvf intends to integrate its offerings with additional services for traveling patients, most of whom are first-time fertility tourists, such as translation service, hotel arrangement and airport pickup services. NewGenIvf plans to enhance its customers’ experience by entering into exclusive cooperation arrangements with local premium hospitality providers.
Continue to Invest in Laboratories and Facilities
NewGenIvf believes laboratories and treatment facilities are critical to supporting its future research, development and clients experience. NewGenIvf currently operates two laboratories that offer IVF services, one in Thailand and one in Cambodia, and plans to continue to scale up its existing laboratories. NewGenIvf plans to continue to invest in upgrading its laboratories and facilities to complement its growth and expansion, which it believes will help NewGenIvf maintain an edge over its competitors with regard to technology, operational efficiency, scalability, and client experience.
NewGenIvf intends to develop advanced facilities for its existing laboratories, which will be conducting research on ARS related basic science and experiments relating to emerging technologies to improve ARS success rates and lower costs. NewGenIvf also plans to correlate its data on patient treatment protocols to the embryo physiologic data and the pregnancy success rate-related data to identify better treatment protocols to increase ARS success rates. NewGenIvf intends to continue to actively promote technological cooperation with tertiary institutions to discover ways to improve its IVF success rates. Furthermore, NewGenIvf seeks to actively deploy the technology that it possesses to expand the services it provides.
NewGenIvf has accumulated experience in treating patients over 40 years old with premature ovarian failure and patients who have had recurrent ARS implementation failure, by, for the example, injecting platelet rich plasma into the ovaries to stimulate and support growth of the follicles. NewGenIvf is also implementing certain technological advancements relevant to the ARS industry, including microfluidics, automated sperm analysers, time lapsed incubators, non-invasive preimplantation genetic testing (“PGT”) of cell-free DNA in spent media, automated systems for oocyte/embryo vitrification to reduce reagent consumption and decrease labor intensity, mitochondria replacement therapy to reconstruct oocytes by nuclear transfer of polar body genome from an MII oocyte into an enucleated donor MII cytoplasm, to increase the number of oocytes available for the treatment of infertile women, preimplantation methylome screening. There are also breakthrough developments in science including organ culture systems, induced pluripotent stem cells, embryonic stem cells, spermatogonial stem cells for creation of functional gametes, but these techniques are not yet ready for human clinical trials.
NewGenIvf also intends to develop clinically customised interior design concepts for its medical facilities, including improved service rooms, consultation rooms, reception areas, nutrition food areas, and traditional Chinese medicine (such as acupuncture) facilities.
Increase Brand Awareness and Market Share
NewGenIvf intends to maintain and strengthen its brand awareness and market share in Asia Pacific. In order to expand its reach and increase patient numbers, NewGenIvf plans to collaborate with local hospitals, companies, premium hospitality providers and other key players in the ARS industry in Asia Pacific. Additionally, NewGenIvf intends to increase brand awareness through social media promotions and marketing initiatives, and establishing its business development team with the goal of attracting new patients and partners across Asia Pacific. Meanwhile, NewGenIvf intends to provide innovative treatment services to attract more clients. For example, NewGenIvf plans to introduce IVF mental health services, which allows clients who fail in IVF treatments to access online consultation for further treatment plans such as egg donation and surrogacy. These new treatments services aim to enable NewGenIvf to attract potential clients. By adopting a comprehensive strategy to expand its market share, NewGenIvf aims to strengthen its reputation as a long-standing ARS provider and capture additional market share of the growingly ARS market in Asia-Pacific.
Expand Service Reach Through Acquisitions and Partnerships
Leveraging its reputation and footprint in its current markets, NewGenIvf intends to expand its reach, services offering and client base through strategic acquisitions and/or partnerships in Asia Pacific. Acquisitions of or by companies offering similar services could not only allow NewGenIvf to diversify its client base, but also allow it to benefit from potential economies of scale and increasing efficiency through consolidation. NewGenIvf could also leverage the acquired or acquiring company’s customer base, reputation and expertise to further improve its offerings and operations. NewGenIvf intends to focus on ARS providers in Asia Pacific which possess all conventional licenses and locally recognized brands. For the global market beyond Asia Pacific, NewGenIvf intends to expand its footprint through partnerships with other IVF clinics.
In addition, NewGenIvf plans to explore expanding its client base by offering its fertility services as part of corporate benefit programs in Asia. NewGenIvf believes that there is potential in Asia in offering fertility treatments as a benefit for employees, particularly in companies with a large number of female employees of childbearing age. By partnering with corporate clients to provide fertility benefits, NewGenIvf can increase its market reach, enhance its brand reputation, and drive client growth. NewGenIvf’s broad range of fertility services, including IVF and egg freezing, can help corporate partners differentiate their employee benefits in the competitive employment landscape, which could make them more attractive to potential employees. Additionally, by offering these services, companies can help address the growing concern of delayed childbearing, which is becoming more common among women according to CIC. NewGenIvf plans to collaborate with potential corporate clients to develop customized fertility benefit programs that cater to their specific needs, and to provide comprehensive support and counselling throughout the process.
Meanwhile, NewGenIvf also intends to attract more clients by establishing its “home country gynecologist partnership program”. Under the program, NewGenIvf may, subject to its discretion and screening process, offer treatment services to clients with reduced time requirements to be spent overseas. Depending on local laws, the potential clients may be able to complete their treatments with gynecologists NewGenIvf partners with, in their home countries.
On February 28, 2025, NewGenIvf completed its acquisition of the MicroSort technology from Genetics & IVF Institute, Inc. (“GIVF”). Pursuant to a Purchase Agreement dated January 21, 2025 between NewGenIvf and GIVF (“Purchase Agreement”), NewGenIvf purchased all of the Assets (as defined in the Purchase Agreement) and IP Licenses (as defined in the Purchase Agreement) relating to the MicroSort technology (as described below from GIVF for a cash consideration of $750,000 and a share consideration of 125,000 Class A Ordinary Shares (equivalent to 42 Class A Ordinary Shares post Sixth Reverse Stock Split) (“MicroSort Acquisition”).
On July 24, 2025, NewGenIvf signed a Purchase Agreement to acquire certain tangible assets, company domain and six patents from Nodexus Inc. The technology associated with the patents is related to cell sorting to be performed via a device called NX One and could be applied in different fields including In Vitro Fertilization. The Patents would allow NewGenIvf to penetrate the IVF market in the United States by leasing the NX One to clinics that provide IVF treatments in the United States. The clinics would utilize the NX One to perform gender selection services and detect potential genetic diseases during the IVF process and charge a service fee from customers.
Business Model
With a focus on providing fertility treatments to fulfil couples and individuals’ dreams of raising children, NewGenIvf offers mainly two services, namely: (i) IVF treatment service, comprising traditional IVF and egg donation; and (ii) fertility referral services. The following table sets forth NewGenIvf’s revenue by service offerings and as a percentage of total revenue for the periods indicated:
| For the Year ended December 31, | ||||||||||||||||
| 2024 | 2025 | |||||||||||||||
| US$ | % | US$ | % | |||||||||||||
| IVF Treatment Service | 5,433,375 | 100.0 | 3,905,863 | 82.6 | ||||||||||||
| Fertility Referral Services | — | — | 820,570 | 17.4 | ||||||||||||
| Total Revenue | 5,433,375 | 100.0 | 4,726,433 | 100.0 | ||||||||||||
IVF Treatment Service
NewGenIvf primarily provides its clients with conventional IVF/ICSI and embryo transfer services. NewGenIvf is also able to, through MicroSort technology, help fulfill the family-balancing dreams of its clients and avoiding certain gender-related hereditary diseases.
IVF treatments that NewGenIvf provides address tubal factor, ovulatory dysfunction, diminished ovarian reserve, endometriosis, uterine factor, male factor, unexplained infertility and other causes. IVF bypasses the function of the fallopian tube by achieving fertilization within a laboratory environment. Ovarian hyper-stimulation is common with IVF treatments to recruit numerous follicles to increase the chances for success. Follicles are retrieved trans-vaginally using a vaginal probe and ultrasound guidance. Anaesthesia is frequently used due to the number of follicles retrieved and the resulting discomfort experienced by the patient. The eggs are identified in the follicular fluid and combined with sperm and culture medium in culture dishes, which are placed in an incubator with a temperature and gas environment designed to mimic the condition of the fallopian tubes. Once the embryos develop, typically over a 3-to-5-day period, they are transferred to the uterine cavity.
For the years ended December 31, 2024 and 2025, the revenue from NewGenIvf’s IVF treatments was US$5,433,375 and US$3,905,863, respectively, representing 100% and 82.6% of its total revenue in the corresponding periods.
IVF Treatments Process
A typical IVF treatment process mainly includes two stages, the pre-IVF treatment stage and the IVF treatment stage. During the IVF treatment process, NewGenIvf also provides support services such as nutrition guidance and psychological counselling. The flow chart below shows the stages involved in a typical IVF treatment process:

At the pre-IVF treatment stage, clients attend an initial consultation, undergo pre-IVF tests, and undergo treatment for gynaecological and andrological diseases, if needed. At the initial consultation, a physician reviews the clients’ detailed medical history to collect more information relating to the potential cause of their infertility. The client then undergoes various pre-IVF tests, which may include, among other things, blood pressure, hormone level, ultrasound, infectious disease screening, uterine evaluation and male fertility test. The physician will then design treatment plans based on the client’s medical history and results of the tests. If the client is satisfied with treatment plan and the test results are acceptable to the physician, the physician will prescribe medications and start stimulation treatment.
The first step of the cycle is to boost egg production through injecting synthetic hormones. Over about one week of ovarian stimulation, clients are monitored on a regular basis with blood test and transvaginal ultrasound. If follicles have reached at least 10 mm in size, an additional antagonist drug will be added into the daily injection schedule. This is used to prevent ovulation before ovum pickup time. After another few days of ovarian simulation, if follicle growth is consistent and majority of follicles are around 16 mm to 17 mm, the final injection of a human chorionic gonadotropin will be administered. The trigger injection is the final step of the stimulation process and is for the maturation of the eggs in the follicles before they are collected. The next major step is to retrieve the eggs with a minor surgical procedure called Trans Vaginal Follicle Aspiration conducted under anaesthesia. At the same time the male partner collects the sperms for fertilizing the eggs in the laboratory by a process known as intracytoplasmic sperm injection. The fertilized embryos are cultured in the laboratory for two to six days. Embryos that grow well are biopsied and tested by PGT to detect potential genetic diseases.
The final step is to transfer the embryos into the uterus using a catheter. Within eight days after the embryo transfer, a blood test can be conducted to detect whether the implantation was successful.
MicroSort Technology
MicroSort technology is a preconception process developed by the Genetics and IVF Institute, Inc. that aims to improve the chances that the baby to be conceived will be of the desired gender and prevents certain gender-related hereditary diseases.
Semen samples usually contain equal amounts of sperm carrying the Y chromosome (which will produce a boy), and sperm carrying the X chromosome (which will produce a girl). During the MicroSort process, the sperm sample is washed to remove seminal liquid and nonmotile cells. After the washing, the sample is stained with a special fluorescent material that attaches to the DNA contained in the sperm. The stained sperm cells are analyzed one by one by a flow cytometer, in which cells pass through a laser to make the stain attach to the DNA fluoresce. The sperm containing the X chromosome (which have more DNA and therefore more stain) will shine brighter than the sperm containing the Y chromosome. The flow cytometer uses a special software to identify X and Y chromosome sperm based on their fluorescence signature. The sperm carrying the chromosome that will produce the desired gender are separated from the rest of the sample -resulting in an enriched sperm sample ready for use.
NewGenIvf held an exclusive license granted by a division of GIVF, MicroSort International, to use the MicroSort technology in Thailand and Cambodia. MicroSort’s licenses for NewGenIvf’s operation in Thailand and Cambodia were each provided under a lease and service agreement. In April 2019, First Fertility PGS entered into a Lease and Services Agreement with MicroSort International to use MicroSort equipment in Thailand and in March 2019, Phnom Penh Center entered into a Lease and Services Agreement with MicroSort International to use MicroSort equipment in Cambodia (together, the “Lease and Services Agreements”). Pursuant to the Lease and Services Agreements, First Fertility PGS and Phnom Penh Center each had the exclusive right to utilize the MicroSort equipment and to market and sell MicroSort sperm sorting services in Thailand and Cambodia, respectively. MicroSort International was responsible for the maintenance of MicroSort equipment and technical and engineering support. The term of each Lease and Service Agreements was initially from 2019 to 2024, which shall be automatically renewed for one year unless a written notice of at least 180 days prior to the intended termination date is provided. The consideration under each of the Lease and Services Agreements was US$9,000 per month after six months. On February 28, 2025, NewGenIvf completed its acquisition of the MicroSort technology from Genetics & IVF Institute, Inc. (“GIVF”).
The flow chart below shows the process involved in MicroSort:

Nodexus Technology
During 2025, NewGenIvf acquired patents and machines from Nodexus Inc, which is a biotechnology company revolutionizing live cell analysis and isolation through its advanced microfluidic technology. The company’s flagship NX One platform is an automated, benchtop system designed to make high-precision cell sorting more accessible and affordable for a wide range of laboratories, from academic institutions to biopharma firms. Unlike traditional, high-pressure sorters that can damage fragile cells, Nodexus utilizes a proprietary technology that operates at incredibly low pressures (under 1 psi), ensuring high cell viability during the sorting process. The system employs single-use, closed cartridges that maintain sterility, prevent cross-contamination, and support the sorting of diverse particle types, from small yeast cells and nuclei to large, delicate organoids up to 200 micrometers in size. This versatile and gentle approach has critical applications in cutting-edge research areas, including CRISPR gene editing, stem cell therapy development, cancer biology, and single-cell genomics. This technology can facilitate the gender selection as well as avoiding genetic diseases in the IVF treatment.
Preimplantation Genetic Screening
PGS is used in parallel with an IVF treatment cycle. PGS is the practice of determining the presence of aneuploidy (either too many or too few chromosomes) in a developing embryo. PGS improves success rates of in vitro fertilization by ensuring the transfer of euploid embryos that have a higher chance of implantation and resulting in a live birth. PGS has improved clinical outcomes for NewGenIvf by achieving a higher implantation rate of 70.9% and reducing miscarriage rates by 26.6%.
Next-Generation Sequencing
NGS is a high-throughput technology for determining the sequence of deoxyribonucleic acid DNA or RNA to study genetic variation associated with diseases or other biological phenomena. NGS determines the sequence of a sample all at once by using parallel sequencing. Traditional Sanger sequencing determines the sequence of a sample one section at a time. Sequencing thousands of gene fragments simultaneously with NGS reduces time and cost associated with sequencing and increases the coverage quality and data output.
Preimplantation Genetic Diagnosis
Similar to PGS, PGD is also used in parallel with an IVF treatment cycle. But PGD is a more enhanced process than PGS since it scans for individual genes. PGD is the practice of evaluating embryos for specific genetic abnormalities, such as sickle cell disease or cystic fibrosis, where carrier status has been documented in each of the parents. By using this technique, physicians are able to check the genes or chromosomes for a specific genetic condition. PGD can decrease the risk of miscarriage and this technology can help women achieve a healthy pregnancy. Individuals who suspect or know they carry genes for serious medical conditions may opt to screen for healthy embryos ahead of time.
Egg Freezing Service
NewGenIvf also provides egg freezing services which is a fertility preservation service that allows individuals to freeze and store their eggs for future use. The process begins with hormonal stimulation to encourage multiple egg production, followed by a minor surgical procedure to retrieve the eggs under sedation. The eggs are then rapidly frozen using vitrification, a technique that prevents ice crystal formation, and stored in liquid nitrogen at ultra-low temperatures. This service is ideal for those delaying childbearing due to career, medical reasons (like cancer treatment), or personal choice. When ready to conceive, the eggs can be thawed, fertilized with sperm, and transferred as embryos. While success rates vary by age and clinic, egg freezing offers a proactive option for preserving fertility.
Fertility Referral Services
NewGenIvf also generated revenue from fertility referral services in Kyrgyzstan. For the period ended December 31, 2025 and the year ended December 31, 2024, we generated approximately 17.4% and Nil %, of our revenue from fertility referral services. For customers who were interested in surrogacy services in Kyrgyzstan, we referred the customers to our agents and the surrogacy mother services were provided by our agents independently while we received the referral commission from our agents. We believe that this arrangement enabled us to avoid the risk of surrogacy business while we could enjoy the referral income.
Network of Facilities
As of December 31, 2025, NewGenIvf had one accounting and general administrative support office located in Hong Kong and four clinics located in Thailand, in Cambodia, and in Kyrgyzstan, respectively. In the United States, we have not had office yet but acquired MicroSort Lab Services LLC from Genetic IVF Institute during 2025. The integration of the medical facilities in Thailand help NewGenIvf provide a more seamless one-stop experience to its clients. Set out below is an illustration of the locations of NewGenIvf’s clinics and accounting/general administrative office:
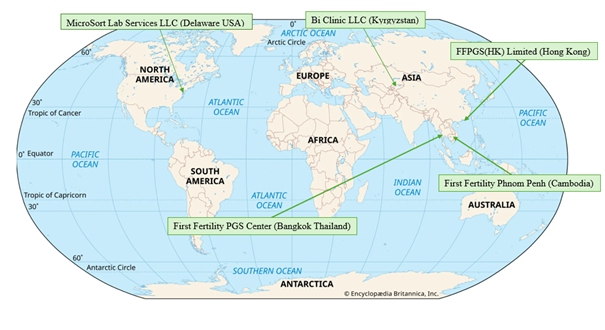
The following table sets forth the approximate aggregate average gross floor area (“G.F.A.”) of each of NewGenIvf’s clinics that were under lease and actively used for client service as of December 31, 2025:
| As of December 31, 2025 |
||||
| (Square Feet) | ||||
| Thailand | ||||
| First Fertility PGS Center Co., Ltd. – PS Tower (“First Fertility PGS Center”) |
14,750 | |||
| First Fertility PGS Center Co., Ltd – Erawan Hotel (“First Fertility PGS Center”) |
2,615 | |||
| Cambodia | ||||
| First Fertility Phnom Penh Center (“Phnom Penh Center”) |
18,567 | |||
| Kyrgyzstan | ||||
| Bi Clinic Limited Liability Company (“Bi Clinic”) |
2,164 | |||
| Aggregate G.F.A | 38,096 | |||
Currently, IVF treatments are performed in its Thailand and Cambodia clinics, egg donation services are provided in its Cambodia clinic, and fertility referral services are provided in its Kyrgyzstan clinic. The following table summarises the services available at NewGenIvf’s clinics:
| IVF Treatments |
Fertility Referral Services |
|||
| Thailand | ||||
| First Fertility PGS Center | √ | × | ||
| Cambodia | ||||
| Phnom Penh Center | √ | × | ||
| Kyrgyzstan | ||||
| Bi Clinic | √ | √ |
| √ | — | Yes |
| × | — | No |
The following table sets forth a breakdown of revenue from services performed at NewGenIvf’s medical centers for the periods indicated:
| For the Year ended December 31, | ||||||||||||||||
| 2024 | 2025 | |||||||||||||||
| US$ | % | US$ | % | |||||||||||||
| HK SAR | - | - | - | - | ||||||||||||
| Thailand | 2,175,253 | 40.0 | 1,691,862 | 43.3 | ||||||||||||
| Cambodia | 601,526 | 11.1 | 676,743 | 17.4 | ||||||||||||
| Kyrgyzstan | 2,656,596 | 48.9 | 1,491,778 | 38.2 | ||||||||||||
| Other third party clinics | - | - | 45,480 | 1.1 | ||||||||||||
| Total Revenue | 5,433,375 | 100.0 | 3,905,863 | 100.0 | ||||||||||||
Thailand Clinic
As of December 31, 2025, NewGenIvf had two clinics in Thailand. At the clinic in Thailand, NewGenIvf offers its clients customized fertility treatment solutions including IVF/ICSI, embryo culture, hormonal blood tests, infectious diseases tests, chromosome screening by PGT, hysteroscopy, sperm analysis, sorting, washing and freezing, and egg freezing. Its medical and operational personnel are organized into specialized teams according to the different stages of the IVF treatment process and different patient profiles. When clients are admitted, they are assigned to a team which NewGenIvf believes is better suited the clients after taking into account the clients’ diagnosis and preferences. Furthermore, NewGenIvf also provides related value-added services such as nutrition guidance, psychological counselling, acupuncture, and translation interpreters to supplement the IVF treatment. NewGenIvf prides itself on providing quality and customized treatment to its clients on a day-to-day basis.
As of December 31, 2025, the clinic in Thailand had 8 nurses, 8 full time lab physicians and embryologists, 22 administrative staff, totaling 38 staff members.
Cambodia Clinic
NewGenIvf has one clinic, Phnom Penh Center, in Cambodia. Phnom Penh Center is staffed with 3 Cambodian physician, 2 embryologists, 8 nurses and 12 other staff, and offers similar IVF treatments as in Thailand and egg donation services. Phnom Penh Center operates under a license issued by Cambodia MOH for the Cambodian physician, who has entered into an agreement with Phnom Penh Center for the exclusive use of such license.
After more than ten years of development since its opening in 2015, Phnom Penh Center has become one of the long-standing ARS providers in Cambodia. Phnom Penh Center’s IVF philosophy concentrates on three key points in the treatment process: the mother’s wellbeing, the technology used to assist mothers deliver a strong and healthy baby and the medical science used to ensure every chance of success for women in various age spectrums.
Clinic in Kyrgyzstan
Through Bi Clinic, NewGenIvf also provide fertility referral services by referring customers who are interested in surrogacy services which are legal in Kyrgyzstan.
Although Physician at Bi Clinic has expertise in sourcing surrogate mothers, techniques of embryo transfers, prenatal care, baby delivery, and postnatal care, NewGenIvf refers customers to independent agents for fertility service like surrogacy so as to reduce the operation risks in Kyrgyzstan.
As of December 31, 2025, Bi Clinic had 1 full-time physician, 1 embryologist, 1 nurse, and 7 other staff.
Professionals
Licensed Physicians
As of December 31, 2025, NewGenIvf employed five licensed physicians, among which one was based in Cambodia, one was based in Kyrgyzstan and the other three were based in Thailand. Most of NewGenIvf’s physicians had over 10 years of experience or above. The following table summarizes the number and types of such licensed physicians as of December 31, 2025.
| Country | Licensed physician | Licenses and Approvals |
Effective Period | Issuing Authority |
||||
| Cambodia | Mr. Keut Serey | Decision on permission for beauty treatment operation | December 14, 2022 – December 14, 2026 | The Ministry of Health of Cambodia | ||||
| Thailand | Dr Keatthisak Boonsimma | Number 31801 Medical Practitioner License | April 1, 2005 – Indefinite | Royal Thai College of Obstetricians and Gynaecologists of Thailand | ||||
| Number 22624/2554 OB-Gyn License |
July 1, 2014 – Indefinite | Medical Council of Thailand | ||||||
| Number 40962/2563 Reproductive Medicine Diploma | July 1, 2020 – Indefinite | Medical Council of Thailand | ||||||
| Thailand | Dr. Anurach Kulvanitchaiyanunt | Practice License No. 11755 | February 1 2025 – Indefinite | Medical Council of Thailand | ||||
| Thailand | Dr Wiphawee Luangtangvarodom | Number 38347/2562 OB-Gyn License |
August 1, 2019 – Indefinite | Medical Council of Thailand | ||||
| Number 43217/2564 Reproductive Medicine License | July 1, 2021 – Indefinite | Medical Council of Thailand | ||||||
| Number 48510 Medical Practitioner License | April 1, 2014 – Indefinite | Medical Council of Thailand | ||||||
| Kyrgyzstan | Dr Myrzalymbekova A.B. | Number & numero; CO170001836 Medical Certificate | June 23. 2017 - Indefinite | Medical Council of Kyrgyzstan | ||||
| Number №0002953 Medical Practitioner | 25 April 2018 - Indefinite | Medical Council of Kyrgyzstan | ||||||
| Number №11373\2565 Medical Certificate | 16 August 2024 - Indefinite | Medical Council of Kyrgyzstan | ||||||
| Number №0003447 Medical Certificate | 12 April 2023 - Indefinite | Medical Council of Kyrgyzstan |
Customers
For the years ended December 31, 2024 and 2025, the majority of NewGenIvf’s clients were from China (including mainland China and Hong Kong). NewGenIvf enters into a verbal contract with each of its customers that outline, among other things, the scope of services, service fees, payment terms and rights, responsibilities and obligations of each party. Consent is obtained from the patients prior to the provision of the various treatments. Customers are not entitled to enjoy the relevant services until outstanding amounts have been settled pursuant to the relevant contract. Sales to individual consumers did not vary significantly and none of the customers contribute more than 10% of NewGenIvf’s revenue for the years ended December 31, 2024 and 2025.
In addition to significant customers using NewGenIvf’s IVF treatment services and ancillary caring services, NewGenIvf also has customers who utilize the access to the freezing and storage facility and other relatively insignificant services, such as check-ups services, blood test services and other minor services (the latter category of customers are referred to as “consultation customers”). NewGenIvf also refers customers to other IVF and/or Surrogacy agents who will provide fertility referral income for NewGenIvf.
Sales and Marketing
For the years ended December 31, 2024 and 2025, NewGenIvf promoted brand awareness through its sales teams and, in many cases, through cooperating with third-party agencies and partners.
NewGenIvf’s sales teams have broad experience in fertility services and are responsible for identifying potential clients and managing the overall sales process. NewGenIvf’s sales team primarily relies on social media marketing, word-of-mouth referrals, recognition of its brand, printed advertisements and marketing events. NewGenIvf spends marketing expenses on placing advertisements through popular social media platforms, maintaining the official website of NewGenIvf and sending information through its official accounts on social media platforms.
Supply and Procurement
NewGenIvf’s procurement is mainly for medications, laboratory media and reagents, laboratory consumables, and blood test reagents. As of December 31, 2024 and 2025, there was no single supplier individually contributed more than 10% of the Group’s trade payable respectively. For both the years ended December 31, 2024 and 2025, no vendor contributed more than 10% of total direct cost of the Group. NewGenIvf’s procurement team is experienced in selecting cost-effective supplies as well as selecting reliable suppliers. NewGenIvf’s major suppliers are pharmaceutical companies.
Competition
NewGenIvf believes that it is a long-standing provider of ARS in Asia Pacific that competes primarily based on the following competitive factors:
| ● | the value and comprehensiveness of the solutions; |
| ● | treatment that is effective and achieves desired outcomes; |
| ● | clients’ experience, including dedicated patient education, clinical guidance and emotional support; and |
| ● | access to a network of high-quality fertility specialists. |
NewGenIvf competes primarily with other regional fertility service providers. While NewGenIvf does not believe any single competitor offers a comparably robust and integrated fertility solution package as NewGenIvf in the regions that it operates, NewGenIvf’s competitors may compete in a variety of ways, including by providing better services, having established local connections, fulfilling the evolving client’s needs, as well as conducting brand promotions and other marketing activities.
As NewGenIvf may introduce new ancillary services and other companies may introduce similar fertility services as NewGenIvf’s, NewGenIvf may become subject to additional competition.
Facilities
As of December 31, 2025, in addition to its clinics, NewGenIvf leased one property in Hong Kong with an aggregate square footage of approximately 8,000 for its accounting and office administration support offices. NewGenIvf also operates its medical facilities as described above in “— Network of Facilities” above. NewGenIvf believes that its existing facilities are suitable and adequate to meet its current needs.
C. Organizational Structure
The following is a list of our principal subsidiaries and consolidated affiliated entities as of the date of this Report:
| Name | Place of Formation | Relationship | ||
| NewGenIvf Limited | Cayman Islands | Wholly-owned subsidiary | ||
| Incorporated on January 16, 2019 | ||||
| MicroSort Lab Services LLC | U.S.A. | Wholly-owned subsidiary | ||
| Acquired on February 28, 2025 | ||||
| NewGenDigital Limited | British Virgin Islands | Wholly-owned subsidiary | ||
| Incorporated on June 16, 2025 | ||||
| FFPGS (HK) Ltd | Hong Kong | Indirect subsidiary, wholly owned by NewGenIvf Limited | ||
| Incorporated on December 19, 2019 | ||||
| NewGenProperty Limited | British Virgin Islands | Wholly-owned subsidiary | ||
| Incorporated on June 16, 2025 | ||||
| NewGenBiz Limited | British Virgin Islands | Wholly-owned subsidiary | ||
| Incorporated on January 13, 2026 | ||||
| NewGenOman Limited | British Virgin Islands | Wholly-owned subsidiary | ||
| Incorporated on February 2, 2026 | ||||
| Alfred Siu Charitable Foundation Limited | Hong Kong | Wholly-owned subsidiary | ||
| Incorporated on February 27, 2026 |
| Bi Clinic LLC | Kyrgyzstan | Indirect subsidiary, wholly owned by NewGenIvf Limited | ||
| Acquired on December 17, 2024 | ||||
| First Fertility PGS Center Limited | Thailand | Indirect subsidiary, 74% owned by Well Image Limited HK directly and indirectly | ||
| Incorporated on March 6, 2014 | ||||
| First Fertility Phnom Penh Ltd | Kingdom of Cambodia | Indirect subsidiary, wholly owned by NewGenIvf Limited | ||
| Incorporated on August 10, 2015 | ||||
| Med Holdings Limited | Thailand | Indirect subsidiary, 48.99% owned by Well Image Limited HK | ||
| Incorporated on January 21, 2015 | ||||
| Well Image Limited HK | Hong Kong | Indirect subsidiary, wholly owned by NewGenIvf Limited | ||
| Incorporated on July 11, 2008 | ||||
| HyFi Innovations Limited | Hong Kong | Indirect subsidiary, wholly owned by NewGenDigital Limited | ||
| Incorporated on October 20, 2025 |
The following subsidiaries were disposed of:
| 深圳前海豐泰仁匯健康科技有限公司 (Shenzhen Qianhai Fengtai Renhui Health Technology Co., Ltd) Disposed of on July 29, 2025 |
China | Indirect subsidiary, wholly owned by FFPGS (HK) Ltd |
D. Property, Plants and Equipment
The Company leases the premises for its principal executive office located at 36/39-36/40, 13th Floor, PS Tower, Sukhumvit 21 Road (Asoke) Khlong Toei Nuea Sub-district, Watthana District, Bangkok 10110, Thailand. The Company leases one property in Hong Kong with an aggregate square footage of approximately 8,000 for its administration support offices.
The Company also leases several premises to operate its clinics in various countries. The Company operates the Bi Clinic Limited Liability Company starting from December 17, 2024 in Kyrgyzstan, which premises have an aggregate area of 2,164 square feet. In Cambodia, the Company operates the First Fertility Phnom Penh Center, which premises have an aggregate area of 18,567 square feet. In Thailand, the Company operates a clinic named First Fertility PGS Center Co., Ltd., which premises have an aggregate area of 14,750 square feet.
The Company also leases premises located in Thailand for its Erawan Consultation Clinic clinic, with an aggregate area of approximately 2,615 square feet. This property has been used as the Company’s second clinic in Thailand since early 2025 but will stop operating in March 2026 as the landlord has decided to lease it to other parties.
ITEM 4A. UNRESOLVED STAFF COMMENTS
Not applicable.
ITEM 5. OPERATING AND FINANCIAL REVIEW AND PROSPECTS
The following discussion of our financial condition and results of operations is based upon, and should be read in conjunction with, our audited consolidated financial statements and the related notes included in this Report. This Report contains forward-looking statements. See “Forward-Looking Information.” In evaluating our business, you should carefully consider the information provided under the caption “Item 3 Key Information — D. Risk Factors” in this Report. We caution you that our businesses and financial performance are subject to substantial risks, changes and uncertainties.
Unless the context otherwise requires, all references in this Item 5 to “NewGenIvf” refers to NewGenIvf Group Limited and its subsidiaries pursuant to a Business Combination which came to effect on April 3, 2024.
We incorporated by reference the discussion of the operating results comparing the years ended December 31, 2025 and 2024.
Overview
NewGenIvf is an assisted reproductive services (“ARS”) provider in Asia Pacific. Since the establishment of its first clinic in Thailand in 2014, it has established itself as a long-standing ARS provider in the region. NewGenIvf’s mission is to assist couples and individuals across Asia Pacific, regardless of fertility challenges that they may face, to fulfil their dreams of building families and to increase their access to fertility treatments. Its strategic presence in Thailand, Cambodia, and Kyrgyzstan positions the company to take advantage of opportunities across Asia Pacific.
NewGenIvf is still in the early stage of materializing its long-term objective of building a comprehensive, sophisticated and high-end ARS platform for its clients and providing personalized solutions based on NewGenIvf’s brands and client-generated services. NewGenIvf plans to offer full fertility services for fertility tourists across Asia Pacific, continue to invest in laboratories and facilities updates, increase its brand awareness and market share, as well as expand service reach through acquisitions and partnerships, which NewGenIvf believes will help expand its client base and enhance expertise attraction, and in turn strengthen NewGenIvf’s monetization capabilities.
Key Factors Affecting NewGenIvf’s Results of Operations
NewGenIvf’s results of operation are principally affected by the following factors:
Regulatory environment
The ARS market in Asia-Pacific region is highly regulated. The implementation and enforcement of laws, regulations and government policies in Thailand, Cambodia, Kyrgyzstan and other applicable jurisdictions significantly impact the design, pricing and sale of fertility services and cost of compliance for clinics across Asia Pacific. Medical facilities providing fertility services generally must be filed and registered with the relevant supervision institutions and such filing and registration must be renewed periodically. Any change in laws, regulations or policies in relation to such filing or registration could affect NewGenIvf’s ability and plans to launch new services and renew registration for existing services. The regulatory framework for medical facilities and services, especially those involving ARS, is, and will continue, evolving. Any changes in the applicable regulatory frameworks in the jurisdictions where NewGenIvf operates may materially affect its financial condition and results of operations.
Growth and competitive landscape of Asia Pacific’s ARS market
NewGenIvf’s revenue has historically been primarily derived from clients in Asia Pacific. As such, NewGenIvf’s financial performance and future growth depend primarily on the demand for ARS, as well as changes in its competitive landscape, in Asia Pacific. Population growth, infertility rates, and demand for facility treatments in the region will ultimately determine the demand for NewGenIvf’s services. Infertility is increasingly becoming prevalent globally, primarily driven by increasing average age of first birth, as well as various lifestyle and environmental factors. Driven by an increased infertility rate and growing demand for children without birth defects, resulting from improving living standards and improved awareness about birth defects and prevention, the global ARS market is expected to continue to grow. Furthermore, a growing number of governments around the world has granted legal recognition to same-sex marriages, which brings more desires for having children to form a complete family. Because of the fertility rate and recent government incentive policies, such as the Three-child Policy of China in 2021, the ARS market increased significantly in Asia Pacific. Leveraging its status as a long-standing ARS provider in Asia Pacific, NewGenIvf expects to continue to be well positioned to capture the expected growth in the demand for ARS in the area.
To date, NewGenIvf owns and uses MicroSort technology in Thailand and Cambodia, which is a form of pre-conception gender selection technology for humans. While NewGenIvf expects to benefit from first-mover advantages for this technology in the two regions, market entry by potential competitors or faster-than-expected development of potential competitors may affect its market position and demand for its services and cause downward pricing pressure on its treatments, which may in turn materially and adversely affect its results of operations. Meanwhile, ARS market could also be affected by the macroeconomic environment and geopolitical events. Uncertainty in the macroeconomic environment and the outbreak of US and Israel war with Iran in March 2026, resulting from a range of events and trends, including the rise in global inflation and interest rates, supply chain disruptions, geopolitical pressures, including the unknown impact of current and future trade regulations, changes in Asian-Pacific relations, fluctuation in foreign exchange rates, and associated global economic conditions may result in volatility in ARS market and NewGenIvf’s operating performance. For example, NewGenIvf derives a substantial portion of its revenue from Chinese clients and as such, its failure to maintain PRC-sourced revenues and access to new and existing clients from the PRC could materially and adversely affect its results of operations and competitive position. However, the near-term growth prospects of the PRC economy are unclear due to the uncertain effects of ongoing economic stress caused by trade and national security policies, and the elevated levels of private and public indebtedness, among others. A prolonged downturn or slow-growth in the PRC economy generally could materially and adversely affect NewGenIvf’s results of operations and there is a significant likelihood that NewGenIvf’s actual results over the time periods and under the scenarios covered by the projections would be different.
Fluctuation of costs
NewGenIvf’s costs primarily include clinic costs, cost of goods sold, selling and marketing expenses and general and administrative expenses, details of which are set out below.
| ● | Clinic costs. NewGenIvf’s clinic costs primarily consisted of sub-contracting charges, office supplies and staff salaries and bonus, most of which are recognized during the provision of IVF services. Its clinic costs represented approximately 54.8% and 66.6% of its revenue for the years ended December 31, 2024 and 2025, respectively. As NewGenIvf gradually expands the scale of its operation and presence in Asia Pacific, its clinic costs is expected to increase in the foreseeable future, which will affect its profitability. |
| ● | Cost of goods sold. NewGenIvf’s cost of goods sold primarily consisted of purchase and direct cost for IVF treatment services and surrogacy and ancillary caring services, most of which are recognized during the provision of IVF treatment services. Its cost of goods sold represented approximately 11.6% and 13.2% of the revenue for the years ended December 31, 2024 and 2025, respectively. NewGenIvf expects its cost of goods sold to increase in the foreseeable future as it gradually grows its revenues and expand its sales network. |
| ● | Selling and marketing expenses. NewGenIvf’s selling and marketing expenses primarily consisted of social media expense. Its selling and marketing expenses represented approximately 3.8% and 25.2% of its revenue for the years ended December 31, 2024 and 2025, respectively. NewGenIvf expects its selling and marketing expenses to increase as it plans to expand its sales and scale its operation in Asia-Pacific. |
| ● | General and administrative expenses. NewGenIvf’s general and administrative expenses primarily consisted of write-off of agency deposit, legal and professional fees, depreciation in operating lease right-of-use (“ROU”) assets, staff salaries and director fees. Its general and administrative expenses represented approximately 51.2% and 222.01% of its revenue for the years ended December 31, 2024 and 2025, respectively. NewGenIvf expects its general and administrative expenses to increase in line with its expansion plan. |
NewGenIvf expects its cost structure to evolve as it develops and expands its business. As NewGenIvf continues to develop new services and technologies, NewGenIvf expects to incur additional costs in relation to its raw materials procurement, production and sales and marketing, among other things. Moreover, to support NewGenIvf’s business growth, it expects to increase its headcount, particularly for its lab and nurse team, and incur higher staff costs as a result.
Ability to maintain trust of clients and reputation in the industry
The success of NewGenIvf’s business will depend to a large extent on its ability to gain broad acceptance of its services from clients. Reputation is crucial in keeping existing clients and attracting new clients. NewGenIvf’s reputation depends on a number of factors, including for example the success, effectiveness, quality and pricing of its services, service offerings of its competitors, the effectiveness of its marketing efforts to drive awareness and the demand for fertility services, which eventually will affect its ability to maintain clients and attract new clients. Therefore, NewGenIvf’s success will depend to a large extent on its ability to maintain its reputation in the industry and its clients’ trust, which would affect the number of its clients and treatment cycles that will in turn affect its revenues.
NewGenIvf believes that the medical facilities in its network are increasingly recognized among clients, for their service quality, technological expertise and patient experience. NewGenIvf also hopes to keep its clients by providing discounts in treatment services and via the “success guarantee” program for egg donation services in Cambodia and surrogacy services in Kyrgyzstan, which provides treatments to clients until a success is achieved.
Based on its increasingly recognized reputation, NewGenIvf believes that there is substantial opportunity to continue to grow its revenue through attracting new clients. NewGenIvf’s addressable market is couples who want to have children, egg freezing patients, LGBT groups and couples with genetic abnormalities, particularly those in Asia Pacific. NewGenIvf believes that its current client base represents a small percentage of its total market opportunity. NewGenIvf intends to attract new clients by, among other things, making significant investments in sales and marketing to engage, educate and drive awareness of the unmet need of fertility treatment among its potential clients and by its customer-reference discounts mechanism. Additionally, NewGenIvf believes that its expanding presence has resulted in a heightened awareness of the need to offer fertility services and the value it provides to its clients, which it believes will help facilitate its growth. In addition, NewGenIvf is continuously utilizing its established client relationships to evaluate other potential services that could benefit its clients and simultaneously drive its growth.
International traveling conditions
The revenue from international clients is a critical component of NewGenIvf’s revenue. International traveling to Thailand, Cambodia and Kyrgyzstan may be affected by a number of factors, including local and global political, economic and cultural conditions. In Thailand, the political landscape has recently consolidated under a conservative government following February 2026 elections, with Prime Minister Anutin Charnvirakul’s Bhumjaithai Party securing victory by emphasizing nationalism and border security amid ongoing tensions with Cambodia. These border disputes have prompted multiple governments, including Austria and the UK, to issue advisories warning against travel to the Thai-Cambodia border region and parts of southern Thailand. In Cambodia, authorities strongly assert the country remains “safe, secure, and stable” for tourists, with major destinations like Phnom Penh and Siem Reap operating normally despite diplomatic tensions with Thailand. However, the government has made defending territorial integrity its “highest priority” as it seeks international attention regarding the border dispute. In Kyrgyzstan, travelers face a different set of considerations, including recent security agency restructuring and detentions that signal political consolidation at the highest levels. The Chinese embassy has advised visitors to exercise heightened vigilance, respect local laws, avoid border areas prone to natural hazards, and be aware of potential scams, reflecting ongoing regional stability concerns. The recent war involving the US, Israel, and Iran has severely disrupted international travel, beginning with the immediate closure of airspace over several Middle Eastern countries and the suspension of flights through critical hubs like Dubai and Doha, which has stranded hundreds of thousands of passengers. This has forced airlines to cancel thousands of flights or take longer, more expensive routes, leading to a surge in ticket prices and the imposition of new fuel surcharges that are affecting travelers globally. Governments have responded by issuing widespread “do not travel” warnings for the region and organizing evacuations, while embassies have halted routine visa services, further complicating international mobility. The compounded effect is a climate of uncertainty where even trips not destined for the Middle East face potential delays, higher costs, and complex rebooking challenges. Together, these factors create a dynamic travel environment where regional tensions, domestic politics, and economic conditions intersect to shape the risk landscape for international visitors. These conditions may cause NewGenIvf difficulty in attracting clients from the PRC to travel to Thailand, Cambodia and Kyrgyzstan for NewGenIvf’s services, which could materially and adversely affect NewGenIvf’s operations and financial results.
Given the uncertainty of the local and global conditions and the countries’ future policy regarding international traveling, all of which are beyond NewGenIvf’s control, NewGenIvf’s results of operation may be materially and adversely affected by any changes in international travelling conditions.
Key Components of Results of Operations
NewGenIvf’s revenues were derived from two types of services: IVF treatment services and fertility referral services.
Revenue
The following table sets forth a breakdown of NewGenIvf’s revenue by the types of services, in absolute amounts and as percentages of total revenue, for the periods indicated.
| For the Year ended December 31, | ||||||||||||||||
| 2024 | 2025 | |||||||||||||||
| US$ | % | US$ | % | |||||||||||||
| IVF treatment services(1) | 5,433,375 | 100.0 | 3,905,863 | 82.6 | ||||||||||||
| Fertility referral services | - | - | 820,570 | 17.4 | ||||||||||||
| Total revenues | 5,433,375 | 100.0 | 4,726,433 | 100.0 | ||||||||||||
| (1) | Include an insignificant amount of revenue derived from consultation customers who used NewGenIvf’s non-IVF treatment and insignificant services, such as check-ups services, blood test services and other minor services. |
NewGenIvf generated revenue from facilities located in various geographic regions. The following table sets forth a breakdown of NewGenIvf’s revenue based on the locations where the revenue originated, in absolute amounts and as percentages of total revenue, for the periods indicated.
| For the Year ended December 31, | ||||||||||||||||
| 2024 | 2025 | |||||||||||||||
| US$ | % | US$ | % | |||||||||||||
| China/HK SAR | - | - | 41,555 | 0.9 | ||||||||||||
| Kyrgyzstan | 2,656,596 | 48.9 | 2,312,348 | 48.9 | ||||||||||||
| U.S.A. | - | - | 45,480 | 1.0 | ||||||||||||
| Cambodia | 601,526 | 11.1 | 635,188 | 13.4 | ||||||||||||
| Thailand | 2,175,253 | 40.0 | 1,691,862 | 35.8 | ||||||||||||
| Total revenues | 5,433,375 | 100.0 | 4,726,433 | 100.00 | ||||||||||||
NewGenIvf’s revenue results are affected by, among others, changes in sales price and the fluctuation of foreign currency rates with US dollars. Based on the breakdown of the revenue contribution in terms of currencies used by customers for 2025, a 10% change in foreign currency rates with US dollars would cause approximately 9.99% change in NewGenIvf’s revenue.
IVF treatment services
NewGenIvf generated revenue from IVF treatment services provided at facilities that NewGenIvf operated in Thailand and Cambodia. In addition, NewGenIvf’s revenue from IVF treatment service amounted to US$5,433,375 and US$3,905,863, representing approximately 100% and 82.6% of its total revenues in 2024 and 2025, respectively.
IVF treatment involves the performance of a series of medical treatment as well as procedures and eventually brings benefits to clients when embryo is successfully implanted. Revenue from IVF treatment is recognized at a point in time when different treatment and/or procedure completed in clinic. The completion of the respective treatments and/or procedures are evidenced by treatment cards and reports maintained in the patient files indicating successful completion of respective promised obligations.
Fertility Referral services
NewGenIvf also generated revenue of US$820,570 from fertility referral services, approximately 17.4% of its total revenue in 2025. Fertility referral service revenue is recognized at a point in time when the customers are referred to our agents and commission is received from the agents.
Cost of revenue
The following table sets forth a breakdown of NewGenIvf’s cost of revenue by the nature of the cost, in absolute amounts and as percentages of total cost of revenues, for the periods indicated.
| For the Year ended December 31, | ||||||||||||||||
| 2024 | 2025 | |||||||||||||||
| US$ | % | US$ | % | |||||||||||||
| Cost of revenues | ||||||||||||||||
| Cost of goods sold | 629,620 | 17.5 | 622,386 | 16.5 | ||||||||||||
| Clinic costs | 2,976,861 | 82.5 | 3,148,201 | 83.5 | ||||||||||||
| Total cost of revenues | 3,606,481 | 100.0 | 3,770,587 | 100.0 | ||||||||||||
Cost of goods sold. Cost of goods sold primarily consisted of purchase and direct cost for IVF treatment services and ancillary caring services. NewGenIvf’s cost of goods was mostly recognized during the provision of IVF treatment services.
Clinic costs. Clinic costs primarily consisted of sub-contracting charges, office supplies and staff salaries and bonus of NewGenIvf’s clinics. Sub-contracting charges represented fees paid to agents who supervised and managed the whole IVF services for customers who utilized third party independent clinics, recruited surrogate mothers and assisted in the documentation, consulting and medical treatment arrangement throughout treatment procedure of surrogacy service.
Gross profit and gross margin
The following table sets forth NewGenIvf’s gross profit in absolute amounts and its gross margin as percentages of total revenues, for the periods indicated.
| For the Year ended December 31, | ||||||||||||||||
| 2024 | 2025 | |||||||||||||||
| US$ | % | US$ | % | |||||||||||||
| Gross profit | 1,826,894 | 33.6 | 955,846 | 20.2 | ||||||||||||
| Revenues | 5,433,375 | — | 4,726,433 | — | ||||||||||||
NewGenIvf expects that gross profit and gross margin will continue to be affected by various factors including the geographic locations where treatments are performed, as well as the pricing with its clients, agent subcontracting charges and the costs of the supplies provided by major pharmaceutical companies, all of which are negotiated separately.
Operating expenses
NewGenIvf’s operating expenses consist of selling and marketing expenses and general and administrative expenses. NewGenIvf’s selling and marketing expenses are primarily social media expenses. NewGenIvf’s general and administrative expenses mainly include depreciation in operating lease ROU assets, consultant and legal fees, embedded derivative fair value expense, write off of certain receivables and staff salaries.
Other income (expense)
NewGenIvf’s other income consists primarily of bargain purchase gain on acquisition of the MicroSort and Nodexus businesses, offset partially by the losses arising from investment in digital assets and financial assets.
Interest expense
NewGenIvf’s interest expense is incurred mainly in relation to its interest-bearing convertible bonds.
Taxation
British Virgin Islands
NewGenIvf is incorporated in the British Virgin Islands and is not subject to tax on income or capital gains under current British Virgin Islands law. In addition, upon payment of dividends to shareholders, no British Virgin Islands withholding tax will be imposed.
Hong Kong
Under the two-tiered profits tax rates regime, Hong Kong tax residents are subject to Hong Kong profits tax in respect of profits arising in or derived from Hong Kong at 8.25% for the first HK$2 million of profits of the qualifying group entity, and profits above HK$2 million will be taxed at 16.5%. The profits of group entities not qualifying for the two-tiered profits tax rates regime will continue to be taxed at a flat rate of 16.5%.
Accordingly, the Hong Kong profits tax is calculated at 8.25% on the first HK$2 million of the estimated assessable profits and at 16.5% on the remaining estimated assessable profits.
Thailand
The companies incorporated in Thailand are taxed on worldwide income. A company incorporated outside of Thailand is taxed on its profits arising from or in consequence of the business carried on in Thailand. The Thailand corporate income tax rate for locally incorporated company is 20%. A foreign company not carrying on business in Thailand is subject to a final withholding tax on certain types of assessable income (e.g., interest, dividends, royalties, rentals, and service fees) paid from or in Thailand. The rate of withholding tax is generally 15%, except for dividends, which is 10%, while other rates may apply under the provisions of a double tax treaty.
Cambodia
The standard rate of corporate income tax for companies and permanent establishments in Cambodia who are classified as medium and large taxpayers is 20%. For companies and permanent establishments who are classified as small taxpayers, the corporate income tax rates are progressive rates from 0% to 20%. In view of the annual turnover of the company, which ranges from KHR1 billion to KHR6 billion for service and commercial sectors, the company is considered a medium-sized company.
Kyrgyzstan
NewGenIvf is subject to a corporate income tax on its aggregate annual income earned worldwide. Non-resident legal entities carrying out business activities through a permanent establishment in Kyrgyzstan are subject to profit tax on the income attributed to the activities of that permanent establishments. Profit tax for locally incorporated company is calculated at a rate of 10% of aggregate annual income less allowed deductions.
U.S.A.
NewGenIvf is subject to a corporate income tax on its aggregate annual income earned worldwide from its U.S. operations at a flat 21% on net income. In addition to federal obligations, there is a graduated state income tax rate that applies to corporate income, with a top rate of 6.6% on income over $60,000. Moreover, when the Company’s US operation pays a dividend to its offshore parent company, the payment is generally subject to a US federal withholding tax, typically at a statutory rate of 30% on the gross amount.
Results of Operations
| For the Year ended December 31, |
||||||||
| 2024 | 2025 | |||||||
| US$ | ||||||||
| Revenues | 5,433,375 | 4,726,433 | ||||||
| Cost of revenues | (3,606,481 | ) | (3,770,587 | ) | ||||
| Gross profit | 1,826,894 | 955,846 | ||||||
| Operating expenses | ||||||||
| Selling and marketing expenses | (206,314 | ) | (1,188,711 | ) | ||||
| General and administrative expenses | (2,781,075 | ) | (10,493,596 | ) | ||||
| Total operating expenses | (2,987,389 | ) | (11,682,306 | ) | ||||
| Operating (loss) income | (1,160,495 | ) | (10,726,461 | ) | ||||
| Other income (expenses), net | ||||||||
| Other income (expense), net | 971,391 | (8,099 | ) | |||||
| Bargain purchase gain | - | 21,653,835 | ||||||
| Loss on digital assets, net | - | (332,480 | ) | |||||
| Loss on financial assets, net | - | (700,631 | ) | |||||
| Gain on embedded derivative | - | 456,731 | ||||||
| Interest income | 6,953 | 2,251 | ||||||
| Finance costs | (778,656 | ) | (616,423 | ) | ||||
| Total other income (expenses), net | 199,688 | 20,455,184 | ||||||
| (Loss) Income before taxes | (960,807 | ) | 9,728,723 | |||||
| Tax income (expense) | 486,706 | - | ||||||
| Net (loss) income | (474,101 | ) | 9,728,723 | |||||
| Less: net income attributable to non-controlling interests | 50,542 | (150,685 | ) | |||||
| Net (loss) income attributable to the shareholders of the Company | (524,643 | ) | 9,879,408 | |||||
| Other comprehensive income (loss) | ||||||||
| Foreign currency translation adjustment | 32,529 | (222,944 | ) | |||||
| Total comprehensive (loss) income | (441,572 | ) | 9,505,779 | |||||
| Less: Total comprehensive income (loss) attributable to non-controlling interests | 56,908 | (211,683 | ) | |||||
| Total comprehensive (loss) income attributable to the shareholders of the Company | (498,480 | ) | 9,717,462 | |||||
| Earnings per share – basic | (4,408.76 | ) | 214.73 | |||||
| – diluted | (4,408.76 | ) | 3.04 | |||||
| Weighted average shares outstanding * – basic | 119 | 46,009 | ||||||
| - diluted | 119 | 3,250,020 | ||||||
| * | Subsequent to 2024 year end, the Company carried out a 1-for-20 reverse stock split, a 1-for-10 reverse stock split, a 1-for-5 reverse stock split, a 1-for-5 reverse stock split, a 1-for-3 reverse stock split and a 1-for-4 reverse stock split of its issued and unissued shares which was effected on February 11, 2025, May 5, 2025, August 4, 2025, December 1, 2025, January 26, 2026 and March 16, 2026 respectively. The shares above have been adjusted retrospectively for these reverse stock splits. |
Year Ended December 31, 2024 Compared with Year Ended December 31, 2025
Revenue
NewGenIvf’s revenue decreased by approximately 13.01% from US$5,433,375 in 2024 to US$4,726,433 in 2025.
IVF treatment services
NewGenIvf’s IVF treatment service revenue decreased by approximately 28.11% from US$5,433,375 in 2024 to US$3,905,863 in 2025. This decrease was primarily the result of change of management in Thailand clinic and new clinic set-up in Kyrgyzstan focusing on IVF services in late 2024.
Fertility Referral services
In 2025, NewGenIvf’s derived fertility referral services of US$820,570 revenue from referring customers to outside agents for IVF and surrogacy services while no such revenue was recorded in 2024.
Cost of revenue
NewGenIvf’s cost of revenue increased by approximately 4.55% from US$3,606,481 in 2024 to US$3,770,587 in 2025.
Cost of goods sold
NewGenIvf’s cost of goods sold decreased by approximately 1.15% from US$629,620 in 2024 to US$622,386 in 2025, primarily attributed to the decrease on procurement costs.
Clinic costs
NewGenIvf’s clinic costs increased by approximately 5.76% from US$2,976,861 in 2024 to US$3,148,201 in 2025, primarily due to clinic staff turnover in Thailand.
Gross profit
NewGenIvf’s gross profit decreased by approximately 47.68% from US$1,826,894 in 2024 to US$955,846 in 2025, primarily attributable to a reorganizing of our cooperation model with subcontractors and the decreased efficiency of our marketing services, resulting in a increase in unit service costs per customer, directly leading to decreases in gross profit margins.
NewGenIvf’s gross margin decreased from 33.6% in 2024 to 20.2% in 2025. This is primarily due to the drop of sales revenue while the overhead costs of clinic was not reduced accordingly.
Operating expenses
NewGenIvf’s operating expenses increased by approximately 291.05% from US$2,987,389 in 2024 to US$11,682,307 in 2025, primarily attributable to (i) professional fees of US$1,861,572 incurred in 2025 being related to real estate development project in United Arab of Emirates, valuation fees on acquisition of Nodexus (ii) staff and director salary and personnel cost of US$2,910,173 in 2025 due to enlargement of sales/marketing force and administrative staff to implement our business expansion pan and bonus related to maintaining listing status in Nasdaq and increasing share capitalization; and (iii) write-off of certain agency deposit and artwork of US$1,105,370.
Other income
NewGenIvf’s other income increased from US$971,391 in 2024 to US$21,069,356 in 2025, primarily attributable to a non-cash, bargain purchase gain recognized on the acquisition of Nodexus and Microsort businesses of $21,653,835, gain on embedded derivative of $456,731, loss on investment in digital assets, net of $332,480 and loss on investment on financial assets, net of $700,631.
Finance Costs
NewGenIvf’s finance costs decreased by approximately 20.84% from US$778,656 in 2024 to US$616,423 in 2025 as a result of securing a lower interest rate on convertible notes issued during the year and some convertible notes being issued second half of 2025 for financing new business initiatives including real estate development project in United Arab of Emirates, supplement business and tokenization project.
Tax income (expense)
NewGenIvf made no provision for income taxes in 2024 and 2025. There was no assessable income generated from Hong Kong, Thailand and Cambodia.
During the year ended December 31, 2024 excess provision of US$486,706 was reversed as they are no longer deemed to be payable.
Net income
NewGenIvf generated a net income of US$9,728,723 in 2025 while there was a net loss of US$(474,101) in 2024.
Liquidity and Capital Resources
Cash flows and working capital
NewGenIvf’s principal sources of liquidity have been cash flows generated from its business operations and external financing via various instruments. As of December 31, 2024 and 2025, NewGenIvf had US$457,740 and US$758,621, respectively, in cash and cash equivalents. NewGenIvf had working capital (defined as total current assets deducted by total current liabilities) of a surplus of $452,391 and US$5,017,221 respectively, as of December 31, 2024 and 2025.
As of December 31, 2025, NewGenIvf owed US$6,421 to related parties. Nevertheless, NewGenIvf is able to generate sufficient cash flow from its business operations and financing activities to operate and grow its business.
NewGenIvf continually seeks to monetize from positive cash flow contracts and increase revenue from its operating activities. NewGenIvf monitors its current and expected liquidity requirements to help ensure that it maintains sufficient cash balances to meet its existing and reasonably likely long-term liquidity needs.
NewGenIvf intends to finance its future working capital requirements and capital expenditures from cash generated from operating activities, in addition to funds raised from financing activities. NewGenIvf may, however, require additional cash due to changing business conditions or other future developments, including any investments or acquisitions it may decide to pursue. If its existing cash is insufficient to meet its requirements, NewGenIvf may seek to issue debt or equity securities or obtain additional credit facilities. Financing may be unavailable in the amounts NewGenIvf needs or on terms acceptable to it, if at all. Issuance of additional equity securities, including convertible debt securities, would dilute NewGenIvf’s earnings per share. The incurrence of debt would divert cash for working capital and capital expenditures to service debt obligations and could result in operating and financial covenants that restrict NewGenIvf’s operations and its ability to pay dividends to its shareholders. If NewGenIvf is unable to obtain additional equity or debt financing as required, its business operations and prospects may suffer. Please see “Risk Factors — Risks Relating to NewGenIvf’s Business and Industry — NewGenIvf requires a significant amount of capital to fund its operations and growth. If NewGenIvf cannot obtain sufficient capital on acceptable terms, its business, financial condition, and prospects may be materially and adversely affected.”
The following table presents NewGenIvf’s selected consolidated cash flow data for the periods indicated.
| For the Year ended December 31, |
||||||||
| 2024 | 2025 | |||||||
| US$ | ||||||||
| Net cash used in operating activities | (8,264,074 | ) | (10,926,056 | ) | ||||
| Net cash used in investing activities | (53,045 | ) | (7,559,774 | ) | ||||
| Net cash provided by financing activities | 8,675,790 | 18,992,758 | ||||||
| Net increase in cash and cash equivalents | 358,671 | 506,928 | ||||||
| Effect of foreign currency translation on cash and cash equivalents | 44,965 | (206,047 | ) | |||||
| Cash and cash equivalents, beginning of year | 54,104 | 457,740 | ||||||
| Cash and cash equivalents, end of year | 457,740 | 758,621 | ||||||
Operating activities
Net cash used in operating activities was US$10,926,056 for the year ended December 31, 2025. The difference between NewGenIvf’s net profit of US$9,728,723 for the year ended December 31, 2025 and the net cash used in operating activities was primarily attributable to bargain purchase gain of US$21.7m on acquisition of Nodexus and Microsort businesses.
Net cash used in operating activities was US$8,264,074 for the year ended December 31, 2024. The difference between NewGenIvf’s net loss of US$474,101 for the year ended December 31, 2024 and the net cash used in operating activities was primarily attributable to (i) an increase of agent’s receivable of US$1,191,795, (ii) a cash deposit of US$1,000,000 with a digital trading platform and settlement of charges in relation to the de-spac and business reorganisation.
Investing activities
Net cash used in investing activities in 2025 was US$7,559,774, primarily representing reservation money of US$4.6m paid for the real estate development project in United Arab of Emirates, as well as cash paid for investment in digital assets of $1,000,000, amounts held in custody for investments of $760,000 and cash paid for acquisition of businesses in 2025 of $1,050,000.
Net cash used in investing activities in 2024 was US$53,045, primarily representing purchase of plant and equipment.
Financing activities
Net cash provided by financing activities in 2025 was US$18,992,758, primarily representing issuance of share capital of US$12.6m utilizing equity line of credit, convertible note issuance of US$6.8m and settlement of $500,000 for a promissory note.
Net cash provided by financing activities in 2024 was US$8,675,790, primarily representing issuance of share capital utilizing equity line of credit, convertible note issuance and proceeds from issuance of promissory notes all totaling to an amount of $8,583,597.
Contractual Obligations
The following table sets forth NewGenIvf’s main contractual obligations and commitments as of December 31, 2024 and December 31, 2025.
| December 31, | ||||||||
| 2024 | 2025 | |||||||
| Operating lease | US$ | |||||||
| - Payable less than 1 years | 111,321 | 262,244 | ||||||
| - Payable 1 – 2 years | 10,300 | 184,041 | ||||||
| - Payable 2.- 5 years | — | 118,800 | ||||||
| Total | 121,621 | 565,085 | ||||||
| Convertible bonds- | - | |||||||
| 2025 | 516,250 | - | ||||||
| 2026 | 457,250 | 664,000 | ||||||
| 2027 | 457,250 | - | ||||||
| 2028 | 507,250 | 3,150,000 | ||||||
| 2029 | 3,269,625 | - | ||||||
| 2030 | - | 1,475,000 | ||||||
| Total | 5,207,625 | 5,289,000 | ||||||
The convertible bonds have the option of being converted to Class A ordinary shares at the discretion of the holder, any time after issuance. The conversion, however, is subject to certain terms and conditions, including the requirement of the shareholdings not to exceed 4.99 % or 9.99% of the Company’s shares based on the noteholder. As of December 31, 2025, this criterion has been met.
Off-Balance Sheet Commitments and Arrangements
NewGenIvf has not entered into any financial guarantees or other commitments to guarantee the payment obligations of any third parties, nor any derivative contracts that are indexed to its shares and classified as shareholder’s equity or that are not reflected in its consolidated financial statements. Furthermore, NewGenIvf does not have any retained or contingent interest in assets transferred to an unconsolidated entity that serves as credit, liquidity or market risk support to such entity. NewGenIvf does not have any variable interest in any unconsolidated entity that provides financing, liquidity, market risk or credit support to it or engages in leasing, hedging or product development services with it.
Dividend Policy
NewGenIvf Group Limited is a holding company with no material operations of its own. NewGenIvf Group Limited conducts all of its operations through its subsidiaries. As a result, NewGenIvf Group Limited’s ability to pay dividends depends upon dividends paid by its subsidiaries. If our subsidiaries or any newly formed subsidiaries incur debt on their own behalf in the future, the instruments governing their debt may restrict their ability to pay dividends to the Company.
NewGenIvf Group Limited is permitted under BVI law to provide funding to its subsidiaries in Hong Kong, Thailand, Cambodia and Kyrgyzstan through loans or capital contributions without restrictions on the amount of the funds.
In addition, the Company’s subsidiaries are currently permitted to pay dividends to the Company in accordance with relevant laws and regulations. Payment of dividends requirements in a company incorporated under the laws of Thailand is governed by the Civil and Commercial Code of Thailand. For example, the company may not declare dividends if the company has incurred losses, the company must appropriate to a reserved fund at each dividend contribution of dividend of at least one-twentieth of the profits until the fund reaches one-tenth of the capital, or the dividends payment must be made to the shareholders within one (1) month from the dividend declaration date. On the capital remittance or payment of dividends to the shareholders from outside of Thailand, it is regulated by the regulations issued by the Bank of Thailand, including the Exchange Control Act B.E. 2485 (1942). The fund remittance from Thailand to a foreign jurisdiction may require an approval from the Bank of Thailand or require notifying the Bank of Thailand for such transfer, depending on the types of the remittance transactions, through the commercial bank in the country. For a company incorporated under the laws of Kyrgyzstan, under Kyrgyz regulations of dividends (net profit), the dividends can be paid once a year depending on the results of the financial year of the company.
Critical Accounting Estimates
NewGenIvf prepares its financial statements in conformity with U.S. GAAP, which requires NewGenIvf to make judgments, estimates and assumptions. NewGenIvf continually evaluates these estimates and assumptions based on the most recently available information, its historical experience and various other assumptions that NewGenIvf’s management believes to be reasonable under the circumstances. Since the use of estimates is an integral component of the financial reporting process, actual results could differ from its expectations as a result of changes in NewGenIvf’s estimates.
The selection of critical accounting estimates and other uncertainties affecting application of those policies and the sensitivity of reported results to changes in conditions and assumptions are factors that should be considered when reviewing NewGenIvf’s financial statements. NewGenIvf’s management believes the following accounting estimates involve the most significant estimates used in the preparation of their financial statements.
Business combination and gain on bargain purchase
The Company adopted ASC 805, the accounting for business combinations which requires the application of the acquisition method. This method mandates that the acquiring entity recognize all assets acquired and liabilities assumed at their acquisition-date fair values. Gain on a business acquisition is recognized in earnings only in the specific circumstance of a “bargain purchase.” This occurs when the fair value of the identifiable net assets acquired exceeds the total consideration transferred (including the fair value of any non-controlling interest). It requires the acquirer to first meticulously reassess all measurements—including the identification and valuation of assets, liabilities, and the consideration—to ensure the gain is not a measurement error. Once confirmed, the resulting bargain purchase gain is recognized in profit or loss on the acquisition date. This treatment reflects the economic benefit obtained by the acquirer from purchasing net assets for less than their aggregate fair value. Under US GAAP, the distinction between acquiring a business and acquiring a group of assets is determined by a defined screen test. A transaction is accounted for as a business combination only if the acquired set meets the specific definition of a business, which requires an integrated set of activities with, at a minimum, an input and a substantive process that together significantly contribute to the ability to create outputs.
Intangible Assets
Intangible assets acquired from third parties are measured initially at fair value and those with an infinite life are not amortized. The Company annually evaluates the recoverability of the infinite-lived intangible assets for possible impairment whenever events or circumstances indicate that the carrying amount of such assets may not be recoverable. To test indefinite-lived intangible assets for impairment, the Company first performs a qualitative assessment to determine if it is more likely than not that the carrying amount of each of its indefinite-lived intangible assets exceeds its fair value. If it is, a quantitative assessment is required. Alternatively, the Company may bypass the qualitative assessment and perform a quantitative impairment test. The qualitative assessment requires the consideration of factors such as recent market transactions, macroeconomic conditions and changes in projected future cash flows. The quantitative assessment compares the fair value of an indefinite-lived intangible asset to its carrying amount. If the carrying amount of an indefinite-lived intangible asset exceeds its fair value, an impairment loss is recognized for the excess. Fair values of indefinite-lived intangible assets are determined based on discounted cash flows or appraised values, as appropriate. If such a review indicates that the carrying amount of intangible assets is not recoverable, the carrying amount of such assets is reduced to fair value.
As of December 31, 2025, and 2024, the Company did not record an impairment on the intangible assets.
Convertible Instruments
Convertible Instruments are categorized as equity or debt based on the terms of the notes. Convertible Notes are recorded at amounts equal to the proceeds of the issuance, including the embedded conversion feature, and net of discounts and unamortized debt issuance in accordance with ASC 480-10-55-44 on the consolidated balance sheets. An evaluation of all conversion, purchase and redemption features contained in a debt instrument is performed to determine if there are any embedded features that require bifurcation as a derivative. The conversion feature is recorded separately as a derivative liability at its fair value, calculated using the Black-Scholes model.
Debt issuance and offering costs are amortized over the contractual term of the Convertible Notes, to the consolidated statements of operations in accordance with ASC 835-30-45-1A.
The convertible notes are subsequently recorded at amortized cost, with interest expense recognized using the effective interest method. The derivative liability, if any is remeasured at fair value at each reporting date and any gain or loss on fair value is recognized in the statement of comprehensive income.
Recent accounting pronouncements
The FASB has introduced expanded income tax disclosure requirements under ASU 2023-09 to improve transparency. Companies will now need to provide a detailed reconciliation of their effective tax rate, breaking down federal, state, and foreign taxes, as well as specific categories like tax credits and foreign earnings. Additionally, businesses must disclose income taxes paid by jurisdiction, offering investors greater clarity on tax obligations. These changes apply to both public and private companies, with annual reporting periods beginning after December 15, 2024 (2025 for calendar-year entities). This update aims to reduce ambiguity in tax reporting and align disclosures with investor needs.
A major shift in digital asset accounting, ASU 2023-08 requires companies to measure certain crypto assets (e.g., Bitcoin, Ethereum) at fair value rather than applying the previous impairment-only model. This means entities must recognize quarterly fair value adjustments in their financial statements, increasing volatility in reported earnings but improving transparency. The standard applies to fiscal years beginning after December 15, 2024, and impacts both corporate treasuries and investment firms holding cryptocurrencies. This change aligns GAAP closer to fair value accounting seen in other investment holdings, addressing criticisms of the old impairment approach.
In November 2024, the FASB issued ASU 2024-03, *Income Statement—Reporting Comprehensive Income—Expense Disaggregation Disclosures (Subtopic 220-40): Disaggregation of Income Statement Expenses*, which requires public entities to disclose, in a tabular format in the notes to the financial statements, specific information about certain costs included in common expense captions. This standard mandates the disaggregation of natural expenses—such as employee compensation, depreciation, and inventory costs—from broader captions like “cost of services” or “selling, general and administrative expenses.” Although this ASU is effective for annual reporting periods beginning after December 15, 2026, and interim periods thereafter, with early adoption permitted, the Company is currently evaluating the potential impact of this new guidance on its financial statement disclosures.
In July 2025, the FASB issued ASU 2025-05, Financial Instruments—Credit Losses (Topic 326): Measurement of Credit Losses for Accounts Receivable and Contract Assets, which introduces a practical expedient to simplify the application of the current expected credit loss (CECL) model for current accounts receivable and contract assets arising from ASC 606 revenue transactions. Such practical expedient permits entities to assume that current economic conditions as of the balance sheet date will remain unchanged for the remaining life of these short-term assets, eliminating the need to develop and document forward-looking macroeconomic forecasts. For healthcare entities with significant patient accounts receivable, third-party payor contract assets, or government reimbursement receivables, this simplification reduces compliance costs and complexity while continuing to require consideration of historical loss experience and current conditions such as customer-specific financial distress or changes in credit policies. The guidance is effective for annual periods beginning after December 15, 2025, applied prospectively, and early adoption is permitted. The Company plans to adopt this practical expedient and will update its credit loss estimation policies and related disclosures accordingly.
ITEM 6. DIRECTORS, SENIOR MANAGEMENT AND EMPLOYEES
| A. | Directors and Senior Management. |
The following table sets forth information regarding our executive officers and directors as of the date of this Report.
| Name | Age | Title | ||
| Wing Fung Alfred Siu | 71 | Chairman of the Board of Directors, Chief Executive Officer | ||
| Hei Yue Tina Fong | 44 | Director, Chief Marketing Officer | ||
| Hok Man Jefferson Au | 45 | Independent Director | ||
| Florianna Ann Chi Wan Chan | 45 | Independent Director | ||
| Chun Wa Tam | 62 | Independent Director | ||
| Ho Fai Chung | 62 | Chief Financial Officer |
Wing Fung Alfred Siu. NewGenIvf’s co-founder, Mr. Wing Fung Alfred Siu, has served as the Chairman of the Board and the Chief Executive Officer of NewGenIvf (before the Closing, Legacy NewGenIvf) since 2019. Prior to establishing Legacy NewGenIvf in 2019, Mr. Siu served as a director of First Fertility PGS Center Co., Ltd. since 2014. Mr. Siu received his master’s degree in science and bachelor’s degree in science from Stanford University.
Hei Yue Tina Fong. Ms. Fong has served as a Director and the Chief Marketing Officer of NewGenIvf (before the Closing, Legacy NewGenIvf) since 2019. Prior to establishing NewGenIvf in 2019, Ms. Fong served as a director of First Fertility PGS Center Co., Ltd. since 2014. Ms. Fong received her bachelor’s degree in marketing from Indiana University.
Ho Fai Chung. Mr. Chung has served as NewGenIvf’s Chief Financial Officer since October 10, 2024. Mr. Chung has over 30 years of working experience in Asia. He is a certified public accountant in the U.S. Born in Hong Kong, Mr. Chung holds a Bachelor of Law degree from University of London and a Master’s degree in Accounting and Finance from Lancaster University (UK). Mr. Chung also holds a Master’s degree in International and Public Affairs from Hong Kong University. He started his career with Price Waterhouse (“PwC”) in Hong Kong and then joined a number of companies to take up financial control as well as general management jobs in industries including fashion, office products, telecommunications/internet and advertising. He had worked and based in China, Taiwan, Singapore and Malaysia before and had extensive regional financial controlling exposure in Asia.
Hok Man Jefferson Au. Mr. Au has served as NewGenIVF’s independent director since April 3, 2024. Mr. Au has served as the Assistant Financial Controller and the Company Secretary at Coolpoint Innonism Holding Limited since May 2017 and a director of JWMG CPA Limited, Certified Public Accountants since August 2014. He previously worked as the audit supervisor at Clement C.W. Chan & Co., Certified Public Accountants from September 2010 to March 2014. Mr. Au obtained his honours diploma in accounting from Hong Kong Shue Yan University (formerly known as Hong Kong Shue Yan College) and received his Master of Science in professional accountancy from the University of London. Mr. Au is a member of the Hong Kong Institute of Certified Public Accountants and an associate of the Association of Chartered Certified Accountants.
Florianna Ann Chi Wan Chan has served as NewGenIVF’s independent director since April 15, 2025. Ms Chan has over 20 years of experience in project management, real estate development, and luxury hospitality, she is a proven leader in driving growth and operational excellence. Since 2015, Ms. Chan has led Lab Concept Company Limited, a subsidiary of The Lane Crawford Joyce Group, where she successfully restructured operations, spearheaded rebranding, and digitized processes, achieving significant revenue growth. Previously, she held senior roles at VCC Company Limited, Eton Properties, and Crown Macau, excelling in real estate development, marketing, and VIP services. She holds a Bachelor of Hospitality Management from Central Queensland University in Australia and an Advanced Diploma from William Angliss Institute of TAFE. Fluent in Cantonese, Mandarin, and English, Ms. Chan brings a deep understanding of Asia Pacific markets and a strategic vision that will drive the Company’s continued growth and success.
Chun Wa Tam. Mr. Tam has served as NewGenIvf’s independent director since November 29, 2024. Mr. Tam is currently the chief financial officer, the company secretary, and the authorised representative of China Asia Valley Group Limited since December 2023. Mr. Tam was appointed as an independent non-executive director of Green Energy Group Limited on 24 August 2011. Mr. Tam was also the chief financial officer, the company secretary and the authorised representative of Perfect Group International Holdings Limited from February 2017 to November 2023. The shares of these three companies are listed on the Main Board of the Stock Exchange with stock codes 63, 979 and 3326 respectively. He was the executive director of Chinasing Investment Holdings Limited from February 2009 to August 2015, a company whose shares were listed on the Main Board of Singapore Exchange Limited. Mr. Tam obtained a Master degree of Business Administration from the University of Sydney. Mr. Tam is also a member of Hong Kong Institute of Certified Public Accountants, CPA (Australia) and Institute of Singapore Chartered Accountants. Mr. Tam has more than 30 years in the areas of auditing, accounting, tax, investment banking and company secretarial work.
Election of Officers
Our executive officers are appointed by, and serve at the discretion of, the Board of Directors.
Family Relationships
Mr. Wing Fung Alfred Siu and Ms. Hei Yue Tina Fong are husband and wife. Other than as disclosed in this Report, none of the directors or executive officers has a family relationship as defined in Item 401 of Regulation S-K.
Involvement in Certain Legal Proceedings
To the best of our knowledge, none of our directors or executive officers has, during the past ten years, been involved in any legal proceedings described in subparagraph (f) of Item 401 of Regulation S-K. Our directors and officers have not been involved in any transactions with us or any of our affiliates or associates which are required to be disclosed pursuant to the rules and regulations of the SEC.
B. Compensation
Compensation of Directors and Officers
In 2025, we paid an aggregate of about US$1.8m in cash compensation, including benefits, to our directors and executive officers as a group, respectively. Our directors and executive officers do not receive pension, retirement or other similar benefits, and we have not set aside or accrued any amount to provide such benefits to our executive officers. Our subsidiary in Hong Kong is required by the applicable local laws and regulations to make contributions to Mandatory Provident Fund.
Share Incentive Plans
We adopted a share incentive plan on April 3, 2024, which was subsequently amended by the approval of our Board on March 28, 2025 (the “Share Incentive Plan” or “Plan”). The following summarizes the material terms of the Share Incentive Plan:
Shares Subject to the Plan.
The maximum aggregate number of Shares that are available for awards shall initially be 1,054,260 ordinary shares of the Company (351 ordinary shares as adjusted for reverse stock splits in May 2025, August 2025, December 2025, January 2026 and March 2026), and has been increased by the Board to 454,812 ordinary shares (113,703 ordinary shares as adjusted by reverse stock split in 16 March 2026) of the Company as of the date of this Report. Under the Plan, the total maximum Shares subject to the Plan may be increased by the Board or the Compensation Committee of the Board (the “Committee”) from time to time to allow for the total maximum number of Shares subject to the Plan to be 20% of the then outstanding ordinary shares of the Company at the time of such increase. The number of Shares may be made available from Shares held in treasury or authorized but unissued shares of the Company not reserved for any other purpose.
Administration
The Share Incentive Plan shall be administered by the Committee. The Committee shall have full discretionary authority to administer the Share Incentive Plan, including but not limited to the authority to: (i) interpret the provisions of the Share Incentive Plan, (ii) prescribe, amend and rescind rules and regulations relating to the Share Incentive Plan, (iii) correct any defect, supply any omission, or reconcile any inconsistency in any Award or agreement covering an Award in the manner and to the extent it deems desirable to carry the Share Incentive Plan into effect, and (iv) make all other determinations necessary or advisable for the administration of the Share Incentive Plan. The Committee’s decisions (including any failure to make decisions) shall be binding upon all persons, including the Company, shareholders, Employers, and each Employee, Director, Consultant or Participant, and shall be given deference in any proceeding with respect thereto.
Eligibility
The Share Incentive Plan is open to any Employee, Director or Consultant who, in the opinion of the Committee, has the capacity to contribute to the success of the Company is eligible to be a Participant.
Effective Date, Amendment, Modification and Termination
This Share Incentive Plan shall become effective on the date of its adoption by the Board or a committee of the Board duly authorized by the Board (the “Effective Date”). The Share Incentive Plan will expire on the tenth anniversary of the Effective Date, and no Award may be granted pursuant to the Share Incentive Plan after, the tenth anniversary of the Effective Date, unless otherwise determined by the Committee. Any Awards that are outstanding on the tenth anniversary of the Effective Date shall remain in force according to the terms of the Share Incentive Plan and the applicable Award Agreement.
At any time and from time to time, the Board or the Committee may terminate, amend or modify the Share Incentive Plan; provided, however, that to the extent necessary to comply with Applicable Laws, the Company shall obtain shareholder approval of any Plan amendment in such a manner and to such a degree as required, unless the Board decides to follow home country practice not to seek the shareholder approval for any amendment or modification of the Share Incentive Plan.
C. Board Practices
Committees of the Board of Directors
We established three committees under the Board: an audit committee (“Audit Committee”), a Compensation Committee (“Compensation Committee”) and a nominating and corporate governance committee (“Nominating and Corporate Governance Committee). We have adopted a charter for each of the three committees. Each committee’s members and functions are described below.
Audit Committee. Our Audit Committee consists of Mr. Hok Man Jefferson Au, Mr Tam Chun Wa and Ms Florianna Ann Chi Wan Chan. We have determined that all of these individuals satisfy the “independence” requirements of NASDAQ Rule 5605 and Rule 10A-3 under the Exchange Act. Our Board has determined that Mr. Hok Man Jefferson Au qualifies as an audit committee financial expert and has the accounting or financial management expertise as required under Item 407(d)(5)(ii) and (iii) of Regulation S-K. The audit committee will oversee our accounting and financial reporting processes and the audits of the financial statements of our company. The Audit Committee will be responsible for, among other things:
| ● | establishing clear hiring policies for employees or former employees of the independent auditors; |
| ● | reviewing and recommending to the Board for approval, the appointment, reappointment or removal of the independent auditor, after considering its annual performance evaluation of the independent auditor; |
| ● | approving the remuneration and terms of engagement of the independent auditor and pre-approving all auditing and non-auditing services permitted to be performed by the Company’s independent auditors at least annually; |
| ● | obtaining a written report from the Company’s independent auditor describing matters relating to its independence and quality control procedures; |
| ● | reviewing with the independent registered public accounting firm any audit problems or difficulties and management’s response; |
| ● | discussing with the Company’s independent auditor, among other things, the audits of the financial statements, including whether any material information should be disclosed, in addition to issues regarding accounting and auditing principles and practices; |
| ● | reviewing and approving all proposed related party transactions, as defined in Item 404 of Regulation S-K under the Securities Act; |
| ● | discussing the annual audited financial statements with management and the independent registered public accounting firm; |
| ● | reviewing policies with respect to risk assessment and risk management; |
| ● | reviewing the adequacy and effectiveness of the Company’s accounting and internal control policies and procedures and any special steps taken to monitor and control major financial risk exposures; |
| ● | periodically reviewing and reassessing the adequacy of the committee charter; |
| ● | approving annual audit plans, and undertaking an annual performance evaluation of the internal audit function; |
| ● | establishing and overseeing procedures for the handling of complaints and whistleblowing; |
| ● | meeting separately and periodically with management, the internal auditors and the independent registered public accounting firm; |
| ● | monitoring compliance with the Company’s code of business conduct and ethics, including reviewing the adequacy and effectiveness of its procedures to ensure proper compliance; |
| ● | reporting periodically to the Board; and |
| ● | such other matters that are specifically delegated to the Company’s Audit Committee by the Board from time to time. |
A copy of the audit committee’s current charter is available at our corporate website at www.newgenivf.com.
Compensation Committee. Our Compensation Committee (“Compensation Committee”) consists of Mr. Wing Fung Alfred Siu, Ms Florianna Anne Chi Wan Chan and Ms. Hei Yue Tina Fong. The Chairman of the Compensation Committee is Mr. Siu. The Company has determined that Ms. Chan satisfies the “independence” requirements of Rule 5605(c)(2) of the Nasdaq Stock Market Listing Rules. Ms Florianna Anne Chi Wan Chan, having satisfied the requirements as mentioned, was appointed to the Committee on April 15,2025. The Compensation Committee assists the Board in reviewing and approving compensation structure, including all forms of compensation relating to the Company’s directors and executive officers. The Company’s Chief Executive Officer may not be present at any committee meeting during which their compensation is deliberated upon. The Compensation Committee is responsible for, among other things:
| ● | reviewing and evaluating the Company’s executive compensation and benefits policies generally; |
| ● | reviewing and recommending any incentive compensation or equity plans, programs or other similar arrangements; |
| ● | periodically reviewing and reassessing the adequacy of the Compensation Committee charter; |
| ● | selecting compensation consultant, legal counsel or other adviser only after taking into consideration all factors relevant to that person’s independence from management; |
| ● | reporting periodically to the Board; and |
| ● | such other matters that are specifically delegated to the Compensation Committee by the Board from time to time. |
A copy of the Compensation Committee’s current charter is available at our corporate website at: www.newgenivf.com.
Nominating and Corporate Governance Committee. The Company’s Nominating and Corporate Governance Committee consist of Mr. Wing Fung Alfred Siu, Ms. Hei Yue Tina Fong, and Mr. Hok Man Jefferson Au. The Chairman of the Nominating and Corporate Governance Committee is Mr. Siu. The Company has determined that Mr. Au satisfies the “independence” requirements of Rule 5605(c)(2) of the Nasdaq Stock Market Listing Rules. The Nominating and Corporate Governance Committee assists the Board of Directors in selecting individuals qualified to become the Company’s directors and in determining the composition of the Board of Directors and its committees. The Nominating and Corporate Governance Committee is responsible for, among other things:
| ● | recommending nominees to the Board for election or re-election to the Board, or for appointment to fill any vacancy or newly created directorships on the Board; |
| ● | reviewing periodically with the Board the current composition of the Board with regards to characteristics such as judgment, experience, expertise, diversity and background; |
| ● | recommending to the Board of criteria with respect to nomination or appointment of members of its Board of Directors and chairs and members of its committees or other corporate governance matters as may be required pursuant to any SEC or Nasdaq Stock Market Listing Rules, or otherwise considered desirable and appropriate; |
| ● | recommending to the Board the names of directors to serve as members of the Audit Committee and the Compensation Committee, as well as of the Nominating and Corporate Governance Committee itself; |
| ● | periodically and reassessing the adequacy of the committee charter; |
| ● | overseeing compliance with the corporate governance guidelines and code of business conduct and ethics; and |
| ● | overseeing and leading the self-evaluation of the Board in its performance and |
| ● | effectiveness as a whole. |
A copy of the Nominating and Corporate Governance Committee’s current charter is available at our corporate website at www.newgenivf.com.
Duties and Functions of Directors
Under the laws of the British Virgin Islands, the Company’s directors owe fiduciary duties to the Company, including duty to act honestly and in good faith in what the directors believe to be in the best interests of the company, duty to exercise powers for a proper purpose and directors shall not act, or agree to act, in a matter that contravenes the BVI Companies Act or the Memorandum and Articles of Association, duty to exercise the care, diligence and skill that a reasonable director would exercise in the circumstances, and duty to avoid conflicts of interest. In fulfilling their duty of care to the Company, the Company’s directors must ensure compliance with the Company’s Memorandum and Articles of Association, as amended and restated from time to time. The Company has the right to seek damages if a duty owed by its directors is breached. In limited exceptional circumstances, a shareholder may have the right to seek damages in the Company’s name if a duty owed by the Company’s directors is breached. The functions and powers of the Board include, among other things, (i) convening shareholder meetings at such times and in such manner and places as the director considers necessary or desirable, (ii) declaring dividends, (iii) appointing directors or officers and determining their terms of offices and responsibilities, and (iv) approving the transfer of shares of the Company, including the registering of such shares in the Company’s share register.
Terms of Directors and Officers
The Company’s officers are elected by and serve at the discretion of the Board. Each director holds office for the term fixed by the resolution of shareholders or the resolution of directors appointing him until such time as his successor takes office or until the earlier of his death, resignation or removal from office by resolution of directors with or without cause or by shareholders for cause only by a resolution approved at a duly convened and constituted meeting of the shareholders of the Company by the affirmative vote of not less than 75% of the votes entitled to vote thereon. The directors may at any time appoint any person to be a director either to fill a vacancy or as an addition to the existing directors. Where the directors appoint a person as director to fill a vacancy, the term shall not exceed the term that remained when the person who has ceased to be a director ceased to hold office. A vacancy in relation to directors occurs if a director dies or otherwise ceases to hold office prior to the expiration of his term of office.
Interested Transactions
A director may, subject to any separate requirements for Audit Committee approval under applicable laws or applicable Nasdaq Stock Market Listing Rules, vote on a matter relating to the transaction in which he or she is interested, provided that the interest of any directors in such transaction is disclosed by him or her to all other directors.
Director Agreements
We have entered into director agreements with our directors, which require us to maintain director and officer liability insurance for our directors, provide reimbursements for business related travel and accommodation and other reasonable expenses, and an annual remuneration of between $20,000 for our independent directors, and $325,000 for our executive directors.
D. Employees
NewGenIvf has 87 full-time employees as of December 31, 2025, of which 72 are based in Thailand, Cambodia and Kyrgyzstan. NewGenIvf aims to attract and retain employees with the skills, and experience necessary to implement its growth strategy. The following table sets forth the number of its employees in Thailand, Cambodia and Kyrgyzstan by function as of December 31, 2025:
| Function | Number of employees |
|||
| Thailand | ||||
| Medical professionals | 16 | |||
| Administrative staff and others | 22 | |||
| Sub-total | 38 | |||
| Cambodia | ||||
| Medical professionals | 13 | |||
| Administrative staff and others | 12 | |||
| Sub-total | 25 | |||
| Kyrgyzstan | ||||
| Medical professionals | 3 | |||
| Administrative staff and others | 7 | |||
| Sub-total | 10 | |||
| Hong Kong | ||||
| Administrative staff and others | 14 | |||
| Total | 87 | |||
We believe that we maintain a good working relationship with our employees and we have not experienced any significant labor disputes.
E. Share Ownership
Except as specifically noted, the following table sets forth information with respect to the beneficial ownership of our ordinary shares as of the date of this annual report by:
| ● | each of our directors and executive officers; and | |
| ● | each of our principal shareholders who beneficially own more than 5% of our total outstanding ordinary shares; |
The calculations in the table below are based on 568,542 ordinary shares outstanding, including 568,473 Class A Ordinary Shares and 69 Class B Ordinary Shares, as of March 27, 2026. Unless otherwise indicated, each person has sole investment and voting power with respect to all shares shown as beneficially owned. The term “beneficial owner” of securities refers to any person who, even if not the record owner of the securities, has or shares the underlying benefits of ownership. These benefits include the power to direct the voting or the disposition of the securities or to receive the economic benefit of ownership of the securities. A person also is considered to be the “beneficial owner” of securities that the person has the right to acquire within 60 days by option or other agreement. Beneficial owners include persons who hold their securities through one or more trustees, brokers, agents, legal representatives or other intermediaries, or through companies in which they have a “controlling interest”, which means the direct or indirect power to direct the management and policies of the entity. The Company’s directors and executive officers do not have different voting rights than other shareholders of the Company.
| Name of Beneficial Owner | Number of Class A Ordinary Shares |
Number of class B Ordinary Shares |
% of Outstanding Shares |
%
of Voting Power |
||||||||||||
| Directors and Named Executive Officers: | ||||||||||||||||
| Wing Fung Alfred Siu | 30 | 0.005 | % | 0.517 | % | |||||||||||
| Hei Yue Tina Fong | 39 | 0.007 | % | 0.678 | % | |||||||||||
| Hok Man Jefferson Au | - | - | ||||||||||||||
| Chun Wa Tam | - | - | ||||||||||||||
| Florianna Anne Chi Wan Chan | ||||||||||||||||
| Ho Fai Chung | - | - | ||||||||||||||
| All Directors and Executive Officers as Group | 69 | 0.012 | % | 1.195 | % | |||||||||||
F. Disclosure of a registrant’s action to recover erroneously awarded compensation.
Not applicable.
ITEM 7. MAJOR SHAREHOLDERS AND RELATED PARTY TRANSACTIONS
A. Major Shareholders
Please refer to “Item 6.E. Directors, Senior Management and Employees—Share Ownership.”
B. Related Party Transactions
A summary of related parties of the Company is as follows:
| Relationship | ||
| Mr. Siu, Wing Fung Alfred and Ms. Fong, Hei Yue Tina* | Shareholders and directors | |
| Harcourt Limited | Controlled by Mr. Siu | |
| Mr. Daniel Wai To Siu | Mr. Siu’s son | |
| Mr. Jaspar Siu | Mr. Siu’s son |
| * | Ms. Fong is the spouse of Mr. Siu |
Transaction with Mr. Wing Fung Alfred Siu and Ms. Hei Yue Tina Fong
NewGenIvf recorded remuneration to its directors, Mr. Siu and Ms. Fong. The total remuneration to Mr. Siu, Wing Fung Alfred and Ms Hei Yue Tina Fong was US$380,000 and US$1.76m (including a one-time bonus of $750,000 for maintaining Nasdaq listing status and achieving targeted share capitalization level and benefits paid) during the year ended December 31, 2024 and 2025, respectively. The remuneration during the years ended December 31, 2024 and 2025 was all in the nature of the fair value of the services provided by Mr. Siu and Ms. Fong.
Transaction with Harcourt Limited
During the year ended December 31, 2025, NewGenIvf entered into a service agreement with Harcourt Limited, which is controlled by Mr Siu and Ms Fong, and paid a total of $230,772 as administrative support fee. This service agreement was duly approved by the Board.
Transaction with Mr. Daniel Siu and Mr. Jaspar Siu
During the year ended December 31, 2025, Mr. Daniel Siu and Mr. Jaspar Siu provided consulting services for NewGenIvf with respect to fund raising and other capital activities, and received a total of $219,045 and $65,000 respectively.
C. Interests of Experts and Counsel
No disclosure is required in response to this Item.
ITEM 8. FINANCIAL INFORMATION
A. Consolidated Statements and Other Financial Information
Financial Statements
We have appended consolidated financial statements filed as part of this Report.
Legal Proceedings
We are currently not a party to any material legal or administrative proceedings. We have been, and may from time to time be involved in various legal proceedings arising from the normal course of business activities. The results of litigation cannot be predicted with certainty, and regardless of the outcome, litigation can have an adverse impact on our business, financial condition and/or operations because of defense and settlement costs, diversion of management resources and other factors.
Dividend Policy
We have not declared or paid any cash dividend on our Class A Ordinary Shares as of the date of this Report. We currently intend to retain any future earnings and do not expect to pay any dividends in the near future. Any further determination to pay dividends on our ordinary shares would be at the discretion of our Board of Directors, subject to applicable laws, and would depend on our financial condition, results of operations, capital requirements, general business conditions, and other factors that our Board of Directors may deem relevant.
B. Significant Changes
We have not experienced any significant changes since the date of our audited consolidated financial statements included in this Report.
ITEM 9. THE OFFER AND LISTING
A. Offer and Listing Details
Our Class A ordinary shares were listed on the Nasdaq Global Market since April 4, 2024 under the symbol “NIVF”. On February 28, 2025, we transferred our listing to the Nasdaq Capital Market. Our Warrants are listed on the Nasdaq Capital Market under the symbol “NIVFW.”
B. Plan of Distribution
Not applicable.
C. Markets
Our Class A Ordinary Shares are listed on the Nasdaq Capital Market under the symbol “NIVF” and our warrants are listed on the Nasdaq Capital Market under the symbol “NIVFW.”
D. Selling Shareholders
Not applicable.
E. Dilution
Not applicable.
F. Expenses of the Issue
Not applicable.
ITEM 10. ADDITIONAL INFORMATION
A. Share Capital
Not applicable.
B. Memorandum and Articles of Association
A copy of our amended and restated memorandum and articles of association is filed as Exhibit 1.1 to this Report.
C. Material Contracts
Material Contracts Relating to our Operations
Other than contracts entered into in the ordinary course of business and other than those described under “Item 4. Information on the Company,” “Item 7. Major Shareholders and Related Party Transactions” or described elsewhere in this annual report, the following contracts summarized below are the material contracts that the Company has been a party to for the two years preceding the publication of this Annual Report.
JAK Securities Purchase Agreement
On August 7, 2024, the Company entered into a Securities Purchase Agreement (“Securities Purchase Agreement”) with JAK OPPORTUNITIES VI LLC (“JAK”), pursuant to which, amongst other things: (i) the Company agreed to sell, at an initial closing, (a) a senior convertible note in the aggregate original principal amount not exceeding $1,100,000, convertible into Class A Ordinary Shares pursuant to its terms, (b) a warrant to purchase 1,325,301 Class A Ordinary Shares (22 Shares after adjusted for all the Reverse Stock Splits done as of this Report), and (c) a warrant to purchase 180,722 Class A Ordinary Shares (3 Shares after adjusted for all the Reverse Stock Splits done as of this Report); and (ii) the Company may require JAK (or JAK may require the Company, as applicable) to participate in the sale of (a) one or more additional convertible notes (which aggregate original principal amount for all additional convertible notes shall not exceed $9,500,000) and (b) related Warrants. A copy of the Securities Purchase Agreement is filed as Exhibit 4.25 to this Report.
On April 1, 2025, the Company entered into a new Securities Purchase Agreement (“2025 Securities Purchase Agreement”) with JAK, pursuant to which, amongst other things: (i) the Company agreed to sell, at an initial closing, a senior convertible note in the aggregate original principal amount not exceeding $3,200,000, convertible into Class A Ordinary Shares pursuant to its terms; and (ii) the Company may require JAK (or JAK may require the Company, as applicable) to participate in the sale of one or more additional convertible notes (which aggregate original principal amount for all additional convertible notes shall not exceed $25,600,000). A copy of the 2025 Securities Purchase Agreement is filed as Exhibit 4.33 to this Report.
White Lion Purchase Agreement
On November 21, 2024, the Company entered into a Common Shares Purchase Agreement (the “White Lion Purchase Agreement”) with White Lion Capital, LLC (“White Lion”) and a related Registration Rights Agreement (the “RRA”). Pursuant to the White Lion Purchase Agreement, the Company has the right, but not the obligation, to require White Lion to purchase, from time to time, up to One Hundred Million Dollars ($100,000,000) in aggregate gross purchase price of newly issued Class A Ordinary Shares, with an automatic increase to Three Hundred Million Dollars ($300,000,000) upon any substantial M&A or Material Transaction (as defined in the White Lion Purchase Agreement) and a further option to increase to Five Hundred Million Dollars ($500,000,000) after Two Hundred and Fifty Million Dollars ($250,000,000) has been issued and sold to White Lion under the White Lion Purchase Agreement, subject to certain limitations and conditions set forth in the White Lion Purchase Agreement. A copy of each of the White Lion Purchase Agreement and RRA is filed as Exhibits 4.27 and Exhibits 4.28 to this Report.
On October 31, 2025, the Company entered into a digital-assets purchase agreement with White Lion to acquire up to 600,000 Solana tokens valued at over $110 Million.
MicroSort Purchase Agreement
On January 21, 2025, the Company entered into a Purchase Agreement with Genetics & IVF Institute, Inc. (the “Purchase Agreement,” a copy of which is filed as Exhibit 4. 30 to this Report), pursuant to which the Company purchased all of the Assets (as defined in the Purchase Agreement), IP Licenses (as defined in the Purchase Agreement) and key personnel relating to the MicroSort Business (as defined in the Purchase Agreement) from Genetics & IVF Institute, Inc. for a cash consideration of $750,000 and a share consideration of 125,000 Class A Ordinary Shares.
ASPAC Consulting Services Agreement
On February 2, 2025, the Company entered into a consulting services agreement (the “Consulting Services Agreement”) with A SPAC (Holdings) Group Corp (“ASPAC”), pursuant to which the Company engaged ASPAC for the provision of certain consulting services for a cash consideration of $300,000 and a share consideration of 3,000,000 (equivalent to 50 post Sixth Reverse Stock Split) Class A Ordinary Shares. A copy of the Consulting Services Agreement is filed as Exhibit 4.31 to this Report.
Nodexus Purchase Agreement
On July 29, 2025, the Company acquired advanced cytometry intellectual property and equipment from Nodexus. An independent valuation by a “big-four” accounting firm received in October 2025 confirmed a fair value of $17.9 million for the IP, resulting in a total bargain purchase gain of $19.16 million. The technology associated with the patents is related to cell sorting to be performed via a device called NX One and could be applied in different fields including In Vitro Fertilization. The Patents would allow NewGenIvf to penetrate the IVF market in the United States by leasing the NX One to clinics that provide IVF treatments in the United States.
Joint Venture Agreement with BNW Real Estate Development LLC (“BNW”)
On October 6, 2025, NewGenProperty Limited (“NewGenProperty”), a wholly owned subsidiary of the Company, entered into a joint venture agreement with BNW to develop the Plot. Under the terms of the joint venture agreement, NewGenProperty shall hold 60% of the joint venture, and be responsible for funding 36% of the purchase price of the Plot to the Master Developer, while BNW shall be responsible for all the other cashflow and financing requirements of the project. In the event that BNW pays the balance of the Purchase Price of the Plot to the Master Developer, NewGenProperty shall reimburse BNW in accordance with the provisions of the joint venture agreement. NewGenProperty’s entitlement is an amount equivalent to 36% of the Gross Sales Revenue or Gross Sellable Area generated from the project, with deductions including (i) up to 10% of External and Internal Real Estate Agent and Brokerage Fees; (ii) 2% of Gross Sales Revenue for BNW’s Marketing Fees; and (iii) any amounts paid by the Developer towards the balance of the Purchase Price of the Plot on behalf of NewGenProperty.
Financing Agreements
The clinics would utilize the NX One to perform gender selection services and detect potential genetic diseases during the IVF process and charge a service fee from customers On October 15, 2025, November 4, 2025, and January 22, 2026, the Company entered into three securities purchase agreements (“Vanquish SPAs”) with Vanquish Funding Group Inc. (“Vanquish”). Pursuant to the terms of the Vanquish SPAs, the Company agreed to issue three convertible promissory notes to Vanquish in the principal amounts of $257,000, $157,000 and $107,000. Effective October 15, 2025, November 4, 2025, and January 22, 2026, the Company issued three convertible promissory notes (“Vanquish Notes”) to Vanquish II consistent with the terms of the Vanquish SPAs. The Vanquish Notes bear interest at 10% and mature by June 30, 2026, June 30, 2026, and October 30, 2026, respectively. Pursuant to the terms of the Vanquish Notes, the outstanding principal and accrued interest on the Note shall be convertible into shares of the Company’s common stock as set forth therein. Copies of the Vanquish SPAs are filed as Exhibits 10.33, 10.34, 10.35 to this Report.
On November 12, 2025, the Company entered into a securities purchase agreement (“Labrys SPA”) with Labrys Fund II, L.P. (“Labrys”). Pursuant to the terms of the Labrys SPA, the Company agreed to issue a convertible promissory note to Labrys at a purchase price of $232,500, with an original issue discount of $17,500. On November 12, 2025, the Company issued a convertible promissory note to Labrys (“Labrys Note”) consistent with the terms of the Labrys SPA. The Labrys Note bears interest at 10% and matures 12 months from the date of issuance on the Labrys Note. A copy of the Labrys SPA is filed as Exhibit 10.36 to this Report.
On January 22, 2026, the Company entered into a securities purchase agreement (“Boot Capital SPA”) with Boot Capital LLC (“Boot Capital”). Pursuant to the terms of the Boot Capital SPA, the Company agreed to issue a convertible promissory note to Boot Capital at a purchase price of $50,000. On January 22, 2026, the Company issued a convertible promissory note to Boot Capital (“Boot Capital Note”) consistent with the terms of the Boot Capital SPA. The Boot Capital Note bears interest at 10% and matures on October 30, 2026. Copies of the Boot Capital SPA and the Boot Capital Note are filed as Exhibits 10.37 and 10.38 to this Report, respectively.
On March 18, 2026, the Company entered into a securities purchase agreement (“CFI Capital SPA”) with CFI Capital LLC (“CFI Capital”). Pursuant to the terms of the CFI Capital SPA, the Company agreed to issue a 6% convertible redeemable note to CFI Capital at a purchase price of $180,000, with an original issue discount of $20,000, for a principal amount of $200,000. On March 18, 2026, the Company issued a 6% convertible redeemable note to CFI Capital (“CFI Capital Note”) consistent with the terms of the CFI Capital SPA. The CFI Capital Note bears interest at 6% and matures on March 18, 2027. Copies of the CFI Capital SPA and CFI Capital Note are filed as Exhibits 10.39 and 10.40 to this Report, respectively.
On March 25, 2026, the Company entered into a securities purchase agreement (“FirstFire SPA”) with FirstFire Global Opportunities Fund, LLC (“FirstFire”). Pursuant to the terms of the FirstFire SPA, the Company agreed to issue a convertible promissory note to FirstFire at a purchase price of $200,000, with an original issue discount of $20,000, for a principal amount of $220,000. The Company also agreed to issue 20,000 shares of Class A Ordinary Shares to FirstFire as commitment shares. On March 25, 2026, the Company issued a convertible promissory note to FirstFire (“FirstFire Note”) consistent with the terms of the FirstFire SPA. The FirstFire Note bears interest at 12% and matures on March 25, 2027. Copies of the FirstFire SPA and FirstFire Note are filed as Exhibits 10.41 and 10.42 to this Report, respectively.
D. Exchange Controls and Other Limitations Affecting Security Holders
Under the laws of the British Virgin Islands, there are currently no restrictions on the export or import of capital, including foreign exchange controls or restrictions that affect the remittance of dividends, interest or other payments to non-resident holders of our ordinary shares.
E. Taxation
The following is a general discussion of the material U.S. federal income tax consequences of the ownership and disposition of the Class A Ordinary Shares and Warrants (collectively, the “Company Securities”).
This discussion is based on provisions of the Code, the Treasury Regulations promulgated thereunder (whether final, temporary, or proposed), administrative rulings of the IRS, and judicial decisions, all as in effect on the date hereof, and all of which are subject to differing interpretations or change, possibly with retroactive effect. This discussion does not purport to be a complete analysis or listing of all potential U.S. federal income tax considerations that may apply to a securityholder of the Company as a result of the ownership and disposition of the Company Securities. In addition, this discussion does not address all aspects of U.S. federal income taxation that may be relevant to particular holders nor does it take into account the individual facts and circumstances of any particular holder that may affect the U.S. federal income tax consequences to such holder, and accordingly, is not intended to be, and should not be construed as, tax advice. This discussion does not address the U.S. federal 3.8% Medicare tax imposed on certain net investment income or any aspects of U.S. federal taxation other than those pertaining to the income tax, nor does it address any tax consequences arising under any U.S. state and local, or non-U.S. tax laws, or, except as discussed here, any tax reporting obligations of a holder of the Company Securities. Holders should consult their own tax advisors regarding such tax consequences in light of their particular circumstances.
No ruling has been requested or will be obtained from the IRS regarding the U.S. federal income tax consequences discussed below; thus, there can be no assurance that the IRS will not challenge the U.S. federal income tax treatment described below or that, if challenged, such treatment will be sustained by a court.
This summary is limited to considerations relevant to U.S. Holders that hold the Company Securities as “capital assets” within the meaning of section 1221 of the Code (generally, property held for investment). This discussion does not address all aspects of U.S. federal income taxation that may be important to holders in light of their individual circumstances, including holders subject to special treatment under the U.S. tax laws, such as, for example:
| ● | banks or other financial institutions, underwriters, or insurance companies; |
| ● | traders in securities who elect to apply a mark-to-market method of accounting; |
| ● | real estate investment trusts and regulated investment companies; |
| ● | tax-exempt organizations, qualified retirement plans, individual retirement accounts, or other tax- deferred accounts; |
| ● | expatriates or former citizens or long-term residents of the United States; |
| ● | subchapter S corporations, partnerships or other pass-through entities or investors in such entities; |
| ● | any holder that is not a U.S. Holder; |
| ● | dealers or traders in securities, commodities or currencies; |
| ● | grantor trusts; |
| ● | persons subject to the alternative minimum tax; |
| ● | U.S. persons whose “functional currency” is not the U.S. dollar; |
| ● | persons who receive stock of the Company through the issuance of restricted share under an incentive plan or through a tax-qualified retirement plan or otherwise as compensation; |
| ● | U.S. shareholders of controlled foreign corporations, as those terms are defined in Sections 951(b) and 957(a), respectively; |
| ● | persons who own (directly or through attribution) 5% or more (by vote or value) of the outstanding Class A Ordinary Shares (excluding treasury shares); |
| ● | holders holding ASCA securities, or, after the Business Combination, the Company Securities, as a position in a “straddle,” as part of a “synthetic security” or “hedge,” as part of a “conversion transaction,” or other integrated investment or risk reduction transaction. |
As used in this Report, the term “U.S. Holder” means a beneficial owner of the Company Securities, that is, for U.S. federal income tax purposes:
| ● | an individual who is a citizen or resident of the United States; |
| ● | a corporation (or other entity that is classified as a corporation for U.S. federal income tax purposes) that is created or organized in or under the laws of the United States or any State thereof or the District of Columbia; |
| ● | an estate the income of which is subject to U.S. federal income tax regardless of its source; or |
| ● | a trust (i) if a court within the United States is able to exercise primary supervision over the administration of the trust and one or more U.S. persons have the authority to control all substantial decisions of the trust, or (ii) that has a valid election in effect under applicable Treasury Regulations to be treated as a U.S. person for U.S. federal income tax purposes. |
If a partnership, including for this purpose any entity or arrangement that is treated as a partnership for U.S. federal income tax purposes, holds the Company Securities, the U.S. federal income tax treatment of a partner in such partnership will generally depend on the status of the partner and the activities of the partnership. A holder that is a partnership and the partners in such partnership should consult their own tax advisors with regard to the U.S. federal income tax consequences of ownership and disposition of the Company Securities.
THIS SUMMARY DOES NOT PURPORT TO BE A COMPREHENSIVE ANALYSIS OR DESCRIPTION OF ALL POTENTIAL U.S. FEDERAL INCOME TAX CONSEQUENCES OF OWNERSHIP AND DISPOSITION OF THE COMPANY SECURITIES. IN ADDITION, THE U.S. FEDERAL INCOME TAX TREATMENT OF THE BENEFICIAL OWNERS OF THE COMPANY SECURITIES MAY BE AFFECTED BY MATTERS NOT DISCUSSED HEREIN AND DEPENDS IN SOME INSTANCES ON DETERMINATIONS OF FACT AND INTERPRETATIONS OF COMPLEX PROVISIONS OF U.S. FEDERAL INCOME TAX LAW FOR WHICH NO CLEAR PRECEDENT OR AUTHORITY MAY BE AVAILABLE. HOLDERS OF THE COMPANY SECURITIES SHOULD CONSULT WITH THEIR TAX ADVISORS REGARDING THE PARTICULAR TAX CONSEQUENCES TO THEM OF THE OWNERSHIP AND DISPOSITION OF THE COMPANY SECURITIES, INCLUDING THE APPLICABILITY AND EFFECTS OF U.S. FEDERAL, STATE, LOCAL, AND OTHER TAX LAWS.
Distribution on the Class A Ordinary Shares
Subject to the PFIC rules discussed below “— Passive Foreign Investment Company Status,” the gross amount of any distribution on the Class A Ordinary Shares that is made out of the Company’s current and accumulated earnings and profits (as determined for U.S. federal income tax purposes) will generally be taxable to a U.S. Holder as ordinary dividend income on the date such distribution is actually or constructively received by such U.S. Holder. Any such dividends paid to corporate U.S. Holders generally will not qualify for the dividends-received deduction that may otherwise be allowed under the Code.
Dividends received by non-corporate U.S. Holders, including individuals, from a “qualified foreign corporation” may be eligible for reduced rates of taxation, provided that certain holding period requirements and other conditions are satisfied. For these purposes, a non-U.S. corporation will be treated as a qualified foreign corporation with respect to dividends paid by that corporation on shares that are readily tradable on an established securities market in the United States. U.S. Treasury Department guidance indicates that shares listed on Nasdaq will be considered readily tradable on an established securities market in the United States. Even if the Class A Ordinary Shares are listed on Nasdaq, there can be no assurance that the Class A Ordinary Shares will be considered readily tradable on an established securities market in future years. Non-corporate U.S. Holders that do not meet a minimum holding period requirement or that elect to treat the dividend income as “investment income” pursuant to Section 163(d)(4) of the Code (dealing with the deduction for investment interest expense) will not be eligible for the reduced rates of taxation regardless of the Company’s status as a qualified foreign corporation. In addition, the rate reduction will not apply to dividends if the recipient of a dividend is obligated to make related payments with respect to positions in substantially similar or related property. This disallowance applies even if the minimum holding period has been met. Finally, the Company will not constitute a qualified foreign corporation for purposes of these rules if it is a PFIC for the taxable year in which it pays a dividend or for the preceding taxable year. See the discussion below under “— Passive Foreign Investment Company Status.
The amount of any dividend paid in foreign currency will be the U.S. dollar value of the foreign currency distributed by the Company, calculated by reference to the exchange rate in effect on the date the dividend is includible in the U.S. Holder’s income, regardless of whether the payment is in fact converted into U.S. dollars on the date of receipt. Generally, a U.S. Holder should not recognize any foreign currency gain or loss if the foreign currency is converted into U.S. dollars on the date the payment is received. However, any gain or loss resulting from currency exchange fluctuations during the period from the date the U.S. Holder includes the dividend payment in income to the date such U.S. Holder actually converts the payment into U.S. dollars will be treated as ordinary income or loss. That currency exchange income or loss (if any) generally will be income or loss from U.S. sources for foreign tax credit limitation purposes.
To the extent that the amount of any distribution made by the Company on the Class A Ordinary Shares exceeds the Company’s current and accumulated earnings and profits for a taxable year (as determined under U.S. federal income tax principles), the distribution will first be treated as a tax-free return of capital, causing a reduction in the adjusted basis of the U.S. Holder’s the Class A Ordinary Shares, and to the extent the amount of the distribution exceeds the U.S. Holder’s tax basis, the excess will be taxed as capital gain recognized on a sale or exchange as described below under “— Sale, Exchange, Redemption or Other Taxable Disposition of the Company Securities.”
Sale, Exchange, Redemption or Other Taxable Disposition of the Company Securities
Subject to the discussion below under “— Passive Foreign Investment Company Status,” a U.S. Holder will generally recognize gain or loss on any sale, exchange, redemption, or other taxable disposition of the Class A Ordinary Shares and the Warrants in an amount equal to the difference between the amount realized on the disposition and such U.S. Holder’s adjusted tax basis in such the Class A Ordinary Shares or Warrants. Any gain or loss recognized by a U.S. Holder on a taxable disposition of the Class A Ordinary Shares or Warrants will generally be capital gain or loss and will be long-term capital gain or loss if the holder’s holding period in the Class A Ordinary Shares or Warrants exceeds one year at the time of the disposition. Preferential tax rates may apply to long-term capital gains of non-corporate U.S. Holders (including individuals). The deductibility of capital losses is subject to limitations. Any gain or loss recognized by a U.S. Holder on the sale or exchange of the Class A Ordinary Shares or the Warrants will generally be treated as U.S. source gain or loss.
Exercise or Lapse of a Warrant
Except as discussed below with respect to the cashless exercise of a Warrant, a U.S. Holder generally will not recognize gain or loss upon the acquisition of an ordinary share of the Company on the exercise of a Warrant for cash. A U.S. Holder’s tax basis in an ordinary share received upon exercise of the Warrant generally will be an amount equal to the sum of the U.S. Holder’s tax basis in the Warrant exchanged therefor and the exercise price. The U.S. Holder’s holding period for an ordinary share received upon exercise of the Warrant will begin on the date following the date of exercise (or possibly the date of exercise) of the Warrants and will not include the period during which the U.S. Holder held the Warrants. If a Warrant is allowed to lapse unexercised, a U.S. Holder generally will recognize a capital loss equal to such holder’s tax basis in the Warrant.
The tax consequences of a cashless exercise of a warrant are not clear under current tax law. A cashless exercise may be tax-free, either because the exercise is not a gain realization event or because the exercise is treated as a recapitalization for U.S. federal income tax purposes. In either tax-free situation, a U.S. Holder’s basis in the Class A Ordinary Shares received would equal the holder’s basis in the Warrant. If the cashless exercise were treated as not being a gain recognition event, a U.S. Holder’s holding period in the Class A Ordinary Shares would be treated as commencing on the date following the date of exercise (or possibly the date of exercise) of the Warrant. If the cashless exercise were treated as a recapitalization, the holding period of the Class A Ordinary Share would include the holding period of the Warrant.
It is also possible that a cashless exercise could be treated in part as a taxable exchange in which gain or loss would be recognized. In such event, a U.S. Holder would recognize gain or loss with respect to the portion of the exercised Warrants treated as surrendered to pay the exercise price of the Warrants (the “surrendered warrants”). The U.S. Holder would recognize capital gain or loss with respect to the surrendered warrants in an amount generally equal to the difference between (i) the fair market value of the Class A Ordinary Shares that would have been received with respect to the surrendered warrants in a regular exercise of the Warrants and (ii) the sum of the U.S. Holder’s tax basis in the surrendered warrants and the aggregate cash exercise price of such warrants (if they had been exercised in a regular exercise). In this case, a U.S. Holder’s tax basis in the Class A Ordinary Shares received would equal the U.S. Holder’s tax basis in the Warrants exercised plus (or minus) the gain (or loss) recognized with respect to the surrendered warrants. A U.S. Holder’s holding period for the Class A Ordinary Shares would commence on the date following the date of exercise (or possibly the date of exercise) of the Warrant.
Due to the absence of authority on the U.S. federal income tax treatment of a cashless exercise, there can be no assurance which, if any, of the alternative tax consequences and holding periods described above would be adopted by the IRS or a court of law. Accordingly, U.S. Holders should consult their tax advisors regarding the tax consequences of a cashless exercise.
Passive Foreign Investment Company Status
Certain adverse U.S. federal income tax consequences could apply to a U.S. Holder if the Company or any of its subsidiaries is treated as a PFIC for any taxable year during which the U.S. Holder holds the Company Securities. A non-U.S. corporation will be classified as a PFIC for any taxable year (a) if at least 75% of its gross income in a taxable year, including its pro rata share of the gross income of any entity in which it is considered to own at least 25% of the interest by value, is passive income, or (b) if at least 50% of its assets in a taxable year of the foreign corporation, ordinarily determined based on fair market value and averaged quarterly over the year, including its pro rata share of the assets of any entity in which it is considered to own at least 25% of the interest by value, are held for the production of, or produce, passive income. Passive income generally includes dividends, interest, rents and royalties (other than rents or royalties derived from the active conduct of a trade or business) and gains from the disposition of passive assets.
If the Company is not a PFIC in the 2024 taxable year, such U.S. Holder would likely recognize gain (but not loss if the Reincorporation Merger qualifies as a “reorganization”) upon the exchange of ASCA securities for The Company securities pursuant to the Reincorporation Merger. The gain (or loss) would be computed as described above under “— If the Reincorporation Merger Does Not Qualify as a Reorganization.” Any such gain recognized by such U.S. Holder on the exchange of ASCA securities for The Company securities would be allocated ratably over the U.S. Holder’s holding period for the ASCA securities. Such amounts allocated for the current taxable year and any taxable year prior to the first taxable year in which ASCA was a PFIC would be treated as ordinary income, and not as capital gain, in the U.S. Holder’s taxable year, and such amounts allocated to each other taxable year beginning with the year that ASCA became a PFIC would be taxed at the highest tax rate in effect for each year to which the gain was allocated, together with a special interest charge on the tax attributable to each such year.
Whether the Company is a PFIC for any taxable year is a factual determination that depends on, among other things, the composition of the Company’s income and assets, the market value of its assets, and potentially the composition of the income and assets of one or more of the Company’s subsidiaries and the market value of their assets in that year. Whether a Company subsidiary is a PFIC for any taxable year is likewise a factual determination that depends on, among other things, the composition of the subsidiary’s income and assets and the market value of such assets in that year. One or more changes in these factors may cause the Company and/or one or more of its subsidiaries to become a PFIC for a taxable year even though it has not been a PFIC for one or more prior taxable years. Whether the Company or a subsidiary is treated as a PFIC for U.S. federal income tax purposes is a factual determination that must be made annually at the close of each taxable year and, thus, is subject to significant uncertainty. Moreover, there can be no assurance that the Company will timely provide a PFIC annual information statement for 2024 or going forward. The failure to provide such information on an annual basis could preclude U.S. Holders from making or maintaining a “qualified electing fund” election under Section 1295 of the Code.
If the Company were determined to be a PFIC for any taxable year (or portion thereof) that is included in the holding period of a U.S. Holder of Class A Ordinary Shares, the U.S. Holder did not make a valid “mark-to-market” election, such U.S. Holder generally will be subject to special rules with respect to:
| ● | any gain recognized by the U.S. Holder on the sale or other disposition of the Company Securities (including a redemption treated as a sale or exchange); and |
| ● | any “excess distribution” made to the U.S. Holder (generally, any distributions to such U.S. Holder during a taxable year of the U.S. Holder that are greater than 125% of the average annual distributions received by such U.S. Holder in respect of the Class A Ordinary Shares during the three preceding taxable years of such U.S. Holder or, if shorter, such U.S. Holder’s holding period for such ordinary shares). |
Under these rules:
| ● | the U.S. Holder’s gain or excess distribution will be allocated ratably over the U.S. Holder’s Company Securities; |
| ● | the amount allocated to the U.S. Holder’s taxable year in which the U.S. holder recognized gain or received the excess distribution, or to the period in the U.S. Holder’s holding period before the first day of the Company’s first taxable year in the Company is a PFIC, will be taxed as ordinary income; |
| ● | the amount allocated to other taxable years (or portions thereof) of the U.S. Holder and included in its holding period will be taxed at the highest tax rate in effect for that year and applicable to the U.S. Holder; and |
| ● | the interest charge generally applicable to underpayments of tax will be imposed in respect of the tax attributable to each such other taxable year of the U.S. Holder. |
Although a determination as to the Company’s PFIC status will be made annually, an initial determination that the Company is a PFIC will generally apply for subsequent years to a U.S. Holder who held Company Securities while the Company was a PFIC, whether or not the Company meets the test for PFIC status in those subsequent years.
If a U.S. Holder, at the close of its taxable year, owns shares in a PFIC that are treated as marketable stock, the U.S. Holder may make a mark-to-market election with respect to such shares for such taxable year. If the U.S. Holder makes a valid mark-to-market election for the first taxable year of the U.S. Holder in which the U.S. Holder holds (or is deemed to hold) the Class A Ordinary Shares and for which the Company is determined to be a PFIC, such holder generally will not be subject to the PFIC rules described above in respect to the Class A Ordinary Shares as long as such shares continue to be treated as marketable stock. Instead, in general, the U.S. Holder will include as ordinary income each year that the Company is treated as a PFIC the excess, if any, of the fair market value of its Class A Ordinary Shares at the end of its taxable year over the adjusted basis in its Class A Ordinary Shares. The U.S. Holder also will be allowed to take an ordinary loss in respect of the excess, if any, of the adjusted basis of its Class A Ordinary Shares over the fair market value of its Class A Ordinary Shares at the end of its taxable year (but only to the extent of the net amount of previously recognized income as a result of the mark-to-market election). The U.S. Holder’s adjusted tax basis in its Class A Ordinary Shares will be adjusted to reflect any such income or loss amounts, and any further gain recognized on a sale or other taxable disposition of the Class A Ordinary Shares in a taxable year in which the Company is treated as a PFIC will be treated as ordinary income. Special tax rules may also apply if a U.S. Holder makes a mark-to-market election for a taxable year after the first taxable year in which the U.S. Holder holds (or is deemed to hold) its Class A Ordinary Shares and for which the Company is treated as a PFIC. Currently, a mark-to-market election may not be made with respect to the Warrants.
The mark-to-market election is available only for stock that is regularly traded on a national securities exchange that is registered with the SEC, including Nasdaq (on which the Company Securities are traded), or on a foreign exchange or market that the IRS determines has rules sufficient to ensure that the market price represents a legitimate and sound fair market value. Such stock generally will be “regularly traded” for any calendar year during which such stock is traded, other than in de minimis quantities, on at least 15 days during each calendar quarter, but no assurances can be given in this regard with respect to the Class A Ordinary Shares. U.S. Holders should consult their own tax advisors regarding the availability and tax consequences of a mark-to-market election in respect of the Class A Ordinary Shares under their particular circumstances.
If the Company is a PFIC and, at any time, has a foreign subsidiary that is classified as a PFIC, U.S. Holders generally would be deemed to own a portion of the shares of such lower-tier PFIC, and generally could incur liability for the deferred tax and interest charge described above if the Company were to receive a distribution from, or dispose of all or part of the Company’s interest in, the lower-tier PFIC (even though such U.S. Holder would not receive the proceeds of those distributions or dispositions) or the U.S. Holders otherwise were deemed to have disposed of an interest in the lower-tier PFIC. A mark-to-market election generally would not be available with respect to such lower-tier PFIC. U.S. Holders are urged to consult their own tax advisors regarding the tax issues raised by lower-tier PFICs.
A U.S. Holder that owns (or is deemed to own) shares in a PFIC during any taxable year of the U.S. Holder, may have to file an IRS Form 8621 (whether or not a mark-to-market election is or has been made) with such U.S. Holder’s U.S. federal income tax return and provide any such other information as may be required by the U.S. Treasury Department. Failure to do so, if required, will extend the statute of limitations until such required information is furnished to the IRS.
The rules dealing with PFICs and mark-to-market elections are very complex and are affected by various factors in addition to those described above. Accordingly, U.S. Holders of Company Securities should consult their own tax advisors concerning the application of the PFIC rules to the Company Securities under the U.S. Holders’ particular circumstances.
Information Reporting and Backup Withholding
In general, information reporting requirements may apply to dividends received by U.S. Holders of the Class A Ordinary Shares (including constructive dividends), and the proceeds received on sale or other taxable disposition of the Class A Ordinary Shares or Warrants effected within the United States (and, in certain cases, outside the United States), in each case, other than U.S. Holders that are exempt recipients (such as corporations). Backup withholding (currently at a rate of 24%) may apply to such amounts if the U.S. Holder fails to provide an accurate taxpayer identification number (generally on an IRS Form W-9 provided to the paying agent or the U.S. Holder’s broker) or is otherwise subject to backup withholding.
Certain U.S. Holders holding specified foreign financial assets with an aggregate value in excess of the applicable dollar threshold are required to report information to the IRS relating to the Company Securities, subject to certain exceptions (including an exception for the Company Securities held in accounts maintained by U.S. financial institutions), by attaching a complete IRS Form 8938, Statement of Specified Foreign Financial Assets, with their tax return, for each year in which they hold the Company Securities. In addition to these requirements, U.S. Holders may be required to annually file FinCEN Report 114 (Report of Foreign Bank and Financial Accounts) with the U.S. Department of Treasury. U.S. Holders should consult their own tax advisors regarding information reporting requirements relating to their ownership of the Company Securities.
Backup withholding is not an additional tax. Any amounts withheld under the backup withholding rules may be allowed as a refund or credit against a holder’s U.S. federal income tax liability, if any, provided the required information is timely furnished to the IRS.
F. Dividends and Paying Agents
Not applicable.
G. Statement by Experts
Not applicable.
H. Documents on Display
We previously filed with the SEC our shell company report on Form 20-F with respect to the Business Combination.
Since we are a “foreign private issuer,” we are exempt from the rules and regulations under the Exchange Act prescribing the furnishing and content of proxy statements, and our officers, directors and principal shareholders are exempt from the reporting and “short-swing” profit recovery provisions contained in Section 16 of the Exchange Act, with respect to their purchase and sale of our shares. In addition, we are not required to file reports and financial statements with the SEC as frequently or as promptly as U.S. companies whose securities are registered under the Exchange Act. However, we are required to file with the SEC an Annual Report on Form 20-F containing financial statements audited by an independent accounting firm. The SEC also maintains a website at http://www.sec.gov that contains reports and other information that we file with or furnish electronically with the SEC.
We are subject to the periodic reporting and other informational requirements of the Exchange Act. Under the Exchange Act, we are required to file reports and other information with the SEC. Specifically, we are required to file annually a Form 20-F within four months after the end of each fiscal year, which is December 31. Copies of reports and other information, when so filed, may be inspected without charge and may be obtained at prescribed rates at the public reference facilities maintained by the SEC at 100 F Street, N.E., Room 1580, Washington, D.C. 20549. The public may obtain information regarding the Washington, D.C. Public Reference Room by calling the Commission at 1-800-SEC-0330. The SEC also maintains a website at www.sec.gov that contains reports, proxy and information statements, and other information regarding registrants that make electronic filings with the SEC using its EDGAR system. As a foreign private issuer, we are exempt from the rules under the Exchange Act prescribing the furnishing and content of quarterly reports and proxy statements, and officers, directors and principal shareholders are exempt from the reporting and short-swing profit recovery provisions contained in Section 16 of the Exchange Act.
In accordance with NASDAQ Stock Market Rule 5250(d), we will post this Report on our website at https://newgenivf.gcs-web.com/financials/annual-reports.
I. Subsidiary Information
Please refer to “Item 4. Information on the Company - C. Organizational Structure.”
J. Annual Report to Security Holders.
Not applicable.
ITEM 11. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISKS
Accounts receivable
In most cases, NewGenIvf requires customers to pay before service is rendered. Thus, the risks of unrecoverable account receivable is minimized. In order to further reduce the credit risk, NewGenIvf’s management team monitors and ensures that follow-up action is taken to recover overdue debts. NewGenIvf considers the probability of default upon initial recognition of the asset and whether there has been a significant increase in credit risk on an ongoing basis throughout each reporting period. To assess whether there is a significant increase in credit risk, NewGenIvf compares the risk of a default occurring on the asset as at the reporting date with the risk of default as at the date of initial recognition. It considers available reasonable and supportive forwarding-looking information, such as GDP growth rate and nominal GDP per capita. Based on the impairment assessment performed by NewGenIvf, the directors considered the loss allowance for account receivables as of December 31, 2025 and December 31, 2024 as immaterial, respectively.
Cash and cash equivalents
NewGenIvf is exposed to concentration of credit risk on liquid funds which are deposited with several banks with high credit ratings. The credit risk on liquid funds is limited because the counterparties are banks with high credit ratings assigned by international credit-rating agencies.
Deposits and other receivables
NewGenIvf assessed the impairment for deposits and other receivables based on internal credit rating and aging of these debtors which, in the opinion of the directors, have no significant increase in credit risk since initial recognition. Based on the impairment assessment performed by the Company, the loss allowance for deposits and other receivables as of December 31, 2024 and December 31, 2025 were both immaterial respectively.
Cash flow interest rate risk
NewGenIvf is exposed to cash flow interest rate risk through the changes in interest rates related mainly to its variable-rates bank balances.
NewGenIvf currently does not have any interest rate hedging policy in relation to fair value interest rate risk and flow interest rate risk. The directors monitor NewGenIvf’s exposures on an ongoing basis and will consider hedging the interest rate should the need arises.
NewGenIvf has an outstanding convertible note of $5,289,000 as of December 31, 2025. The convertible note carries an annual fixed interest rate. Thus, the interest rate fluctuation risk has been controlled.
Sensitivity analysis
The sensitivity analysis below has been determined by assuming that a change in interest rates had occurred at the end of the reporting period and had been applied to the exposure to interest rates for financial instruments in existence at that date. 1% increase or decrease is used when reporting interest rate risk internally to key management personnel and represents management’s assessment of the reasonably possible change in interest rates.
If interest rates had been 1% higher or lower and all other variables were held constant, NewGenIvf’s post tax loss for the years ended December 31, 2025 and 2024 would have increased or decreased by approximately US$52,890 and US$26,894, respectively.
Foreign currency risk
Foreign currency risk is the risk that the holding of foreign currency assets will affect NewGenIvf’s financial position as a result of a change in foreign currency exchange rates.
NewGenIvf’s monetary assets and liabilities are mainly denominated in HK$ and THB which are the same as the functional currencies of the relevant group entities. Hence, in the opinion of the directors of NewGenIvf, the currency risk of US$ is considered insignificant. NewGenIvf currently does not have a foreign currency hedging policy to eliminate currency exposures. However, the directors monitor the related foreign currency exposure closely and will consider hedging significant foreign currency exposures should the need arise.
Market Liquidity Risk
NewGenIvf is exposed to market liquidity risk with respect to certain assets held on our balance sheet. Market liquidity risk is the risk that we cannot realize the fair value of our assets when needed due to insufficient market depth, disruption, or the absence of willing buyers. As of December 31, 2025, we held Solana as treasury investment. These assets trade in markets that may experience periods of reduced liquidity, particularly during periods of economic stress or market disruption.
Economic and political risks
NewGenIvf’s operations are mainly conducted in Thailand, Cambodia and Kyrgyzstan. Accordingly, NewGenIvf’s business, financial condition, and results of operations may be influenced by changes in the political, economic, and legal environments in Thailand, Cambodia and Kyrgyzstan.
NewGenIvf’s operations in Thailand, Cambodia and Kyrgyzstan are subject to special considerations and significant risks not typically associated with companies in North America and Western Europe. These include risks associated with, among other things, the political, economic and legal environment and foreign currency exchange. NewGenIvf’s results may be adversely affected by changes in the political and social conditions in Thailand, Cambodia and Kyrgyzstan, and by changes in governmental policies with respect to laws and regulations, anti-inflationary measures, currency conversion, remittances abroad, and rates and methods of taxation, among other things.
Travel restriction risk
International clients contribute a large portion of NewGenIvf’s revenue. International clients need to travel to Thailand, Cambodia and Kyrgyzstan for treatment services, where NewGenIvf’s operations are mainly conducted. International traveling to Thailand, Cambodia and Kyrgyzstan may be affected by a number of factors, including local and global political and economic conditions. Furthermore, an outbreak, or threatened outbreak, of any severe contagious disease may also in turn significantly reduce the demand of traveling or cause extensive travel restrictions. NewGenIvf’s results may be materially and adversely affected if travel restriction was imposed or difficulties in cross-border flow arose.
The recent war involving the US, Israel, and Iran has severely disrupted international travel, beginning with the immediate closure of airspace over several Middle Eastern countries and the suspension of flights through critical hubs like Dubai and Doha, which has stranded hundreds of thousands of passengers. This has forced airlines to cancel thousands of flights or take longer, more expensive routes, leading to a surge in ticket prices and the imposition of new fuel surcharges that are affecting travelers globally. Governments have responded by issuing widespread “do not travel” warnings for the region and organizing evacuations, while embassies have halted routine visa services, further complicating international mobility. The compounded effect is a climate of uncertainty where even trips not destined for the Middle East face potential delays, higher costs, and complex rebooking challenges.
Inflation risk
Management of NewGenIvf monitors changes in prices levels. Historically inflation has not materially impacted NewGenIvf’s consolidated financial statements; however, significant increases in the price of labor that cannot be passed to NewGenIvf’s customers could adversely impact its results of operations. Moreover, the recent escalation of the US-Israel-Iran conflict poses a significant threat to the global economy, primarily through the disruption of oil supplies, with cascading effects on the real estate sector in the UAE and a broad economic downturn across Asia, including Hong Kong and China. As the conflict intensifies around the Strait of Hormuz—through which nearly 20% of the world’s oil passes—global oil prices have surged past $100 per barrel, raising fears of an extended energy crisis. Such increase in oil prices might lead to domino effect on the overall costs of business operation for NewGenIvf and NewGenIvf might not be able to pass the cost pressure to its customers.
ITEM 12. DESCRIPTION OF SECURITIES OTHER THAN EQUITY SECURITIES
No disclosure is required in response to this Item.
PART II
ITEM 13. DEFAULTS, DIVIDEND ARREARAGES AND DELINQUENCIES
Not applicable.
ITEM 14. MATERIAL MODIFICATIONS TO THE RIGHTS OF SECURITY HOLDERS AND USE OF PROCEEDS
Our authorized and issued ordinary shares are divided into Class A Ordinary Shares, Class B Ordinary Shares. Each Class A Ordinary Share is entitled to one (1) vote, while each Class B Ordinary Share is entitled to one hundred (100) votes with all Ordinary Shares voting together as a single class on most matters. Each Class B Ordinary Share is convertible into one Class A Ordinary Share at any time by the holder thereof, while Class A Ordinary Shares are not convertible into Class B Ordinary Shares under any circumstances.
Aside from the above, there have been no modifications to the rights of security holders and there is no other information to disclose in response to this Item.
ITEM 15. CONTROLS AND PROCEDURES
(a) Disclosure Controls and Procedures
Under the supervision and with the participation of our management, including our chief executive officer and chief financial officer, we carried out an evaluation of the effectiveness of our disclosure controls and procedures, which is defined in Rules 13a-15(e) of the Exchange Act, as of December 31, 2025. Based on that evaluation, our chief executive officer and chief financial officer concluded that our disclosure controls and procedures as of December 31, 2025, were not effective.
(b) Management’s Annual Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as defined in Rule 13a-15(f) and 15d-15(f) of the Exchange Act. Our internal control over financial reporting is designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with U.S. GAAP and includes those policies and procedures that (1) pertain to the maintenance of records that in reasonable detail accurately and fairly reflect our transactions; (2) provide reasonable assurance that our transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles and that our receipts and expenditures are being made only in accordance with appropriate authorizations; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of our assets that could have a material effect on our financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. In addition, projections of any evaluation of effectiveness of our internal control over financial reporting to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies and procedures may deteriorate.
Under the supervision of and with the participation of our management, we assessed the effectiveness of our internal control over financial reporting as of December 31, 2025, using the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) in Internal Control-Integrated Framework (2013).
In relation to the examination of our consolidated financial statements presented in this annual report, we have identified a significant weakness in our internal control over financial reporting. This identified material weakness is associated with a lack of adequately skilled staff possessing U.S. GAAP knowledge for financial reporting purposes, thereby affecting the proper adherence to U.S. GAAP and SEC requirements. A comprehensive assessment of our internal control, aimed at identifying and reporting material weaknesses and other deficiencies, was not conducted by our independent registered public accounting firm. Performing such an assessment or having an audit of our internal control over financial reporting might have revealed additional deficiencies.
To address the identified material weakness stemming from the audit of our consolidated financial statements for the year ended December 31, 2025, we intend to implement various measures, including the hiring of additional accounting personnel to enhance the financial reporting function and the establishment of a financial and system control framework. We also intend to initiate regular U.S. GAAP and SEC financial reporting training programs for our accounting and financial personnel. Moreover, we are in the process of developing and implementing a set of policies and procedures for period-end financial reporting. However, we cannot provide assurance that these measures will be entirely effective in remediating the material weakness in a timely manner or at all. To mitigate the risk, the Company engages consultants and professionals to provide advice and assistance.
We qualify as an “emerging growth company” under the JOBS Act. An emerging growth company is entitled to certain reduced reporting and other requirements that are typically applicable to public companies. These provisions include exemption from the auditor attestation requirement under Section 404 of the Sarbanes-Oxley Act of 2002 concerning the assessment of the emerging growth company’s internal control over financial reporting.
(c) Changes in Internal Control Over Financial Reporting
Management is committed to improving the internal controls over financial reporting and will undertake consistent improvements or enhancements on an ongoing basis. Except as described above, there were no changes in our internal controls over financial reporting during the fiscal year ended December 31, 2025 that have materially affected, or are reasonably likely to material affect, our internal control over financial reporting.
ITEM 16. [RESERVED]
Not applicable.
ITEM 16A. AUDIT COMMITTEE FINANCIAL EXPERT
In general, an “audit committee financial expert” within the meaning of Item 407(d)(5) of Regulation S-K, is an individual member of the Audit Committee who:
| ● | understands generally accepted accounting principles and financial statements, | |
| ● | is able to assess the general application of such principles in connection with accounting for estimates, accruals and reserves, |
| ● | has experience preparing, auditing, analyzing or evaluating financial statements comparable to the breadth and complexity to our financial statements, | |
| ● | understands internal controls over financial reporting, and | |
| ● | understands Audit Committee functions. |
An “audit committee financial expert” may acquire the foregoing attributes through:
| ● | education and experience as a principal financial officer, principal accounting officer, controller, public accountant, auditor or person serving similar functions; |
| ● | experience actively supervising a principal financial officer, principal accounting officer, controller, public accountant, auditor or person serving similar functions; experience overseeing or assessing the performance of companies or public accounts with respect to the preparation, auditing or evaluation of financial statements; or | |
| ● | other relevant experience. |
The Board has determined that Mr. Hok Man Jefferson Au qualifies as an audit committee financial expert and has the accounting or financial management expertise as required under Item 407(d)(5)(ii) and (iii) of Regulation S-K. He is independent as that term is used in NASDAQ Marketplace Rule 5605(a)(2).
ITEM 16B. CODE OF ETHICS
A Code of Ethics is a written standard designed to deter wrongdoing and to promote:
| ● | honest and ethical conduct, | |
| ● | full, fair, accurate, timely and understandable disclosure in regulatory filings and public statements, | |
| ● | compliance with applicable laws, rules and regulations, | |
| ● | the prompt reporting violation of the code, and | |
| ● | accountability for adherence to the Code of Business Conduct and Ethics. |
We have adopted a Code of Conduct that complies with the descriptions set forth above for a Code of Ethics. Our Code of Conduct is applicable to all of our employees, and also contains provisions that set forth a higher level of expectations from our leaders. A copy of our Code of Conduct is incorporated by reference as an exhibit to this Report and posted on our website at www.newgenivf.com.
ITEM 16C. PRINCIPAL ACCOUNTANT FEES AND SERVICES
The following table shows the fees that we paid for audit and other services of years ended December 31, 2025 and 2024 provided by J&S Associate PLT respectively.
| Year Ended December 31, 2025 |
Year Ended December 31, 2024 |
|||||||
| Audit Fees | $ | 165,000 | $ | 130,000 | ||||
| Audit-Related Fees | — | — | ||||||
| Tax Fees | — | — | ||||||
| All Other Fees | — | — | ||||||
Audit Fees —This category includes the audit of our annual financial statements and services that are normally provided by the independent auditor in connection with engagements for those fiscal years.
Audit-Related Fees — This category consists of assurance and related services by the independent auditor that are reasonably related to the performance of the audit or review of our financial statements and are not reported above under “Audit Fees.”
Tax Fees — This category consists of professional services rendered by the independent registered public accounting firm for tax compliance and tax advice. The services for the fees disclosed under this category include tax return preparation and technical tax advice.
All Other Fees — This category consists of fees for other miscellaneous items.
ITEM 16D. EXEMPTIONS FROM THE LISTING STANDARDS FOR AUDIT COMMITTEES
Not applicable.
ITEM 16E. PURCHASES OF EQUITY SECURITIES BY THE ISSUER AND AFFILIATED PURCHASERS
There have been no purchases of equity securities required to be disclosed in response to this Item.
ITEM 16F. CHANGE IN REGISTRANT’S CERTIFYING ACCOUNTANT
When Onestop Assurance PAC (“Onestop”) was dismissed effective September 3, 2024, J & S Associate PLT (“J & S”) was appointed as the auditor for the Company on the same day. Since then, J & S has been engaged as the Company’s independent auditor for the years ended December 31, 2023, December 31, 2024 and December 31, 2025.
During the year end December 31, 2025, neither the Company, nor anyone on its behalf, consulted J & S regarding either (i) the application of accounting principles to a specified transaction, either completed or proposed, or the type of audit opinion that might be rendered with respect to the financial statements of the Company and neither a written report was provided to the Company or oral advice was provided that J & S concluded was an important factor considered by the Company in reaching a decision as to the accounting, auditing or financial reporting issue; or (ii) any matter that was either the subject of a “disagreement,” as that term is defined in Item 16F(a)(1)(iv) of Form 20-F and the related instructions to Item 16F of Form 20-F, or a “reportable event,” as that term is described in Item 16F(a)(1)(v) of Form 20-F.
ITEM 16G. CORPORATE GOVERNANCE
Our Class A Ordinary Shares were listed on the NASDAQ Global Market until February 27, 2025 and have then been listed on the Nasdaq Capital Market since February 28, 2025. Our Warrants are listed on the Nasdaq Capital Market. We are a foreign private issuer and a “controlled company” as defined under the Nasdaq rules. Our Chairman of the Board and the Chief Executive Officer, Mr. Wing Fung Alfred Siu and our Director and the Chief Marketing Officer, Ms. Hei Yue Tina Fong, who are husband and wife, jointly own more than 50% of the total voting power of all issued and outstanding ordinary shares. For so long as we remain a foreign private issuer or a “controlled company” under that definition, we are permitted to elect to rely, and may rely, on certain exemptions from certain corporate governance rules, including: an exemption from the rule that a majority of the board of directors must be independent directors; an exemption from the rule that director nominees must be selected or recommended solely by independent directors or by a nominations committee that is comprised entirely of independent directors; an exemption from the rule that our board of directors must have a compensation committee that is comprised solely of independent directors; an exemption from the requirement that an audit committee be comprised of at least three members; an exemption from the requirement that an annual general meeting must be held; an exemption from the requirement that we must obtain shareholder approval prior to a plan or other equity compensation arrangement is established or materially amended; an exemption from the requirement to obtain shareholder approval prior to an issuance of securities in connection with certain acquisition of stock or assets of another company; and an exemption from the requirement to obtain shareholder approval for issuing additional securities exceeding 20% of our outstanding ordinary shares.
We currently rely on home country practice exemption with respect to the requirement of (i) having a nominating committee composed entirely of independent directors; (ii) having a compensation committee composed entirely of independent directors ; (iii) holding an annual meeting of shareholders no later than one year after the end of the issuer’s fiscal year-end; (iv) obtaining shareholder approval prior to a plan or other equity compensation arrangement is established or materially amended; (v) obtaining shareholder approval prior to an issuance of securities in connection with certain acquisition of stock or assets of another company; and (vi) obtaining shareholder approval for issuing additional securities exceeding 20% of our outstanding ordinary shares
ITEM 16H. MINE SAFETY DISCLOSURE
Not applicable.
ITEM 16I. DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS.
Not applicable.
ITEM 16J. INSIDER TRADING POLICIES
We have adopted an insider trading policy governing the purchase, sale, and other dispositions of our securities by directors, senior management, and employees. A copy of the insider trading policy is attached as an exhibit to this Annual Report.
ITEM 16K. CYBERSECURITY
The Company’s executive officers oversee the strategic processes to safeguard data and comply with relevant regulations and has overall responsibility for evaluating cybersecurity risks, as well as related policies and risks in connection with the company’s supply chain, suppliers and other service providers. The Company does not currently engage any assessors, consultants, auditors, or other third parties in connection with any such processes, given the size and scale of the Company, the resources available to it, the anticipated expenditures, and the risks it faces in terms of cybersecurity. The Company’s executive officers are responsible for overseeing and periodically reviewing and identifying risks from cybersecurity threats associated with its use of any third-party service provider.
Since the start of its latest completed fiscal year and up to the date of this Annual Report, the Company is not aware of any risks from cybersecurity threats, including as a result of any previous cybersecurity incidents, have materially affected or are reasonably likely to materially affect the registrant, including its business strategy, results of operations, or financial condition.
The Board is collectively responsible for oversight of risks from cybersecurity threats. The Company’s executive officers oversee the overall processes to safeguard data and comply with relevant regulations and will report material cybersecurity incidents to the board. The Company’s executive officers have limited experience in the area of cybersecurity, but where necessary in the view of the Company’s executive officers, the Company will consult with external advisers to manage and remediate any cybersecurity incidents. For material cybersecurity incidents, the Company’s executive officers will promptly inform, update, and seek the instructions of the board.
PART III
ITEM 17. FINANCIAL STATEMENTS
We have elected to provide financial statements pursuant to Item 18.
ITEM 18. FINANCIAL STATEMENTS
The following financial statements are filed as a part of this Report.
ITEM 19. EXHIBITS
| * | Filed herewith. |
SIGNATURES
The registrant hereby certifies that it meets all of the requirements for filing its annual report on Form 20-F and that it has duly caused and authorized the undersigned to sign this annual report on its behalf.
| Signature | Title | Date | ||
| /s/ Wing Fung Alfred Siu | Chairman, Chief Executive Officer | March 31, 2026 | ||
| Wing Fung Alfred Siu | (Principal Executive Officer and Duly Authorized Officer) | |||
| /s/ Ho Fai Chung | Chief Financial Officer | March 31, 2026 | ||
| Ho Fai Chung | (Principal Financial Officer and Principal Accounting Officer) |
NEWGENIVF LIMITED
F-
 |
J&S ASSOCIATE PLT | |||
| 202206000037 (LLP0033395-LCA) & AF002380 | ||||
| (Registered with PCAOB and MIA) | Tel: | +603-4813 9469 | ||
| B-11-14, Megan Avenue II | Email : | info@jns-associate.com | ||
| 12, Jalan Yap Kwan Seng, 50450, Kuala Lumpur, Malaysia | Website : | jns-associate.com |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To: The Board of Directors and Shareholders of NewGenIvf Group Limited
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheet of NewGenivf Group Limited and its subsidiaries (collectively, the “Company”) as of December 31, 2025 and 2024, and the related consolidated statements of operations and comprehensive income (loss), changes in shareholders’ equity, and cash flows for each of the three years in the period ended December 31, 2025, and the related notes to the consolidated financial statements and schedule (collectively, the financial statements). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2025 and 2024, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2025, in conformity with accounting principles generally accepted in the United States of America.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audit. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audit, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audit included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audit also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audit provides a reasonable basis for our opinion.
/s/ J&S Associate PLT
Certified Public
Accountants
Firm ID: 6743
We have served as the Company’s auditor since 2024. Kuala Lumpur, Malaysia NEWGENIVF GROUP LIMITED AUDITED CONSOLIDATED BALANCE SHEETS
March 31, 2026
F-
AS OF DECEMBER 31, 2025 AND 2024
(Stated in US Dollars)
| December 31, | December 31, | |||||||
| 2025 | 2024 | |||||||
| ASSETS | ||||||||
| Current assets | ||||||||
| Cash and cash equivalents | $ | 758,621 | $ | 457,740 | ||||
| Accounts receivable, net | 166,531 | 49,245 | ||||||
| Inventories | 335,360 | 80,813 | ||||||
| Deposits, prepayments and other receivables, net | 503,588 | 393,152 | ||||||
| Deposit for land acquisition | 4,614,749 | |||||||
| Deposit with a digital asset custodian | 34,987 | 1,000,000 | ||||||
| Deposit with futures broker, net | 59,369 | |||||||
| Receivable from agents, net | 794,116 | 1,191,795 | ||||||
| Deferred offering costs | 116,250 | |||||||
| Total current assets | 7,383,571 | 3,172,745 | ||||||
| Non-current assets | ||||||||
| Plant and equipment, net | 1,789,110 | 273,096 | ||||||
| Right-of-use assets, net | 504,192 | 98,570 | ||||||
| Intangible assets | 21,415,069 | |||||||
| Digital asset, at fair value | 1,630,827 | |||||||
| Other receivable, deposit and other receivables, net | 33,333 | |||||||
| Total non-current assets | 25,339,198 | 404,999 | ||||||
| TOTAL ASSETS | $ | 32,722,769 | $ | 3,577,744 | ||||
| LIABILITIES AND SHAREHOLDERS’ EQUITY | ||||||||
| Current liabilities | ||||||||
| Accounts payable | $ | 779,280 | $ | 1,298,964 | ||||
| Accrued liabilities and other payables | 711,892 | 500,729 | ||||||
| Contract liabilities | 111,981 | 63,489 | ||||||
| Due to related parties | 6,421 | 154,453 | ||||||
| Operating lease liabilities, current | 238,105 | 108,526 | ||||||
| Convertible notes | 256,056 | 82,447 | ||||||
| Embedded derivative liability | 262,616 | |||||||
| Promissory note | 500,000 | |||||||
| Taxes payable | 11,746 | |||||||
| Total current liabilities | 2,366,351 | 2,720,354 | ||||||
| Non-current liabilities | ||||||||
| Operating lease liabilities, non-current | 272,359 | 10,231 | ||||||
| Convertible notes, non-current | 4,097,366 | 2,328,916 | ||||||
| Total non-current liabilities | 4,369,725 | 2,339,147 | ||||||
| Total liabilities | $ | 6,736,076 | $ | 5,059,501 | ||||
| Shareholders’ equity | ||||||||
| $ | $ | |||||||
| Subscription receivable | (204,000 | ) | ||||||
| Additional paid-in capital | 17,881,176 | 122,505 | ||||||
| Retained earnings / (Accumulated deficit) | 8,893,414 | (985,994 | ) | |||||
| Accumulated other comprehensive (loss) income | (143,071 | ) | 18,875 | |||||
| Equity attributable to the shareholders of the Company | 26,631,519 | (1,048,614 | ) | |||||
| Non-controlling interests | (644,826 | ) | (433,143 | ) | ||||
| Total shareholders’ equity | 25,986,693 | (1,481,757 | ) | |||||
| TOTAL LIABILITIES AND SHAREHOLDERS’ EQUITY | $ | 32,722,769 | $ | 3,577,744 | ||||
| * | The shares as presented have been adjusted retrospectively for Reverse Share Splits effected in February 2025 of 1 share for every 20 existing shares issued, in May 2025 of 1 share for every 10 existing share issued, in August 2025 of 1 share for every 5 shares issued, in December 2025 of 1 share for every 5 shares issued, in January 2026 of 1 share for every 3 shares issued and in March 2026 of 1 share for every 4 shares issued respectively. |
The accompanying notes are an integral part of these consolidated financial statements.
F-
NEWGENIVF GROUP LIMITED
AUDITED CONSOLIDATED STATEMENTS OF OPERATIONS AND
COMPREHENSIVE INCOME (LOSS)
FOR THE YEARS ENDED DECEMBER 31, 2025, 2024 AND 2023
(Stated in US Dollars)
| December 31, | ||||||||||||
| 2025 | 2024 | 2023 | ||||||||||
| Revenues | 4,726,433 | $ | 5,433,375 | $ | 5,136,153 | |||||||
| Cost of revenues | (3,770,587 | ) | (3,606,481 | ) | (3,454,368 | ) | ||||||
| Gross profit | 955,846 | 1,826,894 | 1,681,785 | |||||||||
| Operating expenses | ||||||||||||
| Selling and marketing expenses | (1,188,711 | ) | (206,314 | ) | (18,030 | ) | ||||||
| General and administrative expenses | (10,493,596 | ) | (2,781,075 | ) | (1,621,513 | ) | ||||||
| Total operating expenses | (11,682,307 | ) | (2,987,389 | ) | (1,639,543 | ) | ||||||
| Operating (loss) income | (10,726,461 | ) | (1,160,495 | ) | 42,242 | |||||||
| Other income (expenses), net | ||||||||||||
| Other income, net | (8,099 | ) | 971,391 | 111,837 | ||||||||
| Bargain purchase gain | 21,653,835 | |||||||||||
| Loss on digital assets, net | (332,480 | ) | ||||||||||
| Loss on financial assets, net | (700,631 | ) | ||||||||||
| Gain on embedded derivative | 456,731 | |||||||||||
| Interest income | 2,251 | 6,953 | 518 | |||||||||
| Interest expense | (616,423 | ) | (778,656 | ) | (46,179 | ) | ||||||
| Total other income (expenses), net | 20,455,184 | 199,688 | 66,176 | |||||||||
| Income (Loss) before taxes | 9,728,723 | (960,807 | ) | 108,418 | ||||||||
| Tax income (expense) | 486,706 | |||||||||||
| Net income (loss) | 9,728,723 | (474,101 | ) | 108,418 | ||||||||
| Less: net (loss) income attributable to non-controlling interests | (150,685 | ) | 50,542 | (21,775 | ) | |||||||
| Net income (loss) attributable to the shareholders of the Company | 9,879,408 | $ | (524,643 | ) | $ | 130,193 | ||||||
| Other comprehensive (loss) income | ||||||||||||
| Foreign currency translation adjustment | (222,944 | ) | 32,529 | (22,704 | ) | |||||||
| Total comprehensive (loss) income | 9,505,779 | (441,572 | ) | 85,714 | ||||||||
| Less: total comprehensive (loss) income attributable to non-controlling interests | (211,683 | ) | 56,908 | (27,621 | ) | |||||||
| Total comprehensive income (loss) attributable to the shareholders of the Company | 9,717,462 | $ | (498,480 | ) | $ | 113,335 | ||||||
| Earnings per share – basic | 214.73 | $ | (4,408.76 | ) | $ | 13,019.30 | ||||||
| – diluted | 3.04 | (4,408.76 | ) | 13,019.30 | ||||||||
| Weighted average shares outstanding *- basic | 46,009 | 119 | 10 | |||||||||
| - diluted | 3,250,020 | 119 | 10 | |||||||||
| * | The shares as presented have been adjusted retrospectively for Reverse Share Splits effected in February 2025 of 1 share for every 20 existing shares issued, in May 2025 of 1 share for every 10 existing share issued, in August 2025 of 1 share for every 5 shares issued, in December 2025 of 1 share for every 5 shares issued, in January 26, 2026 of 1 share for every 3 shares issued and in March 2026 of 1 share for every 4 shares issued respectively. |
The accompanying notes are an integral part of these consolidated financial statements.
F-
NEWGENIVF GROUP LIMITED
AUDITED CONSOLIDATED STATEMENTS OF CHANGES IN SHAREHOLDERS’ EQUITY
FOR THE YEARS ENDED DECEMBER 31, 2025, 2024 AND 2023
(Stated in US Dollars)
| Number of shares* |
Ordinary shares |
Subscription receivable |
Additional paid-in capital |
Retained earnings (Accumulated deficit) |
Accumulated other comprehensive income/(loss) |
Total attributable to the shareholders of the Company |
Non- controlling interests |
Total | ||||||||||||||||||||||||||||
| Balance, January 1, 2023 | 167 | $ | (319,872 | ) | $ | 1,464,959 | $ | (591,544 | ) | 9,570 | 563,113 | $ | (462,430 | ) | $ | 100,683 | ||||||||||||||||||||
| Net loss | — | 130,193 | 130,193 | (21,775 | ) | 108,418 | ||||||||||||||||||||||||||||||
| Foreign currency translation adjustment | — | (16,858 | ) | (16,858 | ) | (5,846 | ) | (22,704 | ) | |||||||||||||||||||||||||||
| Settlement of Subscription receivable | — | 192,308 | 192,308 | 192,308 | ||||||||||||||||||||||||||||||||
| Issuance of shares | 2 | 2,866,856 | 2,866,856 | 2,866,856 | ||||||||||||||||||||||||||||||||
| Balance, December 31, 2023 | 169 | $ | $ | (127,564 | ) | $ | 4,331,815 | $ | (461,351 | ) | $ | (7,288 | ) | $ | 3,735,612 | $ | (490,051 | ) | $ | 3,245,561 | ||||||||||||||||
| Net loss | — | (524,643 | ) | (524,643 | ) | 50,542 | (474,101 | ) | ||||||||||||||||||||||||||||
| Foreign currency translation adjustment | — | 26,163 | 26,163 | 6,366 | 32,529 | |||||||||||||||||||||||||||||||
| Reverse Capitalization | — | (6,028,690 | ) | (6,028,690 | ) | (6,028,690 | ) | |||||||||||||||||||||||||||||
| Settlement of Subscription receivable | — | 127,564 | 127,564 | 127,564 | ||||||||||||||||||||||||||||||||
| Remeasurement of share based compensation | — | (2,766,856 | ) | (2,766,856 | ) | (2,766,856 | ) | |||||||||||||||||||||||||||||
| Issuance of shares under ELOC/Note Conversion arrangement | 211 | (204,000 | ) | 4,586,236 | 4,382,236 | 4,382,236 | ||||||||||||||||||||||||||||||
| Balance, December 31, 2024 | 380 | $ | $ | (204,000 | ) | $ | 122,505 | $ | (985,994 | ) | $ | 18,875 | $ | (1,048,614 | ) | $ | (433,143 | ) | $ | (1,481,757 | ) | |||||||||||||||
| Net Profit | — | 9,879,408 | 9,879,408 | (150,685 | ) | 9,728,723 | ||||||||||||||||||||||||||||||
| Foreign currency translation adjustment | — | (161,946 | ) | (161,946 | ) | (60,998 | ) | (222,944 | ) | |||||||||||||||||||||||||||
| Issuance of shares under ELOC | 141,661 | 12,620,494 | 12,620,494 | 12,620,494 | ||||||||||||||||||||||||||||||||
| Issuance of shares under conversion | 58,424 | 204,000 | 4,860,427 | 5,064,427 | 5,064,427 | |||||||||||||||||||||||||||||||
| Issuance of shares for business acquisition | 42 | 257,500 | 257,500 | 257,500 | ||||||||||||||||||||||||||||||||
| Issuance of shares for share-based compensation to consultant | 50 | 20,250 | 20,250 | 20,250 | ||||||||||||||||||||||||||||||||
| Issuance of shares for warrant exercise | 40,065 | |||||||||||||||||||||||||||||||||||
| Balance, December 31, 2025 | 240,622 | 17,881,176 | 8,893,414 | (143,071 | ) | 26,631,519 | (644,826 | ) | 25,986,693 | |||||||||||||||||||||||||||
| * | The shares as presented have been adjusted retrospectively for Reverse Share Splits effected in February 2025 of 1 share for every 20 existing shares issued, in May 2025 of 1 share for every 10 existing share issued, in August 2025 of 1 share for every 5 shares issued, in December 2025 of 1 share for every 5 shares issued, in January 2026 of 1 share for every 3 shares issued and in March 2026 of 1 share for every 4 shares issued respectively. |
The accompanying notes are an integral part of these consolidated financial statements.
F-
NEWGENIVF GROUP LIMITED
CONSOLIDATED STATEMENTS OF CASH FLOWS
FOR THE YEARS ENDED DECEMBER 31, 2025, 2024 AND 2023
(Stated in US Dollars)
| December 31, | ||||||||||||
| 2025 | 2024 | 2023 | ||||||||||
| CASH FLOWS FROM OPERATING ACTIVITIES | ||||||||||||
| Net income (loss) | 9,728,723 | $ | (474,101 | ) | $ | 108,418 | ||||||
| Adjustments to reconcile net income (loss) to net cash provided by operating activities: | ||||||||||||
| Depreciation of plant and equipment | 188,109 | 19,502 | 31,173 | |||||||||
| Amortization of right-of-use assets | 311,390 | 186,762 | 198,535 | |||||||||
| Loss on digital assets, fair value | 332,480 | |||||||||||
| Loss on financial assets | 700,631 | |||||||||||
| Amortisation of share-based compensation expense | 53,583 | 33,334 | ||||||||||
| Non cash discount on convertible notes | 402,500 | |||||||||||
| Provision of expected credit loss allowance | 625 | |||||||||||
| Interest expense | 616,423 | 778,656 | 46,179 | |||||||||
| Impairment on receivable from agency | 818,780 | |||||||||||
| Loss on disposal of subsidiary | 47,702 | |||||||||||
| Waiver of related party balance | (88,151 | ) | ||||||||||
| Gain on lease modification | (13,092 | ) | ||||||||||
| Gain on promissory note | (953,861 | ) | ||||||||||
| Gain on bargain purchase | (21,653,835 | ) | ||||||||||
| Embedded derivative gain | (456,731 | ) | ||||||||||
| Legal and professional fee | 27,320 | |||||||||||
| (Gain on) Provision for income taxes | (486,706 | ) | ||||||||||
| Changes in operating assets and liabilities: | ||||||||||||
| Accounts receivable | (117,285 | ) | (39,871 | ) | 1,166 | |||||||
| Inventories | (254,547 | ) | 45,451 | (80,665 | ) | |||||||
| Deposit and other receivables, net | (110,436 | ) | (2,000,851 | ) | (448,266 | ) | ||||||
| Amount due from agency | (421,101 | ) | ||||||||||
| Deferred issuance cost | (116,250 | ) | ||||||||||
| Accounts payable | (519,684 | ) | 1,126,338 | 71,362 | ||||||||
| Accrued liabilities and other payables | 211,161 | (6,750,587 | ) | (51,167 | ) | |||||||
| Contract liabilities | 48,492 | 55,552 | (1,352,231 | ) | ||||||||
| Operating lease liabilities | (321,915 | ) | (204,846 | ) | (230,433 | ) | ||||||
| Tax paid | (11,746 | ) | 11,746 | |||||||||
| Net cash used in operating activities | (10,926,056 | ) | (8,264,074 | ) | (1,766,135 | ) | ||||||
| CASH FLOWS FROM INVESTING ACTIVITIES | ||||||||||||
| Purchase of plant and equipment | (135,025 | ) | (53,045 | ) | (69,848 | ) | ||||||
| Investment in financial asset | (760,000 | ) | ||||||||||
| Investment in digital assets | (1,000,000 | ) | ||||||||||
| Deposit paid for acquisition of land | (4,614,749 | ) | ||||||||||
| Acquisition of businesses | (1,050,000 | ) | ||||||||||
| Net cash used in investing activities | (7,559,774 | ) | (53,045 | ) | (69,848 | ) | ||||||
| CASH FLOWS FROM FINANCING ACTIVITIES | ||||||||||||
| Amount due from A SPAC I | 140,000 | (140,000 | ) | |||||||||
| Finance lease | (6,446 | ) | (9,317 | ) | ||||||||
| Settlement of Promissory note | (500,000 | ) | ||||||||||
| Issuance of convertible notes, net | 6,842,500 | 8,583,597 | ||||||||||
| Issuance of shares under ELOC | 12,620,496 | 192,308 | ||||||||||
| Interest paid | (26,206 | ) | (677,663 | ) | (24,704 | ) | ||||||
| Amount with related parties | (148,032 | ) | 508,738 | 1,863,206 | ||||||||
| Subscription receivable | 204,000 | 127,564 | ||||||||||
| Net cash provided by financing activities | 18,992,758 | 8,675,790 | 1,881,493 | |||||||||
| Net increase/(decrease) in cash and cash equivalents | 506,928 | 358,671 | 45,510 | |||||||||
| Effect of foreign currency translation on cash and cash equivalents | (206,047 | ) | 44,965 | (18,962 | ) | |||||||
| Cash and cash equivalents, beginning of year | 457,740 | 54,104 | 27,556 | |||||||||
| Cash and cash equivalents, end of year | 758,621 | $ | 457,740 | $ | 54,104 | |||||||
| Supplementary cash flow information: | ||||||||||||
| Taxes paid | $ | $ | ||||||||||
| Interest paid | (26,206 | ) | $ | (677,663 | ) | $ | (24,704 | ) | ||||
During the year ended December 31, 2025, no cash was exchanged in respect of these transactions:
| a. | $4,860,427 of convertible debt was converted into equity. |
| b. | $20,250 was credited to APIC in relation to shares issued for share-based compensation |
| c. | $257,500 was credited to APIC in relation to shares issued for acquisition of a business during the year. |
During the year ended December 31, 2024, no cash was exchanged in respect of these transactions:
| a. | $2,650,000 of convertible debt was converted into equity. |
| b. | Reversal arising from remeasurement of share-based compensation within prepayment amounting to $2,739,856. |
The accompanying notes are an integral part of these consolidated financial statements.
F-
NEWGENIVF GROUP LIMITED
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
FOR THE YEARS ENDED DECEMBER 31, 2025 AND DECEMBER 31, 2024
(Stated in US Dollars)
NOTE 1 — ORGANIZATION AND PRINCIPAL ACTIVITIES
Prior to the Business Combination, on April 29, 2021, A SPAC I Acquisition Corp. (“ASCA”), was incorporated as a British Virgin Islands business company, specifically a blank check company formed for the purpose of effecting a merger, share exchange, asset acquisition, share purchase, recapitalization, reorganization or similar business combination with one or more target businesses.
The Business Combination
On February 15, 2023, ASCA entered into the Merger Agreement (as amended on June 12, 2023 and December 6, 2023, the “Merger Agreement,” and the transactions contemplated thereunder, the “Business Combination”) with A SPAC I Mini Acquisition Corp., Merger Sub, NewGenIvf Limited, a Cayman Islands exempted company (“Legacy NewGenIvf”) and certain shareholders of Legacy NewGenIvf. Pursuant to the Merger Agreement, the Business Combination was effected in two steps: (i) ASCA was reincorporated to the British Virgin Islands by merging with and into A SPAC I Mini Acquisition Corp. (such transaction, the “Reincorporation Merger”) and then the listed company was renamed as NewGenIvf Group Limited; and (ii) Merger Sub merged with and into Legacy NewGenIvf, resulting in Legacy NewGenIvf being a wholly-owned subsidiary of the Company (such second step in isolation, the “Acquisition Merger”). The surviving entity of the Business Combination, together with its subsidiaries is referred to in this prospectus as “NewGenIvf,” the “Company,” “we,” “our,” or “us,” unless the context otherwise requires.
On June 12, 2023, the parties to the Merger Agreement entered into the First Amendment to Merger Agreement (the “First Amendment”), pursuant to which Legacy NewGenIvf agreed to provide non-interest bearing loans in an aggregate principal amount of up to $560,000 (the “Loan”) to ASCA to fund any amount that would be required in order to further extend the period of time available for ASCA to consummate a business combination and for ASCA’s working capital, payment of professional, administrative and operational fees and expenses, and other purposes as mutually agreed by ASCA and Legacy NewGenIvf. Such loans were to become repayable upon the closing of the Acquisition Merger. In addition, pursuant to the First Amendment, subject to receipt of at least $140,000 as part of the Loan from Legacy NewGenIvf, ASCA agreed to waive its termination rights and the right to receive any break-up fee due to Legacy NewGenIvf’s failure to deliver audited financial statements by no later than February 28, 2023.
On December 6, 2023, the parties to the Merger Agreement entered into the Second Amendment to the Merger Agreement (the “Second Amendment”) which amended and modified the Merger Agreement to, among other things, (i) reduce the size of NewGenIvf’s board of directors following the consummation of the Business Combination to five (5) directors, two (2) of whom would be executive directors designated by NewGenIvf and three (3) of whom will be designated by NewGenIvf to serve as independent directors in accordance with Nasdaq requirements, (ii) provide for the conversion of NewGenIvf shares issued by NewGenIvf following the original date of the Merger Agreement into Class A Ordinary Shares in connection with the Acquisition Merger, and (iii) remove the condition that ASCA have in excess of $5,000,000 in net tangible assets immediately after the consummation of the Business Combination.
On April 3, 2024, the Business Combination was consummated with the Company as the surviving entity.
F-
The following is an organization chart of the Company and its subsidiaries as of December 31, 2025:

The Company’s subsidiaries are detailed in the table as follows:
| Name | Background | Ownership % | Principal activity | |||
| NewGenivf Limited | ● A Cayman Islands company ● Incorporated on 16 January, 2019 | 100% | Investment holding | |||
| FFPGS (HK) Limited | ● A Hong Kong company ● Incorporated on December 19, 2019 | 100% | Marketing and administrative services | |||
| Well Image Limited | ● A Hong Kong company ● Incorporated on July 11, 2008 | 100% | Investment holding | |||
| Med Holdings Limited (“Med Holdings”) (Note) | ● A Thailand company ● Incorporated on January 21, 2015 | 49%* | Investment holding | |||
| First Fertility PGS Center Limited (“FFC”) (Note) | ● A Thailand company ● Incorporated on March 6, 2014 | 74% | Provision of IVF treatment | |||
| First Fertility Phnom Penh Limited (“FFPP”) | ● A Cambodia company ● Incorporated on August 10, 2015 | 100% | Provision of IVF treatment | |||
| Bi Clinic Ltd (“FFBi”) | ● A Kyrgyzstan company ● Incorporated on December 16, 2021 ● Acquired on December 17, 2024 | 100% | Provision of IVF treatment, surrogacy and ancillary caring services | |||
| MicroSort Lab Services LLC | ● A US company ● Incorporated on June 16, 2025 | 100% | Specialized applications, in provision of IVF treatment | |||
| NewGenDigital Limited | ● A BVI company ● Incorporated on January 21, 2025 | 100% | Investment Holding | |||
| NewGenProperty Limited | ● A BVI company ● Incorporated on September 18, 2025 | 100% | Investment Holding | |||
| HyFi Innovations Limited | ● A Hong Kong company ● Incorporated on October 20, 2025 | 100% | Tokenization Service | |||
| Incorporated subsequent to the financial year end: | ||||||
| NewGenBiz Limited | ● A BVI company ● Incorporated on January 13, 2026 | 100% | Investment Holding | |||
| NewGenOman Limited | ● A BVI company ● Incorporated on February 2, 2026 | 100% | Investment Holding | |||
| Alfred Siu Charitable Foundation Limited | ● A Hong Kong Foundation ● Incorporated on February 27, 2026 | 100% | Charitable activities | |||
| The following subsidiary was disposed during the year | ||||||
| 深圳前海豐泰仁匯健康科技有限公司 (Shenzhen Qianhai Fengtai Renhui Health Technology Co., Ltd.) | ● A PRC China company ● Disposed on July 29, 2025 | 100% | Marketing services | |||
| * | Where less than 50% of the equity of an investee is held, the Company (through its subsidiaries) holds significantly more voting rights than any other vote holder or organized company of vote holders. An assessment has been made, taking into account all the factors relevant to the relationship with the investee, to ascertain control has been established and the investee should be consolidated as a subsidiary of the Company. |
F-
Note:
According to Thailand’s Foreign Business Act (the “FBA”), the majority shareholdings of limited company incorporated in Thailand is required to be owned by Thai nationals.
With reference to the capital structure and voting rights structure of ordinary shares and preference shares (the “Share Structure”) of Med Holdings and FFC, all the preference share capital shall be owned by a Thai national. No preference shares, however, have been issued to date. The ordinary shares and preference shares have the same rights and status in all respects except for the distribution of profits by way of dividends with details as follow:
| (a) | Dividends from profits of Med Holdings and FFC shall be allocated to the holders of preference shares at a rate fixed from time to time by the board of directors prior to allocating to the holders of ordinary shares. In any event, such dividends to be allocated to the holders of preference shares shall not exceed 15% of the total amount of dividends declared from time to time; |
| (b) | After allocation of dividends as per (a) above, the rest of the dividends shall be distributed equally amongst the holders of ordinary shares according to their shareholding ratio; |
| (c) | The holders of preferred shares shall be entitled to dividends only in respect of the years for which the Company has declared a dividend payment, and there shall be no cumulative dividends; and |
| (d) | Dividends allocated to the holders of preferred shares in each year shall be limited at the rate as stated in (a) only. No additional dividends shall be paid to the holders of preferred shares. |
Based upon the management’s judgement on the Shares Structure, as the Company is able to exercise majority voting power in any board meeting, the Company accounts for Med Holdings and FFC as subsidiaries on the ground that the Company is able to control Med Holdings and FFC by exercising its majority voting power in any board meetings.
On April 17, 2024, the Company entered into a non-binding term sheet (the “Non-Binding Term Sheet”) with European Wellness Investment Holdings Limited (“EWIHL”) for (i) the potential acquisition of the entire equity interest of EWIHL by the Company for a consideration of US$268,000,000 to be payable by issuing 53,600,000 ordinary shares of the of the Company to the shareholder(s) of EWIHL or its associate and (ii) the fund-raising activity by the Company from public or private shareholders, and in a form mutually acceptable to the parties, including structured equity investment for up to US$30 million. On December 11, 2024, NewGenIvf announced its entry into a binding term sheet with European Wellness Investment Holdings Limited (“EWIHL”) for the above proposed reverse merger, completion of which was subject to, among other conditions, the completion of due diligence, the negotiation of a definitive agreement, and obtaining adequate financing. On March 31, 2025, the Company terminated the binding term sheet with healthcare company EWIHL regarding their previously announced reverse merger transaction on the grounds of non fulfilment of certain criteria as contained in the term sheet.
On May 24, 2024, the Company received a deficiency letter from the Listing Qualifications Department of The Nasdaq Stock Market LLC (“Nasdaq”) notifying the Company of its non-compliance with two (2) listing requirements for continued listing on Nasdaq pursuant to Nasdaq Listing Rules. On November 21, 2024, a delisting notice was received from the continued non-compliance. The Company had filed to appeal the delisting determination and undertook several strategic actions to regain compliance with Nasdaq’s listing require. On February 27, 2025, the Company received approval for the transfer of the Company’s securities from the Nasdaq Global Market to the Nasdaq Capital Market and on March 10, 2025 met its compliance with the listing requirements thereof.
On June 3, 2024, the Company announced the execution of a non-binding term sheet (the “Term Sheet”) regarding a proposed reverse merger (the “Proposed Transaction”) with pharmaceutical company COVIRIX Medical Pty Ltd (“COVIRIX”). The consideration was to be settled by way of the issuance of issue 102,890,000 of its ordinary shares to the shareholder(s) of COVIRIX or their respective nominees (the “COVIRIX Shareholders”) in exchange for 100% equity interest of COVIRIX, at a deemed price per share of US$6, representing an aggregate amount of US$617,340,000. Simultaneously, it is proposed that COVIRIX undertakes to introduce investors to raise US$6 million at US$6 per share for NIVF, in a form mutually acceptable to both NewGen and COVIRIX. Following stockholder approval of the Proposed Transaction, COVIRIX Shareholders are expected to hold approximately 85.8% equity interest in NewGen. However, on September 21, 2024, COVIRIX withdrew from the Proposed Transaction, as such the Proposed Transaction was terminated with no cost to the Company.
On August 7, 2024, the Company entered into a Securities Purchase Agreement with certain investors named therein (collectively, the “Buyers”), pursuant to which, amongst other things: (i) the Company agreed to sell, at an initial closing with JAK Opportunities VI LLC (“JAK” and such initial closing, the “Initial Closing”), pursuant to which the Company agreed to sell to JAK (a) a senior convertible note (the “Initial Note”) in the aggregate original principal amount not exceeding $1,100,000), and which terms are further set forth below under the subheading “(ii) Initial Closing with JAK”), (b) a warrant to purchase 1,325,301 Class A Ordinary Shares of the Company, no par value (“Class A Shares” and such warrant, the Series A Warrant), and (c) a warrant to purchase 180,722 Class B Ordinary Shares of the Company, no par value (“Class B Shares” and such warrant, the Series B Warrant, and the Series B Warrants, together with the Series A Warrants, the “Warrants”); and (ii) the Company may require each Buyer (or each Buyer may require the Company, as applicable) to participate in the sale of (a) one or more additional convertible notes (which aggregate original principal amount for all additional convertible notes shall not exceed $9,500,000) (the “Additional Notes,” and, together with the Initial Note, the “Notes”).
F-
On August 12, 2024, the Company and JAK consummated the Initial Closing. The Initial Note sold to JAK in connection with the Securities Purchase Agreement bears an interest rate of 14.75% per annum and is convertible into the Company’s Class A Shares as follows: the Conversion Amount (as defined below) into validly issued, fully paid and non-assessable Class A at the Conversion Rate determined by dividing the aggregate of the principal sum plus the interest rates (including late interest charges, if any) and the Make-Whole Amount, if any, by conversion price of $0.83.
At the Initial Closing, the Company also sold to JAK a Series A Warrant to purchase 1,325,301 Class A Shares and a Series B Warrant to purchase 180,722 Class B Shares subject to Reverse Stock Split adjustments
Additionally, in connection with the Securities Purchase Agreement, the Company entered into amendment and exchange agreements with certain holders of its convertible promissory notes (the “Existing Notes” and each of such amendment and exchange agreements, “Amendment and Exchange Agreement”), pursuant to which the Company will exchange the Existing Notes by issuing, among other things, (i) senior convertible notes in the aggregate principal amount of $2,700,000 (the “Exchange Notes”) and (b) a series of warrants to initially acquire up to a certain number of ordinary shares to the holders of the Existing Notes set forth therein or in the Amendment and Exchange Agreement (the “Exchange Warrants”)
On August 28, 2024, the Company consummated the second tranche of its debt financing under the terms of the Securities Purchase Agreement. At the closing of the second tranche, the Company sold to JAK Opportunities VI LLC (“JAK”) a senior convertible note (the “Note”) in the principal amount of $500,000.
On November 11, 2024, the Company consummated the third tranche of its debt financing under the terms of the Securities Purchase Agreement (“SPA”) referenced in the current report on Form 6-K filed with the United States Securities and Exchange Commission (the “SEC”) on August 16, 2024. The Form 6-K filed with the SEC on August 16, 2024 is incorporated by reference herein. Pursuant to the terms of the SPA, the Company may elect at the second additional mandatory closing to sell and the institutional investor party to the SPA shall be required to purchases, subject to certain conditions, an additional note (“Second Additional Mandatory Note”) in the principal amount of $1,500,000, after the effective date of the Registration Statement (as defined in the SPA). The sale of the Second Additional Mandatory Note resulted in $1,395,000 of gross proceeds to the company before fees and expenses. The Notes bears an interest rate of 14.75% per annum and may be adjustable from time to time pursuant to its terms, with maturity at the 4.5 years anniversary of the date of issuance, subject to extension at the option of the holders in certain circumstances. The Second Additional Note are convertible at any time, at an initial conversion price of $0.658.
On November. 18, 2024, the Company entered into a binding term sheet (the “Term Sheet”) with White Lion Capital, LLC, (“White Lion”) a California-based institutional investor focused on high-growth, early-stage public companies, setting out the principal terms and conditions for a $100 million equity line of credit, expandable to $500 million. Pursuant to the Term Sheet, NewGen will have the option, but not the obligation, to sell to White Lion up to $100.0 million in shares of common stock over an initial 36-month period, with the potential to increase to $300.0 million upon substantial M&A or merger activity, and further to $500.0 million after $250.0 million has been drawn.
On February 28, 2025, the Company completed its acquisition of the MicroSort business from Genetics & IVF Institute, Inc. (“GIVF”). Pursuant to a Purchase Agreement dated January 21, 2025 between the Company and GIVF (“Purchase Agreement”), the Company purchased all of the Assets (as defined in the Purchase Agreement) and IP Licenses (as defined in the Purchase Agreement) relating to the MicroSort business from GIVF for a total cash consideration of for US$5 million, which was satisfied through a combination of US$750,000 in cash and the issuance of 2,500,000 ordinary shares (as was adjusted to 42 ordinary shares after all the Reverse Stock Splits done as of this Report date at date of completion) at a deemed value of US$1.70 per share.
On February 18, 2025, the Company entered into a cooperation agreement with FERTILITY GROUP LLC (“BOBCARE”) to jointly develop fertility services in the Kyrgyzstan market. The collaboration aims to combine NewGen’s technical expertise in fertility treatments with BOBCARE’s market resources in the region. This strategic partnership is anticipated to contribute to NewGen’s market presence over the next three years, improving the competitive position of both parties in the Kyrgyzstan market. Both companies will work together on several initiatives, including enhancing clinical protocols and patient management systems, knowledge exchange between fertility specialists, coordinated marketing and brand-building activities in Kyrgyzstan, and developing specialized fertility treatment options for the regional market.
F-
On March 31, 2025, the Company’s Board approved certain amendments to its Share Incentive Plan of 2024, the awards of which are valid for a period of 10 years from March 28, 2025, whereby the maximum aggregate number of shares with respect to which Awards may be granted under the Plan shall be 1,054,260 Shares (351 ordinary shares as adjusted for reverse stock splits in May 2025, August 2025, December 2025, January 2026 and March 2026) and has been increased in March 2026 by the Board to 454,812 ordinary shares (113,703 ordinary shares as adjusted by reverse stock split in 16 March 2026) of the Company as of the date of this Report. which may be increased from time to time as determined by the Board or Committee of the Board, in an amount equal to 20% of the then outstanding ordinary shares of the Company at the time of such increase. Shares may be made available from Shares held in treasury or authorized but unissued shares of the Company not reserved for any other purpose. To date, no shares have been awarded under this Plan.
On April 1, 2025, the Company entered into a new Securities Purchase Agreement (“2025 Securities Purchase Agreement”) with JAK, pursuant to which, amongst other things: (i) the Company agreed to sell, at an initial closing, a senior convertible note in the aggregate original principal amount not exceeding $3,200,000, convertible into Class A Ordinary Shares pursuant to its terms; and (ii) the Company may require JAK (or JAK may require the Company, as applicable) to participate in the sale of one or more additional convertible notes (which aggregate original principal amount for all additional convertible notes shall not exceed $25,600,000).
The 2025 Securities Purchase Agreement is filed as Exhibit 4.1 of the Company’s current report on Form 6-K dated April 3, 2025 and is incorporated by reference herein. The first tranche of $3,200,000 was received on June 3, 2025
On April 2, 2025 and July 16, 2025, the Company consummated the fourth and fifth tranche of its debt financing under the terms of its existing Securities Purchase Agreement with the investor, At the closing of the fourth and fifth tranche, the Company sold to the investor a senior convertible note in the principal amount of $2,000,000 each. The Note bears an interest rate of 14.75% per annum and may be adjustable from time to time pursuant to its terms. The funding from these arrangements with the investor is intended to be used to finance the establishment of a fertility clinic in Dubai.
On July 24, 2025, NewGenIvf signed a Purchase Agreement to acquire the Nodexus business which included certain tangible assets, company domain and six patents from Nodexus Inc, a distressed company, for a consideration of $300,000. The technology associated with the patents is related to cell sorting to be performed via a device called NX One and could be applied in different fields including In Vitro Fertilization. The Patents would allow NewGenIvf to penetrate the IVF market in the United States by leasing the NX One to clinics that provide IVF treatments in the United States. The clinics would utilize the NX One to perform gender selection services and detect potential genetic diseases during the IVF process and charge a service fee from customers.
On October 6, 2025, NewGenProperty Limited (“NewGenProperty”), a wholly owned subsidiary of the Company, entered into a joint venture agreement with BNW to develop the Plot. Under the terms of the joint venture agreement, NewGenProperty shall hold 60% of the joint venture, and be responsible for funding 36% of the purchase price of the Plot to the Master Developer, while BNW shall be responsible for all the other cashflow and financing requirements of the project. In the event that BNW pays the balance of the Purchase Price of the Plot to the Master Developer, NewGenProperty shall reimburse BNW in accordance with the provisions of the joint venture agreement. NewGenProperty’s entitlement is an amount equivalent to 36% of the Gross Sales Revenue or Gross Sellable Area generated from the project, with deductions including (i) up to 10% of External and Internal Real Estate Agent and Brokerage Fees; (ii) 2% of Gross Sales Revenue for BNW’s Marketing Fees; and (iii) any amounts paid by the Developer towards the balance of the Purchase Price of the Plot on behalf of NewGenProperty.
On October 15, 2025, November 4, 2025 and January 22, 2026, the Company entered into three Convertible Promissory notes with Vanguish Funding Group, in amount to $257,000, 157,000 and $107,000 respectively and also November 12, 2025, the Company entered another Convertible Promissory notes with Labrys Fund II, L.P. Note, totaling to $250,000.
F-
On November 3, 2025, the Company announced the execution of a non-binding term sheet regarding a proposed reverse merger (the “Proposed RTO”) with SAXA, Inc. (“SAXA”), an international holdings company focused on mining and processing operations. On November 5, 2025, the Company announced the signing of an service agreement with SAXA to act as the worldwide agent for the tokenization of an initial US$100 million tranche of SAXA’s gold-backed assets (“Tokenization Engagement”). The Tokenization Engagement was intended to be the first part of a two-part transaction culminating in the Proposed RTO. The Proposed RTO was subject to completion of due diligence, and negotiation of a definitive agreement, among other conditions. As the conditions set out in the term sheet, in particular, with respect to due diligence and execution of definitive agreements, had not been satisfied by SAXA by the later of: (i) the end of the 30-day period as set out in the term sheet; or (ii) the tentative timetable with completion by January 28, 2026 as set out in the term sheet, the Company will not be proceeding with the Proposed RTO, or the Tokenization Engagement. No material agreements have been entered into between SAXA and the Company, and the Company is not pursuing any transaction with SAXA.
On November 10, 2025, Board of Directors (the “Board”) has authorized a share repurchase program under which the Company may repurchase up to US$2 million of its outstanding Class A ordinary shares over the next 24 months (“Share Repurchase Program”). The Company plans to adopt and implement this Share Repurchase Program in accordance with applicable rules and requirements under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), and the Company’s insider trading policy.
Mr. Yip Eng Jeremy Foo, a director of NewGenIvf Group Limited (the “Company”), has resigned from the Board of Directors of the Company, the Audit Committee of the Company, and the Compensation Committee of the Company due to personal reasons, effective April 4, 2025. Mr. Foo’s resignation did not result from any disagreement with the Board or the Company on any matter relating to the Company’s operations, policies or practices.
Ms. Florianna Ann Chi Wan Chan was appointed as a director of the Company, effective April 15, 2025, to fill the vacancy on the Audit Committee and the Compensation Committee created by Mr. Foo’s resignation. Ms Chan has over 20 years of experience in project management, real estate development, and luxury hospitality, and is a proven leader in driving growth and operational excellence. Since 2015, Ms. Chan has led Lab Concept Company Limited, a subsidiary of The Lane Crawford Joyce Group, where she successfully restructured operations, spearheaded rebranding, and digitized processes, achieving significant revenue growth. Previously, she held senior roles at VCC Company Limited, Eton Properties, and Crown Macau, excelling in real estate development, marketing, and VIP services. She holds a Bachelor of Hospitality Management from Central Queensland University in Australia and an Advanced Diploma from William Angliss Institute of TAFE. Fluent in Cantonese, Mandarin, and English, Ms. Chan brings a deep understanding of Asia Pacific markets and a strategic vision that will drive the Company’s continued growth and success.
NOTE 2 — SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Principles of consolidation and basis of preparation
The accompanying consolidated financial statements reflect the accounts of the Company and all of its subsidiaries in which a controlling interest is maintained. All inter-company balances and transactions have been eliminated in consolidation.
The business combination transaction between Legacy NewGenIvf and SPAC I was accounted for as a reverse recapitalization under ASC 805, Business Combinations, with NewGenIvf Group Limited, and deemed to be the accounting acquirer. As SPAC I did not meet the definition of a business under ASC 805, the transaction was not treated as a business combination. Instead, it was accounted for as a recapitalization.
Accordingly, the consolidated assets, liabilities and results of operations of the accounting acquirer will become the historical financial statements of the Company, and the accounting acquirer’s assets, liabilities and results of operations will be consolidated with the Company beginning on the acquisition date. The Legacy NewGenIvf was the legal acquiree but deemed to be the accounting acquirer. The Company was the legal acquirer but deemed to be the accounting acquiree in the reverse merger. The historical financial statements prior to the acquisition are those of the accounting acquirer (Legacy NewGenivf). After completion of the Share Exchange Transaction, the Company’s consolidated financial statements include the assets and liabilities, the operations and cash flow of the accounting acquirer. Any excess of the value of shares issued by the Company over the net book value of the accounting acquirer will be recognized as a reduction to equity (APIC).
F-
Business combination and gain on bargain purchase
The Company adopted ASC 805, the accounting for business combinations which requires the application of the acquisition method. This method mandates that the acquiring entity recognize all assets acquired and liabilities assumed at their acquisition-date fair values. Gain on a business acquisition is recognized in earnings only in the specific circumstance of a “bargain purchase.” This occurs when the fair value of the identifiable net assets acquired exceeds the total consideration transferred (including the fair value of any non-controlling interest). It requires the acquirer to first meticulously reassess all measurements—including the identification and valuation of assets, liabilities, and the consideration—to ensure the gain is not a measurement error. Once confirmed, the resulting bargain purchase gain is recognized in profit or loss on the acquisition date. This treatment reflects the economic benefit obtained by the acquirer from purchasing net assets for less than their aggregate fair value. Under US GAAP, the distinction between acquiring a business and acquiring a group of assets is determined by a defined screen test. A transaction is accounted for as a business combination only if the acquired set meets the specific definition of a business, which requires an integrated set of activities with, at a minimum, an input and a substantive process that together significantly contribute to the ability to create outputs.
Segmental Reporting
As a public entity with securities listed on the Nasdaq Stock Market, the Company is required to apply the provisions of ASC 280, Segment Reporting. The Company determines its operating segments based on the information used by the Chief Operating Decision Maker (CODM) to allocate resources and assess performance. The CODM has been identified as the Chief Executive Officer.
The CODM reviews financial information at the operating segment level on a regular basis. Based on this review process, the Company has determined its reportable segments to be IVF Revenue and Fertility Referral Revenue. These segments reflect how the CEO manages the business, evaluates performance, and makes strategic decisions regarding resource allocation.
The CODM evaluates the performance of each segment based on segment contribution margin. Significant expenses are reviewed by CEO and it is determined that expenses related to Fertility Referral Revenue are immaterial and thus there is no need to segregate the expenses under different segments. Other segment items represent general and administrative and other miscellaneous expenses not separately reported to the CODM.
As a public entity with securities listed on the Nasdaq Stock Market, the Company applies the provisions of ASC 280, Segment Reporting. Segment assets are not regularly provided to the CODM and are therefore not reported in the segment disclosures.
Use of estimates
The preparation of the consolidated financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosures of contingent assets and liabilities as of the date of the consolidated financial statements and the reported amounts of revenues and expenses during the periods presented. Actual results could differ from those estimates. Significant accounting estimates reflected in the Company’s consolidated financial statements includes the impairment of long-lived assets including the intangible assets, revenue recognition, fair value measurement of digital assets, assessment of whether an acquisition is that of a business or an asset and recognition of bargain purchase gain, determination of embedded derivatives requiring bifurcation and valuation thereof.
Foreign currency translation
The accompanying consolidated financial statements are presented in United States dollar (“$”), which is the reporting currency of the Company. The functional currency of the Company and its subsidiaries, FFPGS (HK) Limited and Well Image Limited, are Hong Kong dollar (“HK$”). Med Holdings and FFC use Thai baht (“THB”) as their functional currencies. The other subsidiaries use United States dollar (“USD”) as their functional currencies.
F-
Assets and liabilities denominated in currencies other than the reporting currency are translated into the reporting currency at the rates of exchange prevailing at the balance sheet date. Monetary assets and liabilities denominated in currencies other than the functional currency are translated into the functional currency using the applicable exchange rates at the balance sheet dates. Translation gains and losses are recognized in the consolidated statements of operations and comprehensive income as other comprehensive income or loss.
Transactions in currencies other than the reporting currency are measured and recorded in the reporting currency at the exchange rate prevailing on the transaction date. The cumulative gain or loss from foreign currency transactions is reflected in the consolidated statements of operations and comprehensive income as other income (other expenses).
The value of foreign currencies including, the HK$, THB and RMB, may fluctuate against the United States dollar. Any significant variations of the aforementioned currencies relative to the United States dollar may materially affect the Company’s financial condition in terms of reporting in USD. The following table outlines the currency exchange rates that were used in preparing the accompanying consolidated financial statements:
| 2025 | 2024 | 2023 | ||||||||||||
| Period-end | $: HK$ | 7.8000 | 7.8000 | 7.8000 | ||||||||||
| Period average | $: HK$ | 7.8000 | 7.8000 | 7.8000 | ||||||||||
| Period-end | $: THB | 31.4215 | 34.3353 | 34.2265 | ||||||||||
| Period average | $: THB | 31.7436 | 35.2262 | 34.7867 | ||||||||||
| Period-end | $: RMB | 7.2001 | 7.2994 | 7.0971 | ||||||||||
| Period average | $: RMB | 7.2327 | 7.1946 | 7.0835 | ||||||||||
Cash and cash equivalents
Cash and cash equivalents include cash on hand, deposits held at call with financial institutions, other short-term deposits with original maturities of three months or less that are readily convertible to known amounts of cash and which are subject to an insignificant risk of changes in value.
Deposits, prepayments other receivables, net
Deposits, other receivables, net primarily include deposits paid to suppliers and prepaid expenses.
Legal and professional fees incurred in connection with issuing convertible debt are deferred and amortized over the life of the debt. These costs are presented as a direct deduction from the carrying amount of the debt liability on the balance sheet (per ASC 835-30).
Digital assets, fair value
The Company accounts for crypto assets in accordance with ASC 350-60, Intangibles — Goodwill and Other — Crypto Assets. We have ownership of and control over our digital assets and use third-party custodial services to store the private keys that provide access to the digital assets in its digital wallets. Digital assets are initially recorded at cost and are subsequently remeasured at fair value, with changes in fair value recognized in net income in the period of change.
We determine and record the fair value of our digital assets in accordance with ASC 820, Fair Value Measurement (“ASC 820”), based on quoted prices on the active exchange(s) that we have determined is the principal market for such assets (Level I inputs). We determine the cost basis (using the first-in, first-out (“FIFO”) method) of our digital assets using the specific identification of each unit received. Realized and unrealized gains and losses are recorded in Other income (expense), net in the Consolidated Statements of Operations.
F-
Share based compensation
The Company accounts for stock-based compensation in accordance with ASC Topic 718-10, Compensation-Stock Compensation, which requires the measurement and recognition of compensation expense for all share-based payment arrangements related to the acquisition of goods and services from both non-employees and employees based on fair values of the shares at grant date represented by the closing prices of the Company’s common stock at the respective grant date in accordance with ASC 718. Stock-based compensation expense recognized during the period is based on the value of the portion of share-based payment awards that relate to the service period during the year.
Prepaid share-based compensation is recorded when the Company issues fully vested, nonforfeitable share-based payment awards to nonemployees on the grant date, when the nonemployee earns the right to receive and where no specific performance is required by the nonemployee to retain those equity instruments, in accordance with ASC 718-10-45-3. The prepayment is recorded where the shares are issued before the financial year end in accordance with the grant date and represents the fair value of goods or services that are to be received from the nonemployee subsequent to the financial period end.
Compensation cost for awards that do not have an established accounting grant date, but for which the service inception date has been established is based on the best estimate of fair value of Company’s common stock at the end of each reporting period and a corresponding accrual is recorded.
Share-based compensation cost is recognized over the requisite service period for employees and over the service period for non employees.
Property and equipment, net
Plant and equipment are stated at cost less accumulated depreciation. Depreciation is provided over their estimated useful lives, using the straight-line method. The Company typically applies a salvage value of 0%. The estimated useful lives of the plan and equipment are as follows:
| Furniture and fixtures | 3 – 5 years | |
| Leasehold improvements | the lesser of useful life or term of lease | |
| Medical instruments | 3 – 10 years | |
| Motor vehicle | 3 – 5 years | |
| Office equipment | 3 – 5 years |
The cost and related accumulated depreciation of assets sold or otherwise retired are eliminated from the accounts, and any gain or loss are included in the Company’s results of operations. The costs of maintenance and repairs are expensed as incurred. Significant renewals and betterments that extend the useful life of an assets are capitalized.
Intangible Assets
Intangible assets acquired from third parties are measured initially at fair value and those with an infinite live are not amortized. The Company annually evaluates the recoverability of the infinite-lived intangible assets for possible impairment whenever events or circumstances indicate that the carrying amount of such assets may not be recoverable. To test indefinite-lived intangible assets for impairment, the Company first performs a qualitative assessment to determine if it is more likely than not that the carrying amount of each of its indefinite-lived intangible assets exceeds its fair value. If it is, a quantitative assessment is required. Alternatively, the Company may bypass the qualitative assessment and perform a quantitative impairment test. The qualitative assessment requires the consideration of factors such as recent market transactions, macroeconomic conditions and changes in projected future cash flows. The quantitative assessment compares the fair value of an indefinite-lived intangible asset to its carrying amount. If the carrying amount of an indefinite-lived intangible asset exceeds its fair value, an impairment loss is recognized for the excess. Fair values of indefinite-lived intangible assets are determined based on discounted cash flows or appraised values, as appropriate. If such a review indicates that the carrying amount of intangible assets is not recoverable, the carrying amount of such assets is reduced to fair value.
As of December 31, 2025, and 2024, the Company did not record an impairment on the intangible assets.
F-
Impairment of long-lived assets
In accordance with the provisions of ASC Topic 360, Impairment or Disposal of Long-Lived Assets, all long-lived assets such as property, plant and equipment owned and held by the Company are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. Recoverability of assets to be held and used is evaluated by a comparison of the carrying amount of an asset to its estimated future undiscounted cash flows expected to be generated by the asset. If such assets are considered to be impaired, the impairment to be recognized is measured by the amount by which the carrying amounts of the assets exceed the fair value of the assets.
There was no impairment loss on plant and equipment for the year ended December 31, 2025 and 2024.
Deposit with futures broker, net
The Company accounts for financial assets, derivative instruments, and broker margin balances in accordance with Accounting Standards Codification (“ASC”) 815, Derivatives and Hedging, ASC 820, Fair Value Measurement. The Company has engaged in trading of exchange-traded equity index futures through a margin account maintained with a regulated futures broker. Futures contracts are accounted for as derivative instruments under ASC 815.
The Company does not designate any derivative instruments as hedging instruments for accounting purposes. Accordingly, all derivatives are accounted for at fair value with changes in fair value recognized in earnings as incurred. Realised and unrealised gains and losses arising from derivative trading activities are recorded within other income (expense) in the consolidated statement of comprehensive income.
Cash deposited with a futures broker to support margin requirements is included within deposit held by futures broker. The margin account reflects the accumulation of realised gains and losses from futures transactions, brokerage commissions, margin interest, and other related charges. Broker margin accounts may include balances denominated in multiple currencies. Amounts related to trading activity are primarily accumulated in Japanese yen (“JPY”), while margin deposits are funded principally in U.S. dollars (“USD”).
Upon closure of a margin account, cash proceeds are expected to be received net of outstanding broker charges and financing costs.
Interest incurred on negative balances within broker margin accounts is recognized as interest expense in the period incurred and included within other income (expense) or finance costs, as applicable.
Exchange-traded futures contracts are measured at fair value using quoted prices in active markets. When outstanding, such instruments are classified within Level 1 of the fair value hierarchy described in ASC 820.
Inventories
Inventories are stated at the lower of cost and net realizable value. Costs are determined on a first-in, first-out basis. Net realizable value is based on the estimated selling prices less any estimated costs to be incurred to completion and disposal. A provision for excess and obsolete inventory will be made based primarily on products approaching expiry period and forecasts of product demand. The excess balance above the product demand as determined by this analysis becomes the basis for excess inventory charge and the written-down value of the inventory becomes its cost. Written-down inventory would not be reversed if market conditions improve.
Deferred offering costs
Deferred offering costs, which consist of legal and other expenses incurred through the balance sheet date that are directly related to the proposed public offering, are capitalized, and will be charged against the gross proceeds of the offering and recorded as reduction of shareholders’ equity upon the completion of the proposed offering. Should the proposed public offering prove to be unsuccessful, these deferred costs, as well as additional expenses incurred, will be charged to the statements of operations and comprehensive income (loss).
F-
Other borrowings
Other borrowings are recognized initially at fair value, net of debt issuance costs incurred. Other borrowings are subsequently stated at amortized cost; any difference between the proceeds (net of debt issuance costs) and the redemption value is recognized in the consolidated statements of operations over the period of the borrowings using the effective interest method.
Convertible Instruments
Convertible Instruments are categorized as equity or debt based on the terms of the notes. Convertible Notes are recorded at amounts equal to the proceeds of the issuance, including the embedded conversion feature, and net of discounts and unamortized debt issuance in accordance with ASC 480-10-55-44 on the consolidated balance sheets. An evaluation of all conversion, purchase and redemption features contained in a debt instrument is performed to determine if there are any embedded features that require bifurcation as a derivative. The conversion feature is recorded separately as a derivative liability at its fair value, calculated using the Black-Scholes model.
Debt issuance and offering costs are amortized over the contractual term of the Convertible Notes, to the consolidated statements of operations in accordance with ASC 835-30-45-1A and presented as a reduction of the amortised cost of the Convertible Note.
The convertible notes are subsequently recorded at amortized cost, with interest expense recognized using the effective interest method. The derivative liability, if any is remeasured at fair value at each reporting date and any gain or loss on fair value is recognized in the statement of comprehensive income.
Warrants
The Company accounts for warrants as either equity-classified or liability-classified instruments based on an assessment of the warrant’s specific terms and applicable authoritative guidance in ASC Topic 480, Distinguishing Liabilities from Equity (“ASC 480”) and ASC Topic 815, Derivatives and Hedging (“ASC 815”). The assessment considers whether the warrants are freestanding financial instruments pursuant to ASC 480, meet the definition of a liability pursuant to ASC 480, and whether the warrants meet all of the requirements for equity classification under ASC 815, including whether the warrants are indexed to the Company’s own common stock and whether the warrant holders could potentially require “net cash settlement” in a circumstance outside of our control, among other conditions for equity classification. This assessment, which requires the use of professional judgment, is conducted at the time of warrant issuance and as of each subsequent period end date while the warrants are outstanding.
Equity-classified
For issued or modified warrants that meet all of the criteria for equity classification, the warrants are required to be recorded as a component of equity at the time of issuance. Warrants classified as equity instruments are initially recognized at fair value within additional paid in capital and are not subsequently remeasured. The warrants have been assessed to meet the requirements for equity classification and accounted for as equity. Upon exercise of the warrants, the Company will record the proceeds received, together with the carrying value of the warrants, as an increase to common stock and additional paid-in capital. Warrants that expire unexercised will be derecognized with no impact on the statement of operations
Liability-classified
For issued or modified warrants that do not meet all the criteria for equity classification, the warrants are required to be recorded as liabilities at their initial fair value on the date of issuance and are remeasured at each reporting date until settlement. Changes in fair value is recognized as a component of change in fair value of warrant liability in the condensed consolidated statements of operations and as a non-cash gain or loss in the condensed consolidated statement of comprehensive loss. Transaction costs allocated to warrants that are presented as a liability are immediately expensed in the consolidated statements of operations and comprehensive loss.
No liability classified warrants have been issued as of December 31, 2025.
Promissory Notes
Promissory notes, are of non-interest bearing and recorded at original cost. They are subsequently measured at amortised cost, with interest expense recognized using the effective interest method in the consolidated statement of income.
F-
Ordinary shares
The Company’s ordinary shares are stated at no par value. The difference between the consideration received, net of issuance cost, is recorded in additional paid-in capital.
Pursuant to ASC 505, reverse stock splits are applied retrospectively. Consequently, all share and per share amounts for all periods presented in the accompanying consolidated financial statements and notes thereto have been adjusted to reflect the reverse stock split. This retrospective adjustment includes the recalculation of shares within equity and ensures that basic and diluted earnings per share, as well as all other share-based information such as stock options and warrants, are presented as if the reverse stock split had been in effect for all periods disclosed.
The equity of the Company as of December 31, 2025 represents 240,622 ordinary shares with par value of $Nil. The number of shares as above have taken into consideration the reverse stock split effected on February 11, 2025, May 6, 2025, August 4, 2025, December 1, 2025, January 26, 2026 and March 16, 2026 at an exchange ratio of one (1) share for twenty (20) shares, one (1) share for ten (10) shares, one (1) share for five (5) shares, one (1) share for five (5) shares, one (1) share for three (3) shares and one (1) share for four (4) shares respectively.
Revenue recognition
The Company adopted ASC Topic 606, Revenue from Contracts with Customers, and all subsequent ASUs that modified ASC 606 on April 1, 2017 using the full retrospective method which requires the Company to present the financial statements for all periods as if Topic 606 had been applied to all prior periods. The Company derives revenue principally from provision of In vitro fertilization (“IVF”) treatment and surrogacy and ancillary caring services. Revenue from contracts with customers is recognized using the following five steps:
| (1) | identify its contracts with customers; | |
| (2) | identify its performance obligations under those contracts; | |
| (3) | determine the transaction prices of those contracts; | |
| (4) | allocate the transaction prices to its performance obligations in those contracts; and | |
| (5) | recognize revenue when each performance obligation under those contracts is satisfied. Revenue is recognized when promised services are transferred to the client in an amount that reflects the consideration expected in exchange for those services. |
The Company enters into verbal agreements with its customers that outline the rights, responsibilities, and obligations of each party. The agreements also identify the scope of services, service fees, and payment terms. Agreements are acknowledged and consent forms are signed by the customers prior to each promised service or bundle of services are inter dependent. All the contracts have commercial substance, and it is probable that the Company will collect considerations from its customers for service component as settlement is predominantly required prior to performance of the promised service.
The Company derives its revenues from two sources: (1) revenue from IVF treatment, and (2) revenue from fertility referral services.
Revenue from IVF treatment
In vitro fertilization (“IVF”) treatment is an assisted reproductive technique where eggs and sperm are collected and fertilized in laboratory to become embryo. Fertilized embryo is then implanted to the customer or a surrogate mother. IVF treatment involves the performance of a series of medical treatment as well as procedures and brings benefits to clients as the service of bundles service is completed. Revenue from IVF treatment is recognized at a point in time when different treatment and/or procedure or bundles thereof, are completed in clinic. The full completion of the various procedures and treatments are evidenced by treatment cards and reports included within the patient files indicating successful completion of the service. Collections from customer who are individuals are received in advance of the service, whereas for entities, a credit term of 30 days is provided.
Revenue from Fertility Referral services
The Company arranges fertility referral services for customers seeking IVF and/or surrogacy services and earns referral fee income. Upon a customer inquiry, the Company evaluates the customer’s needs and, if appropriate, refers them to independent third-party agents.
In these arrangements, the Company’s performance obligation is satisfied upon the successful referral of the customer to the independent agent. Referral fee revenue is recognized when the Company transfers control of the service by completing the referral, as this represents the point at which the Company’s obligation is fulfilled and the customer (the agent) simultaneously receives and consumes the benefits.
The transaction price consists of a referral fee, which is negotiated with the agent on a case-by-case basis. As the referral fee is contingent upon the agent’s acceptance of the referral and subsequent income received from the agent, revenue is constrained until it is probable that a significant reversal will not occur. Accordingly, revenue is recognized at the point in time when the referral fee is received from the agent, as this is when the uncertainty surrounding the consideration is resolved and the Company’s right to consideration becomes unconditional.
F-
Revenue from egg freezing and storage facility
The Company provides access the facility to its customers. Upon request for the service, which is agreed verbally and followed by signed consent form from the customer, the Company makes available access to the facility with no further substantial involvement. Revenue is recognized at a point in time when the facility is made available to the customer at the agreed consideration by the provision of specific address within the facility as maintained in the patient file. The receipt of consideration is assured as payment is required upfront.
Principal versus agency considerations
The Company follows the guidance provided in ASC 606, Revenue from Contracts with Customers, for determining whether the Company is the principal or an agent in arrangements with customers that involve another party that contributes to the provision of services to a customer. In these instances, the Company determines whether it has promised to provide the service itself (as principal) or to arrange for the specified service to be provided by another party (as an agent). This determination is a matter of judgment that depends on the facts and circumstances of each arrangement. The Company recognizes revenue from the performance of the procedures and treatment on a gross basis as the Company is responsible for the fulfillment, controls the delivery of the promised service, and has full discretion in establishing prices and therefore is the principal in the arrangement.
Contract related assets and liabilities are classified as current assets and current liabilities.
Account receivables, net
Accounts receivable, net are stated at the original amount less an allowance for expected credit loss on such receivables. The allowance for expected credit loss is estimated based upon the Company’s assessment of various factors including historical experience, the age of the accounts receivable balances, current general economic conditions, future expectations and customer specific quantitative and qualitative factors that may affect the Company’s customers’ ability to pay. An allowance is also made when there is objective evidence for the Company to reasonably estimate the amount of probable loss.
Contract liabilities
Contract liabilities represent considerations received from customers in advance of satisfying the Company’s performance obligations under the contract. These amounts are expected to be earned within 12 months and are classified as current liabilities.
Expected credit loss
ASU No. 2016-13, Financial Instruments — Credit Losses (Topic 326): Measurement of Credit Losses on Financial Instruments requires entities to use a current lifetime expected credit loss methodology to measure impairments of certain financial assets. Using this methodology will result in earlier recognition of losses than under the current incurred loss approach, which requires waiting to recognize a loss until it is probable of having been incurred. There are other provisions within the standard that affect how impairments of other financial assets may be recorded and presented, and that expand disclosures. Expected credit losses are probability-weighted estimates of credit losses. Credit losses are measured at the present value of all cash shortfalls (i.e., the difference between the cash flows due to the entity in accordance with the contract and the cash flows that the Company expects to receive). ECLs are discounted at the effective interest rate of the financial asset.
Retirement benefits
Retirement benefits in the form of mandatory government-sponsored defined contribution plans are charged to either expense as incurred or allocated to wages as part of cost of revenues.
F-
Leases
The Company measured the lease in accordance to ASU 2016-02, “Leases” (Topic 842). Lease terms used to calculate the present value of lease payments generally do not include any options to extend, renew, or terminate the lease, as the Company does not have reasonable certainty at lease inception that these options will be exercised. The Company generally considers the economic life of its operating lease ROU assets to be comparable to the useful life of similar owned assets. The Company has elected the short-term lease exception, therefore operating lease ROU assets and liabilities do not include leases with a lease term of twelve months or less. Its leases generally do not provide a residual guarantee. The operating lease ROU asset also excludes lease incentives. Lease expense is recognized on a straight-line basis over the lease term.
Income Taxes
The Company recognizes deferred income tax assets or liabilities for expected future tax consequences of events recognized in the consolidated financial statements or tax returns. Under this method, deferred income tax assets and liabilities are determined based on the differences between the financial reporting and income tax bases of assets and liabilities and are measured using the income tax rates that will be in effect when the differences are expected to reverse. Valuation allowances are provided when it is more likely than not that a deferred tax asset is not realizable or recoverable in the future.
The Company determines that the tax position is more likely than not to be sustained and records the largest amount of benefit that is more likely than not to be realized when the tax position is settled. the Company recognizes interest and penalties, if any, related to uncertain tax positions in income tax expense.
Comprehensive Income
The Company presents comprehensive income in accordance with ASC Topic 220, Comprehensive Income. ASC Topic 220 states that all items that are required to be recognized under accounting standards as components of comprehensive income be reported in the consolidated financial statements. The components of comprehensive income were the net income for the years and the foreign currency translation adjustments.
Earnings per share
The Company computes earnings per share (“EPS”) following ASC Topic 260, “Earnings per share”. Basic EPS is measured as the income or loss available to common shareholders divided by the weighted average common shares outstanding for the period. Diluted EPS presents the dilutive effect on a per-share basis from the potential conversion of convertible securities or the exercise of options and or warrants; the dilutive impacts of potentially convertible securities are calculated using the as-if method. Potentially anti-dilutive securities (i.e., those that increase income per share or decrease loss per share) are excluded from diluted EPS calculation.
F-
Related parties
The Company adopted ASC 850, Related Party Disclosures, for the identification of related parties and disclosure of related party transactions.
Commitments and contingencies
In the normal course of business, the Company is subject to contingencies, including legal proceedings and claims arising out of the business that relate to a wide range of matters, such as government investigations and tax matters. The Company recognizes its liability for such contingency if it determines it is probable that a loss has occurred and a reasonable estimate of the loss can be made. The Company may consider many factors in making these assessments including historical and the specific facts and circumstances of each matter.
Non-controlling interests
Non-controlling interests are presented as a separate component of equity on the consolidated balance sheets and net (loss) income and other comprehensive loss are attributed to controlling and non-controlling interests respectively.
Concentration of risks
Concentration of credit risk
Financial instruments that potentially expose us to concentrations of credit risk consist primarily of cash and cash equivalents and account receivable. The Company places cash and cash equivalents with financial institutions with high credit ratings and quality.
Accounts receivable primarily comprise of amounts receivable from the service customers. The Company conducts credit evaluations of customers, and generally does not require collateral or other security from its customers. The Company establishes an allowance for doubtful accounts primarily based upon the factors surrounding the credit risk of specific customers. Furthermore, the risk is mitigated by ascertaining upfront payments for services prior to their performance.
Financial instruments
The Company’s financial instruments, including cash and cash equivalents, accounts receivables, net, deposits, other receivables and deferred IPO cost, net, loan to A SPAC I, accounts payables, accrued liabilities and other payables, and due from (to) shareholders, have carrying amounts that approximate their fair values due to their short maturities. ASC Topic 820, “Fair Value Measurements and Disclosures” requires disclosing the fair value of financial instruments held by the Company. ASC Topic 825, “Financial Instruments” defines fair value and establishes a three-level valuation hierarchy for disclosures of fair value measurement that enhances disclosure requirements for fair value measures. The carrying amounts reported in the consolidated balance sheets for cash and cash equivalents, accounts and other receivables, accounts and other payables, accrued liabilities and amounts due from (to) related parties each qualify as financial instruments and are a reasonable estimate of their fair values because of the short period between the origination of such instruments and their expected realization and their current market rate of interest. The three levels of valuation hierarchy are defined as follows:
| ● | Level 1 — inputs to the valuation methodology used quoted prices for identical assets or liabilities in active markets. |
| ● | Level 2 — inputs to the valuation methodology include quoted prices for similar assets and liabilities in active markets and information that are observable for the asset or liability, either directly or indirectly, for substantially the financial instrument’s full term |
| ● | Level 3 — inputs to the valuation methodology are unobservable and significant to the fair value measurement. |
The Company analyzes all financial instruments with features of both liabilities and equity under ASC 480, “Distinguishing Liabilities from Equity” and ASC 815.
F-
Recently issued accounting pronouncements
The FASB has introduced expanded income tax disclosure requirements under ASU 2023-09 to improve transparency. Companies will now need to provide a detailed reconciliation of their effective tax rate, breaking down federal, state, and foreign taxes, as well as specific categories like tax credits and foreign earnings. Additionally, businesses must disclose income taxes paid by jurisdiction, offering investors greater clarity on tax obligations. These changes apply to both public and private companies, with annual reporting periods beginning after December 15, 2024 (2025 for calendar-year entities). This update aims to reduce ambiguity in tax reporting and align disclosures with investor needs.
A major shift in digital asset accounting, ASU 2023-08 requires companies to measure certain crypto assets (e.g., Bitcoin, Ethereum) at fair value rather than applying the previous impairment-only model. This means entities must recognize quarterly fair value adjustments in their financial statements, increasing volatility in reported earnings but improving transparency. The standard applies to fiscal years beginning after December 15, 2024, and impacts both corporate treasuries and investment firms holding cryptocurrencies. This change aligns GAAP closer to fair value accounting seen in other investment holdings, addressing criticisms of the old impairment approach.
In November 2024, the FASB issued ASU 2024-03, *Income Statement—Reporting Comprehensive Income—Expense Disaggregation Disclosures (Subtopic 220-40): Disaggregation of Income Statement Expenses*, which requires public entities to disclose, in a tabular format in the notes to the financial statements, specific information about certain costs included in common expense captions. This standard mandates the disaggregation of natural expenses—such as employee compensation, depreciation, and inventory costs—from broader captions like “cost of services” or “selling, general and administrative expenses.” Although this ASU is effective for annual reporting periods beginning after December 15, 2026, and interim periods thereafter, with early adoption permitted, the Company is currently evaluating the potential impact of this new guidance on its financial statement disclosures.
In July 2025, the FASB issued ASU 2025-05, Financial Instruments—Credit Losses (Topic 326): Measurement of Credit Losses for Accounts Receivable and Contract Assets, which introduces a practical expedient to simplify the application of the current expected credit loss (CECL) model for current accounts receivable and contract assets arising from ASC 606 revenue transactions. Such practical expedient permits entities to assume that current economic conditions as of the balance sheet date will remain unchanged for the remaining life of these short-term assets, eliminating the need to develop and document forward-looking macroeconomic forecasts. For healthcare entities with significant patient accounts receivable, third-party payor contract assets, or government reimbursement receivables, this simplification reduces compliance costs and complexity while continuing to require consideration of historical loss experience and current conditions such as customer-specific financial distress or changes in credit policies. The guidance is effective for annual periods beginning after December 15, 2025, applied prospectively, and early adoption is permitted. The Company plans to adopt this practical expedient and will update its credit loss estimation policies and related disclosures accordingly.
Save for elsewhere disclosed, the Company does not believe other recently issued but not yet effective accounting standards, if currently adopted, would have a material effect on the Company’s consolidated balance sheet, statement of operations and comprehensive income (loss) and statement of cash flows.
NOTE 3 — ACCOUNTS RECEIVABLE, NET
Accounts receivable, net consists of the following:
| December 31, 2025 |
December 31, 2024 |
|||||||
| Accounts receivable | $ | 166,552 | $ | 49,264 | ||||
| Less: allowance for expected credit loss | (21 | ) | (19 | ) | ||||
| $ | 166,531 | $ | 49,245 | |||||
F-
As of the end of each of the financial year, the aging analysis of accounts receivable, net of allowance for expected credit loss, based on the invoice date is as follows:
| December 31, 2025 |
December 31, 2024 |
|||||||
| Current | 25,548 | - | ||||||
| Within 60 days | 6,677 | |||||||
| Within 90 days | 1,941 | 49,245 | ||||||
| More than 90 days | $ | 132,365 | $ | |||||
| $ | 166,531 | $ | 49,245 | |||||
The movement of allowances for expected credit loss is as follow:
| December 31, 2025 |
December 31, 2024 |
|||||||
| Balance at beginning of the year | $ | (19 | ) | $ | (19 | ) | ||
| Reversal of expected credit losses | (2 | ) | ||||||
| Ending balance | $ | (21 | ) | $ | (19 | ) | ||
NOTE 4 — INVENTORIES
Inventories, at cost, consist of the following:
| December 31, 2025 |
December 31, 2024 |
|||||||
| Medicines, consumables and reagents for clinical and laboratory analyses | $ | 77,296 | $ | 80,813 | ||||
| Supplements | 258,064 | |||||||
| $ | 335,360 | $ | 80,813 | |||||
NOTE 5 — DEPOSITS, PREPAYMENT AND OTHER RECEIVABLES, NET
Deposits, prepayment and other receivables cost, net consist of the following:
| December 31, 2025 |
December 31, 2024 |
|||||||
| Current | ||||||||
| Other receivables | $ | 33,442 | $ | 121,533 | ||||
| Deposits | 199,935 | 73,917 | ||||||
| Prepayment | 120,542 | 197,706 | ||||||
| Loan receivable from a third party (a) | 75,000 | |||||||
| Advanced payment to supplier | 74,674 | |||||||
| Less: allowance for expected credit loss | (5 | ) | (4 | ) | ||||
| $ | 503,588 | $ | 393,152 | |||||
| Deposit with a digital asset broker | $ | 34,987 | $ | 1,000,000 | ||||
| Receivables from agents | $ | 794,116 | $ | 1,191,795 | ||||
| Deposit for land acquisition (b) | $ | 4,614,749 | $ | |||||
| Non-current | ||||||||
| Prepayment | $ | $ | 33,333 | |||||
| (a) | The loan is unsecured, interest free and is repayable on demand. |
| (b) | No definitive agreement has been concluded to date regarding this deposit paid for the acquisition of land in Ras Al Khaimah, UAE. |
F-
The movement of allowances for expected credit loss is as follow:
| December 31, 2025 |
December 31, 2024 |
|||||||
| Balance at beginning of the year | $ | (4 | ) | $ | (14 | ) | ||
| Provision during the year | (1 | ) | ||||||
| Reversal of provision (Provision) | 10 | |||||||
| Ending balance | $ | (5 | ) | $ | (4 | ) | ||
NOTE 6 — PROPERTY AND EQUIPMENT, NET
Plant and equipment, net consist of the following:
| December 31, 2025 |
December 31, 2024 |
|||||||
| At cost: | ||||||||
| Building improvement | $ | 247,515 | $ | 196,929 | ||||
| Furniture and fixtures | 1,550,061 | 25,390 | ||||||
| Medical instruments | 1,052,578 | 785,776 | ||||||
| Motor vehicle | 142,936 | |||||||
| Office equipment | 167,234 | 361,385 | ||||||
| 3,017,388 | 1,512,416 | |||||||
| Less: accumulated depreciation | (1,228,278 | ) | (1,239,320 | ) | ||||
| Total | $ | 1,789,110 | $ | 273,096 | ||||
Depreciation expenses for the years ended December 31, 2025 and December 31, 2024 were $188,109 and $19,502, respectively.
No impairment loss was recorded for the years ended December 31, 2025 and December 31, 2024
F-
NOTE 7 — INTANGIBLE ASSETS
The intangible assets comprise:
| (ii) | A trademark and technology upon the acquisition of MicroSort Lab Services, LLC (“MicroSort”) on February 28, 2025 valued at $3,495,335, refer Note 21, and |
| (ii) | An intellectual property upon the acquisition of the business of Nodexus Inc (“Nodexus”) on July 29, 2025 valued at $19,166,000, refer Note 21. |
NewGenIvf exclusively owns the MicroSort Technology. MicroSort technology is a form of pre-conception gender selection technology for humans. MicroSort technology aims to separate male sperm cells based on which gender chromosome they contain, which results in separated semen samples that contain a higher percentage of sperm cells that carry the same gender chromosome. The technology ultimately helps couples choose the gender of their future child by choosing semen samples that predominately contain sperm with the X chromosome for a female or Y chromosome for a male. Traditionally and naturally, gender selection occurs after conception, meaning after the eggs are fertilized. As a result, some fertilized eggs will go unused. However, with MicroSort technology, NewGenIvf is able to increase the ratio of male or female embryos, based on the patient’s preference. Eggs are more likely to be fertilized according to the preferences of the parents. Other improvements that MicroSort treatment could help achieve include prevention of certain gender-related hereditary diseases.
NewGenIvf acquired patents from Nodexus Inc, in 2025, which is a biotechnology company revolutionizing live cell analysis and isolation through its advanced microfluidic technology. The company’s flagship NX One platform is an automated, benchtop system designed to make high-precision cell sorting more accessible and affordable for a wide range of laboratories, from academic institutions to biopharma firms. Unlike traditional, high-pressure sorters that can damage fragile cells, Nodexus utilizes a proprietary technology that operates at incredibly low pressures (under 1 psi), ensuring high cell viability during the sorting process. The system employs single-use, closed cartridges that maintain sterility, prevent cross-contamination, and support the sorting of diverse particle types, from small yeast cells and nuclei to large, delicate organoids up to 200 micrometers in size. This versatile and gentle approach has critical applications in cutting-edge research areas, including CRISPR gene editing, stem cell therapy development, cancer biology, and single-cell genomics. This technology can facilitate the gender selection as well as avoiding genetic diseases in the IVF treatment.
The Company has identified these intangible assets as indefinite life intangible assets as there are currently no legal, competitive, economic or other factors that materially limit the useful life of the Company’s intangible assets.
ASC 350-30-35, Intangibles - Goodwill and Other, provides for an option to first perform a qualitative assessment to determine whether it is more likely than not that an asset is impaired. If the qualitative assessment supports that it is more likely than not that the fair value of the asset exceeds its carrying value, a quantitative impairment test is not required. If the qualitative assessment does not support the fair value of the asset, then a quantitative assessment is performed. Our annual impairment assessment of our identifiable indefinite lived intangible assets is performed as of the fourth quarter of each year. An assessment is performed at other times if an event occurs or circumstances change that would more likely than not reduce the fair value of the asset below its carrying value. If the carrying value of the intangible assets exceeds its fair value, an impairment loss is recognized in an amount equal to that excess.
As of December 31, 2025, the Company performed a qualitative assessment, during the fourth quarter of 2025, to determine whether it was more likely than not that the carrying amounts of its intangible assets were impaired. This assessment considered a number of qualitative factors, including but not limited to:
| - | Macroeconomic conditions, |
| - | Industry and market trends, |
| - | Cost factors, |
| - | Overall financial performance of the asset, |
| - | Legal and regulatory environment, and |
| - | Relevant internal reporting and management’s plans |
F-
Based on this qualitative assessment, management did not identify any events or changes in circumstances that would indicate that the carrying amounts of the Company’s intangible assets are not recoverable. The qualitative impairment assessment of the indefinite intangible assets indicated that the fair value of such assets exceeded their carrying value and therefore were not at risk of impairment. Accordingly, no quantitative impairment test was deemed necessary, and no impairment losses were recognized for the year ended December 31, 2025.
NOTE 9 — DIGITAL ASSETS, FAIR VALUE
During the year ended December 31, 2025, the Company began investing excess treasury balances in certain digital assets, comprising Solana (“SOL”), Bitcoin (“BTC”), Ethereum (“ETH”), and a cryptographic token native to the XRP Ledger (“XRP”), all of which are blockchain-based crypto assets, as part of its capital allocation strategy. The Company’s primary business remains the provision of in-vitro fertilization (“IVF”) services, and digital asset activities are not part of its core operating activities. The Company does not engage in mining, staking, lending, nor other yield-generating crypto asset activities.
Investments and dispositions in digital assets during the year ended December 31, 2025 are made in cash through a third-party trading platform. The Company did not receive digital assets from customers nor use digital assets as payment for goods or services.
Digital Asset Holdings as at December 31, 2025
The following table presents information about the Company’s crypto asset holdings at December 31, 2025:
| Crypto asset | Units held | Cost basis (USD) |
Fair value (USD) |
|||||||||
| Solana (SOL) | 13,000.23 | $ | 2,906,222 | $ | 1,630,827 | |||||||
| Bitcoin (BTC) | ||||||||||||
| Ethereum (ETH) | ||||||||||||
| XRP | ||||||||||||
| Total | $ | 2,906,222 | $ | 1,630,827 | ||||||||
The fair value per unit of Solana as at December 31, 2025 was approximately $125, based on quoted prices in active markets.
Reconciliation of Digital Asset Activity
The following table summarizes activity in digital assets for the year ended December 31, 2025 (in units):
| SOL Units | BTC Units | ETH Units | XRP Units | |||||||||||||
| Balance at January 1, 2025 | ||||||||||||||||
| Purchases during the year | 46,118.55 | 8.77 | 8.95 | 146,432.14 | ||||||||||||
| Sales during the year | (33,118.32 | ) | (8.77 | ) | (8.95 | ) | (146,432.14 | ) | ||||||||
| Balance at December 31, 2025 | 13,000.23 | |||||||||||||||
| SOL (USD) | BTC (USD) | ETH (USD) | XRP (USD) | Total (USD) | ||||||||||||||||
| Realised gains (Losses) | 919,720 | 12,569 | 223 | 10,849 | 942,915 | |||||||||||||||
| Unrealized gains (losses) | (1,275,395 | ) | (1,275,395 | ) | ||||||||||||||||
| Net Gain/Loss | (355,675 | ) | 12,569 | 223 | 10,849 | (332,480 | ) | |||||||||||||
The company recorded and presented the Net loss as a separate line item within other income (expense), net in the Consolidated Statements of Operations.
F-
Only Solana remained held at year-end; all BTC, ETH, and XRP positions were fully liquidated prior to December 31, 2025.
As of December 31, 2025, the Company’s crypto assets were not subject to contractual sale restrictions, pledges, or lock-up arrangements.
Fair Value Measurement
Crypto assets are measured using Level 1 inputs within the fair value hierarchy under ASC 820, based on quoted prices for identical assets in active markets.
NOTE 10 — DEPOSIT WITH FUTURES BROKER, NET
During the year ended December 31, 2025, the Company engaged in trading of exchange-traded equity index futures through a margin account maintained with a regulated futures broker. These contracts were accounted for as derivative instruments under ASC 815, Derivatives and Hedging, and were not designated as hedging instruments for accounting purposes.
The margin deposit, which is denominated in US$, represents cash funds placed with the futures broker to support margin requirements and trading activities denominated in Japanese yen (“JPY) within the margin account. Realised gains and losses from futures transactions, along with brokerage fees, margin interest, and other associated charges, were accumulated within the margin account.
As of December 31, 2025, the Company had a margin deposit held with the broker of US$ 760,000, and a margin account of accumulated trading losses and margin-related charges of JPY 109,875,291 (US$700,631), which are presented as a net amount of US59,368.
Prior to the financial year end, the Company has ceased all derivative trading activities. Upon subsequent closure of the margin account in the near term, any remaining cash balances are expected to be remitted to the Company net of outstanding broker charges and financing costs.
All realised and unrealised gains and losses arising from futures trading activities during the year were recognized in earnings within other income (expense) in the consolidated statement of comprehensive income.
Fair Value Measurements
The futures contracts traded during the year were exchange-traded instruments valued using quoted prices in active markets and, when outstanding, would have been classified within Level 1 of the fair value hierarchy under ASC 820, Fair Value Measurement.
As of December 31, 2025, the Company held no derivative assets or liabilities subject to recurring fair value measurement.
F-
NOTE 11 — ACCRUED LIABILITIES AND OTHER PAYABLES
Accrued liabilities and other payables consist of the following:
| December 31, 2025 |
December 31, 2024 |
|||||||
| Accrued expenses | $ | 514,169 | $ | 201,455 | ||||
| Customer deposit | 6,349 | |||||||
| Withholding tax payable | 1,889 | 4,361 | ||||||
| Independent director fee payable | 20,000 | |||||||
| Compensation payable | 105,446 | 100,802 | ||||||
| Other payables | 90,388 | 167,762 | ||||||
| $ | 711,892 | $ | 500,729 | |||||
Compensation payable represents a claim relating to an employee of the subsidiary.
NOTE 12 — CONTRACT LIABILITIES
Contract liabilities consist of the following:
| December 31, 2025 |
December 31, 2024 |
|||||||
| Balance at beginning of year | $ | 63,489 | $ | 7,937 | ||||
| Advance billings to customers during the year | 57,274 | 75,340 | ||||||
| Recognized to revenue during the year | (8,782 | ) | (19,788 | ) | ||||
| Balance at end of year | $ | 111,981 | $ | 63,489 | ||||
F-
NOTE 13 — CONVERTIBLE NOTES AND DERIVATIVE LIABILITY AND WARRANTS
Convertible Notes
| December 31, 2025 |
December 31, 2024 |
|||||||
| Convertible notes, at amortised cost are as follows: | ||||||||
| Convertible notes issued | $ | 4,701,738 | 3,059,595 | |||||
| Less: Deferred debt issuance cost | $ | (348,316 | ) | (648,232 | ) | |||
| Convertible notes, net | $ | 4,353,422 | 2,411,363 | |||||
| Presented as: | ||||||||
| Current liability | $ | 256,056 | 82,447 | |||||
| Non-current liability | 4,097,366 | 2,328,916 | ||||||
| Convertible notes, net | $ | 4,353,422 | 2,411,363 | |||||
As of December 31, 2025, the Company had the following outstanding convertible notes:
| Note Holders | Principal Amount | Conversion Price | Coupon Rate | Issuance Date | Maturity Date | Fair Value at December 31, 2025 |
Fair Value level | ||||||||||||||
| JAK & affiliated holders | $ | 3,150,000 | 1.7 | 10 | % | 2025-06-03 | 2028-06-02 | $ | 2,831,611 | Level 2 | |||||||||||
| JAK & affiliated holders | 1,475,000 | 0.63 | 14.75 | % | 2025-07-16 | 2030-01-16 | 1,736,145 | Level 2 | |||||||||||||
| Vanquish Funding Group Inc | 257,000 | 25% discount of lowest traded price during 10 trading days immediately preceding the conversion date | 10 | % | 2025-10-15 | 2026-07-30 | 257,673 | Level 2 | |||||||||||||
| Labrys Fund II, LP | 250,000 | 15% discount of lowest traded price during 10 trading days immediately preceding the conversion date | USD 39,285.71 for 6 instalments | 2025-11-12 | 2026-11-11 | 156,063 | Level 2 | ||||||||||||||
| Vanquish Funding Group Inc | 157,000 | 25% discount of lowest traded price during 10 trading days immediately preceding the conversion date | 10 | % | 2025-11-04 | 2026-07-30 | 235,900 | Level 2 | |||||||||||||
F-
During the year period ended December 31, 2025, a total of $7,200,000 of convertible promissory notes were issued with a discount of 7% to JAK. These Notes are to be settled by way of cash or may be converted to Class A ordinary shares. The conversion of these notes is subject to certain criterions as mentioned in the Convertible Note Agreements, which include a beneficiary cap of 9.9% of shareholdings in the Company by JAK.
On October 15, 2025, the Company entered into an additional Securities Purchase Agreement with Vanquish Funding Group Inc. (“Vanquish SPA”), pursuant to which, amongst other things, (i) the Company agreed to sell a convertible note in the aggregate principal amount of $257,000, convertible into Class A Ordinary Shares pursuant to its terms; and (ii) the parties may agree to additional tranches of funding up to $2,200,000 during the following twelve months. The first tranche of $257,000 was received on October 15, 2025. The second tranche of $157,000 was received on November 10, 2025. The third tranche of $107,000 was received on January 30, 2026.
On November 12, 2025, the Company entered into a Securities Purchase Agreement with Labrys Fund II, L.P. (“Labrys SPA”), pursuant to which, amongst other things, (i) the Company agreed to sell a convertible note in the aggregate principal amount of $250,000, convertible into Class A Ordinary Shares pursuant to its terms and the funding of $250,000 was received on November 15, 2025.
During the year ended December 31, 2025, a total of $ 2,725,000 of convertible notes were converted into 58,424 ordinary shares by the note holder (adjusted by Reverse Stock Splits in 2025 and 2026 as of this report date), thereby increasing our shareholder equity by the same amount.
During the year ended December 31, 2024, a total of $5,800,000 of convertible promissory notes were issued with a discount of 7%. These Notes are to be settled by way of cash or may be converted to Class A ordinary shares. The conversion of these notes are subject to certain criterions as mentioned in the Convertible Note Agreements, which include a beneficiary cap of 9.99% of shareholdings in the Company by JAK.
During the year ended December 31, 2024, a total of $2,650,000 of convertible notes were converted into 117 (adjusted by four Reverse Stock Splits in 2025 and two Reverse Stock Splits on January 26, 2026 and March 16, 2026) ordinary shares by the note holder, thereby increasing our shareholder equity by the same amount.
Interest expense amounted to $504,136 and $765,177 for the period ended December 31, 2025 and December 31, 2024 respectively and has been calculated based on the effective interest rate for each convertible note.
The transaction costs incurred on issuance of the Notes are capitalized and amortised over the term of the Notes as follows:
| December 31, 2025 |
December 31, 2024 |
|||||||
| Balance at beginning of the year | $ | 648,232 | $ | |||||
| Add: capitalized during the year | 564,000 | 929,500 | ||||||
| Less: charged to additional paid in capital for converted notes during the year | (777,836 | ) | ||||||
| Less: amortized during the year | (86,080 | ) | (281,268 | ) | ||||
| Ending balance | $ | 348,316 | $ | 648,232 | ||||
Derivative liability
Pursuant to ASC 815, a derivative liability had arisen from the issuance of convertible bonds which have the option of being converted to or exchanged for Class A ordinary shares at any time from date of issuance. The derivative liability is assessed to be a debt requiring to be bifurcated from the host contract and recorded at the fair value.
However, the conversion to ordinary shares is subject to certain terms and criteria as set within the Agreement, which includes restriction of conversion if the shareholdings by the note holder before and after the conversion exceeds 9.99%, for JAK & affiliated holders and Labrys Fund II, LP, as revised and 4.99% for Vanquish Funding Group Inc. As of December 31, 2025, the threshold has not been met, and accordingly the derivative is assessed to have a value of US$262,616 for the portions convertible to the extent of threshold, where relevant.
F-
Free standing instruments – warrants
During the financial year ending December 31, 2025 and 2024, JAK & affiliated holders were issued with 2,142,940 and 3,555,678 warrants respectively to purchase additional Class A ordinary shares at an exercise price of $2.71 and $2.71, respectively (as adjusted for the effects of the reverse stock split in May 2025).
The exercise of these warrants is subject to a 9.99% beneficial cap, as revised, which restricts the holder from exceeding shareholdings in excess of the cap in the Company. As of December 31, 2025 and 2024, the derivative is assessed to have a nil value in view of the cap assessed to have been met with the exercise of the tab convertible notes above.
| December 31, 2025 |
December 31, 2024 |
|||||||
| Warrant units: | ||||||||
| Balance at beginning of the year* | 3,555,678 | |||||||
| Warrants issued | 2,142,940 | 3,555,678 | ||||||
| Less : Exercised | (4,474,290 | ) | ||||||
| Ending balance | 1,224,328 | 3,555,678 | ||||||
| * | Adjusted for the effects of the RSS in May 2025. |
Public and Private – warrants
In connection with its initial public offering, the Company had issued 5,175,000 public warrants and 3,145,000 private warrants in a private placement transaction. Each warrant entitles the holder to purchase one ordinary share of the Company at an exercise price of $11.50 per share (subject to adjustment for share capital reorganisations and reverse stock splits).
The warrants may be exercised for cash or on a cashless basis at the holder’s option and are not redeemable by the Company. The warrants are exercisable commencing on the later of:
| ● | twelve months after the date of the IPO prospectus, and |
| ● | the completion of the Company’s initial business combination. |
The warrants expire five years after the completion of the business combination, or earlier upon liquidation of the Company.
As at 31 December 2025 and 2024, there were 5,175,000 public warrants and 3,145,000 private warrants outstanding.
During the periods since issuance, the Company completed multiple reverse stock splits. As a result, the number of ordinary shares issuable upon exercise of the warrants and the exercise price have been proportionately adjusted in accordance with the warrant agreement.
As at 31 December 2025, the market price of the Company’s ordinary shares was significantly below the adjusted exercise price of the warrants. Accordingly, the warrants were out of the money as at the reporting date. Management does not expect the warrants to be exercised unless the market price of the ordinary shares exceeds the exercise price prior to expiry.
NOTE 14 — LEASES
The Company has various operating leases for clinics and office spaces.
As of December 31, 2025 and December 31, 2024, the right-of-use assets totaled $504,192, and $98,570, respectively.
As of December 31, 2025 and December 31, 2024, lease liabilities consist of the following:
| 2025 | 2024 | |||||||
| Operating lease | ||||||||
| Lease liabilities – current portion | $ | 238,105 | $ | 108,526 | ||||
| Lease liabilities – non-current portion | 272,359 | 10,231 | ||||||
| Total | $ | 510,464 | $ | 118,757 | ||||
The following is a schedule of future minimum payments under operating leases as of December 31, 2025:
| Not later than 1 year | $ | 262,244 | ||
| Between 1 to 2 years | 184,041 | |||
| Between 2 to 5 years | 118,800 | |||
| Total lease payments | 565,085 | |||
| Less: imputed interest | (54,621 | ) | ||
| Total operating lease liabilities, net of interest | $ | 510,464 |
F-
Other lease information is as follows:
| 2025 | 2024 | |||||||
| 2.37 years | 2.75 years | |||||||
| Weighted-average discount rate – operating leases | 5 | % | 5 | % | ||||
| Short term lease cost | $ | 207,787 | $ | 234,767 | ||||
As of December 31, 2025 and December 31, 2024, there were $504,192 and $98,570 right of use (“ROU”) assets and $510,464 and $118,757 lease liabilities based on the present value of the future minimum rental payments of leases, respectively. The Company’s management believes that using an incremental borrowing rate of the minimum loan rate and the Hong Kong Dollar Best Lending Rate (“BLR”) minus 0.125% was the most indicative rate of the Company’s borrowing cost for the calculation of the present value of the lease payments; the rate used by the Company was 6.6% and 5.5% respectively.
NOTE 15 — EQUITY
Ordinary shares
As at December 31, 2025 and December 31, 2024, the Company is authorized to issue unlimited and 50,000,000 ordinary shares respectively. Each ordinary share is entitled to one vote. The holders of ordinary shares are also entitled to receive dividends whenever funds are legally available and when declared by the Board of Directors of the Company. On July 4, 2025, the Board of Directors of the Company approved unlimited authorized ordinary share.
The equity of the Company as of December 31, 2025 and December 31, 2024 represents 240,622 and 380 ordinary shares with par value of $Nil. The number of shares as above have taken into consideration the reverse stock split effected on February 11, 2025, May 6, 2025 and August 4, 2025, December 1, 2025, January 26, 2026 and March 16, 2026 at an exchange ratio of one (1) share for twenty (20) shares, one (1) share for ten (10) shares and one (1) share for five (5) share, one (1) share for five (5) shares, one (1) share for three (3) and one (1) share for four (4) shares, respectively. Pursuant to ASC 505 the reverse stock split has been applied retrospectively.
Additional paid-in capital
| Balance as at January 1,2023 | $ | 1,464,959 | ||
| Issuance of shares as compensation for services (Note 1) | 2,866,856 | |||
| Balance as at December 31, 2023 | 4,331,815 | |||
| Issuance of shares through Equity Line of Credit (Note 2) | 1,936,236 | |||
| Issuance of shares through conversion of convertible notes | 2,650,000 | |||
| Decrease in subscription receivable due to reduction of work by professionals (Note 1) | (2,766,856 | ) | ||
| Business Combination (Note 3) | (6,028,690 | ) | ||
| Balance as at December 31, 2024 | $ | 122,505 | ||
| Issuance of shares for share-based compensation to consultant (Note 4) | 20,250 | |||
| Issuance of shares through Equity Line of Credit (Note 5) | 12,620,494 | |||
| Issuance of shares through conversion of convertible notes | 4,860,427 | |||
| Issuance of shares for business acquisition | 257,500 | |||
| Balance as at December 31, 2025 | $ | 17,881,176 |
| * | the number of shares below have been amended to apply the reverse stock split in February 11, May 6, 2025 and August 4, 2025, December 1, 2025, January 26, 2026 and March 16, 2026 retrospectively. |
Note 1: On January 10, 2023, the Company issued 1,365 ordinary shares (0.45 Ordinary Shares as adjusted by all Reverse Stock Splits done as of this report) to professional party for consulting service of 10 years, increasing the additional paid-in capital by $812,300. On December 4, 2023, the Company issued additional 3,450 shares (1.15 Ordinary Shares as adjusted by all Reverse Stock Splits done as of this report) to DoubleClick Services Limited for consulting service of 10 years, increasing the additional paid-in capital by $2,054,556. During 2024, the service scope and duration were reduced and the service value was adjusted accordingly pursuant to addendums to respective agreements Note 2: On November 21, 2024, the Company entered into a Common Shares Purchase Agreement (the “White Lion Purchase Agreement”) with White Lion Capital, LLC (“White Lion”) and a related Registration Rights Agreement (the “RRA”).
F-
Pursuant to the White Lion Purchase Agreement, the Company has the right, but not the obligation, to require White Lion to purchase, within 36 months from the Agreement effective date up to One Hundred Million Dollars ($100,000,000) in aggregate gross purchase price of newly issued Ordinary Shares, with an automatic increase to Three Hundred Million Dollars ($300,000,000) upon any substantial M&A or Material Transaction (as defined in the White Lion Purchase Agreement) and a further option to increase to Five Hundred Million Dollars ($500,000,000) after Two Hundred and Fifty Million Dollars ($250,000,000) has been issued and sold to White Lion under the White Lion Purchase Agreement, subject to certain limitations and conditions set forth in the White Lion Purchase Agreement. The White Lion Purchase Agreement was included as Exhibit 10.27 of the Form F-1 registration statement filed on December 5, 2024.
On December 10, 2024, 35,000 Class A Ordinary Shares (0.58 Ordinary Shares as adjusted by all reverse stock splits done as of this report) were issued to White Lion amounting $276,850, in consideration for White Lion’s commitment pursuant to the White Lion Purchase Agreement. In addition, on December 12, 2024, pursuant to this Agreement, the Company issued 9,500 (0.15 Ordinary Shares as adjusted by all reverse stock splits done as of this report) Class A Ordinary Shares for a purchase price of $1,572,186. On December 24, 2024, the Company issued 25,000 Class A Ordinary Shares (0.42 Ordinary Shares as adjusted by all reverse stock splits done as of this report) for a purchase price of $160,050. On December 31, 2024, the Company issued 30,000 Class A Ordinary Shares (0.5 Ordinary Shares as adjusted by all reverse stock splits done as of this report) for a purchase price of $204,000.
Note 3: Upon de-spac business combination, additional paid-in capital and capital reserve of Legacy NewGenIvf was combined with additional paid-in capital and accumulated net losses of the A SPAC I Acquisition Corp and added into the additional paid-in capital of NewGenIvf Group upon the reverse take-over.
Note 4: The Company had issued 50 Class A Ordinary Shares to A SPAC (Holdings) Group Corp, pursuant to a one year consulting services agreement commencing on February 3, 2025.
Note 5: During the year ended December 31, 2025, the Company had issued 141,661 shares after taking account of the reverse stock split on Feb 11, 2025 and May 6, 2025, August 3, 2025, December 1, 2025, January 26, 2026 and March 16, 2026 respectively.
Note 6: The Company had issued 125,000 Class A Ordinary Shares (42 Ordinary Shares as adjusted by all reverse stock splits done as of this report) valued at US$2.06 per share for acquiring of MicroSort Reproductive Technology on January 21, 2025.
NOTE 16 — EMPLOYEE BENEFIT PLANS
HK SAR
The Company has a defined contribution pension scheme for its qualifying employees. The scheme assets are held under a provident fund managed by an independent fund manager. The Company and its employees are each required to make contributions to the scheme calculated at 5% of the employees’ basic salaries on monthly basis.
Thailand
The Company is obliged to make social security payments within the first 15 days of the month over which it is accrued. During period ended December 31 2025, the Company and its employees are each required to make contributions to the scheme calculated at 5% of the employee’s salary with a cap of THB750 per month.
F-
Cambodia
Every business employing one or more workers must register its business and workers with the National Social Security Fund (the “NSSF”) for the Occupational Risk Scheme (for work-related accidents and occupational diseases), the Health Care Scheme and the Pension Scheme.
Once registered, the business must pay to the NSSF:
| ● | A monthly contribution equivalent to 0.8% of each worker’s monthly average wages (between $0.40 and $2.40 per month per worker) for the Occupational Risk Scheme. |
| ● | A monthly contribution equivalent to 2.6% of a worker’s monthly average wages (between $1.30 and $7.80 per month per worker) for the Health Care Scheme. |
| ● | A monthly contribution to the compulsory Pension Scheme, which is jointly paid by the employer and the employee at the same rate of 2% (total of 4%) of the contributable wage for the first five years. The contributable wage for the Pension Scheme ranges from between KHR400,000 (approximately $100) up to KHR1,200,000 (approximately $300). |
Kyrgyzstan
The Company has a defined contribution pension scheme for its qualifying employees. The scheme assets are held under a provident fund managed by an independent fund manager. The Company and its employees are each required to make contributions to the scheme calculated at 15% and 8%, respectively of the employees’ basic salaries on monthly basis.
NOTE 17 — PROVISION FOR INCOME TAXES
Cayman Islands
NewGenIvf Limited was incorporated in the Cayman Islands and is not subject to tax on income or capital gains under current Cayman Islands law. In addition, upon payment of dividends by these entities to the shareholders, no Cayman Islands withholding tax will be imposed.
HK SAR
Under the two-tiered profits tax rates regime, Hong Kong tax residents are subject to Hong Kong Profits Tax in respect of profits arising in or derived from Hong Kong at 8.25% for the first HK$2 million of profits of the qualifying group entity, and profits above HK$2 million will be taxed at 16.5%. The profits of group entities not qualifying for the two-tiered profits tax rates regime will continue to be taxed at a flat rate of 16.5%.
Accordingly, the HK SAR profits tax is calculated at 8.25% on the first HK$2 million of the estimated assessable profits and at 16.5% on the remaining estimated assessable profits.
Thailand
The companies incorporated in Thailand are taxed on worldwide income. A company incorporated abroad is taxed on its profits arising from or in consequence of the business carried on in Thailand. The corporate income tax (CIT) rate is 20%. A foreign company not carrying on business in Thailand is subject to a final withholding tax (WHT) on certain types of assessable income (e.g. interest, dividends, royalties, rentals, and service fees) paid from or in Thailand. The rate of tax is generally 15%, except for dividends, which is 10%, while other rates may apply under the provisions of a double tax treaty (DTT).
F-
Cambodia
The standard rate of corporate income tax (“CIT”) for companies and permanent establishments who are classified as medium and large taxpayers is 20%. For companies and permanent establishments who are classified as small taxpayers, the CIT rates are progressive rates from 0% to 20%. In view of the annual turnover of the company, the annual turnover ranges from KHR1 billion to KHR6 billion for service and commercial sectors, the company shall consider as the medium-sized company.
Kyrgyzstan
The company is subject to a corporate income tax on their aggregate annual income earned worldwide. Non-resident legal entities carrying out business activities through a permanent establishment in Kyrgyzstan are subject to profit tax on the income attributed to the activities of that permanent establishments. During the period ended June 30, 2025, no tax payable is accrued and reversed accordingly.
Profit tax is calculated at a rate of 10% of aggregate annual income less allowed deductions.
U.S.A.
NewGenIvf is subject to a corporate income tax on its aggregate annual income earned worldwide from its U.S. operations at a flat 21% on net income. In addition to federal obligations, there is a graduated state income tax rate that applies to corporate income, with a top rate of 6.6% on income over $60,000. Moreover, when the Company’s US operation pays a dividend to its offshore parent company, the payment is generally subject to a US federal withholding tax, typically at a statutory rate of 30% on the gross amount.
Significant components of the provisions for income taxes for the years ended December 31, 2024, and December 31, 2025 were as follows:
| December 30, 2025 |
December 31, 2024 |
|||||||
| Current tax provision Kyrgyzstan | $ | $ | ||||||
| Over provision of tax in prior year Kyrgyzstan | (486,706 | ) | ||||||
| Current tax provision Cambodia | ||||||||
| Total tax (income) expense | $ | $ | (486,706 | ) | ||||
| December 31, 2025 |
December 31, 2024 |
|||||||
| Profit / (Loss) Income before taxes | $ | 9,728,723 | $ | (960,807 | ) | |||
| Tax credit (expense) at the effective tax rates | 1,705,862 | (179,863 | ) | |||||
| Tax effect on non-taxable income | (3,682,167 | ) | (81,323 | ) | ||||
| Tax effect on non-deductible expenses | 1,975,290 | 129,814 | ||||||
| Change in valuation allowance | 131,991 | |||||||
| Tax effect on utilization of tax losses | (127,521 | ) | ||||||
| Tax losses unable to be utilized | 1,015 | 126,902 | ||||||
| Over provision of tax in prior year | (486,706 | ) | ||||||
| Tax (income) expense | $ | $ | (486,706 | ) | ||||
F-
Deferred tax asset, net
Significant components of deferred tax assets, net were as follows:
| December 31, 2025 |
December 31, 2024 |
|||||||
| USD | USD | |||||||
| Deferred tax assets: | ||||||||
| – Net operating loss carry forward | 160,432 | |||||||
| Less: valuation allowance | (160,432 | ) | ||||||
| Deferred tax assets, net | ||||||||
As of December 31, 2025 and December 31, 2024, the Company had net operating loss carry forward of Nil and $160,432. The Company believes it is less likely than not that its operations will be able to fully utilize its deferred tax assets related to the net operating loss carry forward. As a result, the Company provided 100% allowance on deferred tax assets on net operating loss.
NOTE 18 — DISAGGREGATED REVENUES
The Company’s main business operations are to provide: (i) IVF treatment service; and (ii) fertility referral services.
| Revenue from external customers | December 31, 2025 |
December 31, 2024 |
||||||
| IVF treatment service | $ | 3,905,863 | $ | 5,433,375 | ||||
| Fertility referral services | 820,570 | |||||||
| Total revenues | $ | 4,726,433 | $ | 5,433,375 | ||||
Geographical information
| Revenue from external customers originated from | December 31, 2025 |
December 31, 2024 |
||||||
| China / HK SAR | 41,555 | $ | ||||||
| Kyrgyzstan | 2,312,348 | 2,656,596 | ||||||
| Cambodia | 635,188 | 601,526 | ||||||
| Thailand | 1,691,862 | 2,175,253 | ||||||
| US | 45,480 | |||||||
| Total revenues | 4,726,433 | $ | 5,433,375 | |||||
The revenue information above is based on the locations where the revenue originated.
| Long-lived assets located at | December 31, 2025 |
December 31, 2024 |
||||||
| HK SAR | $ | 209,630 | 3,081 | |||||
| US | 22,832,236 | |||||||
| China | 8,370 | |||||||
| Cambodia | 354,660 | 47,902 | ||||||
| Thailand | 311,845 | 312,313 | ||||||
| $ | 23,708,371 | 371,666 | ||||||
The Company’s long-lived assets consist of plant and equipment, net, intangible assets, net and operating leases right-of-use assets, net.
F-
NOTE 19 — RISKS
A. Credit risk
Accounts receivable
In order to minimize the credit risk, the management of the Company monitors and ensures that follow-up action is taken to recover overdue debts. The Company considers the probability of default upon initial recognition of asset and whether there has been a significant increase in credit risk on an ongoing basis throughout each reporting period. To assess whether there is a significant increase in credit risk, the Company compares the risk of a default occurring on the asset as at the reporting date with the risk of default as at the date of initial recognition. It considers available reasonable and supportive forward-looking information, such as GDP growth rate and nominal GDP per capita. Based on the impairment assessment performed by the Company, the directors consider the loss allowance for account receivables as of December 31, 2025 and December 31, 2024 to be $21 and $19 respectively.
Cash and cash equivalents
The credit risk on liquid funds is limited because the counterparties are banks with high credit ratings assigned by international credit-rating agencies. The Company is exposed to concentration of credit risk on liquid funds which are deposited with several banks with high credit ratings.
Deposits, prepayments and other receivables
The Company assessed the impairment for deposits and other receivables, due from shareholders individually based on internal credit rating and ageing of these debtors which, in the opinion of the directors, have no significant increase in credit risk since initial recognition. Based on the impairment assessment performed by the Company, the management consider the loss allowance for deposits and other receivables, due from shareholders as of December 31, 2025 and December 31, 2024 is $5, and $4, respectively. The loss allowance for deposits and other receivables, due from shareholders as of December 30, 2025 and December 31, 2024 is $0, and $0, respectively.
Receivables from agents
The Company assessed the impairment for receivables from agents based on ageing of these debts and the recoverability of the deposits distributed by the agents to their sub-agents. Based on the assessment performed by the Company, the management have written off an amount of $818,780 during the year which were deemed not recoverable. The loss allowance for receivables from agents as of December 31, 2025 and December 31, 2024 were $nil.
B. Interest risk
Cash flow interest rate risk
The Company is exposed to cash flow interest rate risk through the changes in interest rates related mainly to the Company’s variable-rates bank balances.
The Company currently does not have any interest rate hedging policy in relation to fair value interest rate risk and cash flow interest rate risk. The directors monitor the Company’s exposures on an ongoing basis and will consider hedging the interest rate should the need arises.
Sensitivity analysis
The sensitivity analysis below has been determined by assuming that a change in interest rates had occurred at the end of the reporting period and had been applied to the exposure to interest rates for financial instruments in existence at that date. 1% increase or decrease is used when reporting interest rate risk internally to key management personnel and represents management’s assessment of the reasonably possible change in interest rates.
F-
If interest rates had been 1% higher or lower and all other variables were held constant, the Company’s net (loss) income for the period/years ended December 31, 2025 and December 31, 2024 would have increased or decreased by approximately $52,890 and $26,894, respectively.
Foreign currency risk
Foreign currency risk is the risk that the holding of foreign currency assets will affect the Company’s financial position as a result of a change in foreign currency exchange rates.
The Company’s monetary assets and liabilities are mainly denominated in HK$, THB and RMB which are the same as the functional currencies of the relevant group entities. Hence, in the opinion of the directors of the Company, the currency risk of US$ is considered insignificant. The Company currently does not have a foreign currency hedging policy to eliminate currency exposures. However, the directors monitor the related foreign currency exposure closely and will consider hedging significant foreign currency exposures should the need arise.
Market Liquidity Risk
NewGenIvf is exposed to market liquidity risk with respect to certain assets held on our balance sheet. Market liquidity risk is the risk that we cannot realize the fair value of our assets when needed due to insufficient market depth, disruption, or the absence of willing buyers. As of December 31, 2025, we held Solana as treasury investment. These assets trade in markets that may experience periods of reduced liquidity, particularly during periods of economic stress or market disruption.
C. Economic and political risks
The Company’s operations are mainly conducted in Thailand, Cambodia and Kyrgyzstan. Accordingly, the Company’s business, financial condition, and results of operations may be influenced by changes in the political, economic, and legal environments in Thailand, Cambodia and Kyrgyzstan.
The Company’s operations in Thailand, Cambodia and Kyrgyzstan are subject to special considerations and significant risks. These include risks associated with, among others, the political, economic and legal environment and foreign currency exchange. The Company’s results may be adversely affected by changes in the political and social conditions in Thailand, Cambodia and Kyrgyzstan, and by changes in governmental policies with respect to laws and regulations, anti-inflationary measures, currency conversion, remittances abroad, and rates and methods of taxation, among other things including natural disasters and wars.
The recent war involving the US, Israel, and Iran has severely disrupted international travel, beginning with the immediate closure of airspace over several Middle Eastern countries and the suspension of flights through critical hubs like Dubai and Doha, which has stranded hundreds of thousands of passengers. This has forced airlines to cancel thousands of flights or take longer, more expensive routes, leading to a surge in ticket prices and the imposition of new fuel surcharges that are affecting travelers globally. Governments have responded by issuing widespread “do not travel” warnings for the region and organizing evacuations, while embassies have halted routine visa services, further complicating international mobility. The compounded effect is a climate of uncertainty where even trips not destined for the Middle East face potential delays, higher costs, and complex rebooking challenges.
D. Inflation risk
Management monitors changes in prices levels. Historically inflation has not materially impacted the Company’s consolidated financial statements; however, significant increases in the price of labor that cannot be passed to the Company’s customers could adversely impact the Company’s results of operations. Moreover, the recent escalation of the US-Israel-Iran conflict poses a significant threat to the global economy, primarily through the disruption of oil supplies, with cascading effects on the real estate sector in the UAE and a broad economic downturn across Asia, including Hong Kong and China. As the conflict intensifies around the Strait of Hormuz—through which nearly 20% of the world’s oil passes—global oil prices have surged past $100 per barrel, raising fears of an extended energy crisis. Such increase in oil prices might lead to domino effect on the overall costs of business operation for NewGenIvf and NewGenIvf might not be able to pass the cost pressure to its customers.
F-
NOTE 20 — RELATED PARTY BALANCES AND TRANSACTIONS
The summary of amount due from and due to related parties as the following:
| Relationship | December 31, 2025 |
December 31, 2024 |
||||||||
| Due from shareholders consist of the following: | ||||||||||
| Mr. Siu Wing Fung, Alfred (“Mr. Siu”) | Shareholder and director | $ | $ | 404,549 | ||||||
| Harcourt Limited | A related party (note 1) | $ | 110,232 | |||||||
| Due to a related party consist of the following: | ||||||||||
| Mr. Siu Wing Fung, Alfred (“Mr. Siu”) | Shareholder and director | (116,653 | ) | |||||||
| Ms. Fong Hei Yue, Tina (“Ms. Fong”) | Shareholder and director | (497,200 | ) | |||||||
| Harcourt Limited | A related party (note 1) | $ | $ | (61,802 | ) | |||||
Note:
| (1) | The directors and shareholders of Harcourt Limited are Mr. Siu and Ms. Fong, Harcourt Limited therefore has the common ultimate beneficial owners with the Company. |
The balance due from shareholders consist of the following:
| December 31, 2025 |
December 31, 2024 |
|||||||
| Due from/ (to) shareholders/ related party | $ | (6,421 | ) | $ | (154,453 | ) | ||
| Less: allowance for expected credit loss | ||||||||
| $ | (6,421 | ) | $ | (154,453 | ) | |||
The movement of allowances for expected credit loss is as follow:
| December 31, 2025 |
December 31, 2024 |
|||||||
| Balance at beginning of the year | $ | $ | (17,818 | ) | ||||
| Reversal of Provision / (Provision) | (17,818 | ) | ||||||
| Ending balance | $ | $ | ||||||
In addition to the transactions and balances detailed elsewhere in these consolidated financial statements, the Company had the following transactions with related parties:
| December 31, 2025 |
December 31, 2024 |
|||||||
| Directors’ remuneration to Mr. Siu Wing Fung, Alfred | $ | 325,000 | $ | 190,000 | ||||
| Directors’ remuneration to Ms. Fong Hei Yue, Tina | 325,000 | 190,000 | ||||||
| Director’s bonus of Mr. Siu Wing Fung, Alfred | 750,000 | |||||||
| Director’s welfare, accommodation and insurance to Mr. Siu Wing Fung, Alfred | 393,103 | |||||||
| Advisory services on fund raising project to Mr Siu, Daniel | 219,045 | |||||||
| Consulting services on fund raising project to Mr Siu, Jasper | 65,000 | |||||||
| Marketing service fee to Harcourt Limited | 230,769 | |||||||
F-
NOTE 21 — BUSINESS COMBINATIONS
Acquisition of MicroSort Lab Services, LLC
On February 28, 2025, NewGenIvf Group Limited (the “Company”) completed the acquisition of 100% of the outstanding equity interests of MicroSort Lab Services, LLC (“MicroSort”), a Delaware limited liability company established by Genetics & IVF Institute, Inc. (“Seller”). The Seller transferred to MicroSort a portfolio of assets including registered trademarks, domain names, customer contracts, and a consultant agreement. MicroSort technology is a form of pre-conception gender selection technology for humans and this Preimplantation Genetic Screening (“PGS”) is used in parallel with an IVF treatment cycle. Prior to the acquisition, the Company had been a licensee of the MicroSort® technology in Thailand and Cambodia. The company’s strategic acquisition of the MicroSort technology provide the company with the exclusive right to this technology for its continued use within its clinics as well as to licence it out to other clinics globally. In accordance with ASC 805, The acquisition was accounted for as a business combination in accordance with ASC 805, Business Combinations, as the Company obtained control of an integrated set of activities and assets that constitute a business.
The total purchase consideration amounted to US$5,000,000, consisting of cash consideration of US$750,000 and 2,500,000 Class A Ordinary Shares of the Company (42 shares, post–reverse stock splits) at a deemed share price of $1.7 per share. After the 2nd reverse stock split in February 2025, the Company issued 125,000 shares were when the share price was $2.06 per share. Accordingly, the share consideration was valued at US$257,500 at the acquisition date. The total fair value of the purchase consideration on acquisition date amounts to $1,007,500 measured in accordance with ASC 805-30-30-7. No contingent consideration or seller financing arrangements were included in the transaction.
The Company engaged an independent valuation firm, to determine the fair value of the acquired trademarks and technology. Using the income approach (discounted cash flow method), the fair value of the trademarks and technology were determined to be US$5,284,000 as of December 31, 2024. The acquired trademark was valued at $5,284,000, using an income approach, primarily applying a discounted cash flow methodology, which considered projected revenues attributable to the trademark, licensing income and expenses, and a risk-adjusted discount rate.
The company obtained control over the Company and its related business and assets on February 28, 2025. Accordingly, February 28, 2026 which been determined by management as the acquisition date. No liabilities were assumed as part of the transaction. As at acquisition date management had re-evaluated the fair value assessment of the trademarks and determined the fair value to be $3.5 million. As the fair value of net assets acquired exceeded the fair value of the purchase consideration, the Company recognized a bargain purchase gain of US$2.5 million, in accordance with ASC 805-30-25-2. In accordance with ASC 805-30-25-2, the Company reassessed the identification and measurement of all acquired assets and liabilities and the consideration transferred. Management concluded that the bargain purchase gain resulted primarily from the Seller’s decision to exit non-core operations, as well as a decline in the Company’s stock price between the signing date and the Acquisition Date.
The fair values of the identifiable assets and liabilities of MicroSort, as at the date of acquisition were as follows:
| Fair value recognized on acquisition |
||||
| Intangible Asset – intellectual property and trademark | 3,495,335 | |||
| Liabilities assumed | ||||
| Total identifiable net assets at fair value | 3,495,335 | |||
| Cash consideration | (750,000 | ) | ||
| Share consideration | (257,500 | ) | ||
| Bargain purchase gain | 2,487,835 | |||
F-
MicroSort did not have any trade receivables outstanding as of the Acquisition Date. Accordingly, no receivables were recognized as part of the business combination.
Acquisition-related transaction costs were immaterial and were expensed as incurred within general and administrative expenses in the consolidated statements of operations
The intangible assets have been classified as indefinite-lived intangible assets acquired will be subject to annual impairment testing in accordance with ASC 350. The bargain purchase gain has been which has been recorded as a separate line item within Other income (expense), net in the Consolidated Statements of Operations for the year ended December 31, 2025.
The MicroSort business, while being established by the Seller in prior years, it had not generated significant revenue and earnings prior to the acquisition due to MicroSort not being the core business focus of the Seller Accordingly, the unaudited pro forma consolidated revenue and net income of the Company, as if the acquisition had occurred on January 1, 2024, would not have differed materially from the amounts reported in the Company’s consolidated financial statements.
Acquisition of Nodexus business.
On July 24, 2025, NewGenIvf Group Limited (the “Company”) acquired substantially all of the operating assets of Nodexus Inc (“Nodexus”) through an assignment for the benefit of creditors process administered by Sherwood Partners, Inc. The acquired assets included a patent portfolio and proprietary cell-sorting technology (five granted U.S. patents and one granted Japan patent), 18 Nodexus NX One units, partially constructed NX One units, cartridges, spare parts, and the Nodexus.com domain. Nodexus technology is a form of pre-conception gender selection technology for humans and this Preimplantation Genetic Screening (“PGS”) is used in parallel with an IVF treatment cycle. The company’s strategic acquisition of the Nodexus technology provide the company with the exclusive right to this technology for its continued use within its clinics as well as to license it out to other clinics globally. In accordance with ASC 805, The acquisition was accounted for as a business combination in accordance with ASC 805, Business Combinations, as the Company obtained control of an integrated set of activities and assets that constitute a business. Although Nodexus did not generate revenue and did not have outputs as of the acquisition date, management concluded that the acquired set met the definition of a business under ASC 805-10-55-5A through 55-9 because it included inputs and a substantive process that together are capable of producing outputs.
The Company engaged an independent valuation of the acquired patents. Using the relief-from-royalty method, the fair value of the patents was determined to be US$17,900,000 primarily applying a relief-from-royalty method, which estimates the present value of royalty payments avoided by owning the technology rather than licensing it. Key assumptions included estimated market-participant revenue projections, an indicative royalty rate, a discount rate reflective of early-stage technological risk. The Company also estimated the fair value of other acquired assets at US$1,566,000. No liabilities were assumed as part of the transaction. The aggregate fair value of net assets acquired was US$19,466,000. As the fair value of net assets acquired significantly exceeded the consideration transferred, the Company recognized a bargain purchase gain of US$19,166,000 in accordance with ASC 805-30-25-2. The gain reflects the distressed nature of the sale process, limited market participants, and the Seller’s inability to execute commercialization, rather than any measurement error.
F-
The total cash consideration paid was US$300,000. The company obtained control over the Company and its related business and assets on July 29, 2025. Accordingly, July 29, 2025 has been determined by management as the acquisition date. No liabilities were assumed as part of the transaction. The Company also estimated the fair value of other acquired assets at US$1,566,000. No liabilities were assumed as part of the transaction. The aggregate fair value of net assets acquired was US$19.5 million. As the fair value of net assets acquired significantly exceeded the consideration transferred, the Company recognized a bargain purchase gain of US$19,166,000 in accordance with ASC 805-30-25-2. In accordance with ASC 805-30-25-2, the Company reassessed the identification and measurement of all acquired assets and liabilities and the consideration transferred. Management concluded that the gain represents a unique, non-recurring event arising from the acquisition of high-potential assets in a forced liquidation process at a price substantially below fair value.
The fair values of the identifiable assets and liabilities of Nodexus, as at the date of acquisition were as follows:
| Fair value recognized on acquisition |
||||
| Intangible assets - patented Technology | 17,900,000 | |||
| Nodexus NX One Unit | 1,530,000 | |||
| Catridges for Nodexus NX | 16,000 | |||
| Nodexus.com domain and hosting access | 20,000 | |||
| Liabilities assumed | ||||
| Total identifiable net assets at fair value | 19,466,000 | |||
| Cash consideration | (300,000 | ) | ||
| Bargain purchase gain | 19,166,000 | |||
Nodexus did not have any trade receivables outstanding as of the Acquisition Date. Accordingly, no receivables were recognized as part of the business combination.
Acquisition-related transaction costs were immaterial and were expensed as incurred within general and administrative expenses in the consolidated statements of operations.
The tangible assets will be amortized on a straight-line basis over their estimated useful life of 5 years in accordance with ASC 360. The intangible assets have been classified as indefinite-lived intangible assets acquired will be subject to annual impairment testing in accordance with ASC 350. The bargain purchase gain has been which has been recorded as a separate line item within Other income (expense), net in the Consolidated Statements of Operations for the year ended December 31, 2025.
Nodexus was a development-stage entity and did not generate revenue or earnings prior to the acquisition. Accordingly, the unaudited pro forma consolidated revenue and net income of the Company, as if the acquisition had occurred on January 1, 2024, would not have differed materially from the amounts reported in the Company’s consolidated financial statements for the periods presented. While the Nodexus business had been established in prior years, it had not generated significant revenue and earnings prior to the acquisition due to it being used primarily for research and development purposes. Accordingly, the unaudited pro forma consolidated revenue and net income of the Company, as if the acquisition had occurred on January 1, 2024, would not have differed materially from the amounts reported in the Company’s consolidated financial statements.
F-
NOTE 22 — DISPOSAL OF SUBSIDIARY
On July 29, 2025, the Company completed the sale of its Shenzhen Qianhai Fengtai Renhui Health Technology Co., Ltd. (“SZ QianHai”) to an unrelated third party for total consideration of US$ 0 (RMB 1), consisting of cash.
As part of the disposal, all the outstanding account payables and receivables as well as all outstanding balances were transferred to the Buyer. Any tax liabilities in People’s Republic of China (PRC) arising from the transfer of these balances shall be borne by the Buyer.
The following table summarizes the financial impact of the disposal:
| ● | Consideration received: Cash of US$ 0 (RMB 1) |
| ● | Net assets disposed of $47,702 (RMB 352,826) |
| ● | Loss on disposal of subsidiary of $47,702 (RMB 352,826) |
The disposal was accounted for in accordance with ASC 810, Consolidation. As a result, the Company deconsolidated Shenzhen Qianhai Fengtai Renhui Health Technology Co. and recognized a loss on disposal of subsidiary (after netting against the cash consideration received) in the amount of US$47,702 in other income, net in the consolidated statements of operations for the year ended December 31, 2025.
The disposal did not represent a strategic shift and, therefore, was not classified as a discontinued operation under ASC 205-20.
NOTE 23 — COMMITMENTS & CONTINGENCIES
As of December 31, 2025 and December 31, 2024, the Company was not a party to any legal or administrative proceedings. As of December 31, 2025, the Company had commitment as described in Notes 13 and 14 to the financial statements.
The Company is also committed to honor its obligations pursuant to the convertible note agreements as described in Note 13.
NOTE 24 — SUBSEQUENT EVENTS
The Company evaluated subsequent events and transactions that occurred after the balance sheet date up to the date that the financial statements were issued. Based upon this review, other than as described below, the Company did not identify any subsequent events that would have required adjustment or disclosure in the financial statements.
Subsequent to December 31, 2025 and to the date of this report, $600,000 convertible notes outstanding as of December 31, 2025 had been converted to additional 190,319 Class A ordinary shares (adjusted by Reverse Stock Splits on December 2, 2025, January 26, 2026 and March 16, 2026).
On January 13, 2026, NewGenBiz Limited, a 100% directly owned subsidiary of NewGenIvf Group Limited, was incorporated in British Virgin Islands as an investment holding company for future business.
On January 22, 2026, the Company entered into a Securities Purchase Agreement with Vanquish Funding Group Inc., pursuant to which, amongst other things, the Company agreed to sell a convertible note in the aggregate principal amount of $107,000, convertible into Class A Ordinary Shares pursuant to its terms.
On January 22, 2026, the Company entered into a Securities Purchase Agreement with Boot Capital LLC, pursuant to which, amongst other things, the Company agreed to sell a convertible note in the aggregate principal amount of $50,000, convertible into Class A Ordinary Shares pursuant to its terms.
On February 2, 2026, NewGenOman Limited, a 100% directly owned subsidiary of NewGenIvf Group Limited, was incorporated in British Virgin Islands as an investment holding company for future business.
On February 27, 2026, NewGenBiz Limited, a 100% directly owned subsidiary of NewGenIvf Group Limited, was incorporated in Hong Kong as a foundation for charitable activities.
On March 17, 2026, the Company entered into a Securities Purchase Agreement with CFI Capital LLC, pursuant to which, amongst other things, the Company agreed to sell a convertible note in the aggregate principal amount of $200,000, convertible into Class A Ordinary Shares pursuant to its terms.
On March 25, 2026, the Company entered into a Securities Purchase Agreement with Firstfire Global Opportunities Fund, LLC, pursuant to which, amongst other things, the Company agreed to sell a convertible note in the aggregate principal amount of $220,000, convertible into Class A Ordinary Shares pursuant to its terms.
F-
Exhibit 2.5
DESCRIPTION OF THE
REGISTRANT’S SECURITIES REGISTERED PURSUANT TO
SECTION 12 OF THE SECURITIES EXCHANGE ACT OF 1934
As of December 31, 2025, NewGenIvf Group Limited (the “Company”, “we”, “us” and “our”) had the following series of securities registered pursuant to Section 12(b) of the Exchange Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||
| Class A ordinary shares, no par value per share | NIVF | The Nasdaq Stock Market LLC | ||
| Warrants | NIVFW | The Nasdaq Stock Market LLC |
Description of Ordinary Shares
The following is a summary of material provisions of our amended and restated memorandum and articles of association (the “Memorandum and Articles of Association”), as well as the BVI Business Companies Act (As Revised) (the “BVI Act”) insofar as they relate to the material terms of our Ordinary Shares. Notwithstanding this, because it is a summary, it may not contain all the information that you may otherwise deem important. For more complete information, you should read the entire Memorandum and Articles of Association, which is filed under Exhibit 1.1 to this Report.
Type and Class of Securities (Item 9.A.5 of Form 20-F)
The Memorandum and Articles of Association authorize the issuance of unlimited Class A Ordinary Shares, unlimited Class B Ordinary Shares and unlimited preferred shares with no par value (“Preferred Shares”). As of the date of this Report, we have 568,473 Class A Ordinary Shares outstanding, 69 Class B Ordinary Shares outstanding, and no Preferred Shares outstanding. All of our outstanding Class A Ordinary Shares are validly issued, and fully paid.
Preemptive Rights (Item 9.A.3 of Form 20-F)
Our shareholders do not have preemptive rights.
Limitations or Qualifications (Item 9.A.6 of Form 20-F)
We have a dual-class voting structure such that our issued Ordinary Shares consist of Class A Ordinary Shares and Class B Ordinary Shares. In respect of all matters upon which holders of Ordinary Shares are entitled to vote, each Class A Ordinary Share are entitled to one (1) vote and each Class B Ordinary Share are entitled to one hundred (100) votes. Holders of Class A Ordinary Shares and Class B Ordinary Shares generally have the same rights and powers except for voting and conversion rights.
Hei Yue Tina Fong and Wing Fung Alfred Siu control the voting power of all of the outstanding Class B Ordinary Shares. Although Ms. Fong and Mr. Siu control the voting power of all of the outstanding Class B Ordinary Shares, their control over those shares is not permanent and is subject to reduction or elimination at any time or after certain periods as a result of a variety of factors. As further described below, upon any transfer of Class B Ordinary Shares by a holder thereof to any person which is not a Affiliate (as defined below) of such holder, those shares will automatically and immediately convert into Class A Ordinary Shares. In addition, all Class B Ordinary Shares will automatically convert to Class A Ordinary Shares in other events described below. See “— Optional and Mandatory Conversion.”
Rights of Class A Ordinary Shares (Item 10.B.3 of Form 20-F)
Classes of Ordinary Shares
Our Ordinary Shares are divided into Class A Ordinary Shares and Class B Ordinary Shares (and a further class of authorized but undesignated shares). Except for conversion rights and voting rights, the Class A Ordinary Shares and Class B Ordinary Shares shall generally carry equal rights and rank pari passu with one another.
Conversion
Subject to any applicable adjustment pursuant to the Memorandum and Articles of Association, each Class B Ordinary Share is convertible into one (1) Class A Ordinary Share at any time at the option of the holder thereof. The right to convert shall be exercisable by the holder of the Class B Ordinary Share delivering a written notice to the Company that such holder elects to convert a specified number of Class B Ordinary Shares into Class A Ordinary Shares. In no event shall Class A Ordinary Shares be convertible into Class B Ordinary Shares.
Dividends
The holders of Ordinary Shares are entitled to such dividends as the board of directors may in its discretion lawfully declare from time to time, if the board of directors is satisfied on a reasonable grounds, that immediately after the payment of such dividends, the value of the Company’s assets will exceed its liabilities and the Company will be able to pay its debt and when they fall due.
Class A Ordinary Shares and Class B Ordinary Shares rank equally as to dividends and other distributions. Dividends may be paid either in cash or in specie.
Voting Rights
Holders of Class A Ordinary Shares and Class B Ordinary Shares have the right to receive notice of, attend, speak and vote at general meetings of the shareholders. In respect of all matters upon which holders of our shares are entitled to vote, each Class A Ordinary Share are entitled to one (1) vote and each Class B Ordinary Share are entitled to one hundred (100) votes. At any meeting of shareholders, a resolution put to the vote of the meeting shall be decided by way of a poll.
A meeting of the shareholders is duly constituted if, at the commencement of the meeting, there are present in person or by proxy not less than 50 per cent of the votes of the shares entitled to vote on resolutions of the shareholders to be considered at the meeting. As a BVI business company, the Company is not obliged by the BVI Act to call shareholders’ annual general meetings. The Memorandum and Articles of Association provide that the Company may (but is not obliged to) in each calendar year hold a general meeting as its annual general meeting in which case the Company will specify the meeting as such in the notices calling it, and the annual general meeting will be held at such time and place as may be determined by its directors. Each general meeting, other than an annual general meeting, shall be an extraordinary general meeting.
Shareholders’ annual general meetings and any other general meetings of the Company’s shareholders may be convened by any director or, upon a requisition of shareholders holding at the date of deposit of the requisition not less than 30 percent of the votes attaching to the issued and outstanding shares entitled to vote at general meetings in respect of the matter for which the meeting is requested, in which case the directors are obliged to convene such meeting; however, the Memorandum and Articles of Association do not provide its shareholders with any right to put any proposals before any annual general meetings or any extraordinary general meetings not called by such shareholders. Advance notice of at least ten (10) days and not more than sixty (60) days is required for the convening of the Company’s annual general meeting and other general meetings unless such notice is waived in accordance with its articles of association.
Any resolution to be passed at a meeting by the shareholders requires the affirmative vote of a simple majority of the votes attaching to the shares cast by those shareholders entitled to vote who are present in person or by proxy at a general meeting.
Transfer of Ordinary Shares
Subject to applicable laws, including securities laws, and the restrictions contained in the Memorandum and Articles of Association, any shareholders may transfer all or any of their Class A Ordinary Shares by an instrument of transfer in the usual or common form, in a form prescribed by NASDAQ or any other form approved by our board of directors.
Class B Ordinary Shares may be transferred only to an Affiliate (as defined below) of the holder and any Class B Ordinary Shares transferred otherwise will be converted into Class A Ordinary Shares as described above. See “— Conversion.”
An “Affiliate” with respect to the Class B Ordinary Shares means in respect of a person or entity, any other person or entity that, directly or indirectly (including through one or more intermediaries), controls, is controlled by, or is under common control with, such person or entity, and (i) in the case of a natural person, shall include, without limitation, such person’s spouse, parents, children, siblings, mother-in-law and father-in-law and brothers and sisters-in-law, a trust solely for the benefit of any of the foregoing, a company, partnership or entity wholly owned by one or more of the foregoing, and (ii) in the case of an entity, shall include a partnership, a corporation or any natural person or entity which directly, or indirectly through one or more intermediaries, controls, is controlled by, or is under common control with, such entity. The term “control” in this definition shall mean the ownership, directly or indirectly, of securities possessing more than fifty percent (50%) of the voting power of the corporation, or the partnership or other entity (other than, in the case of corporation, securities having such power only by reason of the happening of a contingency not within the reasonable control of such partnership, corporation, natural person or entity), or having the power to control the management or elect a majority of members to the board of directors or equivalent decision-making body of such corporation, partnership or other entity.
Liquidation
As permitted by the BVI Act and the Memorandum and Articles of Association, the Company may be voluntarily liquidated under Part XII of the BVI Act by resolution of directors and resolution of shareholders if the Company’s assets exceed the Company’s liabilities and the Company is able to pay the Company’s debts as they fall due. The Company may also be wound up in circumstances where the Company is insolvent in accordance with the terms of the BVI Insolvency Act (As Revised).
If the Company is wound up and the assets available for distribution among the Company’s shareholders are more than sufficient to repay all amounts paid to the Company on account of the issue of shares immediately prior to the winding up, the excess shall be distributable pari passu among those shareholders in proportion to the amount paid up immediately prior to the winding up on the shares held by them, respectively. If the Company is wound up and the assets available for distribution among the shareholders as such are insufficient to repay the whole of the amounts paid to the Company on account of the issue of shares, those assets shall be distributed so that, to the greatest extent possible, the losses shall be borne by the shareholders in proportion to the amounts paid up immediately prior to the winding up on the shares held by them, respectively. If the Company is wound up, the liquidator appointed by the Company may, in accordance with the BVI Act, divide among the Company’s shareholders in specie or kind the whole or any part of the Company’s assets (whether they shall consist of property of the same kind or not) and may, for such purpose, set such value as the liquidator deems fair upon any property to be divided and may determine how such division shall be carried out as between the shareholders or different classes of shareholder
Redemption, Repurchase and Surrender of Ordinary Shares
The Company may issue shares on terms that such shares are subject to redemption, at the Company’s option or at the option of the holders thereof, on such terms and in such manner as may be determined, before the issue of such shares, by the Board of Directors. The Company may also repurchase any of its shares provided that the Company may not purchase, redeem or otherwise acquire its own shares without the consent of the shareholder whose Shares are to be purchased, redeemed or otherwise acquired unless the Company is permitted or required by the Act or any other provision in the Memorandum or Articles to purchase, redeem or otherwise acquire the Shares without such consent.
Requirements to Change the Rights of Holders of Class A Ordinary Shares (Item 10.B.4 of Form 20-F)
Variations of Rights of Shares
If at any time the Company’s share are divided into different classes or series of shares, the rights attached to any class or series of shares, whether or not the Company is being wound-up, may be varied by a resolution passed at a meeting by the holders of more than fifty percent of the issued shares of that class that have voted (and are entitled to vote thereon) in relation to any such resolution, unless otherwise provided by the terms of issue of such class. The rights conferred upon the holders of the shares of any class issued shall not, unless otherwise expressly provided by the terms of issue of the shares of that class, be deemed to be varied by the creation or issue of further shares ranking pari passu with such existing class of shares.
Limitations on the Rights to Own Class A Ordinary Shares (Item 10.B.6 of Form 20-F)
There are no limitations under the laws of the British Virgin Islands or under the Memorandum and Articles of Association that limit the right of non-resident or foreign shareholders to hold or exercise voting rights on our Class A Ordinary Shares.
Provisions Affecting Any Change of Control (Item 10.B.7 of Form 20-F)
Anti-Takeover Provisions in the Memorandum and Articles of Association. Some provisions of our Memorandum and Articles of Association may discourage, delay or prevent a change of control of our company or management that shareholders may consider favorable, including provisions that authorize our board of directors to issue preferred shares in one or more series and to designate the price, rights, preferences, privileges and restrictions of such preferred shares without any further vote or action by our shareholders. However, under the laws of the British Virgin Islands, our directors may only exercise the rights and powers granted to them under our Memorandum and Articles of Association for a proper purpose and for what they believe in good faith to be in the best interests of our company.
Transfer of Class B Ordinary Shares. Class B Ordinary Shares may be transferred without conversion by a resolution of the directors, and an affirmative vote of a majority of the Class B Ordinary Shares, if such transfer will result in a change of control of our company.
Ownership Threshold (Item 10.B.8 of Form 20-F)
There are no provisions under BVI law applicable to the Company, or under the Memorandum and Articles of Association, that require the Company to disclose shareholder ownership above any particular ownership threshold.
Differences Between the Law of Different Jurisdictions (Item 10.B.9 of Form 20-F)
British Virgin Islands companies are governed by the BVI Act. The BVI Act is modelled on the laws of England and Wales but does not follow recent statutory enactments, and differs from laws applicable to United States corporations and their shareholders.
Set forth below is a comparison of select provisions of the corporate laws of Delaware and the British Virgin Islands showing the default positions in each jurisdiction that govern shareholder rights.
| DELAWARE CORPORATE LAW | BVI CORPORATE LAW |
| Class actions and derivative actions generally are available to shareholders of a Delaware corporation for, among other things, breach of fiduciary duty, corporate waste and actions not taken in accordance with applicable law. In such actions, the court has discretion to permit the winning party to recover attorneys’ fees incurred in connection with such action. |
Class actions and derivative actions are generally not available to shareholders under British Virgin Islands law.
The British Virgin Islands courts, however, would ordinarily be expected to permit a shareholder to commence an action in the name of a company to remedy a wrong to the company where the act complained of is alleged to be beyond the corporate power of the company or illegal, or would result in the violation of the company’s memorandum and articles of association. Furthermore, consideration would be given by a British Virgin Islands court to acts that are alleged to constitute a fraud against the minority shareholders or, for instance, where an act requires the approval of a greater percentage of the company’s shareholders than that which actually approved it.
When the affairs of a company are being conducted in a manner which is oppressive or prejudicial to the interests of some part of the shareholders, one or more shareholders may apply to the High Court of the British Virgin Islands, which may make such order as it sees fit, including an order regulating the conduct of the company’s affairs in the future or ordering the purchase of the shares of any shareholders by other shareholders or by the company. |
| Under the Delaware General Corporation Law, the board of directors has the authority to fix the compensation of directors, unless otherwise restricted by the certificate of incorporation or bylaws. | The Articles contain a provision that the board of directors has the power to determine the remuneration, if any, of the directors. |
|
Unless directors are elected by written consent in lieu of an annual meeting, directors are elected in an annual meeting of stockholders on a date and at a time designated by or in the manner provided in the bylaws. Re-election is possible.
Classified boards are permitted. |
The Articles provide that the directors shall be appointed by resolution of shareholders or by resolution of directors and will hold office for the term fixed by the resolution of shareholders or resolution of directors appointing them or until their earlier death, resignation or removal.
The directors of the Company may appoint directors where there is a vacancy or as an addition to the existing directors. |
| DELAWARE CORPORATE LAW | BVI CORPORATE LAW |
|
The Delaware General Corporation Law provides that a certificate of incorporation may contain a provision eliminating or limiting the personal liability of directors (but not other controlling persons) of the corporation for monetary damages for breach of a fiduciary duty as a director, except no provision in the certificate of incorporation may eliminate or limit the liability of a director for:
● any breach of a director’s duty of loyalty to the corporation or its shareholders;
● acts or omissions not in good faith or which involve intentional misconduct or a knowing violation of law;
● statutory liability for unlawful payment of dividends or unlawful stock purchase or redemption; or
● any transaction from which the director derived an improper personal benefit.
A Delaware corporation may indemnify any person who was or is a party or is threatened to be made a party to any proceeding, other than an action by or on behalf of the corporation, because the person is or was a director or officer, against liability incurred in connection with the proceeding if the director or officer acted in good faith and in a manner reasonably believed to be in, or not opposed to, the best interests of the corporation; and the director or officer, with respect to any criminal action or proceeding, had no reasonable cause to believe his or her conduct was unlawful.
Unless ordered by a court, any foregoing indemnification is subject to a determination that the director or officer has met the applicable standard of conduct:
● by a majority vote of the directors who are not parties to the proceeding, even though less than a quorum;
● by a committee of directors designated by a majority vote of the eligible directors, even though less than a quorum;
● by independent legal counsel in a written opinion if there are no eligible directors, or if the eligible directors so direct; or
● by the shareholders.
Moreover, a Delaware corporation may not indemnify a director or officer in connection with any proceeding in which the director or officer has been adjudged to be liable to the corporation unless and only to the extent that the court determines that, despite the adjudication of liability but in view of all the circumstances of the case, the director or officer is fairly and reasonably entitled to indemnity for those expenses which the court deems proper. |
Section 132 of the BVI Act, and regulation 16 of the Articles, provide that, subject to certain limitations, the Company shall indemnify its directors and officers against all expenses, including legal fees, and against all judgments, fines and amounts paid in settlement and reasonably incurred in connection with legal, administrative or investigative proceedings. Such indemnity only applies if the person acted honestly and in good faith with a view to the best interests of the company and, in the case of criminal proceedings, the person had no reasonable cause to believe that their conduct was unlawful.
Section 133 of the BVI Act permits a company to purchase and maintain insurance for the benefit of any officer or director in respect of any loss or liability attaching to them in respect of any negligence, default, breach of duty or breach of trust, whether or not we may otherwise indemnify such officer or director. |
| DELAWARE CORPORATE LAW | BVI CORPORATE LAW |
|
A director of a Delaware corporation has a fiduciary duty to the corporation and its shareholders. This duty has two components:
● the duty of care; and
● the duty of loyalty. |
The laws of the BVI impose a duty on directors and officers of a British Virgin Islands company:
● to act honestly and in good faith and in what the director believes to be in the best interests of the company when exercising their powers as a director;
● to exercise the reasonable care, diligence, and skill that a reasonable director would exercise in the same circumstances taking into account, but without limitation: i. the nature of the company; ii. the nature of the decision; and iii. the position of the director and the nature of their responsibilities;
● to exercise their duties for proper purpose and in accordance with the BVI Act and the memorandum and articles of association of the company; and
● to disclose any interest which they have in a transaction entered into or to be entered into by the company.
The statutory duties imposed on directors, by the BVI Act, are further supplemented by common law duties established (over centuries) of case law. There is considerable overlap between the common law and the BVI Act and in most circumstances it is not necessary to consider the two separately.
In addition, the BVI Act imposes various duties on directors and officers of a company with respect to certain matters of management and administration of the company. |
|
The duty of care requires that a director act in good faith, with the care that an ordinarily prudent person would exercise under similar circumstances. Under this duty, a director must inform himself of, and disclose to shareholders, all material information reasonably available regarding a significant transaction. The duty of loyalty requires that a director act in a manner he reasonably believes to be in the best interests of the corporation. He must not use his corporate position for personal gain or advantage. This duty prohibits self-dealing by a director and mandates that the best interest of the corporation and its shareholders take precedence over any interest possessed by a director, officer or controlling shareholder and not shared by the shareholders generally. In general, actions of a director are presumed to have been made on an informed basis, in good faith and in the honest belief that the action taken was in the best interests of the corporation. However, this presumption may be rebutted by evidence a breach of one of the fiduciary duties.
Should such evidence be presented concerning a transaction by a director, a director must prove the procedural fairness of the transaction, and that the transaction was of fair value to the corporation. |
The laws of the BVI also impose a duty on directors and officers of a British Virgin Islands company to:
● act honestly and in good faith with a view to the best interests of the company; and
● exercise the care, diligence and skill that a reasonable director or officer would exercise in the same circumstances.
In addition, the BVI Act imposes various duties on directors and officers of a company with respect to certain matters of management and administration of the company. |
| A Delaware corporation may, in its certificate of incorporation, eliminate the right of shareholders to act by written consent. | Our Memorandum and Articles of Association provides that any action required or permitted to be taken by shareholders of the Company must be effected by a duly convened meeting of the Company. |
| A shareholder of a Delaware corporation has the right to put any proposal before the annual meeting of shareholders, provided it complies with the notice provisions in the governing documents. A special meeting may be called by the board of directors or any other person authorized to do so in the governing documents, but shareholders may be precluded from calling special meetings. | Under the Articles, shareholders entitled to exercise 30% or more of the voting rights, in respect of the matter for which the meeting is requested, can require the directors to convene a meeting of shareholders. |
| DELAWARE CORPORATE LAW | BVI CORPORATE LAW |
| Under the Delaware General Corporation Law, cumulative voting for elections of directors is not permitted unless the corporation’s certificate of incorporation provides for it. |
Under British Virgin Islands law, the voting rights of shareholders are regulated by the company’s memorandum and articles of association and, in certain circumstances, by the BVI Act.
The Articles do not provide for cumulative voting. |
| A Delaware corporation with a classified board may be removed only for cause with the approval of a majority of the outstanding shares entitled to vote, unless the certificate of incorporation provides otherwise. |
Under the Articles, a director may be removed:
● by resolution of directors with or without cause; or
● by resolution of shareholders for cause only by a resolution approved at a duly convened and constituted meeting of the shareholders of the Company by the affirmative vote of not less than 75% of the votes of the Ordinary Shares entitled to vote thereon which were present at the meeting and were voted. |
| The Delaware General Corporation Law generally prohibits a Delaware corporation from engaging in certain business combinations with an “interested shareholder” for three years following the date that such person becomes an interested shareholder. An interested shareholder generally is a person or group who or which owns or owned 15.0% or more of the corporation’s outstanding voting stock within the past three years. | There is no similar law in the British Virgin Islands. |
| Unless the board of directors of a Delaware corporation approves the proposal to dissolve, dissolution must be approved by shareholders holding 100.0% of the total voting power of the corporation. Only if the dissolution is initiated by the board of directors may it be approved by a simple majority of the corporation’s outstanding shares. Delaware law allows a Delaware corporation to include in its certificate of incorporation a supermajority voting requirement in connection with dissolutions initiated by the board. |
As permitted by the BVI Act and our Articles, we may be voluntarily liquidated under Part XII of the BVI Act by resolution of directors or resolution of shareholders if we have no liabilities or we are able to pay our debts as they fall due and the value of our assets equals or exceeds our liabilities.
A company may also be wound up where a court deems it just and equitable to do so and in circumstances where they are insolvent in accordance with the terms of the BVI Insolvency Act. |
| A Delaware corporation may vary the rights of a class of shares with the approval of a majority of the outstanding shares of such class, unless the certificate of incorporation provides otherwise. | If at any time the Company’s share are divided into different classes or series of shares, the rights attached to any class or series of shares, whether or not the Company is being wound-up, may be varied by a resolution passed at a meeting by the holders of more than fifty percent of the issued shares of that class that have voted (and are entitled to vote thereon) in relation to any such resolution, unless otherwise provided by the terms of issue of such class. The rights conferred upon the holders of the shares of any class issued shall not, unless otherwise expressly provided by the terms of issue of the shares of that class, be deemed to be varied by the creation or issue of further shares ranking pari passu with such existing class of shares. |
| A Delaware corporation’s governing documents may be amended with the approval of a majority of the outstanding shares entitled to vote, unless the certificate of incorporation provides otherwise. | A British Virgin Islands company’s memorandum and articles of association may be amended by resolutions of the board of directors or the shareholders, subject to the BVI Act and the memorandum and articles of association. Certain amendments can only be made by resolution of shareholder. |
| DELAWARE CORPORATE LAW | BVI CORPORATE LAW |
| Shareholders of a Delaware corporation, upon written demand under oath stating the purpose thereof, have the right during the usual hours for business to inspect for any proper purpose, and to obtain copies of list(s) of shareholders and other books and records of the corporation and its subsidiaries, if any, to the extent the books and records of such subsidiaries are available to the corporation. |
Under the BVI Act, members of the general public, on payment of a nominal fee, can obtain copies of the public records of a company available at the office of the BVI Registrar which will include the company’s certificate of incorporation, its memorandum and articles of association (with any amendments), a list of the current directors and records of license fees paid to date and will also disclose any articles of dissolution, articles of merger and a register of charges if the company has elected to file such a register.
A shareholder of a company is entitled, on giving written notice to the company, to inspect:
● the memorandum and articles;
● the register of members;
● the register of directors; and
● the minutes of meetings and resolutions of members and of those classes of members of which they are a member; and to make copies of or take extracts from the documents and records referred to in above.
Subject to the memorandum and articles of association, the directors may, if they are satisfied that it would be contrary to the company’s interests to allow a shareholder to inspect any document, or part of a document, specified above, refuse to permit the member to inspect the document or limit the inspection of the document, including limiting the making of copies or the taking of extracts from the records.
Where a company fails or refuses to permit a shareholder to inspect a document or permits a shareholder to inspect a document subject to limitations, that shareholder may apply to a British Virgin Islands Court for an order that they should be permitted to inspect the document or to inspect the document without limitation. |
|
The board of directors may approve a dividend without shareholder approval. Subject to any restrictions contained in its certificate of incorporation, the board may declare and pay dividends upon the shares of its capital stock either:
● out of its surplus, or
● in case there is no such surplus, out of its net profits for the fiscal year in which the dividend is declared and/or the preceding fiscal year.
Stockholder approval is required to authorize capital stock in excess of that provided in the charter. Directors may issue authorized shares without stockholder approval. |
Under British Virgin Islands law, the board of directors may declare a dividend without shareholder approval, but a company may not declare or pay dividends if there are reasonable grounds for believing that:
● the company is, or would after the payment be, unable to pay its debts as they fall due; or
● that the value of the company’s assets would be less than its liabilities. |
| All creation of shares require the board of directors to adopt a resolution or resolutions, pursuant to authority expressly vested in the board of directors by the provisions of the company’s certificate of incorporation. |
The number of shares that a British Virgin Islands company is authorized to issue is set out in the memorandum and articles of association.
The Memorandum provides that the company is authorized to issue a maximum of 500,000,000 shares with no par value divided into three classes of shares as follows:
● 400,000,000 Class A Ordinary Shares;
● 99,950,000 Class B Shares Ordinary Shares; and
● 50,000 preferred shares with no par value. |
| DELAWARE CORPORATE LAW | BVI CORPORATE LAW |
| Under the Delaware General Corporation Law, with certain exceptions, a merger, consolidation, sale, lease or transfer of all or substantially all of the assets of a corporation must be approved by the board of directors and a majority of the outstanding shares entitled to vote thereon. A shareholder of a Delaware corporation participating in certain major corporate transactions may, under certain circumstances, be entitled to appraisal rights pursuant to which such shareholder may receive cash in the amount of the fair value of the shares held by such shareholder (as determined by a court) in lieu of the consideration such shareholder would otherwise receive in the transaction. The Delaware General Corporation Law also provides that a parent corporation, by resolution of its board of directors, may merge with any subsidiary, of which it owns at least 90.0% of each class of capital stock without a vote by the shareholders of such subsidiary. Upon any such merger, dissenting shareholders of the subsidiary would have appraisal rights. |
The consolidation or merger of a British Virgin Islands company with another company or corporation (other than certain affiliated companies) requires the consolidation or merger to be approved by the company’s board of directors and by its shareholders. Unless the company’s memorandum and articles of association provide otherwise, the approval of a majority of the shareholders voting at a meeting of shareholders is required to approve the consolidation or merger agreement.
Under British Virgin Islands law, in the event of a consolidation or merger of a British Virgin Islands company with another company or corporation, a shareholder of the British Virgin Islands company who did not vote in favor of the amalgamation or merger and who is not satisfied that fair value has been offered for such shareholder’s shares may seek fair value for those shares in accordance with Section 179 of the BVI Act. |
Changes in Capital (Item 10.B.10 of Form 20-F)
We may from time to time by resolution of the directors:
| ● | authorise and create additional classes of shares, and issue such amount of shares as the resolution will prescribe; |
| ● | consolidate and divide all or any of our shares into shares of a larger amount than existing shares; or |
| ● | sub-divide our existing shares or any of them into shares of a smaller amount. |
Debt Securities (Item 12.A of Form 20-F)
Not applicable.
Warrants (Item 12.B of Form 20-F)
Public Warrants
The number of warrants that were outstanding as of December 31, 2025 is 5,175,000. Our warrants are issued in registered (book-entry) form under the warrant agreement dated February 14, 2022 between A SPAC I Acquisition Corp., a British Virgin Islands company (the “Company”), and Continental Stock Transfer & Trust Company, a New York corporation, as warrant agent (“ASCA Warrant Agreement”).
In connection with and upon the consummation of the Business Combination, each warrant outstanding immediately prior to the Business Combination was assumed by the Company and converted into a warrant entitling the holder thereof to purchase one Class A Ordinary Share upon exercise. Each Warrant continues to have and be subject to substantially the same terms and conditions as were applicable to such warrant immediately prior to the consummation of the Business Combination (including any repurchase rights and cashless exercise provisions).
The following summary is not complete and is subject to, and is qualified in its entirety by reference to, the provisions of the Memorandum and Articles of Association and the Warrant Agreement.
General
Each warrant entitles the holder thereof to purchase 1/60,000th of a Class A ordinary share (post-Reverse Stock Splits) at an effective price of $690,000 per whole share. The Company will not issue fractional shares. Our warrants became exercisable 30 days after the completion of the Business Combination and 12 months from the date of the IPO, and will expire five years after the consummation of the Business Combination.
Exercise
A warrant may be exercised by delivering to the warrant agent (i) the warrant, (ii) an election to purchase form, and (iii) the payment in full of the Exercise Price and any and all applicable taxes due in connection with the exercise.
As soon as practicable after the exercise of any warrant we will issue a book-entry position or certificate, as applicable, for the Class A Ordinary Shares. All Class A Ordinary Shares issued upon the proper exercise of a warrant in conformity with the Warrant Agreement will be validly issued, fully paid and non-assessable.
A warrant holder may notify us in writing of the holder’s election to be subject to a provision of the Warrant Agreement preventing the holder from exercising a warrant, to the extent that, after giving effect to such exercise, the holder (together with its affiliates), to the warrant agent’s actual knowledge, would beneficially own in excess of 9.8% (or such other amount as a holder may specify) (the “Maximum Percentage”) of our outstanding Class A Ordinary Shares immediately after giving effect to such exercise. By written notice to us, a warrant holder may increase or decrease the Maximum Percentage to any other percentage specified in such notice; provided, however, that any such increase will not be effective until the sixty-first (61st) day after such notice is delivered to us.
Notwithstanding the above, we are not obligated to deliver any Class A Ordinary Shares pursuant to the exercise of a warrant and do not have the obligation to settle such warrant exercise unless a registration statement under the Securities Act with respect to the Class A Ordinary Shares underlying the warrants is then effective and a prospectus relating thereto is current, subject to our satisfaction of obligations with respect to registration under the Warrant Agreement, or a valid exemption from registration is available. No warrant will be exercisable and we will not be obligated to issue a Class A Ordinary Share upon exercise of a warrant unless the Class A Ordinary Share issuable upon such warrant exercise has been registered, qualified or deemed to be exempt under the securities laws of the state of residence of the registered holder of the warrants.
Adjustments
The number of Class A Ordinary Shares issuable upon the exercise of the warrants is subject to customary adjustments in certain circumstances, such as a capitalization or share dividend of Ordinary Shares, or by a sub-division of Ordinary Shares or other similar event, as described in the Warrant Agreement. In the event the number of Class A Ordinary Shares purchasable upon the exercise of the warrants is adjusted, the Exercise Price will be adjusted (to the nearest cent) by multiplying the Exercise Price immediately prior to such adjustment, by a fraction (x) the numerator of which shall be the number of Ordinary Shares purchasable upon the exercise of the warrants immediately prior to such adjustment, and (y) the denominator of which shall be the number of Ordinary Shares so purchasable immediately thereafter.
In addition, if we issue an extraordinary dividend (as defined in the Warrant Agreement), then the Warrant Price shall be decreased, effective immediately after the effective date of such Extraordinary Dividend, by the amount of cash and the fair market value (as determined by the Company’s Board of Directors, in good faith) of any securities or other assets paid in respect of such Extraordinary Dividend divided by all outstanding shares of the Company at such time (whether or not any stockholders waived their right to receive such dividend)
If, by reason of any adjustment made pursuant to the events described above, the holder of any warrant would be entitled, upon the exercise of such warrant, to receive a fractional interest in a share, we will, upon such exercise, round down to the nearest whole number the number of Ordinary Shares to be issued to such holder.
Warrant holders also have replacement rights in the case of certain reclassification, reorganization, merger, consolidation or sale transactions involving our company or substantially all of our assets (each a “Replacement Event”). Upon the occurrence of any Replacement Event, warrant holders will have the right to purchase and receive (in lieu of our Class A Ordinary Shares) the kind and amount of stock or other securities or property (including cash) receivable upon such Replacement Event that the holder would have received if the warrants were exercised immediately prior to such event.
Upon any adjustment of the Exercise Price or the number of shares issuable upon exercise of a warrant, we will provide written notice of such adjustment to the warrant agent stating the Exercise Price resulting from such adjustment and the increase or decrease, if any, in the number of Class A Ordinary Shares purchasable at such price upon the exercise of a warrant.
Redemption
We may redeem the outstanding warrants, in whole and not in part, at a price of $0.01 per warrant:
| ● | at any time while the warrants are exercisable; |
| ● | upon a minimum of 30 days’ prior written notice of redemption; |
| ● | if, and only if, the last sales price of Class A Ordinary Shares equals or exceeds $990,000 per share (post-Reverse Stock Splits) for any 20 trading days within a 30 trading day period ending three trading days before we send the notice of redemption; and |
| ● | if, and only if, a registration statement under the Securities Act covering the Class A ordinary shares issuable upon exercise of the warrants is effective and a current prospectus relating to those Class A ordinary shares is available throughout the 30-day redemption period, except if the warrants may be exercised on a cashless basis and such cashless exercise is exempt from registration under the Securities Act. |
If the foregoing conditions are satisfied and the Company issues a notice of redemption, each warrant holder can exercise his, her or its warrant prior to the scheduled redemption date. However, the price of the Ordinary Shares may fall below the $990,000 trigger price as well as the $690,000 warrant exercise price per full share after the redemption notice is issued and not limit the Company’s ability to complete the redemption.
In the event the Company elects to redeem all of the warrants that are subject to redemption, the Company will fix a date for the redemption. Notice of redemption shall be mailed by first class mail, postage prepaid, by the Company not less than 30 days prior to the date fixed for redemption to the registered holders of the warrants to be redeemed at their last addresses as they appear on the warrant register. Any notice mailed in the aforesaid manner will be conclusively presumed to have been duly given, whether or not the registered holder received such notice.
Cashless Exercise
If the Company calls the warrants for redemption as described above, the Company’s management will have the option to require all warrant holders that wish to exercise warrants to do so on a “cashless basis.” In such event, each warrant holder would pay the exercise price by surrendering the whole warrant for that number of Class A Ordinary Shares equal to the quotient obtained by dividing (x) the product of the number of Class A Ordinary Shares underlying the warrants, multiplied by the difference between the exercise price of the warrants and the “fair market value” (as defined below) by (y) the fair market value. The “fair market value” shall mean the average reported last sale price of the Class A Ordinary Shares for the 10 trading days ending on the third trading day prior to the date on which the notice of redemption is sent to the warrant holders. Whether the Company will exercise its option to require all warrant holders to exercise their warrants on a “cashless basis” will depend on a variety of factors including the price of the Class A Ordinary Shares at the time the warrants are called for redemption, the Company’s cash needs at such time and concerns regarding dilutive share issuances.
Other Securities (Item 12.C of Form 20-F)
Not applicable.
Description of American Depositary Shares (Items 12.D.1 and 12.D.2 of Form 20-F)
Not applicable.
Exhibit 8.1
List of subsidiaries
| Subsidiaries | Place of incorporation | |
| NewGenIvf Limited | Cayman Islands | |
| MicroSort Lab Services LLC | U.S.A. | |
| NewGenDigital Limited | British Virgin Islands | |
| FFPGS (HK) Limited | Hong Kong | |
| NewGenProperty Limited | British Virgin Islands | |
| NewGenBiz Limited | British Virgin Islands | |
| Bi Clinic LLC | Kyrgyzstan | |
| First Fertility PGS Center Limited | Thailand | |
| First Fertility Phnom Penh Limited | Cambodia | |
| Med Holdings Limited | Thailand | |
| Well Image Limited | Hong Kong | |
| HyFi Innovations Limited | Hong Kong | |
| NewGenOman Limited | British Virgin Islands | |
| Alfred Siu Charitable Foundation Limited | Hong Kong |
Exhibit 10.39
SECURITIES PURCHASE AGREEMENT
This SECURITIES PURCHASE AGREEMENT (the “Agreement”), dated as of March 18, 2026, by and between NEWGENIVF GROUP LIMITED, a British Virgin Islands Limited Company, with headquarters located at 36/39-36/40, 13th Floor, PS Tower, Sukhumvit 21 Road (Asoke), Watthana District, B 10110, Thailand (the “Company”), and CFI CAPITAL LLC, a Florida limited liability company, with its address at 2151 West Hillsboro Blvd., Suite 209, Deerfield Beach, FL 33442 (the “Buyer”).
WHEREAS:
A. The Company and the Buyer are executing and delivering this Agreement in reliance upon the exemption from securities registration afforded by the rules and regulations as promulgated by the United States Securities and Exchange Commission (the “SEC”) under the Securities Act of 1933, as amended (the “1933 Act”);
B. Buyer desires to purchase and the Company desires to issue and sell, upon the terms and conditions set forth in this Agreement a 6% convertible redeemable note of the Company, in the form attached hereto as Exhibit A, in the aggregate principal amount of $200,000.00 (together with any note(s) issued in replacement thereof or as a dividend thereon or otherwise with respect thereto in accordance with the terms thereof, the “Note”), convertible into shares of common stock, of the Company (the “Common Stock”), upon the terms and subject to the limitations and conditions set forth in such Note. The Note shall contain an original issue discount of $20,000.00, such that the purchase price of the Note shall be $180,000.00.
C. The Buyer wishes to purchase, upon the terms and conditions stated in this Agreement, such principal amount of Note as is set forth immediately below its name on the signature page hereto; and
NOW THEREFORE, the Company and the Buyer severally (and not jointly) hereby agree as follows:
1. Purchase and Sale of the Note.
a. Purchase of the Note. On the Closing Date (as defined below), the Company shall issue and sell to the Buyer, and the Buyer agrees to purchase from the Company such Note as is set forth immediately below the Buyer’s name on the signature pages hereto.
b. Form of Payment. On the Closing Date (as defined below), (i) the Buyer shall pay the purchase price for the Note to be issued and sold to it at the Closing (as defined below) (the “Purchase Price”) by wire transfer of immediately available funds to the Company, in accordance with the Company’s written wiring instructions, against delivery of the Note in the principal amount equal to the Purchase Price as is set forth immediately below the Buyer’s name on the signature pages hereto, and (ii) the Company shall deliver such duly executed Note on behalf of the Company to the Buyer, against delivery of such Purchase Price.
c. Closing Date. The date and time of the issuance and sale of the Note pursuant to this Agreement (the “Closing Date”) shall be on or about March 18, 2026, or such other mutually agreed upon time. The closing of the transactions contemplated by this Agreement (the “Closing”) shall occur on the Closing Date at such location as may be agreed to by the parties.
Company Initials
2. Buyer’s Representations and Warranties. The Buyer represents and warrants to the Company that:
a. Investment Purpose. As of the date hereof, the Buyer is purchasing the Note and the shares of Common Stock issuable upon conversion of or otherwise pursuant to the Note, such shares of Common Stock being collectively referred to herein as the “Conversion Shares” and, collectively with the Note, the “Securities”) for its own account and not with a present view towards the public sale or distribution thereof, except pursuant to sales registered or exempted from registration under the 1933 Act; provided, however, that by making the representations herein, the Buyer does not agree to hold any of the Securities for any minimum or other specific term and reserves the right to dispose of the Securities at any time in accordance with or pursuant to an effective registration statement with respect to such Securities or an exemption under the 1933 Act.
b. Accredited Investor Status. The Buyer is an “accredited investor” as that term is defined in Rule 501(a) of Regulation D (an “Accredited Investor”). Any of Buyer’s transferees, assignees, or purchasers must be “accredited investors” in order to qualify as prospective transferees, permitted assignees in the case of Buyer’s or Holder’s transfer, assignment, or sale of the Note.
c. Reliance on Exemptions. The Buyer understands that the Securities are being offered and sold to it in reliance upon specific exemptions from the registration requirements of United States federal and state securities laws and that the Company is relying upon the truth and accuracy of, and the Buyer’s compliance with, the representations, warranties, agreements, acknowledgments, and understandings of the Buyer set forth herein in order to determine the availability of such exemptions and the eligibility of the Buyer to acquire the Securities.
d. Information. The Buyer and its advisors, if any, have been furnished with all materials relating to the business, finances, and operations of the Company and materials relating to the offer and sale of the Securities which have been requested by the Buyer or its advisors. The Buyer and its advisors, if any, have been afforded the opportunity to ask questions of the Company. Notwithstanding the foregoing, the Company has not disclosed to the Buyer any material nonpublic information and will not disclose such information unless such information is disclosed to the public prior to or promptly following such disclosure to the Buyer. Neither such inquiries nor any other due diligence investigation conducted by Buyer or any of its advisors or representatives shall modify, amend, or affect Buyer’s right to rely on the Company’s representations and warranties contained in Section 3 below. The Buyer understands that its investment in the Securities involves a significant degree of risk. The Buyer is not aware of any facts that may constitute a breach of any of the Company’s representations and warranties made herein.
e. Governmental Review. The Buyer understands that no United States federal or state agency or any other government or governmental agency has passed upon or made any recommendation or endorsement of the Securities.
f. Transfer or Re-sale. The Buyer understands that (i) the sale or re-sale of the Securities has not been and is not being registered under the 1933 Act or any applicable state securities laws, and the Securities may not be transferred unless (a) the Securities are sold pursuant to an effective registration statement under the 1933 Act, (b) the Buyer shall have delivered to the Company, at the cost of the Buyer, an opinion of counsel that shall be in form, substance and scope customary for opinions of counsel in comparable transactions to the effect that the Securities to be sold or transferred may be sold or transferred pursuant to an exemption from such registration, which opinion may be accepted by the Company in its reasonable discretion, (c) the Securities are sold or transferred to an “affiliate” (as defined in Rule 144 promulgated under the 1933 Act (or a successor rule) (“Rule 144”)) of the Buyer who agrees to sell or otherwise transfer the Securities only in accordance with this Section 2(f) and who is an Accredited Investor, or (d) the Securities are sold pursuant to Rule 144 or Regulation S under the 1933 Act (or a successor rule) (“Regulation S”), and the Buyer shall have delivered to the Company, at the cost of the Buyer, an opinion of counsel that shall be in form, substance and scope customary for opinions of counsel in corporate transactions, which opinion may be accepted by the Company in its reasonable discretion; (ii) any sale of such Securities made in reliance on Rule 144 may be made only in accordance with the terms of said Rule 144 and further, if said Rule 144 is not applicable, any re-sale of such Securities under circumstances in which the selling Buyer (or the person through whom the sale is made) may be deemed to be an underwriter (as that term is defined in the 1933 Act) may require compliance with some other exemption under the 1933 Act or the rules and regulations of the SEC thereunder; and (iii) neither the Company nor any other person is under any obligation to register such Securities under the 1933 Act or any state securities laws or to comply with the terms and conditions of any exemption thereunder (in each case).
g. Legends. The Buyer understands that the Note and, until such time, if any, as the Conversion Shares have been registered under the 1933 Act may be sold pursuant to Rule 144 or Regulation S without any restriction as to the number of securities as of a particular date that have been sold, the Conversion Shares shall bear a restrictive legend in substantially the following form (and a stop-transfer order may be placed against the transfer of the certificates for such Securities):
“NEITHER THE ISSUANCE AND SALE OF THE SECURITIES REPRESENTED HEREBY NOR THE SECURITIES INTO WHICH THESE SECURITIES ARE CONVERTIBLE HAVE BEEN REGISTERED UNDER THE SECURITIES ACT OF 1933, AS AMENDED, OR APPLICABLE STATE SECURITIES LAWS. THE SECURITIES MAY NOT BE OFFERED FOR SALE, SOLD, TRANSFERRED, OR ASSIGNED (I) IN THE ABSENCE OF (A) AN EFFECTIVE REGISTRATION STATEMENT FOR THE SECURITIES UNDER THE SECURITIES ACT OF 1933, AS AMENDED, OR (B) AN OPINION OF COUNSEL (WHICH COUNSEL SHALL BE SELECTED BY THE HOLDER), IN A FORM ACCEPTABLE TO THE COMPANY, THAT REGISTRATION IS NOT REQUIRED UNDER SAID ACT OR (II) UNLESS SOLD PURSUANT TO RULE 144 OR RULE 144A UNDER SAID ACT.”
The legend set forth above shall be removed and the Company shall issue a certificate without such legend to the holder of any Security upon which it is stamped, if, unless otherwise required by applicable state securities laws, (a) such Security is registered for sale under an effective registration statement filed under the 1933 Act or otherwise may be sold pursuant to Rule 144 or Regulation S without any restriction as to the number of securities as of a particular date that can then be immediately sold, or (b) such holder provides the Company with an opinion of counsel, in form, substance, and scope customary for opinions of counsel in comparable transactions, to the effect that a public sale or transfer of such Security may be made without registration under the 1933 Act, which opinion shall be accepted by the Company in its reasonable discretion so that the sale or transfer is effected. The Buyer agrees to sell all Securities, including those represented by a certificate(s) from which the legend has been removed, in compliance with applicable prospectus delivery requirements, if any.
h. Authorization; Enforcement. This Agreement has been duly and validly authorized. This Agreement has been duly executed and delivered on behalf of the Buyer, and this Agreement constitutes a valid and binding agreement of the Buyer enforceable in accordance with its terms.
i. Residency. The Buyer is a resident of the jurisdiction set forth immediately below the Buyer’s name on the signature pages hereto.
3. Representations and Warranties of the Company. The Company represents and warrants to the Buyer that:
a. Organization and Qualification. The Company and each of its subsidiaries, if any, is a Limited Company duly organized, validly existing, and in good standing under the laws of the jurisdiction in which it is incorporated, with full power and authority (corporate and other) to own, lease, use and operate its properties and to carry on its business as and where now owned, leased, used, operated and conducted, except for those jurisdictions in which failure to have such authority would not have a Material Adverse Effect.
b. Authorization; Enforcement. (i) The Company has all requisite corporate power and authority to enter into and perform this Agreement, the Note and to consummate the transactions contemplated hereby and thereby and to issue the Securities, in accordance with the terms hereof and thereof, (ii) the execution and delivery of this Agreement and the Note by the Company and the consummation by it of the transactions contemplated hereby and thereby (including without limitation, the issuance of the Note and the issuance and reservation for issuance of the Conversion Shares issuable upon conversion or exercise thereof) have been duly authorized by the Company’s Board of Directors and no further consent or authorization of the Company, its Board of Directors, or its shareholders is required, (iii) this Agreement has been duly executed and delivered by the Company by its authorized representative, and such authorized representative is the true and official representative with authority to sign this Agreement and the other documents executed in connection herewith and bind the Company accordingly, and (iv) this Agreement constitutes, and upon execution and delivery by the Company of the Note, each of such instruments will constitute, a legal, valid and binding obligation of the Company enforceable against the Company in accordance with its terms, except as limited by bankruptcy, insolvency or other laws of general application relating to or affecting the enforcement of creditors’ rights generally and general principles of equity.
c. Issuance of Shares. The shares reserved for conversion of the Note shall be duly authorized and reserved for issuance as soon as practicable after the Company has increased its shares of authorized Common Stock in an amount equal to or greater than that permitting it to reserve such shares. Upon conversion of the Note in accordance with its respective terms, Conversion Shares will be validly issued, fully paid and non-assessable, and free from all taxes, liens, claims and encumbrances with respect to the issue thereof and shall not be subject to preemptive rights or other similar rights of shareholders of the Company and will not impose personal liability upon the holder thereof.
d. Acknowledgment of Dilution. The Company understands and acknowledges the potentially dilutive effect to the Common Stock upon the issuance of the Conversion Shares upon conversion of the Note. The Company further acknowledges that its obligation to issue Conversion Shares upon conversion of the Note in accordance with this Agreement and the Note is absolute and unconditional regardless of the dilutive effect that such issuance may have on the ownership interests of other shareholders of the Company.
e. No Conflicts. The execution, delivery and performance of this Agreement and the Note by the Company and the consummation by the Company of the transactions contemplated hereby and thereby (including, without limitation, the issuance and reservation for issuance of the Conversion Shares) will not (i) conflict with or result in a violation of any provision of the Certificate of Incorporation or By-laws, (ii) violate or conflict with, or result in a breach of any provision of, or constitute a default (or an event which with notice or lapse of time or both could become a default) under, or give to others any rights of termination, amendment, acceleration or cancellation of, any agreement, indenture, patent, patent license or instrument to which the Company or any of its subsidiaries is a party, or (iii) result in a violation of any law, rule, regulation, order, judgment or decree (including federal and state securities laws and regulations and regulations of any self-regulatory organizations to which the Company or its securities are subject) applicable to the Company or any of its subsidiaries or by which any property or asset of the Company or any of its subsidiaries is bound or affected (except for such conflicts, defaults, terminations, amendments, accelerations, cancellations and violations as would not, individually or in the aggregate, have a Material Adverse Effect). Except for applicable blue sky state notice filings, all consents, authorizations, orders, filings and registrations which the Company is required to obtain pursuant to the preceding sentence have been obtained or effected on or prior to the date hereof. The Company is not in violation of the eligibility requirements of the Nasdaq Stock Market (“Nasdaq”) and does not reasonably anticipate that the Common Stock will be delisted or suspended from trading on the Nasdaq in the foreseeable future, nor are the Company’s securities “chilled” by DTC. The Company and its subsidiaries are unaware of any facts or circumstances which might give rise to any of the foregoing. For purposes of this Agreement, “Material Adverse Effect” means an event or combination of events, which individually or in the aggregate, would reasonably be expected to (a) adversely affect the legality, validity or enforceability of the Agreement or the Note, or (b) have or result in a material adverse effect on the results of operations, assets, or financial condition of the Company, taken as a whole.
f. Absence of Litigation. Except as disclosed to the Buyer or in the Company’s filings with the SEC, there is no action, suit, claim, proceeding, inquiry or investigation before or by any court, public board, government agency, self-regulatory organization or body pending or, to the knowledge of the Company or any of its subsidiaries, threatened against or affecting the Company or any of its subsidiaries, or their officers or directors in their capacity as such, that could have a Material Adverse Effect.
g. Acknowledgment Regarding Buyer’s Purchase of Securities. The Company acknowledges and agrees that the Buyer is acting solely in the capacity of arm’s length purchasers with respect to this Agreement and the transactions contemplated hereby. The Company further acknowledges that the Buyer is not acting as a financial advisor or fiduciary of the Company (or in any similar capacity) with respect to this Agreement and the transactions contemplated hereby and any statement made by the Buyer or any of its respective representatives or agents in connection with this Agreement and the transactions contemplated hereby is not advice or a recommendation and is merely incidental to the Buyer’ purchase of the Securities.
h. No Integrated Offering. Neither the Company, nor any of its affiliates, nor any person acting on its or their behalf, has directly or indirectly made any offers or sales in any security or solicited any offers to buy any security under circumstances that would require registration under the 1933 Act of the issuance of the Securities to the Buyer.
i. Title to Property. The Company and its subsidiaries have good and marketable title in fee simple to all real property and good and marketable title to all personal property owned by them which is material to the business of the Company and its subsidiaries, in each case free and clear of all liens, encumbrances and defects except such as are described in Schedule 3(i) or such as would not have a Material Adverse Effect. Any real property and facilities held under lease by the Company and its subsidiaries are held by them under valid, subsisting and enforceable leases with such exceptions as would not have a Material Adverse Effect.
j. Bad Actor. No officer or director of the Company would be disqualified under Rule 506(d) of the Securities Act as amended on the basis of being a “bad actor” as that term is established in the September 19, 2013 Small Entity Compliance Guide published by the Securities and Exchange Commission.
k. Breach of Representations and Warranties by the Company. If the Company breaches any of the representations or warranties set forth in this Section 3 in any material respect (subject to a 10-day cure period from the date that the Buyer notifies the Company in writing of such breach with reasonable detail), and in addition to any other remedies available to the Buyer pursuant to this Agreement, it will be considered an Event of Default under the Note.
4. COVENANTS.
a. Expenses. The Company agrees that Buyer can deduct $6,000.00 (six Thousand Dollars) from the $180,000.00 payment due under the Note, at the time of cash funding, to be applied to the legal expenses of Buyer.
b. Listing. The Company shall promptly secure the listing of the Conversion Shares upon each national securities exchange or automated quotation system, if necessary, upon which shares of Common Stock are then listed or quoted (subject to official notice of issuance) and, so long as the Buyer owns any of the Securities, shall maintain, so long as any other shares of Common Stock shall be so listed or quoted, such listing or quotation of all Conversion Shares from time to time issuable upon conversion of the Note. The Company will obtain and, so long as the Buyer owns any of the Securities, maintain (i) the listing and trading of its Common Stock on the Nasdaq Stock Market (“Nasdaq”) or the New York Stock Exchange (“NYSE”), or (ii) if the Common Stock is no longer listed on Nasdaq or NYSE or any other national securities exchange, the quotation and trading of its Common Stock on the OTC Markets Group Inc. marketplace (“OTC Markets”) or any other recognised trading market or quotation system, and will comply in all respects with the Company’s reporting, filing and other obligations under the bylaws or rules of the Financial Industry Regulatory Authority (“FINRA”) and such exchanges, as applicable. The Company shall promptly provide to the Buyer copies of any notices it receives, if any, from the Nasdaq, the NYSE, the OTC Markets, and any other markets on which the Common Stock is then listed regarding the continued eligibility of the Common Stock for listing on such markets.
c. Corporate Existence. So long as the Buyer beneficially owns any Note, the Company shall maintain its corporate existence and shall not sell all or substantially all of the Company’s assets, except in the event of a merger, consolidation, or sale of all or substantially all of the Company’s assets, where the surviving or successor entity in such transaction (i) assumes the Company’s obligations hereunder and under the agreements and instruments entered into in connection herewith and (ii) is a publicly traded corporation whose Common Stock is (A) listed for trading on Nasdaq, NYSE, or any other national securities exchange registered under Section 6 of the Securities Exchange Act of 1934, as amended, or (B) if not so listed, quoted and traded on the OTC Markets or any other recognised trading market or quotation system.
d. No Integration. The Company shall not make any offers or sales of any security (other than the Securities) under circumstances that would require registration of the Securities being offered or sold hereunder under the 1933 Act or cause the offering of the Securities to be integrated with any other offering of securities by the Company for the purpose of any stockholder approval provision applicable to the Company or its securities.
e. 20-F or 6-K Filing. The Company will disclose this transaction in its next 20-F or 6-K filing (whichever is next) in a manner sufficient to specifically disclose the material contents of the transaction. The Company will not “lump” this Note with other similar instruments for purposes of disclosure but will specifically identify the parties to this transaction and all material terms.
f. Breach of Covenants. If the Company breaches any of the covenants set forth in this Section 4 (subject to a 5-day cure period from the date that the Buyer notifies the Company in writing of such breach with reasonable detail), and in addition to any other remedies available to the Buyer pursuant to this Agreement, it will be considered an event of default under the Note.
5. Governing Law; Miscellaneous.
a. Governing Law. This Agreement shall be governed by and construed in accordance with the laws of the State of Florida without regard to principles of conflicts of laws. Any action brought by either party against the other concerning the transactions contemplated by this Agreement shall be brought only in the state courts of Florida located in Miami, Florida or in the federal courts located in the state of Florida in the Southern District of Florida. The parties to this Agreement hereby irrevocably waive any objection to jurisdiction and venue of any action instituted hereunder and shall not assert any defense based on lack of jurisdiction or venue or based upon forum non conveniens. The Company and Buyer waive the trial by jury. The prevailing party shall be entitled to recover from the other party its reasonable attorney’s fees and costs. In the event that any provision of this Agreement or any other agreement delivered in connection herewith is invalid or unenforceable under any applicable statute or rule of law, then such provision shall be deemed inoperative to the extent that it may conflict therewith and shall be deemed modified to conform with such statute or rule of law. Any such provision which may prove invalid or unenforceable under any law shall not affect the validity or enforceability of any other provision of any agreement. Each party hereby irrevocably waives personal service of process and consents to process being served in any suit, action, or proceeding in connection with this Agreement or any other Transaction Document by mailing a copy thereof via registered or certified mail or overnight delivery (with evidence of delivery) to such party at the address in effect for notices to it under this Agreement and agrees that such service shall constitute good and sufficient service of process and notice thereof. Nothing contained herein shall be deemed to limit in any way any right to serve process in any other manner permitted by law.
b. Counterparts; Signatures by Facsimile. This Agreement may be executed in one or more counterparts, each of which shall be deemed an original but all of which shall constitute one and the same agreement and shall become effective when counterparts have been signed by each party and delivered to the other party. This Agreement, once executed by a party, may be delivered to the other party hereto by facsimile transmission of a copy of this Agreement bearing the signature of the party so delivering this Agreement.
c. Headings. The headings of this Agreement are for the convenience of reference only and shall not form part of, or affect the interpretation of, this Agreement.
d. Severability. In the event that any provision of this Agreement is invalid or unenforceable under any applicable statute or rule of law, then such provision shall be deemed inoperative to the extent that it may conflict therewith and shall be deemed modified to conform with such statute or rule of law. Any provision hereof which may prove invalid or unenforceable under any law shall not affect the validity or enforceability of any other provision hereof.
e. Entire Agreement; Amendments. This Agreement and the instruments referenced herein contain the entire understanding of the parties with respect to the matters covered herein and therein and, except as specifically set forth herein or therein, neither the Company nor the Buyer makes any representation, warranty, covenant, or undertaking with respect to such matters. No provision of this Agreement may be waived or amended other than by an instrument in writing signed by the majority in the interest of the Buyer.
f. Notices. All notices, demands, requests, consents, approvals, and other communications required or permitted hereunder shall be in writing and, unless otherwise specified herein, shall be (i) personally served, (ii) deposited in the mail, registered or certified, return receipt requested, postage prepaid, (iii) delivered by reputable air courier service with charges prepaid, (iv) via electronic mail or (v) transmitted by hand delivery, telegram, or facsimile, addressed as set forth below or to such other address as such party shall have specified most recently by written notice. Any notice or other communication required or permitted to be given hereunder shall be deemed effective (a) upon hand delivery or delivery by facsimile, with accurate confirmation generated by the transmitting facsimile machine, at the address or number designated below (if delivered on a business day during normal business hours where such notice is to be received) or delivery via electronic mail, or the first business day following such delivery (if delivered other than on a business day during normal business hours where such notice is to be received) or (b) on the second business day following the date of mailing by express courier service, fully prepaid, addressed to such address, or upon actual receipt of such mailing, whichever shall first occur. The addresses for such communications shall be:
If to the Company, to:
NEWGENIVF GROUP LIMITED
36/39-36/40, 13th Floor, PS Tower, Sukhumvit 21 Road (Asoke),
Watthana District, B 10110, Thailand
Attn: Wing Fung Alfred Siu, CEO
If to the Buyer:
CFI CAPITAL LLC
2151 West Hillsboro Blvd.
Suite 209
Deerfield Beach, FL 33442
Attn: Ahron Fraiman, Manager
Each party shall provide notice to the other party of any change in address.
g. Successors and Assigns. This Agreement shall be binding upon and inure to the benefit of the parties and their successors and assigns. Neither the Company nor the Buyer shall assign this Agreement or any rights or obligations hereunder without the prior written consent of the other, which consent shall not be unreasonably withheld, conditioned, or delayed. Notwithstanding the foregoing, the Buyer may assign its rights hereunder to any affiliate (as that term is defined under the 1933 Act) of the Buyer without the consent of the Company, provided that (i) the Buyer delivers written notice to the Company at least five (5) business days prior to such assignment, (ii) such affiliate assignee delivers to the Company a written assumption agreement confirming that it is bound by all terms and conditions of this Agreement and the Note, and (iii) the Buyer provides the Company with an opinion of counsel from a reputable law firm confirming that such assignment is in compliance with applicable securities laws. Any purported assignment in violation of this Section 5(g) shall be null and void.
h. Third Party Beneficiaries. This Agreement is intended for the benefit of the parties hereto and their respective permitted successors and assigns, and is not for the benefit of, nor may any provision hereof be enforced by, any other person.
i. Survival. The representations and warranties of the Company and the agreements and covenants set forth in this Agreement shall survive the closing hereunder notwithstanding any due diligence investigation conducted by or on behalf of the Buyer. The Company agrees to indemnify and hold harmless the Buyer and all their officers, directors, employees, and agents for loss or damage arising as a result of or related to any breach or alleged breach by the Company of any of its representations, warranties, and covenants set forth in this Agreement or any of its covenants and obligations under this Agreement.
j. Further Assurances. Each party shall do and perform, or cause to be done and performed, all such further acts and things, and shall execute and deliver all such other agreements, certificates, instruments, and documents, as the other party may reasonably request in order to carry out the intent and accomplish the purposes of this Agreement and the consummation of the transactions contemplated hereby.
k. No Strict Construction. The language used in this Agreement will be deemed to be the language chosen by the parties to express their mutual intent, and no rules of strict construction will be applied against any party.
l. Remedies. The Company acknowledges that a breach by it of its obligations hereunder will cause irreparable harm to the Buyer by vitiating the intent and purpose of the transaction contemplated hereby. Accordingly, the Company acknowledges that the remedy at law for a breach of its obligations under this Agreement will be inadequate and agrees, in the event of a breach or threatened breach by the Company of the provisions of this Agreement, that the Buyer shall be entitled, in addition to all other available remedies at law or in equity, and in addition to the penalties assessable herein, to an injunction or injunctions restraining, preventing or curing any breach of this Agreement and to enforce specifically the terms and provisions hereof, without the necessity of showing economic loss and without any bond or other security being required.
[Signature page follows]
IN WITNESS WHEREOF, the undersigned Buyer and the Company have caused this Agreement to be duly executed as of the date first above written.
| NEWGENIVF GROUP LIMITED | ||
| By: | /s/ Wing Fung Alfred Siu | |
| Name: | Wing Fung Alfred Siu | |
| Title: | CEO | |
| CFI CAPITAL LLC | ||
| By: | /s/ Ahron Fraiman | |
| Name: | Ahron Fraiman | |
| Title: | Manager | |
AGGREGATE SUBSCRIPTION AMOUNT:
| Aggregate Principal Amount of the Note: | $200,000.00 |
| Aggregate Purchase Price: |
Note: $200,000.00, less $20,000.00 in original issue discount, and less $6,000.00 in legal fees.
EXHIBIT A
144 NOTE - $200,000
Exhibit 10.40
THIS NOTE AND THE COMMON STOCK ISSUABLE UPON CONVERSION OF THIS NOTE HAVE NOT BEEN AND WILL NOT BE REGISTERED WITH THE UNITED STATES SECURITIES AND EXCHANGE COMMISSION OR THE SECURITIES COMMISSION OF ANY STATE PURSUANT TO AN EXEMPTION FROM REGISTRATION PROVIDED UNDER THE SECURITIES ACT OF 1933, AS AMENDED, AND THE RULES AND REGULATIONS PROMULGATED THEREUNDER (THE “1933 ACT”)
| US $200,000.00 |
NEWGENIVF GROUP LIMITED
6% CONVERTIBLE REDEEMABLE NOTE
DUE MARCH 18, 2027
FOR VALUE RECEIVED, NEWGENIVF GROUP LIMITED (the “Company”) promises to pay to the order of CFI CAPITAL LLC and its authorized successors and Permitted Assigns, defined below, (the “Holder”), the aggregate principal face amount of Two Hundred Thousand Dollars exactly (U.S. $200,000.00) on March 18, 2027 (the “Maturity Date”) and to pay interest on the principal amount outstanding hereunder at the rate of 6% per annum commencing on March 18, 2026 (the “Issuance Date”). The interest will be paid to the Holder in whose name this 6% Convertible Redeemable Note (this “Note”) is registered on the records of the Company regarding registration and transfers of this Note. This Note shall contain a $20,000.00 original issue discount, such that the purchase price shall be $180,000.00. The principal of, and interest on, this Note are payable at 2151 West Hillsboro Blvd, Suite 209, Deerfield Beach, FL 33442, initially, and if changed, last appearing on the records of the Company as designated in writing by the Holder hereof from time to time. The Company will pay each interest payment and the outstanding principal due upon this Note before or on the Maturity Date, less any amounts required by law to be deducted or withheld, to the Holder of this Note by check or wire transfer addressed to such Holder at the last address appearing on the records of the Company. The forwarding of such check or wire transfer shall constitute a payment of the outstanding principal hereunder and shall satisfy and discharge the liability for the principal on this Note to the extent of the sum represented by such check or wire transfer. Interest shall be payable in Common Stock (as defined below) pursuant to paragraph 4(b) herein. Permitted Assigns means any “qualified person”, “permitted assigns” or “prospective transferee” acquiring all or a portion of this Note accompanied by an Opinion of Counsel, all in accordance with the terms provided in Sections 2(f) and 5(g) of the Securities Purchase Agreement by and between the Holder and the Company dated as of March 18, 2026 (the “Securities Purchase Agreement”).
This Note is subject to the following additional provisions:
1. This Note is exchangeable for an equal aggregate principal amount of Notes of different authorized denominations, as requested by the Holder surrendering the same. No service charge will be made for such registration or transfer, or exchange, except that Holder shall pay any tax or other governmental charges payable in connection therewith and for the cost of any Opinion of Counsel as maybe required under Section 5(g) of the Securities Purchase Agreement. To the extent that Holder subsequently transfers, assigns, sells or exchanges any of the multiple lesser denomination notes, Holder acknowledges that it will provide the Company with an Opinion of Counsel as provided for in Sections 2(f) and 5(g) of the Securities Purchase Agreement (“Opinions of Counsel”).
2. The Company shall be entitled to withhold from all payments any amounts required to be withheld under applicable laws.
3. This Note may be transferred or exchanged only in compliance with the Securities Act of 1933, as amended (“Act”), applicable state securities laws and Sections 2(f) and 5(f) of the Securities Purchase Agreement. Any attempted transfer to a non-qualifying party shall be treated by the Company as void. Prior to due presentment for transfer of this Note, the Company and any agent of the Company may treat the person in whose name this Note is duly registered on the Company’s records as the owner hereof for all other purposes, whether or not this Note be overdue, and neither the Company nor any such agent shall be affected or bound by notice to the contrary. Any Holder of this Note electing to exercise the right of conversion set forth in Section 4(a) hereof, in addition to the requirements set forth in Section 4(a), and any prequalified prospective transferee of this Note, also is required to give the Company written confirmation that this Note is being converted (“Notice of Conversion”) in the form annexed hereto as Exhibit A. The date of receipt (including receipt by telecopy) of such Notice of Conversion shall be the Conversion Date. All notices of conversion will be accompanied by an Opinion of Counsel of the Holder’s counsel, which the Company shall not reasonably reject.
| Initials | ||
4. (a) The Holder of this Note is entitled, at its option, at any time after the 6th monthly anniversary of this Note, to convert all or any amount of the principal face amount of this Note then outstanding into shares of the Company’s common stock (the “Common Stock”) at a price (“Conversion Price”) for each share of Common Stock equal to 75% of the lowest trading price of the Common Stock as reported on (i) the Nasdaq stock market or any other national securities exchange up on which the Common Stock is then listed, or (ii) if the Common Stock is not then listed on any national securities exchange, the OTC Markets or any other trading platform or quotation system on which the Common Stock is then traded or quoted (the “Exchange”) , for the ten prior trading days including the day upon which a Notice of Conversion is received by the Company (provided such Notice of Conversion is duly executed by the Holder and is delivered together with a duly executed Opinion of Counsel, by fax or other electronic method of communication to the Company after 4 P.M. Eastern Standard or Daylight Savings Time if the Holder wishes to include the same day closing price). For purposes of the above calculations, a day shall not be considered a trading day if there was no trading volume for the Company’s Common Stock for that particular day. If the shares have not been delivered within 2 business days, the Notice of Conversion may be rescinded. Such conversion shall be effectuated by the Company delivering the shares of Common Stock to the Holder within 2 business days of receipt by the Company of the Notice of Conversion. Accrued, but unpaid interest shall be subject to conversion. No fractional shares or scrip representing fractions of shares will be issued on conversion, but the number of shares issuable shall be rounded to the nearest whole share. To the extent the Conversion Price of the Company’s Common Stock closes below the par value per share, the Company will take all steps necessary to solicit the consent of the stockholders to reduce the par value to the lowest value possible under law or to conduct a reverse split at a ratio determined by the Company’s board of directors. The Company agrees to honor all conversions submitted pending this increase or such stock split, as applicable. In the event the Company experiences a DTC “Chill” on its shares, the conversion price shall be decreased to 65% instead of 75% while that “Chill” is in effect. In no event shall the Holder be allowed to effect a conversion if such conversion, along with all other shares of Company Common Stock beneficially owned by the Holder and its affiliates would exceed 4.99% of the outstanding shares of the Common Stock of the Company (which may be increased up to 9.9% upon 60 days’ prior written notice by the Holder to the Company).
(b) Interest on any unpaid principal balance of this Note shall be paid at the rate of 6% per annum. Interest shall be paid by the Company in Common Stock (“Interest Shares”). The Holder may, at any time, send in a Notice of Conversion to the Company for Interest Shares based on the formula provided in Section 4(a) above. The dollar amount converted into Interest Shares shall be all or a portion of the accrued interest calculated on the unpaid principal balance of this Note to the date of such notice.
(c) The Note may be prepaid or assigned with the following penalties/premiums:
| PREPAY DATE | PREPAY AMOUNT |
| ≤ 60 days | 105% of principal plus accrued interest |
| 60-120 days | 115% of principal plus accrued interest |
| 120-150 days | 120% of principal plus accrued interest |
| 150-180 days | 125% of principal plus accrued interest |
Such redemption must be closed and funded within 3 days of giving notice of redemption, or the right to redeem shall be null and void. Any partial prepayments will be made in accordance with the formula set forth in the chart above with respect to principal, premium, and interest.
|
|
||
| Initials | ||
(d) Upon (i) a transfer of all or substantially all of the assets of the Company to any person in a single transaction or series of related transactions, (ii) a reclassification, capital reorganization (excluding an increase in authorized capital) or other change or exchange of outstanding shares of the Common Stock, other than a forward or reverse stock split or stock dividend, or (iii) any consolidation or merger of the Company with or into another person or entity in which the Company is not the surviving entity (other than a merger which is effected solely to change the jurisdiction of incorporation of the Company and results in a reclassification, conversion or exchange of outstanding shares of Common Stock solely into shares of Common Stock) (each of items (i), (ii) and (iii) being referred to as a “Sale Event”), then, in each case, the Company shall, upon request of the Holder, redeem this Note in cash for the prepayment price set forth in Section 4(c), above, or at the election of the Holder, such Holder may convert the unpaid principal amount of this Note (together with the amount of accrued but unpaid interest) into shares of Common Stock immediately prior to such Sale Event at the Conversion Price. Notwithstanding the foregoing, the following transactions shall not constitute a “Sale Event” for purposes of this Section 4(d) and shall not trigger any redemption or conversion right of the Holder under this Section 4(d) (each, a “Permitted Transaction”): any merger, consolidation, reverse takeover, or business combination transaction approved by the Board of Directors of the Company in which the surviving or successor entity (1) assumes the Company’s obligations under this Note and the Securities Purchase Agreement pursuant to a written assumption agreement delivered to the Holder prior to or simultaneously with the closing of such transaction, and (2) has its shares of common stock or equivalent equity securities listed or quoted on a national securities exchange or recognized trading market following the closing of such transaction. For the avoidance of doubt, any Permitted Transaction shall not give rise to any right of the Holder to demand redemption, accelerate payment, adjust the Conversion Price, or exercise any other remedy under this Note solely by reason of the occurrence of such Permitted Transaction, provided that the successor or surviving entity has delivered the assumption agreement referred to above.
(e) In the event the Company is not able to pay the payment section forth in Section 4(d), above, then in case of any Sale Event (not to include a sale of all or substantially all of the Company’s assets) in connection with which this Note is not redeemed or converted, the Company shall cause effective provision to be made so that the Holder of this Note shall have the right thereafter, by converting this Note, to purchase or convert this Note into the kind and number of shares of stock or other securities or property (including cash) receivable upon such reclassification, capital reorganization or other change, consolidation or merger by a holder of the number of shares of Common Stock that could have been purchased upon exercise of the Note and at the same Conversion Price, as defined in this Note, immediately prior to such Sale Event. The foregoing provisions shall similarly apply to successive Sale Events. If the consideration received by the holders of Common Stock is other than cash, the value shall be as determined by the Board of Directors of the Company or successor person or entity acting in good faith.
5. No provision of this Note shall alter or impair the obligation of the Company, which is absolute and unconditional, to pay the principal of, and interest on, this Note at the time, place, and rate, and in the form, herein prescribed.
|
|
||
| Initials | ||
6. The Company hereby expressly waives demand and presentment for payment, notice of non-payment, protest, notice of protest, notice of dishonor, notice of acceleration or intent to accelerate, and diligence in taking any action to collect amounts called for hereunder and shall be directly and primarily liable for the payment of all sums owing and to be owing hereto.
7. The Company agrees to pay all reasonable costs and expenses, including reasonable attorneys’ fees and expenses, which may be incurred by the Holder in collecting any amount due under this Note.
8. If one or more of the following described “Events of Default” shall occur:
(a) The Company shall default in the payment of principal or interest on this Note or any other note issued to the Holder by the Company; or
(b) Any of the representations or warranties made by the Company herein or in any certificate or financial or other written statements heretofore or hereafter furnished by or on behalf of the Company in connection with the execution and delivery of this Note, or the Securities Purchase Agreement under which this note was issued shall be false or misleading in any material respect; or
(c) The Company shall fail to perform or observe, in any respect, any covenant, term, provision, condition, agreement or obligation of the Company under this Note; or
(d) The Company shall (1) become insolvent (which does not include a “going concern opinion”); (2) admit in writing its inability to pay its debts generally as they mature; (3) make an assignment for the benefit of creditors or commence proceedings for its dissolution; (4) apply for or consent to the appointment of a trustee, liquidator or receiver for its or for a substantial part of its property or business; (5) file a petition for bankruptcy relief, consent to the filing of such petition or have filed against it an involuntary petition for bankruptcy relief, all under federal or state laws as applicable; or
(e) A trustee, liquidator, or receiver shall be appointed for the Company or for a substantial part of its property or business without its consent and shall not be discharged within sixty (60) days after such appointment; or
(f) Any governmental agency or any court of competent jurisdiction at the instance of any governmental agency shall assume custody or control of the whole or any substantial portion of the properties or assets of the Company; or
(g) One or more money judgments, writs or warrants of attachment, or similar process, in excess of two hundred fifty thousand dollars ($250,000) in the aggregate, shall be entered or filed against the Company or any of its properties or other assets and shall remain unpaid, unvacated, unbonded or unstayed for a period of fifteen (15) days or in any event later than five (5) days prior to the date of any proposed sale thereunder; or
(h) The Company has defaulted on or breached any term of any purchase agreement, convertible note, or similar debt instrument entered into between the Company and the Holder and failed to cure such default within the appropriate grace period; or
(i) The Company shall have its Common Stock delisted from the Nasdaq Stock Market or any other national securities exchange on which the Common Stock is then listed,or trading in the Common Stock shall be suspended for more than 10 consecutive days or ceases to file its reports under the Securities Exchange Act of 1934, as amended, with the SEC;
|
|
||
| Initials | ||
(j) The Company shall not deliver to the Holder the Common Stock pursuant to paragraph 4 herein without a restrictive legend within 2 business days of its receipt of a Notice of Conversion which includes a duly executed Opinion of Counsel from a reputable lawyer or law firm expressing an opinion which supports the removal of a restrictive legend; or
(k) The Company shall not replenish the reserve set forth in Section 12, within 2 business days of the written request of the Holder.
(l) The Company shall be delinquent in its periodic report filings with the Securities and Exchange Commission (subject to applicable extensions); or
(m) The Common Stock shall cease to have a publicly displayed bid price on any trading platform or exchange on which it is then listed or quoted, for a period of five (5) or more consecutive trading days, other than as a result of a general market closure, exchange-wide trading halt, or suspension not specific to the Company’s securities; or; or
(n) Terminate its existing transfer agent relationship without the prior written consent of the Holder; or
(l) Supplemental to 8(i), above, the Company shall have its stock delisted from the Nasdaq stock exchange.
Then, or at any time thereafter, unless cured within 10 business days, and in each and every such case, unless such Event of Default shall have been waived in writing by the Holder (which waiver shall not be deemed to be a waiver of any subsequent default) at the option of the Holder and in the Holder’s sole discretion, the Holder may consider this Note immediately due and payable, without presentment, demand, protest or (further) notice of any kind (other than notice of acceleration), all of which are hereby expressly waived, anything herein or in any note or other instruments contained to the contrary notwithstanding, and the Holder may immediately, and without expiration of any period of grace, enforce any and all of the Holder’s rights and remedies provided herein or any other rights or remedies afforded by law. Upon an Event of Default, interest shall accrue at a default interest rate at the highest rate of interest permitted by law, and the Conversion Price shall be adjusted from 75% to 60% (the conversion price discount shall increase by 15%). In the event of a breach of Section 8(j), the penalty shall be $500 per day if the shares are not issued beginning on the 3rd day after the conversion notice was delivered to the Company. This penalty shall increase to $1,000 per day beginning on the 10th day. In the event of a breach of Section 8(h), the Holder may elect to utilize the same remedy available under the defaulted interest, and such remedy shall be incorporated by reference into the terms of this Note. Further, if a breach of Section 8(l) occurs or is continuing after the 6-month anniversary of the Note, then the Holder shall be entitled to use the lowest closing bid price during the delinquency period as a base price for the conversion. For example, if the lowest closing bid price during the delinquency period is $0.01 per share and the conversion discount is 50%, the Holder may elect to convert future conversions at $0.005 per share. In the event of a breach of Section 8(l) then the Conversion Price set forth in Section 4(a) shall be changed from a discount of 75% of the lowest trading price of the Common Stock as reported on (i) the Nasdaq stock market or any other national securities exchange on which the Common Stock is then listed, or (ii) if the Common Stock is not then listed on any national securities exchange, the OTC Markets or any other trading platform or quotation system on which the Common Stock is then traded or quoted (the “Exchange”), for the ten prior trading days to a discount of 65% of the lowest trading price of the Common Stock as reported on the (i) the Nasdaq Stock Market or any other national securities exchange on which the Common Stock is then listed, or (ii) if the Common Stock is not then listed on any national securities exchange, the OTC Markets or any other trading platform or quotation system on which the Common Stock is then traded or quoted (the “Exchange”), for the twenty prior trading days, and the prepayment penalty set forth in Section 4(c) shall be increased by 10%.
|
|
||
| Initials | ||
If the Holder shall commence an action or proceeding to enforce any provisions of this Note, including, without limitation, engaging an attorney, then if the Holder prevails in such action, the Holder shall be reimbursed by the Company for its attorneys’ fees and other costs and expenses incurred in the investigation, preparation, and prosecution of such action or proceeding.
9. In case any provision of this Note is held by a court of competent jurisdiction to be excessive in scope or otherwise invalid or unenforceable, such provision shall be adjusted rather than voided, if possible, so that it is enforceable to the maximum extent possible, and the validity and enforceability of the remaining provisions of this Note will not in any way be affected or impaired thereby. Specifically, notwithstanding any provision to the contrary, the parties acknowledge and agree that the Company is a corporation and the loan evidenced by this Note is made solely for business and commercial purposes. Accordingly, pursuant to Florida Statutes § 687.031 and applicable Florida case law, the provisions of Florida usury law, including §§ 687.02 and 687.03, shall not apply to this transaction. The Holder and Company further acknowledge that no natural person is guaranteeing the obligations under this Note, and the proceeds are not intended for consumer or personal use. In entering into this Note, each party has independently evaluated the terms, including any conversion rights, and agrees that the structure and pricing reflect a bona fide commercial transaction outside the scope of Florida usury laws.
10. Neither this Note nor any term hereof may be amended, waived, discharged, or terminated other than by a written instrument signed by the Company and the Holder.
11. The Company represents that it is not a “shell” issuer and has never been a “shell” issuer or that if it previously has been a “shell” issuer at least 12 months have passed since the Company has reported form 10 type information indicating it is no longer a “shell” issuer. Further, the Company will instruct its counsel to either (i) write a 144 opinion to allow for the salability of the conversion shares or (ii) accept such opinion from Holder’s counsel.
12. The Company shall issue irrevocable transfer agent instructions reserving 489,297 shares of its Common Stock for conversions under this Note (the “Share Reserve”). Upon full conversion of this Note, any shares remaining in the Share Reserve shall be canceled. The Company should at the Holder’s request, reserve a four times the amount of shares required if the note would be fully converted. The Holder may reasonably request increases from time to time to reserve such amounts to maintain such four times coverage, and the Company will be responsible for all fees associated with the increase in the Share Reserve. The Company will instruct its transfer agent to provide the outstanding share information to the Holder in connection with its conversions, along with shareholder information statements and other shareholder reservations that exist. The Holder shall be entitled to deduct $1,000 per conversion to adequately cover all transfer agent costs and legal fees associated with issuing and delivering the shares to the Holder.
13. If it shall be found that any interest or other amount deemed interest due hereunder violates the applicable law governing usury, the applicable provision shall automatically be revised to equal the maximum rate of interest or other amount deemed interest permitted under applicable law. The Company covenants (to the extent that it may lawfully do so) that it will not seek to claim or take advantage of any law that would prohibit or forgive the Company from paying all or a portion of the principal or interest on this Note.
|
|
||
| Initials | ||
15. (a) Nasdaq Listing Condition. The provisions of this Section 15 shall only take effect, and shall only be operative, upon and following the date on which the Common Stock is delisted from the Nasdaq Stock Market and is no longer listed or admitted to trading on any national securities exchange registered under Section 6 of the Securities Exchange Act of 1934, as amended (a “Delisting Event”). For so long as the Common Stock remains listed on the Nasdaq Stock Market or any other such national securities exchange, this Section 15 shall be of no force or effect.
(b) MFN Rights Following a Delisting Event. Following a Delisting Event, any issuance by the Company or any of its subsidiaries of a convertible promissory note or other convertible debt instrument with any term more favorable to the holder of such instrument or with a term (including without limitation any Conversion Price) in favor of the holder of such instrument that was not similarly provided to the Holder in this Note (other than a future financing with the Holder), then the Company shall notify the Holder of such additional or more favorable term and such term, at the Holder’s option, shall become a part of the Note with the Holder. The types of terms contained in such convertible debt instrument that may be more favorable to the holder of such instrument include, but are not limited to, terms addressing conversion discounts, prepayment rate, conversion lookback periods, interest rates, original issue discounts.
(c) Excluded Issuances. Notwithstanding Section 15(b), the following issuances shall not trigger any MFN right of the Holder under this Section 15, whether before or after a Delisting Event: (i) any issuance of equity securities (including ordinary shares, preferred shares, or warrants) pursuant to a registered direct offering or any other offering registered under the Securities Act of 1933, as amended; (ii) any issuance of securities in connection with a merger, acquisition, reverse takeover, business combination, joint venture, or other strategic transaction approved by the Board of Directors of the Company (each, a “Permitted Issuance”); and (iii) any issuance of securities to employees, officers, directors, or consultants pursuant to a Board-approved equity incentive plan.
16. This Note shall be governed by and construed in accordance with the laws of the State of Florida without regard to principles of conflicts of laws. Any action brought by either party against the other concerning the transactions contemplated by this Agreement shall be brought only in the state courts of Florida located in Miami, Florida, or in the federal courts located in the state of Florida in the Southern District of Florida. The parties to this Agreement hereby irrevocably waive any objection to jurisdiction and venue of any action instituted hereunder and shall not assert any defense based on lack of jurisdiction or venue or based upon forum non conveniens. The Company and Buyer waive the trial by jury. The prevailing party shall be entitled to recover from the other party its reasonable attorney’s fees and costs. In the event that any provision of this Agreement or any other agreement delivered in connection herewith is invalid or unenforceable under any applicable statute or rule of law, then such provision shall be deemed inoperative to the extent that it may conflict therewith and shall be deemed modified to conform with such statute or rule of law. Any such provision which may prove invalid or unenforceable under any law shall not affect the validity or enforceability of any other provision of any agreement. Each party hereby irrevocably waives personal service of process and consents to process being served in any suit, action, or proceeding in connection with this Agreement or any other Transaction Document by mailing a copy thereof via registered or certified mail or overnight delivery (with evidence of delivery) to such party at the address in effect for notices to it under this Agreement and agrees that such service shall constitute good and sufficient service of process and notice thereof. Nothing contained herein shall be deemed to limit in any way any right to serve process in any other manner permitted by law. This Agreement may be executed in counterparts, and the facsimile transmission of an executed counterpart to this Agreement shall be effective as an original.
[Signature page follows]
|
|
||
| Initials | ||
IN WITNESS WHEREOF, the Company has caused this Note to be duly executed by an officer thereunto duly authorized.
| Dated: | March 18, 2026 |
| NEWGENIVF GROUP LIMITED | ||
| By: | /s/ Wing Fung Alfred Siu | |
| Name: | Wing Fung Alfred Siu | |
| Title: | CEO | |
| Initials | ||
EXHIBIT A
NOTICE OF CONVERSION
(To be Executed by the Registered Holder in order to Convert the Note)
The undersigned hereby irrevocably elects to convert *$___________ of the above Note into _________ Shares of Common Stock of NEWGENIVF GROUP LIMITED (“Shares”) according to the conditions set forth in such Note, as of the date written below.
If Shares are to be issued in the name of a person other than the undersigned, the undersigned will pay all transfer and other taxes and charges payable with respect thereto.
Date of Conversion:
Applicable Conversion Price:
Signature:
[Print Name of Holder and Title of Signer]
Address:
SSN or EIN:
Shares are to be registered in the following name:
Name:
Address:
Tel:
Fax:
SSN or EIN:
Shares are to be sent or delivered to the following account:
Account Name:
Address:
* consist of $____ in principal, $____ in interest and $___ in fees pursuant to section 12 of the Note
| Initials | ||
Exhibit 10.41
SECURITIES PURCHASE AGREEMENT
This SECURITIES PURCHASE AGREEMENT (the “Agreement”), dated as of March 25, 2026, by and between NewGenIvf Group Limited, a British Virgin Islands company, with headquarters located at 36/39-36/40, 13th Floor, PS Tower, Sukhumvit 21 Road (Asoke), Khlong Toei Nuea Sub-district, Watthana District, Bangkok 10110, Thailand (the “Company”), and FIRSTFIRE GLOBAL OPPORTUNITIES FUND, LLC, a Delaware limited liability company, with its address at 1040 First Avenue, Suite 190, New York, NY 10022 (the “Buyer”).
WHEREAS:
A. The Company and the Buyer are executing and delivering this Agreement in reliance upon the exemption from securities registration afforded by Section 4(a)(2) of the Securities Act of 1933, as amended (the “1933 Act”) and Rule 506(b) promulgated by the United States Securities and Exchange Commission (the “SEC”) under the 1933 Act;
B. Buyer desires to purchase from the Company, and the Company desires to issue and sell to the Buyer, upon the terms and conditions set forth in this Agreement, a promissory note of the Company, in the aggregate principal amount of $220,000.00 (as the principal amount thereof may be increased pursuant to the terms thereof, and together with any note(s) issued in replacement thereof or as a dividend thereon or otherwise with respect thereto in accordance with the terms thereof, in the form attached hereto as Exhibit A, the “Note”), convertible into Class A ordinary shares of the Company, no par value per share (the “Common Stock”), upon the terms and subject to the limitations and conditions set forth in such Note; and
C. The Buyer wishes to purchase, upon the terms and conditions stated in this Agreement, such principal amount of the Note as is set forth in this Agreement; and
D. The Company wishes to issue 20,000 shares of Common Stock (the “Commitment Shares”) to the Buyer as additional consideration for the purchase of the Note, which all shall be earned in full as of the Closing Date, as further provided herein.
NOW THEREFORE, in consideration of the foregoing and of the agreements and covenants herein contained, and for other good and valuable consideration, the receipt and sufficiency of which is hereby acknowledged, the Company and the Buyer hereby agree as follows:
1. Purchase and Sale of Note.
a. Purchase of Note. On the Closing Date (as defined below), the Company shall issue and sell to the Buyer, and the Buyer agrees to purchase from the Company, the Note, as further provided herein. As used in this Agreement, the term “business day” shall mean any day other than a Saturday, Sunday, or a day on which commercial banks in the city of New York, New York are authorized or required by law or executive order to remain closed.
b. Form of Payment. On the Closing Date: (i) the Buyer shall pay the purchase price of $200,000.00 (the “Purchase Price”) for the Note, to be issued and sold to it at the Closing (as defined below), by wire transfer of immediately available funds to the Company, in accordance with the Company’s written wiring instructions, against delivery of the Note, and (ii) the Company shall deliver such duly executed Note on behalf of the Company, to the Buyer, against delivery of such Purchase Price. On the Closing Date, the Buyer shall withhold $6,000.00 from the Purchase Price to cover the Buyer’s legal fees in connection with the transactions contemplated by this Agreement.
c. Closing Date. Subject to the satisfaction (or written waiver) of the conditions thereto set forth in Section 6 and Section 7 below, the date and time of the issuance and sale of the Note pursuant to this Agreement (the “Closing Date”) shall be on the date that the Purchase Price for the Note is paid by Buyer pursuant to terms of this Agreement.
d. Closing. The closing of the transactions contemplated by this Agreement (the “Closing”) shall occur on the Closing Date at such location as may be agreed to by the parties (including via exchange of electronic signatures).
e. Commitment Shares. On or before the Closing Date, the Company shall issue the Commitment Shares to the Buyer, which shall be earned in full as of the Closing Date.
2. Buyer’s Representations and Warranties. The Buyer represents and warrants to the Company as of the Closing Date that:
a. Investment Purpose. As of the Closing Date, the Buyer is purchasing the Note (the Note, Commitment Shares, and shares of Common Stock issuable upon conversion of or otherwise pursuant to the Note (the “Conversion Shares”) shall collectively be referred to herein as the “Securities”) for its own account and not with a present view towards the public sale or distribution thereof, except pursuant to sales registered or exempted from registration under the 1933 Act; provided, however, that by making the representations herein, the Buyer does not agree to hold any of the Securities for any minimum or other specific term and reserves the right to dispose of the Securities at any time in accordance with or pursuant to a registration statement or an exemption under the 1933 Act.
b. Accredited Investor Status. The Buyer is an “accredited investor” as that term is defined in Rule 501(a) of Regulation D (an “Accredited Investor”).
c. Reliance on Exemptions. The Buyer understands that the Securities are being offered and sold to it in reliance upon specific exemptions from the registration requirements of United States federal and state securities laws and that the Company is relying upon the truth and accuracy of, and the Buyer’s compliance with, the representations, warranties, agreements, acknowledgments and understandings of the Buyer set forth herein in order to determine the availability of such exemptions and the eligibility of the Buyer to acquire the Securities.
d. Information. The Buyer and its advisors, if any, have been, and for so long as the Note remains outstanding will continue to be, furnished with all materials relating to the business, finances and operations of the Company and materials relating to the offer and sale of the Securities which have been requested by the Buyer or its advisors. The Buyer and its advisors, if any, have been, and for so long as the Note remains outstanding will continue to be, afforded the opportunity to ask questions of the Company regarding its business and affairs. Notwithstanding the foregoing, the Company has not disclosed to the Buyer any material nonpublic information regarding the Company or otherwise and will not disclose such information unless such information is disclosed to the public prior to or promptly following such disclosure to the Buyer. Neither such inquiries nor any other due diligence investigation conducted by Buyer or any of its advisors or representatives shall modify, amend or affect Buyer’s right to rely on the Company’s representations and warranties contained in Section 3 below.
e. Governmental Review. The Buyer understands that no United States federal or state agency or any other government or governmental agency has passed upon or made any recommendation or endorsement of the Securities.
f. Transfer or Re-sale. The Buyer understands that (i) the sale or resale of the Securities has not been and is not being registered under the 1933 Act or any applicable state securities laws, and the Securities may not be transferred unless (a) the Securities are sold pursuant to an effective registration statement under the 1933 Act, (b) the Buyer shall have delivered to the Company, at the cost of the Company, an opinion of counsel (which may be the Legal Counsel Opinion (as defined below)) that shall be in form, substance and scope customary for opinions of counsel in comparable transactions to the effect that the Securities to be sold or transferred may be sold or transferred pursuant to an exemption from such registration, which opinion shall be accepted by the Company, (c) the Securities are sold or transferred to an “affiliate” (as defined in Rule 144 promulgated under the 1933 Act (or a successor rule) (“Rule 144”)) of the Buyer who agrees to sell or otherwise transfer the Securities only in accordance with this Section 2(f) and who is an Accredited Investor, (d) the Securities are sold pursuant to Rule 144 or other applicable exemption, or (e) the Securities are sold pursuant to Regulation S under the 1933 Act (or a successor rule) (“Regulation S”), and the Buyer shall have delivered to the Company, at the cost of the Company, an opinion of counsel that shall be in form, substance and scope customary for opinions of counsel in corporate transactions, which opinion shall be accepted by the Company; (ii) any sale of such Securities made in reliance on Rule 144 may be made only in accordance with the terms of said Rule and further, if said Rule is not applicable, any re-sale of such Securities under circumstances in which the seller (or the person through whom the sale is made) may be deemed to be an underwriter (as that term is defined in the 1933 Act) may require compliance with some other exemption under the 1933 Act or the rules and regulations of the SEC thereunder; and (iii) neither the Company nor any other person is under any obligation to register such Securities under the 1933 Act or any state securities laws or to comply with the terms and conditions of any exemption thereunder (in each case). Notwithstanding the foregoing or anything else contained herein to the contrary, the Securities may be pledged in connection with a bona fide margin account or other lending arrangement secured by the Securities, and such pledge of Securities shall not be deemed to be a transfer, sale or assignment of the Securities hereunder, and the Buyer in effecting such pledge of Securities shall not be required to provide the Company with any notice thereof or otherwise make any delivery to the Company pursuant to this Agreement or otherwise.
g. Legends. The Buyer understands that until such time as the Note and/or Conversion Shares have been registered under the 1933 Act or may be sold pursuant to Rule 144, Rule 144A under the 1933 Act, Regulation S, or other applicable exemption without any restriction as to the number of securities as of a particular date that can then be immediately sold, the Securities may bear a restrictive legend in substantially the following form (and a stop-transfer order may be placed against transfer of such Securities):
“NEITHER THE ISSUANCE AND SALE OF THE SECURITIES REPRESENTED BY THIS CERTIFICATE NOR THE SECURITIES INTO WHICH THESE SECURITIES ARE CONVERTIBLE HAVE BEEN REGISTERED UNDER THE SECURITIES ACT OF 1933, AS AMENDED, OR APPLICABLE STATE SECURITIES LAWS. THE SECURITIES MAY NOT BE OFFERED FOR SALE, SOLD, TRANSFERRED OR ASSIGNED (I) IN THE ABSENCE OF (A) AN EFFECTIVE REGISTRATION STATEMENT FOR THE SECURITIES UNDER THE SECURITIES ACT OF 1933, AS AMENDED, OR (B) AN OPINION OF COUNSEL (WHICH COUNSEL SHALL BE SELECTED BY THE HOLDER), IN A GENERALLY ACCEPTABLE FORM, THAT REGISTRATION IS NOT REQUIRED UNDER SAID ACT OR (II) UNLESS SOLD PURSUANT TO RULE 144, RULE 144A, REGULATION S, OR OTHER APPLICABLE EXEMPTION UNDER SAID ACT. NOTWITHSTANDING THE FOREGOING, THE SECURITIES MAY BE PLEDGED IN CONNECTION WITH A BONA FIDE MARGIN ACCOUNT OR OTHER LOAN OR FINANCING ARRANGEMENT SECURED BY THE SECURITIES.”
The legend set forth above shall be removed and the Company shall issue a certificate or book entry statement for the applicable shares of Common Stock without such legend to the holder of any Security upon which it is stamped or (as requested by such holder) issue the applicable shares of Common Stock to such holder by electronic delivery by crediting the account of such holder’s broker with The Depository Trust Company (“DTC”), if, unless otherwise required by applicable state securities laws, (a) such Security is registered for sale under an effective registration statement filed under the 1933 Act or otherwise may be sold pursuant to Rule 144, Rule 144A, Regulation S, or other applicable exemption without any restriction as to the number of securities as of a particular date that can then be immediately sold, or (b) the Company or the Buyer provides the Legal Counsel Opinion (as contemplated by and in accordance with Section 4(l) hereof) to the effect that a public sale or transfer of such Security may be made without registration under the 1933 Act, which opinion shall be accepted by the Company so that the sale or transfer is effected. The Company shall be responsible for the fees of its transfer agent and all DTC fees associated with any such issuance. The Buyer agrees to sell all Securities, including those represented by a certificate(s) from which the legend has been removed, in compliance with applicable prospectus delivery requirements, if any. In the event that the Company does not accept the opinion of counsel provided by the Buyer with respect to the transfer of Securities pursuant to an exemption from registration, such as Rule 144, Rule 144A, Regulation S, or other applicable exemption at the Deadline (as defined in the Note), it will be considered an Event of Default pursuant to Section 3.2 of the Note.
h. Authorization; Enforcement. This Agreement has been duly and validly authorized by the Buyer and has been duly executed and delivered on behalf of the Buyer, and this Agreement constitutes a valid and binding agreement of the Buyer enforceable in accordance with its terms, except as enforcement may be limited by bankruptcy, insolvency, reorganization, moratorium or other similar laws affecting creditors’ rights generally and except as may be limited by the exercise of judicial discretion in applying principles of equity.
3. Representations and Warranties of the Company. The Company represents and warrants to the Buyer as of the Closing Date that:
a. Organization and Qualification. The Company and each of its Subsidiaries (as defined below), if any, is a corporation duly organized, validly existing and in good standing under the laws of the jurisdiction in which it is incorporated or formed, with full power and authority (corporate and other) to own, lease, use and operate its properties and to carry on its business as and where now owned, leased, used, operated and conducted. The SEC Documents set forth a list of all of the Subsidiaries of the Company and the jurisdiction in which each is incorporated. The Company and each of its Subsidiaries is duly qualified to do business and is in good standing in every jurisdiction in which its ownership or use of property or the nature of the business conducted by it makes such qualification necessary except where the failure to be so qualified or in good standing would not have a Material Adverse Effect. “Material Adverse Effect” means any material adverse effect on the business, operations, assets, financial condition or prospects of the Company or its Subsidiaries, if any, taken as a whole, or on the transactions contemplated hereby or by the agreements or instruments to be entered into in connection herewith. “Subsidiaries” means any corporation or other organization, whether incorporated or unincorporated, in which the Company owns, directly or indirectly, any equity or other ownership interest.
b. Authorization; Enforcement. The Company and Subsidiaries have all requisite corporate power and authority to enter into and perform the Transaction Documents and to consummate the transactions contemplated hereby and thereby and to issue the Securities, in accordance with the terms hereof and thereof. The Company represents and warrants that (i) the execution and delivery of the Transaction Documents, the Note, and Conversion Shares by the Company and the consummation by it of the transactions contemplated hereby and thereby (including without limitation, the issuance of the Note as well as the issuance and reservation for issuance of the Conversion Shares issuable upon conversion of the Note) have been duly authorized by the Company’s Board of Directors and no further consent or authorization of the Company, its Board of Directors, its shareholders, or its debt holders is required, (i) the Transaction Documents (together with any other instruments executed in connection herewith or therewith) have been duly executed and delivered by the Company and Subsidiaries by its authorized representatives, and such authorized representatives are the true and official representative with authority to sign the Transaction Documents and the other instruments documents executed in connection herewith or therewith and bind the Company and Subsidiaries accordingly, and (iii) the Transaction Documents constitute, and upon execution and delivery by the Company and Subsidiaries as applicable, each of such instruments will constitute, a legal, valid and binding obligation of the Company and Subsidiaries, enforceable against the Company and Subsidiaries in accordance with their terms.
c. Capitalization; Governing Documents. As of March 25, 2026, the authorized capital stock of the Company consists of: (i) unlimited authorized shares of Common Stock, of which 568,473 shares were issued and outstanding, (ii) unlimited authorized shares of Class B ordinary shares, of which 69 shares were issued and outstanding, and (iii) unlimited authorized shares of preferred stock, of which 0 shares were issued and outstanding. All of such outstanding shares of capital stock of the Company and the Conversion Shares are, or upon issuance will be, duly authorized, validly issued, fully paid and non-assessable. No shares of capital stock of the Company are subject to preemptive rights or any other similar rights of the shareholders of the Company or any liens or encumbrances imposed through the actions or failure to act of the Company. As of the effective date of this Agreement, other than: (i) as publicly announced prior to such date and reflected in the SEC Documents of the Company and; (ii) the securities issued to CFI Capital LLC pursuant to a securities purchase agreement dated on March 18, 2026, (i) there are no outstanding options, warrants, scrip, rights to subscribe for, puts, calls, rights of first refusal, agreements, understandings, claims or other commitments or rights of any character whatsoever relating to, or securities or rights convertible into or exchangeable for any shares of capital stock of the Company or any of its Subsidiaries, or arrangements by which the Company or any of its Subsidiaries is or may become bound to issue additional shares of capital stock of the Company or any of its Subsidiaries, (ii) there are no agreements or arrangements under which the Company or any of its Subsidiaries is obligated to register the sale of any of its or their securities under the 1933 Act and (iii) there are no anti-dilution or price adjustment provisions contained in any security issued by the Company (or in any agreement providing rights to security holders) that will be triggered by the issuance of any of the Securities. The Company has furnished to the Buyer true and correct copies of the Company’s Certificate of Incorporation as in effect on the date hereof (“Certificate of Incorporation”), the Company’s By-laws, as in effect on the date hereof (the “By-laws”), and the terms of all securities convertible into or exercisable for Common Stock of the Company and the material rights of the holders thereof in respect thereto.
d. Issuance of Conversion Shares. The Conversion Shares are duly authorized and reserved for issuance and, upon conversion of the Note in accordance with its terms, will be validly issued, fully paid and non-assessable, and free from all taxes, liens, claims and encumbrances with respect to the issue thereof and shall not be subject to preemptive rights or other similar rights of shareholders of the Company and will not impose personal liability upon the holder thereof.
e. Commitment Shares. The issuance of the Commitment Shares are duly authorized and will be validly issued, fully paid and non-assessable, and free from all taxes, liens, claims and encumbrances with respect to the issue thereof and shall not be subject to preemptive rights or other similar rights of shareholders of the Company and will not impose personal liability upon the holder thereof.
f. Acknowledgment of Dilution. The Company understands and acknowledges the potentially dilutive effect of the Commitment Shares, Conversion Shares upon the conversion of the Note to the Common Stock. The Company further acknowledges that its obligation to issue, upon conversion of the Note, the Conversion Shares, are absolute and unconditional regardless of the dilutive effect that such issuance may have on the ownership interests of other shareholders of the Company.
g. No Conflicts. The execution, delivery and performance of the Transaction Documents by the Company and Subsidiaries, and the consummation by the Company and Subsidiaries of the transactions contemplated hereby and thereby (including, without limitation, the issuance and reservation for issuance of the Conversion Shares) will not (i) conflict with or result in a violation of any provision of the Certificate of Incorporation or By-laws, or (ii) violate or conflict with, or result in a breach of any provision of, or constitute a default (or an event which with notice or lapse of time or both could become a default) under, or give to others any rights of termination, amendment, acceleration or cancellation of, any agreement, note, evidence of indebtedness, indenture, patent, patent license or instrument to which the Company or any of its Subsidiaries is a party, or (iii) result in a violation of any law, rule, regulation, order, judgment or decree (including federal and state securities laws and regulations and regulations of any self-regulatory organizations to which the Company or its securities is subject) applicable to the Company or any of its Subsidiaries or by which any property or asset of the Company or any of its Subsidiaries is bound or affected (except for such conflicts, defaults, terminations, amendments, accelerations, cancellations and violations as would not, individually or in the aggregate, have a Material Adverse Effect), or (iv) trigger any anti-dilution and/or ratchet provision contained in any other contract in which the Company is a party thereto or any security issued by the Company. Neither the Company nor any of its Subsidiaries is in violation of its Certificate of Incorporation, By-laws or other organizational documents and neither the Company nor any of its Subsidiaries is in default (and no event has occurred which with notice or lapse of time or both could put the Company or any of its Subsidiaries in default) under, and neither the Company nor any of its Subsidiaries has taken any action or failed to take any action that would give to others any rights of termination, amendment, acceleration or cancellation of, any agreement, indenture or instrument to which the Company or any of its Subsidiaries is a party or by which any property or assets of the Company or any of its Subsidiaries is bound or affected, except for possible defaults as would not, individually or in the aggregate, have a Material Adverse Effect. The businesses of the Company and its Subsidiaries, if any, are not being conducted, and shall not be conducted so long as the Buyer owns any of the Securities, in violation of any law, ordinance or regulation of any governmental entity. Except as specifically contemplated by this Agreement and as required under the 1933 Act and any applicable state securities laws, the Company is not required to obtain any consent, authorization or order of, or make any filing or registration with, any court, governmental agency, regulatory agency, self-regulatory organization or stock market or any third party in order for it to execute, deliver or perform any of its obligations under this Agreement and the Note in accordance with the terms hereof or thereof or to issue and sell the Note in accordance with the terms hereof and, upon conversion of the Note, issue Conversion Shares. All consents, authorizations, orders, filings and registrations which the Company is required to obtain pursuant to the preceding sentence have been obtained or effected on or prior to the date hereof. The Company is not in violation of the listing requirements of the Principal Market (as defined herein) and does not reasonably anticipate that the Common Stock will be delisted by the Principal Market in the foreseeable future. The Company and its Subsidiaries are unaware of any facts or circumstances which might give rise to any of the foregoing. The “Principal Market” shall mean the principal securities exchange or trading market where such Common Stock is listed or traded, including but not limited to any tier of the OTC Markets, any tier of the NASDAQ Stock Market (including NASDAQ Capital Market), or the NYSE American, or any successor to such markets.
h. SEC Documents; Financial Statements. The Company has filed all reports, schedules, forms, statements and other documents required to be filed by it with the SEC pursuant to the reporting requirements of the Securities Exchange Act of 1934, as amended (the “1934 Act”) (all of the foregoing filed prior to the date hereof and all exhibits included therein and financial statements and schedules thereto and documents (other than exhibits to such documents) incorporated by reference therein, being hereinafter referred to herein as the “SEC Documents”). As of their respective dates, the SEC Documents complied in all material respects with the requirements of the 1934 Act and the rules and regulations of the SEC promulgated thereunder applicable to the SEC Documents, and none of the SEC Documents, at the time they were filed with the SEC, contained any untrue statement of a material fact or omitted to state a material fact required to be stated therein or necessary in order to make the statements therein, in light of the circumstances under which they were made, not misleading. None of the statements made in any such SEC Documents is, or has been, required to be amended or updated under applicable law (except for such statements as have been amended or updated in subsequent filings prior the date hereof). As of their respective dates, the financial statements of the Company included in the SEC Documents complied as to form in all material respects with applicable accounting requirements and the published rules and regulations of the SEC with respect thereto. Such financial statements have been prepared in accordance with United States generally accepted accounting principles, consistently applied, during the periods involved and fairly present in all material respects the consolidated financial position of the Company and its consolidated Subsidiaries as of the dates thereof and the consolidated results of their operations and cash flows for the periods then ended (subject, in the case of unaudited statements, to normal year-end audit adjustments). Except as set forth in the financial statements of the Company included in the SEC Documents, the Company has no liabilities, contingent or otherwise, other than (i) liabilities incurred in the ordinary course of business subsequent to September 30, 2025, and (ii) obligations under contracts and commitments incurred in the ordinary course of business and not required under generally accepted accounting principles to be reflected in such financial statements, which, individually or in the aggregate, are not material to the financial condition or operating results of the Company. The Company is subject to the reporting requirements of the 1934 Act.
i. Absence of Certain Changes. Since September 30, 2025, there has been no material adverse change and no material adverse development in the assets, liabilities, business, properties, operations, financial condition, results of operations, prospects or 1934 Act reporting status of the Company or any of its Subsidiaries.
j. Absence of Litigation. There is no action, suit, claim, proceeding, inquiry or investigation before or by any court, public board, government agency, self-regulatory organization or body pending or, to the knowledge of the Company or any of its Subsidiaries, threatened against or affecting the Company or any of its Subsidiaries, or their officers or directors in their capacity as such, that could have a Material Adverse Effect. The SEC Documents contain a complete list and summary description of any pending or, to the knowledge of the Company, threatened proceeding against or affecting the Company or any of its Subsidiaries, without regard to whether it would have a Material Adverse Effect. The Company and its Subsidiaries are unaware of any facts or circumstances which might give rise to any of the foregoing.
k. Intellectual Property. The Company and each of its Subsidiaries owns or possesses the requisite licenses or rights to use all patents, patent applications, patent rights, inventions, know-how, trade secrets, trademarks, trademark applications, service marks, service names, trade names and copyrights (“Intellectual Property”) necessary to enable it to conduct its business as now operated (and, as presently contemplated to be operated in the future); there is no claim or action by any person pertaining to, or proceeding pending, or to the Company’s knowledge threatened, which challenges the right of the Company or of a Subsidiary with respect to any Intellectual Property necessary to enable it to conduct its business as now operated (and, as presently contemplated to be operated in the future); to the best of the Company’s knowledge, the Company’s or its Subsidiaries’ current and intended products, services and processes do not infringe on any Intellectual Property or other rights held by any person; and the Company is unaware of any facts or circumstances which might give rise to any of the foregoing. The Company and each of its Subsidiaries have taken reasonable security measures to protect the secrecy, confidentiality and value of their Intellectual Property.
l. No Materially Adverse Contracts, Etc. Neither the Company nor any of its Subsidiaries is subject to any charter, corporate or other legal restriction, or any judgment, decree, order, rule or regulation which in the judgment of the Company’s officers has or is expected in the future to have a Material Adverse Effect. Neither the Company nor any of its Subsidiaries is a party to any contract or agreement which in the judgment of the Company’s officers has or is expected to have a Material Adverse Effect.
m. Tax Status. The Company and each of its Subsidiaries has made or filed all federal, state and foreign income and all other tax returns, reports and declarations required by any jurisdiction to which it is subject (unless and only to the extent that the Company and each of its Subsidiaries has set aside on its books provisions reasonably adequate for the payment of all unpaid and unreported taxes) and has paid all taxes and other governmental assessments and charges that are material in amount, shown or determined to be due on such returns, reports and declarations, except those being contested in good faith and has set aside on its books provisions reasonably adequate for the payment of all taxes for periods subsequent to the periods to which such returns, reports or declarations apply. There are no unpaid taxes in any material amount claimed to be due by the taxing authority of any jurisdiction, and the officers of the Company know of no basis for any such claim. The Company has not executed a waiver with respect to the statute of limitations relating to the assessment or collection of any foreign, federal, state or local tax. None of the Company’s tax returns is presently being audited by any taxing authority.
n. Transactions with Affiliates. Except for arm’s length transactions pursuant to which the Company or any of its Subsidiaries makes payments in the ordinary course of business upon terms no less favorable than the Company or any of its Subsidiaries could obtain from third parties and other than the grant of stock options described in the SEC Documents, none of the officers, directors, or employees of the Company is presently a party to any transaction with the Company or any of its Subsidiaries (other than for services as employees, officers and directors), including any contract, agreement or other arrangement providing for the furnishing of services to or by, providing for rental of real or personal property to or from, or otherwise requiring payments to or from any officer, director or such employee or, to the knowledge of the Company, any corporation, partnership, trust or other entity in which any officer, director, or any such employee has a substantial interest or is an officer, director, trustee or partner.
o. Disclosure. All information relating to or concerning the Company or any of its Subsidiaries set forth in this Agreement and provided to the Buyer pursuant to Section 2(d) hereof and otherwise in connection with the transactions contemplated hereby is true and correct in all material respects and the Company has not omitted to state any material fact necessary in order to make the statements made herein or therein, in light of the circumstances under which they were made, not misleading. No event or circumstance has occurred or exists with respect to the Company or any of its Subsidiaries or its or their business, properties, prospects, operations or financial conditions, which, under applicable law, rule or regulation, requires public disclosure or announcement by the Company but which has not been so publicly announced or disclosed (assuming for this purpose that the Company’s reports filed under the 1934 Act are being incorporated into an effective registration statement filed by the Company under the 1933 Act).
p. Acknowledgment Regarding Buyer’s Purchase of Securities. The Company acknowledges and agrees that the Buyer is acting solely in the capacity of arm’s length purchaser with respect to this Agreement and the transactions contemplated hereby. The Company further acknowledges that the Buyer is not acting as a financial advisor or fiduciary of the Company (or in any similar capacity) with respect to this Agreement and the transactions contemplated hereby and any statement made by the Buyer or any of its respective representatives or agents in connection with this Agreement and the transactions contemplated hereby is not advice or a recommendation and is merely incidental to the Buyer’s purchase of the Securities. The Company further represents to the Buyer that the Company’s decision to enter into this Agreement has been based solely on the independent evaluation of the Company and its representatives.
q. No Integrated Offering. Neither the Company, nor any of its affiliates, nor any person acting on its or their behalf, has directly or indirectly made any offers or sales in any security or solicited any offers to buy any security under circumstances that would require registration under the 1933 Act of the issuance of the Securities to the Buyer. The issuance of the Securities to the Buyer will not be integrated with any other issuance of the Company’s securities (past, current or future) for purposes of any shareholder approval provisions applicable to the Company or its securities.
r. No Brokers; No Solicitation. Except with respect to the Placement Agent, the Company has taken no action which would give rise to any claim by any person for brokerage commissions, transaction fees or similar payments relating to this Agreement or the transactions contemplated hereby. The Company represents and warrants that neither the Buyer nor its employee(s), member(s), beneficial owner(s), or partner(s) solicited the Company to enter into this Agreement and consummate the transactions described in this Agreement. The Company represents and warrants that neither the Buyer nor its employee(s), member(s), beneficial owner(s), or partner(s) is required to be registered as a broker-dealer under the Securities Exchange Act of 1934 in order to (i) enter into or consummate the transactions encompassed by this Agreement, the Note, and the related transaction documents entered into in connection herewith (the “Transaction Documents”), (ii) fulfill the Buyer’s obligations under the Transaction Documents, or (iii) exercise any of the Buyer’s rights under the Transaction Documents (including but not limited to the sale of the Securities).
s. Permits; Compliance. The Company and each of its Subsidiaries is in possession of all franchises, grants, authorizations, licenses, permits, easements, variances, exemptions, consents, certificates, approvals and orders necessary to own, lease and operate its properties and to carry on its business as it is now being conducted (collectively, the “Company Permits”), and there is no action pending or, to the knowledge of the Company, threatened regarding suspension or cancellation of any of the Company Permits. Neither the Company nor any of its Subsidiaries is in conflict with, or in default or violation of, any of the Company Permits, except for any such conflicts, defaults or violations which, individually or in the aggregate, would not reasonably be expected to have a Material Adverse Effect. Since September 30, 2025, neither the Company nor any of its Subsidiaries has received any notification with respect to possible conflicts, defaults or violations of applicable laws, except for notices relating to possible conflicts, defaults or violations, which conflicts, defaults or violations would not have a Material Adverse Effect.
t. Environmental Matters.
(i) There are, to the Company’s knowledge, with respect to the Company or any of its Subsidiaries or any predecessor of the Company, no past or present violations of Environmental Laws (as defined below), releases of any material into the environment, actions, activities, circumstances, conditions, events, incidents, or contractual obligations which may give rise to any common law environmental liability or any liability under the Comprehensive Environmental Response, Compensation and Liability Act of 1980 or similar federal, state, local or foreign laws and neither the Company nor any of its Subsidiaries has received any notice with respect to any of the foregoing, nor is any action pending or, to the Company’s knowledge, threatened in connection with any of the foregoing. The term “Environmental Laws” means all federal, state, local or foreign laws relating to pollution or protection of human health or the environment (including, without limitation, ambient air, surface water, groundwater, land surface or subsurface strata), including, without limitation, laws relating to emissions, discharges, releases or threatened releases of chemicals, pollutants contaminants, or toxic or hazardous substances or wastes (collectively, “Hazardous Materials”) into the environment, or otherwise relating to the manufacture, processing, distribution, use, treatment, storage, disposal, transport or handling of Hazardous Materials, as well as all authorizations, codes, decrees, demands or demand letters, injunctions, judgments, licenses, notices or notice letters, orders, permits, plans or regulations issued, entered, promulgated or approved thereunder.
(ii) Other than those that are or were stored, used or disposed of in compliance with applicable law, no Hazardous Materials are contained on or about any real property currently owned, leased or used by the Company or any of its Subsidiaries, and no Hazardous Materials were released on or about any real property previously owned, leased or used by the Company or any of its Subsidiaries during the period the property was owned, leased or used by the Company or any of its Subsidiaries, except in the normal course of the Company’s or any of its Subsidiaries’ business.
(iii) There are no underground storage tanks on or under any real property owned, leased or used by the Company or any of its Subsidiaries that are not in compliance with applicable law.
u. Title to Property. The Company and its Subsidiaries have good and marketable title in fee simple to all real property and good and marketable title to all personal property owned by them which is material to the business of the Company and its Subsidiaries, in each case free and clear of all liens, encumbrances and defects except such as would not have a Material Adverse Effect. Any real property and facilities held under lease by the Company and its Subsidiaries are held by them under valid, subsisting and enforceable leases with such exceptions as would not have a Material Adverse Effect.
v. Insurance. The Company and each of its Subsidiaries are insured by insurers of recognized financial responsibility against such losses and risks and in such amounts as management of the Company believes to be prudent and customary in the businesses in which the Company and its Subsidiaries are engaged. Neither the Company nor any such Subsidiary has any reason to believe that it will not be able to renew its existing insurance coverage as and when such coverage expires or to obtain similar coverage from similar insurers as may be necessary to continue its business at a cost that would not have a Material Adverse Effect. Upon written request the Company will provide to the Buyer true and correct copies of all policies relating to directors’ and officers’ liability coverage, errors and omissions coverage, and commercial general liability coverage.
w. Internal Accounting Controls. The Company and each of its Subsidiaries maintain a system of internal accounting controls sufficient, in the judgment of the Company’s board of directors, to provide reasonable assurance that (i) transactions are executed in accordance with management’s general or specific authorizations, (ii) transactions are recorded as necessary to permit preparation of financial statements in conformity with generally accepted accounting principles and to maintain asset accountability, (iii) access to assets is permitted only in accordance with management’s general or specific authorization and (iv) the recorded accountability for assets is compared with the existing assets at reasonable intervals and appropriate action is taken with respect to any differences.
x. Foreign Corrupt Practices. Neither the Company, nor any of its Subsidiaries, nor any director, officer, agent, employee or other person acting on behalf of the Company or any Subsidiary has, in the course of his actions for, or on behalf of, the Company, used any corporate funds for any unlawful contribution, gift, entertainment or other unlawful expenses relating to political activity; made any direct or indirect unlawful payment to any foreign or domestic government official or employee from corporate funds; violated or is in violation of any provision of the U.S. Foreign Corrupt Practices Act of 1977, as amended, or made any bribe, rebate, payoff, influence payment, kickback or other unlawful payment to any foreign or domestic government official or employee.
y. Solvency. The Company (after giving effect to the transactions contemplated by this Agreement) is solvent (i.e., its assets have a fair market value in excess of the amount required to pay its probable liabilities on its existing debts as they become absolute and matured) and currently the Company has no information that would lead it to reasonably conclude that the Company would not, after giving effect to the transaction contemplated by this Agreement, have the ability to, nor does it intend to take any action that would impair its ability to, pay its debts from time to time incurred in connection therewith as such debts mature. The Company’s financial statements for its most recent fiscal year end and interim financial statements have been prepared assuming the Company will continue as a going concern, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business.
z. No Investment Company. The Company is not, and upon the issuance and sale of the Securities as contemplated by this Agreement will not be an “investment company” required to be registered under the Investment Company Act of 1940 (an “Investment Company”). The Company is not controlled by an Investment Company.
aa. No Off Balance Sheet Arrangements. There is no transaction, arrangement, or other relationship between the Company or any of its Subsidiaries and an unconsolidated or other off balance sheet entity that is required to be disclosed by the Company in its 1934 Act filings and is not so disclosed or that otherwise could be reasonably likely to have a Material Adverse Effect.
bb. No Disqualification Events. None of the Company, any of its predecessors, any affiliated issuer, any director, executive officer, other officer of the Company participating in the offering hereunder, any beneficial owner of 20% or more of the Company’s outstanding voting equity securities, calculated on the basis of voting power, nor any promoter (as that term is defined in Rule 405 under the 1933 Act) connected with the Company in any capacity at the time of sale (each, an “Issuer Covered Person”) is subject to any of the “Bad Actor” disqualifications described in Rule 506(d)(1)(i) to (viii) under the 1933 Act (a “Disqualification Event”), except for a Disqualification Event covered by Rule 506(d)(2) or (d)(3). The Company has exercised reasonable care to determine whether any Issuer Covered Person is subject to a Disqualification Event.
cc. Manipulation of Price. The Company has not, and to its knowledge no one acting on its behalf has: (i) taken, directly or indirectly, any action designed to cause or to result, or that could reasonably be expected to cause or result, in the stabilization or manipulation of the price of any security of the Company to facilitate the sale or resale of any of the Securities, (ii) sold, bid for, purchased, or paid any compensation for soliciting purchases of, any of the Securities, or (iii) paid or agreed to pay to any person any compensation for soliciting another to purchase any other securities of the Company.
dd. Bank Holding Company Act. Neither the Company nor any of its Subsidiaries is subject to the Bank Holding Company Act of 1956, as amended (the “BHCA”) and to regulation by the Board of Governors of the Federal Reserve System (the “Federal Reserve”). Neither the Company nor any of its Subsidiaries or affiliates owns or controls, directly or indirectly, five percent (5%) or more of the outstanding shares of any class of voting securities or twenty-five percent (25%) or more of the total equity of a bank or any entity that is subject to the BHCA and to regulation by the Federal Reserve. Neither the Company nor any of its Subsidiaries or affiliates exercises a controlling influence over the management or policies of a bank or any entity that is subject to the BHCA and to regulation by the Federal Reserve.
ee. Illegal or Unauthorized Payments; Political Contributions. Neither the Company nor any of its Subsidiaries nor, to the Company’s knowledge, any of the officers, directors, employees, agents or other representatives of the Company or any of its Subsidiaries or any other business entity or enterprise with which the Company or any Subsidiary is or has been affiliated or associated, has, directly or indirectly, made or authorized any payment, contribution or gift of money, property, or services, whether or not in contravention of applicable law, (i) as a kickback or bribe to any person or (ii) to any political organization, or the holder of or any aspirant to any elective or appointive public office except for personal political contributions not involving the direct or indirect use of funds of the Company or any of its Subsidiaries.
ff. Breach of Representations and Warranties by the Company. The Company agrees that if the Company breaches any of the representations or warranties set forth in this Section 3 and in addition to any other remedies available to the Buyer pursuant to this Agreement, it will be considered an Event of Default under Section 3.4 of the Note.
4. ADDITIONAL COVENANTS, AGREEMENTS AND ACKNOWLEDGEMENTS.
a. Best Efforts. The parties shall use their best efforts to satisfy timely each of the conditions described in Section 6 and 7 of this Agreement.
b. Form D; Blue Sky Laws. The Company agrees to file a Form D with respect to the Securities if required under Regulation D and to provide a copy thereof to the Buyer promptly after such filing. The Company shall, on or before the Closing Date, take such action as the Company shall reasonably determine is necessary to qualify the Securities for sale to the Buyer at the applicable closing pursuant to this Agreement under applicable securities or “blue sky” laws of the states of the United States (or to obtain an exemption from such qualification), and shall provide evidence of any such action so taken to the Buyer on or prior to the Closing Date.
c. Use of Proceeds. The Company shall use the Purchase Price for business development and general working capital, and not in violation or contravention of any applicable law, rule or regulation.
d. [Intentionally Omitted].
e. Usury. To the extent it may lawfully do so, the Company hereby agrees not to insist upon or plead or in any manner whatsoever claim, and will resist any and all efforts to be compelled to take the benefit or advantage of, usury laws wherever enacted, now or at any time hereafter in force, in connection with any action or proceeding that may be brought by the Buyer in order to enforce any right or remedy under this Agreement, the Note and any document, agreement or instrument contemplated thereby. Notwithstanding any provision to the contrary contained in this Agreement, the Note and any document, agreement or instrument contemplated thereby, it is expressly agreed and provided that the total liability of the Company under this Agreement, the Note or any document, agreement or instrument contemplated thereby for payments which under applicable law are in the nature of interest shall not exceed the maximum lawful rate authorized under applicable law (the “Maximum Rate”), and, without limiting the foregoing, in no event shall any rate of interest or default interest, or both of them, when aggregated with any other sums which under applicable law in the nature of interest that the Company may be obligated to pay under this Agreement, the Note and any document, agreement or instrument contemplated thereby exceed such Maximum Rate. It is agreed that if the maximum contract rate of interest allowed by law applicable to this Agreement, the Note and any document, agreement or instrument contemplated thereby is increased or decreased by statute or any official governmental action subsequent to the date hereof, the new maximum contract rate of interest allowed by law will be the Maximum Rate applicable to this Agreement, the Note and any document, agreement or instrument contemplated thereby from the effective date thereof forward, unless such application is precluded by applicable law. If under any circumstances whatsoever, interest in excess of the Maximum Rate is paid by the Company to the Buyer with respect to indebtedness evidenced by this Agreement, the Note and any document, agreement or instrument contemplated thereby, such excess shall be applied by the Buyer to the unpaid principal balance of any such indebtedness or be refunded to the Company, the manner of handling such excess to be at the Buyer’s election.
f. Restriction on Activities. Commencing as of the date first above written, and until the earlier of payment of the Note in full or full conversion of the Note, the Company shall not, directly or indirectly, without the Buyer’s prior written consent, which consent shall not be unreasonably withheld, sell, divest, acquire, or dispose of all or substantially all of its assets other than in the ordinary course of business.
g. Listing. The Company will, so long as the Buyer owns any of the Securities, maintain the listing and trading of its Common Stock on the Principal Market or any equivalent replacement exchange or electronic quotation system (including but not limited to the Pink Sheets electronic quotation system) and will comply in all respects with the Company’s reporting, filing and other obligations under the bylaws or rules of the Financial Industry Regulatory Authority (“FINRA”) and such exchanges, as applicable. The Company shall promptly provide to the Buyer copies of any notices it receives from the Principal Market and any other exchanges or electronic quotation systems on which the Common Stock is then traded regarding the continued eligibility of the Common Stock for listing on such exchanges and quotation systems if requested in writing by the Buyer.
h. Corporate Existence. The Company will, so long as the Buyer beneficially owns any of the Securities, maintain its corporate existence and shall not sell all or substantially all of the Company’s assets, except in the event of a Fundamental Transaction (as defined in the Note), where the surviving or successor entity in such transaction (i) assumes the Company’s obligations under the Transaction Documents and (ii) is a publicly traded corporation whose Common Stock is listed for trading or quotation on the Principal Market, any tier of the NASDAQ Stock Market, the New York Stock Exchange or the NYSE American.
i. No Integration. The Company shall not make any offers or sales of any security (other than the Securities) under circumstances that would require registration of the Securities being offered or sold hereunder under the 1933 Act or cause the offering of the Securities to be integrated with any other offering of securities by the Company for the purpose of any stockholder approval provision applicable to the Company or its securities.
j. Compliance with 1934 Act; Public Information Failures. For so long as the Buyer beneficially owns the Note or any Conversion Shares, the Company shall comply with the reporting requirements of the 1934 Act; and the Company shall continue to be subject to the reporting requirements of the 1934 Act.
k. [Intentionally Omitted].
l. Legal Counsel Opinions. Upon the request of the Buyer from to time to time, the Company shall be responsible (at its cost) for promptly supplying to the Company’s transfer agent and the Buyer a customary legal opinion letter of its counsel (the “Legal Counsel Opinion”) to the effect that the resale of the Conversion Shares by the Buyer or its affiliates, successors and assigns is exempt from the registration requirements of the 1933 Act pursuant to Rule 144 (provided the requirements of Rule 144 are satisfied and provided the Conversion Shares are not then registered under the 1933 Act for resale pursuant to an effective registration statement) or other applicable exemption (provided the requirements of such other applicable exemption are satisfied). In addition, the Buyer may (at the Company’s cost) at any time secure its own legal counsel to issue the Legal Counsel Opinion, and the Company will instruct its transfer agent to accept such opinion.
m. [Intentionally Omitted].
n. Non-Public Information. The Company covenants and agrees that neither it, nor any other person acting on its behalf will provide the Buyer or its agents or counsel with any information that constitutes, or the Company reasonably believes constitutes, material non-public information, unless prior thereto the Buyer shall have consented to the receipt of such information and agreed with the Company to keep such information confidential. The Company understands and confirms that the Buyer shall be relying on the foregoing covenant in effecting transactions in securities of the Company. To the extent that the Company delivers any material, non-public information to the Buyer without such Buyer’s consent, the Company hereby covenants and agrees that such Buyer shall not have any duty of confidentiality to the Company, any of its Subsidiaries, or any of their respective officers, directors, agents, employees or affiliates, not to trade on the basis of, such material, non- public information, provided that the Buyer shall remain subject to applicable law. To the extent that any notice provided, information provided, or any other communications made by the Company, to the Buyer, constitutes or contains material non-public information regarding the Company or any Subsidiaries, the Company shall simultaneously file such notice or other material information with the SEC pursuant to a Current Report on Form 8-K. In addition to any other remedies provided by this Agreement or the related transaction documents, if the Company provides any material non-public information to the Buyer without their prior written consent, and it fails to immediately (no later than that business day) file a Form 8-K disclosing this material non-public information, it shall pay the Buyer as partial liquidated damages and not as a penalty a sum equal to $3,000 per day beginning with the day the information is disclosed to the Buyer and ending and including the day the Form 8-K disclosing this information is filed.
o. No Broker-Dealer Acknowledgement. Absent a final adjudication from a court of competent jurisdiction stating otherwise, the Company shall not to any person, institution, governmental or other entity, state, claim, allege, or in any way assert, that Buyer is currently, or ever has been, a broker-dealer under the Securities Exchange Act of 1934.
p. Breach of Covenants. The Company acknowledges and agrees that if the Company breaches any of the covenants set forth in this Section 4, in addition to any other remedies available to the Buyer pursuant to this Agreement, it will be considered an Event of Default under Section 3.3 of the Note.
5. Transfer Agent Instructions. The Company shall issue irrevocable instructions to the Company’s transfer agent to issue certificates and/or issue shares electronically at the Buyer’s option, registered in the name of the Buyer or its nominee, upon conversion of the Note, the Conversion Shares, in such amounts as specified from time to time by the Buyer to the Company in accordance with the terms thereof (the “Irrevocable Transfer Agent Instructions”). In the event that the Company proposes to replace its transfer agent, the Company shall provide, prior to the effective date of such replacement, a fully executed Irrevocable Transfer Agent Instructions in a form as initially delivered pursuant to this Agreement (including but not limited to the provision to irrevocably reserved shares of Common Stock in the Reserved Amount (as defined in the Note)) signed by the successor transfer agent to the Company and the Company. Prior to registration of the Conversion Shares under the 1933 Act or the date on which the Conversion Shares may be sold pursuant to Rule 144, Rule 144A, Regulation S, or other applicable exemption without any restriction as to the number of Securities as of a particular date that can then be immediately sold, all such certificates or book entry shares shall bear the restrictive legend specified in Section 2(g) of this Agreement. The Company warrants that: (i) no instruction other than the Irrevocable Transfer Agent Instructions referred to in this Section 5 will be given by the Company to its transfer agent and that the Securities shall otherwise be freely transferable on the books and records of the Company as and to the extent provided in this Agreement and the Note; (ii) it will not direct its transfer agent not to transfer or delay, impair, and/or hinder its transfer agent in transferring (or issuing)(electronically or in certificated form) any certificate for Securities to be issued to the Buyer upon conversion of or otherwise pursuant to the Note as and when required by the Note and this Agreement; (iii) it will not fail to remove (or directs its transfer agent not to remove or impairs, delays, and/or hinders its transfer agent from removing) any restrictive legend (or to withdraw any stop transfer instructions in respect thereof) on any certificate for any Securities issued to the Buyer upon conversion of or otherwise pursuant to the Note as and when required by the Note and/or this Agreement and (iv) it will provide any required corporate resolutions and issuance approvals to its transfer agent within 6 hours of each conversion of the Note. Nothing in this Section shall affect in any way the Buyer’s obligations and agreement set forth in Section 2(g) hereof to comply with all applicable prospectus delivery requirements, if any, upon re-sale of the Securities. If the Buyer provides the Company, at the cost of the Company, with (i) an opinion of counsel in form, substance and scope customary for opinions in comparable transactions, to the effect that a public sale or transfer of such Securities may be made without registration under the 1933 Act and such sale or transfer is effected or (ii) the Buyer provides reasonable assurances that the Securities can be sold pursuant to 144, Rule 144A, Regulation S, or other applicable exemption, the Company shall permit the transfer, and, in the case of the Securities, promptly instruct its transfer agent to issue one or more certificates, free from restrictive legend, in such name and in such denominations as specified by the Buyer. The Company acknowledges that a breach by it of its obligations hereunder will cause irreparable harm to the Buyer, by vitiating the intent and purpose of the transactions contemplated hereby. Accordingly, the Company acknowledges that the remedy at law for a breach of its obligations under this Section 5 may be inadequate and agrees, in the event of a breach or threatened breach by the Company of the provisions of this Section, that the Buyer shall be entitled, in addition to all other available remedies, to an injunction restraining any breach and requiring immediate transfer, without the necessity of showing economic loss and without any bond or other security being required.
6. Conditions to the Company’s Obligation to Sell. The obligation of the Company hereunder to issue and sell the Note to the Buyer at the Closing is subject to the satisfaction, at or before the Closing Date, of each of the following conditions thereto, provided that these conditions are for the Company’s sole benefit and may be waived by the Company at any time in its sole discretion:
a. The Buyer shall have executed the Transaction Documents and delivered the same to the Company.
b. The Buyer shall have delivered the Purchase Price in accordance with Section 1(b) above.
c. The representations and warranties of the Buyer shall be true and correct in all material respects as of the date when made and as of the Closing Date, as though made at that time (except for representations and warranties that speak as of a specific date), and the Buyer shall have performed, satisfied and complied in all material respects with the covenants, agreements and conditions required by this Agreement to be performed, satisfied or complied with by the Buyer at or prior to the Closing Date.
d. No litigation, statute, rule, regulation, executive order, decree, ruling or injunction shall have been enacted, entered, promulgated or endorsed by or in any court or governmental authority of competent jurisdiction or any self-regulatory organization having authority over the matters contemplated hereby which prohibits the consummation of any of the transactions contemplated by this Agreement.
e. The Buyer shall have delivered to the Company all information reasonably necessary and requested by the Company to comply with applicable federal and state securities laws in connection with the Agreement, which shall include, but not be limited to, the Buyer’s principal place of residence or domicile for tax and securities law purposes and sufficient information to verify the Buyer’s status as an accredited investor.
7. Conditions to The Buyer’s Obligation to Purchase. The obligation of the Buyer hereunder to purchase the Note, on the Closing Date, is subject to the satisfaction, at or before the Closing Date, of each of the following conditions, provided that these conditions are for the Buyer’s sole benefit and may be waived by the Buyer at any time in its sole discretion:
a. The Company and Subsidiaries, as applicable, shall have executed the Transaction Documents and delivered the same to the Buyer.
b. The Company shall have delivered to the Buyer the duly executed Note in such denominations as the Buyer shall request and in accordance with Section 1(b) above.
c. The Company shall have delivered to the Buyer the Commitment Shares.
d. The Irrevocable Transfer Agent Instructions, in form and substance satisfactory to the Buyer, shall have been delivered to and acknowledged in writing by the Company’s Transfer Agent.
e. The representations and warranties of the Company shall be true and correct in all material respects as of the date when made and as of Closing Date, as though made at such time (except for representations and warranties that speak as of a specific date) and the Company shall have performed, satisfied and complied in all material respects with the covenants, agreements and conditions required by this Agreement to be performed, satisfied or complied with by the Company at or prior to the Closing Date.
f. No litigation, statute, rule, regulation, executive order, decree, ruling or injunction shall have been enacted, entered, promulgated or endorsed by or in any court or governmental authority of competent jurisdiction or any self-regulatory organization having authority over the matters contemplated hereby which prohibits the consummation of any of the transactions contemplated by this Agreement.
g. No event shall have occurred which could reasonably be expected to have a Material Adverse Effect on the Company including but not limited to a change in the 1934 Act reporting status of the Company or the failure of the Company to be timely in its 1934 Act reporting obligations.
h. Trading in the Common Stock on the Principal Market shall not have been suspended by the SEC, FINRA or the Principal Market.
i. The Company shall have delivered to the Buyer (i) a certificate evidencing the formation and good standing of the Company in such entity’s jurisdiction of formation issued by the Secretary of State (or comparable office) of such jurisdiction, as of a date within ten (10) days of the Closing Date and (ii) resolutions adopted by the Company’s Board of Directors at a duly called meeting or by unanimous written consent authorizing this Agreement and all other documents, instruments and transactions contemplated hereby.
8. Governing Law; Miscellaneous.
a. Arbitration of Claims; Governing Law; Venue. The Company and Buyer shall submit all Claims (as defined in Exhibit B of this Purchase Agreement) (the “Claims”) arising under this Agreement or any other agreement between the Company and Buyer or their respective affiliates (including but not limited to the Transaction Documents) or any Claim relating to the relationship of the Company and Buyer or their respective affiliates to binding arbitration pursuant to the arbitration provisions set forth in Exhibit B of the Purchase Agreement (the “Arbitration Provisions”). The Company and Buyer hereby acknowledge and agree that the Arbitration Provisions are unconditionally binding on the Company and Buyer hereto and are severable from all other provisions of this Agreement. By executing this Agreement, Company represents, warrants and covenants that Company has reviewed the Arbitration Provisions carefully, consulted with legal counsel about such provisions (or waived its right to do so), understands that the Arbitration Provisions are intended to allow for the expeditious and efficient resolution of any dispute hereunder, agrees to the terms and limitations set forth in the Arbitration Provisions, and that Company will not take a position contrary to the foregoing representations. Company acknowledges and agrees that Buyer may rely upon the foregoing representations and covenants of Company regarding the Arbitration Provisions. This Agreement shall be construed and enforced in accordance with, and all questions concerning the construction, validity, interpretation and performance of this Agreement shall be governed by, the internal laws of the State of Delaware, without giving effect to any choice of law or conflict of law provision or rule (whether of the State of Delaware or any other jurisdictions) that would cause the application of the laws of any jurisdictions other than the State of Delaware. The Company and Buyer consent to and expressly agree that the exclusive venue for arbitration of any Claims arising under this Agreement or any other agreement between the Company and Buyer or their respective affiliates (including but not limited to the Transaction Documents) or any Claim relating to the relationship of the Company and Buyer or their respective affiliates shall be in the State of Delaware. Without modifying the Company’s and Buyer’s mandatory obligations to resolve disputes hereunder pursuant to the Arbitration Provisions, for any litigation arising in connection with any of the Transaction Documents (and notwithstanding the terms (specifically including any governing law and venue terms) of any transfer agent services agreement or other agreement between the Company’s transfer agent and the Company, such litigation specifically includes, without limitation any action between or involving Company and the Company’s transfer agent under the Irrevocable Transfer Agent Instructions or otherwise related to Buyer in any way (specifically including, without limitation, any action where Company seeks to obtain an injunction, temporary restraining order, or otherwise prohibit the Company’s transfer agent from issuing shares of Common Stock to Buyer for any reason)), each party hereto hereby (i) consents to and expressly submits to the exclusive personal jurisdiction of any state or federal court sitting in the State of Delaware, (ii) expressly submits to the exclusive venue of any such court for the purposes hereof, (iii) agrees to not bring any such action (specifically including, without limitation, any action where Company seeks to obtain an injunction, temporary restraining order, or otherwise prohibit the Company’s transfer agent from issuing shares of Common Stock to Buyer for any reason) outside of any state or federal court sitting in the State of Delaware, and (iv) waives any claim of improper venue and any claim or objection that such courts are an inconvenient forum or any other claim, defense or objection to the bringing of any such proceeding in such jurisdiction or to any claim that such venue of the suit, action or proceeding is improper. Notwithstanding anything in the foregoing to the contrary, nothing herein shall limit, or shall be deemed or construed to limit, the ability of the Buyer to realize on any collateral or any other security, or to enforce a judgment or other court ruling in favor of the Buyer, including through a legal action in any court of competent jurisdiction. The Company hereby irrevocably waives, and agrees not to assert in any suit, action or proceeding, any objection to jurisdiction and venue of any action instituted hereunder, any claim that it is not personally subject to the jurisdiction of any such court, and any claim that such suit, action or proceeding is brought in an inconvenient forum or that the venue of such suit, action or proceeding is improper (including but not limited to based upon forum non conveniens). THE COMPANY HEREBY IRREVOCABLY WAIVES ANY RIGHT IT MAY HAVE, AND AGREES NOT TO REQUEST, A JURY TRIAL FOR THE ADJUDICATION OF ANY DISPUTE HEREUNDER OR IN CONNECTION WITH OR ARISING OUT OF THIS AGREEMENT OR ANY TRANSACTIONS CONTEMPLATED HEREBY. The Company irrevocably waives personal service of process and consents to process being served in any suit, action or proceeding in connection with this Agreement or any other agreement, certificate, instrument or document contemplated hereby or thereby by mailing a copy thereof via registered or certified mail or overnight delivery (with evidence of delivery) to Company at the address in effect for notices to it under this Agreement and agrees that such service shall constitute good and sufficient service of process and notice thereof. Nothing contained herein shall be deemed to limit in any way any right to serve process in any other manner permitted by law. The prevailing party in any action or dispute brought in connection with this Agreement or any other agreement, certificate, instrument or document contemplated hereby or thereby shall be entitled to recover from the other party its reasonable attorney’s fees and costs. If any provision of this Agreement shall be invalid or unenforceable in any jurisdiction, such invalidity or unenforceability shall not affect the validity or enforceability of the remainder of this Agreement in that jurisdiction or the validity or enforceability of any provision of this Agreement in any other jurisdiction.
b. Counterparts. This Agreement may be executed in one or more counterparts, each of which shall be deemed an original but all of which shall constitute one and the same agreement and shall become effective when counterparts have been signed by each party and delivered to the other party. A facsimile or .pdf signature shall be considered due execution and shall be binding upon the signatory thereto with the same force and effect as if the signature were an original, not a facsimile or .pdf signature. Delivery of a counterpart signature hereto by facsimile or email/.pdf transmission shall be deemed validly delivery thereof.
c. Construction; Headings. This Agreement shall be deemed to be jointly drafted by the Company and the Buyer and shall not be construed against any person as the drafter hereof. The headings of this Agreement are for convenience of reference only and shall not form part of, or affect the interpretation of, this Agreement.
d. Severability. In the event that any provision of this Agreement, the Note, or any other agreement or instrument delivered in connection herewith is invalid or unenforceable under any applicable statute or rule of law, then such provision shall be deemed inoperative to the extent that it may conflict therewith and shall be deemed modified to conform with such statute or rule of law. Any such provision which may prove invalid or unenforceable under any law shall not affect the validity or enforceability of any other provision of this Agreement, the Note, or any other agreement, certificate, instrument or document contemplated hereby or thereby.
e. Entire Agreement; Amendments. This Agreement, the Note, and the instruments referenced herein contain the entire understanding of the parties with respect to the matters covered herein and therein and, except as specifically set forth herein or therein, neither the Company nor the Buyer makes any representation, warranty, covenant or undertaking with respect to such matters. No provision of this Agreement or any agreement or instrument contemplated hereby may be waived or amended other than by an instrument in writing signed by the Buyer.
f. Notices. All notices, demands, requests, consents, approvals, and other communications required or permitted hereunder shall be in writing and, unless otherwise specified herein, shall be (i) personally served, (ii) deposited in the mail, registered or certified, return receipt requested, postage prepaid, (iii) delivered by reputable air courier service with charges prepaid, or (iv) transmitted by hand delivery, telegram, e-mail or facsimile, addressed as set forth below or to such other address as such party shall have specified most recently by written notice. Any notice or other communication required or permitted to be given hereunder shall be deemed effective (a) upon hand delivery or delivery by e-mail or facsimile, with accurate confirmation generated by the transmitting facsimile machine, at the address or number designated below (if delivered on a business day during normal business hours where such notice is to be received), or the first business day following such delivery (if delivered other than on a business day during normal business hours where such notice is to be received) or (b) on the second business day following the date of mailing by express courier service, fully prepaid, addressed to such address, or upon actual receipt of such mailing, whichever shall first occur. The addresses for such communications shall be:
If to the Company, to:
NewGenIvf Group Limited
36/39-36/40, 13th Floor, PS Tower
Sukhumvit 21 Road (Asoke)
Khlong Toei Nuea Sub-district
Watthana District, Bangkok 10110
Attention: Wing Fung Alfred Siu
e-mail: Alfred.Siu@newgenivf.com
If to the Buyer:
FIRSTFIRE GLOBAL OPPORTUNITIES FUND, LLC
1040 First Avenue, Suite 190
New York, NY 10022
e-mail: eli@firstfirecapital.com
g. Successors and Assigns. This Agreement shall be binding upon and inure to the benefit of the parties and their successors and assigns. The Company shall not assign this Agreement or any rights or obligations hereunder without the prior written consent of the Buyer. The Buyer may assign its rights hereunder to any “accredited investor” (as defined in Rule 501(a) of the 1933 Act) in a private transaction from the Buyer or to any of its “affiliates,” as that term is defined under the 1934 Act, without the consent of the Company but shall provide prior notice to the Company of such assignment.
h. Third Party Beneficiaries. This Agreement is intended for the benefit of the parties hereto and their respective permitted successors and assigns, and is not for the benefit of, nor may any provision hereof be enforced by, any other person.
i. Survival. The representations and warranties of the Company and the agreements and covenants set forth in this Agreement shall survive the closing hereunder notwithstanding any due diligence investigation conducted by or on behalf of the Buyer. The Company agrees to indemnify and hold harmless the Buyer and all their officers, directors, employees and agents for loss or damage arising as a result of or related to any breach or alleged breach by the Company of any of its representations, warranties and covenants set forth in this Agreement or any of its covenants and obligations under this Agreement, including advancement of expenses as they are incurred.
j. Publicity. The Company, and the Buyer shall have the right to review a reasonable period of time before issuance of any press releases, SEC, Principal Market or FINRA filings, or any other public statements with respect to the transactions contemplated hereby; provided, however, that the Company shall be entitled, without the prior approval of the Buyer, to make any press release or SEC, Principal Market (or other applicable trading market) or FINRA filings with respect to such transactions as is required by applicable law and regulations (although the Buyer shall be consulted by the Company in connection with any such press release prior to its release and shall be provided with a copy thereof and be given an opportunity to comment thereon).
k. Further Assurances. Each party shall do and perform, or cause to be done and performed, all such further acts and things, and shall execute and deliver all such other agreements, certificates, instruments and documents, as the other party may reasonably request in order to carry out the intent and accomplish the purposes of this Agreement and the consummation of the transactions contemplated hereby.
l. No Strict Construction. The language used in this Agreement will be deemed to be the language chosen by the parties to express their mutual intent, and no rules of strict construction will be applied against any party.
m. Indemnification. In consideration of the Buyer’s execution and delivery of this Agreement and acquiring the Securities hereunder, and in addition to all of the Company’s other obligations under this Agreement or the Note, the Company shall defend, protect, indemnify and hold harmless the Buyer and its stockholders, partners, members, officers, directors, employees and direct or indirect investors and any of the foregoing persons’ agents or other representatives (including, without limitation, those retained in connection with the transactions contemplated by this Agreement) (collectively, the “Indemnitees”) from and against any and all actions, causes of action, suits, claims, losses, costs, penalties, fees, liabilities and damages, and expenses in connection therewith (irrespective of whether any such Indemnitee is a party to the action for which indemnification hereunder is sought), and including reasonable attorneys’ fees and disbursements (the “Indemnified Liabilities”), incurred by any Indemnitee as a result of, or arising out of, or relating to (a) any misrepresentation or breach of any representation or warranty made by the Company in this Agreement, the Note or any other agreement, certificate, instrument or document contemplated hereby or thereby, (b) any breach of any covenant, agreement or obligation of the Company contained in this Agreement, the Note or any other agreement, certificate, instrument or document contemplated hereby or thereby or (c) any cause of action, suit or claim brought or made against such Indemnitee by a third party (including for these purposes a derivative action brought on behalf of the Company) and arising out of or resulting from (i) the execution, delivery, performance or enforcement of this Agreement, the Note or any other agreement, certificate, instrument or document contemplated hereby or thereby, (ii) any transaction financed or to be financed in whole or in part, directly or indirectly, with the proceeds of the issuance of the Securities, or (iii) the status of the Buyer or holder of the Securities as an investor in the Company pursuant to the transactions contemplated by this Agreement. To the extent that the foregoing undertaking by the Company may be unenforceable for any reason, the Company shall make the maximum contribution to the payment and satisfaction of each of the Indemnified Liabilities that is permissible under applicable law.
n. Remedies. The Company acknowledges that a breach by it of its obligations hereunder will cause irreparable harm to the Buyer by vitiating the intent and purpose of the transaction contemplated hereby. Accordingly, the Company acknowledges that the remedy at law for a breach of its obligations under this Agreement, the Note, or any other agreement, certificate, instrument or document contemplated hereby or thereby will be inadequate and agrees, in the event of a breach or threatened breach by the Company of the provisions of this Agreement, the Note, or any other agreement, certificate, instrument or document contemplated hereby or thereby, that the Buyer shall be entitled, in addition to all other available remedies at law or in equity, and in addition to the penalties assessable herein, to an injunction or injunctions restraining, preventing or curing any breach of this Agreement, the Note, or any other agreement, certificate, instrument or document contemplated hereby or thereby, and to enforce specifically the terms and provisions hereof and thereof, without the necessity of showing economic loss and without any bond or other security being required.
o. Payment Set Aside. To the extent that the (i) Company makes a payment or payments to the Buyer hereunder, pursuant to the Note or pursuant to any other agreement, certificate, instrument or document contemplated hereby or thereby, or (ii) the Buyer enforces or exercises its rights hereunder or pursuant to the Note, or pursuant to any other agreement, certificate, instrument or document contemplated hereby or thereby, and such payment or payments or the proceeds of such enforcement or exercise or any part thereof (including but not limited to the sale of the Securities) are for any reason (i) subsequently invalidated, declared to be fraudulent or preferential, set aside, recovered from, or disgorged by the Buyer, or (ii) are required to be refunded, repaid or otherwise restored to the Company, a trustee, receiver, government entity, or any other person or entity under any law (including, without limitation, any bankruptcy law, foreign, state or federal law, common law or equitable cause of action), then (i) to the extent of any such restoration the obligation or part thereof originally intended to be satisfied shall be revived and continued in full force and effect as if such payment had not been made or such enforcement or setoff had not occurred and (ii) the Company shall immediately pay to the Buyer a dollar amount equal to the amount that was for any reason (i) subsequently invalidated, declared to be fraudulent or preferential, set aside, recovered from, or disgorged by the Buyer, or (ii) required to be refunded, repaid or otherwise restored to the Company, a trustee, receiver, government entity, or any other person or entity under any law (including, without limitation, any bankruptcy law, foreign, state or federal law, common law or equitable cause of action).
p. Failure or Indulgence Not Waiver. No failure or delay on the part of the Buyer in the exercise of any power, right or privilege hereunder shall operate as a waiver thereof, nor shall any single or partial exercise of any such power, right or privilege preclude other or further exercise thereof or of any other right, power or privileges. All rights and remedies of the Buyer existing hereunder are cumulative to, and not exclusive of, any rights or remedies otherwise available.
q. Electronic Signature. This Agreement may be executed and delivered in one or more counterparts (including by facsimile or electronic mail or in .pdf or any other form of electronic delivery (including any electronic signature complying with U.S. federal ESIGN Act of 2000)) and by different parties in separate counterparts, with the same effect as if all parties hereto had signed the same document. All counterparts so executed and delivered shall be construed together and shall constitute one and the same agreement.
[Signature Page Follows]
IN WITNESS WHEREOF, the undersigned Buyer and the Company have caused this Agreement to be duly executed as of the date first above written.
| NEWGENIVF GROUP LIMITED | |||
| By: | /s/ Wing Fung Alfred Siu | ||
| Name: | WING FUNG ALFRED SIU | ||
| Title: | CHIEF EXECUTIVE OFFICER | ||
|
FIRSTFIRE GLOBAL OPPORTUNITIES FUND, LLC |
|||
| By: FirstFire Capital Management LLC, its manager | |||
| By: | /s/ Eli Fireman | ||
| Name: | ELI FIREMAN | ||
EXHIBIT A
FORM OF NOTE
[attached hereto]
EXHIBIT B
ARBITRATION PROVISIONS
1. Dispute Resolution. Each party consents to and expressly agrees that the exclusive venue for arbitration of any dispute arising out of or relating to any of the Transaction Documents or the relationship of the parties or their affiliates shall be in the State of Delaware. For purposes of this Exhibit B, the term “Claims” means any disputes, claims, demands, causes of action, requests for injunctive relief, requests for specific performance, questions regarding severability of any provisions of the Transaction Documents, liabilities, damages, losses, or controversies whatsoever arising from, related to, or connected with the transactions contemplated in the Transaction Documents and any communications between the parties related thereto, including without limitation any claims of mutual mistake, mistake, fraud, misrepresentation, failure of formation, failure of consideration, promissory estoppel, unconscionability, failure of condition precedent, rescission, and any statutory claims, tort claims, contract claims, or claims to void, invalidate or terminate the Agreement (or these Arbitration Provisions (defined below)) or any of the other Transaction Documents. The parties to this Agreement (the “parties”) hereby agree that the Claims may be arbitrated in one or more Arbitrations pursuant to these Arbitration Provisions (one for an injunction or injunctions and a separate one for all other Claims). The parties hereby agree that the arbitration provisions set forth in this Exhibit B (“Arbitration Provisions”) are binding on each of them. As a result, any attempt to rescind the Agreement (or these Arbitration Provisions) or any other Transaction Document) or declare the Agreement (or these Arbitration Provisions) or any other Transaction Document invalid or unenforceable pursuant to Section 29 of the 1934 Act or for any other reason is subject to these Arbitration Provisions. These Arbitration Provisions shall also survive any termination or expiration of the Agreement. Any capitalized term not defined in these Arbitration Provisions shall have the meaning set forth in the Agreement.
2. Arbitration. Except as otherwise provided herein, all Claims must be submitted to arbitration (“Arbitration”) to be conducted exclusively in the State of Delaware and pursuant to the terms set forth in these Arbitration Provisions. Subject to the arbitration appeal right provided for in Paragraph 5 below (the “Appeal Right”), the parties agree that the award of the arbitrator rendered pursuant to Paragraph 4 below (the “Arbitration Award”) shall be (a) final and binding upon the parties, (b) the sole and exclusive remedy between them regarding any Claims, counterclaims, issues, or accountings presented or pleaded to the arbitrator, and (c) promptly payable in United States dollars free of any tax, deduction or offset (with respect to monetary awards). Subject to the Appeal Right, any costs or fees, including without limitation attorneys’ fees, incurred in connection with or incident to enforcing the Arbitration Award shall, to the maximum extent permitted by law, be charged against the party resisting such enforcement. The Arbitration Award shall include Default Interest (as defined or otherwise provided for in the Note, “Default Interest”) (with respect to monetary awards) at the rate specified in the Note for Default Interest both before and after the Arbitration Award. Judgment upon the Arbitration Award will be entered and enforced by any state or federal court sitting in the State of Delaware.
3. The Arbitration Act. The parties hereby incorporate herein the provisions and procedures set forth in the Delaware Uniform Arbitration Act, Title 10 Chapter 57 (as amended or superseded from time to time, the “Arbitration Act”). Notwithstanding the foregoing, pursuant to, and to the maximum extent permitted by, the Arbitration Act, in the event of conflict or variation between the terms of these Arbitration Provisions and the provisions of the Arbitration Act, the terms of these Arbitration Provisions shall control and the parties hereby waive or otherwise agree to vary the effect of all requirements of the Arbitration Act that may conflict with or vary from these Arbitration Provisions.
4. Arbitration Proceedings. Arbitration between the parties will be subject to the following:
4.1 Initiation of Arbitration. The parties agree that a party may initiate Arbitration by giving written notice to the other party (“Arbitration Notice”) in the same manner that notice is permitted under Section 8(f) of the Agreement; provided, however, that the Arbitration Notice may not be given by email or fax. Arbitration will be deemed initiated as of the date that the Arbitration Notice is deemed physically delivered to such other party under Section 8(f) of the Agreement (the “Service Date”). After the Service Date, information may be delivered, and notices may be given, by email or fax pursuant to Section 8(f) of the Agreement or any other method permitted thereunder. The Arbitration Notice must describe the nature of the controversy, the remedies sought, and the election to commence Arbitration proceedings. All Claims in the Arbitration Notice must be pleaded consistent with the Delaware Rules of Civil Procedure.
4.2 Selection and Payment of Arbitrator.
(a) Within ten (10) calendar days after the Service Date, Buyer shall select and submit to Company the names of four (4) arbitrators that are designated as “neutrals” or qualified arbitrators by American Arbitration Association (“AAA”) (https://www.adr.org/) or other arbitration service provider agreed upon by the parties (such four (4) designated persons hereunder are referred to herein as the “Proposed Arbitrators”). Within five (5) calendar days after Buyer has submitted to Company the names of the Proposed Arbitrators, Company must select, by written notice to Buyer, one (1) of the Proposed Arbitrators to act as the arbitrator for the parties under these Arbitration Provisions. If Company fails to select one of the Proposed Arbitrators in writing within such 5-day period, then Buyer may select the arbitrator from the Proposed Arbitrators by providing written notice of such selection to Company. The date that the Proposed Arbitrator selected pursuant to this Paragraph 4.2 agrees in writing (including via email) delivered to both parties to serve as the arbitrator hereunder is referred to herein as the “Arbitration Commencement Date”. If an arbitrator resigns or is unable to act during the Arbitration, a replacement arbitrator shall be chosen by Buyer in accordance with this Paragraph 4.2 to continue the Arbitration. If AAA or other arbitration service provider agreed upon by the parties ceases to exist or to provide a list of neutrals and there is no successor thereto, then the arbitrator shall be selected under the then prevailing rules of the American Arbitration Association.
4.3 Applicability of Certain Delaware Rules. The parties agree that the Arbitration shall be conducted generally in accordance with the Delaware Rules of Civil Procedure and the Delaware Rules of Evidence. More specifically, the Delaware Rules of Civil Procedure shall apply, without limitation, to the filing of any pleadings, motions or memoranda, the conducting of discovery, and the taking of any depositions. The Delaware Rules of Evidence shall apply to any hearings, whether telephonic or in person, held by the arbitrator. Notwithstanding the foregoing, it is the parties’ intent that the incorporation of such rules will in no event supersede these Arbitration Provisions. In the event of any conflict between the Delaware Rules of Civil Procedure or the Delaware Rules of Evidence and these Arbitration Provisions, these Arbitration Provisions shall control.
4.4 Answer and Default. An answer and any counterclaims to the Arbitration Notice shall be required to be delivered to the party initiating the Arbitration within twenty (20) calendar days after the Arbitration Commencement Date. If an answer is not delivered by the required deadline, the arbitrator must provide written notice to the defaulting party stating that the arbitrator will enter a default award against such party if such party does not file an answer within five (5) calendar days of receipt of such notice. If an answer is not filed within the five (5) day extension period, the arbitrator must render a default award, consistent with the relief requested in the Arbitration Notice, against a party that fails to submit an answer within such time period.
4.5 [Intentionally Omitted].
4.6 Discovery. The parties agree that discovery shall be conducted in accordance with the Arbitration Act.
4.6 Dispositive Motions. Each party shall have the right to submit dispositive motions pursuant to the Delaware Rules of Civil Procedure (a “Dispositive Motion”). The party submitting the Dispositive Motion may, but is not required to, deliver to the arbitrator and to the other party a memorandum in support (the “Memorandum in Support”) of the Dispositive Motion. Within seven (7) calendar days of delivery of the Memorandum in Support, the other party shall deliver to the arbitrator and to the other party a memorandum in opposition to the Memorandum in Support (the “Memorandum in Opposition”). Within seven (7) calendar days of delivery of the Memorandum in Opposition, as applicable, the party that submitted the Memorandum in Support shall deliver to the arbitrator and to the other party a reply memorandum to the Memorandum in Opposition (“Reply Memorandum”). If the applicable party shall fail to deliver the Memorandum in Opposition as required above, or if the other party fails to deliver the Reply Memorandum as required above, then the applicable party shall lose its right to so deliver the same, and the Dispositive Motion shall proceed regardless.
4.7 Confidentiality. All information disclosed by either party (or such party’s agents) during the Arbitration process (including without limitation information disclosed during the discovery process or any Appeal (defined below)) shall be considered confidential in nature. Each party agrees not to disclose any confidential information received from the other party (or its agents) during the Arbitration process (including without limitation during the discovery process or any Appeal) unless (a) prior to or after the time of disclosure such information becomes public knowledge or part of the public domain, not as a result of any inaction or action of the receiving party or its agents, (b) such information is required by a court order, subpoena or similar legal duress to be disclosed if such receiving party has notified the other party thereof in writing and given it a reasonable opportunity to obtain a protective order from a court of competent jurisdiction prior to disclosure, or (c) such information is disclosed to the receiving party’s agents, representatives and legal counsel on a need to know basis who each agree in writing not to disclose such information to any third party.
4.8 Authorization; Timing; Scheduling Order. Subject to all other portions of these Arbitration Provisions, the parties hereby authorize and direct the arbitrator to take such actions and make such rulings as may be necessary to carry out the parties’ intent for the Arbitration proceedings to be efficient and expeditious. The parties hereby agree that an Arbitration Award must be made within one hundred twenty (120) calendar days after the Arbitration Commencement Date.
4.9 Relief. The arbitrator shall have the right to award or include in the Arbitration Award (or in a preliminary ruling) any relief which the arbitrator deems proper under the circumstances, including, without limitation, specific performance and injunctive relief, provided that the arbitrator may not award exemplary or punitive damages.
4.10 Fees and Costs. As part of the Arbitration Award, the arbitrator is hereby directed to require the losing party (the party being awarded the least amount of money by the arbitrator, which, for the avoidance of doubt, shall be determined without regard to any statutory fines, penalties, fees, or other charges awarded to any party) to (a) pay the full amount of any unpaid costs and fees of the Arbitration, and (b) reimburse the prevailing party for all reasonable attorneys’ fees, arbitrator costs and fees, deposition costs, other discovery costs, and other expenses, costs or fees paid or otherwise incurred by the prevailing party in connection with the Arbitration.
5. Arbitration Appeal.
5.1 Initiation of Appeal. Following the entry of the Arbitration Award, either party (the “Appellant”) shall have a period of thirty (30) calendar days in which to notify the other party (the “Appellee”), in writing, that the Appellant elects to appeal (the “Appeal”) the Arbitration Award (such notice, an “Appeal Notice”) to a panel of arbitrators in accordance with the Arbitration Act.
5.2 Fees and Costs. As part of the Appeal Panel Award, the Appeal Panel is hereby directed to require the losing party (the party being awarded the least amount of money by the arbitrator, which, for the avoidance of doubt, shall be determined without regard to any statutory fines, penalties, fees, or other charges awarded to any party) to (a) pay the full amount of any unpaid costs and fees of the Arbitration and the Appeal Panel, and (b) reimburse the prevailing party (the party being awarded the most amount of money by the Appeal Panel, which, for the avoidance of doubt, shall be determined without regard to any statutory fines, penalties, fees, or other charges awarded to any part) the reasonable attorneys’ fees, arbitrator and Appeal Panel costs and fees, deposition costs, other discovery costs, and other expenses, costs or fees paid or otherwise incurred by the prevailing party in connection with the Arbitration (including without limitation in connection with the Appeal).
6. Miscellaneous.
6.1 Severability. If any part of these Arbitration Provisions is found to violate or be illegal under applicable law, then such provision shall be modified to the minimum extent necessary to make such provision enforceable under applicable law, and the remainder of the Arbitration Provisions shall remain unaffected and in full force and effect.
6.2 Governing Law. Except as otherwise expressly provided for in these Arbitration Provisions, these Arbitration Provisions shall be governed by the laws of the State of Delaware without regard to the conflict of laws principles therein.
6.3 Interpretation. The headings of these Arbitration Provisions are for convenience of reference only and shall not form part of, or affect the interpretation of, these Arbitration Provisions.
6.4 Waiver. No waiver of any provision of these Arbitration Provisions shall be effective unless it is in the form of a writing signed by the party granting the waiver.
6.5 Time is of the Essence. Time is expressly made of the essence with respect to each and every provision of these Arbitration Provisions.
[Remainder of page intentionally left blank]
Exhibit 10.42
NEITHER THE ISSUANCE AND SALE OF THE SECURITIES REPRESENTED BY THIS CERTIFICATE NOR THE SECURITIES INTO WHICH THESE SECURITIES ARE CONVERTIBLE HAVE BEEN REGISTERED UNDER THE SECURITIES ACT OF 1933, AS AMENDED, OR APPLICABLE STATE SECURITIES LAWS. THE SECURITIES MAY NOT BE OFFERED FOR SALE, SOLD, TRANSFERRED OR ASSIGNED (I) IN THE ABSENCE OF (A) AN EFFECTIVE REGISTRATION STATEMENT FOR THE SECURITIES UNDER THE SECURITIES ACT OF 1933, AS AMENDED, OR (B) AN OPINION OF COUNSEL (WHICH MAY BE THE LEGAL COUNSEL OPINION (AS DEFINED IN THE PURCHASE AGREEMENT)), IN A GENERALLY ACCEPTABLE FORM, THAT REGISTRATION IS NOT REQUIRED UNDER SAID ACT OR (II) UNLESS SOLD PURSUANT TO RULE 144, RULE 144A OR REGULATION S UNDER SAID ACT OR OTHER APPLICABLE EXEMPTION. NOTWITHSTANDING THE FOREGOING, THE SECURITIES MAY BE PLEDGED IN CONNECTION WITH A BONA FIDE MARGIN ACCOUNT OR OTHER LOAN OR FINANCING ARRANGEMENT SECURED BY THE SECURITIES.
| Principal Amount: $220,000.00 | Issue Date: March 25, 2026 |
| Actual Amount of Purchase Price: $200,000.00 |
PROMISSORY NOTE
FOR VALUE RECEIVED, NewGenIvf Group Limited, a British Virgin Islands company (hereinafter called the “Borrower” or the “Company”), hereby promises to pay to the order of FIRSTFIRE GLOBAL OPPORTUNITIES FUND, LLC, a Delaware limited liability company, or registered assigns (the “Holder”), in the form of lawful money of the United States of America, the principal sum of $220,000.00 (the “Principal Amount”) (subject to adjustment herein), which includes the purchase price of $200,000.00 plus an original issue discount in the amount of $20,000.00 (the “OID”), and to pay a one-time interest charge on the Principal Amount hereof at the rate of twelve percent (12%) (the “Interest Rate”) (which is equal to $26,400.00 and shall be guaranteed and earned in full as of the date hereof (the “Issue Date”)), when such amounts become due and payable, whether at maturity or upon acceleration or by prepayment or otherwise, as further provided herein. The maturity date shall be twelve (12) months from the Issue Date (the “Maturity Date”), and is the date upon which the Principal Amount (which includes the OID) and any accrued and unpaid interest and other fees, shall be due and payable.
This Note may not be prepaid or repaid in whole or in part except as otherwise explicitly set forth herein.
Any Principal Amount or interest on this Note which is not paid when due shall bear interest at the rate of the lesser of (i) eighteen percent (18%) per annum and (ii) the maximum amount permitted by law from the due date thereof until the same is paid (“Default Interest”). Interest and Default Interest shall be computed on the basis of a 365-day year and the actual number of days elapsed.
All payments due hereunder (to the extent not converted into Class A ordinary shares of the Borrower, no par value per share (the “Common Stock”) in accordance with the terms hereof) shall be made in lawful money of the United States of America. All payments shall be made at such address as the Holder shall hereafter give to the Borrower by written notice made in accordance with the provisions of this Note. Whenever any amount expressed to be due by the terms of this Note is due on any day which is not a business day, the same shall instead be due on the next succeeding day which is a business day.
Each capitalized term used herein, and not otherwise defined, shall have the meaning ascribed thereto in that certain Securities Purchase Agreement, dated as of the Issue Date, pursuant to which this Note was originally issued (the “Purchase Agreement”). As used in this Note, the term “business day” shall mean any day other than a Saturday, Sunday or a day on which commercial banks in the city of New York, New York are authorized or required by law or executive order to remain closed. As used herein, the term “Trading Day” means any day that shares of Common Stock are listed for trading or quotation on the Principal Market (as defined in the Purchase Agreement), provided, however, that if the Common Stock is not then listed or quoted on any Principal Market, then any calendar day.
This Note is free from all taxes, liens, claims and encumbrances with respect to the issue thereof and shall not be subject to preemptive rights or other similar rights of shareholders of the Borrower and will not impose personal liability upon the holder thereof.
The following terms shall also apply to this Note:
ARTICLE I. CONVERSION RIGHTS
1.1 Conversion Right. The Holder shall have the right, on any calendar day, at any time on or following the date that is one hundred eighty (180) calendar days after the Issue Date, to convert all or any portion of the then outstanding and unpaid Principal Amount and interest (including any Default Interest) into fully paid and non-assessable shares of Common Stock, as such Common Stock exists on the Issue Date, or any shares of capital stock or other securities of the Borrower into which such Common Stock shall hereafter be changed or reclassified, at the Conversion Price (as defined below) determined as provided herein (a “Conversion”), by submitting to the Borrower a Notice of Conversion (as defined in this Note) by facsimile, e-mail or other reasonable means of communication dispatched on the Conversion Date (as defined in this Note) prior to 11:59 p.m., New York, New York time; provided, however, that notwithstanding anything to the contrary contained herein, the Holder shall not have the right to convert any portion of this Note, pursuant to Section 1 or otherwise, to the extent that after giving effect to such issuance after conversion as set forth on the applicable Notice of Conversion, the Holder (together with the Holder’s affiliates (the “Affiliates”), and any other Persons (as defined below) acting as a group together with the Holder or any of the Holder’s Affiliates (such Persons, “Attribution Parties”)), would beneficially own in excess of the Beneficial Ownership Limitation (as defined below). For purposes of the foregoing sentence, the number of shares of Common Stock beneficially owned by the Holder and Attribution Parties shall include the number of shares of Common Stock issuable upon conversion of this Note with respect to which such determination is being made, but shall exclude the number of shares of Common Stock which would be issuable upon (i) conversion of the remaining, nonconverted portion of this Note beneficially owned by the Holder or any of its Affiliates or Attribution Parties and (ii) exercise or conversion of the unexercised or nonconverted portion of any other securities of the Company subject to a limitation on conversion or exercise analogous to the limitation contained herein beneficially owned by the Holder or any of its Affiliates or Attribution Parties. Except as set forth in the preceding sentence, for purposes of this Section 1.1, beneficial ownership shall be calculated in accordance with Section 13(d) of the Securities Exchange Act of 1934, as amended (the “1934 Act”) and the rules and regulations promulgated thereunder, it being acknowledged by the Holder that the Holder is solely responsible for any schedules required to be filed in accordance therewith. In addition, a determination as to any group status as contemplated above shall be determined in accordance with Section 13(d) of the 1934 Act and the rules and regulations promulgated thereunder. For purposes of this Section 1.1, in determining the number of outstanding shares of Common Stock, the Holder may rely on the number of outstanding shares of Common Stock as reflected in (A) the Company’s most recent periodic or annual report filed with the Commission, as the case may be, (B) a more recent public announcement by the Company or (C) a more recent written notice by the Company or the Transfer Agent setting forth the number of shares of Common Stock outstanding. Upon the written or oral request of the Holder, the Company shall within two Trading Days confirm orally and in writing to the Holder the number of shares of Common Stock then outstanding. In any case, the number of outstanding shares of Common Stock shall be determined after giving effect to the conversion or exercise of securities of the Company, including this Note, by the Holder or its Affiliates or Attribution Parties since the date as of which such number of outstanding shares of Common Stock was reported. The “Beneficial Ownership Limitation” shall be 4.99% of the number of shares of the Common Stock outstanding at the time of the respective calculation hereunder, provided, however, that the Holder may from time to time increase or decrease the Beneficial Ownership Limitation to any other percentage not in excess of 9.99% by delivering written notice of such to the Company, with such increase or decrease not effective until the sixty-first (61st) day after delivery of such written notice. “Person” and “Persons” means an individual, a limited liability company, a partnership, a joint venture, a corporation, a trust, an unincorporated organization, any other entity and any governmental entity or any department or agency thereof. The limitations contained in this paragraph shall apply to a successor holder of this Note. The number of Conversion Shares to be issued upon each conversion of this Note shall be determined by dividing the Conversion Amount (as defined below) by the applicable Conversion Price then in effect on the date specified in the notice of conversion, in the form attached hereto as Exhibit A (the “Notice of Conversion”), delivered to the Borrower by the Holder in accordance with the terms of this Note; provided that the Notice of Conversion is submitted by facsimile or e-mail (or by other means resulting in, or reasonably expected to result in, notice) to the Borrower before 11:59 p.m., New York, New York time on such conversion date (the “Conversion Date”). The term “Conversion Amount” means, with respect to any conversion of this Note, the sum of (1) the Principal Amount of this Note to be converted in such conversion plus (2) at the Holder’s option, accrued and unpaid interest, if any, on such Principal Amount at the Interest Rate to the Conversion Date, plus (3) at the Holder’s option, Default Interest, if any, on the amounts referred to in the immediately preceding clauses (1) and/or (2).
1.2 Conversion Price.
(a) Calculation of Conversion Price. The per share conversion price into which Principal Amount and interest (including any Default Interest) under this Note shall be convertible into shares of Common Stock hereunder as further described in this Note (the “Conversion Price”) shall equal the lesser of (i) $1.60 (subject to appropriate adjustment for any stock dividend, stock split, stock combination, rights offerings, reclassification or similar transaction that proportionately decreases or increases the Common Stock as further provided in this Note) or (ii) Market Price (as defined in this Note), subject to adjustment as provided in this Note. “Market Price” shall mean 80% of the lowest closing price of the Common Stock on the Principal Market during the ten (10) Trading Day period immediately preceding the respective Conversion Date, provided, however, that at all times on and after the date that an Event of Default (as defined in this Note) occurs under this Note, Market Price shall instead mean 70% of the lowest traded price of the Common Stock on the Principal Market on any Trading Day during the fifteen (15) Trading Days prior to the respective Conversion Date. If at any time the Conversion Price as determined hereunder for any conversion would be less than the par value of the Common Stock, then at the sole discretion of the Holder, the Conversion Price hereunder may equal such par value for such conversion and the Conversion Amount for such conversion may be increased to include Additional Principal, where “Additional Principal” means such additional amount to be added to the Conversion Amount to the extent necessary to cause the number of conversion shares issuable upon such conversion to equal the same number of conversion shares as would have been issued had the Conversion Price not been adjusted by the Holder to the par value price. Holder shall be entitled to deduct $750.00 from the conversion amount of not less than $50,000 in each Notice of Conversion to cover Holder’s fees associated with each Notice of Conversion. All such Conversion Price determinations are to be appropriately adjusted for any stock dividend, stock split, stock combination, rights offerings, reclassification or similar transaction that proportionately decreases or increases the Common Stock. If the Company, at any time while this Note is outstanding: (i) pays a stock dividend or otherwise makes a distribution or distributions payable in shares of Common Stock on shares of Common Stock or any Common Stock Equivalents, (ii) subdivides outstanding shares of Common Stock into a larger number of shares, (iii) combines (including by way of a reverse stock split) outstanding shares of Common Stock into a smaller number of shares or (iv) issues, in the event of a reclassification of shares of the Common Stock, any shares of capital stock of the Company, then the Conversion Price shall be multiplied by a fraction of which the numerator shall be the number of shares of Common Stock (excluding any treasury shares of the Company) outstanding immediately before such event, and of which the denominator shall be the number of shares of Common Stock outstanding immediately after such event. Any adjustment made pursuant to the immediately preceding sentence shall become effective immediately after the record date for the determination of shareholders entitled to receive such dividend or distribution and shall become effective immediately after the effective date in the case of a subdivision, combination or re-classification. “Common Stock Equivalents” means any securities of the Company or the Company’s Subsidiaries (as defined in the Purchase Agreement) which would entitle the holder thereof to acquire at any time Common Stock, including, without limitation, any debt, preferred stock, right, option, warrant or other instrument that is at any time convertible into or exercisable or exchangeable for, or otherwise entitles the holder thereof to receive, Common Stock.
(b) Voluntary Adjustment By Company. Subject to the rules and regulations of the Principal Market, the Company may at any time while this Note is outstanding, with the prior written consent of the Holder, reduce the then applicable Conversion Price to any amount and for any period of time deemed appropriate by the Board of Directors of the Company. For the avoidance of doubt, the Holder shall not be required to effectuate such conversion in the event of any reduction in Conversion Price by the Company.
1.3 Authorized and Reserved Shares. The Borrower shall use commercially reasonable endeavors to ensure that at all times beginning on the Issue Date and continuing until the Note is extinguished in the entirety, the Borrower will reserve from its authorized and unissued Common Stock a sufficient number of shares, free from preemptive rights, to provide for the issuance of a number of Conversion Shares equal to the greater of: (a) 2,000,000 shares of Common Stock or (b) the sum of (i) the number of Conversion Shares issuable upon the full conversion of this Note at a conversion price equal to the Conversion Price (assuming no payment of Principal Amount or interest) multiplied by (ii) four (4) (the “Reserved Amount”). For the avoidance of doubt, the Reserved Amount shall be required to be reserved by the Borrower pursuant to the immediately preceding sentence even if the Note is not yet convertible into Common Stock by the Holder under Section 1.1 of this Note. The Borrower represents that upon issuance, the Conversion Shares will be duly and validly issued, fully paid, as applicable, and non-assessable. The Borrower (i) acknowledges that it has irrevocably instructed its transfer agent to issue certificates for the Conversion Shares or instructions to have the Conversion Shares issued as contemplated by Section 1.4(e) hereof, and (ii) agrees that its issuance of this Note shall constitute full authority to its officers and agents who are charged with the duty of executing stock certificates or cause the Company to electronically issue shares of Common Stock to execute and issue the necessary certificates for the Conversion Shares or cause the Conversion Shares to be issued as contemplated by Section 1.4(e) hereof in accordance with the terms and conditions of this Note.
1.4 Method of Conversion.
(a) Surrender of Note Upon Conversion. Notwithstanding anything to the contrary set forth herein, upon conversion of this Note in accordance with the terms hereof, the Holder shall not be required to physically surrender this Note to the Borrower unless the entire unpaid Principal Amount is so converted. The Holder and the Borrower shall maintain records showing the Principal Amount so converted and the dates of such conversions or shall use such other method, reasonably satisfactory to the Holder and the Borrower, so as not to require physical surrender of this Note upon each such conversion. In the event of any dispute or discrepancy, such records of the Holder shall, prima facie, be controlling and determinative in the absence of manifest error.
(b) Payment of Taxes. The Borrower shall not be required to pay any tax which may be payable in respect of any transfer involved in the issue and delivery of shares of Common Stock or other securities or property on conversion of this Note in a name other than that of the Holder (or in street name), and the Borrower shall not be required to issue or deliver any such shares or other securities or property unless and until the person or persons (other than the Holder or the custodian in whose street name such shares are to be held for the Holder’s account) requesting the issuance thereof shall have paid to the Borrower the amount of any such tax or shall have established to the satisfaction of the Borrower that such tax has been paid.
(c) Delivery of Common Stock Upon Conversion. Upon receipt by the Borrower from the Holder of an e-mail (or other reasonable means of communication) of a Notice of Conversion meeting the requirements for conversion as provided in this Section 1.4, the Borrower shall issue and deliver or cause to be issued and delivered to or upon the order of the Holder certificates for the Conversion Shares (or cause the electronic delivery of the Conversion Shares as contemplated by Section 1.4(e) hereof) within one (1) Trading Day after such receipt (the “Deadline”) (and, solely in the case of conversion of the entire unpaid Principal Amount and interest (including any Default Interest) under this Note, surrender of this Note). If the Company shall fail for any reason or for no reason to issue to the Holder on or prior to the Deadline a certificate for the number of Conversion Shares or to which the Holder is entitled hereunder and register such Conversion Shares on the Company’s share register or to credit the Holder’s balance account with DTC (as defined below) for such number of Conversion Shares to which the Holder is entitled upon the Holder’s conversion of this Note (a “Conversion Failure”), then, in addition to all other remedies available to the Holder, (i) the Company shall pay in cash to the Holder on each day after the Deadline and during such Conversion Failure an amount equal to 2.0% of the product of (A) the sum of the number of Conversion Shares not issued to the Holder on or prior to the Deadline and to which the Holder is entitled and (B) the closing sale price of the Common Stock on the Trading Day immediately preceding the last possible date which the Company could have issued such Conversion Shares to the Holder without violating this Section 1.4(c); and (ii) the Holder, upon written notice to the Company, may void all or any portion of such Notice of Conversion; provided that the voiding of all or any portion of a Notice of Conversion shall not affect the Company’s obligations to make any payments which have accrued prior to the date of such notice. In addition to the foregoing, if on or prior to the Deadline the Company shall fail to issue and deliver a certificate to the Holder and register such Conversion Shares on the Company’s share register or credit the Holder’s balance account with DTC for the number of Conversion Shares to which the Holder is entitled upon the Holder’s exercise hereunder or pursuant to the Company’s obligation pursuant to clause (ii) below, and if on or after such Trading Day the Holder purchases (in an open market transaction or otherwise) shares of Common Stock to deliver in satisfaction of a sale by the Holder of shares of Common Stock issuable upon such exercise that the Holder anticipated receiving from the Company, then the Company shall, within two (2) Trading Days after the Holder’s request and in the Holder’s discretion, either (i) pay cash to the Holder in an amount equal to the Holder’s total purchase price (including brokerage commissions and other reasonable and customary out-of-pocket expenses, if any) for the shares of Common Stock so purchased (the “Buy-In Price”), at which point the Company’s obligation to deliver such certificate (and to issue such Conversion Shares) or credit such Holder’s balance account with DTC for such Conversion Shares shall terminate, or (ii) promptly honor its obligation to deliver to the Holder a certificate or certificates representing such Conversion Shares or credit such Holder’s balance account with DTC and pay cash to the Holder in an amount equal to the excess (if any) of the Buy-In Price over the product of (A) such number of shares of Common Stock, times (B) the closing sales price of the Common Stock on the date of exercise. Nothing shall limit the Holder’s right to pursue any other remedies available to it hereunder, at law or in equity, including, without limitation, a decree of specific performance and/or injunctive relief with respect to the Company’s failure to timely deliver certificates representing the Conversion Shares (or to electronically deliver such Conversion Shares) upon the conversion of this Note as required pursuant to the terms hereof.
(d) Obligation of Borrower to Deliver Common Stock. At the time that the Holder submits the Notice of Conversion to the Borrower, the Holder shall be deemed to be the holder of record of the Conversion Shares issuable upon such conversion, the outstanding Principal Amount and the amount of accrued and unpaid interest (including any Default Interest) under this Note shall be reduced to reflect such conversion, and, unless the Borrower defaults on its obligations under this Article I, all rights with respect to the portion of this Note being so converted shall forthwith terminate except the right to receive the Common Stock or other securities, cash or other assets, as herein provided, on such conversion. If the Holder shall have given a Notice of Conversion as provided herein, the Borrower’s obligation to issue and deliver the certificates for the Conversion Shares (or cause the electronic delivery of the Conversion Shares as contemplated by Section 1.4(e) hereof) shall be absolute and unconditional, irrespective of the absence of any action by the Holder to enforce the same, any waiver or consent with respect to any provision thereof, the recovery of any judgment against any person or any action to enforce the same, any failure or delay in the enforcement of any other obligation of the Borrower to the holder of record, or any setoff, counterclaim, recoupment, limitation or termination, or any breach or alleged breach by the Holder of any obligation to the Borrower, and irrespective of any other circumstance which might otherwise limit such obligation of the Borrower to the Holder in connection with such conversion. The Conversion Date specified in the Notice of Conversion shall be the Conversion Date so long as the Notice of Conversion is sent to the Borrower before 11:59 p.m., New York, New York time, on such date.
(e) Delivery of Conversion Shares by Electronic Transfer. In lieu of delivering physical certificates representing the Conversion Shares issuable upon conversion hereof, provided the Borrower is participating in the Depository Trust Company (“DTC”) Fast Automated Securities Transfer or Deposit/Withdrawal at Custodian programs, upon request of the Holder and its compliance with the provisions contained in Section 1.1 and in this Section 1.4, the Borrower shall use its best efforts to cause its transfer agent to electronically transmit the Conversion Shares issuable upon conversion hereof to the Holder by crediting the account of Holder’s Prime Broker with DTC through its Deposit Withdrawal Agent Commission system.
1.5 Concerning the Shares. The Conversion Shares issuable upon conversion of this Note may not be sold or transferred unless (i) such shares are sold pursuant to an effective registration statement under the 1933 Act or (ii) the Borrower or its transfer agent shall have been furnished with an opinion of counsel (which opinion shall be the Legal Counsel Opinion (as defined in the Purchase Agreement)) to the effect that the shares to be sold or transferred may be sold or transferred pursuant to an exemption from such registration or (iii) such shares are sold or transferred pursuant to Rule 144, Rule 144A, Regulation S, or other applicable exemption, or (iv) such shares are transferred to an “affiliate” (as defined in Rule 144) of the Borrower who agrees to sell or otherwise transfer the shares only in accordance with this Section 1.5 and who is an Accredited Investor (as defined in the Purchase Agreement). Except as otherwise provided in the Purchase Agreement (and subject to the removal provisions set forth below), until such time as the Conversion Shares have been registered under the 1933 Act or otherwise may be sold pursuant to Rule 144, Rule 144A, Regulation S, or other applicable exemption without any restriction as to the number of securities as of a particular date that can then be immediately sold, each certificate for the Conversion Shares that has not been so included in an effective registration statement or that has not been sold pursuant to an effective registration statement or an exemption that permits removal of the legend, shall bear a legend substantially in the following form, as appropriate:
“NEITHER THE ISSUANCE AND SALE OF THE SECURITIES REPRESENTED BY THIS CERTIFICATE NOR THE SECURITIES INTO WHICH THESE SECURITIES ARE EXERCISABLE HAVE BEEN REGISTERED UNDER THE SECURITIES ACT OF 1933, AS AMENDED, OR APPLICABLE STATE SECURITIES LAWS. THE SECURITIES MAY NOT BE OFFERED FOR SALE, SOLD, TRANSFERRED OR ASSIGNED (I) IN THE ABSENCE OF (A) AN EFFECTIVE REGISTRATION STATEMENT FOR THE SECURITIES UNDER THE SECURITIES ACT OF 1933, AS AMENDED, OR (B) AN OPINION OF COUNSEL (WHICH MAY BE THE LEGAL COUNSEL OPINION (AS DEFINED IN THE PURCHASE AGREEMENT)), IN A GENERALLY ACCEPTABLE FORM, THAT REGISTRATION IS NOT REQUIRED UNDER SAID ACT OR (II) UNLESS SOLD PURSUANT TO RULE 144, RULE 144A, REGULATION S UNDER SAID ACT, OR OTHER APPLICABLE EXEMPTION. NOTWITHSTANDING THE FOREGOING, THE SECURITIES MAY BE PLEDGED IN CONNECTION WITH A BONA FIDE MARGIN ACCOUNT OR OTHER LOAN OR FINANCING ARRANGEMENT SECURED BY THE SECURITIES.”
The legend set forth above shall be removed and the Company shall issue to the Holder a certificate for the applicable Conversion Shares without such legend upon which it is stamped or (as requested by the Holder) issue the applicable Conversion Shares by electronic delivery by crediting the account of such holder’s broker with DTC, if, unless otherwise required by applicable state securities laws: (a) such Conversion Shares are registered for sale under an effective registration statement filed under the 1933 Act or otherwise may be sold pursuant to Rule 144, Rule 144A, Regulation S, or other applicable exemption without any restriction as to the number of securities as of a particular date that can then be immediately sold, or (b) the Company or the Holder provides the Legal Counsel Opinion (as contemplated by and in accordance with Section 4(m) of the Purchase Agreement) to the effect that a public sale or transfer of such Conversion Shares may be made without registration under the 1933 Act, which opinion shall be accepted by the Company so that the sale or transfer is effected. The Company shall be responsible for the fees of its transfer agent and all DTC fees associated with any such issuance. The Holder agrees to sell all Conversion Shares, including those represented by a certificate(s) from which the legend has been removed, in compliance with applicable prospectus delivery requirements, if any. In the event that the Company does not accept the opinion of counsel provided by the Holder with respect to the transfer of Conversion Shares pursuant to an exemption from registration, such as Rule 144, Rule 144A, Regulation S, or other applicable exemption, at the Deadline, notwithstanding that the conditions of Rule 144, Rule 144A, Regulation S, or other applicable exemption, as applicable, have been met, it will be considered an Event of Default under this Note.
1.6 Effect of Certain Events.
(a) Effect of Merger, Consolidation, Etc. At the option of the Holder, the sale, conveyance or disposition of all or substantially all of the assets of the Borrower, or the consolidation, merger or other business combination of the Borrower with or into any other Person (as defined below) or Persons when the Borrower is not the survivor shall be treated pursuant to Section 1.6(b) hereof. “Person” shall mean any individual, corporation, limited liability company, partnership, association, trust or other entity or organization.
(b) Adjustment Due to Merger, Consolidation, Etc. If, at any time when this Note is issued and outstanding and prior to conversion of all of this Note, there shall be any merger, consolidation, exchange of shares, recapitalization, reorganization, or other similar event, as a result of which shares of Common Stock of the Borrower shall be changed into the same or a different number of shares of another class or classes of stock or securities of the Borrower or another entity, or in case of any sale or conveyance of all or substantially all of the assets of the Borrower other than in connection with a plan of complete liquidation of the Borrower (each a “Fundamental Transaction”), then the Holder of this Note shall thereafter have the right to receive upon conversion of this Note, upon the basis and upon the terms and conditions specified herein and in lieu of the shares of Common Stock immediately theretofore issuable upon conversion, such stock, securities or assets which the Holder would have been entitled to receive in such transaction had this Note been converted in full immediately prior to such transaction (without regard to any limitations on conversion set forth herein), and in any such case appropriate provisions shall be made with respect to the rights and interests of the Holder of this Note to the end that the provisions hereof (including, without limitation, provisions for adjustment of the Conversion Price and of the number of shares issuable upon conversion of the Note) shall thereafter be applicable, as nearly as may be practicable in relation to any securities or assets thereafter deliverable upon the conversion hereof.
(c) Adjustment Due to Distribution. If the Borrower shall declare or make any distribution of its assets (or rights to acquire its assets) to holders of Common Stock as a dividend, stock repurchase, by way of return of capital or otherwise (including any dividend or distribution to the Borrower’s shareholders in cash or shares (or rights to acquire shares) of capital stock of a subsidiary (i.e., a spin-off)) (a “Distribution”), then the Holder of this Note shall be entitled, upon any conversion of this Note after the date of record for determining shareholders entitled to such Distribution, to receive the amount of such assets which would have been payable to the Holder with respect to the shares of Common Stock issuable upon such conversion had such Holder been the holder of such shares of Common Stock on the record date for the determination of shareholders entitled to such Distribution.
(d) Purchase Rights. If, at any time when all or any portion of this Note is issued and outstanding, the Borrower issues any convertible securities or rights to purchase stock, warrants, securities or other property (the “Purchase Rights”) pro rata to the record holders of any class of Common Stock, then the Holder of this Note will be entitled to acquire, upon the terms applicable to such Purchase Rights, the aggregate Purchase Rights which such Holder could have acquired if such Holder had held the number of shares of Common Stock acquirable upon complete conversion of this Note (without regard to any limitations on conversion contained herein) immediately before the date on which a record is taken for the grant, issuance or sale of such Purchase Rights or, if no such record is taken, the date as of which the record holders of Common Stock are to be determined for the grant, issue or sale of such Purchase Rights.
(e) [Intentionally Omitted].
(f) Notice of Adjustments. Upon the occurrence of each adjustment or readjustment of the Conversion Price as a result of the events described in Section 1.6 of this Note, the Borrower shall, at its expense and within one (1) calendar day after the occurrence of each respective adjustment or readjustment of the Conversion Price, compute such adjustment or readjustment and prepare and furnish to the Holder a certificate setting forth (i) the Conversion Price in effect at such time, (ii) the number of shares of Common Stock and the amount, if any, of other securities or property which at the time would be received upon conversion of the Note, (iii) the detailed facts upon which such adjustment or readjustment is based, and (iv) copies of the documentation (including but not limited to relevant transaction documents) that evidences the adjustment or readjustment. In addition, the Borrower shall, within one (1) calendar day after each written request from the Holder, furnish to such Holder a like certificate setting forth (i) the Conversion Price in effect at such time, (ii) the number of shares of Common Stock and the amount, if any, of other securities or property which at the time would be received upon conversion of the Note, (iii) the detailed facts upon which such adjustment or readjustment is based, and (iv) copies of the documentation (including but not limited to relevant transaction documents) that evidences the adjustment or readjustment. For the avoidance of doubt, each adjustment or readjustment of the Conversion Price as a result of the events described in Section 1.6 of this Note shall occur without any action by the Holder and regardless of whether the Borrower complied with the notification provisions in Section 1.6 of this Note.
1.7 Status as Shareholder. Upon submission of a Notice of Conversion by the Holder, (i) the Conversion Shares covered thereby shall be deemed converted into shares of Common Stock and (ii) the Holder’s rights as the Holder of such converted portion of this Note shall cease and terminate, excepting only the right to receive certificates for such shares of Common Stock and to any remedies provided herein or otherwise available at law or in equity to such Holder because of a failure by the Borrower to comply with the terms of this Note. Notwithstanding the foregoing, if the Holder has not received certificates for all shares of Common Stock prior to the tenth (10th) business day after the expiration of the Deadline with respect to a conversion of any portion of this Note for any reason, then (unless the Holder otherwise elects to retain its status as a holder of Common Stock by so notifying the Borrower) the Holder shall regain the rights of a Holder of this Note with respect to such unconverted portions of this Note and the Borrower shall, as soon as practicable, return such unconverted Note to the Holder or, if the Note has not been surrendered, adjust its records to reflect that such portion of this Note has not been converted. In all cases, the Holder shall retain all of its rights and remedies for the Borrower’s failure to convert this Note.
1.8 Prepayment. At any time prior to the date that an Event of Default occurs under this Note, the Borrower shall have the right, exercisable on three (3) Trading Days prior written notice to the Holder of the Note, to prepay the outstanding Principal Amount and interest then due under this Note in accordance with this Section 1.8. Any notice of prepayment hereunder (an “Optional Prepayment Notice”) shall be delivered to the Holder of the Note at its registered addresses and shall state: (1) that the Borrower is exercising its right to prepay the Note, and (2) the date of prepayment which shall be three (3) Trading Days from the date of the Optional Prepayment Notice (the “Optional Prepayment Date”). The Holder shall have the right, during the period beginning on the date of Holder’s receipt of the Optional Prepayment Notice and until the Holder’s actual receipt of the full prepayment amount on the Optional Prepayment Date, to instead convert all or any portion of the Note pursuant to the terms of this Note, including the amount of this Note to be prepaid by the Borrower in accordance with this Section 1.8, even if the Note is not yet convertible into Common Stock by the Holder under Section 1.1 of this Note. On the Optional Prepayment Date, the Borrower shall make payment of the amounts designated below to or upon the order of the Holder as specified by the Holder in writing to the Borrower. If the Borrower exercises its right to prepay the Note in accordance with this Section 1.8, the Borrower shall make payment to the Holder of an amount in cash equal to the sum of: (w) the prepayment percentage set forth in the table immediately following this paragraph for the applicable prepayment period set forth in the table immediately following this paragraph (“Prepayment Percentage”) multiplied by the Principal Amount then outstanding plus (x) the Prepayment Percentage multiplied by the accrued and unpaid interest on the Principal Amount to the Optional Prepayment Date.
Prepayment Period |
Prepayment Percentage |
| 1. At any time prior to the date that an Event of Default occurs under this Note. | 110% |
ARTICLE II. RANKING AND CERTAIN COVENANTS
2.1 Ranking. This Note shall be an unsecured obligation of the Borrower.
2.2 Distributions on Capital Stock. So long as the Borrower shall have any obligation under this Note, the Borrower shall not without the Holder’s written consent (a) pay, declare or set apart for such payment, any dividend or other distribution (whether in cash, property or other securities) on shares of capital stock other than dividends on shares of Common Stock solely in the form of additional shares of Common Stock or (b) directly or indirectly or through any subsidiary make any other payment or distribution in respect of its capital stock except for distributions pursuant to any shareholders’ rights plan which is approved by a majority of the Borrower’s disinterested directors.
ARTICLE III. EVENTS OF DEFAULT
It shall be considered an event of default if any of the following events listed in this Article III (each, an “Event of Default”) shall occur on or after the Issue Date, subject, in each case (except Section 3.2 under this Note), to a five (5) business day cure period:
3.1 Failure to Pay Principal or Interest. The Borrower fails to pay the Principal Amount hereof or interest thereon when due on this Note, whether at maturity, upon acceleration or otherwise, or fails to fully comply with Section 1.10 of this Note.
3.2 Conversion and the Shares. The Borrower (i) fails to issue Conversion Shares to the Holder (or announces or threatens in writing that it will not honor its obligation to do so) upon exercise by the Holder of the conversion rights of the Holder in accordance with the terms of this Note, (ii) fails to transfer or cause its transfer agent to transfer (issue) (electronically or in certificated form) any certificate for the Conversion Shares issuable to the Holder upon conversion of or otherwise pursuant to this Note as and when required by this Note, (iii) fails to reserve the Reserved Amount at all times, (iv) the Borrower directs its transfer agent not to transfer or delays, impairs, and/or hinders its transfer agent in transferring (or issuing) (electronically or in certificated form) any certificate for the Conversion Shares issuable to the Holder upon conversion of or otherwise pursuant to this Note as and when required by this Note, or fails to remove (or directs its transfer agent not to remove or impairs, delays, and/or hinders its transfer agent from removing) any restrictive legend (or to withdraw any stop transfer instructions in respect thereof) on any certificate for any Conversion Shares issued to the Holder upon conversion of or otherwise pursuant to this Note as and when required by this Note (or makes any written announcement, statement or threat that it does not intend to honor the obligations described in this paragraph) and any such failure shall continue uncured (or any written announcement, statement or threat not to honor its obligations shall not be rescinded in writing) for two (2) Trading Days after the Holder shall have delivered a Notice of Conversion, and/or (v) fails to remain current in its obligations to its transfer agent (including but not limited to payment obligations to its transfer agent). It shall be an Event of Default of this Note, if a conversion of this Note is delayed, hindered or frustrated due to a balance owed by the Borrower to its transfer agent. If at the option of the Holder, the Holder advances any funds to the Borrower’s transfer agent in order to process a conversion, such advanced funds shall be added to the principal balance of the Note.
3.3 Breach of Agreements and Covenants. The Borrower breaches any covenant, agreement, or other term or condition contained in the Purchase Agreement, this Note, Irrevocable Transfer Agent Instructions, or in any agreement, statement or certificate given in writing pursuant hereto or in connection herewith or therewith.
3.4 Breach of Representations and Warranties. Any representation or warranty of the Borrower made in the Purchase Agreement, this Note, Irrevocable Transfer Agent Instructions, or in any agreement, statement or certificate given in writing pursuant hereto or in connection herewith or therewith shall be false or misleading in any material respect when made.
3.5 Receiver or Trustee. The Borrower or any subsidiary of the Borrower shall make an assignment for the benefit of creditors, or apply for or consent to the appointment of a receiver or trustee for it or for a substantial part of its property or business, or such a receiver or trustee shall otherwise be appointed.
3.6 Judgments. Any money judgment, writ or similar process shall be entered or filed against the Borrower or any subsidiary of the Borrower or any of its property or other assets for more than $100,000, and shall remain unvacated, unbonded or unstayed for a period of twenty (20) days unless otherwise consented to by the Holder, which consent will not be unreasonably withheld.
3.7 Bankruptcy. Bankruptcy, insolvency, reorganization or liquidation proceedings or other proceedings, voluntary or involuntary, for relief under any bankruptcy law or any law for the relief of debtors shall be instituted by or against the Borrower or any subsidiary of the Borrower.
3.8 Failure to Comply with the 1934 Act. At any time after the Issue Date, the Borrower shall fail to comply with the reporting requirements of the 1934 Act and/or the Borrower shall cease to be subject to the reporting requirements of the 1934 Act.
3.9 Liquidation. Any dissolution, liquidation, or winding up of Borrower or any substantial portion of its business.
3.10 Cessation of Operations. Any cessation of operations by Borrower or Borrower admits it is otherwise generally unable to pay its debts as such debts become due, provided, however, that any disclosure of the Borrower’s ability to continue as a “going concern” shall not be an admission that the Borrower cannot pay its debts as they become due.
3.11 Maintenance of Assets. The failure by Borrower to maintain any material intellectual property rights, personal, real property or other assets which are necessary to conduct its business (whether now or in the future).
3.12 Replacement of Transfer Agent. In the event that the Borrower proposes to replace its transfer agent, the Borrower fails to provide, prior to the effective date of such replacement, a fully executed Irrevocable Transfer Agent Instructions in a form as initially delivered pursuant to the Purchase Agreement (including but not limited to the provision to irrevocably reserve shares of Common Stock in the Reserved Amount) signed by the successor transfer agent to Borrower and the Borrower.
3.13 Inside Information. Any actual transmittal, conveyance, or disclosure by the Borrower or its officers, directors, and/or affiliates of, material non-public information concerning the Borrower, to the Holder or its successors and assigns, which is not cured by Borrower’s filing of a Form 6-K pursuant to Regulation FD.
3.14 Unavailability of Rule 144. If, at any time on or after the date that is six (6) calendar months after the Issue Date, the Holder is unable to (i) obtain a standard “144 legal opinion letter” from an attorney reasonably acceptable to the Borrower’s transfer agent in order to facilitate the Holder’s conversion of any portion of the Note into free trading shares of the Borrower’s Common Stock pursuant to Rule 144, and/or (ii) thereupon deposit such shares into the Holder’s brokerage account.
3.15 Delisting, Suspension, or Quotation of Trading of Common Stock. If, at any time on or after the Issue Date, the Borrower’s Common Stock (i) is suspended from trading, (ii) halted from trading, and/or (iii) fails to be listed or quoted on a Principal Market.
3.16 Rights and Remedies Upon an Event of Default. Upon the occurrence of any Event of Default specified in this Article III, this Note shall become immediately due and payable, and the Borrower shall pay to the Holder, in full satisfaction of its obligations hereunder, an amount equal to the Principal Amount then outstanding plus accrued interest (including any Default Interest) through the date of full repayment multiplied by 150% (collectively the “Default Amount”), as well as all costs, including, without limitation, legal fees and expenses, of collection, all without demand, presentment or notice, all of which hereby are expressly waived by the Borrower. Holder may, in Holder’s sole discretion, convert all or any portion of this Note (including the Default Amount) into Common Stock pursuant to the terms of this Note (for the avoidance of doubt, this shall apply even if such conversion occurs after the Maturity Date). The Holder shall be entitled to exercise all other rights and remedies available at law or in equity.
ARTICLE IV. MISCELLANEOUS
4.1 Failure or Indulgence Not Waiver. No failure or delay on the part of the Holder in the exercise of any power, right or privilege hereunder shall operate as a waiver thereof, nor shall any single or partial exercise of any such power, right or privilege preclude other or further exercise thereof or of any other right, power or privileges. All rights and remedies of the Holder existing hereunder are cumulative to, and not exclusive of, any rights or remedies otherwise available.
4.2 Notices. All notices, demands, requests, consents, approvals, and other communications required or permitted hereunder shall be in writing and, unless otherwise specified herein, shall be (i) personally served, (ii) deposited in the mail, registered or certified, return receipt requested, postage prepaid, (iii) delivered by reputable air courier service with charges prepaid, or (iv) transmitted by hand delivery, telegram, e-mail or facsimile, addressed as set forth below or to such other address as such party shall have specified most recently by written notice. Any notice or other communication required or permitted to be given hereunder shall be deemed effective (a) upon hand delivery or delivery by e-mail or facsimile, with accurate confirmation generated by the transmitting facsimile machine, at the address or number designated below (if delivered on a business day during normal business hours where such notice is to be received), or the first business day following such delivery (if delivered other than on a business day during normal business hours where such notice is to be received) or (b) on the second business day following the date of mailing by express courier service, fully prepaid, addressed to such address, or upon actual receipt of such mailing, whichever shall first occur. The addresses for such communications shall be:
If to the Borrower, to:
NewGenIvf Group Limited
36/39-36/40, 13th Floor, PS Tower
Sukhumvit 21 Road (Asoke)
Khlong Toei Nuea Sub-district
Watthana District, Bangkok 10110
Attention: Wing Fung Alfred Siu
e-mail: Alfred.Siu@newgenivf.com
If to the Holder:
FIRSTFIRE GLOBAL OPPORTUNITIES FUND, LLC
1040 First Avenue, Suite 190
New York, NY 10022
e-mail: eli@firstfirecapital.com
4.3 Amendments. This Note and any provision hereof may only be amended by an instrument in writing signed by the Borrower and the Holder. The term “Note” and all reference thereto, as used throughout this instrument, shall mean this instrument as originally executed, or if later amended or supplemented, then as so amended or supplemented.
4.4 Assignability. This Note shall be binding upon the Borrower and its successors and assigns, and shall inure to be the benefit of the Holder and its successors and assigns. The Borrower shall not assign this Note or any rights or obligations hereunder without the prior written consent of the Holder. The Holder may assign its rights hereunder to any “accredited investor” (as defined in Rule 501(a) of the 1933 Act) in a private transaction from the Holder or to any of its “affiliates”, as that term is defined under the 1934 Act, without the consent of the Borrower.
4.5 Cost of Collection. If default is made in the payment of this Note, the Borrower shall pay the Holder hereof costs of collection, including reasonable attorneys’ fees.
4.6 Arbitration of Claims; Governing Law; Venue; Attorney’s Fees. The Company and Holder shall submit all Claims (as defined in Exhibit B of the Purchase Agreement) (the “Claims”) arising under this Note or any other agreement between the parties and their affiliates or any Claim relating to the relationship of the parties to binding arbitration pursuant to the arbitration provisions set forth in Exhibit B of the Purchase Agreement (the “Arbitration Provisions”). The Company and Holder hereby acknowledge and agree that the Arbitration Provisions are unconditionally binding on the Company and Holder hereto and are severable from all other provisions of this Note. By executing this Note, Company represents, warrants and covenants that Company has reviewed the Arbitration Provisions carefully, consulted with legal counsel about such provisions (or waived its right to do so), understands that the Arbitration Provisions are intended to allow for the expeditious and efficient resolution of any dispute hereunder, agrees to the terms and limitations set forth in the Arbitration Provisions, and that Company will not take a position contrary to the foregoing representations. The Company acknowledges and agrees that Holder may rely upon the foregoing representations and covenants of the Company regarding the Arbitration Provisions. This Note shall be construed and enforced in accordance with, and all questions concerning the construction, validity, interpretation and performance of this Note shall be governed by, the internal laws of the State of Delaware, without giving effect to any choice of law or conflict of law provision or rule (whether of the State of Delaware or any other jurisdictions) that would cause the application of the laws of any jurisdictions other than the State of Delaware. The Company and Holder consent to and expressly agree that the exclusive venue for arbitration of any Claims arising under this Note or any other agreement between the Company and Holder or their respective affiliates (including but not limited to the Transaction Documents) or any Claim relating to the relationship of the Company and Holder or their respective affiliates shall be in the State of Delaware. Without modifying the Company’s and Holder’s obligations to resolve disputes hereunder pursuant to the Arbitration Provisions, for any litigation arising in connection with any of the Transaction Documents (and notwithstanding the terms (specifically including any governing law and venue terms) of any transfer agent services agreement or other agreement between the Company’s transfer agent and the Company, such litigation specifically includes, without limitation any action between or involving Company and the Company’s transfer agent under the Irrevocable Transfer Agent Instructions (as defined in the Purchase Agreement) or otherwise related to Holder in any way (specifically including, without limitation, any action where Company seeks to obtain an injunction, temporary restraining order, or otherwise prohibit the Company’s transfer agent from issuing shares of Common Stock to Holder for any reason)), each party hereto hereby (i) consents to and expressly submits to the exclusive personal jurisdiction of any state or federal court sitting in the State of Delaware, (ii) expressly submits to the exclusive venue of any such court for the purposes hereof, (iii) agrees to not bring any such action (specifically including, without limitation, any action where Company seeks to obtain an injunction, temporary restraining order, or otherwise prohibit the Company’s transfer agent from issuing shares of Common Stock to Holder for any reason) outside of any state or federal court sitting in the State of Delaware, and (iv) waives any claim of improper venue and any claim or objection that such courts are an inconvenient forum or any other claim, defense or objection to the bringing of any such proceeding in such jurisdiction or to any claim that such venue of the suit, action or proceeding is improper. Notwithstanding anything in the foregoing to the contrary, nothing herein shall limit, or shall be deemed or construed to limit, the ability of the Holder to realize on any collateral or any other security, or to enforce a judgment or other court ruling in favor of the Holder, including through a legal action in any court of competent jurisdiction. The Company hereby irrevocably waives, and agrees not to assert in any suit, action or proceeding, any objection to jurisdiction and venue of any action instituted hereunder, any claim that it is not personally subject to the jurisdiction of any such court, and any claim that such suit, action or proceeding is brought in an inconvenient forum or that the venue of such suit, action or proceeding is improper (including but not limited to based upon forum non conveniens). THE COMPANY HEREBY IRREVOCABLY WAIVES ANY RIGHT IT MAY HAVE, AND AGREES NOT TO REQUEST, A JURY TRIAL FOR THE ADJUDICATION OF ANY DISPUTE HEREUNDER OR IN CONNECTION WITH OR ARISING OUT OF THIS NOTE OR ANY TRANSACTIONS CONTEMPLATED HEREBY. The Company irrevocably waives personal service of process and consents to process being served in any suit, action or proceeding in connection with this Note or any other agreement, certificate, instrument or document contemplated hereby or thereby by mailing a copy thereof via registered or certified mail or overnight delivery (with evidence of delivery) to Company at the address in effect for notices to it under this Note and agrees that such service shall constitute good and sufficient service of process and notice thereof. Nothing contained herein shall be deemed to limit in any way any right to serve process in any other manner permitted by law. The prevailing party in any action or dispute brought in connection with this Note or any other agreement, certificate, instrument or document contemplated hereby or thereby shall be entitled to recover from the other party its reasonable attorney’s fees and costs. If any provision of this Note shall be invalid or unenforceable in any jurisdiction, such invalidity or unenforceability shall not affect the validity or enforceability of the remainder of this Note in that jurisdiction or the validity or enforceability of any provision of this Note in any other jurisdiction.
4.7 Certain Amounts. Whenever pursuant to this Note the Borrower is required to pay an amount in excess of the outstanding Principal Amount (or the portion thereof required to be paid at that time) plus accrued and unpaid interest plus Default Interest on such interest, the Borrower and the Holder agree that the actual damages to the Holder from the receipt of cash payment on this Note may be difficult to determine and the amount to be so paid by the Borrower represents stipulated damages and not a penalty and is intended to compensate the Holder in part for loss of the opportunity to convert this Note and to earn a return from the sale of shares of Common Stock acquired upon conversion of this Note at a price in excess of the price paid for such shares pursuant to this Note. The Borrower and the Holder hereby agree that such amount of stipulated damages is not plainly disproportionate to the possible loss to the Holder from the receipt of a cash payment without the opportunity to convert this Note into shares of Common Stock.
4.8 Purchase Agreement. The Company and the Holder shall be bound by the applicable terms of the Purchase Agreement, and the Transaction Documents entered into in connection herewith and therewith.
4.9 Remedies. The Borrower acknowledges that a breach by it of its obligations hereunder will cause irreparable harm to the Holder, by vitiating the intent and purpose of the transaction contemplated hereby. Accordingly, the Borrower acknowledges that the remedy at law for a breach of its obligations under this Note will be inadequate and agrees, in the event of a breach or threatened breach by the Borrower of the provisions of this Note, that the Holder shall be entitled, in addition to all other available remedies at law or in equity, and in addition to the penalties assessable herein, to an injunction or injunctions restraining, preventing or curing any breach of this Note and to enforce specifically the terms and provisions thereof, without the necessity of showing economic loss and without any bond or other security being required.
4.10 Construction; Headings. This Note shall be deemed to be jointly drafted by the Company and all the Holder and shall not be construed against any person as the drafter hereof. The headings of this Note are for convenience of reference and shall not form part of, or affect the interpretation of, this Note.
4.11 Usury. To the extent it may lawfully do so, the Company hereby agrees not to insist upon or plead or in any manner whatsoever claim, and will resist any and all efforts to be compelled to take the benefit or advantage of, usury laws wherever enacted, now or at any time hereafter in force, in connection with any action or proceeding that may be brought by the Holder in order to enforce any right or remedy under this Note. Notwithstanding any provision to the contrary contained in this Note, it is expressly agreed and provided that the total liability of the Company under this Note for payments which under the applicable law are in the nature of interest shall not exceed the maximum lawful rate authorized under applicable law (the “Maximum Rate”), and, without limiting the foregoing, in no event shall any rate of interest or default interest, or both of them, when aggregated with any other sums which under the applicable law in the nature of interest that the Company may be obligated to pay under this Note exceed such Maximum Rate. It is agreed that if the maximum contract rate of interest allowed by applicable law and applicable to this Note is increased or decreased by statute or any official governmental action subsequent to the Issue Date, the new maximum contract rate of interest allowed by law will be the Maximum Rate applicable to this Note from the effective date thereof forward, unless such application is precluded by applicable law. If under any circumstances whatsoever, interest in excess of the Maximum Rate is paid by the Company to the Holder with respect to indebtedness evidenced by this the Note, such excess shall be applied by the Holder to the unpaid principal balance of any such indebtedness or be refunded to the Company, the manner of handling such excess to be at the Holder’s election.
4.12 Severability. In the event that any provision of this Note is invalid or unenforceable under any applicable statute or rule of law (including any judicial ruling), then such provision shall be deemed inoperative to the extent that it may conflict therewith and shall be deemed modified to conform with such statute or rule of law. Any such provision which may prove invalid or unenforceable under any law shall not affect the validity or enforceability of any other provision of this Note.
4.13 Terms of Future Financings. So long as this Note is outstanding, upon any issuance by the Borrower or any of its Subsidiaries of any Common Stock Equivalents, or amendment to any Common Stock Equivalents that was originally issued before the Issue Date, with any term that the Holder reasonably believes is more favorable to the holder of such security or with a term in favor of the holder of such security that the Holder reasonably believes was not similarly provided to the Holder in this Note (even if the holder of such other security does not receive the benefit of such more favorable term until a default occurs under such other security), then (i) the Borrower shall notify the Holder of such additional or more favorable term within one (1) business day of the issuance and/or amendment (as applicable) of the respective security, and (ii) such term, at Holder’s option, shall become a part of the transaction documents with the Holder (regardless of whether the Borrower complied with the notification provision of this Section 4.13). The types of terms contained in another security that may be more favorable to the holder of such security include, but are not limited to, terms addressing prepayment rate, interest rates, conversion rate, and original issue discount. Notwithstanding the foregoing, this Section 4.13 of this Note shall not apply to (i) the Borrower’s offering of equity securities pursuant to a registration statement of the Borrower; (ii) in connection with the Borrower’s acquisition of all or part of another entity by stock acquisition, merger, consolidation or other reorganization, or by the purchase of all or part of the assets of such other entity approved by the Board of Directors of the Borrower and provided that such transactions are not for the purpose of raising capital); (iii) pursuant to the issuance, vesting and/or exercise of options, warrants, restricted stock, restricted stock units or other common stock purchase rights issued (or to be issued) to employees, officers, directors, or employees of the Borrower for compensatory purposes pursuant to any stock purchase plan, stock option plan, equity incentive plan or other plan or arrangement approved by the Board of Directors of the Borrower at any time; and (iv) pursuant to the exercise or conversion of Common Stock Equivalents outstanding prior to the Issue Date so long as (a) such Common Stock Equivalents were disclosed in the SEC Documents prior to the Issue Date and (b) the terms or provisions of such Common Stock Equivalents were not altered, amended, or changed on or after the Issue Date.
[signature page follows]
IN WITNESS WHEREOF, Borrower has caused this Note to be signed in its name by its duly authorized officer on March 25, 2026.
NewGenIvf Group Limited
| By: | /s/ Wing Fung Alfred Siu | ||
| Name: | Wing Fung Alfred Siu | ||
| Title: | Chief Executive Officer | ||
EXHIBIT A -- NOTICE OF CONVERSION
The undersigned hereby elects to convert $ ____________ principal amount of the Note (defined below) into that number of shares of Common Stock to be issued pursuant to the conversion of the Note (“Common Stock”) as set forth below, of NewGenIvf Group Limited, a British Virgin Islands company (the “Borrower”), according to the conditions of the promissory note of the Borrower dated as of March 25, 2026 (the “Note”), as of the date written below. No fee will be charged to the Holder for any conversion, except for transfer taxes, if any.
Box Checked as to applicable instructions:
| ☐ | The Borrower shall electronically transmit the Common Stock issuable pursuant to this Notice of Conversion to the account of the undersigned or its nominee with DTC through its Deposit Withdrawal Agent Commission system (“DWAC Transfer”). | |
| Name of DTC Prime Broker: | ||
| Account Number: |
| ☐ | The undersigned hereby requests that the Borrower issue a certificate or certificates for the number of shares of Common Stock set forth below (which numbers are based on the Holder’s calculation attached hereto) in the name(s) specified immediately below or, if additional space is necessary, on an attachment hereto: |
| Date of Conversion: | ||||
| Applicable Conversion Price: | $ | |||
| Number of Shares of Common Stock to be Issued Pursuant to Conversion of the Note: | ||||
| Amount of Principal Balance Due remaining Under the Note after this conversion: | ||||
| By: | ||
| Name: | ||
| Title: | ||
| Date: |
Exhibit 12.1
Certification by the Principal Executive Officer
Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002
I, Siu Wing Fung Alfred, certify that:
| 1. | I have reviewed this annual report on Form 20-F of NewGenIvf Group Limited: |
| 2. | Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report; |
| 3. | Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the company as of, and for, the periods presented in this report; |
| 4. | The company’s other certifying officer(s) and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the company and have: |
| (a) | Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the company, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared; |
| (b) | Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles; |
| (c) | Evaluated the effectiveness of the company’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and |
| (d) | Disclosed in this report any change in the company’s internal control over financial reporting that occurred during the period covered by the annual report that has materially affected, or is reasonably likely to materially affect, the company’s internal control over financial reporting; and |
| 5. | The company’s other certifying officer(s) and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the company’s auditors and the audit committee of the company’s board of directors (or persons performing the equivalent functions): |
| (a) | All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the company’s ability to record, process, summarize and report financial information; and |
| (b) | Any fraud, whether or not material, that involves management or other employees who have a significant role in the company’s internal control over financial reporting. |
| Date: March 31, 2026 | ||
| By: | /s/ Siu Wing Fung Alfred | |
| Name: | Siu Wing Fung Alfred | |
| Title: | Chief Executive Officer | |
Exhibit 12.2
Certification by the Principal Financial Officer
Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002
I, Ho Fai Chung, certify that:
| 1. | I have reviewed this annual report on Form 20-F of NewGenIvf Group Limited: |
| 2. | Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report; |
| 3. | Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the company as of, and for, the periods presented in this report; |
| 4. | The company’s other certifying officer(s) and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the company and have: |
| (a) | Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the company, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared; |
| (b) | Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles; |
| (c) | Evaluated the effectiveness of the company’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and |
| (d) | Disclosed in this report any change in the company’s internal control over financial reporting that occurred during the period covered by the annual report that has materially affected, or is reasonably likely to materially affect, the company’s internal control over financial reporting; and |
| 5. | The company’s other certifying officer(s) and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the company’s auditors and the audit committee of the company’s board of directors (or persons performing the equivalent functions): |
| (a) | All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the company’s ability to record, process, summarize and report financial information; and |
| (b) | Any fraud, whether or not material, that involves management or other employees who have a significant role in the company’s internal control over financial reporting. |
| Date: March 31, 2026 | ||
| By: | /s/ Ho Fai Chung | |
| Name: | Ho Fai Chung | |
| Title: | Chief Financial Officer | |
Exhibit 13.1
Certification by the Principal Executive Officer
Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002
In connection with the Annual Report of NewGenIvf Group Limited (the “Company”) on Form 20-F for the year ended December 31, 2025 as filed with the Securities and Exchange Commission on the date hereof (the “Report”), I, Siu Wing Fung Alfred, Chief Executive Officer of the Company, certify, pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, that to my knowledge:
| (1) | The Report fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934; and |
| (2) | The information contained in the Report fairly presents, in all material respects, the financial condition and results of operations of the Company. |
Date: March 31, 2026
| By: | /s/ Siu Wing Fung Alfred | |
| Name: | Siu Wing Fung Alfred | |
| Title: | Chief Executive Officer |
Exhibit 13.2
Certification by the Principal Financial Officer
Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002
In connection with the Annual Report of NewGenIvf Group Limited (the “Company”) on Form 20-F for the year ended December 31, 2025 as filed with the Securities and Exchange Commission on the date hereof (the “Report”), I, Ho Fai Chung, Chief Financial Officer of the Company, certify, pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, that to my knowledge:
| (1) | The Report fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934; and |
| (2) | The information contained in the Report fairly presents, in all material respects, the financial condition and results of operations of the Company. |
Date: March 31, 2026
| By: | /s/ Ho Fai Chung | |
| Name: | Ho Fai Chung | |
| Title: | Chief Financial Officer |