UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16
UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the Month of January 2026
Commission File Number: 001-39957
NEWCELX LTD.
(Translation of registrant’s name into English)
The Circle 6
8058 Zurich, Switzerland
(Address of principal executive office)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F:
CONTENTS
Press Release
☒ Form 20-F ☐ Form 40-F On January 29, 2026, NewcelX Ltd., a corporation incorporated under the laws of Switzerland (the “Company”), issued a press release titled: “NewcelX Announces Updated Corporate Presentation Ahead of Key Spring 2026 Investor and Partnering Conferences” A copy of this press release is furnished herewith as Exhibit 99.1.
Investor Presentation
On January 29, 2026, the Company made available an investor presentation on its website. A copy of the investor presentation is furnished herewith as Exhibit 99.2.
EXHIBIT INDEX
| Exhibit Number |
Description of Document | |
| 99.1 | Press release titled: “NewcelX Announces Updated Corporate Presentation Ahead of Key Spring 2026 Investor and Partnering Conferences” | |
| 99.2 | Investor Presentation |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| NewcelX Ltd. | |||
| Date: January 29, 2026 | By: | /s/ Ronen Twito | |
| Name: | Ronen Twito | ||
| Title: | Chief Executive Officer | ||
Exhibit 99.1

NewcelX Announces Updated Corporate Presentation Ahead of Key Spring 2026 Investor and Partnering Conferences
Reinforces strategic focus on Type 1 Diabetes cell therapy and highlights expansion of Scientific Advisory Board and leadership team
Zurich, Switzerland – January 29, 2026 — NewcelX Ltd. (NASDAQ: NCEL) (“NewcelX” or the “Company”), a clinical-stage cell therapy company advancing transformative treatments for chronic metabolic and neurodegenerative diseases, today announced the release of an updated corporate presentation in advance of its participation in several major spring 2026 investor and partnering conferences, including BIO-Europe Spring in Lisbon.
The updated corporate deck reflects NewcelX’s sharpened strategic focus on its lead cell therapy programs for Type 1 Diabetes, highlighting recent scientific progress, clinical development priorities, and the Company’s differentiated approach to restoring endogenous insulin production through regenerative medicine.
In addition, the presentation introduces several highly experienced and internationally recognized additions to NewcelX’s Scientific Advisory Board and leadership team. These appointments significantly strengthen the Company’s expertise across cell therapy development, translational medicine, clinical strategy, and commercialization, and further position NewcelX to execute on its near- and mid-term value creation milestones.
“Our updated corporate presentation clearly articulates NewcelX’s mission to deliver disease-modifying cell therapies, with Type 1 Diabetes now firmly established as our primary clinical focus,” said Ronen Twito, Executive Chairman & CEO of NewcelX. “Equally important, we are proud to welcome outstanding new scientific and operational leaders whose depth of experience materially enhances our ability to advance our pipeline and engage strategically with partners and investors. As we head into an active spring conference season, we look forward to sharing our progress and vision with the global biotech community.”
Management will be available for one-on-one meetings during upcoming spring conferences, including BIO-Europe Spring 2026 in Lisbon, where the Company plans to engage with potential partners, investors, and collaborators.

About NewcelX Ltd.
The updated presentation is available on the Company’s website at: Corporate Presentation NewcelX Ltd. (Nasdaq: NCEL) is a clinical-stage biotechnology company focused primarily on developing cell-based therapies for Type 1 Diabetes, supported by a broader platform spanning neurodegenerative and metabolic diseases. The Company’s proprietary stem-cell technologies enable the scalable production of functional human cells for “off-the-shelf” therapeutic use, complemented by strategic collaborations and targeted in-house innovation. NewcelX is headquartered in Zurich, Switzerland, with research and development operations in Ness Ziona, Israel.
Social Media: LinkedIn, Facebook, X, Instagram
Website: www.newcelx.com
Investor & Media Contacts
Sarah Bazak, Investors relations
InvestorRelations@newcelx.com
Forward-Looking Statements
This press release contains expressed or implied forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 and other applicable securities laws. For example, NewcelX is using forward-looking statements when it discusses advancing its pipeline and engaging strategically with partners and investors. These forward-looking statements and their implications are based on the current expectations of the management of NewcelX and are subject to a number of factors and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. The following factors, among others, could cause actual results to differ materially from those described in the forward-looking statements: changes in technology and market requirements; potential delays or obstacles in launching or completing clinical trials; products that may not be approved by regulatory agencies; technologies that may not be validated or accepted by the scientific community; the inability to retain or attract key employees; unforeseen scientific difficulties with products in development; higher-than-expected product costs; results in the laboratory that do not translate to clinical success; insufficient patent protection; possible adverse safety outcomes; legislative changes; delays in developing or introducing new technologies, products, or applications; and competitive pressures that could reduce market share or pricing. Except as otherwise required by law, NewcelX does not undertake any obligation to publicly release revisions to these forward-looking statements to reflect events or circumstances after the date hereof or to reflect the occurrence of unanticipated events. More detailed information about the risks and uncertainties affecting the Company is contained under the heading “Risk Factors” in its Annual Report on Form 20-F for the year ended December 31, 2024, filed with the U.S. Securities and Exchange Commission (“SEC”) and available at www.sec.gov, as well as in subsequent filings made by NewcelX, including under the heading “Risk Factors” in its proxy statement/prospectus filed with the SEC on November 6, 2025.
Exhibit 99.2

JANUARY 2026 ( NASDAQ : NCEL ) ©2026 N EW C EL X L TD .

REGENERATIVE MEDICINE TO CURE TYPE 1 DIABETES 72:$5'$&85()257<3( ',$%(7(6 Disclaimer This presentation contains express or implied forward - looking statements within the Private Securities Litigation Reform Act of 1995 and other U . S . Federal securities laws . For example, NewCelX Ltd . (“ NewCelX ,” “we,” “our” or “us”) is using forward - looking statements when we discuss the timing and outcome of our planned clinical trials, including the Phase IIa trial of AstroRx ® in ALS ; our expectations regarding the pre - IND and IND filings for IsletRx ; the development and regulatory path for the DOXA platform ; our ability to obtain regulatory approvals ; our ability to secure and maintain patent protection ; our plans for collaborations and partnerships ; our anticipated market opportunities ; and, our prospects for future growth and revenue generation . These forward - looking statements and their implications are based on the current expectations of our management only and are subject to a number of factors and uncertainties that could cause actual results to differ materially from those described in the forward - looking statements . The following factors, among others, could cause actual results to differ materially from those described in the forward - looking statements : changes in technology and market requirements ; we may encounter delays or obstacles in launching and/or successfully completing our clinical trials ; our products may not be approved by regulatory agencies, our technology may not be validated as it progresses further and its methods may not be accepted by the scientific community ; we may be unable to retain or attract key employees whose knowledge is essential to the development of our products ; unforeseen scientific difficulties may develop with our process ; our products may wind up being more expensive than we anticipates results in the laboratory may not translate to equally good results in real clinical settings ; results of preclinical studies may not correlate with the results of human clinical trials ; our patents may not be sufficient ; our products may harm recipients ; changes in legislation may adversely impact us ; inability to timely develop and introduce new technologies, products and applications ; loss of market share and pressure on pricing resulting from competition, which could cause our actual results or performance to differ materially from those contemplated in such forward - looking statements . Except as otherwise required by law, we undertake no obligation to publicly release any revisions to these forward - looking statements to reflect events or circumstances after the date hereof or to reflect the occurrence of unanticipated events . More detailed information about the risks and uncertainties affecting us is contained under the heading "Risk Factors" in our annual report on Form 20 - F for the year ended December 31 , 2024 filed with the Securities and Exchange Commission (SEC), which is available on the SEC's website, www . sec . gov, and in subsequent filings made by us with the SEC, including under the heading “Risk Factors” in our proxy statement/prospectus, filed with the SEC on September 10 , 2025 . Our logo and some of our trademarks and tradenames are used or incorporated by reference in this presentation . This presentation also includes trademarks, tradenames and service marks that are the property of other organizations . Solely for convenience, trademarks, tradenames and service marks referred to in this presentation may appear without the ®, TM and SM symbols, but those references are not intended to indicate in any way that we will not assert to the fullest extent under applicable law our rights or the rights of the applicable licensor to these trademarks, tradenames and service marks . We obtained the statistical data, market data and other industry data and forecasts described by reference in this presentation from market research, publicly available information and industry publications . Industry publications generally state that they obtain their information from sources that they believe to be reliable, but they do not guarantee the accuracy and completeness of the information . Similarly, while we believe that the statistical data, industry data and forecasts and market research are reliable, we have not independently verified the data, and we do not make any representation as to the accuracy of the information .


2 2 3 Cure Type 1 diabetes We are a clinical stage regenerative medicine company focused on Type 1 Diabetes Cell Therapy using IsletRx Plus highly specialized stem - cell derived insulin producing cells as cell replacement therapy 3 72:$5'$&85()257<3( ',$%(7(6 The NewcelX Factor Today, Value Driven CORE PLATFORM Stem Cell Technology • GMP Manufacturing • Clinical - Grade Bio - Banks • Off - the - Shelf Therapies • Scalable Multi - Indication Platform PRIMARY VALUE DRIVER Type 1 Diabetes IsletRx • Insulin - dependent diabetes • Pre - clinical / IND - enabling • Key partnering focus PLATFORM VALIDATION Neurology – ALS AstroRx ® • Clinical - stage - FDA approved IND for Phase 2a • Proof of concept for stem cell platform ADJACENT CNS PLATFORM DOX A (Dual Orexin Agonist) • Sleep disorders • Neurodegenerative disease optionality STRATEGIC ALLIANCES 4 • Healthy and functional islet cells can produce and secrete insulin in a regulated manner • Cadaveric donor islet cell therapy is a safe and clinically validated treatment for Insulin - Dependent Diabetes 1 • Patients treated achieved Insulin independence for ~2 years following treatment 4 • Main challenges remaining : a severe shortage of donor islet cells and immune suppression that is unhealthy and not always prevent immune rejection Even with strict insulin treatment regimens, patients experience: • Frequent episodes of severe, undetected hypoglycemia • S evere glycemic lability • Progressive diabetic complications: Neuropathies Heart Disease R etinopathy Kidney failure Stroke Durable glycemic control requires functional beta - cell replacement, as demonstrated by donor islet transplantation Insulin therapy and glucose management manages symptoms, but are not a cure Restoring patient’s ability to naturally produce insulin Why Cell Therapy in Type 1 Diabetes - Unmet Need in Insulin Dependent Diabetes 5


Global Market and Facts For Type 1 Diabetes Living with Type 1 Diabetes worldwide 9.5 million people 1. 8 million Children and adolescents below 20 years have type 1 diabetes 2 174,000 Premature Deaths attributed to Type 1 Diabetes 813B$ Lifetime cost in US for health expenditure in Type 1 diabetes patients 4 6 72:$5'$&85()257<3( ',$%(7(6 The global insulin pump market size was valued at USD 6.08 billion in 2024 and is projected to grow to USD 21.65 billion by 2032. North America dominated the insulin pump market with a market share of 57.79% in 2023 5 .

An Active Market – Key Recent Transactions, Developments & Validations 2019 Semma Therapeutics 2019 Acquired by Vertex $950M Preclinical islet cell therapy 1 Diabetes 20 21 Diabetes and cell therapy Sana Biotechnology 2021 Aspect Biosystems /Novo Nordisk Lead asset licensing deal $75M upfr ont $750M total 7 2026 Diabetes ViaCyte 2022 Acquired by Vertex $ 320 M 4 IPO – Nasdaq $ 6.4B Market Cap at IPO 2 20 22 Lineage Cell Therapeutics / Roche (Genentech) Global collaboration & license ( OpRegen ®) $50M upfr ont $670M total potential value 3 2021 / 2022 Vertex / CRISPR Therapeutics 2023 Vertex will get non - exclusive rights to CRISPR Therapeutics ’ CRISPR/Cas 9 gene editing technology.
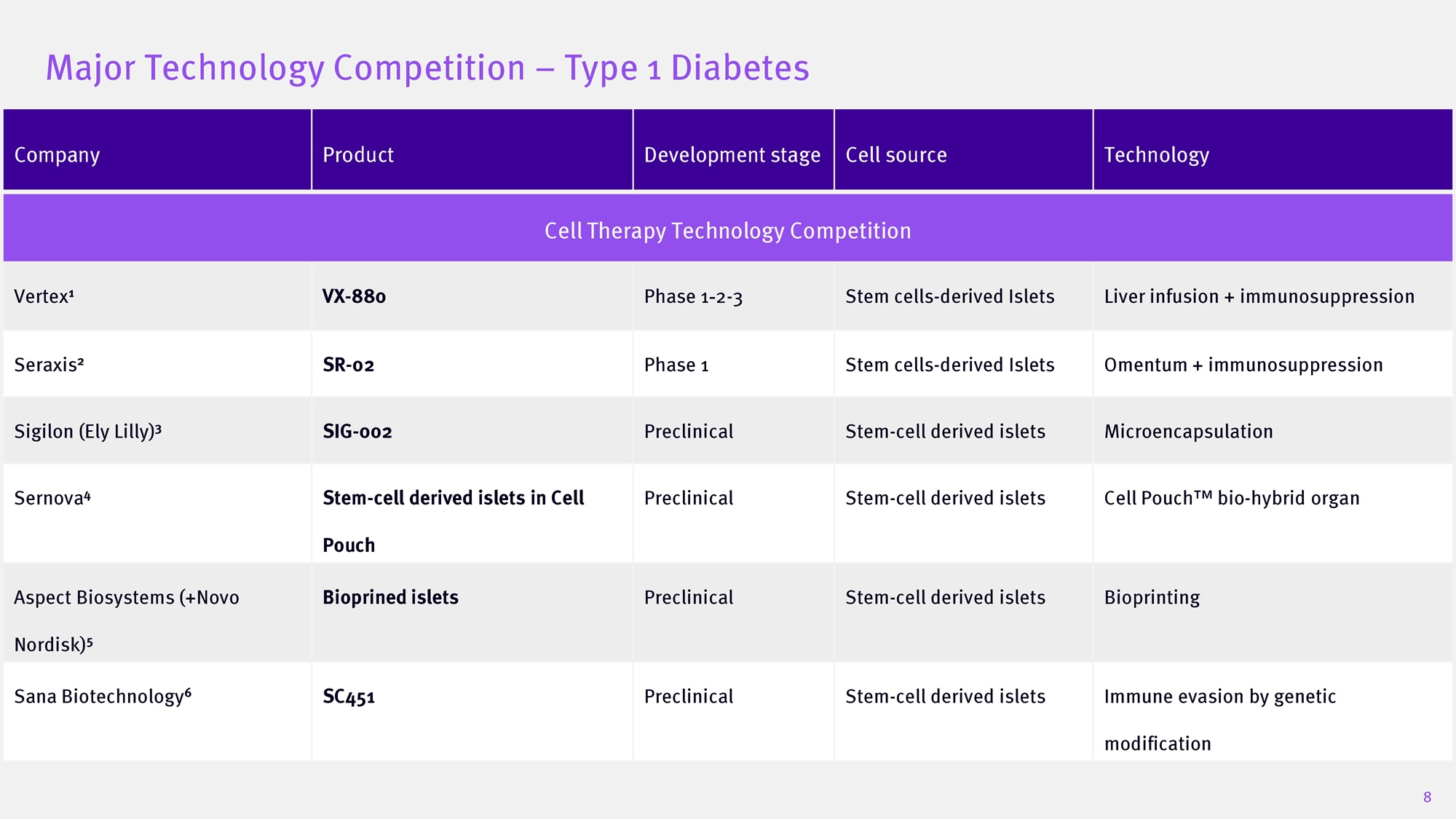
$ 230 M 5 20 23 Diabetes Diabetes and Cell Therapy Diabetes 20 2 6 2025 20 25 SAB Bio private placement Diabetes $175 M upfront $284M total potential value 6 20 20 20 24 7 Major Technology Competition – Type 1 Diabetes Technology Cell source Development stage Product Company Cell Therapy Technology Competition Liver infusion + immunosuppression Stem cells - derived Islets Phase 1 2 - 3 VX - 880 Vertex 1 Omentum + immunosuppression Stem cells - derived Islets Phase 1 SR - 02 Seraxis 2 Microencapsulation Stem - cell derived islets Preclinical SIG - 002 Sigilon (Ely Lilly) 3 Cell Pouch bio - hybrid organ Stem - cell derived islets Preclinical Stem - cell derived islets in Cell Pouch Sernova 4 Bioprinting Stem - cell derived islets Preclinical Bioprined islets Aspect Biosystems (+Novo Nordisk) 5 Immune evasion by genetic modification Stem - cell derived islets Preclinical SC451 Sana Biotechnology 6 8 Differentiation of PSCs into islet - like clusters 9 Pluripotent Definitive Endoderm Endocrine progenitors Posterior Foregut Pancreatic progenitors Primitive gut tube Immature Endocrine S S S S S S S 7 - stage protocol, ~28 days for production Recapitulating pancreatic development stages Final enrichment step - capturing the elite cells for optimized clinical performance β cells enrichment IsletRx Plus IsletRx IsletRxPlus: Functional Islet Cells to cure diabetes from unlimited source - GMP PSCs line PSC 72:$5'$&85()257<3( ',$%(7(6


iTOL 102 – With Enriched IsletRx Plus Cells • Functional insulin - secreting islet cells • Physiologic glucose - responsive insulin release • Unlimited and standardized cell supply • Efficacy and safety in preclinical studies • Durable – long term function in diabetic animal models • Localized immune tolerance via FasL - based approach • Avoids lifelong systemic immunosuppression • Designed for integration with partner technologies Immune Protection Strategy 10 Regulatory Pathway IND in Q 1 2027 FIH Q 2 2027 72:$5'$&85()257<3( ',$%(7(6 Flagship Diabetes Program 10 • Regenerative medicine without the need for life - long immunosuppression • Leverages the naturally occurring protein , Fas Ligand ( FasL ) • Creates localized immune privilege environment • BIRD Grant funded (~$ 1 M) • Successful POC results (Q 2 2024 ) • Breakthrough designation by DRI • Successful Pre - IND Meeting with the FDA (Q 1 2025 ) iTOL - 102 : Kadimastem & iTolerance co - development Local Immune tolerance to allow IsletRx Plus survival and function - Eliminating the need for systemic immunosuppression 11


IsletRx Co - development & Projects Pipeline D EVELOPMENTS T YPE 1 D IABETES 12 In house product using encapsulation: EnCap - IsletRx Plus Collaboration project for h ypo immune gene edited cells : NCX 007 Collaboration project for device: In - Scaffold - IsletRx Plus INDICATION • Long - term therapeutic effect was achieved in an immunocompetent animal model (C 57 BL/ 6 mice ) • IsletRx Plus cells well protected from host immune system In House Product Development: Encap - IsletRx Plus Preclinical Efficacy Encap - IsletRx Plus treated diabetic mice (STZ) demonstrated balanced and normal blood glucose levels Days post treatment Blood Glucose (mg/dl) https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2021.635405/full 13


• Co - Development with TargetGene Biotechnologies Inc. Genome editing to evade immune system recognition allowing graft persistence. • Using precise, validated, gene editing platform, with minimal off - target editing • Enables the production of hypo immune cells for the treatment of Type 1 Diabetes • No need for immune suppression • Increased safety and efficacy Next generation cell line NCX 007 – hypoimmune cells 14 In - Scaffold - IsletRx Plus • Bioengineered 3 D Printed Device: IsletRx cells can be implanted in a bioengineered device featuring a 3 D printed scaffold for easier surgical retrieval and immuno - isolation • Innovative 3 D Printing Process: The scaffold utilizes melt electrospinning writing (MEW), a novel solvent - less technique that creates micrometer - thin fibers with highly controllable architectures • Successful Preclinical Results: Encapsulated islets in scaffolds demonstrated production of human insulin and maintained viability for approximately 3 months in preclinical studies Intact IsletRx cells Expansion and differentiation protocols Potential wide range of cells treating a wide range of diseases.

Pluripotent Stem Cell Expansion Bio - Banking GMP quality Starting Material . Clinical - grade Embryonic Stem Cell line Unlimited # of Pluripotent Stem Cell Bio - banks Large scale Key Platform Attributes • Renewable pluripotent cell source enabling consistent supply • No genetic modification required (unless indication - specific) • Residual pluripotent cells controlled below detection limits • Scalable for late - stage clinical development and commercialization • Supports multi - indication expansion from a single platform Proprietary Guided Differentiation (in - house cGMP) • Controlled differentiation into target cell types • Core focus: – Pancreatic / islet - like cells (Type 1 Diabetes) • Platform validation: – Astrocytes (ALS – clinical - stage) • Release - tested, ready - to - use formulation Proprietary expansion and differentiation processes 15 Proof of platform product execution from bench to bed: FDA - approved IND, phase 2 a using AstroRx® cell therapy product for clinical trial in ALS patients 16 72:$5'$&85()257<3( ',$%(7(6

Astrocytes - Cell Therapy Treatment for Neurodegenerative Diseases a Validated Platform AstroRx ® AstroRx® Cell Product AstroRx ® has Orphan drug designation from the U.S.

Food and Drug Administration ( “ FDA ” ) 17 72:$5'$&85()257<3( ',$%(7(6 Why Use Astrocytes for ALS - AstroRx ® In neurodegeneration, neurons die because their support cells (Astrocytes) fail AstroRx ® contains functional healthy astrocytes to protect ALS - diseased motor neurons using multiple mechanisms of action In ALS, the patient ’ s own astrocytes fail to support motor neuron survival 1. Izrael et al, 2020 Front Neurosci . 2020 ; 14 : 824 . doi : 10.3389 /fnins. 2020.00824 2. Izrael et al, 2021 Front Med (Lausanne) 2021 ; 8 : 740071 . doi : 10.3389 /fmed. 2021.740071 AstroRx ® Healthy, Functional Astrocytes ALS Patients’ Astrocytes Mechanism of Action Secrete neurotrophic factors 1 Remove toxic factors (i.e.
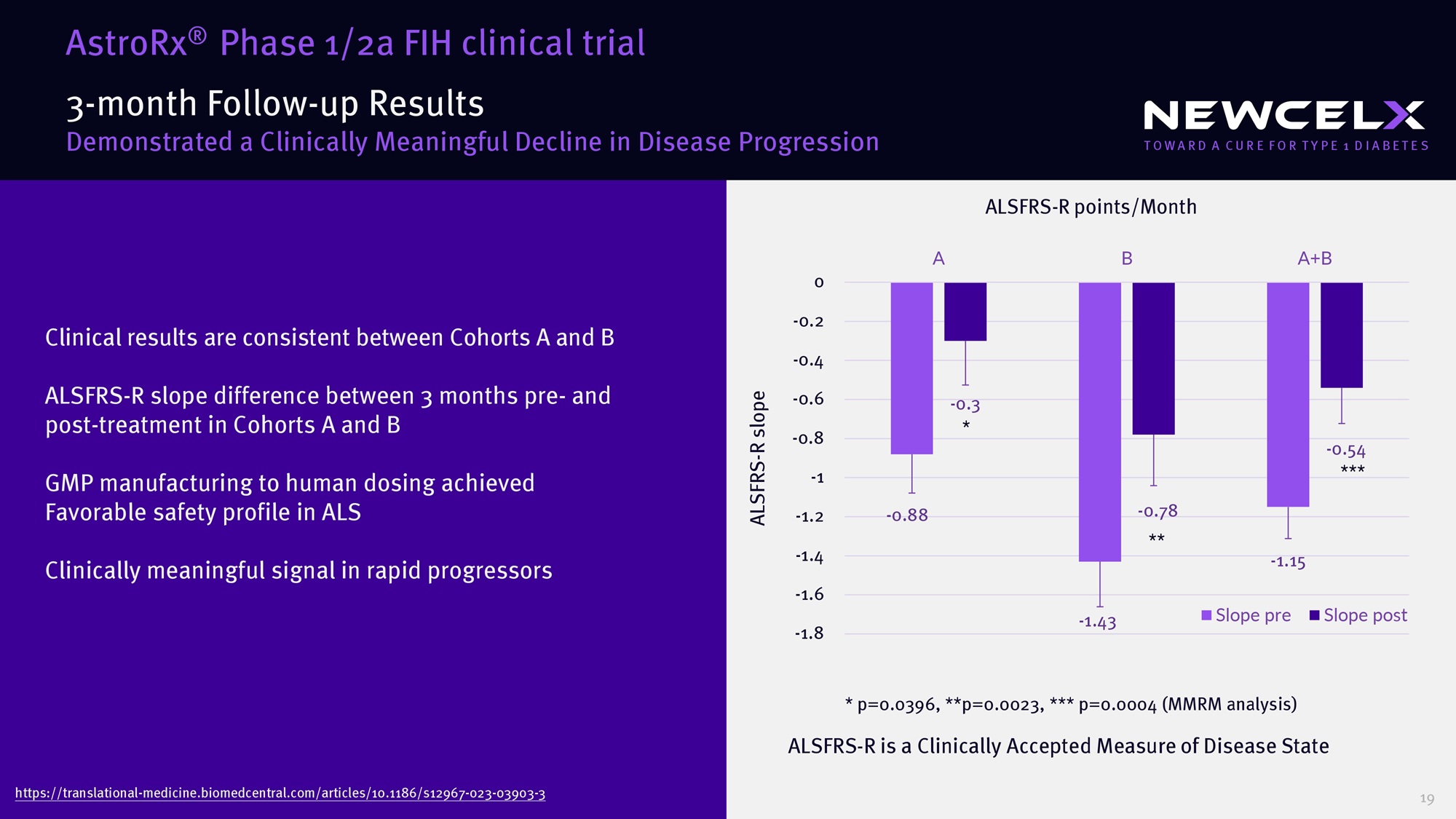
glutamate) 1 Regulate oxidative stress 1 Immune - modulation 2 Injecting AstroRx ® Healthy, Functional Astrocytes in the CNS ALS Patients’ CNS Environment Astrocyte Capillary Neuron AstroRx® Support motor neurons by secreting neurotrophic factors and clearing toxic environment Healthy CNS Environment Astrocytes malfunction in ALS 18 3 - month Follow - up Results Demonstrated a Clinically Meaningful Decline in Disease Progression Clinical results are consistent between Cohorts A and B ALSFRS - R slope difference between 3 months pre - and post - treatment in Cohorts A and B GMP manufacturing to human dosing achieved Favorable safety profile in ALS Clinically meaningful signal in rapid progressors ALSFRS - R is a Clinically Accepted Measure of Disease State * p = 0.0396 , **p= 0.0023 , *** p= 0.0004 (MMRM analysis) - 0.88 - 1.43 - 1.15 - 0.3 - 0.78 - 0.54 -1.8 -1.6 -1.4 -1.2 -1 -0.8 -0.6 -0.4 -0.2 0 A B A+B ALSFRS - R slope Slope pre Slope post * ** *** ALSFRS - R points/Month https://translational - medicine.biomedcentral.com/articles/ 10.1186 /s 12967 - 023 - 03903 - 3 AstroRx ® Phase 1 / 2 a FIH clinical trial 19 Strategic Enhancements Positioned for potential out - licensing or strategic partnerships Target indications: • Targeting sleep – wake regulation • Rare Sleep Disorders (Narcolepsy, Idiopathic Hypersomnia) • Neurodegenerative / neuroinflammation diseases (such as ߙ - synucleinopathies ) • Diabetes - Associated Neurological Disorders (DANS) DOXA (Dual Orexin - Receptor Agonist) Platform 20 72:$5'$&85()257<3( ',$%(7(6


21 21 Scalability & Clinical Manufacturing Capabilities 21 From Art to Industry Long - term success in cell therapy through reproducible expansion and differentiation technology, manufacturing systems that are scalable, and cost - efficient Manufacturing Strategy Allogeneic, Off - the - Shelf Model Enables standardized production and broad patient access, proven ability for frozen product developed Core Advantage GMP Master Cell Banks Freezing technology - easily shipped worldwide Scale up manufacturing capabilities Agreement with well - established CDMO for clinical manufacturing 72:$5'$&85()257<3( ',$%(7(6 22 22 IND Preclinical Discovery Indication Target Program Priority Program Type 1 Diabetes Allogeneic Cell Therapy, without chronic immuno supression ITOL102 – IsletRxPlus (Priority Program) Adjacent Programs ALS (Amyotrophic Lateral Sclerosis) Allogeneic Astrocytes AstroRx ® (Platform Validation) Neurodegenerative Diseases (MS, PD) Narcolepsy, Sleep Disorders (PoC), Dual Orexin Agonist (Small Molecule) DOXA Platform Neurodegenerative Diseases (PD) NewcelX Pipeline Focus – Type 1 Diabetes & CNS Disorders 22


Patent Protection Patents in major countries/markets such as the US, EU and other countries providing NewCelX with robust technology protection Methods for differentiating and purifying pancreatic endocrine cells Insulin producing cells derived from pluripotent stem cells Methods of generating glial and neuronal cells and use of same for the treatment of medical conditions of the CNS Methods of generating glial and neuronal cells Directed differentiation of astrocytes from human pluripotent stem cells for use in drug screening and the treatment of amyotrophic lateral sclerosis (ALS) Dihydro - quinazoline , - benzothiazine and - benzoxazine derivatives and use thereof as orexin receptors agonists for treating or preventing neurological diseases (Chemical matter / synthesis ) Dihydro - quinazoline , - benzothiazine and - benzoxazine derivatives and use thereof as orexin receptors agonists for treating or preventing neurological diseases ( Therapeutic use / indications) PCT Phase PCT Phase Granted 2 Granted Granted Granted Granted 1 1. Excluding certain patents in the U.S. and Brazil. 2. Excluding certain patents in the U.S., China, and Canada. 23 The Partnership "Blueprints" Model: We aim to monetize the platform in three ways: 1.

Program Licensing: Regional deals for IsletRx / AstroRx 2. Platform Partnerships: Licensing the "Manufacturing Blueprints" to partners (e.g., Pharma wants Dopamine neurons; we build them) 3. Delivery Partnerships : Device co - development Corporate 24 72:$5'$&85()257<3( ',$%(7(6 Leadership Team 25 Prof.

Michel Revel Director & CSO Ronen Twito Executive Chairman & CEO Prof. Ariel Revel, MD Medical Director Alex Zwyer , MBA Director & CBO Eric Konofal , MD, PhD Head of DOXA Program Kfir Molakandov , PhD VP of R&D Omri Hagai CFO Scientific Advisory Board Prof. Tamir Ben Hur M.D., Ph.D. Associate Director, Hadassah BrainLabs Jeremy M. Shefner, MD, PhD Chief Medical Officer, Barrow Neurological Institute Julien Boisdron , MD Chief Medical Officer, Roche The Circle 6 | P.O. Box CH - 8058 Zurich Switzerland Headquarter in Switzerland : +41 44 512 21 50 Main office in Israel : +972 - 73 - 797 - 1613 info@newcelx.com 26 72:$5'$&85()257<3( ',$%(7(6


References page 1. https://www.diabetesresearchclinicalpractice.com/article/S 0168 - 8227 ( 25 ) 00291 - 8 /fulltext 2. https://diabetesatlas.org/data - by - indicator/type - 1 - diabetes - estimates/people - with - type - 1 - diabetes - 0 - 19 - y/ 3. https://www.diabetesresearchclinicalpractice.com/article/S 0168 - 8227 ( 25 ) 00291 - 8 /fulltext 4. https://www.liebertpub.com/doi/ 10.1089 /dia. 2019.0398 5. https://www.fortunebusinessinsights.com/insulin - pump - market - 102735 #:~:text=KEY% 20 MARKET% 20 INSIGHTS,share% 20 of% 2057.79 % 25 % 20 in% 202023 . Page 6 27 1. https://investors.vrtx.com/news - releases/news - release - details/vertex - acquire - semma - therapeutics - goal - developing - curative - cell 2. https://www.biopharmadive.com/news/sana - biotech - ipo - record - pace - 2021 / 594496 / 3. https://www.businesswire.com/news/home/ 20211220005028 /en/Lineage - Establishes - Exclusive - Worldwide - Collaboration - With - Genentech - for - the - Development - and - Commercialization - of - OpRegen - RPE - Cell - Therapy - for - the - Treatment - of - Ocular - Disorders 4. https://www.fiercebiotech.com/biotech/vertex - absorbs - viacyte - 320 m - clearing - out - competition - stem - cell - based - diabetes - treatments 5. https://www.pharmaceutical - technology.com/news/vertex - crispr - diabetes - therapies/ 6. https://www.globenewswire.com/news - release/ 2025 / 07 / 21 / 3118633 / 0 /en/SAB - BIO - Announces - Oversubscribed - 175 - Million - Private - Placement.html 7. https://www.theglobeandmail.com/business/article - aspect - biosystems - expands - novo - nordisk - partnership/ Page 7 Page 8 1. https://news.vrtx.com/news - releases/news - release - details/vertex - presents - positive - data - zimislecel - type - 1 - diabetes 2. https://seraxis.com/2024/10/15/oct - 15 - 2024/ 3. https://pryzm.ozmosi.com/product/26467 4. https://sernova.com/press_releases/sernova - biotherapeutics - provides - positive - interim - data - from - ongoing - phase - 1 - 2 - clinical - trial - of - cell - pouch - bio - hybrid - organ - in - patients - living - with - type - 1 - diabetes/ 5. https://www.aspectbiosystems.com/news - resources/novo - nordisk - partnership - to - develop - bioprinted - tissue - therapeutics - for - diabetes - and - obesity 6. https://ir.sana.com/static - files/33b2aa85 - 6fb6 - 47cf - baa5 - f97b587c8227