UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
Report of Foreign Private Issuer
Pursuant to Rule 13a-16 or 15d-16
of the Securities Exchange Act of 1934
For the Month of November 2025
Commission File Number 001-35948
Kamada Ltd.
(Translation of registrant’s name into English)
2
Holzman Street
Science Park, P.O. Box 4081
Rehovot 7670402
Israel
(Address of principal executive offices)
Indicate by check mark whether the registrant files or will file annual reports under cover Form 20-F or Form 40-F.
Form 20-F ☒ Form 40-F ☐
This Form 6-K is being incorporated by reference into the Registrant’s Form S-8 Registration Statements, File Nos. 333-192720, 333-207933, 333-215983, 333-222891, 333-233267 and 333-265866.
The following exhibits are attached:
| 99.1 | Kamada Reports Strong Third Quarter and Nine Month 2025 Financial Results with over 30% Year-over-Year Profitability Growth | |
| 99.2 | Company’s Presentation – November 2025 | |
| 99.3 | Kamada Ltd’s Consolidated Financial Statements as of September 30, 2025 (Unaudited) | |
| 101.INS | Inline XBRL Instance Document | |
| 101.SCH | Inline XBRL Taxonomy Extension Schema Document | |
| 101.CAL | Inline XBRL Taxonomy Extension Calculation Linkbase Document | |
| 101.DEF | Inline XBRL Taxonomy Extension Definition Linkbase Document | |
| 101.LAB | Inline XBRL Taxonomy Extension Label Linkbase Document | |
| 101.PRE | Inline XBRL Taxonomy Extension Presentation Linkbase Document | |
| 104 | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) |
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| Date: November 10, 2025 | KAMADA LTD. | |
| By: | /s/ Nir Livneh | |
|
Nir Livneh Vice
President General Counsel and |
||
EXHIBIT INDEX
| EXHIBIT NO. | DESCRIPTION | |
| 99.1 | Kamada Reports Strong Third Quarter and Nine Month 2025 Financial Results with over 30% Year-over-Year Profitability Growth | |
| 99.2 | Company’s Presentation – November 2025 | |
| 99.3 | Kamada Ltd’s Consolidated Financial Statements as of September 30, 2025 (Unaudited) | |
| 101.INS | Inline XBRL Instance Document | |
| 101.SCH | Inline XBRL Taxonomy Extension Schema Document | |
| 101.CAL | Inline XBRL Taxonomy Extension Calculation Linkbase Document | |
| 101.DEF | Inline XBRL Taxonomy Extension Definition Linkbase Document | |
| 101.LAB | Inline XBRL Taxonomy Extension Label Linkbase Document | |
| 101.PRE | Inline XBRL Taxonomy Extension Presentation Linkbase Document | |
| 104 | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) |
4
Exhibit 99.1
Kamada Reports Strong Third Quarter and Nine Month 2025 Financial Results with over 30% Year-over-Year Profitability Growth
| ● | Third Quarter Revenues of $47.0 Million, up 13% Year-over-Year, and Adjusted EBITDA of $11.7 Million, up 34% Year-Over Year |
| ● | Nine Month Revenue of $135.8 Million, up 11% Year-over-Year; Adjusted EBITDA of $34.2 Million, up 35% Year-over-Year |
| ● | Positive Outlook for Remainder of 2025 Based on the Company’s Diverse Product Portfolio Supports Full-Year Revenue Guidance of $178 Million-$182 Million and Adjusted EBITDA of $40 Million-$44 Million |
| ● | Generated $17.9 Million of Cash from Operations During First Nine Months of 2025; as of September 30, 2025, had $72.0 Million of Available Cash |
| ● | Company Continues to Focus on Advancing Business Development Opportunities to Support Continued Long-Term Annual Double-Digit Profitable Growth |
| ● | Interim Futility Analysis of the Pivotal Phase 3 InnovAATe Clinical Trial for the Inhaled AAT Therapy to be Conducted in Current Quarter |
| ● | Conference Call and Live Webcast Today at 8:30am ET |
REHOVOT, Israel, and HOBOKEN, NJ – November 10, 2025 -- Kamada Ltd. (NASDAQ: KMDA; TASE: KMDA.TA), a global biopharmaceutical company with a portfolio of marketed products indicated for rare and serious conditions and a leader in the specialty plasma-derived field, today announced financial results for the three months and nine months ended September 30, 2025.
“Our strong operational and financial momentum continues as we delivered the strongest quarter of the year,” said Amir London, Kamada’s Chief Executive Officer. “We continue to execute on our strategy and generate significant profitable growth through the diversity of our product portfolio. Total revenues for the first nine months of the year of $135.8 million, representing an 11% year-over-year increase, and adjusted EBITDA of $34.2 million, up 35% year-over-year, representing a 25% margin of revenues. Based on our consistent, strong performance in the first nine months of the year, disciplined management of operational expenses, and positive outlook for the remainder of 2025, we are reiterating our full-year 2025 revenue guidance of between $178 million to $182 million and our annual adjusted EBITDA guidance of $40 million to $44 million.”
“We have consistently demonstrated our ability to convert profitability to operational cash flow, as we generated $17.9 million of cash from operating activities during the first nine months of the year, contributing to our strong cash position as of the end of the quarter of $72.0 million. As we advance our business development initiatives, we plan to leverage our financial strength to enrich our portfolio of marketed products and support our continued long-term annual double-digit profitable growth,” added Mr. London.
“Earlier this year we announced the launch of a comprehensive post-marketing research program for CYTOGAM® with the aim of generating key data in support of the benefits of the product in the management of cytomegalovirus (CMV) in solid organ transplantation. In connection with this program, we announced last week the initiation of the investigator-initiated SHIELD study, which will be conducted by leading experts in CMV and organ transplantation, investigating the benefits of CYTOGAM administered at the conclusion of the anti-viral prophylaxis to reduce the risk of clinically significant late CMV in kidney transplant recipients,” continued Mr. London.
“We continue to invest in our plasma collection operations and recently reported the receipt of an FDA approval for our Houston facility. We are ramping up plasma collection at our three operational centers and are in active discussions with potential customers to secure long-term sales agreements for normal source plasma. Lastly, we continue to advance our ongoing pivotal Phase 3 InnovAATe clinical trial for our inhaled Alpha-1 Antitrypsin therapy, with the interim futility analysis to be conducted by the end of this year,” concluded Mr. London.
Financial Highlights for the Three Months Ended September 30, 2025
| ● | Total revenues were $47.0 million in the third quarter of 2025, up 13% compared to $41.7 million in the third quarter of 2024. Consistent with previous quarters of the year, the increase in revenues was driven by the diversity of the Company’s portfolio, primarily attributable to increased sales of GLASSIA® in ex-U.S. markets, increased sales in the Distribution segment, as well as VARIZIG® U.S. sales. |
| ● | Gross profit and gross margins were $19.8 million and 42%, respectively, in the third quarter of 2025, compared to $17.2 million and 41%, respectively, in the third quarter of 2024. The increase in both metrics is attributable to the continued improved sales mix and overall increased commercial scale. |
| ● | Operating expenses, including R&D, S&M, G&A and other expenses, totaled $11.9 million in the third quarter of 2025, consistent with the level of these expenses in the third quarter of 2024. The reduction in R&D expenses during the quarter was mainly related to development projects timing changes. |
| ● | Net income was $5.3 million, or $0.09 per diluted share, in the third quarter of 2025, up 37% as compared to $3.9 million, or $0.07 per diluted share, in the third quarter of 2024. |
| ● | Adjusted EBITDA, as detailed in the tables below, was $11.7 million in the third quarter of 2025, up 34% over the $8.8 million achieved in the third quarter of 2024. |
| ● | Cash provided by operating activities was $10.4 million in the third quarter of 2025, as compared to cash provided by operating activities of $22.2 million in the third quarter of 2024. The decrease is associated with the increase in working capital that supports the Company’s continued growth. |
Financial Highlights for the Nine Months Ended September 30, 2025
| ● | Total revenues for the first nine months of 2025 were $135.8 million, an 11% increase from the $121.9 million generated in the first nine months of 2024. The overall increase in revenues was driven by the diversity of the Company’s portfolio, primarily attributable to increased sales of GLASSIA® in ex U.S. markets, increased sales in the Distribution segment, as well as VARIZIG® U.S. sales. |
| ● | Gross profit and gross margins for the first nine months of 2025 were $59.4 million and 44%, respectively, compared to $52.9 million and 43%, respectively, in the first nine months of 2024. The increase in gross profit is in line with the continued improved sales mix and overall increased commercial scale. |
| ● | Operating expenses, including R&D, S&M, G&A and other expenses, totaled $36.8 million in the first nine months of 2025, as compared to $38.0 million in the first nine months of 2024. The reduction in R&D expenses, year over-year, was mainly related to development projects timing changes. |
| ● | Net income for the first nine months of 2025 was $16.6 million, or $0.29 per diluted share, a 56% increase as compared to net income of $10.7 million or $0.18 per diluted share, in the first nine months of 2024. |
| ● | Adjusted EBITDA, as detailed in the tables below, was $34.2 million in the first nine months of 2025, a 35% increase as compared to $25.4 million in the first nine months of 2024. |
| ● | Cash provided by operating activities during the first nine months of 2025 was approximately $17.9 million, as compared to $37.2 million during the first nine months of 2024. The decrease was associated with the increase in working capital that supports the Company’s continued growth. |
Balance Sheet Highlights
As of September 30, 2025, Kamada had cash and cash equivalents of $72.0 million, as compared to $78.4 million as of December 31, 2024. While cash provided by operating activities totaled $17.9 million, net cash used in investment activities of $7.1 million and net cash used in financing activities of $17.2 million, of which $11.5 million was associated with the payment of a special cash dividend, collectively resulted in an overall decrease in cash balance.
Recent Corporate Highlights
| ● | Announced that the first patient was enrolled into an investigator-initiated post-marketing clinical trial of CYTOGAM® (CMV-IGIV) to prevent late CMV infection, a common post-transplant infectious complication that remains an unaddressed medical need despite recent advances in anti-viral drug therapies. The Strategic Help with Immunoglobulin to Enhance protection against Late Disease (CMV), or SHIELD study, is a prospective, randomized, controlled multicenter investigator-initiated study in CMV high-risk kidney transplant recipients. |
| ● | Announced that the FDA has approved the supplement to the Company’s existing Biologics License Application (BLA) for Kamada Plasma’s collection center in Houston, TX. The approval was obtained following an on-site inspection made by the FDA during the second quarter of this year. The center is now cleared to commence commercial sales of normal source plasma. The 12,000 square foot Houston facility supports 50 donor beds, with a planned capacity of approximately 50,000 liters per year and is anticipated to be one of the largest collection centers for specialty plasma in the U.S. Kamada intends to seek a subsequent inspection and approval by the European Medicines Agency (EMA) of this site. |
Fiscal 2025 Guidance
Kamada is reiterating its annual financial guidance comprising of 2025 total revenues in the range of $178 million to $182 million and adjusted EBITDA guidance in the range of $40 million to $44 million, representing double digit top- and bottom-line growth year-over-year.
Conference Call Details
Kamada’s management will host an investment community conference call on Monday, November 10, at 8:30am Eastern Time to discuss these results and answer questions. Shareholders and other interested parties may participate in the call by dialing 1-877-407-0792 (from within the U.S.), 1-809-406-247 (from Israel), or 1-201-689- 8263 (International) using conference I.D. 13756537. The call will be webcast live on the internet at: https://viavid.webcasts.com/starthere.jsp?ei=1738840&tp_key=92257bc662.
Non-IFRS financial measures
We present EBITDA and adjusted EBITDA because we use these non-IFRS financial measures to assess our operational performance, for financial and operational decision-making, and as a means to evaluate period-to-period comparisons on a consistent basis. Management believes these non-IFRS financial measures are useful to investors because: (1) they allow for greater transparency with respect to key metrics used by management in its financial and operational decision-making and provide investors with a meaningful perspective on the current underlying performance of the Company’s core ongoing operations; and (2) they exclude the impact of certain items that are not directly attributable to our core operating performance and that may obscure trends in the core operating performance of the business. Non-IFRS financial measures have limitations as an analytical tool and should not be considered in isolation from, or as a substitute for, our IFRS results. We expect to continue reporting non-IFRS financial measures, adjusting for the items described below, and we expect to continue to incur expenses similar to certain of the non-cash, non-IFRS adjustments described below. Accordingly, unless otherwise stated, the exclusion of these and other similar items in the presentation of non-IFRS financial measures should not be construed as an inference that these items are unusual, infrequent or non-recurring. EBITDA and adjusted EBITDA are not recognized terms under IFRS and do not purport to be an alternative to IFRS terms as an indicator of operating performance or any other IFRS measure. Moreover, because not all companies use identical measures and calculations, the presentation of EBITDA and adjusted EBITDA may not be comparable to other similarly titled measures of other companies. EBITDA is defined as net income (loss), plus income tax expense, plus or minus financial income or expenses, net, plus or minus income or expense in respect of securities measured at fair value, net, plus or minus income or expenses in respect of currency exchange differences and derivatives instruments, net, plus depreciation and amortization expense, whereas adjusted EBITDA is the EBITDA plus non-cash share-based compensation expenses and certain other costs.
For the projected 2025 adjusted EBITDA information presented herein, the Company is unable to provide a reconciliation of this forward measure to the most comparable IFRS financial measure because the information for these measures is dependent on future events, many of which are outside of the Company’s control. Additionally, estimating such forward-looking measures and providing a meaningful reconciliation consistent with the Company’s accounting policies for future periods is meaningfully difficult and requires a level of precision that is unavailable for these future periods and cannot be accomplished without unreasonable effort. Forward-looking non-IFRS measures are estimated in a manner consistent with the relevant definitions and assumptions noted in the Company’s adjusted EBITDA for historical periods.
About Kamada
Kamada Ltd. (the “Company”) is a global biopharmaceutical company with a portfolio of marketed products indicated for rare and serious conditions and a leader in the specialty plasma-derived therapies field. The Company’s strategy is focused on driving profitable growth through four primary growth pillars: First, organic growth from its commercial activities, including continued investment in the commercialization and life cycle management of its proprietary products, which include six FDA-approved specialty plasma-derived products: KEDRAB®, CYTOGAM®, GLASSIA®, WINRHO SDF®, VARIZIG® and HEPAGAM B®, as well as KAMRAB®, KAMRHO (D)® and two types of equine-based anti-snake venom products, and the products in the distribution segment portfolio, mainly through the launch of several biosimilar products in Israel. Second: the Company aims to secure significant new business development, in-licensing, collaboration and/or merger and acquisition opportunities, which are anticipated to enhance the Company’s marketed products portfolio and leverage its financial strength and existing commercial infrastructure to drive long-term growth. Third: the Company is expanding its plasma collection operations to support revenue growth through the sale of normal source plasma to other plasma-derived manufacturers, and to support its increasing demand for hyper-immune plasma. The Company currently owns three operating plasma collection centers in the United States, in Beaumont Texas, Houston Texas, and San Antonio, Texas. Lastly, the Company is leveraging its manufacturing, research and development expertise to advance the development and commercialization of additional product candidates, targeting areas of significant unmet medical need, with the lead product candidate Inhaled AAT, for which the Company is continuing to progress the InnovAATe clinical trial, a randomized, double-blind, placebo-controlled, pivotal Phase 3 trial. FIMI Opportunity Funds, the leading private equity firm in Israel, is the Company’s controlling shareholder, beneficially owning approximately 38% of the outstanding ordinary shares.
Cautionary Note Regarding Forward-Looking Statements
This release includes forward-looking statements within the meaning of Section 21E of the U.S. Securities Exchange Act of 1934, as amended, and the safe harbor provisions of the U.S. Private Securities Litigation Reform Act of 1995. Forward-looking statements are statements that are not historical facts, including statements regarding: 1) positive outlook for the remainder of 2025, supporting the reiteration of our full-year 2025 revenue guidance of between $178 million to $182 million and our annual adjusted EBITDA guidance of $40 million to $44 million; 2) leveraging the Company’s financial strength to enrich our portfolio of marketed products and support our continued long-term annual double-digit profitable growth; 3) futility analysis of the Pivotal Phase 3 InnovAATe Clinical Trial for the Inhaled AAT Therapy to be conducted by the end of 2025; 4) launching a comprehensive post marketing research program for CYTOGAM® with the aim of generating key data in support of the benefits of the product in the management of cytomegalovirus (CMV) in solid organ transplantation; 5) discussions with potential customers to secure long-term sales agreements for normal source plasma; 6) the site in Houston, TX planned capacity of approximately 50,000 liters per year, anticipated to be one of the largest collection centers for specialty plasma in the U.S.; and 7) the intention to seek a subsequent inspection and approval by the European Medicines Agency (EMA) of the Houston site. Forward-looking statements are based on Kamada’s current knowledge and its present beliefs and expectations regarding possible future events and are subject to risks, uncertainties and assumptions. Actual results and the timing of events could differ materially from those anticipated in these forward-looking statements as a result of several factors including, but not limited to the evolving nature of the conflicts in the Middle East and the impact of such conflicts in Israel, the Middle East and the rest of the world, the impact of these conflicts on market conditions and the general economic, industry and political conditions in Israel, the U.S. and globally, effect of potential imposed tariff on overall international trade and specifically on Kamada’s ability to continue maintaining expected sales and profit levels in light of such potential tariff, the effect on establishment and timing of business initiatives, Kamada’s ability to leverage new business opportunities and integrate it with its existing product portfolio, unexpected results of clinical and development programs, regulatory approvals and regulatory delays, and other risks detailed in Kamada’s filings with the U.S. Securities and Exchange Commission (the “SEC”) including those discussed in its most recent Annual Report on Form 20-F and in any subsequent reports on Form 6-K, each of which is on file or furnished with the SEC and available at the SEC’s website at www.sec.gov. The forward-looking statements made herein speak only as of the date of this announcement and Kamada undertakes no obligation to update publicly such forward-looking statements to reflect subsequent events or circumstances, except as otherwise required by law.
CONTACTS:
Chaime Orlev
Chief Financial Officer
IR@kamada.com
Brian Ritchie
LifeSci Advisors, LLC
212-915-2578
britchie@LifeSciAdvisors.com
---tables to follow---
CONSOLIDATED STATEMENTS OF FINANCIAL POSITION
| As of September 30, |
As of December 31, |
|||||||||||
| 2025 | 2024 | 2024 | ||||||||||
| Unaudited | ||||||||||||
| Assets | ||||||||||||
| Current Assets | ||||||||||||
| Cash and cash equivalents | $ | 71,997 | $ | 72,001 | $ | 78,435 | ||||||
| Trade receivables, net | 31,379 | 16,295 | 21,547 | |||||||||
| Other accounts receivables | 3,945 | 4,555 | 5,546 | |||||||||
| Inventories | 85,413 | 71,558 | 78,819 | |||||||||
| Total Current Assets | 192,734 | 164,409 | 184,347 | |||||||||
| Non-Current Assets | ||||||||||||
| Property, plant and equipment, net | 38,100 | 33,746 | 36,245 | |||||||||
| Right-of-use assets | 9,189 | 9,854 | 9,617 | |||||||||
| Intangible assets and other long-term assets | 99,186 | 104,728 | 103,226 | |||||||||
| Goodwill | 30,313 | 30,313 | 30,313 | |||||||||
| Contract assets | 7,688 | 8,159 | 8,019 | |||||||||
| Deferred taxes | - | - | 488 | |||||||||
| Total Non-Current Assets | 184,476 | 186,800 | 187,908 | |||||||||
| Total Assets | $ | 377,210 | $ | 351,209 | $ | 372,255 | ||||||
| Liabilities | ||||||||||||
| Current Liabilities | ||||||||||||
| Current maturities of lease liabilities | 1,912 | 1,586 | 1,631 | |||||||||
| Current maturities of other long term liabilities | 10,585 | 9,480 | 10,181 | |||||||||
| Trade payables | 24,875 | 14,786 | 27,735 | |||||||||
| Other accounts payables | 9,443 | 8,104 | 9,671 | |||||||||
| Deferred revenues | 1,022 | 41 | 171 | |||||||||
| Total Current Liabilities | 47,837 | 33,997 | 49,389 | |||||||||
| Non-Current Liabilities | ||||||||||||
| Lease liabilities | 9,558 | 9,574 | 9,431 | |||||||||
| Contingent consideration | 19,730 | 17,630 | 20,646 | |||||||||
| Other long-term liabilities | 32,539 | 34,121 | 32,816 | |||||||||
| Deferred taxes | 1,723 | - | - | |||||||||
| Employee benefit liabilities, net | 591 | 618 | 509 | |||||||||
| Total Non-Current Liabilities | 64,141 | 61,943 | 63,402 | |||||||||
| Shareholder’s Equity | ||||||||||||
| Ordinary shares | 15,077 | 15,024 | 15,028 | |||||||||
| Additional paid in capital net | 268,222 | 266,588 | 266,933 | |||||||||
| Capital reserve due to translation to presentation currency | (3,490 | ) | (3,490 | ) | (3,490 | ) | ||||||
| Capital reserve from hedges | 346 | 16 | 51 | |||||||||
| Capital reserve from share-based payments | 5,339 | 6,394 | 6,316 | |||||||||
| Capital reserve from employee benefits | 374 | 283 | 364 | |||||||||
| Accumulated deficit | (20,636 | ) | (29,546 | ) | (25,738 | ) | ||||||
| Total Shareholder’s Equity | 265,232 | 255,269 | 259,464 | |||||||||
| Total Liabilities and Shareholder’s Equity | $ | 377,210 | $ | 351,209 | $ | 372,255 | ||||||
CONSOLIDATED STATEMENTS OF PROFIT OR LOSS AND OTHER COMPREHENSIVE INCOME
| Nine months period ended | Three months period ended | Year ended | ||||||||||||||||||
| September 30, | September 30, | December 31, | ||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | 2024 | ||||||||||||||||
| Unaudited | Unaudited | |||||||||||||||||||
| Revenues from proprietary products | $ | 117,976 | $ | 110,032 | $ | 39,523 | $ | 37,128 | $ | 141,447 | ||||||||||
| Revenues from distribution | 17,806 | 11,916 | 7,487 | 4,612 | 19,506 | |||||||||||||||
| Total revenues | 135,782 | 121,948 | 47,010 | 41,740 | 160,953 | |||||||||||||||
| Cost of revenues from proprietary products | 61,464 | 59,207 | 20,884 | 20,869 | 73,708 | |||||||||||||||
| Cost of revenues from distribution | 14,878 | 9,805 | 6,364 | 3,637 | 17,278 | |||||||||||||||
| Total cost of revenues | 76,342 | 69,012 | 27,248 | 24,506 | 90,986 | |||||||||||||||
| Gross profit | 59,440 | 52,936 | 19,762 | 17,234 | 69,967 | |||||||||||||||
| Research and development expenses | 10,101 | 12,512 | 2,636 | 3,414 | 15,185 | |||||||||||||||
| Selling and marketing expenses | 13,573 | 13,862 | 4,505 | 4,501 | 18,428 | |||||||||||||||
| General and administrative expenses | 13,084 | 11,578 | 4,819 | 4,014 | 15,702 | |||||||||||||||
| Other expenses (income) | - | 11 | (14 | ) | 11 | 601 | ||||||||||||||
| Operating income | 22,682 | 14,973 | 7,816 | 5,294 | 20,051 | |||||||||||||||
| Financial income | 1,479 | 1,434 | 492 | 646 | 2,118 | |||||||||||||||
| Income (expenses) in respect of currency exchange differences and derivatives instruments, net | (766 | ) | 255 | (43 | ) | (60 | ) | (94 | ) | |||||||||||
| Financial Income (expense) in respect of contingent consideration and other long- term liabilities. | (4,057 | ) | (5,316 | ) | (1,677 | ) | (1,766 | ) | (8,081 | ) | ||||||||||
| Financial expenses | (605 | ) | (471 | ) | (221 | ) | (167 | ) | (660 | ) | ||||||||||
| Income (expense) before tax on income | 18,733 | 10,875 | 6,367 | 3,947 | 13,334 | |||||||||||||||
| Taxes on income | (2,097 | ) | (221 | ) | (1,071 | ) | (84 | ) | 1,128 | |||||||||||
| Net income (loss) | $ | 16,636 | $ | 10,654 | $ | 5,296 | $ | 3,863 | $ | 14,462 | ||||||||||
| Other comprehensive income (loss) : | ||||||||||||||||||||
| Amounts that will be or that have been reclassified to profit or loss when specific conditions are met: | ||||||||||||||||||||
| Gain (loss) from securities measured at fair value through other comprehensive income | ||||||||||||||||||||
| Gain (loss) on cash flow hedges | 770 | (63 | ) | 207 | 32 | (30 | ) | |||||||||||||
| Net amounts transferred to the statement of profit or loss for cash flow hedges | (475 | ) | (61 | ) | (317 | ) | (4 | ) | (59 | ) | ||||||||||
| Items that will not be reclassified to profit or loss in subsequent periods: | ||||||||||||||||||||
| Remeasurement gain (loss) from defined benefit plan | 10 | 8 | - | - | 89 | |||||||||||||||
| Total comprehensive income (loss) | $ | 16,941 | $ | 10,538 | $ | 5,186 | $ | 3,891 | $ | 14,462 | ||||||||||
| Earnings per share attributable to equity holders of the Company: | ||||||||||||||||||||
| Basic net earnings per share | $ | 0.29 | $ | 0.19 | $ | 0.09 | $ | 0.07 | $ | 0.25 | ||||||||||
| Diluted net earnings per share | $ | 0.29 | $ | 0.18 | $ | 0.09 | $ | 0.07 | $ | 0.25 | ||||||||||
CONSOLIDATED STATEMENTS OF CASH FLOWS
| Nine months period Ended | Three months period Ended | Year Ended | ||||||||||||||||||
| September, 30 | September, 30 | December 31, | ||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | 2024 | ||||||||||||||||
| Unaudited | ||||||||||||||||||||
| U.S Dollars In thousands | ||||||||||||||||||||
| Cash Flows from Operating Activities | ||||||||||||||||||||
| Net income | $ | 16,636 | $ | 10,654 | $ | 5,296 | $ | 3,863 | $ | 14,462 | ||||||||||
| Adjustments to reconcile net income to net cash provided by (used in) operating activities: | ||||||||||||||||||||
| Adjustments to the profit or loss items: | ||||||||||||||||||||
| Depreciation and impairment | 11,117 | 9,708 | 3,760 | 3,242 | 13,808 | |||||||||||||||
| Financial expenses, net | 3,949 | 4,098 | 1,449 | 1,347 | 6,717 | |||||||||||||||
| Cost of share-based payment | 387 | 700 | 117 | 224 | 874 | |||||||||||||||
| Taxes on income | 2,097 | 221 | 1,071 | 84 | (1,128 | ) | ||||||||||||||
| Loss (gain) from sale of property and equipment | (8 | ) | 11 | - | 12 | 11 | ||||||||||||||
| Change in employee benefit liabilities, net | 91 | 6 | 17 | 17 | 52 | |||||||||||||||
| 17,633 | 14,744 | 6,414 | 4,926 | 20,334 | ||||||||||||||||
| Changes in asset and liability items: | ||||||||||||||||||||
| Decrease (increase) in trade receivables, net | (9,705 | ) | 3,249 | (1,035 | ) | 10,004 | (1,977 | ) | ||||||||||||
| Decrease in other accounts receivables | 1,666 | 1,452 | 588 | 510 | 593 | |||||||||||||||
| Decrease (increase) in inventories | (6,593 | ) | 16,920 | (3,333 | ) | 7,155 | 9,659 | |||||||||||||
| Decrease in deferred expenses | 331 | 336 | 119 | 97 | 476 | |||||||||||||||
| Increase (decrease) in trade payables | (3,497 | ) | (10,747 | ) | 634 | (5,655 | ) | 1,226 | ||||||||||||
| Increase (decrease) in other accounts payables | (253 | ) | (157 | ) | 630 | 881 | 1,413 | |||||||||||||
| Increase (decrease) in deferred revenues | 851 | (107 | ) | 845 | 14 | 23 | ||||||||||||||
| (17,200 | ) | 10,946 | (1,552 | ) | 13,006 | 11,413 | ||||||||||||||
| Cash received (paid) during the period for: | ||||||||||||||||||||
| Interest paid | (605 | ) | (424 | ) | (221 | ) | (158 | ) | (594 | ) | ||||||||||
| Interest received | 1,479 | 1,434 | 492 | 646 | 2,118 | |||||||||||||||
| Taxes paid | (19 | ) | (158 | ) | (13 | ) | (70 | ) | (139 | ) | ||||||||||
| 855 | 852 | 258 | 418 | 1,385 | ||||||||||||||||
| Net cash provided by operating activities | $ | 17,924 | $ | 37,196 | $ | 10,416 | $ | 22,213 | $ | 47,594 | ||||||||||
CONSOLIDATED STATEMENTS OF CASH FLOWS (continued)
| Nine months period Ended | Three months period Ended | Year Ended | ||||||||||||||||||
| September, 30 | September, 30 | December 31, | ||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | 2024 | ||||||||||||||||
| Unaudited | Audited | |||||||||||||||||||
| U.S Dollars In thousands | ||||||||||||||||||||
| Cash Flows from Investing Activities | ||||||||||||||||||||
| Purchase of property and equipment and intangible assets | $ | (7,071 | ) | $ | (7,816 | ) | $ | (3,589 | ) | $ | (2,124 | ) | $ | (10,740 | ) | |||||
| Proceeds from sale of property and equipment | 8 | 1 | - | - | 1 | |||||||||||||||
| Net cash used in investing activities | (7,063 | ) | (7,815 | ) | (3,589 | ) | (2,124 | ) | (10,739 | ) | ||||||||||
| Cash Flows from Financing Activities | ||||||||||||||||||||
| Proceeds from exercise of share base payments | 49 | 3 | - | 1 | 7 | |||||||||||||||
| Repayment of lease liabilities | (833 | ) | (890 | ) | (415 | ) | (319 | ) | (1,251 | ) | ||||||||||
| Repayment of other long-term liabilities | (4,848 | ) | (12,316 | ) | (339 | ) | (4,468 | ) | (12,667 | ) | ||||||||||
| Dividends Paid | (11,534 | ) | - | - | - | - | ||||||||||||||
| Net cash used in financing activities | (17,166 | ) | (13,203 | ) | (754 | ) | (4,786 | ) | (13,911 | ) | ||||||||||
| Exchange differences on balances of cash and cash equivalent | (133 | ) | 182 | (61 | ) | 151 | (150 | ) | ||||||||||||
| Increase (decrease) in cash and cash equivalents | (6,438 | ) | 16,360 | 6,012 | 15,454 | 22,794 | ||||||||||||||
| Cash and cash equivalents at the beginning of the period | 78,435 | 55,641 | 65,985 | 56,547 | 55,641 | |||||||||||||||
| Cash and cash equivalents at the end of the period | $ | 71,997 | $ | 72,001 | $ | 71,997 | $ | 72,001 | $ | 78,435 | ||||||||||
| Significant non-cash transactions | ||||||||||||||||||||
| Right-of-use asset recognized with corresponding lease liability | $ | 870 | $ | 3,163 | $ | 360 | $ | 2,642 | $ | 3,304 | ||||||||||
| Purchase of property and equipment and Intangible assets | $ | 555 | $ | 1,040 | $ | (475 | ) | $ | 1,040 | $ | 1,955 | |||||||||
NON-IFRS MEASURES
| Nine months period ended | Three months period ended | Year ended | ||||||||||||||||||
| September 30, | September 30, | December 31, | ||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | 2024 | ||||||||||||||||
| U.S Dollars In thousands | ||||||||||||||||||||
| Net income | $ | 16,636 | $ | 10,654 | $ | 5,296 | $ | 3,863 | $ | 14,462 | ||||||||||
| Taxes on income | 2,097 | 221 | 1,071 | 84 | (1,128 | ) | ||||||||||||||
| Financial expense (income), net | 3,949 | 4,098 | 1,449 | 1,347 | 6,717 | |||||||||||||||
| Depreciation and amortization expense | 11,122 | 9,708 | 3,765 | 3,242 | 13,218 | |||||||||||||||
| Non-cash share-based compensation expenses | 387 | 700 | 117 | 224 | 867 | |||||||||||||||
| Adjusted EBITDA | $ | 34,191 | $ | 25,381 | $ | 11,698 | $ | 8,760 | $ | 34,136 | ||||||||||
Exhibit 99.2

November 2025 9M/2025 & Q3/2025 Investors Call NASDAQ: KMDA; TASE: KMDA.TA

2 FORWARD - LOOKING STATEMENT This presentation is not intended to provide investment or medical advice . It should be noted that some products under development described herein have not been found safe or effective by any regulatory agency and are not approved for any use outside of clinical trials . This presentation contains forward - looking statements, which express the current beliefs and expectations of Kamada’s management . Such statements include 2025 financial guidance ; growth strategy and plans for double digit growth ; growth prospects related to CYTOGAM®, GLASSIA®, and the Israeli distribution business segment ; success in identifying and integrating M&A targets for growth ; advancement and future expected revenues driven by our plasma collection operation ; and continued progression of the inhaled AAT clinical study, its benefits and advantages, potential market size, reduction of the study sample to approximately 180 patients, and the plan to conduct an interim futility analysis by the end of 2025 . These statements involve a number of known and unknown risks and uncertainties that could cause Kamada's future results, performance or achievements to differ significantly from the projected results, performances or achievements expressed or implied by such forward - looking statements . Important factors that could cause or contribute to such differences include, but are not limited to, risks relating to Kamada's ability to successfully develop and commercialize its products and product candidates, progress and results of any clinical trials, introduction of competing products, continued market acceptance of Kamada’s commercial products portfolio, impact of geo - political environment in the middle east, impact of any changes in regulation and legislation that could affect the pharmaceutical industry, difficulty in predicting, obtaining or maintaining U . S . Food and Drug Administration, European Medicines Agency and other regulatory authority approvals, restrains related to third parties’ IP rights and changes in the health policies and structures of various countries, success of M&A strategies, environmental risks, changes in the worldwide pharmaceutical industry and other factors that are discussed under the heading “Risk Factors” of Kamada’s 2024 Annual Report on Form 20 - F (filed on March 5 , 2025 ), as well as in Kamada’s recent Forms 6 - K filed with the U . S . Securities and Exchange Commission . This presentation includes certain non - IFRS financial information, which is not intended to be considered in isolation or as a substitute for, or superior to, the financial information prepared and presented in accordance with IFRS . The non - IFRS financial measures may be calculated differently from, and therefore may not be comparable to, similarly titled measures used by other companies . In accordance with the requirement of the SEC regulations a reconciliation of these non - IFRS financial measures to the comparable IFRS measures is included in an appendix to this presentation . Management uses these non - IFRS financial measures for financial and operational decision - making and as a means to evaluate period - to - period comparisons . Management believes that these non - IFRS financial measures provide meaningful supplemental information regarding Kamada’s performance and liquidity . Forward - looking statements speak only as of the date they are made, and Kamada undertakes no obligation to update any forward - looking statement to reflect the impact of circumstances or events that arise after the date the forward - looking statement was made, except as required by applicable law .

3 9 M - 25 CONTINUING THE GROWTH YoY DOUBLE DIGIT REVENUE AND PROFITABILITY INCREASE Paid special cash dividend of $0.20 per share (totaling approximately $11.5M) on April 7, 2025 GROSS PROFIT REVENUE 12% 2024 $52.9 2025 $ 59.4 11% 2024 $121.9 2025 $ 135.8 Adj. EBITDA EPS 35% 2024 $25.4 2025 $ 34.2 61% 2024 $0.18 2025 $0.29

4 6 18 24 34 40 - 44 2021 2022 2023 2024 2025 104 129 142 161 178 - 182 2021 2022 2023 2024 2025 ADJUSTED EBITDA US$M 61% CAGR 2025 represents annual guidance 2025 represents annual guidance ANNUAL DOUBLE - DIGIT GROWTH TRAJECTORY REVENUES US$M 15% CAGR Quarter - End Strong Cash Position of $72.0 Million 9 M $ 136 M ( 75 %) 9 M $ 34 M ( 81 %)

DELIVERING ON OUR COMMITMENTS 5

6 KAMADA ’ S ROADMAP FOR CONTINUED ANNUAL DOUBLE - DIGIT GROWTH Organic Growth Portfolio of 6 FDA - approved products; Over 30 territories; and Distribution portfolio in Israel M&A Transactions Support growth through M&A transactions Plasma Collection Centers Each new center expected to contribute annual revenues of $ 8 M - $ 10 M at peak capacity Inhaled AAT Phase III pivotal clinical study, targeting a market of over $ 2 B

$ 150 M Total U.S HRIG market size, KEDRAB presents double - digit growth YoY Only anti - Rabies IgG product with FDA approved label confirming safety and effectiveness in children KEDRAB /KAMRAB $ 50 M 2024 U.S. Revenues; $ 135 M Minimum sales in the U.S. expected in 2025 - 2027 Only 2 FDA approved products Leading HRIG in Canada, Australia, Israel, Latin America and additional territories A GLOBAL LEADER IN ANTI - RABIES IMMUNE GLOBULIN (HRIG) For Important Safety Information, visit https://kedrab.com/ 7

8 Licensed to Takeda in the USA, Canada, Australia and New Zealand Commencing in 2022 , Takeda is paying Kamada royalties at a rate of 12 % on its net market sales through August 2025 , and 6 % thereafter until 2040 Projected royalties in the range of $ 10 M to $ 20 M per year Outside the Takeda territories, GLASSIA is marketed by Kamada through a network of partners and distributors . Key countries include Argentina, Switzerland, Russia, Israel, and other international markets . Sales in these territories is expected to continue growing, as result of better disease awareness and patients’ diagnosis . GLASSIA $ 17 M 2024 Royality Income; Up 5 % over 2023 LIQUID AAT FOR THE TREATMENT OF AAT DEFICIENCY (AATD) $ 15 M 2024 Glassia sales by Kamada; Up 205 % over 2023

9 CYTOGAM is the only plasma - derived IgG approved in the U.S. and Canada for prophylaxis of CMV disease after Solid Organ Transplantation. CMV is the leading cause for organ rejection post - transplant. CYTOGAM $ 23 M 2024 Revenues; Up 31 % over 2023 CMV IMMUNE GLOBULIN Growth Continued growth expected in the U.S. and Canada markets Launched, in collaboration with multiple KOLs, a post - marketing research program aimed at generating key data in support of the benefits of CYTOGAM in the management of CMV in solid organ transplantation. Initiated the investigator - initiated SHIELD study, conducted by leading experts and KOLs in CMV and organ transplantation, investigating the benefits of CYTOGAM in reducing the risk of late CMV in kidney transplant recipients.

10 DISTRIBUTION SEGMENT GROWTH More than 25 products exclusively licensed from leading international pharmaceutical companies, marketed in the Israeli market EXCLUSIVE DISTRIBUTOR IN ISRAEL FOR LEADING BIOPHARMACEUTICAL COMPANIES EXPANDING THE DISTRIBUTION SEGMENT MODEL TO THE MENA REGION Key areas : plasma - derived, respiratory, rare diseases, infectious diseases, biosimilar portfolio of several product candidates, mainly from Alvotech First biosimilar launched in 2024 and two additional expected to be launched in Israel in the coming months Additional biosimilar products are expected to be launched in Israel over the coming years, at a rate of 1 - 3 products per year Biosimilar portfolio expected to generate annual sales of $ 15 - 20 M within the next five years

11 M&A TRANSACTIONS EXPECT TO SECURE NEW BUSINESS DEVELOPMENT AND M&A TRANSACTIONS DURING 2025 ; LEVERAGING OVERALL FINANCIAL STRENGTH AND COMMERCIAL INFRASTRUCTURE Screening strategic business development opportunities to identify potential acquisition or in - licensing to accelerate long - term growth Focusing on products synergistic to our existing commercial and/or production activities as well as marketing infrastructure Strong financial position, commercial infrastructure and proven successful M&A capabilities
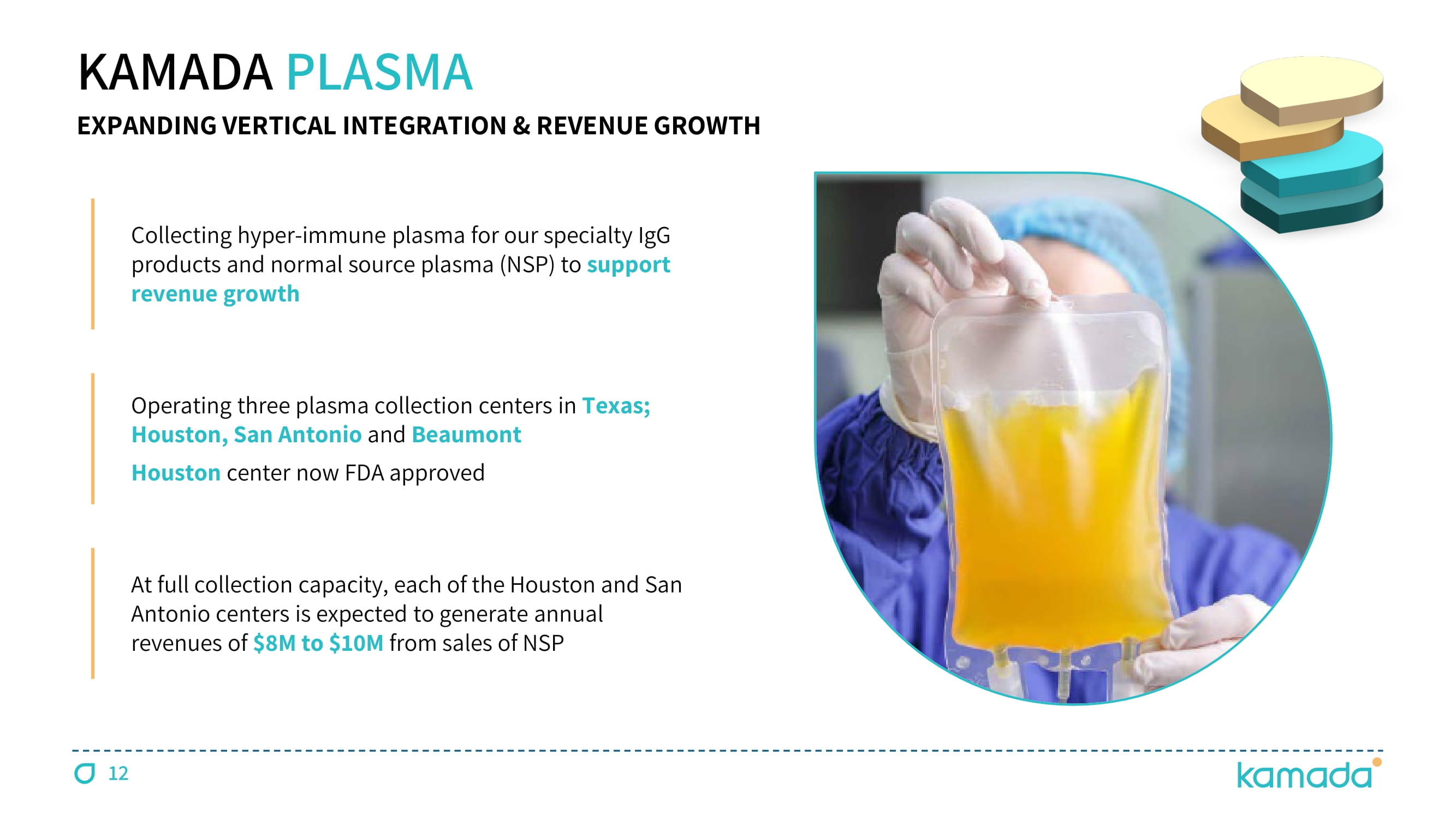
12 KAMADA PLASMA EXPANDING VERTICAL INTEGRATION & REVENUE GROWTH Collecting hyper - immune plasma for our specialty IgG products and normal source plasma (NSP) to support revenue growth Operating three plasma collection centers in Texas; Houston, San Antonio and Beaumont Houston center now FDA approved At full collection capacity, each of the Houston and San Antonio centers is expected to generate annual revenues of $ 8 M to $ 10 M from sales of NSP
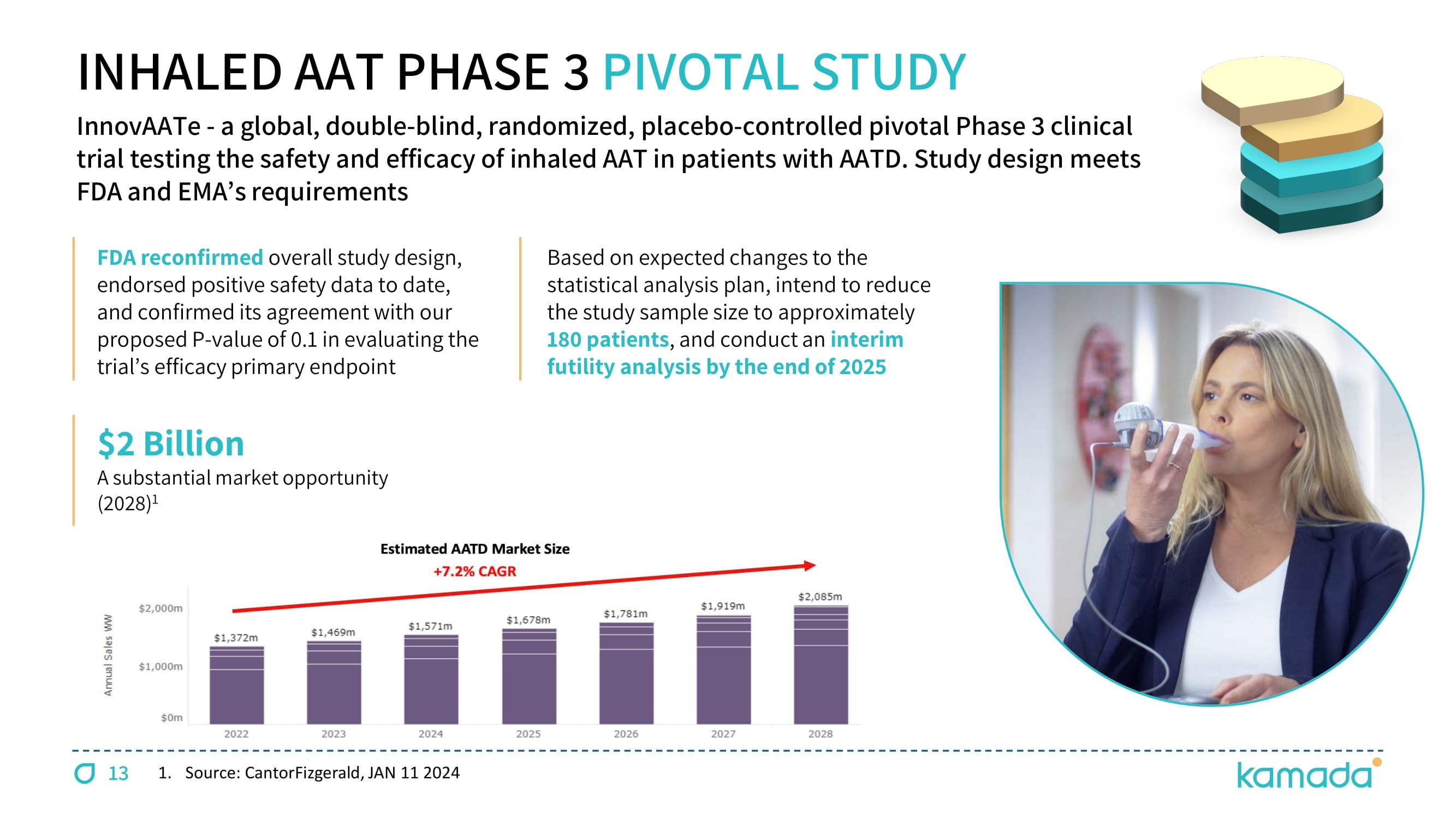
13 $ 2 Billion A substantial market opportunity ( 2028 ) č Based on expected changes to the statistical analysis plan, intend to reduce the study sample size to approximately 180 patients , and conduct an interim futility analysis by the end of 2025 FDA reconfirmed overall study design, endorsed positive safety data to date, and confirmed its agreement with our proposed P - value of 0.1 in evaluating the trial’s efficacy primary endpoint INHALED AAT PHASE 3 PIVOTAL STUDY InnovAATe - a global, double - blind, randomized, placebo - controlled pivotal Phase 3 clinical trial testing the safety and efficacy of inhaled AAT in patients with AATD. Study design meets FDA and EMA ’ s requirements 1. Source: CantorFizgerald, JAN 11 2024
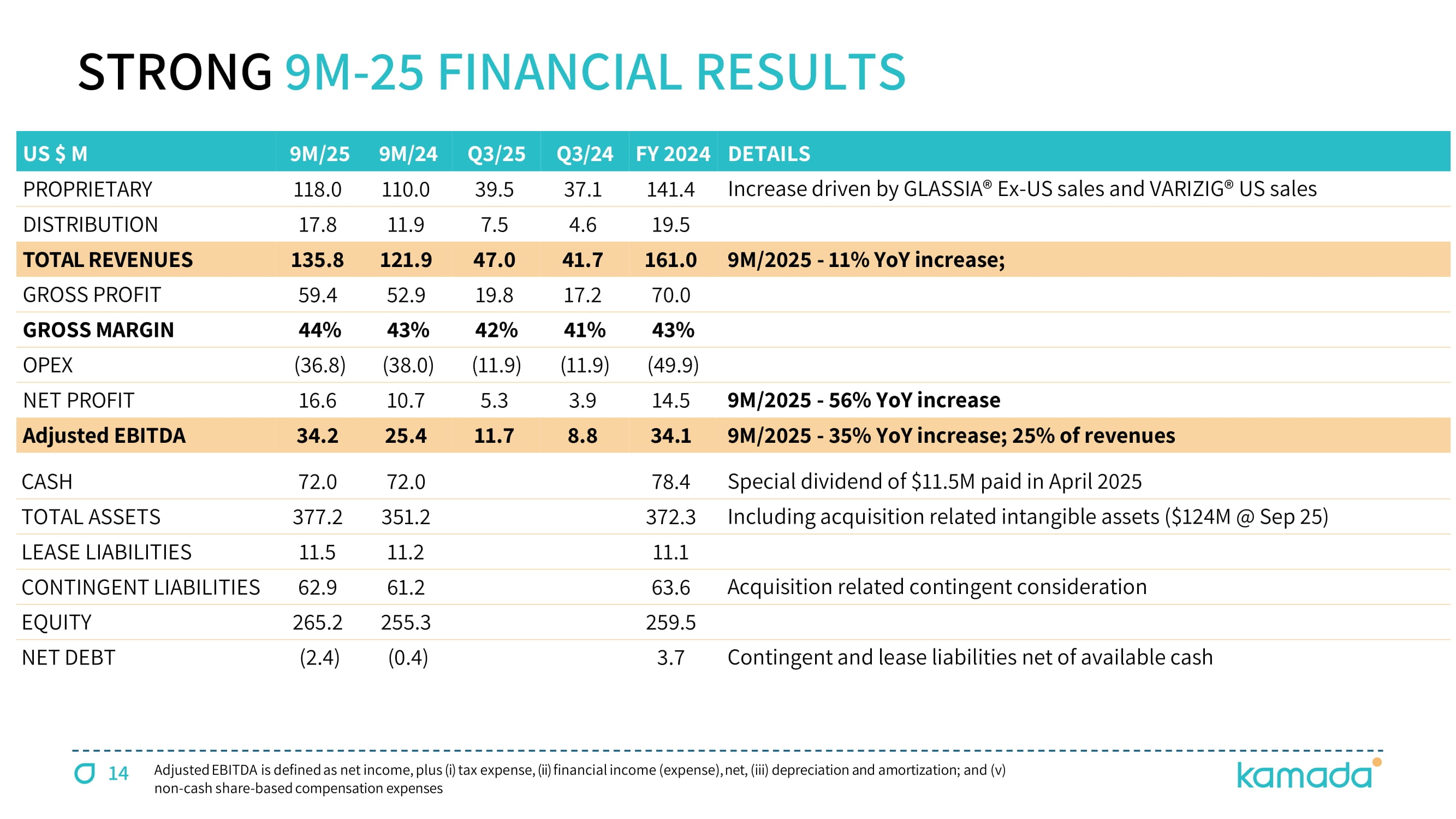
14 Adjusted EBITDA is defined as net income, plus ( i ) tax expense, (ii) financial income (expense), net, (iii) depreciation and amortization ; and (v) non - cash share - based compensation expenses STRONG 9 M - 25 FINANCIAL RESULTS DETAILS FY 2024 Q3/24 Q3/25 9M/24 9M/25 US $ M Increase driven by GLASSIA® Ex - US sales and VARIZIG® US sales 141.4 37.1 39.5 110.0 118.0 PROPRIETARY 19.5 4.6 7.5 11.9 17.8 DISTRIBUTION 9M/2025 - 11% YoY increase; 161.0 41.7 47.0 121.9 135.8 TOTAL REVENUES 70.0 17.2 19.8 52.9 59.4 GROSS PROFIT 43 % 41 % 42 % 43 % 44 % GROSS MARGIN ( 49.9 ) ( 11.9 ) ( 11.9 ) ( 38.0 ) ( 36.8 ) OPEX 9M/2025 - 56% YoY increase 14.5 3.9 5.3 10.7 16.6 NET PROFIT 9M/2025 - 35% YoY increase; 25% of revenues 34.1 8.8 11.7 25.4 34.2 Adjusted EBITDA Special dividend of $11.5M paid in April 2025 78.4 72.0 72.0 CASH Including acquisition related intangible assets ($124M @ Sep 25) 372.3 351.2 377.2 TOTAL ASSETS 11.1 11.2 11.5 LEASE LIABILITIES Acquisition related contingent consideration 63.6 61.2 62.9 CONTINGENT LIABILITIES 259.5 255.3 265.2 EQUITY Contingent and lease liabilities net of available cash 3.7 ( 0.4 ) ( 2.4 ) NET DEBT

KEDRAB® CYTOGAM® HEPGAM B® VARIZIG® WINRHO® GLASSIA® KAMADA - A GLOBAL BIOPHARMACEUTICAL COMPANY 6 FDA - Approved Products 15 % CAGR (from 2021) $ 178 - 182 M 2025 Revenues Guidance $ 40 - 4 Đ M 2025 Adj. EBIDTA Guidance 4 Pillars of Growth A LEADER IN SPECIALTY PLASMA THERAPIES, WITH A PORTFOLIO OF MARKETED PRODUCTS INDICATED FOR RARE AND SERIOUS CONDITIONS $ 72.0 M Cash @ Sep 30 , 2025 15 Organic Growth M&A Transactions Inhaled AAT Pivotal Study Plasma Collection Centers

THANK YOU www.kamada.com NASDAQ: KMDA; TASE: KMDA.TA

17 NON - IFRS MEASURES – ADJUSTED EBITDA Adjusted EBITDA is defined as net income, plus ( i ) tax expense, (ii) financial income (expense), net, (iii) depreciation and amortization ; and (v) non - cash share - based compensation expenses FY 2024 Q3/24 Q3/25 9M/24 9 M/ 25 US $ M 14.5 3.9 5.3 10.7 16.6 NET PROFIT (1.1) 0.1 1.1 0.2 2.1 TAXES ON INCOME 8.1 1.8 1.7 5.3 4.1 REVALUATION OF ACQUISITION RELATED CONTINGENT CONSIDERATION (1.4) ( 0.4 ) ( 0.2 ) ( 1.2 ) ( 0.1 ) OTHER FINANCIAL EXPENSE, NET 7.1 1.8 1.8 5.3 5.3 AMORTIZATION OF ACQUISITION RELATED INTANGIBLE ASSETS 6.2 1.5 2.0 4.4 5.8 OTHER DEPRECIATION AND AMORTIZATION EXPENSES 0.9 0.2 0.1 0.7 0.4 NON - CASH SHARE - BASED COMPENSATION EXPENSES 34.1 8.8 11.7 25.4 34.2 ADJUSTED EBITDA

18 6 FDA - APPROVED SPECIALTY PLASMA PRODUCTS KEDRAB® [Rabies Immune Globulin (Human)] Post exposure prophylaxis of rabies infection CYTOGAM® [Cytomegalovirus Immune Globulin (Human)] Prophylaxis of CMV disease associated with transplants HEPGAM B® [Hepatitis B Immune Globulin (Human)] Prevention of HBV recurrence following liver transplants VARIZIG® [Varicella Zoster Immune Globulin (Human)] Post - exposure prophylaxis of varicella in high - risk patients WINRHO® [Rho(D) Immune Globulin (Human)] Treatment of ITP & suppression of Rh isoimmunization (HDN) KEY FOCUS ON TRANSPLANTS & RARE CONDITIONS For Important Safety Information, visit www.Kamada.com GLASSIA® [Alpha 1 - Proteinase Inhibitor (Human)] Augmentation therapy for Alpha - 1 Antitrypsin Deficiency (AATD)
Exhibit 99.3
KAMADA LTD.
CONSOLIDATED FINANCIAL STATEMENTS
AS OF September 30, 2025
TABLE OF CONTENTS
- - - - - - - - - - -
KAMADA LTD.
CONSOLIDATED STATEMENTS OF FINANCIAL POSITION
| As of September 30, |
As of December 31, |
|||||||||||
| 2025 | 2024 | 2024 | ||||||||||
| Unaudited | ||||||||||||
| Assets | ||||||||||||
| Current Assets | ||||||||||||
| Cash and cash equivalents | $ | 71,997 | $ | 72,001 | $ | 78,435 | ||||||
| Trade receivables, net | 31,379 | 16,295 | 21,547 | |||||||||
| Other accounts receivables | 3,945 | 4,555 | 5,546 | |||||||||
| Inventories | 85,413 | 71,558 | 78,819 | |||||||||
| Total Current Assets | 192,734 | 164,409 | 184,347 | |||||||||
| Non-Current Assets | ||||||||||||
| Property, plant and equipment, net | 38,100 | 33,746 | 36,245 | |||||||||
| Right-of-use assets | 9,189 | 9,854 | 9,617 | |||||||||
| Intangible assets and other long-term assets | 99,186 | 104,728 | 103,226 | |||||||||
| Goodwill | 30,313 | 30,313 | 30,313 | |||||||||
| Contract assets | 7,688 | 8,159 | 8,019 | |||||||||
| Deferred taxes | 488 | |||||||||||
| Total Non-Current Assets | 184,476 | 186,800 | 187,908 | |||||||||
| Total Assets | $ | 377,210 | $ | 351,209 | $ | 372,255 | ||||||
| Liabilities | ||||||||||||
| Current Liabilities | ||||||||||||
| Current maturities of lease liabilities | 1,912 | 1,586 | 1,631 | |||||||||
| Current maturities of other long term liabilities | 10,585 | 9,480 | 10,181 | |||||||||
| Trade payables | 24,875 | 14,786 | 27,735 | |||||||||
| Other accounts payables | 9,443 | 8,104 | 9,671 | |||||||||
| Deferred revenues | 1,022 | 41 | 171 | |||||||||
| Total Current Liabilities | 47,837 | 33,997 | 49,389 | |||||||||
| Non-Current Liabilities | ||||||||||||
| Lease liabilities | 9,558 | 9,574 | 9,431 | |||||||||
| Contingent consideration | 19,730 | 17,630 | 20,646 | |||||||||
| Other long-term liabilities | 32,539 | 34,121 | 32,816 | |||||||||
| Deferred taxes | 1,723 | |||||||||||
| Employee benefit liabilities, net | 591 | 618 | 509 | |||||||||
| Total Non-Current Liabilities | 64,141 | 61,943 | 63,402 | |||||||||
| Shareholder’s Equity | ||||||||||||
| Ordinary shares | 15,077 | 15,024 | 15,028 | |||||||||
| Additional paid in capital net | 268,222 | 266,588 | 266,933 | |||||||||
| Capital reserve due to translation to presentation currency | (3,490 | ) | (3,490 | ) | (3,490 | ) | ||||||
| Capital reserve from hedges | 346 | 16 | 51 | |||||||||
| Capital reserve from share-based payments | 5,339 | 6,394 | 6,316 | |||||||||
| Capital reserve from employee benefits | 374 | 283 | 364 | |||||||||
| Accumulated deficit | (20,636 | ) | (29,546 | ) | (25,738 | ) | ||||||
| Total Shareholder’s Equity | 265,232 | 255,269 | 259,464 | |||||||||
| Total Liabilities and Shareholder’s Equity | $ | 377,210 | $ | 351,209 | $ | 372,255 | ||||||
The accompanying Notes are an integral part of the Consolidated Financial Statements.
KAMADA LTD.
CONSOLIDATED STATEMENTS OF PROFIT OR LOSS AND OTHER COMPREHENSIVE INCOME
| Nine months period ended | Three months period ended | Year ended | ||||||||||||||||||
| September 30, | September 30, | December 31, | ||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | 2024 | ||||||||||||||||
| Unaudited | Unaudited | |||||||||||||||||||
| Revenues from proprietary products | $ | 117,976 | $ | 110,032 | $ | 39,523 | $ | 37,128 | $ | 141,447 | ||||||||||
| Revenues from distribution | 17,806 | 11,916 | 7,487 | 4,612 | 19,506 | |||||||||||||||
| Total revenues | 135,782 | 121,948 | 47,010 | 41,740 | 160,953 | |||||||||||||||
| Cost of revenues from proprietary products | 61,464 | 59,207 | 20,884 | 20,869 | 73,708 | |||||||||||||||
| Cost of revenues from distribution | 14,878 | 9,805 | 6,364 | 3,637 | 17,278 | |||||||||||||||
| Total cost of revenues | 76,342 | 69,012 | 27,248 | 24,506 | 90,986 | |||||||||||||||
| Gross profit | 59,440 | 52,936 | 19,762 | 17,234 | 69,967 | |||||||||||||||
| Research and development expenses | 10,101 | 12,512 | 2,636 | 3,414 | 15,185 | |||||||||||||||
| Selling and marketing expenses | 13,573 | 13,862 | 4,505 | 4,501 | 18,428 | |||||||||||||||
| General and administrative expenses | 13,084 | 11,578 | 4,819 | 4,014 | 15,702 | |||||||||||||||
| Other expenses (income) | 11 | (14 | ) | 11 | 601 | |||||||||||||||
| Operating income | 22,682 | 14,973 | 7,816 | 5,294 | 20,051 | |||||||||||||||
| Financial income | 1,479 | 1,434 | 492 | 646 | 2,118 | |||||||||||||||
| Income (expenses) in respect of currency exchange differences and derivatives instruments, net | (766 | ) | 255 | (43 | ) | (60 | ) | (94 | ) | |||||||||||
| Financial Income (expense) in respect of contingent consideration and other long- term liabilities. | (4,057 | ) | (5,316 | ) | (1,677 | ) | (1,766 | ) | (8,081 | ) | ||||||||||
| Financial expenses | (605 | ) | (471 | ) | (221 | ) | (167 | ) | (660 | ) | ||||||||||
| Income (expense) before tax on income | 18,733 | 10,875 | 6,367 | 3,947 | 13,334 | |||||||||||||||
| Taxes on income | (2,097 | ) | (221 | ) | (1,071 | ) | (84 | ) | 1,128 | |||||||||||
| Net income (loss) | $ | 16,636 | $ | 10,654 | $ | 5,296 | $ | 3,863 | $ | 14,462 | ||||||||||
| Other comprehensive income (loss) : | ||||||||||||||||||||
| Amounts that will be or that have been reclassified to profit or loss when specific conditions are met: | ||||||||||||||||||||
| Gain (loss) from securities measured at fair value through other comprehensive income | ||||||||||||||||||||
| Gain (loss) on cash flow hedges | 770 | (63 | ) | 207 | 32 | (30 | ) | |||||||||||||
| Net amounts transferred to the statement of profit or loss for cash flow hedges | (475 | ) | (61 | ) | (317 | ) | (4 | ) | (59 | ) | ||||||||||
| Items that will not be reclassified to profit or loss in subsequent periods: | ||||||||||||||||||||
| Remeasurement gain (loss) from defined benefit plan | 10 | 8 | 89 | |||||||||||||||||
| Total comprehensive income (loss) | $ | 16,941 | $ | 10,538 | $ | 5,186 | $ | 3,891 | $ | 14,462 | ||||||||||
| Earnings per share attributable to equity holders of the Company: | ||||||||||||||||||||
| Basic net earnings per share | $ | 0.29 | $ | 0.19 | $ | 0.09 | $ | 0.07 | $ | 0.25 | ||||||||||
| Diluted net earnings per share | $ | 0.29 | $ | 0.18 | $ | 0.09 | $ | 0.07 | $ | 0.25 | ||||||||||
The accompanying Notes are an integral part of the Consolidated Financial Statements.
KAMADA LTD.
CONSOLIDATED STATEMENTS OF CHANGES IN EQUITY
| Share capital |
Additional paid in capital |
Capital reserve due to translation to presentation currency |
Capital reserve from hedges |
Capital reserve from sharebased payments |
Capital reserve from employee benefits |
Accumulated deficit |
Total equity |
|||||||||||||||||||||||||
| Unaudited | ||||||||||||||||||||||||||||||||
| In thousands | ||||||||||||||||||||||||||||||||
| Balance as of January 1, 2025 (audited) | $ | 15,028 | $ | 266,933 | $ | (3,490 | ) | $ | 51 | $ | 6,316 | $ | 364 | $ | (25,738 | ) | $ | 259,464 | ||||||||||||||
| Net income | - | - | - | - | - | - | 16,636 | 16,636 | ||||||||||||||||||||||||
| Other comprehensive income (loss) | - | - | - | 295 | - | 10 | - | 305 | ||||||||||||||||||||||||
| Total comprehensive income (loss) | - | - | - | 295 | - | 10 | 16,636 | 16,941 | ||||||||||||||||||||||||
| Exercise and forfeiture of share-based payment into shares | 49 | 1,289 | - | - | (1,364 | ) | - | - | (26 | ) | ||||||||||||||||||||||
| Cost of share-based payment | - | - | - | - | 387 | - | - | 387 | ||||||||||||||||||||||||
| Dividend | - | - | - | - | - | - | (11,534 | ) | (11,534 | ) | ||||||||||||||||||||||
| Balance as of September 30, 2025 | $ | 15,077 | 268,222 | (3,490 | ) | 346 | 5,339 | 374 | (20,636 | ) | 265,232 | |||||||||||||||||||||
| Share capital |
Additional paid in capital |
Capital reserve due to translation to presentation currency |
Capital reserve from hedges |
Capital reserve from sharebased payments |
Capital reserve from employee benefits |
Accumulated deficit |
Total equity |
|||||||||||||||||||||||||
| Unaudited | ||||||||||||||||||||||||||||||||
| In thousands | ||||||||||||||||||||||||||||||||
| Balance as of January 1, 2024 (audited) | $ | 15,021 | $ | 265,848 | $ | (3,490 | ) | $ | 140 | $ | 6,427 | $ | 275 | $ | (40,200 | ) | $ | 244,021 | ||||||||||||||
| Net income | 10,654 | 10,654 | ||||||||||||||||||||||||||||||
| Other comprehensive income (loss) | (124 | ) | 8 | (116 | ) | |||||||||||||||||||||||||||
| Total comprehensive income (loss) | (124 | ) | 8 | 10,654 | 10,538 | |||||||||||||||||||||||||||
| Exercise and forfeiture of share-based payment into shares | 3 | 740 | (740 | ) | 3 | |||||||||||||||||||||||||||
| Cost of share-based payment | 707 | 707 | ||||||||||||||||||||||||||||||
| Balance as of September 30, 2024 | $ | 15,024 | 266,588 | (3,490 | ) | 16 | 6,394 | 283 | (29,546 | ) | 255,269 | |||||||||||||||||||||
The accompanying Notes are an integral part of the Consolidated Financial Statements.
KAMADA LTD.
CONSOLIDATED STATEMENTS OF CHANGES IN EQUITY
| Share capital |
Additional paid in capital |
Capital reserve due to translation to presentation currency |
Capital reserve from hedges |
Capital reserve from sharebased payments |
Capital reserve from employee benefits |
Accumulated deficit |
Total equity |
|||||||||||||||||||||||||
| Unaudited | ||||||||||||||||||||||||||||||||
| In thousands | ||||||||||||||||||||||||||||||||
| Balance as of July 1, 2025 | $ | 15,077 | $ | 268,243 | $ | (3,490 | ) | $ | 456 | $ | 5,226 | $ | 374 | $ | (25,932 | ) | $ | 259,954 | ||||||||||||||
| Net income | 5,296 | 5,296 | ||||||||||||||||||||||||||||||
| Other comprehensive income (loss) | (110 | ) | (110 | ) | ||||||||||||||||||||||||||||
| Total comprehensive income (loss) | (110 | ) | 5,296 | 5,186 | ||||||||||||||||||||||||||||
| Exercise and forfeiture of share-based payment into shares | (21 | ) | (4 | ) | (25 | ) | ||||||||||||||||||||||||||
| Cost of share-based payment | 117 | 117 | ||||||||||||||||||||||||||||||
| Balance as of September 30, 2025 | $ | 15,077 | 268,222 | (3,490 | ) | 346 | 5,339 | 374 | (20,636 | ) | 265,232 | |||||||||||||||||||||
| Share capital |
Additional paid in capital |
Capital reserve due to translation to presentation currency |
Capital reserve from hedges |
Capital reserve from sharebased payments |
Capital reserve from employee benefits |
Accumulated deficit |
Total equity |
|||||||||||||||||||||||||
| Unaudited | ||||||||||||||||||||||||||||||||
| In thousands | ||||||||||||||||||||||||||||||||
| Balance as of July 1, 2024 | $ | 15,023 | $ | 266,313 | $ | (3,490 | ) | $ | (12 | ) | $ | 6,444 | $ | 283 | $ | (33,409 | ) | $ | 251,152 | |||||||||||||
| Net income | 3,863 | 3,863 | ||||||||||||||||||||||||||||||
| Other comprehensive income (loss) | 28 | 28 | ||||||||||||||||||||||||||||||
| Total comprehensive income (loss) | 28 | 3,863 | 3,891 | |||||||||||||||||||||||||||||
| Exercise and forfeiture of share-based payment into shares | 1 | 275 | (275 | ) | 1 | |||||||||||||||||||||||||||
| Cost of share-based payment | 225 | 225 | ||||||||||||||||||||||||||||||
| Balance as of September 30, 2024 | $ | 15,024 | $ | 266,588 | $ | (3,490 | ) | $ | 16 | $ | 6,394 | $ | 283 | $ | (29,546 | ) | $ | 255,269 | ||||||||||||||
KAMADA LTD.
CONSOLIDATED STATEMENTS OF CHANGES IN EQUITY
| Share | Additional paid in |
Capital reserve due to translation to presentation |
Capital reserve from |
Capital reserve from share based |
Capital reserve from employee |
Accumulated | Total | |||||||||||||||||||||||||
| capital | capital | currency | hedges | payments | benefits | deficit | equity | |||||||||||||||||||||||||
| U.S. Dollars in Thousands | ||||||||||||||||||||||||||||||||
| Balance as of January 1, 2024 (audited) | $ | 15,021 | $ | 265,848 | $ | (3,490 | ) | $ | 140 | $ | 6,427 | $ | 275 | $ | (40,200 | ) | $ | 244,021 | ||||||||||||||
| Net income | 14,462 | 14,462 | ||||||||||||||||||||||||||||||
| Other comprehensive income (loss), net of tax) | (89 | ) | 89 | |||||||||||||||||||||||||||||
| Total comprehensive income (loss) | (89 | ) | 89 | 14,462 | 14,462 | |||||||||||||||||||||||||||
| Exercise and forfeiture of share-based payment into shares | 7 | 985 | (985 | ) | 7 | |||||||||||||||||||||||||||
| Cost of share-based payment | 874 | 874 | ||||||||||||||||||||||||||||||
| Income tax impact associated with issuance of shares | 100 | 100 | ||||||||||||||||||||||||||||||
| Balance as of December 31, 2024 | $ | 15,028 | $ | 266,933 | $ | (3,490 | ) | $ | 51 | $ | 6,316 | $ | 364 | $ | (25,738 | ) | $ | 259,464 | ||||||||||||||
The accompanying Notes are an integral part of the Consolidated Financial Statements.
KAMADA LTD.
CONSOLIDATED STATEMENTS OF CASH FLOWS
| Nine months period Ended | Three months period Ended | Year Ended | ||||||||||||||||||
| September, 30 | September, 30 | December 31, | ||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | 2024 | ||||||||||||||||
| Unaudited | ||||||||||||||||||||
| U.S Dollars In thousands | ||||||||||||||||||||
| Cash Flows from Operating Activities | ||||||||||||||||||||
| Net income | $ | 16,636 | $ | 10,654 | $ | 5,296 | $ | 3,863 | $ | 14,462 | ||||||||||
| Adjustments to reconcile net income to net cash provided by (used in) operating activities: | ||||||||||||||||||||
| Adjustments to the profit or loss items: | ||||||||||||||||||||
| Depreciation and impairment | 11,117 | 9,708 | 3,760 | 3,242 | 13,808 | |||||||||||||||
| Financial expenses, net | 3,949 | 4,098 | 1,449 | 1,347 | 6,717 | |||||||||||||||
| Cost of share-based payment | 387 | 700 | 117 | 224 | 874 | |||||||||||||||
| Taxes on income | 2,097 | 221 | 1,071 | 84 | (1,128 | ) | ||||||||||||||
| Loss (gain) from sale of property and equipment | (8 | ) | 11 | 12 | 11 | |||||||||||||||
| Change in employee benefit liabilities, net | 91 | 6 | 17 | 17 | 52 | |||||||||||||||
| 17,633 | 14,744 | 6,414 | 4,926 | 20,334 | ||||||||||||||||
| Changes in asset and liability items: | ||||||||||||||||||||
| Decrease (increase) in trade receivables, net | (9,705 | ) | 3,249 | (1,035 | ) | 10,004 | (1,977 | ) | ||||||||||||
| Decrease in other accounts receivables | 1,666 | 1,452 | 588 | 510 | 593 | |||||||||||||||
| Decrease (increase) in inventories | (6,593 | ) | 16,920 | (3,333 | ) | 7,155 | 9,659 | |||||||||||||
| Decrease in deferred expenses | 331 | 336 | 119 | 97 | 476 | |||||||||||||||
| Increase (decrease) in trade payables | (3,497 | ) | (10,747 | ) | 634 | (5,655 | ) | 1,226 | ||||||||||||
| Increase (decrease) in other accounts payables | (253 | ) | (157 | ) | 630 | 881 | 1,413 | |||||||||||||
| Increase (decrease) in deferred revenues | 851 | (107 | ) | 845 | 14 | 23 | ||||||||||||||
| (17,200 | ) | 10,946 | (1,552 | ) | 13,006 | 11,413 | ||||||||||||||
| Cash received (paid) during the period for: | ||||||||||||||||||||
| Interest paid | (605 | ) | (424 | ) | (221 | ) | (158 | ) | (594 | ) | ||||||||||
| Interest received | 1,479 | 1,434 | 492 | 646 | 2,118 | |||||||||||||||
| Taxes paid | (19 | ) | (158 | ) | (13 | ) | (70 | ) | (139 | ) | ||||||||||
| 855 | 852 | 258 | 418 | 1,385 | ||||||||||||||||
| Net cash provided by operating activities | $ | 17,924 | $ | 37,196 | $ | 10,416 | $ | 22,213 | $ | 47,594 | ||||||||||
KAMADA LTD.
CONSOLIDATED STATEMENTS OF CASH FLOWS (continued)
| Nine months period Ended | Three months period Ended | Year Ended | ||||||||||||||||||
| September, 30 | September, 30 | December 31, | ||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | 2024 | ||||||||||||||||
| Unaudited | Audited | |||||||||||||||||||
| U.S Dollars In thousands | ||||||||||||||||||||
| Cash Flows from Investing Activities | ||||||||||||||||||||
| Purchase of property and equipment and intangible assets | $ | (7,071 | ) | $ | (7,816 | ) | $ | (3,589 | ) | $ | (2,124 | ) | $ | (10,740 | ) | |||||
| Proceeds from sale of property and equipment | 8 | 1 | 1 | |||||||||||||||||
| Net cash used in investing activities | (7,063 | ) | (7,815 | ) | (3,589 | ) | (2,124 | ) | (10,739 | ) | ||||||||||
| Cash Flows from Financing Activities | ||||||||||||||||||||
| Proceeds from exercise of share base payments | 49 | 3 | 1 | 7 | ||||||||||||||||
| Repayment of lease liabilities | (833 | ) | (890 | ) | (415 | ) | (319 | ) | (1,251 | ) | ||||||||||
| Repayment of other long-term liabilities | (4,848 | ) | (12,316 | ) | (339 | ) | (4,468 | ) | (12,667 | ) | ||||||||||
| Dividends Paid | (11,534 | ) | ||||||||||||||||||
| Net cash used in financing activities | (17,166 | ) | (13,203 | ) | (754 | ) | (4,786 | ) | (13,911 | ) | ||||||||||
| Exchange differences on balances of cash and cash equivalent | (133 | ) | 182 | (61 | ) | 151 | (150 | ) | ||||||||||||
| Increase (decrease) in cash and cash equivalents | (6,438 | ) | 16,360 | 6,012 | 15,454 | 22,794 | ||||||||||||||
| Cash and cash equivalents at the beginning of the period | 78,435 | 55,641 | 65,985 | 56,547 | 55,641 | |||||||||||||||
| Cash and cash equivalents at the end of the period | $ | 71,997 | $ | 72,001 | $ | 71,997 | $ | 72,001 | $ | 78,435 | ||||||||||
| Significant non-cash transactions | ||||||||||||||||||||
| Right-of-use asset recognized with corresponding lease liability | $ | 870 | $ | 3,163 | $ | 360 | $ | 2,642 | $ | 3,304 | ||||||||||
| Purchase of property and equipment and Intangible assets | $ | 555 | $ | 1,040 | $ | (475 | ) | $ | 1,040 | $ | 1,955 | |||||||||
The accompanying Notes are an integral part of the Consolidated Financial Statements.
KAMADA LTD.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Note 1:- General
General description of the Company and its activity
Kamada Ltd (the “Company”) is a global biopharmaceutical company with a portfolio of marketed products indicated for rare and serious conditions and a leader in the specialty plasma-derived therapies field. The Company’s strategy is focused on driving profitable growth through four primary growth pillars: First, organic growth from its commercial activities, including continued investment in the commercialization and life cycle management of its proprietary products, which include six FDA-approved specialty plasma-derived products: KEDRAB®, CYTOGAM®, GLASSIA®, WINRHO SDF®, VARIZIG® and HEPAGAM B®, as well as KAMRAB®, KAMRHO (D)® and two types of equine-based anti-snake venom products, and the products in the Distribution segment portfolio, mainly through the launch of several biosimilar products in Israel. Second, the Company aims to secure significant new business development, in-licensing, collaboration and/or merger and acquisition opportunities, which are anticipated to enhance the Company’s marketed products portfolio and leverage its financial strength and existing commercial infrastructure to drive long-term growth. Third, the Company is expanding its plasma collection operations to support revenue growth through the sale of normal source plasma to other plasma-derived manufacturers, and to support its increasing demand for hyper-immune plasma. The Company currently owns three operating plasma collection centers in the United States, in Beaumont Texas, Houston Texas, and San Antonio, Texas. Lastly, the Company is leveraging its manufacturing, research and development expertise to advance the development and commercialization of additional product candidates, targeting areas of significant unmet medical need, with its lead product candidate Inhaled AAT, for which the Company is continuing to progress the InnovAATe clinical trial, a randomized, double-blind, placebo-controlled, pivotal Phase 3 trial.
In November 2021, the Company acquired, pursuant to an Asset Purchase Agreement, CYTOGAM, WINRHO SDF, VARIZIG and HEPGAM B from Saol Therapeutics Ltd. The acquisition of this portfolio furthered the Company’s core objective to become a fully integrated specialty plasma company with strong commercial capabilities in the U.S. market, as well as to expand to new markets, mainly in the Middle East/North Africa region, and to broaden the Company’s portfolio offering in existing markets. The Company’s wholly owned U.S. subsidiary, Kamada Inc., is responsible for the commercialization of the four products in the U.S. market, including direct sales to wholesalers and local distributers.
In accordance with an agreement with Takeda Pharmaceuticals Company Limited (“Takeda”), starting from the first quarter of 2022, Takeda pays the Company royalties on sales of GLASSIA manufactured by Takeda in the United States and, commencing in 2024, in Canada, at a rate of 12% on net sales through August 2025 and at a rate of 6% thereafter until 2040, with a minimum of $5 million annually for each year from 2022 to 2040. The Company will also be entitled to royalty income on sales of GLASSIA by Takeda in Australia and New Zealand, to the extent that GLASSIA will be approved, and sales will be generated in these markets by Takeda in the future.
The Company’s ordinary shares are listed for trading on the Tel Aviv Stock Exchange and the NASDAQ Global Select Market.
FIMI Opportunity Funds (“FIMI”), the leading private equity firm in Israel beneficially owns approximately 38% of the Company’s outstanding ordinary shares and is a controlling shareholder of the Company; within the meaning of the Israeli Companies Law, 1999.
The Company’s activity is divided into two operating segments:
| Proprietary Products | Manufacturing, sales and distribution of plasma-derived protein therapeutics. | |
| Distribution | Distribute imported drug products in Israel, which are manufactured by third parties. |
The Company has four wholly-owned subsidiaries – Kamada Inc., Kamada Plasma LLC (wholly owned by Kamada Inc.), KI Biopharma LLC and Kamada Ireland Limited. In addition, the Company owns 74% of Kamada Assets Ltd. (“Kamada Assets”).
KAMADA LTD.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Note 2:- Material Accounting Policies
| a. | Basis of preparation of the interim consolidated financial statements: |
The interim consolidated financial statements have been prepared in accordance with generally accepted accounting principles for the preparation of financial statements for interim periods, as prescribed in IAS 34, “Interim Financial Reporting”.
| b. | Forthcoming requirements |
| Presentation and Disclosure in Financial Statements – IFRS 18 |
In April 2024, the IASB issued IFRS 18 Presentation and Disclosure in Financial Statements (“IFRS 18”) which replaces IAS 1 Presentation of Financial Statements. IFRS 18 requires an entity to classify all income and expenses within its statement of profit and loss into one of five categories: operating; investing; financing; income taxes; and discontinued operations. The first three categories are new. These categories are complemented by the requirement to present subtotals for “operating profit or loss,” profit or loss before financing income and taxes” and “profit or loss” IFRS 18, and the amendments to the other standards, is effective for reporting periods beginning on or after January 1, 2027, but earlier application is permitted.
The Company is currently assessing the impact of the Standard on its financial statements. As of September 30, 2025, the Company does not have impact on its financial statement.
Note 3:- Significant events in the reporting period
On March 5, 2025, the Company announced that its Board of Directors had declared a special cash dividend of $0.20 (NIS 0.73) per share on the Company’s common stock, amounting to approximately $11.5 million in total. The dividend was paid on April 7, 2025, to shareholders of record as of the close of business on March 17, 2025.
Note 4:- Operating Segments
| a. | General: |
The company has two operating segments, as follows:
| Proprietary Products | - | Development, manufacturing, sales and distribution of proprietary plasma-derived protein therapeutics. |
| Distribution | - | Distribute imported drug products in Israel, which are manufactured by third parties. |
KAMADA LTD.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Note 4:- Operating Segments (cont.)
| b. | Reporting on operating segments: |
| Nine months period ended September 30, 2025 |
||||||||||||
| Proprietary Products |
Distribution | Total | ||||||||||
| U.S Dollars in thousands | ||||||||||||
| Unaudited | ||||||||||||
| Revenues | $ | 117,976 | $ | 17,806 | $ | 135,782 | ||||||
| Gross profit | $ | 56,512 | $ | 2,928 | $ | 59,440 | ||||||
| Unallocated corporate expenses | (36,758 | ) | ||||||||||
| Finance expenses, net | (3,949 | ) | ||||||||||
| Income before taxes on income | $ | 18,733 | ||||||||||
| Nine months period ended September 30, 2024 |
||||||||||||
| Proprietary Products |
Distribution | Total | ||||||||||
| U.S Dollars in thousands | ||||||||||||
| Unaudited | ||||||||||||
| Revenues | $ | 110,032 | $ | 11,916 | $ | 121,948 | ||||||
| Gross profit | $ | 50,825 | $ | 2,111 | $ | 52,936 | ||||||
| Unallocated corporate expenses | (37,963 | ) | ||||||||||
| Finance expenses, net | (4,098 | ) | ||||||||||
| Income before taxes on income | $ | 10,875 | ||||||||||
| Three months period ended September 30, 2025 |
||||||||||||
| Proprietary Products |
Distribution | Total | ||||||||||
| U.S Dollars in thousands | ||||||||||||
| Unaudited | ||||||||||||
| Revenues | $ | 39,523 | $ | 7,487 | $ | 47,010 | ||||||
| Gross profit | $ | 18,639 | $ | 1,123 | $ | 19,762 | ||||||
| Unallocated corporate expenses | (11,946 | ) | ||||||||||
| Finance expenses, net | (1,449 | ) | ||||||||||
| Income before taxes on income | $ | 6,367 | ||||||||||
| Three months period ended September 30, 2024 |
||||||||||||
| Proprietary Products |
Distribution | Total | ||||||||||
| U.S Dollars in thousands | ||||||||||||
| Unaudited | ||||||||||||
| Revenues | $ | 37,128 | $ | 4,612 | $ | 41,740 | ||||||
| Gross profit | $ | 16,259 | $ | 975 | $ | 17,234 | ||||||
| Unallocated corporate expenses | (11,940 | ) | ||||||||||
| Finance expenses, net | (1,347 | ) | ||||||||||
| Income before taxes on income | $ | 3,947 | ||||||||||
KAMADA LTD.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Note 4:- Operating Segments (cont.)
| b. | Reporting on operating segments: |
| Year Ended December 31, 2024 | ||||||||||||
| Proprietary Products |
Distribution | Total | ||||||||||
| U.S Dollars in thousands | ||||||||||||
| Audited | ||||||||||||
| Revenues | $ | 141,447 | $ | 19,506 | $ | 160,953 | ||||||
| Gross profit | $ | 67,739 | $ | 2,228 | $ | 69,967 | ||||||
| Unallocated corporate expenses | (49,916 | ) | ||||||||||
| Finance expenses, net | (6,717 | ) | ||||||||||
| Income before taxes on income | $ | 13,334 | ||||||||||
| c. | Reporting on operating segments by geographic region: |
| Nine months period ended September 30, 2025 |
||||||||||||
| Proprietary Products |
Distribution | Total | ||||||||||
| U.S Dollars in thousands | ||||||||||||
| Unaudited | ||||||||||||
| Geographical markets | ||||||||||||
| U.S.A | $ | 79,230 | $ | $ | 79,230 | |||||||
| Israel | 4,746 | 17,806 | 22,552 | |||||||||
| Latin America | 16,324 | 16,324 | ||||||||||
| Canada | 7,982 | 7,982 | ||||||||||
| Europe | 6,310 | 6,310 | ||||||||||
| Asia | 3,362 | 3,362 | ||||||||||
| Others | 22 | 22 | ||||||||||
| $ | 117,976 | $ | 17,806 | $ | 135,782 | |||||||
| c. | Reporting on operating segments by geographic region: |
| Nine months period ended September 30, 2024 |
||||||||||||
| Proprietary Products |
Distribution | Total | ||||||||||
| U.S Dollars in thousands | ||||||||||||
| Unaudited | ||||||||||||
| Geographical markets | ||||||||||||
| U.S.A | $ | 84,779 | $ | $ | 84,779 | |||||||
| Israel | 4,701 | 11,916 | 16,617 | |||||||||
| Canada | 7,873 | 7,873 | ||||||||||
| Latin America | 7,588 | 7,588 | ||||||||||
| Europe | 3,220 | 3,220 | ||||||||||
| Asia | 1,837 | 1,837 | ||||||||||
| Others | 34 | 34 | ||||||||||
| $ | 110,032 | $ | 11,916 | $ | 121,948 | |||||||
KAMADA LTD.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Note 4:- Operating Segments (cont.)
| Three months period ended September 30, 2025 |
||||||||||||
| Proprietary Products |
Distribution | Total | ||||||||||
| U.S Dollars in thousands | ||||||||||||
| Unaudited | ||||||||||||
| Geographical markets | ||||||||||||
| U.S.A | $ | 23,736 | $ | $ | 23,736 | |||||||
| Israel | 1,710 | 7,487 | 9,197 | |||||||||
| Latin America | 6,443 | 6,443 | ||||||||||
| Europe | 3,858 | 3,858 | ||||||||||
| Canada | 2,386 | 2,386 | ||||||||||
| Asia | 1,390 | 1,390 | ||||||||||
| Others | ||||||||||||
| $ | 39,523 | $ | 7,487 | $ | 47,010 | |||||||
| Three months period ended September 30, 2024 |
||||||||||||
| Proprietary Products |
Distribution | Total | ||||||||||
| U.S Dollars in thousands | ||||||||||||
| Unaudited | ||||||||||||
| Geographical markets | ||||||||||||
| U.S.A | $ | 29,610 | $ | $ | 29,610 | |||||||
| Israel | 1,144 | 4,612 | 5,756 | |||||||||
| Latin America | 2,353 | 2,353 | ||||||||||
| Canada | 2,108 | 2,108 | ||||||||||
| Europe | 1,542 | 1,542 | ||||||||||
| Asia | 337 | 337 | ||||||||||
| Others | 34 | 34 | ||||||||||
| $ | 37,128 | $ | 4,612 | $ | 41,740 | |||||||
| Year ended December 31, 2024 | ||||||||||||
| Proprietary Products |
Distribution | Total | ||||||||||
| U.S Dollars in thousands | ||||||||||||
| Audited | ||||||||||||
| Geographical markets | ||||||||||||
| U.S.A | $ | 100,504 | $ | $ | 100,504 | |||||||
| Israel | 5,506 | 19,506 | 25,012 | |||||||||
| Canada | 18,606 | 18,606 | ||||||||||
| Europe | 9,457 | 9,457 | ||||||||||
| Latin America | 4,936 | 4,936 | ||||||||||
| Asia | 2,376 | 2,376 | ||||||||||
| Others | 62 | 62 | ||||||||||
| $ | 141,447 | $ | 19,506 | $ | 160,953 | |||||||
KAMADA LTD.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Note 5:- Financial Instruments
| a. | Classification of financial instruments by fair value hierarchy |
Financial assets (liabilities) measured at fair value
| Level 1 | Level 2 | Level 3 | ||||||||||
| U.S Dollars in thousands | ||||||||||||
| September 30, 2025 | ||||||||||||
| Derivatives instruments | $ | $ | 363 | $ | ||||||||
| Contingent consideration | (22,571 | ) | ||||||||||
| September 30, 2024 | ||||||||||||
| Derivatives instruments | 27 | |||||||||||
| Contingent consideration | $ | $ | $ | (20,472 | ) | |||||||
| December 31, 2024 | ||||||||||||
| Derivatives instruments | $ | $ | 49 | $ | ||||||||
| Contingent consideration | $ | $ | $ | (23,566 | ) | |||||||
During the Nine months ended on September 30, 2023 there were no transfers due to the fair value measurement of any financial instrument from Level 1 to Level 2, and furthermore, there were no transfers to or from Level 3 due to the fair value measurement of any financial instrument.
Note 6:- Contingent Liabilities and Commitments
In connection with a Statement of Claim filed on June 13, 2023 against the Company by a third-party previously engaged to distribute the Company’s propriety products in Russia and Ukraine (the “Distributor”), with the tribunal of first instance in Geneva, on April 22, 2025, the tribunal dismissed the Distributer’s action and declared it inadmissible due to a lack of jurisdiction in Switzerland. On May 27, 2025, the Distributor challenged the decision rendered by the tribunal of first instance, claiming that the tribunal of first instance wrongly declined jurisdiction. The allegations raised by the Distributer in this appeal are largely reiterations of what it argued in front of the tribunal of first instance. The Company plans to submit its response to the appeal by mid-September 2025. Once that is submitted, the Court of Appeal will decide whether a second exchange of briefs is necessary before ruling on the Distributor’s appeal. For more information about this claim please refer to note 17h to the Company’s 2024 annual financial statements included in the 2024 form 20-F filed with the Securities and Exchange Commission on March 5, 2025.
Note 7:- Subsequent events
On October 22, 2025, the Company’s Board of Directors approved the grant of 1,590,000 options to purchase ordinary shares of the Company, under the 2011 Plan and the US Appendix.
Under the Israeli Share Option Plan:
1,324,000 options to purchase ordinary shares of the Company were granted at an exercise price of NIS 23.75 (USD 7.20) per share. The fair value of these options, as estimated on the grant date, was $3,249 thousands. The grant included 400 thousand options awarded to the Chief Executive Officer, pursuant to approval by the Annual General Meeting.
Under the US Appendix:
266,000 options to purchase ordinary shares of the Company were granted at an exercise price of USD 7.14 per share. The fair value of these options, as estimated on the grant date, was $653 thousands.
13