
First Quarter 2026 Financial Results and Business Update April 30, 2026 Exhibit 99.2

Forward-looking statements disclosure This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Words such as "may," "will," "expect," "plan," "anticipate" and similar expressions (as well as other words or expressions referencing future events, progress, timing or circumstances) are intended to identify forward-looking statements. All statements other than statements of historical facts contained in this presentation, including statements regarding future operations, financial results and the financial condition of Syndax Pharmaceuticals, Inc. (“Syndax” or the “Company”), including financial position, strategy and plans, the progress, timing, clinical development and scope of clinical trials and the reporting of clinical data for Syndax’s product candidates, the progress of regulatory submissions and approvals and subsequent commercialization and the potential use of Syndax’s product candidates to treat various cancer indications and fibrotic diseases, and Syndax’s expectations for liquidity and future operations, are forward-looking statements. Many factors may cause differences between current expectations and actual results, including unexpected safety or efficacy data observed during preclinical studies or clinical trials, clinical site activation rates or clinical trial enrollment rates that are lower than expected; changes in expected or existing competition; the impact of macroeconomic conditions (the Russia-Ukraine war, inflation, among others) on Syndax’s business and that of the third parties on which Syndax depends, including delaying or otherwise disrupting Syndax’s clinical trials and preclinical studies, manufacturing and supply chain, or impairing employee productivity; failure of our collaborators to support or advance collaborations or product candidates and unexpected litigation or other disputes. Moreover, Syndax operates in a very competitive and rapidly changing environment. Other factors that may cause our actual results to differ from current expectations are discussed in Syndax’s filings with the U.S. Securities and Exchange Commission, including the “Risk Factors” sections contained therein. New risks emerge from time to time. It is not possible for Syndax’s management to predict all risks, nor can Syndax assess the impact of all factors on its business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statement. In light of these risks, uncertainties and assumptions, the forward-looking events and circumstances discussed in this presentation may not occur and actual results could differ materially and adversely from those anticipated or implied. Except as required by law, neither Syndax nor any other person assumes responsibility for the accuracy and completeness of the forward- looking statements. Syndax undertakes no obligation to update publicly any forward-looking statements for any reason after the date of this presentation to conform these statements to actual results or to changes in Syndax’s expectations.

$49M Revuforj net revenue in 1Q26, highlighting leadership in menin inhibition $55M Niktimvo net revenue in 1Q26, resulting in $16M in collaboration revenue $65M in total revenue to Syndax in 1Q26, up 224% vs. 1Q25 Positioned to be 1st to frontline (1L) AML with a menin inhibitor Major presence planned at upcoming medical meetings with new real-world, 1L, R/R, and post-HSCT data expected in 2Q26 and 2H26 Topline data from Phase 2 trials of axatilimab in IPF and newly diagnosed cGVHD anticipated in 4Q26 Growing product revenues from two first- and best-in-class medicines Robust balance sheet and stable expense outlook On the road to profitability Strong 1Q26 results with multiple drivers for continued growth in 2026 and beyond 3 ROBUST COMMERCIAL DEMAND EXCELLENT PIPELINE PROGRESS STRONG FINANCIAL POSITION

1Q26 Revuforj results highlight leadership in menin inhibition and growing use in R/R NPM1m AML TRx, total prescriptions; HSCT, hematopoietic stem cell transplant; Tx, treatment EXCEPTIONAL LAUNCH >$180M net revenue ~1,380 patients treated Cumulative since launch in Nov ‘24: 1Q26 Revuforj results $48.9M net revenue ≥30% >1,300 TRx Growth in new patient starts driven by expanding uptake in R/R NPM1m AML of net revenuefrom NPM1 business 11% growth vs. 4Q25 ~13% growth vs. 4Q25, even with nearly 50% of KMT2A pts pausing Tx to proceed to HSCT New patients per quarter ~300 ~250 ~200 ~330 ~200 ≥40% of 1Q26 new patients were NPM1m

Revuforj is positioned for success with an outstanding commercial foundation All covered lives includes all commercially covered lives plus Medicare and Medicaid lives. Excellent payer coverage with no meaningful barriers to access of all covered lives with formulary coverage for both Revuforj indications 97% Robust and growing prescriber base with >85% of Tier 1+2 accounts activated Tier 1+2 account activation

Evolving clinical practice will drive continued Revuforj growth in 2026 1 2 Robust transplant rate in KMT2Ar and usage post-transplant Growing adoption in R/R NPM1m AML 3 patient population Tx durations Nearly 50% of KMT2A patients proceed to HSCT and ~45% have resumed Tx thus far R/R, relapsed/refractory; 2L, second line; 3L, third line; HSCT, hematopoietic stem cell transplantation; Tx, treatment Use in early lines of R/R treatment andin combinations Tx durations ~70% of use in 2L/3L ~40% of use in combination Average duration of therapy expected to extend as treatment patterns mature and an increasing number of patients return to therapy post-HSCT Significant room for further growth within a ~4,500 annual incident patient population
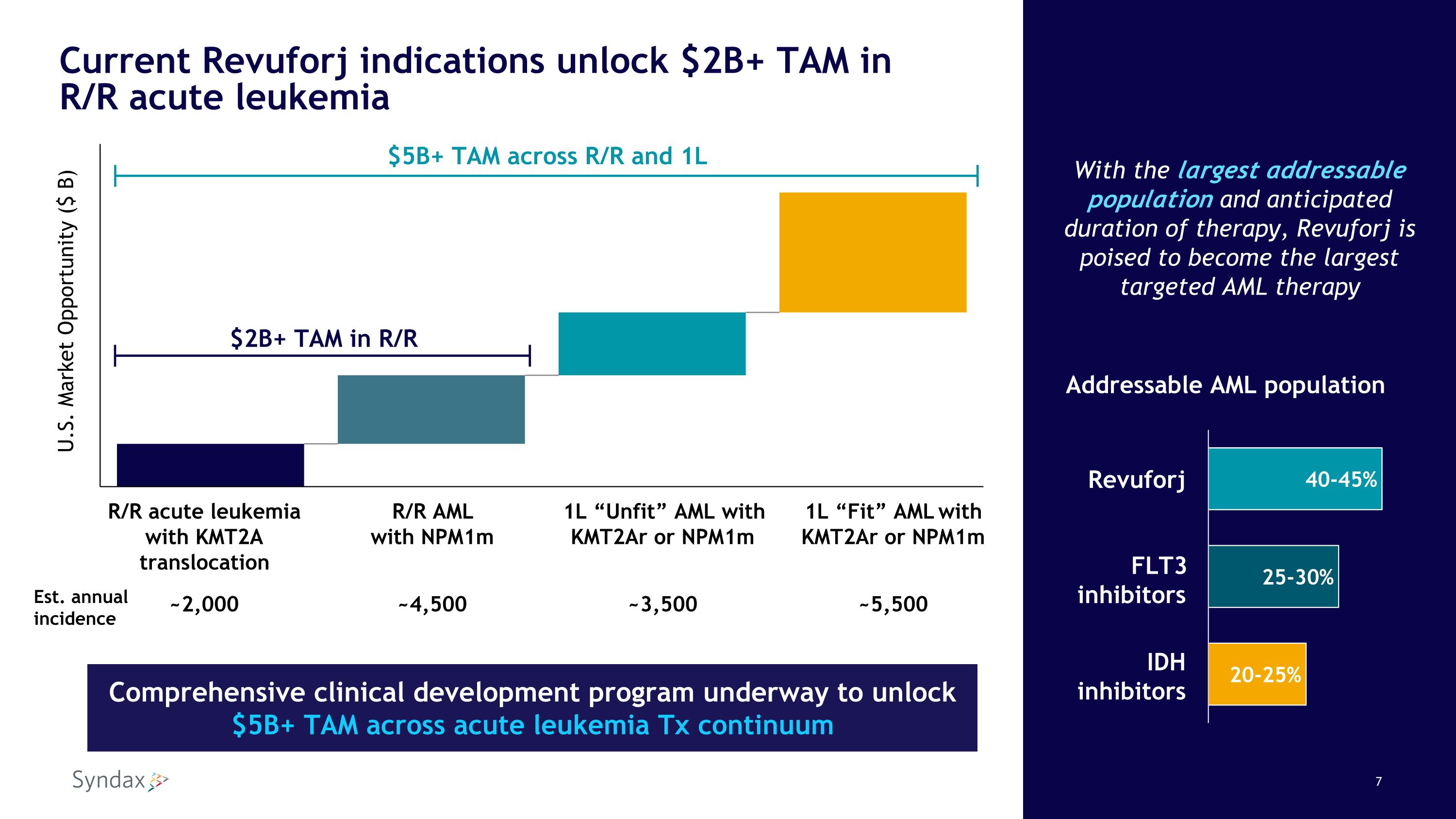
Current Revuforj indications unlock $2B+ TAM inR/R acute leukemia With the largest addressable population and anticipated duration of therapy, Revuforj is poised to become the largest targeted AML therapy R/R acute leukemia with KMT2A translocation R/R AML with NPM1m 1L “Unfit” AML with KMT2Ar or NPM1m 1L “Fit” AML with KMT2Ar or NPM1m ~2,000 ~4,500 ~3,500 ~5,500 U.S. Market Opportunity ($ B) $5B+ TAM across R/R and 1L $2B+ TAM in R/R Addressable AML population Revuforj FLT3 inhibitors IDH inhibitors Comprehensive clinical development program underway to unlock $5B+ TAM across acute leukemia Tx continuum Est. annualincidence

Building off an excellent first year, Niktimvo delivered strong 1Q26 results driven by robust demand ~300 1Q26 new patient starts ~5,000 1Q26 infusions administered Syndax records 50% of the Niktimvo net commercial profit, defined as net revenue (recorded by Incyte) minus the cost of sales and commercial expenses. $55.1M 1Q26 net revenueto INCY Compared to $13.6M in 1Q25, the first partial quarter of launch $15.9M 1Q26 collaboration revenue to SNDX Compared to -$0.2M in 1Q25, the first partial quarter of launch REMARKABLE LAUNCH $207M net revenue >1,700 patients treated Cumulative since launch in late Jan ‘25: Performance reflects strong, consistent new patient starts and solid persistency, offset by natural attrition among the large cohort of predominantly later-line patients who started in the first quarter of launch last year

Multiple drivers support continued Niktimvo growth in 2026 1 Continued adoption in 4L and growing usage in 3L cGVHD 2 Potential for extended treatment durations to address chronic disease Broad and growing prescriber base & strong commercial synergies 3 3L, third line; 4L, fourth line; BMT, bone marrow transplant centers; Tx, treatment ~32% share of 3L+ cGVHD market within 1 year of launch ~60-70% of pts who started in 1Q25 remained on Tx in 1Q26 Tx durations population and Tx durations Nearly every U.S. BMT center has ordered and become a repeat customer utilization 3

Initial Niktimvo indication represents a $2B U.S. market opportunity, with substantial opportunities for label and geographic expansion cGVHD, chronic graft-versus-host disease; 1. Internal data on file; 2. SmartImmunology Insights cGVHD report March 2020; 3. SmartImmunology Insights IPF report March 2020. * IPF trial will be conducted and funded by Syndax. ~17,000 U.S. cGVHD patients1 ~6,500 currently treated 3L+ U.S. cGVHD patients ~35,000 cGVHD patients W.W.2 Idiopathic pulmonary fibrosis (IPF) patients3 ~150,000 U.S. | ~280,000 W.W. $2B TAM with initial Niktimvo indication Ongoing combo trials could support future expansion into 1L cGVHD Ph 2 IPF trial underway, the firstof several potential areas for further pipeline expansion Ph 3 cGVHD trial underwayoutside the U.S. $5B+ TAM
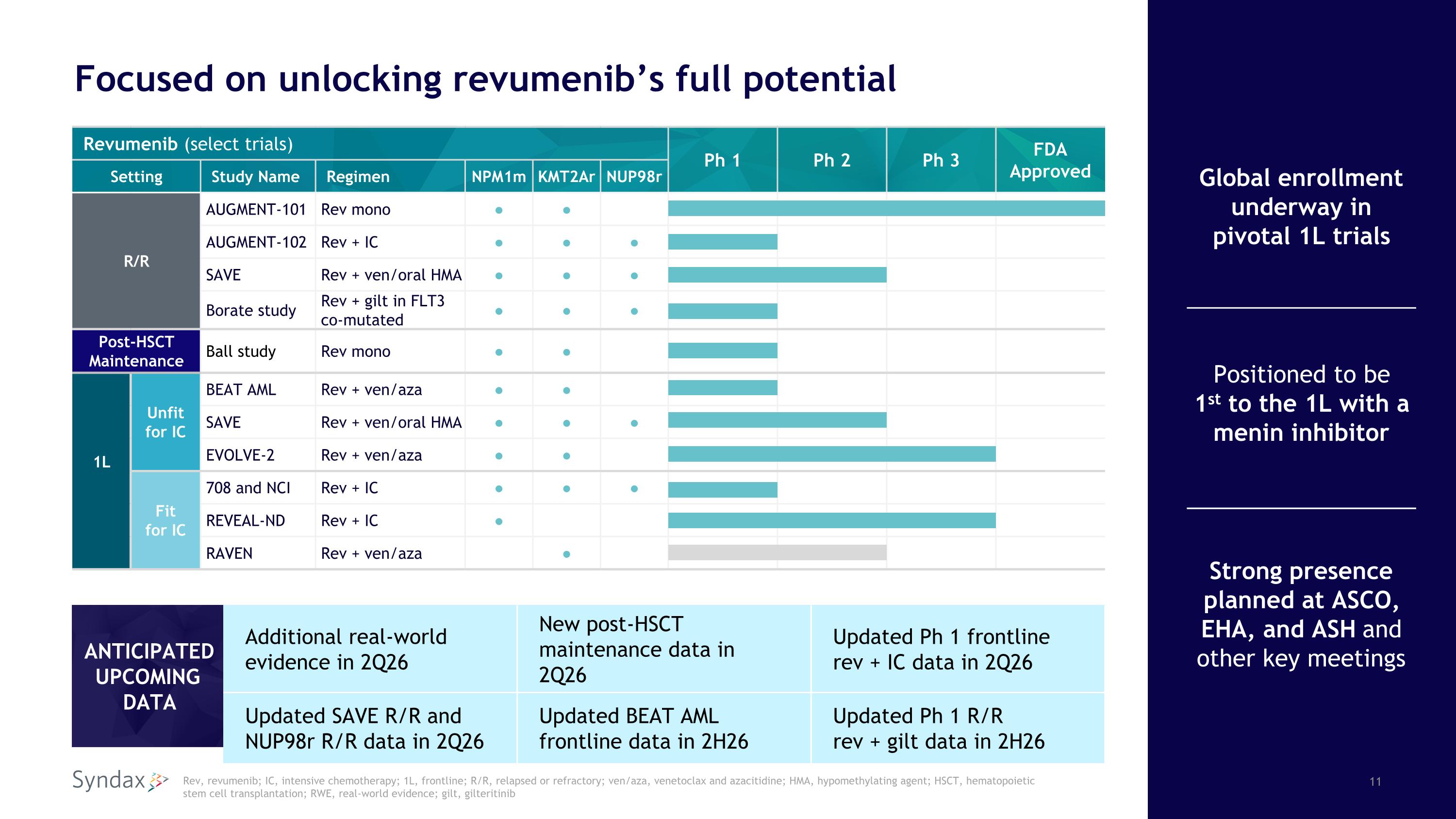
Revumenib (select trials) Ph 1 Ph 2 Ph 3 FDA Approved Setting Study Name Regimen NPM1m KMT2Ar NUP98r R/R AUGMENT-101 Rev mono ● ● AUGMENT-102 Rev + IC ● ● ● SAVE Rev + ven/oral HMA ● ● ● Borate study Rev + gilt in FLT3 co-mutated ● ● ● Post-HSCT Maintenance Ball study Rev mono ● ● 1L Unfitfor IC BEAT AML Rev + ven/aza ● ● SAVE Rev + ven/oral HMA ● ● ● EVOLVE-2 Rev + ven/aza ● ● Fitfor IC 708 and NCI Rev + IC ● ● ● REVEAL-ND Rev + IC ● RAVEN Rev + ven/aza ● Focused on unlocking revumenib’s full potential Rev, revumenib; IC, intensive chemotherapy; 1L, frontline; R/R, relapsed or refractory; ven/aza, venetoclax and azacitidine; HMA, hypomethylating agent; HSCT, hematopoieticstem cell transplantation; RWE, real-world evidence; gilt, gilteritinib Global enrollment underway in pivotal 1L trials Positioned to be1st to the 1L with amenin inhibitor Strong presence planned at ASCO, EHA, and ASH and other key meetings ANTICIPATED UPCOMINGDATA Additional real-world evidence in 2Q26 New post-HSCT maintenance data in 2Q26 Updated Ph 1 frontlinerev + IC data in 2Q26 Updated SAVE R/R and NUP98r R/R data in 2Q26 Updated BEAT AML frontline data in 2H26 Updated Ph 1 R/Rrev + gilt data in 2H26

Axatilimab (select trials) Ph 1 Ph 2 Ph 3 FDA Approved Setting Study Name Regimen R/R cGVHD AGAVE-201 Axatilimab (axa) monotherapy 1L cGVHD AXemplify-357* Axatilimab + corticosteroids NCT06388564* Axatilimab + ruxolitinib IPF MAXPIRe Axatilimab on top of SOC Robust clinical development plan underway to unlock the potential for axatilimab in 1L cGVHD, IPF, and beyond *Trials led by Incyte. List is not inclusive of ongoing ex-U.S. trials. ANTICIPATED UPCOMINGDATA Topline Ph 2 MAXPIRe IPF data in 4Q26 Topline Ph 2 axa + rux data now in 4Q26 Topline Ph 3 axa + steroids data in early 2028 Near-term Phase 2 data readouts in IPF and newly diagnosed cGVHD could open transformative opportunities

A growing body of evidence points to CSF1-dependent monocyte-derived alveolar macrophages as a promising new target in IPF Key discoveries: Monocyte-derived alveolar macrophages drive lung fibrosis Colony stimulating factor-1 receptor (CSF1R) signaling is a key regulatorof monocytes and macrophages Multiple studies implicate the CSF1R pathway in IPF: Higher CSF1R levels observed in IPF patients vs. healthy controls Higher CSF1R levels predict shorter survival in IPF patients Higher monocyte levels predict shorter survival in IPF patients Misharin J Exp Med 2017; Byrne Trends Mol Med 2016; Joshi Eur Respir J 2020; Scott Respir Med 2019; Oldham ERJ Open Res 2023; Michalaki ERS 2024.

Development in IPF supported by lung responses observed in cGVHD Axatilimab is a CSF1R-blocking antibody targeting monocyte-derived alveolar macrophages in IPF Blocking CSF-1R with axatilimab: Reduces levels of circulating profibrotic and proinflammatory monocytes and monocyte-derived macrophages Inhibits the activity of pathogenic macrophages in tissues Among cGVHD pts with lung involvement who received axa 0.3 mg/kg Q2W, nearly 50% achieved a lung response and >90% reported improvements in shortness of breath at rest Lung responses were observed across all subgroups, including patients with difficult-to-treat, severe disease Salhotra et al., oral Presentation at ATS 2025; Salhotra et al., poster presentation at ERS 2024.

Topline data from MAXPIRe Phase 2 trial of axatilimab in IPF anticipatedin 4Q26 A randomized, double-blind, placebo-controlled, multi-center international trial Key eligibility criteria: ≥40 yrs of age HRCT confirming IPF diagnosis FVC ≥45% of predicted normal (PN) FEV1/FVC ≥0.7 DLCO ≥30% and ≤90% PN Stable background use of pirfenidone or nintedanib allowed (N≈135) Placebo Q2W(n≈45) ClinicalTrials.gov ID: NCT06132256; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; SGRQ, St. George's Respiratory Questionnaire; DLCO, diffusion capacity for carbon monoxide; HRCT, high-resolution computed tomography PRIMARY ENDPOINT: Annualized rate of decline in FVC over 26 weeks (ml) Axatilimab 0.3 mg/kg Q2W (n≈90) Randomized 2:1 to axatilimab or placebo; stratified by background antifibrotic therapy (pirfenidone, nintedanib, or none) SECONDARY ENDPOINTS: Disease progression, SGRQ (quality of life measures), change in FVC % predicted, DLCO 26-week treatment

Strong financial position driven by growing revenue and stable expense outlook Financial Summary ($ in millions) Three Months EndedMarch 31 2026 2025 Product revenue, net 48.9 20.0 Collaboration revenue, net 15.9 — Total revenues 64.9 20.0 Cost of product sales (2.6) (0.9) Research & development (R&D) (58.8) (61.6) Selling, general and administrative (SG&A) (37.6) (41.0) Total operating expenses (99.1) (103.8)* Other (expense) income, net (8.5) (1.1) Net loss (42.7) (84.8) Totals may not sum due to rounding; 1. Includes short-term investments; 2. Includes pre-funded warrants to purchase 285,714 common shares (rounded). *Total operating expenses includes $0.2M in collaboration loss associated with a Niktimvo net commercial loss in 1Q25. AS OF 31 MAR 2026: $352.1M in cash and equivalents1 88.8Mshares outstanding2 2026 R&D + SG&A EXPENSE GUIDANCE: $400M, excluding $50M in expected stock option expense On the road to profitability

Continued focus on driving revenue growth and pipeline progress, with another data-rich year ahead 2026 Anticipated Milestones 2025 Key Accomplishments Advance global enrollment in pivotal 1L trials of revumenib Advance leadership in menin inhibition with new 1L, maintenance, R/R, and real-world evidence for revumenib Report topline Ph 2 axatilimab data in IPF and newly diagnosed cGVHD in 4Q26 Initiate RAVEN 1L trial of revumenib in fit KMT2Ar in 2H26 Initiate a program to generate proof-of-principle clinical data with revumenib in myelofibrosis Executed two strong product launches Expanded Revuforj into 2nd indication Initiated 1st pivotal 1L trial of a menin inhibitor Presented first RWE for a menin inhibitor Initiated managed access program, expanding access to Revuforj in certain OUS regions 1L, frontline; RWE, real-world evidence; OUS, outside the United States



















