UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): January 9, 2026
Foghorn Therapeutics Inc.
(Exact name of registrant as specified in its charter)
| Delaware | 001-39634 | 47-5271393 | ||
|
(State or other jurisdiction of incorporation) |
(Commission File Number) |
(IRS Employer Identification No.) |
||
| 99 Coolidge Ave, Suite 500 | ||||
| Watertown, MA | 02472 | |||
| (Address of principal executive offices) | (Zip Code) | |||
(Registrant’s telephone number, including area code): (617) 586-3100
500 Technology Square, Suite 700, Cambridge, MA 02139
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| ☐ | Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ☐ | Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ☐ | Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ☐ | Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
Title of each class |
Trading Symbol(s) |
Name of each exchange on which registered |
||
| Common Stock, $0.0001 par value per share | FHTX | The Nasdaq Global Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
| Item 2.02 | Results of Operations and Financial Condition. |
On January 9, 2026, Foghorn Therapeutics Inc. (the “Company”) issued a press release announcing certain business updates, including certain preliminary financial information as of December 31, 2025, ahead of the 44th Annual J.P. Morgan Healthcare Conference. The press release is attached as Exhibit 99.1 hereto and incorporated by reference herein.
The information in this Item 2.02 (including Exhibit 99.1 attached hereto) is being furnished and shall not be deemed “filed” for purposes of Section 18 of the Exchange Act, or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference into any filing by the Company under the Securities Act or the Exchange Act, except as expressly set forth by specific reference in such filing.
| Item 7.01 | Regulation FD Disclosure. |
Additionally, the Company intends to use the presentation attached as Exhibit 99.2 hereto in meetings with or presentations to investors.
The information in this Item 7.01 (including Exhibits 99.2 attached hereto) is being furnished and shall not be deemed “filed” for purposes of Section 18 of the Exchange Act, or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference into any filing by the Company under the Securities Act or the Exchange Act, except as expressly set forth by specific reference in such filing.
| Item 8.01 | Other Events. |
Purchase Agreements
On January 9, 2026, the Company entered into securities purchase agreements (the “Purchase Agreements”) with certain leading life sciences investors (the “Investors”), relating to the issuance and sale of 2,030,314 shares of its common stock, par value $0.0001 per share (“Common Stock”) and, in lieu of Common Stock, pre-funded warrants to purchase 5,421,250 shares of Common Stock (the “Pre-Funded Warrants”). The Company sold the shares of Common Stock and Pre-Funded Warrants together with two series of warrants, Series 1 Warrants and Series 2 Warrants, to purchase an aggregate of 7,451,564 shares of the Common Stock (the “Series Warrants”). The Pre-Funded Warrants are exercisable immediately upon issuance at an initial exercise price of $0.0001 per share and have a term of 20 years. The shares of Common Stock, or Pre-Funded Warrants, and the accompanying Series Warrants are immediately separable and will be issued separately, but they will be purchased together in this offering.
The Series Warrants are immediately exercisable. Each Series 1 Warrant has an initial exercise price of $13.42 per share of Common Stock, subject to certain adjustments, and expires on June 30, 2027. Each Series 2 Warrant has an initial exercise price of $20.13 per share of Common Stock, subject to certain adjustments, and expires on December 31, 2030. For the Series Warrants, the Investor may elect to receive, in lieu of shares of Common Stock, pre-funded warrants to purchase an equivalent number of shares of Common Stock.
Subject to certain exclusions, if, prior to the June 30, 2027, the Company sells shares of capital stock or derivative securities convertible into or exercisable for capital stock in one or more related transactions primarily for the purpose of raising capital at a Weighted-Average Price (as described below) below $13.42 per share, then the initial exercise price of a Series Warrant will be automatically reset upon exercise to an exercise price (the “Adjusted Exercise Price”) that is the midpoint between the initial exercise price and the lowest Weighted-Average Price per share at which the Company sells capital stock or derivative securities convertible into or exercisable for capital stock in a subsequent offering prior to the exercise date; provided, however, that the Adjusted Exercise Price will not be reduced below $6.71 per share. The “Weighted-Average Price” shall be calculated as the weighted-average common stock equivalent price of the equity securities sold in such transaction(s) (excluding any derivative securities with an exercise or conversion price that is above the closing sale price as of the time of pricing such offering(s)). In no event will the exercise price for a Series Warrant be adjusted more than once pursuant to this adjustment mechanism.
The offering price for the shares of Common Stock is $6.71 per share (or $6.7099 for each Pre-Funded Warrant, which equals the price per share of the Common Stock less the exercise price of the Pre-Funded Warrants). The aggregate gross proceeds to the Company from this offering were approximately $50.0 million, excluding any proceeds the Company may receive upon exercise of the Pre-Funded Warrants and Series Warrants. No underwriter or placement agent participated in the offering.
The number of shares of Common Stock purchasable upon the exercise of the Pre-Funded Warrants and the exercise price and the number of shares of Common Stock purchasable upon the exercise of the Series Warrants are subject to adjustment upon the occurrence of specific events, including stock dividends, stock splits, reclassifications and combinations of the Company’s Common Stock.
The offering was made pursuant to an effective registration statement on Form S-3 (Registration Statement No. 333-284476), as previously filed with the Securities and Exchange Commission, and a related prospectus. The offering is expected to close on January 13, 2026.
The Purchase Agreement contains customary representations, warranties and agreements by the Company, customary conditions to closing, indemnification obligations of the Company and the Investors. The representations, warranties and covenants contained in the Purchase Agreement were made only for purposes of such agreements and as of specific dates, were solely for the benefit of the parties to such agreement, and may be subject to limitations agreed upon by the contracting parties.
The form of Purchase Agreement, form of Pre-Funded Warrant and the form of Series Warrant are filed as Exhibit 99.3, Exhibit 4.1 and Exhibit 4.2, respectively, and the description of the terms of the Purchase Agreement, the Pre-Funded Warrants and the Series Warrants are qualified in their entirety by reference to such exhibit. A copy of the opinions of Ropes & Gray LLP relating to the legality of the issuance and sale of the shares, Pre-Funded Warrants and Series Warrants are attached as Exhibit 5.1 hereto.
| Item 9.01. | Financial Statements and Exhibits. |
(d) Exhibits
| Exhibit No. |
Description | |
| 4.1 | Form of Series Warrant | |
| 4.2 | Form of Pre-Funded Warrant | |
| 5.1 | Opinion of Ropes & Gray LLP | |
| 23.1 | Consent of Ropes & Gray LLP (contained in Exhibit 5.1) | |
| 99.1 | Press release issued on January 9, 2026 | |
| 99.2 | Investor Presentation dated January 2026 | |
| 99.3 | Form of Securities Purchase Agreement between the Company and the Investors, dated January 9, 2026 | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document). | |
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| FOGHORN THERAPEUTICS INC. | ||
| By: | /s/ Michael J. LaCascia |
|
| Michael J. LaCascia | ||
| Chief Legal Officer | ||
Dated: January 12, 2026
Exhibit 4.1
FOGHORN THERAPEUTICS INC.
FORM OF WARRANT TO PURCHASE COMMON STOCK
| Number of Shares: [ ] (subject to adjustment) |
||
| Series [1][2] Warrant No. [1][2][-[ ] | Original Issue Date: [ ] |
Foghorn Therapeutics Inc., a Delaware corporation (the “Company”), hereby certifies that, for good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, [ ] or its permitted registered assigns (the “Holder”), is entitled, subject to the terms set forth below, to purchase from the Company up to a total of [ ] shares of common stock, $0.0001 par value per share (the “Common Stock”), of the Company (each such share, a “Warrant Share” and all such shares, the “Warrant Shares”) at an exercise price per share equal to $[ ] per share (as adjusted from time to time as provided in Section 9 herein, the “Exercise Price”), upon surrender of this Warrant to Purchase Common Stock (including any Warrants to Purchase Common Stock issued in exchange, transfer or replacement hereof, the “Warrant”) at any time and from time to time on or after the date hereof (the “Original Issue Date”) and through and including 5:30 P.M., New York City time, on Expiration Date (as defined herein), and subject to the following terms and conditions:
1. Definitions. For purposes of this Warrant, the following terms shall have the following meanings:
(a) “Affiliate” means any Person directly or indirectly controlled by, controlling or under common control with, a Holder, but only for so long as such control shall continue. For purposes of this definition, “control” (including, with correlative meanings, “controlled by,” “controlling” and “under common control with”) means, with respect to a Person, possession, direct or indirect, of (a) the power to direct or cause direction of the management and policies of such Person (whether through ownership of securities or partnership or other ownership interests, by contract or otherwise), or (b) at least 50% of the voting securities (whether directly or pursuant to any option, warrant or other similar arrangement) or other comparable equity interests.
(b) “Commission” means the United States Securities and Exchange Commission.
(c) “Closing Sale Price” means, for any security as of any date, the last trade price for such security on the Principal Trading Market for such security, as reported by Bloomberg Financial Markets, or, if such Principal Trading Market begins to operate on an extended hours basis and does not designate the last trade price, then the last trade price of such security prior to 4:00 P.M., New York City time, as reported by Bloomberg Financial Markets, or if the foregoing do not apply, the last trade price of such security in the over-the-counter market on the electronic bulletin board for such security as reported by Bloomberg Financial Markets. If the Closing Sale Price cannot be calculated for a security on a particular date on any of the foregoing bases, the Closing Sale Price of such security on such date shall be the fair market value as mutually determined by the Company and the Holder. If the Company and the Holder are unable to agree upon the fair market value of such security, then the Board of Directors of the Company shall use its good faith judgment to determine the fair market value. The Board of Directors’ determination shall be binding upon all parties absent demonstrable error. All such determinations shall be appropriately adjusted for any stock dividend, stock split, stock combination or other similar transaction during the applicable calculation period.
(d) “Exchange Act” means the Securities Exchange Act of 1934, as amended.
(e) “Principal Trading Market” means the national securities exchange or other trading market on which the Common Stock is primarily listed on and quoted for trading, which, as of the Original Issue Date, shall be The Nasdaq Global Market.
(f) “Registration Statement” means the Company’s Registration Statement on Form S-3 (File No. 333-284476), declared effective on January 31, 2025.
(g) “Securities Act” means the Securities Act of 1933, as amended.
(h) “Trading Day” means any weekday on which the Principal Trading Market is open for trading.
(i) “Transfer Agent” means Computershare Trust Company, N.A., the Company’s transfer agent and registrar for the Common Stock, and any successor appointed in such capacity.
2. Registration of Warrants. The Company shall register this Warrant, upon records to be maintained by the Company for that purpose (the “Warrant Register”), in the name of the record Holder (which shall include the initial Holder or, as the case may be, any registered assignee to which this Warrant is permissibly assigned hereunder) from time to time. The Company may deem and treat the registered Holder of this Warrant as the absolute owner hereof for the purpose of any exercise hereof or any distribution to the Holder, and for all other purposes, absent actual notice to the contrary.
3. Registration of Transfers. Subject to compliance with all applicable securities laws, the Company shall, or will cause its Transfer Agent to, register the transfer of all or any portion of this Warrant in the Warrant Register, upon surrender of this Warrant, and payment for all applicable transfer taxes (if any). Upon any such registration or transfer, a new warrant to purchase Common Stock in substantially the form of this Warrant (any such new warrant, a “New Warrant”) evidencing the portion of this Warrant so transferred shall be issued to the transferee, and a New Warrant evidencing the remaining portion of this Warrant not so transferred, if any, shall be issued to the transferring Holder. The acceptance of the New Warrant by the transferee thereof shall be deemed the acceptance by such transferee of all of the rights and obligations in respect of the New Warrant that the Holder has in respect of this Warrant. The Company shall, or will cause its Transfer Agent to, prepare, issue and deliver at the Company’s own expense any New Warrant under this Section 3. Until due presentment for registration of transfer, the Company may treat the registered Holder hereof as the owner and holder for all purposes, and the Company shall not be affected by any notice to the contrary.
4. Exercise and Duration of Warrants.
(a) This Warrant shall expire on [ ] (the “Expiration Date”). All or any part of this Warrant shall be exercisable by the registered Holder in any manner permitted by Section 10 of this Warrant at any time and from time to time on or after the Original Issue Date and through and including 5:30 P.M. New York City time, on the Expiration Date. At 5:30 P.M., New York City time, on the Expiration Date, the portion of this Warrant not exercised prior thereto shall be and become void and of no value and this Warrant shall be terminated and no longer outstanding.
(b) The Holder may exercise this Warrant by delivering to the Company (i) an exercise notice, in the form attached as Schedule 1 hereto (the “Exercise Notice”), completed and duly signed, and (ii) payment of the Exercise Price for the number of Warrant Shares as to which this Warrant is being exercised (which may take the form of a “cashless exercise” if so indicated in the Exercise Notice pursuant to Section 10 below), and the date on which the last of such items is delivered to the Company (as determined in accordance with the notice provisions hereof) is an “Exercise Date.” The Holder shall not be required to deliver the original Warrant in order to effect an exercise hereunder. Execution and delivery of the Exercise Notice shall have the same effect as cancellation of the original Warrant and issuance of a New Warrant evidencing the right to purchase the remaining number of Warrant Shares.
2
5. Delivery of Warrant Shares.
(a) Upon exercise of this Warrant, the Company shall promptly (but in no event later than one Trading Day after the Exercise Date), upon the request of the Holder, credit such aggregate number of shares of Common Stock to which the Holder is entitled pursuant to such exercise to the Holder’s or its designee’s balance account with The Depository Trust Company (“DTC”) through its Deposit Withdrawal Agent Commission system, or if the Transfer Agent is not participating in the Fast Automated Securities Transfer Program (the “FAST Program”) or if the certificates are required to bear a legend regarding restriction on transferability, issue and dispatch by overnight courier to the address as specified in the Exercise Notice, a certificate, registered in the Company’s share register in the name of the Holder or its designee, for the number of shares of Common Stock to which the Holder is entitled pursuant to such exercise. The Holder, or any natural person or legal entity (each, a “Person”) permissibly so designated by the Holder to receive Warrant Shares, shall be deemed to have become the holder of record of such Warrant Shares as of the Exercise Date, irrespective of the date such Warrant Shares are credited to the Holder’s or its designee’s DTC account or the date of delivery of the certificates evidencing such Warrant Shares, as the case may be.
(b) If by the close of the Trading Day after the Exercise Date, the Company fails to deliver to the Holder a certificate representing the required number of Warrant Shares in the manner required pursuant to Section 5(a) or fails to credit the Holder’s or its designee’s balance account with DTC for such number of Warrant Shares to which the Holder is entitled, and if after such Trading Day and prior to the receipt of such Warrant Shares, the Holder purchases (in an open market transaction or otherwise) shares of Common Stock to deliver in satisfaction of a sale by the Holder of the Warrant Shares which the Holder anticipated receiving upon such exercise (a “Buy-In”), then the Company shall, within one Trading Day after the Holder’s request and in the Holder’s sole discretion, either (1) pay in cash to the Holder an amount equal to the Holder’s total purchase price (including brokerage commissions, if any) for the shares of Common Stock so purchased (the “Buy-In Price”), at which point the Company’s obligation to deliver such certificate (and to issue such Warrant Shares) shall terminate or (2) promptly honor its obligation to deliver to the Holder a certificate or certificates representing such Warrant Shares or credit the Holder’s or its designee’s balance account with DTC for such Warrant Shares and pay cash to the Holder in an amount equal to the excess (if any) of Holder’s total purchase price (including brokerage commissions, if any) for the shares of Common Stock so purchased in the Buy-In over the product of (A) the number of shares of Common Stock purchased in the Buy-In, times (B) the Closing Sale Price of a share of Common Stock on the Exercise Date.
(c) To the extent permitted by law and subject to Section 5(b), the Company’s obligations to issue and deliver Warrant Shares in accordance with and subject to the terms hereof (including the limitations set forth in Section 11 below) are absolute and unconditional, irrespective of any action or inaction by the Holder to enforce the same, any waiver or consent with respect to any provision hereof, the recovery of any judgment against any Person or any action to enforce the same, or any setoff, counterclaim, recoupment, limitation or termination, or any breach or alleged breach by the Holder or any other Person of any obligation to the Company or any violation or alleged violation of law by the Holder or any other Person, and irrespective of any other circumstance that might otherwise limit such obligation of the Company to the Holder in connection with the issuance of Warrant Shares. Subject to Section 5(b), nothing herein shall limit the Holder’s right to pursue any other remedies available to it hereunder, at law or in equity including, without limitation, a decree of specific performance and/or injunctive relief with respect to the Company’s failure to timely deliver certificates representing shares of Common Stock upon exercise of the Warrant as required pursuant to the terms hereof.
6. Charges, Taxes and Expenses. Issuance and delivery of certificates for shares of Common Stock upon exercise of this Warrant shall be made without charge to the Holder for any issue or transfer tax, transfer agent fee or other incidental tax or expense (excluding any applicable stamp duties) in respect of the issuance of such certificates, all of which taxes and expenses shall be paid by the Company; provided, however, that the Company shall not be required to pay any tax that may be payable in respect of any transfer involved in the registration of any certificates for Warrant Shares or the Warrants in a name other than that of the Holder or an Affiliate thereof. The Holder shall be responsible for all other tax liability that may arise as a result of holding or transferring this Warrant or receiving Warrant Shares upon exercise hereof.
7. Replacement of Warrant. If this Warrant is mutilated, lost, stolen or destroyed, the Company shall issue or cause to be issued in exchange and substitution for and upon cancellation hereof, or in lieu of and substitution for this Warrant, a New Warrant, but only upon receipt of evidence reasonably satisfactory to the Company of such loss, theft or destruction (in such case) and, in each case, a customary and reasonable indemnity and surety bond, if requested by the Company. Applicants for a New Warrant under such circumstances shall also comply with such other reasonable regulations and procedures and pay such other reasonable third-party costs as the Company may prescribe. If a New Warrant is requested as a result of a mutilation of this Warrant, then the Holder shall deliver such mutilated Warrant to the Company as a condition precedent to the Company’s obligation to issue the New Warrant.
3
8. Reservation of Warrant Shares. The Company covenants that it will at all times while this Warrant is outstanding reserve and keep available out of the aggregate of its authorized but unissued and otherwise unreserved Common Stock, solely for the purpose of enabling it to issue Warrant Shares upon exercise of this Warrant as herein provided, the number of Warrant Shares that are initially issuable and deliverable upon the exercise of this entire Warrant, free from preemptive rights or any other contingent purchase rights of persons other than the Holder (taking into account the adjustments and restrictions of Section 9). The Company covenants that all Warrant Shares so issuable and deliverable shall, upon issuance and the payment of the applicable Exercise Price in accordance with the terms hereof, be duly and validly authorized, issued and fully paid and nonassessable. The Company will take all such action as may be reasonably necessary to assure that such shares of Common Stock may be issued as provided herein without violation of any applicable law or regulation, or of any requirements of any securities exchange or automated quotation system upon which the Common Stock may be listed.
9. Certain Adjustments. The number of Warrant Shares issuable upon exercise of this Warrant is subject to adjustment from time to time as set forth in this Section 9.
(a) One-Time Exercise Price Adjustment. If, prior to June 30, 2027, the Company sells shares of capital stock or derivative securities convertible into or exercisable for capital stock (other than Exempted Securities) in one or more related transactions primarily for the purpose of raising capital (a “Subsequent Offering”) at a Weighted-Average Price (as defined below) below $13.42 per share (as may be adjusted by paragraphs (b) through (h)), then the initial Exercise Price of this Warrant (the “Initial Exercise Price”) will be automatically reset upon exercise of this Warrant to an exercise price (the “Adjusted Exercise Price”) that is the midpoint between the Initial Exercise Price and the lowest Weighted-Average Price per share at which the Company sells capital stock or derivative securities convertible into or exercisable for capital stock in a Subsequent Offering prior to the exercise date; provided, however, that the Adjusted Exercise Price will not be reduced below $6.71. For purposes of this Section 9(a), the “Weighted-Average Price” shall be calculated as the weighted-average common stock equivalent price of the equity securities sold in such transaction(s) (excluding any derivative securities with an exercise or conversion price that is above the Closing Sale Price as of the time of pricing such offering(s)). For example, if the Company were to conduct a capital raising transaction where it sold 100 shares of Common Stock at $8.00 per share, but also provided 25% warrant coverage with an exercise price of $6.00 per share, then the weighted average common stock equivalent price would be $7.60 per share (i.e., $950 in total potential proceeds, divided by 125 total potential shares), and the Adjusted Exercise Price would equal midpoint between the Initial Exercise Price and $7.60 per share (rounded up to the nearest cent). In no event will the exercise price for this Warrant be adjusted more than once pursuant to this Section 9(a). “Exempted Securities” means (a) shares of Common Stock or rights, warrants or options to purchase Common Stock issued in connection with any acquisition of another corporation by the Company by merger, purchase of substantially all of the assets or other reorganization or to a joint venture agreement, provided that such issuances are approved by the Board of Directors, (b) equity securities issued by reason of a dividend, stock split, split-up or other distribution on shares of Common Stock, (c) shares of Common Stock or rights, warrants or options to purchase Common Stock issued to employees or directors of, or consultants or advisors to, the Company or any of its subsidiaries pursuant to a plan, agreement or arrangement approved by the Board of Directors (“Equity Plans”), (d) shares of Common Stock issued upon the exercise of options or upon the conversion or exchange of any securities convertible into Common Stock, including any pre-funded warrants outstanding as of the Original Issue Date, any pre-funded warrants to be issued pursuant to Section 15(i) of this Warrant and any pre-funded warrants issued pursuant to the Securities Purchase Agreement dated January 9, 2026 between the Company and certain Investors, in each case provided that such issuance is pursuant to the terms of the applicable option or convertible security, (e) shares of Common Stock or rights, warrants or options to purchase Common Stock issued to banks, equipment lessors or other financial institutions, real property lessors, or to other persons engaged in the business of making loans, pursuant to a debt financing, equipment leasing or real property leasing transaction approved by the Board of Directors, (f) shares of Common Stock or rights, warrants or options to purchase Common Stock issued in connection with sponsored research, collaboration, technology license, development, marketing or other similar agreements or strategic partnerships approved by the Board of Directors, or (g) shares of Common Stock issued under an at-the-market facility (an “ATM Program”), provided that the aggregate gross proceeds from sales under such program do not exceed $10,000,000 (the “ATM Threshold”). For the avoidance of doubt, all sales under an ATM Program in excess of the ATM Threshold shall constitute a single Subsequent Offering and any adjustment related thereto shall be calculated by determining the weighted-average price of such sales. For the avoidance of doubt, the adjustments to the initial Exercise Price described in this Section 9(a) shall apply only to a single Subsequent Offering and in no event shall the Exercise Price be adjusted more than once pursuant to this Section 9(a).
4
(b) Stock Dividends and Splits. If the Company, at any time while this Warrant is outstanding, (i) pays a stock dividend on its Common Stock or otherwise makes a distribution on any class of capital stock issued and outstanding on the Original Issue Date and in accordance with the terms of such stock on the Original Issue Date or as amended, that is payable in shares of Common Stock, (ii) subdivides its outstanding shares of Common Stock into a larger number of shares of Common Stock, (iii) combines its outstanding shares of Common Stock into a smaller number of shares of Common Stock or (iv) issues by reclassification of shares of capital stock any additional shares of Common Stock of the Company, then in each such case the number of Warrant Shares then underlying this Warrant shall be divided by a fraction, the numerator of which shall be the number of shares of Common Stock outstanding immediately before such event and the denominator of which shall be the number of shares of Common Stock outstanding immediately after such event. Any adjustment made pursuant to clause (i) of this paragraph shall become effective immediately after the record date for the determination of stockholders entitled to receive such dividend or distribution, provided, however, that if such record date shall have been fixed and such dividend is not fully paid on the date fixed therefor, the number of Warrant Shares shall be recomputed accordingly as of the close of business on such record date and thereafter the Warrant Shares shall be adjusted pursuant to this paragraph as of the time of actual payment of such dividends. Any adjustment pursuant to clause (ii) or (iii) of this paragraph shall become effective immediately after the effective date of such subdivision or combination. Simultaneously with any adjustment made pursuant to this Section 9(b), the Exercise Price shall be increased or decreased proportionately, so that after such adjustment the aggregate Exercise Price payable hereunder for the increased or decreased number of Warrant Shares shall be the same as the aggregate Exercise Price in effect immediately prior to such adjustment. Notwithstanding the foregoing, in no event may the Exercise Price be adjusted below the par value of the Common Stock then in effect.
(c) Pro Rata Distributions. If the Company, at any time while this Warrant is outstanding, distributes to all holders of Common Stock for no consideration (i) evidences of its indebtedness, (ii) any security (other than a distribution of Common Stock covered by the preceding paragraph), (iii) rights or warrants to subscribe for or purchase any security, or (iv) cash or any other asset (in each case, “Distributed Property”), then, upon any exercise of this Warrant that occurs after the record date fixed for determination of stockholders entitled to receive such distribution, the Holder shall be entitled to receive, in addition to the Warrant Shares otherwise issuable upon such exercise (if applicable), the Distributed Property that such Holder would have been entitled to receive in respect of such number of Warrant Shares had the Holder been the record holder of such Warrant Shares immediately prior to such record date without regard to any limitation on exercise contained therein.
(d) Purchase Rights. In addition to any adjustments pursuant to the other subsections of this Section 9, if at any time on or after the Original Issue Date, the Company grants, issues or sells any Options, Convertible Securities or rights to purchase stock, warrants, securities or other property, in each case pro rata to the record holders of any class of Common Stock (the “Purchase Rights”), then the Holder will be entitled to acquire, upon the terms applicable to such Purchase Rights, the aggregate Purchase Rights which the Holder could have acquired if the Holder had held the number of shares of Common Stock acquirable upon complete exercise of this Warrant (without regard to any limitations or restrictions on exercise of this Warrant, including without limitation, the Maximum Percentage) immediately before the date on which a record is taken for the grant, issuance or sale of such Purchase Rights, or, if no such record is taken, the date as of which the record holders of Common Stock are to be determined for the grant, issuance or sale of such Purchase Rights (provided, that to the extent that the Holder’s right to participate in any such Purchase Right would result in the Holder and its Affiliates exceeding the Maximum Percentage, then the Holder shall not be entitled to participate in such Purchase Right to such extent (and shall not be entitled to beneficial ownership of such Common Stock as a result of such Purchase Right (and beneficial ownership) to such extent) and such Purchase Right to such extent shall be held in abeyance for the benefit of the Holder until such time or times as its right thereto would not result in the Holder and its Affiliates exceeding the Maximum Percentage, at which time or times the Holder shall be granted such right (and any Purchase Right granted, issued or sold on such initial Purchase Right or on any subsequent Purchase Right to be held similarly in abeyance) to the same extent as if there had been no such limitation). As used in this Section 9(d), (i) “Options” means any rights, warrants or options to subscribe for or purchase shares of Common Stock or Convertible Securities and (ii) “Convertible Securities” mean any stock or securities (other than Options) directly or indirectly convertible into or exercisable or exchangeable for shares of Common Stock.
5
(e) Fundamental Transactions. If, at any time while this Warrant is outstanding (i) the Company effects any merger or consolidation of the Company with or into another Person, in which the Company is not the surviving entity or the stockholders of the Company immediately prior to such merger or consolidation do not own, directly or indirectly, at least 50% of the voting power of the surviving entity immediately after such merger or consolidation, (ii) the Company effects any sale to another Person of all or substantially all of its assets in one transaction or a series of related transactions, (iii) pursuant to any tender offer or exchange offer (whether by the Company or another Person), holders of capital stock who tender shares representing more than 50% of the voting power of the capital stock of the Company and the Company or such other Person, as applicable, accepts such tender for payment, (iv) the Company consummates a stock purchase agreement or other business combination (including, without limitation, a reorganization, recapitalization, spin-off or scheme of arrangement) with another Person whereby such other Person acquires more than the 50% of the voting power of the capital stock of the Company (except for any such transaction in which the stockholders of the Company immediately prior to such transaction maintain, in substantially the same proportions, the voting power of such Person immediately after the transaction) or (v) the Company effects any reclassification of the Common Stock or any compulsory share exchange pursuant to which the Common Stock is effectively converted into or exchanged for other securities, cash or property (other than as a result of a subdivision or combination of shares of Common Stock covered by Section 9(b) above) (in any such case, a “Fundamental Transaction”), then following such Fundamental Transaction the Holder shall have the right to receive, upon exercise of this Warrant, the same amount and kind of securities, cash or property as it would have been entitled to receive upon the occurrence of such Fundamental Transaction if it had been, immediately prior to such Fundamental Transaction, the holder of the number of Warrant Shares then issuable upon exercise in full of this Warrant without regard to any limitations on exercise contained herein (the “Alternate Consideration”). The Company shall not effect any Fundamental Transaction in which the Company is not the surviving entity or the Alternate Consideration includes securities of another Person unless (i) the Alternate Consideration is solely cash and the Company provides for the simultaneous “cashless exercise” of this Warrant pursuant to Section 10 below or (ii) prior to or simultaneously with the consummation thereof, any successor to the Company, surviving entity or other Person (including any purchaser of assets of the Company) shall assume the obligation to deliver to the Holder, such Alternate Consideration as, in accordance with the foregoing provisions, the Holder may be entitled to receive, and the other obligations under this Warrant. The provisions of this paragraph (e) shall similarly apply to subsequent transactions analogous of a Fundamental Transaction type.
(f) Calculations. All calculations under this Section 9 shall be made to the nearest share.
(g) Notice of Adjustments. Upon the occurrence of each adjustment pursuant to this Section 9, the Company at its expense will, at the written request of the Holder, promptly compute such adjustment, in good faith, in accordance with the terms of this Warrant and prepare a certificate setting forth such adjustment, including a statement of the adjusted number or type of Warrant Shares or other securities issuable upon exercise of this Warrant (as applicable), describing the transactions giving rise to such adjustments and showing in detail the facts upon which such adjustment is based. Upon written request, the Company will promptly deliver a copy of each such certificate to the Holder and to the Company’s transfer agent.
(h) Notice of Corporate Events. If, while this Warrant is outstanding, the Company (i) declares a dividend or any other pro rata distribution of cash, securities or other property in respect of its Common Stock, including, without limitation, any granting of rights or warrants to subscribe for or purchase any capital stock of the Company or any subsidiary, (ii) authorizes or approves, enters into any agreement contemplating or solicits stockholder approval for any Fundamental Transaction or (iii) authorizes the voluntary dissolution, liquidation or winding up of the affairs of the Company, then, except if such notice and the contents thereof shall be deemed to constitute material non-public information, the Company shall deliver to the Holder a notice of such transaction at least ten (10) days prior to the applicable record or effective date on which a Person would need to hold Common Stock in order to participate in or vote with respect to such transaction; provided, however, that the failure to deliver such notice or any defect therein shall not affect the validity of the corporate action required to be described in such notice. In addition, if while this Warrant is outstanding, the Company authorizes or approves, enters into any agreement contemplating or solicits stockholder approval for any Fundamental Transaction contemplated by Section 9(e), other than a Fundamental Transaction under clause (iii) of Section 9(e), the Company shall deliver to the Holder a notice of such Fundamental Transaction at least thirty (30) days prior to the date such Fundamental Transaction is consummated. Holder agrees to maintain any information disclosed pursuant to this Section 9(h) in confidence until such information is publicly available, and shall comply with applicable law with respect to trading in the Company’s securities following receipt of any such information.
6
10. Payment of Exercise Price. Notwithstanding anything contained herein to the contrary, the Holder may, in its sole discretion, satisfy its obligation to pay the Exercise Price through a “cashless exercise,” in which event the Company shall issue to the Holder the number of Warrant Shares determined as follows:
X = Y [(A-B)/A]
where:
“X” equals the number of Warrant Shares to be issued to the Holder;
“Y” equals the total number of Warrant Shares with respect to which this Warrant is then being exercised;
“A” equals the average of the Closing Sale Prices of the shares of Common Stock (as reported by Bloomberg Financial Markets) for the five (5) consecutive Trading Days ending on the date immediately preceding the Exercise Date; and
“B” equals the Exercise Price then in effect for the applicable Warrant Shares at the time of such exercise.
For purposes of Rule 144 promulgated under the Securities Act, it is intended, understood and acknowledged that the Warrant Shares issued in a “cashless exercise” transaction shall be deemed to have been acquired by the Holder, and the holding period for the Warrant Shares shall be deemed to have commenced, on the date this Warrant was originally issued (provided that the Commission continues to take the position that such treatment is proper at the time of such exercise). Except as set forth in Section 5(b) (Buy-In remedy) and Section 12 (payment of cash in lieu of fractional shares), in no event will the exercise of this Warrant be settled in cash.
In the event that the Registration Statement, or another registration statement registering the issuance of the Warrant Shares is, for any reason, not effective at the time of exercise of this Warrant, then the Warrant may only be exercised through a cashless exercise, as set forth in this Section 10. If Warrant Shares are issued in such a cashless exercise, the Company acknowledges and agrees that in accordance with Section 3(a)(9) of the Securities Act of 1933, as amended, the Warrant Shares shall take on the registered characteristics of the Warrants being exercised, and the holding period of the Warrants being exercised may be tacked on to the holding period of the Warrant Shares.
11. Limitations on Exercise.
(a) Notwithstanding anything to the contrary contained herein, the number of Warrant Shares that may be acquired by the Holder upon any exercise of this Warrant (or otherwise in respect hereof) shall be limited to the extent necessary to ensure that, following such exercise , the total number of shares of Common Stock then beneficially owned by the Holder and its Affiliates and any other Persons whose beneficial ownership of Common Stock would be aggregated with the Holder’s for purposes of Section 13(d) of the Exchange Act, does not exceed [9.99%][19.99% (if exceeding such limit would result in a change of control under Nasdaq Listing Rule 5635(b) or any successor rule)] of the total number of then issued and outstanding shares of Common Stock (including for such purpose the shares of Common Stock issuable upon such exercise) (the “Maximum Percentage”), it being acknowledged by the Holder that the Company is not representing to such Holder that such calculation is in compliance with Section 13(d) of the Exchange Act and such Holder is solely responsible for any schedules required to be filed in accordance therewith. To the extent that the limitation contained in this Section 11(a) applies, the determination of whether this Warrant is exercisable (in relation to other securities owned by such Holder) and of which a portion of this Warrant is exercisable shall be in the sole discretion of the Holder, and the submission of a Notice of Exercise shall be deemed to be the Holder’s determination of whether this Warrant is exercisable (in relation to other securities owned by such Holder) and of which portion of this Warrant is exercisable, in each case subject to such aggregate percentage limitation, and the Company shall have no obligation to verify or confirm the accuracy of such determination.
7
In addition, a determination under this Section 11(a) as to any group status shall be determined in accordance with Section 13(d) of the Exchange Act and the rules and regulations promulgated thereunder. For purposes of this Section 11(a), in determining the number of outstanding shares of Common Stock, the Holder may rely on the number of outstanding shares of Common Stock as reflected in (x) the Company’s most recent Form 10-Q or Form 10-K, as the case may be, (y) a more recent public announcement by the Company or (z) any other notice to the Holder by the Company or the Transfer Agent setting forth the number of shares of Common Stock outstanding. Upon the written request of the Holder, the Company shall, within three (3) Trading Days, confirm orally and in writing to such Holder the number of shares of Common Stock then outstanding. By written notice to the Company, which will not be effective until the sixty-first (61st) day after such notice is delivered to the Company, the Holder may waive the provisions of this Section 11(a) or increase the beneficial ownership limitation to such percentage of the number of shares of the Common Stock outstanding immediately after giving effect to the issuance of shares of Common Stock upon exercise of this Warrant as the Holder shall determine, in its sole discretion, subject to Section 11(b), and the provisions of this Section 11(a) shall continue to apply. Upon such a change by a Holder of the beneficial ownership limitation from such 9.99% limitation to such other percentage limitation, the beneficial ownership limitation may not be further waived or increased by such Holder without first providing the minimum notice required by this Section 11(a). Notwithstanding the foregoing, at any time following notice of a Fundamental Transaction under Section 9(h)(ii) with respect to a Section 9(e)(iii) Fundamental Transaction, the Holder may waive and/or change the beneficial ownership limitation effective immediately upon written notice to the Company and may reinstitute a beneficial ownership limitation at any time thereafter effective immediately upon written notice to the Company. For purposes of this Section 11(a), the aggregate number of shares of Common Stock or voting securities beneficially owned by the Holder and its Affiliates and any other Persons whose beneficial ownership of Common Stock would be aggregated with the Holder’s for purposes of Section 13(d) of the Exchange Act shall include the shares of Common Stock issuable upon the exercise of this Warrant with respect to which such determination is being made, but shall exclude the number of shares of Common Stock which would be issuable upon (x) the exercise of the remaining unexercised and non-cancelled portion of this Warrant by the Holder and (y) the exercise or conversion of the unexercised, non-converted or non-cancelled portion of any other securities of the Company that do not have voting power (including without limitation (i) any securities of the Company which would entitle the holder thereof to acquire at any time Common Stock, and (ii) any debt, preferred stock, right, option, warrant or other instrument that is at any time convertible into or exercisable or exchangeable for, or otherwise entitles the holder thereof to receive, Common Stock), is subject to a limitation on conversion or exercise analogous to the limitation contained herein and is beneficially owned by the Holder or any of its Affiliates and other Persons whose beneficial ownership of Common Stock would be aggregated with the Holder’s for purposes of Section 13(d) of the Exchange Act.
(b) Notwithstanding anything to the contrary contained herein, including Section 11(a), the Company shall not effect any exercise of this Warrant, and the Holder shall not be entitled to exercise this Warrant for a number of Warrant Shares in excess of that number of Warrant Shares which, upon giving effect to such exercise, would cause (i) the aggregate number of shares of Common Stock beneficially owned by the Holder and its Affiliates and any other Persons whose beneficial ownership of Common Stock would be aggregated with the Holder’s for purposes of Section 13(d) of the Exchange Act, to exceed 19.99% of the total number of issued and outstanding shares of Common Stock of the Company following such exercise (if exceeding such limit would result in a change of control under Nasdaq Listing Rule 5635(b) or any successor rule), or (ii) the combined voting power of the securities of the Company beneficially owned by the Holder and its Affiliates and any other Persons whose beneficial ownership of Common Stock would be aggregated with the Holder’s for purposes of Section 13(d) of the Exchange Act to exceed 19.99% of the combined voting power of all of the securities of the Company then outstanding following such exercise (if exceeding such limit would result in a change of control under Nasdaq Listing Rule 5635(b) or any successor rule). For purposes of this Section 11(b), the aggregate number of shares of Common Stock or voting securities beneficially owned by the Holder and its Affiliates and any other Persons whose beneficial ownership of Common Stock would be aggregated with the Holder’s for purposes of Section 13(d) of the Exchange Act shall include the shares of Common Stock issuable upon the exercise of this Warrant with respect to which such determination is being made, but shall exclude the number of shares of Common Stock which would be issuable upon (x) the exercise of the remaining unexercised and non-cancelled portion of this Warrant by the Holder and (y) the exercise or conversion of the unexercised, non-converted or non-cancelled portion of any other securities of the Company that do not have voting power (including without limitation (i) any securities of the Company which would entitle the holder thereof to acquire at any time Common Stock, and (ii) any debt, preferred stock, right, option, warrant or other instrument that is at any time convertible into or exercisable or exchangeable for, or otherwise entitles the holder thereof to receive, Common Stock), is subject to a limitation on conversion or exercise analogous to the limitation contained herein and is beneficially owned by the Holder or any of its Affiliates and other Persons whose beneficial ownership of Common Stock would be aggregated with the Holder’s for purposes of Section 13(d) of the Exchange Act.
8
(c) This Section 11 shall not restrict the number of shares of Common Stock which a Holder may receive or beneficially own in order to determine the amount of securities or other consideration that such Holder may receive in the event of a Fundamental Transaction as contemplated in Section 9 of this Warrant.
12. No Fractional Shares. No fractional Warrant Shares will be issued in connection with any exercise of this Warrant. In lieu of any fractional shares that would otherwise be issuable, the number of Warrant Shares to be issued shall be rounded down to the next whole number and the Company shall pay the Holder in cash the fair market value (based on the Closing Sale Price) for any such fractional shares.
13. Notices. Any and all notices or other communications or deliveries hereunder (including, without limitation, any Exercise Notice) shall be in writing and shall be deemed given and effective on the earliest of (i) the date of transmission, if such notice or communication is delivered via facsimile or confirmed e-mail at the facsimile number or e-mail address specified below prior to 5:30 P.M., New York City time, on a Trading Day, (ii) the next Trading Day after the date of transmission, if such notice or communication is delivered via facsimile or confirmed e-mail at the facsimile number or e-mail address specified below on a day that is not a Trading Day or later than 5:30 P.M., New York City time, on any Trading Day, (iii) the Trading Day following the date of mailing, if sent by nationally recognized overnight courier service specifying next business day delivery, or (iv) upon actual receipt by the Person to whom such notice is required to be given, if by hand delivery.
14. Warrant Agent. The Company shall initially serve as warrant agent under this Warrant. Upon thirty (30) days’ notice to the Holder, the Company may appoint a new warrant agent. Any corporation into which the Company or any new warrant agent may be merged or any corporation resulting from any consolidation to which the Company or any new warrant agent shall be a party or any corporation to which the Company or any new warrant agent transfers substantially all of its corporate trust or shareholders services business shall be a successor warrant agent under this Warrant without any further act. Any such successor warrant agent shall promptly cause notice of its succession as warrant agent to be mailed (by first class mail, postage prepaid) to the Holder at the Holder’s last address as shown on the Warrant Register.
15. Miscellaneous.
(a) No Rights as a Stockholder. The Holder, solely in such Person’s capacity as a holder of this Warrant, shall not be entitled to vote or receive dividends or be deemed the holder of share capital of the Company for any purpose, nor shall anything contained in this Warrant be construed to confer upon the Holder, solely in such Person’s capacity as the Holder of this Warrant, any of the rights of a stockholder of the Company or any right to vote, give or withhold consent to any corporate action (whether any reorganization, issue of stock, reclassification of stock, consolidation, merger, amalgamation, conveyance or otherwise), receive notice of meetings, receive dividends or subscription rights, or otherwise, prior to the issuance to the Holder of the Warrant Shares which such Person is then entitled to receive upon the due exercise of this Warrant. In addition, nothing contained in this Warrant shall be construed as imposing any liabilities on the Holder to purchase any securities (upon exercise of this Warrant or otherwise) or as a stockholder of the Company, whether such liabilities are asserted by the Company or by creditors of the Company.
(b) Authorized Shares.
(i) Except and to the extent as waived or consented to by the Holder, the Company shall not by any action, including, without limitation, amending its certificate or articles of incorporation or through any reorganization, transfer of assets, consolidation, merger, dissolution, issue or sale of securities or any other voluntary action, avoid or seek to avoid the observance or performance of any of the terms of this Warrant, but will at all times in good faith assist in the carrying out of all such terms and in the taking of all such actions as may be necessary or appropriate to protect the rights of Holder as set forth in this Warrant against impairment.
9
Without limiting the generality of the foregoing, the Company will (a) not increase the par value of any Warrant Shares above the amount payable therefor upon such exercise immediately prior to such increase in par value, (b) take all such action as may be necessary or appropriate in order that the Company may validly and legally issue fully paid and nonassessable Warrant Shares upon the exercise of this Warrant, and (c) use commercially reasonable efforts to obtain all such authorizations, exemptions or consents from any public regulatory body having jurisdiction thereof as may be necessary to enable the Company to perform its obligations under this Warrant.
(ii) Before taking any action which would result in an adjustment in the number of Warrant Shares for which this Warrant is exercisable or in the Exercise Price, the Company shall obtain all such authorizations or exemptions thereof, or consents thereto, as may be necessary from any public regulatory body or bodies having jurisdiction thereof.
(c) Successors and Assigns. Subject to the restrictions on transfer set forth in this Warrant and the restrictions on transfer set forth in this Warrant and compliance with applicable securities laws, this Warrant may be assigned by the Holder. This Warrant may not be assigned by the Company without the written consent of the Holder except to a successor in the event of a Fundamental Transaction. This Warrant shall be binding on and inure to the benefit of the Company and the Holder and their respective successors and assigns. Subject to the preceding sentence, nothing in this Warrant shall be construed to give to any Person other than the Company and the Holder any legal or equitable right, remedy or cause of action under this Warrant. This Warrant may be amended only in writing signed by the Company and the Holder, or their successors and assigns.
(d) Amendment and Waiver. Except as otherwise provided herein, the provisions of this Warrant may be amended and the Company may take any action herein prohibited, or omit to perform any act herein required to be performed by it, only if the Company has obtained the written consent of the Holder.
(e) Acceptance. Receipt of this Warrant by the Holder shall constitute acceptance of and agreement to all of the terms and conditions contained herein.
(f) Governing Law; Jurisdiction. ALL QUESTIONS CONCERNING THE CONSTRUCTION, VALIDITY, ENFORCEMENT AND INTERPRETATION OF THIS WARRANT SHALL BE GOVERNED BY AND CONSTRUED AND ENFORCED IN ACCORDANCE WITH THE LAWS OF THE STATE OF NEW YORK WITHOUT REGARD TO THE PRINCIPLES OF CONFLICTS OF LAW THEREOF. EACH OF THE COMPANY AND THE HOLDER HEREBY IRREVOCABLY SUBMITS TO THE EXCLUSIVE JURISDICTION OF THE STATE AND FEDERAL COURTS SITTING IN THE CITY OF NEW YORK, BOROUGH OF MANHATTAN, FOR THE ADJUDICATION OF ANY DISPUTE HEREUNDER OR IN CONNECTION HEREWITH OR WITH ANY TRANSACTION CONTEMPLATED HEREBY OR DISCUSSED HEREIN (INCLUDING WITH RESPECT TO THE ENFORCEMENT OF ANY OF THE TRANSACTION DOCUMENTS), AND HEREBY IRREVOCABLY WAIVES, AND AGREES NOT TO ASSERT IN ANY SUIT, ACTION OR PROCEEDING, ANY CLAIM THAT IT IS NOT PERSONALLY SUBJECT TO THE JURISDICTION OF ANY SUCH COURT. EACH OF THE COMPANY AND THE HOLDER HEREBY IRREVOCABLY WAIVES PERSONAL SERVICE OF PROCESS AND CONSENTS TO PROCESS BEING SERVED IN ANY SUCH SUIT, ACTION OR PROCEEDING BY MAILING A COPY THEREOF VIA REGISTERED OR CERTIFIED MAIL OR OVERNIGHT DELIVERY (WITH EVIDENCE OF DELIVERY) TO SUCH PERSON AT THE ADDRESS IN EFFECT FOR NOTICES TO IT AND AGREES THAT SUCH SERVICE SHALL CONSTITUTE GOOD AND SUFFICIENT SERVICE OF PROCESS AND NOTICE THEREOF. NOTHING CONTAINED HEREIN SHALL BE DEEMED TO LIMIT IN ANY WAY ANY RIGHT TO SERVE PROCESS IN ANY MANNER PERMITTED BY LAW. EACH OF THE COMPANY AND THE HOLDER HEREBY WAIVES ALL RIGHTS TO A TRIAL BY JURY.
(g) Headings. The headings herein are for convenience only, do not constitute a part of this Warrant and shall not be deemed to limit or affect any of the provisions hereof.
(h) Severability. In case any one or more of the provisions of this Warrant shall be invalid or unenforceable in any respect, the validity and enforceability of the remaining terms and provisions of this Warrant shall not in any way be affected or impaired thereby, and the Company and the Holder will attempt in good faith to agree upon a valid and enforceable provision which shall be a commercially reasonable substitute therefor, and upon so agreeing, shall incorporate such substitute provision in this Warrant.
10
(i) Pre-Funded Warrants in lieu of Common Stock. Notwithstanding anything to the contrary in this Warrant, the Holder may choose, in lieu of receiving Warrant Shares upon exercise of this Warrant, to receive a pre-funded warrant in the form attached hereto as EXHIBIT A (the “Pre-Funded Warrant”) to purchase an identical number of shares of common stock it would have receive upon the exercise of this Warrant for shares of common stock, provided, however that the Exercise Price shall instead be the Exercise Price less $0.0001 per share, and the resulting issued Pre-Funded Warrant shall have an exercise price of $0.0001 per share.
[REMAINDER OF PAGE INTENTIONALLY LEFT BLANK]
11
IN WITNESS WHEREOF, the Company has caused this Warrant to be duly executed by its authorized officer as of the date first indicated above.
| FOGHORN THERAPEUTICS INC. | ||
| By: |
|
|
| Name: | ||
| Title: | ||
12
SCHEDULE 1
FORM OF EXERCISE NOTICE
[To be executed by the Holder to purchase shares of Common Stock under the Warrant]
Ladies and Gentlemen:
| (1) | The undersigned is the Holder of Warrant No. (the “Warrant”) issued by Foghorn Therapeutics Inc., a Delaware corporation (the “Company”). Capitalized terms used herein and not otherwise defined herein have the respective meanings set forth in the Warrant. | |
| (2) | The undersigned hereby exercises its right to purchase Warrant Shares pursuant to the Warrant. | |
| (3) | The Holder intends that payment of the Exercise Price shall be made as (check one): | |
| ☐ Cash Exercise |
||
| ☐ “Cashless Exercise” under Section 10 of the Warrant |
||
| (4) | If the Holder has elected a Cash Exercise, the Holder shall pay the sum of $ in immediately available funds to the Company in accordance with the terms of the Warrant. | |
| (5) | Except as set forth in paragraph (6), pursuant to this Exercise Notice, the Company shall deliver to the Holder Warrant Shares determined in accordance with the terms of the Warrant. | |
| (6) | The Holder hereby elects to receive a Pre-Funded Warrant (in the form attached hereto as EXHIBIT A) to purchase an identical number of shares of common stock that it would receive if it had exercised this Warrant for shares of Common Stock in lieu of receiving the Warrant Shares in accordance with this Warrant. ☐ | |
| (7) | By its delivery of this Exercise Notice, the undersigned represents and warrants to the Company that in giving effect to the exercise evidenced hereby the Holder will not beneficially own in excess of the number of shares of Common Stock (as determined in accordance with Section 13(d) of the Securities Exchange Act of 1934, as amended) permitted to be owned under Section 11(a) or Section 11(b), as applicable, of the Warrant to which this notice relates. | |
| Dated: |
|
|
| Name of Holder: |
|
|
| By: |
|
|
| Name: |
|
|
| Title: |
|
|
(Signature must conform in all respects to name of Holder as specified on the face of the Warrant)
13
Exhibit 4.2
FOGHORN THERAPEUTICS INC.
FORM OF PRE-FUNDED WARRANT TO PURCHASE COMMON STOCK
| Number of Shares: [ ] (subject to adjustment) |
||
| Warrant No. [ ] | Original Issue Date: [ ] |
Foghorn Therapeutics Inc., a Delaware corporation (the “Company”), hereby certifies that, for good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, [ ]. or its permitted registered assigns (the “Holder”), is entitled, subject to the terms set forth below, to purchase from the Company up to a total of [ ] shares of common stock, $0.0001 par value per share (the “Common Stock”), of the Company (each such share, a “Warrant Share” and all such shares, the “Warrant Shares”) at an exercise price per share equal to $0.0001 per share (the “Exercise Price”), upon surrender of this Warrant to Purchase Common Stock (including any Warrants to Purchase Common Stock issued in exchange, transfer or replacement hereof, the “Warrant”) at any time and from time to time on or after the date hereof (the “Original Issue Date”) and through and including 5:30 P.M., New York City time, on the date that is 20 years following the Original Issue Date (the “Expiration Date”), and subject to the following terms and conditions:
1. Definitions. For purposes of this Warrant, the following terms shall have the following meanings:
(a) “Affiliate” means any Person directly or indirectly controlled by, controlling or under common control with, a Holder, but only for so long as such control shall continue. For purposes of this definition, “control” (including, with correlative meanings, “controlled by,” “controlling” and “under common control with”) means, with respect to a Person, possession, direct or indirect, of (a) the power to direct or cause direction of the management and policies of such Person (whether through ownership of securities or partnership or other ownership interests, by contract or otherwise), or (b) at least 50% of the voting securities (whether directly or pursuant to any option, warrant or other similar arrangement) or other comparable equity interests.
(b) “Commission” means the United States Securities and Exchange Commission.
(c) “Closing Sale Price” means, for any security as of any date, the last trade price for such security on the Principal Trading Market for such security, as reported by Bloomberg Financial Markets, or, if such Principal Trading Market begins to operate on an extended hours basis and does not designate the last trade price, then the last trade price of such security prior to 4:00 P.M., New York City time, as reported by Bloomberg Financial Markets, or if the foregoing do not apply, the last trade price of such security in the over-the-counter market on the electronic bulletin board for such security as reported by Bloomberg Financial Markets. If the Closing Sale Price cannot be calculated for a security on a particular date on any of the foregoing bases, the Closing Sale Price of such security on such date shall be the fair market value as mutually determined by the Company and the Holder. If the Company and the Holder are unable to agree upon the fair market value of such security, then the Board of Directors of the Company shall use its good faith judgment to determine the fair market value. The Board of Directors’ determination shall be binding upon all parties absent demonstrable error. All such determinations shall be appropriately adjusted for any stock dividend, stock split, stock combination or other similar transaction during the applicable calculation period.
(d) “Exchange Act” means the Securities Exchange Act of 1934, as amended.
(e) “Principal Trading Market” means the national securities exchange or other trading market on which the Common Stock is primarily listed on and quoted for trading, which, as of the Original Issue Date, shall be The Nasdaq Global Market.
(f) “Registration Statement” means the Company’s Registration Statement on Form S-3 (File No. 333-284476), declared effective on January 31, 2025.
(g) “Securities Act” means the Securities Act of 1933, as amended.
(h) “Trading Day” means any weekday on which the Principal Trading Market is open for trading.
(i) “Transfer Agent” means Computershare Trust Company, N.A., the Company’s transfer agent and registrar for the Common Stock, and any successor appointed in such capacity.
2. Registration of Warrants. The Company shall register this Warrant, upon records to be maintained by the Company for that purpose (the “Warrant Register”), in the name of the record Holder (which shall include the initial Holder or, as the case may be, any registered assignee to which this Warrant is permissibly assigned hereunder) from time to time. The Company may deem and treat the registered Holder of this Warrant as the absolute owner hereof for the purpose of any exercise hereof or any distribution to the Holder, and for all other purposes, absent actual notice to the contrary.
3. Registration of Transfers. Subject to compliance with all applicable securities laws, the Company shall, or will cause its Transfer Agent to, register the transfer of all or any portion of this Warrant in the Warrant Register, upon surrender of this Warrant, and payment for all applicable transfer taxes (if any). Upon any such registration or transfer, a new warrant to purchase Common Stock in substantially the form of this Warrant (any such new warrant, a “New Warrant”) evidencing the portion of this Warrant so transferred shall be issued to the transferee, and a New Warrant evidencing the remaining portion of this Warrant not so transferred, if any, shall be issued to the transferring Holder. The acceptance of the New Warrant by the transferee thereof shall be deemed the acceptance by such transferee of all of the rights and obligations in respect of the New Warrant that the Holder has in respect of this Warrant. The Company shall, or will cause its Transfer Agent to, prepare, issue and deliver at the Company’s own expense any New Warrant under this Section 3. Until due presentment for registration of transfer, the Company may treat the registered Holder hereof as the owner and holder for all purposes, and the Company shall not be affected by any notice to the contrary.
4. Exercise and Duration of Warrants.
(a) All or any part of this Warrant shall be exercisable by the registered Holder in any manner permitted by Section 10 of this Warrant at any time and from time to time on or after the Original Issue Date and through and including 5:30 P.M. New York City time, on the Expiration Date. At 5:30 P.M., New York City time, on the Expiration Date, the portion of this Warrant not exercised prior thereto shall be and become void and of no value and this Warrant shall be terminated and no longer outstanding.
(b) The Holder may exercise this Warrant by delivering to the Company (i) an exercise notice, in the form attached as Schedule 1 hereto (the “Exercise Notice”), completed and duly signed, and (ii) payment of the Exercise Price for the number of Warrant Shares as to which this Warrant is being exercised (which may take the form of a “cashless exercise” if so indicated in the Exercise Notice pursuant to Section 10 below), and the date on which the last of such items is delivered to the Company (as determined in accordance with the notice provisions hereof) is an “Exercise Date.” The Holder shall not be required to deliver the original Warrant in order to effect an exercise hereunder. Execution and delivery of the Exercise Notice shall have the same effect as cancellation of the original Warrant and issuance of a New Warrant evidencing the right to purchase the remaining number of Warrant Shares.
5. Delivery of Warrant Shares.
(a) Upon exercise of this Warrant, the Company shall promptly (but in no event later than one Trading Day after the Exercise Date), upon the request of the Holder, credit such aggregate number of shares of Common Stock to which the Holder is entitled pursuant to such exercise to the Holder’s or its designee’s balance account with The Depository Trust Company (“DTC”) through its Deposit Withdrawal Agent Commission system, or if the Transfer Agent is not participating in the Fast Automated Securities Transfer Program (the “FAST Program”) or if the certificates are required to bear a legend regarding restriction on transferability, issue and dispatch by overnight courier to the address as specified in the Exercise Notice, a certificate, registered in the Company’s share register in the name of the Holder or its designee, for the number of shares of Common Stock to which the Holder is entitled pursuant to such exercise. The Holder, or any natural person or legal entity (each, a “Person”) permissibly so designated by the Holder to receive Warrant Shares, shall be deemed to have become the holder of record of such Warrant Shares as of the Exercise Date, irrespective of the date such Warrant Shares are credited to the Holder’s or its designee’s DTC account or the date of delivery of the certificates evidencing such Warrant Shares, as the case may be.
2
(b) If by the close of the Trading Day after the Exercise Date, the Company fails to deliver to the Holder a certificate representing the required number of Warrant Shares in the manner required pursuant to Section 5(a) or fails to credit the Holder’s or its designee’s balance account with DTC for such number of Warrant Shares to which the Holder is entitled, and if after such Trading Day and prior to the receipt of such Warrant Shares, the Holder purchases (in an open market transaction or otherwise) shares of Common Stock to deliver in satisfaction of a sale by the Holder of the Warrant Shares which the Holder anticipated receiving upon such exercise (a “Buy-In”), then the Company shall, within one Trading Day after the Holder’s request and in the Holder’s sole discretion, either (1) pay in cash to the Holder an amount equal to the Holder’s total purchase price (including brokerage commissions, if any) for the shares of Common Stock so purchased (the “Buy-In Price”), at which point the Company’s obligation to deliver such certificate (and to issue such Warrant Shares) shall terminate or (2) promptly honor its obligation to deliver to the Holder a certificate or certificates representing such Warrant Shares or credit the Holder’s or its designee’s balance account with DTC for such Warrant Shares and pay cash to the Holder in an amount equal to the excess (if any) of Holder’s total purchase price (including brokerage commissions, if any) for the shares of Common Stock so purchased in the Buy-In over the product of (A) the number of shares of Common Stock purchased in the Buy-In, times (B) the Closing Sale Price of a share of Common Stock on the Exercise Date.
(c) To the extent permitted by law and subject to Section 5(b), the Company’s obligations to issue and deliver Warrant Shares in accordance with and subject to the terms hereof (including the limitations set forth in Section 11 below) are absolute and unconditional, irrespective of any action or inaction by the Holder to enforce the same, any waiver or consent with respect to any provision hereof, the recovery of any judgment against any Person or any action to enforce the same, or any setoff, counterclaim, recoupment, limitation or termination, or any breach or alleged breach by the Holder or any other Person of any obligation to the Company or any violation or alleged violation of law by the Holder or any other Person, and irrespective of any other circumstance that might otherwise limit such obligation of the Company to the Holder in connection with the issuance of Warrant Shares. Subject to Section 5(b), nothing herein shall limit the Holder’s right to pursue any other remedies available to it hereunder, at law or in equity including, without limitation, a decree of specific performance and/or injunctive relief with respect to the Company’s failure to timely deliver certificates representing shares of Common Stock upon exercise of the Warrant as required pursuant to the terms hereof.
6. Charges, Taxes and Expenses. Issuance and delivery of certificates for shares of Common Stock upon exercise of this Warrant shall be made without charge to the Holder for any issue or transfer tax, transfer agent fee or other incidental tax or expense (excluding any applicable stamp duties) in respect of the issuance of such certificates, all of which taxes and expenses shall be paid by the Company; provided, however, that the Company shall not be required to pay any tax that may be payable in respect of any transfer involved in the registration of any certificates for Warrant Shares or the Warrants in a name other than that of the Holder or an Affiliate thereof. The Holder shall be responsible for all other tax liability that may arise as a result of holding or transferring this Warrant or receiving Warrant Shares upon exercise hereof.
7. Replacement of Warrant. If this Warrant is mutilated, lost, stolen or destroyed, the Company shall issue or cause to be issued in exchange and substitution for and upon cancellation hereof, or in lieu of and substitution for this Warrant, a New Warrant, but only upon receipt of evidence reasonably satisfactory to the Company of such loss, theft or destruction (in such case) and, in each case, a customary and reasonable indemnity and surety bond, if requested by the Company. Applicants for a New Warrant under such circumstances shall also comply with such other reasonable regulations and procedures and pay such other reasonable third-party costs as the Company may prescribe. If a New Warrant is requested as a result of a mutilation of this Warrant, then the Holder shall deliver such mutilated Warrant to the Company as a condition precedent to the Company’s obligation to issue the New Warrant.
8. Reservation of Warrant Shares. The Company covenants that it will at all times while this Warrant is outstanding reserve and keep available out of the aggregate of its authorized but unissued and otherwise unreserved Common Stock, solely for the purpose of enabling it to issue Warrant Shares upon exercise of this Warrant as herein provided, the number of Warrant Shares that are initially issuable and deliverable upon the exercise of this entire Warrant, free from preemptive rights or any other contingent purchase rights of persons other than the Holder (taking into account the adjustments and restrictions of Section 9).
3
The Company covenants that all Warrant Shares so issuable and deliverable shall, upon issuance and the payment of the applicable Exercise Price in accordance with the terms hereof, be duly and validly authorized, issued and fully paid and nonassessable. The Company will take all such action as may be reasonably necessary to assure that such shares of Common Stock may be issued as provided herein without violation of any applicable law or regulation, or of any requirements of any securities exchange or automated quotation system upon which the Common Stock may be listed.
9. Certain Adjustments. The number of Warrant Shares issuable upon exercise of this Warrant is subject to adjustment from time to time as set forth in this Section 9.
(a) Stock Dividends and Splits. If the Company, at any time while this Warrant is outstanding, (i) pays a stock dividend on its Common Stock or otherwise makes a distribution on any class of capital stock issued and outstanding on the Original Issue Date and in accordance with the terms of such stock on the Original Issue Date or as amended, that is payable in shares of Common Stock, (ii) subdivides its outstanding shares of Common Stock into a larger number of shares of Common Stock, (iii) combines its outstanding shares of Common Stock into a smaller number of shares of Common Stock or (iv) issues by reclassification of shares of capital stock any additional shares of Common Stock of the Company, then in each such case the number of Warrant Shares then underlying this Warrant shall be divided by a fraction, the numerator of which shall be the number of shares of Common Stock outstanding immediately before such event and the denominator of which shall be the number of shares of Common Stock outstanding immediately after such event. Any adjustment made pursuant to clause (i) of this paragraph shall become effective immediately after the record date for the determination of stockholders entitled to receive such dividend or distribution, provided, however, that if such record date shall have been fixed and such dividend is not fully paid on the date fixed therefor, the number of Warrant Shares shall be recomputed accordingly as of the close of business on such record date and thereafter the Warrant Shares shall be adjusted pursuant to this paragraph as of the time of actual payment of such dividends. Any adjustment pursuant to clause (ii) or (iii) of this paragraph shall become effective immediately after the effective date of such subdivision or combination.
(b) Pro Rata Distributions. If the Company, at any time while this Warrant is outstanding, distributes to all holders of Common Stock for no consideration (i) evidences of its indebtedness, (ii) any security (other than a distribution of Common Stock covered by the preceding paragraph), (iii) rights or warrants to subscribe for or purchase any security, or (iv) cash or any other asset (in each case, “Distributed Property”), then, upon any exercise of this Warrant that occurs after the record date fixed for determination of stockholders entitled to receive such distribution, the Holder shall be entitled to receive, in addition to the Warrant Shares otherwise issuable upon such exercise (if applicable), the Distributed Property that such Holder would have been entitled to receive in respect of such number of Warrant Shares had the Holder been the record holder of such Warrant Shares immediately prior to such record date without regard to any limitation on exercise contained therein.
(c) Purchase Rights. In addition to any adjustments pursuant to the other subsections of this Section 9, if at any time on or after the Original Issue Date, the Company grants, issues or sells any Options, Convertible Securities or rights to purchase stock, warrants, securities or other property, in each case pro rata to the record holders of any class of Common Stock (the “Purchase Rights”), then the Holder will be entitled to acquire, upon the terms applicable to such Purchase Rights, the aggregate Purchase Rights which the Holder could have acquired if the Holder had held the number of shares of Common Stock acquirable upon complete exercise of this Warrant (without regard to any limitations or restrictions on exercise of this Warrant, including without limitation, the Maximum Percentage) immediately before the date on which a record is taken for the grant, issuance or sale of such Purchase Rights, or, if no such record is taken, the date as of which the record holders of Common Stock are to be determined for the grant, issuance or sale of such Purchase Rights (provided, that to the extent that the Holder’s right to participate in any such Purchase Right would result in the Holder and its Affiliates exceeding the Maximum Percentage, then the Holder shall not be entitled to participate in such Purchase Right to such extent (and shall not be entitled to beneficial ownership of such Common Stock as a result of such Purchase Right (and beneficial ownership) to such extent) and such Purchase Right to such extent shall be held in abeyance for the benefit of the Holder until such time or times as its right thereto would not result in the Holder and Affiliates exceeding the Maximum Percentage, at which time or times the Holder shall be granted such right (and any Purchase Right granted, issued or sold on such initial Purchase Right or on any subsequent Purchase Right to be held similarly in abeyance) to the same extent as if there had been no such limitation). As used in this Section 9(c), (i) “Options” means any rights, warrants or options to subscribe for or purchase shares of Common Stock or Convertible Securities and (ii) “Convertible Securities” mean any stock or securities (other than Options) directly or indirectly convertible into or exercisable or exchangeable for shares of Common Stock.
4
(d) Fundamental Transactions. If, at any time while this Warrant is outstanding (i) the Company effects any merger or consolidation of the Company with or into another Person, in which the Company is not the surviving entity or the stockholders of the Company immediately prior to such merger or consolidation do not own, directly or indirectly, at least 50% of the voting power of the surviving entity immediately after such merger or consolidation, (ii) the Company effects any sale to another Person of all or substantially all of its assets in one transaction or a series of related transactions, (iii) pursuant to any tender offer or exchange offer (whether by the Company or another Person), holders of capital stock who tender shares representing more than 50% of the voting power of the capital stock of the Company and the Company or such other Person, as applicable, accepts such tender for payment, (iv) the Company consummates a stock purchase agreement or other business combination (including, without limitation, a reorganization, recapitalization, spin-off or scheme of arrangement) with another Person whereby such other Person acquires more than the 50% of the voting power of the capital stock of the Company (except for any such transaction in which the stockholders of the Company immediately prior to such transaction maintain, in substantially the same proportions, the voting power of such Person immediately after the transaction) or (v) the Company effects any reclassification of the Common Stock or any compulsory share exchange pursuant to which the Common Stock is effectively converted into or exchanged for other securities, cash or property (other than as a result of a subdivision or combination of shares of Common Stock covered by Section 9(a) above) (in any such case, a “Fundamental Transaction”), then following such Fundamental Transaction the Holder shall have the right to receive, upon exercise of this Warrant, the same amount and kind of securities, cash or property as it would have been entitled to receive upon the occurrence of such Fundamental Transaction if it had been, immediately prior to such Fundamental Transaction, the holder of the number of Warrant Shares then issuable upon exercise in full of this Warrant without regard to any limitations on exercise contained herein (the “Alternate Consideration”). The Company shall not effect any Fundamental Transaction in which the Company is not the surviving entity or the Alternate Consideration includes securities of another Person unless (i) the Alternate Consideration is solely cash and the Company provides for the simultaneous “cashless exercise” of this Warrant pursuant to Section 10 below or (ii) prior to or simultaneously with the consummation thereof, any successor to the Company, surviving entity or other Person (including any purchaser of assets of the Company) shall assume the obligation to deliver to the Holder, such Alternate Consideration as, in accordance with the foregoing provisions, the Holder may be entitled to receive, and the other obligations under this Warrant. The provisions of this paragraph (d) shall similarly apply to subsequent transactions analogous of a Fundamental Transaction type.
(e) Calculations. All calculations under this Section 9 shall be made to the nearest share.
(f) Notice of Adjustments. Upon the occurrence of each adjustment pursuant to this Section 9, the Company at its expense will, at the written request of the Holder, promptly compute such adjustment, in good faith, in accordance with the terms of this Warrant and prepare a certificate setting forth such adjustment, including a statement of the adjusted number or type of Warrant Shares or other securities issuable upon exercise of this Warrant (as applicable), describing the transactions giving rise to such adjustments and showing in detail the facts upon which such adjustment is based. Upon written request, the Company will promptly deliver a copy of each such certificate to the Holder and to the Company’s transfer agent.
(g) Notice of Corporate Events. If, while this Warrant is outstanding, the Company (i) declares a dividend or any other pro rata distribution of cash, securities or other property in respect of its Common Stock, including, without limitation, any granting of rights or warrants to subscribe for or purchase any capital stock of the Company or any subsidiary, (ii) authorizes or approves, enters into any agreement contemplating or solicits stockholder approval for any Fundamental Transaction or (iii) authorizes the voluntary dissolution, liquidation or winding up of the affairs of the Company, then, except if such notice and the contents thereof shall be deemed to constitute material non-public information, the Company shall deliver to the Holder a notice of such transaction at least ten (10) days prior to the applicable record or effective date on which a Person would need to hold Common Stock in order to participate in or vote with respect to such transaction; provided, however, that the failure to deliver such notice or any defect therein shall not affect the validity of the corporate action required to be described in such notice. In addition, if while this Warrant is outstanding, the Company authorizes or approves, enters into any agreement contemplating or solicits stockholder approval for any Fundamental Transaction contemplated by Section 9(d), other than a Fundamental Transaction under clause (iii) of Section 9(d), the Company shall deliver to the Holder a notice of such Fundamental Transaction at least thirty (30) days prior to the date such Fundamental Transaction is consummated. Holder agrees to maintain any information disclosed pursuant to this Section 9(g) in confidence until such information is publicly available, and shall comply with applicable law with respect to trading in the Company’s securities following receipt of any such information.
5
10. Payment of Exercise Price. Notwithstanding anything contained herein to the contrary, the Holder may, in its sole discretion, satisfy its obligation to pay the Exercise Price through a “cashless exercise,” in which event the Company shall issue to the Holder the number of Warrant Shares determined as follows:
X = Y [(A-B)/A]
where:
“X” equals the number of Warrant Shares to be issued to the Holder;
“Y” equals the total number of Warrant Shares with respect to which this Warrant is then being exercised;
“A” equals the average of the Closing Sale Prices of the shares of Common Stock (as reported by Bloomberg Financial Markets) for the five (5) consecutive Trading Days ending on the date immediately preceding the Exercise Date ; and
“B” equals the Exercise Price then in effect for the applicable Warrant Shares at the time of such exercise.
For purposes of Rule 144 promulgated under the Securities Act, it is intended, understood and acknowledged that the Warrant Shares issued in a “cashless exercise” transaction shall be deemed to have been acquired by the Holder, and the holding period for the Warrant Shares shall be deemed to have commenced, on the date this Warrant was originally issued (provided that the Commission continues to take the position that such treatment is proper at the time of such exercise). Except as set forth in Section 5(b) (Buy-In remedy) and Section 12 (payment of cash in lieu of fractional shares), in no event will the exercise of this Warrant be settled in cash.
In the event that the Registration Statement, or another registration statement registering the issuance of the Warrant Shares is, for any reason, not effective at the time of exercise of this Warrant, then the Warrant may only be exercised through a cashless exercise, as set forth in this Section 10. If Warrant Shares are issued in such a cashless exercise, the Company acknowledges and agrees that in accordance with Section 3(a)(9) of the Securities Act of 1933, as amended, the Warrant Shares shall take on the registered characteristics of the Warrants being exercised, and the holding period of the Warrants being exercised may be tacked on to the holding period of the Warrant Shares.
11. Limitations on Exercise.
(a) Notwithstanding anything to the contrary contained herein, the number of Warrant Shares that may be acquired by the Holder upon any exercise of this Warrant (or otherwise in respect hereof) shall be limited to the extent necessary to ensure that, following such exercise, the total number of shares of Common Stock then beneficially owned by the Holder and its Affiliates and any other Persons whose beneficial ownership of Common Stock would be aggregated with the Holder’s for purposes of Section 13(d) of the Exchange Act, does not exceed [9.99%][19.99% (if exceeding such limit would result in a change of control under Nasdaq Listing Rule 5635(b) or any successor rule)] of the total number of then issued and outstanding shares of Common Stock (including for such purpose the shares of Common Stock issuable upon such exercise) (the “Maximum Percentage”), it being acknowledged by the Holder that the Company is not representing to such Holder that such calculation is in compliance with Section 13(d) of the Exchange Act and such Holder is solely responsible for any schedules required to be filed in accordance therewith.
6
To the extent that the limitation contained in this Section 11(a) applies, the determination of whether this Warrant is exercisable (in relation to other securities owned by such Holder) and of which a portion of this Warrant is exercisable shall be in the sole discretion of the Holder, and the submission of a Notice of Exercise shall be deemed to be the Holder’s determination of whether this Warrant is exercisable (in relation to other securities owned by such Holder) and of which portion of this Warrant is exercisable, in each case subject to such aggregate percentage limitation, and the Company shall have no obligation to verify or confirm the accuracy of such determination. In addition, a determination under this Section 11(a) as to any group status shall be determined in accordance with Section 13(d) of the Exchange Act and the rules and regulations promulgated thereunder. For purposes of this Section 11(a), in determining the number of outstanding shares of Common Stock, the Holder may rely on the number of outstanding shares of Common Stock as reflected in (x) the Company’s most recent Form 10-Q or Form 10-K, as the case may be, (y) a more recent public announcement by the Company or (z) any other notice to the Holder by the Company or the Transfer Agent setting forth the number of shares of Common Stock outstanding. Upon the written request of the Holder, the Company shall, within three (3) Trading Days, confirm orally and in writing to such Holder the number of shares of Common Stock then outstanding. By written notice to the Company, which will not be effective until the sixty-first (61st) day after such notice is delivered to the Company, the Holder may waive the provisions of this Section 11(a) or increase the beneficial ownership limitation to such percentage of the number of shares of the Common Stock outstanding immediately after giving effect to the issuance of shares of Common Stock upon exercise of this Warrant as the Holder shall determine, in its sole discretion, subject to Section 11(b), and the provisions of this Section 11(a) shall continue to apply. Upon such a change by a Holder of the beneficial ownership limitation from such 9.99% limitation to such other percentage limitation, the beneficial ownership limitation may not be further waived or increased by such Holder without first providing the minimum notice required by this Section 11(a). Notwithstanding the foregoing, at any time following notice of a Fundamental Transaction under Section 9(f)(ii) with respect to a Section 9(d)(iii) Fundamental Transaction, the Holder may waive and/or change the beneficial ownership limitation effective immediately upon written notice to the Company and may reinstitute a beneficial ownership limitation at any time thereafter effective immediately upon written notice to the Company. For purposes of this Section 11(a), the aggregate number of shares of Common Stock or voting securities beneficially owned by the Holder and its Affiliates and any other Persons whose beneficial ownership of Common Stock would be aggregated with the Holder’s for purposes of Section 13(d) of the Exchange Act shall include the shares of Common Stock issuable upon the exercise of this Warrant with respect to which such determination is being made, but shall exclude the number of shares of Common Stock which would be issuable upon (x) the exercise of the remaining unexercised and non-cancelled portion of this Warrant by the Holder and (y) the exercise or conversion of the unexercised, non-converted or non-cancelled portion of any other securities of the Company that do not have voting power (including without limitation (i) any securities of the Company which would entitle the holder thereof to acquire at any time Common Stock, and (ii) any debt, preferred stock, right, option, warrant or other instrument that is at any time convertible into or exercisable or exchangeable for, or otherwise entitles the holder thereof to receive, Common Stock), is subject to a limitation on conversion or exercise analogous to the limitation contained herein and is beneficially owned by the Holder or any of its Affiliates and other Persons whose beneficial ownership of Common Stock would be aggregated with the Holder’s for purposes of Section 13(d) of the Exchange Act.
(b) Notwithstanding anything to the contrary contained herein, including Section 11(a), the Company shall not effect any exercise of this Warrant, and the Holder shall not be entitled to exercise this Warrant for a number of Warrant Shares in excess of that number of Warrant Shares which, upon giving effect to such exercise, would cause (i) the aggregate number of shares of Common Stock beneficially owned by the Holder and its Affiliates and any other Persons whose beneficial ownership of Common Stock would be aggregated with the Holder’s for purposes of Section 13(d) of the Exchange Act, to exceed 19.99% of the total number of issued and outstanding shares of Common Stock of the Company following such exercise (if exceeding such limit would result in a change of control under Nasdaq Listing Rule 5635(b) or any successor rule), or (ii) the combined voting power of the securities of the Company beneficially owned by the Holder and its Affiliates and any other Persons whose beneficial ownership of Common Stock would be aggregated with the Holder’s for purposes of Section 13(d) of the Exchange Act to exceed 19.99% of the combined voting power of all of the securities of the Company then outstanding following such exercise (if exceeding such limit would result in a change of control under Nasdaq Listing Rule 5635(b) or any successor rule). For purposes of this Section 11(b), the aggregate number of shares of Common Stock or voting securities beneficially owned by the Holder and its Affiliates and any other Persons whose beneficial ownership of Common Stock would be aggregated with the Holder’s for purposes of Section 13(d) of the Exchange Act shall include the shares of Common Stock issuable upon the exercise of this Warrant with respect to which such determination is being made, but shall exclude the number of shares of Common Stock which would be issuable upon (x) the exercise of the remaining unexercised and non-cancelled portion of this Warrant by the Holder and (y)
7
the exercise or conversion of the unexercised, non-converted or non-cancelled portion of any other securities of the Company that do not have voting power (including without limitation (i) any securities of the Company which would entitle the holder thereof to acquire at any time Common Stock, and (ii) any debt, preferred stock, right, option, warrant or other instrument that is at any time convertible into or exercisable or exchangeable for, or otherwise entitles the holder thereof to receive, Common Stock), is subject to a limitation on conversion or exercise analogous to the limitation contained herein and is beneficially owned by the Holder or any of its Affiliates and other Persons whose beneficial ownership of Common Stock would be aggregated with the Holder’s for purposes of Section 13(d) of the Exchange Act.
(c) This Section 11 shall not restrict the number of shares of Common Stock which a Holder may receive or beneficially own in order to determine the amount of securities or other consideration that such Holder may receive in the event of a Fundamental Transaction as contemplated in Section 9 of this Warrant.
12. No Fractional Shares. No fractional Warrant Shares will be issued in connection with any exercise of this Warrant. In lieu of any fractional shares that would otherwise be issuable, the number of Warrant Shares to be issued shall be rounded down to the next whole number and the Company shall pay the Holder in cash the fair market value (based on the Closing Sale Price) for any such fractional shares.
13. Notices. Any and all notices or other communications or deliveries hereunder (including, without limitation, any Exercise Notice) shall be in writing and shall be deemed given and effective on the earliest of (i) the date of transmission, if such notice or communication is delivered via facsimile or confirmed e-mail at the facsimile number or e-mail address specified below prior to 5:30 P.M., New York City time, on a Trading Day, (ii) the next Trading Day after the date of transmission, if such notice or communication is delivered via facsimile or confirmed e-mail at the facsimile number or e-mail address specified below on a day that is not a Trading Day or later than 5:30 P.M., New York City time, on any Trading Day, (iii) the Trading Day following the date of mailing, if sent by nationally recognized overnight courier service specifying next business day delivery, or (iv) upon actual receipt by the Person to whom such notice is required to be given, if by hand delivery.
14. Warrant Agent. The Company shall initially serve as warrant agent under this Warrant. Upon thirty (30) days’ notice to the Holder, the Company may appoint a new warrant agent. Any corporation into which the Company or any new warrant agent may be merged or any corporation resulting from any consolidation to which the Company or any new warrant agent shall be a party or any corporation to which the Company or any new warrant agent transfers substantially all of its corporate trust or shareholders services business shall be a successor warrant agent under this Warrant without any further act. Any such successor warrant agent shall promptly cause notice of its succession as warrant agent to be mailed (by first class mail, postage prepaid) to the Holder at the Holder’s last address as shown on the Warrant Register.
15. Miscellaneous.
(a) No Rights as a Stockholder. The Holder, solely in such Person’s capacity as a holder of this Warrant, shall not be entitled to vote or receive dividends or be deemed the holder of share capital of the Company for any purpose, nor shall anything contained in this Warrant be construed to confer upon the Holder, solely in such Person’s capacity as the Holder of this Warrant, any of the rights of a stockholder of the Company or any right to vote, give or withhold consent to any corporate action (whether any reorganization, issue of stock, reclassification of stock, consolidation, merger, amalgamation, conveyance or otherwise), receive notice of meetings, receive dividends or subscription rights, or otherwise, prior to the issuance to the Holder of the Warrant Shares which such Person is then entitled to receive upon the due exercise of this Warrant. In addition, nothing contained in this Warrant shall be construed as imposing any liabilities on the Holder to purchase any securities (upon exercise of this Warrant or otherwise) or as a stockholder of the Company, whether such liabilities are asserted by the Company or by creditors of the Company.
(b) Authorized Shares.
(i) Except and to the extent as waived or consented to by the Holder, the Company shall not by any action, including, without limitation, amending its certificate or articles of incorporation or through any reorganization, transfer of assets, consolidation, merger, dissolution, issue or sale of securities or any other voluntary action, avoid or seek to avoid the observance or performance of any of the terms of this Warrant, but will at all times in good faith assist in the carrying out of all such terms and in the taking of all such actions as may be necessary or appropriate to protect the rights of Holder as set forth in this Warrant against impairment.
8
Without limiting the generality of the foregoing, the Company will (a) not increase the par value of any Warrant Shares above the amount payable therefor upon such exercise immediately prior to such increase in par value, (b) take all such action as may be necessary or appropriate in order that the Company may validly and legally issue fully paid and nonassessable Warrant Shares upon the exercise of this Warrant, and (c) use commercially reasonable efforts to obtain all such authorizations, exemptions or consents from any public regulatory body having jurisdiction thereof as may be necessary to enable the Company to perform its obligations under this Warrant.
(ii) Before taking any action which would result in an adjustment in the number of Warrant Shares for which this Warrant is exercisable or in the Exercise Price, the Company shall obtain all such authorizations or exemptions thereof, or consents thereto, as may be necessary from any public regulatory body or bodies having jurisdiction thereof.
(c) Successors and Assigns. Subject to the restrictions on transfer set forth in this Warrant and the restrictions on transfer set forth in this Warrant and compliance with applicable securities laws, this Warrant may be assigned by the Holder. This Warrant may not be assigned by the Company without the written consent of the Holder except to a successor in the event of a Fundamental Transaction. This Warrant shall be binding on and inure to the benefit of the Company and the Holder and their respective successors and assigns. Subject to the preceding sentence, nothing in this Warrant shall be construed to give to any Person other than the Company and the Holder any legal or equitable right, remedy or cause of action under this Warrant. This Warrant may be amended only in writing signed by the Company and the Holder, or their successors and assigns.
(d) Amendment and Waiver. Except as otherwise provided herein, the provisions of this Warrant may be amended and the Company may take any action herein prohibited, or omit to perform any act herein required to be performed by it, only if the Company has obtained the written consent of the Holder.
(e) Acceptance. Receipt of this Warrant by the Holder shall constitute acceptance of and agreement to all of the terms and conditions contained herein.
(f) Governing Law; Jurisdiction. ALL QUESTIONS CONCERNING THE CONSTRUCTION, VALIDITY, ENFORCEMENT AND INTERPRETATION OF THIS WARRANT SHALL BE GOVERNED BY AND CONSTRUED AND ENFORCED IN ACCORDANCE WITH THE LAWS OF THE STATE OF NEW YORK WITHOUT REGARD TO THE PRINCIPLES OF CONFLICTS OF LAW THEREOF. EACH OF THE COMPANY AND THE HOLDER HEREBY IRREVOCABLY SUBMITS TO THE EXCLUSIVE JURISDICTION OF THE STATE AND FEDERAL COURTS SITTING IN THE CITY OF NEW YORK, BOROUGH OF MANHATTAN, FOR THE ADJUDICATION OF ANY DISPUTE HEREUNDER OR IN CONNECTION HEREWITH OR WITH ANY TRANSACTION CONTEMPLATED HEREBY OR DISCUSSED HEREIN (INCLUDING WITH RESPECT TO THE ENFORCEMENT OF ANY OF THE TRANSACTION DOCUMENTS), AND HEREBY IRREVOCABLY WAIVES, AND AGREES NOT TO ASSERT IN ANY SUIT, ACTION OR PROCEEDING, ANY CLAIM THAT IT IS NOT PERSONALLY SUBJECT TO THE JURISDICTION OF ANY SUCH COURT. EACH OF THE COMPANY AND THE HOLDER HEREBY IRREVOCABLY WAIVES PERSONAL SERVICE OF PROCESS AND CONSENTS TO PROCESS BEING SERVED IN ANY SUCH SUIT, ACTION OR PROCEEDING BY MAILING A COPY THEREOF VIA REGISTERED OR CERTIFIED MAIL OR OVERNIGHT DELIVERY (WITH EVIDENCE OF DELIVERY) TO SUCH PERSON AT THE ADDRESS IN EFFECT FOR NOTICES TO IT AND AGREES THAT SUCH SERVICE SHALL CONSTITUTE GOOD AND SUFFICIENT SERVICE OF PROCESS AND NOTICE THEREOF. NOTHING CONTAINED HEREIN SHALL BE DEEMED TO LIMIT IN ANY WAY ANY RIGHT TO SERVE PROCESS IN ANY MANNER PERMITTED BY LAW. EACH OF THE COMPANY AND THE HOLDER HEREBY WAIVES ALL RIGHTS TO A TRIAL BY JURY.
(g) Headings. The headings herein are for convenience only, do not constitute a part of this Warrant and shall not be deemed to limit or affect any of the provisions hereof.
9
(h) Severability. In case any one or more of the provisions of this Warrant shall be invalid or unenforceable in any respect, the validity and enforceability of the remaining terms and provisions of this Warrant shall not in any way be affected or impaired thereby, and the Company and the Holder will attempt in good faith to agree upon a valid and enforceable provision which shall be a commercially reasonable substitute therefor, and upon so agreeing, shall incorporate such substitute provision in this Warrant.
[REMAINDER OF PAGE INTENTIONALLY LEFT BLANK]
10
IN WITNESS WHEREOF, the Company has caused this Warrant to be duly executed by its authorized officer as of the date first indicated above.
| FOGHORN THERAPEUTICS INC. | ||
| By: |
|
|
| Name: | ||
| Title: | ||
[Signature Page to Pre-Funded Warrant]
SCHEDULE 1
FORM OF EXERCISE NOTICE
[To be executed by the Holder to purchase shares of Common Stock under the Warrant]
Ladies and Gentlemen:
| (1) | The undersigned is the Holder of Warrant No. (the “Warrant”) issued by Foghorn Therapeutics Inc., a Delaware corporation (the “Company”). Capitalized terms used herein and not otherwise defined herein have the respective meanings set forth in the Warrant. |
| (2) | The undersigned hereby exercises its right to purchase Warrant Shares pursuant to the Warrant. |
| (3) | The Holder intends that payment of the Exercise Price shall be made as (check one): |
| ☐ | Cash Exercise |
| ☐ | “Cashless Exercise” under Section 10 of the Warrant |
| (4) | If the Holder has elected a Cash Exercise, the Holder shall pay the sum of $ in immediately available funds to the Company in accordance with the terms of the Warrant. |
| (5) | Pursuant to this Exercise Notice, the Company shall deliver to the Holder Warrant Shares determined in accordance with the terms of the Warrant. |
| (6) | By its delivery of this Exercise Notice, the undersigned represents and warrants to the Company that in giving effect to the exercise evidenced hereby the Holder will not beneficially own in excess of the number of shares of Common Stock (as determined in accordance with Section 13(d) of the Securities Exchange Act of 1934, as amended) permitted to be owned under Section 11(a) or Section 11(b), as applicable, of the Warrant to which this notice relates. |
| Dated: |
|
|
| Name of Holder: |
|
|
| By: |
|
|
| Name: |
|
|
| Title: |
|
|
(Signature must conform in all respects to name of Holder as specified on the face of the Warrant)
12
Exhibit 5.1

|
ROPES & GRAY LLP PRUDENTIAL TOWER 800 BOYLSTON STREET BOSTON, MA 02199-3600 WWW.ROPESGRAY.COM |
January 12, 2026
Foghorn Therapeutics Inc.
99 Coolidge Ave, Suite 500
Watertown, MA 024729
Re: Registration Statement on Form S-3 (File No. 333-284476)
Ladies and Gentlemen:
We have acted as counsel to Foghorn Therapeutics Inc., a Delaware corporation (the “Company”), in connection with the issuance and sale of (i) 2,030,314 shares (the “Shares”) of the common stock, $0.0001 par value (“Common Stock”), of the Company, (ii) pre-funded warrants (the “Pre-Funded Warrants”) to purchase 5,421,250 shares of Common Stock (the “Pre-Funded Warrant Shares”), and (iii) Series 1 Warrants (the “Series 1 Warrants”) and Series 2 Warrants (the “Series 2 Warrants”) to purchase an aggregate of 7,451,564 shares of Common Stock (the “Series Warrant Shares”), or, at the option of the Investor (as defined below), in lieu of the Series Warrant Shares, pre-funded warrants to purchase shares of Common Stock (the “Underlying Pre-Funded Warrants” and, together with the Pre-Funded Warrants, the Series 1 Warrants and the Series 2 Warrants, the “Warrants” and the shares issuable pursuant to the Underlying Pre-Funded Warrants, together with the Pre-Funded Warrant Shares and the Series Warrant Shares, the “Warrant Shares”), pursuant to the above-referenced registration statement (the “Registration Statement”) filed by the Company with the Securities and Exchange Commission (the “Commission”) under the Securities Act of 1933, as amended (the “Securities Act”). The Shares and Warrants are being sold pursuant to securities purchase agreements, dated January 9, 2026 (the “Securities Purchase Agreements”), among the Company and the investors set forth on the signature pages thereto (collectively, the “Investors” and each, an “Investor”).
In connection with this opinion letter, we have examined such certificates, documents and records and have made such investigation of fact and such examination of law as we have deemed appropriate in order to enable us to render the opinions set forth herein. In conducting such investigation, we have relied, without independent verification, upon certificates of officers of the Company, public officials and other appropriate persons.
The opinions expressed below are limited to the Delaware General Corporation Law.
Based upon and subject to the foregoing, we are of the opinion that (i) the Shares have been duly authorized and, when issued and delivered pursuant to the Securities Purchase Agreements against payment of the consideration set forth therein, will be validly issued, fully paid and non-assessable, (ii) provided that the Warrants have been duly executed and delivered by the Company and duly delivered to the purchaser thereof against payment therefor, then the Warrants, when issued and sold pursuant to the Securities Purchase Agreements, will be a valid and legally binding obligation of the Company, enforceable against the Company in accordance with its terms, except as enforcement thereof may be limited by bankruptcy, insolvency, reorganization, moratorium or other similar laws relating to or affecting creditors’ rights generally and by general equitable principles (regardless of whether such enforceability is considered in a proceeding at law or in equity) and implied covenants of good faith and fair dealing, and (iii) the Warrant Shares, when issued upon exercise of the Warrants in accordance with their respective terms and assuming a sufficient number of Warrant Shares are at the time available for issuance, will have been duly authorized and validly issued and will be fully paid and non-assessable.
We hereby consent to your filing this opinion as an exhibit to a Current Report on Form 8-K to be incorporated by reference into the Registration Statement and to the use of our name under the caption “Legal Matters” in the Registration Statement. In giving such consent, we do not thereby admit that we are in the category of persons whose consent is required under Section 7 of the Securities Act or the rules and regulations of the Commission thereunder.
Very truly yours,
/s/ Ropes & Gray LLP
Ropes & Gray LLP
Exhibit 99.1
Foghorn Therapeutics Highlights January Equity Financing, Program Progress and Strategic Objectives for 2026
Recently raised $50 million with BVF Partners, Deerfield Management, founding investor Flagship Pioneering and a leading biotech mutual fund in a transaction that will close January 13th, 2026
Phase 1 dose-escalation trial of FHD-909 (LY4050784) advancing as planned, targeting SMARCA4 (BRG1)-mutant cancers with a focus on non-small cell lung cancer (NSCLC)
Selective CBP degrader program with potential in ER+ breast cancer on track to be IND-ready in 2026
Selective EP300 degrader program shows preclinical superior anti-tumor efficacy and tolerability over dual CBP/EP300; tracking to IND-enabling studies in 2026
Strong balance sheet with cash, cash equivalents, and marketable securities of $208.9 million*, including proceeds from the equity financing, which is anticipated to close on January 13, 2026, and allows for continued investment in the pipeline and extends cash into the first half of 2028
WATERTOWN, Mass. — (GLOBE NEWSWIRE) – January 9, 2026 — Foghorn® Therapeutics Inc. (Nasdaq: FHTX), a clinical-stage biotechnology company pioneering a new class of medicines that treat serious diseases by correcting abnormal gene expression, today announced its strategic objectives for 2026.
“We are pleased to have raised $50 million in an equity financing, priced at a 30% premium to the closing stock price on January 9, 2026. As part of the financing, we issued premium-priced warrants with an exercise price of 2 and 3 times the issue price. This equity raise represents an important vote of confidence from key biotech investors in our vision and execution,” said Adrian Gottschalk, President and Chief Executive Officer of Foghorn. “We continue to execute across our first-in-class pipeline focused on developing new treatment options for cancers with significant unmet need. For FHD-909, our partnered program with Lilly, the Phase 1 dose-escalation trial is on track. The trial is enrolling patients with SMARCA4-mutated cancers, particularly those with NSCLC where prognosis is poor and worsens with each additional line of therapy. We are also making strong progress across our degrader portfolio as we advance our Selective CBP degrader, with promise in ER+ breast cancer, and our Selective EP300 degrader, with potential in hematologic malignancies, toward anticipated Investigational New Drug (INDs) filings. With unique programs across our partnered and proprietary pipeline, we look forward to providing updates during the coming year.”
| * | Unaudited and estimated. |
Corporate Update
Strengthens Balance Sheet with Equity Financing to Advance Pipeline. On January 9, 2026, Foghorn entered into agreements with BVF Partners, Deerfield Management, founding investor Flagship Pioneering and a leading biotech mutual fund for the purchase and sale of 2,030,314 shares of its common stock at a purchase price of $6.71 per share and in lieu of common stock to certain investors, pre-funded warrants to purchase up to an aggregate of 5,421,250 shares of its common stock at a price of $6.7099 per pre-funded warrant, which represents the per share offering price for the common stock less the $0.0001 per share exercise price for each such pre-funded warrant as well as warrants to purchase up to 3,725,782 shares of common stock at an exercise price of $13.42 per share and up to 3,725,782 shares of common stock at an exercise price of $20.13 per share. The purchase price of the shares of common stock to be sold in the offering represents a premium of 30% to the last reported sale price of our common stock on the Nasdaq Global Market on January 9, 2026. (The offering is expected to close on or about January 13, 2026, subject to satisfaction of customary closing conditions). All of the shares of common stock in the offering are to be sold by Foghorn.
Program Overview and Upcoming Milestones
FHD-909 (LY4050784). FHD-909 is a first-in-class oral SMARCA2 selective inhibitor that has demonstrated in preclinical studies to have high selectivity over its closely related paralog SMARCA4, two proteins that are the catalytic engines across all forms of the BAF complex. Selectively blocking SMARCA2 activity is a promising synthetic lethal strategy intended to induce tumor death while sparing healthy cells. SMARCA4 is mutated in up to 10% of NSCLC alone and implicated in a significant number of solid tumors. Across lines of therapy, significant unmet needs remain for patients with SMARCA4 (BRG1)-mutant cancers with both poor response rates and short progression-free survival.
| • | Phase 1 trial on track. Enrollment in the first-in-human Phase 1 multi-center trial of FHD-909 is progressing well. The trial in patients with NSCLC as the primary target population is on track, following the dosing of the first patient in October 2024. |
| • | Synergistic preclinical data of FHD-909 in combination with pembrolizumab and KRAS inhibitors. Preclinical data supports enhanced anti-tumor activity of FHD-909 in combination with standard-of-care (SoC) chemotherapies, anti-PD-1 pembrolizumab and several novel KRAS inhibitors in NSCLC animal models. |
| • | Pending successful Phase 1 dose escalation results, Foghorn and Lilly anticipate evaluating FHD-909 in combination studies in the front-line setting of NSCLC. |
Ongoing strategic collaboration with Lilly. Foghorn is collaborating with Lilly to develop novel oncology medicines, including a 50/50 U.S. co-development and co-commercialization agreement for its selective SMARCA2 oncology program that includes both a selective inhibitor and a selective degrader, as well as an additional undisclosed oncology target. The collaboration also includes three discovery programs from Foghorn’s proprietary Gene Traffic Control® platform.
Selective CBP degrader program. Foghorn’s Selective CBP degrader selectively targets CBP, an acetyltransferase closely related to EP300. CBP lineage dependencies are established in several cancers, including breast cancer and there is also a synthetic relationship in EP300-mutated cancers, which include endometrial, cervical, ovarian, bladder, and colorectal cancer. Attempts to selectively drug CBP have been challenging due to the high level of similarity between the two proteins, while dual inhibition of CBP/EP300 has been associated with dose-limiting toxicities.
| • | CBP degrader program – IND-ready anticipated in 2026. In October 2025, preclinical data for Selective CBP degraders CBP-dependent cancers and ER+ breast cancer was presented during a Foghorn virtual investor event, which included: |
| • | Highly potent and selective lead candidate CBPd-171 in ongoing dose range finding toxicology studies |
| • | Anti-tumor activity in EP300 mutant solid tumors and in CBP-dependent cancers, including promising potential in ER+ breast cancer |
| • | No impact on platelet counts and spared megakaryocytes with CBPd-171 |
| • | Long Acting Injectable (LAI) formulation optimized for subcutaneous injection weekly or every other week for convenient administration |
Selective EP300 degrader program. Foghorn is developing a Selective EP300 degrader for the treatment of hematological malignancies and prostate cancer. Attempts to selectively drug EP300 have been challenging due to the high level of similarity between EP300 and CBP, while dual inhibition of CBP/EP300 has been associated with dose limiting toxicities. EP300 lineage dependencies are established in diffuse large b-cell lymphoma (DLBCL) and multiple myeloma (MM).
| • | EP300 degrader program – IND-enabling studies expected in 2026, with a focus in MM and DLBCL. In October 2025, efficacy and safety data of Selective EP300 degraders in preclinical models of hematological malignancies was presented during a Foghorn virtual investor event which included: |
| • | Broad anti-tumor activity in over 70% of all heme sub-lineages tested |
| • | VHL-based selective degrader shows impressive efficacy in MM without hematological toxicities including thrombocytopenia |
| • | EP300 degraders show full efficacy in IMiD-resistant MM cell lines |
| • | Tolerability profile with widespread potential for combinations |
Selective ARID1B degrader program. Foghorn’s Selective ARID1B degrader selectively targets and degrades ARID1B in ARID1A-mutated cancers. ARID1A is the most mutated subunit in the BAF complex and amongst the most mutated proteins in cancer. These mutations lead to a dependency on ARID1B in several types of cancer, including endometrial, gastric, gastroesophageal junction, bladder and NSCLC. Attempts to selectively drug ARID1B have been challenging because of the high degree of similarity between ARID1A and ARID1B and the fact that ARID1B has no enzymatic activity to target. ARID1B is a major synthetic lethal target implicated in up to 5% of all solid tumors.
| • | First-in-class Selective ARID1B degrader program advancing towards in vivo proof of concept in 2026. In October 2025, progress for the Selective ARID1B degrader was presented during a Foghorn virtual investor event which included: |
| • | Developed VHL and cereblon based bifunctional degraders with potential for oral delivery |
| • | Selective degradation of ARID1B achieved |
| • | Modulation of downstream target genes following ARID1B degradation |
Strong Balance Sheet and Cash Runway.
As of January 13, 2026, the Company expects to have approximately $208.9 million (unaudited) in cash, cash equivalents, and marketable securities, inclusive of proceeds from the recent equity financing, allowing for continued investment in the pipeline and extending cash into the first half of 2028.
The securities described under Corporate Update are being offered by Foghorn pursuant to a shelf registration statement on Form S-3 declared effective by the Securities and Exchange Commission (“SEC”) on January 31, 2025. This press release does not constitute an offer to sell or a solicitation of an offer to buy the securities in this offering, nor shall there be any sale of these securities in any state or other jurisdiction in which such offer, solicitation, or sale would be unlawful prior to the registration or qualification under the securities laws of any such state or other jurisdiction. A prospectus supplement and accompanying prospectus relating to the offering will be filed with the SEC and will be available on the SEC’s website at www.sec.gov.
About Foghorn Therapeutics
Foghorn® Therapeutics is discovering and developing a novel class of medicines targeting genetically determined dependencies within the chromatin regulatory system. Through its proprietary, scalable Gene Traffic Control® platform, Foghorn is systematically studying, identifying, and validating potential drug targets within the chromatin regulatory system. The Company is developing multiple product candidates in oncology. Visit our website at www.foghorntx.com for more information on the Company, and follow us on X and LinkedIn.
Forward-Looking Statements
This press release contains “forward-looking statements.” Forward-looking statements include statements regarding the Company’s ongoing Phase 1 trial of FHD-909 in SMARCA4-mutated cancers, preclinical product candidates, expected timing of clinical data, expected cash runway, expected timing of regulatory filings, and research efforts and other statements identified by words such as “could,” “may,” “might,” “will,” “likely,” “anticipates,” “intends,” “plans,” “seeks,” “believes,” “estimates,” “expects,” “continues,” “projects” and similar references to future periods. Forward-looking statements are based on our current expectations and assumptions regarding capital market conditions, our business, the economy and other future conditions. Because forward-looking statements relate to the future, by their nature, they are subject to inherent uncertainties, risks and changes in circumstances that are difficult to predict. As a result, actual results may differ materially from those contemplated by the forward-looking statements. Important factors that could cause actual results to differ materially from those in the forward-looking statements include regional, national or global political, economic, business, competitive, market and regulatory conditions, including risks relating to our clinical trials and other factors set forth under the heading “Risk Factors” in the Company’s Annual Report on Form 10-K for the year ended December 31, 2024, as filed with the Securities and Exchange Commission. Any forward-looking statement made in this press release speaks only as of the date on which it is made.
Contact:
Karin Hellsvik, Foghorn Therapeutics Inc.
khellsvik@foghorntx.com
Exhibit 99.2

Unique biology Precision therapeutics Broad impact FHTX January 2026 NASDAQ LISTED

2 | Forward Looking Statements This currently presentation available contains to management forward.- looking All statements statements other that than are statements based on of management’s historical facts beliefs contained and assumptions in this presentation and on are information forward-looking “anticipates,” statements “intends,” . In some “plans,” cases, “seeks,” you can “believes,” identify forward “estimates,” -looking “expects,” statements “continues,” by terms “projects” such as “could,” or the “may,” negative “might,” of these “will,” terms “likely,” or other are not similar limited expressions, to, statements although concerning: not all forward the potential -looking outcomes statements from contain our collaboration these words agreement . Forward- looking with Lilly; statements the initiation, include, timing, but Phase progress 1 dose and results escalation of our trial research of FHD —and 909 development with Lilly; our programs ability to advance and pre- clinical product studies candidates and clinical that we trials, may including develop and with to respect successfully to our complete Gene Traffic preclinical Control and Platform clinical ®; studies; the impact our of ability exogeneous to leverage factors, our initial including programs macroeconomic to develop additional and geopolitical product circumstances, candidates using on our our and related our collaborators’ business operations, including our research and development programs and pre-clinical studies; developments to our competitors and our industry; our ability to expand the target populations of our programs and the availability of patients regulatory for clinical authorities; testing; our our ability ability to to obtain identify regulatory and enter approval into future for license FHD-909 agreements and any future and collaborations; product candidates our ability from to the continue FDA and to rely other on our our CDMOs ability to and attract CROs and for retain our key manufacturing scientific and and management research needs; personnel; regulatory the scope developments of protection in the we United are able States to establish, and foreign maintain countries; and enforce proceeds for from intellectual capital- raising property transactions, rights covering estimates FHD- 909, of our our expenses, future products capital and requirements, our Gene Traffic and needs Control for Platform; additional and financing our use . You of should, presentation therefore, . Additional not rely important on these forward factors -looking to be considered statements in as connection representing with our views forward as -looking of any date statements subsequent are to described the date in of this the Annual Company’s Report filings on with Form the 10 Securities -K for the and fiscal Exchange year ended Commission, December including 31, 2024 .withing Any forward the section -looking entitled statements “Risk Factors” represent in the the Company’s Company’s views Company only explicitly as of the date disclaims of this any presentation obligation and to should update not any be forward relied upon -looking as representing statements. its The views Company’s as of any business subsequent is subject date. The to substantial risks and uncertainties.
Foghorn is a Leader in Chromatin Biology, Successfully Drugging Challenging Targets Multi-billion $ First-and-Best-in-Class Selective Target Strategic Partnership: Opportunities Approaches Engagement Multiple Programs Targeting chromatin Unlocking Innovating selective Leveraging a proven regulation selectivity of protein degradation drug development previously with capabilities in platform with Implicated in up to undruggable targets induced proximity expansive potential 50% of all tumors 3 |
Chromatin Remodeling Complexes – specialized multiprotein machines that Chromatin – compacted Transcription Factors – proteins allow access to DNA form of DNA inside the that help turn specific genes “on” or Targets: SMARCA2, ARID1B nucleus of the cell “off” by working in concert with the chromatin remodeling complex to bind to DNA Targets: Multiple TFs Chromatin Regulatory System Orchestrates Gene Expression: Multiple Opportunities for Targets and Therapeutics Chromatin Regulatory System genes are implicated across a wide range of cancers Other Chromatin Binding Proteins involved in gene expression / function Targets: CBP, EP300 Leveraging synthetic lethality and lineage dependencies 4 |
2. Chemistry, Biochemistry & Formulations Track record Track record with previously of delivering undruggable targets precision therapeutics Foghorn’s Platform Has Delivered Precision First-in-Class Therapeutics, and is Poised to Unlock New Biology 1. Chromatin Biology Deep mechanistic understanding of chromatin regulatory system 3. Degraders / Selective Targeting Expansion into Expertise in rules of degrader New TAs optimization & Induced Proximity 5
Modality Program and Partner Disease Discovery Pre-Clinical Phase 1 Anticipated Milestones FHD-909* SMARCA4-mutant cancers (e.g., NSCLC) Ph 1 Monotherapy Data; Enzyme (Selective SMARCA2) Confidential Inhibitors Partnered** Transition to Lilly; Undisclosed Undisclosed Confidential Transition to Lilly; Selective SMARCA2 SMARCA4-mutant cancers (e.g., NSCLC) Confidential ER+ Breast and EP300-mutant cancers Selective CBP (e.g., endometrial, cervical, ovarian, IND-Ready; 2026 Protein bladder) Degraders IND-enabling Studies; Selective EP300 MM and DLBCL and other heme cancers 2026 ARID1A-mutant cancers (e.g., Selective ARID1B In vivo PoC; 2026 endometrial, gastric, bladder, NSCLC) Transcription Factor Undisclosed Undisclosed Disruptors 3 Discovery Undisclosed Undisclosed Programs Foghorn: Precision Therapies with Potential for Multi-Billion Dollar Opportunities Across Oncology *LY4050784, 50/50 U.S. economic split, ex-U.S. royalties. ** Pending Lilly decision to proceed, 50/50 U.S. economic split, ex-U.S. royalties. SMARCA2 = BRM 6 | DLBCL: Diffuse Large B-Cell Lymphoma; ER+: Estrogen Receptor-positive; MM: Multiple Myeloma; NSCLC: Non-Small Cell Lung Cancer

FHD-909 is Being Developed in Collaboration with Lilly; Landmark Agreement Signed in December 2021 Significant Upfront and Strong Momentum and Ongoing Discovery Economics Shared Vision Programs • $300 million cash • $80 million in Foghorn common stock at a price of $20 per share • 50/50 U.S. economic split on SMARCA2-target and another undisclosed program • Tiered ex-U.S. royalties ranging from low double-digit into 20s • Lilly is a leading oncology company with a track record of innovation and execution • Lilly selected FHD-909 for development and initiated the first clinical trial in 2024 • Thorough evaluation of FHD-909 in models of SMARCA2-dependent tumors • Three additional programs as part of collaboration (undisclosed) • Potential to earn royalties and up to $1.3 billion in potential milestones across these three programs 7 |
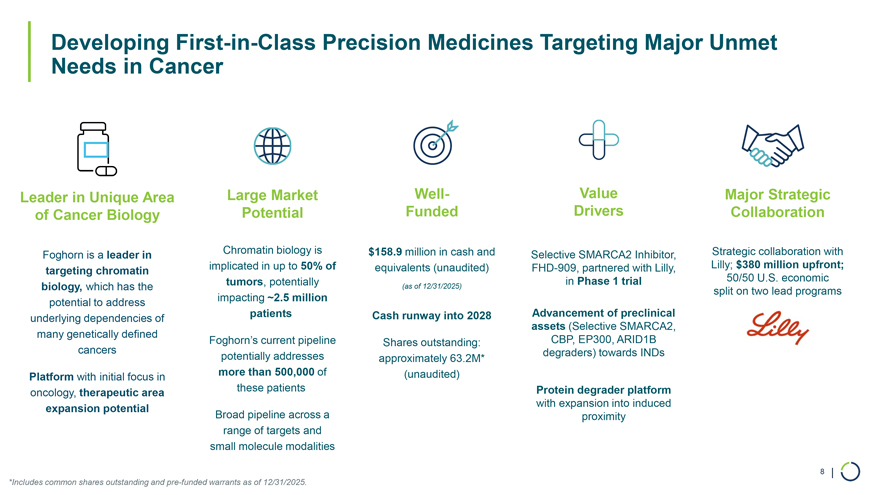
Large Market Well-Potential Funded Chromatin biology is $158.9 million in cash and implicated in up to 50% of equivalents (unaudited) tumors, potentially (as of 12/31/2025) impacting ~2.5 million patients Cash runway into 2028 Foghorn’s current pipeline Shares outstanding: potentially addresses approximately 63.2M* more than 500,000 of (unaudited) these patients Broad pipeline across a range of targets and small molecule modalities Developing First-in-Class Precision Medicines Targeting Major Unmet Needs in Cancer Leader in Unique Area of Cancer Biology Foghorn is a leader in targeting chromatin biology, which has the potential to address underlying dependencies of many genetically defined cancers Platform with initial focus in oncology, therapeutic area expansion potential Value Major Strategic Drivers Collaboration Selective SMARCA2 Inhibitor, Strategic collaboration with FHD-909, partnered with Lilly, Lilly; $380 million upfront; in Phase 1 trial 50/50 U.S. economic split on two lead programs Advancement of preclinical assets (Selective SMARCA2, CBP, EP300, ARID1B degraders) towards INDs Protein degrader platform with expansion into induced proximity 8 | *Includes common shares outstanding and pre-funded warrants as of 12/31/2025.

For SMARCA4-mutant Cancers Selective SMARCA2 Program • FHD-909 (LY4050784) – Selective SMARCA2 Inhibitor 9

Selective SMARCA2 Selective SMARCA2 Inhibitor FHD-909* Degrader Exploit the synthetic lethal relationship between SMARCA2 and Biology mutated SMARCA4 SMARCA2: Clinical-stage FHD-909 Selective SMARCA2 Inhibitor and Preclinical Selective SMARCA2 Degrader Phase 1 monotherapy dose escalation Advancing through late Status trial ongoing preclinical development Opportunity SMARCA4-mutated cancer including ~10% of NSCLC and up to 5% of all solid tumors Lilly 50/50 global R&D cost share | 50/50 U.S. economics | tiered ex-U.S. royalties starting Partnership in the low double-digit range and escalating into the twenties 10 | *LY4050784

Selective SMARCA2 Selective SMARCA2 Inhibitor FHD-909* Degrader Exploit the synthetic lethal relationship between SMARCA2 and Biology mutated SMARCA4 SMARCA2: Clinical-stage FHD-909 Selective SMARCA2 Inhibitor and Preclinical Selective SMARCA2 Degrader Phase 1 monotherapy dose escalation Advancing through late Status trial ongoing preclinical development Opportunity SMARCA4-mutated cancer including ~10% of NSCLC and up to 5% of all solid tumors Lilly 50/50 global R&D cost share | 50/50 U.S. economics | tiered ex-U.S. royalties starting Partnership in the low double-digit range and escalating into the twenties 10 | *LY4050784 Selective SMARCA2 Inhibition: Promising Strategy to Exploit Synthetic Lethal Relationship Between SMARCA2 and Mutant SMARCA4 Healthy Cells Cancer Cells BAF BAF BAF Complex Complex Complex SMARCA2i SMARCA2 Mutant SMARCA4 SMARCA4 SMARCA2 SMARCA4 No SMARCA4 Mutations SMARCA4 Mutation SMARCA4 Mutation and SMARCA2i Cell Survival Cell Death Precision medicine targeting synthetic lethal relationships is a proven clinical approach now used in multiple cancers (e.g., PARP inhibitors) 11 |

SMARCA4 mutated across a broad range of tumors Accounts for ~5% of solid tumors 12 | SMARCA4 is Mutated in Up to 10% of NSCLC; Up to 5% of Solid Tumors AACR GENIE via cBioPortal

FHD-909: Overall Goal is to Become a First-Line Treatment for SMARCA4-mutated NSCLC Relevant treatment regimens in each line of therapy for metastatic NSCLC* 1st Line NSCLC Pembrolizumab-based regimens Other immune checkpoint 2nd Line NSCLC inhibitor-based regimens Chemotherapy-based Chemotherapy-based regimens regimens Targeted agents Ramucirumab (anti-VEGF) + 3rd Line NSCLC chemotherapy Checkpoint inhibitor Chemotherapy-based monotherapy regimens Other targeted agents + KRAS inhibitors Agents evaluated preclinically in chemotherapy combination with FHD-909 Note: *Generalized across squamous and non-squamous metastatic NSCLC without driver mutation 13 | Source: CancerMPact 2024 US NSCLC TA report
Significant Unmet Need 21.9% 15.0 months 8.1 months 6.1 months 2.7 months 1L (SMARCA4 WT) 1L (SMARCA4 Mut) Significant Unmet Medical Need in NSCLC Metastatic Setting for SMARCA4 Patients Frontline NSCLC Metastatic – PD1 + Chemo1 Response Rate (%) PFS (months) OS (months) 39.1% Significant Unmet Need in SMARCA4-mutated NSCLC • Poor response to 1L chemo-immunotherapy for patients with SMARCA4 mutations1 • 2L outcomes with docetaxel are poor for all patients (ORR 10-20% depending on agent/s2,3; PFS ~5-months) • SMARCA4-mutated patients are expected to fare even worse in the 2L setting • 3L+ setting – experience suggests less than 10% ORR and 1 to 2 months PFS Source: 1. Alessi, J Thorac Oncol, 2023 2. Rittmeyer, Lancet, 2017; Fehrenbacher, J Thorac Oncol, 2018; Mazieres, J Thorac Oncol, 2021 Herbst, Lancet, 2016; Herbst, Abs OA03.07 3. S1800A – Lung-MAP Sub-study – 14 | ASCO 2022; Garon, Lancet, 2014
SMARCA4 10% KRAS 29% EGFR 26% ALK 7% Survival(%) RET 4% MET 7% Overall SMARCA4 Mutations are Consequential – in NSCLC, Patients with Mutated SMARCA4 Have Significantly Worse Clinical Outcomes Overall Survival for SMARCA4wt vs SMARCA4mut1; Frontline Metastatic NSCLC w/ Chemoimmunotherapy SMARCA4 Mutated in Up to 10% of NSCLC Tumors, Minimal Overlap w/ Other Mutations2 Months NSCLC patients with SMARCA4 mutations: — Poor prognosis — Shorter overall survival — Less responsive to immune checkpoint inhibitors — Clinically definable, high unmet need population Supporting references: • Gandhi, et al, 2025; DOI: 10.1016/j.jtho.2025.01.016 • Alessi, et al, 2023; DOI: 10.1016/j.jtho.2023.01.091 • Negrao, et al, 2023; DOI: 10.1158/2159-8290.Cd-22-1420 • Liu, et al, 2021; DOI: 10.1002/1878-0261.12831 • Fernando, et al, 2020; DOI: 10.1038/s41467-020-19402-8 • Schoenfeld, et al, 2020DOI: 10.1158/1078-0432.ccr-20-1825 15 | Source: 1. Alessi et al DOI: 10.1016/j.jtho.2023.01.091; 2. TCGA via cBioPortal
When SMARCA4 and KRAS Mutations Co-Occur, Patients Have Even Worse Outcomes to Standard of Care Treatment Overall Response Rate (ORR) Progression-Free Survival (PFS) Overall Survival (OS) Median PFS, months (95% CI) Median OS, months (95% CI) P = 0.03 KRAS mut + SMARCA4 wt (n:159): 4.1 (2.8–6.0) KRAS mut + SMARCA4 wt (n:159): 15.1 (11.2–20.1) KRAS mut + SMARCA4 mut (n:17): 1.4 (0.9–2.0) KRAS mut + SMARCA4 mut (n:17): 3.0 (1.0–9.3) 22.0% (35/159) HR = 0.25 [95% CI: 0.14–0.42] HR = 0.29 [95% CI: 0.17–0.50] P < 0.001 P < 0.001 0.0% (0/17) In response to PD(L)-1 therapy, patients with co-occurring SMARCA4 and KRAS mutations have a shorter ORR, PFS, and OS than patients with only KRASmut Source: Alessi et al DOI: 10.1016/j.jtho.2021.03.024 16 |
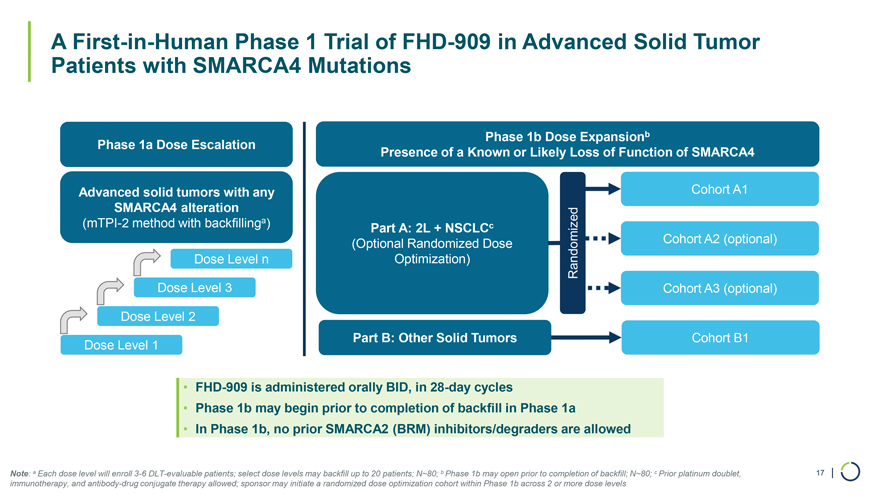
A First-in-Human Phase 1 Trial of FHD-909 in Advanced Solid Tumor Patients with SMARCA4 Mutations Phase 1b Dose Expansionb Phase 1a Dose Escalation Presence of a Known or Likely Loss of Function of SMARCA4 Advanced solid tumors with any Cohort A1 SMARCA4 alteration (mTPI-2 method with backfillinga) Part A: 2L + NSCLCc (Optional Randomized Dose Cohort A2 (optional) Dose Level n Optimization) Randomized Dose Level 3 Cohort A3 (optional) Dose Level 2 Part B: Other Solid Tumors Cohort B1 Dose Level 1 • FHD-909 is administered orally BID, in 28-day cycles • Phase 1b may begin prior to completion of backfill in Phase 1a • In Phase 1b, no prior SMARCA2 (BRM) inhibitors/degraders are allowed Note: a Each dose level will enroll 3-6 DLT-evaluable patients; select dose levels may backfill up to 20 patients; N~80; b Phase 1b may open prior to completion of backfill; N~80; c Prior platinum doublet, 17 | immunotherapy, and antibody-drug conjugate therapy allowed; sponsor may initiate a randomized dose optimization cohort within Phase 1b across 2 or more dose levels

Degrader Programs • Selective CBP Degrader • Selective EP300 Degrader • Selective ARID1B Degrader 18
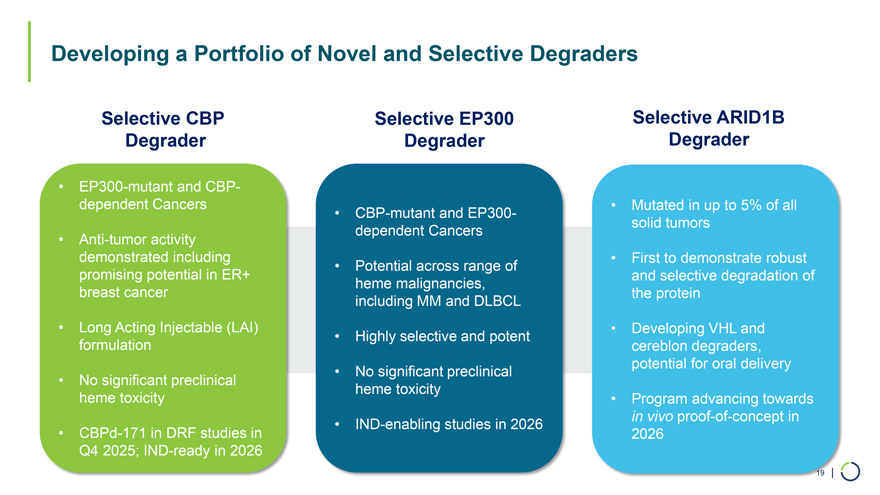
• EP300-mutant and CBP-dependent Cancers • Mutated in up to 5% of all • CBP-mutant and EP300-solid tumors dependent Cancers • Anti-tumor activity demonstrated including • First to demonstrate robust • Potential across range of promising potential in ER+ and selective degradation of heme malignancies, breast cancer the protein including MM and DLBCL • Long Acting Injectable (LAI) • Developing VHL and • Highly selective and potent formulation cereblon degraders, potential for oral delivery • No significant preclinical • No significant preclinical heme toxicity heme toxicity • Program advancing towards • IND-enabling studies in 2026 in vivo proof-of-concept in • CBPd-171 in DRF studies in 2026 Q4 2025; IND-ready in 2026 Developing a Portfolio of Novel and Selective Degraders Selective CBP Selective EP300 Selective ARID1B Degrader Degrader Degrader 19 |
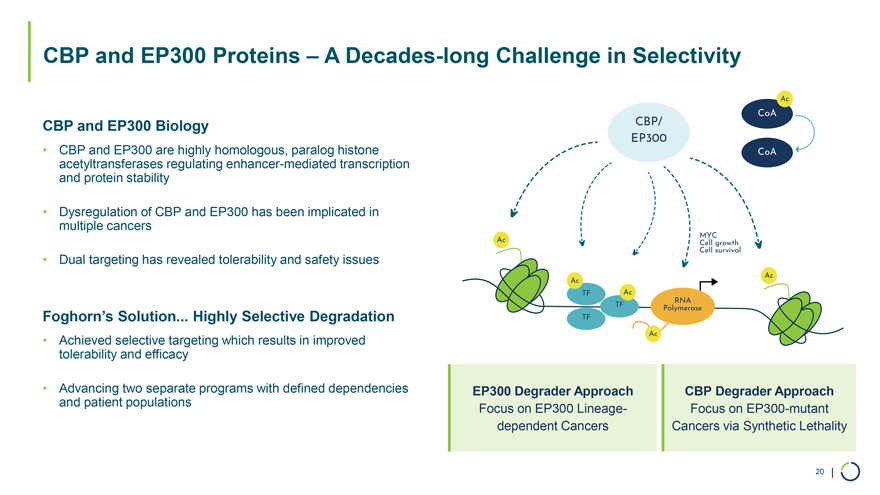
• Dysregulation of CBP and EP300 has been implicated in multiple cancers • Dual targeting has revealed tolerability and safety issues CBP and EP300 Proteins – A Decades-long Challenge in Selectivity CBP and EP300 Biology • CBP and EP300 are highly homologous, paralog histone acetyltransferases regulating enhancer-mediated transcription and protein stability Foghorn’s Solution... Highly Selective Degradation • Achieved selective targeting which results in improved tolerability and efficacy • Advancing two separate programs with defined dependencies and patient populations Ac CoA CBP/ EP300 CoA Ac MYC Cell growth Cell survival Ac Ac TF Ac TF RNA TF Polymerase Ac EP300 Degrader Approach CBP Degrader Approach Focus on EP300 Lineage- Focus on EP300-mutant dependent Cancers Cancers via Synthetic Lethality 20 |

Selective CBP Degrader, FHT-171 For EP300-mutant and CBP-dependent Cancers 21
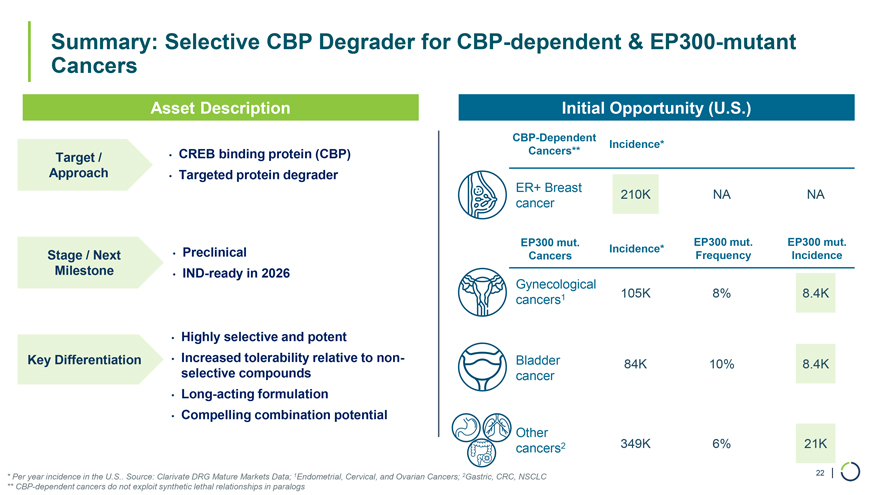
• CREB binding protein (CBP) • Targeted protein degrader • Preclinical • IND-ready in 2026 • Highly selective and potent • Increased tolerability relative to non-selective compounds • Long-acting formulation • Compelling combination potential Summary: Selective CBP Degrader for CBP-dependent & EP300-mutant Cancers Target / Approach Stage / Next Milestone Key Differentiation Asset Description Initial Opportunity (U.S.) CBP-Dependent Incidence* Cancers** ER+ Breast 210K NA NA cancer EP300 mut. EP300 mut. EP300 mut. Incidence* Cancers Frequency Incidence Gynecological cancers1 105K 8% 8.4K Bladder 84K 10% 8.4K cancer Other cancers2 349K 6% 21K * Per year incidence in the U.S.. Source: Clarivate DRG Mature Markets Data; 1Endometrial, Cervical, and Ovarian Cancers; 2Gastric, CRC, NSCLC 22 | ** CBP-dependent cancers do not exploit synthetic lethal relationships in paralogs
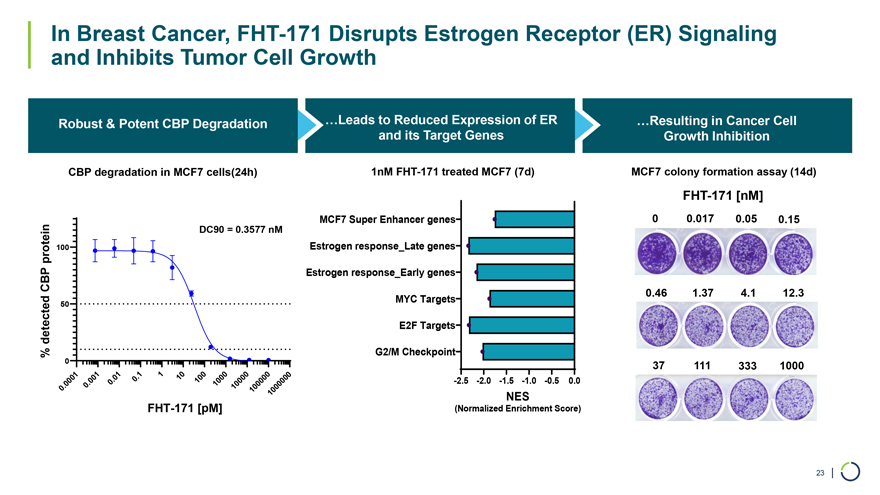
Robust & Potent CBP Degradation …Leads to Reduced Expression of ER …Resulting in Cancer Cell and its Target Genes Growth Inhibition CBP degradation in MCF7 cells(24h) 1nM FHT-171 treated MCF7 (7d) MCF7 colony formation assay (14d) FHT-171 [nM] DC90 = 0.3577 nM MCF7 Super Enhancer genes 0 0.017 0.05 0.15 protein 100 Estrogen response_Late genes CBP Estrogen response_Early genes MYC Targets 0.46 1.37 4.1 12.3 50 detected E2F Targets % G2/M Checkpoint 0 37 111 333 1000 1 . 1 1 0 0 0 0 0 01 . 01 0 10 0 0 0 00 . 00 0 1 0 0 -2.5 -2.0 -1.5 -1.0 -0.5 0.0 . 0 100 0 0 0 1000 0 0 1 0 1 NES FHT-171 [pM] (Normalized Enrichment Score) In Breast Cancer, FHT-171 Disrupts Estrogen Receptor (ER) Signaling and Inhibits Tumor Cell Growth 23 |
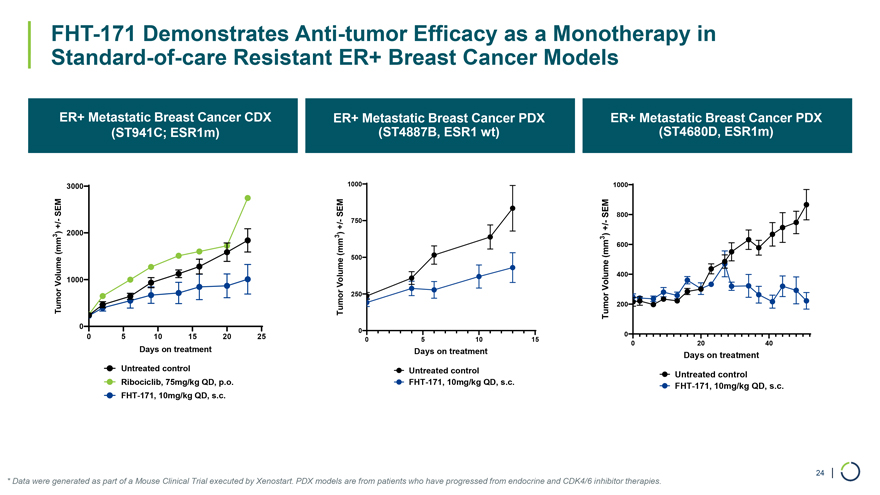
FHT-171 Demonstrates Anti-tumor Efficacy as a Monotherapy in Standard-of-care Resistant ER+ Breast Cancer Models ER+ Metastatic Breast Cancer CDX ER+ Metastatic Breast Cancer PDX ER+ Metastatic Breast Cancer PDX (ST941C; ESR1m) (ST4887B, ESR1 wt) (ST4680D, ESR1m) 3000 1000 1000 SEMSEM SEM 800——750—+/ +/ +/ ) 2000 ) ) 3 3 (mm 3 600 (mm 500 (mm 400 Volume 1000 Volume Volume 250 Tumor Tumor Tumor 200 0 0 0 0 5 10 15 20 25 0 5 10 15 0 20 40 Days on treatment Days on treatment Days on treatment Untreated control Untreated control Untreated control Ribociclib, 75mg/kg QD, p.o. FHT-171, 10mg/kg QD, s.c. FHT-171, 10mg/kg QD, s.c. FHT-171, 10mg/kg QD, s.c. 24 | * Data were generated as part of a Mouse Clinical Trial executed by Xenostart. PDX models are from patients who have progressed from endocrine and CDK4/6 inhibitor therapies.
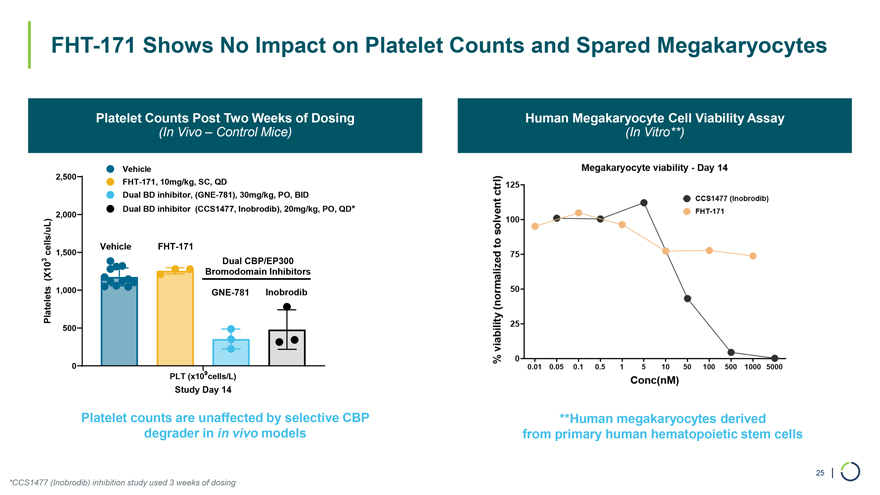
FHT-171 Shows No Impact on Platelet Counts and Spared Megakaryocytes Platelet Counts Post Two Weeks of Dosing Human Megakaryocyte Cell Viability Assay (In Vivo – Control Mice) (In Vitro**) Vehicle 2,500 FHT-171, 10mg/kg, SC, QD Dual BD inhibitor, (GNE-781), 30mg/kg, PO, BID Dual BD inhibitor (CCS1477, Inobrodib), 20mg/kg, PO, QD* 2,000 cells/uL) Vehicle FHT-171 1,500 3 Dual CBP/EP300 (X10 Bromodomain Inhibitors Platelets 1,000 GNE-781 Inobrodib 500 0 9 PLT (x10 cells/L) Study Day 14 Platelet counts are unaffected by selective CBP **Human megakaryocytes derived degrader in in vivo models from primary human hematopoietic stem cells *CCS1477 (Inobrodib) inhibition study used 3 weeks of dosing 25 |
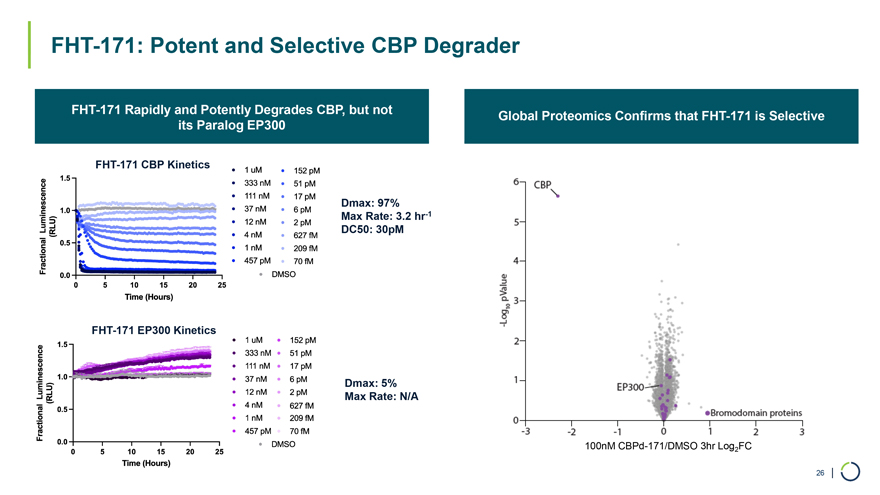
FHT-171 CBP Kinetics Dmax: 97% Max Rate: 3.2 hr-1 DC50: 30pM FHT-171 EP300 Kinetics Dmax: 5% Max Rate: N/A 100nM CBPd-171/DMSO 3hr Log2FC FHT-171: Potent and Selective CBP Degrader FHT-171 Rapidly and Potently Degrades CBP, but not its Paralog EP300 Global Proteomics Confirms that FHT-171 is Selective 26 |
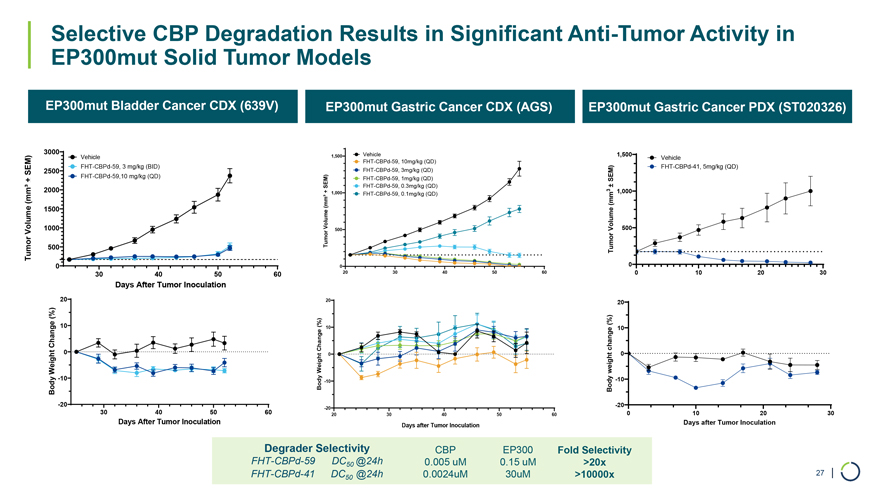
EP300mut Bladder Cancer CDX (639V) EP300mut Gastric Cancer CDX (AGS) EP300mut Gastric Cancer PDX (ST020326) 3000 Vehicle 1,500 Vehicle 1,500 FHT-CBPd-59, 10mg/kg (QD) Vehicle FHT-CBPd-59, 3 mg/kg (BID) FHT-CBPd-41, 5mg/kg (QD) SEM) 2500 FHT-CBPd-59, 3mg/kg (QD) + FHT-CBPd-59,10 mg/kg (QD) FHT-CBPd-59, 1mg/kg (QD) SEM) SEM) FHT-CBPd-59, 0.3mg/kg (QD)± (mm³ 2000+ 1,000 FHT-CBPd-59, 0.1mg/kg (QD) 3 1,000 (mm³(mm 1500 Volume 1000 500 Volume 500 Volume Tumor Tumor 500 Tumor 0 0 0 30 40 50 60 20 30 40 50 60 0 10 20 30 Days After Tumor Inoculation 20 20 20 (%) 10 (%)(%) Change 10 10 Change change 0 0 0 WeightWeightweight -10 -10 Body Body -10 Body -20 -20 -20 30 40 50 60 20 30 40 50 60 0 10 20 30 Days After Tumor Inoculation Days after Tumor Inoculation Days after Tumor Inoculation Degrader Selectivity CBP EP300 Fold Selectivity FHT-CBPd-59 DC50 @24h 0.005 uM 0.15 uM >20x FHT-CBPd-41 DC @24h 0.0024uM 30uM >10000x 27 | 50 Selective CBP Degradation Results in Significant Anti-Tumor Activity in EP300mut Solid Tumor Models
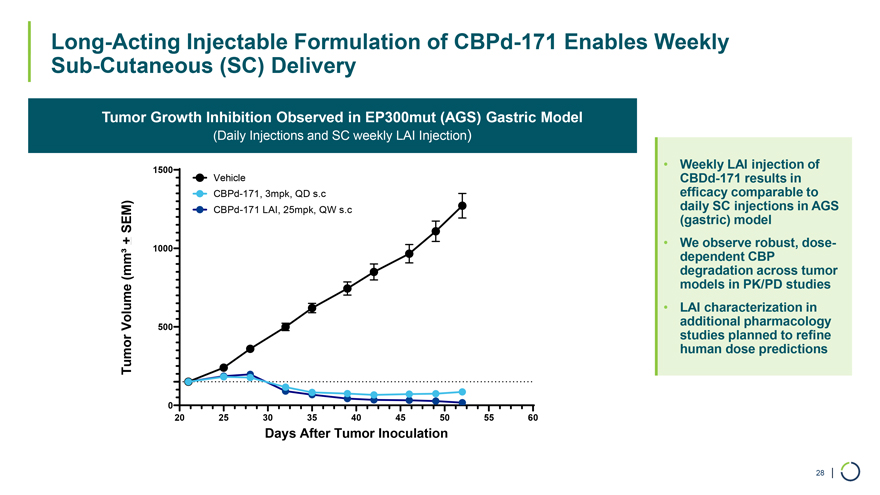
Long-Acting Injectable Formulation of CBPd-171 Enables Weekly Sub-Cutaneous (SC) Delivery Tumor Growth Inhibition Observed in EP300mut (AGS) Gastric Model (Daily Injections and SC weekly LAI Injection) 1500 Vehicle CBPd-171, 3mpk, QD s.c SEM) CBPd-171 LAI, 25mpk, QW s.c + 1000 (mm³ Volume 500 Tumor 0 20 25 30 35 40 45 50 55 60 Days After Tumor Inoculation • Weekly CBDd-171 LAI results injection in of daily efficacy SC comparable injections in to AGS (gastric) model • We dependent observe CBP robust, dose- degradation models in PK/PD across studies tumor • LAI additional characterization pharmacology in studies human dose planned predictions to refine 28 |

Selective EP300 Degrader For CBP-mutant and EP300-dependent Cancers 29

Asset Description Initial Opportunity (U.S.) EP300-Dependent Target / • E1A binding protein p300 (EP300) Hematological Incidence* Approach Malignancies • Targeted protein degrader MM 31K • Broad range of heme malignancies Indications focused on MM and DLBCL • AR+ prostate • Bladder, melanoma, others DLBCL 32K Stage / Next • Preclinical Milestone • IND-enabling studies in 2026 • Deeper efficacy response vs non- AML + MDS 38K Key Differentiation selective molecules • Improved tolerability profile vs non-selective molecules • Patient selection biomarker for (DLBCL) 30 | Summary: Selective EP300 Degrader for CBP-Mutant & EP300-Dependent Cancers * Per year incidence in the U.S.. Source: Clarivate DRG Mature Markets Data
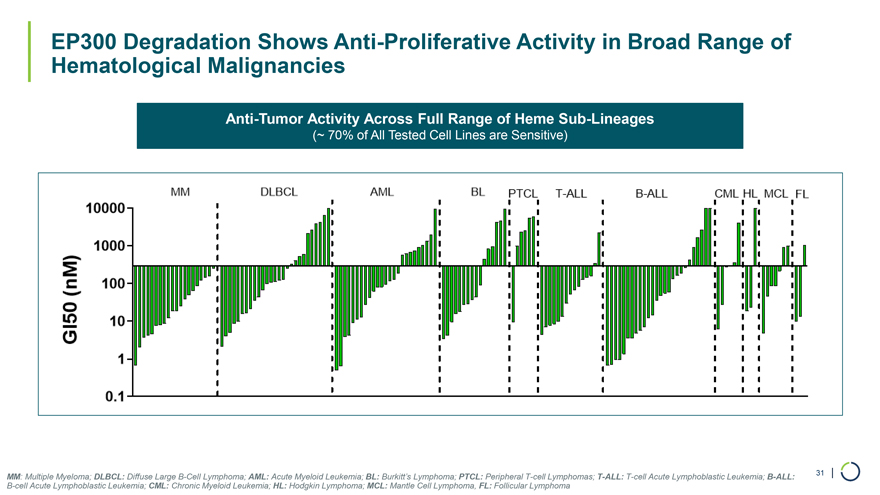
EP300 Degradation Shows Anti-Proliferative Activity in Broad Range of Hematological Malignancies Anti-Tumor Activity Across Full Range of Heme Sub-Lineages (~ 70% of All Tested Cell Lines are Sensitive) MM: Multiple Myeloma; DLBCL: Diffuse Large B-Cell Lymphoma; AML: Acute Myeloid Leukemia; BL: Burkitt’s Lymphoma; PTCL: Peripheral T-cell Lymphomas; T-ALL: T-cell Acute Lymphoblastic Leukemia; B-ALL: 31 | B-cell Acute Lymphoblastic Leukemia; CML: Chronic Myeloid Leukemia; HL: Hodgkin Lymphoma; MCL: Mantle Cell Lymphoma, FL: Follicular Lymphoma
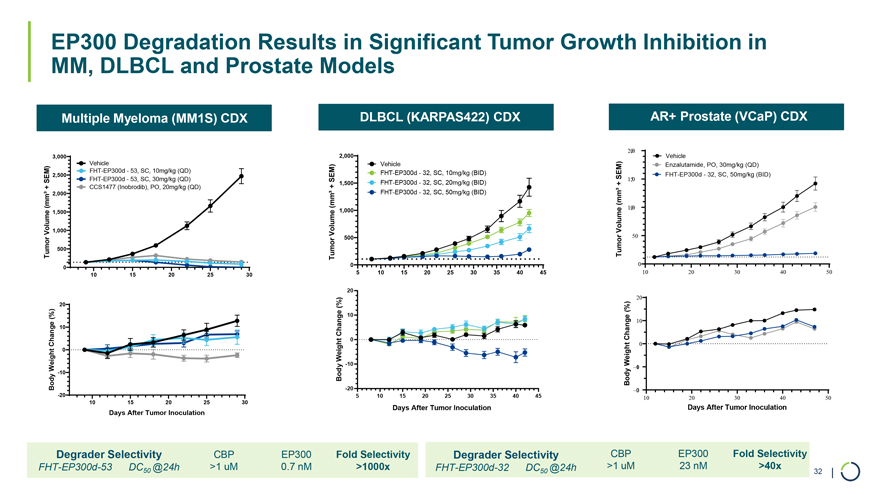
Multiple Myeloma (MM1S) CDX DLBCL (KARPAS422) CDX AR+ Prostate (VCaP) CDX 2,00 3,000 2,000 Vehicle Vehicle Vehicle Enzalutamide, PO, 30mg/kg (QD) FHT-EP300d—53, SC, 10mg/kg (QD) FHT-EP300d—32, SC, 10mg/kg (BID) 2,500 SEM) FHT-EP300d—32, SC, 50mg/kg (BID) SEM) FHT-EP300d—53, SC, 30mg/kg (QD) SEM) 1,50 1,500 FHT-EP300d—32, SC, 20mg/kg (BID) + CCS1477 (Inobrodib), PO, 20mg/kg (QD) + + 2,000 FHT-EP300d—32, SC, 50mg/kg (BID) (mm³ (mm³ (mm³ 1,00 1,500 1,000 Volume 1,000 VolumeVolume 500 50 Tumor 500 Tumor Tumor 0 0 0 10 15 20 25 30 5 10 15 20 25 30 35 40 45 10 20 30 40 50 Days After Tumor Inoculation Days After Tumor Inoculation Days After Tumor Inoculation 20 20 (%) (%) 10 10 Change Change 0 0 Weight Weight -0 1 Body -10 Body -2 0 -20 10 20 30 40 50 10 15 20 25 30 Days After Tumor Inoculation Days After Tumor Inoculation Degrader Selectivity CBP EP300 Fold Selectivity Degrader Selectivity CBP EP300 Fold Selectivity FHT-EP300d-53 DC @24h >1 uM 0.7 nM >1000x FHT-EP300d-32 DC @24h >1 uM 23 nM >40x 50 50 32 | EP300 Degradation Results in Significant Tumor Growth Inhibition in MM, DLBCL and Prostate Models
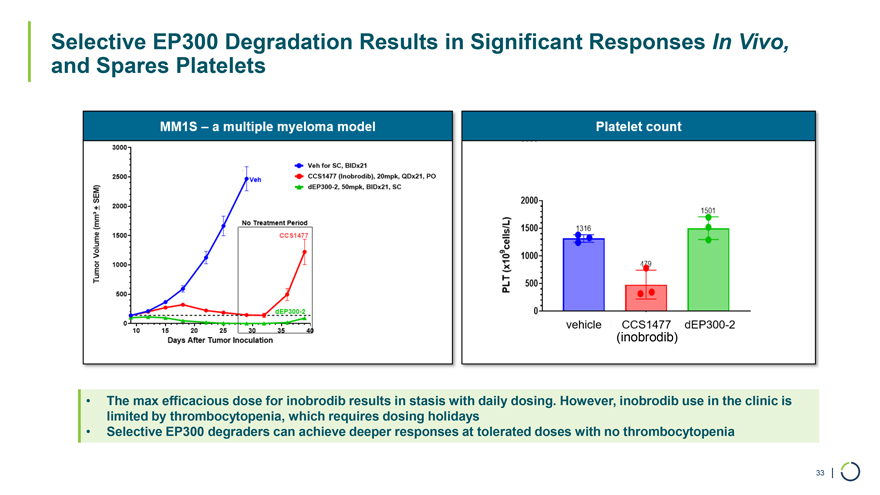
Selective EP300 Degradation Results in Significant Responses In Vivo, and Spares Platelets (inobrodib) • The max efficacious dose for inobrodib results in stasis with daily dosing. However, inobrodib use in the clinic is limited by thrombocytopenia, which requires dosing holidays • Selective EP300 degraders can achieve deeper responses at tolerated doses with no thrombocytopenia 33 |

No Change in Platelets, Red Blood Cells or Neutrophils Following 14 Days of Daily Dosing in Immunocompetent Mice Selective EP300 Degrader Spares Platelets, Red Blood Cells and Neutrophils 2000 1500 1283.3 1172.7 1000 500 Vehicle 20 EP300d-351, 30mpk, QD, s.c 15 10.8 10 8.8 5.6 5 3.3 0 0.5 0.9 ) ) ) ) /L L /L /L ls ì ls ls l l s/ l l e e e 9c el 9 c 9 c 6 c 0 0 10 1 1 x x10 ( x x(( ( t BC C LT eu P N W RB WBC: White Blood Cells; RBC: Red Blood Cells; PLT: Platelets; Neut: Neutrophils 34 |
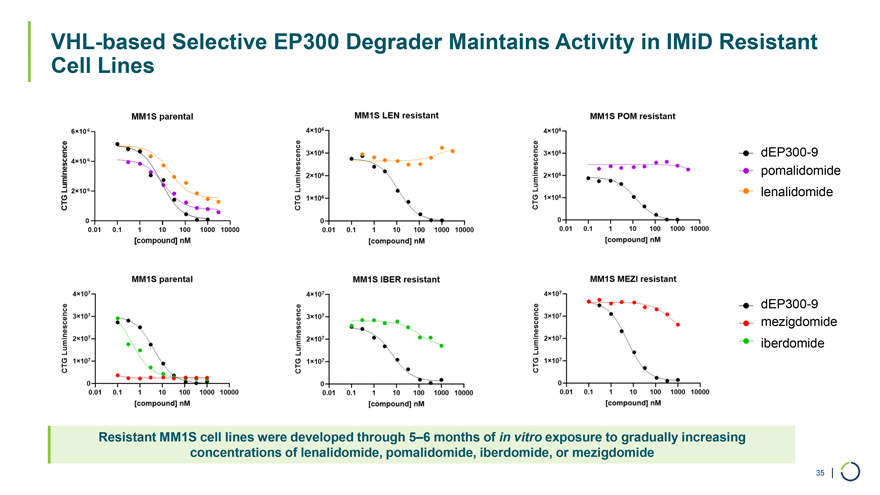
VHL-based Selective EP300 Degrader Maintains Activity in IMiD Resistant Cell Lines dEP300-9 pomalidomide lenalidomide dEP300-9 mezigdomide iberdomide Resistant MM1S cell lines were developed through 5–6 months of in vitro exposure to gradually increasing concentrations of lenalidomide, pomalidomide, iberdomide, or mezigdomide 35 |
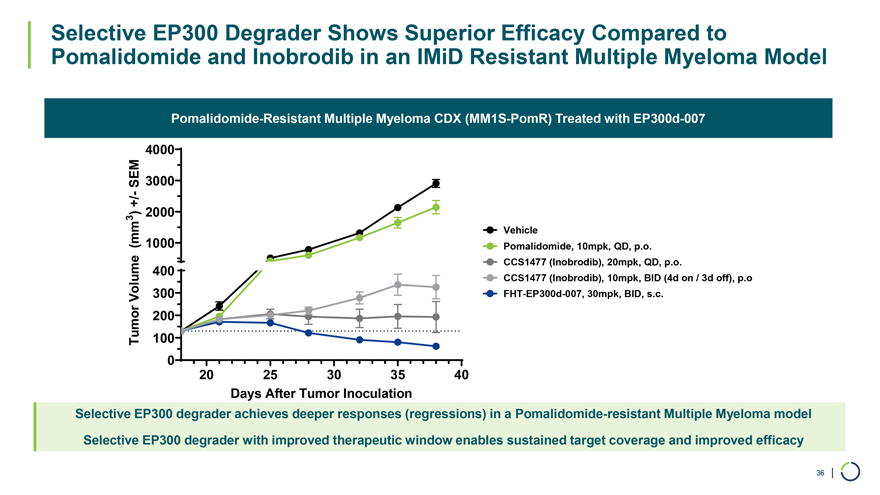
Pomalidomide-Resistant Multiple Myeloma CDX (MM1S-PomR) Treated with EP300d-007 Selective EP300 Degrader Shows Superior Efficacy Compared to Pomalidomide and Inobrodib in an IMiD Resistant Multiple Myeloma Model 4000 SEM 3000— +/ 3 ) 2000 (mm 1000 Vehicle Pomalidomide, 10mpk, QD, p.o. CCS1477 (Inobrodib), 20mpk, QD, p.o. 400 CCS1477 (Inobrodib), 10mpk, BID (4d on / 3d off), p.o Volume 300 FHT-EP300d-007, 30mpk, BID, s.c. 200 Tumor 100 0 20 25 30 35 40 Days After Tumor Inoculation Selective EP300 degrader achieves deeper responses (regressions) in a Pomalidomide-resistant Multiple Myeloma model Selective EP300 degrader with improved therapeutic window enables sustained target coverage and improved efficacy 36 |
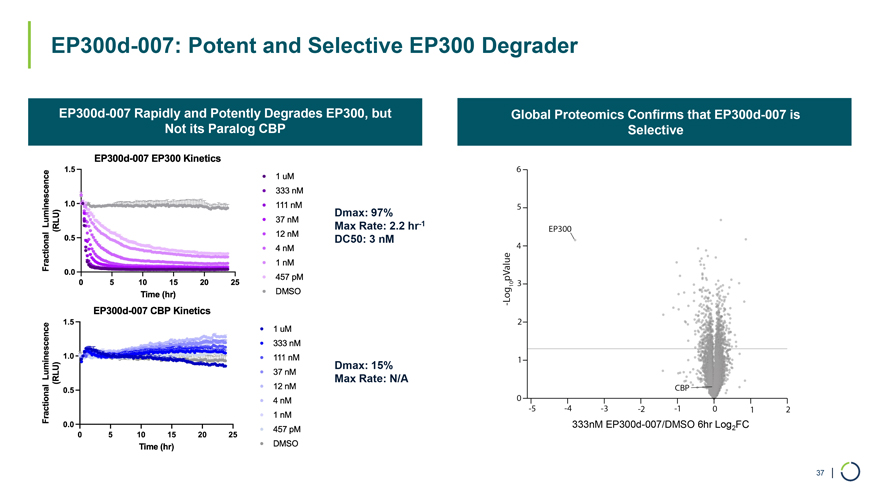
EP300d-007 Rapidly and Potently Degrades EP300, but Not its Paralog CBP EP300d-007: Potent and Selective EP300 Degrader Global Proteomics Confirms that EP300d-007 is Selective Dmax: 97% Max Rate: 2.2 hr-1 DC50: 3 nM Dmax: 15% Max Rate: N/A 333nM EP300d-007/DMSO 6hr Log2FC 37 |

Selective ARID1B Degrader For ARID1A-mutant Cancers 38

Asset Description Initial Opportunity (U.S.) ARID1A mut. ARID1A mut. Incidence* Frequency Incidence Target / • ARID1B Approach • Targeted protein degrader Endometrial cancers 66K 38% 25K ARID1B is a Major Synthetic Lethal Target with Potential in Up To 5% of All Solid Tumors Stage / Next • Preclinical Gastric 37K 20% 7K Milestone In vivo proof-of- in 2026 cancers • concept Bladder 84K 24% 20K • Multiple ARID1B binders with nM cancer Key Differentiation affinity and selectivity • Selective ARID1B degradation Non-small cell lung 195K 7% 14K cancer 39 | * Per year incidence in the U.S.. Source: Clarivate DRG Mature Markets Data.
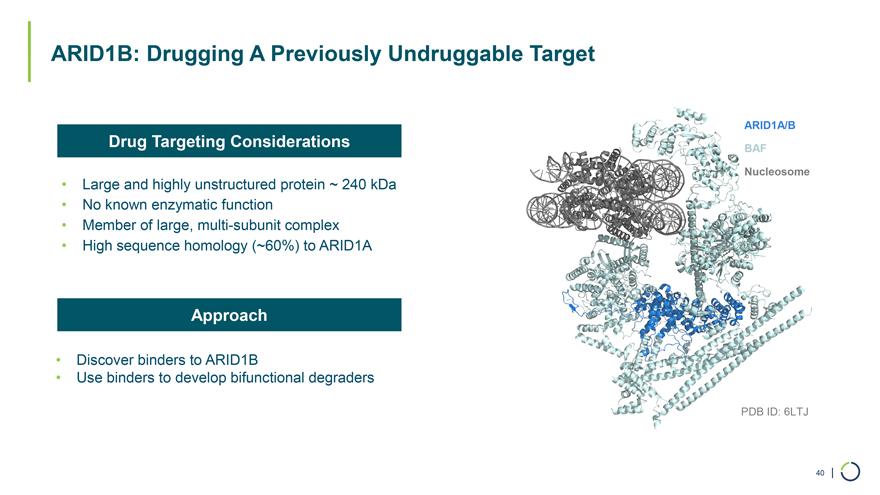
• Large and highly unstructured protein ~ 240 kDa • No known enzymatic function • Member of large, multi-subunit complex • High sequence homology (~60%) to ARID1A ARID1B: Drugging A Previously Undruggable Target Drug Targeting Considerations Approach • Discover binders to ARID1B • Use binders to develop bifunctional degraders ARID1A/B BAF Nucleosome PDB ID: 6LTJ 40 |
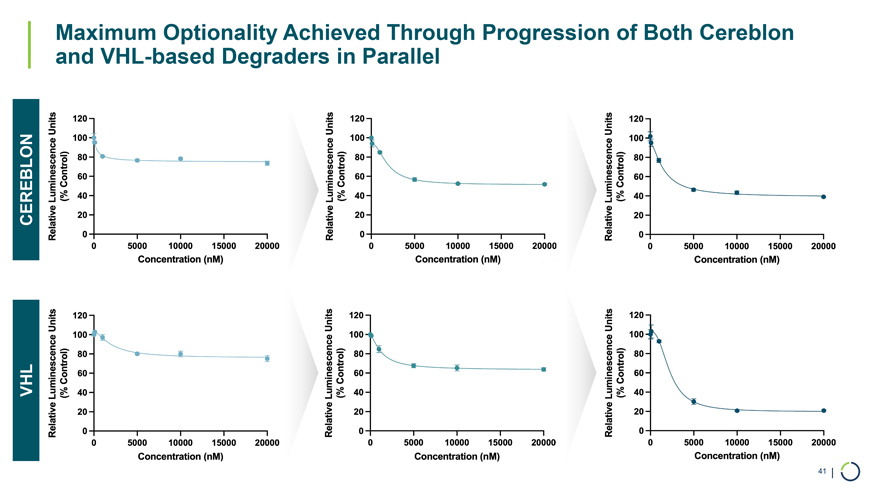
Maximum Optionality Achieved Through Progression of Both Cereblon and VHL-based Degraders in Parallel VHL CEREBLON 41 |
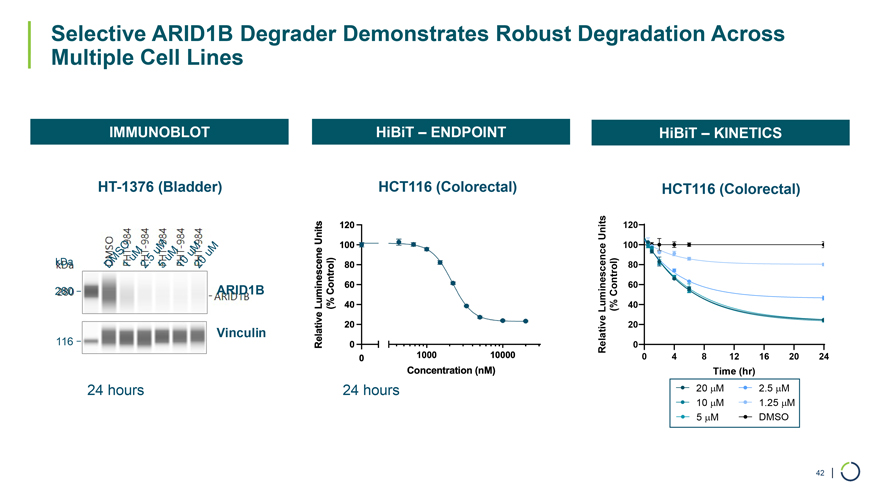
IMMUNOBLOT HiBiT – ENDPOINT HiBiT – KINETICS HT-1376 (Bladder) HCT116 (Colorectal) HCT116 (Colorectal) Units 120 100 kDa 80 ARID1B 60 280 Control) Luminescence(% 40 20 Vinculin 116 Relative 0 0 4 8 12 16 20 24 Time (hr) 24 hours 24 hours 20 µM 2.5 µM 10 µM 1.25 µM 5 µM DMSO Selective ARID1B Degrader Demonstrates Robust Degradation Across Multiple Cell Lines 42 |

ARID1B Degradation is On-Mechanism Via Ubiquitin-Proteasome System Inhibitors of the UPS Pathway VHL-inactive Degrader Does Not Affect ARID1B Levels Rescue Degradation Degradation Cell Viability Degradation ARID1B degrader + 750 nM MG132 (Proteosome inhibitor) + 250 nM Bortezomib (Velcade, Proteosome inhibitor) + 750 nM MLN4924 (Neddylation inhibitor) + 750 nM TAK-243 (Ub activating enzyme inhibitor) 43 |

44 | Selective ARID1B Degradation Demonstrated by Global Proteomics
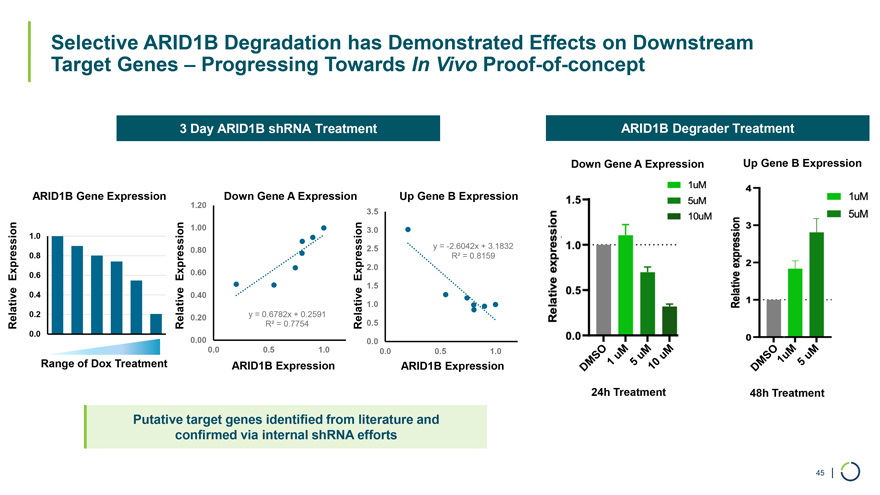
Selective ARID1B Degradation has Demonstrated Effects on Downstream Target Genes – Progressing Towards In Vivo Proof-of-concept 3 Day ARID1B shRNA Treatment ARID1B Degrader Treatment Down Gene A Expression Up Gene B Expression ARID1B Gene Expression 1.20 Down Gene A Expression Up Gene B Expression 3.5 1.0 1.00 3.0 0.80 2.5 y = -2.6042x + 3.1832 0.8 R² = 0.8159 2.0 Expression 0.6 Expression 0.60 Expression 1.5 0.4 0.40 1.0 0.2 0.20 y = 0.6782x + 0.2591 0.5 Relative Relative R² = 0.7754 Relative 0.0 6 0.00 0.0 0.0 0.5 1.0 0.0 0.5 1.0 Range of Dox Treatment ARID1B Expression ARID1B Expression 24h Treatment 48h Treatment Putative target genes identified from literature and confirmed via internal shRNA efforts 45 |
Developing First-in-Class Precision Medicines Targeting Major Unmet Needs in Cancer Leader in Unique Area of Cancer Biology Foghorn is a leader in targeting chromatin biology, which has the potential to address underlying dependencies of many genetically defined cancers Platform with initial focus in oncology, therapeutic area expansion potential Large Market Well-Potential Funded Chromatin biology is $158.9 million in cash and implicated in up to 50% of equivalents (unaudited) tumors, potentially (as of 12/31/2025) impacting ~2.5 million patients Cash runway into 2028 Foghorn’s current pipeline Shares outstanding: potentially addresses approximately 63.2M* more than 500,000 of (unaudited) these patients Broad pipeline across a range of targets and small molecule modalities Value Major Strategic Drivers Collaboration Selective SMARCA2 Inhibitor, Strategic collaboration with FHD-909, partnered with Lilly, Lilly; $380 million upfront; in Phase 1 trial 50/50 U.S. economic split on two lead programs Advancement of preclinical assets (Selective SMARCA2, CBP, EP300, ARID1B degraders) towards INDs Protein degrader platform with expansion into induced proximity 46 | *Includes common shares outstanding and pre-funded warrants as of 12/31/2025.

Unique biology Precision therapeutics Broad impact January 2026 47

Appendix 48

SMARCA2 Program 49
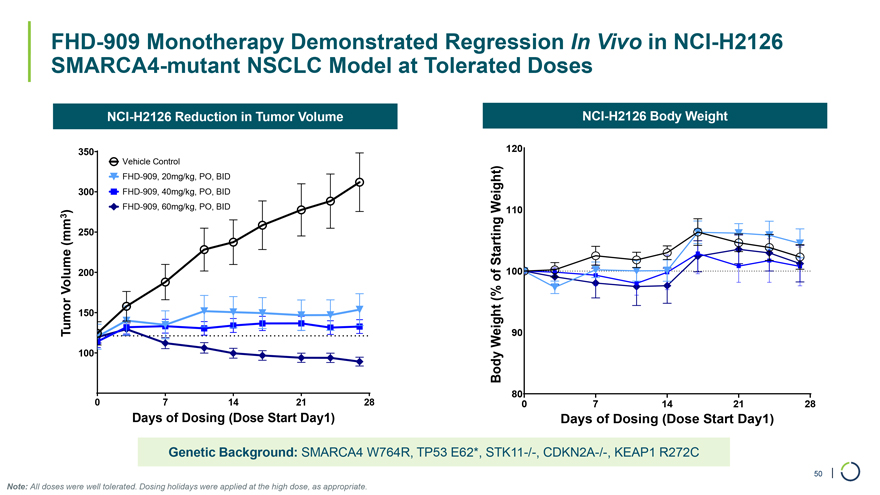
FHD-909 Monotherapy Demonstrated Regression In Vivo in NCI-H2126 SMARCA4-mutant NSCLC Model at Tolerated Doses NCI-H2126 Reduction in Tumor Volume NCI-H2126 Body Weight 350 120 Vehicle Control FHD-909, 20mg/kg, PO, BID 300 FHD-909, 40mg/kg, PO, BID ) FHD-909, 60mg/kg, PO, BID Weight) 110 3 (mm 250 Starting 200 of 100 Volume (% 150 Tumor Weight 90 100 Body 80 0 7 14 21 28 0 7 14 21 28 Days of Dosing (Dose Start Day1) Days of Dosing (Dose Start Day1) Genetic Background: SMARCA4 W764R, TP53 E62*, STK11-/-, CDKN2A-/-, KEAP1 R272C 50 | Note: All doses were well tolerated. Dosing holidays were applied at the high dose, as appropriate.
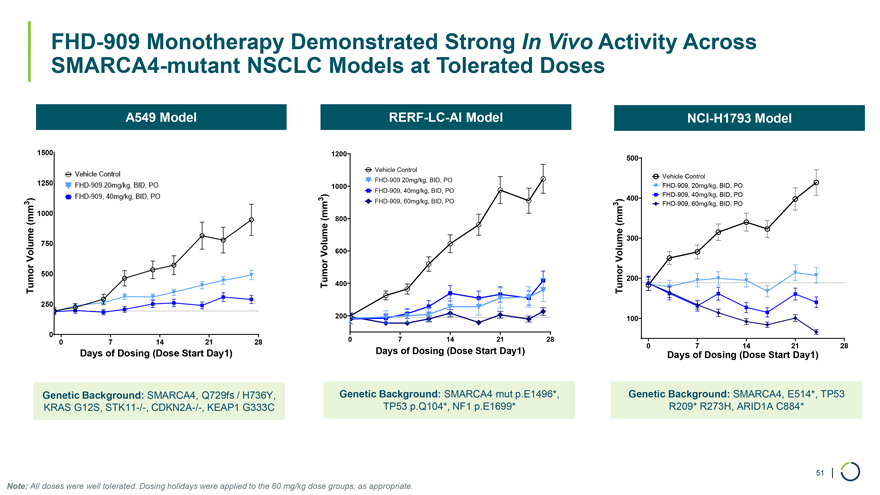
A549 Model RERF-LC-AI Model NCI-H1793 Model 1500 1200 500 Vehicle Control Vehicle Control 1250 FHD-909 20mg/kg, BID, PO 1000 FHD-909, 20mg/kg, BID, PO FHD-909, 40mg/kg, BID, PO ) 400 FHD-909, 40mg/kg, BID, PO 3 ) 3 FHD-909, 60mg/kg, BID, PO ) 3 FHD-909, 60mg/kg, BID, PO 1000 (mm 800 (mm (mm 300 750 Volume 600 Volume Volume 500 200 Tumor Tumor 400 Tumor FHD-909 Monotherapy Demonstrated Strong In Vivo Activity Across SMARCA4-mutant NSCLC Models at Tolerated Doses 250 200 100 0 0 7 14 21 28 0 7 14 21 28 Days of Dosing (Dose Start Day1) 0 7 14 21 28 Days of Dosing (Dose Start Day1) Days of Dosing (Dose Start Day1) Genetic Background: SMARCA4, Q729fs / H736Y, Genetic Background: SMARCA4 mut p.E1496*, Genetic Background: SMARCA4, E514*, TP53 KRAS G12S, STK11-/-, CDKN2A-/-, KEAP1 G333C TP53 p.Q104*, NF1 p.E1699* R209* R273H, ARID1A C884* 51 | Note: All doses were well tolerated. Dosing holidays were applied to the 60 mg/kg dose groups, as appropriate.
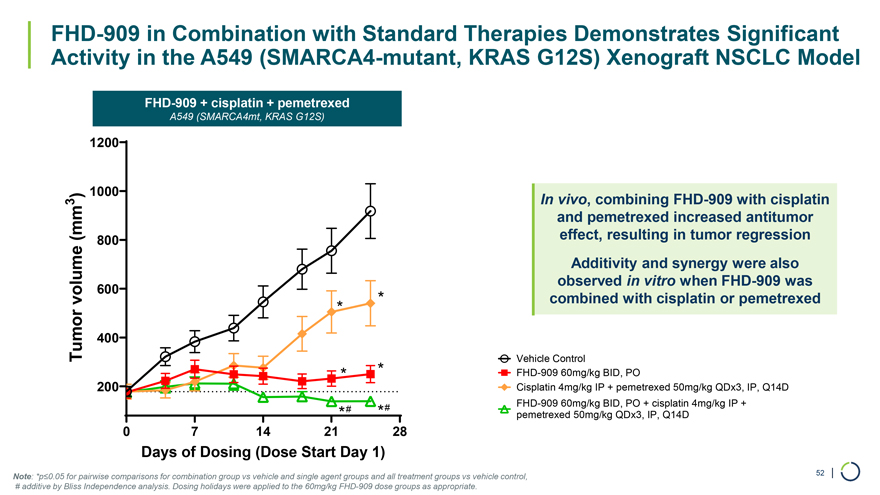
FHD-909 + cisplatin + pemetrexed A549 (SMARCA4mt, KRAS G12S) 1200 ) 1000 3 (mm 800 volume 600 * * Tumor 400 * * 200 *# *# 0 7 14 21 28 FHD-909 in Combination with Standard Therapies Demonstrates Significant Activity in the A549 (SMARCA4-mutant, KRAS G12S) Xenograft NSCLC Model In vivo, combining FHD-909 with cisplatin and pemetrexed increased antitumor effect, resulting in tumor regression Additivity and synergy were also observed in vitro when FHD-909 was combined with cisplatin or pemetrexed Vehicle Control FHD-909 60mg/kg BID, PO Cisplatin 4mg/kg IP + pemetrexed 50mg/kg QDx3, IP, Q14D FHD-909 60mg/kg BID, PO + cisplatin 4mg/kg IP + pemetrexed 50mg/kg QDx3, IP, Q14D Days of Dosing (Dose Start Day 1) Note: *p≤0.05 for pairwise comparisons for combination group vs vehicle and single agent groups and all treatment groups vs vehicle control, 52 | # additive by Bliss Independence analysis. Dosing holidays were applied to the 60mg/kg FHD-909 dose groups as appropriate.
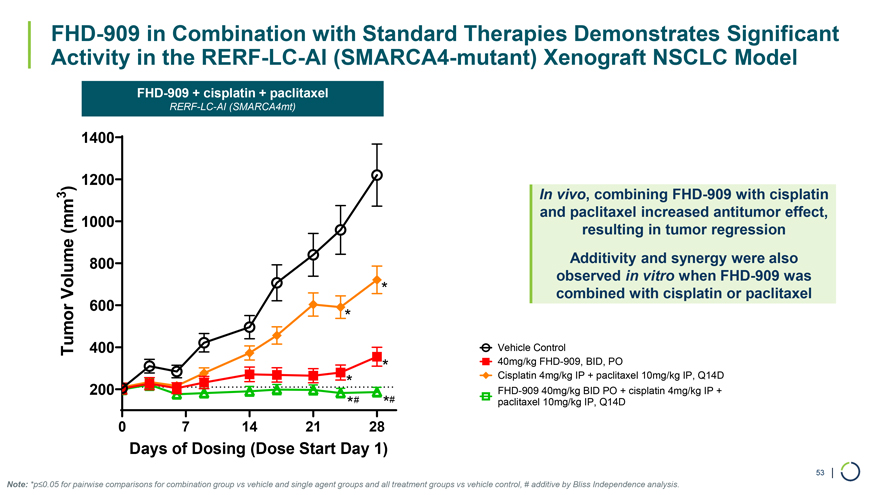
1400 1200 3 ) In vivo, combining FHD-909 with cisplatin and paclitaxel increased antitumor effect, (mm 1000 resulting in tumor regression 800 Additivity and synergy were also observed in vitro when FHD-909 was Volume * combined with cisplatin or paclitaxel 600 * Tumor 400 Vehicle Control * 40mg/kg FHD-909, BID, PO * Cisplatin 4mg/kg IP + paclitaxel 10mg/kg IP, Q14D 200 FHD-909 40mg/kg BID PO + cisplatin 4mg/kg IP + *# *# paclitaxel 10mg/kg IP, Q14D 0 7 14 21 28 Days of Dosing (Dose Start Day 1) 53 | FHD-909 in Combination with Standard Therapies Demonstrates Significant Activity in the RERF-LC-AI (SMARCA4-mutant) Xenograft NSCLC Model FHD-909 + cisplatin + paclitaxel RERF-LC-AI (SMARCA4mt) Note: *p≤0.05 for pairwise comparisons for combination group vs vehicle and single agent groups and all treatment groups vs vehicle control, # additive by Bliss Independence analysis.
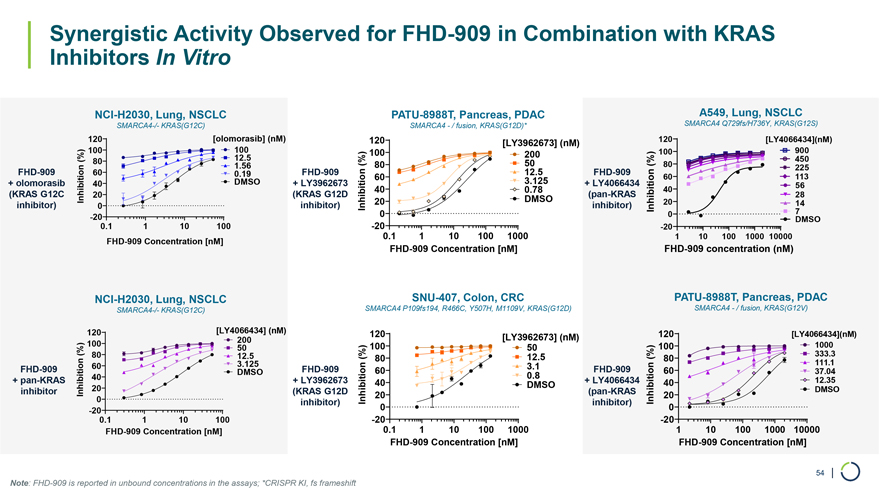
Synergistic Activity Observed for FHD-909 in Combination with KRAS Inhibitors In Vitro NCI-H2030, Lung, NSCLC PATU-8988T, Pancreas, PDAC A549, Lung, NSCLC SMARCA4-/- KRAS(G12C) SMARCA4—/ fusion, KRAS(G12D)* SMARCA4 Q729fs/H736Y, KRAS(G12S) 120 [olomorasib] (nM) 120 [LY3962673] (nM) 120 [LY4066434](nM) 100 100 100 200 100 900 (%) 80 12.5 450 (%) 80 50 (%) 80 FHD-909 60 1.56 FHD-909 12.5 FHD-909 225 0.19 60 60 113 + olomorasib 40 DMSO + LY3962673 3.125 + LY4066434 56 (KRAS G12C (KRAS G12D 40 0.78 (pan-KRAS 40 Inhibition 20 DMSO 28 inhibitor) 0 inhibitor) Inhibition 20 inhibitor) Inhibition 20 14 0 0 7 -20 DMSO 0.1 1 10 100 -200.1 1 10 100 1000 -20 1 10 100 1000 10000 FHD-909 Concentration [nM] FHD-909 Concentration [nM] FHD-909 concentration (nM) NCI-H2030, Lung, NSCLC SNU-407, Colon, CRC PATU-8988T, Pancreas, PDAC SMARCA4-/- KRAS(G12C) SMARCA4 P109fs194, R466C, Y507H, M1109V, KRAS(G12D) SMARCA4—/ fusion, KRAS(G12V) 120 [LY4066434] (nM) 120 120 [LY4066434](nM) 200 [LY3962673] (nM) 100 50 100 50 100 1000 (%) 80 12.5 (%) 12.5 (%) 333.3 80 80 111.1 60 3.125 3.1 FHD-909 DMSO FHD-909 60 FHD-909 60 37.04 + pan-KRAS 40 + LY3962673 0.8 + LY4066434 12.35 40 DMSO 40 inhibitor Inhibition 20 (KRAS G12D (pan-KRAS DMSO 0 Inhibition 20 Inhibition 20 inhibitor) 0 inhibitor) 0 -20 0.1 1 10 100 -200.1 1 10 100 1000 -20 1 10 100 1000 10000 FHD-909 Concentration [nM] FHD-909 Concentration [nM] FHD-909 Concentration [nM] 54 | Note: FHD-909 is reported in unbound concentrations in the assays; *CRISPR KI, fs frameshift
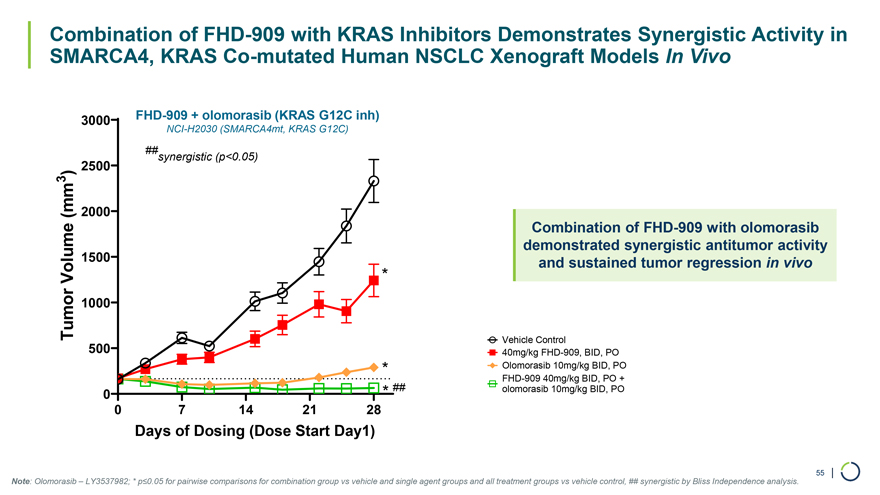
3000 FHD-909 + olomorasib (KRAS G12C inh) NCI-H2030 (SMARCA4mt, KRAS G12C) ## synergistic (p<0.05) 2500 3 ) (mm 2000 Combination of FHD-909 with olomorasib demonstrated synergistic antitumor activity 1500 and sustained tumor regression in vivo Volume * Tumor 1000 Vehicle Control 500 40mg/kg FHD-909, BID, PO * Olomorasib 10mg/kg BID, PO FHD-909 40mg/kg BID, PO + 0 * ## olomorasib 10mg/kg BID, PO 0 7 14 21 28 Days of Dosing (Dose Start Day1) 55 | Combination of FHD-909 with KRAS Inhibitors Demonstrates Synergistic Activity in SMARCA4, KRAS Co-mutated Human NSCLC Xenograft Models In Vivo Note: Olomorasib – LY3537982; * p≤0.05 for pairwise comparisons for combination group vs vehicle and single agent groups and all treatment groups vs vehicle control, ## synergistic by Bliss Independence analysis.
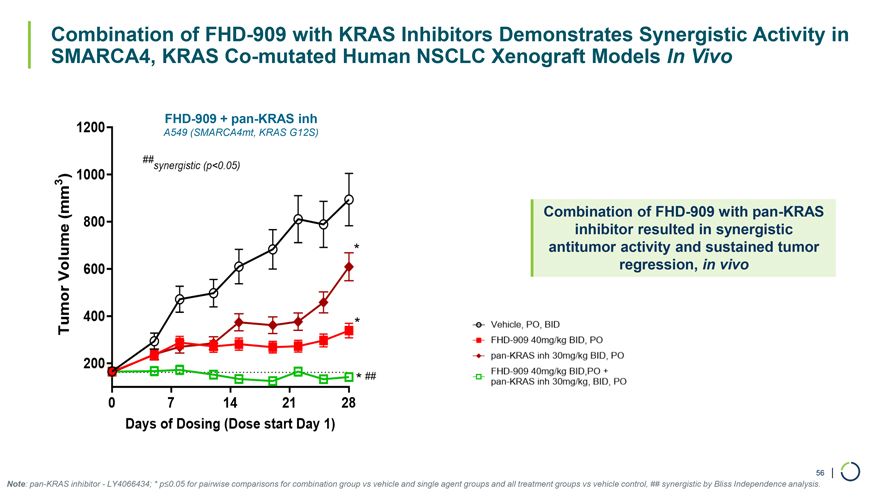
Combination of FHD-909 with KRAS Inhibitors Demonstrates Synergistic Activity in SMARCA4, KRAS Co-mutated Human NSCLC Xenograft Models In Vivo FHD-909 + pan-KRAS inh A549 (SMARCA4mt, KRAS G12S) Combination of FHD-909 with pan-KRAS inhibitor resulted in synergistic antitumor activity and sustained tumor regression, in vivo 56 | Note: pan-KRAS inhibitor—LY4066434; * p≤0.05 for pairwise comparisons for combination group vs vehicle and single agent groups and all treatment groups vs vehicle control, ## synergistic by Bliss Independence analysis.
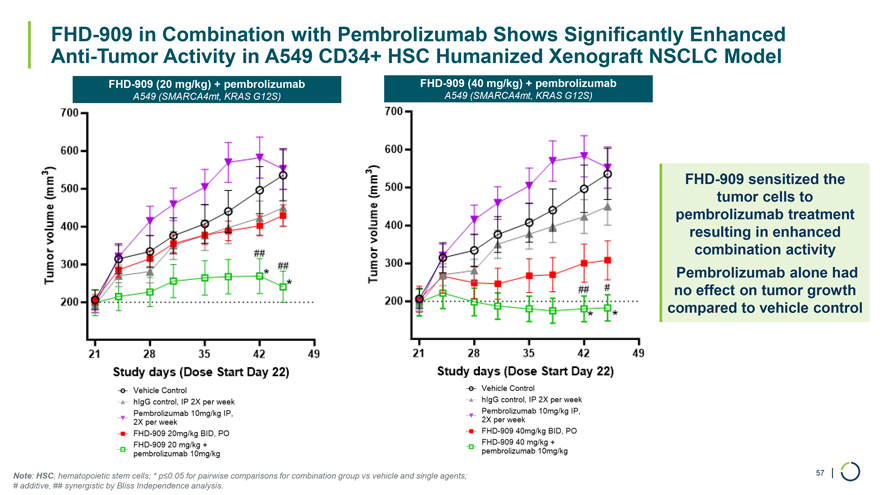
FHD-909 in Combination with Pembrolizumab Shows Significantly Enhanced Anti-Tumor Activity in A549 CD34+ HSC Humanized Xenograft NSCLC Model FHD-909 (20 mg/kg) + pembrolizumab FHD-909 (40 mg/kg) + pembrolizumab A549 (SMARCA4mt, KRAS G12S) A549 (SMARCA4mt, KRAS G12S) FHD-909 sensitized the tumor cells to pembrolizumab treatment resulting in enhanced combination activity Pembrolizumab alone had no effect on tumor growth compared to vehicle control Note: HSC, hematopoietic stem cells; * p≤0.05 for pairwise comparisons for combination group vs vehicle and single agents; 57 | # additive, ## synergistic by Bliss Independence analysis.
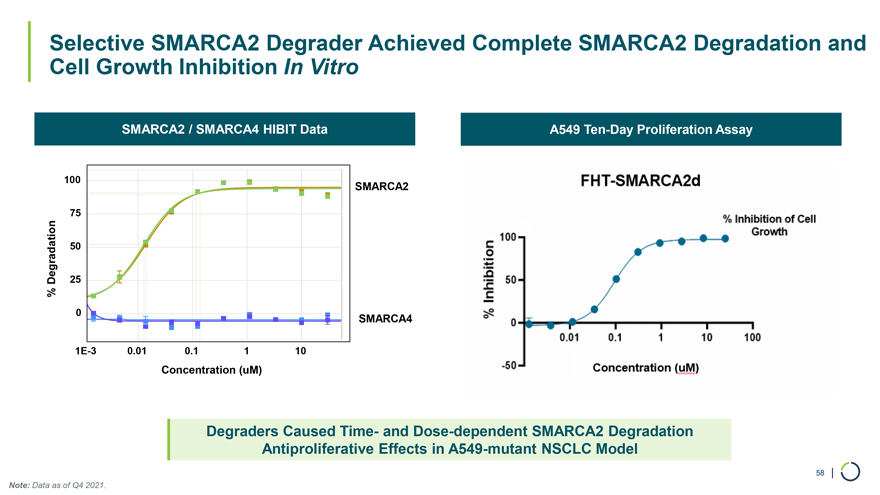
Selective SMARCA2 Degrader Achieved Complete SMARCA2 Degradation and Cell Growth Inhibition In Vitro SMARCA2 / SMARCA4 HIBIT Data A549 Ten-Day Proliferation Assay 100 SMARCA2 75 50 Degradation 25 % 0 SMARCA4 1E-3 0.01 0.1 1 10 Concentration (uM) Degraders Caused Time- and Dose-dependent SMARCA2 Degradation Antiproliferative Effects in A549-mutant NSCLC Model 58 | Note: Data as of Q4 2021.

Protein Degradation Importance of Rate Analysis FHD-609 is a Selective, Potent, Protein Degrader of the BRD9 component of the BAF complex 59
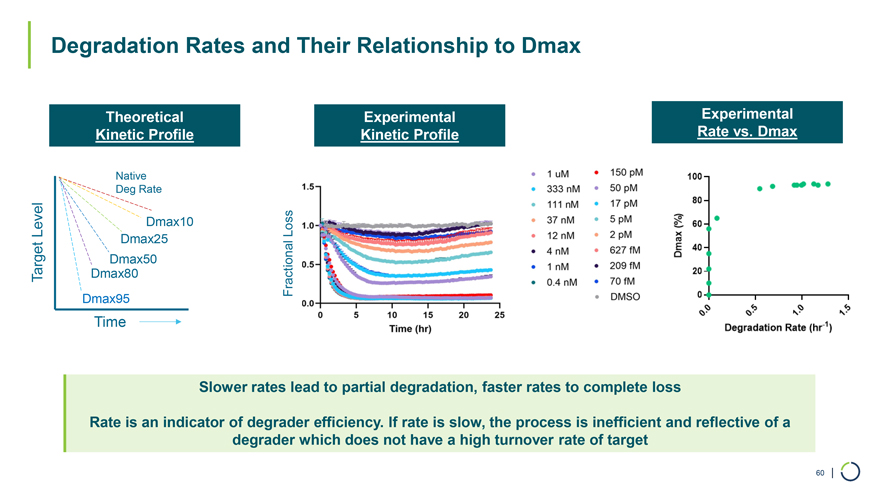
Theoretical Experimental Experimental Kinetic Profile Kinetic Profile Rate vs. Dmax Native Deg Rate Level Dmax10 Loss Dmax25 Dmax50 Target Dmax80 Fractional Dmax95 Time Degradation Rates and Their Relationship to Dmax Slower rates lead to partial degradation, faster rates to complete loss Rate is an indicator of degrader efficiency. If rate is slow, the process is inefficient and reflective of a degrader which does not have a high turnover rate of target 60 |

Foghorn BRD9 Program Foghorn CBP Program Foghorn EP300 Program Degradation Rate Dictates Dmax – Program Independent Large scale experimental kinetic analysis for a program reveals the relationship between rate and Dmax 61 |
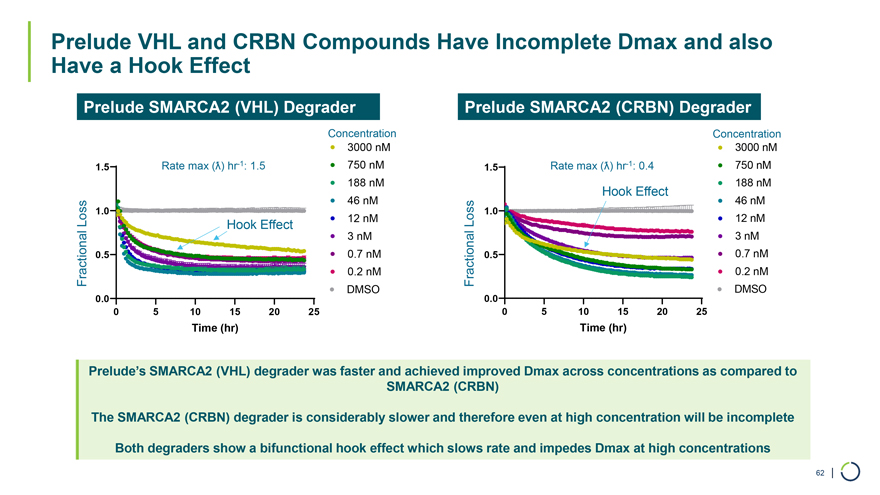
Prelude SMARCA2 (VHL) Degrader Prelude SMARCA2 (CRBN) Degrader Concentration Concentration 3000 nM 3000 nM 1.5 Rate max hr-1: 1.5 750 nM 1.5 Rate max hr-1: 0.4 750 nM 188 nM 188 nM Hook Effect 46 nM 46 nM 1.0 1.0 Loss Hook Effect 12 nM Loss 12 nM Luminescence 3 nM Luminescence 3 nM (RLU) 0.7 nM (RLU) 0.7 nM 0.5 0.5 Fractional 0.2 nM Fractional 0.2 nM Fractional DMSO Fractional DMSO 0.0 0.0 0 5 10 15 20 25 0 5 10 15 20 25 Time (hr) Time (hr) Prelude VHL and CRBN Compounds Have Incomplete Dmax and also Have a Hook Effect Prelude’s SMARCA2 (VHL) degrader was faster and achieved improved Dmax across concentrations as compared to SMARCA2 (CRBN) The SMARCA2 (CRBN) degrader is considerably slower and therefore even at high concentration will be incomplete Both degraders show a bifunctional hook effect which slows rate and impedes Dmax at high concentrations 62 |
Foghorn Analysis and Dmax Results Match Published Prelude Data Prelude Published Data Foghorn Replicated Data from Prelude Patents Prelude PRT3789 (VHL) 1 Prelude (VHL) Degrader VHL degrader (2 hours) 100 VHL degrader (24 hours) 100 SMARCA2 80 80 60 60 Degradation 40 Degradation 40 % % 20 20 0 0 0.01 0.1 1 10 100 1000 10000 0.01 0.1 1 10 100 1000 10000 Concentration (nM) Concentration (nM) Prelude PRT7732 (CRBN) 2 Prelude (CRBN) Degrader 100 SMARCA2 SMARCA4 Protein 50 % 0 0.01 0.1 1 10 100 1000 10000 Concentration (nM) 1. AACR-NCI-EORTC 2023 Poster B113 – Ito et al. 63 | 2. AACR 2024 Poster 4503 -Shvartsbart et al.
Exhibit 99.3
SECURITIES PURCHASE AGREEMENT
THIS SECURITIES PURCHASE AGREEMENT (the “Agreement”) is made as of the 9th day of January, 2026, by and among Foghorn Therapeutics Inc., a Delaware corporation (the “Company”), and each of the investors set forth on the signature pages hereto (the “Investors”).
THE PARTIES HEREBY AGREE AS FOLLOWS:
1. Purchase and Sale of Securities.
1.1 Sale and Issuance. Subject to the terms and conditions of this Agreement, each Investor, severally and not jointly, agrees to purchase at the Closing and the Company agrees to sell and issue to such Investor at the Closing (i) the number of shares of the Company’s common stock, $0.0001 par value (the “Common Stock”), set forth in Exhibit A hereto at a purchase price of $6.71 per share (the “Shares”); provided, however, that, at the option of the Investor, in lieu of purchasing Common Stock, such Investor may elect to purchase pre-funded warrants to purchase shares of Common Stock with an exercise price of $0.0001 per share (the “Pre-Funded Warrants”) at a purchase price of $6.71 minus the $0.0001 per share, and (ii) (x) a warrant to purchase a number of shares of Common Stock, or, at the option of the Investor, in lieu thereof, pre-funded warrants to purchase a number of shares of Common Stock (the “Series 1 Underlying Pre-Funded Warrants”), equal to fifty percent (50%) of the aggregate number of Shares and shares underlying the Pre-Funded Warrants, if any, purchased by such Investor, as indicated in Exhibit A hereto (each, a “Series 1 Warrant”) with an initial exercise price of $13.42 per share and (y) a warrant to purchase a number of shares of Common Stock, or, at the option of the Investor, in lieu thereof, pre-funded warrants to purchase a number of shares of Common Stock (the “Series 2 Underlying Pre-Funded Warrants” and, together with the Series 1 Underlying Pre-Funded Warrants, the “Underlying Pre-Funded Warrants”), equal to fifty percent (50%) of the number of Shares and shares underlying the Pre-Funded Warrants, if any, purchased by such Investor, as indicated in Exhibit A hereto (each, a “Series 2 Warrant” and, together with the Series 1 Warrant, the “Series Warrants”) with an initial exercise price of $20.13 per share (the Series Warrants together with the Shares and the Pre-Funded Warrants, the “Securities”). The shares of Common Stock issuable upon exercise of the Series Warrants, the Pre-Funded Warrants, and the Underlying Pre-Funded Warrants are collectively referred to herein as the “Warrant Shares.”
1.2 Closing; Conditions to Closing. The purchase and sale of the Securities shall take place at the offices of Ropes & Gray LLP located at 800 Boylston Street, Boston, MA 02199 at 10:00 A.M. Eastern Time, on January 13, 2026, or at such other time and place as the Company and the Investors may mutually agree upon in writing (which time and place are designated as the “Closing”). At the Closing, the Company shall (a) cause its transfer agent to deliver to each Investor, on an expedited basis via The Depository Trust Company Deposit or Withdrawal at Custodian system (“DWAC”) in accordance with the instructions for such Investor provided in writing to the Company, the Shares, (b) deliver to each investor the Series Warrants and, if applicable, the Pre-Funded Warrants, in each case that such Investor is purchasing against payment of the purchase price therefor by wire transfer of immediately available funds to an account specified by the Company in writing to the Investors, and (c) cause its outside legal counsel, Ropes & Gray LLP, to deliver to each Investor a legal opinion in a form to be agreed to between the Company and the Investors.
Notwithstanding the foregoing and anything in this Agreement to the contrary and as may be agreed to among the Company and one or more Investors, if an Investor notifies the Company on or prior to the date hereof that it is (i) an investment company registered under the Investment Company Act of 1940, as amended, (ii) advised by an investment adviser subject to regulation under the Investment Advisers Act of 1940, as amended, or (iii) otherwise subject to internal policies and/or procedures relating to the timing of funding and issuance of securities that would require it, such Investor shall not be required to wire such funds until the Closing and until it confirms receipt of the Shares via DWAC and evidence acceptable to the Investor evidencing the issuance of the Series Warrants and Pre-Funded Warrants, if applicable on and as of the date of the Closing.
The obligations of each Investor hereunder in connection with the Closing are subject to satisfaction (or waiver in writing by each Investor solely as to such Investor) of the following conditions: (i) the representations and warranties of the Company contained herein shall be true and correct in all respects as of the date hereof and as of the Closing as if made as of the Closing (unless such representation or warranty was made as of a specific date, in which case such representation and warranty shall be true and correct in all respects as of such date); (ii) all obligations, covenants and agreements to be performed or complied with by the Company on or prior to the Closing shall have been performed or complied with by it; (iii) the delivery by the Company of (A) the items set forth in this Section 2.1 and (B) the Base Prospectus and Prospectus Supplement (which may be delivered in accordance with Rule 172 under the Securities Act); (iv) no Material Adverse Effect shall have occurred since the date hereof; (v) from the date hereof to the Closing, trading in the Common Stock shall not have been suspended by the Commission or Nasdaq, nor shall suspension have been threatened by the Commission or Nasdaq; (vi) no stop order suspending the effectiveness of the Registration Statement or any part thereof, or preventing or suspending the use of the Base Prospectus or the Prospectus Supplement or any part thereof, shall have been issued, and no proceedings for that purpose or otherwise under the Securities Act, shall have been initiated or threatened by the Commission, and (vii) the Company shall have filed with Nasdaq a Notification Form: Listing of Additional Shares for the listing of the Shares and Warrant Shares and no objection shall have been raised by the Nasdaq with respect to the consummation of the transactions contemplated by this Agreement.
2. Representations and Warranties of the Company. The Company hereby represents and warrants to each Investor as of the date hereof and as of the Closing that:
2.1 The Company meets the requirements for use of Form S-3 under the Securities Act of 1933, as amended (the “Securities Act”), and has filed with the Securities and Exchange Commission (the “Commission”) a registration statement on such Form S-3 (Registration File No. 333-284476), which became effective as of January 31, 2025, for the registration under the Securities Act of the Securities, the Underlying Pre-Funded Warrants and the Warrant Shares. Such registration statement meets the requirements set forth in Rule 415(a)(1)(x) under the Securities Act and complies with said Rule. The Company will file with the Commission pursuant to Rule 424(b) under the Securities Act, and the rules and regulations (the “Rules and Regulations”) of the Commission promulgated thereunder, a supplement to the form of prospectus filed with the Commission on January 24, 2025. Such registration statement, including the exhibits thereto, as amended at the date of this Agreement, is hereinafter called the “Registration Statement”; such prospectus in the form filed with the Commission on January 24, 2025, is hereinafter called the “Base Prospectus”; and the form of prospectus supplement, in the form in which it will be filed with the Commission pursuant to Rule 424(b) (including the Base Prospectus as so supplemented) is hereinafter called the “Prospectus Supplement.” Any reference herein to the Registration Statement, the Base Prospectus or the Prospectus Supplement shall be deemed to refer to and include the documents incorporated by reference therein (the “Incorporated Documents”) pursuant to Item 12 of Form S-3 which were filed under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), on or before the date of this Agreement, or the issue date of the Base Prospectus or the Prospectus Supplement, as the case may be; and any reference herein to the terms “amend,” “amendment” or “supplement” with respect to the Registration Statement, the Base Prospectus or the Prospectus Supplement shall be deemed to refer to and include the filing of any document under the Exchange Act after the date of this Agreement, or the issue date of the Base Prospectus or the Prospectus Supplement, as the case may be, deemed to be incorporated therein by reference. All references in this Agreement to financial statements and schedules and other information which is “contained,” “included,” “described,” “set forth” or “stated” in the Registration Statement, the Base Prospectus or the Prospectus Supplement (and all other references of like import) shall be deemed to mean and include all such financial statements and schedules and other information which is or is deemed to be incorporated by reference in the Registration Statement, the Base Prospectus or the Prospectus Supplement, as the case may be. No stop order suspending the effectiveness of the Registration Statement or the use of the Base Prospectus or the Prospectus Supplement has been issued, and no proceeding for any such purpose is pending or has been initiated or, to the Company’s knowledge, is threatened by the Commission.
2
2.2 The Registration Statement contains all exhibits and schedules as required by the Securities Act. Each of the Registration Statement and any post-effective amendment thereto, at the time it became effective, complied in all material respects with the Securities Act and the Exchange Act and the applicable Rules and Regulations and did not, as amended or supplemented, if applicable, contain any untrue statement of a material fact or omit to state a material fact required to be stated therein or necessary to make the statements therein not misleading. The Base Prospectus and the Prospectus Supplement, each as of its respective date, complied in all material respects with the Securities Act and the Exchange Act and the applicable Rules and Regulations. Each of the Base Prospectus and the Prospectus Supplement, as amended or supplemented, did not contain as of the date thereof any untrue statement of a material fact or omit to state a material fact necessary in order to make the statements therein, in light of the circumstances under which they were made, not misleading. The Incorporated Documents, when they were filed with the Commission, conformed in all material respects to the requirements of the Exchange Act and the applicable Rules and Regulations and none of such Incorporated Documents, when they were filed with the Commission, contained any untrue statement of a material fact or omitted to state a material fact necessary to make the statements therein, in light of the circumstances under which they were made, not misleading. Notwithstanding the foregoing, the Company makes no representations or warranties as to information, if any, contained in or omitted from the Prospectus Supplement or any amendment thereof or supplement thereto in reliance upon and in conformity with information furnished in writing to the Company by or on behalf of any Investor specifically for use in the Registration Statement or the Prospectus Supplement. No post-effective amendment to the Registration Statement reflecting any facts or events arising after the date thereof which represent, individually or in the aggregate, a fundamental change in the information set forth therein is required to be filed with the Commission. There are no documents required to be filed with the Commission in connection with the transaction contemplated hereby that have not been filed as required pursuant to the Securities Act. The financial statements of the Company incorporated by reference in the Registration Statement, comply in all material respects with applicable accounting requirements and the rules and regulations of the Commission with respect thereto as in effect at the time of filing. Such financial statements have been prepared in accordance with United States generally accepted accounting principles applied on a consistent basis during the periods involved (“GAAP”), except as may be otherwise specified in such financial statements or the notes thereto and except that unaudited financial statements may not contain all footnotes required by GAAP, and fairly present in all material respects the financial position of the Company as of and for the dates thereof and the results of operations and cash flows for the periods then ended, subject, in the case of unaudited statements, to normal year-end audit adjustments.
2.3 Neither the Company nor any of its directors and officers has distributed and none of them will distribute, prior to the Closing, any offering material in connection with the offering and sale of the Securities other than the Base Prospectus, the Prospectus Supplement, the Registration Statement, copies of the documents incorporated by reference therein and any other materials permitted by the Securities Act.
2.4 The Company has been duly organized and is validly existing as a corporation and is in good standing under the laws of the State of Delaware as of the date hereof, and is duly qualified to do business and as in good standing in each other jurisdiction in which its ownership or lease of property or the conduct of business requires such qualification, except where the failure to qualify, singularly or in the aggregate, would not have or reasonably be expected to result in a material adverse effect on the business, properties, operations, condition (financial or otherwise) or results of operations of the Company taken as a whole, or in its ability to perform its obligations under this Agreement (a “Material Adverse Effect”). All direct and indirect subsidiaries of the Company (“Subsidiaries”) are duly organized and in good standing under the laws of the place of organization or incorporation, and each Subsidiary is in good standing in each jurisdiction in which its ownership or lease of property or the conduct of business requires such qualification, except where the failure to qualify would not have a Material Adverse Effect on the assets, business or operations of the Company taken as a whole.
2.5 The Company has the requisite corporate power and authority to enter into and to consummate the transactions contemplated by this Agreement and otherwise to carry out its obligations hereunder. The execution and delivery of this Agreement by the Company and the consummation by it of the transactions contemplated hereby have been duly authorized by all necessary corporate action on the part of the Company and no further corporate consent or action is required to be obtained by the Company, its Board of Directors or its stockholders in connection therewith. This Agreement has been (or upon delivery will have been) duly executed by the Company and, when delivered in accordance with the terms hereof, will constitute the legally valid and binding obligation of the Company enforceable against the Company in accordance with its terms except (i) as limited by general equitable principles and applicable bankruptcy, insolvency, reorganization, moratorium and other laws of general application affecting enforcement of creditors’ rights generally, (ii) as limited by laws relating to the availability of specific performance, injunctive relief or other equitable remedies and (iii) insofar as indemnification and contribution provisions may be limited by applicable law. Subject to the accuracy of the representations and warranties of each Investor set forth in Section 3 hereof, the execution, delivery and performance by the Company of this Agreement and the offer, issuance and sale of the Securities require no consent of, action by or in respect of, or filing with, any person, governmental body, agency, or official other than (a) pursuant to applicable state and federal securities laws, (b) filings pursuant to the rules and regulations of Nasdaq, each of which the Company has filed or undertakes to file within the applicable time and (c) the requisite consent of any persons that the Company has obtained in connection with the transactions contemplated hereby.
3
2.6 The Securities have been duly authorized for issuance and sale pursuant to this Agreement and, when issued and delivered by the Company pursuant to this Agreement, will be validly issued, fully paid and non-assessable, and free and clear of all liens imposed by the Company. The Warrant Shares have been duly authorized and reserved for issuance and sale pursuant to the terms of the Series Warrant, Pre-Funded Warrant or Underlying Pre-Funded Warrant, as applicable, and when issued against payment therefor pursuant to the terms of such warrant, will be validly issued, fully paid and non-assessable, and free and clear of all liens imposed by the Company. Neither the issuance of the Securities nor the Warrant Shares is subject to any preemptive rights and will not result in any adjustment (automatic, at the election of any Person or otherwise) of the exercise, conversion, exchange or reset price under, or any other anti-dilution adjustment pursuant to, any outstanding securities of the Company.
2.7 The Company and its Board of Directors have taken all necessary action, if any, in order to render inapplicable any control share acquisition, business combination, poison pill (including any distribution under a rights agreement), or other similar anti-takeover provision pursuant to the Third Amended and Restated Certificate of Incorporation of the Company, the Amended and Restated By-laws of the Company or the laws of its state of incorporation that is or could become applicable to the Investors as a result of the Investors and the Company fulfilling their obligations or exercising their rights pursuant to the Agreement and the transactions contemplated hereby, including without limitation, as a result of the Company’s issuance of the Securities and the Investors’ ownership of the Securities. All such anti-takeover provisions in effect as of the date hereof are summarized generally in the Base Prospectus under the caption, “Description of Common Stock – Anti-Takeover Effects of our Certificate of Incorporation and our By-Laws.”
2.8 All issued and outstanding securities of the Company issued prior to the transactions contemplated by this Agreement have been duly authorized and validly issued and are fully paid and non-assessable; the holders thereof have no rights of rescission with respect thereto, and are not subject to personal liability by reason of being such holders; and none of such securities were issued in violation of the preemptive rights of any holders of any security of the Company or similar contractual rights granted by the Company.
2.9 The Common Stock is registered pursuant to Section 12(b) or 12(g) of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), and the Company has taken no action designed to, or which to its knowledge is likely to have the effect of, terminating the registration of the Common Stock pursuant to the Exchange Act nor has the Company received any notification that the Commission is currently contemplating terminating such registration. The Company is currently in compliance with all applicable Nasdaq listing and maintenance requirements and, except as disclosed in its filings with the Commission, the Company has not, in the twelve (12) months preceding the date hereof, received notice from Nasdaq to the effect that the Company is not in compliance with such listing or maintenance requirements.
2.10 The Company is in material compliance with any and all applicable requirements of the Sarbanes-Oxley Act of 2002 that are effective as of the date hereof, and any and all applicable rules and regulations promulgated by the Commission thereunder that are effective as of the date hereof and as of the Closing. The Company maintains a system of internal accounting controls designed to provide reasonable assurance that: (i) transactions are executed in accordance with management’s general or specific authorizations, (ii) transactions are recorded as necessary to permit preparation of financial statements in conformity with GAAP and to maintain asset accountability, (iii) access to assets is permitted only in accordance with management’s general or specific authorization, and (iv) the recorded accountability for assets is compared with the existing assets at reasonable intervals and appropriate action is taken with respect to any differences. The Company has established disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) for the Company and designed such disclosure controls and procedures to ensure that information required to be disclosed by the Company in the reports it files or submits under the Exchange Act is recorded, processed, summarized and reported, within the time periods specified in the Commission’s rules and forms and (ii) there have been no material weaknesses in the Company’s internal control over financial reporting (whether or not remediated) and no change in the Company’s internal control over financial reporting that has materially affected, or would reasonably be expected to materially affect, the Company’s internal control over financial reporting.
4
2.11 The Company: (i) is not in default under or in violation of (and no event has occurred that has not been waived that, with notice or lapse of time or both, would reasonably be expected to result in a default by the Company or any of its subsidiaries under), nor has the Company received notice of a claim that it is in default under or that it is in violation of, any indenture, loan or credit agreement or any other agreement or instrument to which it is a party or by which it or any of its properties is bound (whether or not such default or violation has been waived), (ii) is not in violation of any judgment, decree or order of any court, arbitrator or other governmental authority and (iii) is not and has not been in violation of any statute, rule, ordinance or regulation of any governmental authority, including without limitation all foreign, federal, state and local laws relating to taxes, environmental protection, occupational health and safety, product quality and safety and employment and labor matters, except in each case as would not have or would not reasonably be expected to have a Material Adverse Effect. Since September 30, 2025, except as identified and described in the Registration Statement, Base Prospectus, Prospectus Supplement and/or Incorporated Documents, there has not been any event or condition of any character that has had or would reasonably be expected to have a Material Adverse Effect.
3. Representations and Warranties of the Investor. Each Investor hereby represents and warrants to the Company that the Investor has full right, power and authority to enter into this Agreement and to consummate the transactions contemplated hereby and has taken all necessary action to authorize the execution, delivery and performance of this Agreement. This Agreement constitutes a valid and binding obligation of the Investor enforceable against the Investor in accordance with its terms, except as enforceability may be limited by applicable bankruptcy, insolvency, reorganization, moratorium or similar laws affecting creditors’ and contracting parties’ rights generally and except as enforceability may be subject to general principles of equity (regardless of whether such enforceability is considered in a proceeding in equity or at law).
4. Miscellaneous.
4.1 Integration. After this transaction, the Company shall not sell, offer for sale or solicit offers to buy or otherwise negotiate in respect of any security (as defined in Section 2 of the Securities Act) that would be integrated with the offer or sale of the Securities such that the rules of The Nasdaq Stock Market would require stockholder approval of this transaction prior to the closing of such other transaction unless stockholder approval is obtained before the closing of such subsequent transaction.
4.2 Successors and Assigns. Except as otherwise provided herein, the terms and conditions of this Agreement shall inure to the benefit of and be binding upon the respective successors and assigns of the parties. Nothing in this Agreement, express or implied, is intended to confer upon any party other than the parties hereto or their respective successors and assigns any rights, remedies, obligations, or liabilities under or by reason of this Agreement, except as expressly provided in this Agreement.
4.3 Governing Law. This Agreement shall be governed by and construed under the laws of the State of New York as applied to agreements among New York residents entered into and to be performed entirely within New York.
4.4 Execution. This Agreement may be executed in two or more counterparts, each of which shall be deemed an original, but all of which together shall constitute one and the same instrument. Counterparts may be delivered via electronic mail (including pdf or any electronic signature complying with the U.S. federal ESIGN Act of 2000, e.g., www.docusign.com) or other transmission method and any counterpart so delivered shall be deemed to have been duly and validly delivered and be valid and effective for all purposes.
4.5 Titles and Subtitles. The titles and subtitles used in this Agreement are used for convenience only and are not to be considered in construing or interpreting this Agreement.
4.6 Notices. Unless otherwise provided, any notice required or permitted under this Agreement shall be given in writing (including electronic mail as permitted in this Agreement) and shall be deemed effectively given upon personal delivery to the party to be notified, when sent, if sent by electronic mail during normal business hours of the recipient, and if not sent during normal business hours, then on the recipient’s next business day, or upon deposit with the United States Post Office, by registered or certified mail, postage prepaid and addressed to the party to be notified at the address indicated for such party on the signature page hereof, or at such other address as such party may designate by ten (10) days’ advance written notice to the other parties.
5
4.7 Consent to Electronic Notice. Each Investor consents to the delivery of any notice pursuant this Agreement at the e-mail address set forth below such Investor’s name on the signature page, as updated from time to time by notice to the Company. To the extent that any notice given by means of electronic mail is returned or undeliverable for any reason, the foregoing consent shall be deemed to have been revoked until a new or corrected e-mail address has been provided, and such attempted electronic notice shall be ineffective and deemed to not have been given. Each party agrees to promptly notify the other parties of any change in its e-mail address, and that failure to do so shall not affect the foregoing.
4.8 Finders’ Fee. Each party represents that it neither is nor will be obligated for any finders’ fee or commission in connection with this transaction. Each Investor agrees to indemnify and to hold harmless the Company from any liability for any commission or compensation in the nature of a finders’ fee (and the costs and expenses of defending against such liability or asserted liability) for which such Investor or any of its officers, partners, employees, or representatives is responsible. The Company agrees to indemnify and hold harmless each Investor from any liability for any commission or compensation in the nature of a finders’ fee (and the costs and expenses of defending against such liability or asserted liability) for which the Company or any of its officers, employees or representatives is responsible.
4.9 Amendments and Waivers. Any term of this Agreement may be amended and the observance of any term of this Agreement may be waived (either generally or in a particular instance and either retroactively or prospectively), only with the written consent of the Company and each Investor; provided, however, that any provision of this Agreement may be waived by any waiving party on such party’s own behalf, without the consent of any other Investor.
4.10 Severability. If one or more provisions of this Agreement are held to be unenforceable under applicable law, such provision shall be excluded from this Agreement and the balance of the Agreement shall be interpreted as if such provision were so excluded and shall be enforceable in accordance with its terms.
4.11 Entire Agreement. This Agreement and the other documents referred to herein constitute the entire agreement among the parties and no party shall be liable or bound to any other party in any manner by any warranties, representations, or covenants except as specifically set forth herein or therein.
4.12 Indemnification. Subject to the provisions of this Section 4.12, the Company will indemnify and hold each Investor and its directors, officers, shareholders, members, partners, advisers, employees and agents (and any other persons with a functionally equivalent role of a person holding such titles notwithstanding a lack of such title or any other title), each person who controls such Investor (within the meaning of Section 15 of the Securities Act and Section 20 of the Exchange Act), and the directors, officers, shareholders, agents, members, partners, advisers or employees (and any other persons with a functionally equivalent role of a person holding such titles notwithstanding a lack of such title or any other title) of such controlling persons (each, a “Investor Party”) harmless from any and all losses, liabilities, obligations, claims, contingencies, damages, costs and expenses, including all judgments, amounts paid in settlements, court costs and reasonable and documented attorneys’ fees and costs of investigation that any such Investor Party may suffer or incur as a result of or relating to (a) any breach of any of the representations, warranties, covenants or agreements made by the Company in this Agreement or (b) any action instituted against an Investor, or any of them or their respective affiliates, by any stockholder of the Company who is not an affiliate of such Investor or any governmental or regulatory agency, with respect to any of the transactions contemplated by this Agreement (unless such action is based upon a material breach of such Investor’s representations, warranties or covenants in this Agreement or any material violations by the Investor of state or federal securities laws or any conduct by such Investor (or any Investor Party related to such Investor) which constitutes fraud, gross negligence, willful misconduct or malfeasance). If any action shall be brought against any Investor Party in respect of which indemnity may be sought pursuant to this Agreement, such Investor Party shall promptly notify the Company in writing, and the Company shall have the right to assume the defense thereof with counsel of its own choosing reasonably acceptable to the Investor Party. Any Investor Party shall have the right to engage separate counsel in any such action and participate in the defense thereof, but the fees and expenses of such counsel shall be at the expense of such Investor Party except to the extent that (i) the engagement thereof has been specifically authorized by the Company in writing, (ii) the Company has failed after a reasonable period of time to assume such defense and to employ counsel or (iii) in such action there is, in the reasonable opinion of such separate counsel, a material conflict on any material issue between the position of the Company and the position of such Investor Party, in which case the Company shall be responsible for the reasonable fees and expenses of no more than one such separate counsel. The Company will not be liable to any Investor Party under this Agreement (i) for any settlement by a Investor Party effected without the Company’s prior written consent, which shall not be unreasonably withheld or delayed, or (ii) to the extent, but only to the extent, that a loss, claim, damage or liability is attributable to any Investor Party’s breach of any of the representations, warranties, covenants or agreements made by such Investor Party in this Agreement. To the extent that an Investor Party wishes to seek indemnification under this Section 4.12, such Investor Party must provide the Company with written notice asserting a claim under this Section 4.12, with such notice to be promptly provided and in any event within one year from the Closing. If an Investor Party fails to provide such written notice within this one-year period, the Investor Party shall no longer be entitled to indemnification by the Company hereunder.
6
4.13 Expenses. Each party shall pay the fees and expenses of its advisers, counsel, accountants and other experts, if any, and all other expenses incurred by such party incident to the negotiation, preparation, execution, delivery and performance of this Agreement; provided, however, that the Company shall pay the Investors’ reasonable and documented legal fees and expenses incurred in connection with the transactions contemplated by this Agreement (including any post-Closing expenses) up to an aggregate amount of $75,000. The Company shall pay all transfer agent fees, stamp taxes and other taxes and duties levied in connection with the delivery of the Securities to the Investors.
4.14 Construction. The parties agree that each of them and/or their respective counsel has reviewed and had an opportunity to revise the Agreement and, therefore, the normal rule of construction to the effect that any ambiguities are to be resolved against the drafting party shall not be employed in the interpretation of the Agreement or any amendments hereto.
4.15 Securities Law Disclosure; Publicity. The Company shall by 9:00 a.m., New York City time, on the first business day immediately following the date hereof (provided that, if this Agreement is signed between midnight and 9:00 a.m. (New York City time) on any business day, the Company shall by no later than 9:01 a.m. on the date hereof), issue a press release and/or file with the Commission on a Current Report on Form 8-K or other filing disclosing the material terms of the transactions contemplated hereby and any other material non-public information concerning the Company disclosed to the Investors. From and after the issuance of such press release or filing, the Company represents to the Investors that it shall have publicly disclosed all material, non-public information delivered to any of the Investors by the Company, or any of its officers, directors, employees or agents in connection with the transactions contemplated by this Agreement. In addition, effective upon the issuance of such press release and/or filing, the Company acknowledges and agrees that any and all confidentiality or similar obligations under this Agreement, or an agreement entered into in connection with the transactions contemplated this Agreement, whether written or oral, between the Company or any of its officers, directors, agents, employees or affiliates, on the one hand, and any of the Investors or any of their respective officers, directors, agents, employees or investment advisers, on the other hand, shall terminate. The Company shall not issue any press release or otherwise make any such public statement regarding the transactions contemplated hereby that discloses the name of an Investor or any of its affiliates or advisers without the prior consent of such Investor, except if such disclosure is required by law, in which case the Company shall promptly provide such Investor with prompt prior notice of, and an opportunity to comment on, such public statement or communication.
4.16 Termination. This Agreement may be terminated (i) by written agreement of an Investor as to such Investor’s obligations and the Company, or (ii) by any Investor, as to such Investor’s obligations hereunder only and without any effect whatsoever on the obligations between the Company and other Investor, by written notice to the other parties, if the Closing has not been consummated on or before the fifth (5th) Trading Day following the date hereof; provided, however, that no such termination will affect the right of any party to sue for any breach by any other party (or parties).
[Signature Pages Follow]
7
IN WITNESS WHEREOF, the parties hereto have executed this Securities Purchase Agreement as of the day and year first above written.
| FOGHORN THERAPEUTICS INC. | ||
| By: |
|
|
| Name: |
|
|
| Title: |
|
|
| Address: | ||
[Company Signature Page to Purchase Agreement]
IN WITNESS WHEREOF, the parties hereto have executed this Securities Purchase Agreement as of the day and year first above written.
Name of Investor:
| Signature of Authorized Signatory of Investor: |
|
Name of Authorized Signatory:
Title of Authorized Signatory:
Email Address of Authorized Signatory:
Address for Notice of Investor:
Telephone:
With a copy to (which shall not constitute notice):
Attn:
[Investor Signature Page to Purchase Agreement]
Exhibit A
| Investor |
Number of Shares |
Number of Pre- Funded Warrants |
Number of Series 1 Warrants |
Number of Series 2 Warrants |
Total Purchase Price |
Instructions for Delivery of Shares
| Investor |
Address (if not same as address for notice) |
EIN | DWAC Delivery Instructions |