
Connecting Innovation to Purpose Corporate Presentation December 11, 2025 Exhibit 99.2

This presentation contains certain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934 and Private Securities Litigation Reform Act, as amended, including those relating to the Company’s trial results, product development, clinical and regulatory timelines, market opportunity, competitive position, possible or assumed future results of operations, business strategies, potential growth opportunities, including timing or completion of trials and presentation of data and other statements that are predictive in nature. These forward-looking statements are based on current expectations, estimates, forecasts and projections about the industry and markets in which we operate and management’s current beliefs and assumptions. These statements may be identified by the use of forward-looking expressions, including, but not limited to, “expect,” “anticipate,” “intend,” “plan,” “believe,” “estimate,” “potential,” “predict,” “project,” “should,” “would” and similar expressions and the negatives of those terms. These statements relate to future events or our financial performance and involve known and unknown risks, uncertainties, and other factors, on our operations, clinical development plans and timelines, which may cause actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. Such factors include those set forth in the Company’s filings with the Securities and Exchange Commission. Prospective investors are cautioned not to place undue reliance on such forward-looking statements, which speak only as of the date of this presentation. The Company undertakes no obligation to publicly update any forward-looking statement, whether as a result of new information, future events or otherwise. This presentation includes limited observations derived from separate clinical settings that are not, and should not be interpreted as, direct or indirect head‑to‑head comparisons of CRB‑701, CRB‑913 or CRB-601 with any other product. The observations described herein are subject to change as additional data become available, and future clinical trials of CRB‑701, CRB‑913 or CRB-601 may not reproduce, validate, or otherwise confirm these observations. All product names, logos, brands and company names are trademarks or registered trademarks of their respective owners. Their use does not imply affiliation or endorsement by these companies. Forward-Looking Statements

Clinical data readouts expected for all three drug candidates in 2nd half of 2025 $173M Cash, cash equivalents and investments as of November 3, 2025 and approximately 17.6M common shares issued and outstanding (~20.5M fully diluted shares) SAD/MAD data: Q4 2025 CRB-913 Dose escalation: Q4 2025 CRB-601 ESMO 2025: Clinical update in HNSCC, Cervical and Bladder CRB-701

Therapy Disease Indication Sponsor Pre-Clinical Phase 1 Phase 2 Phase 3 Milestones Next-Generation Nectin-4 targeting ADC CRB-701 Next-generation Nectin-4 targeting ADC Nectin-4 positivesolid tumors CSPC(China) Phase 3 in cervical Corbus(US + Europe) Dose optimization in mUC, HNSCC & cervical Highly peripherally-restricted CB1R inverse agonist CRB-913 CB1 inverse agonist Obesity and related conditions Corbus SAD/MAD dosing completed Anti-Integrin mAb CRB-601Anti-⍺vβ8 mAb(TGFβ-targeting) ⍺vβ8 enriched solid tumors Corbus First patient dosed December 2024 A diversified pipeline with differentiated clinical risk profiles FDA Fast Track Designation granted HNSCC and Cervical

CRB-701 Next Generation Nectin-4 Targeting ADC

CRB-701: Re-imagining a Nectin-4 ADC Extend ADC half-life Reduce dosing frequency Convenience Lower DAR + longer half-life Dose higher + longer than PADCEV® Efficacy Markedly reduce PADCEV®-associated toxicities Safety Focus on non-mUC tumors Avoid competing with PADCEV® Strategy Summary

CRB-701: Proprietary components novel design MMAE = Monomethyl auristatin E ADCC = antibody-dependent cellular cytotoxicity. CDC = complement dependent cytotoxicity Source(s): Modified image from Corbus data on file; Corbus data on file Novel Nectin-4 Antibody ADCC + CDC functionality Glutamine Focused Side chain conjugation Payload: MMAE Microtubule disruption Cathepsin-B Cleavage Site An Improved ADC Construct Precise & stable DAR of 2 —> Longer half life Improved binding affinity & selectivity —> 2x rate of internalization vs. PADCEV® Improved linker stability —> Reduced free MMAE Structure

Best responses seen in tumors with highest Nectin-4 expression-mUC, cervical & HNSCC1 Source(s): 1. Corbus data on file; 2. AACR 2023 Poster CRB-701 demonstrates better efficacy than EV in patient-derived tumor model expressing low levels of Nectin-42 Elevated Nectin-4 expression: urothelial, cervical, head and neck. breast, ovarian, colorectal, rectal, esophageal, gastric, lung, thyroid, prostate, cholangiocarcinoma, pancreatic cancer, testicular cancer Other highly expressing tumors BLCA=Bladder Cancer (urothelial) CESC=Cervical Cancer (squamous) HNSC = Head and neck Cancer (Squamous) Mean Tumor Volume ± SEM CRB-701 PADCEV® 74.5% p<0.05 53.7% p=0.70 Nectin-4

Company 21-day PK Comparison % ADC % Free MMAE Cmax AUC0-21d Cmax AUC0-21d PADCEV® 1.24 mg/kg Q1W x 3 PADCEV® Benchmark 100% 100% 100% 100% 2.7 mg/kg Q3W Matched for MMAE dose (DAR) 183% 274% 35% 38% 3.6 mg/kg Q3W 2.9-fold PADCEV® ADC Dose® 228% 361% 59% 62% Key differentiator: Lower levels of free MMAE for CRB-701 vs. PADCEV® Source(s): PADCEV® reference data from BLA761137 17 December 2019 Corbus data: ESMO 01 Sep 2025 Data cut PK

CRB-701: Best-in-class dosing regimen Source(s): Corbus data on file; PADCEV® Prescribing Information as of Dec 2023 CRB-701 Clinical Cycle Comparison Patient / Physician Convenience Combination Flexibility Dosing

CRB-701: Corbus study design (U.S. + Europe) 1.8 mg/kg 2.7 mg/kg 3.6 mg/kg 4.5 mg/kg Completed Dose expansion (Data maturation for pivotal trial planning) Dose escalation Project Optimus (Dose optimization) Randomized to 2.7 or 3.6 mg/kg monotherapy in: HNSCC Cervical mUC CRB-701 + Keytruda® Ongoing Completed Ongoing HNSCC Cervical Study design

Head & Neck Squamous Cell Carcinoma Cervical Cancer mUC 5-Year Survival Not pursuing as a stand-alone company U.S. Incident Patient Population Funnel 2025 Current Treatment Emerging indications of interest: HNSCC + cervical cancer Source: SEER Bladder Cancer; Census.gov; Weir et al., 2021; American Cancer Society; Chu et al., 2022; Hoffman-Censits et al., 2022. SEER Cervical Cancer; Census.gov; Weir et al., 2021; American Cancer Society; Mizuho Analyst Report; Corbus Corporate Deck. SEER Oral Cavity & Pharynx Cancer; SEER Laryngeal Cancer; American Cancer Society; Sanders et al., 2022. LifeSci Consulting Qualitative Market Research Localized Regional Distant 86% 66% 37% ~68% locally adv / metastatic ~80% 1L-treated ~60% progress to 2L+ ~55% locally adv / metastatic ~95% 1L-treated ~37% progress to 2L+ Localized Regional Distant 88% 70% 39% Prevalence 1L Carbo + Paclitaxel + Beva +/- Keytruda® Keytruda® + Platinum + Paclitaxel 2L Tivdak® Single-Agent Chemo Single-Agent or Combo Chemotherapy Keytruda® + Platinum + 5-FU

ESMO 2025: Key characteristics & tumor types Source(s): ESMO 01 Sep 2025 Data cut Baseline characteristic (as of 9/1/25 data cut) Median age (range) 60 (32-90) Sex (M/F) 50.3% / 49.7% ECOG PS 0, 1, 2 43.1% , 55.1% , 1.8% Weight in kg mean (range) 72 (32.1-132.8) Prior therapies median (range) 3 (1-9) Safety Population n=167 Safety Population dosed with monotherapy CRB-701 n=163 Efficacy evaluable population (participants with at least 1 post-baseline scans) HNSCC Cervical La/mUC Other tumor types n=122 n=41 n=37 n=23 n=21 Enrolled tumor types (n=167) HNSCC 60 Cervical 54 Locally advanced/ mUC 27 NSCLC 7 TNBC 1 Endometrial 3 Prostate 1 Penile 2 Ovarian 4 Pancreatic 7 Missing 1 ECOG = Eastern Cooperative Oncology Group Performance Status; HNSCC = Head and Neck Squamous Cell Carcinoma; La/mUC = locally advance or metastatic urothelial cancer; NSCLC = Non-small cell lung cancer, TNBC=Triple negative breast canceer Baseline

ESMO 2025: TEAEs ≥15% (n=167) Sources: ESMO 01 Sep 2025 Data cut Adverse Events of Interest N=167 (%) Peripheral neuropathy Broad Terms* 8.4% Eye Overall 56.9% Grade 3 9% Grade 4 & 5 0 Skin Pruritus 14.4% Dry skin 10.2% Rash 9.0% Rash maculo-papular 4.8% Dermatitis acneiform 3.6% Erythema 1.8% Dermatitis bullous 1.2% Rash pustular 1.2% Rash erythematous 0.6% Rash macular 0.6% Rash pruritic 0.6% Skin disorder 0.6% Skin reaction 0.6% Skin ulcer 0.6% No related Grade 4 or 5 *Standardized MedDRA Category Search Safety

ESMO 2025: Favorable emerging safety profile vs. Nectin-4-MMAE peers Source(s): 1. NDA/BLA Multidisciplinary Review and Evaluation BLA 761137 PADCEV® (enfortumab vedotin) 2. Torras, O. Reig, et al. "652P BT8009 monotherapy in enfortumab vedotin (EV)-naïve patients with metastatic urothelial carcinoma (mUC): Updated results of Duravelo-1."Annals of Oncology 35 (2024): S515-S516. 3. ASCO 2024, Zhang, et al. 4. SGO plenary March 2024, Yang et al. 5. ESMO 01 Sep 2025 Data cut *Rash (Broad terms): Skin and subcutaneous tissue disorders SOC, excluding alopecia PADCEV® 1 BT80092 9MW-28213,4 CRB-7015 Upper dose limit 1.25 mg/kg 5 mg/m2 1.25 mg/kg 2.7mg/kg 3.6mg/kg Schedule D1, D8, D15 /28 days Q1W D1, D8, D15 /28 days Q3W ≥ Grade 3 AE rate 62.5% (n=237/379) 53% (n=24/45) 70% 35.7% (n=25/70) 35.5% (n=27/76) Peripheral neuropathy (broad terms) 48% (n=182/379) 36% (n=16/45) 22.5% (n=54/240) 8.6% (6/70) 6.6% (5/76) Rash (broad terms*) 50.7% (n=192/379) 18% (n=8/45) 30% (n=72/240) 32.9% (n=23/70) 23.7% (n=18/76) Neutropenia (Gr 3) 10% (31/310) 4% (n=2/45) 27.9% (n=67/240) 0% 0% Dose reduction 27.7% (n=105/379) 27% (n=12/45) Not released 10% (7/70) 19.7% (15/76) Dose interruptions 55.9% (n=212/379) 53% (n=24/45) Not released 38.6% (27/70) 51.3% (39/76) Discontinuations 20.6% (78/379) 4% (n=2/45) Not released 5.7% (4/70) 7.9% (6/76) Safety

Safety Summary *Standardized MedDRA Category Search; **decubitus ulcer, general rash, Bullous dermatitis Safety 28.7% (broad-terms excluding alopecia) Low numbers of Grade ≥3 events (3/167**) Discontinuations due to eye toxicities have been low (4.2%) Best-in-class for peripheral neuropathy 8.4% (all grade 1 or 2)* Low rates of skin adverse events Eye toxicities have been manageable with prophylaxis and dose modifications

HNSCC baseline characteristics vs. peers Source(s): * ESMO 01 Sep 2025 Data cut; **ESMO ASIA data Dec 2024; *** Swiecicki et al, 2024 Baseline characteristic CRB-701* Petosemtamab** HNSCC PADCEV®*** Median age (range) 62 (35-76) 60 (31–77) 65 (33-81) Sex (M/F) 90% / 10% 79% / 21% 87% / 13% ECOG PS 0,1, 2 48.3%, 50%, 1.7% 30%, 70%, 0% 34.8%, 65.2%, 0% Prior lines median (range) 3 (1-9) 2 (1-4) 1 line 15.2% 2 lines 17.4% ≥3 lines 67.4% HPV/P16 Status (Positive/Negative/Missing) 28.3% / 15.0% / 56.7% 46% / 46% / 8% 43.5% / 13% / 43.5% Disease status at Study Entry (Locally Recurrent/Metastatic) 15% / 85% Not disclosed Not disclosed Nectin-4 H-Score (Range) 13-285 N/A 20-300 PD-L1 Criteria Agnostic PD1(L1)-1 Positive Agnostic HNSCC

ESMO 2025: HNSCC waterfall plot (n=41) Source: ESMO 01 Sep 2025 Data cut, ,Note: NE = Non-Evaluable. 2.7 mg/kg 3.6 mg/kg ORR 33.3% (4/12) 47.6% (10/21) DCR 75% 61.9% Response confirmation All confirmed 7 confirmed 3 unconfirmed: 1 discontinued and 2 ongoing ORR% = ( CR+PR) / Response evaluable patients DCR% = (CR+PR+SD) /Response evaluable patients 8 patients on Waterfall plot excluded from ORR & DCR calculations 4 non-evaluable patients 1 patient received combination of CRB-701 and pembrolizumab (+24% PD) 3 patients dosed at 1.8mg/kg * HNSCC

ESMO 2025: HNSCC swimmer plots (n=58) Source(s): ESMO 01 Sep 2025, *based on confirmed overall response PFS and DOR: too early to assess HNSCC HNSCC

CRB-701 biomarker populations: Observed efficacy Nectin-4 Responses seen across wide range of IHC H-score expressions HPV Responses seen in HPV positive and negative patients PD(L)-1 Responses in PD(L)-1 positive and negative patients Source(s): Corbus data on file HNSCC

Case Study #1: Clinical improvement in participant with resistant disease Source(s): Corbus Data on file As of 22 Sep 2025 – Participant is ongoing with a PR and tumor reduction of -73% with negative NavDx ctDNA. Remaining disease is PET negative/cold – being considered as a clinical (not formal) CR. Prior therapies Carboplatin+docetaxel+5FU 3 weeks (PD) then Cisplatin 4 weeks (PD) then pembrolizumab 6 weeks (PD) then experimental bispecific antibody (PD) 61-year-old male patient with HNSCC PD-L1 <1 recently had 1 year tumor assessment images. He was previously suffering with significantly reduced performance status (ECOG 2) and on supplemental oxygen, now riding his bicycle, off oxygen and has gained 15 pounds with an ECOG of 0. – USA Study Physician HNSCC

Source: Corbus data on file Case Study #2: Response seen in patient pre-treated with petosemtamab Patient had a partial response (after previously showing stable disease while on petosemtamab) Patient was heavily pre-treated with 4 lines of prior therapy HNSCC Adjuvant 1st line (completed) Cisplatin Locally advanced (completed) Local advanced (progressed) pembrolizumab Locally advanced (progressed) petosemtamab Metastatic (ongoing treatment) CRB-701 PR (-60.8% ) ongoing Cisplatin paclitaxel pembrolizumab

CRB-701 compared to petosemtamab or PADCEV® in 2L HNSCC Source(s): *ESMO 01 Sep 2025 Data cut; ** Swiecicki et al, 2024; ***ESMO ASIA data Dec 2024. Petosemtamab*** HNSCC PADCEV®** CRB-701* Dosing regimen 1500mg Q2W 1.25mg/kg on d1/8/15 of 28-day 3.6mg/kg Q3W Target population PD(L)-1 +ve only (HPV+/-) PD(L)-1 agnostic (HPV+/-) PD(L)-1 agnostic (HPV+/-) Efficacy (ORR) 36% 23.9% 47.6% TEAEs Grade 3 & greater 59% 34.8% 35.5% HNSCC

Target patient populations for CRB-701 in HNSCC 1L Multiple MOAs being evaluated CRB-701 combo data with pembrolizumab expected mid-2026 24,000* annual cases in USA No ADCs approved Orthogonal mechanism to EGFR Existing late line Tx ORR ~10% Petosemtamab ORR 36% 2L+ Source(s): LifeSci Consulting Qualitative Market Research HNSCC

CRB-701 HNSCC: Next steps planned Convenience kit components Mid-2026 Start registrational studies Monotherapy update Q1 2026 Registrational pathway update following FDA engagement 2nd Half 2026 CRB-701 + pembrolizumab data HNSCC

ESMO 2025: Waterfall plot (n=37) Source: ESMO 01 Sep 2025 Data cut Note: NE =Non-evaluable ORR %=( CR+PR)/ Response evaluable patients DCR % = (CR+PR+SD) /Response evaluable patients 2.7 mg/kg 3.6 mg/kg ORR 22.2% (4/18) 37.5% (6/16) DCR 66.6% 68.8% Response confirmation 2 confirmed 2 unconfirmed and ongoing 3 confirmed 3 unconfirmed: 1 discontinued and 2 ongoing 3 patients excluded from ORR and DCR Calculations 2 non-evaluable patients 1 patient dosed at 1.8mg/kg Cervical Cancer
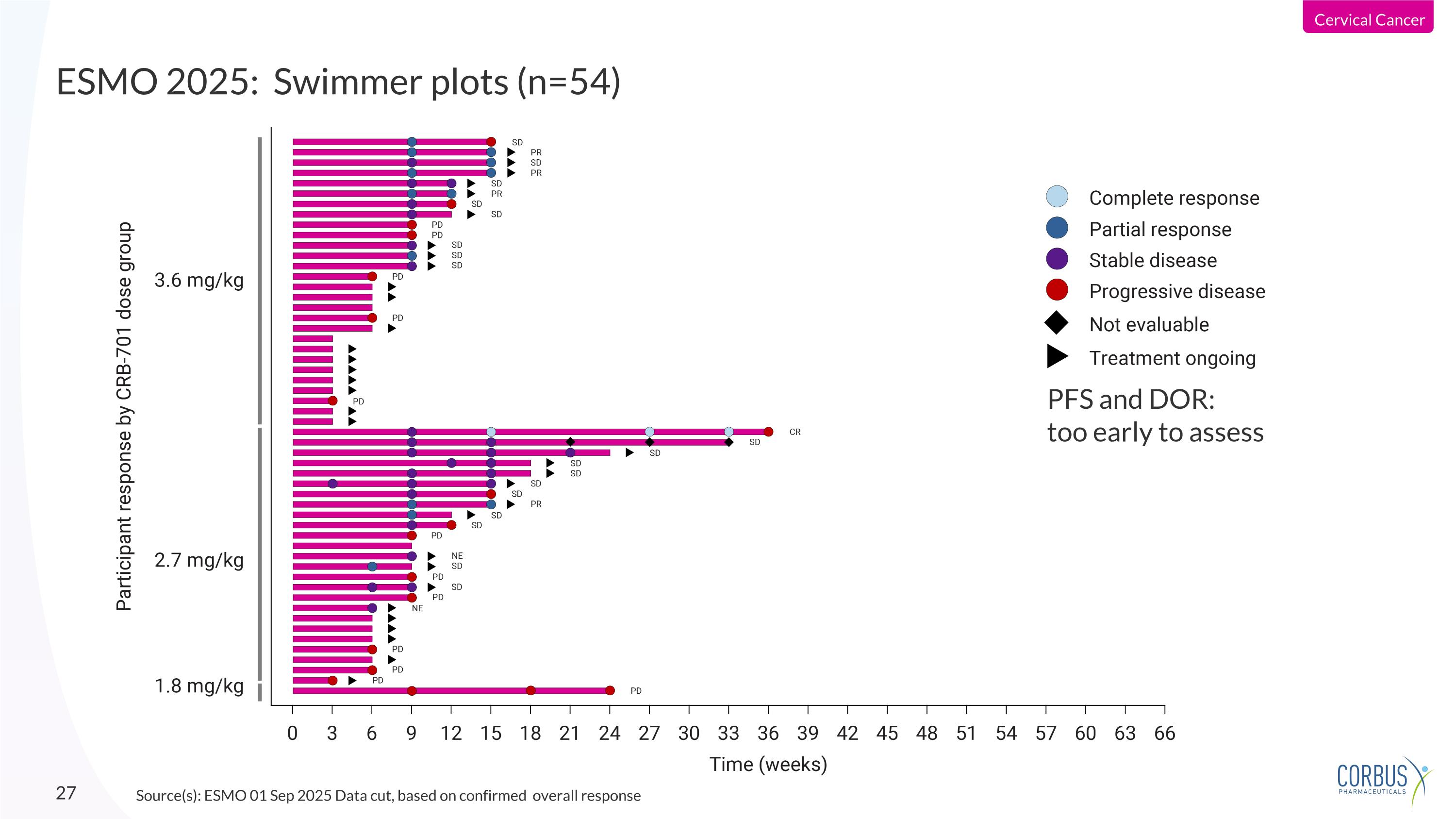
ESMO 2025: Swimmer plots (n=54) Source(s): ESMO 01 Sep 2025 Data cut, based on confirmed overall response PFS and DOR: too early to assess Cervical Cancer

ESMO 2025: CRB-701 compared to Tivdak® Sources: ESMO 01 Sep 2025 Data cut*Tivdak® Package Insert CRB-701 Tivdak® Mechanism Nectin-4 ADC with MMAE payload (DAR 2) Tissue factor ADC with MMAE payload (DAR 4) Target population 2L 2L Median Age 54 (32-78) 51 (26-80) ECOG (0, 1, 2, missing) 51.9%, 48.1%, 0%, 0% 61%, 39%, 0%, 0% Prior lines of therapy median (range) 3 (1, 8) 1 line: 61% 2 lines: 38% Unknown: 1% Dosing regimen 3.6 mg/kg Q3W 2 mg/kg Q3W Efficacy (ORR) 37.5% 17.8%* TEAEs Grade 3 & greater 35.5% (n=76) 46% (n=405) Cervical Cancer

Post-1L therapy represents unmet need with few effective modalities Tivdak® considered “a standard of care” in 2L with current annualized sales of $314 million* Side effect profile + poor efficacy are limitations on Tivdak® commercial success FDA has granted CRB-701 Fast Track Status in cervical cancer Potential use of CRB-701 in cervical cancer Source(s): *Genmab Q2 YTD sales of Tivdak® were $78 million *Pfizer Q2 YTD sales of Tivdak® were $79 million **Keytruda prescription label-Keynote 826 study 1L Keytruda® + chemo Efficacy (ORR ~68%**) Tivdak® Modest efficacy (ORR 17.8%) and poor tolerability 2L+ Cervical Cancer

ESMO 2025: Waterfall plot (n=23) Source: ESMO 01 Sep 2025 Data cut Note: NE = Non-Evaluable ORR% = ( CR+PR) / Response evaluable patients DCR% = (CR+PR+SD)/ Response evaluable patients 2.7 mg/kg 3.6 mg/kg ORR 50.0% (4/8) 55.6% (5/9) DCR 75.0% 88.9% Response confirmation 2 confirmed 2 unconfirmed and ongoing 3 confirmed 2 unconfirmed: 1 discontinued and 1 ongoing 6 patients on Waterfall plot excluded from the ORR and DCR calculation 1 patient with a tumor reduction of -60.7% (PR) excluded due to missing data 2 non-evaluable patients 3 patients dosed at 1.8mg/kg Bladder Cancer

ESMO 2025 Swimmer plots (n=27) Source(s): ESMO 01 Sep 2025 Data cut, based on confirmed overall response PFS and DOR: too early to assess Bladder Cancer

ESMO 2025: CRB-701 compared to PADCEV® monotherapy Sources: *ESMO 01 Sep 2025 Data cut **PADCEV® data ***All grade 3, no Grade 4/5: 1 x rash, 1 x decubitus ulcer, 1 x dermatitis bullous CRB-701* PADCEV®** Mechanism Nectin-4 ADC with MMAE payload (DAR 2) Nectin-4 ADC with MMAE payload (DAR ~3.8) Dosing regimen 3.6mg/kg Q3W 1.25mg/kg on d1/8/15 of 28-day Target population 2nd line 2nd line Efficacy-ORR 55.6% 44% Pooled safety database n=76 n=310 (1.25mg/kg dose) Grade 3 or greater AE rate 35.5% 58% Peripheral neuropathy 6.6% 49% Rash & skin reactions (broad terms) 29.3% (2.4% Grade 3***) 54% (7% Grade 3) Discontinuation rates 7.9% 19.4% Bladder Cancer
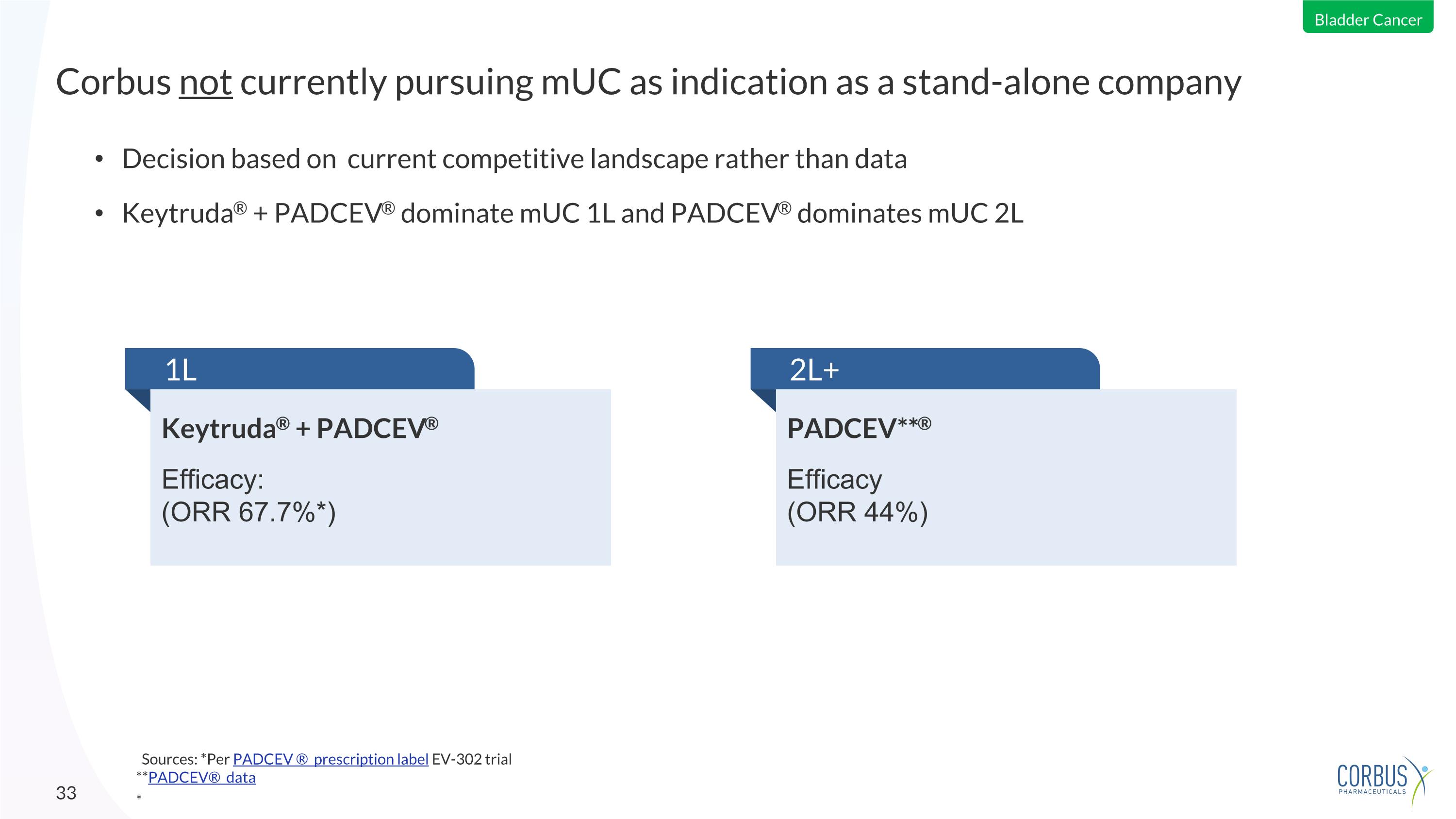
Decision based on current competitive landscape rather than data Keytruda® + PADCEV® dominate mUC 1L and PADCEV® dominates mUC 2L Corbus not currently pursuing mUC as indication as a stand-alone company * 1L Keytruda® + PADCEV® Efficacy: (ORR 67.7%*) PADCEV**® Efficacy (ORR 44%) 2L+ Bladder Cancer Sources: *Per PADCEV ® prescription label EV-302 trial **PADCEV® data

CRB-913 Daily oral small molecule targeting chronic obesity management Data from Phase 1a SAD/MAD study

CB1 is a well-understood receptor in metabolism >9K papers in PubMed on CB1 and metabolism Source(s): Targeting the endocannabinoid system in diabesity: Fact or fiction?, Drug Discovery Today, Deeba et al. Mar 2021.

Monlunabant CRB-913 Next-generation CB1 inverse agonists are peripherally restricted Rimonabant Otenabant Ibipinabant Taranabant Source(s): Cinar et al 2020 First-generation (2000-2007) Next-generation (2020 onwards) Designed to target the brain with high BBB penetration FDA rejection due to safety concerns (2007) Designed to be peripherally restricted with minimal BBB penetration avoid safety issues

1/50th Brain:plasma ratio CRB-913vs rimonabant 1/15th Brain level CRB-913 vs monlunabant Murine data demonstrates CRB-913 is best-in-class peripheral restriction Reference: Morningstar M, et al. Obesity 2023;31: 2676-2688.

CRB-913 SAD/MAD study (Phase 1 unit in USA, total n=112) 9 mg 25 mg 75 mg 150 mg 300 mg 600 mg 150 mg participants with obesity 7-Day Multiple Ascending Dose (MAD) (n=48) Single Ascending Dose (SAD) (n=64) # Per Cohort Placebo: 2 CRB-913: 6 # Per Cohort Placebo: 3 CRB-913: 9 25 mg 75 mg 150 mg 150 mg participants with obesity Context: Rimonabant efficacious dose: 20 mg QD Monlunabant efficacious dose: 10 mg QD 150 mg fed

Monlunabant (Phase 2a, n= 240) CRB-913 (Phase 1a, n=112) >90% of GI AEs in first 14 days GI AE rates: 51-79% depending on dose 2nd most common cause of dropout in study 1 case of possibly related diarrhea (mild) CRB-913 demonstrated a differentiated and favorable GI tolerability profile Monlunabant 50 mg Monlunabant 20mg Monlunabant 10 mg placebo Reference: Knop, F.K. , The Lancet Diabetes & Endocrinology, 13, 11, 911 - 923 GI AE CRB-913 Monlunabant Nausea None 36%-50% Diarrhea N=1 25%-37% Vomiting None 16%-20%

Monlunabant (Phase 2a, n= 240) CRB-913 (Phase 1a cohorts, n=112) >60% of psych AEs in first 14 days >95% of psych AEs in first 60 days 180 patients on treatment experienced 111 events Cases of depression noted in phase 1 and phase 2 Most common cause of dropout in study *Transient and fully resolved without any intervention CRB-913: daily neuropsych assessments negative at all timepoints Monlunabant 50 mg Monlunabant 20mg Monlunabant 10 mg placebo Reference: Knop, F.K. , The Lancet Diabetes & Endocrinology, 13, 11, 911 - 923 All SAD cohorts 25 mg/day x7 days 75 mg/day x7 days 150 mg/day x7 days Subjects with obesity 150 mg/day x7 days CSSRS Negative Negative Negative Negative Negative PHQ-9 Negative Negative Negative Negative Negative GAD-7 Negative Negative Negative Negative Negative Suicidality, depression, dysphoria Negative Negative Negative Negative Negative Insomnia Negative Negative Negative Negative Negative Mild anxiety* (negative GAD-7, PHQ9 and CSSRS) None None None None N=3 Mild irritability* (negative GAD-7, PHQ-9 and CSSRS) None None None None N=1 Weight loss? No No Yes Yes Yes

150 mg/day OD for 7 days of dosing + additional 7 days observation = 14 days in-clinic Emerging weight loss with CRB-913 in subjects with obesity (150 mg dedicated MAD cohort) 2.9% average placebo-adjusted weight loss @ day 14 Participants reported reductions in food-related thoughts and cravings Note: Baseline is defined as the last available measurement taken prior to the first dose of study drug. Percent change in body weight is defined as body weight at Day 14 minus body weight at baseline divided by body weight at baseline multiplied by 100.
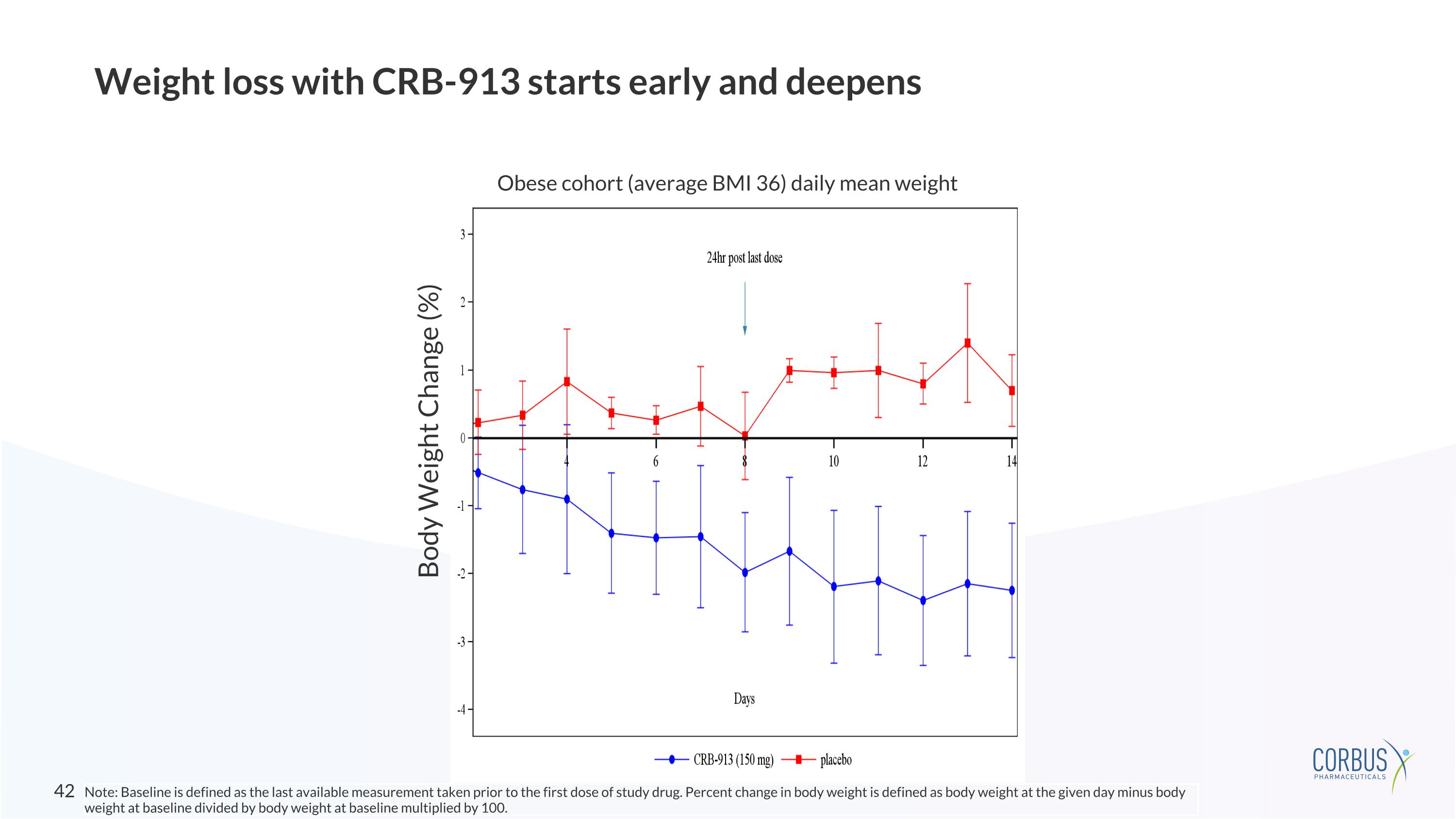
Weight loss with CRB-913 starts early and deepens Obese cohort (average BMI 36) daily mean weight Body Weight Change (%) Note: Baseline is defined as the last available measurement taken prior to the first dose of study drug. Percent change in body weight is defined as body weight at the given day minus body weight at baseline divided by body weight at baseline multiplied by 100.

Signals of weight loss in non-obese MAD cohorts at lower doses Average BMI of 28 lower potential for weight loss Placebo-adjusted weight loss 0% 2.0% 1.5% BMI range 23.5 to 31.4 22.3 to 31.8 24.4 to 31.3 Note: Baseline is defined as the last available measurement taken prior to the first dose of study drug. Percent change in body weight is defined as body weight at Day 14 minus body weight at baseline divided by body weight at baseline multiplied by 100.

Emerging efficacy of CRB-913 vs Monlunabant vs Rimonabant N =346* N =60** N = 20*** Placebo-adjusted weight loss cross-trial comparison Change in weights (%) N=9 Source: All comparator data points are approximated and based on extracted figures reported from * RIO North America (Pi Snuyer et al 2006), ** Crater et al 2023, *** Knop et al 2025 and **** Wharton et al 2025 weeks N =60** N =60** Context: Orfoglipron 45 mg QD weight loss @ day 28 = 3% **** Rimonabant (20 mg) * Monlunabant (10 mg)** Monlunabant (20 mg)** Monlunabant (50 mg)** Monlunabant (25 mg)*** CRB-913 (150 mg)
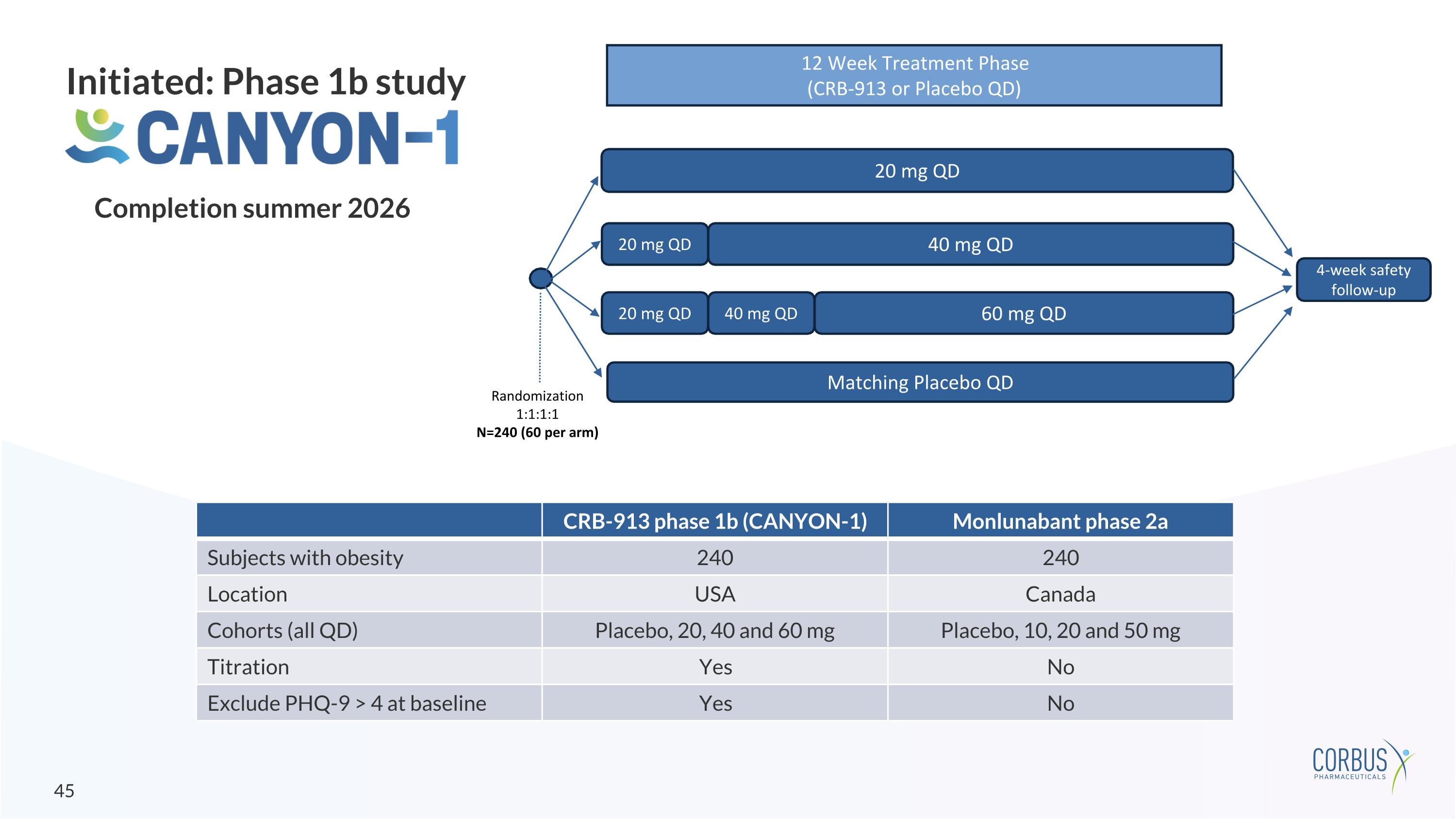
Initiated: Phase 1b study 20 mg QD 20 mg QD Matching Placebo QD 12 Week Treatment Phase (CRB-913 or Placebo QD) Randomization 1:1:1:1 N=240 (60 per arm) 4-week safety follow-up 40 mg QD 20 mg QD 40 mg QD 60 mg QD CRB-913 phase 1b (CANYON-1) Monlunabant phase 2a Subjects with obesity 240 240 Location USA Canada Cohorts (all QD) Placebo, 20, 40 and 60 mg Placebo, 10, 20 and 50 mg Titration Yes No Exclude PHQ-9 > 4 at baseline Yes No Completion summer 2026

High peripheral restriction associated with favorable safety and tolerability CRB-913 elicits weight loss that starts early and deepens Weight loss is not driven by GI AEs Weight loss is associated with restriction to the periphery What did we learn from the CRB-913 SAD/MAD data?

CRB-601 Potential “best-in-class” ⍺vβ8 mAb

CRB-601 has the potential to enhance checkpoint iinhibition Convenience kit components Focus on adopting a precision-targeted approach Novel mechanism to target TGFb in the tumor microenvironment Large opportunity potential if POC is validated

Targeting the integrin ⍺vβ8 represents a novel approach to regulating TGFβ Source(s): Huang et al., 2021. Recent progress in TGFβ inhibitors for cancer therapy.

CRB-601 PF-06940434 SRK-181 ABBV-151 RG6440 MOA ⍺vβ8 ⍺vβ8 L-TGFβ GARP (TGFβ1) L-TGFβ Clinical Stage Phase 1 Phase 1/2 –study completed December 2024 Phase 1 Phase 2 HCC (read-out in 2025) Expanded Ph2 trials into muC & NSCLC Phase 1 Indications Solid Tumors Solid Tumors Solid Tumors HCC Solid Tumors Type Monoclonal Antibody Monoclonal Antibody Monoclonal Antibody Monoclonal Antibody Monoclonal Antibody ROA IV IV IV IV IV mAbs targeting TGFβ activation in the clinic Source(s): Company websites. Clinicaltrials.gov. Internal analysis.

LeadershipUpcoming CatalystsFinancials

Management team Yuval Cohen, PhD Chief Executive Officer, Director Sean Moran, CPA, MBA Chief Financial Officer Dominic SmethurstChief Medical Officer, MA MRCP Ian Hodgson, PhD Chief Operating Officer Christina Bertsch, M.A. Head of Human Resources Corbus co-founder and Chief Executive Officer since 2014. Previously the President and co-founder of Celsus Therapeutics from 2005. Corbus co-founder and Chief Financial Officer since 2014. Prior senior financial management experience in emerging biotech and medical device companies. Dr. Smethurst, MA MRCP, joined Corbus as our Chief Medical Officer in February 2024. He most recently served as CMO of Bicycle Therapeutics. Dr. Hodgson joined Corbus in 2022. Previously he held senior leadership positions in biotech and contract research organizations. Most recently served as V.P., Head of Clinical Services at TMC Pharma. Accomplished senior human resource executive providing strategic HR consulting services to both large and small businesses across a variety of industries.

Board of Directors Winston Kung, MBA Director More than 20 years of senior financial, business development and investment banking experience; currently CFO of ArriVent. (NASDAQ: AVBP) Yuval Cohen, PhD Chief Executive Officer, Director Corbus co-founder and Chief Executive Officer since 2014. Previously the President and co-founder of Celsus Therapeutics from 2005. Anne Altmeyer, PhD, MBA, MPH Director Greater than 25 years of experience advancing oncology R&D programs and leading impactful corporate development transactions; former CEO of TigaTx (acquired by Epsilogen Ltd) Yong (Ben) Ben, MD, MBA Director 25 years of oncology R&D experience across industry and academia. CMO of BridgeBio Oncology Therapeutics and former CMO of BeiGene. John K. Jenkins, MD Director Distinguished 25-year career serving at the U.S. FDA, including 15 years of senior leadership in CDER and OND. Rachelle Jacques Chair of the Board More than 30-year professional career, experience in U.S. and global biopharmaceutical commercial leadership, including multiple high-profile product launches in rare diseases; Former CEO of Enzyvant Therapeutics (now Sumitomo Pharma) and Akari Therapeutics (NASDAQ: AKTX) Amb. Alan Holmer Ret. Director More than two decades of public service in Washington, D.C. including Special Envoy to China; Former CEO of PhRMA.

CRB-701 CRB-913 CRB-601 Upcoming anticipated corporate milestones Complete Ph1 SAD/MAD Q4 2025 Start Ph1B study Q4 2025 Complete Phase 1B Summer 2026 Ph1 dose escalation Q4 2025 Regulatory update Q1 2026 Start HNSCC monotherapy Ph2/3 registrational study Mid 2026 Phase 1/2 monotherapy data Mid 2026 CRB-701+ pembrolizumab data 2nd half 2026






















































