UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): November 11, 2025
enGene Holdings Inc.
(Exact name of Registrant as Specified in Its Charter)
| British Columbia | 001-41854 | Not applicable | ||
| (State or Other Jurisdiction of Incorporation) |
(Commission File Number) |
(IRS Employer Identification No.) |
||
| 4868 Rue Levy, Suite 220 | ||||
| Saint-Laurent, Quebec, Canada | H4R 2P1 | |||
| (Address of Principal Executive Offices) | (Zip Code) | |||
Registrant’s Telephone Number, Including Area Code: 514 332-4888
Not Applicable
(Former Name or Former Address, if Changed Since Last Report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| ☐ | Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ☐ | Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ☐ | Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ☐ | Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
Title of each class |
Trading |
Name of each exchange |
||
| Common Shares | ENGN | The Nasdaq Stock Market LLC | ||
| Warrants, each exercisable for one Common Share, at an exercise price of $11.50 per Share | ENGNW | The Nasdaq Stock Market LLC |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company ☒
| Item 7.01 | Regulation FD Disclosure. |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☒ On November 11, 2025, enGene Holdings, Inc. (the “Company”) issued a press release announcing updates regarding the pivotal cohort of its ongoing Phase 2 LEGEND trial of detalimogene voraplasmid (also known as “detalimogene” and previously EG-70) in high-risk, Bacillus Calmette-Guérin (“BCG”)-unresponsive patients with carcinoma in situ (“CIS”) with or without concomitant papillary disease, including new preliminary clinical data. In addition, as announced in a press release issued November 10, 2025, the Company hosted a conference call on November 11, 2025 to discuss the preliminary clinical data and made available a slide presentation for the conference call, as well as an updated Corporate Presentation to be used during future investor meetings. A copy of the press releases issued November 10th and 11th, the slide presentation for the conference call and the Corporate Presentation are being furnished as Exhibits 99.1, 99.2, 99.3 and 99.4, respectively, to this Current Report on Form 8-K.
The information in this Item 7.01, including Exhibits 99.1, 99.2, 99.3 and 99.4, shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Securities Act of 1933, as amended, or the Exchange Act, except as expressly set forth by specific reference in such filing.
| Item 8.01 | Other Information. |
Detalimogene is being evaluated in the ongoing, open-label, multi-cohort, Phase 2 LEGEND trial to establish its safety and efficacy in high-risk NMIBC. LEGEND’s pivotal cohort (“Cohort 1”) has completed enrollment of 125 patients with high-risk, BCG-unresponsive NMIBC with CIS with or without papillary disease. In addition to Cohort 1, LEGEND includes three additional cohorts, which include NMIBC patients with CIS who are naïve to treatment with BCG (“Cohort 2a”); NMIBC patients with CIS who have been exposed to BCG but have not received adequate BCG treatment (“Cohort 2b”); and BCG-unresponsive high-risk NMIBC patients with papillary-only disease (“Cohort 3”).
Following recent discussions with the U.S. Food and Drug Administration (the “FDA”) the primary endpoint for Cohort 1 will change to complete response (“CR”) at any time from its previous primary endpoint of landmark 12-month CR rate, with its key secondary endpoint becoming duration of response rate. This updated primary endpoint is consistent with other recent programs for BCG-unresponsive NMIBC registered with FDA.
Preliminary Efficacy Data
On November 11, 2025, the Company announced updated preliminary efficacy data from two patient subpopulations of the intent-to-treat (“ITT”) patient population of Cohort 1 consisting of (i) patients who received at least one dose of detalimogene and had at least one post-baseline disease assessment and were enrolled prior to the amendment of the protocol in the fourth quarter of 2024 (the “Pre-Protocol Amendment Patients”) and (ii) patients who received at least one dose of detalimogene and had at least one post-baseline disease assessment and were enrolled following the amendment of the protocol in the fourth quarter of 2024 (the “Post-Protocol Amendment Patients”). As of October 24, 2025, the ITT population consisted of 31 Pre-Protocol Amendment Patients and 62 Post-Protocol Amendment Patients. The table below summarizes preliminary efficacy results from the two separate patient subpopulations:
ITT Population* |
Any Time (N=62) |
3 Month (N=62) |
6 Month** |
|||||
| Post-Protocol Amendment Patients | CR Rate | 63% (CI: 51-74) | 56% (CI: 44-68) | 62% (CI: 46-76) | ||||
| Any Time (N=31) | 3 Month (N=31) |
6 Month (N=27) |
||||||
| Pre-Protocol Amendment Patients | CR Rate | 55% (CI: 38-71) | 55% (CI: 38-71) | 41% (CI: 25-59) |
Data as of October 24, 2025.
| * | ITT: Intent-To-Treat population includes all Pre-Protocol Amendment Patients and Post-Protocol Amendment Patients, respectively, who received at least 1 dose of treatment and had at least 1 post-baseline disease assessment. |
| ** | CR rates at 6 months include only patients who were evaluable at the 6-month timepoint or had disease progression prior to the 6-month assessment. |
CI: 95% Confidence Interval
The preliminary efficacy data for Pre-Protocol Amendment Patients demonstrated a markedly lower 12-month CR rate than those of FDA-approved products for BCG-unresponsive NMIBC patients.
Among the Post-Protocol Amendment Patients, four patients that did not have a CR at the 3-month assessment converted to a CR at the 6-month assessment. In addition, of the 23 patients in this subpopulation with a CR at the 6-month assessment, five patients remained in CR at the 9-month assessment, 17 patients are pending their 9-month assessment and one patient dropped out of the study prior to the 9-month assessment.
Preliminary Safety Data
Detalimogene’s overall tolerability profile was favorable. Of the 125 patients assessed for safety in Cohort 1, as of October 24, 2025, 53 patients (42%) experienced at least one treatment-related adverse event (“TRAE”), which were mainly Grade 1/2 in severity, except for three patients (2.4%) that experienced Grade 3 TRAEs. The most common TRAEs were Bladder Spasm (10.4%), Dysuria (12.0%), Fatigue (16.8%), Micturition urgency (10.4%), and Pollakiuria (10.4%). There were no Grade 4 or Grade 5 TRAEs reported. Among the three patients with Grade 3 TRAEs, there were no drug discontinuations related to the severe adverse events. The rate of TRAEs leading to dose interruptions was 1.6% and to dose discontinuations was 0.8%.
Additional LEGEND Trial Updates
On November 11, 2025, the Company also announced updates regarding enrollment in the other cohorts of the LEGEND trial:
| • | Cohort 2a, evaluating detalimogene in NMIBC patients with CIS who are naïve to treatment with BCG, has enrolled 30 patients; |
| • | Cohort 2b, evaluating detalimogene in high-risk NMIBC patients with CIS who have been exposed to BCG but have not received adequate BCG treatment, has enrolled 45 patients; and |
| • | Cohort 3, evaluating detalimogene in BCG-unresponsive high-risk NMIBC patients with papillary-only disease, has enrolled 36 patients. |
Cash and Liquidity Update
As of October 31, 2025, the Company had cash, cash equivalents and short-term investments of $202.4 million.
| Item 9.01 | Financial Statements and Exhibits. |
(d) Exhibits
Exhibit |
Description |
|
| 99.1 | Press Release of the Company, dated November 10, 2025 | |
| 99.2 | Press Release of the Company, dated November 11, 2025 | |
| 99.3 | Slide Presentation, dated November 11, 2025 | |
| 99.4 | Corporate Presentation, dated November 11, 2025 | |
| 104 | Cover Page Interactive Data File (Formatted as Inline XBRL) | |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| ENGENE HOLDINGS INC. | ||||||
| Date: November 12, 2025 | By: | /s/ Ronald H.W. Cooper |
||||
| Name: | Ronald H.W. Cooper | |||||
| Title: | Chief Executive Officer and President | |||||
Exhibit 99.1

enGene to Host Conference Call to Provide Update on Pivotal Cohort of LEGEND Trial
BOSTON & MONTREAL, November 10, 2025 – enGene Holdings Inc. (Nasdaq: ENGN or “enGene” or the “Company”), a clinical-stage, non-viral genetic medicines company, today announced that it will host a conference call and webcast tomorrow, November 11, 2025, at 8:00 a.m. ET to discuss new preliminary data from its pivotal cohort in the ongoing LEGEND trial of its novel, non-viral gene therapy candidate, detalimogene voraplasmid (also known as detalimogene, and previously EG-70) for patients with high-risk, Bacillus Calmette-Guérin (BCG)-unresponsive non-muscle invasive bladder cancer with carcinoma in situ.
The live call can be accessed by registering as a participant here. Upon registration, participants will receive conference dial-in information. A link to the live webcast of the call is available here and is also accessible on the Events and Presentations page of the Company’s Investor website: https://engene.com/presentations/. A slide deck to accompany the call will be posted to the Events and Presentations page approximately 30 minutes prior to the start of the conference call. A replay of the webcast will be available on the Company’s website for one year.
About enGene
enGene is a clinical-stage biotechnology company mainstreaming genetic medicines through the delivery of therapeutics to mucosal tissues and other organs, with the goal of creating new ways to address diseases with high clinical needs. enGene’s lead program is detalimogene voraplasmid (also known as detalimogene, and previously EG-70) for patients with Non-Muscle Invasive Bladder Cancer (NMIBC), a disease with a high clinical burden. Detalimogene is being evaluated in the ongoing multi-cohort LEGEND Phase 2 trial, which includes a pivotal cohort studying detalimogene in high-risk, Bacillus Calmette-Guérin (BCG)-unresponsive patients with carcinoma in situ (CIS) with or without concomitant papillary disease. Detalimogene was developed using enGene’s proprietary Dually Derivatized Oligochitosan (DDX) platform, which enables penetration of mucosal tissues and delivery of a wide range of sizes and types of cargo, including DNA and various forms of RNA.
To learn more, please visit enGene.com and follow us on LinkedIn, X and BlueSky.
Contact
For media contact:
media@engene.com
For investor contact:
investors@engene.com
Exhibit 99.2

Detalimogene Demonstrates Improved Complete Response Rate of 62% at 6 Months
Low rates of treatment-related adverse events (42%) and dose interruptions (1.6%)
Emerging detalimogene profile supports potential first line use in patients with high-risk,
BCG-unresponsive non-muscle invasive bladder cancer with CIS
LEGEND trial pivotal cohort completed enrollment with 125 patients, exceeding target
enGene to host conference call and webcast at 8:00 a.m. ET
BOSTON & MONTREAL, November 11, 2025 – enGene Holdings Inc. (Nasdaq: ENGN or “enGene” or the “Company”), a clinical-stage, non-viral genetic medicines company, today reported additional preliminary data from the pivotal cohort of its ongoing, Phase 2 LEGEND trial of detalimogene voraplasmid (also known as detalimogene and previously EG-70) in high-risk, Bacillus Calmette-Guérin (BCG)-unresponsive non-muscle invasive bladder cancer (NMIBC) patients with carcinoma in situ (CIS) with or without concomitant papillary disease. Detalimogene’s preliminary data to date and differentiated profile continue to support the Company’s planned Biologics License Application (BLA) submission in the second half of 2026.
The preliminary analysis included 62 patients at 3 months and 37 patients at 6 months. All patients included in this analysis were evaluated under LEGEND’s amended protocol, which went into effect in the fourth quarter of 2024, and was designed to more closely align the LEGEND trial with the American Urological Association’s Guidelines and standard of care.
Data from 62 patients enrolled under the amended protocol with at least one post-baseline disease assessment demonstrated:
| • | 63% complete response (CR) rate at any time (n=62); |
| • | 56% CR rate at 3 months (n=62); |
| • | 62% CR rate at 6 months (n=37), with 4 patients having successfully converted to CR post reinduction; and |
| • | All 5 patients who completed the 9-month assessment had a CR. |
The Company completed enrollment of 125 patients in the pivotal cohort, exceeding its target by 25%. Data from these patients demonstrated a favorable tolerability profile:
| • | 42% of patients experienced a treatment-related adverse event (TRAE); |
| • | 1.6% of patients experienced dose interruptions due to TRAEs; and |
| • | 0.8% of patients experienced dose discontinuations due to TRAEs. |
“We are pleased to report an improved 6-month CR rate for patients being treated with detalimogene under our amended protocol,” said Hussein Sweiti, M.D., MSc, Chief Medical Officer. “With a competitive preliminary efficacy profile and potential for best-in-class tolerability and ease of use, we believe detalimogene could emerge as the first-line therapy for patients with high-risk, BCG-unresponsive NMIBC.”
“Careful selection of an appropriate bladder-sparing therapy is of utmost importance in creating a long-term strategy to maintain a patient’s disease control and quality of life, while minimizing the logistical burden on patient and practice,” remarked Suzanne Merrill, M.D., Senior Physician, Urologic Oncologist and Bladder Cancer Regional Lead, at Colorado Urology. “I am pleased to see the positive trajectory of detalimogene’s efficacy and tolerability data. Combined with its ease of use, detalimogene would be an attractive option to both patient and a busy urology practice.”
In September 2024, enGene reported preliminary data from 21 patients in the pivotal cohort of LEGEND who were enrolled prior to implementing a protocol amendment in the fourth quarter of 2024. An additional 10 patients were enrolled under the prior protocol for a total of 31 patients. The table below provides summary efficacy from the two separate patient subpopulations.
| ITT Population* |
Any Time (N=62) |
3 Month (N=62) |
6 Month** (N=37) |
|||||
| Post-Protocol Amendment |
CR Rate | 63% (CI: 51-74) |
56% (CI: 44-68) |
62% (CI: 46-76) |
| Any Time (N=31) |
3 Month (N=31) |
6 Month (N=27) |
||||||
| Pre-Protocol Amendment |
CR Rate | 55% (CI: 38-71) |
55% (CI: 38-71) |
41% (CI: 25-59) |
Data as of October 24, 2025.
| * | ITT: Intent-To-Treat population includes all post/pre-protocol amendment patients, respectively, who received at least 1 dose of treatment and had at least 1 post-baseline disease assessment. |
| ** | CR rates at 6 months include only patients who were evaluable at the 6-month timepoint or had disease progression prior to the 6-month assessment. |
CI: 95% Confidence Interval.
Preliminary efficacy data for patients enrolled prior to implementing the protocol amendment demonstrated a markedly lower 12-month CR rate than those of U.S. Food and Drug Administration (FDA)-approved products for BCG-unresponsive NMIBC. The Company is encouraged by the strong improvement demonstrated in the preliminary 6-month CR rate of patients enrolled under the amended protocol.
Additional LEGEND trial updates
| • | Following recent discussions with the FDA, the primary endpoint for LEGEND’s pivotal cohort will change to CR rate at any time from a primary endpoint of landmark 12-month CR rate, with its key secondary endpoint becoming duration of response (DOR) for patients in CR. This updated primary endpoint is consistent with other recent programs registered with FDA. |
| • | The Company plans to engage with FDA on a statistical analysis plan (SAP) to determine which patients will be included in the final efficacy evaluable population. |
| • | Cohort 2a, evaluating detalimogene in NMIBC patients with CIS who are naïve to treatment with BCG, has enrolled 30 patients. |
| • | Cohort 2b, evaluating detalimogene in high-risk NMIBC patients with CIS who have been exposed to BCG but have not received adequate BCG treatment, has enrolled 45 patients. |
| • | Cohort 3, evaluating detalimogene in BCG-unresponsive high-risk NMIBC patients with papillary-only disease, has enrolled 36 patients. |
“We are highly encouraged with the preliminary data from our LEGEND study, which support our planned BLA filing,” said Ron Cooper, President and CEO. “Based on the emerging clinical profile and detalimogene’s differentiated ease of use, we continue to believe there is a substantial commercial opportunity for detalimogene if approved.”
Anticipated upcoming milestones
| • | Following agreement with the FDA on the SAP and the accumulation of sufficient 12-month CR data points, enGene expects to provide a data update on the LEGEND trial’s pivotal cohort in the second half of 2026. |
| • | The Company continues to expect to file the BLA in the second half of 2026, with a potential FDA approval in 2027. |
Safety Information
Detalimogene’s overall tolerability profile was favorable. Of the 125 patients assessed for safety in Cohort 1 as of October 24, 2025, 53 patients (42%) experienced at least one TRAE, which were mainly Grade 1/2 in severity, except for 3 patients with Grade 3 TRAEs. The most common TRAEs were Bladder Spasm (10.4%), Dysuria (12.0%), Fatigue (16.8%), Micturition Urgency (10.4%), and Pollakiuria (10.4%). There were no Grade 4 or Grade 5 TRAEs reported. Among the 3 patients with Grade 3 TRAEs, there were no drug discontinuations related to the severe adverse events.
Conference Call
enGene will host a conference call and live webcast at 8:00 a.m. ET today, November 11, 2025. A link to the live webcast of the call is available here and is also accessible on the Events and Presentations page of the Company’s Investor website along with an accompanying slide deck: https://engene.com/presentations/. The live call can be accessed by registering as a participant here. Upon registration, participants will receive conference dial-in information. A replay of the webcast will be available on the Company’s website for one year.
About Non-Muscle Invasive Bladder Cancer (NMIBC)
Non-muscle invasive bladder cancer (NMIBC) is a disease that poses a significant burden on both patients and clinics and has a massive economic impact on the healthcare system. NMIBC occurs when cancer cells grow in the tissues that line the interior of the bladder, but the cancer has not yet penetrated the muscle of the bladder wall. NMIBC can present as papillary outgrowths from the bladder wall, which are typically resected, or as carcinoma in situ (CIS), which consists of flat, multifocal lesions that cannot be resected. The two forms can also co-occur. About 75%-80% of new bladder cancer diagnoses are NMIBC. Patients suffering from high-risk NMIBC who are unresponsive to the standard of care, Bacillus Calmette-Guérin (BCG), face high rates of disease recurrence (50%-70%) and are potentially subject to full removal of the bladder (cystectomy) as a curative but life-altering next step.
About Detalimogene Voraplasmid
Detalimogene is a novel, investigational, non-viral gene therapy for patients with high-risk, non-muscle invasive bladder cancer (NMIBC), including Bacillus Calmette-Guérin (BCG)-unresponsive disease. It is designed to be instilled in the bladder and elicit a powerful yet localized anti-tumor immune response.
Detalimogene was developed using the Company’s Dually Derivatized Oligochitosan® (DDX) platform, a technology designed to transform how gene therapies are accessed by patients and utilized by clinicians. Medicines developed with the DDX platform can potentially overcome the limitations of viral-based gene therapies, reduce complexities related to safe handling and cold storage, and streamline both manufacturing processes and administration paradigms.
Detalimogene has received Regenerative Medicine Advanced Therapy (RMAT) and Fast Track designations from the U.S. Food and Drug Administration (FDA) based on its potential to address the high unmet medical need for patients with BCG-unresponsive, carcinoma in situ (CIS) NMIBC with or without resected papillary tumors who are unable to undergo cystectomy. The RMAT program is intended to expedite the development and review of regenerative medicine therapies for serious or life-threatening conditions, where preliminary clinical evidence suggests the potential to address unmet medical needs. Similarly, Fast Track designation is a process designed to facilitate the development and expedite the review of drugs to treat serious conditions and fill an unmet medical need.
About the LEGEND Trial
Detalimogene is being evaluated in the ongoing, open-label, multi-cohort, Phase 2 LEGEND trial to establish its safety and efficacy in high-risk NMIBC. LEGEND’s pivotal cohort (Cohort 1) consists of 125 patients with high-risk, BCG-unresponsive NMIBC with CIS (with or without papillary disease) and is designed to serve as the basis of the Company’s planned Biologics License Application (BLA) filing. In addition to this pivotal cohort, LEGEND includes three additional cohorts, including NMIBC patients with CIS who are naïve to treatment with BCG (Cohort 2a); NMIBC patients with CIS who have been exposed to BCG but have not received adequate BCG treatment (Cohort 2b); and BCG-unresponsive high-risk NMIBC patients with papillary-only disease (Cohort 3). The LEGEND trial is actively enrolling patients with sites participating in the USA, Canada, Europe, and the Asia-Pacific region.
About enGene
enGene is a clinical-stage biotechnology company mainstreaming genetic medicines through the delivery of therapeutics to mucosal tissues and other organs, with the goal of creating new ways to address diseases with high clinical needs. enGene’s lead program is detalimogene voraplasmid (also known as detalimogene, and previously EG-70) for patients with Non-Muscle Invasive Bladder Cancer (NMIBC), a disease with a high clinical burden. Detalimogene is being evaluated in the ongoing multi-cohort LEGEND Phase 2 trial, which includes a pivotal cohort studying detalimogene in high-risk, Bacillus Calmette-Guérin (BCG)-unresponsive patients with carcinoma in situ (CIS) with or without concomitant papillary disease. Detalimogene was developed using enGene’s proprietary Dually Derivatized Oligochitosan (DDX) platform, which enables penetration of mucosal tissues and delivery of a wide range of sizes and types of cargo, including DNA and various forms of RNA.
To learn more, please visit enGene.com and follow us on LinkedIn, X and BlueSky.
Forward-Looking Statements
Certain statements contained in this press release may constitute “forward-looking statements” within the meaning of the safe harbor provisions of the Private Securities Litigation Reform Act of 1995, and “forward-looking information” within the meaning of Canadian securities laws (collectively, “forward-looking statements”). enGene’s forward-looking statements include, but are not limited to, statements regarding enGene’s management team’s expectations, hopes, beliefs, intentions, goals, or strategies regarding the future. In addition, any statements that refer to projections, forecasts or other characterizations of future events or circumstances, including any underlying assumptions, are forward-looking statements. The words “anticipate”, “appear”, “approximate”, “believe”, “continue”, “could”, “estimate”, “expect”, “foresee”, “goal”, “intends”, “may”, “might”, “plan”, “possible”, “potential”, “predict”, “project”, “seek”, “should”, “would”, and similar expressions (or the negative version of such words or expressions) may identify forward-looking statements, but the absence of these words does not mean that a statement is not forward-looking. Forward-looking statements may include, for example, statements about: the potential benefits of the amended protocol, including the potential for patients enrolled under the amended protocol to experience better long-term CR rates, detalimogene’s potential safety and ease of use profile, the development of detalimogene, the potential benefits of detalimogene, including its ability to become a first line therapy in BCG-unresponsive NMIBC and its attractiveness to urologists, plans regarding regulatory interactions and potential BLA submission for detalimogene, plans regarding updates on the LEGEND study, including clinical data and engagement with the FDA, and the potential benefits of medicines developed with the DDX platform. Such statements are subject to numerous important factors, risks and uncertainties, many of which are beyond enGene’s control, that may cause actual events or results to differ materially from enGene’s current expectations. For example, there can be no guarantee that detalimogene will successfully complete necessary clinical development phases, including achieving positive results in the pivotal cohort of the LEGEND study, or that those results or any feedback from regulatory authorities will ultimately lead to BLA submission for, and the approval of, detalimogene.
Management’s expectations and, therefore, any forward-looking statements in this press release could also be affected by risks, uncertainties and assumptions relating to a number of other factors, which could cause the Company’s actual results, performance or achievements to differ materially from those expressed or implied by the forward-looking statements, including, without limitation, the inability of preliminary clinical data to predict the final results of the trial, changes in the results from enGene’s clinical trials, including due to new data collected from the ongoing LEGEND study or future studies, subsequent analysis of existing data, and audit and verification procedures; the content and timing of decisions made by the FDA and other regulatory authorities; the Company’s ability to recruit and retain qualified scientific and management personnel, establish clinical trial sites and enroll patients in its clinical trials, execute on the Company’s clinical development plans; and ability to secure regulatory approval on anticipated timelines, and other risks and uncertainties detailed in filings with Canadian securities regulators on SEDAR+ and with the U.S. Securities and Exchange Commission (“SEC”) on EDGAR, including those described in the “Risk Factors” section of the Company’s Annual Report on Form 10-K for the fiscal year ended October 31, 2024 (copies of which may be obtained at www.sedarplus.ca or www.sec.gov).
You should not place undue reliance on any forward-looking statements, which speak only as of the date on which they are made. enGene anticipates that subsequent events and developments will cause enGene’s assessments to change. While enGene may elect to update these forward-looking statements at some point in the future, enGene specifically disclaims any obligation to do so, unless required by applicable law. Nothing in this press release should be regarded as a representation by any person that the forward-looking statements set forth herein will be achieved or that any of the contemplated results of such forward-looking statements will be achieved.
###
Media contact:
media@engene.com
Investor contact:
investors@engene.com

LEGEND Data Update November 11, 2025 Exhibit 99.3

Disclaimers Cautionary Statement Regarding Forward-Looking Statements This Presentation contains certain forward-looking statements within the meaning of the federal securities laws and "forward-looking information" within the meaning of Canadian securities laws (collectively, "forward-looking statements"). Forward-looking statements may be identified by the use of the words such as “plan”, “forecast”, “intend”, “development”, “expect”, “anticipate”, “become”, “believe”, “continue”, “could”, “estimate”, “expect”, “goa”, “intends”, “may”, “might”, “plan”, “possible”, “project”, “should”, “would”, “strategy”, “future”, “potential”, “opportunity”, “target”, “term”, “will”, “would”, “will be” or similar expressions that predict or indicate future events or trends or that are not statements of historical matters. These forward-looking statements include, but are not limited to, statements regarding the potential benefits of the amended protocol for cohort 1 of the LEGEND study, including the potential for patients enrolled under the amended protocol to experience better long-term complete response (“CR”) rates, detalimogene’s potential safety and ease of use profile, the development of detalimogene, the potential benefits of detalimogene, including its ability to become a first line therapy in BCG-unresponsive NMIBC, plans regarding regulatory interactions and a potential biologics license application (“BLA”) submission for detalimogene, estimates and forecasts of financial and performance metrics, projections of market opportunity and market share, the anticipated market acceptance of detalimogene, expectations and timing related to regulatory submissions and commercial product launches and the prospects for regulatory approval of detalimogene. These forward-looking statements are based on various estimates and assumptions, whether or not identified in this presentation, and on the current expectations of the management of enGene Holdings Inc. ("enGene"), are not predictions of annual performance, and are subject to risks and uncertainties. Such statements are subject to numerous important factors, risks and uncertainties, many of which are beyond enGene's control, that may cause actual events or results to differ materially from enGene's current expectations. For example, there can be no guarantee that detalimogene will successfully complete necessary clinical development phases, including achieving positive results in the pivotal cohort of the LEGEND study, or that those results or any feedback from regulatory authorities will ultimately lead to a BLA submission for, and the approval of, detalimogene. Management's expectations and, therefore, any forward-looking statements in this presentation could also be affected by risks, uncertainties and assumptions relating to a number of other factors, which could cause the Company’s actual results, performance or achievements to differ materially from those expressed or implied by the forward-looking statements, including, without limitation, the inability of preliminary clinical data to predict the final results of the trial, changes in the results from enGene’s clinical trials, including due to new data collected from the ongoing LEGEND study or future studies, subsequent analysis of existing data, and audit and verification procedures; the content and timing of decisions made by the U.S. Food and Drug Administration and other regulatory authorities; the Company’s ability to recruit and retain qualified scientific and management personnel, establish clinical trial sites and enroll patients in its clinical trials, execute on the Company’s clinical development plans; and ability to secure regulatory approval on anticipated timelines, and other risks and uncertainties detailed in filings with Canadian securities regulators on SEDAR+ and with the U.S. Securities and Exchange Commission (“SEC”) on EDGAR, including those described in the “Risk Factors” section of the Company’s Annual Report on Form 10-K for the fiscal year ended October 31, 2024 (copies of which may be obtained at www.sedarplus.ca or www.sec.gov). You should carefully consider the risks and uncertainties described in the “Risk Factors” section of such Annual Report, as well as other documents if and when filed by enGene from time to time with the SEC and Canadian securities regulators. If any of these risks materialize or our assumptions prove incorrect, actual events and results could differ materially from those contained in the forward-looking statements. There may be additional risks that enGene presently knows or that enGene currently believes are immaterial that could also cause actual events and results to differ. In addition, forward-looking statements reflect enGene’s expectations, plans, or forecasts of future events and views as of the date of this presentation. enGene anticipates that subsequent events and developments will cause enGene’s assessments to change. While enGene may elect to update these forward-looking statements at some point in the future, enGene specifically disclaim any obligation to do so, unless required by applicable law. These forward-looking statements should not be relied upon as representing enGene’s assessments as of any date subsequent to the date of this presentation. Accordingly, undue reliance should not be placed upon the forward-looking statements contained herein. Intellectual Property This Presentation contains trademarks, service marks, trade names, copyrights, and products of enGene and other companies, which are the property of their respective owners. The use or display of third parties’ trademarks, service marks, trade names, copyrights, or products in this Presentation is not intended to, and does not, imply a relationship with enGene, or an endorsement of or sponsorship by enGene. Solely for convenience, the trademarks, service marks, and trade names referred to in this Presentation may appear without the ®, TM or SM symbols, but such references are not intended to indicate, in any way, that enGene will not assert, to the fullest extent permitted under applicable law, their rights or the right of the applicable licensor in such trademarks, service marks and trade names. Lead Program (detalimogene voraplasmid) The lead program described herein is an investigational drug therapy that has not been subject to testing designed to demonstrate that the therapy is effective in humans or to provide a basis to predict in advance whether an adequate level of efficacy in humans will be demonstrated in further testing. Although deemed sufficient to permit further testing, the limited, early Phase 1 testing to date is not a sufficient basis on which to predict efficacy or safety. Although the FDA has indicated that the Phase 2 portion of the current LEGEND study may potentially support BLA approval, that outcome will depend entirely on the results of Phase 2 clinical testing, which are not expected to be available until 2026.

Protocol Changes Prior LEGEND Protocol Current LEGEND Protocol T1 disease at pre-enrollment screen Surgically resect lesion via TURBT Enroll patient Perform 2nd resection at lesion site and restage If residual T1 disease present, patient ineligible Ta disease detected at 3 months Response deemed “Progressive Disease” Discontinue patient from study Surgically resect lesion via TURBT Re-induce patient with detalimogene Assessment of Suspected CIS or other disease Patient may be discontinued from study based only on visual impression of CIS Discontinuation requires biopsy confirmation of disease LEGEND Pivotal Cohort Protocol Amendment Q’4 2024 Aligns with Standard of Care and AUA Guidelines

Enrollment Complete: 125 Patients Enrolled in Pivotal Cohort 125 Total Intention to Treat Population (ITT) Pts. 94 Post-Amendment Pts. (75%) 31 Pre-Amendment Pts. (25%) November update based on October 24, 2025 data cut FDA review and agreement on Statistical Analysis Plan (SAP) will impact final patient (n) 62 of 94 Received at least one post-baseline assessment 21 (Sept.’24 Data cut) +10 additional pts.= 31 pts

LEGEND Pivotal Cohort Pre- and Post-Protocol Amendment Characteristics Baseline characteristic POST N=94 PRE N=31 Gender, n (%) Male 76 (80.9) 25 (80.6) Female 18 (19.1) 6 (19.4) Age, years Mean (SD) 69.6 (10.1) 71.8 (10.4) Median (range) 70.5 (35, 85) 74 (46, 90) BCG Doses Median 12.5 12.0 Range 7, 50 6, 33 Tumor Stage, n (%) T1 + CIS 7 (7.4) 3 (9.7) Ta + CIS 33 (35.1) 9 (29.0) CIS 54 (57.4) 19 (61.3) Very High-Risk Characteristics in a High Percentage of Patients ~33% of patients had received additional therapy other than BCG ~42% of patients had CIS + concurrent papillary disease (Ta/T1)
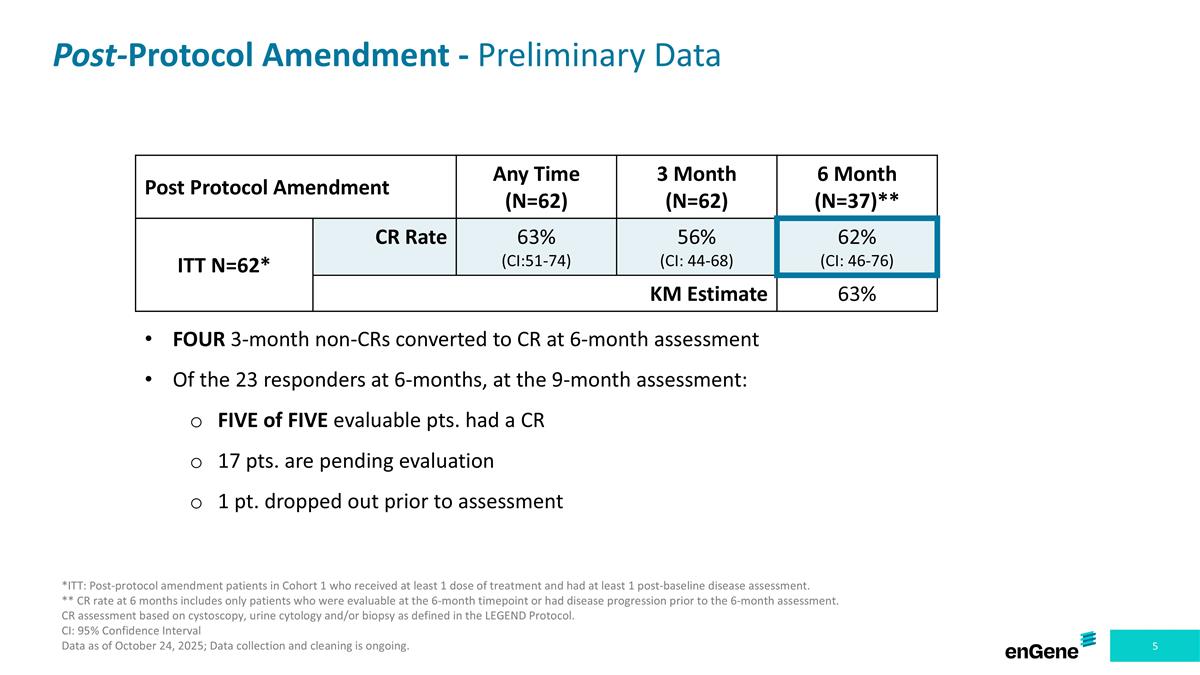
Post-Protocol Amendment - Preliminary Data Post Protocol Amendment Any Time (N=62) 3 Month (N=62) 6 Month (N=37)** ITT N=62* CR Rate 63% (CI:51-74) 56% (CI: 44-68) 62% (CI: 46-76) KM Estimate 63% *ITT: Post-protocol amendment patients in Cohort 1 who received at least 1 dose of treatment and had at least 1 post-baseline disease assessment. ** CR rate at 6 months includes only patients who were evaluable at the 6-month timepoint or had disease progression prior to the 6-month assessment. CR assessment based on cystoscopy, urine cytology and/or biopsy as defined in the LEGEND Protocol. CI: 95% Confidence Interval Data as of October 24, 2025; Data collection and cleaning is ongoing. FOUR 3-month non-CRs converted to CR at 6-month assessment Of the 23 responders at 6-months, at the 9-month assessment: FIVE of FIVE evaluable pts. had a CR 17 pts. are pending evaluation 1 pt. dropped out prior to assessment

Pre-Protocol Amendment Data - Preliminary Data Sept. 2024 Data Cut Updated With Additional Patients N=31 Data Cut Oct. 2025 Any Time (N=31) 3 Month (N=31) 6 Month (N=27)** ITT Population* CR Rate 55% (CI: 38-71) 55% (CI: 38-71) 41% (CI: 25-59) KM Estimate 42% N=21 Data Cut Sept. 2024 Any Time (N=21) 3 Month (N=21) 6 Month (N=17)** ITT Population* CR Rate 71% (CI: 50-86) 67% (CI: 45-83) 47% (CI: 26-69) KM Estimate 51% +10 additional patients not evaluable as of Sept. 2024 cutoff *ITT: Pre-protocol amendment patients in Cohort 1 who received at least 1 dose of treatment and had at least 1 post-baseline disease assessment. ** CR rate at 6 months includes only patients who were evaluable at the 6-month timepoint or had disease progression prior to the 6-month assessment. CR assessment based on cystoscopy, urine cytology and/or biopsy as defined in the LEGEND Protocol. CI: 95% Confidence Interval Data as of October 24, 2025; Data collection and cleaning is ongoing.

Post-Protocol Amendment N=31 as of Oct. 2025 Any Time (N=31) 3 Month (N=31) 6 Month (N=27)** ITT Population* CR Rate 55% (CI: 38-71) 55% (CI: 38-71) 41% (CI: 25-59) KM Estimate 42% Evolution of CR Rate With Protocol Amendment Pre-Protocol Amendment N=62 as of Oct. 2025 Any Time (N=62) 3 Month (N=62) 6 Month (N=37)** ITT Population* CR Rate 63% (CI: 51-74) 56% (CI: 44-68) 62% (CI: 46-76) KM Estimate 63% Pre-protocol amendment pts. had a markedly lower 12-month CR rate than approved products for BCG-unresponsive NMIBC FDA review and agreement on Statistical Analysis Plan (SAP) will impact final patient (n) Following FDA interaction, primary endpoint changed from CR rate at 12-months to CR rate at any time *ITT: Post/pre-protocol amendment patients, respectively, who received at least 1 dose of treatment and had at least 1 post-baseline disease assessment. ** CR rate at 6 months includes only patients who were evaluable at the 6-month timepoint or had disease progression prior to the 6-month assessment. CR assessment based on cystoscopy, urine cytology and/or biopsy as defined in the LEGEND Protocol. CI: 95% Confidence Interval Data as of October 24, 2025; Data collection and cleaning is ongoing.

Any Time CR Rate: Efficacy in Range With Novel Products 6-Month CR Rate Trending Competitively These data are presented for informational purposes and are not based on any head-to-head or comparator clinical studies. Cross-trial comparisons are inherently limited and may suggest misleading similarities or differences. Accordingly, no direct comparisons should be made. *As of the LEGEND Study’s data cut from the pivotal cohort (24 October 2025). Final LEGEND Cohort 1 population may include patients from pre- and post-protocol changes, as well as patients not included in this analysis. 1: CG AUA 2023 Presentation. 2: CG oncology BOND-003 Investor Call presentation, accessed May 1, 2025. 6m CR rate derived from internal calculation based on BOND-003 investor presentation. 3: Based on Inlexzo USPI. 4: Based on Keytruda USPI. 6m CR rates based on internal calculation from data available in Balar et al., Lancet Onc 2021 Jul;22(7):919-930; 5: Based on Adstiladrin USPI. 6m rates from Boorjian et al, Lancet Onc 2021 Jan;22(1):107-117 with N=103. 6: Based on Anktiva USPI. 6m rates from Chamie et al., NEJM 2023 Jan;2(1):EVIDoa2200167. 7: Daneshmand et al., J of Clin Oncol 2025 Jul; 00:1-11. Detalimogene Pre-Amendment Detalimogene Post-Amendment Cretostimogene Inlexzo Keytruda Adstiladrin Anktiva + BCG Trial LEGEND LEGEND BOND-0021 BOND-0032 SunRISe-13 KEYNOTE-0574 NCT027738495 QUILT 3.0326 CR Rate, Any Time 55% 63% 65% 76% 82% 41% 51% 62% Duration of Response ≥ 12 mon.%(n) TBD Insufficient Data Not disclosed Not disclosed 51% (35) 46% (18) 46% (23) 58% (28) CR Rate, 6m 41% 62% 44% 63% 59%7 34% 41% 56% Sample Size N = 31 N = 62 N = 46 N = 110 N = 83 N = 96 N = 98 N = 77

Enrollment Rate Heavily Skewed to Last 10 Months Bolus of 12-month Patient Evaluations in 2H 2026
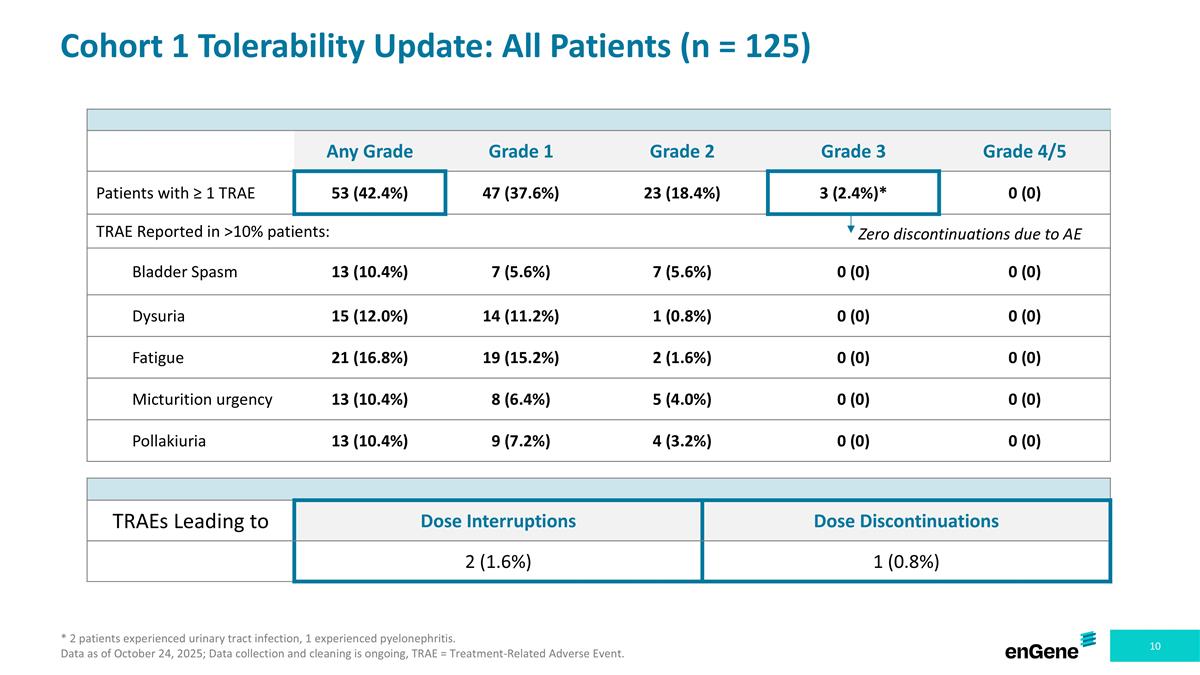
Cohort 1 Tolerability Update: All Patients (n = 125) * 2 patients experienced urinary tract infection, 1 experienced pyelonephritis. Data as of October 24, 2025; Data collection and cleaning is ongoing, TRAE = Treatment-Related Adverse Event. Any Grade Grade 1 Grade 2 Grade 3 Grade 4/5 Patients with ≥ 1 TRAE 53 (42.4%) 47 (37.6%) 23 (18.4%) 3 (2.4%)* 0 (0) TRAE Reported in >10% patients: Bladder Spasm 13 (10.4%) 7 (5.6%) 7 (5.6%) 0 (0) 0 (0) Dysuria 15 (12.0%) 14 (11.2%) 1 (0.8%) 0 (0) 0 (0) Fatigue 21 (16.8%) 19 (15.2%) 2 (1.6%) 0 (0) 0 (0) Micturition urgency 13 (10.4%) 8 (6.4%) 5 (4.0%) 0 (0) 0 (0) Pollakiuria 13 (10.4%) 9 (7.2%) 4 (3.2%) 0 (0) 0 (0) TRAEs Leading to Dose Interruptions Dose Discontinuations 2 (1.6%) 1 (0.8%) Zero discontinuations due to AE

Trending to Best-in-Class Tolerability for Detalimogene Investigational Agents based on TRAEs*** Deta n=125 Creto n = 112 Any Grade 42% 63% Serious 1.6% 1.8% Fatal 0 0 Dose Interruptions 1.6% Not reported Dose Discontinuations 0.8% 0 Source Internal Data* CG Investor Deck** Approved Products based on ADRs*** Inlexzo n=85 Adstiladrin n=157 Anktiva + BCG n=88 Keytruda n=148 84% Not disclosed Not disclosed 66% 24% 11% 16% 28% 1.2% None noted 1.1% None noted 41% 34% 34% 22% 7% 2% 7% 11% USPI USPI USPI USPI *Data Cutoff: October 24, 2025; Data collection and cleaning is ongoing. ** September 9, 2025 Investor Deck. *** TRAEs: Treatment-related Adverse Events; ADRs: Adverse Drug Reactions

Additional Cohorts - Trial in Progress Update All NMIBC cohorts refer to high-risk NMIBC unless otherwise specified. Expected data update, timelines, enrollment targets, and anticipated milestones reflect enGene management's current estimate and are subject to change. Enrollment Status Detalimogene Voraplasmid (intravesical) BCG-Unresponsive NMIBC with CIS Pivotal Cohort 1 Enrollment, n = 125 BCG-Naïve NMIBC with CIS Ph2 Cohort 2a, n ≤ 30 BCG-Exposed NMIBC with CIS Ph2 Cohort 2b, n ≤ 70 BCG-Unresponsive NMIBC, Papillary-Only Ph2 Cohort 3, n = 50-100 30 pts. Enrolled Fully Enrolled 45 pts. Enrolled 36 pts. Enrolled

LEGEND Cohort 1 Update Summary Preliminary data suggest positive impact on efficacy post-protocol amendment Trending to best-in-class tolerability profile FDA review and agreement on Statistical Analysis Plan (SAP) will impact final patient (n) Heavily skewed enrollment curve delays insights into long-term efficacy Long-term durability data is limited to patients enrolled prior to protocol revision

Detalimogene: Designed to Meet the Needs of Urologists and Patients Preliminary evidence of efficacy* Non-viral gene therapy designed for streamlined clinical experience *LEGEND phase 1 reported data and September 2024 and November 2025 preliminary pivotal phase 2 reported data Efficacy Ease-of-Use Safety Generally mild treatment related adverse events*

Corporate Presentation November 2025 Exhibit 99.4

Disclaimers Cautionary Statement Regarding Forward-Looking Statements This Presentation contains certain forward-looking statements within the meaning of the federal securities laws and "forward-looking information" within the meaning of Canadian securities laws (collectively, "forward-looking statements"). Forward-looking statements may be identified by the use of the words such as “plan”, “forecast”, “intend”, “development”, “expect”, “anticipate”, “become”, “believe”, “continue”, “could”, “estimate”, “expect”, “goa”, “intends”, “may”, “might”, “plan”, “possible”, “project”, “should”, “would”, “strategy”, “future”, “potential”, “opportunity”, “target”, “term”, “will”, “would”, “will be” or similar expressions that predict or indicate future events or trends or that are not statements of historical matters. These forward-looking statements include, but are not limited to, statements regarding the potential benefits of the amended protocol for cohort 1 of the LEGEND study, including the potential for patients enrolled under the amended protocol to experience better long-term complete response (“CR”) rates, detalimogene’s potential safety and ease of use profile, the development of detalimogene, the potential benefits of detalimogene, including its ability to become a first line therapy in BCG-unresponsive NMIBC, plans regarding regulatory interactions and a potential biologics license application (“BLA”) submission for detalimogene, estimates and forecasts of financial and performance metrics, projections of market opportunity and market share, the anticipated market acceptance of detalimogene, expectations and timing related to regulatory submissions and commercial product launches and the prospects for regulatory approval of detalimogene. These forward-looking statements are based on various estimates and assumptions, whether or not identified in this presentation, and on the current expectations of the management of enGene Holdings Inc. ("enGene"), are not predictions of annual performance, and are subject to risks and uncertainties. Such statements are subject to numerous important factors, risks and uncertainties, many of which are beyond enGene's control, that may cause actual events or results to differ materially from enGene's current expectations. For example, there can be no guarantee that detalimogene will successfully complete necessary clinical development phases, including achieving positive results in the pivotal cohort of the LEGEND study, or that those results or any feedback from regulatory authorities will ultimately lead to a BLA submission for, and the approval of, detalimogene. Management's expectations and, therefore, any forward-looking statements in this presentation could also be affected by risks, uncertainties and assumptions relating to a number of other factors, which could cause the Company’s actual results, performance or achievements to differ materially from those expressed or implied by the forward-looking statements, including, without limitation, the inability of preliminary clinical data to predict the final results of the trial, changes in the results from enGene’s clinical trials, including due to new data collected from the ongoing LEGEND study or future studies, subsequent analysis of existing data, and audit and verification procedures; the content and timing of decisions made by the U.S. Food and Drug Administration and other regulatory authorities; the Company’s ability to recruit and retain qualified scientific and management personnel, establish clinical trial sites and enroll patients in its clinical trials, execute on the Company’s clinical development plans; and ability to secure regulatory approval on anticipated timelines, and other risks and uncertainties detailed in filings with Canadian securities regulators on SEDAR+ and with the U.S. Securities and Exchange Commission (“SEC”) on EDGAR, including those described in the “Risk Factors” section of the Company’s Annual Report on Form 10-K for the fiscal year ended October 31, 2024 (copies of which may be obtained at www.sedarplus.ca or www.sec.gov). You should carefully consider the risks and uncertainties described in the “Risk Factors” section of such Annual Report, as well as other documents if and when filed by enGene from time to time with the SEC and Canadian securities regulators. If any of these risks materialize or our assumptions prove incorrect, actual events and results could differ materially from those contained in the forward-looking statements. There may be additional risks that enGene presently knows or that enGene currently believes are immaterial that could also cause actual events and results to differ. In addition, forward-looking statements reflect enGene’s expectations, plans, or forecasts of future events and views as of the date of this presentation. enGene anticipates that subsequent events and developments will cause enGene’s assessments to change. While enGene may elect to update these forward-looking statements at some point in the future, enGene specifically disclaim any obligation to do so, unless required by applicable law. These forward-looking statements should not be relied upon as representing enGene’s assessments as of any date subsequent to the date of this presentation. Accordingly, undue reliance should not be placed upon the forward-looking statements contained herein. Intellectual Property This Presentation contains trademarks, service marks, trade names, copyrights, and products of enGene and other companies, which are the property of their respective owners. The use or display of third parties’ trademarks, service marks, trade names, copyrights, or products in this Presentation is not intended to, and does not, imply a relationship with enGene, or an endorsement of or sponsorship by enGene. Solely for convenience, the trademarks, service marks, and trade names referred to in this Presentation may appear without the ®, TM or SM symbols, but such references are not intended to indicate, in any way, that enGene will not assert, to the fullest extent permitted under applicable law, their rights or the right of the applicable licensor in such trademarks, service marks and trade names. Industry and Market Data This Presentation relies on and refers to certain information and statistics based on estimates by enGene’s management and/or obtained from third party sources which enGene believes to be reliable. enGene has not independently verified the accuracy or completeness of any such third party information, which involves elements of subjective judgment and analysis that may or may not prove to be accurate. None enGene, or its affiliates or any third parties that provide information to enGene or its affiliates, such as market research firms, guarantees the accuracy, completeness, timeliness, or availability of any information. None enGene, or its affiliates, or any third parties that provide information to enGene, and its affiliates, such as market research firms, is responsible for any errors or omissions (negligent or otherwise), regardless of the cause, or the results obtained from the use of such content. enGene may have supplemented such information where necessary, taking into account publicly available information about other industry participants and enGene management’s best view as to information that is not publicly available. Neither enGene nor its affiliates give any express or implied warranties with respect to the information included herein, including, but not limited to, any warranties regarding its accuracy or of merchantability or fitness for a particular purpose or use, and they expressly disclaim any responsibility or liability for direct, indirect, incidental, exemplary, compensatory, punitive, special, or consequential damages, costs, expenses, legal fees, or losses (including lost income or profits and opportunity costs) in connection with the use of the information herein. Lead Program (detalimogene voraplasmid) The lead program described herein is an investigational drug therapy that has not been subject to testing designed to demonstrate that the therapy is effective in humans or to provide a basis to predict in advance whether an adequate level of efficacy in humans will be demonstrated in further testing. Although deemed sufficient to permit further testing, the limited, early Phase 1 testing to date is not a sufficient basis on which to predict efficacy or safety. Although the FDA has indicated that the Phase 2 portion of the current LEGEND study may potentially support BLA approval, that outcome will depend entirely on the results of Phase 2 clinical testing, which are not expected to be available until 2026.

Detalimogene Voraplasmid for Non-Muscle Invasive Bladder Cancer Designed for Seamless Integration into High-Volume Urology Practices Transformational Market Opportunity in Area of High Unmet Need Novel class of drug: non-viral gene therapy Powerful and coordinated immune activation across two major axes Clinical benefit and best-in-class ease-of-use Clinical activity, favorable safety, and a streamlined patient experience Near term planned catalysts Data Update: 2H 2026 BLA Submission: 2H 2026 Potential Launch: 2027

DDX Platform: Unique Non-Viral Gene Therapy Without the Headaches Large Genetic Cargo Non-Immunogenic Low Cost of Goods/ No Specialized Handling Detalimogene

NMIBC Represents 75-80% of Bladder Cancer Diagnoses NMIBC Patients Segmentation CIS +/- papillary tumors Papillary tumors only US Incident Bladder Cancer ~85,000/year Non-Muscle Invasive ~65,000/year HR NMIBC ~25,000/year US Prevalent Bladder Cancer ~730,000 Carcinoma in situ Non-invasive papillary tumors Papillary tumor that invades subepithelial connective tissue Outer muscle Inner muscle Lamina propria Bladder Mucosa CIS/Tis Ta T1 Papillary Tumors Sources: SEER database; Knowles et al., Nat Rev Cancer 15, 25–41 (2015); Isharwal and Konety, Indian Journal of Urology, 2015; TNM Classification, 8th Edition; Tan et al., Eur Urol Oncol (2022): https://doi.org/10.1016/j.euo.2022.05.005, Nielson et al. Cancer. 2013 Oct 10;120(1):86–95. doi: 10.1002/cncr.28397.

MIBC = Muscle-invasive bladder cancer Sources: Mossanen M, Gore JL, Curr Opin Urol. 2014 Sep;24(5):487-91; Clark O et al., Pharmacoecon Open. 2024 Aug 18; https://seer.cancer.gov/statfacts/html/common.html#comparison, van den Bosch and Witjes, Eur Urol. 2011 Sep;60(3):493-500. NMIBC: Slow Progression but High Incidence, Cost, and Recurrence Rate Bladder cancer is expensive to manage: >$6.5B/year total cost Bladder cancer is common: 6th most common by incidence NMIBC is highly recurrent but slow progressing: 20% progress to MIBC over 10 years
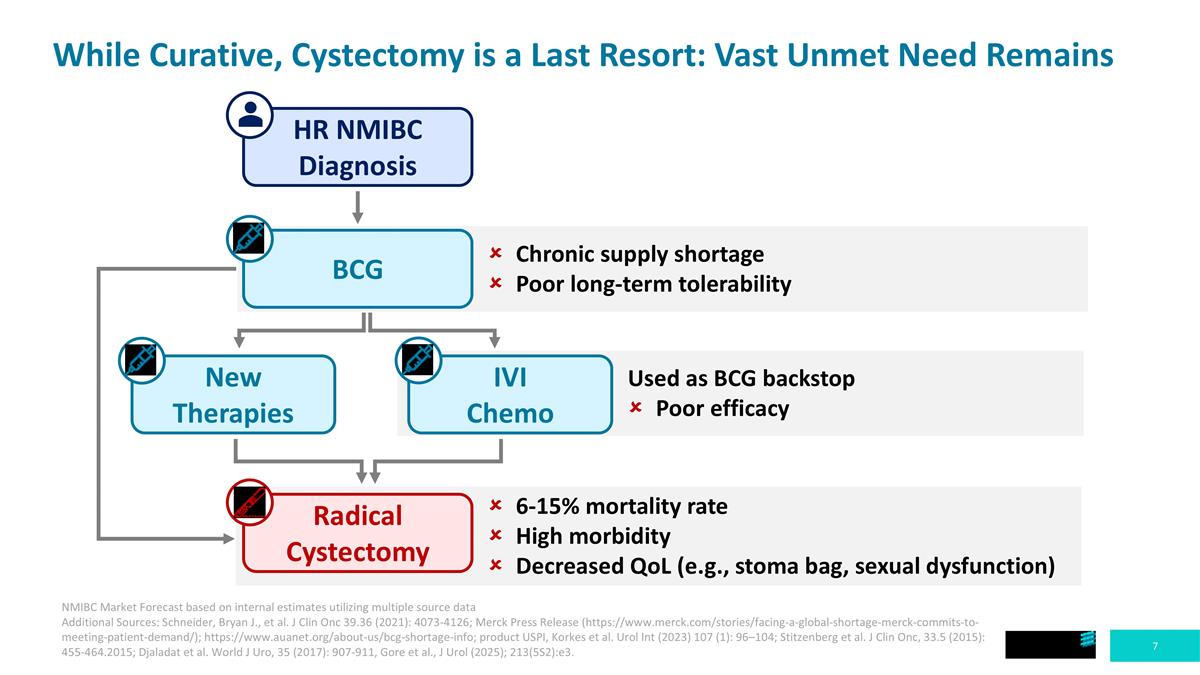
While Curative, Cystectomy is a Last Resort: Vast Unmet Need Remains HR NMIBC Diagnosis BCG New Therapies Radical Cystectomy IVI Chemo Chronic supply shortage Poor long-term tolerability Used as BCG backstop Poor efficacy 6-15% mortality rate High morbidity Decreased QoL (e.g., stoma bag, sexual dysfunction) NMIBC Market Forecast based on internal estimates utilizing multiple source data Additional Sources: Schneider, Bryan J., et al. J Clin Onc 39.36 (2021): 4073-4126; Merck Press Release (https://www.merck.com/stories/facing-a-global-shortage-merck-commits-to-meeting-patient-demand/); https://www.auanet.org/about-us/bcg-shortage-info; product USPI, Korkes et al. Urol Int (2023) 107 (1): 96–104; Stitzenberg et al. J Clin Onc, 33.5 (2015): 455-464.2015; Djaladat et al. World J Uro, 35 (2017): 907-911, Gore et al., J Urol (2025); 213(5S2):e3.
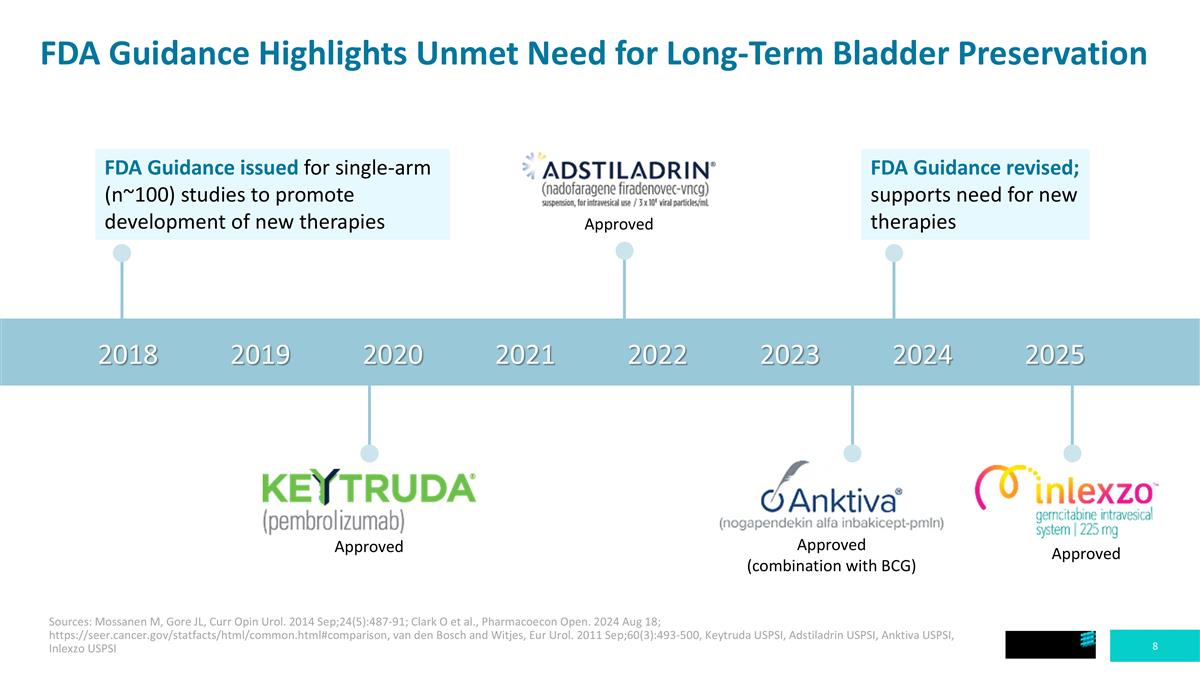
FDA Guidance Highlights Unmet Need for Long-Term Bladder Preservation 2018 2019 2020 2021 2022 2023 2024 2025 FDA Guidance issued for single-arm (n~100) studies to promote development of new therapies Approved FDA Guidance revised; supports need for new therapies Approved Approved (combination with BCG) Approved Sources: Mossanen M, Gore JL, Curr Opin Urol. 2014 Sep;24(5):487-91; Clark O et al., Pharmacoecon Open. 2024 Aug 18; https://seer.cancer.gov/statfacts/html/common.html#comparison, van den Bosch and Witjes, Eur Urol. 2011 Sep;60(3):493-500, Keytruda USPSI, Adstiladrin USPSI, Anktiva USPSI, Inlexzo USPSI

Tolerability 12-month CR Rate 12-month DOR Rate Lack of Freezer Storage & Thaw Time Requirements Cost of Treatment/Coverage Ease of Administration Any time CR Rate Community Urologists Balance a Range of Needs in Selecting Treatments Community Urologist Treatment Considerations: Tolerability, Efficacy & Ease of Use 67% Community Practices Urologist Primary Practice Settings % of Respondents that Listed in Top 5 (of 10 options) Community practice includes private practice, public or private hospital, and health system or HMO. Sources: “The State of the Urology Workforce and Practice in the United States, 2024” American Urological Association Education and Research, Inc. Raymond James & Associates “Community Urologist Survey (n=25) Supportive of enGene’s Strategy” October 2025.

Buy+Bill 5% of revenue in 2008 and grown to 15-20% in 2025 Products requiring fewer clinical resources are preferred Practice Operations are Strained: Time Savings are Economically Valuable Sources: “The State of the Urology Workforce and Practice in the United States, 2024” American Urological Association Education and Research, Inc, McKinsey & Company “Assessing the Lingering Impact of COVID-19 on the Nursing Workforce” 2024, Hyman et al., J of Urol 2025 April 213(5S) Supplement e288, Kopechek et al. Urol Pract 2025 Jul;12(4):380-390, enGene internal estimate. <5 urologists per 100,000 population Nursing vacancies High turnover Staff Shortage Revenue Pressure Reduced reimbursement for surgery Increased cost for procedures
Detalimogene: Designed to Meet the Needs of Urologists and Patients Preliminary evidence of clinical activity* Non-viral gene therapy designed for streamlined clinical experience *LEGEND phase 1 reported data and September 2024 and November 2025 preliminary pivotal phase 2 reported data Efficacy Ease-of-Use Safety Generally mild treatment related adverse events*

Non-Viral Coordinated Immune Activation Across Two Major Axes Plasmid Encodes Three Immune-Stimulatory Genes to Activate Both Innate and Adaptive Pathways Bladder lumen Intracellular Detalimogene DNA plasmid dsRNA agonists of RIG-I Secreted IL-12 Activate intracellular RIG-I receptor Locally stimulates incoming T cells Adaptive immune system activation: secreted IL-12 T-cell–dependent cytokine response promotes tumor killing and immune memory Bladder-restricted production reduces potential for systemic AEs Innate immune system activation: dual RIG-I agonists Activates NK cells and dampens suppressor cells to promote killing Drives T-cell recruitment and neoantigen presentation dsRNA, double-stranded RNA; IL-12, interleukin-12; IVI, intravesical instillation, NK, natural killer; RIG-I, retinoic acid–inducible gene I. Narayan VM, et al. Poster presented at: ASCO Genitourinary Cancers Symposium; February 13-15, 2025; San Francisco, CA.

DDX Platform Designed to Support Industrial-Scale Manufacturing e e e e e - - - - - (Stabilizes Nanoparticle) PEG-PLE Bulk Mixing In-line Mixing PEGylated DDX/DNA Nanoparticle (Non-Viral Drug Product) DDX/DNA Nanoparticle + + + + + + + Non-viral, lyophilized drug product No special delivery or handling Established at-scale manufacturing Regulatory validation batches near completion + + + + + + + DDX: Proprietary Sugar Polymer (Facilitates Cellular Uptake) Plasmid DNA (Encodes Genetic Cargo) - - - - - - - - - + 2 4 3 1

Multiple LEGEND Cohorts Maximize Detalimogene’s Potential Detalimogene Voraplasmid (intravesical) Comprehensive IP Portfolio Through at Least 2040 *These additional non-pivotal cohorts of the LEGEND study provide further opportunity to demonstrate the potential use of detalimogene in clinical settings with unmet need. Progress as of October 24, 2025. All cohorts are currently in Phase 2 clinical trials. All NMIBC cohorts refer to high-risk NMIBC unless otherwise specified Expected data update, timelines, enrollment targets, and anticipated milestones reflect enGene management's current estimate and are subject to change Fully Enrolled 125 patients 30 patients Preclinical activity ongoing 36 patients 45 patients BCG-Unresponsive NMIBC with Cis Key Protocol Amendments Implemented Q4 2024 Pre-Clinical Phase 1 Pivotal Cohort 2a: BCG-Naïve NMIBC with CIS* Cohort 2b: BCG-Exposed NMIBC with CIS* Cohort 3: BCG-Unresponsive NMIBC, Papillary-Only* Urological Targets (undisclosed) Status Completed Active DDX Platform

Pivotal Study Target Enrollment Achieved Patients: BCG-Unresponsive High-risk NMIBC with CIS Design: Global, single-arm, open label N = 125 (fully enrolled) Dosing2: Year 1: 800μg/ml IVI at weeks 1,2,5,6 Q3M Year 2-3 (Maintenance): 800μg/ml IVI at weeks 1,2 Q3M Primary Endpoint: Anytime CR rate Key Secondary Endpoint: Duration of Response 1: IVI = Intravesical. LEGEND allows patients from any cohort who are in complete response at 12 months to continue receiving detalimogene on a dose-reduced maintenance schedule throughout their second year of treatment. At the end of year two, patients may optionally elect to remain on maintenance therapy for another year, for a total of three years of therapy.
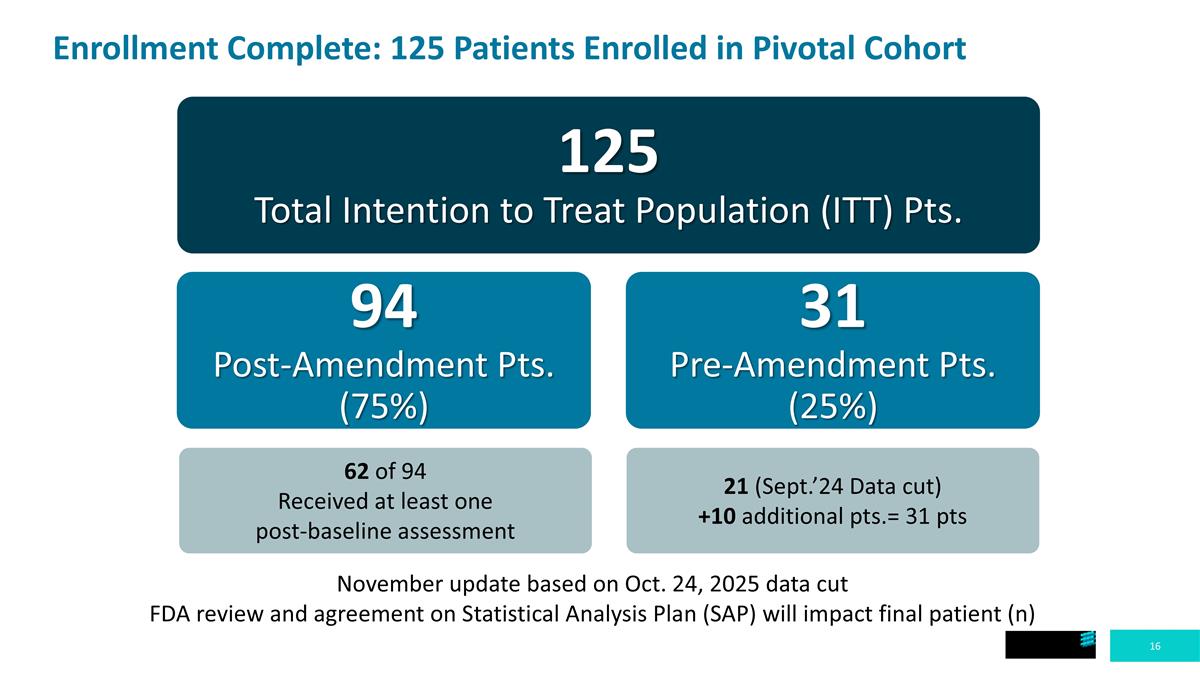
Enrollment Complete: 125 Patients Enrolled in Pivotal Cohort 125 Total Intention to Treat Population (ITT) Pts. 94 Post-Amendment Pts. (75%) 31 Pre-Amendment Pts. (25%) November update based on Oct. 24, 2025 data cut FDA review and agreement on Statistical Analysis Plan (SAP) will impact final patient (n) 62 of 94 Received at least one post-baseline assessment 21 (Sept.’24 Data cut) +10 additional pts.= 31 pts

LEGEND Pivotal Cohort Pre- and Post-Protocol Amendment Characteristics Baseline characteristic POST N=94 PRE N=31 Gender, n (%) Male 76 (80.9) 25 (80.6) Female 18 (19.1) 6 (19.4) Age, years Mean (SD) 69.6 (10.1) 71.8 (10.4) Median (range) 70.5 (35, 85) 74 (46, 90) BCG Doses Median 12.5 12.0 Range 7, 50 6, 33 Tumor Stage, n (%) T1 + CIS 7 (7.4) 3 (9.7) Ta + CIS 33 (35.1) 9 (29.0) CIS 54 (57.4) 19 (61.3) Very High-Risk Characteristics in a High Percentage of Patients ~33% of patients had received additional therapy other than BCG ~42% of patients had CIS + concurrent papillary disease (Ta/T1)

Post-Protocol Amendment - Preliminary Data Post Protocol Amendment Any Time (N=62) 3 Month (N=62) 6 Month (N=37)** ITT N=62* CR Rate 63% (CI:51-74) 56% (CI: 44-68) 62% (CI: 46-76) KM Estimate 63% *ITT: Post-protocol amendment patients in Cohort 1 who received at least 1 dose of treatment and had at least 1 post-baseline disease assessment. ** CR rate at 6 months includes only patients who were evaluable at the 6-month timepoint or had disease progression prior to the 6-month assessment. CR assessment based on cystoscopy, urine cytology and/or biopsy as defined in the LEGEND Protocol. CI: 95% Confidence Interval Data as of October 24, 2025; Data collection and cleaning is ongoing. FOUR 3-month non-CRs converted to CR at 6-month assessment Of the 23 responders at 6-months, at the 9-month assessment: FIVE of FIVE evaluable pts. had a CR 17 pts. are pending evaluation 1 pt. dropped out prior to assessment

Post-Protocol Amendment N=31 as of Oct. 2025 Any Time (N=31) 3 Month (N=31) 6 Month (N=27)** ITT Population* CR Rate 55% (CI: 38-71) 55% (CI: 38-71) 41% (CI: 25-59) KM Estimate 42% Evolution of CR Rate With Protocol Amendment Pre-Protocol Amendment N=62 as of Oct. 2025 Any Time (N=62) 3 Month (N=62) 6 Month (N=37)** ITT Population* CR Rate 63% (CI: 51-74) 56% (CI: 44-68) 62% (CI: 46-76) KM Estimate 63% Pre-protocol amendment pts. had a markedly lower 12-month CR rate than approved products for BCG-unresponsive NMIBC FDA review and agreement on Statistical Analysis Plan (SAP) will impact final patient (n) Following FDA interaction, primary endpoint changed from CR rate at 12-months to CR rate at any time *ITT: Post/pre-protocol amendment patients, respectively, who received at least 1 dose of treatment and had at least 1 post-baseline disease assessment. ** CR rate at 6 months includes only patients who were evaluable at the 6-month timepoint or had disease progression prior to the 6-month assessment. CR assessment based on cystoscopy, urine cytology and/or biopsy as defined in the LEGEND Protocol. CI: 95% Confidence Interval Data as of October 24, 2025; Data collection and cleaning is ongoing.

Cohort 1 Tolerability Update: All Patients (n = 125) * 2 patients experienced urinary tract infection, 1 experienced pyelonephritis. 0 discontinuations due to Grade 3 TRAEs. Data as of October 24, 2025; Data collection and cleaning is ongoing, TRAE = Treatment-Related Adverse Event. Any Grade Grade 1 Grade 2 Grade 3 Grade 4/5 Patients with ≥ 1 TRAE 53 (42.4%) 47 (37.6%) 23 (18.4%) 3 (2.4%)* 0 (0) TRAE Reported in >10% patients: Bladder Spasm 13 (10.4%) 7 (5.6%) 7 (5.6%) 0 (0) 0 (0) Dysuria 15 (12.0%) 14 (11.2%) 1 (0.8%) 0 (0) 0 (0) Fatigue 21 (16.8%) 19 (15.2%) 2 (1.6%) 0 (0) 0 (0) Micturition urgency 13 (10.4%) 8 (6.4%) 5 (4.0%) 0 (0) 0 (0) Pollakiuria 13 (10.4%) 9 (7.2%) 4 (3.2%) 0 (0) 0 (0) TRAEs Leading to Dose Interruptions Dose Discontinuations 2 (1.6%) 1 (0.8%)

Patients Practice Designed to Streamline Experience for Patients and Urology Practices Fewer doses per 12-week cycle than BCG induction 1-hour total chair time in clinical trials and no thaw or pre-wash steps No urine bleaching or close contact precautions with loved ones Storage at 2-8°C* possible Administered by nurses in exam rooms No decontamination or HCP exposure precautions Detalimogene *Subject to confirmation from the FDA.

Any Time CR Rate: Efficacy in Range With Novel Products 6-Month CR Rate Trending Competitively These data are presented for informational purposes and are not based on any head-to-head or comparator clinical studies. Cross-trial comparisons are inherently limited and may suggest misleading similarities or differences. Accordingly, no direct comparisons should be made. *As of the LEGEND Study’s data cut from the pivotal cohort (24 October 2025). Final LEGEND Cohort 1 population may include patients from pre- and post-protocol changes, as well as patients not included in this analysis. 1: CG AUA 2023 Presentation. 2: CG oncology BOND-003 Investor Call presentation, accessed May 1, 2025. 6m CR rate derived from internal calculation based on BOND-003 investor presentation. 3: Based on Inlexzo USPI. 4: Based on Keytruda USPI. 6m CR rates based on internal calculation from data available in Balar et al., Lancet Onc 2021 Jul;22(7):919-930; 5: Based on Adstiladrin USPI. 6m rates from Boorjian et al, Lancet Onc 2021 Jan;22(1):107-117 with N=103. 6: Based on Anktiva USPI. 6m rates from Chamie et al., NEJM 2023 Jan;2(1):EVIDoa2200167. 7: Daneshmand et al., J of Clin Oncol 2025 Jul; 00:1-11. Note: To-date, no head-to-head comparisons of any other products to any of our product candidates in any clinical trial have been completed; results have been obtained from different trials with different designs, endpoints and patient populations; results may not be comparable. Detalimogene Pre-Amendment Detalimogene Post-Amendment Cretostimogene Inlexzo Keytruda Adstiladrin Anktiva + BCG Trial LEGEND LEGEND BOND-0021 BOND-0032 SunRISe-13 KEYNOTE-0574 NCT027738495 QUILT 3.0326 CR Rate, Any Time 55% 63% 65% 76% 82% 41% 51% 62% Duration of Response ≥ 12 mon.%(n) TBD Insufficient Data Not disclosed Not disclosed 51% (35) 46% (18) 46% (23) 58% (28) CR Rate, 6m 41% 62% 44% 63% 59%7 34% 41% 56% Sample Size N = 31 N = 62 N = 46 N = 110 N = 83 N = 96 N = 98 N = 77

Trending to Best-in-Class Tolerability for Detalimogene Investigational Agents based on TRAEs*** Deta n=125 Creto n = 112 Any Grade 42% 63% Serious 1.6% 1.8% Fatal 0 0 Dose Interruptions 1.6% Not reported Dose Discontinuations 0.8% 0 Source Internal Data* CG Investor Deck** Approved Products based on ADRs*** Inlexzo n=85 Adstiladrin n=157 Anktiva + BCG n=88 Keytruda n=148 84% Not disclosed Not disclosed 66% 24% 11% 16% 28% 1.2% None noted 1.1% None noted 41% 34% 34% 22% 7% 2% 7% 11% USPI USPI USPI USPI *Data Cutoff: October 24, 2025; Data collection and cleaning is ongoing. ** September 9, 2025 Investor Deck. *** TRAEs: Treatment-related Adverse Events; ADRs: Adverse Drug Reactions Note: To-date, no head-to-head comparisons of any other products to any of our product candidates in any clinical trial have been completed; results have been obtained from different trials with different designs, endpoints and patient populations; results may not be comparable.

Designed for Logistical and Post-Treatment Protocol Simplicity Ease of Use Non-Viral Gene Therapy Viral Gene Therapy Chemo Device Combination Bacterial Therapy Checkpoint Inhibitor No Complex Shipping & Storage ü û ü ü ü Short Single-Step Administration/Procedure ü û û ü ü No Viral/Bacterium Decontamination or HCP Exposure Precautions ü û ü û ü No Post-Treatment Patient Precautions ü û û û ü No Requirement for Specially Qualified Staff and/or Facility ü ü û ü û Sources: Adstiladrin USPI, Inlexzo USPI, BCG USPI, Keytruda USPI, CG Oncology Corporate Presentation accessed November 5 2025, Raymond James & Associates “Community Urologist Survey Supportive of enGene’s Strategy” October 2025

Logical Sequencing – A Critical Future Consideration for Long-Term Care HR NMIBC Diagnosis BCG Chemotherapy, CPI & Clinical Trials Radical Cystectomy HR NMIBC Diagnosis BCG Non-Viral Gene Therapy Chemotherapy Referral to Academic Center Viral Gene Therapy Checkpoint Radical Cystectomy Future Past

Experience Developing and Commercializing Highly Successful Medicines RON COOPER Chief Executive Officer Chief Executive Officer, Albireo Pharma President, Europe, Bristol Myers Squibb ANTHONY CHEUNG, Ph.D. Chief Scientific Officer Co-founder, enGene Co-inventor All Key enGene Patents ALEX NICHOLS, Ph.D. Chief Strategy & Operations Officer Co-Founder and CEO, Mythic Therapeutics Flagship Pioneering JOAN CONNOLLY Chief Technology Officer Chief Technology Officer, Albireo Pharma SVP Technical Operations, Stemline Therapeutics AMY POTT Chief Global Commercialization Officer Head, Rare Disease & Ophthalmology, Global Commercial, Astellas Head, Internal Medicine & Oncology, Commercial, Shire RYAN DAWS Chief Financial Officer CFO Roles: Concert Therapeutics, Obsidian Therapeutics Investment Banker Roles: Cowen, Stifel, Baird LEE GIGUERE Chief Legal Officer & Secretary CLO, Obsidian Therapeutics General Counsel, Chiasma Inc. Karyopharm Therapeutics, Boston Scientific, Goodwin Procter JILL BUCK Chief Development Officer Head, Clinical Development Operations, Rare Disease, Ipsen Albireo Pharma, Ziopharm Oncology, Synageva Biopharma HUSSEIN SWEITI, M.D. Chief Medical Officer Global Medical Head, Oncology Clinical Development, Johnson & Johnson MATTHEW BOYD Chief Regulatory Officer VP, Head of Regulatory Affairs, Zambon USA VP, Regulatory Affairs, Albireo Pharma

Detalimogene Voraplasmid: Designed to be the First Line After BCG Transformational Opportunity Streamlines Clinical Workflow Designed for Patient Impact Near-Term Planned Catalysts NMIBC market forecasted to be >$20B Non-viral DDX Platform Enables Gene Therapy Without the Headaches Demonstrated Clinical Activity, Favorable Safety, and a Streamlined Patient Experience Data Update Following FDA Interactions: 2H 2026 BLA Filing: 2H 2026 Potential Launch: 2027 Well-Capitalized, with Runway Projected into 2027


Protocol Changes Prior LEGEND Protocol Current LEGEND Protocol T1 disease at pre-enrollment screen Surgically resect lesion via TURBT Enroll patient Perform 2nd resection at lesion site and restage If residual T1 disease present, patient ineligible Ta disease detected at 3 months Response deemed “Progressive Disease” Discontinue patient from study Surgically resect lesion via TURBT Re-induce patient with detalimogene Assessment of Suspected CIS or other disease Patient may be discontinued from study based only on visual impression of CIS Discontinuation requires biopsy confirmation of disease LEGEND Pivotal Cohort Protocol Amendment Q’4 2024 Aligns with Standard of Care and AUA Guidelines

Pre-Protocol Amendment Data - Preliminary Data Sept. 2024 Data Cut Updated With Additional Patients N=31 Data Cut Oct. 2025 Any Time (N=31) 3 Month (N=31) 6 Month (N=27)** ITT Population* CR Rate 55% (CI: 38-71) 55% (CI: 38-71) 41% (CI: 25-59) KM Estimate 42% N=21 Data Cut Sept. 2024 Any Time (N=21) 3 Month (N=21) 6 Month (N=17)** ITT Population* CR Rate 71% (CI: 50-86) 67% (CI: 45-83) 47% (CI: 26-69) KM Estimate 51% +10 additional patients not evaluable as of Sept. 2024 cutoff *ITT: Pre-protocol amendment patients in Cohort 1 who received at least 1 dose of treatment and had at least 1 post-baseline disease assessment. ** CR rate at 6 months includes only patients who were evaluable at the 6-month timepoint or had disease progression prior to the 6-month assessment. CR assessment based on cystoscopy, urine cytology and/or biopsy as defined in the LEGEND Protocol. CI: 95% Confidence Interval Data as of October 24, 2025; Data collection and cleaning is ongoing.

Enrollment Rate Heavily Skewed to Last 10 Months Bolus of 12-month Patient Evaluations in 2H 2026