
Corporate Presentation November 5, 2025 Nasdaq: NAMS

Disclaimer This presentation (together with oral statements made in connection herewith, this “Presentation”) is for informational purposes only. Forward Looking Statements Certain statements included in this Presentation that are not historical facts are forward-looking statements for purposes of the safe harbor provisions under the United States Private Securities Litigation Reform Act of 1995. Forward-looking statements generally are accompanied by words such as “believe,” “may,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “expect,” “should,” “would,” “plan,” “predict,” “potential,” “seem,” “seek,” “future,” “outlook” and similar expressions that predict or indicate future events or trends or that are not statements of historical matters. These forward-looking statements include, but are not limited to, statements by NewAmsterdam Pharma Company N.V. (“NewAmsterdam” or the “Company”) regarding estimates and forecasts of financial and performance metrics and projections of peak sales opportunities; the Company's business and strategic plans; expectations and timing related to the success, cost and timing of product development activities, including timing of initiation, completion and data readouts for clinical trials and the potential approval of the Company’s product candidate; the timing for enrolling patients; the timing and forums for announcing data; the size and growth potential of the markets for the Company’s product candidate; the therapeutic and curative potential of the Company’s product candidate; financing and other business milestones; the Company’s expected cash runway; scope and duration of patent protections and regulatory exclusivities; and the Company’s plans for commercialization. These statements are based on various assumptions, whether or not identified in this Presentation, and on the current expectations of the Company’s management and are not predictions of actual performance. These forward-looking statements are provided for illustrative purposes only and are not intended to serve as and must not be relied on as a guarantee, an assurance, a prediction, or a definitive statement of fact or probability. Actual events and circumstances are difficult or impossible to predict and may differ from assumptions. Many actual events and circumstances are beyond the control of the Company. These forward-looking statements are subject to a number of risks and uncertainties, including changes in domestic and foreign business, market, financial, political, and legal conditions; risks related to the approval of NewAmsterdam’s product candidate and the timing of expected regulatory and business milestones; whether topline, initial or preliminary results from a particular clinical trial will be predictive of the final results of that trial and whether results of early clinical trials will be indicative of the results of later clinical trials; the potential for varying interpretations of the Alzheimer’s disease sub-study and the results of other trials; the potential for varying interpretations relating to potential drivers of observed MACE reduction; ability to negotiate definitive contractual arrangements with potential customers; the impact of competitive product candidates; ability to obtain sufficient supply of materials; global economic and political conditions; the effects of competition on NewAmsterdam’s future business; and those factors discussed in documents filed by the Company with the SEC. Additional risks related to NewAmsterdam’s business include, but are not limited to: uncertainty regarding outcomes of the company’s ongoing clinical trials, particularly as they relate to regulatory review and potential approval for its product candidate; risks associated with the Company’s efforts to commercialize a product candidate; the Company’s ability to negotiate and enter into definitive agreements on favorable terms, if at all; the impact of competing product candidates on the Company’s business; risks and uncertainties relating to intellectual property and regulatory exclusivities; the Company’s ability to attract and retain qualified personnel; and the Company’s ability to continue to source the raw materials for its product candidate, together with the risks described in the Company’s filings made with the U.S. Securities and Exchange Commission from time to time. If any of these risks materialize or NewAmsterdam’s assumptions prove incorrect, actual results could differ materially from the results implied by these forward-looking statements. There may be additional risks that are presently unknown by the Company or that NewAmsterdam currently believes are immaterial that could also cause actual results to differ from those contained in the forward-looking statements. In addition, forward-looking statements reflect NewAmsterdam’s expectations, plans, or forecasts of future events and views as of the date of this Presentation and are qualified in their entirety by reference to the cautionary statements herein. NewAmsterdam anticipates that subsequent events and developments will cause the Company’s assessments to change. These forward-looking statements should not be relied upon as representing NewAmsterdam’s assessments as of any date subsequent to the date of this Presentation. Accordingly, undue reliance should not be placed upon the forward-looking statements. Neither NewAmsterdam nor any of its affiliates undertakes any obligation to update these forward-looking statements, except as required by law. Market Data Certain information contained in this Presentation relates to or is based on third-party studies, publications, data, surveys and NewAmsterdam’s own internal estimates and research. In addition, all of the market data included in this Presentation involves a number of assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. Finally, while NewAmsterdam believes its internal research is reliable, such research has not been verified by any independent source and NewAmsterdam cannot guarantee and makes no representation or warranty, express or implied, as to its accuracy and completeness. Trademarks This Presentation contains trademarks, service marks, trade names, and copyrights of NewAmsterdam and other companies, which are the property of their respective owners. The use or display of third parties’ trademarks, service marks, trade name or products in this Presentation is not intended to, and does not imply, a relationship with NewAmsterdam or an endorsement or sponsorship by or of NewAmsterdam. Solely for convenience, the trademarks, service marks and trade names referred to in this Presentation may appear with the TM or SM symbols, but such references are not intended to indicate, in any way, that NewAmsterdam will not assert, to the fullest extent permitted under applicable law, their rights or the right of the applicable licensor to these trademarks, service marks and trade names.
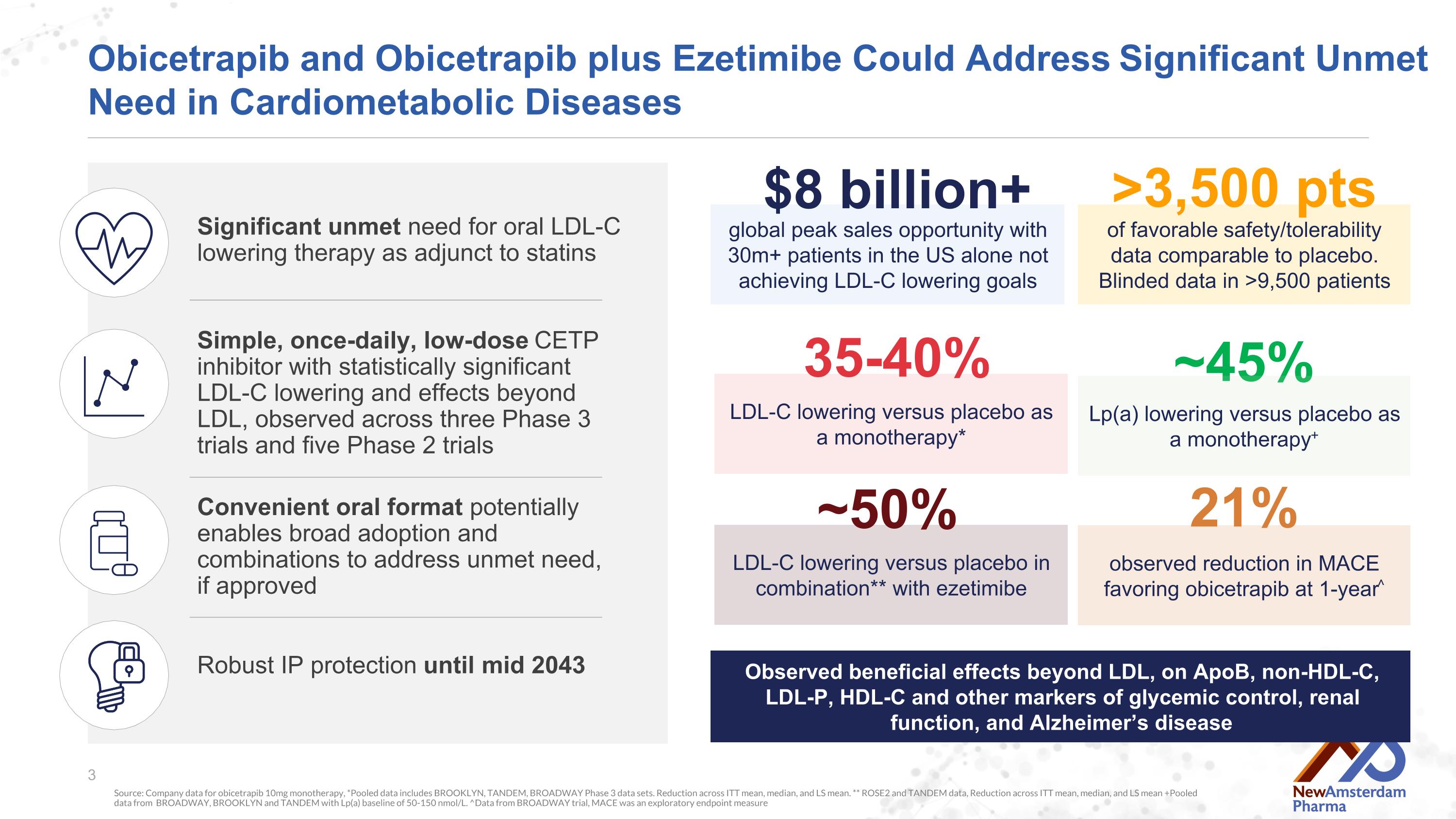
of favorable safety/tolerability data comparable to placebo. Blinded data in >9,500 patients Obicetrapib and Obicetrapib plus Ezetimibe Could Address Significant Unmet Need in Cardiometabolic Diseases Source: Company data for obicetrapib 10mg monotherapy, *Pooled data includes BROOKLYN, TANDEM, BROADWAY Phase 3 data sets. Reduction across ITT mean, median, and LS mean. ** ROSE2 and TANDEM data, Reduction across ITT mean, median, and LS mean +Pooled data from BROADWAY, BROOKLYN and TANDEM with Lp(a) baseline of 50-150 nmol/L. ^Data from BROADWAY trial, MACE was an exploratory endpoint measure Significant unmet need for oral LDL-C lowering therapy as adjunct to statins Simple, once-daily, low-dose CETP inhibitor with statistically significant LDL-C lowering and effects beyond LDL, observed across three Phase 3 trials and five Phase 2 trials Convenient oral format potentially enables broad adoption and combinations to address unmet need, if approved global peak sales opportunity with 30m+ patients in the US alone not achieving LDL-C lowering goals $8 billion+ ~45% Lp(a) lowering versus placebo as a monotherapy+ LDL-C lowering versus placebo as a monotherapy* 35-40% >3,500 pts Robust IP protection until mid 2043 Observed beneficial effects beyond LDL, on ApoB, non-HDL-C, LDL-P, HDL-C and other markers of glycemic control, renal function, and Alzheimer’s disease observed reduction in MACE favoring obicetrapib at 1-year^ 21% LDL-C lowering versus placebo in combination** with ezetimibe ~50%

Secured funding to support US commercial launch, if approved, with cash at 3Q25 of ~$756M Previous 18 Months Were a Time of Groundwork, Goals and Growth PublishedBROADWAY in NEJM Published TANDEM in The Lancet Announced Alzheimer’s topline Full AD data was presented at AAIC on July 30 Building world-class commercial and MSL functions Doubled in size (~100) with new hires and offices in Amsterdam, NL, Miami, FL, and Philadelphia metro area Completed and announced results for BROOKLYN, BROADWAY, and TANDEM Phase 3 studies 1H 2025 2024 Announced topline results for BROOKLYN Phase 3 trial Announced topline results for TANDEM Phase 3 trial Composition of matter IP granted Announced topline results for BROADWAY Phase 3 trial 2H 2025 EMA submission

Obicetrapib Could Address the ~30M Patients in US on Drug but not at Goal US Branded Lipid Lowering Market Potential key factors limiting penetration include product limitations and market access hurdles: Low prescriber enthusiasm for existing TPPs Payors restrict access Of the ~30M treated patients not at goal, ~18M were “far from goal” (greater than 20%) and 6M were not taking statins ASCVD=atherosclerotic cardiovascular disease; HeFH=heterozygous familial hypercholesterolemia; LDL-C=low-density lipoprotein-cholesterol; LLT=lipid lowering treatment.Source: Ortiz et al, Journal of Medical Economics, 2025, Vol 28, No 1, 1616-1625 ~5 million ASCVD not at goal of LDL-C <70 mg/dl ~8 million High risk ASCVD not at goal of LDL-C <55 mg/dl ~18 million Not at goal of LDL-C <100mg/dl ~72 million Adults in US diagnosed with hypercholesterolemia ~43 million Treated primary prevention patients ~19 million Treated secondary prevention patients ~10 million Diagnosed patients not treated with statin or LLT

Currently, Treatment of Hyperlipidemia is One-Dimensional LDL-C, low-density lipoprotein cholesterol; LLT, lipid lower therapy LDL-C ↓ Traditional LLTs

The Future of Hyperlipidemia Treatment Goes Beyond LDL-C Note: NODM = New onset diabetes mellitus ApoE4 LDL-P ↓ Small LDL-P ↓ NODM ↓ HDL-C ↑ ApoA1 ↑ LDL-C ↓ Non-HDL-C ↓ ApoB ↓ sdLDL-C ↓ Lp(a) ↓ Obicetrapib

* Source: Toth, Peter P. et al. Atherosclerosis. Volume 235, Issue 2, 585 - 591. ^Hoogeveen RC, Gaubatz JW, Sun W, Dodge RC, Crosby JR, Jiang J, Couper D, Virani SS, Kathiresan S, Boerwinkle E, Ballantyne CM. Small dense low-density lipoprotein-cholesterol concentrations predict risk for coronary heart disease: the Atherosclerosis Risk In Communities (ARIC) study. Arterioscler Thromb Vasc Biol. 2014 May;34(5):1069-77. doi: 10.1161/ATVBAHA.114.303284. Epub 2014 Feb 20. PMID: 24558110; PMCID: PMC3999643. * Ray K, et al. European Journal of Preventive Cardiology (2021) 28, 1279–1289. ^ Footnote: Prediabetes was defined as fasting plasma glucose values of 100 to 125 mg/dL or A1C values of 5.7% to 6.4%. Reference: National Diabetes Statistics Report | Diabetes | CDC** Tsimikas et al. J Am Coll Cardiol. 2022; 80: 934-946 ^^ Shapiro, M et al. Journal of Clinical Lipidology, Volume 19, Issue 1, 28 – 38 Aβ, amyloid β; AD, Alzheimer’s disease; Apo, apolipoprotein; CVD, cardiovascular disese; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; Lp(a), lipoprotein(a). 1. Abondio P et al. Genes (Basel). 2019;10(3):222. 2. Feringa FM et al. Front Aging Neurosci. 2021;13:690372. 3. Hottman 2017. 4. Jeong W et al. Mol Cells. 2019;42(11):739-746. 5. Williams et al. BMC 2020.c Approximately 43 million patients with hyperlipidemia exhibit elevated levels of LDL-P of > 1300 nmol/L* Prevalence is even higher in individuals with type 2 diabetes^ Presence of LDL-P is a significant independent risk factor for coronary heart disease (CHD), even when traditional lipid measures like LDL-C and triglycerides are within normal ranges^ The Future of Hyperlipidemia Treatment Goes Beyond LDL-C In the US, close to 75 million patients have an elevated Lp(a) score of > 100-125 nmol/L** ^^ An estimated 14.3 million of these patients also have hypercholesterolemia 27.9 million hyperlipidemia patients are estimated to have concomitant diabetes* In addition, there are an estimated 27.2 million^ individuals with hyperlipidemia that are pre-diabetic ApoE4 is the strongest genetic risk factor for late-onset AD and associated with a 22-45% increased risk of CVD1-5 Approximately 20 million patients with hyperlipidemia are at increased risk for AD Prevalence of ApoE4 in AD is up to 66%1-5 LDL Particles Lp(a) Diabetes ApoE4 Obicetrapib was observed to impact multiple risk factors, potentially leading to more comprehensive risk management across additional patient subpopulations

Tailwinds are Making Lipid Lowering Space More Conducive to Commercial Success 1 Very large opportunity – even small amounts of penetration can result in significant financial success We have seen LLT market growth each year for the last 5 years 2 Non-statin growth continues in the high double digits Shift towards more aggressive treatment using alternatives / adding to statins Observations 3 Guidelines* have shifted to recommend more intense intervention not only with statins, but with ezetimibe, PCSK9’s and bempedoic acid Governing bodies and KOL’s are prioritizing the importance of treating to goal 1. Grundy SM, et al. J Am Coll Cardiol. 2019;73(24):3168-3209. 2. Lloyd-Jones DM, et al. J Am Coll Cardiol. 2022;80(14):1366-1418 3. 2025 ACC/AHA/ACEP/NAEMSP/SCAI Guideline for the Management of Patients With Acute Coronary Syndromes: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines | Circulation Key Takeaways Repatha® growth continues to accelerate 10 years post launch with +45% Rx growth in nets sales for 2024 Commercial success is being demonstrated and is accelerating 5 4 Access to PCSK9’s has been increasing, and utilization controls have been decreasing Payers are willing to cover medications in LLT space if priced appropriately

Resulting in Continued Growth and Acceleration of Brand Use Source: Based on third party prescription data on file1. All Lipid Lowering therapies: Statins, Ezetimibe and combinations; PCSK9 and BPA; 2. Non-Statins : Ezetimibe and combinations; PCSK9 and BPA; 3. Branded: PCSK9 and BPA; 4. Merative Marketscan Claims Linked with Lab Data, 2019 - 2022, 12 months continuous data for each patient (6 months LB and 6 months LF from 1st observed statin treatment) Growth of 3.7% in 2024, which is the equivalent of 9 million additional TRx’s over 2023 5 years of consecutive growth Non-statins growing at high double digits Branded agents growing even faster 17.2% Growth 20.2% Growth 2.4% Growth 3.7% Growth 26.0% Growth 31.9% Growth 72 million1 Adults in US diagnosed with hypercholesterolemia ~10 million Diagnosed patients not treated with statin or LLT ~30 million Not at goal Over 40 million patients undertreated / not treated Non-statin2 TRx Branded3 TRx Lipid Lowering Therapy TRx1 Lipid Lowering Therapy TRx4 2 3 4 1
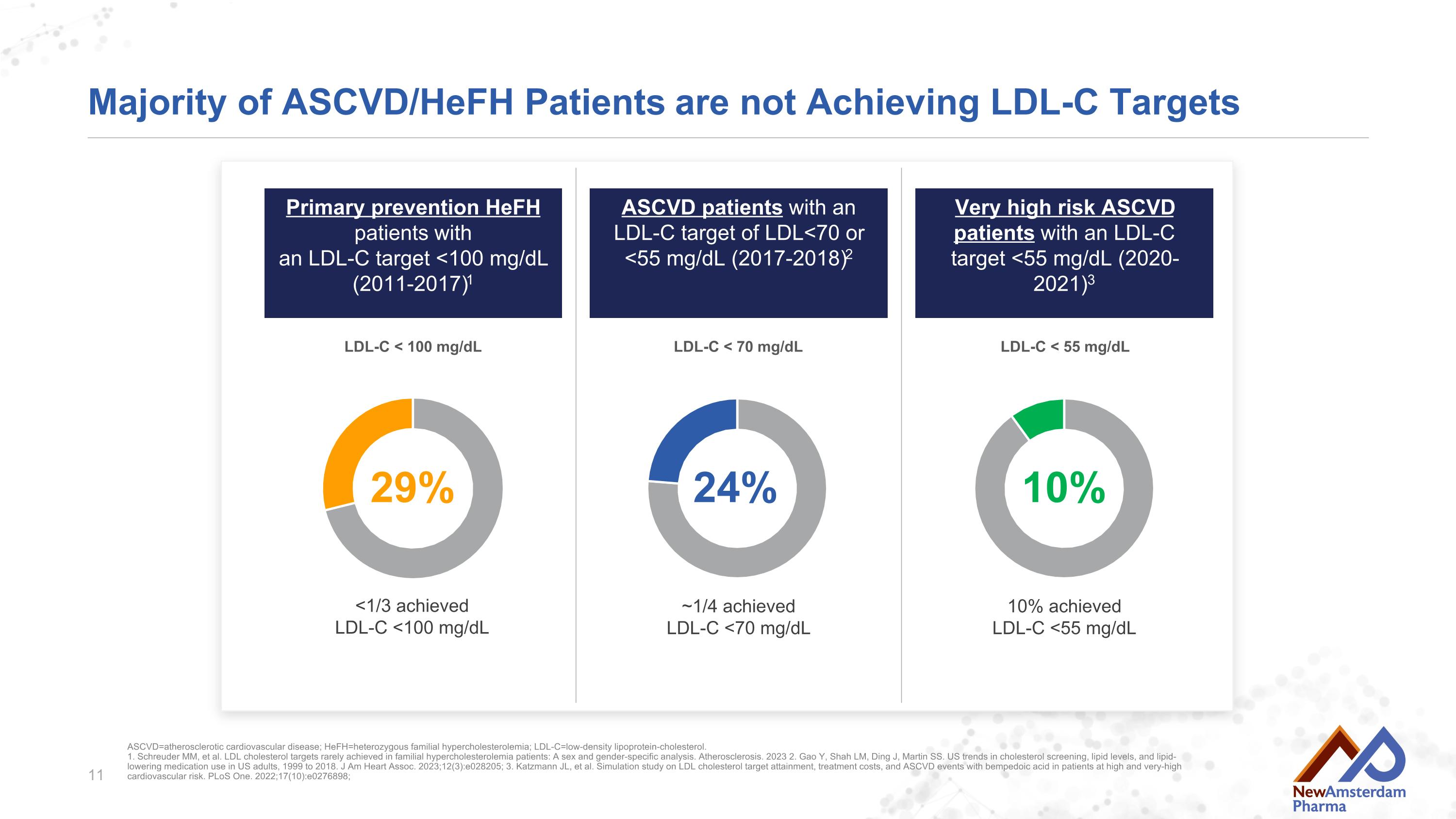
Majority of ASCVD/HeFH Patients are not Achieving LDL-C Targets 10% achievedLDL-C <55 mg/dL 10% 24% ~1/4 achievedLDL-C <70 mg/dL 29% <1/3 achievedLDL-C <100 mg/dL ASCVD=atherosclerotic cardiovascular disease; HeFH=heterozygous familial hypercholesterolemia; LDL-C=low-density lipoprotein-cholesterol.1. Schreuder MM, et al. LDL cholesterol targets rarely achieved in familial hypercholesterolemia patients: A sex and gender-specific analysis. Atherosclerosis. 2023 2. Gao Y, Shah LM, Ding J, Martin SS. US trends in cholesterol screening, lipid levels, and lipid-lowering medication use in US adults, 1999 to 2018. J Am Heart Assoc. 2023;12(3):e028205; 3. Katzmann JL, et al. Simulation study on LDL cholesterol target attainment, treatment costs, and ASCVD events with bempedoic acid in patients at high and very-high cardiovascular risk. PLoS One. 2022;17(10):e0276898; Primary prevention HeFH patients withan LDL-C target <100 mg/dL (2011-2017)1 ASCVD patients with an LDL-C target of LDL<70 or <55 mg/dL (2017-2018)2 Very high risk ASCVD patients with an LDL-C target <55 mg/dL (2020-2021)3 LDL-C < 70 mg/dL LDL-C < 100 mg/dL LDL-C < 55 mg/dL

CV Events Took an Alarming Turn Following Removal of LDL-C Guidelines in 2013 1. Martin SS, Aday AW, Allen NB, et al. 2025 Heart Disease and Stroke Statistics: A Report of US and Global Data From the American Heart Association. Circulation. doi: https://doi.org/10.1161/cir.0000000000001303 | 2. Yumin Gao, Y., et.al., US Trends in Cholesterol Screening, Lipid Levels, and Lipid-Lowering Medication Use in US Adults, 1999 to 2018, J Am Heart Assoc. 2023;12:e028205. DOI: 10.1161/JAHA.122.02820500 CVD Mortality Trends for US Males and Females, 1980 to 20221 LDL-C Goals Removed In 2013 ~75% of ASCVD patients are NOTat their risk-based LDL-C goal Trends in Prevalence of High LDL-C in US Adults, NHANES 1999-2018 with History of ASCVD2 100 75 50 25 0 LDL-C ≥70 mg/dL, % 1999-2000 2001-2002 2003-2004 2005-2006 2007-2008 2009-2010 2011-2012 2013-2014 2015-2016 2017-2018
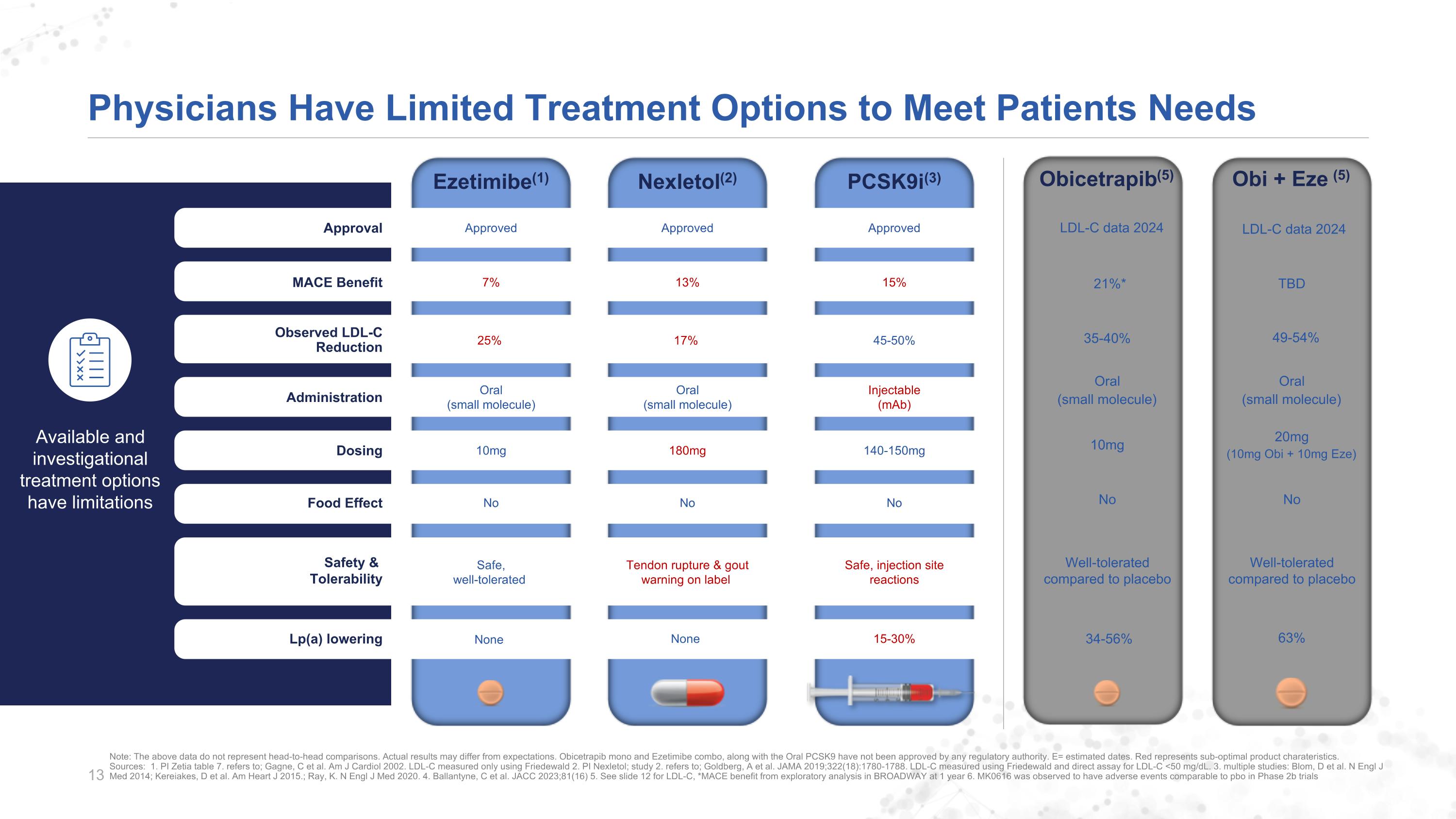
Physicians Have Limited Treatment Options to Meet Patients Needs Approved Approved Approved Approval 15% 13% 7% MACE Benefit 17% 25% Observed LDL-C Reduction Injectable (mAb) Oral (small molecule) Administration 140-150mg 180mg 10mg Dosing No No No Food Effect Safe, injection site reactions Tendon rupture & goutwarning on label Safe,well-tolerated Safety & Tolerability Available and investigational treatment optionshave limitations None Lp(a) lowering 45-50% 15-30% Oral (small molecule) None Note: The above data do not represent head-to-head comparisons. Actual results may differ from expectations. Obicetrapib mono and Ezetimibe combo, along with the Oral PCSK9 have not been approved by any regulatory authority. E= estimated dates. Red represents sub-optimal product charateristics.Sources: 1. PI Zetia table 7. refers to; Gagne, C et al. Am J Cardiol 2002. LDL-C measured only using Friedewald 2. PI Nexletol; study 2. refers to; Goldberg, A et al. JAMA 2019;322(18):1780-1788. LDL-C measured using Friedewald and direct assay for LDL-C <50 mg/dL. 3. multiple studies: Blom, D et al. N Engl J Med 2014; Kereiakes, D et al. Am Heart J 2015.; Ray, K. N Engl J Med 2020. 4. Ballantyne, C et al. JACC 2023;81(16) 5. See slide 12 for LDL-C, *MACE benefit from exploratory analysis in BROADWAY at 1 year 6. MK0616 was observed to have adverse events comparable to pbo in Phase 2b trials Obi + Eze (5) Obicetrapib(5) LDL-C data 2024 LDL-C data 2024 TBD 21%* 49-54% 35-40% Oral (small molecule) Oral (small molecule) 20mg (10mg Obi + 10mg Eze) 10mg No No Well-toleratedcompared to placebo 63% PCSK9i(3) Nexletol(2) Ezetimibe(1) Well-toleratedcompared to placebo 34-56%

Limited New Therapies on the Horizon Approval Current Phase Observed LDL-C Reduction Administration Dosing Food Effect Safety & Tolerability Available and investigational treatment optionshave limitations Lp(a) lowering Note: The above data do not represent head-to-head comparisons. Actual results may differ from expectations. Obicetrapib mono and Ezetimibe combo, along with the Oral PCSK9 have not been approved by any regulatory authority. E= estimated dates. Red represents sub-optimal product charateristics.Sources:. 1. Ballantyne, C et al. JACC 2023;81(16) 2. JACC 2025 3. See slide 12 for LDL-C 6. MK0616 was observed to have adverse events comparable to pbo in Phase 2b trials Obi + Eze (4) LDL-C data 2024 Phase 3 completed 49-54% Oral (small molecule) 20mg (10mg Obi + 10mg Eze) No 63% Well-toleratedcompared to placebo LDL-C data 2026E (CVOT data 2029E) Phase 3 ongoing 380mg (20mg API + 360mg SNAC) Yes (8hr fast & 30min wait) SNAC technology has previously been observed to have tolerability concerns(6) 50-59%(~20% with food) Oral (peptide) 20-25% MK-0616(1) TBD Phase 2b 1mg-30mg No Well-tolerated SAEs and case of AST/ALT > 5x ULN 35%-51% Oral (small molecule) AZD0780(2) Obicetrapib(3) LDL-C data 2024 Phase 3 completed Oral (small molecule) 10mg No Well-toleratedcompared to placebo 34-56% 35-40% 7%-20%

Obicetrapib Program Designed to Overcome Limitations of Prior CETP Inhibitors Note: The above trials and data do not represent head-to-head comparisons. Actual results may differ from expectations. Sources: 1. Barter et al. NEJM.2007; 2. Schwartz et al. NEJM.2012; 3. Lincoff et al. NEJM.2017 4. Bowman et al. NEJM.2017 5. Company Data 6. Per PUC, if available Torcetrapib(1) Dalcetrapib(2) Evacetrapib(3) Anacetrapib(4) Obicetrapib(5) Observed LDL-C reduction(6) 25% 7% 11-21% 17% 35-40% CETP inhibition 35% 30% 65% 80% 97% Dosing 60mg 600mg 100mg 100mg 10mg Blood pressure increase Yes No No No No Aldosterone increase Yes No No No No Lp(a) lowering unknown unknown 20-25% 20-25% 34-56% ApoB lowering 15% None 15-20% 18% 22%-24% OUTCOMES STUDIES Name ILLUMINATE Dal-OUTCOMES ACCELERATE REVEAL PREVAIL Patients 15,067 15,871 12,092 30,449 9,541 Baseline LDL-C (mg/dl) 79.7 76.4 81.1 61 103 LDL-C reduction (mg/dl) 20 NS 25 11 TBD Median follow-up 18 mo 31 mo 26 mo 49 mo 42 mo (expected) Result (HR) 1.25 1.04 1.01 0.91 TBD Explanation Off target tox No LDL-C benefit Short follow-up but mortality benefit (HR 0.84) Low baseline and LDL reduction TBD

Consistent LDL-C Reduction Observed Across Our Phase 2 and Phase 3 Trials, Independent of Background Statin and/or Multiple Lipid Lowering Therapy Usage Obicetrapib 10 mg monotherapy Note: ROSE at week 8, ROSE2, BROOKLYN, BROADWAY, and TANDEM as of week 12. High intensity statin use 100% in Phase 2 trials and over 65% in Phase 3 trials. Obicetrapib plus ezetimibe Monotherapy reductions of 35-40% Combo reductions of 50-60%
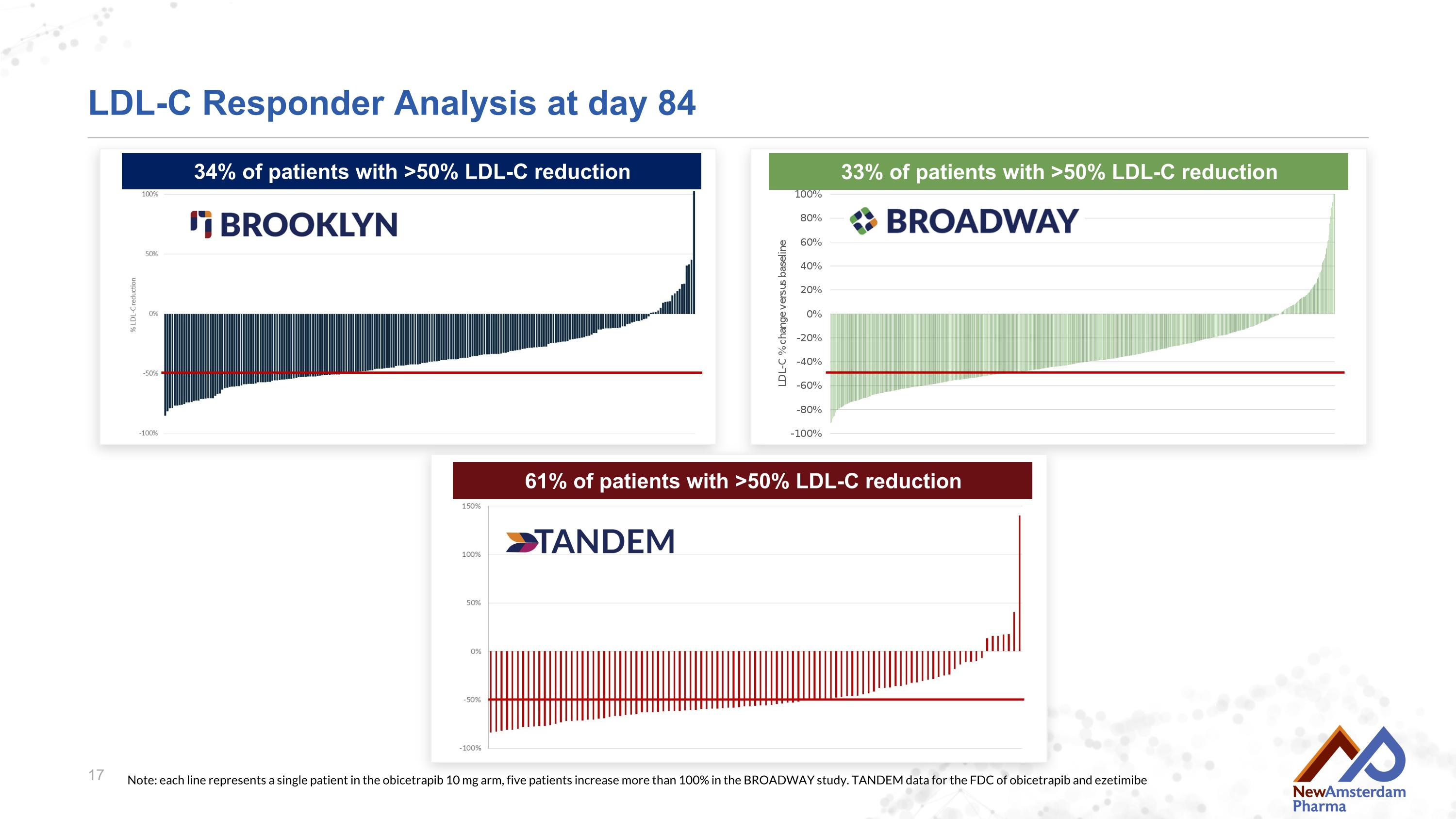
LDL-C Responder Analysis at day 84 34% of patients with >50% LDL-C reduction Note: each line represents a single patient in the obicetrapib 10 mg arm, five patients increase more than 100% in the BROADWAY study. TANDEM data for the FDC of obicetrapib and ezetimibe 33% of patients with >50% LDL-C reduction 61% of patients with >50% LDL-C reduction

Greater Proportion of Patients in Obicetrapib Arm Achieved LDL-C Goal % of patients achieving LDL-C thresholds at Day 84 % of patients achieving LDL-C thresholds at Day 84

Over 70% of Patients on Obicetrapib+Ezetimbe Achieved Less than <55 mg/dL % of patients achieving LDL-C thresholds at Day 84
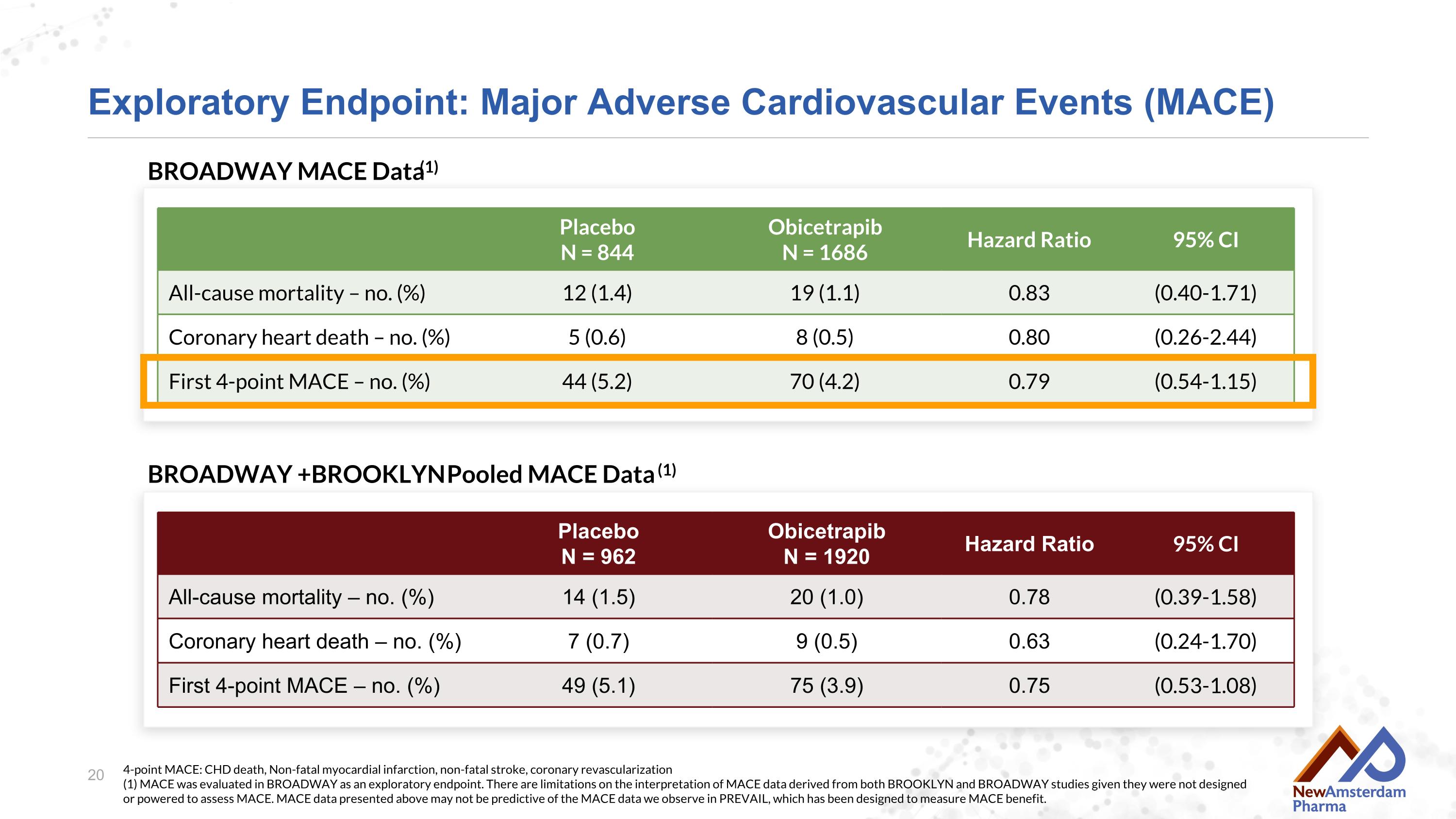
Exploratory Endpoint: Major Adverse Cardiovascular Events (MACE) Placebo N = 844 Obicetrapib N = 1686 Hazard Ratio 95% CI All-cause mortality – no. (%) 12 (1.4) 19 (1.1) 0.83 (0.40-1.71) Coronary heart death – no. (%) 5 (0.6) 8 (0.5) 0.80 (0.26-2.44) First 4-point MACE – no. (%) 44 (5.2) 70 (4.2) 0.79 (0.54-1.15) 4-point MACE: CHD death, Non-fatal myocardial infarction, non-fatal stroke, coronary revascularization(1) MACE was evaluated in BROADWAY as an exploratory endpoint. There are limitations on the interpretation of MACE data derived from both BROOKLYN and BROADWAY studies given they were not designed or powered to assess MACE. MACE data presented above may not be predictive of the MACE data we observe in PREVAIL, which has been designed to measure MACE benefit. Placebo N = 962 Obicetrapib N = 1920 Hazard Ratio 95% CI All-cause mortality – no. (%) 14 (1.5) 20 (1.0) 0.78 (0.39-1.58) Coronary heart death – no. (%) 7 (0.7) 9 (0.5) 0.63 (0.24-1.70) First 4-point MACE – no. (%) 49 (5.1) 75 (3.9) 0.75 (0.53-1.08) BROADWAY MACE Data(1) BROADWAY + BROOKLYN Pooled MACE Data (1)

Kaplan-Meier Curve Separates at Day 200 Driven by Attenuation of Events in the Obicetrapib Arm 4-point MACE: CHD death, Non-fatal myocardial infarction, non-fatal stroke, coronary revascularization (1) While not powered to detect a MACE benefit, we observed positive MACE data as part of our review of the exploratory endpoint. BROADWAY was not powered to measure MACE benefit and the MACE data presented below may not be predictive of the MACE data we observe in PREVAIL, which has been designed to measure MACE benefit. Kaplan Meier Curve Landmark Analysis: Censored at 6 months (1)

Explaining BROADWAY’s Observed 21% MACE Reduction Based on the CTT line, the expectation for a 33% reduction in LDL-C from placebo would be a 9% MACE reduction, after 12 months LDL-P reduction potentially provides 6-7% additional MACE reduction Lp(a) reduction potentially provides an additional 5% MACE reduction These elements may not be additive as they are correlated to some extent. In this scenario, there is room for additional reduction to be driving incremental gains LDL-C Alone LDL-P 9% 21% 1% Lp(a) 5% 6% Potential Drivers ObservedMACE Additional Reduction Obicetrapib’s Observed MACE Reduction Methods of Estimating MACE Reduction Note: Actual results may differ from hypothetical calculation. Source: Cholesterol Treatment Trialists Collaboration; Folse et al. Athero 2014, Quseda et al, Atherosclerosis 35 (2023) 165-177; Berman, A, Biery, D, Besser, S. et al. Lipoprotein(a) and Major Adverse Cardiovascular Events in Patients With or Without Baseline Atherosclerotic Cardiovascular Disease. JACC. 2024 Mar, 83 (9) 873–886; Data on file ** MACE includes CHD death, myocardial infarction, ischemic stroke and coronary revascularization in adults.

Overview of Events of Special Interest Placebo N= 843 n (%) Obicetrapib N= 1685 n (%) AST or ALT > 3 x ULN 8 (0.9) 10 (0.6) Bilirubin > 2 x ULN 4 (0.5) 2 (0.1) CK > 5 x ULN 3 (0.4) 5 (0.3) NODM or worsening of glycemic control 338 (40.1) 592 (35.1) - AE indicating new/worse type 1 or 2 diabetes 30 (3.6) 58 (3.4) - Initiation of diabetes medication 104 (12.3) 186 (11.0) - HbA1c ≥ 6.5% (where baseline HbA1c < 6.5%) 55 (6.5) 84 (5.0) - Two consecutive glucose values > 126 mg/dL 248 (29.4) 459 (27.2) - HbA1c increase from baseline > 0.5% 133 (15.8) 234 (13.9) - Worsening glycemic control 199 (23.6) 350 (20.8) Renal function worsening 77 (9.1) 127 (7.5) - eGFR < 30 mL/min/1.73m2 13 (1.5) 13 (0.8) - 25% decrease in eGFR from baseline: 70 (8.3) 115 (6.8) - Increase of Serum Creatinine ≥ 0.3 mg/dL from baseline 61 (7.2) 91 (5.4) Macular degeneration 0 (0.0) 1 (0.1) (p = 0.015)

BROADWAY Designed to be a Predictor of PREVAIL Key Inclusion Criteria 13-months Placebo Obicetrapib 10 mg (2:1 randomization) N = 2532 13-months 1º endpoint – week 12 Baseline Lipids Baseline Lipid Modifying Therapy Any statin: 91% High intensity statin: 65% Ezetimibe: 26% PCSK9i: 4% SGLT2i: 11% GLP-1: 6% Placebo Obicetrapib 10 mg (1:1 randomization) N = 9541 54-months LDL-C endpoint Source: Data on file Blinded mean Median Blinded mean Median LDL-C≥ 100 mg/dL Maximally tolerated lipid lowering therapy ASCVD or HeFH LDL-C ≥55 mg/dL w/risk factors, or Key Inclusion Criteria LDL-C≥ 100 mg/dL Maximally tolerated lipid lowering therapy ASCVD LDL-C ≥55 mg/dL w/risk factors, or Baseline Lipids Baseline Lipid Modifying Therapy Any statin: >90% High intensity statin: 70% Ezetimibe: 23% PCSK9i: 2% SGLT2i: 14% GLP-1: 6%

PREVAIL Designed to Apply Lessons Learned from Previous CVOTs to Reduce Risk and Demonstrate Obicetrapib’s Full Benefit Greater LDL-C lowering activity anticipated Targeting higher baseline LDL-C patients Higher absolute LDL-C reduction expected to lead to greater MACE benefit Longer duration of follow up Targeting higher-risk patient population Maximizes opportunity for MACE reduction Differentiated secondary endpoints Potentially enhanced commercial profile vs. other LDL-C lowering agents Primary endpoint selection Coronary heart disease (CHD) death, non-fatal MI, fatal and non-fatal ischemic stroke, coronary revascularization

2022 2023 2024 2025E 2026E 1H 2H 1H 2H 1H 2H 1H 2H 1H 2H Phase 3 BROADWAY TrialLipid Mono Study (HeFH or ASCVD; LDL-C ≥ 55 mg/dL; n=2,530) Phase 3 BROOKLYN Trial Lipid Mono Study (HeFH; LDL-C ≥ 70 mg/dL; n=354) Phase 3 CVOT PREVAIL Trial (ASCVD or HeFH; LDL-C ≥ 55 mg/dL; n=9,541) Phase 3 RUBENS Trial Type 2 Diabetes / Metabolic Syndrome Study (T2D/Met Syndrome; LDL ≥ 100 mg/dL; n=300) Phase 2 VINCENT Trial Lp(a) change in combo w/ Repatha (Lp(a) ≥125 nmol/L, n=39; Lp(a) ≥50 nmol/L and <125 nmol/L, n=30) Phase 3 TANDEM Trial (LDL-C ≥ 70 mg/dL; n=407) Phase 3 REMBRANDT Trial Coronary plaque volume study (LDL-C ≥ 70 mg/dL; non–calcified coronary plaque > 75mm3; n=300) * “PREVAIL will continue until the last participant has been followed for a minimum of 2.5 years and the target number of MACE events have occurred. As a result, the earliest the trial could conclude based on the minimum follow-up period is the end of 2026. However, we will continue the trial until the target number of MACE events occur, which could likely require the trial to continue beyond this point. Note: Other than as noted, the pipeline represents trials that are currently ongoing. 2025, 2026 and beyond are projections and subject to inherent limitations. EMA regulatory filings, monotherapy and FDC products, in 2H2025. Actual results may differ from expectations. Obicetrapib + Ezetimibe Fixed-Dose Combination (obicetrapib 10mg + ezetimibe 10mg) Enrollment complete Initiation Ph3 (estimated) CVOT * Ph2 (estimated) LEGEND TANDEM Ph3 Obicetrapib Monotherapy (obicetrapib 10mg) BROADWAY Ph3 BROOKLYN Ph3 PREVAIL CVOT VINCENT Ph2 Multiple Data Readouts to Drive Future Momentum REMBRANDT Ph 3 RUBENS Ph3 EMA Regulatory filings

Obicetrapib andAlzheimer’s Disease

CETPi Bears Therapeutic Potential for Both Multi-infarct Dementia and Alzheimer’s Disease Alzheimer's disease Healthy brain Alzheimer’s disease results in the generalized degeneration of the brain, with over 50M people affected WW and an economic burden of >$1 trillion (2) There is emerging evidence of a vascular component in multi-infarct dementia, implying that LDL-lowering would help reduce atherosclerosis in the brain (1) Multi-infarct dementia (ischemic stroke) Cortical shrinking Enlarged ventricles Shrinking of hippocampus Post-stroke necrosis Source: 1. M Kivipelto et al, Stroke, 2022: 53(2) 2. Alzheimer’s Disease International.

+ Elevated Cholesterol Levels in the Brain Precede Formation of Amyloid-Beta (Aβ) Plaques During early development, oligodendrocytes synthesize large quantities of cholesterol for myelination In adults, when myelination is complete, glial cells and, to a lesser extent, neurons account for the steady-state production of cholesterol The brain uses a unique mechanism for cholesterol recycling and redistribution Involves an ApoE-mediated lipoprotein pathway unique to the CNS whereby cholesterol is turned over Excess oxysterols are ultimately delivered to the liver for secretion into the bile Cholesterol itself does not exit the brain but instead is converted to a metabolite, 24S-hydroxycholesterol 24S-hydroxycholesterol is a proxy for excess cholesterol that will be measured in our phase 2a clinical trial 20% cholesterol APOE, apolipoprotein E; CNS, central nervous system. Mahley RW. Arterioscler Thromb Vasc Biol. 2016;36(7):1305-1315. Amyloid precursor is cleaved,releasing Aβ peptide Aβ peptide leavesmembrane and aggregates Aβ plaque is formed Aβ peptide Cholesterol Cell membrane Aβ plaque formation inside AD brain cells The brain is the most cholesterol-rich organ; it contains only 2% body mass but has 20% of the body’s cholesterol

ApoE4 is a Risk Factor for Both CVD and Alzheimer’s Disease1-5 % of population with this genotype APOE2/E3 11% APOE3/E3 61% APOE2/E2 0.5% APOE4/E2 = 2% APOE4/E3 = 24% APOE4/E4 = 2% Cholesterol transportand clearance Lipid accumulation in brain Aβ aggregation Total cholesterol HDL-C receptor + binding N-term C-term Lipid binding (VLDL) R158 R61 R112 C255 Disruption of neuronal synaptic function Altered astrocytic glucose metabolism Promotion of microglial inflammatory responses Impaired oligodendrocyte remyelination Aβ, amyloid β; AD, Alzheimer’s disease; Apo, apolipoprotein; CVD, cardiovascular disese; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; Lp(a), lipoprotein(a). 1. Abondio P et al. Genes (Basel). 2019;10(3):222. 2. Feringa FM et al. Front Aging Neurosci. 2021;13:690372. 3. Hottman 2017. 4. Jeong W et al. Mol Cells. 2019;42(11):739-746. 5. Williams et al. BMC 2020.c 66% APOE4 carriers exhibit Prevalence of APOE4 in AD ApoE4-induced cholesterol dysregulation has cell-specific effects in the brain that all contribute to AD pathogenesis >25% of population at increased risk Elevated risk is mediated by the profoundeffect of APOE4 on lipid metabolism CVD APOE4 carriers exhibit APOE4 Risk for CVD by 22-45% LDL-C HDL-C ApoC Lp(a)

CETP Loss-of-Function (LoF) Genotype May be Associated with Slower Memory Decline and Lower AD Risk Source :JAMA, January 13, 2010—Vol 303, No. 2 2 LoF CETP genes 1 LoF CETP gene Normal CETP CETP’s potential involvement in CNS cholesterol homeostasis is supported by genomic data CETP LoF may be associated with lower CETP activity & a corresponding increase in HDL levels

+ HDL correlation - LDL correlation NOR score (%) p=0.015 r2=0.5837 p=0.0365 r2=-0.4042 Plasma HDL (mmol/L) 3 2 1 0 0 20 40 60 80 100 NOR score (%) 2.5 1.5 1.0 0 0 20 40 60 80 100 0.5 2.0 Plasma LDL (mmol/L) Control diet 1% cholesterol diet WT CETP CETP transgenic mice have lower plasma HDL-C levels and increased brain cholesterol levels1 1% cholesterol diet Control diet P<0.05 P<0.05 r2=0.5837 P=0.015 r2=-0.4042 P=0.0365 + HDL correlation - LDL correlation A separate study demonstrated that CETP inhibitors can enter the brain4 Novel object discrimination ratio (%) 100 60 40 80 10 0 WT P=0.0376 50% Cognitive impairment CETP APP APP/CETP P=0.0435 Vehicle CETP inhibitor (Evacetrapib) Increased CETP demonstrates 22% increased brain cholesterol levels on high cholesterol diet CETP Knock-in Mice Have Increased Brain Cholesterol Levels and CETPi Observed to Rescue Cognition in Preclinical Models of CETP-induced AD1-3 CETP inhibition with evacetrapib led to cognitive improvement 2,3 Plasma Lipoproteins Were Correlated With NOR Score Positive correlation observed between NOR score and HDL quantification in CETP and APP/CETP-expressing female mice Negative correlation observed between NOR score and LDL quantification in CETP and APP/CETP-expressing female mice AD, Alzheimer’s disease; APP, amyloid precursor protein; CETP, cholesterol ester transfer protein; CETPi, CETP inhibitor; HDL, high-density lipoprotein; HDL-C, high-density lipoprotein cholesterol; LDL, low-density lipoprotein; NOR, novel object recognition; WT, wild-type. 1. Oestereich F et al. J Lipid Res. 2022;63(9):100260. 2. Phénix J et al. Alzheimer’s Dement. 2021;17(suppl3):e051134. 3. Phénix J et al. Poster presented at: Alzheimer’s Association International Conference; July 26-30, 2021; Denver, USA. 4. Phénix J et al. Front Pharmacol. 2023;14:1171937.

Safety FU Safety FU Obicetrapib on Top of Max Tolerated Lipid-Modifying Therapy: Alzheimer’s Biomarker Sub-study Placebo (n=800) Objective: To evaluate the effect of obicetrapib on biomarkers related to Alzheimer’s Disease compared to placebo over 12-months Visit: Days: 1 -28 to -1 6 270 2 1 4 84 3 30 Labs, PK Safety Labs, Safety Labs, PK Safety Eligibility: Labs, Safety 7 365 Labs, PK Safety Study design: Randomized, double-blind, placebo-controlled Main Inclusion Criteria: CV disease < 3 months HoFH Uncontrolled hypertension Main Exclusion Criteria: Biomarker Endpoints: p-tau217 Ratio of Aβ42 to Aβ40 (Aβ42/Aβ40) GFAP NFL p-tau181 . Obicetrapib 10mg (n=1600) Patients (n=2400) HeFH and or ASCVD ≥ 18 years Baseline LDL-C: 98 mg/dL 5 180 Labs, PK Safety Baseline 8 +35 Vitals, conmeds, AEs assessed Endpoint Have a fasting serum LDL-C at Screening (Visit 1) as follows: Have a fasting serum LDL-C ≥ 55 mg/dL (≥ 1.4 mmol/L) to <100 mg/dL (<2.6 mmol/L) OR non-HDL-C ≥ 85 mg/dL (≥ 2.2 mmol/L) to <130 mg/dL (<3.4 mmol/L) with at least 1 risk enhancer OR Have a fasting serum LDL-C ≥ 100 mg/dL (≥ 2.6 mmol/L) OR non-HDL-C ≥ 130 mg/dL (≥ 3.4 mmol/L). Note: Davidson MH et al. 2025. Manuscript in preparation.

Significant Reduction in p-tau217 Progression Over 12 Months with Greater Reductions as ApoE4 Risk Factors Increase Full Analysis Set* (n=1535) 2.84% benefit between obicetrapib and placebo (P=0.025) ApoE4 Carriers* (n=367) 5.74% treatment benefit in ApoE4 carriers (P=0.022) *Results reflect adjustment for mean-centered baseline p-tau217 and mean-centered age. Participants with known ApoE status (based on phenotypic analysis), baseline p-tau217 above the lower limit of quantification (LLQ), and available end-of-study p-tau217 were included in the current analyses. Aβ, amyloid beta; ApoE, apolipoprotein E; p-tau, phosphorylated tau. ApoE4 carriers defined as ApoE3/E4 or ApoE4/E4, based on phenotypic analysis Davidson MH et al. 2025. Journal of Prevention of Alzheimer's Disease Obicetrapib ApoE4 Homozygotes* (n=29) 20.5% treatment benefit in ApoE4 homozygotes (P=0.010)

Intellectual Property

Comprehensive Patent Portfolio with Composition of Matter IP into 2043 Note: Regulatory approval and exclusivity dates are illustrative and shown for information purposes only; actual results may differ. Filled colors = granted patents & dotted lines = pending patents; one patent only to be selected for SPC/PTE; an earlier US approval leads to earlier regulatory expiry & shorter PTE; *including pediatric extension 6m; actual results may differ from expectations. (1) US 12,006,305. (2). Low dose/ species selection patents US 10,653,692, US 11,013,742, US 11,642,344; US 12,186,315 statin combo patent US 10,300,059 2042 2041 2040 2039 2038 2037 2036 2035 2034 2033 2032 2031 2030 2029 2028 2027 2026 2025 2024 2023 Genus 8/27 (U.S.); 4/25 (EU) Species Selection(2) US 2/34 8/39* PTE EU 2/34 8/39* SPC 2/34 COM(1) EU 7/43 Proprietary form (COM) patent family US 7/43 Regulatory exclusivity EU Data exclusivity + market exclusivity (8 + 2 years) EU approval 6 mo. pedi. US NCE exclusivity (5 years) 30 mo. Stay US approval 6 mo. pedi.

Thank You




































