UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): October 30, 2024
Biomea Fusion, Inc.
(Exact name of registrant as specified in its charter)
| Delaware | 001-40335 | 82-2520134 | ||
| (State or other jurisdiction of incorporation) |
(Commission File Number) |
(IRS Employer Identification No.) |
| 900 Middlefield Road, 4th Floor Redwood City, California |
94063 | |||
| (Address of principal executive offices) | (Zip Code) | |||
Registrant’s telephone number, including area code: 650 980-9099
Not Applicable
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| ☐ | Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ☐ | Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ☐ | Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ☐ | Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class |
Trading |
Name of each exchange on which registered |
||
| Common Stock, $0.0001 par value | BMEA | The NASDAQ Global Select Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company ☒
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
| Item 7.01 | Regulation FD Disclosure |
Biomea Fusion, Inc. (the “Company”) hosted a conference call and live webcast to discuss the preclinical data on October 30, 2024 at 4:30 p.m. E.T. The Company has made available a slide presentation to accompany the call, a copy of which is furnished herewith as Exhibit 99.1 to this Current Report on Form 8-K.
The information in this Item 7.01, including Exhibit 99.1 attached hereto, is intended to be furnished and shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Securities Act of 1933, as amended (the “Securities Act”), or the Exchange Act, except as expressly set forth by specific reference in such filing.
| Item 8.01 | Other Events. |
On October 30, 2024, the Company issued a press release titled, “Biomea Fusion Presents Preclinical Data Showing Icovamenib (BMF-219) Enhanced Effectiveness of GLP-1-Based Therapies and Introduces BMF-650, a Next-Generation, Oral Small-Molecule GLP-1 Receptor Agonist Candidate.” A copy of the press release is attached to this Current Report on Form 8-K as Exhibit 99.2 and is incorporated herein by reference.
Forward Looking Statements
Statements made or incorporated by reference in this Current Report on Form 8-K may include statements which are not historical facts and are considered forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended (the “Securities Act”), and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). These statements may be identified by words such as “aims,” “anticipates,” “believes,” “could,” “estimates,” “expects,” “forecasts,” “goal,” “intends,” “may,” “plans,” “possible,” “potential,” “seeks,” “will,” and variations of these words or similar expressions that are intended to identify forward-looking statements. Any such statements in this press release that are not statements of historical fact, including statements regarding the clinical and therapeutic potential of the Company’s product candidates and development programs, and their potential relative to approved products marketed by third parties; the Company’s research, development and regulatory plans, the progress of the Company’s ongoing and upcoming clinical trials, the progress and timing of pre-clinical development in the Company’s programs; the anticipated enrollment of patients and availability of data from the Company’s clinical trials, anticipated milestones, and the timing of such events, may be deemed to be forward-looking statements. The Company intends these forward-looking statements to be covered by the safe harbor provisions for forward-looking statements contained in Section 27A of the Securities Act and Section 21E of the Exchange Act and is making this statement for purposes of complying with those safe harbor provisions.
Any forward-looking statements made or incorporated by reference in this Current Report on Form 8-K are based on the Company’s current expectations, estimates and projections only as of the date of this Current Report on Form 8-K are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in or implied by such forward-looking statements, including the risk that preliminary or interim results of preclinical studies or clinical trials may not be predictive of future or final results in connection with future clinical trials and the risk that the Company may encounter delays in preclinical or clinical development, patient enrollment and in the initiation, conduct and completion of its ongoing and planned clinical trials and other research and development activities. These risks concerning the Company’s business and operations are described in additional detail in its periodic filings with the U.S. Securities and Exchange Commission (the “SEC”), including its most recent periodic report filed with the SEC and subsequent filings thereafter. The Company explicitly disclaims any obligation to update any forward-looking statements except to the extent required by law.
| Item 9.01. | Financial Statements and Exhibits. |
(d) Exhibits.
| Exhibit Number |
Description | |
| 99.1 | Corporate Presentation titled “Preclinical Data Combining icovamenib with a GLP-1-Based Therapy & Biomea’s Oral GLP-1 RA Candidate BMF-650,” dated October 30, 2024, furnished herewith. | |
| 99.2 | Press release titled “Biomea Fusion Presents Preclinical Data Showing Icovamenib (BMF-219) Enhanced Effectiveness of GLP-1-Based Therapies and Introduces BMF-650, a Next-Generation, Oral Small-Molecule GLP-1 Receptor Agonist Candidate” on October 30, 2024. | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) | |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
| Biomea Fusion Inc. | ||||||
| Date: October 31, 2024 | By: | /s/ Thomas Butler |
||||
| Thomas Butler | ||||||
| Principal Executive Officer | ||||||

BIOMEA cONFERENCE Call Preclinical Data Combining icovamenib with a GLP-1-Based Therapy & Biomea’s Oral GLP-1 RA Candidate BMF-650 October 30, 2024 Exhibit 99.1

Legal Disclaimer & Forward-Looking Statements Certain statements in this presentation and the accompanying oral commentary are forward-looking statements. These statements relate to future events or the future business and financial performance of Biomea Fusion, Inc. (the “Company”) and involve known and unknown risks, uncertainties and other factors that may cause the actual results, levels of activity, performance or achievements of the Company or its industry to be materially different from those expressed or implied by any forward-looking statements. In some cases, forward-looking statements can be identified by terminology such as “may,” “will,” “could,” “would,” “should,” “expect,” “plan,” “anticipate,” “intend,” “believe,” “estimate,” “predict,” “potential” or other comparable terminology. All statements other than statements of historical fact could be deemed forward-looking, including any projections of financial information or profitability, the initiation, timing and results of pending or future preclinical studies and clinical trials, the actual or potential actions of the FDA, the status and timing of ongoing research, development and corporate partnering activities, any statements about historical results that may suggest trends for the Company's business; any statements of the plans, strategies, and objectives of management for future operations; any statements of expectation or belief regarding future events, potential markets or market size, or technology developments, unfavorable global economic conditions, including inflationary pressures, market volatility, acts of war and civil and political unrest, and other factors affecting the Company's financial condition or operations. The Company has based these forward-looking statements on its current expectations, assumptions, estimates and projections. While the Company believes these expectations, assumptions, estimates and projections are reasonable, such forward-looking statements are only predictions and involve known and unknown risks and uncertainties, many of which are beyond the Company's control. For a discussion of these and other risks and uncertainties, and other important factors, any of which could cause our actual results to differ from those contained in the forward-looking statements, see the section entitled "Risk Factors" in our most recent periodic report filed with the Securities and Exchange Commission, as well as discussions of potential risks, uncertainties, and other important factors in our other subsequent filings with the Securities and Exchange Commission. The forward-looking statements in this presentation are made only as of the date hereof. Except as required by law, the Company assumes no obligation and does not intend to update these forward-looking statements or to conform these statements to actual results or to changes in the Company's expectations. This presentation also contains estimates and other statistical data made by independent parties and by us relating to market size and growth and other data about our industry. This data involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such estimates. In addition, projections, assumptions, and estimates of our future performance and the future performance of the markets in which we operate are necessarily subject to a high degree of uncertainty and risk. Page Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib

Agenda Page Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib Ramses Erdtmann Chief Operating Officer & Co-Founder of Biomea Thomas Butler Chief Executive Officer, Chairman of the Board & Co-Founder of Biomea Juan Frias, M.D. Chief Medical Director of Biomea Mini Balakrishnan, Ph.D. Executive Director of Biology, Biomea Thorsten Kirschberg, Ph.D. Executive Vice President of Chemistry, Biomea Thomas Butler Chief Executive Officer, Chairman of the Board & Co-Founder of Biomea Introduction Executive Summary GLP-1 Based Therapy Background & Overview Icovamenib in combination with GLP-1 Based Therapy BMF-650 Preclinical Summary Closing Remarks
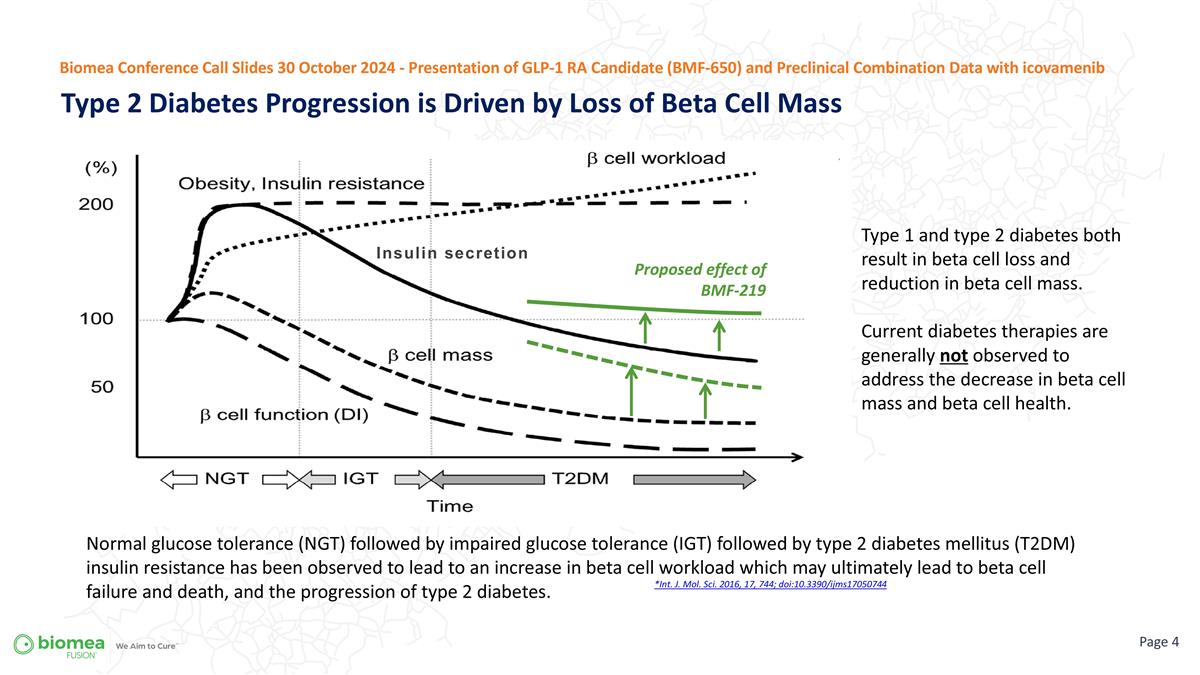
*Int. J. Mol. Sci. 2016, 17, 744; doi:10.3390/ijms17050744 Type 1 and type 2 diabetes both result in beta cell loss and reduction in beta cell mass. Current diabetes therapies are generally not observed to address the decrease in beta cell mass and beta cell health. Normal glucose tolerance (NGT) followed by impaired glucose tolerance (IGT) followed by type 2 diabetes mellitus (T2DM) insulin resistance has been observed to lead to an increase in beta cell workload which may ultimately lead to beta cell failure and death, and the progression of type 2 diabetes. Type 2 Diabetes Progression is Driven by Loss of Beta Cell Mass Page Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib Proposed effect of BMF-219 Insulin secretion
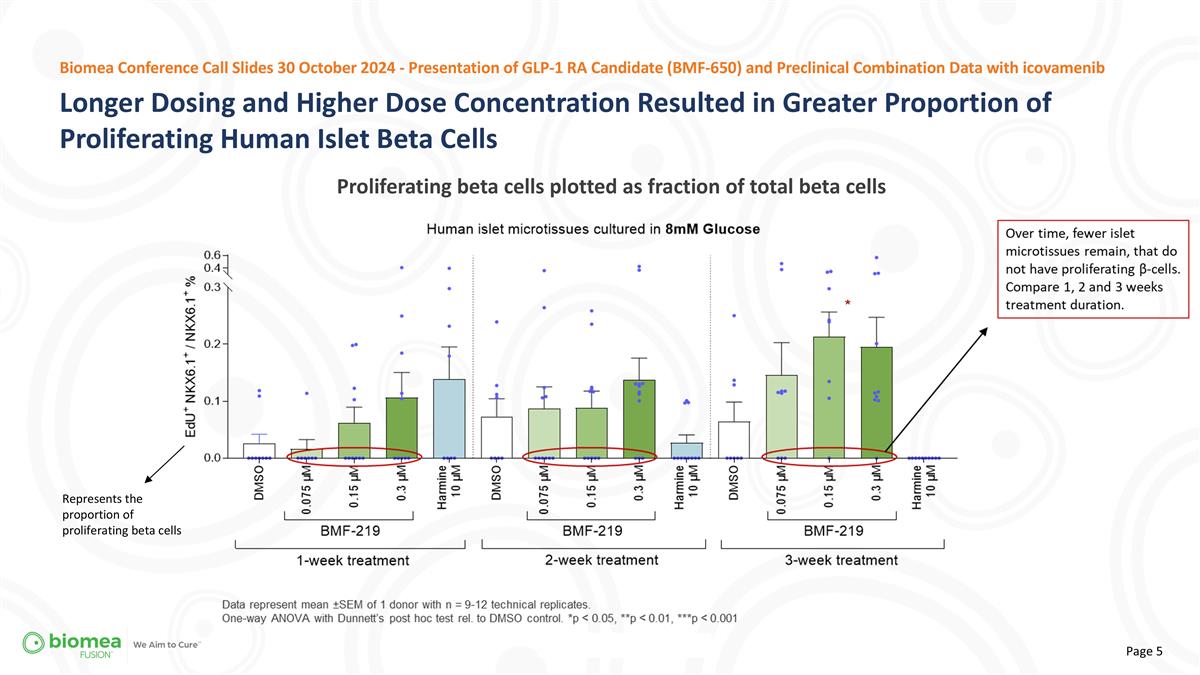
Longer Dosing and Higher Dose Concentration Resulted in Greater Proportion of Proliferating Human Islet Beta Cells Proliferating beta cells plotted as fraction of total beta cells Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib Represents the proportion of proliferating beta cells
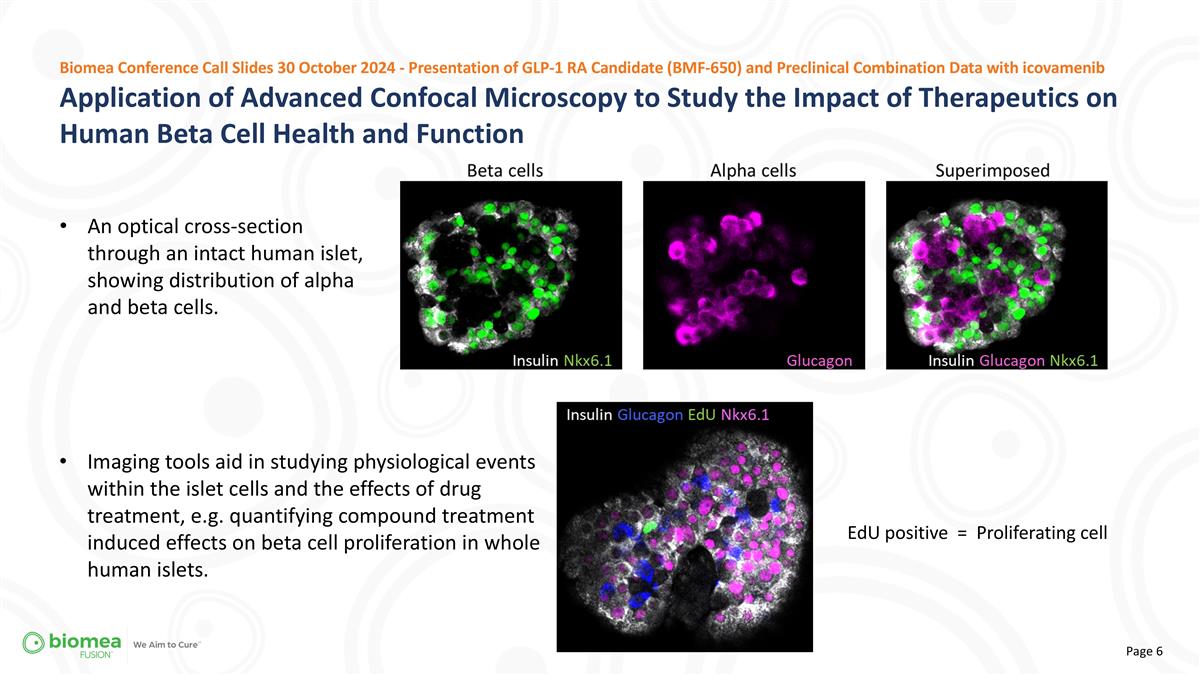
Application of Advanced Confocal Microscopy to Study the Impact of Therapeutics on Human Beta Cell Health and Function Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib An optical cross-section through an intact human islet, showing distribution of alpha and beta cells. Imaging tools aid in studying physiological events within the islet cells and the effects of drug treatment, e.g. quantifying compound treatment induced effects on beta cell proliferation in whole human islets. EdU positive = Proliferating cell

Icovamenib (BMF-219) in Combination with a GLP-1 Based Therapy – An Overview of Pre-Clinical Findings
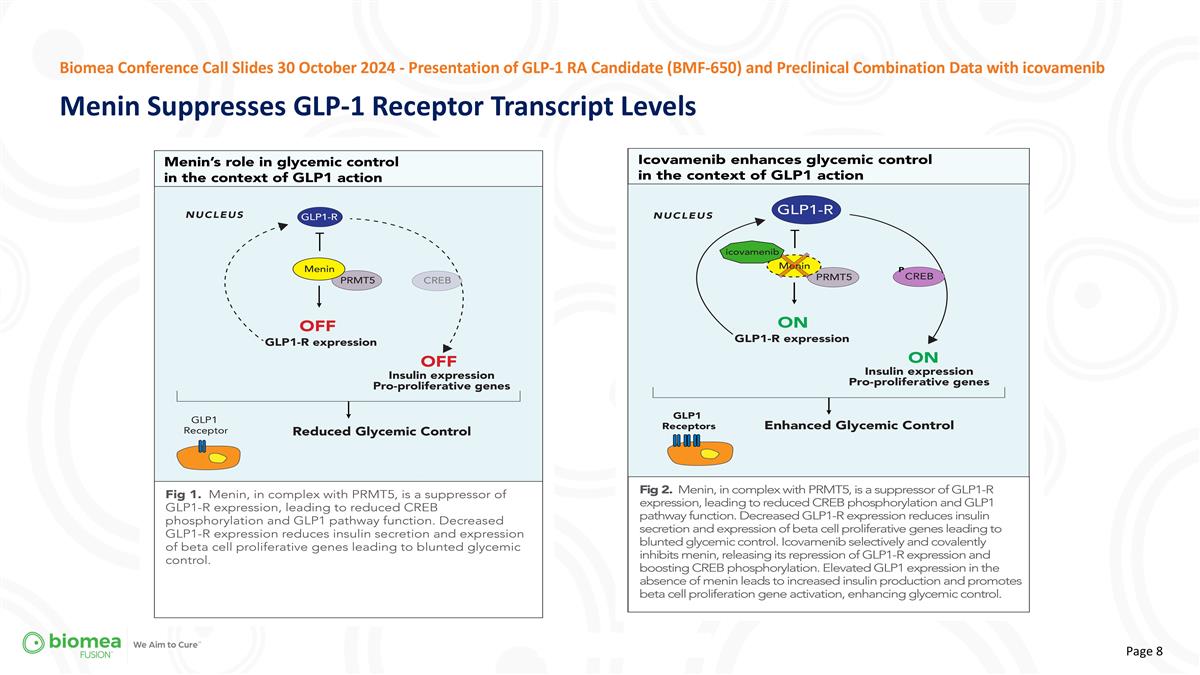
Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib Menin Suppresses GLP-1 Receptor Transcript Levels
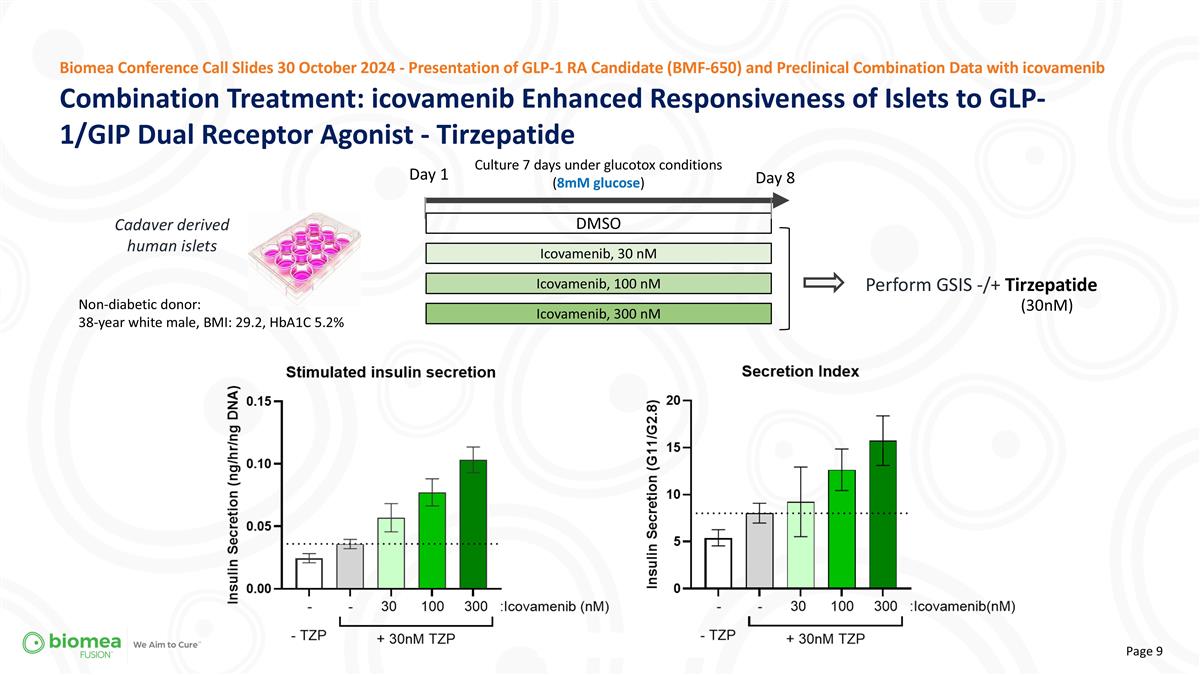
Cadaver derived human islets Combination Treatment: icovamenib Enhanced Responsiveness of Islets to GLP-1/GIP Dual Receptor Agonist - Tirzepatide Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib Day 1 Perform GSIS -/+ Tirzepatide Culture 7 days under glucotox conditions (8mM glucose) DMSO Icovamenib, 30 nM Icovamenib, 100 nM Day 8 Icovamenib, 300 nM Non-diabetic donor: 38-year white male, BMI: 29.2, HbA1C 5.2% (30nM)
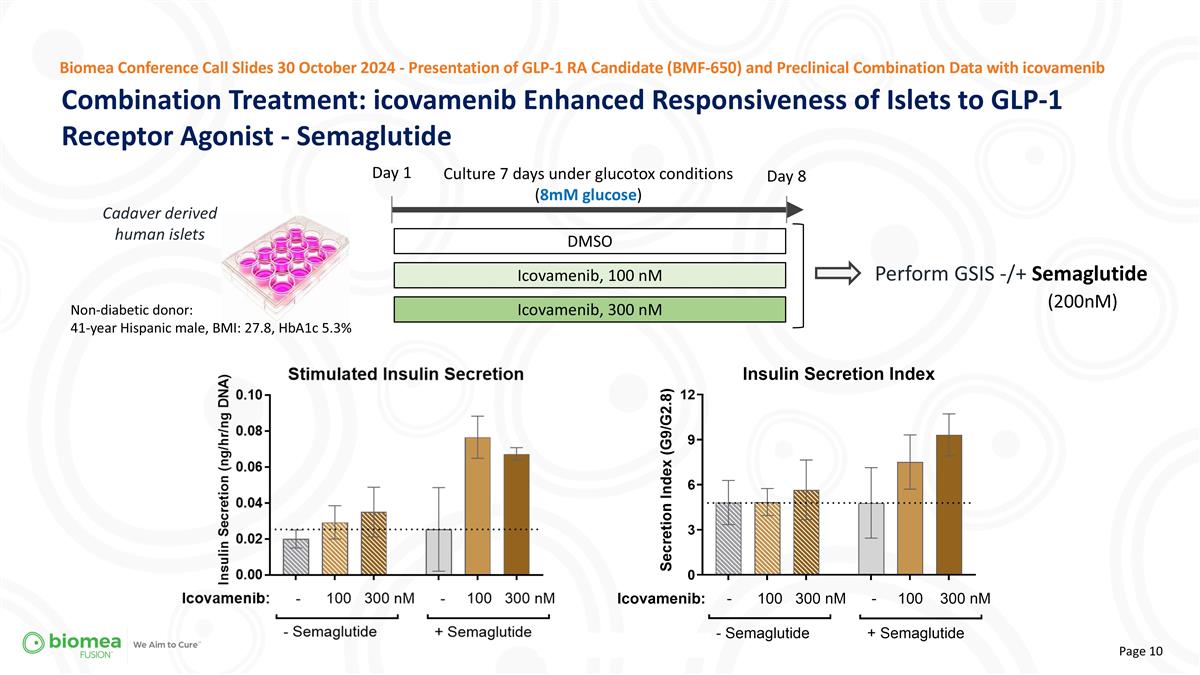
Combination Treatment: icovamenib Enhanced Responsiveness of Islets to GLP-1 Receptor Agonist - Semaglutide Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib Day 1 Perform GSIS -/+ Semaglutide (200nM) Culture 7 days under glucotox conditions (8mM glucose) DMSO Icovamenib, 100 nM Icovamenib, 300 nM Day 8 Non-diabetic donor: 41-year Hispanic male, BMI: 27.8, HbA1c 5.3% Cadaver derived human islets
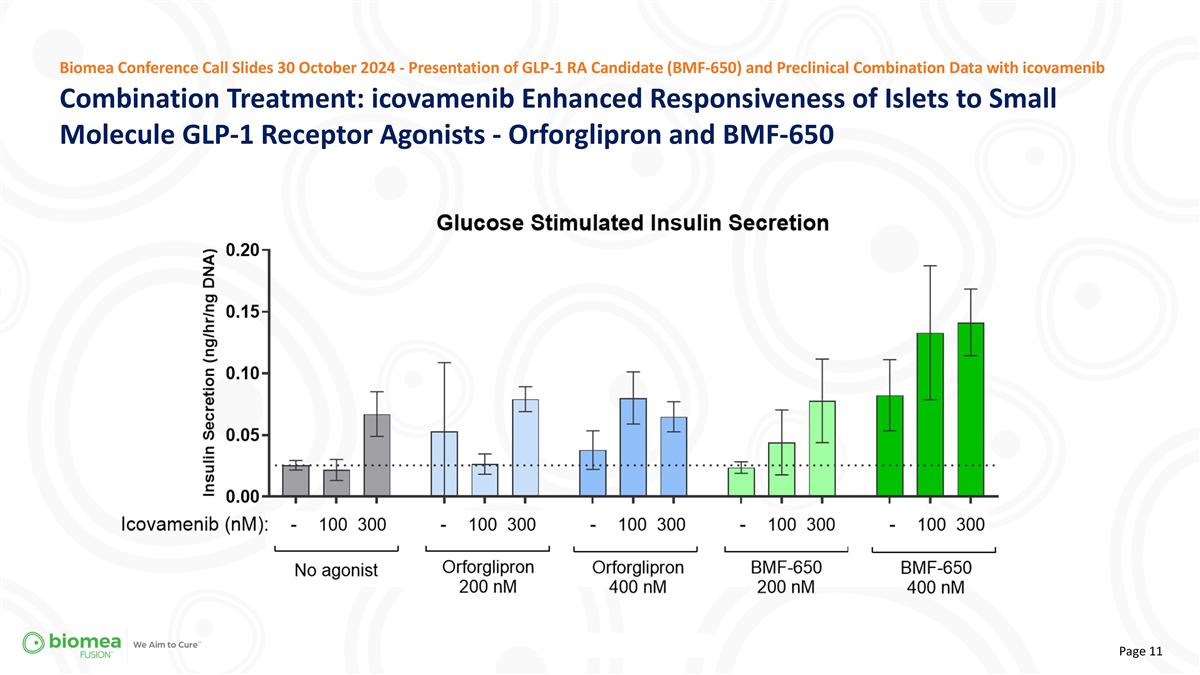
Combination Treatment: icovamenib Enhanced Responsiveness of Islets to Small Molecule GLP-1 Receptor Agonists - Orforglipron and BMF-650 Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib

Potential benefits of using icovamenib together with approved GLP-1 based therapeutics: Lower dosing requirements of existing GLP-1 based therapy Improved tolerability Improved adherence Improved therapeutic window Improved initial responsiveness - Greater patient persistence and treatment results with GLP-1 based therapeutics Next steps in Biomea’s clinical development: - COVALENT-211 (icovamenib in combination with GLP-1 based therapeutics) Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib New Treatment Potential in Diabetes and for Obesity Combining Covalent Binding Menin Inhibitor icovamenib with GLP-1 Based Therapy

BMF-650 – An Investigational, Next-Generation, Oral Small Molecule GLP-1 Receptor Agonist

Attributes of Biomea’s GLP-1 Receptor Agonist Development Candidate: Less PK variability Greater bioavailability Greater protein binding Less side effects Why a greater “Therapeutic Window”? Only 3 of 10 patients in the real-world setting are staying on a GLP-1 based therapy after 12 months Drive for a Greater “Therapeutic Window” with our Next-Generation GLP-1 Receptor Agonist – BMF-650 Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib
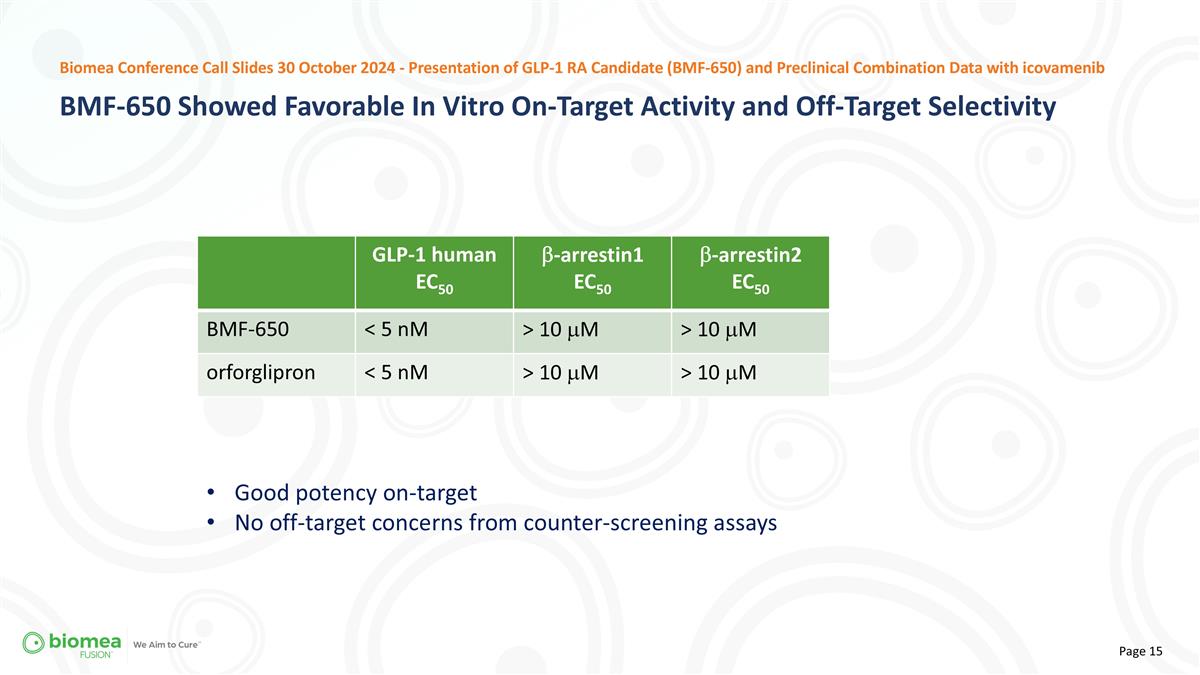
GLP-1 human EC50 b-arrestin1 EC50 b-arrestin2 EC50 BMF-650 < 5 nM > 10 mM > 10 mM orforglipron < 5 nM > 10 mM > 10 mM Good potency on-target No off-target concerns from counter-screening assays BMF-650 Showed Favorable In Vitro On-Target Activity and Off-Target Selectivity Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib
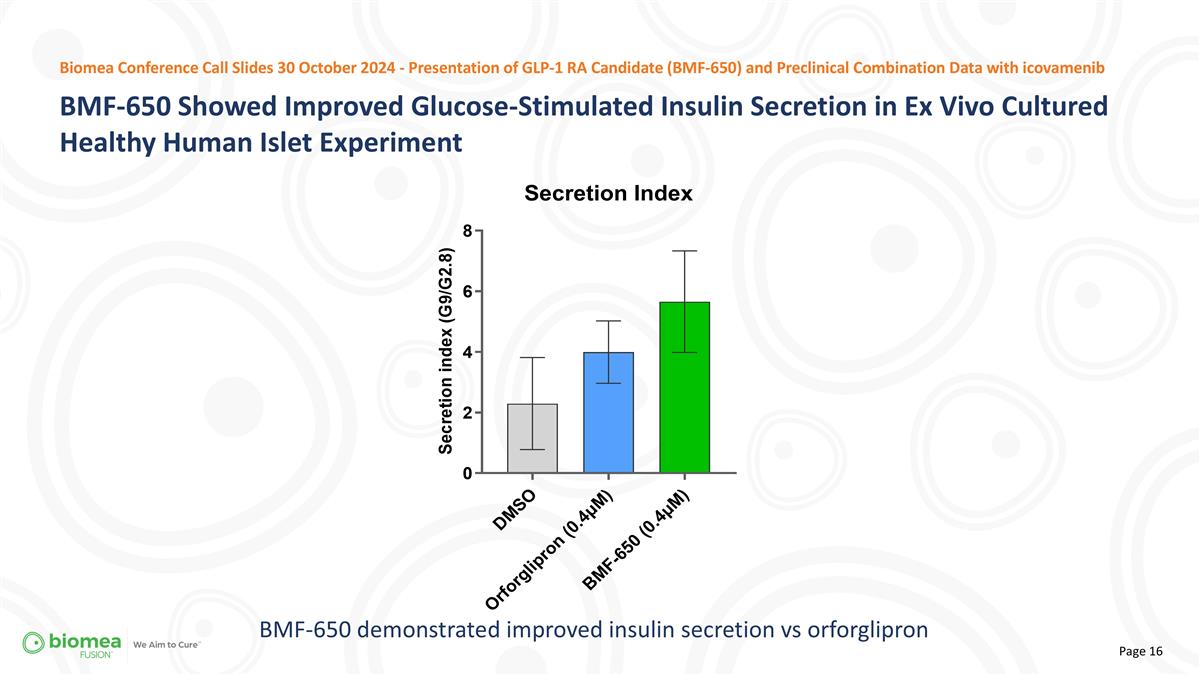
BMF-650 demonstrated improved insulin secretion vs orforglipron BMF-650 Showed Improved Glucose-Stimulated Insulin Secretion in Ex Vivo Cultured Healthy Human Islet Experiment Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib
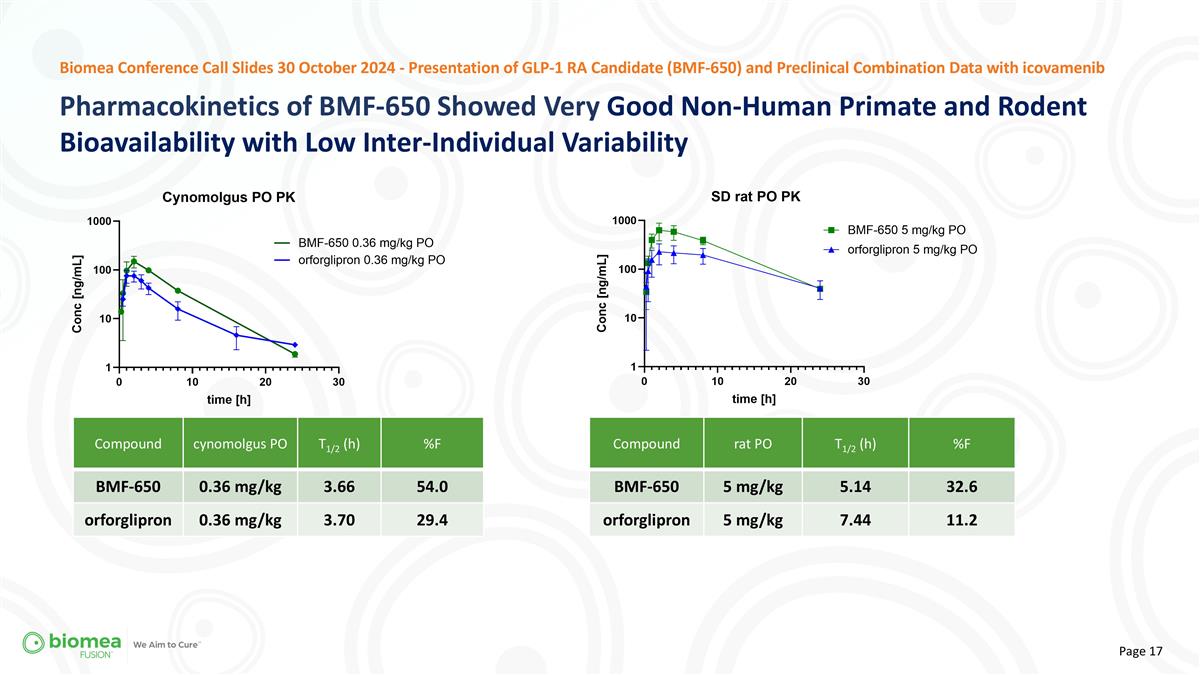
Pharmacokinetics of BMF-650 Showed Very Good Non-Human Primate and Rodent Bioavailability with Low Inter-Individual Variability Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib Compound cynomolgus PO T1/2 (h) %F BMF-650 0.36 mg/kg 3.66 54.0 orforglipron 0.36 mg/kg 3.70 29.4 Compound rat PO T1/2 (h) %F BMF-650 5 mg/kg 5.14 32.6 orforglipron 5 mg/kg 7.44 11.2

Projected Human Dose for BMF-650 Similar Among the Oral Agents Dosages Used in Cynomolgus Monkeys are Species Dependent and Specific to Properties of Compounds Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib Orforglipron Eli Lilly BMF-650 Biomea GSBR-1290 Structure Therapeutics CT-996 Roche (Carmot) Doses tested in cynomolgus monkeys to address food intake HD LD: 0.1 & 0.05 mg/kg 2 and 10 mg/kg 2 to 10 mg/kg 3 to 30 mg/kg Clinical titration target 45 mg 100 mg (projected) 120 mg 120 mg

Period of Anesthesia of Cynomolgus Monkeys Compound: Bolus and continuous infusion to steady-state level Measure glucose and Insulin for 40 min Glucose IV Cynomolgus monkeys (n=4) were anesthetized. Compounds were administered via IV route. Glucose was infused. Blood Glucose and Insulin levels were measured during the following 40 minutes window. Study designed to capture both glucose lowering and insulin release properties Primary Evaluation of Preclinical Activity Set up of Intravenous Glucose Tolerance Test in Cynomolgus Monkeys Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib
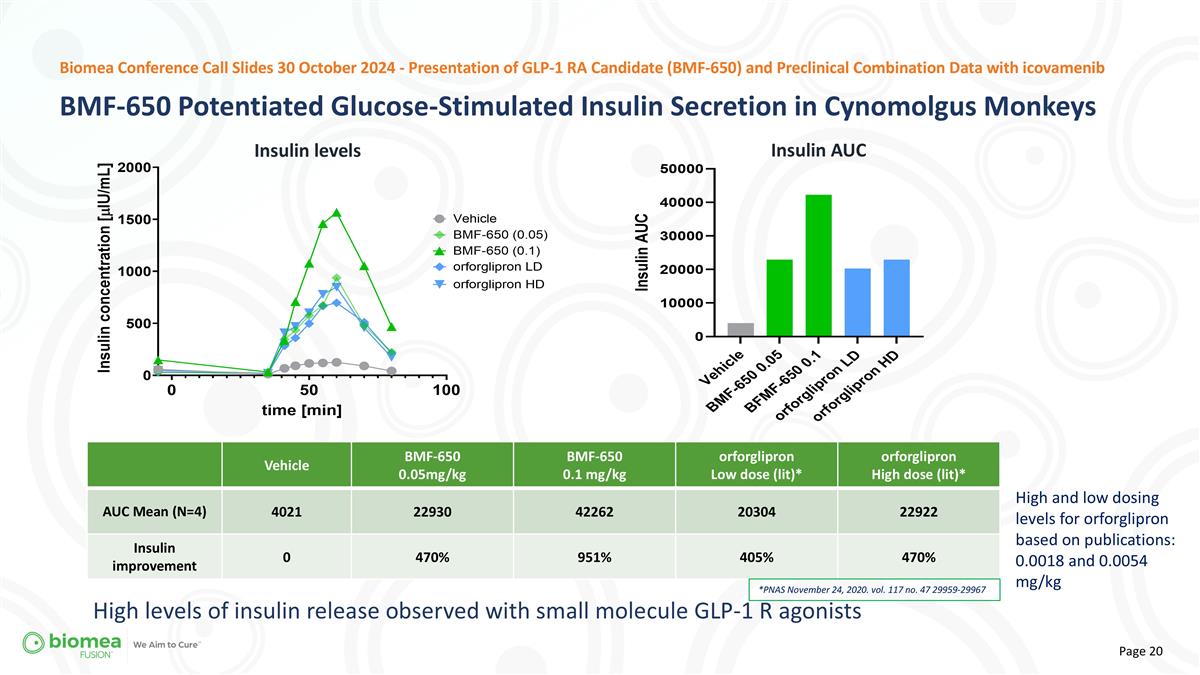
Vehicle BMF-650 0.05mg/kg BMF-650 0.1 mg/kg orforglipron Low dose (lit)* orforglipron High dose (lit)* AUC Mean (N=4) 4021 22930 42262 20304 22922 Insulin improvement 0 470% 951% 405% 470% *PNAS November 24, 2020. vol. 117 no. 47 29959-29967 High and low dosing levels for orforglipron based on publications: 0.0018 and 0.0054 mg/kg High levels of insulin release observed with small molecule GLP-1 R agonists BMF-650 Potentiated Glucose-Stimulated Insulin Secretion in Cynomolgus Monkeys Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib Insulin AUC Insulin levels
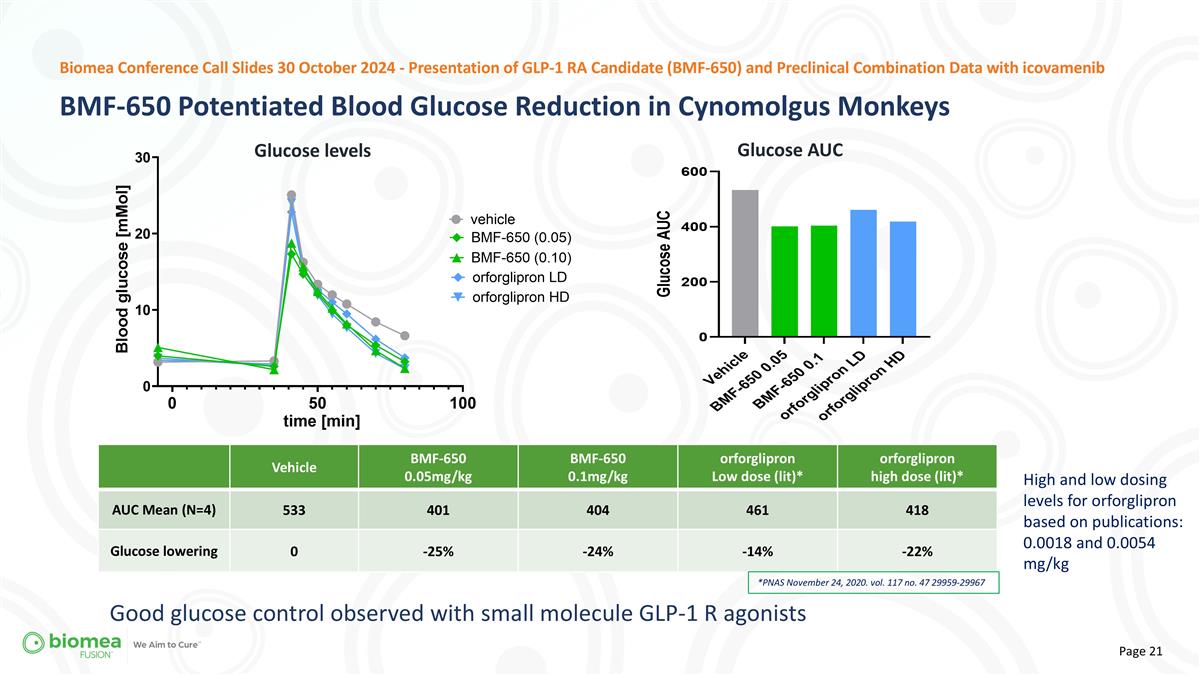
Vehicle BMF-650 0.05mg/kg BMF-650 0.1mg/kg orforglipron Low dose (lit)* orforglipron high dose (lit)* AUC Mean (N=4) 533 401 404 461 418 Glucose lowering 0 -25% -24% -14% -22% *PNAS November 24, 2020. vol. 117 no. 47 29959-29967 High and low dosing levels for orforglipron based on publications: 0.0018 and 0.0054 mg/kg Good glucose control observed with small molecule GLP-1 R agonists BMF-650 Potentiated Blood Glucose Reduction in Cynomolgus Monkeys Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib Glucose levels Glucose AUC

24 hours period PO 3 hours 90 min Rest of day No food Food Fasted monkeys (n=4) were dosed orally (PO) with compound. Food was presented starting at 3 hours post dose. Recorded food intake for time window 3 hours to 4.5 hours and the whole day. Design of study as multiday of active drug and multiday of washout Primary Evaluation of Preclinical Activity Set up of Appetite Suppression Test in Cynomolgus Monkeys Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib Study designed to capture both peak and daily food intake reduction.
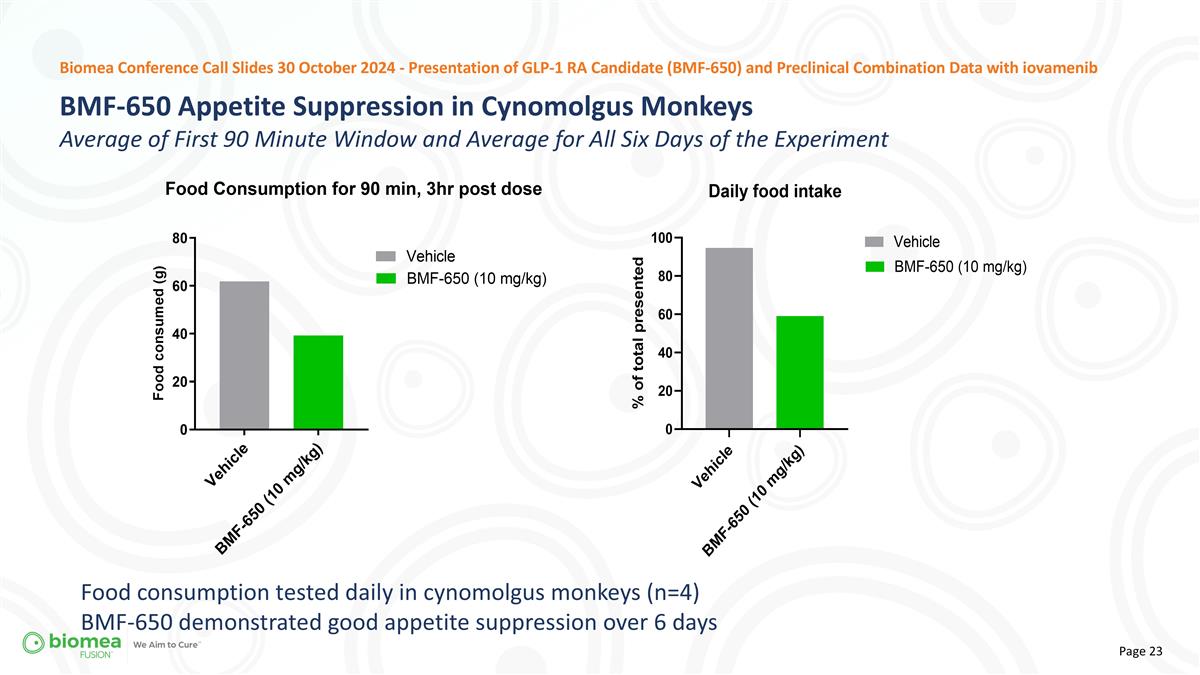
Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with iovamenib Food consumption tested daily in cynomolgus monkeys (n=4) BMF-650 demonstrated good appetite suppression over 6 days BMF-650 Appetite Suppression in Cynomolgus Monkeys Average of First 90 Minute Window and Average for All Six Days of the Experiment
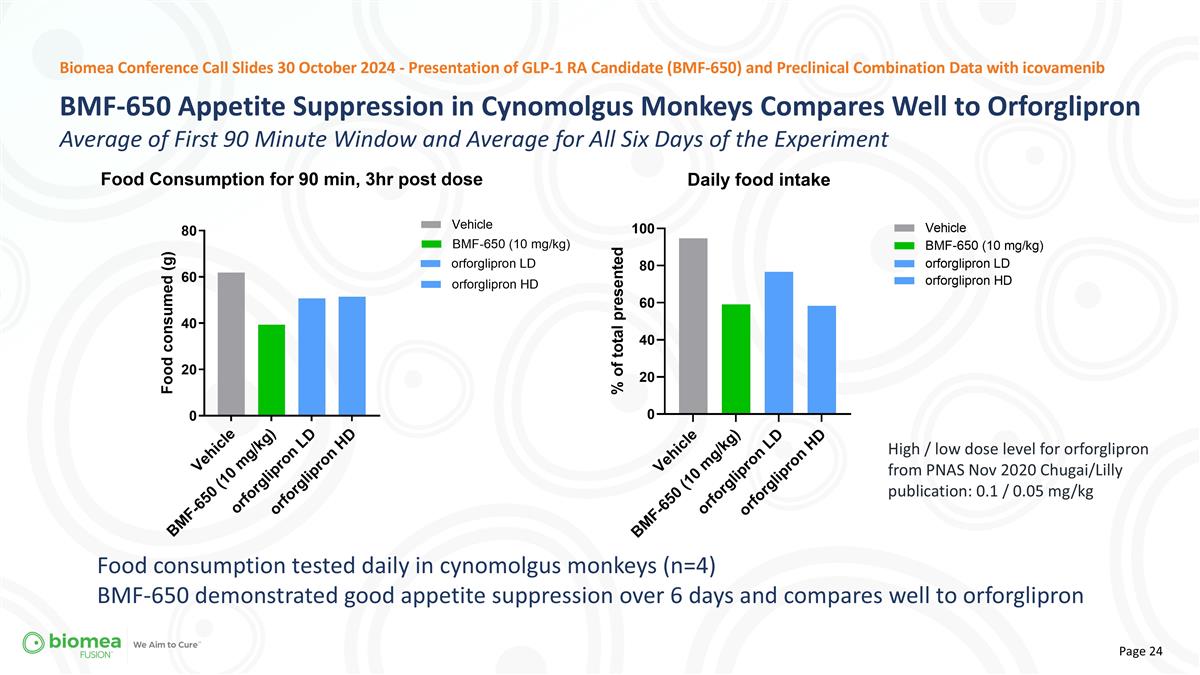
High / low dose level for orforglipron from PNAS Nov 2020 Chugai/Lilly publication: 0.1 / 0.05 mg/kg Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib Food consumption tested daily in cynomolgus monkeys (n=4) BMF-650 demonstrated good appetite suppression over 6 days and compares well to orforglipron BMF-650 Appetite Suppression in Cynomolgus Monkeys Compares Well to Orforglipron Average of First 90 Minute Window and Average for All Six Days of the Experiment
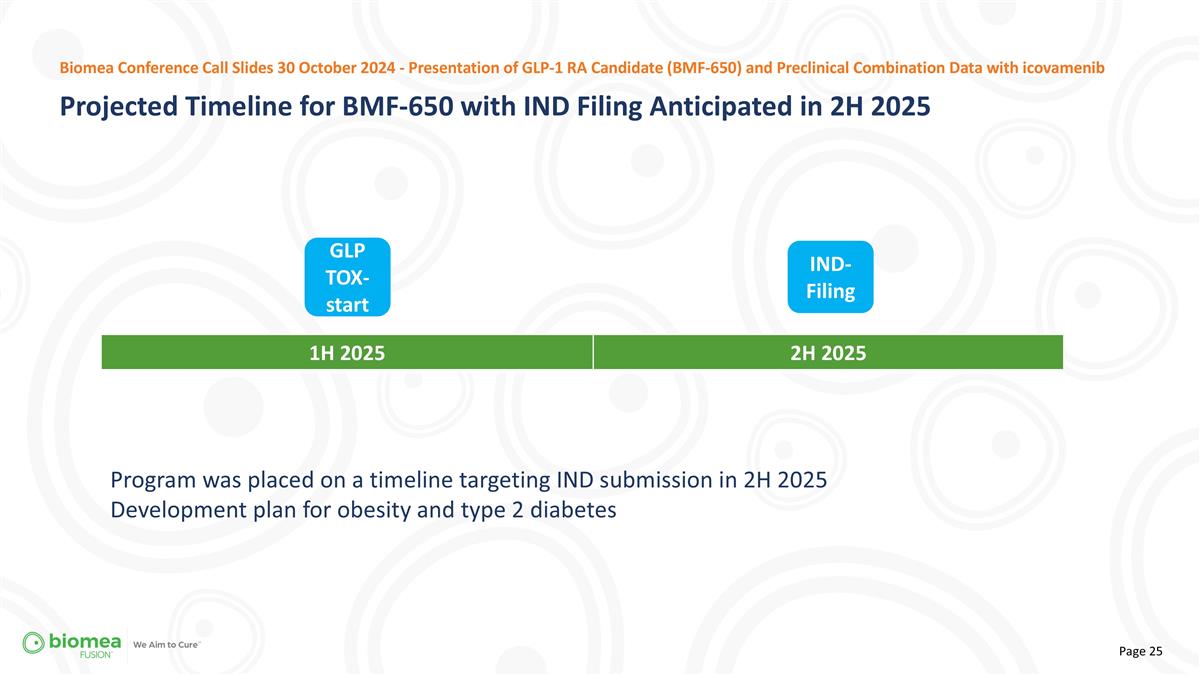
Program was placed on a timeline targeting IND submission in 2H 2025 Development plan for obesity and type 2 diabetes 1H 2025 2H 2025 IND-Filing GLP TOX-start Projected Timeline for BMF-650 with IND Filing Anticipated in 2H 2025 Biomea Conference Call Slides 30 October 2024 - Presentation of GLP-1 RA Candidate (BMF-650) and Preclinical Combination Data with icovamenib

THANK YOU Biomea Fusion 900 Middlefield Road, 4th floor Redwood City, CA, 94063 biomeafusion.com
Exhibit 99.2
Biomea Fusion Presents Preclinical Data Showing Icovamenib (BMF-219) Enhanced Effectiveness of GLP-1-Based Therapies and Introduces BMF-650, a Next-Generation, Oral Small-Molecule GLP-1 Receptor Agonist Candidate
| • | Preclinical data from ex vivo human islet experiments showed that icovamenib (BMF-219) was able to enhance the activity of glucagon-like peptide-1 (GLP-1)-based therapies, potentially leading to increased insulin secretion and improved glycemic control in patients with diabetes |
| • | Phase II study (COVALENT-211), combining icovamenib with a GLP-1-based therapy, planned to begin in 2025 |
| • | BMF-650, an investigational next-generation, oral small-molecule GLP-1 receptor agonist (GLP-1 RA), demonstrated positive early preclinical activity, including improved glucose-stimulated insulin secretion, reduction in blood glucose concentration, and appetite suppression in cynomolgus monkeys |
REDWOOD CITY, Calif., October 30, 2024 (GLOBE NEWSWIRE) — Biomea Fusion, Inc. (“Biomea” or “the Company”) (Nasdaq: BMEA), a clinical-stage biopharmaceutical company dedicated to discovering and developing oral covalent small molecules to improve the lives of patients with diabetes, obesity, and genetically defined cancers, today presented preclinical data showing icovamenib enhanced the activity of GLP-1-based therapies, along with early preclinical efficacy and pharmacokinetic data for BMF-650, a next-generation, oral small-molecule GLP-1 RA candidate.
“Menin plays a central role in the pancreas, not only impacting the proliferation of beta cells but also the expression of GLP-1 receptors. We observed that icovamenib, when combined with either of the two most commonly used GLP-1-based therapies, tirzepatide or semaglutide, enhanced the responsiveness of human islets to the GLP-1-based therapy, leading to substantial insulin secretion under hyperglycemic conditions. The dose-dependent improvements, where glucose-stimulated insulin secretion more than doubled, are highly promising,” stated Juan Pablo Frias, MD, Biomea Fusion’s Chief Medical Officer. “Additionally, we believe the results we’ve seen with our own GLP-1 RA product candidate, BMF-650, highlight a strong potential as a next-generation, oral GLP-1 RA for both diabetes and obesity. We believe these findings open exciting new avenues for treatment.”
Icovamenib (BMF-219) in Combination with a GLP-1-Based Therapy Key Highlights:
| • | Preclinical studies evaluated insulin secretion by GLP-1-based therapies (tirzepatide and semaglutide) using human islets cultured ex vivo under hyperglycemic conditions, with and without icovamenib treatment. |
| • | We hypothesized that menin inhibition would enhance the effectiveness of GLP-1-based therapies and showcased functional data, indicating stronger insulin responses with both tirzepatide and semaglutide when combined with icovamenib treatment. |
| • | Further studies with orforglipron also indicated that icovamenib pretreatment improved insulin secretion, approximately doubling the effect-size depending on the dose. |
| • | Additionally, BMF-650, Biomea’s next-generation, oral small molecule GLP-1 RA product candidate, used alone or in combination with icovamenib, yielded even further improved results supporting the potential for therapeutic benefits. |
| • | The initiation of a Phase II study, COVALENT-211, to evaluate the combination of icovamenib with a GLP-1-based therapy, is planned for 2025. |
In addition, the Company announced additional details about its investigational, next-generation, oral small-molecule GLP-1 RA candidate, BMF-650.
BMF-650 - an Investigational, Next-Generation, Oral Small Molecule GLP-1 Receptor Agonist—Key Highlights:
| • | We conducted preclinical studies to evaluate the properties of BMF-650 in comparison to a leading oral GLP-1 RA. BMF-650 exhibited higher bioavailability and a less variable pharmacokinetic profile, which may translate to improved tolerability and support successful dose escalation in patients. The estimated human dose will be approximately 100 mg once daily. |
| • | In human donor islet studies, BMF-650 significantly enhanced glucose-stimulated insulin secretion. |
| • | In cynomolgus monkey studies, BMF-650 showed significant improvements in glucose stimulated insulin secretion, in line with findings from the human donor islet experiments. BMF-650 also demonstrated superior glucose control. |
| • | Appetite suppression studies revealed that daily oral BMF-650 dosing significantly reduced food intake during peak drug concentration, with sustained effects throughout the day for a six-day study period. |
| • | These findings highlight BMF-650’s potential as an oral treatment for diabetes and obesity. |
“Our preclinical findings about the inhibition of menin in the GLP-1 pathway, announced today, may support the profile of icovamenib as a potential combination agent for GLP-1-based therapies. With the combination of icovamenib, less GLP-1 RA dosing may be required, which may support the overall benefits of these therapeutics. We believe, icovamenib may contribute to improved efficacy, tolerability and adherence, which ultimately will lead to more patients having longer benefits from these agents. We plan to clinically evaluate icovamenib as an adjunct to GLP-1-based therapies,” stated Thomas Butler, Biomea Fusion’s Chief Executive Officer and Chairman of the Board. “We are equally excited with the early results of our newest asset, BMF-650, which demonstrated clear advantages, including when compared to a leading GLP-1 RA in our preclinical studies. BMF-650 has shown superior insulin secretion, better glucose control, a smoother pharmacokinetic profile and higher bioavailability; all of which point to the potential for a greater therapeutic window and support our plans to evaluate BMF-650 as a next-generation oral treatment for diabetes. The appetite suppression results were particularly exciting, as they signal a new and impactful profile which we believe may support an impact on obesity.”
Conference Call and Webcast Details
Webcast, and related presentation, of Biomea’s investor update on Wednesday, October 30 at 4:30 pm ET will be available to registered attendees under the Investors and Media section of the company’s website at https://investors.biomeafusion.com/news-events/events. A replay of the presentation will be available on Biomea’s site following the event.
About Obesity
Obesity is a chronic disease necessitating long-term management, associated with diminished life expectancy and a spectrum of severe health complications. These include metabolic disorders such as type 2 diabetes and non-alcoholic fatty liver disease; cardiovascular diseases like heart attack, stroke, and hypertension; and increased risks of chronic kidney disease, certain cancers, and chronic inflammation. The CDC estimates that over 40% of adults in the U.S. are considered obese, contributing to a significant burden on public health and healthcare systems. Globally, over 650 million adults are living with obesity, and these numbers are steadily rising.
About GLP-1 Receptor Agonists
Glucagon-like peptide-1 (GLP-1) is a naturally occurring incretin hormone that plays a vital role in glucose homeostasis and appetite regulation. GLP-1 receptor agonists (GLP-1 RAs) are a class of medications that bind to and activate GLP-1 receptors, mimicking the effects of native GLP-1. These agents have demonstrated robust clinical efficacy in improving glycemic control, promoting weight loss, and enhancing insulin sensitivity in individuals with type 2 diabetes and obesity.
About Biomea Fusion
Biomea Fusion is a clinical-stage biopharmaceutical company focused on the discovery and development of oral covalent small molecules to improve the lives of patients with diabetes, obesity, and genetically defined cancers. A covalent small molecule is a synthetic compound that forms a permanent bond to its target protein and offers a number of potential advantages over conventional non-covalent drugs, including greater target selectivity, lower drug exposure, and the ability to drive a deeper, more durable response.
We are utilizing our proprietary FUSION™ System to discover, design and develop a pipeline of next-generation covalent-binding small-molecule medicines designed to maximize clinical benefit for patients. We aim to have an outsized impact on the treatment of disease for the patients we serve. We aim to cure.
Visit us at biomeafusion.com and follow us on LinkedIn, X and Facebook.
Forward-Looking Statements
Statements we make in this press release may include statements which are not historical facts and are considered forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended (the “Securities Act”), and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). These statements may be identified by words such as “aims,” “anticipates,” “believes,” “could,” “estimates,” “expects,” “forecasts,” “goal,” “intends,” “may,” “plans,” “possible,” “potential,” “seeks,” “will,” and variations of these words or similar expressions that are intended to identify forward-looking statements. Any such statements in this press release that are not statements of historical fact, including statements regarding the clinical and therapeutic potential of our product candidates and development programs, programs and their potential relative to approved products marketed by third parties; our research, development and regulatory plans, the progress of our ongoing and upcoming clinical trials, the progress and timing of pre-clinical development in our programs; the anticipated enrollment of patients and availability of data from our clinical trials, anticipated milestones, and the timing of such events may be deemed to be forward-looking statements. We intend these forward-looking statements to be covered by the safe harbor provisions for forward-looking statements contained in Section 27A of the Securities Act and Section 21E of the Exchange Act and are making this statement for purposes of complying with those safe harbor provisions.
Any forward-looking statements in this press release are based on our current expectations, estimates and projections only as of the date of this release and are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in or implied by such forward-looking statements, including the risk that preliminary or interim results of preclinical studies or clinical trials may not be predictive of future or final results in connection with future clinical trials and the risk that we may encounter delays in preclinical or clinical development, patient enrollment and in the initiation, conduct and completion of our ongoing and planned clinical trials and other research and development activities. These risks concerning Biomea Fusion’s business and operations are described in additional detail in its periodic filings with the U.S. Securities and Exchange Commission (SEC), including its most recent periodic report filed with the SEC and subsequent filings thereafter. Biomea Fusion explicitly disclaims any obligation to update any forward-looking statements except to the extent required by law.
Contact:
Investor & Media Relations
Ramses Erdtmann
re@biomeafusion.com