UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 OR 15(d)
of The Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): January 4, 2024
NewAmsterdam Pharma Company N.V.
(Exact name of registrant as specified in its charter)
| The Netherlands | 001-41562 | N/A | ||
| (State or other jurisdiction of incorporation) |
(Commission File Number) |
(I.R.S. Employer Identification No.) |
| Gooimeer 2-35 Naarden The Netherlands |
1411 DC | |
| (Address of principal executive offices) | (Zip Code) |
+31 (0) 35 206 2971
(Registrant’s telephone number, including area code)
Not Applicable
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| ☐ | Written communication pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ☐ | Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ☐ | Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ☐ | Pre-commencements communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class |
Trading |
Name of each exchange on which registered |
||
| Ordinary Shares, nominal value €0.12 per share | NAMS | The Nasdaq Stock Market LLC | ||
| Warrants to purchase Ordinary Shares | NAMSW | The Nasdaq Stock Market LLC |
☒ Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
☐ If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act.
| Item 5.02 | Departure of Directors or Certain Officers; Election of Directors; Appointment of Certain Officers; Compensatory Arrangements of Certain Officers. |
On January 4, 2024, the board of directors (the “Board”) of NewAmsterdam Pharma Company N.V. (the “Company”) increased the size of the Board to ten and appointed Willian H. Lewis, J.D., M.B.A., as Chair of the Board. Mr. Lewis will serve as a temporary non-executive director until his formal appointment at the Company’s next general meeting of shareholders. The Board has determined that Mr. Lewis meets the requirements for independence under the applicable listing standards of the Nasdaq Stock Market LLC and the Securities and Exchange Act of 1934, as amended, and has appointed him to serve on the Audit Committee and Compensation Committee of the Board.
Mr. Lewis has served as Chief Executive Officer of Insmed Incorporated, a global commercial-stage biopharmaceutical company, since 2012, and as Chair of Insmed Incorporated’s board of directors since 2018. Mr. Lewis succeeds Sander Slootweg, Managing Partner at Forbion, who has served as Chairman of the Board since inception.
As consideration for his service on the Board, Mr. Lewis will be eligible to receive an annual cash retainer of $40,000, an additional $30,000 annually for serving as Chair of the Board and an additional $20,000 annually for serving as lead independent director. Mr. Lewis will also be granted options to subscribe for 100,000 of the Company’s ordinary shares under the Company’s Long-Term Incentive Plan (the “Initial Grant”). The first 25% of the ordinary shares underlying the Initial Grant will vest on the first anniversary of the vesting start date and the remaining shares will vest in equal monthly installments thereafter for three years, subject to his continued service on the Board. Mr. Lewis will also be entitled to receive an annual retainer of $7,500 and $5,000 for serving on the Audit Committee and Compensation Committee, respectively.
There is no arrangement between Mr. Lewis and any person pursuant to which he was selected as a director. Mr. Lewis has no direct or indirect material interest in any existing or currently proposed transaction that would require disclosure under Item 404(a) of Regulation S-K.
| Item 7.01 | Regulation FD Disclosure. |
On January 8, 2024, the Company issued a press release announcing Mr. Lewis’ appointment to the Board. The press release is attached hereto as Exhibit 99.1 and incorporated herein by reference.
On January 8, 2024, the Company posted an updated corporate investor presentation on its website (https://www.newamsterdampharma.com/). A copy of the corporate investor presentation is furnished as Exhibit 99.2 to this Current Report on Form 8-K. The information contained on, or that can be accessed from, the Company’s website is not incorporated into, and does not constitute a part of, this Current Report on Form 8-K.
The information contained in this Item 7.01, including Exhibits 99.1 and 99.2, is being “furnished” and shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liability of that Section or Sections 11 and 12(a)(2) of the Securities Act of 1933, as amended (the “Securities Act”). The information contained in this Item 7.01, including Exhibit 99.2, shall not be incorporated by reference into any registration statement or other document pursuant to the Securities Act or into any filing or other document pursuant to the Exchange Act, except as otherwise expressly stated in any such filing.
| Item 9.01 | Financial Statements and Exhibits. |
(d) Exhibits.
| EXHIBIT NUMBER |
EXHIBIT DESCRIPTION |
|
| 99.1 | Press Release, dated January 8, 2024 | |
| 99.2 | NewAmsterdam Pharma Company N.V. Corporate Presentation | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) | |
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| NewAmsterdam Pharma Company N.V. | ||
| By: | /s/ Michael Davidson |
|
| Michael Davidson | ||
| Chief Executive Officer | ||
Dated: January 8, 2024
Exhibit 99.1
NewAmsterdam Pharma Appoints William H. Lewis, J.D., M.B.A. as Chair of its Board of Directors
Naarden, the Netherlands and Miami, USA; January 8, 2024 – NewAmsterdam Pharma Company N.V. (Nasdaq: NAMS or “NewAmsterdam” or the “Company”), a clinical-stage biopharmaceutical company developing oral, non-statin medicines for patients at high risk of cardiovascular disease with residual elevation of low-density lipoprotein cholesterol (“LDL-C”), for whom existing therapies are not sufficiently effective or well-tolerated, today announced the appointment of William H. Lewis, J.D., M.B.A., as Chair of its Board of Directors. Mr. Lewis has served as Chief Executive Officer of Insmed Incorporated, a global commercial-stage biopharmaceutical company, since 2012, and as Chair of Insmed’s Board of Directors since 2018. Mr. Lewis succeeds Sander Slootweg, Managing Partner at Forbion, who has served as Chairman of NewAmsterdam’s Board of Directors since inception.
“Will is a leader in the biotechnology industry, widely recognized for his commitment to putting patients first, championing those who are underserved by existing treatment options, and translating breakthrough science into first-in-disease medicines,” said Michael Davidson, M.D., Chief Executive Officer of NewAmsterdam. “I have long admired Will for his deep knowledge of the clinical and regulatory landscape, as well as his proven ability to advance new therapies for serious and rare diseases. He will be a tremendous partner, who I have known for many years, and as Chair of our Board I look forward to his contributions as we advance our CETP inhibitor toward market and work to deliver a simple, oral, once-daily option to millions of people living with dyslipidemia. I would also like to thank Sander for his support and contribution as our Chairman over the last several years. His guidance has been invaluable in getting NewAmsterdam to where we are today.”
“Over the past several years, I have had the unique privilege of partnering with Will as both an investor and colleague. He is a remarkable leader, with experience advancing novel molecules through late-stage development, launching into sizeable markets and establishing new standards-of-care,” commented Mr. Slootweg. “I am confident he is the right partner to support NewAmsterdam as it enters its next phase of growth, with multiple pivotal data readouts expected in 2024 and, if approved, its plans to commercialize obicetrapib.”
Mr. Lewis has more than 30 years of executive experience in the pharmaceutical and finance industries both in the U.S. and internationally. Prior to joining Insmed in 2012, Mr. Lewis served as Co-Founder, President, and Chief Financial Officer of Aegerion Pharmaceuticals, which was acquired by Amryt in 2019. Prior to Aegerion, he spent more than 10 years working in investment banking in the U.S. and Europe. He also previously worked for the U.S. government. Mr. Lewis holds a J.D. with Honors and an M.B.A., both from Case Western Reserve University, and a B.A., cum laude, from Oberlin College. He is a member of the Board of Trustees of Case Western Reserve University and of BioNJ, the life sciences association for New Jersey.
“NewAmsterdam was founded in hopes of delivering a new option to high-risk cardiovascular disease patients, many of whom fail to achieve their risk-based LDL-C goals despite treatment with statin therapy,” said Mr. Lewis. “Based on data generated from its five Phase 2 trials to date, it is clear that obicetrapib is designed to be a powerful therapy, with the potential to safely and effectively improve LDL-C, as well as other key markers of cardiovascular disease risk. I am excited to join NewAmsterdam’s Board of Directors and look forward to partnering closely with management to complete the ongoing Phase 3 program and bring obicetrapib forward.”
About Obicetrapib
Obicetrapib is a novel, oral, low-dose CETP inhibitor that NewAmsterdam is developing to overcome the limitations of current LDL-lowering treatments. The Company believes that obicetrapib has the potential to be a once-daily oral CETP inhibitor for lowering LDL-C, if approved. In the Company’s Phase 2b ROSE trial, obicetrapib demonstrated a 51% lowering of LDL-C from baseline at a 10 mg dose level on top of high-intensity statins and, in the Company’s Phase 2 ROSE2 trial, the combination of a 10 mg dose of obicetrapib and a 10 mg dose of ezetimibe demonstrated a 63% lowering of LDL-C from baseline. In all five of the Company’s Phase 2 trials, ROSE2, TULIP, ROSE, OCEAN, and TA-8995-203, evaluating obicetrapib as monotherapy or combination therapy, the Company observed statistically significant LDL-lowering combined with a side effect profile similar to that of placebo, including no increase in blood pressure or muscle related side effects. Obicetrapib has demonstrated strong tolerability in more than 800 patients with elevated lipid levels (“dyslipidemia”) in NewAmsterdam’s clinical trials to date. The Company is conducting two Phase 3 pivotal trials, BROADWAY and BROOKLYN, to evaluate obicetrapib as a monotherapy used as an adjunct to maximally tolerated lipid-lowering therapies to provide additional LDL-lowering for high-risk cardiovascular disease (“CVD”) patients.
The Company began enrolling patients in BROADWAY in January 2022 and in BROOKLYN in July 2022 and completed enrollment of BROOKLYN in April 2023 and BROADWAY in July 2023. The Company also commenced the Phase 3 PREVAIL cardiovascular outcomes trial in March 2022, which is designed to assess the potential of obicetrapib to reduce occurrences of major adverse cardiovascular events, including cardiovascular death, non-fatal myocardial infarction, non-fatal stroke and non-elective coronary revascularization.
About NewAmsterdam
Based in the Netherlands, NewAmsterdam (Nasdaq: NAMS) is a clinical-stage biopharmaceutical company whose mission is to improve patient care in populations with metabolic diseases where currently approved therapies have not been sufficiently adequate or well tolerated. We seek to fill a significant unmet need for a safe, cost-effective and convenient LDL-lowering therapy as an adjunct to statins, a class of lipid-lowering medications that are the current standard of care for high-risk CVD patients with high cholesterol. NewAmsterdam is investigating obicetrapib, an oral, low-dose and once-daily CETP inhibitor, as the preferred LDL-C lowering therapy to be used as an adjunct to maximally tolerated statin therapy for high-risk cardiovascular disease patients.
Forward-Looking Statements
Certain statements included in this document that are not historical facts are forward-looking statements for purposes of the safe harbor provisions under the United States Private Securities Litigation Reform Act of 1995. Forward-looking statements generally are accompanied by words such as “believe,” “may,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “expect,” “should,” “would,” “plan,” “predict,” “potential,” “position,” “seem,” “seek,” “future,” “outlook” and similar expressions that predict or indicate future events or trends or that are not statements of historical matters. These forward-looking statements include, but are not limited to, statements regarding the Company’s business and strategic plans, cash runway, the therapeutic and curative potential of the Company’s product candidate, the Company’s clinical trials and the timing for enrolling patients, the timing and forums for announcing data, the achievement and timing of regulatory approvals and plans for commercialization. These statements are based on various assumptions, whether or not identified in this document, and on the current expectations of the Company’s management and are not predictions of actual performance. These forward-looking statements are provided for illustrative purposes only and are not intended to serve as and must not be relied on as a guarantee, an assurance, a prediction, or a definitive statement of fact or probability. Actual events and circumstances are difficult or impossible to predict and may differ from assumptions. Many actual events and circumstances are beyond the control of the Company. These forward-looking statements are subject to a number of risks and uncertainties, including changes in domestic and foreign business, market, financial, political, and legal conditions; risks relating to the uncertainty of the projected financial information with respect to the Company; risks relating to the uncertainty of the projected financial information with respect to the Company; risks related to the approval of the Company’s product candidate and the timing of expected regulatory and business milestones, including potential commercialization; ability to negotiate definitive contractual arrangements with potential customers; the impact of competitive product candidates; ability to obtain sufficient supply of materials; global economic and political conditions; the effects of competition on the Company’s future business; and those factors described in the Company’s public filings with the U.S. Securities and Exchange Commission. Additional risks related to the Company’s business include, but are not limited to: uncertainty regarding outcomes of the Company’s ongoing clinical trials, particularly as they relate to regulatory review and potential approval for its product candidate; risks associated with the Company’s efforts to commercialize a product candidate; the Company’s ability to negotiate and enter into definitive agreements on favorable terms, if at all; the impact of competing product candidates on the Company’s business; intellectual property related claims; the Company’s ability to attract and retain qualified personnel; ability to continue to source the raw materials for its product candidate. If any of these risks materialize or the Company’s assumptions prove incorrect, actual results could differ materially from the results implied by these forward-looking statements. There may be additional risks that the Company does not presently know or that the Company currently believes are immaterial that could also cause actual results to differ from those contained in the forward-looking statements.
In addition, forward-looking statements reflect the Company’s expectations, plans, or forecasts of future events and views as of the date of this document and are qualified in their entirety by reference to the cautionary statements herein. The Company anticipates that subsequent events and developments may cause the Company’s assessments to change. These forward-looking statements should not be relied upon as representing the Company’s assessment as of any date subsequent to the date of this communication. Accordingly, undue reliance should not be placed upon the forward-looking statements. Neither the Company nor any of its affiliates undertakes any obligation to update these forward-looking statements, except as may be required by law.
Company Contact
Matthew Philippe
P: 1-917-882-7512
matthew.philippe@newamsterdampharma.com
Media Contact
Spectrum Science on behalf of NewAmsterdam
Jen Gordon
P: 1-202-957-7795
jgordon@spectrumscience.com
Investor Contact
Stern Investor Relations on behalf of NewAmsterdam
Hannah Deresiewicz
P: 1-212-362-1200
hannah.deresiewicz@sternir.com

Exhibit 99.2 January 2024

Disclaimer This presentation (together with oral statements made in connection herewith, this “Presentation”) is for informational purposes only. This Presentation shall not constitute an offer to sell, or the solicitation of an offer to buy, any securities, nor shall there be any sale of securities in any states or jurisdictions in which such offer, solicitation or sale would be unlawful. Forward Looking Statements Certain statements included in this Presentation that are not historical facts are forward-looking statements for purposes of the safe harbor provisions under the United States Private Securities Litigation Reform Act of 1995. Forward-looking statements generally are accompanied by words such as “believe,” “may,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “expect,” “should,” “would,” “plan,” “predict,” “potential,” “seem,” “seek,” “future,” “outlook” and similar expressions that predict or indicate future events or trends or that are not statements of historical matters. These forward-looking statements include, but are not limited to, statements by NewAmsterdam Pharma Company N.V. (“NewAmsterdam” or the “Company”) regarding estimates and forecasts of other financial and performance metrics and projections of market opportunity; expectations and timing related to the success, cost and timing of product development activities, including timing of initiation, completion and data readouts for clinical trials and the potential approval of the Company’s product candidate; the timing for enrolling patients; the timing and forums for announcing data; the size and growth potential of the markets for the Company’s product candidate; the therapeutic and curative potential of the Company’s product candidate; financing and other business milestones; the Company’s expected cash runway; and the Company’s plans for commercialization. These statements are based on various assumptions, whether or not identified in this Presentation, and on the current expectations of the Company’s management and are not predictions of actual performance. These forward-looking statements are provided for illustrative purposes only and are not intended to serve as and must not be relied on as a guarantee, an assurance, a prediction, or a definitive statement of fact or probability. Actual events and circumstances are difficult or impossible to predict and may differ from assumptions. Many actual events and circumstances are beyond the control of the Company. These forward-looking statements are subject to a number of risks and uncertainties, including changes in domestic and foreign business, market, financial, political, and legal conditions; risks related to the approval of NewAmsterdam’s product candidate and the timing of expected regulatory and business milestones; whether topline, initial or preliminary results from a particular clinical trial will be predictive of the final results of that trial and whether results of early clinical trials will be indicative of the results of later clinical trials; ability to negotiate definitive contractual arrangements with potential customers; the impact of competitive product candidates; ability to obtain sufficient supply of materials; global economic and political conditions, including the Russia-Ukraine conflict, and the war in Israel; the effects of competition on NewAmsterdam’s future business; and those factors discussed in documents filed by the Company with the SEC. Additional risks related to NewAmsterdam’s business include, but are not limited to: uncertainty regarding outcomes of the company’s ongoing clinical trials, particularly as they relate to regulatory review and potential approval for its product candidate; risks associated with the Company’s efforts to commercialize a product candidate; the Company’s ability to negotiate and enter into definitive agreements on favorable terms, if at all; the impact of competing product candidates on the Company’s business; intellectual property-related claims; the Company’s ability to attract and retain qualified personnel; and the Company’s ability to continue to source the raw materials for its product candidate, together with the risks described in the Company’s filings made with the U.S. Securities and Exchange Commission from time to time. If any of these risks materialize or NewAmsterdam’s assumptions prove incorrect, actual results could differ materially from the results implied by these forward-looking statements. There may be additional risks that are presently unknown by the Company or that NewAmsterdam currently believes are immaterial that could also cause actual results to differ from those contained in the forward-looking statements. In addition, forward-looking statements reflect NewAmsterdam’s expectations, plans, or forecasts of future events and views as of the date of this Presentation and are qualified in their entirety by reference to the cautionary statements herein. NewAmsterdam anticipates that subsequent events and developments will cause the Company’s assessments to change. These forward-looking statements should not be relied upon as representing NewAmsterdam’s assessments as of any date subsequent to the date of this Presentation. Accordingly, undue reliance should not be placed upon the forward-looking statements. Neither NewAmsterdam nor any of its affiliates undertakes any obligation to update these forward-looking statements, except as required by law. Market Data Certain information contained in this Presentation relates to or is based on third-party studies, publications, surveys and NewAmsterdam’s own internal estimates and research. In addition, all of the market data included in this Presentation involves a number of assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. Finally, while NewAmsterdam believes its internal research is reliable, such research has not been verified by any independent source and NewAmsterdam cannot guarantee and makes no representation or warranty, express or implied, as to its accuracy and completeness. Trademarks This Presentation contains trademarks, service marks, trade names, and copyrights of NewAmsterdam and other companies, which are the property of their respective owners. The use or display of third parties’ trademarks, service marks, trade name or products in this Presentation is not intended to, and does not imply, a relationship with NewAmsterdam or an endorsement or sponsorship by or of NewAmsterdam. Solely for convenience, the trademarks, service marks and trade names referred to in this Presentation may appear with the TM or SM symbols, but such references are not intended to indicate, in any way, that NewAmsterdam will not assert, to the fullest extent permitted under applicable law, their rights or the right of the applicable licensor to these trademarks, service marks and trade names. 2
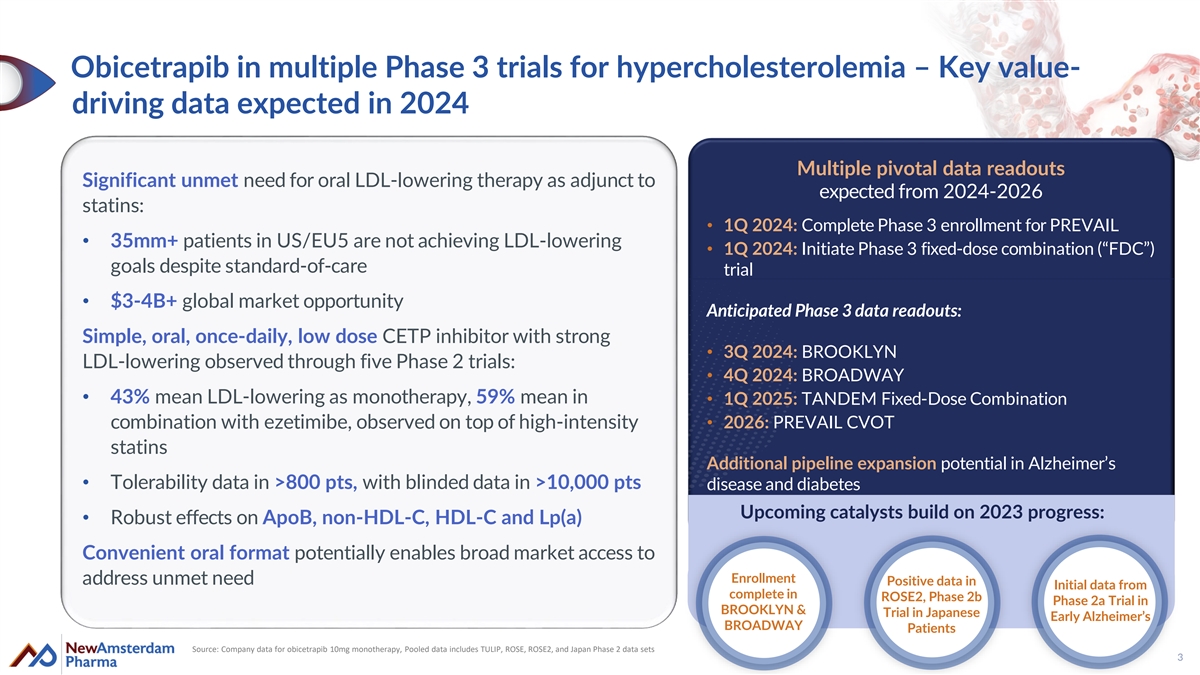
Obicetrapib in multiple Phase 3 trials for hypercholesterolemia – Key value- driving data expected in 2024 Multiple pivotal data readouts Significant unmet need for oral LDL-lowering therapy as adjunct to expected from 2024-2026 statins: • 1Q 2024: Complete Phase 3 enrollment for PREVAIL • 35mm+ patients in US/EU5 are not achieving LDL-lowering • 1Q 2024: Initiate Phase 3 fixed-dose combination (“FDC”) goals despite standard-of-care trial • $3-4B+ global market opportunity Anticipated Phase 3 data readouts: Simple, oral, once-daily, low dose CETP inhibitor with strong • 3Q 2024: BROOKLYN LDL-lowering observed through five Phase 2 trials: • 4Q 2024: BROADWAY • 43% mean LDL-lowering as monotherapy, 59% mean in • 1Q 2025: TANDEM Fixed-Dose Combination combination with ezetimibe, observed on top of high-intensity • 2026: PREVAIL CVOT statins Additional pipeline expansion potential in Alzheimer’s • Tolerability data in >800 pts, with blinded data in >10,000 pts disease and diabetes Upcoming catalysts build on 2023 progress: • Robust effects on ApoB, non-HDL-C, HDL-C and Lp(a) Convenient oral format potentially enables broad market access to Enrollment address unmet need Positive data in Initial data from complete in ROSE2, Phase 2b Phase 2a Trial in BROOKLYN & Trial in Japanese Early Alzheimer’s BROADWAY Patients Source: Company data for obicetrapib 10mg monotherapy, Pooled data includes TULIP, ROSE, ROSE2, and Japan Phase 2 data sets 3
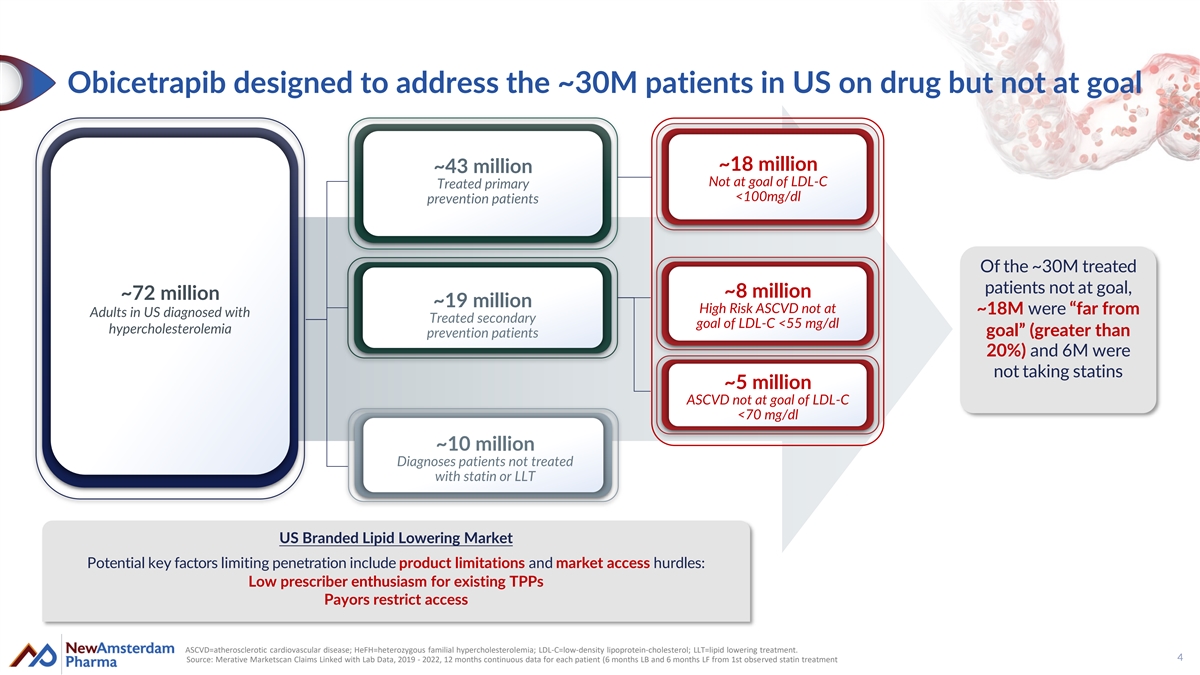
Obicetrapib designed to address the ~30M patients in US on drug but not at goal ~18 million ~43 million Not at goal of LDL-C Treated primary <100mg/dl prevention patients Of the ~30M treated patients not at goal, ~8 million ~72 million ~19 million High Risk ASCVD not at ~18M were “far from Adults in US diagnosed with Treated secondary goal of LDL-C <55 mg/dl hypercholesterolemia goal” (greater than prevention patients 20%) and 6M were not taking statins ~5 million ASCVD not at goal of LDL-C <70 mg/dl ~10 million Diagnoses patients not treated with statin or LLT US Branded Lipid Lowering Market Potential key factors limiting penetration include product limitations and market access hurdles: Low prescriber enthusiasm for existing TPPs Payors restrict access ASCVD=atherosclerotic cardiovascular disease; HeFH=heterozygous familial hypercholesterolemia; LDL-C=low-density lipoprotein-cholesterol; LLT=lipid lowering treatment. 4 Source: Merative Marketscan Claims Linked with Lab Data, 2019 - 2022, 12 months continuous data for each patient (6 months LB and 6 months LF from 1st observed statin treatment
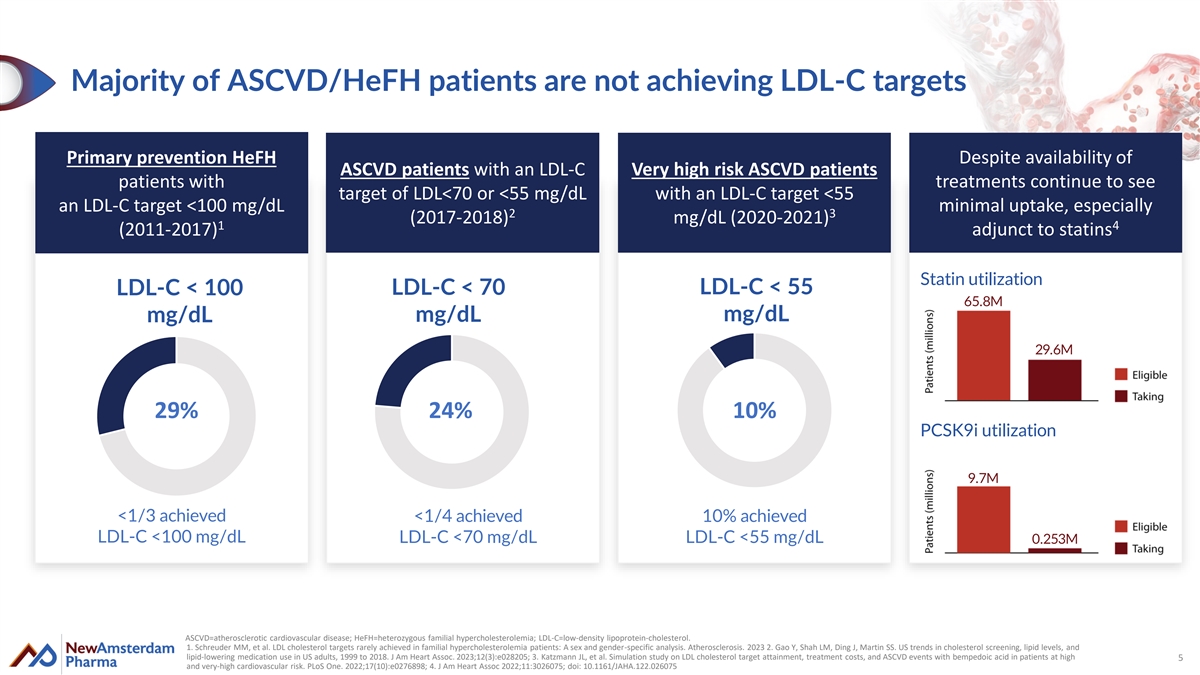
Majority of ASCVD/HeFH patients are not achieving LDL-C targets Primary prevention HeFH Despite availability of ASCVD patients with an LDL-C Very high risk ASCVD patients patients with treatments continue to see target of LDL<70 or <55 mg/dL with an LDL-C target <55 an LDL-C target <100 mg/dL minimal uptake, especially 2 3 (2017-2018) mg/dL (2020-2021) 1 4 (2011-2017) adjunct to statins Statin utilization LDL-C < 100 LDL-C < 70 LDL-C < 55 65.8M mg/dL mg/dL mg/dL 29.6M 29% 24% 10% PCSK9i utilization 9.7M <1/3 achieved <1/4 achieved 10% achieved LDL-C <100 mg/dL LDL-C <70 mg/dL LDL-C <55 mg/dL 0.253M ASCVD=atherosclerotic cardiovascular disease; HeFH=heterozygous familial hypercholesterolemia; LDL-C=low-density lipoprotein-cholesterol. 1. Schreuder MM, et al. LDL cholesterol targets rarely achieved in familial hypercholesterolemia patients: A sex and gender-specific analysis. Atherosclerosis. 2023 2. Gao Y, Shah LM, Ding J, Martin SS. US trends in cholesterol screening, lipid levels, and lipid-lowering medication use in US adults, 1999 to 2018. J Am Heart Assoc. 2023;12(3):e028205; 3. Katzmann JL, et al. Simulation study on LDL cholesterol target attainment, treatment costs, and ASCVD events with bempedoic acid in patients at high 5 and very-high cardiovascular risk. PLoS One. 2022;17(10):e0276898; 4. J Am Heart Assoc 2022;11:3026075; doi: 10.1161/JAHA.122.026075
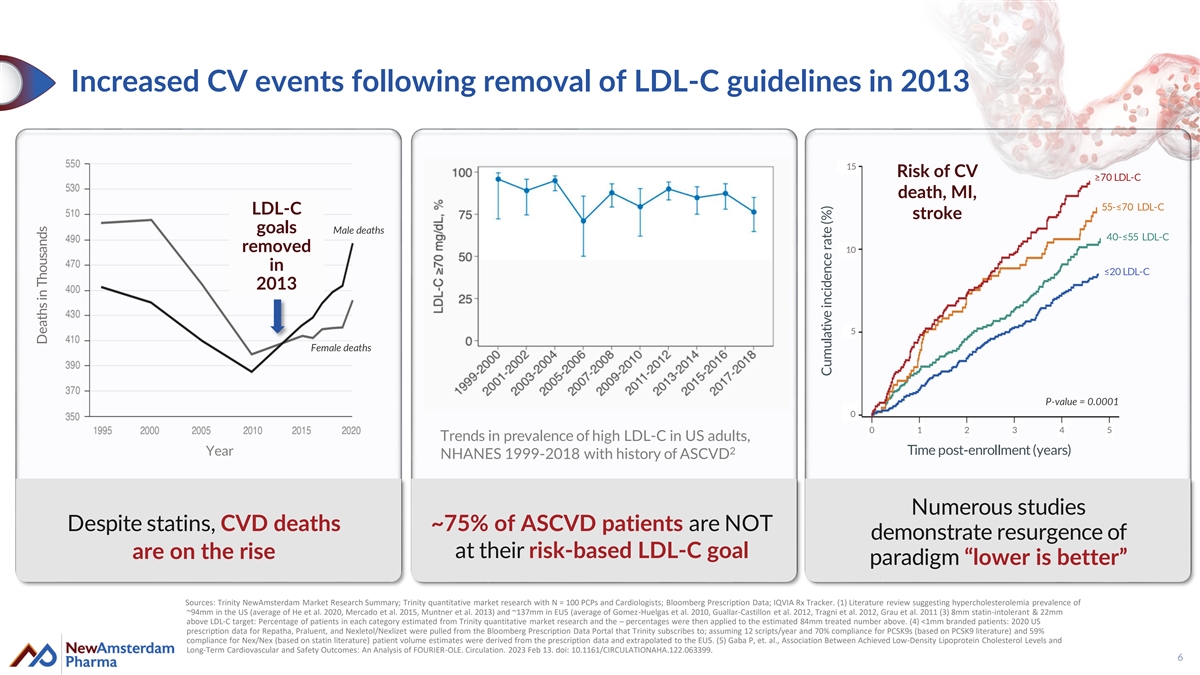
Increased CV events following removal of LDL-C guidelines in 2013 15 Risk of CV ≥70 LDL-C death, MI, 55-≤70 LDL-C LDL-C stroke goals Male deaths 40-≤55 LDL-C removed 10 in ≤20 LDL-C 2013 5 Female deaths P-value = 0.0001 5 0 0 1 2 3 4 5 Trends in prevalence of high LDL-C in US adults, 2 Time post-enrollment (years) Year NHANES 1999-2018 with history of ASCVD Numerous studies Despite statins, CVD deaths ~75% of ASCVD patients are NOT demonstrate resurgence of are on the rise at their risk-based LDL-C goal paradigm “lower is better” Sources: Trinity NewAmsterdam Market Research Summary; Trinity quantitative market research with N = 100 PCPs and Cardiologists; Bloomberg Prescription Data; IQVIA Rx Tracker. (1) Literature review suggesting hypercholesterolemia prevalence of ~94mm in the US (average of He et al. 2020, Mercado et al. 2015, Muntner et al. 2013) and ~137mm in EU5 (average of Gomez-Huelgas et al. 2010, Guallar-Castillon et al. 2012, Tragni et al. 2012, Grau et al. 2011 (3) 8mm statin-intolerant & 22mm above LDL-C target: Percentage of patients in each category estimated from Trinity quantitative market research and the – percentages were then applied to the estimated 84mm treated number above. (4) <1mm branded patients: 2020 US prescription data for Repatha, Praluent, and Nexletol/Nexlizet were pulled from the Bloomberg Prescription Data Portal that Trinity subscribes to; assuming 12 scripts/year and 70% compliance for PCSK9s (based on PCSK9 literature) and 59% compliance for Nex/Nex (based on statin literature) patient volume estimates were derived from the prescription data and extrapolated to the EU5. (5) Gaba P, et. al., Association Between Achieved Low-Density Lipoprotein Cholesterol Levels and Long-Term Cardiovascular and Safety Outcomes: An Analysis of FOURIER-OLE. Circulation. 2023 Feb 13. doi: 10.1161/CIRCULATIONAHA.122.063399. 6 Deaths in Thousands Cumulative incidence rate (%)
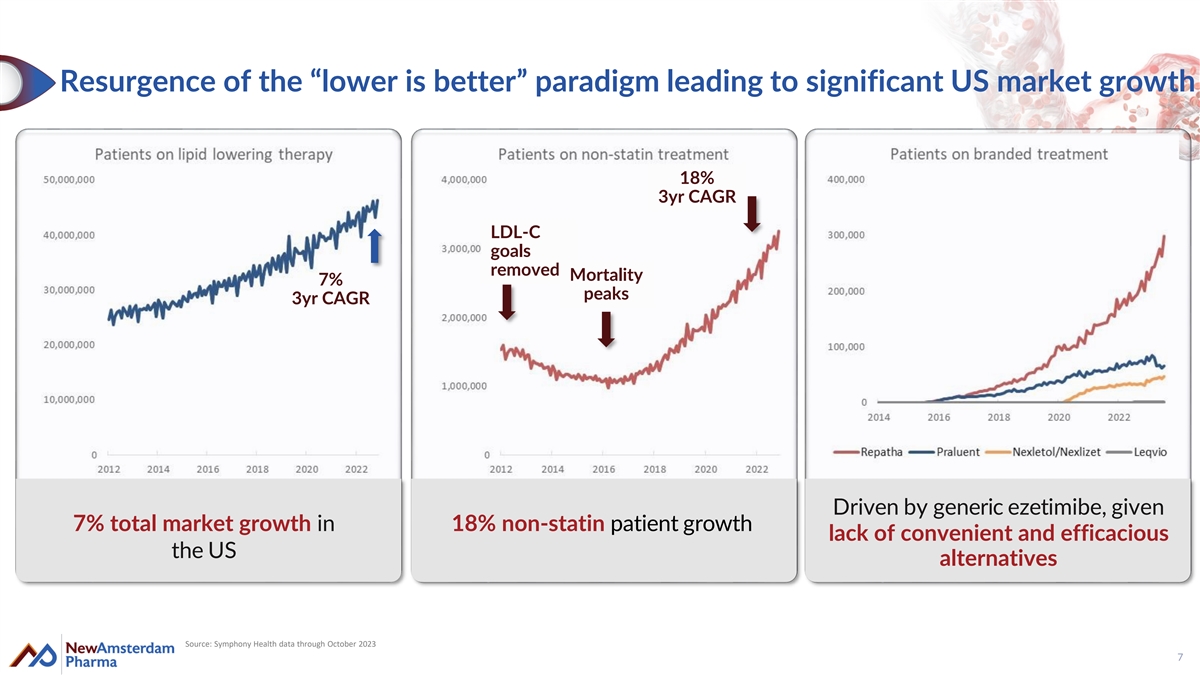
Resurgence of the “lower is better” paradigm leading to significant US market growth 18% 3yr CAGR LDL-C goals removed Mortality 7% peaks 3yr CAGR Driven by generic ezetimibe, given 7% total market growth in 18% non-statin patient growth lack of convenient and efficacious the US alternatives Source: Symphony Health data through October 2023 7
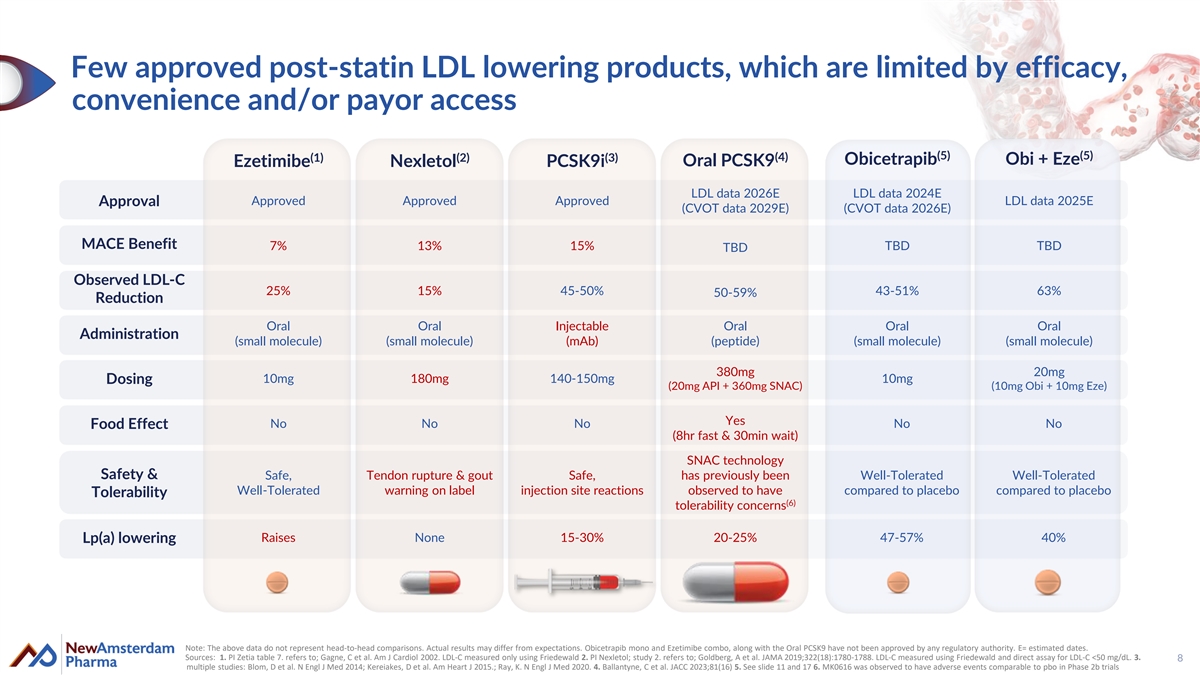
Few approved post-statin LDL lowering products, which are limited by efficacy, convenience and/or payor access (5) (5) (4) (1) (2) (3) Obicetrapib Obi + Eze Oral PCSK9 Ezetimibe Nexletol PCSK9i LDL data 2026E LDL data 2024E Approved Approved Approved LDL data 2025E Approval (CVOT data 2029E) (CVOT data 2026E) MACE Benefit 7% 13% 15% TBD TBD TBD Observed LDL-C 25% 15% 45-50% 43-51% 63% 50-59% Reduction Oral Oral Injectable Oral Oral Oral Administration (small molecule) (small molecule) (mAb) (peptide) (small molecule) (small molecule) 380mg 20mg 10mg 180mg 140-150mg 10mg Dosing (20mg API + 360mg SNAC) (10mg Obi + 10mg Eze) Yes No No No No No Food Effect (8hr fast & 30min wait) SNAC technology Safety & Safe, Tendon rupture & gout Safe, has previously been Well-Tolerated Well-Tolerated Well-Tolerated warning on label injection site reactions observed to have compared to placebo compared to placebo Tolerability (6) tolerability concerns Raises None 15-30% 20-25% 47-57% 40% Lp(a) lowering Note: The above data do not represent head-to-head comparisons. Actual results may differ from expectations. Obicetrapib mono and Ezetimibe combo, along with the Oral PCSK9 have not been approved by any regulatory authority. E= estimated dates. Sources: 1. PI Zetia table 7. refers to; Gagne, C et al. Am J Cardiol 2002. LDL-C measured only using Friedewald 2. PI Nexletol; study 2. refers to; Goldberg, A et al. JAMA 2019;322(18):1780-1788. LDL-C measured using Friedewald and direct assay for LDL-C <50 mg/dL. 3. 8 multiple studies: Blom, D et al. N Engl J Med 2014; Kereiakes, D et al. Am Heart J 2015.; Ray, K. N Engl J Med 2020. 4. Ballantyne, C et al. JACC 2023;81(16) 5. See slide 11 and 17 6. MK0616 was observed to have adverse events comparable to pbo in Phase 2b trials
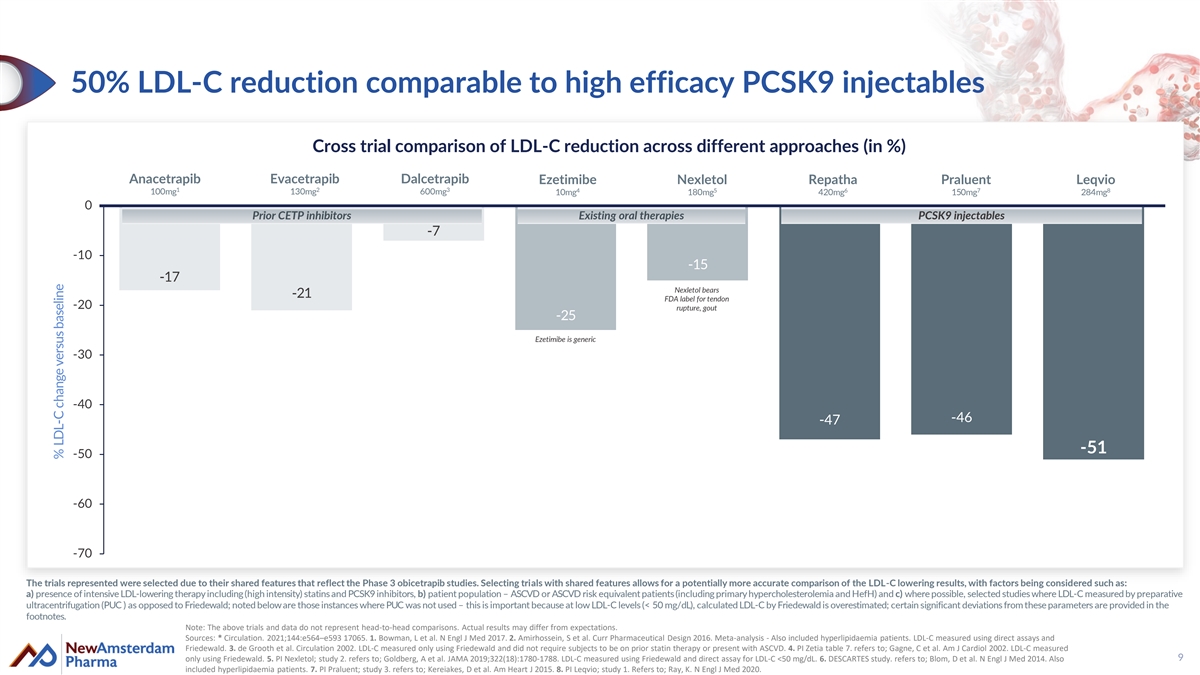
50% LDL-C reduction comparable to high efficacy PCSK9 injectables Cross trial comparison of LDL-C reduction across different approaches (in %) Anacetrapib Evacetrapib Dalcetrapib Ezetimibe Nexletol Repatha Praluent Leqvio 1 2 3 4 5 6 7 8 100mg 130mg 600mg 10mg 180mg 420mg 150mg 284mg 0 Prior CETP inhibitors Existing oral therapies PCSK9 injectables -7 -10 -15 -17 Nexletol bears -21 FDA label for tendon -20 rupture, gout -25 Ezetimibe is generic -30 -40 -46 -47 -51 -50 -60 -70 The trials represented were selected due to their shared features that reflect the Phase 3 obicetrapib studies. Selecting trials with shared features allows for a potentially more accurate comparison of the LDL-C lowering results, with factors being considered such as: a) presence of intensive LDL-lowering therapy including (high intensity) statins and PCSK9 inhibitors, b) patient population – ASCVD or ASCVD risk equivalent patients (including primary hypercholesterolemia and HefH) and c) where possible, selected studies where LDL-C measured by preparative ultracentrifugation (PUC ) as opposed to Friedewald; noted below are those instances where PUC was not used – this is important because at low LDL-C levels (< 50 mg/dL), calculated LDL-C by Friedewald is overestimated; certain significant deviations from these parameters are provided in the footnotes. Note: The above trials and data do not represent head-to-head comparisons. Actual results may differ from expectations. Sources: * Circulation. 2021;144:e564–e593 17065. 1. Bowman, L et al. N Engl J Med 2017. 2. Amirhossein, S et al. Curr Pharmaceutical Design 2016. Meta-analysis - Also included hyperlipidaemia patients. LDL-C measured using direct assays and Friedewald. 3. de Grooth et al. Circulation 2002. LDL-C measured only using Friedewald and did not require subjects to be on prior statin therapy or present with ASCVD. 4. PI Zetia table 7. refers to; Gagne, C et al. Am J Cardiol 2002. LDL-C measured only using Friedewald. 5. PI Nexletol; study 2. refers to; Goldberg, A et al. JAMA 2019;322(18):1780-1788. LDL-C measured using Friedewald and direct assay for LDL-C <50 mg/dL. 6. DESCARTES study. refers to; Blom, D et al. N Engl J Med 2014. Also 9 included hyperlipidaemia patients. 7. PI Praluent; study 3. refers to; Kereiakes, D et al. Am Heart J 2015. 8. PI Leqvio; study 1. Refers to; Ray, K. N Engl J Med 2020. % LDL-C change versus baseline
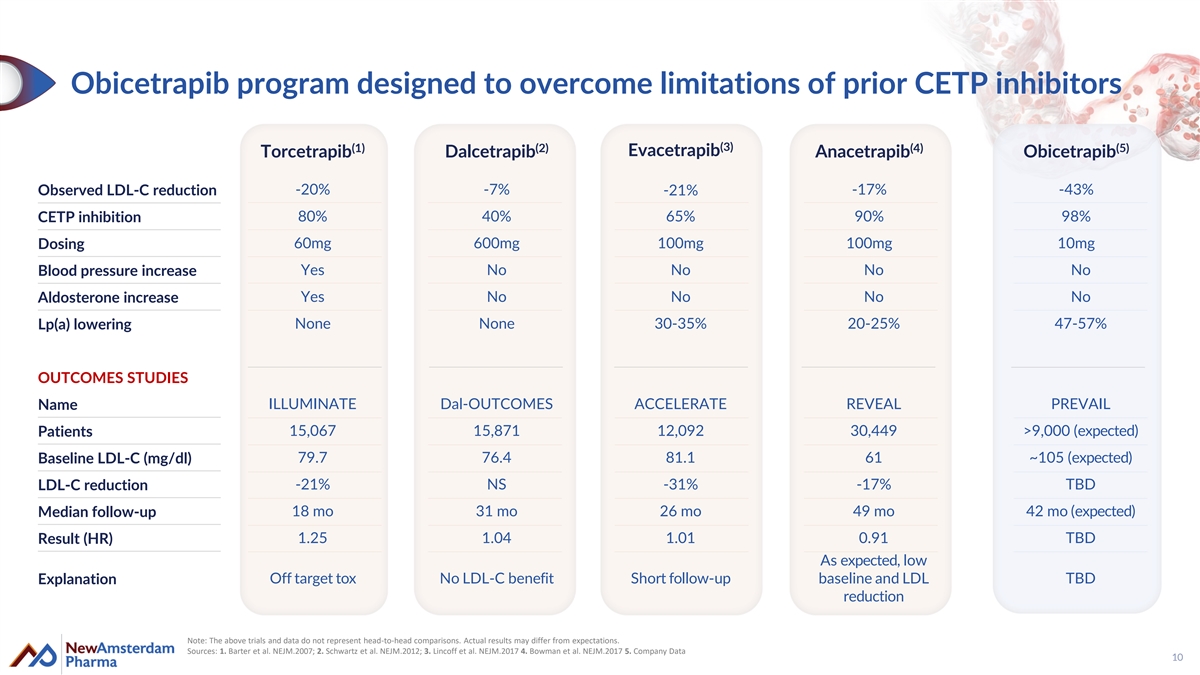
Obicetrapib program designed to overcome limitations of prior CETP inhibitors (3) (1) (2) (4) (5) Evacetrapib Torcetrapib Dalcetrapib Anacetrapib Obicetrapib Observed LDL-C reduction -20% -7% -17% -43% -21% 80% 40% 65% 90% 98% CETP inhibition Dosing 60mg 600mg 100mg 100mg 10mg Yes No No No No Blood pressure increase Aldosterone increase Yes No No No No None None 30-35% 20-25% 47-57% Lp(a) lowering OUTCOMES STUDIES ILLUMINATE Dal-OUTCOMES ACCELERATE REVEAL PREVAIL Name Patients 15,067 15,871 12,092 30,449 >9,000 (expected) 79.7 76.4 81.1 61 ~105 (expected) Baseline LDL-C (mg/dl) LDL-C reduction -21% NS -31% -17% TBD 18 mo 31 mo 26 mo 49 mo 42 mo (expected) Median follow-up Result (HR) 1.25 1.04 1.01 0.91 TBD As expected, low Off target tox No LDL-C benefit Short follow-up baseline and LDL TBD Explanation reduction Note: The above trials and data do not represent head-to-head comparisons. Actual results may differ from expectations. Sources: 1. Barter et al. NEJM.2007; 2. Schwartz et al. NEJM.2012; 3. Lincoff et al. NEJM.2017 4. Bowman et al. NEJM.2017 5. Company Data 10
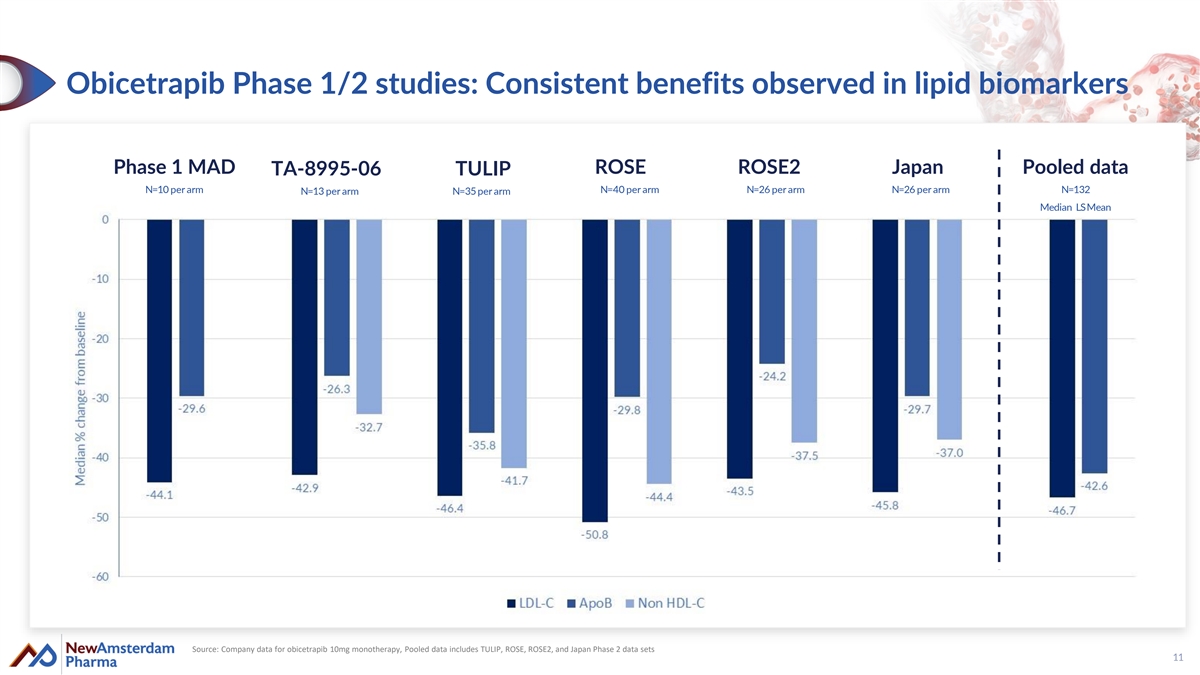
Obicetrapib Phase 1/2 studies: Consistent benefits observed in lipid biomarkers Phase 1 MAD ROSE ROSE2 Japan Pooled data TA-8995-06 TULIP N=10 per arm N=40 per arm N=26 per arm N=26 per arm N=132 N=13 per arm N=35 per arm Median LS Mean Source: Company data for obicetrapib 10mg monotherapy, Pooled data includes TULIP, ROSE, ROSE2, and Japan Phase 2 data sets 11
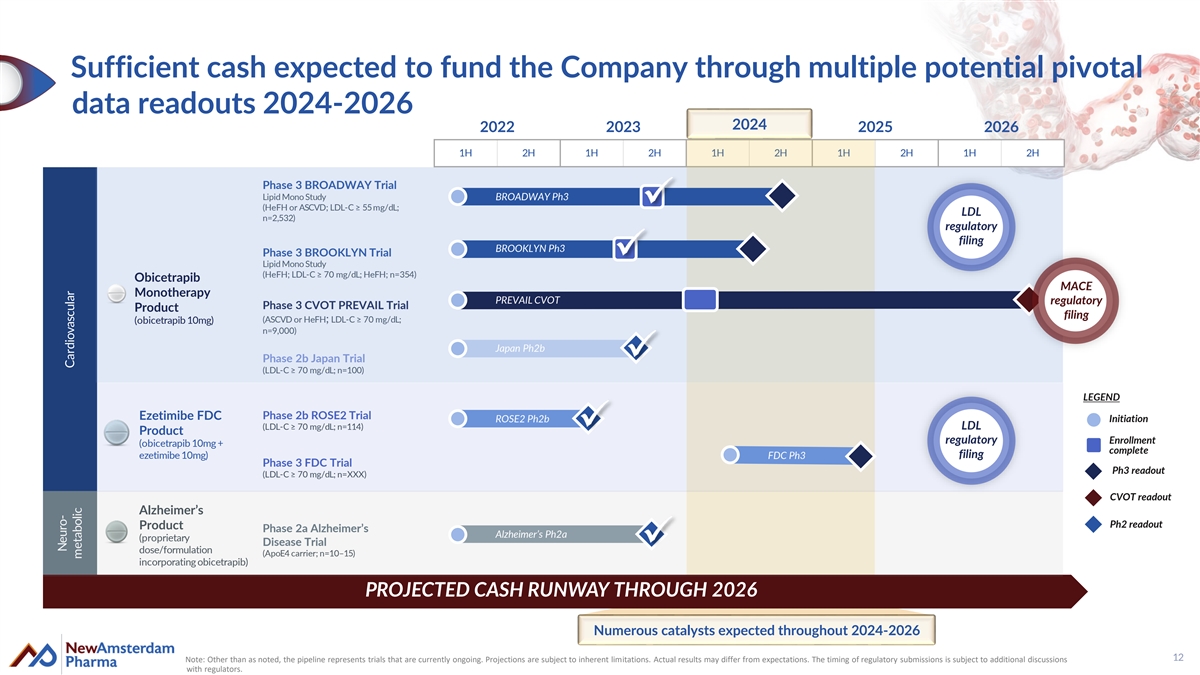
Sufficient cash expected to fund the Company through multiple potential pivotal data readouts 2024-2026 2024 2022 2023 2024 2025 2026 1H 2H 1H 2H 1H 2H 1H 2H 1H 2H Phase 3 BROADWAY Trial Lipid Mono Study BROADWAY Ph3 (HeFH or ASCVD; LDL-C ≥ 55 mg/dL; LDL n=2,532) regulatory filing BROOKLYN Ph3 Phase 3 BROOKLYN Trial Lipid Mono Study (HeFH; LDL-C ≥ 70 mg/dL; HeFH; n=354) Obicetrapib MACE Monotherapy PREVAIL CVOT regulatory Phase 3 CVOT PREVAIL Trial Product filing (ASCVD or HeFH; LDL-C ≥ 70 mg/dL; (obicetrapib 10mg) n=9,000) Japan Ph2b Phase 2b Japan Trial (LDL-C ≥ 70 mg/dL; n=100) LEGEND Ezetimibe FDC Phase 2b ROSE2 Trial ROSE2 Ph2b Initiation (LDL-C ≥ 70 mg/dL; n=114) LDL Product regulatory Enrollment (obicetrapib 10mg + complete ezetimibe 10mg) FDC Ph3 filing Phase 3 FDC Trial Ph3 readout (LDL-C ≥ 70 mg/dL; n=XXX) CVOT readout Alzheimer’s Ph2 readout Product Phase 2a Alzheimer’s Alzheimer’s Ph2a (proprietary Disease Trial dose/formulation (ApoE4 carrier; n=10–15) incorporating obicetrapib) PROJECTED CASH RUNWAY THROUGH 2026 Numerous catalysts expected throughout 2024-2026 12 Note: Other than as noted, the pipeline represents trials that are currently ongoing. Projections are subject to inherent limitations. Actual results may differ from expectations. The timing of regulatory submissions is subject to additional discussions with regulators. Neuro- Cardiovascular metabolic
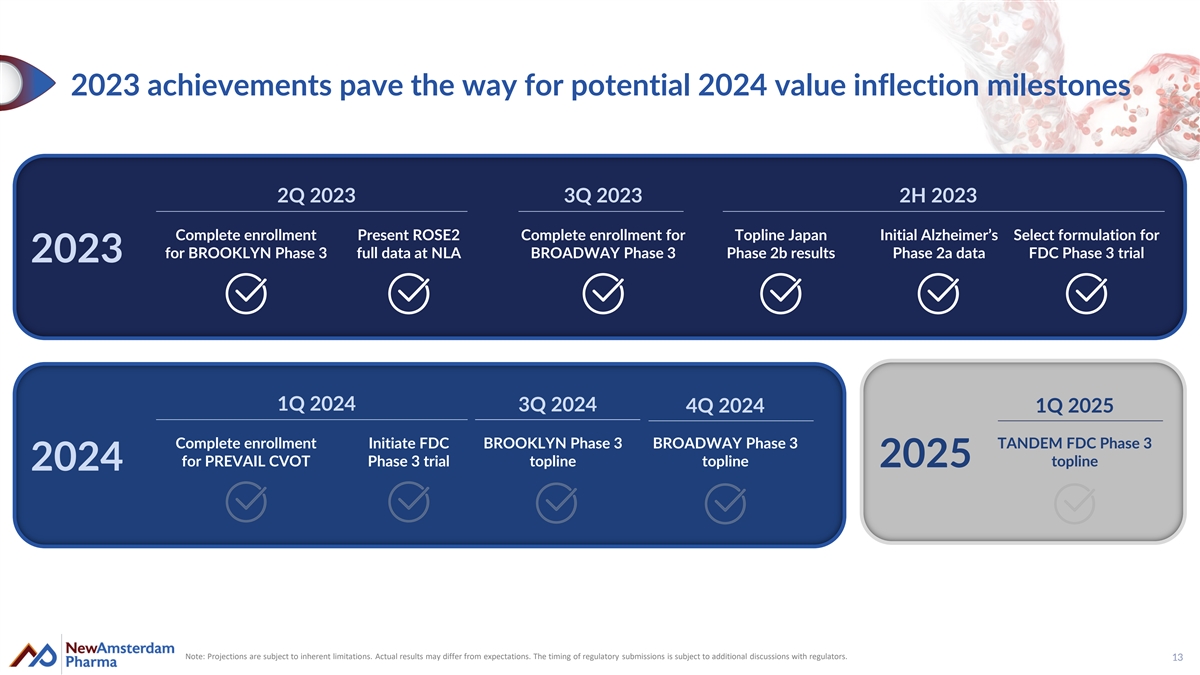
2023 achievements pave the way for potential 2024 value inflection milestones 2Q 2023 3Q 2023 2H 2023 Complete enrollment Present ROSE2 Complete enrollment for Topline Japan Initial Alzheimer’s Select formulation for for BROOKLYN Phase 3 full data at NLA BROADWAY Phase 3 Phase 2b results Phase 2a data FDC Phase 3 trial 2023 1Q 2024 3Q 2024 4Q 2024 1Q 2025 Complete enrollment Initiate FDC BROOKLYN Phase 3 BROADWAY Phase 3 TANDEM FDC Phase 3 for PREVAIL CVOT Phase 3 trial topline topline topline 2025 2024 Note: Projections are subject to inherent limitations. Actual results may differ from expectations. The timing of regulatory submissions is subject to additional discussions with regulators. 13
Obicetrapib for Cardiovascular Disease 14
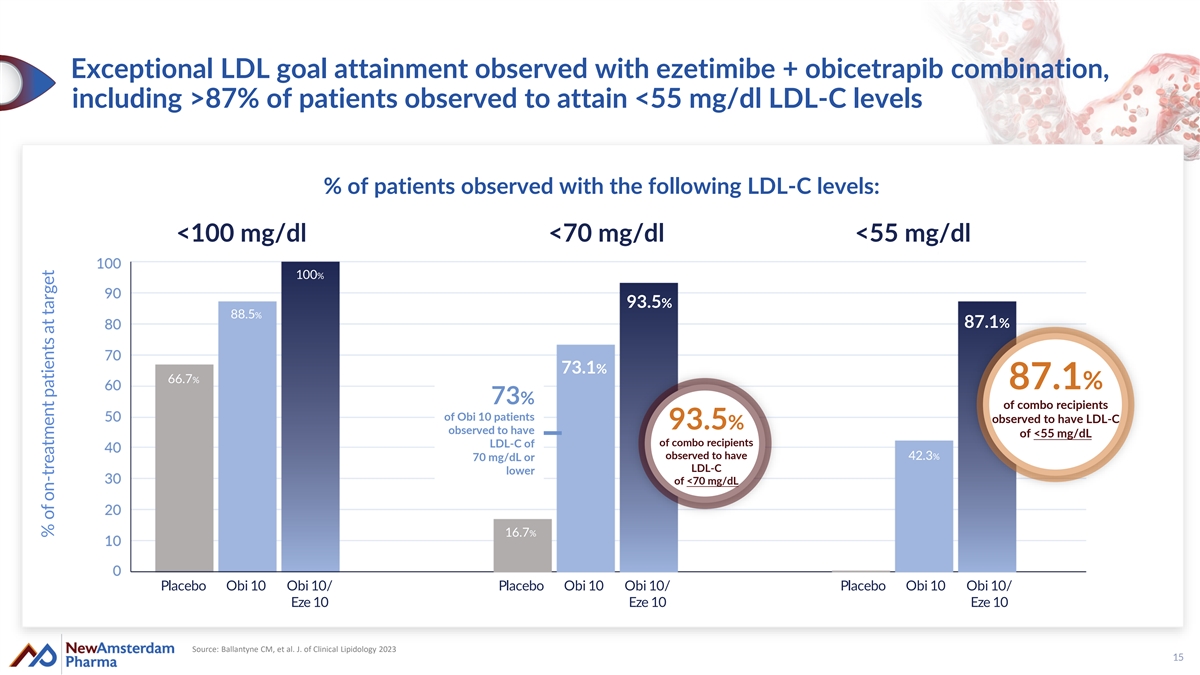
Exceptional LDL goal attainment observed with ezetimibe + obicetrapib combination, including >87% of patients observed to attain <55 mg/dl LDL-C levels % of patients observed with the following LDL-C levels: <100 mg/dl <70 mg/dl <55 mg/dl 100% 93.5% 88.5% 87.1% 73.1% 66.7% 87.1% 73% of combo recipients of Obi 10 patients observed to have LDL-C 93.5% observed to have of <55 mg/dL of combo recipients LDL-C of observed to have 42.3% 70 mg/dL or LDL-C lower of <70 mg/dL 16.7% Placebo Obi 10 Obi 10/ Placebo Obi 10 Obi 10/ Placebo Obi 10 Obi 10/ Eze 10 Eze 10 Eze 10 Source: Ballantyne CM, et al. J. of Clinical Lipidology 2023 15 % of on-treatment patients at target
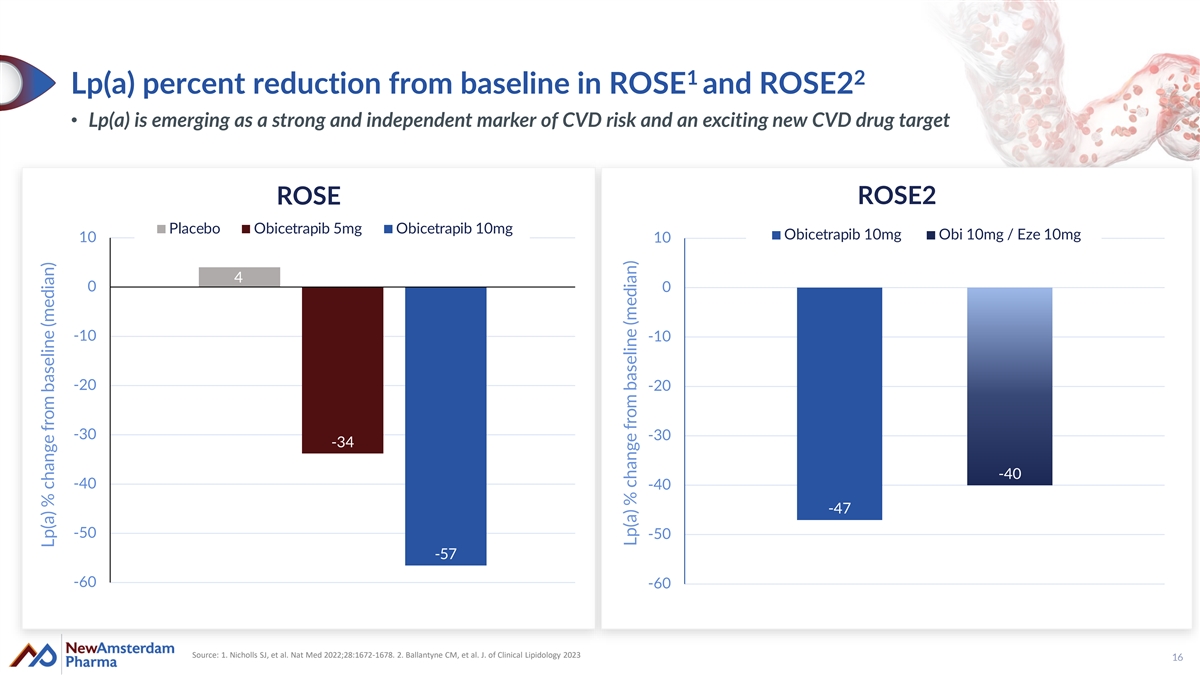
1 2 Lp(a) percent reduction from baseline in ROSE and ROSE2 • Lp(a) is emerging as a strong and independent marker of CVD risk and an exciting new CVD drug target ROSE2 ROSE Placebo Obicetrapib 5mg Obicetrapib 10mg Obicetrapib 10mg Obi 10mg / Eze 10mg 10 10 4 0 0 -10 -10 -20 -20 -30 -30 -34 -40 -40 -40 -47 -50 -50 -57 -60 -60 Source: 1. Nicholls SJ, et al. Nat Med 2022;28:1672-1678. 2. Ballantyne CM, et al. J. of Clinical Lipidology 2023 16 Lp(a) % change from baseline (median) Lp(a) % change from baseline (median)
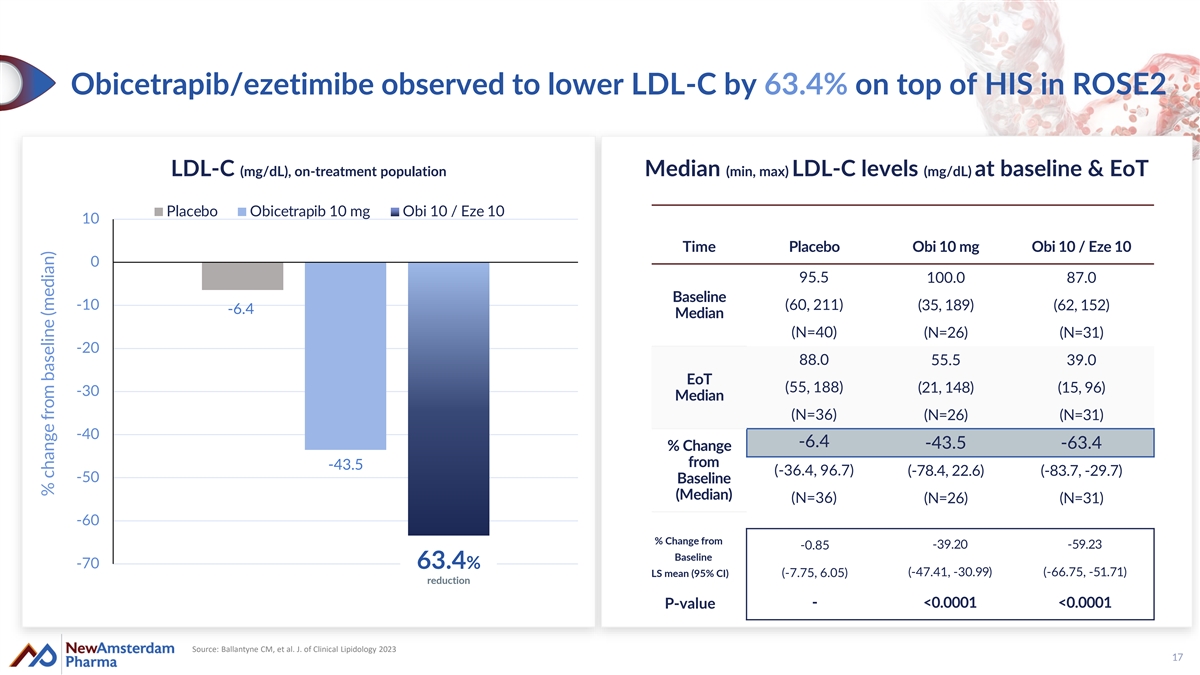
Obicetrapib/ezetimibe observed to lower LDL-C by 63.4% on top of HIS in ROSE2 LDL-C (mg/dL), on-treatment population Median (min, max) LDL-C levels (mg/dL) at baseline & EoT Placebo Obicetrapib 10 mg Obi 10 / Eze 10 10 Time Placebo Obi 10 mg Obi 10 / Eze 10 0 95.5 100.0 87.0 Baseline -10 (60, 211) (35, 189) (62, 152) -6.4 Median (N=40) (N=26) (N=31) -20 88.0 55.5 39.0 EoT (55, 188) (21, 148) (15, 96) -30 Median (N=36) (N=26) (N=31) -40 -6.4 -43.5 -63.4 % Change from -43.5 (-36.4, 96.7) (-78.4, 22.6) (-83.7, -29.7) -50 Baseline (Median) (N=36) (N=26) (N=31) -60 % Change from -39.20 -59.23 -0.85 Baseline -70 63.4% (-47.41, -30.99) (-66.75, -51.71) LS mean (95% CI) (-7.75, 6.05) reduction - <0.0001 <0.0001 P-value Source: Ballantyne CM, et al. J. of Clinical Lipidology 2023 17 % change from baseline (median)
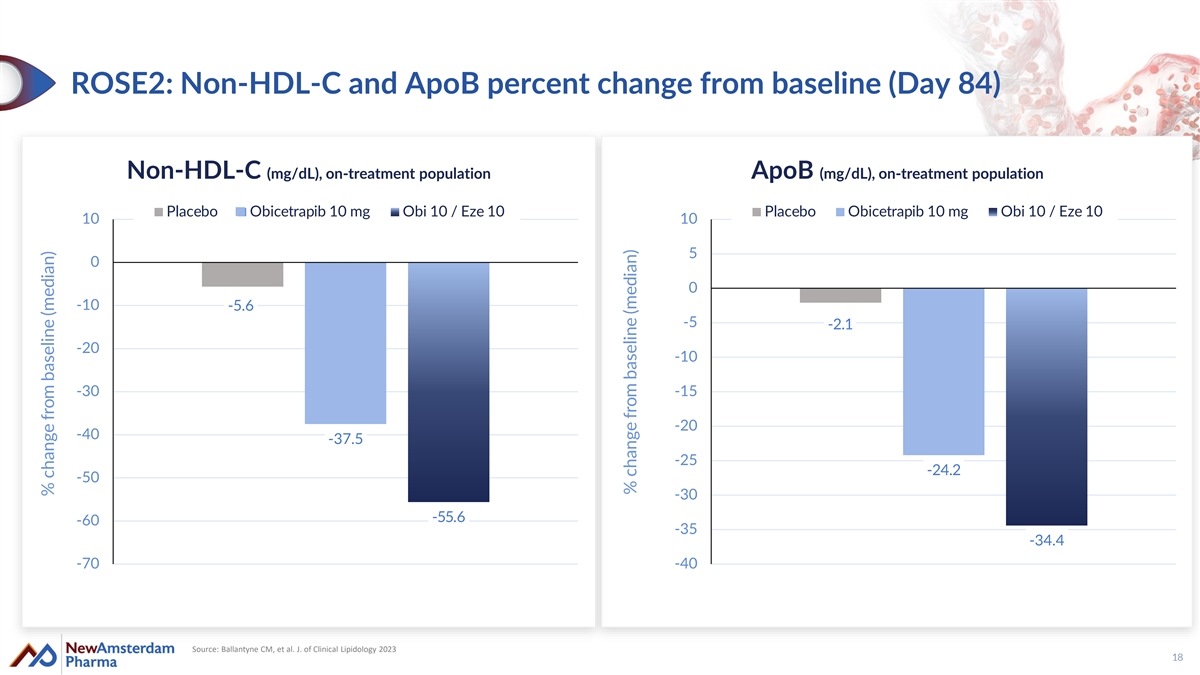
ROSE2: Non-HDL-C and ApoB percent change from baseline (Day 84) Non-HDL-C (mg/dL), on-treatment population ApoB (mg/dL), on-treatment population Placebo Obicetrapib 10 mg Obi 10 / Eze 10 Placebo Obicetrapib 10 mg Obi 10 / Eze 10 10 10 5 0 0 -10 -5.6 -5 -2.1 -20 -10 -30 -15 -20 -40 -37.5 -25 -24.2 -50 -30 -55.6 -60 -35 -34.4 -70 -40 Source: Ballantyne CM, et al. J. of Clinical Lipidology 2023 18 % change from baseline (median) % change from baseline (median)
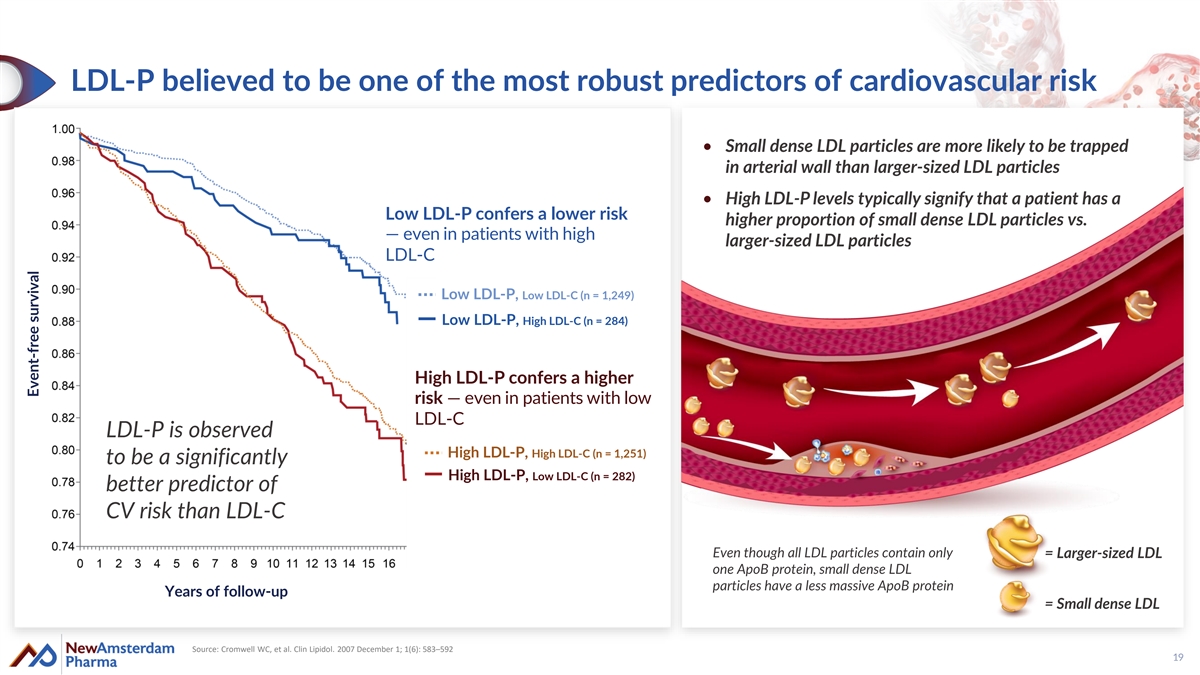
LDL-P believed to be one of the most robust predictors of cardiovascular risk • Small dense LDL particles are more likely to be trapped in arterial wall than larger-sized LDL particles • High LDL-P levels typically signify that a patient has a Low LDL-P confers a lower risk higher proportion of small dense LDL particles vs. — even in patients with high larger-sized LDL particles LDL-C Low LDL-P, Low LDL-C (n = 1,249) Low LDL-P, High LDL-C (n = 284) High LDL-P confers a higher risk — even in patients with low LDL-C LDL-P is observed High LDL-P, High LDL-C (n = 1,251) to be a significantly High LDL-P, Low LDL-C (n = 282) better predictor of CV risk than LDL-C Even though all LDL particles contain only = Larger-sized LDL one ApoB protein, small dense LDL particles have a less massive ApoB protein Years of follow-up = Small dense LDL Source: Cromwell WC, et al. Clin Lipidol. 2007 December 1; 1(6): 583–592 19 Event-free survival
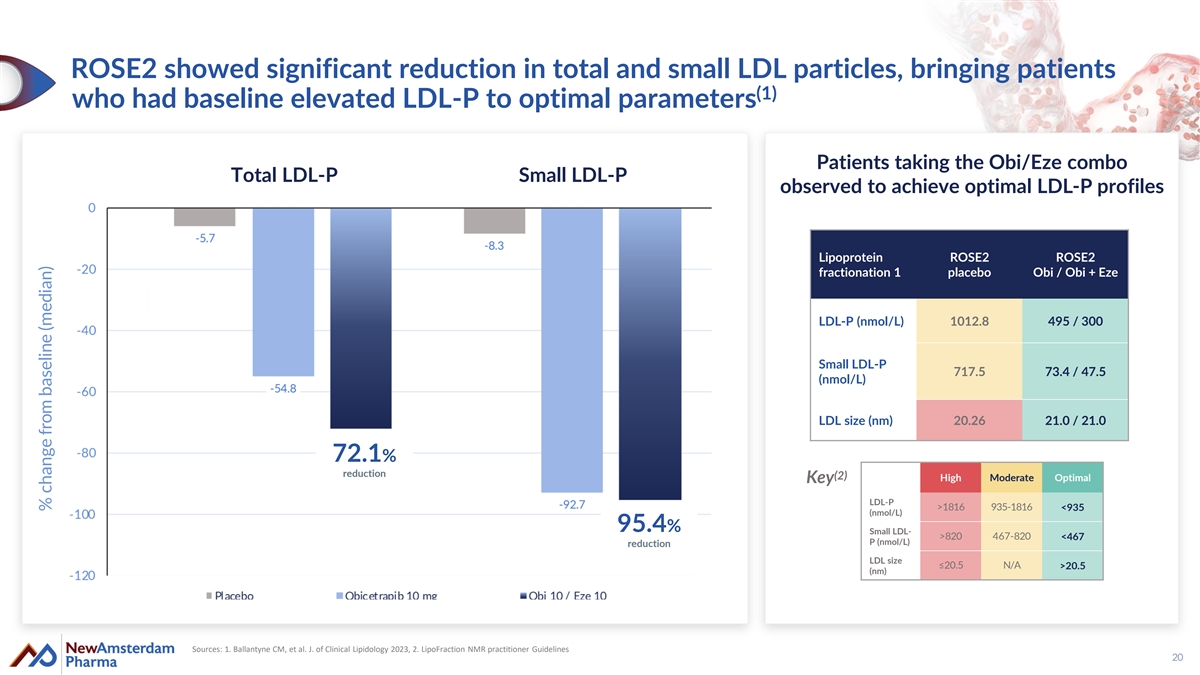
ROSE2 showed significant reduction in total and small LDL particles, bringing patients (1) who had baseline elevated LDL-P to optimal parameters Patients taking the Obi/Eze combo Total LDL-P Small LDL-P observed to achieve optimal LDL-P profiles Lipoprotein ROSE2 ROSE2 fractionation 1 placebo Obi / Obi + Eze LDL-P (nmol/L) 1012.8 495 / 300 Small LDL-P 717.5 73.4 / 47.5 (nmol/L) LDL size (nm) 20.26 21.0 / 21.0 72.1% reduction (2) High Moderate Optimal Key LDL-P >1816 935-1816 <935 (nmol/L) 95.4% Small LDL- >820 467-820 <467 P (nmol/L) reduction LDL size ≤20.5 N/A >20.5 (nm) Sources: 1. Ballantyne CM, et al. J. of Clinical Lipidology 2023, 2. LipoFraction NMR practitioner Guidelines 20 % change from baseline (median)
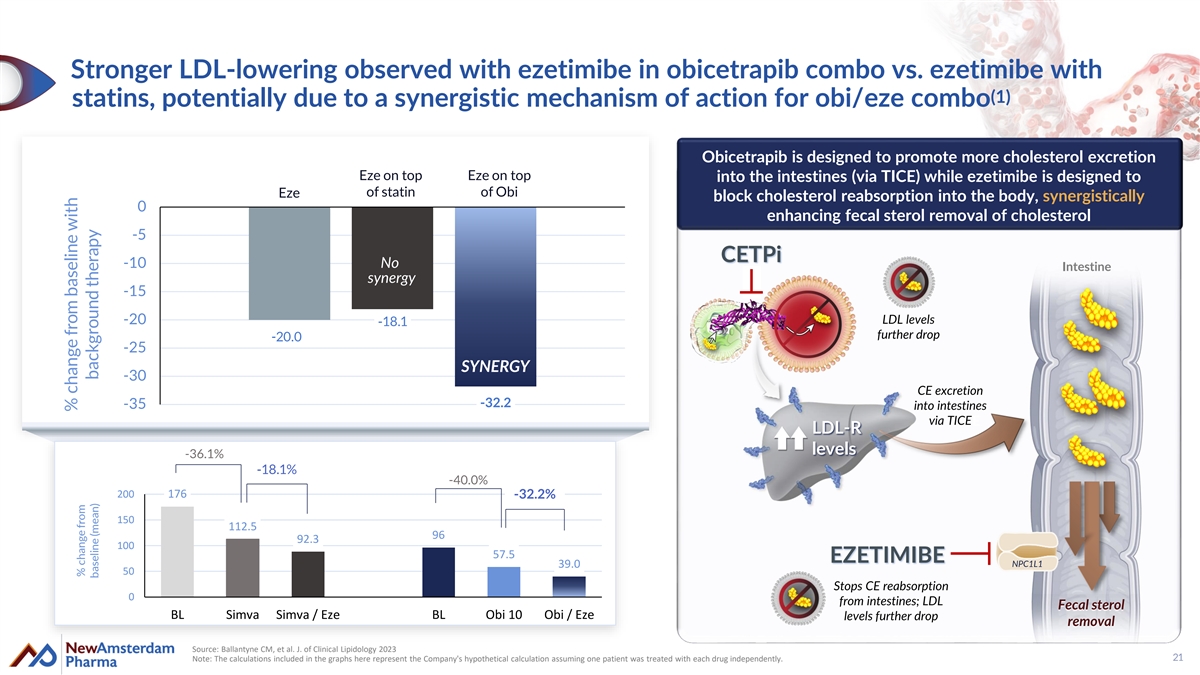
Stronger LDL-lowering observed with ezetimibe in obicetrapib combo vs. ezetimibe with (1) statins, potentially due to a synergistic mechanism of action for obi/eze combo Obicetrapib is designed to promote more cholesterol excretion Eze on top Eze on top into the intestines (via TICE) while ezetimibe is designed to of statin of Obi Eze block cholesterol reabsorption into the body, synergistically 0 enhancing fecal sterol removal of cholesterol -5 CETPi -10 No Intestine synergy -15 LDL levels -20 -18.1 further drop -20.0 -25 SYNERGY -30 CE excretion -32.2 -35 into intestines via TICE TICE LDL-R levels -36.1% -18.1% -40.0% 200 176 -32.2% 150 112.5 96 92.3 100 57.5 EZETIMIBE 39.0 NPC1L1 50 Stops CE reabsorption 0 from intestines; LDL Fecal sterol BL Simva Simva / Eze BL Obi 10 Obi / Eze levels further drop removal Source: Ballantyne CM, et al. J. of Clinical Lipidology 2023 Note: The calculations included in the graphs here represent the Company's hypothetical calculation assuming one patient was treated with each drug independently. 21 % change from baseline with % change from baseline (mean) background therapy
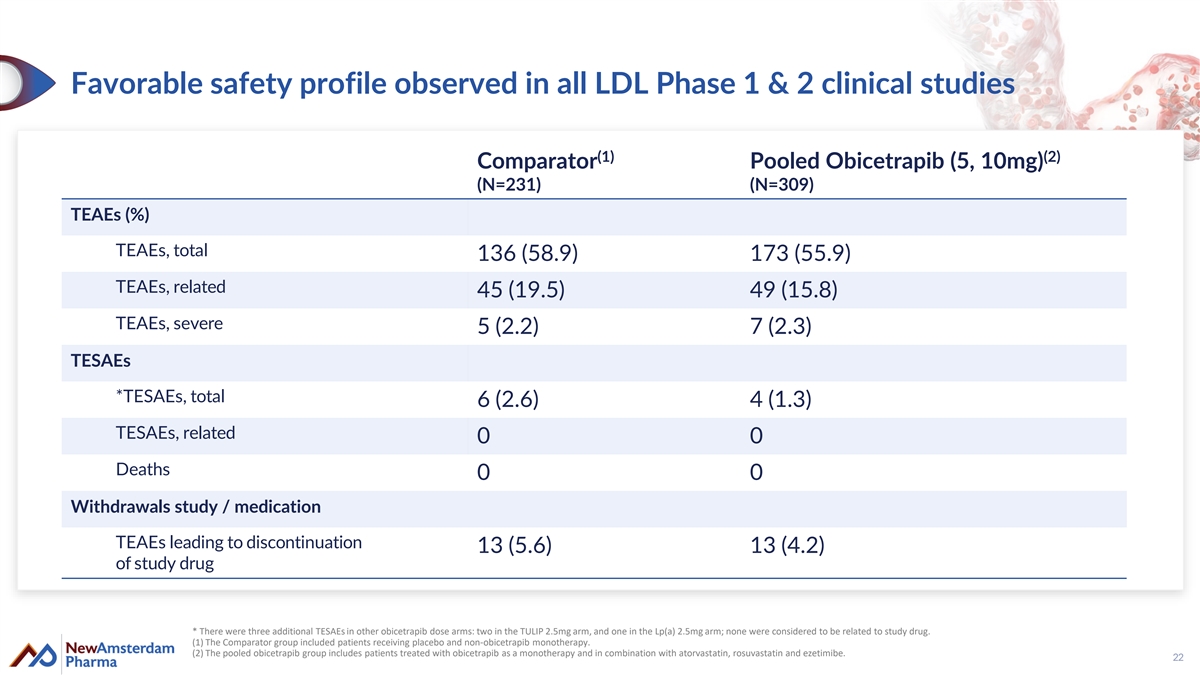
Favorable safety profile observed in all LDL Phase 1 & 2 clinical studies (1) (2) Comparator Pooled Obicetrapib (5, 10mg) (N=231) (N=309) TEAEs (%) TEAEs, total 136 (58.9) 173 (55.9) TEAEs, related 45 (19.5) 49 (15.8) TEAEs, severe 5 (2.2) 7 (2.3) TESAEs *TESAEs, total 6 (2.6) 4 (1.3) TESAEs, related 0 0 Deaths 0 0 Withdrawals study / medication TEAEs leading to discontinuation 13 (5.6) 13 (4.2) of study drug * There were three additional TESAEs in other obicetrapib dose arms: two in the TULIP 2.5mg arm, and one in the Lp(a) 2.5mg arm; none were considered to be related to study drug. (1) The Comparator group included patients receiving placebo and non-obicetrapib monotherapy. (2) The pooled obicetrapib group includes patients treated with obicetrapib as a monotherapy and in combination with atorvastatin, rosuvastatin and ezetimibe. 22
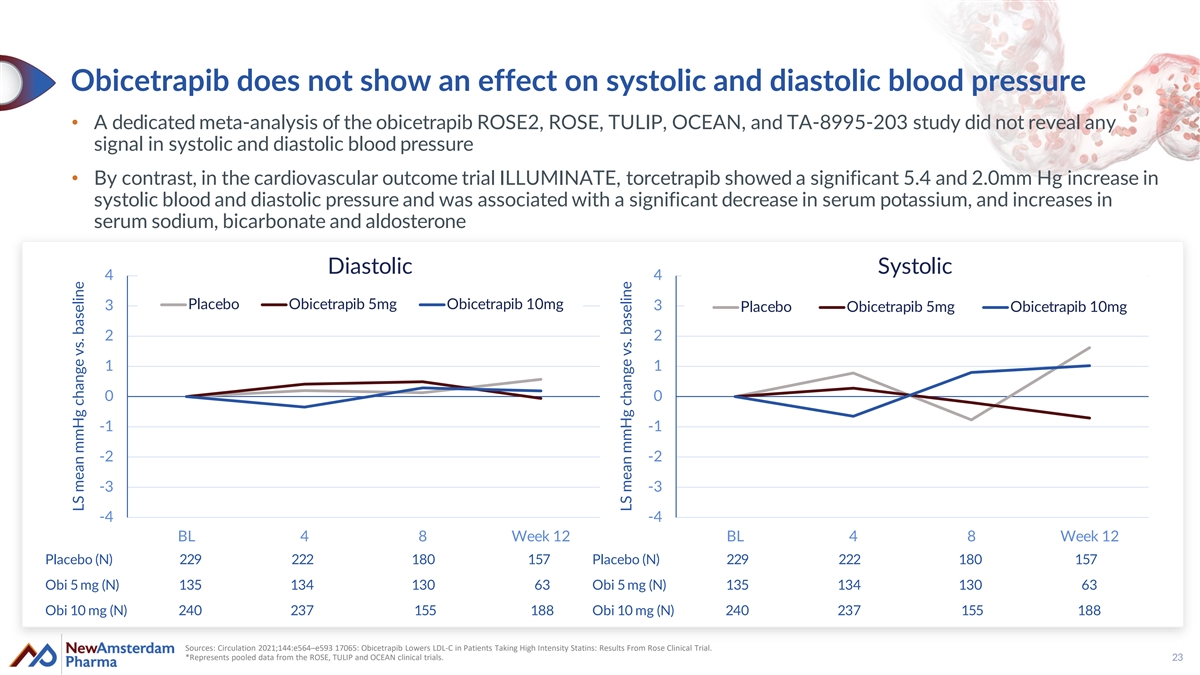
Obicetrapib does not show an effect on systolic and diastolic blood pressure • A dedicated meta-analysis of the obicetrapib ROSE2, ROSE, TULIP, OCEAN, and TA-8995-203 study did not reveal any signal in systolic and diastolic blood pressure • By contrast, in the cardiovascular outcome trial ILLUMINATE, torcetrapib showed a significant 5.4 and 2.0mm Hg increase in systolic blood and diastolic pressure and was associated with a significant decrease in serum potassium, and increases in serum sodium, bicarbonate and aldosterone Diastolic Systolic 4 4 3 Placebo Obicetrapib 5mg Obicetrapib 10mg 3 Placebo Obicetrapib 5mg Obicetrapib 10mg 2 2 1 1 0 0 -1 -1 -2 -2 -3 -3 -4 -4 BL 4 8 Week 12 BL 4 8 Week 12 Placebo (N) 229 222 180 157 Placebo (N) 229 222 180 157 Obi 5 mg (N) 135 134 130 63 Obi 5 mg (N) 135 134 130 63 Obi 10 mg (N) 240 237 155 188 Obi 10 mg (N) 240 237 155 188 Sources: Circulation 2021;144:e564–e593 17065: Obicetrapib Lowers LDL-C in Patients Taking High Intensity Statins: Results From Rose Clinical Trial. *Represents pooled data from the ROSE, TULIP and OCEAN clinical trials. 23 LS mean mmHg change vs. baseline LS mean mmHg change vs. baseline
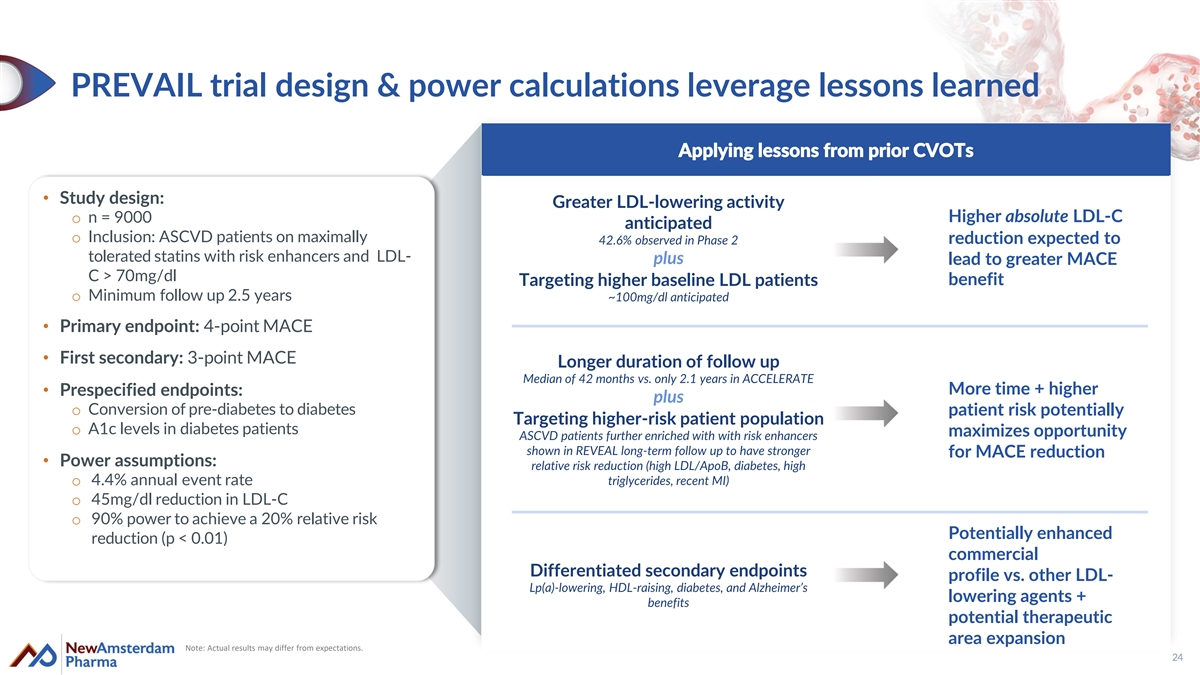
PREVAIL trial design & power calculations leverage lessons learned Applying lessons from prior CVOTs • Study design: Greater LDL-lowering activity o n = 9000 Higher absolute LDL-C anticipated o Inclusion: ASCVD patients on maximally reduction expected to 42.6% observed in Phase 2 tolerated statins with risk enhancers and LDL- plus lead to greater MACE C > 70mg/dl benefit Targeting higher baseline LDL patients o Minimum follow up 2.5 years ~100mg/dl anticipated • Primary endpoint: 4-point MACE • First secondary: 3-point MACE Longer duration of follow up Median of 42 months vs. only 2.1 years in ACCELERATE More time + higher • Prespecified endpoints: plus o Conversion of pre-diabetes to diabetes patient risk potentially Targeting higher-risk patient population o A1c levels in diabetes patients maximizes opportunity ASCVD patients further enriched with with risk enhancers shown in REVEAL long-term follow up to have stronger for MACE reduction • Power assumptions: relative risk reduction (high LDL/ApoB, diabetes, high o 4.4% annual event rate triglycerides, recent MI) o 45mg/dl reduction in LDL-C o 90% power to achieve a 20% relative risk Potentially enhanced reduction (p < 0.01) commercial Differentiated secondary endpoints profile vs. other LDL- Lp(a)-lowering, HDL-raising, diabetes, and Alzheimer’s lowering agents + benefits potential therapeutic area expansion Note: Actual results may differ from expectations. 24

Target biology and class overview: Key lessons learned 25
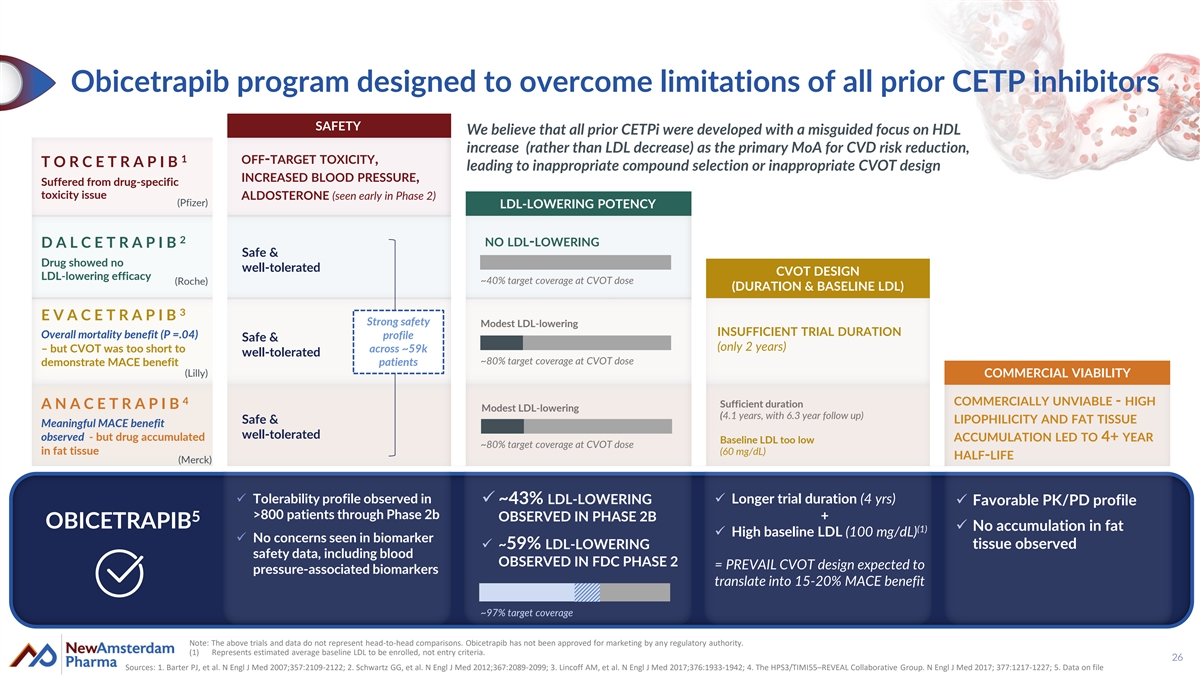
Obicetrapib program designed to overcome limitations of all prior CETP inhibitors SAFETY We believe that all prior CETPi were developed with a misguided focus on HDL increase (rather than LDL decrease) as the primary MoA for CVD risk reduction, 1 OFF-TARGET TOXICITY, T O R C E T R A P I B leading to inappropriate compound selection or inappropriate CVOT design INCREASED BLOOD PRESSURE, Suffered from drug-specific toxicity issue ALDOSTERONE (seen early in Phase 2) (Pfizer) LDL-LOWERING POTENCY 2 NO LDL-LOWERING D A L C E T R A P I B Safe & Drug showed no well-tolerated CVOT DESIGN LDL-lowering efficacy ~40% target coverage at CVOT dose (Roche) (DURATION & BASELINE LDL) 3 E V A C E T R A P I B Strong safety Modest LDL-lowering INSUFFICIENT TRIAL DURATION Overall mortality benefit (P =.04) profile Safe & (only 2 years) – but CVOT was too short to across ~59k well-tolerated ~80% target coverage at CVOT dose demonstrate MACE benefit patients (Lilly) COMMERCIAL VIABILITY 4 COMMERCIALLY UNVIABLE - HIGH Sufficient duration A N A C E T R A P I B Modest LDL-lowering (4.1 years, with 6.3 year follow up) Safe & LIPOPHILICITY AND FAT TISSUE Meaningful MACE benefit well-tolerated observed - but drug accumulated ACCUMULATION LED TO 4+ YEAR Baseline LDL too low ~80% target coverage at CVOT dose in fat tissue (60 mg/dL) HALF-LIFE (Merck) ✓ Tolerability profile observed in ✓ ~43% LDL-LOWERING ✓ Longer trial duration (4 yrs) ✓ Favorable PK/PD profile >800 patients through Phase 2b OBSERVED IN PHASE 2B + 5 OBICETRAPIB ✓ No accumulation in fat (1) ✓ High baseline LDL (100 mg/dL) ✓ No concerns seen in biomarker tissue observed ✓ ~59% LDL-LOWERING safety data, including blood OBSERVED IN FDC PHASE 2 = PREVAIL CVOT design expected to pressure-associated biomarkers translate into 15-20% MACE benefit ~97% target coverage Note: The above trials and data do not represent head-to-head comparisons. Obicetrapib has not been approved for marketing by any regulatory authority. (1) Represents estimated average baseline LDL to be enrolled, not entry criteria. 26 Sources: 1. Barter PJ, et al. N Engl J Med 2007;357:2109-2122; 2. Schwartz GG, et al. N Engl J Med 2012;367:2089-2099; 3. Lincoff AM, et al. N Engl J Med 2017;376:1933-1942; 4. The HPS3/TIMI55–REVEAL Collaborative Group. N Engl J Med 2017; 377:1217-1227; 5. Data on file
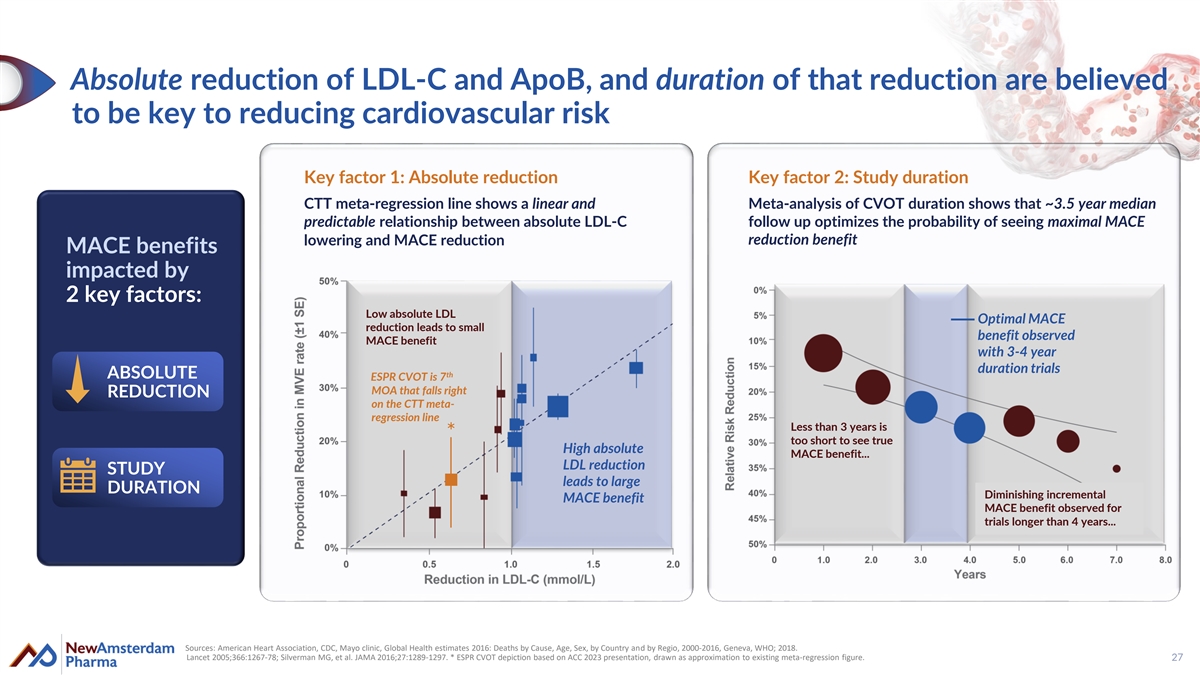
Absolute reduction of LDL-C and ApoB, and duration of that reduction are believed to be key to reducing cardiovascular risk Key factor 1: Absolute reduction Key factor 2: Study duration CTT meta-regression line shows a linear and Meta-analysis of CVOT duration shows that ~3.5 year median predictable relationship between absolute LDL-C follow up optimizes the probability of seeing maximal MACE lowering and MACE reduction reduction benefit MACE MACE benefits benefits impac impac ted ted by by 2 2 key key factors: factors: Low absolute LDL Optimal MACE reduction leads to small benefit observed MACE benefit with 3-4 year duration trials th ABSOLUTE ESPR CVOT is 7 MOA that falls right REDUCTION on the CTT meta- regression line Less than 3 years is * too short to see true High absolute MACE benefit... LDL reduction STUDY leads to large DURATION Diminishing incremental MACE benefit MACE benefit observed for trials longer than 4 years... Sources: American Heart Association, CDC, Mayo clinic, Global Health estimates 2016: Deaths by Cause, Age, Sex, by Country and by Regio, 2000-2016, Geneva, WHO; 2018. Lancet 2005;366:1267-78; Silverman MG, et al. JAMA 2016;27:1289-1297. * ESPR CVOT depiction based on ACC 2023 presentation, drawn as approximation to existing meta-regression figure. 27
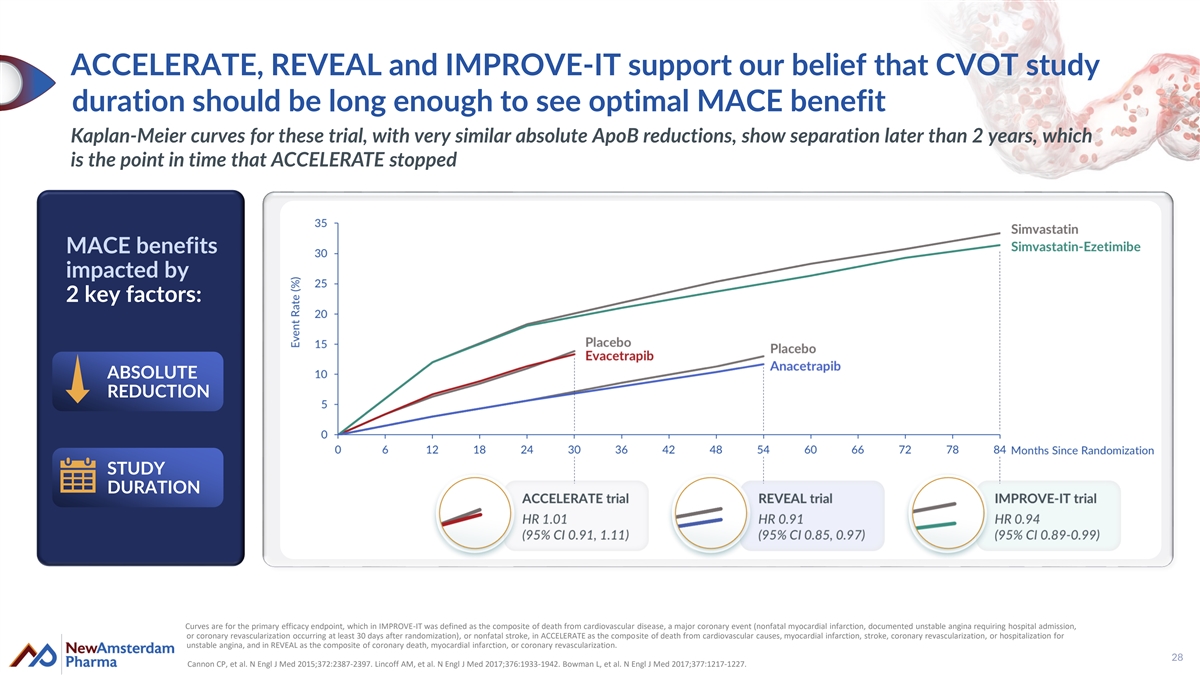
ACCELERATE, REVEAL and IMPROVE-IT support our belief that CVOT study duration should be long enough to see optimal MACE benefit Kaplan-Meier curves for these trial, with very similar absolute ApoB reductions, show separation later than 2 years, which is the point in time that ACCELERATE stopped MACE benefits impacted by 2 key factors: ABSOLUTE REDUCTION STUDY DURATION Curves are for the primary efficacy endpoint, which in IMPROVE-IT was defined as the composite of death from cardiovascular disease, a major coronary event (nonfatal myocardial infarction, documented unstable angina requiring hospital admission, or coronary revascularization occurring at least 30 days after randomization), or nonfatal stroke, in ACCELERATE as the composite of death from cardiovascular causes, myocardial infarction, stroke, coronary revascularization, or hospitalization for unstable angina, and in REVEAL as the composite of coronary death, myocardial infarction, or coronary revascularization. 28 Cannon CP, et al. N Engl J Med 2015;372:2387-2397. Lincoff AM, et al. N Engl J Med 2017;376:1933-1942. Bowman L, et al. N Engl J Med 2017;377:1217-1227.
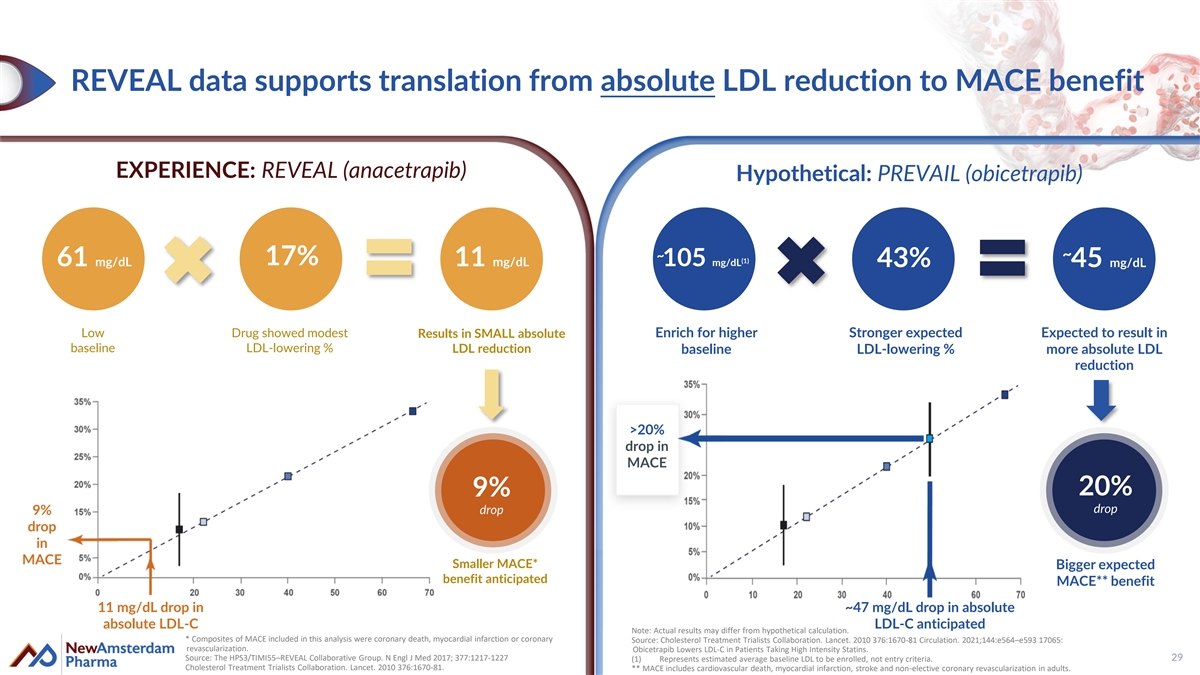
REVEAL data supports translation from absolute LDL reduction to MACE benefit EXPERIENCE: REVEAL (anacetrapib) Hypothetical: PREVAIL (obicetrapib) ~ ~ (1) 17% 61 mg/dL 11 mg/dL 105 mg/dL 43% 45 mg/dL Low Drug showed modest Results in SMALL absolute Enrich for higher Stronger expected Expected to result in baseline LDL-lowering % LDL reduction baseline LDL-lowering % more absolute LDL reduction >20% drop in MACE 20% 9% drop drop 9% drop in MACE Smaller MACE* Bigger expected benefit anticipated MACE** benefit 11 mg/dL drop in ~47 mg/dL drop in absolute absolute LDL-C LDL-C anticipated Note: Actual results may differ from hypothetical calculation. * Composites of MACE included in this analysis were coronary death, myocardial infarction or coronary Source: Cholesterol Treatment Trialists Collaboration. Lancet. 2010 376:1670-81 Circulation. 2021;144:e564–e593 17065: revascularization. Obicetrapib Lowers LDL-C in Patients Taking High Intensity Statins. Source: The HPS3/TIMI55–REVEAL Collaborative Group. N Engl J Med 2017; 377:1217-1227 29 (1) Represents estimated average baseline LDL to be enrolled, not entry criteria. Cholesterol Treatment Trialists Collaboration. Lancet. 2010 376:1670-81. ** MACE includes cardiovascular death, myocardial infarction, stroke and non-elective coronary revascularization in adults.
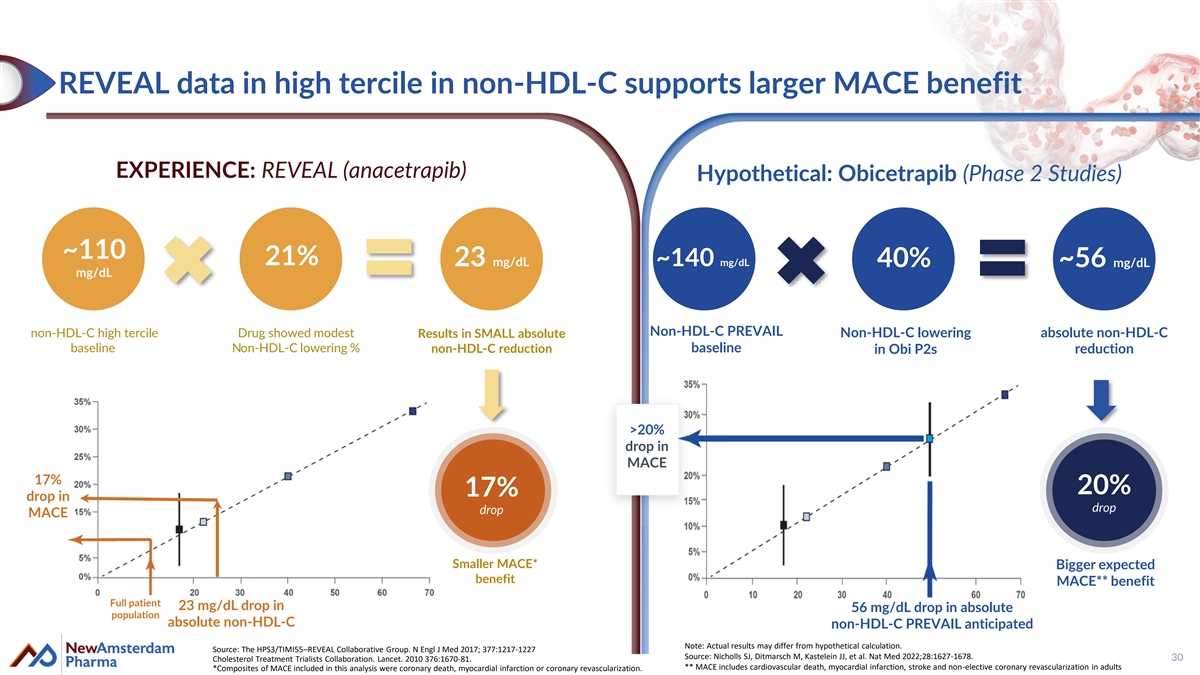
REVEAL data in high tercile in non-HDL-C supports larger MACE benefit EXPERIENCE: REVEAL (anacetrapib) Hypothetical: Obicetrapib (Phase 2 Studies) ~110 21% 23 mg/dL ~140 mg/dL 40% ~56 mg/dL mg/dL Non-HDL-C PREVAIL non-HDL-C high tercile Drug showed modest Results in SMALL absolute Non-HDL-C lowering absolute non-HDL-C baseline Non-HDL-C lowering % non-HDL-C reduction baseline in Obi P2s reduction >20% drop in MACE 17% 20% 17% drop in drop drop MACE Smaller MACE* Bigger expected benefit MACE** benefit Full patient 23 mg/dL drop in 56 mg/dL drop in absolute population absolute non-HDL-C non-HDL-C PREVAIL anticipated Note: Actual results may differ from hypothetical calculation. Source: The HPS3/TIMI55–REVEAL Collaborative Group. N Engl J Med 2017; 377:1217-1227 Source: Nicholls SJ, Ditmarsch M, Kastelein JJ, et al. Nat Med 2022;28:1627-1678. 30 Cholesterol Treatment Trialists Collaboration. Lancet. 2010 376:1670-81. ** MACE includes cardiovascular death, myocardial infarction, stroke and non-elective coronary revascularization in adults *Composites of MACE included in this analysis were coronary death, myocardial infarction or coronary revascularization.
Obicetrapib and Alzheimer’s Disease 31
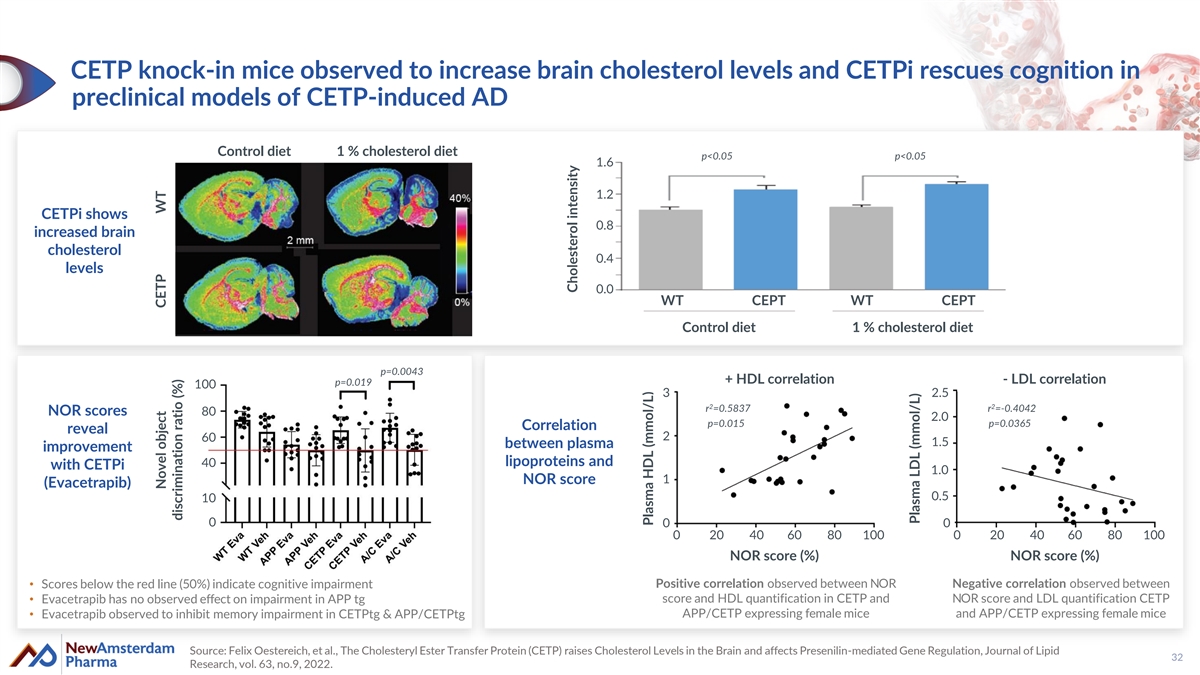
CETP knock-in mice observed to increase brain cholesterol levels and CETPi rescues cognition in preclinical models of CETP-induced AD Control diet 1 % cholesterol diet p<0.05 p<0.05 1.6 1.2 CETPi shows 0.8 increased brain cholesterol 0.4 levels 0.0 WT CEPT WT CEPT Control diet 1 % cholesterol diet p=0.0043 + HDL correlation - LDL correlation p=0.019 100 3 2.5 2 2 r =0.5837 r =-0.4042 NOR scores 80 2.0 p=0.015 p=0.0365 Correlation reveal 2 60 1.5 between plasma improvement lipoproteins and 40 with CETPi 1.0 NOR score 1 (Evacetrapib) 0.5 10 0 0 0 0 20 40 60 80 100 0 20 40 60 80 100 NOR score (%) NOR score (%) Positive correlation observed between NOR Negative correlation observed between • Scores below the red line (50%) indicate cognitive impairment score and HDL quantification in CETP and NOR score and LDL quantification CETP • Evacetrapib has no observed effect on impairment in APP tg APP/CETP expressing female mice and APP/CETP expressing female mice • Evacetrapib observed to inhibit memory impairment in CETPtg & APP/CETPtg Source: Felix Oestereich, et al., The Cholesteryl Ester Transfer Protein (CETP) raises Cholesterol Levels in the Brain and affects Presenilin-mediated Gene Regulation, Journal of Lipid 32 Research, vol. 63, no.9, 2022. Novel object CETP WT discrimination ratio (%) Cholesterol intensity Plasma HDL (mmol/L) Plasma LDL (mmol/L)
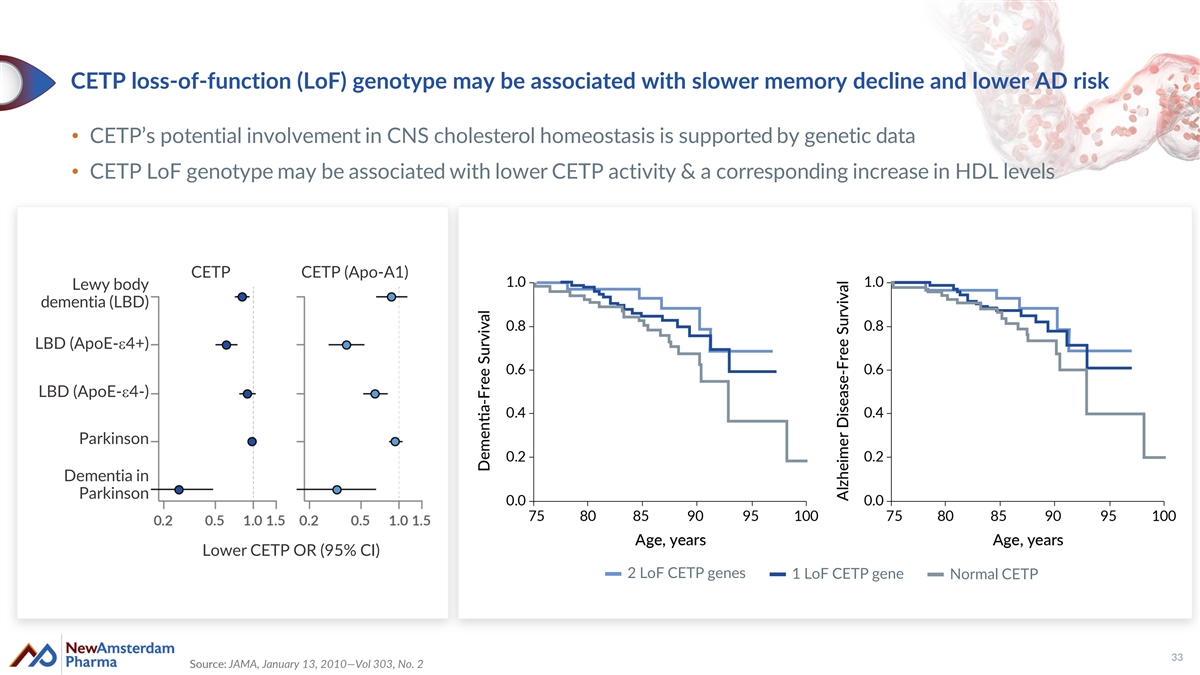
CETP loss-of-function (LoF) genotype may be associated with slower memory decline and lower AD risk • CETP’s potential involvement in CNS cholesterol homeostasis is supported by genetic data • CETP LoF genotype may be associated with lower CETP activity & a corresponding increase in HDL levels CETP CETP (Apo-A1) Lewy body dementia (LBD) LBD (ApoE-e4+) LBD (ApoE-e4-) Parkinson Dementia in Parkinson Lower CETP OR (95% CI) 2 LoF CETP genes 1 LoF CETP gene Normal CETP 33 Source: JAMA, January 13, 2010—Vol 303, No. 2
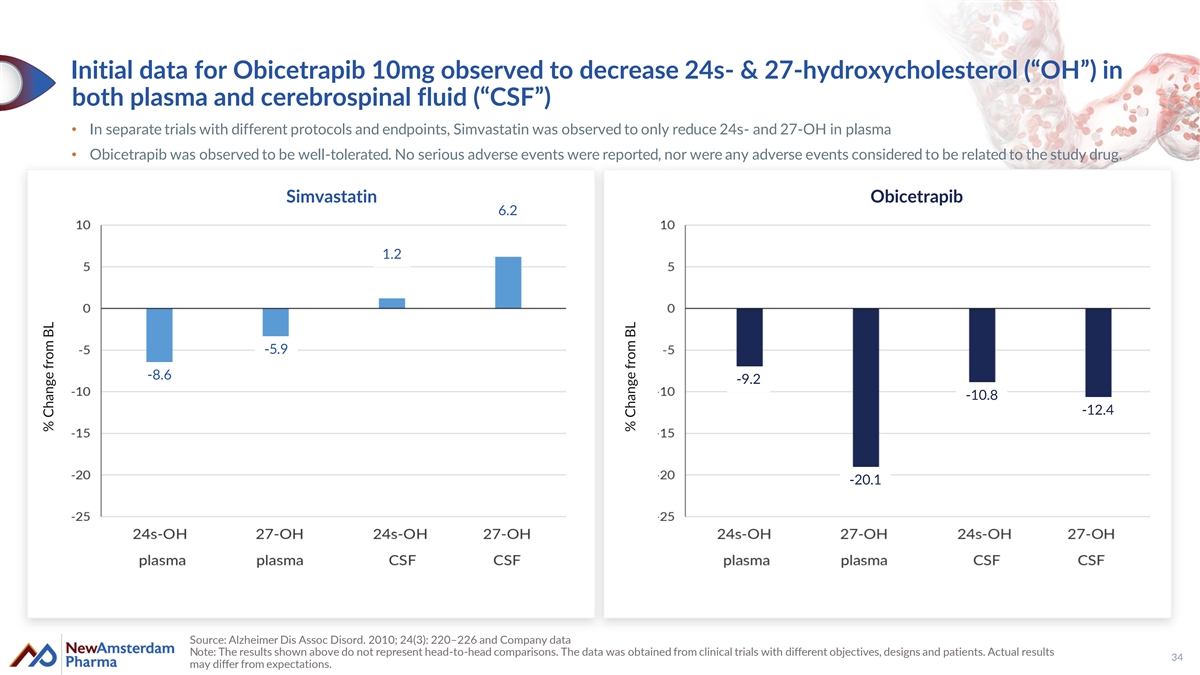
Initial data for Obicetrapib 10mg observed to decrease 24s- & 27-hydroxycholesterol (“OH”) in both plasma and cerebrospinal fluid (“CSF”) • In separate trials with different protocols and endpoints, Simvastatin was observed to only reduce 24s- and 27-OH in plasma • Obicetrapib was observed to be well-tolerated. No serious adverse events were reported, nor were any adverse events considered to be related to the study drug. Simvastatin Obicetrapib 6.2 1.2 -5.9 -8.6 -9.2 -10.8 -12.4 -20.1 Source: Alzheimer Dis Assoc Disord. 2010; 24(3): 220–226 and Company data Note: The results shown above do not represent head-to-head comparisons. The data was obtained from clinical trials with different objectives, designs and patients. Actual results 34 may differ from expectations. % Change from BL % Change from BL

Clinical events and exclusivity timelines 35
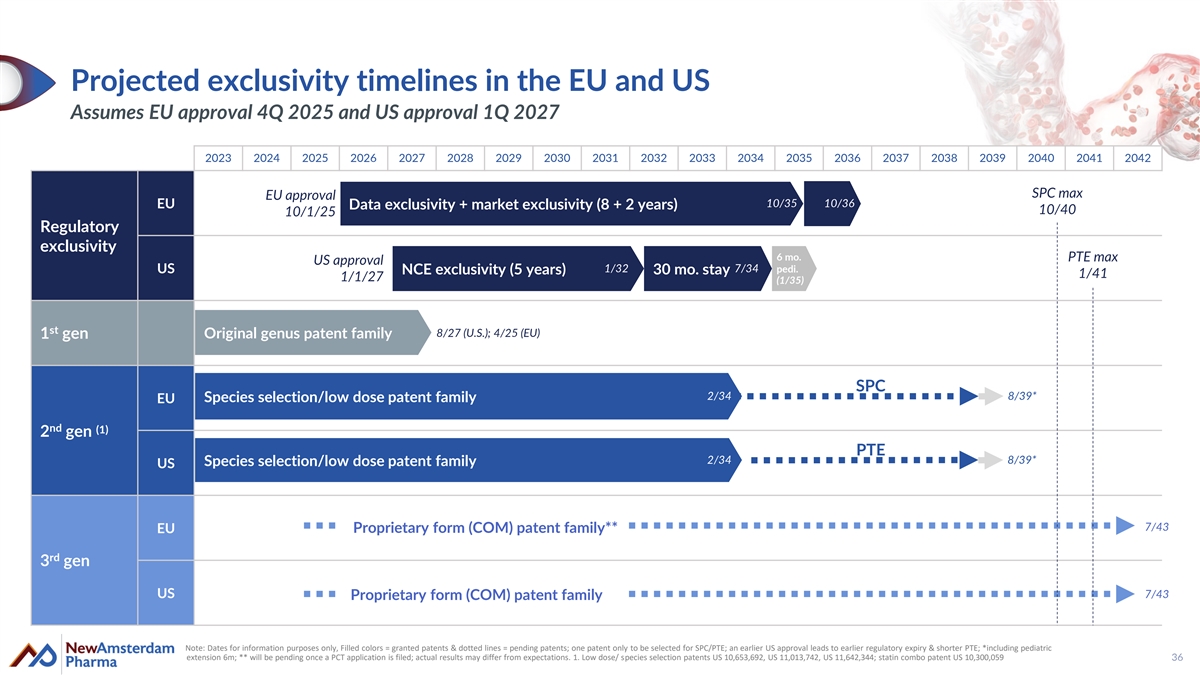
Projected exclusivity timelines in the EU and US Assumes EU approval 4Q 2025 and US approval 1Q 2027 2023 2024 2025 2026 2027 2028 2029 2030 2031 2032 2033 2034 2035 2036 2037 2038 2039 2040 2041 2042 SPC max EU approval 10/35 10/36 EU Data exclusivity + market exclusivity (8 + 2 years) 10/40 10/1/25 Regulatory exclusivity 6 mo. PTE max US approval 1/32 7/34 pedi. US NCE exclusivity (5 years) 30 mo. stay 1/41 1/1/27 (1/35) st 8/27 (U.S.); 4/25 (EU) 1 gen Original genus patent family SPC 2 2/ /3 34 4 8/39* Species selection/low dose patent family EU nd (1) 2 gen PTE 2/34 8/39* Species selection/low dose patent family US 7/43 Proprietary form (COM) patent family** EU rd 3 gen US 7/43 Proprietary form (COM) patent family Note: Dates for information purposes only, Filled colors = granted patents & dotted lines = pending patents; one patent only to be selected for SPC/PTE; an earlier US approval leads to earlier regulatory expiry & shorter PTE; *including pediatric extension 6m; ** will be pending once a PCT application is filed; actual results may differ from expectations. 1. Low dose/ species selection patents US 10,653,692, US 11,013,742, US 11,642,344; statin combo patent US 10,300,059 36

Expert cardiometabolic leadership supported by top investors Michael Davidson, M.D. John Kastelein, M.D. Douglas Kling William ‘BJ’ Jones Ian Somaiya Louise Kooij Marc Ditmarsch, M.D. CEO CSO COO CCO CFO CAO CDO WILLIAM SANDER JULIET JAMIE NICHOLAS LOU MICHAEL JOHN JOHN W. JANNEKE SEASONED LEWIS SLOOTWEG AUDET TOPPER DOWNING LANGE DAVIDSON KASTELEIN SMITHER VAN DER KAMP BOARD OF CEO, Managing Partner, Partner, Managing Partner, Partner, Partner, CEO, NewAmsterdam CSO, NewAmsterdam Independent Independent DIRECTORS: Insmed Forbion Forbion Frazier Life Sciences Bain Capital Asset Management Pharma Pharma Ventures BACKED BY TOP TIER INVESTORS: 37