InflaRx Provides Update on Phase 3 Data Analyses
for Vilobelimab in Pyoderma Gangrenosum
| • |
While the Phase 3 trial of vilobelimab in ulcerative pyoderma gangrenosum (PG) was terminated earlier this year due to futility regarding its prespecified primary endpoint (as previously
disclosed), subsequent post-hoc analyses suggest a positive trend in favor of vilobelimab, with signals indicating a potentially consistent treatment effect
|
| • |
InflaRx anticipates meeting with the FDA to determine a potential development path forward in PG, which the Company anticipates would only be conducted in collaboration with a partner
|
Jena, Germany, December 30, 2025 – InflaRx N.V. (Nasdaq: IFRX), a biopharmaceutical company pioneering anti-inflammatory therapeutics by targeting the complement system, today outlined multiple data analyses of the Phase 3 study for vilobelimab in pyoderma gangrenosum
(PG), which was terminated earlier this year after an Independent Data Monitoring Committee (IDMC) recommended the trial be stopped early due to futility. The analyses disclosed today include the primary intent-to-treat analysis and several
post-hoc analyses on the 54 patients enrolled in the trial at the time of study termination.
Prof. Niels C. Riedemann, Chief Executive Officer and Founder of
InflaRx, said: “Our Phase 3 study was the first randomized placebo-controlled study in pyoderma gangrenosum using complete target ulcer closure on two consecutive visits as a stringent primary clinical
endpoint, which has not been tested before in this rare disease. Our in-depth data analysis reveals signals of efficacy, particularly regarding ulcer volume reduction, which further supports the potential role of the C5a/C5aR pathway in certain
neutrophilic skin diseases, such as PG. Depending on the outcome of our anticipated FDA interactions, our results may provide an opportunity to advance development in collaboration with a partner, which we may pursue in the future.”
Results of Phase 3 data analysis
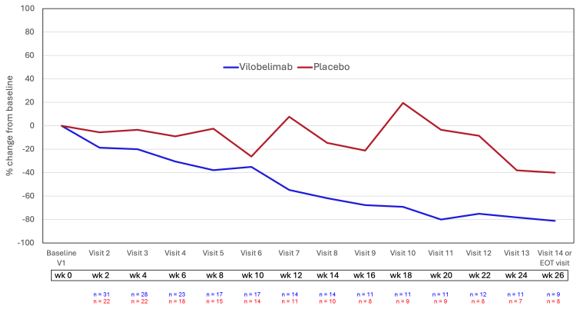
The Phase 3 study had recruited a total of 54 patients at the time the interim analysis was conducted, including 30 patients who had completed 6
months of treatment. The primary clinical endpoint of complete target ulcer closure on two consecutive visits showed a difference in favor of vilobelimab over placebo of 20.8% versus 16.7% (p=NS (not significant)). Key secondary endpoints such as
complete disease remission (complete closure of all ulcers) showed improvement in favor of vilobelimab over placebo (20.8% versus 5.6%, p=NS) and those with >50% reduction of target ulcer volume at week 26 (36.4% versus 16.7%, p=NS). In
addition, patients reported feeling better as measured by the Dermatology Life Quality Index (DLQI) mean percentage change at the end of treatment visit (-31.1% versus 3.4%). Overall, vilobelimab was well tolerated. Observed treatment-emergent
adverse events (TEAEs) were mostly mild to moderate, and patients with serious related on-treatment TEAEs were similarly distributed (6.3% in vilobelimab arm and 4.5% in placebo arm).
In addition, further post-hoc analyses showed that there is an overall treatment effect with vilobelimab when compared to placebo. These include
an MMRM (mixed model repeated measures) for percent change in target ulcer volume, which showed an average effect over all visits in favor of vilobelimab over placebo (-45.4%, p=0.0428, Weeks 2 – 26 overall) when imputing patients with
treatment-related discontinuation reasons, including patient level stopping criteria (PLSC) with a last observation carried forward (LOCF) approach. This analysis yielded a significant treatment difference for every week from Week 14 (-57.6%,
p=0.0357) to Week 26 (-63.2%, p=0.0122) for vilobelimab over placebo.
In addition, ANCOVAs (analyses of covariance) for mean of percentage changes from baseline in volume and area from Week 12 until Week 26 were also
in favor of vilobelimab, including mean of percentage change from baseline in volume (p=0.0111) and area (p=0.0072). These analyses suggest that treatment longer than 26 weeks with vilobelimab may provide improved treatment outcomes in this
difficult-to-treat ulcerative PG population.
Figure 1: Ulcer volume mean percent change from baseline, vilobelimab versus placebo
Alex G. Ortega Loayza, MD, MCR, CWSP, Professor and Interim
Chair, Department of Dermatology, Oregon Health and Science University, said: "I am encouraged by the signals of efficacy observed from the Phase 3 post-hoc analyses of vilobelimab in pyoderma gangrenosum.
The role of targeting C5a/C5aR aligns with prior mechanistic work supporting its use for this devastating inflammatory dermatological disease, which has no FDA-approved therapy. Given the significant unmet need and lack of approved therapies, I
am hopeful these findings will motivate further investigation of this targeted approach."
Benjamin Kaffenberger, MD, Associate Professor, Dermatology, The
Ohio State University Wexner Medical Center, said: “Pyoderma gangrenosum remains a difficult-to-treat rare disease with high unmet medical need. The data for vilobelimab suggest an overall treatment effect,
even if the primary endpoint may not have been achieved within the six-month timeframe required in this first-of-its-kind Phase 3 trial. Unfortunately, there is not a successful precedent for conducting a pivotal Phase 3 placebo-controlled study
in PG, and I believe the futility leading to early discontinuation relates to challenges in trial design rather than lack of efficacy of the therapy. I believe that blocking C5a/C5aR in this neutrophilic disease continues to make scientific and
clinical sense and that there is a strong justification to move forward.”
As next steps, InflaRx anticipates meeting with the FDA to discuss a potential path forward for vilobelimab in PG, including the use of
alternative endpoints that could be utilized for potential future clinical studies. At this time, in an effort to prioritize izicopan (INF904) development, InflaRx does not expect to deploy significant resources towards future vilobelimab
development in PG on its own and will instead consider doing so in collaboration with a partner.
About vilobelimab
Vilobelimab is a first-in-class monoclonal anti-human complement factor C5a antibody, which highly and effectively blocks the biological activity of C5a and
demonstrates high selectivity towards its target in human blood. Thus, vilobelimab leaves the formation of the membrane attack complex (C5b-9) intact as an important defense mechanism of the innate immune system, which is not the case for molecules
blocking C5. In pre-clinical studies, vilobelimab has been shown to control the inflammatory response-driven tissue and organ damage by specifically blocking C5a as a key “amplifier” of this response.
About InflaRx N.V.
InflaRx (Nasdaq: IFRX) is a biopharmaceutical company pioneering anti-inflammatory therapeutics by applying its proprietary anti-C5a and anti-C5aR technologies to
discover, develop and commercialize highly potent and specific inhibitors of the complement activation factor C5a and its receptor, C5aR. C5a is a powerful inflammatory mediator involved in the progression of a wide variety of inflammatory
diseases. InflaRx has developed vilobelimab, a novel, intravenously delivered, first-in-class, anti-C5a monoclonal antibody that selectively binds to free C5a and has demonstrated disease-modifying clinical activity and tolerability in multiple
clinical studies. InflaRx is also developing izicopan (INF904), an orally administered small molecule inhibitor of C5a-induced signaling via the C5a receptor.
InflaRx was founded in 2007, and the group has offices and subsidiaries in Jena and Munich, Germany, as well as Ann Arbor, MI, USA. For further information, please
visit www.inflarx.de. InflaRx GmbH (Germany) and InflaRx Pharmaceuticals Inc. (USA) are wholly owned subsidiaries of InflaRx N.V. (together, InflaRx).
Contacts:
|
InflaRx N.V.
|
MC Services AG
|
|
Jan Medina, CFA
|
Katja Arnold, Laurie Doyle, Dr. Regina Lutz
|
|
Vice President, Head of Investor Relations
|
Email: inflarx@mc-services.eu
|
|
Email: IR@inflarx.de
|
Europe: +49 89-210 2280
|
|
U.S.: +1-339-832-0752
|
FORWARD-LOOKING STATEMENTS
This press release contains forward-looking statements. All statements other than statements of historical fact are forward-looking statements, which are often
indicated by terms such as “may,” “will,” “should,” “expect,” “plan,” “anticipate,” “could,” “intend,” “target,” “estimate,” “believe,” “predict,” “potential” or “continue,” among others. Forward-looking statements appear in a number of places
throughout this press release and may include statements regarding our intentions, beliefs, projections, outlook, analyses and current expectations concerning, among other things: the receptiveness of izicopan as a treatment for HS and CSU by patients and hospitals and related treatment recommendations by medical/healthcare institutes and other third-party organizations; our ability to successfully secure
distribution channels and commercialize GOHIBIC (vilobelimab) as a treatment for COVID-19 patients and our ability to positively influence treatment recommendations by U.S. and European hospitals, guideline bodies and other third-party
organizations; our expectations regarding the size of the patient populations for, market opportunity for, coverage and reimbursement for, estimated returns and return accruals for, and clinical utility of GOHIBIC (vilobelimab) in its approved or
authorized indication or for vilobelimab and any other product candidates, under the Emergency Use Authorization and in the future if approved for commercial use in the United States, Europe or elsewhere; our ability to successfully implement The
InflaRx Commitment Program, the success of our future clinical trials for vilobelimab’s treatment of debilitating or life-threatening inflammatory indications, including acute respiratory distress syndrome and other indications, and any other
product candidates, including izicopan, and whether such clinical results will reflect results seen in previously conducted pre-clinical studies and clinical trials; the timing, progress and results of pre-clinical studies and clinical trials of
vilobelimab, izicopan and any other product candidates, including for the development of vilobelimab in several indications, including to obtain full approval of GOHIBIC (vilobelimab) for COVID-19 and other virally induced ARDS, to treat HS and
CSU, and statements regarding the timing of initiation and completion of studies or trials and related preparatory work, the period during which the results of the trials will become available, the costs of such trials and our research and
development programs generally; our interactions with and the receptiveness and approval by regulators regarding the results of clinical trials and potential regulatory approval or authorization pathways, including our biologics license application
submission for GOHIBIC (vilobelimab); the timing and outcome of any discussions or submission of filings for regulatory approval or authorization of vilobelimab, izicopan or any other product candidate, and the timing of and our ability to obtain
and maintain full regulatory approval, the EUA and/or market authorization of vilobelimab or GOHIBIC (vilobelimab) for any indication; our ability to leverage our proprietary anti-C5a and anti-C5aR technologies to discover and develop therapies to
treat complement-mediated autoimmune and inflammatory diseases; our ability to protect, maintain and enforce our intellectual property protection for vilobelimab, izicopan and any other product candidates, and the scope of such protection; whether
the U.S. Food and Drug Administration, the European Medicines Agency or any comparable foreign regulatory authority will accept or agree with the number, design, size, conduct or implementation of our clinical trials, including any proposed primary
or secondary endpoints for such trials; the success of our future clinical trials for vilobelimab, izicopan and any other product candidates and whether such clinical results will reflect results seen in previously conducted pre-clinical studies
and clinical trials; our expectations regarding the size of the patient populations for, the market opportunity for, the medical need for and clinical utility of vilobelimab, izicopan or any other product candidates, if approved or authorized for
commercial use; our manufacturing capabilities and strategy, including the scalability and cost of our manufacturing methods and processes and the optimization of our manufacturing methods and processes, and our ability to continue to rely on our
existing third-party manufacturers and our ability to engage additional third-party manufacturers for our planned future clinical trials and for commercial supply of vilobelimab and for the finished product GOHIBIC (vilobelimab) in the United
States and Europe; our estimates of our expenses, ongoing losses, future revenue, capital requirements and our needs for or ability to obtain additional financing; our expectations regarding the scope of any approved indication for vilobelimab; our
ability to defend against liability claims resulting from the testing of our product candidates in the clinic or, if approved or authorized, any commercial sales; if any of our product candidates obtain regulatory approval or authorization, our
ability to comply with and satisfy ongoing drug regulatory obligations and continued regulatory overview; our ability to comply with enacted and future legislation in seeking marketing approval or authorization and commercialization; our future
growth and ability to compete, which depends on our retaining key personnel and recruiting additional qualified personnel; our competitive position and the development of and projections relating to our competitors in the development of C5a and
C5aR inhibitors and other therapeutic products being developed in similar medical conditions in which vilobelimab, izicopan or any other of our product candidates is being developed or our industry; and the risks, uncertainties and other factors
described under the heading “Risk Factors” in our periodic filings with the U.S. Securities and Exchange Commission. These statements speak only as of the date of this press release and involve known and unknown risks, uncertainties and other
important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. Given these risks,
uncertainties and other factors, you should not place undue reliance on these forward-looking statements, and we assume no obligation to update these forward-looking statements, even if new information becomes available in the future, except as
required by law.