UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of The Securities Exchange Act of 1934
Date of Report (Date of Earliest Event Reported): January 8, 2026
Voyager Therapeutics, Inc.
(Exact name of registrant as specified in its charter)
| Delaware | 001-37625 | 46-3003182 | ||
| (State
or other jurisdiction of incorporation) |
(Commission File Number) |
(I.R.S.
Employer Identification No.) |
| 75 Hayden Avenue Lexington, Massachusetts |
02421 | |
| (Address of principal executive offices) | (Zip Code) |
Registrant’s telephone number, including area code (857) 259-5340
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
¨ Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425)
¨ Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12)
¨ Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b))
¨ Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c))
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered |
| Common Stock, $0.001 par value | VYGR | Nasdaq Global Select Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ¨
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Item 7.01. Regulation FD Disclosure.
From time to time, Voyager Therapeutics, Inc. (the “Company”) presents or distributes slide presentations to the investment community to provide updates and summaries of its business. The Company is posting a copy of its current corporate investor presentation to the “Investors” portion of its website at https://www.voyagertherapeutics.com/. The Company also plans to distribute a letter summarizing certain of the information included in the corporate investor presentation to specified stockholders. Copies of the corporate investor presentation and letter to stockholders are furnished as Exhibit 99.1 and Exhibit 99.2, respectively, to this Current Report on Form 8-K and are incorporated herein by reference. The information contained on, or accessible through, the Company’s website is not incorporated by reference into this Current Report on Form 8-K and should not be considered to be a part hereof.
The information in this Current Report on Form 8-K (including Item 7.01, Exhibit 99.1 and Exhibit 99.2) shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Exchange Act or the Securities Act of 1933, as amended, except as expressly set forth by specific reference in such a filing.
By providing the information in Item 7.01 of this Current Report on Form 8-K, including Exhibit 99.1 and Exhibit 99.2 hereto, the Company is not making an admission as to the materiality of any information herein. The information contained in this Current Report on Form 8-K is intended to be considered in the context of more complete information included in the Company’s filings with the SEC and other public announcements that the Company has made and may make from time to time by press release or otherwise. The Company undertakes no duty or obligation to update or revise the information contained in this Current Report on Form 8-K, although it may do so from time to time as its management believes is appropriate. Any such updating may be made through the filing of other reports or documents with the SEC, through press releases or through other public disclosures.
Cautionary Note Regarding Forward-Looking Statements
This Current Report on Form 8-K (including Exhibit 99.1 and Exhibit 99.2) contains forward-looking statements for the purposes of the safe harbor provisions under The Private Securities Litigation Reform Act of 1995 and other federal securities laws. The use of words such as “expect,” “anticipate,” “potential,” “may,” “plan,” “future,” “suggest,” “would,” “believe,” “will,” or “continue,” and other similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. These forward-looking statements include, among other things, statements about expectations for the Company’s achievement of preclinical and clinical development milestones for its potential development candidates, such as the identification of lead development candidates, IND and CTA filings, the initiation of clinical trials, and the generation of clinical data and proof-of-concept data; the potential for third-party clinical data to inform the Company’s product development programs; the Company’s ability to expand beyond gene therapy and antibodies into other modalities of neurogenetic medicine, including Voyager NeuroShuttles and small molecule therapies; the Company’s ability to generate near-term and long-term funding through reimbursement, upfront, milestone and royalty-based fees (as applicable) under its existing licensing and collaboration agreements, and to obtain data regarding the performance of its TRACER-derived capsid families licensed to its collaborators and partners under such agreements; the Company’s ability to maintain and advance product development programs under its current partnerships and collaborations, including the anticipated timing of regulatory submissions by collaborators; and the sufficiency of the Company’s cash resources.
These forward-looking statements are only predictions, and the Company may not actually achieve the plans, intentions, or expectations disclosed in the forward-looking statements. All forward-looking statements are based on estimates and assumptions by the Company’s management that, although the Company believes such forward-looking statements to be reasonable, are inherently uncertain and subject to risks and uncertainties that may cause actual results to differ materially from those that the Company expected. Such risks and uncertainties include, among others, the expectations and decisions of regulatory authorities; the timing, initiation, conduct, and outcomes of the Company’s preclinical studies and clinical trials; the availability of data from internal or third-party clinical trials; the success of the Company’s product candidates; the availability or commercial potential of product candidates under collaborations; the willingness and ability of the Company’s collaboration partners to meet obligations under collaboration agreements with the Company; the continued development of the Company’s technology platforms, including the Company’s TRACER capsid discovery platform and its non-viral discovery platform; the Company’s scientific approach and program development progress, and the restricted supply and increased costs of critical research components; the development by third parties of capsid identification platforms and capsids that may be competitive to the Company’s TRACER capsid discovery platform; the Company’s ability to create and protect intellectual property rights associated with the TRACER capsid discovery and non-viral platforms, the capsids and ligands identified by the platforms, and development candidates for the Company’s pipeline programs; the possibility and the timing of the Company’s receipt of program reimbursement, development or commercialization milestones, option exercise, and other payments under the Company’s existing licensing or collaboration agreements; the ability of the Company to negotiate and complete licensing or collaboration agreements with other parties on terms acceptable to the Company and the third parties; the success of programs controlled by third-party collaborators in which the Company retains a financial interest; the ability to attract and retain talented directors, employees, and contractors; and the sufficiency of the Company’s cash resources.
These statements are also subject to a number of material risks and uncertainties that are described in the Company’s most recent Annual Report on Form 10-K filed with the SEC, as updated by its subsequent filings with the SEC. All information in this Form 8-K (including Exhibit 99.1 and Exhibit 99.2) is as of the date of this Form 8-K, and any forward-looking statement speaks only as of the date on which it was made. The Company undertakes no obligation to publicly update or revise this information or any forward-looking statement, whether as a result of new information, future events or otherwise, except as required by law.
Item 9.01. Financial Statements and Exhibits.
(d) Exhibits.
| Exhibit No. | Description | |
| 99.1 | Corporate slide presentation of Voyager Therapeutics, Inc. dated January 8, 2026 | |
| 99.2 | Letter to stockholders of Voyager Therapeutics, Inc. dated January 8, 2026 | |
| 104 | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101). |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| Date: January 8, 2026 | VOYAGER THERAPEUTICS, INC. | |
| By: | /s/ Alfred Sandrock, M.D., Ph.D. | |
| Alfred Sandrock, M.D., Ph.D. | ||
| Chief Executive Officer, President, and Director (Principal Executive Officer) |
||
Exhibit 99.1

Leveraging Genetics to Treat Neurological Diseases Corporate Deck / January 2026

2 | Forward - Looking Statements This presentation contains forward - looking statements for the purposes of the safe harbor provisions under The Private Securities Litigation Reform Act of 1995 and other federal securities laws . The use of words such as “anticipate,” “expect,” “believe,” “plan,” “estimate,” “may,” or “potential,” and other similar expressions are intended to identify forward - looking statements, although not all forward - looking statements contain these identifying words . These forward - looking statements include, among other things, statements about expectations for Voyager’s achievement of preclinical and clinical development milestones for its potential development candidates such as the identification of lead development candidates, IND and CTA filings, the initiation of clinical trials, and the generation of clinical data and proof - of - concept data ; the potential for third - party clinical data to inform Voyager’s product development programs ; Voyager’s ability to expand beyond gene therapy and antibodies into other modalities of neurogenetic medicine, including Voyager NeuroShuttles and small molecule therapies ; Voyager’s ability to generate near - term and long - term funding through reimbursement, upfront, milestone and royalty - based fees (as applicable) under its existing licensing and collaboration agreements, and to obtain data regarding the performance of its TRACER - derived capsid families licensed to its collaborators and partners under such agreements ; Voyager’s ability to maintain and advance product development programs under its current partnerships and collaborations, including the anticipated timing of regulatory submissions by collaborators ; and the sufficiency of Voyager’s cash resources . These forward - looking statements are only predictions, and Voyager may not actually achieve the plans, intentions, or expectations disclosed in the forward - looking statements . All forward - looking statements are based on estimates and assumptions by Voyager’s management that, although Voyager believes such forward - looking statements to be reasonable, are inherently uncertain and subject to risks and uncertainties that may cause actual results to differ materially from those that Voyager expected . Such risks and uncertainties include, among others, the expectations and decisions of regulatory authorities ; the timing, initiation, conduct, and outcomes of Voyager’s preclinical studies and clinical trials ; the availability of data from internal or third - party clinical trials ; the success of Voyager’s product candidates ; the availability or commercial potential of product candidates under collaborations ; the willingness and ability of Voyager's collaboration partners to meet obligations under collaboration agreements with Voyager ; the continued development of Voyager’s technology platforms, including Voyager’s TRACER capsid discovery platform and its non - viral discovery platform ; Voyager’s scientific approach and program development progress, and the restricted supply and increased costs of critical research components ; the development by third parties of capsid identification platforms and capsids that may be competitive to Voyager’s TRACER capsid discovery platform ; Voyager’s ability to create and protect intellectual property rights associated with the TRACER capsid discovery and non - viral platforms, the capsids and ligands identified by the platforms, and development candidates for Voyager’s pipeline programs ; the possibility and the timing of Voyager’s receipt of program reimbursement, development or commercialization milestones, option exercise, and other payments under Voyager’s existing licensing or collaboration agreements ; the ability of Voyager to negotiate and complete licensing or collaboration agreements with other parties on terms acceptable to Voyager and the third parties ; the success of programs controlled by third - party collaborators in which Voyager retains a financial interest ; the ability to attract and retain talented directors, employees, and contractors ; and the sufficiency of Voyager’s cash resources . These statements are also subject to a number of material risks and uncertainties that are described in Voyager’s most recent Annual Report on Form 10 - K filed with the Securities and Exchange Commission, as updated by its subsequent filings with the Securities and Exchange Commission . Any forward - looking statement speaks only as of the date on which this presentation was presented . Voyager undertakes no obligation to publicly update or revise any forward - looking statement, whether as a result of new information, future events or otherwise, except as required by law . © Voyager Therapeutics, Inc .

3 | Voyager (NASDAQ: VYGR) 2026 Investment Rationale Validating brain - targeted capsids in humans Transformative year for tau, with two s hots on goal Demonstrating the value of Voyager NeuroShuttle 1 Based on our current operating plans, cash and cash equivalents and marketable securities as of September 30, 2025, along wit h a mounts expected to be received as reimbursement for development costs under the Neurocrine and Novartis collaborations, and interest income. Cash runway into 2028, not including $ 6.8 B in potential milestone payments from existing partnerships 1 Antibody: VY7523 data in AD patients expected H2 ‘ 26 Knockdown: VY1706 first - in - human dosing expected H2 ‘ 26 following 3 rd - party data ( BIIB080 , expected mid - ’26) Sustained brain delivery vs TfR shuttles ; shuttled antibody has superior target engagement vs unshuttled 2026 data: NHP translatability, safety, program advancement VY1706 for AD first - in - human dosing expected H2 ‘ 26 NBIB - ’223 for Friedreich’s ataxia ( NBIX - partnered ) expected to enter clinic in 2026; dual - targeting of brain and heart 4 | Multi - Modality CNS Pipeline with High Potential Value Approved Pivotal Phase I/II IND - Enabling Research Mechanism / Indication VY7523 (Anti - tau Antibody) / Alzheimer’s Disease VY1706 (Tau Silencing Gene Therapy) (siRNA) / Alzheimer’s Disease APOE Gene Therapy / Alzheimer’s Disease ALPL - VYGR - NeuroShuttle / Undisclosed Neurocrine NBIB - ’223 (FXN Gene Therapy) / Friedreich’s Ataxia Neurocrine GBA1 Gene Therapy / Gaucher’s / Parkinson’s Transition Bio TDP - 43 Small Molecule / ALS / FTD WHOLLY - OWNED OPT - IN RIGHTS LICENSES & COLLABS Option for 100% WW rights Option for 40% US rights Option for 50% US rights 2 in IND - enabling; 1 undisclosed Neurocrine Three Gene Therapies / Undisclosed Undisclosed Novartis Three Gene Therapies / HD, SMA, Undisclosed Undisclosed Alexion One Gene Therapy / Undisclosed



Transformative Year for Tau – Voyager Has Two Approaches 5 | 6 | Tau Critical in Alzheimer’s Disease; Two Ways to Target Braak Staging: Adapted from Jouanne M, Rault S, Voisin - Chiret AS. Eur J Med Chem. 2017 Oct 20;139:153 - 167. Spread of Pathological Tau Corresponds to Alzheimer’s Disease Progression Tau Spread = Disease Worsening Two Approaches to Targeting Tau 1. Intracellular 2. Extracellular Block production of tau within cells, where it aggregates into toxic tangles Block cell - to - cell spread of tau as it moves between neurons Knockdown (ASO, siRNA, etc ) (i.e. VY1706, BIIB080) Intracellular and Extracellular Antibodies (i.e.
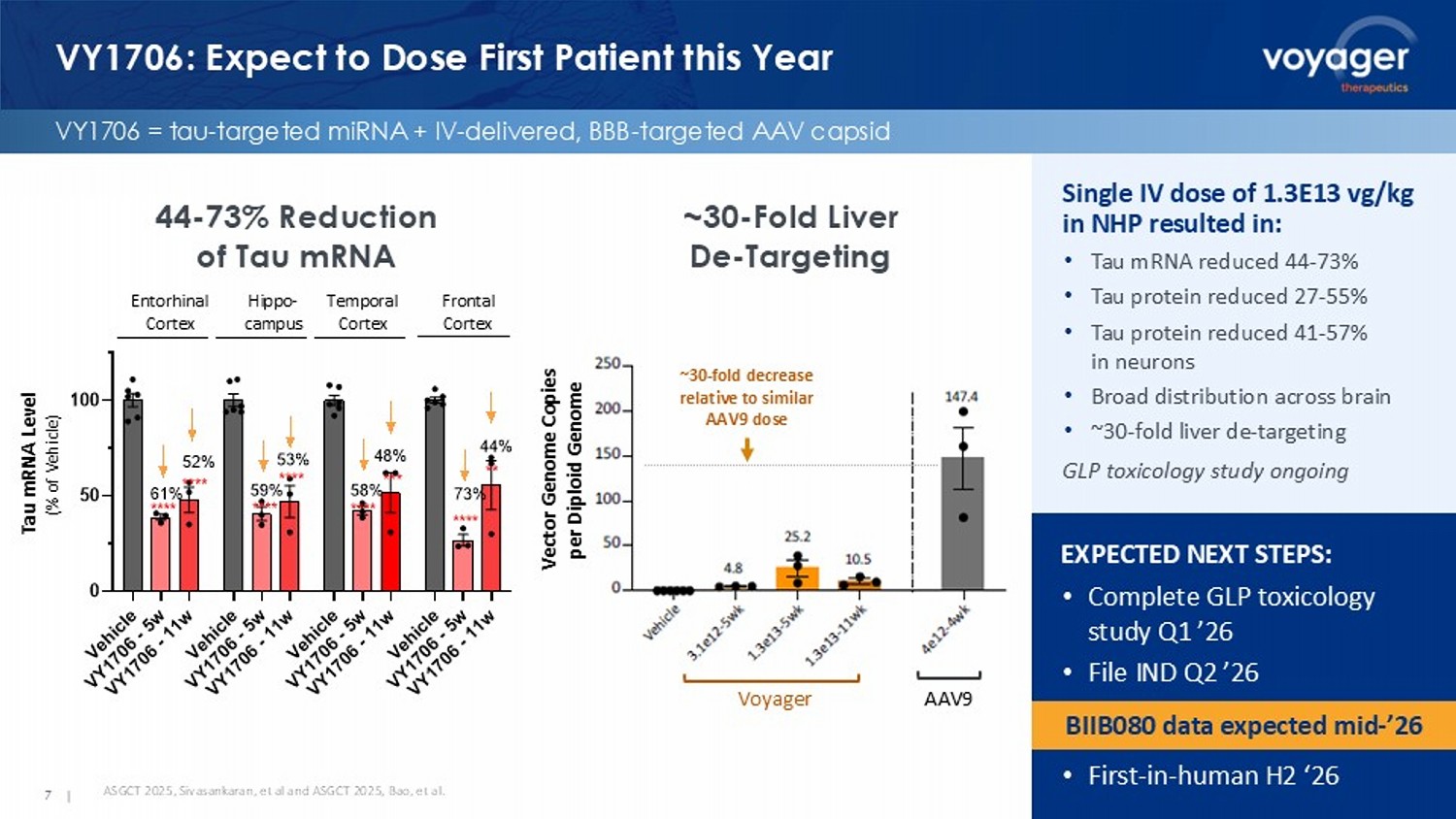
VY7523) Extracellular Voyager AAV9 ~30 - fold decrease relative to similar AAV9 dose Vector Genome Copies per Diploid Genome VY1706: Expect to Dose First Patient this Year VY1706 = tau - targeted miRNA + IV - delivered, BBB - targeted AAV capsid 7 | EXPECTED NEXT STEPS: • Complete GLP toxicology study Q1 ’26 • File IND Q2 ’26 • First - in - human H2 ‘26 ASGCT 2025, Sivasankaran, et al and ASGCT 2025, Bao, et al. Single IV dose of 1.3E13 vg/kg in NHP resulted in: • Tau mRNA reduced 44 - 73% • Tau protein reduced 27 - 55% • Tau protein reduced 41 - 57% in neurons • Broad distribution across brain • ~30 - fold liver de - targeting GLP toxicology study ongoing 44 - 73% Reduction of Tau mRNA ~30 - Fold Liver De - Targeting BIIB080 data expected mid - ’26 V e h i c l e V Y 1 7 0 6 - 5 w V Y 1 7 0 6 - 1 1 w V e h i c l e V Y 1 7 0 6 - 5 w V Y 1 7 0 6 - 1 1 w V e h i c l e V Y 1 7 0 6 - 5 w V Y 1 7 0 6 - 1 1 w V e h i c l e V Y 1 7 0 6 - 5 w V Y 1 7 0 6 - 1 1 w 0 50 100 T a u m R N A L e v e l ( % o f V e h i c l e ) 61% 58%59% 73% EC TC FC HC 52% 53% 44% 48% **** **** **** **** **** *** **** ** Entorhinal Cortex Hippo - campus Temporal Cortex Frontal Cortex Tau mRNA Level (% of Vehicle)
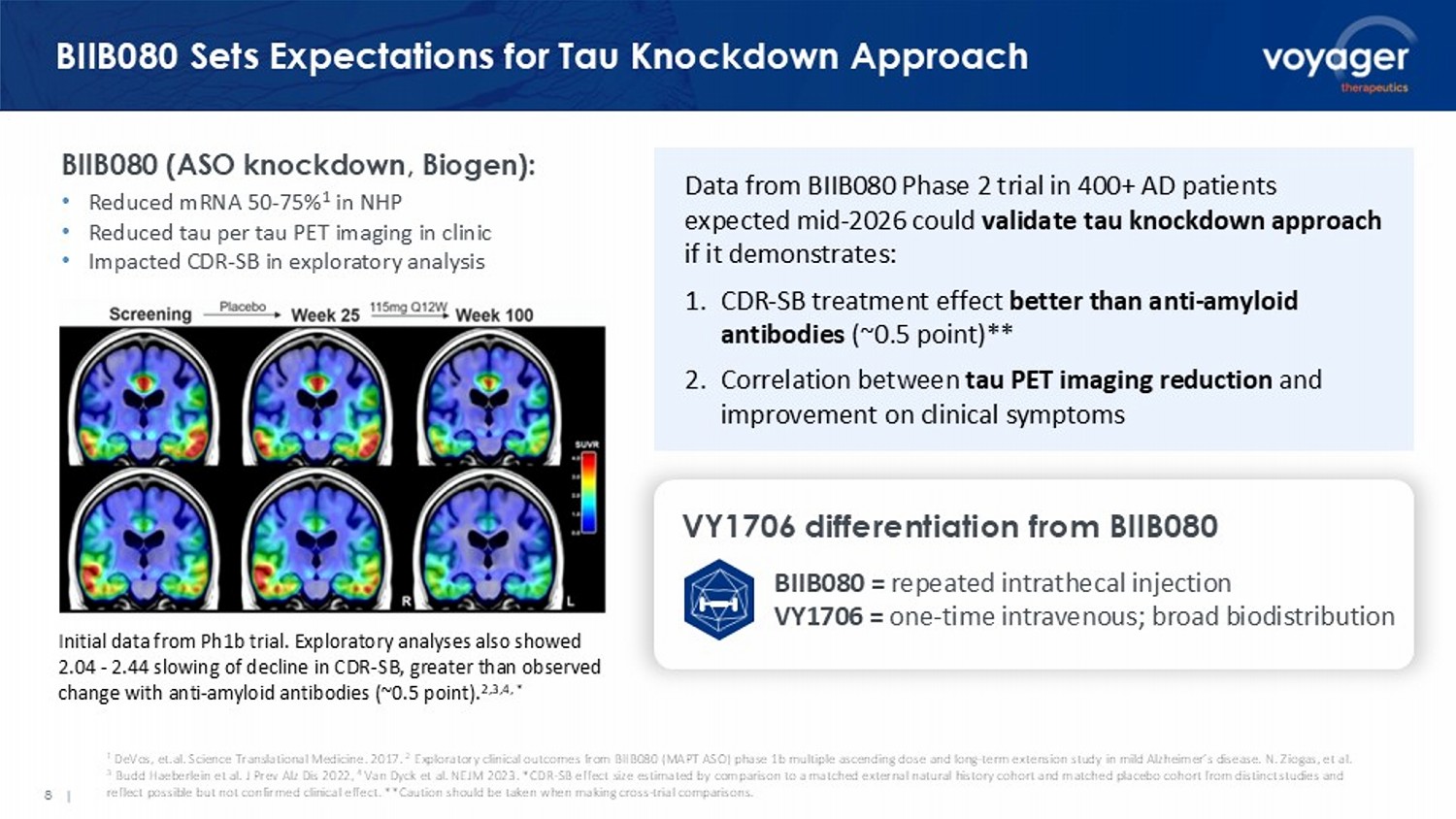
1 DeVos, et.al. Science Translational Medicine. 2017. 2 Exploratory clinical outcomes from BIIB080 (MAPT ASO) phase 1b multiple ascending dose and long - term extension study in mild Al zheimer’s disease. N. Ziogas, et al. 3 Budd Haeberlein et al. J Prev Alz Dis 2022, 4 Van Dyck et al. NEJM 2023. * CDR - SB effect size estimated by comparison to a matched external natural history cohort and matched placebo cohort from distinct studies and reflect possible but not confirmed clinical effect. ** Caution should be taken when making cross - trial comparisons. 8 | BIIB080 Sets Expectations for Tau Knockdown Approach BIIB080 (ASO knockdown, Biogen): • Reduced mRNA 50 - 75% 1 in NHP • Reduced tau per tau PET imaging in clinic • Impacted CDR - SB in exploratory analysis Initial data from Ph1b trial. Exploratory analyses also showed 2.04 - 2.44 slowing of decline in CDR - SB, greater than observed change with anti - amyloid antibodies (~0.5 point). 2,3,4, * Data from BIIB080 Phase 2 trial in 400+ AD patients expected mid - 2026 could validate tau knockdown approach if it demonstrates: 1. CDR - SB treatment effect better than anti - amyloid antibodies (~0.5 point)** 2. Correlation between tau PET imaging reduction and improvement on clinical symptoms VY1706 differentiation from BIIB080 BIIB080 = repeated intrathecal injection VY1706 = one - time intravenous; broad biodistribution 9 | VY7523: Tau PET Imaging Data in AD Patients Expected H2 2026 VY7523 single ascending dose (SAD) trial: • 48 healthy volunteers • No serious/severe adverse events or infusion reactions reported • Half - life supports monthly dosing • Serum concentrations increased in dose - proportionate manner; CSF - to - serum ratio 0.3% CTAD 2025, Ratti, et al.

and CTAD 2025 Butler, et. al. VY7523 Enrollment Complete in MAD Trial Tau PET Imaging Data Expected H2 ‘26 VY7523 Differentiated from Other Anti - Tau Antibodies VY7523 = antibody targeting epitope in C - terminal, highly specific for pathological tau 10 | Epitope Matters 1,2,3 : Pathological Specificity May Matter: 1.
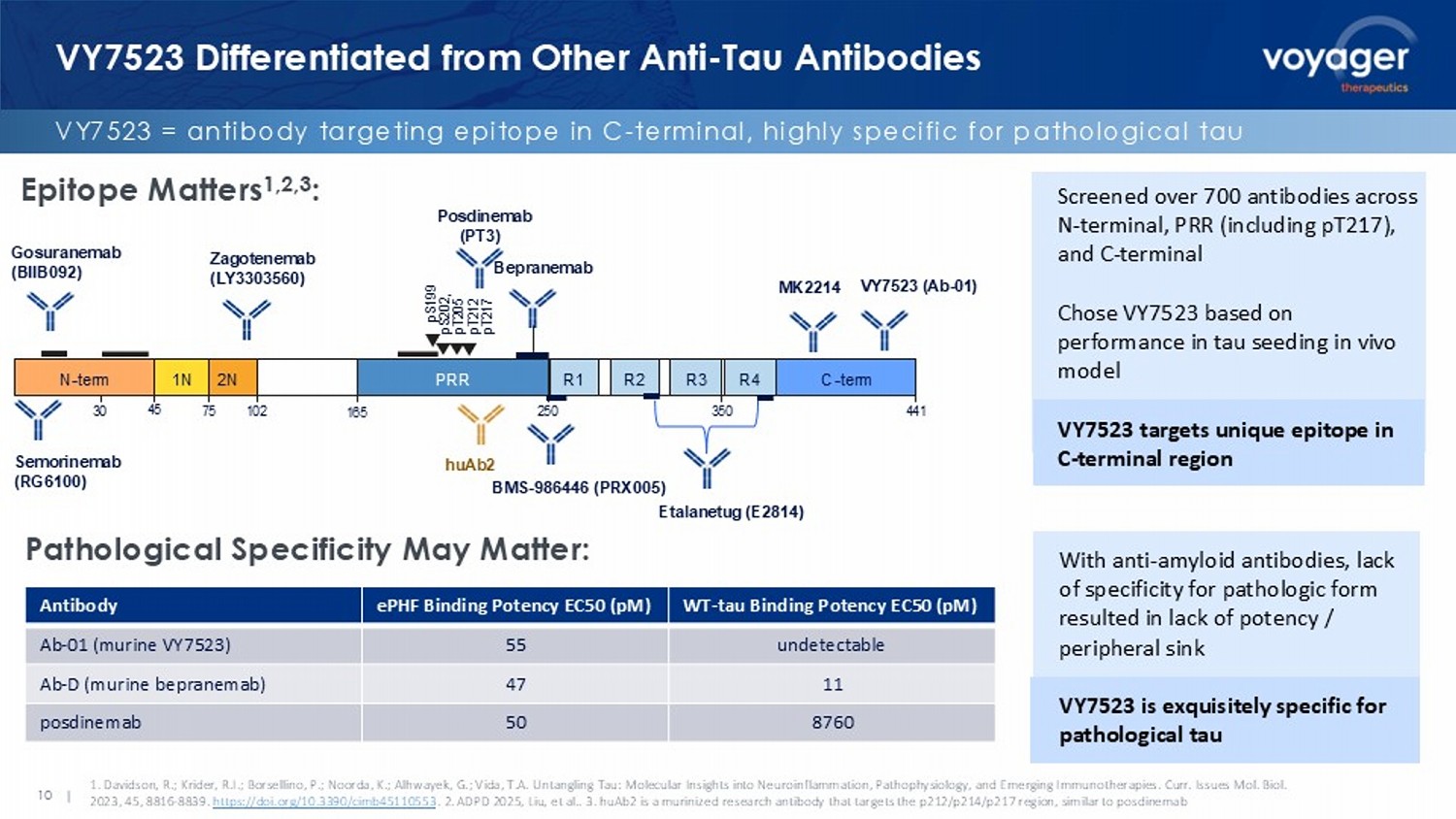
Davidson, R.; Krider, R.I.; Borsellino, P.; Noorda, K.; Alhwayek , G.; Vida, T.A. Untangling Tau: Molecular Insights into Neuroinflammation, Pathophysiology, and Emerging Immunotherapies. Cu rr. Issues Mol. Biol. 2023, 45, 8816 - 8839. https://doi.org/10.3390/cimb45110553 . 2. ADPD 2025, Liu, et al.. 3. huAb2 is a murinized research antibody that targets the p212/p214/p217 region, similar to posdinemab 30 45 250 350 VY 7523 ( Ab - 01 ) N - term 1N 2N PRR R1 R3 R4 R2 C - term pS199 pS202, pT205 pT212 pT217 441 75 102 165 Posdinemab (PT3) B epranemab BMS - 986446 (PRX005) Etalanetug ( E2814) Screened over 700 antibodies across N - terminal, PRR (including pT217), and C - terminal Chose VY7523 based on performance in tau seeding in vivo model VY7523 targets unique epitope in C - terminal region With anti - amyloid antibodies, lack of specificity for pathologic form resulted in lack of potency / peripheral sink VY7523 is exquisitely specific for pathological tau Gosuranemab (BIIB092) Semorinemab (RG6100) Zagotenemab (LY3303560) MK2214 WT - tau Binding Potency EC50 ( pM ) ePHF Binding Potency EC50 ( pM ) Antibody undetectable 55 Ab - 01 (murine VY7523) 11 47 Ab - D (murine bepranemab ) 8760 50 posdinemab huAb2 11 | VY7523: P301S Data Differentiate from Posdinemab , Bepranemab VYGR reduces tau spread; N - terminal antibody does not P301S murine model; all antibodies represent murine versions.

Ab - 01 is murine VY7523; IPN002 is murine BIIB092; Ab - D is murine bepranemab ; huAb2 is a murinized research antibody that targets the p212/p214/p217 region, similar to posdinemab . PHF1 is a positive control antibody. VYGR reduces tau spread; bepranemab does also ADPD 2025, Liu, et al.. *, **, *** and **** indicate p < 0.05, 0.005, 0.0005 and 0.0001, respectively, compared to the vehicl e c ontrol group. huAb2 antibody binding similar epitope as posdinemab does not reduce tau spread I g G C t l P H F 1 h u A b 2 0 50 100 150 200 Contralateral 61% I g G C t r l P H F 1 h u A b 2 0 50 100 150 200 Ipsilateral R e l a t i v e A T 8 S i g n a l ( % o f I g G C o n t r o l ) ** 68% Validating Brain - Targeted Capsids in Humans 12 |


13 | Establishing Novel Capsid Proof - of - Concept in Humans STEP 1: Safety STEP 2. Delivery to Brain STEP 3. Tau Impact Determine if dosing in first few patients warrants continuation DERISKING: x Low doses / liver de - targeting o GLP tox in NHP o Potential for initial dosing in more advanced patients Biomarker - based assessment (i.e.

CSF/ NfL ) to confirm capsid was delivered to brain DERISKING: x Preclinical delivery confirmed cross - species (mouse, multiple NHPs) x Receptor identified for VY1706 capsid; capsid binds human homolog Tau PET assessment to confirm impact on tau and suggest possible efficacy DERISKING: x VY1706 derisked by preclinical efficacy in mouse/NHP o Potential further derisking with BIIB080 data Two Opportunities for First - in - Human Dosing in 2026: • First - in - human anticipated 2026 • Wholly - owned • Tau knockdown for Alzheimer’s disease • First - in - human anticipated 2026 • NBIX partnered (VYGR has opt - in for 40% US) • FXN replacement for Friedreich's ataxia VY1706 NBIB - ’223 14 | Leading Indications for 2026 Gene Therapy Trials Alzheimer’s Disease Impacts approximately 7M Americans and is expected to impact nearly 13M by 2050 1 • Initial anti - amyloid antibodies approved; tau targeting likely to be necessary once tau spread begins 2 • Currently no tau - targeted treatments approved for AD Potential one - time, I.V. gene therapy to knock down tau for Alzheimer’s disease Recent AD Expert KOL Advisory Board: high enthusiasm for VY1706; support for proposed safety approaches and monitoring plan Potential one - time, I.V. gene therapy to replace FXN in brain and heart for FA All VYGR - NBIX partnered gene therapy programs continue to make progress , including the GBA1 program for Gaucher and Parkinson’s disease; updates will be provided. 1. Alzheimer's Association; https://www.alz.org/alzheimers - dementia/facts - figures (accessed July 2025). 2. Sims JR, Zimmer JA, Evans CD, et al. Donanemab in Early Symptomatic Alzheimer Disease: The TRAILBLAZER - ALZ 2 Randomized Clinical Trial. JAMA. 2023;330(6):512 – 527. doi:10.1001/jama.2023.13239 3. Friedreich’s Ataxia Research Alliance (FARA ). What is FA? Available at: https://www.curefa.org/what - is - friedreichs - ataxia. Accessed: May 2024 Friedreich’s Ataxia Impacts approximately 5,000 Americans 3 • All cases caused by mutations of FXN gene. One treatment available but does not replace FXN; unmet need remains.

• No treatments in development thus far have shown brain and heart activity VY1706 NBIB - ’223 TRACER capsid discovery platform derived from evaluation of 200M+ variants of AAV5 and AAV9 TRACER - Derived Capsids to Power Next - Gen Gene Therapy 15 | VCAP - Gen2: 50 - 75% of Cells Transduced Across Diverse Brain Regions at 3E13 vg/kg (ASGCT 2024) Gen2 Capsids: Increased Brain Tropism and Liver Detargeting (ASGCT 2024)

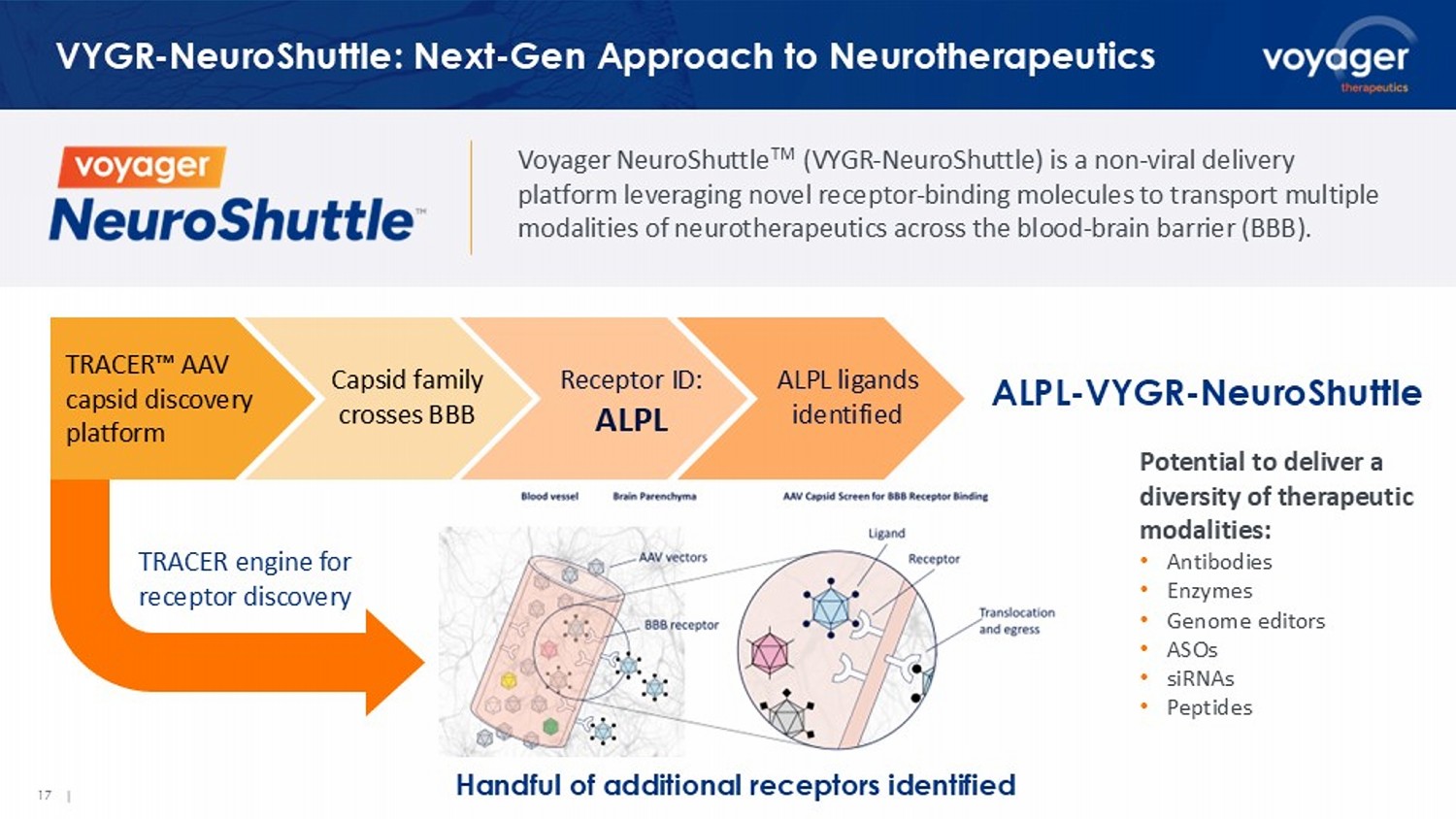
Voyager NeuroShuttle Nonviral Platform 16 | 17 | VYGR - NeuroShuttle : Next - Gen Approach to Neurotherapeutics TRACER AAV capsid discovery platform ALPL - VYGR - NeuroShuttle Handful of additional receptors identified Potential to deliver a diversity of therapeutic modalities: • Antibodies • Enzymes • Genome editors • ASOs • siRNAs • Peptides Voyager NeuroShuttle TM (VYGR - NeuroShuttle ) is a non - viral delivery platform leveraging novel receptor - binding molecules to transport multiple modalities of neurotherapeutics across the blood - brain barrier (BBB). Capsid family crosses BBB Receptor ID: ALPL ALPL ligands identified TRACER engine for receptor discovery Proof of concept murine data 1 show sustained CNS exposure for ALPL versus TfR shuttles ALPL - VYGR - NeuroShuttle Demonstrates Sustained Brain Exposures • Increased antibody concentration in brain; modest impact on peripheral clearance 18 | 1.
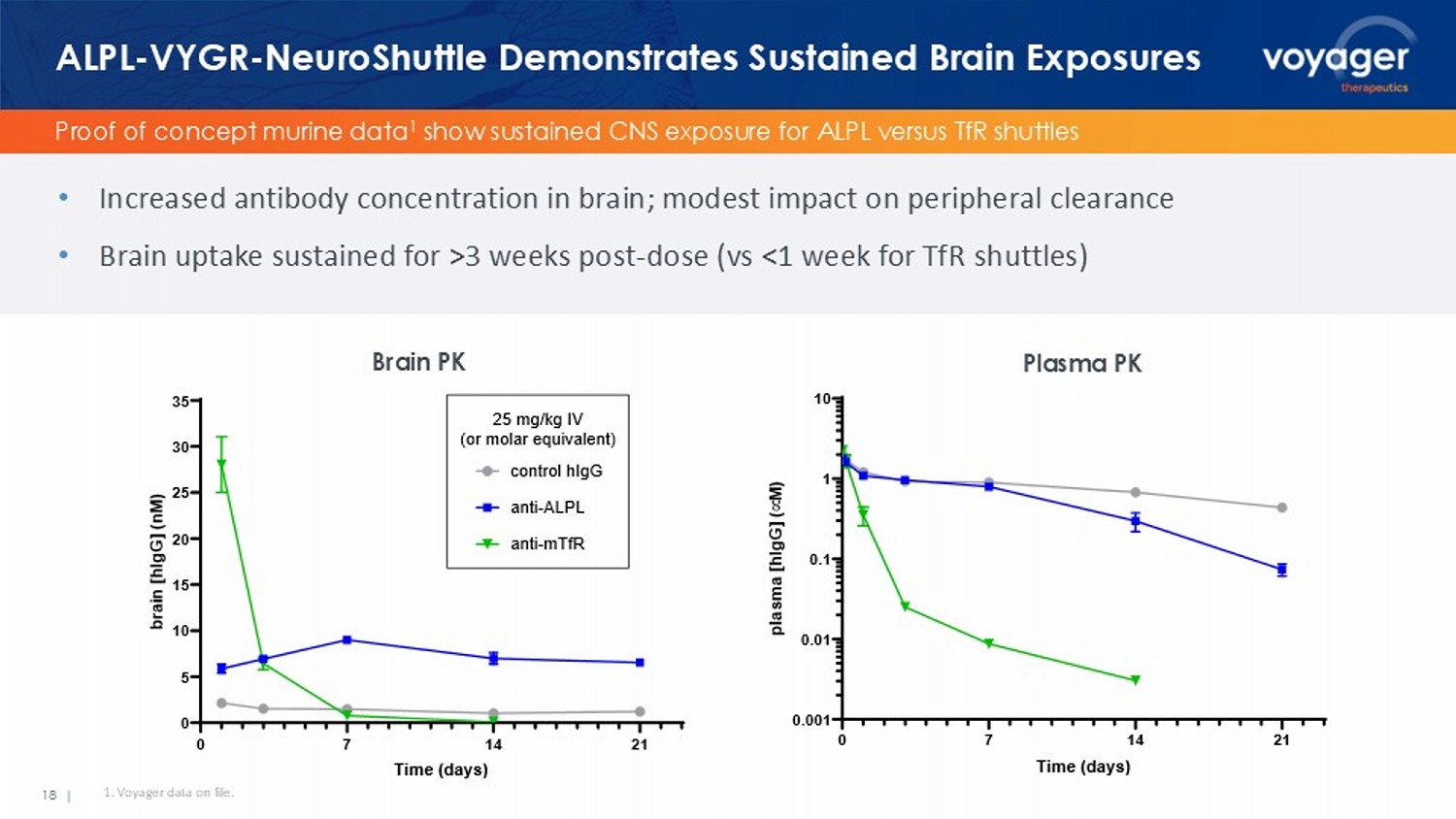
Voyager data on file. Brain PK 7L P H G D \ V EUDL Q > K,J * @ Q0 FRQWUROK,J* DQWL $/3/ DQWL P7I5 PJ NJ,9 RUPRODUHTXLYDOHQW Plasma PK 7L P H G D \ V SO D V P D > K, J * @ v 0 • Brain uptake sustained for >3 weeks post - dose (vs <1 week for TfR shuttles)
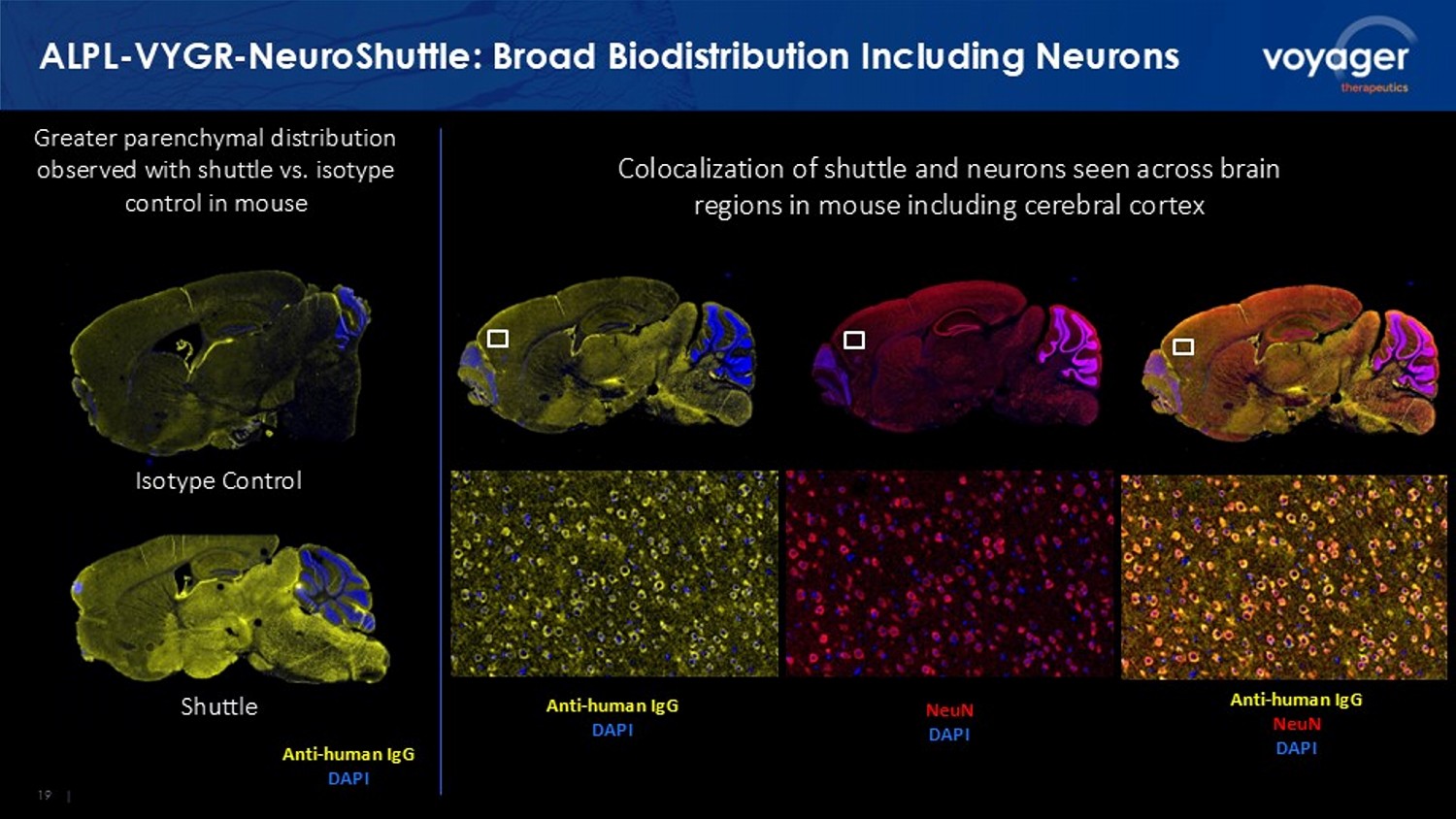
19 | ALPL - VYGR - NeuroShuttle : Broad Biodistribution Including Neurons Anti - human IgG DAPI Anti - human IgG DAPI Anti - human IgG NeuN DAPI Colocalization of shuttle and neurons seen across brain regions in mouse including cerebral cortex Greater parenchymal distribution observed with shuttle vs. isotype control in mouse NeuN DAPI Isotype Control Shuttle 20 | ALPL - VYGR - NeuroShuttle Does Not Impact Reticulocytes 1.
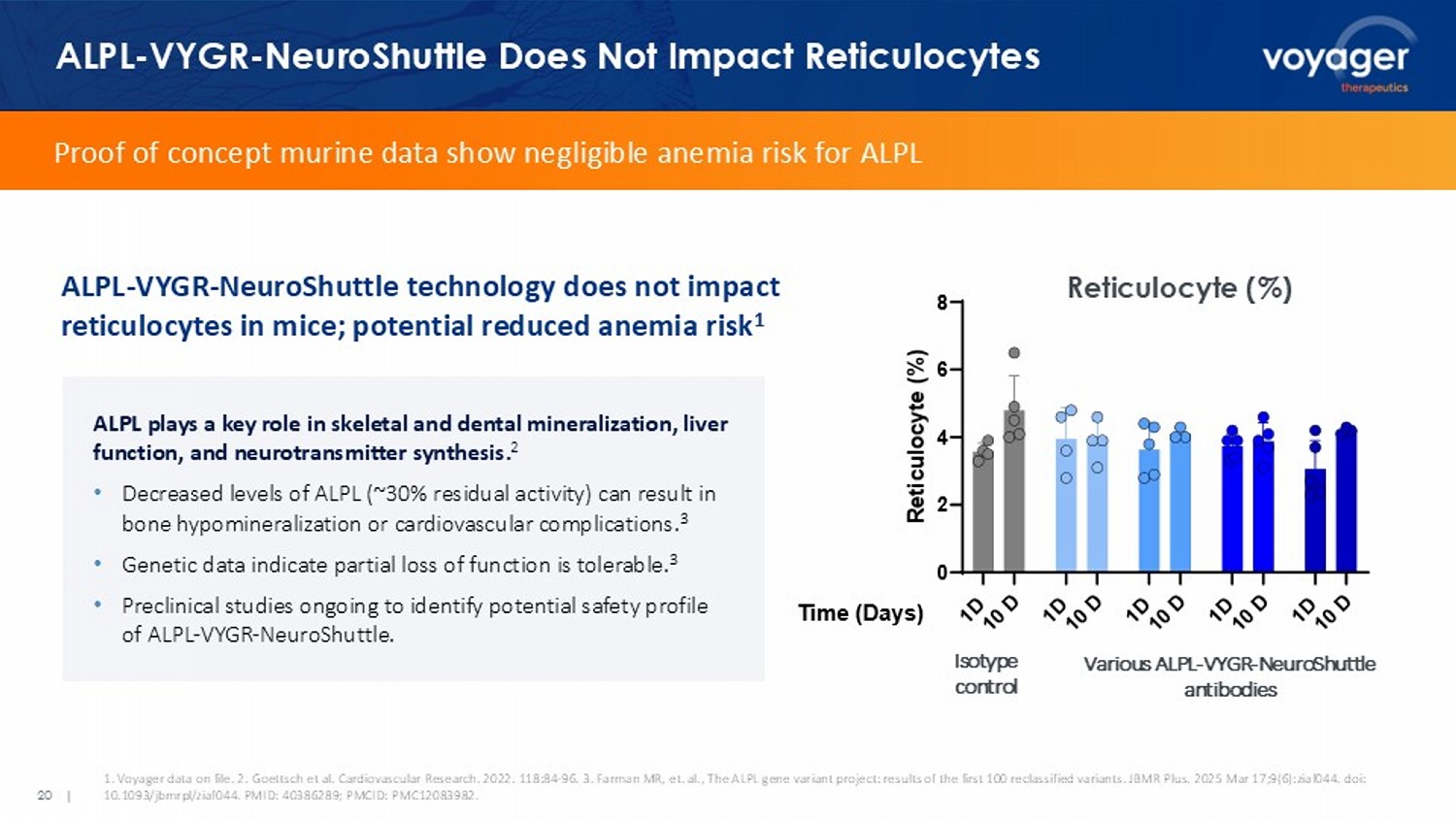
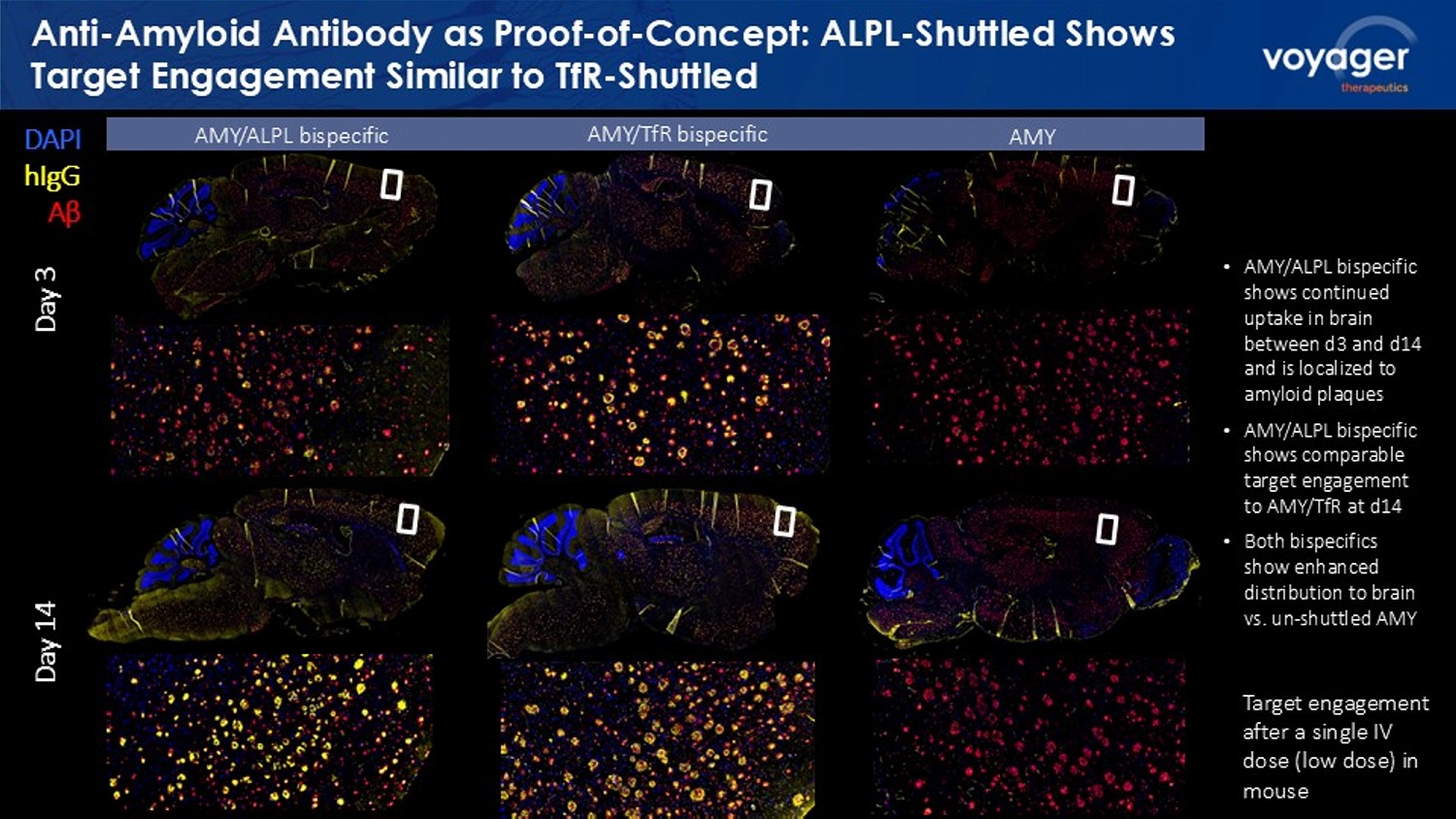
Voyager data on file. 2. Goettsch et al. Cardiovascular Research. 2022. 118:84 - 96. 3. Farman MR, et. al., The ALPL gene variant project: results of the f irst 100 reclassified variants. JBMR Plus. 2025 Mar 17;9(6):ziaf044. doi : 10.1093/ jbmrpl /ziaf044. PMID: 40386289; PMCID: PMC12083982. ALPL plays a key role in skeletal and dental mineralization, liver function, and neurotransmitter synthesis. 2 • Decreased levels of ALPL (~30% residual activity) can result in bone hypomineralization or cardiovascular complications. 3 • Genetic data indicate partial loss of function is tolerable. 3 • Preclinical studies ongoing to identify potential safety profile of ALPL - VYGR - NeuroShuttle . Proof of concept murine data show negligible anemia risk for ALPL ALPL - VYGR - NeuroShuttle technology does not impact reticulocytes in mice; potential reduced anemia risk 1 Reticulocyte (%) 7L P H ' D \ V 5 H W L F X O R F \WH 9DULRXV$/3/ 1HXUR6KXWWOH DQWLERGLHV ,VRW\SH FQWU Various ALPL - VYGR - NeuroShuttle antibodies Isotype control Anti - Amyloid Antibody as Proof - of - Concept: ALPL - Shuttled Shows Target Engagement Similar to TfR - Shuttled DAPI hIgG Aβ Day 3 Day 14 AMY/ALPL bispecific AMY/ TfR bispecific AMY • AMY/ALPL bispecific shows continued uptake in brain between d3 and d14 and is localized to amyloid plaques • AMY/ALPL bispecific shows comparable target engagement to AMY/ TfR at d14 • Both bispecifics show enhanced distribution to brain vs. un - shuttled AMY Target engagement after a single IV dose (low dose) in mouse
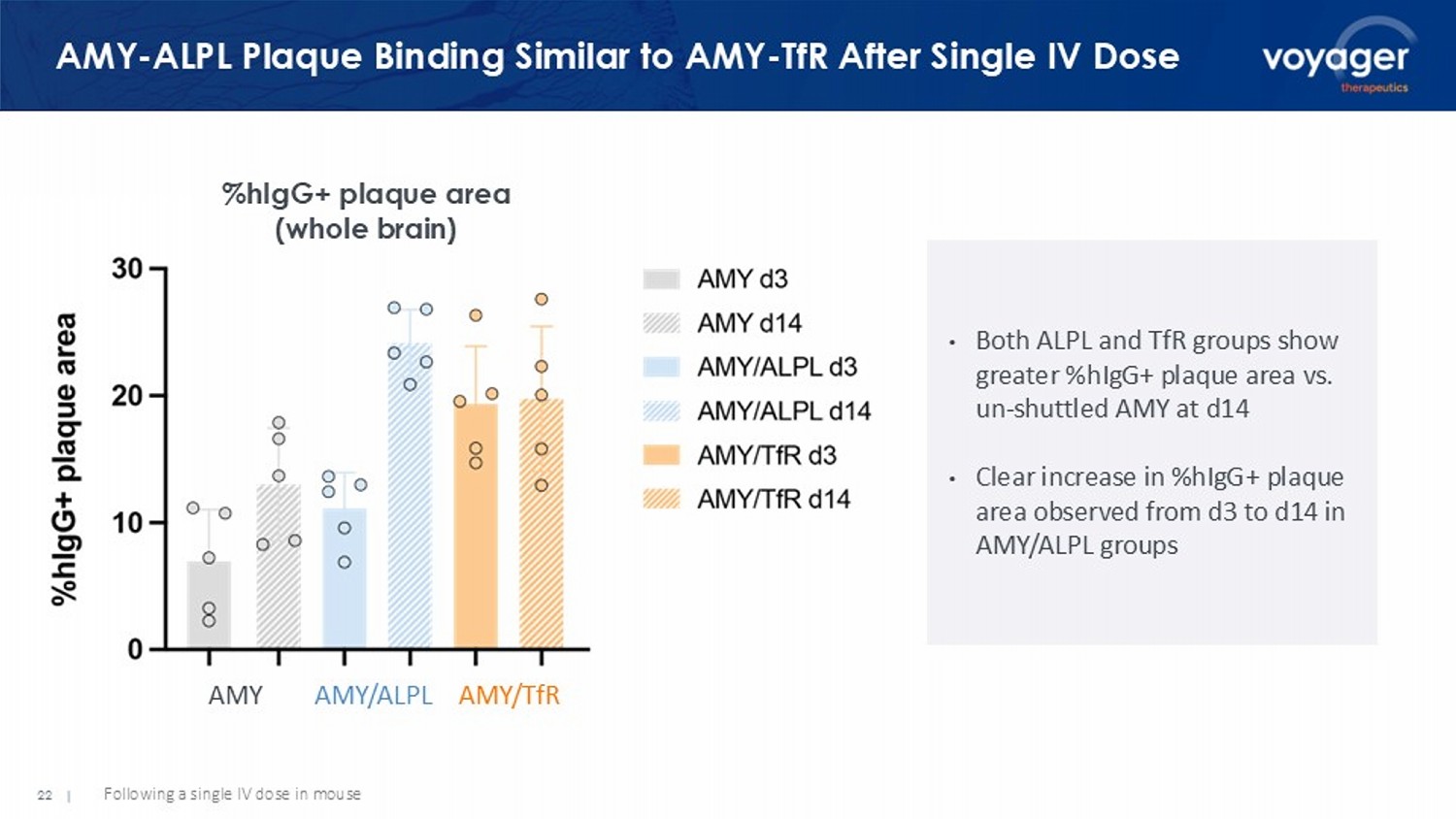
22 | AMY - ALPL Plaque Binding Similar to AMY - TfR After Single IV Dose % hIgG + plaque area (whole brain) • Both ALPL and TfR groups show greater % hIgG + plaque area vs.

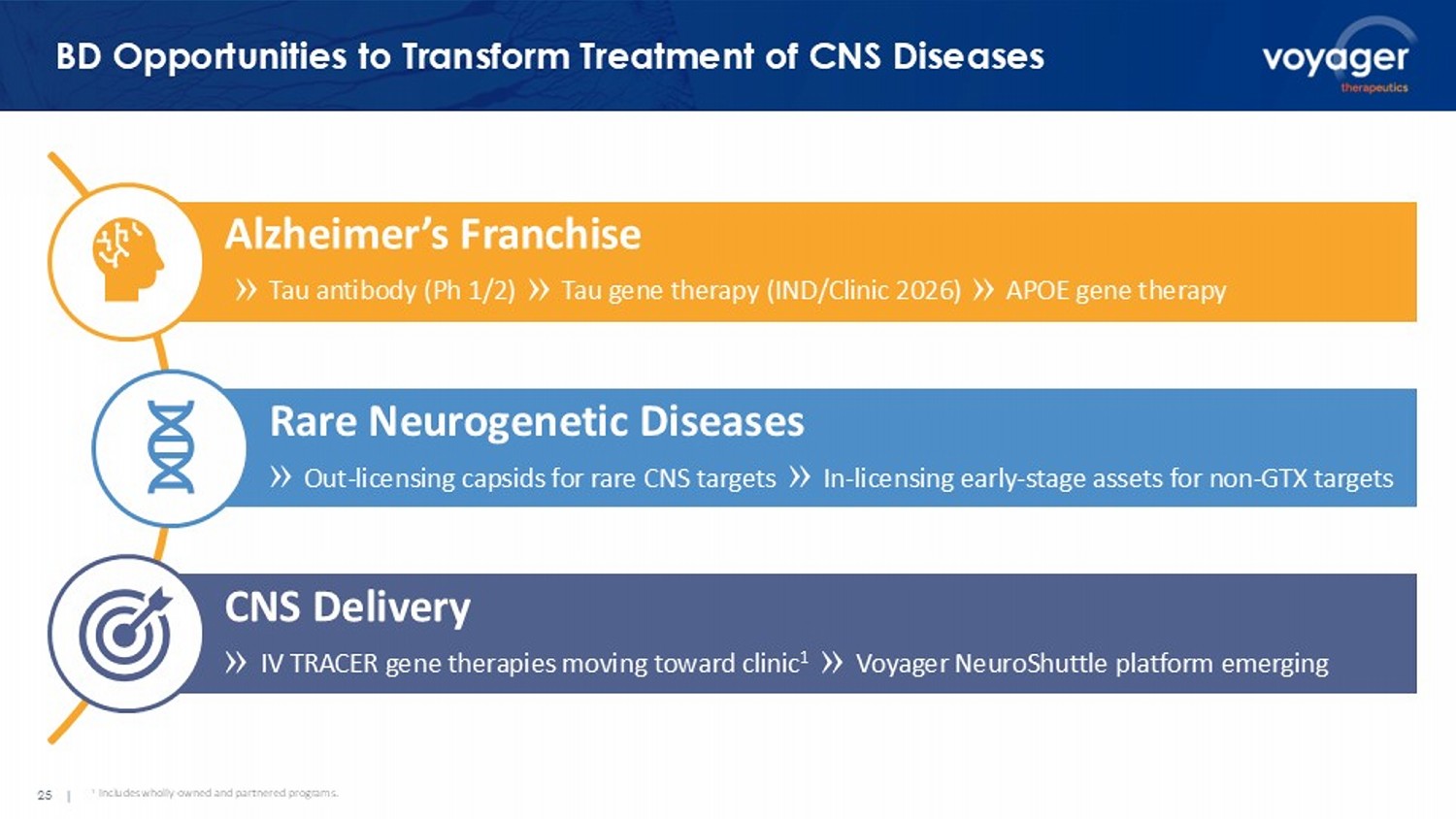
un - shuttled AMY at d14 • Clear increase in % hIgG + plaque area observed from d3 to d14 in AMY/ALPL groups AMY AMY/ALPL AMY/ TfR Following a single IV dose in mouse 23 | Market + Acquirers Appreciate Shuttle Opportunity and Value Multiple potential opportunities for Voyager’s NeuroShuttle platform to create value: • Increase efficacy by increasing on - target delivery • Improve safety by reducing peripheral exposure • Lower COGS by reducing dose needed • Expand opportunity for shuttles: ALPL first of multiple receptors identified VYGR - NeuroShuttle is a priority for Voyager. Platform optimization and program development across therapeutic modalities ongoing, in rodents and non - human primates. Phase Indication # of Asset(s) Value ($mm) Target Partner Preclinical Neurodegenerative Disease Platform $2,720 Phase I Alzheimer’s Disease 1 + Platform $1,400 Preclinical Alzheimer’s Disease 2 $1,350 Preclinical Parkinson’s Disease 1 $1,060 Preclinical Neurodegenerative Disease 1 $800 Registration Neuromuscular Disease 3 + Platform $12,000 Preclinical Neurology Platform $2,000 Select M&A / Licensing Deals in the Shuttling Technology Space M&A Deals 25 | Alzheimer’s Franchise » Tau antibody (Ph 1/2) » Tau gene therapy (IND/Clinic 2026) » APOE gene therapy Rare Neurogenetic Diseases » Out - licensing capsids for rare CNS targets » In - licensing early - stage assets for non - GTX targets CNS Delivery » IV TRACER gene therapies moving toward clinic 1 » Voyager NeuroShuttle platform emerging BD Opportunities to Transform Treatment of CNS Diseases 1 I ncludes wholly - owned and partnered programs .

Business 24 |
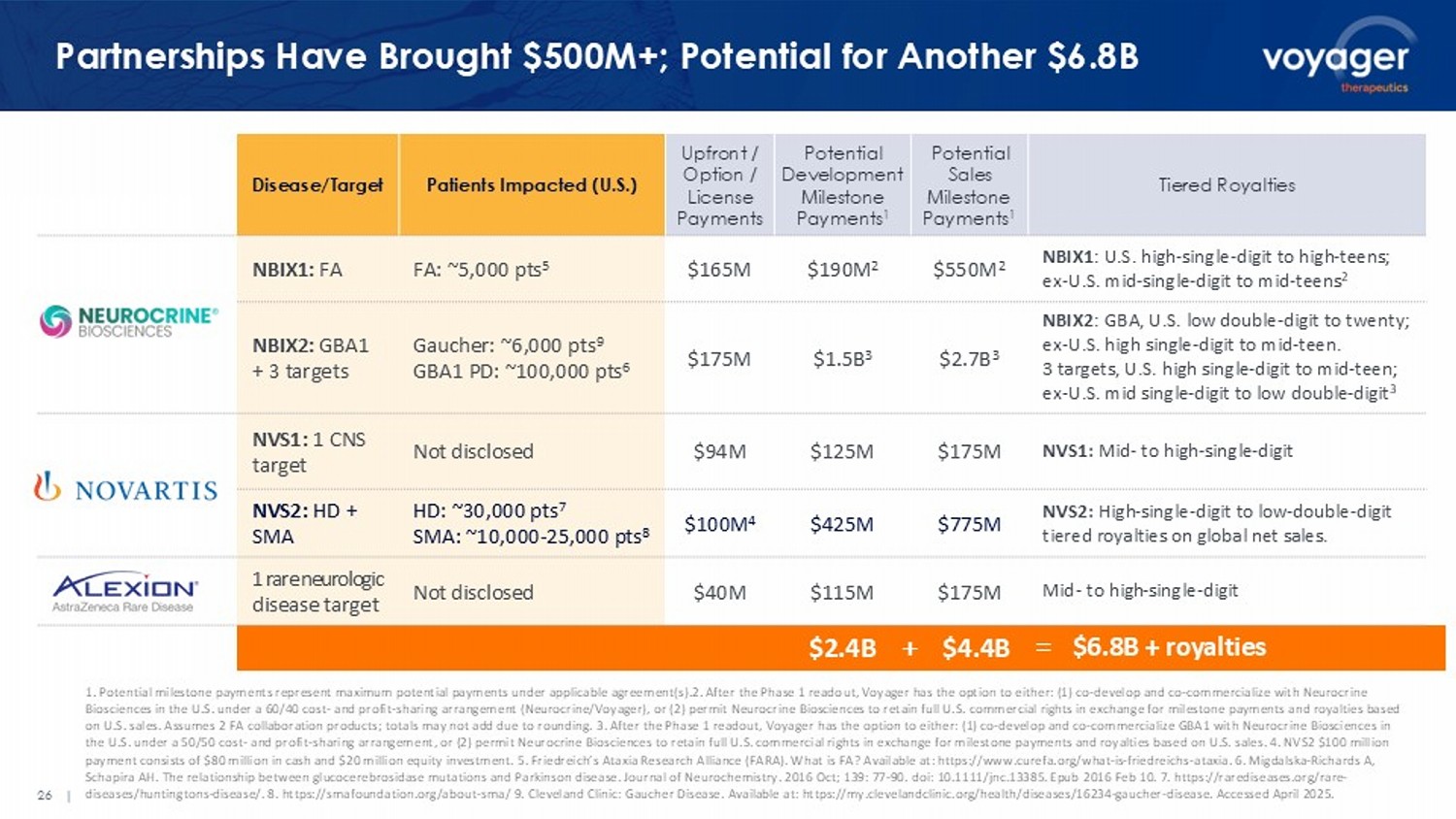
26 | Partnerships Have Brought $500M+; Potential for Another $6.8B Tiered Royalties Potential Sales Milestone Payments 1 Potential Development Milestone Payments 1 Upfront / Option / License Payments Patients Impacted (U.S.) Disease/Target NBIX1 : U.S. high - single - digit to high - teens; ex - U.S. mid - single - digit to mid - teens 2 $550M 2 $190M 2 $165M FA: ~5,000 pts 5 NBIX1: FA NBIX2 : GBA , U.S. low double - digit to twenty; ex - U.S. high single - digit to mid - teen. 3 targets, U.S. high single - digit to mid - teen; ex - U.S. mid single - digit to low double - digit 3 $2.7B 3 $1.5B 3 $175M Gaucher: ~6,000 pts 9 GBA1 PD: ~100,000 pts 6 NBIX2: GBA1 + 3 targets NVS1: Mid - to high - single - digit $175M $125M $94M Not disclosed NVS1: 1 CNS target NVS2: High - single - digit to low - double - digit tiered royalties on global net sales. $775M $425M $100M 4 HD: ~30,000 pts 7 SMA: ~10,000 - 25,000 pts 8 NVS2: HD + SMA Mid - to high - single - digit $175M $115M $40M Not disclosed 1 rare neurologic disease target 1. Potential milestone payments represent maximum potential payments under applicable agreement(s).2. After the Phase 1 reado ut, Voyager has the option to either: (1) co - develop and co - commercialize with Neurocrine Biosciences in the U.S. under a 60/40 cost - and profit - sharing arrangement (Neurocrine/Voyager), or (2) permit Neurocrine Biosci ences to retain full U.S. commercial rights in exchange for milestone payments and royalties based on U.S. sales. Assumes 2 FA collaboration products; totals may not add due to rounding. 3. After the Phase 1 readout, Voyager ha s the option to either: (1) co - develop and co - commercialize GBA1 with Neurocrine Biosciences in the U.S. under a 50/50 cost - and profit - sharing arrangement, or (2) permit Neurocrine Biosciences to retain full U.S. commercial rights in exchange for milestone payments and royalties based on U.S. sales. 4. NVS2 $100 million payment consists of $80 million in cash and $20 million equity investment. 5. Friedreich’s Ataxia Research Alliance (FARA). W hat is FA? Available at: https://www.curefa.org/what - is - friedreichs - ataxia. 6. Migdalska - Richards A, Schapira AH. The relationship between glucocerebrosidase mutations and Parkinson disease. Journal of Neurochemistry. 2016 Oct; 139: 77 - 9 0. doi : 10.1111/jnc.13385. Epub 2016 Feb 10. 7. https://rarediseases.org/rare - diseases/huntingtons - disease/. 8. https://smafoundation.org/about - sma/ 9. Cleveland Clinic: Gaucher Disease. Available at: https ://my.clevelandclinic.org/health/diseases/16234 - gaucher - disease. Accessed April 2025. $2.4B $ 4.4 B + = $ 6.8 B + royalties 27 | Management Team: Extensive Neurology Expertise Trista Morrison Chief Corporate Affairs Officer, Chief of Staff to CEO Todd Carter, Ph.D.

Chief Scientific Officer Robin Swartz Chief Business Officer, Chief Operating Officer Al Sandrock , M.D., Ph.D. Chief Executive Officer Toby Ferguson, M.D. Chief Medical Officer Nathan Jorgensen, Ph.D.

Chief Financial Officer Recent Achievements and Upcoming Milestones Anticipated Voyager NeuroShuttle unveiled; ALPL - VYGR - NeuroShuttle program advanced into pipeline Q3 2025 Enrollment complete in VY7523 (anti - tau antibody) multiple ascending dose trial in AD patients Q4 2025 Complete GLP toxicology study with VY1706 (tau silencing gene therapy) for AD Q1 2026 IND filing with VY1706 Q2 2026 Data from BIIB080 Phase 2 trial in 400+ AD patients; potential to derisk tau knockdown approach Mid - 2026 First - in - human dosing of VY1706 ; potential first TRACER - derived novel capsid in the clinic H2 2026 Initial tau PET imaging data for VY7523 in AD patients H2 2026 NBIX expects clinical trial initiation for NBIB - ’223 for Friedreich’s ataxia 2026 NeuroShuttle data on NHP translatability, safety, program advancement 2026 Potential for additional value - creating partnerships; discussions ongoing Ongoing 28 | 1 Based on our current operating plans, cash and cash equivalents and marketable securities as of Sept. 30, 2025, along with am ou nts expected to be received as reimbursement for development costs under the Neurocrine and Novartis collaborations and interest income. Runway into 2028; this does not include any potential milestone payments from existing partnerships 1 I want to take a moment to share why 2026 is a pivotal year in Voyager’s journey to become a premier multi-modality neurotherapeutics company.

Thank You www.voyagertherapeutics.com
Exhibit 99.2
Dear fellow stakeholders,
In particular, I see three principal pillars of value emerging this year:
| 1. | Transformative year for tau, with two shots on goal |
| · | Biogen’s antisense oligonucleotide BIIB080 laid a compelling foundation for tau targeting, and Phase II data are expected mid-2026. If positive, this would demonstrate that tau knockdown is a viable therapeutic approach. Voyager’s own knockdown program, the one-time intravenous gene therapy VY1706, is expected to achieve first-in-human dosing in H2 2026. | |
| · | Our multiple ascending dose trial of anti-tau antibody VY7523 is fully enrolled with tau PET imaging data expected in H2 2026. While third-party data have been mixed, we believe that the targeted epitope and specificity for pathological forms of the protein will matter, and that VY7523 is differentiated on both fronts. |
| 2. | Validating brain-targeted capsids in humans |
| · | Four of our wholly owned and partnered I.V.-delivered, neuro gene therapy programs have advanced into or completed preclinical toxicology studies. We are optimistic about the potential to address the vast unmet need in neurological disease. | |
| · | In addition to VY1706 (per above), Neurocrine has stated that it intends to initiate a clinical trial with NBIB-‘223 for Friedreich’s ataxia in 2026. In preclinical studies, I.V. administered NBIB-‘223 resulted in protein expression in the brain and heart – a first for the field. |
| 3. | Demonstrating the value of Voyager NeuroShuttle™ |
| · | In a proof-of-concept study using anti-amyloid antibodies, the ALPL-NeuroShuttle showed similar target engagement to a TfR shuttle after a single IV dose, but with more sustained brain exposure. We plan to provide data on NHP translatability, safety, and programs in 2026. | |
| · | Our initial data have generated strong partnering interest – not only in our lead ALPL NeuroShuttle, but in our ability to identify additional novel receptors, a handful of which are already in evaluation. |
In closing, I want to thank our team for our progress in 2025 and setting us up to advance multiple programs, validate our platforms, and build long-term shareholder value in 2026. We appreciate your continued support as we execute on these opportunities.
Sincerely,
Al Sandrock, M.D., Ph.D.
CEO of Voyager