UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, DC 20549
FORM 8-K
CURRENT REPORT
PURSUANT TO SECTION 13 OR 15(d) OF THE
SECURITIES EXCHANGE ACT OF 1934
Date of Report (Date of earliest event reported): November 13, 2025
Traws Pharma, Inc.
(Exact name of Registrant as specified in its charter)
| Delaware | 001-36020 | 22-3627252 | ||
| (State or Other Jurisdiction of Incorporation or Organization) |
(Commission File Number) |
(I.R.S. Employer Identification No.) |
|
12 Penns Trail Newtown, PA 18940 |
| (267) 759-3680 |
(Address,
Including Zip Code, and Telephone Number, Including Area Code, of Registrant’s Principal Executive
Offices)
Not Applicable
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| ¨ | Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ¨ | Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ¨ | Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ¨ | Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered |
| Common stock, par value $.01 per share | TRAW | The Nasdaq Stock Market LLC |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ¨
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Item 2.02 Results of Operations and Financial Condition.
The information provided below in “Item 7.01 - Regulation FD Disclosure” of this Current Report on Form 8-K (this “Current Report”) regarding the Earnings Release is incorporated by reference into this Item 2.02.
Item 7.01 Regulation FD Disclosure.
On November 13, 2025, Traws Pharma, Inc. (the “Company”) issued a press release (the “Earnings Release”) announcing its financial results for the quarter ended September 30, 2025, a copy of which is furnished as Exhibit 99.1 hereto and incorporated herein by reference.
Additionally, on November 13, 2025, the Company began using a new corporate presentation. A copy of that corporate presentation is furnished as Exhibit 99.2 hereto and incorporated herein by reference. The presentation will also be posted to the Company’s website.
The information set forth under Item 7.01 of this Current Report, including Exhibits 99.1 and 99.2 attached hereto, is being furnished and shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of such section. The information in Item 7.01 of this Current Report, including Exhibits 99.1 and 99.2, shall not be incorporated by reference into any filing under the Securities Act of 1933, as amended, or the Exchange Act, regardless of any incorporation by reference language in any such filing, except as expressly set forth by specific reference in such a filing. This Current Report will not be deemed an admission as to the materiality of any information in this Current Report that is required to be disclosed solely by Regulation FD.
Forward-Looking Statements
This Current Report, including Exhibits 99.1 and 99.2, contains certain forward-looking statements that involve substantial risks and uncertainties. When used herein, the terms “anticipates,” “expects,” “estimates,” “believes,” “will” and similar expressions, as they relate to the Company or its management, are intended to identify such forward-looking statements.
Forward-looking statements in this Current Report, including Exhibits 99.1 and 99.2, or hereafter, including in other publicly available documents filed with the Securities and Exchange Commission, reports to the stockholders of the Company and other publicly available statements issued or released by the Company involve known and unknown risks, uncertainties and other factors which could cause the Company’s actual results, performance (financial or operating) or achievements to differ from the future results, performance (financial or operating) or achievements expressed or implied by such forward-looking statements. Such future results are based upon management’s best estimates based upon current conditions and the most recent results of operations. These risks include, but are not limited to, the risks set forth herein and in such other documents filed with the Securities and Exchange Commission, each of which could adversely affect the Company’s business and the accuracy of the forward-looking statements contained herein.
| Item 9.01 | Financial Statements and Exhibits. |
(d) Exhibits.
| Exhibit No. | Description | |
| 99.1 | Press Release, dated November 13, 2025. | |
| 99.2 | Investor Presentation, dated November 2025. | |
| 104 | Cover Page Interactive Data File (embedded within the inline XBRL Document) |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, the Registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| Date: November 13, 2025 | TRAWS PHARMA, INC. | |
| By: | /s/ Iain Dukes | |
| Iain Dukes | ||
| Chief Executive Officer | ||
Exhibit 99.1
Traws Pharma
Reports Third Quarter 2025 Results
and Business Highlights
Progressing Phase 2 studies for ratutrelvir, a ritonavir-free, protease inhibitor regimen in development for Acute and Long COVID with expected top line data by year end
Significant intellectual property and other assets acquired
Significant cost reductions quarter over quarter
NEWTOWN, PA, November 13, 2025 (GLOBE NEWSWIRE) – Traws Pharma, Inc. (NASDAQ: TRAW) (“Traws Pharma”, “Traws” or “the Company”), a clinical-stage biopharmaceutical company developing novel therapies to target critical threats to human health from respiratory viral diseases, today reported financial results for the quarter ended September 30, 2025 and provided recent business highlights for its antiviral programs.
Recent Highlights and Anticipated Milestones:
Product Development
Ratutrelvir (COVID): Potential best-in-class therapy to minimize the risk of viral rebound and Long COVID
A ritonavir-independent investigational oral Main protease (Mpro) inhibitor designed to be administered as a single dose, with potential for once-daily, 10-day dosing
| · | Intended Indication: Treatment of acute COVID infection, with potential to reduce the risk of COVID rebound and Long COVID, including in currently PAXLOVID® -ineligible patients |
| · | U.S. Market Opportunity: Estimated to be a multi-billion dollar opportunity1 |
| · | Recent Updates: |
| o | Phase 2 initiation: Dosing initiated in a Phase 2 non-inferiority study to evaluate the effects of ratutrelvir, compared to PAXLOVID®, in newly diagnosed COVID patients. The program also includes a separate single arm study to evaluate the safety and efficacy of ratutrelvir in patients who are ineligible for treatment with PAXLOVID®. |
| o | Prior Phase 1 studies showed ratutrelvir maintained plasma drug levels four times above the EC90 over a 10-day treatment period in healthy volunteers, with no drug-induced metabolism and no treatment-related adverse events. |
| · | Next Steps: |
| o | Topline data expected by year-end 2025 from both the Phase 2 non-inferiority study and the single-arm study in PAXLOVID®-ineligible subjects |
Tivoxavir Marboxil (TXM, Bird flu and seasonal flu): Potential best-in-class single-dose treatment for bird flu and seasonal flu
A single-dose, investigational CAP-dependent endonuclease inhibitor
| · | Intended Indication: Treatment or prevention of H5N1 bird flu and seasonal flu |
| · | U.S. Market: Estimated to be a multi-billion dollar opportunity, including potential government stockpiling2,3 for pandemic preparedness in the short- to medium-term. |
| · | Recent Updates: |
| o | Prior Phase 1 studies showed that a single dose-maintained plasma blood levels above the EC90 for approximately three weeks, with good overall tolerability. Preclinical data support evaluation of TXM in bird flu and seasonal flu based on compelling protection against mortality and disease in three well accepted flu “challenge” models (mice, ferrets and non-human primates) using an H5N1 virus isolated from a dairy worker. |
| · | Next Steps: |
| o | Stockpiling readiness: Filing of IND to enable advancement of discussions with BARDA regarding the inclusion of TXM in the drug stock piling initiative for pandemic preparedness. |
Legacy Programs: Traws’ strategic objective for its legacy oncology assets (rigosertib and narazaciclib) is to establish additional partnerships for further development.
Rigosertib
| · | Intended Indication: Treatment of recessive dystrophic epidermolysis bullosa associated locally advanced or metastatic squamous cell carcinoma (RDEB SCC) |
| · | U.S. Market: RDEB SCC is an ultra rare, complicated monogenic disease with a highly aggressive, early metastasizing course, and a cumulative risk of death of ~70-79% by 45-55 years of age10,11 |
| · | Recent Updates: |
| o | Publication of key clinical efficacy data for rigosertib in RDEB SCC in the British Journal of Dermatology12 showing a compelling overall response rate of 80%, with complete responses in 50% of evaluable patients and good overall tolerability. These data suggest that rigosertib has the potential to address the substantial unmet need for approved therapies to treat RDEB SCC. |
| · | Next Steps: |
| o | Exploring pathways to advance this important potential medicine: Traws is seeking partnership opportunities to support further development of rigosertib |
Management Updates:
| · | John Leaman, MD, named Independent Board Director on October 6, 2025, bringing significant expertise in finance, M&A and corporate strategy. |
| · | Permanent appointment of Iain Dukes, MA, D Phil, as CEO, and Charles Parker, as CFO, confirmed by the Board on October 6, 2025. |
Financial Results:
Cash and cash equivalents: As of September 30, 2025, the Company had cash and cash equivalents of approximately $6.4 million, compared to approximately $21.3 million as of December 31, 2024.
Intangible Assets for the quarter ended September 30, 2025 were $2.6 million compared to zero for the comparable period in 2024. On September 9, 2025, the Company announced that it had entered into an Asset Purchase Agreement with Viriom, Inc., a related party. The transaction includes the Company’s purchase of key intellectual property and other assets associated with a pyrrolidine antiviral compound. Total consideration paid for the assets was $2.6 million, which includes legal costs incurred in consummating the Asset Purchase Agreement of $0.2 million. The purchased patent has a useful life of 15 years and will be amortized on a straight-line basis over this period, as the economic benefits are expected to be realized evenly over the patent’s useful life.
Revenue for the quarter ended September 30, 2025, was zero, compared to $57 thousand for the comparable period in 2024.
Research and development (R&D) expense for the quarter ended September 30, 2025, totaled $2.3 million, compared to $5.1 million for the comparable period in 2024. This decrease of $2.8 million primarily relates to decrease in expenses related to the virology and oncology programs.
General and administrative (G&A) expense for the quarter ended September 30, 2025, totaled $1.7 million, compared to $3.5 million for the comparable period in 2024. This decrease of $1.8 million was primarily attributable to a decrease in professional and consulting fees.
Net Income (loss): The net loss for the quarter ended September 30, 2025 was $4.0 million, or a net loss of $0.34 per basic common and diluted common share. This compares to a net loss of $8.5 million, or a net loss of $1.49 per basic and diluted common share, for the quarter ended September 30, 2024.
Shares Outstanding: Traws had 7,990,867 shares of common stock outstanding as of November 10, 2025.
About Ratutrelvir
Ratutrelvir is an investigational oral, small molecule Mpro (3CL protease) inhibitor designed to be a broadly acting treatment for SARS-CoV-2/COVID-19 that is used without ritonavir. It has demonstrated in vitro activity against a range of virus strains. Preclinical and Phase 1 studies show that ratutrelvir does not require co-administration with a metabolic inhibitor, such as ritonavir, which could avoid ritonavir-associated drug-drug interactions7, and potentially enable wider patient use. Phase 1 data also show that ratutrelvir’s pharmacokinetic (PK) profile demonstrated maintenance of target blood plasma levels approximately 13 times above the EC50 using the target Phase 2 dosing regimen of 600 mg/day for ten days, which may also reduce the likelihood of clinical rebound and, consequently, reduce the risk for Long COVID8. Industry data indicate that COVID treatment represents a potential multi-billion dollar market opportunity1.
About Tivoxavir Marboxil
Tivoxavir marboxil (TXM) is an investigational oral, small molecule CAP-dependent endonuclease inhibitor designed to be administered as a single-dose for the treatment of bird flu and seasonal influenza. It has shown potent in vitro activity against a range of influenza strains in preclinical studies, including a human isolate of the highly pathogenic avian flu H5N1 (bird flu). Consistent, positive preclinical data from three animal species indicate that a single dose of TXM demonstrated a therapeutic effect against H5N1 bird flu. Seasonal influenza represents an estimated multi-billion dollar antiviral market opportunity, largely driven by global health organizations, practice guidelines and government tenders and inclusion in drug stock piling initiatives2,3, with upside potential from potential pandemic flu outbreaks including H5N1 bird flu. We believe that these data support further development of TXM as a treatment for bird flu.
Source information
| 1. | Pfizer Inc. 10K report 2024, Feb 27, 2025 |
| 2. | Per link |
| 3. | TRAW data on file |
| 4. | Mellerio et al. Br J Dermatol. 2016 Jan; 174(1):56-67. doi: 10.1111/bjd.14104 |
| 5. | Fine et al. J Am Acad Dermatol. 2009 Feb; 60(2):203-11. doi: 10.1016/j.jaad.2008.09.035. |
| 6. | Martin Laimer, Andrew P South, Elisabeth Mayr, Sophie Kitzmueller, Lauren Banner, Michael Alexander, Linda Hosler, Henry Yang, Matthew Parris, Meena Arora, Georg Zimmermann, Gregor Schweighofer-Zwink, Johann W Bauer, Neda Nikbakht, Efficacy and Safety of Rigosertib in Patients with Recessive Dystrophic Epidermolysis Bullosa Associated Advanced/Metastatic Cutaneous Squamous Cell Carcinoma, British Journal of Dermatology, 2025;, ljaf205, https://doi.org/10.1093/bjd/ljaf205 |
| 7. | https://ascpt.onlinelibrary.wiley.com/doi/pdf/10.1002/cpt.2646 |
| 8. | Carly Herbert et al. (2025) Clinical Infectious Diseases. https://doi.org/10.1093/cid/ciae539 = |
Third-party products mentioned herein are the trademarks of their respective owners.
About Traws Pharma, Inc.
Traws Pharma is a clinical stage biopharmaceutical company dedicated to developing novel therapies to target critical threats to human health in respiratory viral diseases. Traws integrates antiviral drug development, medical intelligence and regulatory strategy to meet real world challenges in the treatment of viral diseases. We are advancing novel investigational oral small molecule antiviral agents that have potent activity against difficult to treat or resistant virus strains that threaten human health: COVID-19/Long COVID and bird flu and seasonal influenza. Ratutrelvir is in development as a ritonavir-independent COVID treatment, targeting the Main protease (Mpro or 3CL protease). Tivoxavir marboxil is in development as a single dose treatment for bird flu and seasonal influenza, targeting the influenza cap-dependent endonuclease (CEN).
Traws is actively seeking development and commercialization partners for its legacy clinical oncology programs, rigosertib and narazaciclib. More details can be found on Traws’ website at https://www.ir.trawspharma.com/partnering.
For more information, please visit www.trawspharma.com and follow us on LinkedIn.
Forward-Looking Statements
Some of the statements in this release are forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, Section 21E of the Securities Exchange Act of 1934, as amended, and the Private Securities Litigation Reform Act of 1995, and involve risks and uncertainties including statements regarding the Company, its business and product candidates, including the potential opportunity, market size, benefits, effectiveness, safety, and the clinical and regulatory plans for ratutrelvir and tivoxavir marboxil, as well as plans for its legacy programs. The Company has attempted to identify forward-looking statements by terminology including “believes”, “estimates”, “anticipates”, “expects”, “plans”, “intends”, “may”, “could”, “might”, “will”, “should”, “preliminary”, “encouraging”, “approximately” or other words that convey uncertainty of future events or outcomes. Although Traws believes that the expectations reflected in such forward-looking statements are reasonable as of the date made, expectations may prove to have been materially different from the results expressed or implied by such forward looking statements. These statements are only predictions and involve known and unknown risks, uncertainties, and other factors, including the success and timing of Traws’ clinical trials, including when Traws will report results of the Phase 2 studies of ratutrelvir; the potential efficacy of ratutrelvir for the treatment of COVID-19, including the potential to reduce the risk of COVID rebound and Long COVID; the potential for ratutrelvir to gain market acceptance, if and when regulatory approval is obtained, or to become the new standard of care; Traws’ interactions with the FDA, BARDA and similar foreign regulators; collaborations; market conditions; regulatory requirements and pathways for approval; the ongoing need for improved therapy to reduce the frequency of clinical rebound and the concomitant risk for Long COVID; the extent of the spread and threat of the bird flu; the Company’s cash projections; Traws’ ability to raise additional capital when needed; and those discussed under the heading “Risk Factors” in Traws’ filings with the U.S. Securities and Exchange Commission (SEC). Any forward-looking statements contained in this release speak only as of its date. Traws undertakes no obligation to update any forward-looking statements contained in this release to reflect events or circumstances occurring after its date or to reflect the occurrence of unanticipated events, except to the extent required by law.
Traws Pharma Contact:
Charles Parker
Traws Pharma, Inc.
cparker@trawspharma.com
www.trawspharma.com
Investor Contact:
John Fraunces
LifeSci Advisors, LLC
917-355-2395
jfraunces@lifesciadvisors.com
Traws Pharma, Inc.
Condensed Consolidated Balance Sheets
(unaudited)
| September 30, | December 31, | |||||||
| 2025 | 2024 | |||||||
| Assets | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | 6,420,000 | $ | 21,338,000 | ||||
| Tax incentive and other receivables | 1,991,000 | 1,765,000 | ||||||
| Prepaid expenses and other assets | 1,214,000 | 1,848,000 | ||||||
| Total current assets | 9,625,000 | 24,951,000 | ||||||
| Property and equipment, net | 7,000 | 10,000 | ||||||
| Intangible assets, net | 2,571,000 | — | ||||||
| Other assets | 240,000 | 1,000 | ||||||
| Total assets | $ | 12,443,000 | $ | 24,962,000 | ||||
| Liabilities and stockholders’ equity (deficit) | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 5,105,000 | $ | 8,186,000 | ||||
| Accrued expenses and other liabilities | 2,782,000 | 3,121,000 | ||||||
| Deferred revenue | — | 226,000 | ||||||
| Total current liabilities | 7,887,000 | 11,533,000 | ||||||
| Deferred revenue, non-current | — | 2,565,000 | ||||||
| Warrant liabilities | 11,000 | 42,494,000 | ||||||
| Total liabilities | 7,898,000 | 56,592,000 | ||||||
| Stockholders’ equity (deficit): | ||||||||
| Series C Preferred stock, $0.01 par value, 5,000,000 shares authorized, 7,440 shares issued, 6,737 and 7,398 shares outstanding at September 30, 2025 and December 31, 2024, respectively | — | — | ||||||
| Common stock, $0.01 par value, 250,000,000 shares authorized, 7,131,151 and 3,650,731 shares issued and outstanding at September 30, 2025 and December 31, 2024, respectively | 71,000 | 36,000 | ||||||
| Additional paid-in capital | 637,009,000 | 617,530,000 | ||||||
| Accumulated deficit | (632,541,000 | ) | (649,154,000 | ) | ||||
| Accumulated other comprehensive income (loss) | 6,000 | (42,000 | ) | |||||
| Total stockholders’ equity (deficit) | 4,545,000 | (31,630,000 | ) | |||||
| Total liabilities and stockholders’ equity (deficit) | $ | 12,443,000 | $ | 24,962,000 | ||||
Traws Pharma, Inc.
Condensed Consolidated Statements of Operations (unaudited)
| Three Months Ended September 30, | Nine Months Ended September 30, | |||||||||||||||
| 2025 | 2024 | 2025 | 2024 | |||||||||||||
| Revenue | $ | — | $ | 57,000 | $ | 2,790,000 | $ | 170,000 | ||||||||
| Operating expenses: | ||||||||||||||||
| Acquired in-process research and development | — | — | — | 117,464,000 | ||||||||||||
| Research and development | 2,311,000 | 5,113,000 | 7,108,000 | 10,989,000 | ||||||||||||
| General and administrative | 1,744,000 | 3,480,000 | 6,189,000 | 8,813,000 | ||||||||||||
| Total operating expenses | 4,055,000 | 8,593,000 | 13,297,000 | 137,266,000 | ||||||||||||
| Loss from operations | (4,055,000 | ) | (8,536,000 | ) | (10,507,000 | ) | (137,096,000 | ) | ||||||||
| Change in fair value of warrant liability | (3,000 | ) | — | 26,656,000 | — | |||||||||||
| Other income, net | 96,000 | 61,000 | 464,000 | 495,000 | ||||||||||||
| Net (loss) income | $ | (3,962,000 | ) | $ | (8,475,000 | ) | $ | 16,613,000 | $ | (136,601,000 | ) | |||||
| Net (loss) income attributable to common stockholders, basic and diluted | $ | (3,035,000 | ) | $ | (1,433,000 | ) | $ | 12,263,000 | $ | (33,255,000 | ) | |||||
| Weighted-average shares of common stock outstanding, basic | 8,818,859 | 961,530 | 7,851,515 | 1,043,781 | ||||||||||||
| Net (loss) income per share of common stock, basic | $ | (0.34 | ) | $ | (1.49 | ) | $ | 1.56 | $ | (31.86 | ) | |||||
| Weighted-average shares of common stock outstanding, diluted | 8,818,859 | 961,530 | 8,006,487 | 1,043,781 | ||||||||||||
| Net (loss) income per share of common stock, diluted | $ | (0.34 | ) | $ | (1.49 | ) | $ | 1.53 | $ | (31.86 | ) | |||||
| Net (loss) income attributable to Series C Preferred stockholders, basic and diluted | $ | (927,000 | ) | $ | (7,042,000 | ) | $ | 4,350,000 | $ | (103,346,000 | ) | |||||
| Weighted-average shares of Series C Preferred outstanding, basic and diluted | 6,737 | 11,816 | 6,962 | 8,109 | ||||||||||||
| Net (loss) income per share of Series C Preferred, basic and diluted | $ | (137.60 | ) | $ | (595.97 | ) | $ | 624.82 | $ | (12,744.60 | ) | |||||
Exhibit 99.2


NASDAQ: TRAW November 2025 Corporate Presentation Forward - looking statements 2 This presentation contains, and certain oral statements made by management from time to time may contain, “forward - looking state ments” within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, Section 27A of the Securities Act of 1933, as amended (the “Securiti es Act”), and Section 21E of the Securities Exchange Act of 1934, as amended. Such statements include actions, events, results, strategies and expectations and are often identifiable by use of t he words “believes”, “expects”, “intends”, “anticipates”, “plans”, “seeks”, “estimates”, “projects”, “may”, “will”, “could”, “might”, or “continues” or similar expressions. Forward - looking statem ents within this presentation include, but are not limited to, express or implied statements regarding the nature, strategy, opportunities and focus of Traws Pharma, Inc. (“Traws”); the development, commercial potential and potential benefits of any product candidates; anticipated clinical drug development activities, related timelines and expected milestones; anticipated int eractions with regulatory authorities, including both the FDA and BARDA and similar international regulatory agencies; and other statements that are not historical fact. All statements ot her than statements of historical fact contained in this communication are forward - looking statements. These forward - looking statements are made as of the date they were first issued, a nd were based on the then - current expectations, estimates, forecasts, and projections, as well as the beliefs and assumptions of management. There can be no assurance that f utu re developments affecting the company will be those that have been anticipated. Forward - looking statements are subject to a number of risks and uncertainties, many of which involve factors or circumstances th at are beyond Traws ’ control. Traws ’ actual results could differ materially from those stated or implied in forward - looking statements due to a number of factors, including but not limit ed to ( i ) the uncertainties associated with Traws ’ product candidates, as well as risks associated with the clinical development and regulatory approval of product candidates, includin g p otential delays in the commencement and completion of clinical trials, studies and evaluations; (ii) risks related to the inability of Traws to obtain sufficient additional capital to continue to advance its product candidates and operate its business; (iii) uncertainties in obtaining successful clinical results for product candidates and unexpected costs that may result therefrom; (i v) risks related to the failure to realize any value from product candidates currently being developed and anticipated to be developed in light of inherent risks and difficulties involved in suc cessfully bringing product candidates to market; (v) uncertainties in retaining key personnel, including the executive team and directors; (vi) risks that may stem from changes in the regulato ry, economic and/or political landscape, (vii) disruption and volatility in the global currency, capital, and credit markets; and (viii) risks associated with the possible failure to realize certain an ticipated benefits of Traws ’ 2024 merger with Trawsfynydd Therapeutics, Inc., including with respect to future financial and operating results and the other risks and uncertainties in clu ded in Traws ’ Annual Report on Form 10 - K for the year ended December 31, 2024 and Traws ’ subsequent filings with the U.S. Securities and Exchange Commission (the “SEC”). Actual results and the timing of events co uld differ materially from those anticipated in such forward - looking statements as a result of these risks and uncertainties. You should not place undue reliance on these forward - looking statements, which are made only as of the date hereof or as of the dates indicated in the forward - looking statements. Traws expressly disclaims any obligation or undertaking, unless required by applicable law, to release publicly any updates or revisions to any forward - looking statements contained herein to reflect any change in its expectations with regar d thereto or any change in events, conditions or circumstances on which any such statements are based. This communication does not purport to summarize all of the conditions, risks and oth er attributes of an investment in Traws . This presentation is for informational purposes only and shall not constitute an offer to sell or a solicitation of an offer to buy the securities of the Traws , nor shall there be any sale of any such securities in any state or jurisdiction in which such offer, solicitation, or sale would be unlawful prior to registration or qualification under the se cur ities laws of such state or jurisdiction. No offer of securities shall be made, except by means of a prospectus meeting the requirements of Section 10 of the Securities Act or an exemption therefrom.

Targeting Critical Viral Threats to Human Health 3 Potential first - in - class bird flu program , with an initial goal to build out the US strategic stockpile (estimated 300 million to 600 million doses 2 ) Potential best - in - class COVID program to replace existing therapies (>$6B revenue, 2024), prevent clinical rebound and explore treatment of Long COVID 1 5 F ocusing on small molecule antiviral drugs active against respiratory viruses with pandemic potential Experienced team, supported by recognized institutional investors, including OrbiMed Progressing business development exit strategies for legacy oncology assets (rigosertib and narazaciclib) Source: 1. Pfizer 2024 financial results here, p. 24 , Merck 2024 financial results here, p5 = $5.7B + $964M = >$6B 2. TRAW internal information.

4 Antiviral drugs attack a conserved virus component that does not vary according to seasonal changes in virus Antiviral drugs are small molecules that inhibit the activity of critical viral enzymes and are used specifically to treat active infections or to prevent virus spread to close contacts of an infected person Sources: 1. TRAW information. 2. https://www.nejm.org/doi/full/10.1056/NEJMsb2506929 3. BIKTARVY is a registered trademark of Gilead Science, Inc. 4. PAXLOVID® is a registered trademark of Pfizer, Inc. 5 Antiviral Drugs Represent a Significant Growth Sector for Biopharma 1 V accine use is waning dramatically due to fast development of resistance and regulatory hurdles for approvals, including new guidance restricting broad use in the absence of long - term safety studies 2 Antivirals have experienced continued blockbuster growth (2024 revenue): • BIKTARVY®: $13.4 billion 3 (Gilead: HIV, 2018 introduction) • PAXLOVID®: $5.7 billion 4 (Pfizer: COVID, 2021 introduction) Traws ’ next generation antivirals are designed to maximize efficacy and safety while overcoming resistance to existing agents Comments/Potential Next Steps Phase 3 Phase 2 Phase 1 Preclinical Program/Market Respiratory Viruses • Demonstrated suppression of a broad range of native and resistant strains • Positive Phase 1 safety, PK/PD data in 10 - day regimen without ritonavir • Phase 2 studies initiated in Q4 2025, topline data expected by YE 2025 COVID - 19 (acute treatment and long COVID) Ratutrelvir • Demonstrated protection against mortality and disease in 3 species • Positive Phase 1 safety and PK data • Filing of IND to enable advancement of BARDA discussions on the drug stock piling initiative for pandemic preparedness Bird Flu/Seasonal flu Tivoxavir marboxil (TXM) High - Potential Antiviral Pipeline Focused on Critical Threats to Human Health from Respiratory Viral Diseases 5


Key Program Highlights 6 Source: TRAW data on file. Notes: PK = pharmacokinetics. PAXLOVID® is a registered trademark of Pfizer, Inc. Tamiflu® is a tr ade mark registered of Roche. Xofluza ® is a registered trademark of Genentech Pharmaceuticals COVID - Ratutrelvir Broad potency against many viral strains, including strains resistant to Tamiflu® and XOFLUZA® Positive data from 3 animal models commonly used to assess bird - flu confirm drug activity One dose of TXM showed favorable Phase 1 PK with plasma blood levels > EC 90 for ~3 weeks with good safety Single dose therapy suitable for bird flu or seasonal flu treatment or prophylaxis; strong candidate for inclusion in strategic stockpile for pandemic preparedness Filing of IND to enable advancement of BARDA discussions on the drug stock piling initiative for pandemic preparedness Potent across many SARS - CoV2 viral strains, with superior in vitro suppression of COVID versus benchmarks and emerging drug candidates Ritonavir - independent regimen mitigates patient exclusion due to drug - drug interactions, providing access to protease inhibitor therapy for patients who cannot take PAXLOVID® Single, daily dose showed favorable Phase 1 PK with good safety supporting a proposed 10 - day dose regimen The 10 - day dosing regimen (compared to 5 - day PAXLOVID® ) has the potential to prevent both COVID - rebound and Long COVID H5N1 Bird Flu – Tivoxavir marboxil COVID: Potential b est - in - class agent to replace existing therapies, and prevent clinical rebound, as well as Long COVID.

Expecting increased demand for COVID therapy as vaccine effectiveness/use declines Bird Flu: In short term, US strategic stockpile approx. 300,000,000 - 600,000,000 dose s Seasonal Flu: Potential best in class agent to replace existing therapies addressing emerging resistant strains Cash , cash equivalents and short - term investments as of September 30, 2025, $6.4M 1 Cash position to support planned operations into Q1 2026 Selected Financial Information Milestone Program • Phase 2 studies initiated in Q4 2025, topline data by YE 2025 • Increase focus on preventing clinical rebound and mitigating risk for Long COVID Ratutrelvir • Filing of IND to enable advancement of BARDA discussions on the drug stock piling initiative for pandemic preparedness Tivoxavir Marboxil Finance Dashboard Source: 1. TRAW data on file, Existing therapies include PAXLOVID is a registered trademark of Pfizer, Inc., and LAGREVRIO is tr ademark of Merck Sharpe & Dohme Corp.

Financial information as of TRAW 10Q filed November 13, 2025 Market Potential 1 7 Strong Team with Track Record of Executing and Creating Value Robert R. Redfield, MD Chief Medical Officer C. David Pauza, PhD Chief Science Officer Charles Parker Chief Financial Officer Board of Directors Nikolay Savchuk , PhD Chief Operating Officer, Director Chairman Jack E. Stover CEO, Director Iain D. Dukes, MA D Phil COO, Director Nikolay Savchuk , PhD Special Advisor, Director Werner Cautreels , PhD Director Trafford Clarke Director John Leaman, MD Director Teresa Shoemaker 8 Iain D. Dukes, MA, D Phil CEO, Director 9 COVID - 19 Ratutrelvir (TRX01) Investigational Main protease ( Mpro ) inhibitor Ritonavir - independent,10 - day, once - daily dosing regimen


COVID Continues to be a Critical Threat to Human Health Risk of viral rebound (>20% of PAXLOVID treated cases) and Long COVID (10 - 20% of cases) 10 Notes: PAXLOVID® is a registered trademark of Pfizer, Inc. COVID estimates: https://www.cdc.gov/covid/php/surveillance/burden - estimates.html . Long COVID: Carly Herbert et al. (2025): https://doi.org/10.1093/cid/ciae539 . COVID rebound: https:// www.acpjournals.org / doi /10.7326/M23 - 1756 Long COVID Associates with Slow Viral Clearance and Viral Rebound During Acute SARS - CoV - 2 Infection 11 Symptoms associated with the risk of Long COVID Herbert, C., et al., Poster 0935, 2025 Conference on Retroviruses and Opportunistic Infection (CROI)


Why Improving COVID Therapy Matters Significant cause of human disease and mortality in vulnerable populations 1 12 Increasing clinical and economic burden of Long COVID Many people seeking COVID19 therapy are ineligible for PAXLOVID® due to drug interactions of the boosting agent ritonavir. Estimated that ~20% of persons seeking treatment are denied because they are using common medications that are incompatible with ritonavir Declining vaccine use, regulatory hurdles slowing vaccine approval, and poor match of vaccine with current variants increases virus spread and potential for serious disease Approved PAXLOVID® label limited to 5 - days, which may be insufficient to prevent clinical rebound and Long COVID Sources and Notes: 1. Traws information, 2. Carly Herbert et al. (2025) Clinical Infectious Diseases. https://doi.org/10.1093/cid/ciae539 .

PAXLOVID® is a registered trademark of Pfizer, Inc COVID is now established as a long - term public health challenge with an increasing burden of people suffering from Post - Acute Sequelae of COVID (PASC, aka Long COVID) 2 Ratutrelvir Value Proposition for COVID/Long COVID 13 Differentiating features of ratutrelvir , a potent oral antiviral agent Dose at RP2D maintains favorable PK, with plasma drug levels at > 4 - times the EC 9 0, and we predict that drug concentrations will be higher in lung compared to blood plasma Broadly more active against original COVID strains, variants of concern, and emerging drug - resistant strains compared to nirmatrelvir, ensitrelvir , and ibuzatrelvir A Phase 1 clinical study with 10 continuous days of ratutrelvir dosing demonstrated excellent safety/tolerability, and maintained drug levels above the predicted therapeutic range without the use of ritonavir Good overall tolerability enables increased duration and intensity of treatment, which may reduce the rate of clinical rebound and mitigate the risk of Long COVID Good overall safety profile allows once daily, single tablet dosing for 10 days to prevent rebound, compared to PAXLOVID® with multiple tablets taken twice a day for 5 days where rebound risk was higher than among untreated patients Favorable profile could enable accessibility to a larger number of vulnerable individuals Notes: TRAW data on file. Nirmatrelvir is the active agent in PAXLOVID® (a registered trademark of Pfizer, Inc); ibuzatrelvir is being developed by Pfizer, Inc.

Ratutrelvir is Consistently More Potent than Competitor Agents for Suppressing COVID virus growth in vitro 14 Superior to nirmatrelvir or ibuzatrelvir , across multiple virus strains Notes: Data represents a comparison of literature data v. Traws ’ in vitro assay results. EC50 – effective concentration for 50% inhibition of virus replication in vitro; EC90 – effective concentration for 90% inhibition of virus replication in vitro, COVID = SARS - CoV - 2. Nirmatrelvir is the active ingredi ent in PAXLOVID® a registered trademark of Pfizer, Inc. ibuzatrelvir is being developed by Pfizer, Inc. Traws data on file. EC50 values for Mpro inhibitors against multiple COVID strains (results in nM) Representative EC50 values for Mpro inhibitors and the number of strains tested (results in nM)
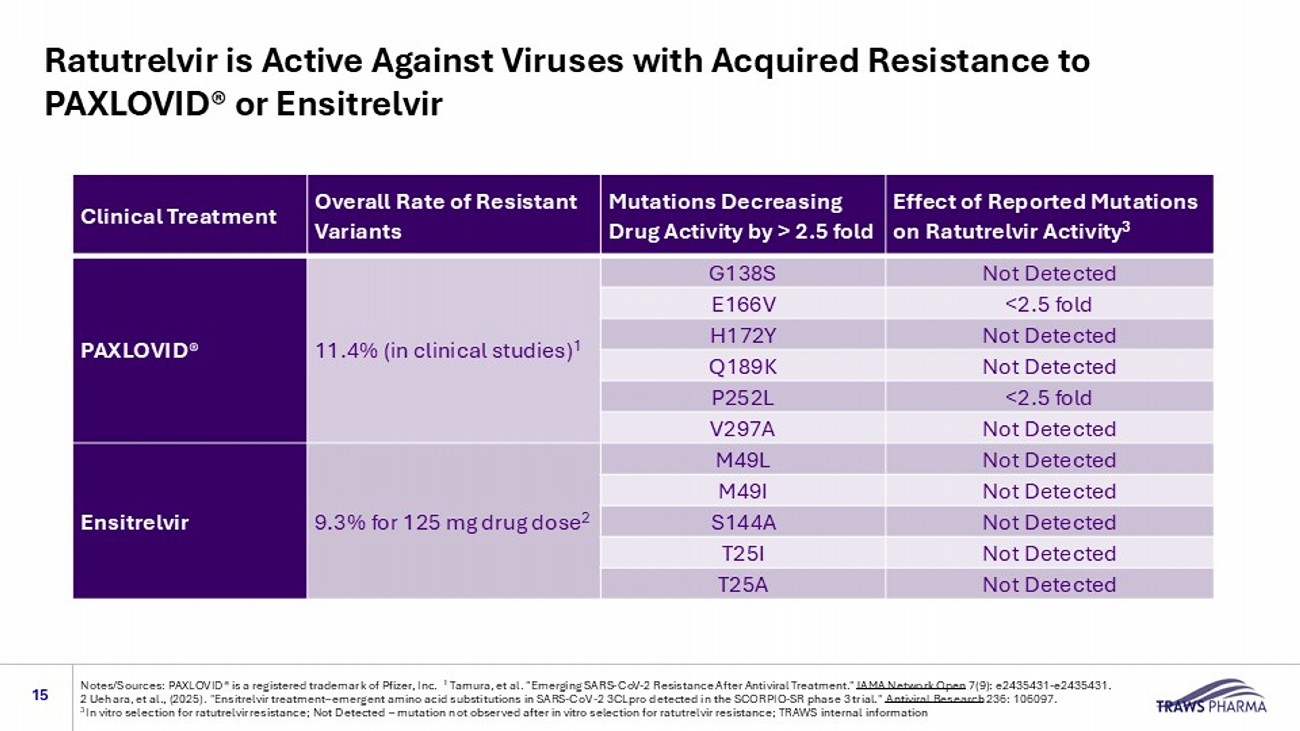
Ratutrelvir is Active Against Viruses with Acquired Resistance to PAXLOVID® or Ensitrelvir 15 Notes/Sources: PAXLOVID® is a registered trademark of Pfizer, Inc. 1 Tamura, et al. "Emerging SARS - CoV - 2 Resistance After Antiviral Treatment." JAMA Network Open 7(9): e2435431 - e2435431. 2 Uehara, et al., (2025). " Ensitrelvir treatment – emergent amino acid substitutions in SARS - CoV - 2 3CLpro detected in the SCORPIO - SR phase 3 trial." Antiviral Research 236: 106097.
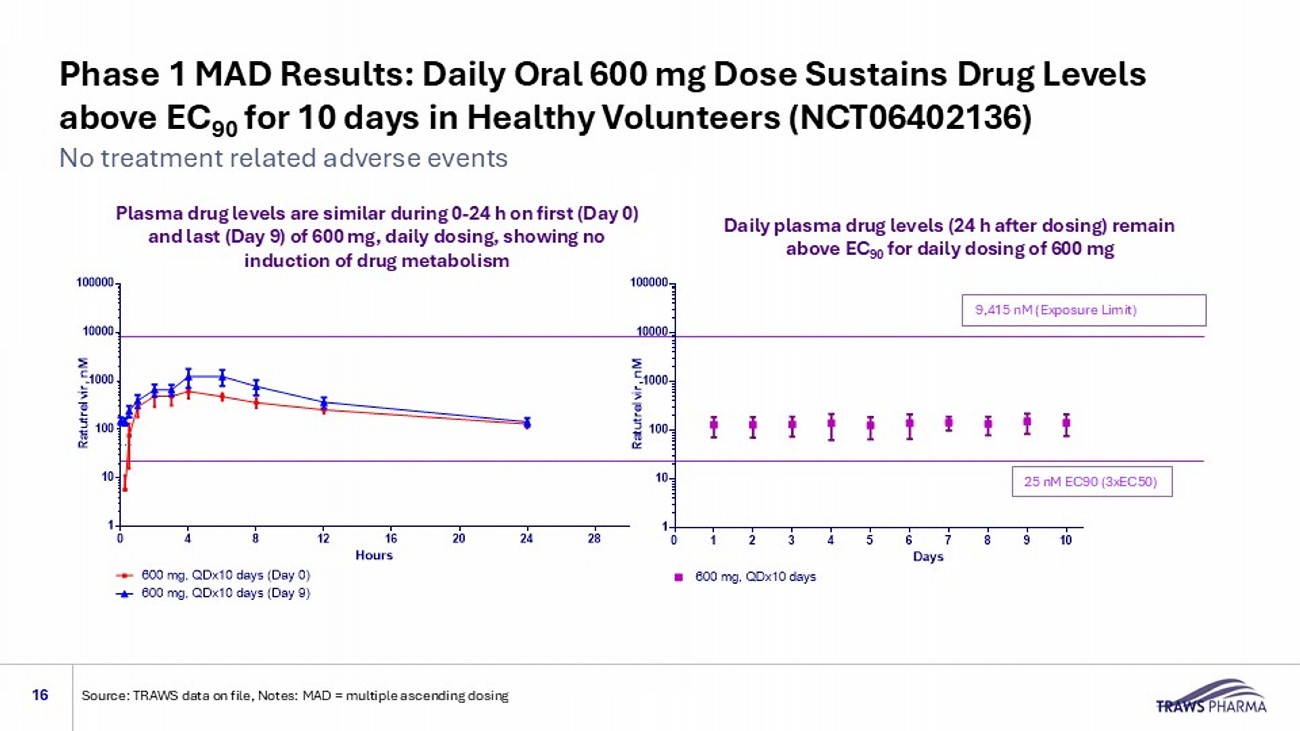
3 In vitro selection for ratutrelvir resistance; Not Detected – mutation not observed after in vitro selection for ratutrelvir resistance; TRAWS internal information Effect of Reported Mutations on Ratutrelvir Activity 3 Mutations Decreasing Drug Activity by > 2.5 fold Overall Rate of Resistant Variants Clinical Treatment Not Detected G138S 11.4% (in clinical studies) 1 PAXLOVID® <2.5 fold E166V Not Detected H172Y Not Detected Q189K <2.5 fold P252L Not Detected V297A Not Detected M49L 9.3% for 125 mg drug dose 2 Ensitrelvir Not Detected M49I Not Detected S144A Not Detected T25I Not Detected T25A Phase 1 MAD Results: Daily Oral 600 mg Dose Sustains Drug Levels above EC 90 for 10 days in Healthy Volunteers (NCT06402136) 16 No treatment related adverse events Source: TRAWS data on file, Notes: MAD = multiple ascending dosing 9,415 nM (Exposure Limit) 25 nM EC90 (3xEC50) Multiple Ascending Doses in human trough concentrations 0 1 2 3 4 5 6 7 8 9 10 11 12 13 1 10 100 1000 10000 100000 Days R a t u t r e l v i r , n M 48 nM 600 mg, QDx10 days Multiple Ascending Doses in human 0 4 8 12 16 20 24 28 1 10 100 1000 10000 100000 Hours R a t u t r e l v i r , n M 48 nM 600 mg, QDx10 days (Day 0) 600 mg, QDx10 days (Day 9) Plasma drug levels are similar during 0 - 24 h on first (Day 0) and last (Day 9) of 600 mg, daily dosing, showing no induction of drug metabolism Daily plasma drug levels (24 h after dosing) remain above EC 90 for daily dosing of 600 mg Consistent High Plasma Drug Levels Could Allow for Protection Against New Variants 17 No treatment related adverse events Source: TRAWS data on file, Notes: MAD = multiple ascending dosing study 9,415 nM (Exposure Limit) 25 nM EC90 (3xEC50) Multiple Ascending Doses in human 0 1 2 3 4 5 6 7 8 9 10 11 12 13 1 10 100 1000 10000 100000 Days R a t u t r e l v i r , n M 48 nM 600 mg, QDx10 days 110 nM (13xEC50) Plasma drug l evels (24 h C trough values for Days 1 - 9) Above EC 90 for virus suppression and within the target PK window (>25 nM and below the exposure limit): based on more intensive data collection after first and last dose Phase 1 Multiple Ascending Dose Study


Phase 2a COVID19 Study Design 18 Enroll 90 COVID19 - positive subjects 18 - 65 years of age Three groups (n=30 each): • Daily 600 mg of ratutrelvir for 10 days in PAXLOVID® eligible patients • Paxlovid for 5 days • Daily 600 mg of ratutrelvir for 10 days in patients ineligible for PAXLOVID® Evaluate time to decrease duration and intensity of symptoms, pharmacokinetics, changes in virus burden, frequency and timing of clinical rebound Randomized, double - blind, controlled, international non - inferiority study to assess safety, tolerability, pharmacokinetics and activity of ratutrelvir , oral, once - daily 600 mg for 10 days in patients eligible or ineligible for PAXLOVID® therapy compared to PAXLOVID® for 5 days Study Design Subjects Overall objectives Dosing Notes: PAXLOVID® is a registered trademark of Pfizer, Inc.

Phase 2a COVID19 Study Design 19 Outcome Measures Include • Symptom relief • Hospitalization • Death • PK • Virus burden • Frequency of clinical rebound • Timing of clinical rebound 20 Phase 2 Studies Underway Phase 1 studies demonstrated excellent overall safety and tolerability and defined a Phase 2 target dose Phase 2 non - inferiority study underway evaluating ratutrelvir compared to PAXLOVID® in newly diagnosed COVID patients.

The program also includes a separate single arm study in patients ineligible for PAXLOVID®. Topline data from both studies expected by YE 2025 Note: TRAW data on file. PAXLOVID® is a registered trademark of Pfizer, Inc.

Bird Flu Tivoxavir marboxil (TXM, TRX100) A single - dose investigational, CAP - dependent endonuclease inhibitor for treatment or prevention of H5N1 bird flu 21 Influenza is a Critical Threat To Human Health Today High pandemic potential with no currently approved treatments 22 Major and increasing agricultural impact of bird flu There are n o approved bird flu therapies 4 5 High historical human mortality from bird flu, with human pandemic potential 1 L imited cross protection provided by current seasonal flu vaccines Inadequate potency of current antiviral agents for bird flu Source: See https:// www.who.int /publications/m/item/cumulative - number - of - confirmed - human - cases - for - avian - influenza - a(h5n1) - reported - to - who -- 2003 - 2025 -- 19 - march - 2025 Tivoxavir Marboxil (TRX100/TXM): Value Proposition for Bird Flu 23 Differentiating features of TXM, a potent oral antiviral drug candidate A single dose reduced disease and prevented death in 3 preclinical animal models used commonly in flu drug development, with large reductions in lun g virus levels and lung pathology A singl e dose maintained plasma levels above the EC 90 for approximately 3 weeks in Phase 1 human studies 4 5 Broadly effective against bird flu strains including “Texas dairy worker strain”, demonstrated through in vivo studies Large safety window enables use of higher TXM doses, if needed to treat future emergent resistant strains


Tivoxavir Potently Inhibits Highly Pathogenic Avian Influenza Viruses 24 Tivoxavir (the active metabolite of TRX100) inhibits replication of HPAI H5N1 avian influenza viruses and reference (H3N2) influenza viruses 2 Note: Based on avian influenza virus circulating in 2022 - 2023, 2. Results from the Influenza replication inhibition neuraminidas e - based assay, provided by the US CDC (Atlanta) Tivoxavir (TRX101) Strain Designation EC 50 (nM) H5N1 viruses 0.40 A/ chicken /Idaho/22 - 011347 - 004/2022 0.52 A/ Canada_ goose /Wyoming/22 - 011671 - 001/2022 0.43 A/ Red_Shoulder_ Hawk /Minnesota/22 - 012000 - 004/2022 0.34 A/ dolphin /Florida/22 - 025319 - 002/2022 0.41 A/ black_ vulture /Florida/22 - 012333 - 001/2022 - c30 Reference A(H3N2) Viruses 1.21 A/Louisiana/50/2017 ( Wt ) 10.28 A/Louisiana/49/2017 (PA - I38M)

Human H5N1 Virus is Resistant to Available Antiviral Drugs Multiple mutations related to increased transmission and growth in mammalian cells and natural resistance to oseltamivir (Tamiflu®) and baloxavir (XOFLUZA® ) 25 Nine Mutations in A/Texas/37/2024 were Not Present in the Bovine Isolate Oseltamivir and Baloxavir are Less Potent Against A/Texas/37/2024 Compared to Other H1N1 and H5N1 Virus Isolates Sources: Adapted from Mostafa, et al (2025) DOI: 10.1080/22221751.2024.2447614 . Tamiflu® is a trademark registered of Roche. Xofluza ® is a registered trademark of Genentech Pharmaceuticals, an independent subsidiary of Roche Mouse antiviral study using bird flu (H5N1) transmitted from cattle to a dairy worker 26 Notes: Challenge dose via intranasal delivery.
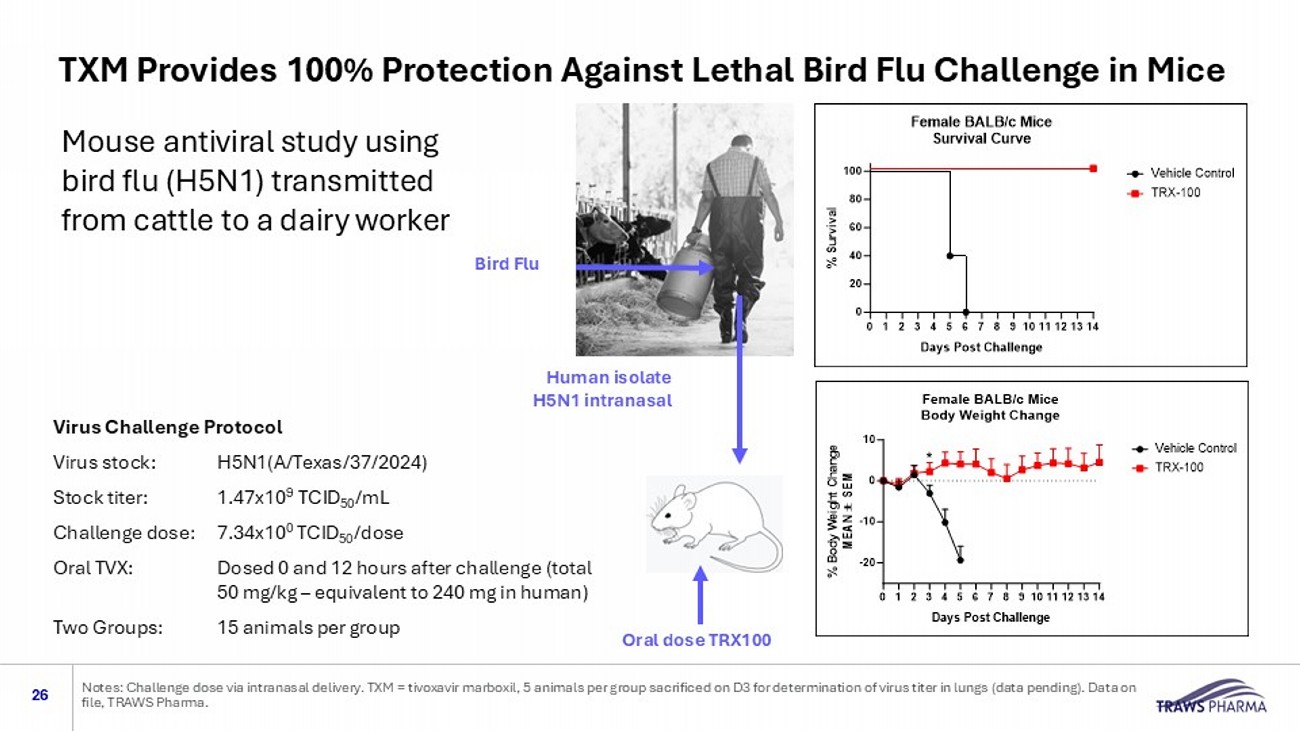
TXM = tivoxavir marboxil , 5 animals per group sacrificed on D3 for determination of virus titer in lungs (data pending). Data on file, TRAWS Pharma. Virus Challenge Protocol Virus stock: H5N1(A/Texas/37/2024) Stock titer: 1.47x10 9 TCID 50 /mL Challenge dose: 7.34x10 0 TCID 50 /dose Oral TVX: Dosed 0 and 12 hours after challenge (total 50 mg/kg – equivalent to 240 mg in human) Two Groups: 15 animals per group Bird Flu Human isolate H5N1 intranasal Oral dose TRX100 TXM Provides 100% Protection Against Lethal Bird Flu Challenge in Mice 27 1.

Virus challenge protocol per slide 14. TCID = tissue culture infectious dose Virus Titer Group Animal 7x10 8 Vehicle 31 1.3x10 7 32 1.3x10 8 33 8.2x10 8 34 1.2x10 7 35 Virus Titer Group Animal BLLQ TXM Day 1 dose of 50 mg/kg 46 BLLQ 47 BLLQ 48 BLLQ 49 BLLQ 50 Virus titer – TCID 50 /gram of tissue; BLLQ – below lower limit of quantitation (2.2x10 3 TCID 50 /gram) Day 3 Lung Virus Titers (TCID 50 ) TXM Achieved Complete Viral Clearance in the Lung Based on murine viral challenge studies 1 Virus Challenge Study in Ferrets: Key Data to Support Clinical and Regulatory Strategy for TXM in Bird Flu 28 Note: TRAW data on file.

intranasal challenge/inoculation. Virus challenge stock: A/Texas/37/2024 (H5N1); 4 animals per group sa crificed at three days after viral inoculation for lung virus burden determinations Oral treatment: 8 hours after virus inoculation Doses: 120 mg human equivalent 240 mg in human equivalent Two Groups: 10 Animals per group (5F/5M), 13 - 15 weeks of age Treated and control Endpoints: Survival, changes in body weight, virus burden in lung and nasal tissues Topline Data: Substantial improvements in disease progression observed after a single dose of TXM • Lethal challenge showed that TXM increased the proportion of surviving animals • Lowered the viral burden in lungs and nasal tissues • Delayed and reduced body weight loss Promising topline data in ferret model using virus isolated from a dairy worker. Viral inoculum: 1X10 3 TCID 50 of A/Texas/37/2024 H5N1 via intranasal instillation Ferret model Vehicle Control Equivalent to 240 mg human dose Equivalent to 120 mg human dose TXM Dose - dependently Increased Survival in Ferrets Infected with Bird Flu 29 Note: TRAW data on file.
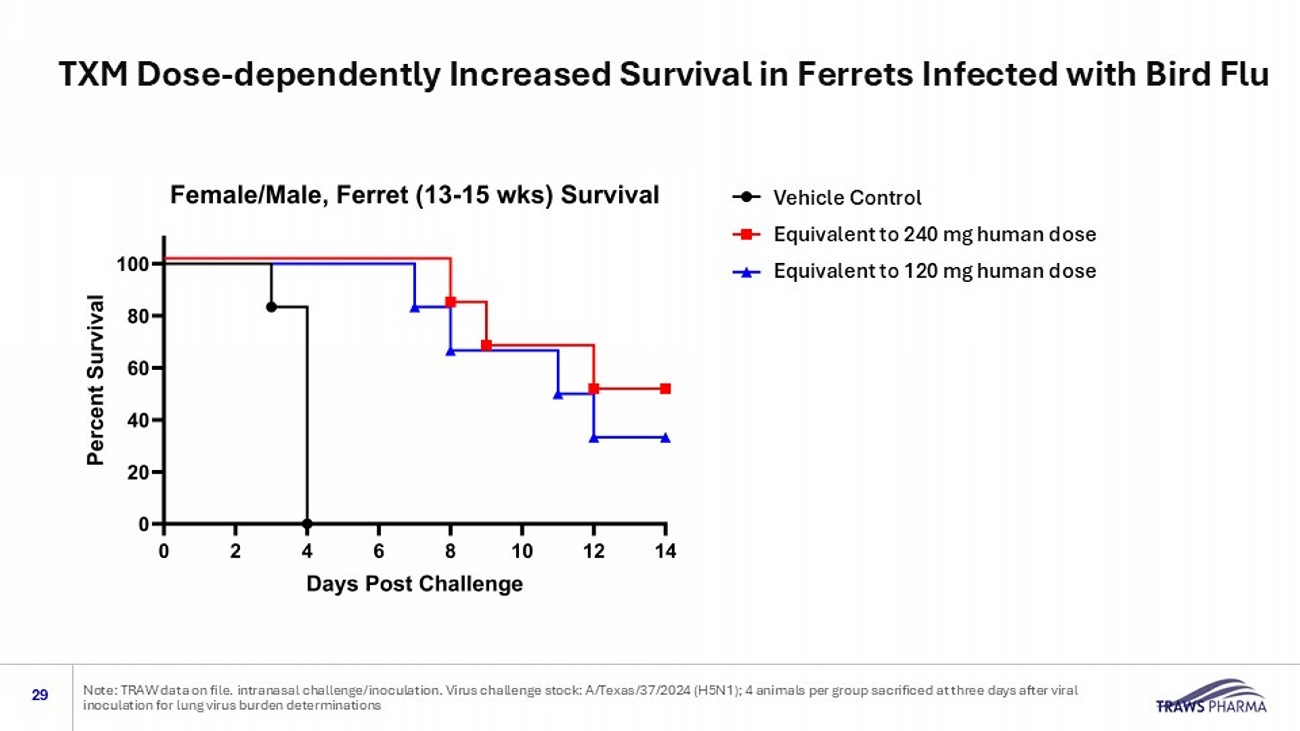
intranasal challenge/inoculation. Virus challenge stock: A/Texas/37/2024 (H5N1); 4 animals per group sa crificed at three days after viral inoculation for lung virus burden determinations Vehicle Control High dose (equivalent to 240 mg human dose Low dose (equivalent to 120 mg human dose) Vehicle Control Equivalent to 240 mg human dose Equivalent to 120 mg human dose TXM Doses Prevented Rapid Body Weight Loss in Ferrets Infected with Bird Flu 30 Note: TRAW data on file.

intranasal challenge/inoculation. Virus challenge stock: A/Texas/37/2024 (H5N1); 4 animals per group sa crificed at three days after viral inoculation for lung virus burden determinations Vehicle Control Equivalent to 240 mg human dose Equivalent to 120 mg human dose Single Oral Dose of TXM Reduced Lung Virus Burden in Ferrets 31 Note: TRAW data on file.

intranasal challenge/inoculation. Virus challenge stock: A/Texas/37/2024 (H5N1); 4 animals per group sa crificed at three days after viral inoculation for lung virus burden determinations.

TCID = tissue culture infectious doses Control – no drug 0% survival Equivalent to 240 mg human dose 50% survival Equivalent to 120 mg human dose 30% survival Viral RNA copies per mL Single Oral Dose of TXM Increased Survival and Reduced Lung Virus Burden in Bird Flu - Infected Ferrets 32 Note: TRAW data on file. intranasal challenge/inoculation. Virus challenge stock: A/Texas/37/2024 (H5N1); 4 animals per group sa crificed at three days after viral inoculation for lung virus burden determinations TXM Treatment Reduced Lung Pathology in Ferrets Infected with Bird Flu Virus: Dose - Dependent Impact on Cellular Infiltration of the Lungs 33 Note: TRAW data on file.

intranasal infection with A/Texas/37/2024 (H5N1); lung pathology assessed three days after infectio n. Note: 1 = TXM dose of 10.6 mg; 2 = TSM dose of 21.2 mg.

HE = human equivalent Day 3: Control Day 3 2 : Human Equivalent 240 mg Day 3 1 : Human Equivalent 120 mg Cellular infiltration/inflammation: basophilic, dark - staining areas (solid arrows ) • Control : Extensive cellular infiltration of lung • TXM 10.6 mg (HE120 mg): Areas of cellular infiltration greater than the 21.2 mg dose animals, far less than untreated controls • TXM 21.2 mg (HE 240 mg): Few areas of cellular infiltration, lung mostly normal with open alveoli Virus Challenge Study in Non - Human Primate: Key Data to Support Clinical and Regulatory Strategy for TXM in Bird Flu 34 Oral treatment 12 hours after non - lethal virus inoculation Dose: One dose (480 mg human equivalent dose) Two Groups: 5 animals per group (treated and control) Endpoints: Virus burden in lung and nasal tissues, body weight Non - human primate model Topline Data: Substantial changes in disease progression observed after a single dose of TXM • Non - lethal challenge showed that tivoxavir marboxil significantly lowered lung virus burden • No virus - induced weight loss Promising topline data in non - human primate (NHP) model using virus isolated from a dairy worker. Viral inoculum:1X10 6 TCID 50 of A/Texas/37/2024 H5N1 with half delivered via intranasal instillation and half delivered to the oropharynx Note: TRAW data on file. Virus challenge stock: A/Texas/37/2024 (H5N1); 5 animals per group Post - Exposure TXM Treatment Prevented Body Weight Loss in A/H5N1 - Infected Non - Human Primates (p < 0.004) 35 Note: TRAW data on file.

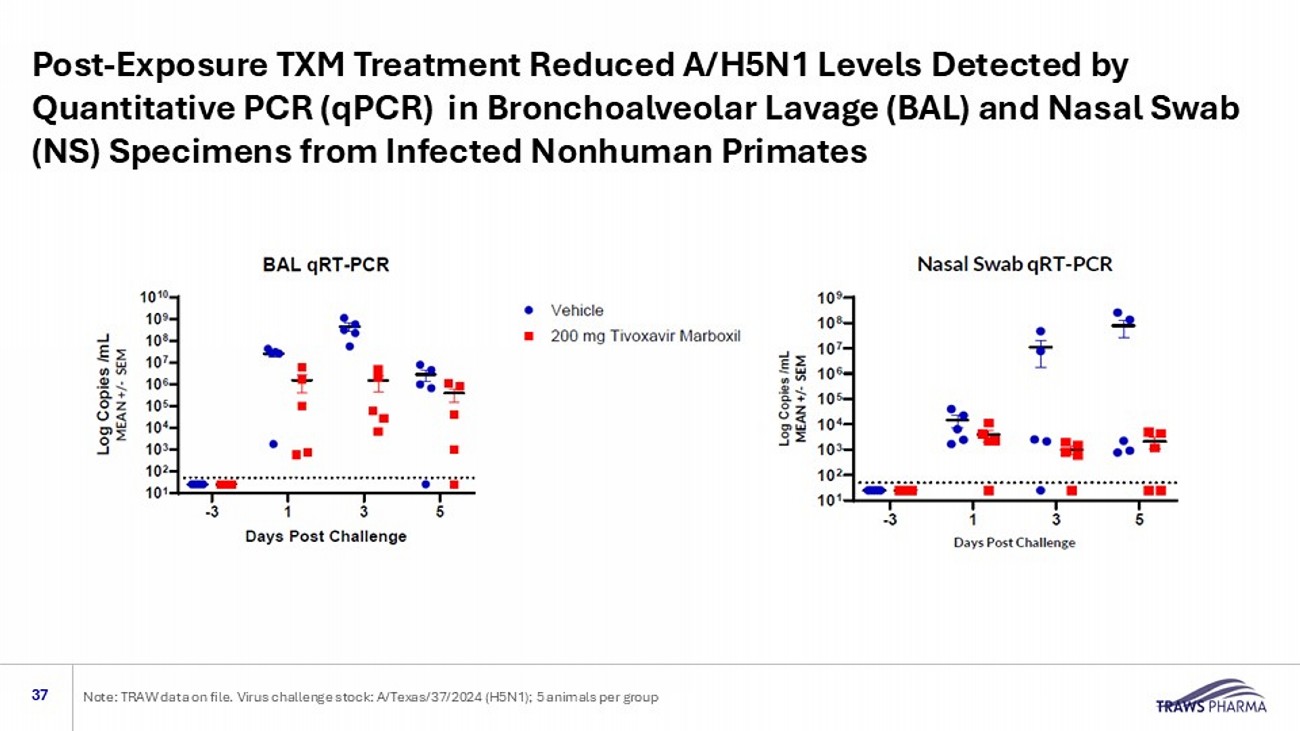
Virus challenge stock: A/Texas/37/2024 (H5N1); 5 animals per group Post - Exposure TXM Treatment Reduced A/H5N1 Titer in Bronchoalveolar Lavage (BAL) and Nasal Swab (NS) Specimens from Infected Nonhuman Primates 36 Note: TRAW data on file.

Virus challenge stock: A/Texas/37/2024 (H5N1); 5 animals per group Post - Exposure TXM Treatment Reduced A/H5N1 Levels Detected by Quantitative PCR (qPCR) in Bronchoalveolar Lavage (BAL) and Nasal Swab (NS) Specimens from Infected Nonhuman Primates 37 Note: TRAW data on file.
Virus challenge stock: A/Texas/37/2024 (H5N1); 5 animals per group Control Animals – no drug treatment • Results for individual animals are depicted • Results from bronchoalveolar lavage samples collected on Study Day 3 and analyzed by TCID 50 assay • < 10e3 – below the lower limit of quantitation Animals treated with equivalent of human dose 480 mg Virus Infectivity per mL Single Oral Dose of TXM Blocks Bird Flu Replication in the Lungs of NHPs 38 Note: TRAW data on file. Virus challenge stock: A/Texas/37/2024 (H5N1); 5 animals per group Tivoxavir Marboxil Treatment Reduced Lung Pathology in Nonhuman Primates Infected with Bird Flu Virus 39 Note: TRAW data on file.
Virus challenge stock: A/Texas/37/2024 (H5N1); 5 animals per group: Based on Day 10 necropsy. X = ma gni fication. 1 = TXM dose at 480 mg human equivalent t dose Cellular infiltration and inflammation: basophilic, dark - staining areas (solid arrows) Untreated TXM - treated 1 Control (untreated) : extensive areas of basophilic staining indicating cellular infiltration and inflammation TXM (Treated) : no areas of cellular infiltration. Lung is normal in appearance with open alveoli 1.25x 10x Phase 1 Study Design for TXM 40 Notes: TRAW data on file.
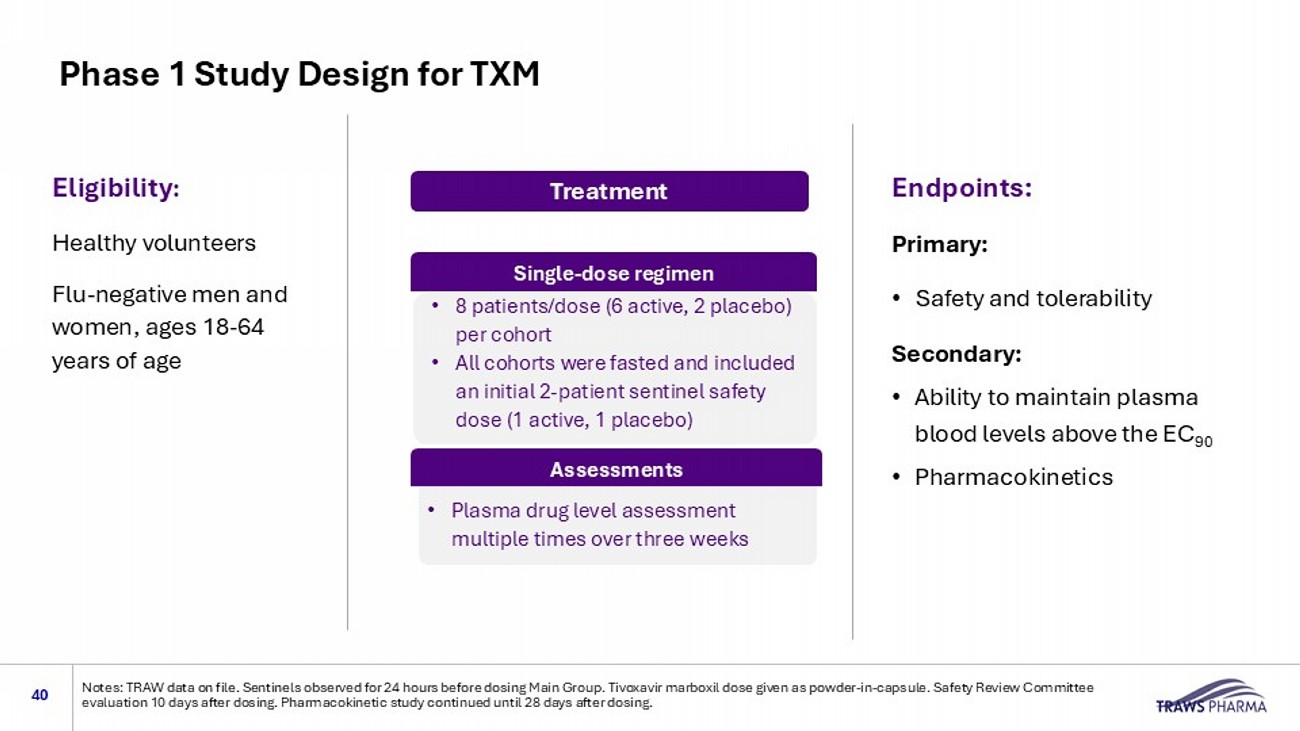
Sentinels observed for 24 hours before dosing Main Group. Tivoxavir marboxil dose given as powder - in - capsule. Safety Review Committee evaluation 10 days after dosing. Pharmacokinetic study continued until 28 days after dosing. Eligibility : Healthy volunteers Flu - negative men and women, ages 18 - 64 years of age Endpoints: Primary: • Safety and tolerability Secondary: • Ability to maintain plasma blood levels above the EC 90 • Pharmacokinetics Treatment 80 mg,120 mg, 240 mg, 480 mg doses • 8 patients/dose (6 active, 2 placebo) per cohort • All cohorts were fasted and included an initial 2 - patient sentinel safety dose (1 active, 1 placebo) Single - dose regimen Assessments • Plasma drug level assessment multiple times over three weeks Phase 1 Data Show that TXM Maintains Plasma Blood Levels > EC 90 for ~3 weeks (NCT06757738) 41 No treatment related adverse events Notes: Four doses evaluated (80, 120, 240, 480mg, all fasted).

Each group included a sentinel dose with 24h observation (1 ac tiv e, 1 placebo), following by the main group (5 active, 1 placebo). Tivoxavir marboxil dosed as powder - in - capsule. Safety Review Committee evaluation 10 days after dosing. Pharmacokinetic study continued until 28 days after dosing.. Data on file, TRAWS Pharma.

Tivoxavir in plasma (ng/mL) Plasma drug levels above EC 90 for ~3 weeks days after single 480 mg dose in healthy volunteers Time (hours) EC90 ~0.82 ng/mL 3 weeks Sources: 1 . b ird flu case fatality rate: WHO information : https://cdn.who.int/media/docs/default - source/wpro --- documents/emergency/surveillance/avian - influenza/ai_20250131.pdf . 2 . Traws information TXM Could Be a Fit for Drug Stock Piling for Pandemic Preparedness Filing of IND to enable advancement of BARDA discussions • Virus evolved to a more virulent strain (dairy worker, mild symptoms) • History of A/H5N1 virus strains with up to 50% case fatality rate 1 • Virus can be 100% fatal in 3 - 5 days in animals (mice, ferrets, NHP) 2 • Placebo - controlled trials would be unethical and not feasible • Preclinical studies used commonly accepted flu models (mice, ferrets and non - human primates) • Positive Phase 1 safety/dosing escalation data (healthy volunteers) includes dose levels that were protective in animal studies • Maintained plasma levels above the EC 90 for more than 3 weeks, with good overall safety Compelling Preclinical Animal Studies in Bird Flu and Seasonal Flu Exceptional Pathogenic Bird Flu Potential Excellent Human Safety and Pharmacokinetics 42 Phase 2a Seasonal/Pandemic Flu Study Design 43 Expected to enroll ~150 influenza - positive subjects 18 - 65 years of age Expected to incorporate five dosing groups (n=30 each): • TXM dose level 1 (seasonal) • TXM dose level 2 (seasonal) • TXM dose level 2 (H5N1) • Baloxavir marboxil (seasonal) • Oseltamivir (H5N1) Evaluate two dose levels of TXM versus baloxavir marboxil in seasonal influenza and TXM versus oseltamivir in H5N1 influenza; assess time to symptom relief, safety, virology, with follow - up out to 28 days Randomized, double - blind, controlled, international non - inferiority study to assess safety, tolerability, pharmacokinetics and activity of a one - time dose of TXM in subjects with acute, uncomplicated, community - acquired influenza compared to standard of care, or TXM for subjects with confirmed H5N1 influenza Study Design Subjects Overall objectives Dosing


Phase 2a Seasonal Flu Study Design: Including H5N1 or H5N2 infections 44

Targeting Critical Viral Threats to Human Health 45 Potential first - in - class bird flu program , with an initial goal to build out the US strategic stockpile (estimated 300 million to 600 million doses 2 ) Potential best - in - class COVID program to replace existing therapies (>$6B revenue, 2024), prevent clinical rebound and explore treatment of Long COVID 1 5 F ocusing on small molecule antiviral drugs active against respiratory viruses with pandemic potential Experienced team, supported by recognized institutional investors, including OrbiMed Progressing business development exit strategies for legacy oncology assets (rigosertib and narazaciclib) Source: 1. Pfizer 2024 financial results here, p. 24 , Merck 2024 financial results here, p5 = $5.7B + $964M = >$6B 2. TRAW internal information.

Thank You! www.trawspharma.com