UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, DC 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of
the
Securities Exchange Act of 1934
Date of report (Date of earliest event reported): October 22, 2025
ARMATA PHARMACEUTICALS, INC.
(Exact name of Registrant as specified in its charter)
| Washington | 001-37544 | 91-1549568 | ||
| (State or other jurisdiction of incorporation or organization) |
(Commission File Number) | (IRS Employer Identification No.) |
| 5005 McConnell Avenue Los Angeles, California |
90066 | ||
| (Address of principal executive offices) | (Zip Code) |
(310) 655-2928
(Registrant’s Telephone number)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the Registrant under any of the following provisions (see General Instruction A.2. below):
| ¨ | Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ¨ | Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ¨ | Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ¨ | Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company ¨
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Securities registered pursuant to Section 12(b) of the Act:
| Title of Each Class | Trading Symbol(s) | Name of Each Exchange on Which Registered | ||
| Common Stock | ARMP | NYSE American |
| Item 7.01 | Regulation FD Disclosure. |
On October 22, 2025, Armata Pharmaceuticals, Inc. (the “Company”) issued a press release announcing the presentation of positive results from its recently completed Phase 2a diSArm study of AP-SA02 as a potential treatment for complicated Staphylococcus aureus bacteremia at IDWeek 2025TM. The full text of the press release issued in connection with this announcement is furnished as Exhibit 99.1 to this Current Report on Form 8-K.
The Company posted to its website a copy of the late-breaking oral presentation delivered on October 22, 2025 by Dr. Loren G. Miller, M.D., M.P.H., at IDWeek 2025TM (the “IDWeek Presentation”). The IDWeek Presentation is furnished as Exhibit 99.2 to this Current Report on Form 8-K.
The information contained in the IDWeek Presentation is summary information that is intended to be considered in the context of the Company’s Securities and Exchange Commission (“SEC”) filings and other public announcements that the Company may make, by press release or otherwise, from time to time. The Company undertakes no duty or obligation to publicly update or revise the information contained in this report, except as may be required by the federal securities laws, although it may do so from time to time as its management believes is warranted. Any such updating may be made through the filing of other reports or documents with the SEC, through press releases or through other public disclosure. In addition, the Company disclaims any inferences regarding the materiality of such information that may arise as a result of it furnishing such information under Item 7.01 of this Current Report on Form 8-K.
The information in this Item 7.01 and the attached Exhibits 99.1 and 99.2 is being furnished and shall not be deemed “filed” for the purposes of Section 18 of the Securities Exchange Act of 1934, as amended, or otherwise subject to the liabilities of that Section. The information in this Item 7.01 and the attached Exhibits 99.1 and 99.2 shall not be incorporated by reference into any registration statement or other document pursuant to the Securities Act of 1933, as amended.
| Item 9.01 | Financial Statements and Exhibits. |
(d) Exhibits.
| Exhibit No. |
Description | |
| 99.1 | Press Release, dated October 22, 2025. | |
| 99.2 | IDWeek Presentation, dated October 22, 2025. | |
| 104 | Cover Page Interactive Data File (embedded within Inline XBRL document). |
-
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| Date: October 22, 2025 | Armata Pharmaceuticals, Inc. | |
| By: | /s/ Deborah L. Birx | |
| Name: | Deborah L. Birx, M.D. | |
| Title: | Chief Executive Officer (Principal Executive Officer) |
|
-
Exhibit 99.1

Armata Pharmaceuticals Highlights Positive Results
from Phase 2a diSArm Study of its
Staphylococcus aureus Bacteriophage Cocktail, AP-SA02, in Late-Breaking Oral Presentation at
IDWeek 2025TM
LOS ANGELES, Calif., October 22, 2025 -- Armata Pharmaceuticals, Inc. (NYSE American: ARMP) (“Armata” or the “Company”), a clinical-stage biotechnology company focused on the development of high-purity, pathogen-specific bacteriophage therapeutics for the treatment of antibiotic-resistant and difficult-to-treat bacterial infections, today highlighted positive results from its recently completed Phase 2a diSArm study of AP-SA02 as a potential treatment for complicated Staphylococcus aureus (“S. aureus”) bacteremia (“SAB”) in a late-breaking oral presentation at IDWeek 2025TM.
The abstract, titled, “A Phase 2a Randomized, Double-Blind, Controlled Trial of the Efficacy and Safety of an Intravenous (IV) Bacteriophage Cocktail (AP-SA02) vs. Placebo in Combination with Best Available Antibiotic Therapy (BAT) in Patients with Complicated Staphylococcus aureus Bacteremia,” was accepted as a highly coveted late-breaking abstract for oral presentation, and was presented by Dr. Loren G. Miller, M.D., M.P.H., Professor of Medicine, David Geffen School of Medicine at UCLA, Chief, Division of Infectious Diseases at Harbor-UCLA Medical Center and the Lundquist Institute.
“The results of the diSArm study confirm, for the first time in a randomized clinical trial, the efficacy of intravenous phage therapy for S. aureus bacteremia, and we are very pleased to highlight these compelling data in an oral presentation at IDWeek,” stated Dr. Miller. “The results of this rigorously designed study provide strong rationale for advancement into a Phase 3 superiority study that, if successful, would support its use in clinical practice for Staphylococcus aureus bacteremia. High-purity, phage-based therapeutics like AP-SA02 have the potential to become the new standard of care for this common, extremely severe, and often deadly infection.”
“The positive results from the diSArm study represent another significant achievement for Armata as we aim to advance AP-SA02 into a pivotal trial,” stated Dr. Deborah Birx, Chief Executive Officer of Armata. “I would like to thank Dr. Miller and the other investigators who contributed to the efficient execution of the diSArm study, and I look forward to working with many of them on a proposed pivotal study next year. I would also like to thank Dr. Vance Fowler who served as the chair of the independent blinded adjudication committee that independently confirmed safety and efficacy findings throughout the Phase 2 trial. Finally, I would like to express my gratitude to the patients who participated in this important study, and acknowledge our partners at the U.S. Department of Defense, and our significant shareholder, Innoviva, who have each provided critical support to make this early breakthrough possible.”
Data highlights:
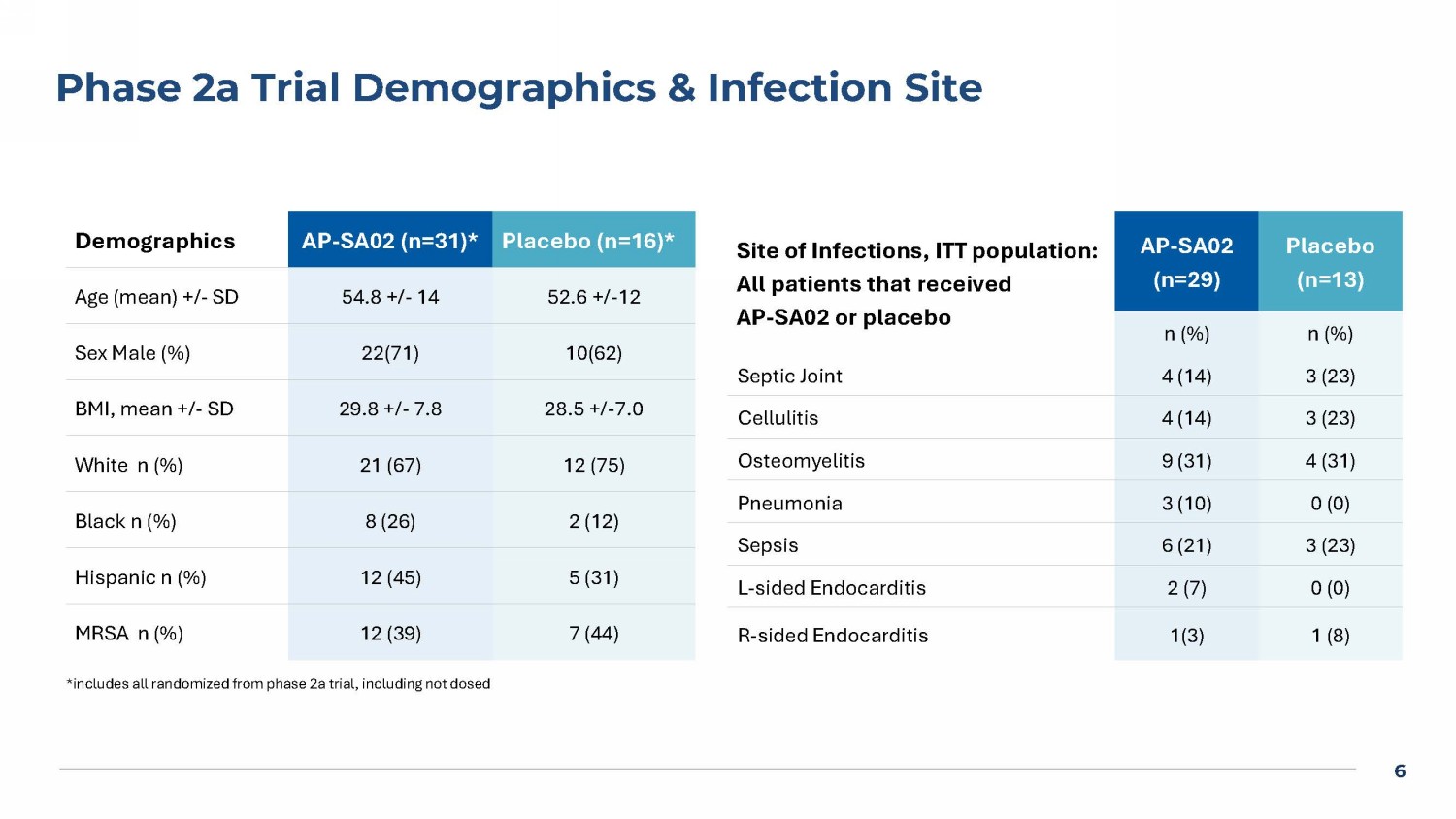
The Phase 2a study enrolled and dosed 42 patients, with 29 randomized to AP-SA02 in addition to BAT and 13 to placebo (BAT alone). Methicillin-resistant S. aureus (“MRSA”) was the causative pathogen in ~38% of both the AP-SA02 and placebo groups.
Clinical response was assessed in the intent-to-treat (ITT) population at Test of Cure (“TOC”) on day 12, one week post-BAT, and End of Study (“EOS”) four weeks after BAT completion. Safety analysis also included data from the Phase 1b portion of the trial (n=8).

Day 12 clinical response rates were higher in the AP-SA02 group — 88% (21/24) versus 58% (7/12) in the placebo group as assessed by blinded site investigators (“PI”) (p = 0.047), and 83% (20/24) in the AP-SA02 group versus 58% (7/12) in the placebo group as assessed by the blinded Adjudication Committee (“AC”).
Non-response/relapse rates were evaluated at the two later timepoints — one week post-BAT and EOS. No patients in the AP-SA02 group experienced non-response or relapse (0%) by either PI or AC assessment. In contrast, the placebo group showed 25% non-response/relapse at both timepoints reported by the PI (p = 0.017) and 22% non-response/relapse at one week post-BAT (p = 0.025) and 25% at EOS (p = 0.02) by the AC.
Patients treated with AP-SA02 showed trends toward rapid normalization of C-reactive protein, shorter time to negative blood culture, quicker time to resolution of signs and symptoms at the infection site, shorter intensive care unit and hospital utilization.
AP-SA02 was well-tolerated with no serious adverse events related to the study drug. Treatment-emergent adverse events occurred in 6% (2/35) and 0% (0/15) in the AP-SA02 and placebo groups, respectively: one patient with transient liver enzyme elevation and one patient with hypersensitivity that resolved with discontinuation of vancomycin.
New findings demonstrate that the defined and reproducible genomic variants present in AP-SA02 Drug Product may provide an immediate advantage, enabling rapid, strain-specific response to each patient’s S. aureus isolate. These characterized variants can expand from as little as 2% to dominance when infecting certain patient isolates in vitro, highlighting that these variants are favored for their enhanced ability to infect those strains and the importance of integrating this diversity into Armata’s phage cocktail from the outset. This inherent flexibility may be central to achieving optimal therapeutic efficacy.
Conclusions:
| · | AP-SA02, combined with BAT, had a higher and earlier cure rate compared to placebo in patients with complicated SAB at day 12 as assessed by both blinded site investigators and independent adjudicators. |
| · | No patients who received AP-SA02 demonstrated non-response or relapse at one week post-BAT or at EOS, as assessed by both blinded site investigators and the independent adjudication committee, compared with approximately 25% in the placebo group. |
| · | AP-SA02 appears safe with clinical efficacy against both MRSA and methicillin-sensitive S. aureus (“MSSA”) and trends toward earlier resolution and shorter hospitalization, with no evidence of relapse four weeks post-therapy. |
| · | Defined phage variants in AP-SA02 Drug Product ensure an intrinsic adaptive mechanism — a flexibility that may be key to achieving effective phage therapy from patient to patient. |
| · | These results strongly support advancement into a pivotal Phase 3 trial that Armata plans to initiate in 2026, subject to review and feedback from the U.S. Food and Drug Administration (the “FDA”). The Company is engaged with the FDA regarding a potential superiority trial design. |

About IDWeek 2025TM
IDWeek 2025TM is a joint annual meeting of the Infectious Diseases Society of America (IDSA), the Society for Healthcare Epidemiology of America (SHEA), the HIV Medicine Association (HIVMA), the Pediatric Infectious Diseases Society (PIDS) and the Society of Infectious Diseases Pharmacists (SIDP). With the theme “Advancing Science, Improving Care,” IDWeek features the latest science and bench-to-bedside approaches in prevention, diagnosis, treatment and epidemiology of infectious diseases, including HIV, across the lifespan. IDWeek 2025TM takes place October 19-22 in Atlanta, GA. For more information, visit www.idweek.org.
About AP-SA02 and diSArm Study
Armata is developing AP-SA02, a fixed multi-phage phage cocktail, for the treatment of complicated bacteremia caused by Staphylococcus aureus (S. aureus), including methicillin-sensitive S. aureus (MSSA) and methicillin-resistant S. aureus (MRSA) strains.
The diSArm study (NCT05184764) was a Phase 1b/2a, multicenter, randomized, double-blind, placebo-controlled, multiple ascending dose escalation study of the safety, tolerability, and efficacy of intravenous AP-SA02 in addition to best available antibiotic therapy (BAT) compared to BAT alone (placebo) for the treatment of adults with complicated S. aureus bacteremia. The results from the diSArm study are an important step forward in Armata’s effort to confirm the potent antimicrobial activity of phage therapy and the completion of the study represents a significant milestone in the development of AP-SA02, moving Armata one step closer to introducing an effective new treatment option to patients suffering from complicated S. aureus bacteremia.
The Phase 1b/2a clinical development of AP-SA02 was partially supported by a $26.2 million Department of Defense (DoD) award, received through the Medical Technology Enterprise Consortium (MTEC) and managed by the Naval Medical Research Command (NMRC) – Naval Advanced Medical Development (NAMD) with funding from the Defense Health Agency and Joint Warfighter Medical Research Program.
About Armata Pharmaceuticals, Inc.
Armata is a clinical-stage biotechnology company focused on the development of high-purity pathogen-specific bacteriophage therapeutics for the treatment of antibiotic-resistant and difficult-to-treat bacterial infections using its proprietary bacteriophage-based technology. Armata is developing and advancing a broad pipeline of natural and synthetic phage candidates, including clinical candidates for Pseudomonas aeruginosa, Staphylococcus aureus, and other important pathogens. Armata is committed to advancing phage therapy with drug development expertise that spans bench to clinic including in-house phage-specific current Good Manufacturing Practices (“cGMP”) manufacturing to support full commercialization.

Forward Looking Statements
This communication contains “forward-looking” statements as defined by the Private Securities Litigation Reform Act of 1995. These statements relate to future events, results or to Armata’s future financial performance and involve known and unknown risks, uncertainties and other factors which may cause Armata’s actual results, performance or events to be materially different from any future results, performance or events expressed or implied by the forward-looking statements. In some cases, you can identify these statements by terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “potential,” “predict,” “project,” “should,” “will,” “would” or the negative of those terms, and similar expressions. These forward-looking statements reflect management’s beliefs and views with respect to future events and are based on estimates and assumptions as of the date of this communication and are subject to risks and uncertainties including risks related to Armata’s development of bacteriophage-based therapies; ability to staff and maintain its production facilities under fully compliant cGMP; ability to meet anticipated milestones in the development and testing of the relevant product; ability to be a leader in the development of phage-based therapeutics; ability to achieve its vision, including improvements through engineering and success of clinical trials; ability to successfully complete preclinical and clinical development of, and obtain regulatory approval of its product candidates and commercialize any approved products on its expected timeframes or at all; and Armata’s estimates regarding anticipated operating losses, capital requirements and needs for additional funds. Additional risks and uncertainties relating to Armata and its business can be found under the caption “Risk Factors” and elsewhere in Armata’s filings and reports with the U.S. Securities and Exchange Commission (the “SEC”), including in Armata’s Annual Report on Form 10-K, filed with the SEC on March 21, 2025, and in its subsequent filings with the SEC.
Armata expressly disclaims any obligation or undertaking to release publicly any updates or revisions to any forward-looking statements contained herein to reflect any change in Armata’s expectations with regard thereto or any change in events, conditions or circumstances on which any such statements are based.
Media Contacts:
At Armata:
Pierre Kyme
ir@armatapharma.com
310-665-2928
Investor Relations:
Joyce Allaire
LifeSci Advisors, LLC
jallaire@lifesciadvisors.com
212-915-2569
Exhibit 99.2

1 A Phase 2a Randomized Double - blind, Controlled Trial of the Efficacy and Safety of an Intravenous Bacteriophage Cocktail (AP - SA02) vs. Placebo in Combination with Best Available Antibiotic Therapy (BAT) in Patients with Complicated Staphylococcus aureus Bacteremia Loren G. Miller (MD MPH, Harbor - UCLA Medical Center/Lundquist Institute), Stacey Kolar (PHD, Armata) John Sanders (MD, Wake Forest University), John Williamson (PharmD, Wake Forest University), Paul Allyn (MD, UCLA), Jihoon Baang (MD, University of Michigan), Paul Riska (MD, Montefiore Medical Center), Saima Aslam (MD, University of California, San Diego), George Alangaden (MD, Henry Ford Health System), Jane Wainaina (MD, Froedtert Hospital and the Medical College of Wisconsin), Colleen Kraft (MD, Emory University Hospital), Nirja Mehta (MD, Emory University Hospital), Deena R. Altman (MD, Icahn School of Medicine at Mount Sinai), Gary Wang (MD, University of Florida), Mehdi Mirsaeidi (MD, University of Florida - Jacksonville), D. Alexander Perry, (MD, University of Arizona, Banner Health), Jose Vazquez (MD, Augusta University), Lindsay Nicholson (MD, Rocky Mountain Regional VA Medical Center), Ralph Rogers (MD, Rhode Island Hospital), Jonathan Iredell (MD, Westmead Hospital), Pierre Kyme (PHD, Armata), Deborah Birx (MD, Armata), Vance G. Fowler, Jr.

(MD, Duke University) IDWeek 2025 TM , Atlanta, GA October 22, 2025 2 This presentation contains “forward - looking” statements that involve risks, uncertainties and assumptions . If the risks or uncertainties materialize or the assumptions prove incorrect, our results may differ materially from those expressed or implied by such forward - looking statements . All statements other than statements of historical fact could be deemed forward - looking, including, but not limited to : our estimates regarding anticipated operating losses, capital requirements and needs for additional funds ; our ability to raise additional capital when needed and to continue as a going concern ; our ability to manufacture, or otherwise secure the manufacture of, sufficient amounts of our product candidates for our preclinical studies and clinical trials ; our clinical development plans, including planned clinical trials ; our research and development plans ; our ability to select combinations of phages to formulate our product candidates ; our development of bacteriophage - based therapies ; the potential use of bacteriophages to treat bacterial infections ; the potential future of antibiotic resistance ; the ability for bacteriophage therapies to disrupt and destroy biofilms and restore sensitivity to antibiotics ; the potential for bacteriophage technology being uniquely positioned to address the global threat of antibiotic resistance ; our planned development strategy, presenting data to regulatory agencies and defining planned clinical studies ; the expected timing of additional clinical trials, including Phase 1 b/Phase 2 or registrational clinical trials ; our ability to manufacture and secure sufficient quantities of our product candidates for clinical trials ; the drug product candidates to be supplied by us for clinical trials ; the safety and efficacy of our product candidates ; our anticipated regulatory pathways for our product candidates ; the activities to be performed by specific parties in connection with clinical trials ; our ability to successfully complete preclinical and clinical development of, and obtain regulatory approval of our product candidates and commercialize any approved products on our expected timeframes or at all ; our pursuit of additional indications ; the content and timing of submissions to and decisions made by the U . S . Food and Drug Administration (the “FDA”) and other regulatory agencies ; our ability to leverage the experience of our management team and to attract and retain management and other key personnel ; the capacities and performance of our suppliers, manufacturers, contract research organizations (“CROs”) and other third parties over whom we have limited control ; our ability to staff and maintain our Los Angeles production facility under fully compliant current Good Manufacturing Practices (”cGMP”) ; the actions of our competitors and success of competing drugs or other therapies that are or may become available ; our expectations with respect to future growth and investments in our infrastructure, and our ability to effectively manage any such growth ; the size and potential growth of the markets for any of our product candidates, and our ability to capture share in or impact the size of those markets ; the benefits of our product candidates ; the potential market growth and market and industry trends ; maintaining collaborations with third parties including our partnerships with the Cystic Fibrosis Foundation (the “CFF”) and the U . S . Department of Defense (the “DoD”) ; potential future collaborations with third parties and the potential markets and market opportunities for product candidates ; our ability to achieve our vision, including improvements through engineering and success of clinical trials ; our ability to meet anticipated milestones in the development and testing of the relevant product ; our ability to be a leader in the development of phage - based therapeutics ; the expected use of proceeds from the $ 26 . 2 million DoD award ; the effects of government regulation and regulatory developments, and our ability and the ability of the third parties with whom we engage to comply with applicable regulatory requirements ; the accuracy of our estimates regarding future expenses, revenues, capital requirements and need for additional financing ; our expectations regarding future planned expenditures ; our ability to achieve and maintain effective internal control over financial reporting in accordance with Section 404 of the Sarbanes - Oxley Act ; our ability to obtain, maintain and successfully enforce adequate patent and other intellectual property protection of any of our products and product candidates ; our ability to protect our intellectual property, including pending and issued patents ; our ability to operate our business without infringing the intellectual property rights of others ; our ability to advance our clinical development programs ; the effects of ongoing conflicts between Ukraine and Russia and in the Middle East, the recent and potential future bank failures or other geopolitical events ; the potential economic and regulatory impacts on the biotechnology, pharmaceutical and drug manufacturing industries ; the effects of artificial intelligence on our business and the industry as a whole ; and statements of belief and any statement of assumptions underlying any of the items mentioned . These statements are based on estimates and information available to us at the time of this presentation and are not guarantees of future performance . Actual results could differ materially from our current expectations as a result of these risks and uncertainties, which include, without limitation, risks related to the ability of our lead clinical candidates, AP - PA 02 and AP - SA 02 (including any modifications thereto) to be more effective than previous candidates ; our ability to enhance AP - PA 02 to treat both CF and NCFB patients ; our ability to develop products as expected ; our expected market opportunity for our products ; our ability to sufficiently fund our operations as expected, including obtaining additional funding as needed, and to refinance, repay or restructure its debt ; and whether Armata will incur unforeseen expenses or liabilities . You should not rely upon forward - looking statements as predictions of future events . Although we believe that the expectations reflected in the forward - looking statements are reasonable, we cannot guarantee that the future results, levels of activity, performance or events and circumstances reflected in the forward - looking statements will be achieved or occur . Moreover, we undertake no obligation to update publicly any forward - looking statements for any reason to conform these statements to actual results or to changes in our expectations except as required by law . We refer you to the documents that we file from time to time with the Securities and Exchange Commission, including our most recently filed Annual Report on Form 10 - K, Quarterly Reports on Form 10 - Q and Current Reports on Form 8 - K . These documents, including the sections therein entitled “Risk Factors,” identify important factors that could cause the actual results to differ materially from those contained in forward - looking statements . In addition, this presentation also contains estimates, projections and other information concerning our industry, our business, and the markets for our product candidates, as well as data regarding market research, estimates and forecasts prepared by our management . Information that is based on estimates, forecasts, projections, market research or similar methodologies is inherently subject to uncertainties and actual events or circumstances may differ materially from events and circumstances reflected in this information . These statements are based upon information available to us as of the date of this presentation, and while we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and our statements should not be read to indicate that we have conducted an exhaustive inquiry into, or review of, all potentially available relevant information . Armata Pharmaceuticals Forward Looking Statements 3 Disclosures Dr. Loren Miller has received Grants/Research Support from: Armata GSK Paratek ContraFect Merck NIH A full list of disclosures is available on the IDWeek 2025 TM website
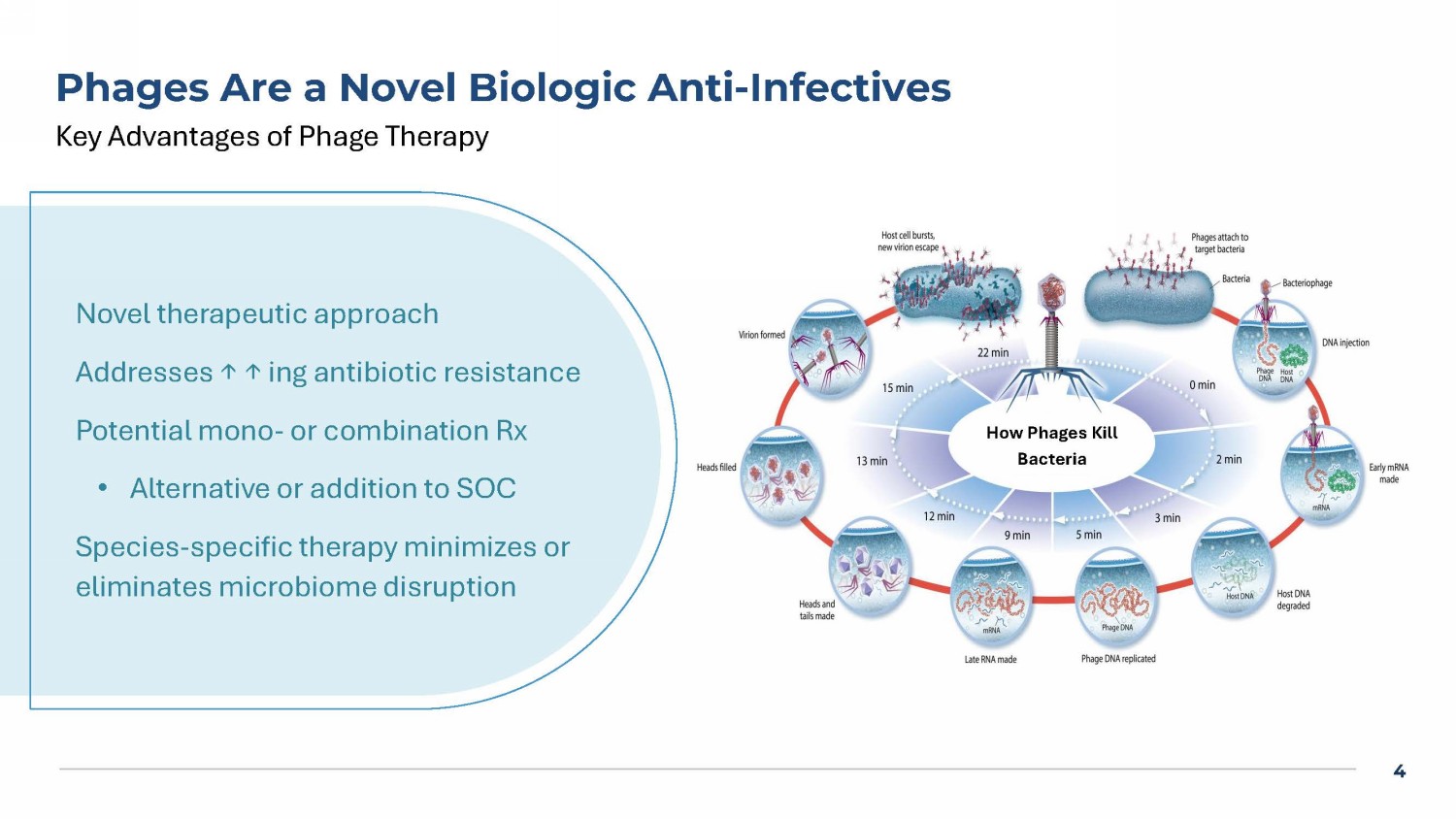

4 Phages Are a Novel Biologic Anti - Infectives Key Advantages of Phage Therapy How Phages Kill Bacteria Novel therapeutic approach Addresses ↑ ↑ ing antibiotic resistance Potential mono - or combination Rx • Alternative or addition to SOC Species - specific therapy minimizes or eliminates microbiome disruption 5 Bacteremia Phase 1b/2a “diSArm” Study Design AP - SA02 5 days q6h * TOC for AP - SA02: 7 days after end of IV AP - SA02 (day 12) PFU = Plaque forming units BAT = Best available antibiotic therapy * < 72 hrs of IV BAT prior to start of AP - SA02 Screening IV BAT 14 - 56 days 28 days post IV BAT TOC for EOS: 28 days after end of IV BAT (~day 39 - 81) TOC for BAT: 7 days after end of IV BAT (~day 18 - 60) Responder defined as all these signs and symptoms resolved from screening: temperature, heart rate, respiratory rate, white b lo od cell count, systolic blood pressure, pain associated with infection site Phase 1 b (n=8; 3:1): dose escalating Phase 2 a (n=42; 2:1): 28 sites Dosed IV push q6 hrs x 5 d + SOC Antibiotics Dose of AP - SA02: 2 x 10 10 PFU or 5 x 10 10 PFU AP - SA02 is a 2 - phage cocktail consisting of ARSA0001 and ARSA0002 ITT: All Phase 2a subjects that received BAT and > 1 dose of AP - SA02 or placebo 6 Phase 2a Trial Demographics & Infection Site Placebo (n=16)* * AP - SA02 (n=31)* Demographics 52.6 +/ - 12 54.8 +/ - 14 Age (mean) +/ - SD 10(62) 22(71) Sex Male (%) 28.5 +/ - 7.0 29.8 +/ - 7.8 BMI, mean +/ - SD 12 (75) 21 (67) White n (%) 2 (12) 8 (26) Black n (%) 5 (31) 12 (45) Hispanic n (%) 7 (44) 12 (39) MRSA n (%) Placebo (n=13) AP - SA02 (n=29) Site of Infections , ITT population: All patients that received AP - SA02 or placebo n (%) n (%) 3 (23) 4 (14) Septic Joint 3 (23) 4 (14) Cellulitis 4 (31) 9 (31) Osteomyelitis 0 (0) 3 (10) Pneumonia 3 (23) 6 (21) Sepsis 0 (0) 2 (7) L - sided Endocarditis 1 (8) 1(3) R - sided Endocarditis *includes all randomized from phase 2a trial, including not dosed

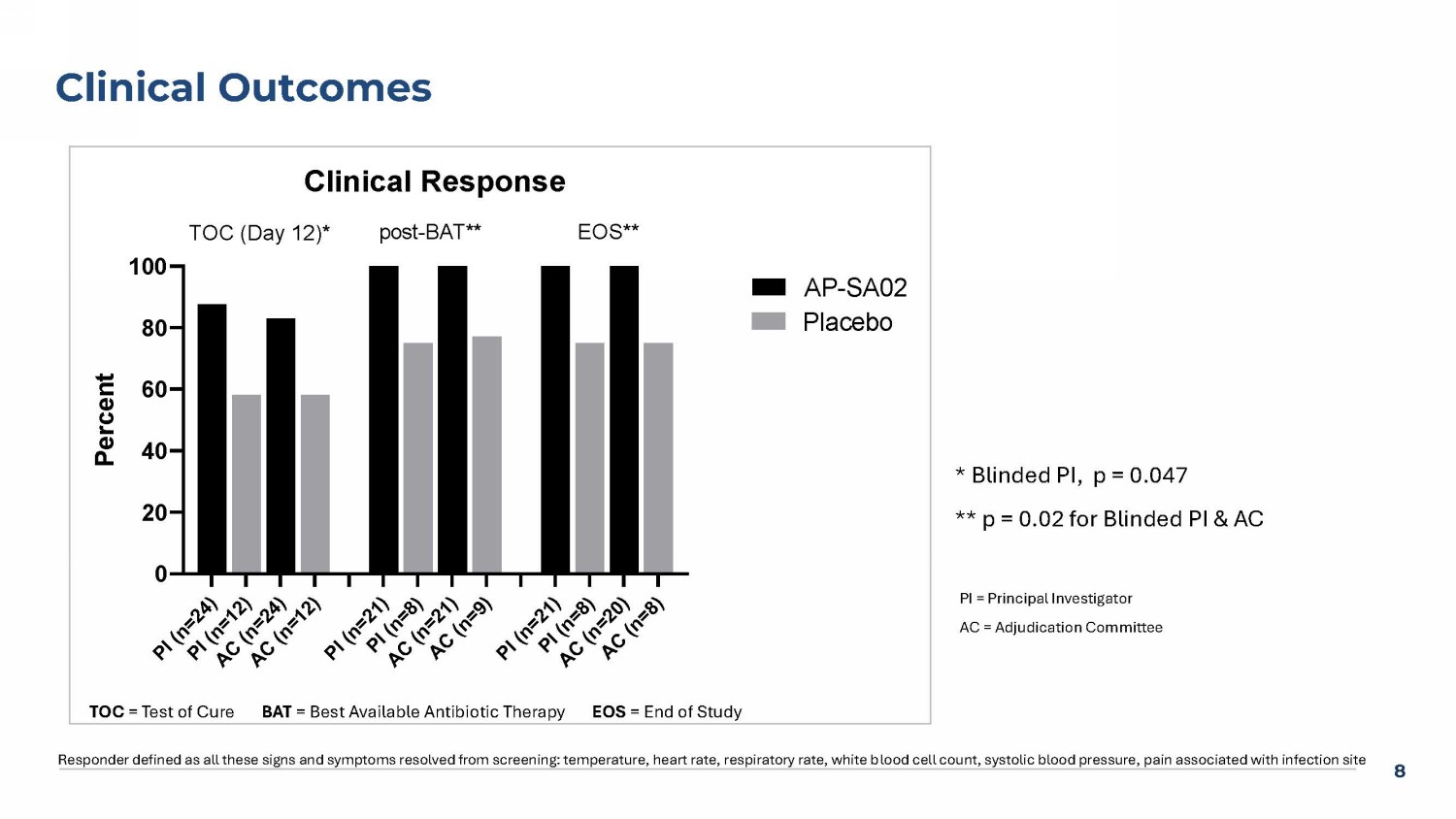
7 Phase 2a Trial BAT Antibiotics Placebo (n=13) n (%) AP - SA02 (n=29) n (%) Antibiotics Used* 8 (62) 18 (62) Cefazolin 6 (46) 12 (41) Vancomycin 2(15) 9 (31) Oxacillin/nafcillin 5 (38) 5 (17) Daptomycin 1(8) 2 (7) Cefepime 0 (0) 4 (14) Ceftriaxone BAT = Best available therapy *Some subjects received >1 antibiotic 8 Clinical Outcomes Responder defined as all these signs and symptoms resolved from screening: temperature, heart rate, respiratory rate, white b loo d cell count, systolic blood pressure, pain associated with infection site PI = Principal Investigator AC = Adjudication Committee * Blinded PI, p = 0.047 ** p = 0.02 for Blinded PI & AC P I ( n = 2 4 ) P I ( n = 1 2 ) A C ( n = 2 4 ) A C ( n = 1 2 ) P I ( n = 2 1 ) P I ( n = 8 ) A C ( n = 2 1 ) A C ( n = 9 ) P I ( n = 2 1 ) P I ( n = 8 ) A C ( n = 2 0 ) A C ( n = 8 ) 0 20 40 60 80 100 Clinical Response P e r c e n t AP-SA02 Placebo TOC (Day 12)* EOS**post-BAT** TOC = Test of Cure BAT = Best Available Antibiotic Therapy EOS = End of Study 9 Response in Subjects With MRSA Cleared Infection, No Evidence of Relapse TOC = Test of Cure BAT = Best Available Antibiotic Therapy EOS = End of Study A P - S A 0 2 ( n = 8 ) P l a c e b o ( n = 5 ) A P - S A 0 2 ( n = 6 ) P l a c e b o ( n = 4 ) A P - S A 0 2 ( n = 5 ) P l a c e b o ( n = 3 ) 0 10 20 30 40 50 60 70 80 90 100 P e r c e n t TOC (Day 12) EOS post-BAT A P - S A 0 2 ( n = 1 6 ) P l a c e b o ( n = 7 ) A P - S A 0 2 ( n = 1 5 ) P l a c e b o ( n = 5 ) A P - S A 0 2 ( n = 1 5 ) P l a c e b o ( n = 5 ) 0 10 20 30 40 50 60 70 80 90 100 P e r c e n t AP-SA02 Placebo TOC (Day 12) EOS post-BAT MRSA MSSA TOC (Day12) Post - BAT EOS TOC (Day12) Post - BAT EOS


10 Clinical Trends for AP - SA02 vs. Placebo Placebo AP - SA02 Clinical Trends 19.2 (13) 11.7 (26) Time to hospital discharge: Mean Day (n) 9.3 (12) 2.7 (25) Time to initial SAB resolution: Mean Day (n ) 97.3 (13) 50.2 (26) Mean CRP Day 12 mg/L (n)

11 AP - SA02 vs. Placebo: Safety & Tolerability Safety Population (N=50) Phase 2a Complicated SAB Phase 1b Uncomplicated SAB Placebo (N=13) AP - SA02 (N=29) Placebo (N=2) AP - SA02 (N=6) n (%) n (%) n (%) n (%) 12 (92) 19 (66) 2 (100) 6 (100) Any adverse events (AEs) 10 (77) 17 (59) 2 (100) 6 (100) Any treatment - emergent AEs (TEAEs) 1 0 1 (3) 0 1 (17) 2 Any study drug related TEAEs 3 (23) 4 (14) 0 (0) 3 (50) Any Best Available Therapy related TEAEs 3 (23) 4 (14) 1 (50) 5 (83) Any serious AEs (SAEs) 9 (69) 9 (31) 1 (50) 5 (83) NCI CTCAE Grade 3/4/5 Aes 7 (54) 8 (28) 1 (50) 5 (83) NCI CTCAE Grade 3/4/5 TEAEs 0 0 0 1 (17) Any TEAEs leading to interruption of study drug 1 (8) 0 0 0 Any TEAEs leading to withdrawal of study drug 0 1 (3) 0 0 Any TEAEs leading to discontinuation of study 0 1 (3) 3 0 0 Any AEs leading to death 4 1. TEAEs are defined as adverse events (AEs) occurring after the first dose of AP - SA02 through TOC (Day 12) or through EOS for S AEs. 2. Concurrent with Vancomycin and resolved with discontinuation of Vancomycin. 3. Transient transaminitis (mean 386 U/L ALT, 316 U/L AST) began on Day 4, persisted through Day 7, and was returned to norma l i n next blood draw on Day 12. 4. Subject was blood culture negative for S. aureus by Day 3/5 of AP - SA02 treatment (8 days before death); fatal (unrelated) event of multiple organ failure determined by study PI to be unrelated to both study drug and vancomycin. NCI CTCAE = National Cancer Institute Common Terminology Criteria for Adverse Events.

12 AP - SA02 Binds, Infects, and Lyses Patient Isolates in vitro Genotypic mapping of ARSA0002 Phage Less Frequent Variants More Frequent Variants 3 2 1 3 2 1 Patient Isolates 4 34 3 57 54 21 01 4 26 3 65 67 16 02 7 29 2 59 58 20 03 5 20 3 55 39 17 04 30 23 27 46 05 42 51 20 46 06 60 1 3 4 3 07 100 08 Defined phage variants in the AP - SA02 drug product ensure an intrinsic adaptive mechanism — a flexibility that may be key to achieving effective phage therapy from patient to patient Genetic Variants of ARSA0002 Phage Production Runs AP - SA02 reference 13 Summary & Conclusion • AP - SA02 phage cocktail safe and well tolerated • In combination with BAT, AP - SA02 had an earlier and higher cure rate compared to placebo in patients with complicated SAB at day 12, post BAT, and at EOS • AP - SA02 was assessed with clinical trends of earlier resolution, shorter hospitalization, and lack of relapse 4 weeks post - therapy • Results support proceeding to a Phase 3 RCT of this novel bacteriophage cocktail for SAB


14 *Armata is the recipient of a $26.2 million DoD award, received through the Medical Technology Enterprise Consortium (MTEC) and managed by the Naval Medical Research Command (NMRC) – Naval Advanced Medical Development (NAMD) with funding from the Defense Health Agency and Joint Warfighter Medical Research Program Acknowledgments Armata’s clinical breakthrough has been made possible with the ongoing financial support and partnership from the U.S. Department of Defense* and Innoviva , and the patients willing to be part of clinical research Proud Sponsor of diSArm Clinical Study 15 Thank you.


If you would be interested in being part of the Phase 3 Trial please contact Jennifer Smith jsmith@armatapharma.com 16 Proposed Superiority Pivotal Trial Design • Primary endpoint clinical response at day 60 (TOC) • Secondary endpoints – Clinical response at day 14 (TOC) – Time to hospital discharge – Microbiologic eradication at time 60 and 14 days – All Cause Mortality • Sample size: 406 total (203 per group) to detect 15% absolute improvement with 90% power, alpha = 0.05 • Provides adequate safety data for BLA