UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 OR 15(d)
of The Securities Exchange Act of 1934
Date of Report (Date of Earliest Event Reported): August 1, 2025
OCUGEN, INC.
(Exact Name of Registrant as Specified in its Charter)
| Delaware | 001-36751 | 04-3522315 | ||
|
(State or Other Jurisdiction of Incorporation) |
(Commission File Number) |
(I.R.S. Employer Identification Number) |
11 Great Valley Parkway
Malvern, Pennsylvania 19355
(484) 328-4701
(Address, including zip code, and telephone number, including area code, of principal executive office)
N/A
(Former Name or Former Address, if Changed Since Last Report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
| ¨ | Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ¨ | Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ¨ | Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ¨ | Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||
| Common Stock, $0.01 par value per share | OCGN |
The Nasdaq Stock Market LLC (The Nasdaq Capital Market) |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ¨
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Item 2.02 Results of Operations and Financial Condition.
On August 1, 2025, Ocugen, Inc. (the "Company") issued a press release announcing certain financial results for the quarter ended June 30, 2025. The Company has scheduled a conference call and webcast for 8:30 a.m. Eastern Time on August 1, 2025, to discuss these financial results and business updates. The Company will use presentation materials in connection with the conference call and webcast, which presentation materials will be posted on the Company's website at www.ocugen.com. Copies of the press release and presentation materials are furnished herewith as Exhibit 99.1 and Exhibit 99.2, respectively, to this Current Report on Form 8-K (this "Report") and incorporated herein by reference.
The information disclosed under Item 2.02 of this Report, including Exhibit 99.1 and Exhibit 99.2, is being furnished and shall not be deemed "filed" for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the "Exchange Act"), or otherwise subject to the liabilities of that section, and shall not be deemed to be incorporated by reference in any Company filing under the Securities Act of 1933, as amended (the "Securities Act"), or the Exchange Act, except as expressly set forth by specific reference in such filing.
Item 9.01 Financial Statements and Exhibits.
The following exhibits are being furnished herewith:
(d) Exhibits
| Exhibit No. | Document | |
| 99.1 | Press Release of Ocugen, Inc. dated August 1, 2025. | |
| 99.2 | Earnings Release Presentation issued August 1, 2025. | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document). |
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
Date: August 1, 2025
| OCUGEN, INC. |
| By: | /s/ Shankar Musunuri | |
| Name: Shankar Musunuri | ||
| Title: Chairman, Chief Executive Officer, & Co-Founder |
Exhibit 99.1
Ocugen Provides Business Update with Second Quarter 2025 Financial Results
Conference Call and Webcast Today at 8:30 a.m. ET
· Initiated dosing in OCU410ST Phase 2/3 GARDian3 pivotal confirmatory clinical trial
· Actively dosing patients in OCU400 Phase 3 liMeliGhT clinical trial and on track for 2026 BLA filing
· OrthoCellix reverse merger intended to unlock the value of NeoCart/regenerative cell therapies and enable the Company to focus capital on modifier gene therapy platform
· Signed binding term sheet for exclusive Korean rights to OCU400 with upfront fees and near-term development milestone payments totaling up to $11 million
MALVERN, Pa., Aug. 01, 2025 (GLOBE NEWSWIRE) – Ocugen, Inc. (Ocugen or the Company) (NASDAQ: OCGN), a pioneering biotechnology leader in gene therapies for blindness diseases, today reported second quarter 2025 financial results along with a business update.
“While our modifier gene therapy clinical trials advance—now with two in late-stage—we are securing strategic partnerships and evolving the business to support three successful Biologics License Application (BLA) filings over the next three years,” said Dr. Shankar Musunuri, Chairman, CEO, and Co-founder of Ocugen. “We have also made important appointments to our Board of Directors, Retina Scientific Advisory Board, and Leadership Team to provide the Company with scientific and strategic know-how to bring us closer to delivering paradigm-changing gene therapies to millions of people with blindness diseases.”
In June, the Company announced a proposed reverse merger with OrthoCellix, a wholly-owned subsidiary, and Carisma Therapeutics, Inc., to create a Nasdaq-listed, late clinical-stage regenerative cell therapy company with a first-in-class technology platform, focused on orthopedic diseases. The combined company will focus on the development of OrthoCellix’s NeoCart® technology for the treatment of articular knee cartilage defects. Previously, NeoCart® received Regenerative Medicine Advanced Therapy (RMAT) designation and concurrence from the U.S. Food and Drug Administration (FDA) on a single, confirmatory Phase 3 clinical trial to enable submission of a BLA.
Aligned with Ocugen’s business development strategy to pursue regional partnerships for OCU400, the Company signed a binding term sheet to negotiate and enter into a licensing agreement with a well-established leader in the pharmaceutical and healthcare sector in Korea for exclusive Korean rights to OCU400. Pursuant to the term sheet, under the license agreement, in addition to the upfront and milestone fees, the Company will be entitled to sales milestones of $1 million for every $15 million of net sales in Korea in addition to a royalty of 25% on net sales of OCU400 generated by Ocugen’s partner. Ocugen will manufacture commercial supply of OCU400 under terms of a supply agreement. A regional approach preserves Ocugen’s rights to larger geographies to maximize total patient reach while also generating return for shareholders.
Following the FDA’s agreement to proceed with a Phase 2/3 GARDian3 pivotal confirmatory trial for OCU410ST for Stargardt disease, the agency granted Rare Pediatric Disease Designation (RPDD) to OCU410ST in May. This designation underscores the urgent need to address Stargardt disease, which remains a significant unmet medical need. Stargardt disease is an inherited retinal disorder that typically presents in childhood and affects approximately 100,000 people in the U.S. and Europe combined, and approximately 1 million globally. Currently, there is no FDA-approved treatment available for Stargardt disease.
The OCU410ST Phase 2/3 GARDian3 clinical trial is progressing well with the first patient dosed in July after FDA clearance in June. The GARDian3 clinical trial builds upon encouraging results and positive data from the Phase 1 GARDian trial, which demonstrated 48% slower lesion growth at 12-month follow-up in evaluable treated eyes compared to untreated eyes. Additionally, evaluable treated eyes showed a statistically significant (p=0.031) and clinically meaningful improvement of nearly 2-line/9-letter gain in best corrected visual acuity (BCVA) at 12-month follow-up when compared to untreated eyes.
Positive preliminary efficacy and safety data from the OCU410 Phase 1 ArMaDa clinical trial at 12 months demonstrated no drug-related serious adverse events (SAEs), 23% slower geographic atrophy (GA) lesion growth in treated eyes versus fellow eyes after a single injection, and 2-line/10-letter gain in visual acuity in treated eyes when compared to untreated fellow eyes. Preliminary results from ongoing Phase 2 clinical trial (N=31), 6-month interim analysis, demonstrated a 27% slower lesion growth and preservation of retinal tissue. These data support the potential for OCU410 to provide a one-time treatment for life for the 2-3 million people in the U.S. & EU combined who suffer from GA.
Patients are actively being recruited in the United States and Canada for the OCU400 Phase 3 liMeliGhT clinical trial, which remains on track for BLA and MAA submissions in 2026. This is the only broad retinitis pigmentosa (RP) gene-agnostic trial to address multiple genetic mutations with a single therapeutic approach. In addition, the European Medicines Agency has granted eligibility to submit the OCU400 Marketing Authorization Application (MAA) through the centralized procedure, based on the current study design and statistical analysis plan.
Regarding the Company’s inhaled vaccines portfolio, the National Institute of Allergy and Infectious Diseases (NIAID) intends to initiate the Phase 1 clinical trial for OCU500 in the third quarter of 2025.
In addition to the notable leadership appointments, Ocugen welcomed the National Security Commission on Emerging Biotechnology (NSCEB) and U.S. Rep. Chrissy Houlahan to its manufacturing facility as part of the NSCEB’s Biotech Across America events, highlighting biotech innovation in Pennsylvania. Rep. Houlahan subsequently announced the bipartisan BIOTech Caucus to build greater awareness and understanding of biotechnology among lawmakers and support transformative advances in healthcare. Dr. Musunuri supports the formation of this very important bipartisan BIOTech Caucus that includes senior congressional leaders such as Rep. Pete Sessions in addition to local leaders, which will prioritize biotechnology at the national level to ensure U.S. leadership globally.
“The meaningful progress Ocugen is making across its novel modifier gene therapy platform, along with strategic leadership changes and significant external alliances are evidence of a strong first half of 2025,” said Dr. Musunuri. “We look forward to providing critical program updates and data in the coming months.”
Modifier Gene Therapy Platform—a Novel First-in-Class Platform
| · | OCU400 for RP – On track to complete enrollment in support of BLA/MAA filings in 2026. Data and Safety Monitoring Board (DSMB) convened and found no SAEs related to OCU400 and recommended to continue study dosing as planned. | |
| · | OCU410ST for Stargardt Disease – FDA granted RPDD for OCU410ST for the treatment of ABCA4-associated retinopathies including Stargardt disease, retinitis pigmentosa 19, and cone-rod dystrophy 3. FDA cleared the Investigational New Drug (IND) amendment to initiate a Phase 2/3 pivotal confirmatory trial of OCU410ST and dosing has been initiated. |
| · | OCU410 for GA – Phase 1 data at 12 months demonstrates reduced lesion growth, preservation of retinal tissue, and—most importantly—a positive effect on the functional visual measure of low luminance visual acuity (LLVA). Interim Phase 2 data at 6 months demonstrated very encouraging results consistent with Phase 1 data. |
Ophthalmic Biologic Product
| · | OCU200 –DSMB approved continuation of dosing in the third cohort and the Company intends to complete the Phase 1 clinical trial in the second half of 2025. |
Second Quarter 2025 Financial Results
| · | The Company’s cash, cash equivalents, and restricted cash totaled $27.3 million as of June 30, 2025, compared to $58.8 million as of December 31, 2024, providing cash runway into the first quarter of 2026. The Company had 292.2 million shares of common stock outstanding as of June 30, 2025. |
| · | Total operating expenses for the three months ended June 30, 2025 were $15.2 million and included research and development expenses of $8.4 million and general and administrative expenses of $6.8 million. This compares to total operating expenses for the three months ended June 30, 2024 of $16.6 million that included research and development expenses of $8.9 million and general and administrative expenses of $7.7 million. |
| · | Ocugen reported a $0.05 net loss per common share for the three months ended June 30, 2025 compared to a $0.06 net loss per common share for the three months ended June 30, 2024. |
Conference Call and Webcast Details
Ocugen has scheduled a conference call and webcast for 8:30 a.m. ET today to discuss the financial results and recent business highlights. Ocugen’s executive leadership team will host the call, which will be open to all listeners. There also will be a question-and-answer session following the prepared remarks.
Attendees are invited to participate on the call or webcast:
Dial-in Numbers: (800) 715-9871 for U.S. callers and (646) 307-1963 for international callers
Conference ID: 9627149
Webcast: Available on the events section of the Ocugen investor site
A replay of the call and archived webcast will be available for approximately 45 days following the event on the Ocugen investor site.
About Ocugen, Inc.
Ocugen, Inc. is a pioneering biotechnology leader in gene therapies for blindness diseases. Our breakthrough modifier gene therapy platform has the potential to address significant unmet medical need for large patient populations through our gene-agnostic approach. Unlike traditional gene therapies and gene editing, Ocugen’s modifier gene therapies address the entire disease—complex diseases that are potentially caused by imbalances in multiple gene networks. Currently we have programs in development for inherited retinal diseases and blindness diseases affecting millions across the globe, including retinitis pigmentosa, Stargardt disease, and geographic atrophy—late stage dry age-related macular degeneration. Discover more at www.ocugen.com and follow us on X and LinkedIn.
Cautionary Note on Forward-Looking Statements
This press release contains forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995, including, but not limited to, strategy, business plans and objectives for Ocugen’s clinical programs, plans and timelines for the preclinical and clinical development of Ocugen’s product candidates, including the therapeutic potential, clinical benefits and safety thereof, expectations regarding timing, success and data announcements of current ongoing preclinical and clinical trials, the ability to initiate new clinical programs; Ocugen’s financial condition and expected cash runway into the first quarter of 2026, statements regarding qualitative assessments of available data, potential benefits, expectations for ongoing clinical trials, anticipated regulatory filings and anticipated development timelines, and Ocugen’s negotiations regarding the license agreement with a Korean partner and Ocugen’s potential merger transaction regarding the OrthoCellix business, which are subject to risks and uncertainties. We may, in some cases, use terms such as “predicts,” “believes,” “potential,” “proposed,” “continue,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should,” or other words that convey uncertainty of future events or outcomes to identify these forward-looking statements. Such statements are subject to numerous important factors, risks, and uncertainties that may cause actual events or results to differ materially from our current expectations, including, but not limited to, the risks that preliminary, interim and top-line clinical trial results may not be indicative of, and may differ from, final clinical data; that unfavorable new clinical trial data may emerge in ongoing clinical trials or through further analyses of existing clinical trial data; that earlier non-clinical and clinical data and testing of may not be predictive of the results or success of later clinical trials; and that that clinical trial data are subject to differing interpretations and assessments, including by regulatory authorities; that a definitive agreement for the license with a Korean partner will be delayed or not executed at all, or that, if executed, it may not be on terms anticipated; that the OrthoCellix merger transaction may not close or, if closed, may not result in the benefits anticipated. These and other risks and uncertainties are more fully described in our annual and periodic filings with the Securities and Exchange Commission (SEC), including the risk factors described in the section entitled “Risk Factors” in the quarterly and annual reports that we file with the SEC. Any forward-looking statements that we make in this press release speak only as of the date of this press release. Except as required by law, we assume no obligation to update forward-looking statements contained in this press release whether as a result of new information, future events, or otherwise, after the date of this press release.
Contact:
Tiffany Hamilton
AVP, Head of Communications
Tiffany.Hamilton@ocugen.com
CONDENSED CONSOLIDATED BALANCE SHEETS
(in thousands, except share and per share amounts)
(Unaudited)
| June 30, 2025 | December 31, 2024 | |||||||
| Assets | ||||||||
| Current assets | ||||||||
| Cash | $ | 27,013 | $ | 58,514 | ||||
| Prepaid expenses and other current assets | 5,870 | 3,168 | ||||||
| Total current assets | 32,883 | 61,682 | ||||||
| Property and equipment, net | 15,445 | 16,554 | ||||||
| Restricted cash | 312 | 307 | ||||||
| Other assets | 4,954 | 3,899 | ||||||
| Total assets | $ | 53,594 | $ | 82,442 | ||||
| Liabilities and stockholders' equity | ||||||||
| Current liabilities | ||||||||
| Accounts payable | $ | 4,237 | $ | 4,243 | ||||
| Accrued expenses and other current liabilities | 12,899 | 15,500 | ||||||
| Operating lease obligations | 853 | 519 | ||||||
| Current portion of long term debt | — | 1,326 | ||||||
| Total current liabilities | 17,989 | 21,588 | ||||||
| Non-current liabilities | ||||||||
| Operating lease obligations, less current portion | 3,945 | 3,313 | ||||||
| Long term debt, net | 28,025 | 27,345 | ||||||
| Other non-current liabilities | 583 | 564 | ||||||
| Total non-current liabilities | 32,553 | 31,222 | ||||||
| Total liabilities | 50,542 | 52,810 | ||||||
| Stockholders' equity | ||||||||
| Common Stock | 2,924 | 2,915 | ||||||
| Treasury stock | (48 | ) | (48 | ) | ||||
| Additional paid-in capital | 370,474 | 366,938 | ||||||
| Accumulated other comprehensive income | 12 | 48 | ||||||
| Accumulated deficit | (370,31 | ) | (340,221 | ) | ||||
| Total stockholders' equity | 3,052 | 29,632 | ||||||
| Total liabilities and stockholders' equity | $ | 53,594 | $ | 82,442 | ||||
OCUGEN, INC.
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
(in thousands, except share and per share amounts)
(Unaudited)
| Three months ended June 30, | Six months ended June 30, | |||||||||||||||
| 2025 | 2024 | 2025 | 2024 | |||||||||||||
| Collaborative arrangement revenue | $ | 1,373 | $ | 1,141 | $ | 2,854 | $ | 2,155 | ||||||||
| Total revenue | 1,373 | 1,141 | 2,854 | 2,155 | ||||||||||||
| Operating expenses | ||||||||||||||||
| Research and development | 8,402 | 8,902 | 17,932 | 15,728 | ||||||||||||
| General and administrative | 6,766 | 7,688 | 13,218 | 14,092 | ||||||||||||
| Total operating expenses | 15,168 | 16,590 | 31,150 | 29,820 | ||||||||||||
| Loss from operations | (13,795 | ) | (15,449 | ) | (28,296 | ) | (27,665 | ) | ||||||||
| Interest (expense) income, net | (1,058 | ) | 173 | (1,972 | ) | 475 | ||||||||||
| Other (expense) income, net | 114 | (4 | ) | 179 | (14 | ) | ||||||||||
| Net loss | $ | (14,739 | ) | $ | (15,280 | ) | $ | (30,089 | ) | $ | (27,204 | ) | ||||
| Other comprehensive income (loss) | ||||||||||||||||
| Foreign currency translation adjustment | (28 | ) | 3 | (36 | ) | 8 | ||||||||||
| Comprehensive loss | $ | (14,767 | ) | $ | (15,277 | ) | $ | (30,125 | ) | $ | (27,196 | ) | ||||
| Net loss attributable to common shareholders— basic and diluted | (14,739 | ) | (15,259 | ) | (30,089 | ) | (27,157 | ) | ||||||||
| Weighted shares used in calculating net loss per common share — basic and diluted | 292,067,192 | 257,353,857 | 292,032,072 | 257,293,247 | ||||||||||||
| Net loss per share attributable to common shareholders — basic and diluted | $ | (0.05 | ) | $ | (0.06 | ) | $ | (0.10 | ) | $ | (0.11 | ) | ||||
| Net loss attributable to Series B Convertible Preferred shareholders — basic and diluted | — | (21 | ) | — | (47 | ) | ||||||||||
| Weighted shares used in calculating net loss per Series B Convertible Preferred Stock — basic and diluted | — | 54,745 | — | 54,745 | ||||||||||||
| Net loss per share attributable to Series B Convertible Preferred shareholders — basic and diluted | — | (0.38 | ) | — | (0.86 | ) | ||||||||||
Exhibit 99.2


Courageous Innovation Dedicated to Bringing Game - Changing Gene Therapies to Market and Working Even Harder to Provide Access to Patients Globally 2Q 2025 Business Update August 1, 2025 2 This presentation contains forward - looking statements within the meaning of The Private Securities Litigation Reform Act of 1995 , including, but not limited to, strategy, business plans and objectives for Ocugen’s clinical programs, plans and timelines for the precl ini cal and clinical development of Ocugen’s product candidates, including the therapeutic potential, clinical benefits and safety thereof, expect ati ons regarding timing, success and data announcements of current ongoing preclinical and clinical trials, the ability to initiate new clinic al programs; Ocugen’s expected cash runway, statements regarding qualitative assessments of available data, potential benefits, expectations for ongoing clinical trials, anticipated regulatory filings and anticipated development timelines and our negotiations regarding the lic ens e agreement with a Korean partner and our potential transaction regarding the OrthoCellix business , which are subject to risks and uncertainties. We may, in some cases, use terms such as “predicts,” “believes,” “potential,” “proposed,” “continue,” “estimates,” “anticipates,” “ex pec ts,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should,” or other words that convey uncertainty of future events or outcomes to ide ntify these forward - looking statements. Such statements are subject to numerous important factors, risks, and uncertainties that may cause a ctual events or results to differ materially from our current expectations, including, but not limited to, the risks that prelimina ry, interim and top - line clinical trial results may not be indicative of, and may differ from, final clinical data; that unfavorable new clinical trial data may emerge in ongoing clinical trials or through further analyses of existing clinical trial data; that earlier non - clinical and clinical d ata and testing of may not be predictive of the results or success of later clinical trials; and that that clinical trial data are subject to differ ing interpretations and assessments, including by regulatory authorities; that a definitive agreement for the license with a Korean partner will be d ela yed or not executed at all, or that, if executed, it may not be on terms anticipated; that the OrthoCellix transaction may not close or, if closed, may not result in the benefits anticipated . These and other risks and uncertainties are more fully described in our annual and periodic filings with the Securities and Exchange Commission (SEC), including the risk factors described in the section entitled “Risk Factors” in the qua rterly and annual reports that we file with the SEC. Any forward - looking statements that we make in this presentation speak only as of the date of this presentation. Except as required by law, we assume no obligation to update forward - looking statements contained in this presenta tion whether as a result of new information, future events, or otherwise, after the date of this presentation.


Forward - Looking Statements Potential for Three Biologics License Applications (BLAs) in the Next Three Years Target BLA/MAA Submission U.S./EU Prevalence Status Product Candidate 2026 300,000 Phase 3 in Progress (Largest Orphan Gene Therapy Clinical Trial) – on Target to File BLA in 2026 Collaboration with Molly Burke to Raise Awareness for the LiMeliGhT Clinical Trial on Social Media O CU400 Retinitis Pigmentosa (RP) (Gene - agnostic targeting >100 genes, broad indication) 2027 100,000 Phase 2/3 Pivotal Confirmatory Initiated Dosing OCU410ST Stargardt Disease (ABCA4 - associated retinopathies >1,200 mutations) 2028 2 - 3 million Completed Phase 2 Recruitment Planning to Initiate Phase 3 in 2026 OCU410 Geographic Atrophy (GA) – Advanced dry age - related macular degeneration ( dAMD ) Leader in Ophthalmology Gene Therapies 3 OCU400 First - in - Class Novel Modifier Gene Therapy for RP Unmet Need ~300,000 people in the U.S./EU and 1.6 million globally suffer from RP, which affects > 100 genes One approved product (Luxturna ® ), a gene therapy, which can address only one gene ( RPE65 ) 4 2,000 298,000 OCU400 Market Potential Luxturna OCU 400 Luxturna peak annual sales $52M* (2023) with a patient population of ~2,000 OCU400 has the potential to treat 298,000 patients First Phase 3 gene therapy clinical trial to receive broad RP indication from FDA Received RMAT & ODD from FDA and OMPD & ATMP from EMA *https://assets.roche.com Potential Solution OCU400 uses a gene - agnostic approach that is delivered via a single, subretinal injection to target all 100 genes associated with RP A traditional gene therapy approach would require developing 100 different products to address all 100 genes Current Status of OCU400: • Largest orphan gene therapy clinical trial (Phase 3) in progress • On track to meet 2026 BLA and MAA filing targets • Process validation (manufacturing) anticipated to be completed this year • The Data and Safety Monitoring Board (DSMB) recently convened and found no Serious Adverse Events (SAEs) related to OCU400 and recommended to continue study dosing as planned Recent Accomplishment • EMA granted eligibility to submit OCU400 MAA via the centralized procedure as an ATMP OCU410ST First - in - Class Novel Modifier Gene Therapy Product for Stargardt Disease 5 100,000 OCU410ST Market Potential OCU410ST Pricing for current gene therapies ranges between $1 - 4M/patient Received ODD & OMPD for ABCA4 - associated Retinopathies 2 https://www.rxcinternational.com/monthly - gene - therapy - review/a - little - earlier - than - expected - sparks - announces - luxturna - pricing - 85 0k 1 P Kohli et al., StatPearls , 2024.

https://www.ncbi.nlm.nih.gov/books/NBK587351/ Unmet Need ~100,000 people in U.S./EU and 1 million globally suffer from ABCA4 - associated retinopathies 1 (>1,200 mutations). Currently there are no approved products in the market. Potential Solution OCU410ST has potential to upregulate key functioning genes in the network and create a heathy environment for cells to survive — therefore a single, one - time product for life can be used to treat all Stargardt disease ( ABCA4 - associated retinopathies) Recent Accomplishments • Positive 6 - month data • ATMP classification from EMA • Receiving RPDD designation in May, IND amendment clearance for registration trial in June Current Status • FDA approved a Phase 2/3 pivotal confirmatory clinical trial for BLA filing • Initiated dosing in July for Phase 2/3 study • Targeting BLA submission by 2027 Stargardt Disease (GARDian3) Clinical Trial 6 • The GARDian3 clinical trial for ABCA4 - related retinopathies including Stargardt disease builds upon encouraging results and positive data from the Phase 1 GARDian trial, which demonstrated 48% slower lesion growth at 12 - month follow - up in evaluable treated eyes compared to untreated eyes • Additionally, evaluable treated eyes showed a statistically significant ( p=0.031 ) and clinically meaningful improvement of nearly 2 - line/9 - letter gain in best corrected visual acuity (BCVA) at 12 - month follow - up when compared to untreated eyes.


Mean Change in Total Atrophy 12M Timeline 48% Unt reated Eye Treated Eye Mean Change in BCVA Improvement: > 5 ETDRS letters ; Stabilization : ± 4 ETDRS letters Data points for M3 (N=6), M6 (N=6); M12 (N=4); *Nearly 9 Letters (Early Treatment Diabetic Retinopathy Scale scale) Two patients with cataract (1 Low, 1 Med), and 1 High dose patient with loss - to - follow up were not included in the analysis OCU410 First - in - Class Novel Modifier Gene Therapy for GA 7 • 2 approved products for GA: FY ‘24 sales SYFOVRE ® (Apellis): $611M (Full Year) IZERVAY (Astellas): $107M • OCU410 has potential to treat 2 - 3 million patients in U.S. and EU • Pricing for current gene therapies ranges between $1 - 4M/patient There remains no approved treatment for GA in Europe ¹DB Rein et al., JAMA Ophthalmol , 2022. doi : 10.1001/jamaophthalmol.2022.4401 ²CJ Thomas et al., Med Clin North Am , 2021. doi : 10.1016/j.mcna.2021.01.003 (2021). Unmet Need ~19 million people in U.S. and EU and 260 million globally suffer from dAMD 1 ~2 - 3 million people in U.S. & EU and ~8 million globally suffer from GA — advanced dAMD 2 Currently, approved products target only one disease pathway associated with GA and address 25% the patient population. These treatments require 6 - 12 injections per year and have associated side effects. Potential Solution OCU410 is designed to regulate all four pathways associated with GA: lipid metabolism, inflammation, oxidative stress, and complement with a single, subretinal injection Current Status of OCU410: • Phase 2 enrollment complete • Planning to initiate Phase 3 in 2026 and targeting BLA submission in 2028 Recent Accomplishments • Phase 1 data at 12 months demonstrated a 23% slower lesion growth in treated eyes in addition to 2 - line (10 - letter) gain in the visual function when compared to untreated eyes • Preliminary data from Phase 2 interim analysis (N=31) at 6M demonstrates 27% slower lesion growth in treated eyes when compared to control untreated eyes GA Lesion Growth after single sub - retinal injection – All Subjects (Phase 1) • 23% slower GA lesion growth in treated eyes versus fellow eyes after single injection Data points at 12M (N=6) GA growth rates per Natural History Studies obtained from Proxima A study 12M 0.0 0.5 1.0 1.5 2.0 M e a n c h a n g e f r o m B a s e l i n e i n G A l e s i o n ( m m 2 ) Treated Eye Untreated Eye 23 % 8


Preservation of Visual Function (LLVA) in OCU410 Treated vs Untreated Eyes (Phase 1) Stabilization : ± 4 letters from Baseline; Improvement: ≥5 Letters from Baseline Data points at 3M (N=8), 6M (N=7), 12M (N=6) BL 3M 6M 12M -25 -20 -15 -10 -5 0 5 10 M e a n c h a n g e i n L L V A ( E T D R S ) f r o m B a s e l i n e Treated Eye Untreated Eye Treated eyes demonstrated improvement compared to untreated in visual function demonstrating treatment benefit Treated eyes demonstrate 2 - line (10 - letter) stabilization/gain in the visual acuity when compared to untreated fellow eyes Meaningful VA decrease Stabilization 9 GA Lesion Growth after single sub - retinal injection: Phase 2 – Preliminary Data 27% slower GA lesion growth in evaluable treated eyes versus fellow eyes after single injection 6M 0.0 0.5 1.0 1.5 M e a n C h a n g e f r o m B a s e l i n e ( ± S E M ) i n G A l e s i o n ( m m 2 ) Control Eyes Treated Eyes GA: geographic atrophy; Data points at 6M , N=9 (Control), N= 22 (Treated) 27 % 10

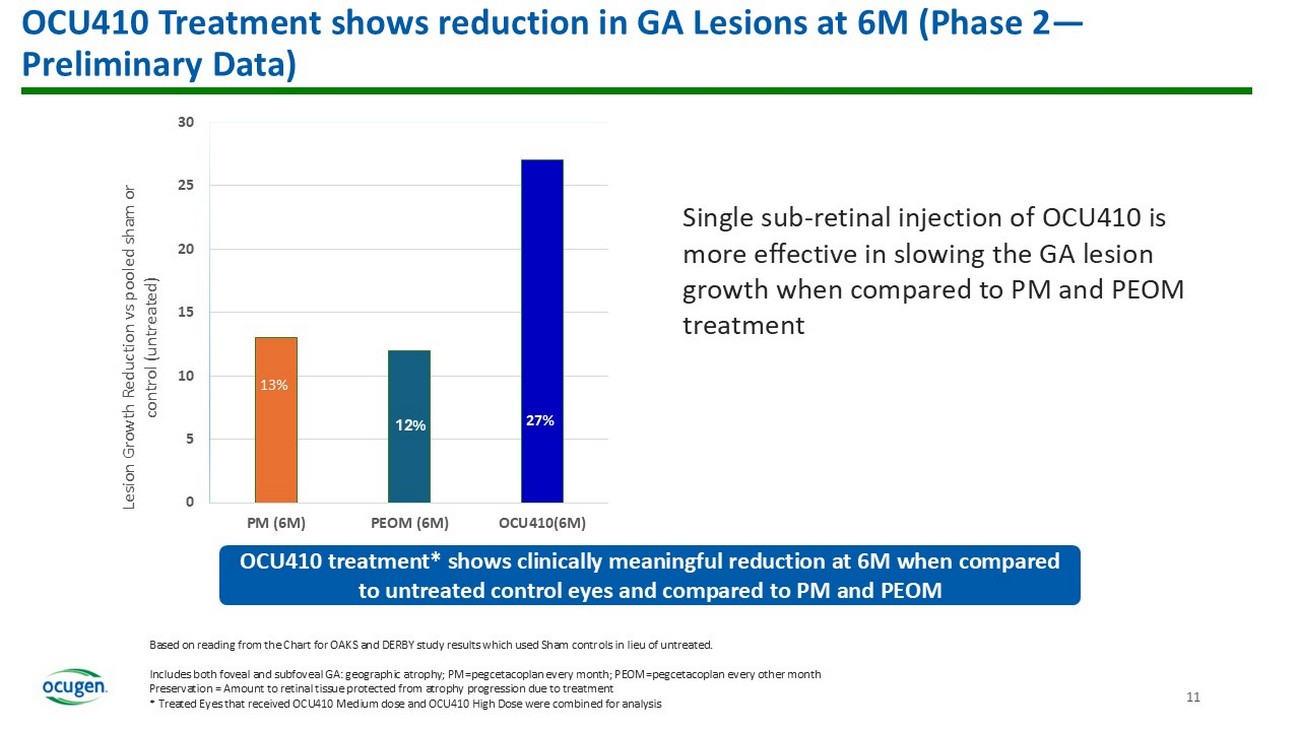
11 13 12% 27% 0 5 10 15 20 25 30 PM (6M) PEOM (6M) OCU410(6M) Lesion Growth Reduction vs pooled sham or control (untreated) OCU410 Treatment shows reduction in GA Lesions at 6M (Phase 2 — Preliminary Data) Based on reading from the Chart for OAKS and DERBY study results which used Sham controls in lieu of untreated. Includes both foveal and subfoveal GA: geographic atrophy; PM= pegcetacoplan every month; PEOM= pegcetacoplan every other month Preservation = Amount to retinal tissue protected from atrophy progression due to treatment * Treated Eyes that received OCU410 Medium dose and OCU410 High Dose were combined for analysis Single sub - retinal injection of OCU410 is more effective in slowing the GA lesion growth when compared to PM and PEOM treatment OCU410 treatment* shows clinically meaningful reduction at 6M when compared to untreated control eyes and compared to PM and PEOM 13% 12 0.14 0.12 0.31 0 0.05 0.1 0.15 0.2 0.25 0.3 0.35 PM (6M) PEOM (6M) OCU410(6M) Difference in Mean GA lesion Size between Treated and Untreated Eyes (mm 2 ) OCU410 Treatment Preserves Retinal Tissue in GA Lesions (Phase 2 – Preliminary Data) Based on reading from the Chart for OAKS and DERBY study results which used Sham controls in lieu of untreated.
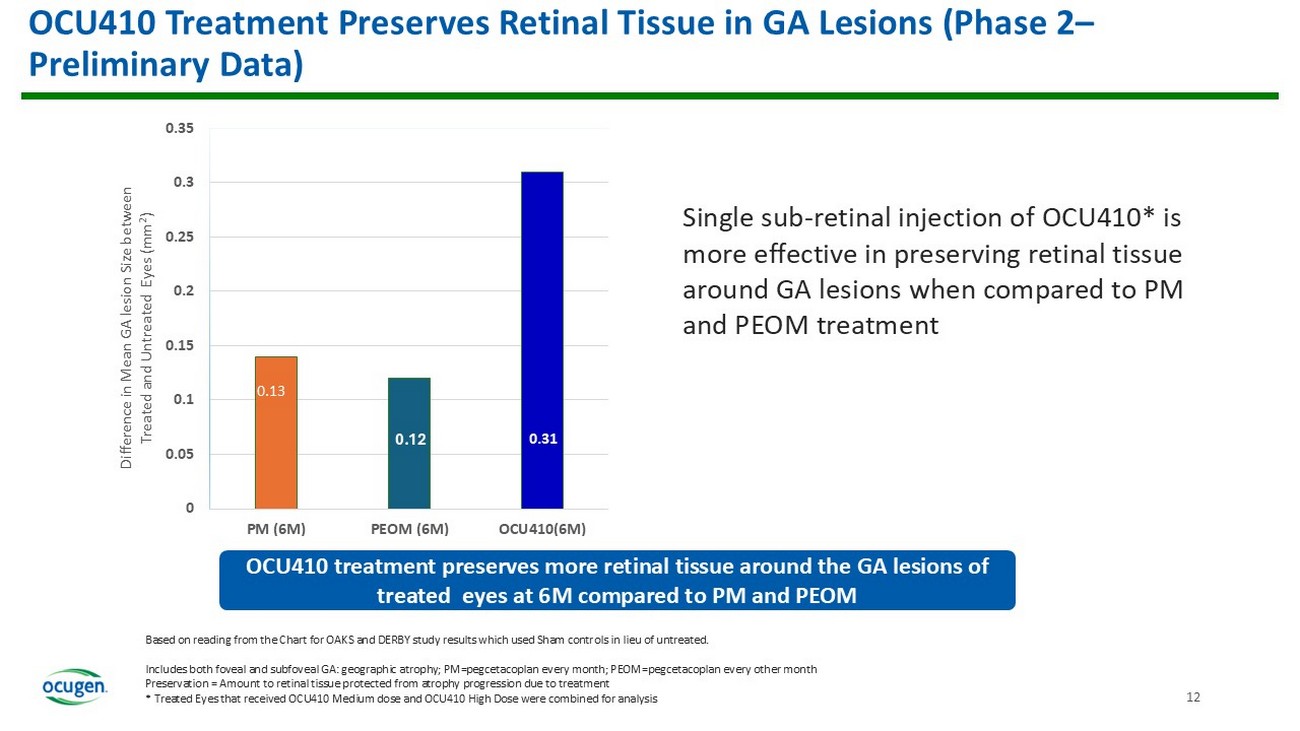

Includes both foveal and subfoveal GA: geographic atrophy; PM= pegcetacoplan every month; PEOM= pegcetacoplan every other month Preservation = Amount to retinal tissue protected from atrophy progression due to treatment * Treated Eyes that received OCU410 Medium dose and OCU410 High Dose were combined for analysis Single sub - retinal injection of OCU410* is more effective in preserving retinal tissue around GA lesions when compared to PM and PEOM treatment OCU410 treatment preserves more retinal tissue around the GA lesions of treated eyes at 6M compared to PM and PEOM 0.13 Binding Term Sheet Signed for our First Regional Partnership for OCU400 and Announced the spin - off of NeoCart into OrthoCellix 13 OCU400 • Company signed a term sheet to negotiate and enter into a licensing agreement with a well - established leader in the pharmaceutical and healthcare sector in Korea • We are in conversations to explore a variety of partnering opportunities for OCU400 and all our gene therapy candidates NeoCart • The planned reverse merger between OrthoCellix (a subsidiary) and Carisma will form a Nasdaq - listed company focused on advanced regenerative cell therapies, using a first - of - its - kind technology • This strategic move is intended to create value for Ocugen stockholders without dilution of Ocugen stock, as OrthoCellix is planned to be funded with $25 million in private financing • Under the Exchange Ratio formula in the Merger Agreement OrthoCellix’s value is estimated at $135 million New Appointments to Board of Directors, Retina SAB & Executive Team 14


Financial Update 15
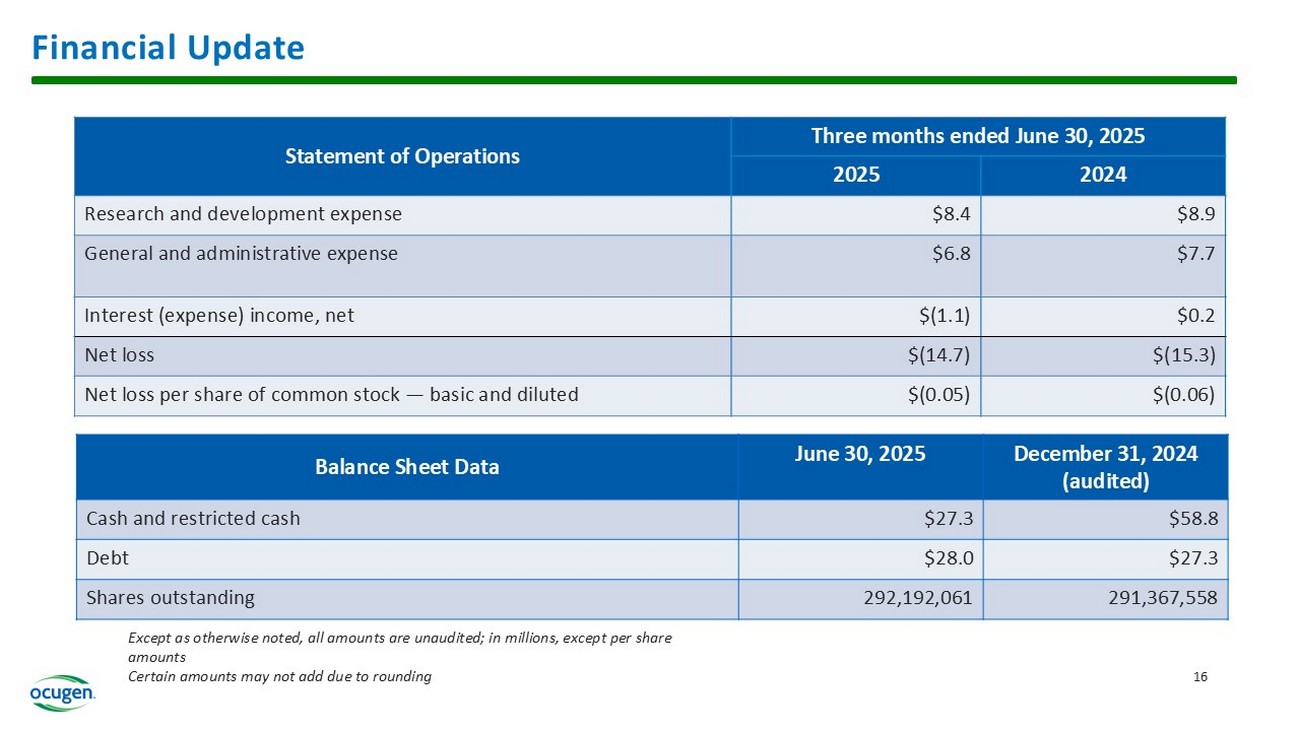
Financial Update Three months ended June 30, 2025 Statement of Operations 2024 2025 $8.9 $8.4 Research and development expense $7.7 $6.8 General and administrative expense $0.2 $(1.1) Interest (expense) income, net $( 15.3) $(14.7) Net loss $(0.06) $(0.05) Net loss per share of common stock — basic and diluted December 31, 2024 (audited) June 30, 2025 Balance Sheet Data $58.8 $27.3 Cash and restricted cash $27.3 $28.0 Debt 291,367,558 292,192,061 Shares outstanding 16 Except as otherwise noted, all amounts are unaudited; in millions, except per share amounts Certain amounts may not add due to rounding 18 Ocugen Vision Fully integrated, patient - centric b iotech company focused on vaccines in support of public health and gene and cell therapies targeting unmet medical needs through Courageous Innovation

Questions & Answers 17
