UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE ISSUER PURSUANT TO RULE 13a-16 OR 15d-16 UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the month of November 2024
Commission File Number: 001-41937
Psyence Biomedical Ltd.
(Translation of registrant’s name into English)
121 Richmond Street West
Penthouse Suite 1300
Toronto, Ontario M5H 2K1
(Address of principal executive office)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F.
x Form 20-F ¨ Form 40-F
EXHIBIT INDEX
| 99.3 | Investor Presentation, dated November 2024. |
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
Dated: November 22, 2024
| Psyence Biomedical Ltd. | ||
| By: | /s/ Dr. Neil Maresky | |
| Name: | Dr. Neil Maresky | |
| Title: | Chief Executive Officer and Director | |
Exhibit 4.1
NEITHER THIS SECURITY NOR THE SECURITIES FOR WHICH THIS SECURITY IS EXERCISABLE HAVE BEEN REGISTERED WITH THE SECURITIES AND EXCHANGE COMMISSION OR THE SECURITIES COMMISSION OF ANY STATE IN RELIANCE UPON AN EXEMPTION FROM REGISTRATION UNDER THE SECURITIES ACT OF 1933, AS AMENDED (THE “SECURITIES ACT”), AND, ACCORDINGLY, MAY NOT BE OFFERED OR SOLD EXCEPT PURSUANT TO AN EFFECTIVE REGISTRATION STATEMENT UNDER THE SECURITIES ACT OR PURSUANT TO AN AVAILABLE EXEMPTION FROM, OR IN A TRANSACTION NOT SUBJECT TO, THE REGISTRATION REQUIREMENTS OF THE SECURITIES ACT AND IN ACCORDANCE WITH APPLICABLE STATE SECURITIES LAWS. THIS SECURITY AND THE SECURITIES ISSUABLE UPON EXERCISE OF THIS SECURITY MAY BE PLEDGED IN CONNECTION WITH A BONA FIDE MARGIN ACCOUNT OR OTHER LOAN SECURED BY SUCH SECURITIES.
UNLESS PERMITTED UNDER SECURITIES LEGISLATION, THE HOLDER OF THIS SECURITY MUST NOT TRADE THE SECURITY BEFORE THE DATE THAT IS 4 MONTHS AND A DAY AFTER THE LATER OF (I) ____________, 202_ AND (II) THE DATE THE ISSUER BECOMES A REPORTING ISSUER IN ANY PROVINCE OR TERRITORY.
COMMON SHARE PURCHASE WARRANT
PSYENCE BIOMEDICAL LTD.
| Warrant Shares:______________________ | Issue Date: ___________, 202__ |
THIS COMMON SHARE PURCHASE WARRANT (the “Warrant”) certifies that, for value received, or its assigns (the “Holder”) is entitled, upon the terms and subject to the limitations on exercise and the conditions hereinafter set forth, at any time on or after the date set forth above (the “Initial Exercise Date”) and on or prior to 5:00 p.m. (New York City time) on _____________, 202_1(the “Termination Date”) but not thereafter, to subscribe for and purchase from Psyence Biomedical Ltd., a corporation existing under the laws of Ontario, Canada (the “Company”), up to ______________ (_______) of the Company’s Common Shares (as subject to adjustment hereunder, the “Warrant Shares”). The purchase price of one Common Share under this Warrant shall be equal to the Exercise Price, as defined in Section 2(b).
Section 1. Definitions. Capitalized terms used and not otherwise defined herein shall have the meanings set forth in that certain Securities Purchase Agreement (the “Purchase Agreement”), dated January 15, 2024, as amended by that certain First Addendum to the Securities Purchase Agreement and Senior Secured Convertible Notes, dated as of August 19, 2024, among the Company and the purchasers signatory thereto. Reference is hereby made to the Termination Agreement entered into by and among the Company and [the Holder], entered into on or about the date hereof.
Section 2. Exercise.
| (a) | Exercise of Warrant. Exercise of the purchase rights represented by this Warrant may be made, in whole or in part, at any time or times on or after the Initial Exercise Date and on or before the Termination Date by delivery to the Company of a duly executed PDF copy submitted by e-mail (or e-mail attachment) of the Notice of Exercise in the form annexed hereto (the “Notice of Exercise”). Within the earlier of (i) one (1) Trading Day and (ii) the number of Trading Days comprising the Standard Settlement Period (as defined in Section 2(d)(i) herein) following the date of exercise as aforesaid, the Holder shall deliver the aggregate Exercise Price for the Warrant Shares specified in the applicable Notice of Exercise by wire transfer or cashier’s check drawn on a United States bank, in either case in immediately available funds. Unless required by the Company’s transfer agent, no ink-original Notice of Exercise shall be required, nor shall any medallion guarantee (or other type of guarantee or notarization) of any Notice of Exercise be required. The Company shall have no obligation to inquire with respect to or otherwise confirm the authenticity of the signature(s) contained on any Notice of Exercise nor the authority of the person so executing such Notice of Exercise. Notwithstanding anything herein to the contrary, the Holder shall not be required to physically surrender this Warrant to the Company until the Holder has purchased all of the Warrant Shares available hereunder and the Warrant has been exercised in full, in which case, the Holder shall surrender this Warrant to the Company for cancellation within three (3) Trading Days of the date on which the final Notice of Exercise is delivered to the Company. Partial exercises of this Warrant resulting in purchases of a portion of the total number of Warrant Shares available hereunder shall have the effect of lowering the outstanding number of Warrant Shares purchasable hereunder in an amount equal to the applicable number of Warrant Shares purchased. The Holder and the Company shall maintain records showing the number of Warrant Shares purchased and the date of such purchases. The Company shall deliver any objection to any Notice of Exercise within one (1) Trading Day of receipt of such notice. The Holder and any assignee, by acceptance of this Warrant, acknowledge and agree that, by reason of the provisions of this paragraph, following the purchase of a portion of the Warrant Shares hereunder, the number of Warrant Shares available for purchase hereunder at any given time may be less than the amount stated on the face hereof. |
1 To be the date that is 12 months following the Issue Date.
a) Exercise Price. The exercise price per Common Share under this Warrant shall be $0.0000001, subject to adjustment hereunder (the “Exercise Price”).
b) Mechanics of Exercise.
i. Delivery of Warrant Shares Upon Exercise. The Company shall cause the Warrant Shares purchased hereunder to be transmitted by the Transfer Agent to the Holder by crediting the account of the Holder’s or its designee’s balance account with The Depository Trust Company through its Deposit or Withdrawal at Custodian system (“DWAC”) if the Company is then a participant in such system and either (A) there is an effective registration statement permitting the issuance of the Warrant Shares to or resale of the Warrant Shares by the Holder or (B) the Warrant Shares are eligible for resale by the Holder without volume or manner-of-sale limitations pursuant to Rule 144, and otherwise by physical delivery of a certificate or a book-entry certificate, registered in the Company’s share register in the name of the Holder or its designee, for the number of Warrant Shares to which the Holder is entitled pursuant to such exercise to the address specified by the Holder in the Notice of Exercise by the date that is the earlier of (i) one (1) Trading Day and (ii) the number of Trading Days comprising the Standard Settlement Period, in each case after the delivery to the Company of the Notice of Exercise and, if applicable, payment of the aggregate Exercise Price (such date, the “Warrant Share Delivery Date”). Upon delivery of the Notice of Exercise, the Holder shall be deemed for all corporate purposes to have become the holder of record of the Warrant Shares with respect to which this Warrant has been exercised, irrespective of the date of delivery of the Warrant Shares, provided that payment of the aggregate Exercise Price is received by the Warrant Share Delivery Date. The Company agrees to maintain a transfer agent that is a participant in the FAST program so long as this Warrant remains outstanding and exercisable. As used herein, “Standard Settlement Period” means the standard settlement period, expressed in a number of Trading Days, on the Company’s primary Trading Market with respect to the Common Shares as in effect on the date of delivery of the Notice of Exercise.
ii. Delivery of New Warrants Upon Exercise. If this Warrant shall have been exercised in part, the Company shall, at the request of a Holder and upon surrender of this Warrant certificate, at the time of delivery of the Warrant Shares, deliver to the Holder a new Warrant evidencing the rights of the Holder to purchase the unpurchased Warrant Shares called for by this Warrant, which new Warrant shall in all other respects be identical with this Warrant.
iii. Rescission Rights. If the Company fails to cause the Transfer Agent to transmit to the Holder the Warrant Shares pursuant to Section 2(d) (i) by the Warrant Share Delivery Date, then the Holder will have the right to rescind such exercise by delivering written notice to the Company at any time prior to the delivery of such Warrant Shares.
iv. Compensation for Buy-In on Failure to Timely Deliver Warrant Shares Upon Exercise. In addition to any other rights available to the Holder, if the Company fails to cause the Transfer Agent to transmit to the Holder the Warrant Shares in accordance with the provisions of Section 2(d) (i) above pursuant to an exercise on or before the Warrant Share Delivery Date, and if after such date the Holder is required by its broker to purchase (in an open market transaction or otherwise) or the Holder’s brokerage firm otherwise purchases, Common Shares to deliver in satisfaction of a sale by the Holder of the Warrant Shares which the Holder anticipated receiving upon such exercise (a “Buy-In”), then the Company shall (A) pay in cash to the Holder the amount, if any, by which (x) the Holder’s total purchase price (including reasonable and customary brokerage commissions, if any) for the Common Shares so purchased exceeds (y) the amount obtained by multiplying (1) the number of Warrant Shares that the Company was required to deliver to the Holder in connection with the exercise at issue times (2) the price at which the sell order giving rise to such purchase obligation was executed, and (B) at the option of the Holder, either reinstate the portion of the Warrant and equivalent number of Warrant Shares for which such exercise was not honored (in which case such exercise shall be deemed rescinded) or deliver to the Holder the number of Common Shares that would have been issued had the Company timely complied with its exercise and delivery obligations hereunder. For example, if the Holder purchases Common Shares having a total purchase price of $11,000 to cover a Buy-In with respect to an attempted exercise of Common Shares with an aggregate sale price giving rise to such purchase obligation of $10,000, under clause (A) of the immediately preceding sentence the Company shall be required to pay the Holder $1,000. The Holder shall provide the Company written notice indicating the amounts payable to the Holder in respect of the Buy-In and, upon request of the Company, evidence of the amount of such loss. Nothing herein shall limit a Holder’s right to pursue any other remedies available to it hereunder, at law or in equity including, without limitation, a decree of specific performance and/or injunctive relief with respect to the Company’s failure to timely deliver Common Shares upon exercise of the Warrant as required pursuant to the terms hereof.
v. No Fractional Shares or Scrip. No fractional shares or scrip representing fractional shares shall be issued upon the exercise of this Warrant. As to any fraction of a share which the Holder would otherwise be entitled to purchase upon such exercise, the Company shall, at its election, either pay a cash adjustment in respect of such final fraction in an amount equal to such fraction multiplied by the Exercise Price or round up to the next whole share.
vi. Charges, Taxes and Expenses. Issuance of Warrant Shares shall be made without charge to the Holder for any issue or transfer tax or other incidental expense in respect of the issuance of such Warrant Shares, all of which taxes and expenses shall be paid by the Company, and such Warrant Shares shall be issued in the name of the Holder or in such name or names as may be directed by the Holder; provided, however, that in the event that Warrant Shares are to be issued in a name other than the name of the Holder, this Warrant when surrendered for exercise shall be accompanied by the Assignment Form attached hereto duly executed by the Holder and the Company may require, as a condition thereto, the payment of a sum sufficient to reimburse it for any transfer tax incidental thereto. The Company shall pay all Transfer Agent fees required for same-day processing of any Notice of Exercise and all fees to the Depository Trust Company (or another established clearing corporation performing similar functions) required for same-day electronic delivery of the Warrant Shares.
vii. Closing of Books. The Company will not close its shareholder books or records in any manner which prevents the timely exercise of this Warrant, pursuant to the terms hereof.
c) Holder’s Exercise Limitations. The Company shall not effect any exercise of this Warrant, and a Holder shall not have the right to exercise any portion of this Warrant, pursuant to Section 2 or otherwise, to the extent that after giving effect to such issuance after exercise as set forth on the applicable Notice of Exercise, the Holder (together with the Holder’s Affiliates, and any other Persons acting as a group together with the Holder or any of the Holder’s Affiliates (such Persons, “Attribution Parties”)), would beneficially own in excess of the Beneficial Ownership Limitation (as defined below). For purposes of the foregoing sentence, the number of Common Shares beneficially owned by the Holder and its Affiliates and Attribution Parties shall include the number of Common Shares issuable upon exercise of this Warrant with respect to which such determination is being made, but shall exclude the number of Common Shares which would be issuable upon (i) exercise of the remaining, nonexercised portion of this Warrant beneficially owned by the Holder or any of its Affiliates or Attribution Parties and (ii) exercise or conversion of the unexercised or nonconverted portion of any other securities of the Company (including, without limitation, any other Common Share Equivalents) subject to a limitation on conversion or exercise analogous to the limitation contained herein beneficially owned by the Holder or any of its Affiliates or Attribution Parties. Except as set forth in the preceding sentence, for purposes of this Section 2(e), beneficial ownership shall be calculated in accordance with Section 13(d) of the Exchange Act and the rules and regulations promulgated thereunder, it being acknowledged by the Holder that the Company is not representing to the Holder that such calculation is in compliance with Section 13(d) of the Exchange Act and the Holder is solely responsible for any schedules required to be filed in accordance therewith and the calculations required under this Section 2(e). To the extent that the limitation contained in this Section 2(e) applies, the determination of whether this Warrant is exercisable (in relation to other securities owned by the Holder together with any Affiliates and Attribution Parties) and of which portion of this Warrant is exercisable shall be in the sole discretion of the Holder, and the submission of a Notice of Exercise shall be deemed to be the Holder’s determination of whether this Warrant is exercisable (in relation to other securities owned by the Holder together with any Affiliates and Attribution Parties) and of which portion of this Warrant is exercisable, in each case subject to the Beneficial Ownership Limitation, and the Company shall have no obligation to verify or confirm the accuracy of such determination. In addition, a determination as to any group status as contemplated above shall be determined in accordance with Section 13(d) of the Exchange Act and the rules and regulations promulgated thereunder. For purposes of this Section 2(e), in determining the number of outstanding Common Shares, a Holder may rely on the number of outstanding Common Shares as reflected in (A) the Company’s most recent periodic or annual report filed with the Commission, as the case may be, (B) a more recent public announcement by the Company or (C) a more recent written notice by the Company or the Transfer Agent setting forth the number of Common Shares outstanding. Upon the written or oral request of a Holder, the Company shall within one (1) Trading Day confirm orally and in writing to the Holder the number of Common Shares then outstanding. In any case, the number of outstanding Common Shares shall be determined after giving effect to the conversion or exercise of securities of the Company, including this Warrant, by the Holder or its Affiliates or Attribution Parties since the date as of which such number of outstanding Common Shares was reported. The “Beneficial Ownership Limitation” shall be 9.99% of the number of Common Shares outstanding immediately after giving effect to the issuance of Common Shares issuable upon exercise of this Warrant. The Holder, upon notice to the Company, may increase or decrease the Beneficial Ownership Limitation provisions of this Section 2(e), provided that the Beneficial Ownership Limitation in no event exceeds 9.99% of the number of Common Shares outstanding immediately after giving effect to the issuance of Common Shares upon exercise of this Warrant held by the Holder and the provisions of this Section 2(e) shall continue to apply. Any increase in the Beneficial Ownership Limitation will not be effective until the 61st day after such notice is delivered to the Company. The provisions of this paragraph shall be construed and implemented in a manner otherwise than in strict conformity with the terms of this Section 2(e) to correct this paragraph (or any portion hereof) which may be defective or inconsistent with the intended Beneficial Ownership Limitation herein contained or to make changes or supplements necessary or desirable to properly give effect to such limitation. The limitations contained in this paragraph shall apply to a successor holder of this Warrant.
Section 3. Certain Adjustments.
a) Share Dividends and Splits. If the Company, at any time while this Warrant is outstanding: (i) pays a share dividend or otherwise makes a distribution or distributions on its Common Shares or any other equity or equity equivalent securities payable in Common Shares (which, for avoidance of doubt, shall not include any Common Shares issued by the Company upon exercise of this Warrant), (ii) subdivides outstanding Common Shares into a larger number of shares, (iii) combines (including by way of reverse share split) outstanding Common Shares into a smaller number of shares, or (iv) issues by reclassification of the Common Shares any capital shares of the Company, then in each case the Exercise Price shall be multiplied by a fraction of which the numerator shall be the number of Common Shares (excluding treasury shares, if any) outstanding immediately before such event and of which the denominator shall be the number of Common Shares outstanding immediately after such event, and the number of shares issuable upon exercise of this Warrant shall be proportionately adjusted such that the aggregate Exercise Price of this Warrant shall remain unchanged. Any adjustment made pursuant to this Section 3(a) shall become effective immediately after the record date for the determination of shareholders entitled to receive such dividend or distribution and shall become effective immediately after the effective date in the case of a subdivision, combination or re- classification.
b) Fundamental Transaction. If, at any time while this Warrant is outstanding, (i) the Company, directly or indirectly, in one or more related transactions effects any merger or consolidation of the Company with or into another Person (other than for the purpose of changing the Company’s name and/or the jurisdiction of incorporation of the Company or a holding company for the Company), (ii) the Company (or any Subsidiary), directly or indirectly, effects any sale, lease, license, assignment, transfer, conveyance or other disposition of all or substantially all of the assets of the Company in one or a series of related transactions, (iii) any, direct or indirect, purchase offer, tender offer or exchange offer (whether by the Company or another Person) is completed pursuant to which holders of Common Shares are permitted to sell, tender or exchange their shares for other securities, cash or property and has been accepted by the holders of 50% or more of the outstanding Common Shares or 50% or more of the voting power of the common equity of the Company, (iv) the Company, directly or indirectly, in one or more related transactions effects any reclassification, reorganization or recapitalization of the Common Shares or any compulsory share exchange pursuant to which the Common Shares are effectively converted into or exchanged for other securities, cash or property, or (v) the Company, directly or indirectly, in one or more related transactions consummates a stock or share purchase agreement or other business combination (including, without limitation, a reorganization, recapitalization, spin-off, merger or scheme of arrangement) with another Person or group of Persons whereby such other Person or group acquires 50% or more of the outstanding Common Shares or 50% or more of the voting power of the common equity of the Company (each a “Fundamental Transaction”), then, upon any subsequent exercise of this Warrant, the Holder shall have the right to receive, for each Warrant Share that would have been issuable upon such exercise immediately prior to the occurrence of such Fundamental Transaction, at the option of the Holder (without regard to any limitation in Section 2(e) on the exercise of this Warrant), the number of Common Shares of the successor or acquiring corporation or of the Company, if it is the surviving corporation, and any additional consideration (the “Alternate Consideration”) receivable as a result of such Fundamental Transaction by a holder of the number of Common Shares for which this Warrant is exercisable immediately prior to such Fundamental Transaction (without regard to any limitation in Section 2(e) on the exercise of this Warrant). For purposes of any such exercise, the determination of the Exercise Price shall be appropriately adjusted to apply to such Alternate Consideration based on the amount of Alternate Consideration issuable in respect of one Common Share in such Fundamental Transaction, and the Company shall apportion the Exercise Price among the Alternate Consideration in a reasonable manner reflecting the relative value of any different components of the Alternate Consideration. If holders of Common Shares are given any choice as to the securities, cash or property to be received in a Fundamental Transaction, then the Holder shall be given the same choice as to the Alternate Consideration it receives upon any exercise of this Warrant following such Fundamental Transaction. The Company shall cause any successor entity in a Fundamental Transaction in which the Company is not the survivor (the “Successor Entity”) to assume in writing all of the obligations of the Company under this Warrant in accordance with the provisions of this Section 3(d) pursuant to written agreements in form and substance reasonably satisfactory to the Holder and approved by the Holder (without unreasonable delay) prior to such Fundamental Transaction and shall, at the option of the Holder, deliver to the Holder in exchange for this Warrant a security of the Successor Entity evidenced by a written instrument substantially similar in form and substance to this Warrant which is exercisable for a corresponding number of capital shares of such Successor Entity (or its parent entity) equivalent to the Common Shares acquirable and receivable upon exercise of this Warrant (without regard to any limitations on the exercise of this Warrant) prior to such Fundamental Transaction, and with an exercise price which applies the exercise price hereunder to such capital shares (but taking into account the relative value of the Common Shares pursuant to such Fundamental Transaction and the value of such capital shares, such number of capital shares and such exercise price being for the purpose of protecting the economic value of this Warrant immediately prior to the consummation of such Fundamental Transaction), and which is reasonably satisfactory in form and substance to the Holder. Upon the occurrence of any such Fundamental Transaction, the Successor Entity shall be added to the term “Company” under this Warrant (so that from and after the occurrence or consummation of such Fundamental Transaction, each and every provision of this Warrant referring to the “Company” shall refer instead to each of the Company and the Successor Entity or Successor Entities, jointly and severally), and the Successor Entity or Successor Entities, jointly and severally with the Company, may exercise every right and power of the Company prior thereto and the Successor Entity or Successor Entities shall assume all of the obligations of the Company prior thereto under this Warrant with the same effect as if the Company and such Successor Entity or Successor Entities, jointly and severally, had been named as the Company herein.
c) Calculations. All calculations under this Section 3 shall be made to the nearest cent or the nearest 1/100th of a share, as the case may be. For purposes of this Section 3, the number of Common Shares deemed to be issued and outstanding as of a given date shall be the sum of the number of Common Shares (excluding treasury shares, if any) issued and outstanding.
d) Notice to Holder.
i. Adjustment to Exercise Price. Whenever the Exercise Price is adjusted pursuant to any provision of this Section 3, the Company shall promptly deliver to the Holder by email a notice setting forth the Exercise Price after such adjustment and any resulting adjustment to the number of Warrant Shares and setting forth a brief statement of the facts requiring such adjustment.
ii. Notice to Allow Exercise by Holder. If (A) the Company shall declare a dividend (or any other distribution in whatever form) on the Common Shares, (B) the Company shall declare a special nonrecurring cash dividend on or a redemption of the Common Shares, (C) the Company shall authorize the granting to all holders of the Common Shares rights or warrants to subscribe for or purchase any capital shares of any class or of any rights, (D) the approval of any shareholders of the Company shall be required in connection with any reclassification of the Common Shares, any consolidation or merger to which the Company (or any of its Subsidiaries) is a party, any sale or transfer of all or substantially all of its assets, or any compulsory share exchange whereby the Common Shares are converted into other securities, cash or property, or (E) the Company shall authorize the voluntary or involuntary dissolution, liquidation or winding up of the affairs of the Company, then, in each case, the Company shall cause to be delivered by email to the Holder at its last email address as it shall appear upon the Warrant Register of the Company, at least twenty (20) calendar days prior to the applicable record or effective date hereinafter specified, a notice stating (x) the date on which a record is to be taken for the purpose of such dividend, distribution, redemption, rights or warrants, or if a record is not to be taken, the date as of which the holders of the Common Shares of record to be entitled to such dividend, distributions, redemption, rights or warrants are to be determined or (y) the date on which such reclassification, consolidation, merger, sale, transfer or share exchange is expected to become effective or close, and the date as of which it is expected that holders of the Common Shares of record shall be entitled to exchange their Common Shares for securities, cash or other property deliverable upon such reclassification, consolidation, merger, sale, transfer or share exchange; provided that the failure to deliver such notice or any defect therein or in the delivery thereof shall not affect the validity of the corporate action required to be specified in such notice. To the extent that any notice provided in this Warrant constitutes, or contains, material, non-public information regarding the Company or any of the Subsidiaries, the Company shall simultaneously file such notice with the Commission pursuant to a Report of Foreign Private Issuer on Form 6-K. The Holder shall remain entitled to exercise this Warrant during the period commencing on the date of such notice to the effective date of the event triggering such notice except as may otherwise be expressly set forth herein.
Section 4. Transfer of Warrant.
a) Transferability. Subject to compliance with any applicable securities laws, this Warrant and all rights hereunder (including, without limitation, any registration rights) are transferable, in whole or in part, upon surrender of this Warrant at the principal office of the Company or its designated agent, together with a written assignment of this Warrant substantially in the form attached hereto duly executed by the Holder or its agent or attorney and funds sufficient to pay any transfer taxes payable upon the making of such transfer. Upon such surrender and, if required, such payment, the Company shall execute and deliver a new Warrant or Warrants in the name of the assignee or assignees, as applicable, and in the denomination or denominations specified in such instrument of assignment, and shall issue to the assignor a new Warrant evidencing the portion of this Warrant not so assigned, and this Warrant shall promptly be cancelled. Notwithstanding anything herein to the contrary, the Holder shall not be required to physically surrender this Warrant to the Company unless the Holder has assigned this Warrant in full, in which case, the Holder shall surrender this Warrant to the Company within three (3) Trading Days of the date on which the Holder delivers an assignment form to the Company assigning this Warrant in full. The Warrant, if properly assigned in accordance herewith, may be exercised by a new holder for the purchase of Warrant Shares without having a new Warrant issued.
b) New Warrants. This Warrant may be divided or combined with other Warrants upon presentation hereof at the aforesaid office of the Company, together with a written notice specifying the names and denominations in which new Warrants are to be issued, signed by the Holder or its agent or attorney. Subject to compliance with Section 4(a), as to any transfer which may be involved in such division or combination, the Company shall execute and deliver a new Warrant or Warrants in exchange for the Warrant or Warrants to be divided or combined in accordance with such notice. All Warrants issued on transfers or exchanges shall be dated the initial issuance date of this Warrant and shall be identical with this Warrant except as to the number of Warrant Shares issuable pursuant thereto.
c) Warrant Register. The Company shall register this Warrant, upon records to be maintained by the Company for that purpose (the “Warrant Register”), in the name of the record Holder hereof from time to time. The Company may deem and treat the registered Holder of this Warrant as the absolute owner hereof for the purpose of any exercise hereof or any distribution to the Holder, and for all other purposes, absent actual notice to the contrary.
d) Representation by the Holder. The Holder, by the acceptance hereof, represents and warrants that it is acquiring this Warrant and, upon any exercise hereof, will acquire the Warrant Shares issuable upon such exercise, for its own account and not with a view to or for distributing or reselling such Warrants or Warrant Shares or any part thereof in violation of the Securities Act or any applicable state securities law or Canadian Securities Laws, except pursuant to sales registered or exempted under the Securities Act, Canadian Securities Laws, in compliance with the Purchase Agreement.
Section 5. Miscellaneous.
a) No Rights as Shareholder Until Exercise; No Settlement in Cash. This Warrant does not entitle the Holder to any voting rights, dividends or other rights as a shareholder of the Company prior to the exercise hereof as set forth in Section 2(d)(i), except as expressly set forth in Section 3. Without limiting any rights of a Holder to receive cash payments pursuant to Section 2(d)(i) and Section 2(d)(iv) herein, in no event shall the Company be required to net cash settle an exercise of this Warrant.
b) Loss, Theft, Destruction or Mutilation of Warrant. The Company covenants that upon receipt by the Company of evidence reasonably satisfactory to it of the loss, theft, destruction or mutilation of this Warrant or any share certificate relating to the Warrant Shares, and in case of loss, theft or destruction, of indemnity or security reasonably satisfactory to it (which, in the case of the Warrant, shall not include the posting of any bond), and upon surrender and cancellation of such Warrant or share certificate, if mutilated, the Company will make and deliver a new Warrant or share certificate of like tenor and dated as of such cancellation, in lieu of such Warrant or share certificate.
c) Saturdays, Sundays, Holidays, etc. If the last or appointed day for the taking of any action or the expiration of any right required or granted herein shall not be a Trading Day, then, such action may be taken or such right may be exercised on the next succeeding Trading Day.
d) Authorized Shares.
The Company covenants that, during the period the Warrant is outstanding, it will reserve from its authorized and unissued Common Shares a sufficient number of shares to provide for the issuance of the Warrant Shares upon the exercise of any purchase rights under this Warrant. The Company further covenants that its issuance of this Warrant shall constitute full authority to its officers who are charged with the duty of issuing the necessary Warrant Shares upon the exercise of the purchase rights under this Warrant. The Company will take all such reasonable action as may be necessary to assure that such Warrant Shares may be issued as provided herein without violation of any applicable law or regulation, or of any requirements of the Trading Market upon which the Common Shares may be listed. The Company covenants that all Warrant Shares which may be issued upon the exercise of the purchase rights represented by this Warrant will, upon exercise of the purchase rights represented by this Warrant and payment for such Warrant Shares in accordance herewith, be duly authorized, validly issued, fully paid and nonassessable and free from all taxes, liens and charges created by the Company in respect of the issue thereof (other than taxes in respect of any transfer occurring contemporaneously with such issue).
Except and to the extent as waived or consented to by the Holder, the Company shall not by any action, including, without limitation, amending its certificate of incorporation or through any reorganization, transfer of assets, consolidation, merger, dissolution, issue or sale of securities or any other voluntary action, avoid or seek to avoid the observance or performance of any of the terms of this Warrant, but will at all times in good faith assist in the carrying out of all such terms and in the taking of all such actions as may be necessary or appropriate to protect the rights of Holder as set forth in this Warrant against impairment. Without limiting the generality of the foregoing, the Company will (i) not increase the par value, if any, of any Warrant Shares above the amount payable therefor upon such exercise immediately prior to such increase in par value, (ii) take all such action as may be necessary or appropriate in order that the Company may validly and legally issue fully paid and nonassessable Warrant Shares upon the exercise of this Warrant and (iii) use commercially reasonable efforts to obtain all such authorizations, exemptions or consents from any public regulatory body having jurisdiction thereof, as may be, necessary to enable the Company to perform its obligations under this Warrant.
Before taking any action which would result in an adjustment in the number of Warrant Shares for which this Warrant is exercisable or in the Exercise Price, the Company shall obtain all such authorizations or exemptions thereof, or consents thereto, as may be necessary from any public regulatory body or bodies having jurisdiction thereof.
e) Jurisdiction. All questions concerning the construction, validity, enforcement and interpretation of this Warrant shall be determined in accordance with the provisions of the Purchase Agreement.
f) Restrictions. The Holder acknowledges that the Warrant Shares acquired upon the exercise of this Warrant, if not registered, will have restrictions upon resale imposed by state and federal securities laws. The Warrant Shares acquired upon exercise of this Warrant will also be subject to restrictions upon resale imposed by Canadian Securities Laws.
g) Nonwaiver and Expenses. No course of dealing or any delay or failure to exercise any right hereunder on the part of Holder shall operate as a waiver of such right or otherwise prejudice the Holder’s rights, powers or remedies, notwithstanding the fact that the right to exercise this Warrant terminates on the Termination Date. Without limiting any other provision of this Warrant, if the Company willfully and knowingly fails to comply with any provision of this Warrant, which results in any material damages to the Holder, the Company shall pay to the Holder such amounts as shall be sufficient to cover any costs and expenses including, but not limited to, reasonable attorneys’ fees, including those of appellate proceedings, incurred by the Holder in collecting any amounts due pursuant hereto or in otherwise enforcing any of its rights, powers or remedies hereunder.
h) Notices. Any notice, request or other document required or permitted to be given or delivered to the Holder by the Company shall be delivered in accordance with the notice provisions of the Purchase Agreement.
i) Limitation of Liability. No provision hereof, in the absence of any affirmative action by the Holder to exercise this Warrant to purchase Warrant Shares, and no enumeration herein of the rights or privileges of the Holder, shall give rise to any liability of the Holder for the purchase price of any Common Shares or as a shareholder of the Company, whether such liability is asserted by the Company or by creditors of the Company.
j) Remedies. The Holder, in addition to being entitled to exercise all rights granted by law, including recovery of damages, will be entitled to specific performance of its rights under this Warrant. The Company agrees that monetary damages would not be adequate compensation for any loss incurred by reason of a breach by it of the provisions of this Warrant and hereby agrees to waive and not to assert the defense in any action for specific performance that a remedy at law would be adequate.
k) Successors and Assigns. Subject to applicable securities laws, this Warrant and the rights and obligations evidenced hereby shall inure to the benefit of and be binding upon the successors and permitted assigns of the Company and the successors and permitted assigns of Holder. The provisions of this Warrant are intended to be for the benefit of any Holder from time to time of this Warrant and shall be enforceable by the Holder or holder of Warrant Shares.
l) Amendment. This Warrant may be modified or amended or the provisions hereof waived with the written consent of the Company, on the one hand, and the Holder of this Warrant, on the other hand.
m) Severability. Wherever possible, each provision of this Warrant shall be interpreted in such manner as to be effective and valid under applicable law, but if any provision of this Warrant shall be prohibited by or invalid under applicable law, such provision shall be ineffective to the extent of such prohibition or invalidity, without invalidating the remainder of such provisions or the remaining provisions of this Warrant.
n) Headings. The headings used in this Warrant are for the convenience of reference only and shall not, for any purpose, be deemed a part of this Warrant.
********************
(Signature Page Follows)
IN WITNESS WHEREOF, the Company has caused this Warrant to be executed by its officer thereunto duly authorized as of the date first above indicated.
| PSYENCE BIOMEDICAL LTD. | ||
| By: | ||
| Name: | ||
| Title: | ||
NOTICE OF EXERCISE
| TO: | PSYENCE BIOMEDICAL LTD. |
(1) The undersigned hereby elects to purchase Warrant Shares of the Company pursuant to the terms of the attached Warrant (only if exercised in full), and tenders herewith payment of the exercise price in full, together with all applicable transfer taxes, if any.
(2) Please issue said Warrant Shares in the name of the undersigned or in such other name as is specified below:
The Warrant Shares shall be delivered to the following DWAC Account Number:
(3) Accredited Investor. The undersigned is an “accredited investor” as defined in Regulation D promulgated under the Securities Act of 1933, as amended.
(4) Non-Canadian. The undersigned is not a resident of any jurisdiction of Canada.
[SIGNATURE OF HOLDER]
Name of Investing Entity:
Signature of Authorized Signatory of Investing Entity:
Name of Authorized Signatory:
Title of Authorized Signatory:
Date:
EXHIBIT B
ASSIGNMENT FORM
(To assign the foregoing Warrant, execute this form and supply required information. Do not use this form to exercise the Warrant to purchase shares.)
In the case of a Warrant certificate that contains a Canadian restrictive legend, the undersigned transferor hereby represents, warrants and certifies that:
(a) the transfer is being made on or through the facilities of an exchange or market outside of Canada; and
(b) neither the undersigned transferor nor anyone acting on its behalf has reason to believe that the assignment has been pre-arranged with a buyer in Canada.
FOR VALUE RECEIVED, the foregoing Warrant and all rights evidenced thereby are hereby assigned to
| Name: | |
| (Please Print) | |
| Address: | |
| (Please Print) | |
| Phone Number: | |
| Email Address: |
Dated: ______________, _________
Holder’s
Signature:__________________________________________________
Holder’s
Address:___________________________________________________
Exhibit 10.1
TERMINATION AGREEMENT
Between
PSYENCE BIOMEDICAL LTD., a corporation existing under the laws of Ontario, Canada (the "Company");
PSYENCE BIOMED II CORP., a corporation existing under the laws of Ontario, Canada ("Biomed II");
HARRADEN CIRCLE INVESTORS, LP, a fund, account, and/other investment vehicle managed by Harraden Circle Investments, LLC ("HCI");
HARRADEN CIRCLE SPECIAL OPPORTUNITIES, LP, a fund, account, and/other investment vehicle managed by Harraden Circle Investments, LLC ("HCSO", and together with HCI the "Holders");
and
Newcourt SPAC Sponsor LLC ("Sponsor")
(collectively, the "Parties")
1 RECITALS
| 1.1 | On January 15, 2024, the Company and Biomed II entered into (i) the Securities Purchase Agreement (as subsequently amended pursuant to that certain First Addendum, by and among the Parties, the "Harraden SPA") with the Holders and the Sponsor, relating to up to four senior secured convertible notes (each a "Note" and collectively the "Notes") obligations under which are guaranteed by certain assets of the Company and Biomed II, issuable to the Holders for the aggregate principal amount of up to $12,500,000 in exchange for up to $10,000,000 in subscription amounts, (ii) the Lock-Up Agreement ("Lock-Up Agreement") with Psyence Group Inc. ("PSYG") restricting certain transactions by PSYG; (iii) the General Security Agreement, dated January 25, 2024 (the "Security Agreement"); and (iv) the Registration Rights Agreement, dated January 25, 2024 (the "RRA" and, collectively with the Harraden SPA, the Lock-Up Agreement, the Security Agreement and the Notes, the "Transaction Documents"). |
| 1.2 | The two first tranche notes, for an aggregate of $3,125,000, were delivered by the Company to the Holders on January 25, 2024, in exchange for an aggregate of $2,500,000 in financing ("First Tranche Notes"). On May 31, 2024, the Notes (the "May Second Tranche Notes") for the initial portion of the second tranche of the financing (the "Second Tranche"), for a total of $312,500 of principal in exchange for a total of $250,000 in subscription amounts, were issued to the Holders. On June 14, 2024, the Company issued two additional Notes (the "June Second Tranche Notes") for an additional portion of the Second Tranche, for an aggregate total of $312,500 principal in exchange for $250,000 provided by the Holders. In July 2024, the Company issued two additional Notes (the "July Second Tranche Notes") for an additional portion of the Second Tranche, for an aggregate total of $625,000 principal in exchange for $500,000 provided by the Holders. The May, June and July Second Tranche Notes shall be collectively referred to as the "Second Tranche Notes". |
| 1.3 | In addition to the terms defined elsewhere in this Termination Agreement (this "Termination Agreement") capitalized terms that are not otherwise defined herein have the meanings given to such terms in the Harraden SPA and the Notes (as the case may be) or in the annexes attached thereto. |
| 1.4 | The Parties wish to terminate all obligations under the Transaction Documents. |
| 1.5 | The Parties wish to record these additional obligations and amendments in writing. |
2 EFFECTIVE DATE AND CONDITIONS PRECEDENT
| 2.1 | This Termination Agreement shall be effective upon on the date that all of the conditions specified in clause 2.2 are satisfied (the "Effective Date"), subject to the fulfilment or waiver (as the case may be) of the suspensive conditions ("Conditions Precedent") contained in clause 2.2. |
| 2.2 | This Termination Agreement shall become effective upon the following: |
| 2.2.1 | by not later than 17h00 on November 30, 2024, the board of directors of PBM approves and ratifies the entering into of this Termination Agreement and all other agreements and transactions contemplated therein; |
| 2.2.2 | the Holder Warrants (as defined below) being issued no later than November 30, 2024; |
| 2.2.3 | the Holder Shares (as defined below) being issued without restrictive legend, no later than November 30, 2024; |
| 2.2.4 | The Holders having taken all steps necessary to terminate the Transaction Documents and full and final release all Security Interests; |
| 2.2.5 | The Company issuing an irrevocable instruction to the Company’s transfer agent authorizing the transfer agent to issue the Holder Warrant Shares to the Holder upon exercise of the Holder Warrants, and counsel to the Company delivering an opinion to the Company’s transfer agent authorizing the issuance of the Holder Warrant Shares to the Holder, each in a form reasonably satisfactory to the Holder. |
| 2.3 | In the event that the provisions of clause 2.2 are not satisfied, this Termination Agreement shall be void and of no further force or effect and any securities issued pursuant to the terms herein shall be cancelled, returned or otherwise rescinded. |
The Company agrees to by 9:00 a.m., New York City time, on the second (2nd) business day immediately following the Effective Date, to file a form 6-k (Disclosure Document) disclosing the Termination Agreement, the then current Common Shares outstanding and any other material, nonpublic information that the Company or their respective representatives have provided to the Holders at any time prior to the filing of the Disclosure Document.
| 2.4 | Unless all the Conditions Precedent have been fulfilled or waived by not later than the relevant dates for fulfilment thereof set out in clause 2.2 (or such later date or dates as may be agreed in writing between the Parties) the provisions of this Termination Agreement, save for clauses 1, 2, 6 and 7, which will remain of full force and effect, will never become of any force or effect and the status quo ante will be restored as near as may be and neither of the Parties will have any claim against the other in terms hereof or arising from the failure of the Conditions Precedent. |
3 TERMINATION OF THE NOTES AND THE HARRADEN SPA
| 3.1 | Full and Final Settlement and Termination. Except for the RRA, which will continue in full force and effect as it relates to the Holder Shares and Holder Warrant Shares and except as otherwise specified in Section 3.2.3 below, upon full and complete performance of all obligations of the Company as outlined in this Termination Agreement, all obligations, covenants, and liabilities of each Party under the Transaction Documents shall be fully and finally terminated, discharged, and deemed satisfied. This includes, without limitation, the termination of any rights, duties, and enforcement options available to any Party under each of the Transaction Documents other than the RRA, including, but not limited to, the full and final release of all Security Interests (as defined in the Security Agreement) by the Holders pursuant to the Security Agreement, the extinguishment of all legal and financial obligations, claims and responsibilities under the Notes. For the avoidance of doubt, except for the RRA, which will continue in full force and effect, following full performance, no further obligations or rights shall survive with respect to any Party. For purposes of this Termination Agreement, claims include, without limitation, any action, application, arbitration, cause of action, complaint, cost, debt due, demand, determination, enquiry, judgment, cost order and verdict at common law; constituting a delict; in equity; arising under contract; and arising under any statute, regulation or otherwise howsoever. The Holders shall, and shall cause their respective affiliates, agents, and representatives to reasonably cooperate with the Company, at the Company’s expense and to the extent permitted by law, in filing any required federal, state, and local regulatory documents, executing necessary agreements, and delivering any additional required documentation to effectuate the termination of the Transaction Documents and the full and final release of the Security Interests. |
| 3.2 | Without limitation to the generality of clause 3.1 - |
| 3.2.1 | the Holders waive any respective rights to call an event of default, breach, or other similar event (including, without limitation, an “Event of Default” under the Notes) under the Transaction Documents; and |
| 3.2.2 | the Holders waive all penalties or liquidated damages payable under the Transaction Documents with respect to any Events of Default that were waived pursuant to clause 3.2.1 (the "Waived Defaults") which are due as at the Effective Date (if any) and undertake not to pursue any rights and remedies provided to them under the Transaction Documents with respect to any penalties or liquidated damages payable under the Transaction Documents with respect to the Waived Defaults which are due as at the Effective Date (if any). |
| 3.2.3 | Notwithstanding anything herein to the contrary, the RRA will remain in full force and effect as it relates to the registration rights of the Holder of the Holder Securities, upon full and complete performance of all obligations of the Company as outlined in this Termination Agreement, all other obligations, terms, conditions and restrictions pursuant to Section 2(i) of the RRA, are hereby terminated, and shall be of no further force and effect. |
| 3.2.4 | Investment Purpose & Canadian Securities Laws. Holder is acquiring the Holder Shares and, when issued, the Holder Warrant Shares, for its own account for investment purposes only and without any view to, or for resale in connection with, any “distribution” thereof within the meaning of the Securities Act. Holder further understands that (i) the Company is not a “reporting issuer” (or the equivalent of a reporting issuer) in any province or territory of Canada, and the Holder Shares, and when issued, the Holder Warrant Shares, have not been qualified for distribution by prospectus in Canada, (ii) the Holder Shares, and when issued, the Holder Warrant Shares, may not be offered or sold in Canada except pursuant to a Canadian prospectus or prospectus exemption); (iii) no representation has been made respecting the applicable hold periods imposed by the securities laws in the provinces and territories of Canada (the “Canadian Securities Laws”) or other resale restrictions applicable to the Holder Shares, and when issued, the Holder Warrant Shares, which restrict the ability of the Holder (or any beneficial purchaser for whom it is contracting hereunder) to resell such securities; (iii) it is solely responsible to find out what these restrictions are; (iv) it is solely responsible (and the Company is not in any way responsible) for compliance with applicable resale restrictions; and (v) it is aware that it may not be able to resell the Holder Shares, and when issued, the Holder Warrant Shares, which in Canada, except in accordance with limited exemptions under the Canadian Securities Laws . The Holder represents and warrants that it is acquiring the Holder Shares, and when issued, the Holder Warrant Shares, which as principal for its own account with investment intent and not with a view to or for distributing or reselling such Holder Shares, and when issued, the Holder Warrant Shares, which or any part thereof in violation of Canadian Securities Laws, has no present intention of distributing any of such Holder Shares, and when issued, the Holder Warrant Shares, which in violation of Canadian Securities Laws, and has no direct or indirect arrangement or understandings with any other persons to distribute or regarding the distribution of such Holder Shares, and when issued, the Holder Warrant Shares, which to or for the benefit of person in Canada. Such Holder is acquiring the Holder Shares, and when issued, the Holder Warrant Shares, hereunder in the ordinary course of its business. |
4 CONSIDERATION
| 4.1 | In consideration for the waiver and abandonment of the Holder's claims and entitlements, the termination of the Transaction Documents and the release of all Security Interests, as contemplated in this Termination Agreement, the Company agrees to pay the Holders as follows: |
| 4.1.1 | Promptly after the signing hereof, the Company shall issue to the Holders or their respective designees an aggregate of 11,325,750 Common Shares ("Holder Shares"), subject to adjustment pursuant to the proposed consolidation of Common Shares to be effected by the Company. An aggregate of 6,742,920 of the Holder Shares shall be issued pursuant to the Company’s obligations under the First Tranche Notes (the “First Tranche Note Shares”) and the remaining 4,582,830 Holder Shares shall be issued pursuant to the Company’s obligations under the Second Tranche Notes (the “Second Tranche Note Shares”). The Second Tranche Note Shares are eligible for resale pursuant to the Registration Statement on Form F-1 filed by the Company with the SEC on October 2, 2024 (File No. 333-282468) and declared effective by the SEC on October 10, 2024. The Second Tranche Note Shares are eligible for resale pursuant to the Registration Statement on Form F-1 (File No. 333-276973) declared effective by the SEC on May 14, 2024. |
| 4.1.2 | Promptly after the signing hereof, the Company shall issue to the Holders or their respective designees warrants (the "Holder Warrants”) to purchase 13,674,250 Common Shares (the “Holder Warrant Shares” and together with the Holder Warrants and Holder Shares, the “Holder Securities”), on the following terms: |
| 4.1.2.1 | The Holder Warrants shall have an exercise price of $0.0000001; and |
| 4.1.2.2 | the Holder Warrants shall be in the form attached to this agreement as Exhibit A. |
| 4.1.3 | The Holder Shares and the Holder Warrant Shares shall be deemed to be "Underlying Shares" and "Registrable Securities" under the terms of the RRA. |
| 4.1.4 | The release and any subsequent sale, transfer, assignment, or disposition of the the Holder Warrants and the Holder Warrant Shares (collectively, the "Holder Securities"), as outlined in this Termination Agreement, shall be subject to and conditioned upon compliance with all applicable federal, state, and foreign securities laws, rules, and regulations, including, without limitation, those governing the registration, sale, or transfer of securities. The Company and the Holder agree to cooperate in good faith to satisfy any regulatory requirements necessary to permit the lawful release, issuance, and sale of the Holder Securities. Furthermore, the Holder agrees not to sell, transfer, assign, or otherwise dispose of the Holder Securities except in transactions that are exempt from registration under the Securities Act of 1933, as amended, or pursuant to an effective registration statement. Prior to the effective date of the Registration Statement, the Company may place appropriate restrictive legends on the Holder Securities and take other necessary steps to ensure compliance with such securities laws. |
5 SAVINGS CLAUSE
Except as expressly stated in this Termination Agreement, all terms, conditions, covenants, and obligations of the Parties under the Transaction Documents are hereby terminated and shall have no further force or effect upon full performance of the obligations outlined herein. In the event of any discrepancy or inconsistency between this Termination Agreement and any of the Transaction Documents, the terms of this Termination Agreement shall govern and prevail. Only those provisions of the Transaction Documents expressly preserved or referenced in this Termination Agreement shall continue in effect, solely to the extent necessary to carry out the terms of this Termination Agreement.
6 COSTS
Each Party will bear and pay its own legal costs and expenses of and incidental to the negotiation, drafting, preparation and implementation of this Termination Agreement.
7 SIGNATURE
Signed on behalf of the parties, each signatory hereto warranting that he/she has due authority to do so.
[signature page follows]
IN WITNESS WHEREOF THE PARTIES HAVE EXECUTED THIS TERMINATION AGREEMENT AS OF THE DATE FIRST WRITTEN ABOVE.
| PSYENCE BIOMEDICAL LTD. | ||
| By: | /s/ Jody Aufrichtig | |
| Name: Jody Aufrichtig | ||
| Title: Director | ||
| PSYENCE BIOMED II CORP. | ||
| By: | /s/ Jody Aufrichtig | |
| Name: Jody Aufrichtig | ||
| Title: Director | ||
| HARRADEN CIRCLE INVESTORS, LP, as Purchaser | ||
| By: | /s/ Frederick V. Fortmiller | |
| Name: Frederick V. Fortmiller | ||
| Title: Authorized Signatory | ||
| HARRADEN CIRCLE SPECIAL OPPORTUNITIES, LP, as Purchaser | ||
| By: | /s/ Frederick V. Fortmiller | |
| Name: Frederick V. Fortmiller | ||
| Title: Authorized Signatory | ||
| NEWCOURT SPAC SPONSOR LLC | ||
| By: | ||
| Name: | ||
| Title: | ||
EXHIBIT A
Form of Warrant
Exhibit 99.1
Psyence Biomedical Announces Effective Date for 1-for-75 Share Consolidation
NEW YORK, November 19, 2024 – Psyence Biomedical Ltd. (Nasdaq: PBM) (“Psyence Biomed” or the “Company”) today announced an effective date for its previously-announced share consolidation. At its Annual General and Special Meeting of Shareholders, which was held on November 12, 2024, Psyence Biomed’s shareholders approved a consolidation of the Company’s common shares at a ratio of up to 1 common share for every 75 common shares held, with the final ratio to be determined by the Company’s board of directors (the “Board”). Also, as previously announced, the Board approved a 1-for-75 consolidation of its outstanding common shares, which the Company intends to be effective at 5:00pm ET on Friday, November 22, 2024.
The Company's common shares will begin trading on a post-consolidated basis at the opening of the market on Monday, November 25, 2024. Following the consolidation, the Company's common shares will continue to trade under the symbol 'PBM' with the new CUSIP number 74449F209. The consolidation is part of the Company's plan to regain compliance with the minimum bid price requirement of Nasdaq Listing Rule 5450(a)(1) (the “Nasdaq Rule 5450”) for continued listing on The Nasdaq Global Market.
At the effective time of the consolidation, every 75 issued and outstanding shares of the Company's common shares will automatically be combined into one issued and outstanding share of the Company's common shares (having no par value per share). The number of shares and the exercise price of the Company’s outstanding warrants will also be adjusted for the consolidation in accordance with the terms of such warrants.
No fractional common shares of the Company will be issued if, as a result of the consolidation, a shareholder would otherwise be entitled to a fractional share. Instead, any fractional common shares resulting from the consolidation will be rounded down to the nearest whole share if the fraction is less than one-half of a share and will be rounded up to the nearest whole share if the fraction is at least one-half or a share. The shares underlying the Company’s outstanding equity awards and warrants will be adjusted accordingly. The consolidation affects all shareholders uniformly and will not alter any shareholder’s percentage interest in the Company’s common shares, except for adjustments that may result from the treatment of fractional shares.
Continental Stock Transfer & Trust Company is acting as the exchange agent and transfer agent for the consolidation. Shareholders holding their shares in book-entry form or in brokerage accounts need not take any action in connection with the consolidation. Beneficial holders are encouraged to contact their bank, broker or custodian with any procedural questions.
About Psyence Biomed:
Psyence Biomedical Ltd. (Nasdaq: PBM) is one of the world’s few vertically integrated biopharmas with a focus on psychedelic-based pharmaceutical therapeutics. The first life science biotechnology company developing nature-derived (non-synthetic) psilocybin-based psychedelic medicine to be listed on Nasdaq, Psyence is initially working to address the unmet needs of patients who suffer from mental health disorders in the context of Palliative Care. The name “Psyence” combines the words “psychedelics” and “science” to affirm Psyence Biomed’s commitment to an evidence-based approach to innovation as it works to develop safe and effective, FDA-approved, nature-derived psychedelic therapeutics to treat a broad range of mental health disorders.
Learn more at www.psyencebiomed.com and on LinkedIn.
Contact Information for Psyence Biomedical Ltd.
Email: ir@psyencebiomed.com
Media Inquiries: media@psyencebiomed.com
General Information: info@psyencebiomed.com
Phone: +1 416-477-1708
Investor Contact:
Jeremy Feffer
Managing Director
LifeSci Advisors
jfeffer@lifesciadvisors.com
Forward Looking Statements
This communication contains "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995. Such statements include, but are not limited to, statements about future financial and operating results, our plans, objectives, expectations and intentions with respect to future operations, products and services; and other statements identified by words such as "will likely result," "are expected to," "will continue," "is anticipated," "estimated," "believe," "intend," "plan," "projection," "outlook" or words of similar meaning.
Forward-looking statements in this communication include statements regarding effective date of the share consolidation referred to in this news release, the expected commencement date of its common shares trading on a split-adjusted basis and the decision regarding the Company’s continued listing on The Nasdaq Global Market. These forward-looking statements are based on a number of assumptions, including the assumption that there will be no delays in effecting the share consolidation.
There are numerous risks and uncertainties that may cause actual results or performance to be materially different from those expressed or implied by these forward-looking statements. These risks and uncertainties include, among others: (i) delays in effecting the share consolidation (ii) the ability of Psyence Biomed to maintain the listing of its common shares and warrants on Nasdaq; (iii) the effectiveness of an increased pool of available shares under the 2023 Plan in incentivizing current employees and attracting future talent; and (iv) volatility in the price of the securities of Psyence Biomed due to a variety of factors, including the proposed share consolidation, changes in the competitive and highly regulated industries in which Psyence Biomed operates, variations in performance across competitors, changes in laws and regulations affecting Psyence Biomed’s business and changes in Psyence Biomed’s capital structure. The foregoing list of factors is not exhaustive. You should carefully consider the foregoing factors and the other risks and uncertainties described in the "Risk Factors" section of the final prospectus (File No. 333-282468) filed with the Securities and Exchange Commission on October 10, 2024 and other documents filed by Psyence Biomed from time to time with the SEC. These filings identify and address other important risks and uncertainties that could cause actual events and results to differ materially from those contained in the forward-looking statements. Actual results and future events could differ materially from those anticipated in such information. Nothing in this communication should be regarded as a representation by any person that the forward-looking statements set forth herein will be achieved or that any of the contemplated results of such forward-looking statements will be achieved. You should not place undue reliance on forward-looking statements, which speak only as of the date they are made. Except as required by law, Psyence Biomed does not intend to update these forward-looking statements.
The Company does not make any medical, treatment or health benefit claims about its proposed products. The U.S. Food and Drug Administration, Health Canada or other similar regulatory authorities have not evaluated claims regarding psilocybin, psilocybin analogues, or other psychedelic compounds or nutraceutical products. The efficacy of such products has not been confirmed by approved research. There is no assurance that the use of psilocybin, psilocybin analogues, or other psychedelic compounds or nutraceuticals can diagnose, treat, cure or prevent any disease or condition. Vigorous scientific research and clinical trials are needed. The Company has not conducted clinical trials for the use of the proposed products. Any references to quality, consistency, efficacy, and safety of potential products do not imply that the Company has verified such in clinical trials or that the Company will complete such trials. If the Company cannot obtain the approvals or research necessary to commercialize its business, it may have a material adverse effect on the Company’s performance and operations.
Exhibit 99.2
Psyence Biomedical Announces Favorable Result of Nasdaq Listing Qualifications Hearing
NEW YORK, November 20, 2024 -- Psyence Biomedical Ltd. (Nasdaq: PBM) (“Psyence Biomed” or the “Company”) today announced the favorable outcome of the Nasdaq Listing Qualifications Hearing that was held on October 31st, 2024. The Nasdaq Hearings Panel has granted Psyence Biomed an extension until December 31st, 2024, to demonstrate compliance with all Nasdaq continued listing rules.
During the Company’s Nasdaq Listing Qualifications Hearing, Psyence Biomed presented a comprehensive plan to regain and maintain compliance with Nasdaq’s continued listing requirements, including 5550(a)(2), the $1 minimum bid price requirement, 5550(a)(5), the $1.0 million minimum market value of publicly held shares requirement, and 5550(b)(1), the $2.5 million minimum stockholders' equity requirement.
As part of its plan, Psyence Biomed requested to be transferred from The Nasdaq Global Market to The Nasdaq Capital Market pursuant to a Panel exception, effective November 15th, 2024. Furthermore, as previously announced, Psyence Biomed obtained shareholder approval during its Annual General Meeting on November 12th, 2024, to effect a 1-for-75 share consolidation that, when complete, will satisfy Nasdaq’s $1 minimum bid price requirement. The Company expects to demonstrate compliance with all the applicable requirements for continued listing on The Nasdaq Capital Market within the Panel’s extension.
“We are very pleased to have gained alignment with the Nasdaq Hearings Panel on our plan to regain compliance with all applicable listing requirements,” stated Dr. Neil Maresky, M.B.,B.Ch., Chief Executive Officer of Psyence Biomed. “Having our shares traded on the Nasdaq exchange is critical to raising awareness of our company among healthcare-focused institutional investors. Once these listing requirements are satisfied, we can focus on advancing our pipeline of nature-derived psilocybin-based therapeutics as potential treatments for underserved conditions in mental health and addiction where more efficacious options are urgently needed.”
The Panel reserves the right to reconsider the terms of this extension based on any event, condition or circumstance that exists or develops that would, in the opinion of the Panel, make continued listing of the Company’s securities on the Exchange inadvisable or unwarranted.
About Psyence Biomed:
Psyence Biomedical Ltd. (Nasdaq: PBM) is one of the world’s few vertically integrated biopharmas with a focus on psychedelic-based pharmaceutical therapeutics. The first life science biotechnology company developing nature-derived (non-synthetic) psilocybin-based psychedelic medicine to be listed on Nasdaq, Psyence is initially working to address the unmet needs of patients who suffer from mental health disorders in the context of Palliative Care. The name “Psyence” combines the words “psychedelics” and “science” to affirm Psyence Biomed’s commitment to an evidence-based approach to innovation as it works to develop safe and effective, FDA-approved, nature-derived psychedelic therapeutics to treat a broad range of mental health disorders.
Learn more at www.psyencebiomed.com and on LinkedIn.
Contact Information for Psyence Biomedical Ltd.
Email: ir@psyencebiomed.com
Media Inquiries: media@psyencebiomed.com
General Information: info@psyencebiomed.com
Phone: +1 416-477-1708
Investor Contact:
Jeremy Feffer
Managing Director
LifeSci Advisors
jfeffer@lifesciadvisors.com
Forward Looking Statements
This communication contains "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995. Such statements include, but are not limited to, statements about future financial and operating results, our plans, objectives, expectations and intentions with respect to future operations, products and services; and other statements identified by words such as "will likely result," "are expected to," "will continue," "is anticipated," "estimated," "believe," "intend," "plan," "projection," "outlook" or words of similar meaning.
Forward-looking statements in this communication include statements regarding effective date of the share consolidation referred to in this news release, the expected commencement date of its common stock trading on a split-adjusted basis and the decision regarding the Company’s continued listing on The Nasdaq Global Market. These forward-looking statements are based on a number of assumptions, including the assumption that there will be no delays in effecting the share consolidation.
There are numerous risks and uncertainties that may cause actual results or performance to be materially different from those expressed or implied by these forward-looking statements. These risks and uncertainties include, among others: (i) delays in effecting the share consolidation (ii) the ability of Psyence Biomed to maintain the listing of its common shares and warrants on Nasdaq; (iii) the 1-for-75 common share split being an effective method by which to comply with the Nasdaq $1 bid price requirement; (iv) the ability to implement the other factors in the Nasdaq compliance plan presented to the Panel and the Panel’s continued opinion that the plan warrants the exception granted; and (v) volatility in the price of the securities of Psyence Biomed due to a variety of factors, including the proposed share consolidation, changes in the competitive and highly regulated industries in which Psyence Biomed operates, variations in performance across competitors, changes in laws and regulations affecting Psyence Biomed’s business and changes in Psyence Biomed’s capital structure. The foregoing list of factors is not exhaustive. You should carefully consider the foregoing factors and the other risks and uncertainties described in the "Risk Factors" section of the final prospectus (File No. 333-282468) filed with the Securities and Exchange Commission on October 10, 2024 and other documents filed by Psyence Biomed from time to time with the SEC. These filings identify and address other important risks and uncertainties that could cause actual events and results to differ materially from those contained in the forward-looking statements. Actual results and future events could differ materially from those anticipated in such information. Nothing in this communication should be regarded as a representation by any person that the forward-looking statements set forth herein will be achieved or that any of the contemplated results of such forward-looking statements will be achieved. You should not place undue reliance on forward-looking statements, which speak only as of the date they are made. Except as required by law, Psyence Biomed does not intend to update these forward-looking statements.
The Company does not make any medical, treatment or health benefit claims about its proposed products. The U.S. Food and Drug Administration, Health Canada or other similar regulatory authorities have not evaluated claims regarding psilocybin, psilocybin analogues, or other psychedelic compounds or nutraceutical products. The efficacy of such products has not been confirmed by approved research. There is no assurance that the use of psilocybin, psilocybin analogues, or other psychedelic compounds or nutraceuticals can diagnose, treat, cure or prevent any disease or condition. Vigorous scientific research and clinical trials are needed. The Company has not conducted clinical trials for the use of the proposed products. Any references to quality, consistency, efficacy, and safety of potential products do not imply that the Company has verified such in clinical trials or that the Company will complete such trials. If the Company cannot obtain the approvals or research necessary to commercialize its business, it may have a material adverse effect on the Company’s performance and operations.
Exhibit 99.3

Investor Presentation Nasdaq: PBM November 2024

2 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature This presentation (“Presentation”) is being issued by Psyence Biomedical Ltd . (the “Company” or “ Psyence ”) for information purposes only . The content of this Presentation has not been approved by any securities regulatory authority . Reliance on this Presentation for the purpose of engaging in any investment activity may expose an individual to a significant risk of losing all of the property or other assets invested . This Presentation is not an admission document, prospectus or an advertisement and is being provided for information purposes only and does not constitute or form part of, and should not be construed as, an offer or invitation to sell or any solicitation of any offer to purchase or subscribe for any securities of the Company in Canada, the United States or any other jurisdiction . Neither this Presentation, nor any part of it nor anything contained or referred to in it, nor the fact of its distribution, should form the basis of or be relied on in connection with or act as an inducement in relation to a decision to purchase or subscribe for or enter into any contract or make any other commitment whatsoever in relation to any securities of the Company . No representation or warranty, express or implied, is given by or on behalf of the Company, its directors, officers and advisors or any other person as to the accuracy, sufficiency or completeness of the information or opinions contained in this Presentation and no liability whatsoever is accepted by the Company, its directors, officers or advisors or any other person for any loss howsoever arising, directly or indirectly, from any use of such information or opinions or otherwise arising in connection therewith . No securities regulatory authority has expressed an opinion about the securities discussed in this Presentation and it is an offense to claim otherwise . The information contained herein does not purport to be all - inclusive . Nothing herein shall be deemed to constitute investment, legal, tax, financial, accounting or other advice . Neither this Presentation nor its delivery to any Recipient shall constitute an offer to sell, invitation or other solicitation of an offer to buy any securities pursuant to the Proposed Investment or otherwise, nor shall there by any sale of securities in any jurisdiction in which the offer, solicitation or sale would be unlawful prior to the registration or qualification under the securities laws of any such jurisdiction . No offer shall be made except by means of a prospectus meeting the requirements of Section 10 of the Securities Act of 1933 , as amended . Only the express provisions of any agreement, if and when it is executed, shall have any legal effect in connection with the Proposed Transaction or the Proposed Investment between the parties thereto . This Presentation is not intended to form the basis of any investment decision . All information herein speaks only as of ( 1 ) the date of this Presentation, in the case of information about Psyence , or ( 2 ) the date of such information, in the case of information from persons other than Psyence . Forward - Looking Statements This Presentation contains “forward - looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995 . Such statements include, but are not limited to, statements about future financial and operating results, our plans, objectives, expectations and intentions with respect to future operations, regulatory approvals, products and services ; and other statements identified by words such as “will likely result,” “are expected to,” “will continue,” “is anticipated,” “estimated,” “believe,” “intend,” “plan,” “projection,” “outlook” or words of similar meaning . Forward - looking statements in this Presentation include statements regarding the future success of the Company to deliver its product candidate to patients . These forward - looking statements are based on a number of assumptions, including the assumptions that Psyence will obtain all such regulatory and other approvals as may be required to pursue its clinical trials on the product candidate referred to in this Presentation, the results of such clinical trials will be positive, and the Company will be able to commercialize its natural psilocybin drug candidate, PEX 010 ( 25 mg), or similar drug candidate, as referred to in this Presentation . There are numerous risks and uncertainties that may cause actual results or performance to be materially different from those expressed or implied by these forward - looking statements . These risks and uncertainties include, among others : demand for the Company’s securities being less than anticipated ; fluctuations in the price of the Company’s common shares ; risks associated with obtaining the necessary regulatory approvals ; and risks associated with conducting the necessary clinical trials referred to in this Presentation . You should carefully consider the foregoing factors and the other risks and uncertainties described in the "Risk Factors" section of the final prospectus (File No . 333 282904 ) filed with the Securities and Exchange Commission on November 12 , 2024 and other documents filed by the Company from time to time with the SEC . These filings identify and address other important risks and uncertainties that could cause actual events and results to differ materially from those contained in the forward - looking statements . There can be no assurance that the data contained herein is reflective of future performance to any degree . You are cautioned not to place undue reliance on forward - looking statements as a predictor of future performance as projected financial information and other information are based on estimates and assumptions that are inherently subject to various significant risks, uncertainties and other factors, many of which are beyond our control . Forward - looking statements speak only as of the date they are made, and Psyence and its respective affiliates disclaim any intention or obligation to update or revise any forward - looking statements, whether as a result of developments occurring after the date of this communication . Forecasts and estimates regarding Psyence’s industry and end markets are based on sources we believe to be reliable, however there can be no assurance these forecasts and estimates will prove accurate in whole or in part . Annualized, pro forma, projected and estimated numbers are used for illustrative purpose only, are not forecasts and may not reflect actual results . Psyence does not make any medical, treatment or health benefit claims about the Psyence’s product candidates . The efficacy of psilocybin, psilocybin analogues, or other psychedelic compounds or nutraceutical products remains the subject of ongoing research . There is no assurance that the use of psilocybin, psilocybin analogues, or other psychedelic compounds or nutraceuticals can diagnose, treat, cure or prevent any disease or condition . Vigorous scientific research and clinical trials are needed . Psyence has not completed clinical trials for the use of its product candidates . Any references to quality, consistency, efficacy, and safety of product candidates do not imply that Psyence verified such in clinical trials or that Psyence will complete such trials . If Psyence cannot obtain the approvals or research necessary to commercialize its business, it may have a material adverse effect on Psyence’s performance and operations .

Forward Looking Statements 3 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature Industry and Market Data This Presentation has been prepared by Psyence and includes market data and other statistical information from third - party sources . Although Psyence believes these third - party sources are reliable as of their respective dates, neither Psyence , nor any of its affiliates has independently verified the accuracy or completeness of this information . Some data are also based on Psyence’s good faith estimates, which are derived from both internal sources and the third - party sources described above . None of Psyence , its affiliates, or its advisors, directors, officers, employees, members, partners, shareholders or agents make any representation or warranty with respect to the accuracy of such information . None of Psyence , its affiliates, or advisors, directors, officers, employees, members, partners, shareholders or agents or the providers of any such third - party information or any other person are responsible for any errors or omissions therein (negligent or otherwise), regardless of the cause, or the results obtained from the use of such content . Psyence and its affiliates, advisors, directors, officers, employees, members, partners, shareholders and agents expressly disclaims any responsibility or liability for any damages or losses in connection with the use of such information herein . Psyence currently has no registered, psilocybin - containing, commercial products and no products as depicted by Psyence are available for sale . Any renderings, depictions and graphic materials of such products contained in this presentation are conceptual only and are for the convenience of reference . These images should not be relied upon as representations, express or implied, of the final detail of the products . Psyence expressly reserves the right to make modifications, revisions, and changes it deems desirable in its sole and absolute discretion .

Forward Looking Statements 4 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature We are a clinical - stage biotechnology company and have incurred significant losses since our inception . We anticipate that we will incur significant losses for the foreseeable future . Psyence has a limited operating history and expects a number of factors to cause its operating results to fluctuate on an annual basis, which may make it difficult to predict the future performance of Psyence . Psyence has never generated revenue and may never be profitable . Psyence will require substantial additional funding to achieve its business goals, and if it is unable to obtain this funding when needed and on acceptable terms, it could be forced to delay, limit or terminate its product development efforts . We depend on our current key personnel and our ability to attract and retain employees . The psychedelic therapy and biotechnology industries are undergoing rapid growth and substantial change, which has resulted in an increase in competitors, consolidation and formation of strategic relationships . Acquisitions or other consolidating transactions could harm Psyence in a number of ways, including by losing strategic partners if they are acquired by or enter into relationships with a competitor, losing customers, revenue and market share, or forcing Psyence to expend greater resources to meet new or additional competitive threats, all of which could harm Psyence’s operating results . Current and future preclinical and clinical studies will be conducted outside the United States, and the FDA may not accept data from such studies to support any NDAs submitted after completing the applicable developmental and regulatory prerequisites (absent an IND) . There is a high rate of failure for product candidates proceeding through clinical trials . Because the results of preclinical studies and earlier clinical trials are not necessarily predictive of future results, Psyence may not have favorable results in its planned and future clinical trials . Negative results from clinical trials or studies of others and adverse safety events involving Psyence’s psychedelic analogs could have a material adverse effect on Psyence’s business . Costs associated with compliance with numerous laws and regulations could impact our financial results . In addition, we could become subject to increased enforcement and/or litigation risks associated with the psychedelic therapeutics industry . We are dependent on licensed intellectual property . If we were to lose our rights to licensed intellectual property, we may not be able to continue developing or commercializing our product candidates, if approved . If we breach any of the agreements under which we license the use, development and commercialization rights to our product candidates or technology from third parties or, in certain cases, we fail to meet certain development deadlines, we could lose license rights that are important to our business . Our prospective products will be subject to the various federal and state laws and regulations relating to health and safety and failure to comply with, or changes in, these laws or regulations could have an adverse impact on our business . If we fail to comply with healthcare regulations, we could face substantial enforcement actions, including civil and criminal penalties and our business, operations and financial condition could be adversely affected . Clinical trials are expensive, time - consuming, uncertain and susceptible to change, delay or termination . The results of clinical trials are open to differing interpretations . Psyence may be subject to federal, state and foreign healthcare laws and regulations and implementation of or changes to such healthcare laws and regulations could adversely affect Psyence’s business and results of operations . Psyence may voluntarily suspend or terminate a clinical trial if at any time its believes that any of its product candidates presents an unacceptable risk to participants, if preliminary data demonstrates that the product candidate is unlikely to receive regulatory approval or unlikely to be successfully commercialized, or if sufficient funds to proceed to the next phases of clinical trials are not raised . The psychedelic therapy industry and market are relatively new, and this industry and market may not continue to exist or grow as anticipated . Negative public opinion and perception of the psychedelic industry could adversely impact Psyence’s ability to operate and Psyence’s growth strategy . The expansion of the use of psychedelics in the medical industry may require new clinical research into effective medical therapies . The psychedelic therapy industry is difficult to quantify and investors will be reliant on their own estimates of the accuracy of market data . Psyence may not be able to adequately protect or enforce its intellectual property rights, which could harm its competitive position . If third parties claim that intellectual property owned or used by Psyence infringes upon their intellectual property, Psyence’s operating profits could be adversely affected . If Psyence is not able to adequately prevent disclosure of trade secrets and other proprietary information, the value of its products could be significantly diminished . If we fail to meet applicable listing requirements, Nasdaq may delist our securities from trading, in which case the liquidity and market price of our securities could decline . The market price and trading volume of our securities may be volatile and could decline significantly . You should carefully consider the foregoing factors and the other risks and uncertainties described in the "Risk Factors" section of the final prospectus (File No . 333 282904 ) filed with the Securities and Exchange Commission on November 12 , 2024 and other documents filed by the Company from time to time with the SEC . These filings identify and address other important risks and uncertainties that could cause actual events and results to differ materially from those contained in the forward - looking statements . There can be no assurance that the data contained herein is reflective of future performance to any degree . You are cautioned not to place undue reliance on forward - looking statements as a predictor of future performance as projected financial information and other information are based on estimates and assumptions that are inherently subject to various significant risks, uncertainties and other factors, many of which are beyond our control . Forward - looking statements speak only as of the date they are made, and Psyence and its respective affiliates disclaim any intention or obligation to update or revise any forward - looking statements, whether as a result of developments occurring after the date of this communication . Forecasts and estimates regarding Psyence’s industry and end markets are based on sources we believe to be reliable, however there can be no assurance these forecasts and estimates will prove accurate in whole or in part . Annualized, pro forma, projected and estimated numbers are used for illustrative purpose only, are not forecasts and may not reflect actual results . Risk Factors Relating to Psyence’s Business 5 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature MISSION To develop nature derived, psilocybin - based therapeutics to address the unmet mental health needs of palliative care and addiction


Company Highlights Phase IIb program, 25mg psilocybin in conjunction with proprietary psychotherapy, being evaluated for Adjustment Disorder in cancer in the Palliative Care context Near - term value - driving catalysts, including first patient randomized in Phase IIb trial in Australia (anticipated in Q4 2024) Leveraging benefits of nature - derived psilocybin, including potential synergistic effects of similar acting molecules not present in synthetic psilocybin* Seasoned leadership team and Board with significant expertise in successful drug development Potential regulatory market and data exclusivity as a New Medicinal Entity (NME) * https://www.nature.com/articles/s41380 - 024 - 02477 - w Acquisition of PsyLabs stake provides vertical integration to support development of second indication, Alcohol Use Disorder

7 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature Projected Upcoming Milestones Phase III Phase II Phase I Pre - clinical First patient enrolled – Q4’24 Top line data – 2025 Adjustment Disorder Initiate Phase IIb pending additional funding Alcohol Use Disorder/ Substance Use Disorder Evaluation of pre - clinical data – H2’24 Undisclosed CNS indication Psilocybin 25mg Development Programs 2 nd Study under evaluation Protocol in Development Phase IIb 8 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature Psychedelics Research is Gaining Acceptance and Momentum


9 Psyence Biomedical Ltd. / Healing Minds with Science .
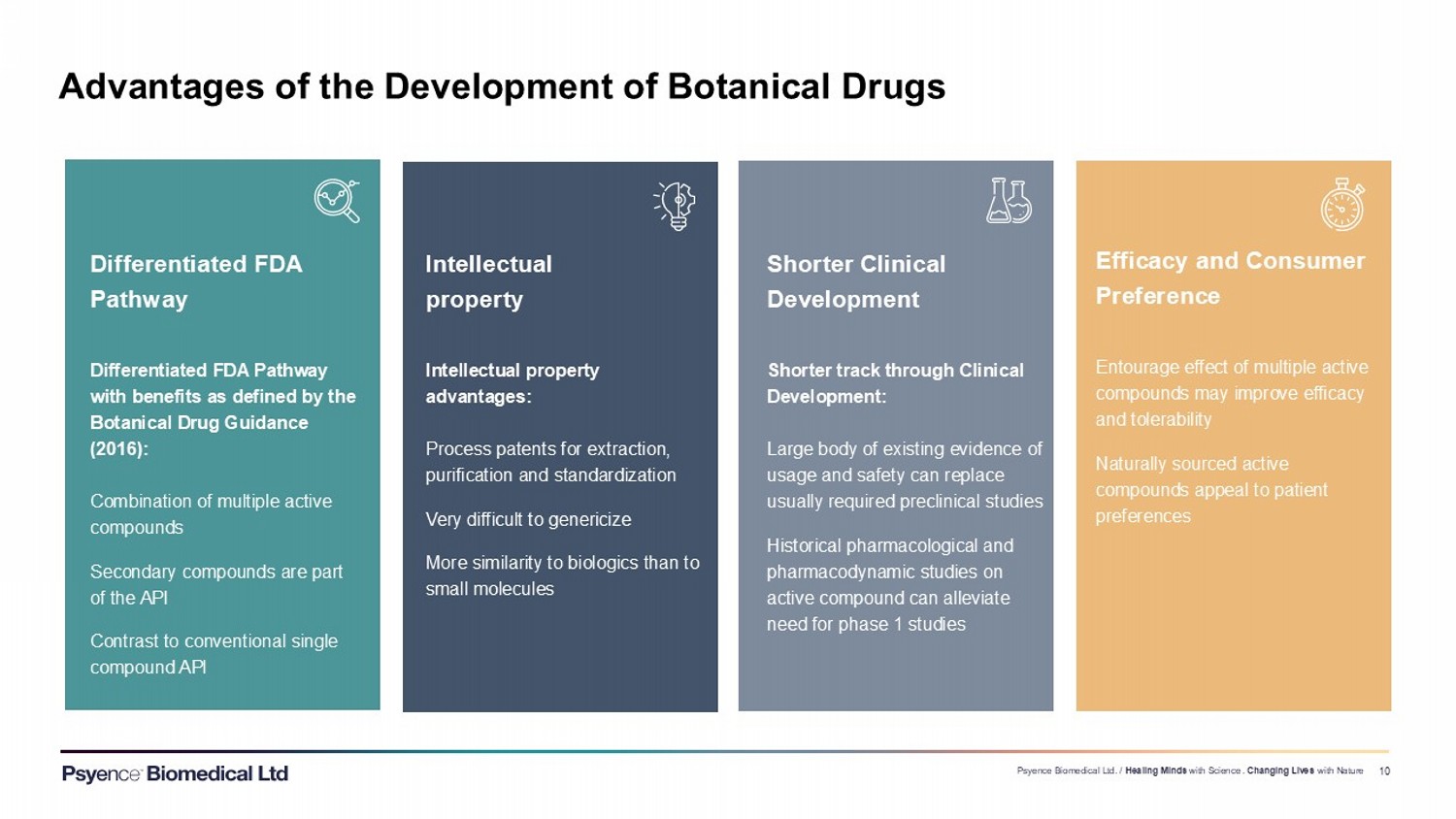
Changing Lives with Nature Accelerating Psychedelics M&A and Growing Institutional Investor Interest 10 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature Differentiated FDA Pathway Differentiated FDA Pathway with benefits as defined by the Botanical Drug Guidance (2016): Combination of multiple active compounds Secondary compounds are part of the API Contrast to conventional single compound API Intellectual property Intellectual property advantages: Process patents for extraction, purification and standardization Very difficult to genericize More similarity to biologics than to small molecules Shorter Clinical Development Shorter track through Clinical Development: Large body of existing evidence of usage and safety can replace usually required preclinical studies Historical pharmacological and pharmacodynamic studies on active compound can alleviate need for phase 1 studies Efficacy and Consumer Preference Entourage effect of multiple active compounds may improve efficacy and tolerability Naturally sourced active compounds appeal to patient preferences Advantages of the Development of Botanical Drugs 11 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature Psyence has licensed proprietary, patent - protected methods of extraction and standardization that may preserve the benefits of nature derived psilocybin with consistent potency ideally suited for therapeutic uses. Nature - Derived Psilocybin vs. Synthetic Psilocybin Nature Derived Contain secondary alkaloids which may contribute to the therapeutic benefit Long history of safe human consumption Proprietary extraction and standardization ensures consistency of dose Synthetic Are typically highly purified, single - molecule compounds Emerging safety/tolerability data Standardized dosing vs


12 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature • A recent paper published in Molecular Psychiatry details results of a preclinical study comparing the biological effects of nature derived psilocybin versus synthetic. • Findings suggest that nature derived psilocybin has a more potent and prolonged effect on neuroplasticity than synthetic. • Neuroplasticity is increasingly recognized as the gold standard by which the efficacy of psychedelic medicines can be measured • This evidence - based and independent finding further validates the Psyence approach to development of nature derived Psilocybin.

Nature Derived Psilocybin May Be Demonstrably More Therapeutic than Synthetic 13 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature *Source: https://www.nia.nih.gov/health/hospice - and - palliative - care/what - are - palliative - care - and - hospice - care Palliative Care is*: • Specialized medical care for people living with a life limiting illness, such as cancer or heart failure. • Patients in Palliative Care may continue to receive treatment as well as care intended to manage their symptoms. • Palliative Care is meant to enhance a person's current care by focusing on quality of life for them, their families and their caregivers. • Cancer patients receiving Palliative Care used significantly less healthcare resources, including emergency department visits, hospital admissions & inpatient days** Palliative Care is NOT End - of - Life (Hospice) Care In Palliative Care, a person does not have to give up treatment that might cure a serious illness. Palliative Care can be provided along with curative treatment and may begin at the time of diagnosis Palliative Care can drive significant savings for payors while improving quality of life for patients ** https://doi.org/10.1038/s41598 - 022 - 23928 - w * https://www.nia.nih.gov/health/hospice - and - palliative - care/what - are - palliative - care - and - hospice - care 14 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature Adjustment Disorder


15 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature *Source: https://www.nia.nih.gov/health/hospice - and - palliative - care/what - are - palliative - care - and - hospice - care Adjustment Disorder ( AjD ) 1. https://www.tandfonline.com/doi/full/10.1080/15622975.2018.1449967?src=recsys 2. Evans SC, Reed GM, Roberts MC, Esparza P, Watts AD, Ritchie PLJ, Maj M, Saxena S. 2013. Psychologists’ perspectives on the di agnostic classification of mental disorders: results from the WHO - IUpsyS Global Survey. Int J Psychol. 48:177 – 193. 3. Reed GM, Correia JM, Esparza P, Saxena S, Maj M. 2011. The WPA - WHO global survey of psychiatrists’ attitudes towards mental disorders classification. World Psychiatry. 10:118 – 131 4. From the Company’s commissioned market research performed by Insight Ace Analytic in Sept. 2022 A global survey of psychiatrists by the WHO revealed 1 : • AjD plays an enormous role in healthcare utilization • AjD ranked 7 th among 44 psychiatric categories 2 they managed at least once a week in their practice • A second global survey of clinical psychiatrists ranked AjD at #9 2 • It ranked higher than alcohol use disorder, at #8 3 • Results were largely consistent across the 44 countries on six continents included in the survey According to Psyence Biomed’s proprietary market research, 2.25 million people (US and EU) with a life limiting cancer diagnosis experience distress each year 4 16 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature *Source: https://www.globenewswire.com/news - release/2024/10/15/2963268/0/en/Palliative - Care - Market - Projected - to - Reach - USD - 23 - 34 - Billion - by - 2032 - Growing - at - a - 9 - 06 - CAGR - SNS - Insider.html Projected Global Palliative Care Market $23.3B with 9.1% CAGR (2032)* The Global Palliative Care Market is Large with a Robust Projected CAGR Other Disease $14.7B* Cancer $8.6B* Cancer = ~37% of Total PC Market
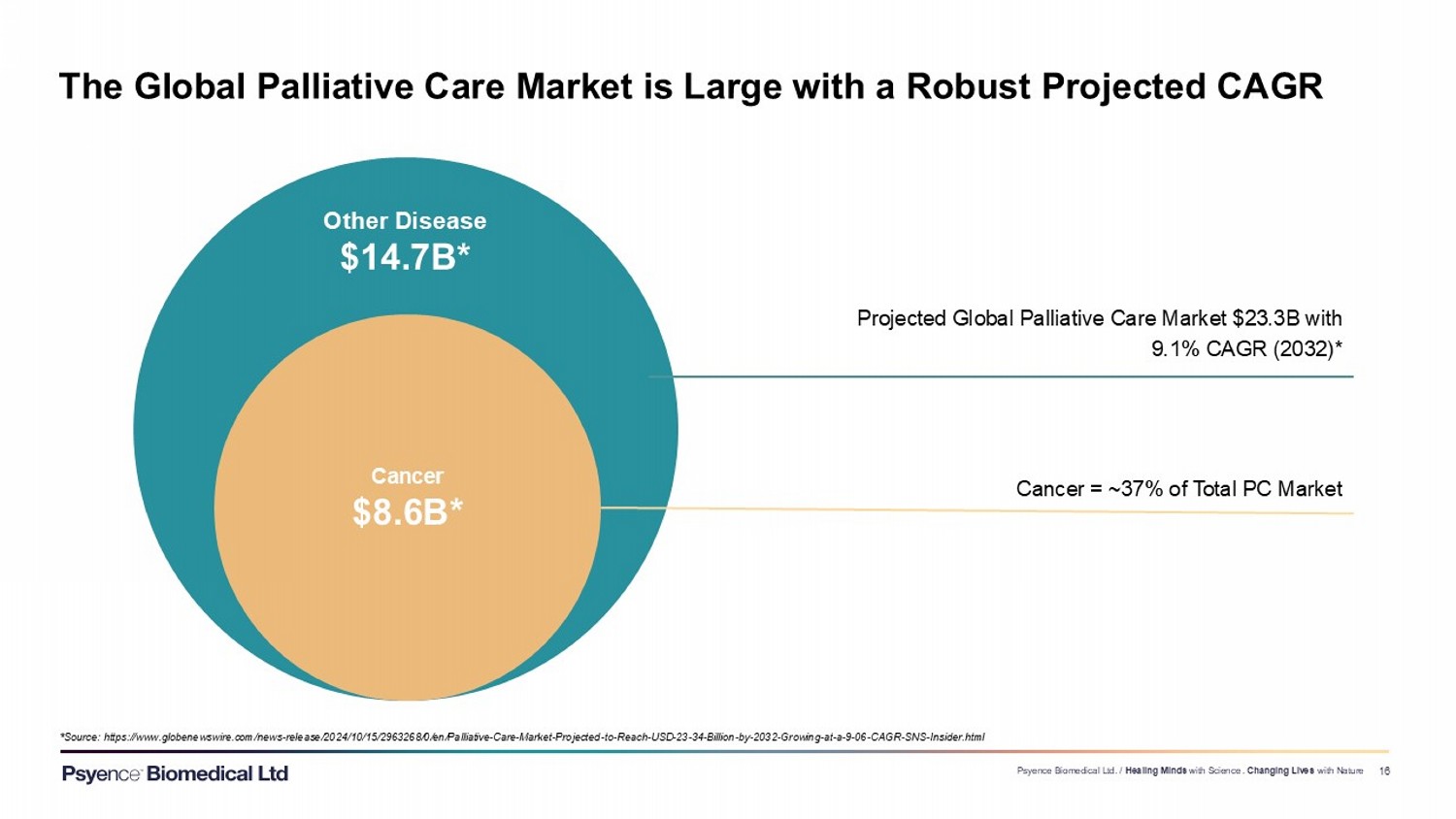
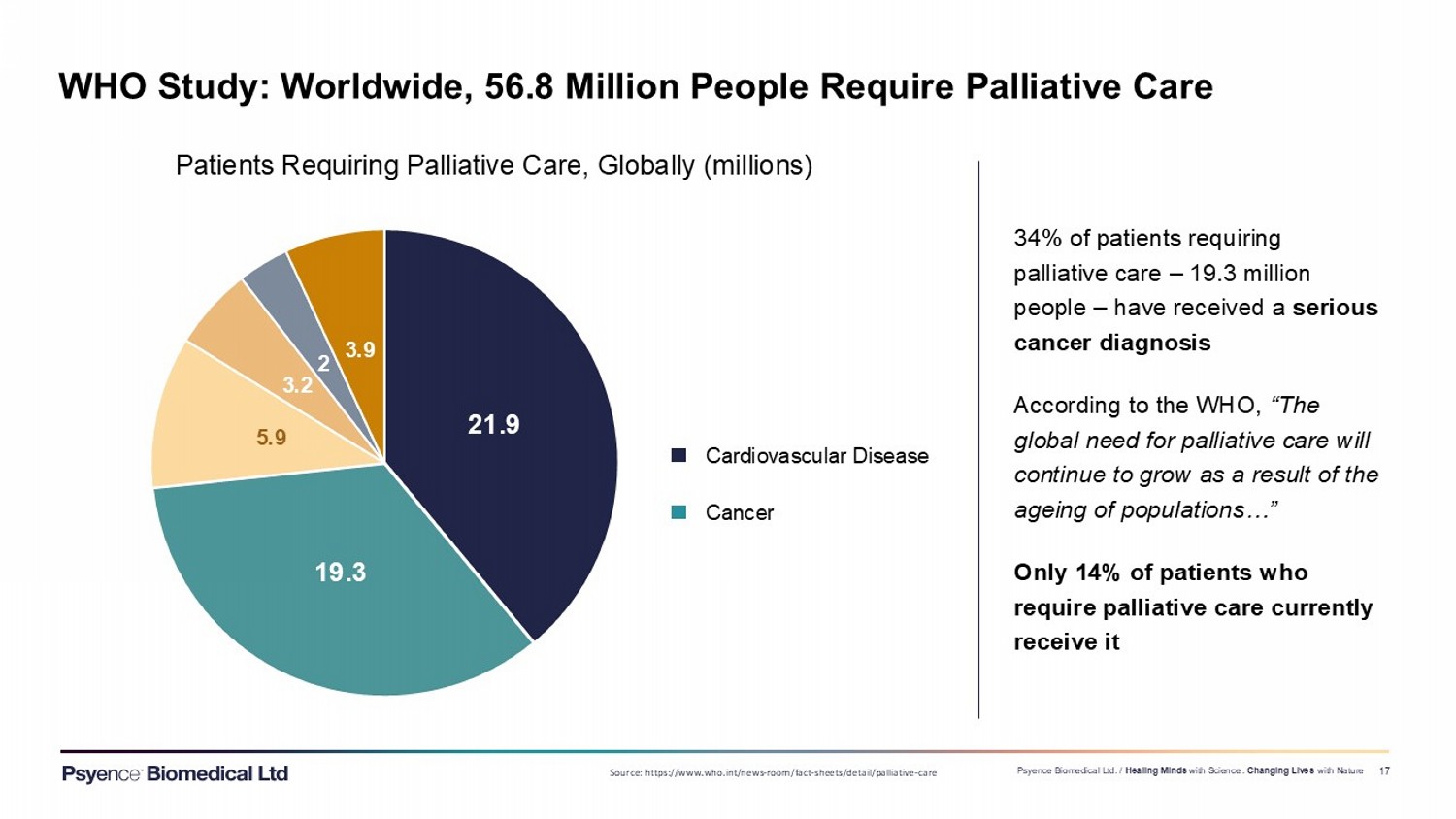
17 Psyence Biomedical Ltd. / Healing Minds with Science .

Changing Lives with Nature Source: https://www.who.int/news - room/fact - sheets/detail/palliative - care 34% of patients requiring palliative care – 19.3 million people – have received a serious cancer diagnosis According to the WHO, “The global need for palliative care will continue to grow as a result of the ageing of populations…” Only 14% of patients who require palliative care currently receive it WHO Study: Worldwide, 56.8 Million People Require Palliative Care 21.9 19.3 5.9 3.2 2 3.9 Patients Requiring Palliative Care, Globally (millions) Cancer Cardiovascular Disease 18 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature *Brennan W and Belser AB (2022), Models of Psychedelic - Assisted Psychotherapy: A Contemporary Assessment and an Introduction to EMBARK, a Transdiagnostic,Trans - Drug Model. Front. Psychol. 13:866018. doi : 10.3389/fpsyg.2022.866018 Patient Safety and Efficacy Enhanced by Psilocybin - Assisted Psychotherapy (PAP) The current standard of care in most uses of psychedelic medicines for the treatment of psychiatric indications includes the provision of a supportive therapeutic context before, during, and after drug administration.* Psyence intends to focus on the psychological distress of palliative care, an important but often ignored and/or poorly treated area. Developing our own unique PAP module to be delivered in Palliative Care Introduction to therapist and process Psychedelic session with therapist for 6 - 8 hours Ongoing psychotherapy or repeat psychedelic depending on response or relapse Integration session Integration session 2 Psilocybin Assisted Psychotherapy (usually 6 - 8 weeks in total) Intention setting Psilocybin Assisted Psychotherapy (usually 6 - 8 weeks in total)

19 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature Prior Proof - of - Concept Studies Provide Strong Rationale for Upcoming Phase IIb At the 6 - month follow - up, these changes were sustained, with about 80% of participants continuing to show clinically significant decreases in depressed mood and anxiety.

At the 6.5 - month follow - up, psilocybin was associated with enduring anxiolytic and antidepressant effects – 60 - 80% of participants continued with clinically significant reductions in depression and anxiety 20 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature Psyence’s clinical trial program is focused on assessing the safety and efficacy of psilocybin - assisted psychotherapy for the treatment of Adjustment Disorder* due to a life - limiting cancer diagnosis versus psychotherapy alone in the Palliative Care context. Trial Leads Dr. Arul Sivanesan Locations(s) Vitalis Research, Melbourne, EMPAX Clinic, Perth and possibly two more sites. CRO Ingenu (www.ingenucro.com). Stage Phase Ilb (Expected to enroll Q4 2024) The study protocol has been approved by the Ethics Board (HREC) using Psyence licensed product FDA response to Pre - IND request received Sample size: 87 patients in three dosage arms – 1mg, 10mg and 25mg. Primary endpoint: HAM - A score at ten weeks Q1 2024 Q4 2024 2025 Australia (Melbourne) 87 PEX010 Botanical Psilocybin * Adjustment disorder is under ICD - 11 and it’s a maladaptive reaction to an identifiable psychosocial stressor or multiple stressors that usually emerges within a month of the stressor. Estimated top line study results PEX010: Australia Clinical Trial Phase IIb Projected Milestones Product and Protocol Overview HREC ethics final approval received HR EC ethics final approval of major amendment received Oct. 24 Estimated timing of first patient randomised Countries/Sites locations: Target Enrollment : Drug Product / imp:

21 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature Study Design: A randomized, double - blind, low - dose comparator - controlled Phase IIb study to investigate the efficacy and safety of psilocybin - assisted psychotherapy (PAP) with 25mg, 10mg and 1mg (low - dose comparator) of PEX010. Key Inclusion Criteria: • A total of 87 subjects, aged 18 - 80 years • Confirmed diagnosis of Adjustment Disorder due to incurable cancer diagnosis • Deemed to be physically capable of undergoing a psychedelic encounter and likely to have a minimum life expectancy of one year Endpoint: Comparison between treatment groups in the change from baseline in the Hamilton Anxiety Rating Scale (HAM - A) total score at Week 10 after a single PAP cycle. PEX010: Phase IIb Trial Design Cohort 1 25mg PEX010 29 subjects PAP cycle (weeks 1 to 10) Dosing occurs on Day 14 Assessment at week 10 Screening Day - 28 to Day - 2 Long Term Follow Up (3 - month post week 10) Responders Non - Responders Cohort 2 10mg PEX010 29 subjects Cohort 3 1mg PEX010 29 subjects Additional PAP cycle (repeat visits 5 - 11) All non - responders will be offered PAP with 25mg PEX010 Long Term Follow Up (3 months post week 10 of second PAP cycle)

22 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature FDA Pre - IND Feedback Received HREC approved Phase IIb study in Adjustment Disorder ( announced March 6 ) Two trial sites selected Last patient Last visit for primary endpoint Top Line Results Planned End of Phase II/Type C meeting with FDA FDA IND for PhIII registrational trials 6 8 2 5 7 Anticipated Developmental Pathway to Approval 1 Q2 2023 H2 2025 H1 2026 Q1 2024 Q3 2024 1 Based on currently anticipated study recruitment rate Q4 2024 First patient Randomised 4 23 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature Alcohol Use Disorder (AUD) and Other Substance Use Disorders (SUDs)

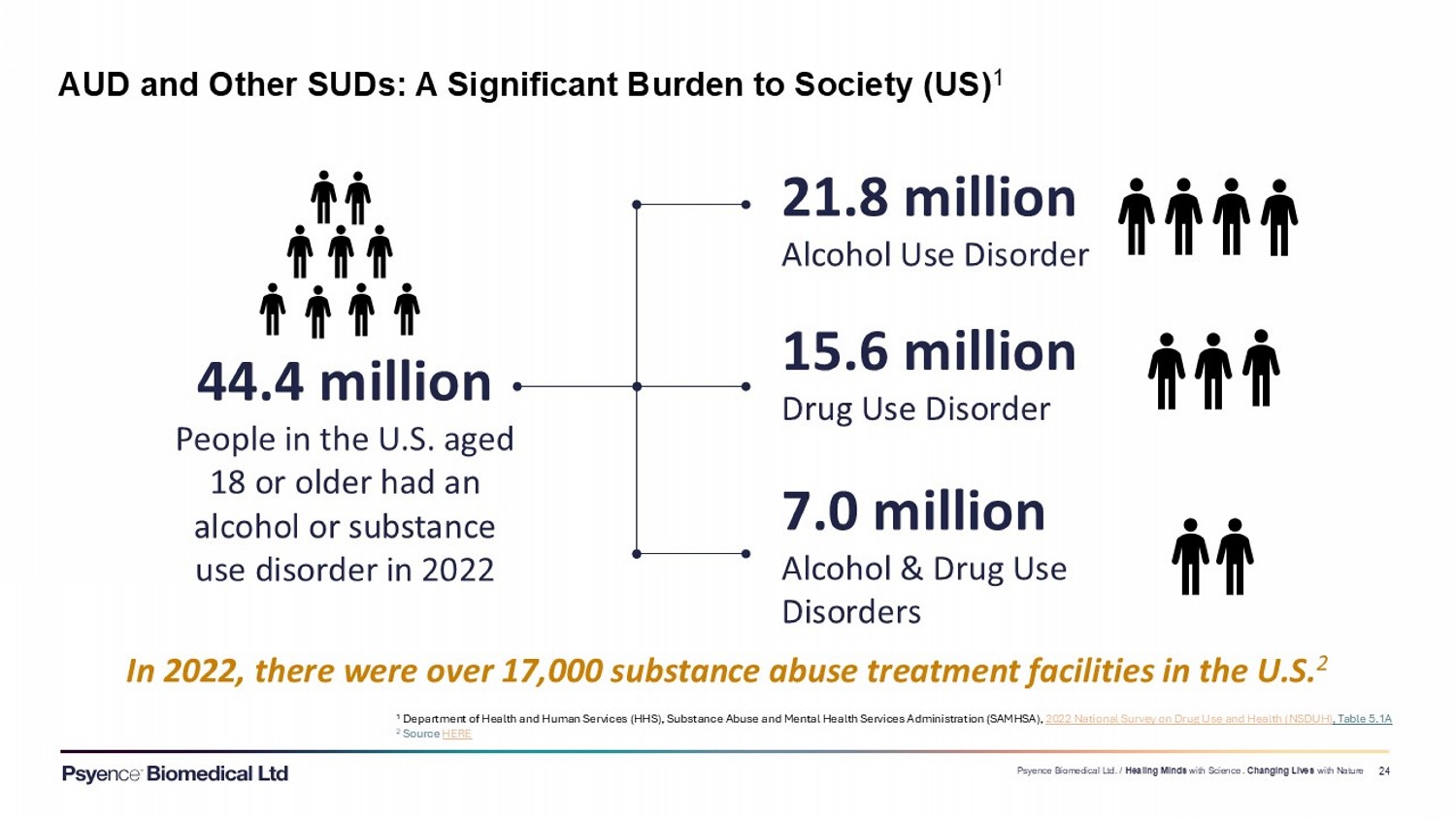
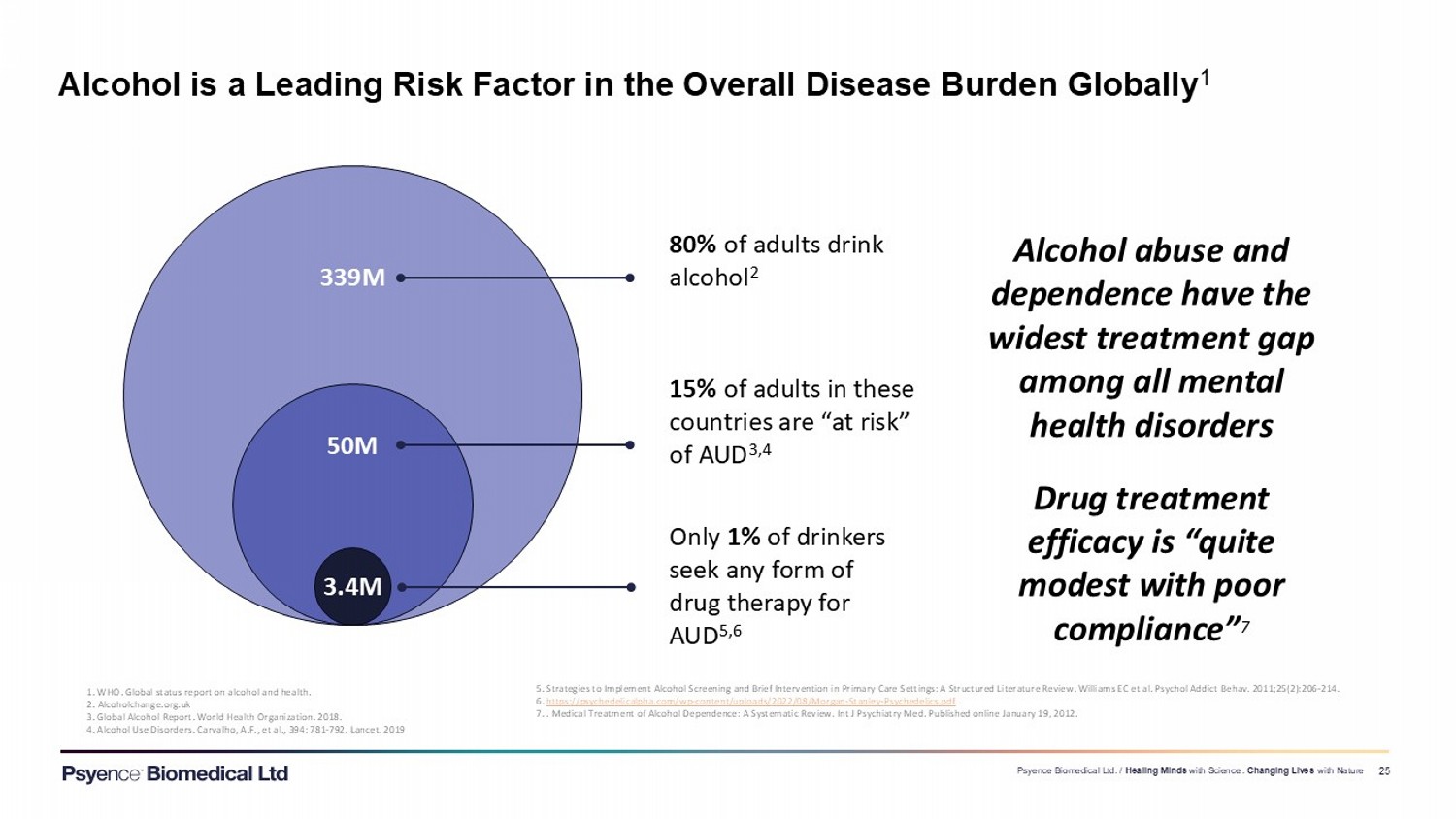
24 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature AUD and Other SUDs: A Significant Burden to Society (US) 1 44.4 million People in the U.S. aged 18 or older had an alcohol or substance use disorder in 2022 21.8 million Alcohol Use Disorder 15.6 million Drug Use Disorder 7.0 million Alcohol & Drug Use Disorders 1 Department of Health and Human Services (HHS), Substance Abuse and Mental Health Services Administration (SAMHSA), 2022 National Survey on Drug Use and Health (NSDUH) , Table 5.1A 2 Source HERE In 2022, there were over 17,000 substance abuse treatment facilities in the U.S. 2 25 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature Alcohol is a Leading Risk Factor in the Overall Disease Burden Globally 1 339M 50M 3.4M 80% of adults drink alcohol 2 15% of adults in these countries are “at risk” of AUD 3,4 Only 1% of drinkers seek any form of drug therapy for AUD 5,6 5. Strategies to Implement Alcohol Screening and Brief Intervention in Primary Care Settings: A Structured Literature Review. Wi lliams EC et al. Psychol Addict Behav . 2011;25(2):206 - 214. 6. https://psychedelicalpha.com/wp - content/uploads/2022/08/Morgan - Stanley - Psychedelics.pdf 7. . Medical Treatment of Alcohol Dependence: A Systematic Review. Int J Psychiatry Med. Published online January 19, 2012. 1. WHO. Global status report on alcohol and health. 2 . Alcoholchange.org.uk 3. Global Alcohol Report. World Health Organization. 2018. 4. Alcohol Use Disorders. Carvalho, A.F., et al., 394: 781 - 792. Lancet. 2019 Alcohol abuse and dependence have the widest treatment gap among all mental health disorders Drug treatment efficacy is “quite modest with poor compliance” 7 26 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature Prior Academic Studies Provide Strong Scientific Rationale for Continued Development of Psilocybin for SUDs 2022 2022 2023


27 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature A Vertically Integrated Psychedelic Biopharma Company Worldwide exclusive licensing agreement with PsyLabs creates consistent supply of nature - derived (non - synthetic) psilocybin for Psyence Biomed’s AUD clinical trial Strategic investment in PsyLabs transformed Psyence Biomed into a vertically integrated developer of psilocybin - based therapeutics PsyLabs develops and produces GMP - compliant, pharmaceutical - grade natural psychedelics for applications in mental health and well - being PsyLabs is federally licensed to produce psilocybin, DMT, Ibogaine and Mescaline for research, clinical trials and commercial production PsyLabs ’ fully operational, on - site lab is ISO22000 certified and audited by the British Standards Institution 28 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature Our intellectual property strategy is built around the following objectives: Ensuring that we have freedom to operate (free of infringing third party patents) to pursue our commercial objectives in our key markets • We conduct ongoing patent freedom to operate studies and watching searches Establishing barriers to entry (patents and regulatory) around our key intellectual property assets • Ensuring that we own the rights arising from our drug product development work and clinical trials • Filing and prosecuting to grant patent applications on key novel and innovative features of our drug product development work and clinical trials • Establishing data and market exclusivity (to the greatest extent possible) arising from a successful clinical trial Psyence’s Intellectual Property within Drug development


29 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature Source : S&P CapitalIQ , Company Filings 1. Market data valuations reflective as of the close on 11 / 1 / 2024 , the valuations listed on this slide reflect valuations of certain companies operating in a similar field as Psyence . Please note that such valuations are for informational purposes only and do not purport to reflect the real, assumed or potential valuation of the combined company . The should not be relied upon in making an investment decision with respect to the potential purchase of the combined company’s securities and all individuals should refer to the disclaimers and the summary of risks related to these valuations on slides 3 and 27 Business Descriptions Market Cap Ticker Company GH Research PLC, a clinical - stage biopharmaceutical company, engages in developing various therapies to treat psychiatric and ne urological disorders. The company develops 5 - Methoxy - N,N - Dimethyltryptamine (5 - MeO - DMT) therapies for the treatment of patients with treatment - resistant depressio n (TRD). Its lead program is GH001, an inhalable mebufotenin product candidate that has completed two Phase 1 clinical trials and Phase 1/2 clinical trial in patients with TRD. $422m NasdaqGM : GHRS GH Research PLC COMPASS Pathways plc operates as a mental health care company primarily in the United Kingdom and the United States. It devel ops COMP360, a psilocybin therapy that is in Phase III clinical trials for the treatment of treatment resistant depression; and is in Phase II clinical trials for the treatment of post - traumatic stress disorder and anorexia nervosa. $319m NasdaqGS : CMPS COMPASS Pathways plc ATAI Life Sciences N.V. develops various therapeutics to treat depression, anxiety, addiction, and other mental health disord ers . Its principal clinical programs include RL - 007, a pro - cognitive neuromodulator for cognitive impairment associated with schizophrenia; GRX - 917, a deuterated etifoxine f or anxiety disorders; VLS - 01 a N,N - Dimethyltryptamine for treatment resistant depression (TRD); DMX - 1002, an oral formulation of ibogaine, a cholinergic, glutamate rgic and monoaminergic receptor modulator that is a naturally occurring psychedelic product isolated from a West African shrub for the treatment of opioid us e d isorders (OUD); and EMP - 01, an oral formulation of an MDMA derivative being developed for the treatment of post - traumatic stress disorder. $186m NasdaqGM : ATAI Atai Life Sciences N.V. MindMed is a clinical - stage biopharmaceutical company developing novel product candidates to treat brain health disorders. Our mission is to be the global leader in the development and delivery of treatments that unlock new opportunities to improve patient outcomes. We are developing a pipelin e o f innovative product candidates, with and without acute perceptual effects, targeting neurotransmitter pathways that play key roles in brain health disorders. $495m NasdaqGS : MNMD MindMed Bright Minds Biosciences is a biotechnology company developing innovative treatments for patients with neurological and psych iat ric disorders. Our pipeline includes novel compounds targeting key receptors in the brain to address conditions with high unmet medical need, including epilepsy, dep ression, and other CNS disorders. Bright Minds is focused on delivering breakthrough therapies that can transform patients’ lives. $339m NasdaqCM : DRUG Bright Minds Biosciences Cybin Inc., a clinical - stage biopharmaceutical company, focuses on developing psychedelic - based therapeutics. The company’s developme nt pipeline includes CYB003, a deuterated psilocybin analog , which is in Phase 1/2a clinical trial to treat major depressive and alcohol use disorders; CYB004, a deuterated dimethyltry pta mine, which is in Phase 1 clinical trial for treating generalized anxiety disorders; and CYB005, a phenethylamine derivative, which in pr ecl inical stage to treat neuroinflammation. $202m NYSEAM: CYBN Cybin Inc. Psyence Biomed Represents a Value Driven Opportunity Versus Peer Companies (1)

30 Psyence Biomedical Ltd. / Healing Minds with Science .

Changing Lives with Nature Synthetic Nature derived Psilocybin Other psychedelics Ayahuasca, Ibogaine Significant Unexplored White Space Exists in the Area of Nature Derived Psilocybin MDMA, Ketamine, LSD Psilocybin Psilocybin 31 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature Dr. Neil Maresky CEO Trained MD in Emergency Medicine and Family Practice VP Scientific Affairs AstraZeneca Canada Inc, Previously at Wyeth, Bayer 25+ years of experience leading Research and Development for Big Pharma Jody Aufrichtig Executive Chairman Chartered Accountant (ex EY) with 25 years private equity experience Corporate governance and listed company experience Ex Canopy Growth CEO Africa Multiple award - winning entrepreneur and business builder creating significant shareholder value in various industries Founded 19 companies over 25 years Awarded Entrepreneur of the year 2023 Warwick Corden - Lloyd Chief Financial Officer Chartered Accountant and Certified Project Manager with over 20 years of experience in the UK, the USA, and South Africa Experienced in IPOs, capital raises, M&A, regulatory, compliance and corporate governance Former Head of Financial Accounting at Capitec Bank Former Senior Financial Manager at Bank of New York Mellon Taryn Vos General Counsel Corporate and commercial attorney (associate (M&A) at Cliffe Dekker Hofmeyr Inc). Formerly Head of Legal (AMEA) for Solar Capital (Pty) Ltd and Business Development (Legal) for Phelan Energy Group Ltd Former Head of Legal for Canopy Growth Africa, then a subsidiary of Canopy Growth Corp Dr. Clive Ward - Able Medical Director Pharmacist and physician with >30 years of international pharmaceutical experience in R&D and Commercialization of over 16 product launches Experience at the executive level in global and affiliate roles for large pharmaceutical and biotechnology companies as well as smaller start - ups Proven Track Record in Innovation with Extensive Pharma Development Experience Psyence Biomedical Ltd Management Team Mary - Elizabeth Gifford Chief of Global Impact Chair of the Global Wellness Institute’s Psychedelics & Healing Initiative Joined Psyence from the DC nonprofit, The Center for Mind - Body Medicine Co - author of the Nature vs.

Synthetic Psilocybin chapter in the recent Springer Nature textbook on therapeutic mushrooms Regularly publishes on palliative psilocybin policy issues Note: company logos indicate prior experience 32 Psyence Biomedical Ltd. / Healing Minds with Science . Changing Lives with Nature Dr. Neil Maresky CEO Trained MD in Emergency Medicine and Family Practice VP Scientific Affairs AstraZeneca Canada Inc, Previously at Wyeth, Bayer 25+ years of experience leading Research and Development for Big Pharma Jody Aufrichtig Executive Chairman Chartered Accountant (ex EY) with 25 years private equity experience Corporate governance and listed company experience Ex Canopy Growth CEO Africa Multiple award - winning entrepreneur and business builder creating significant shareholder value in various industries Founded 19 companies over 25 years Awarded Entrepreneur of the year 2023 Marc Balkin Board Member Partner, DiGame Former Partner, Hasso Plattner Ventures Served on investment committees of Enablis , First National Bank Vumela FutureMaker , Alithea IDF Newcourt Board Member Dr. Seth Feuerstein, MD, JD Board Member Christopher Bull Board Member Internationally top - rated Patent and Technology Lawyer Investor in several successful start - up businesses (Pharma, biotech, chemical processing) Former Chairman of Venture Capital Fund and International IP firm Proven Track Record in Innovation with Extensive Operating Experience Psyence Biomed Board of Directors Yale School of Medicine Lecturer in Psychiatry Faculty Advisor Innovation and Entrepreneurship Assistant Clinical Professor of Psychiatry Note: company logos indicate prior experience

Company Highlights Phase IIb program, 25mg psilocybin in conjunction with proprietary psychotherapy, being evaluated for Adjustment Disorder in cancer in the Palliative Care context Near - term value - driving catalysts, including first patient randomized in Phase IIb trial in Australia (anticipated in Q4 2024) Leveraging benefits of nature - derived psilocybin, including potential synergistic effects of similar acting molecules not present in synthetic psilocybin* Seasoned leadership team and Board with significant expertise in successful drug development Potential regulatory market and data exclusivity as a New Medicinal Entity (NME) * https://www.nature.com/articles/s41380 - 024 - 02477 - w Acquisition of PsyLabs stake provides vertical integration to support development of second indication, Alcohol Use Disorder