| ☒ |
QUARTERLY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
|
| ☐ |
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF SECURITIES EXCHANGE ACT OF 1934
|
|
Delaware
|
33-0336973
|
|
|
(State or other jurisdiction of incorporation or organization)
|
(IRS Employer Identification No.)
|
|
2855 Gazelle Court, Carlsbad, California
|
92010
|
|
|
(Address of Principal Executive Offices)
|
(Zip Code)
|
|
Title of each class
|
Trading symbol
|
Name of each exchange on which registered
|
||
|
Common Stock, $.001 Par Value
|
“IONS”
|
The Nasdaq Stock Market LLC
|
|
Large Accelerated Filer ☒
|
Accelerated Filer ☐
|
|
Non-accelerated Filer ☐
|
Smaller Reporting Company ☐
|
|
Emerging Growth Company ☐
|
|
PART I
|
FINANCIAL INFORMATION
|
|
|
ITEM 1:
|
Financial Statements:
|
|
|
Condensed Consolidated Balance
Sheets as of September 30, 2023 (unaudited) and December 31, 2022
|
3
|
|
|
Condensed Consolidated
Statements of Operations for the three and nine months ended September 30, 2023 and 2022 (unaudited)
|
4
|
|
|
Condensed Consolidated
Statements of Comprehensive Loss for the three and nine months ended September 30, 2023 and 2022 (unaudited)
|
5
|
|
|
Condensed Consolidated Statements of
Stockholders’ Equity for the three and nine months ended September 30, 2023 and 2022 (unaudited)
|
6
|
|
|
Condensed Consolidated Statements of
Cash Flows for the nine months ended September 30, 2023 and 2022 (unaudited)
|
8
|
|
|
9
|
||
|
ITEM 2:
|
||
|
22
|
||
|
24
|
||
|
24
|
||
|
31
|
||
|
ITEM 3:
|
33
|
|
|
ITEM 4:
|
33
|
|
|
PART II
|
33
|
|
|
ITEM 1:
|
33
|
|
|
ITEM 1A:
|
33
|
|
|
ITEM 2:
|
51
|
|
|
ITEM 3:
|
51
|
|
|
ITEM 4:
|
51
|
|
|
ITEM 5:
|
51
|
|
|
ITEM 6:
|
52
|
|
|
53
|
||
|
September 30,
2023
|
December 31,
2022
|
|||||||
|
(unaudited)
|
||||||||
|
ASSETS
|
||||||||
|
Current assets:
|
||||||||
|
Cash and cash equivalents
|
$
|
352,060
|
$
|
276,472
|
||||
|
Short-term investments
|
1,883,522
|
1,710,397
|
||||||
|
Contracts receivable
|
142,359
|
25,538
|
||||||
|
Inventories
|
25,634
|
22,033
|
||||||
|
Other current assets
|
181,075
|
168,254
|
||||||
|
Total current assets
|
2,584,650
|
2,202,694
|
||||||
|
Property, plant and equipment, net
|
70,928
|
74,294
|
||||||
|
Right-of-use assets
|
174,310
|
181,544
|
||||||
|
Deposits and other assets
|
104,083
|
75,344
|
||||||
|
Total assets
|
$
|
2,933,971
|
$
|
2,533,876
|
||||
|
LIABILITIES AND STOCKHOLDERS’ EQUITY
|
||||||||
|
Current liabilities:
|
||||||||
|
Accounts payable
|
$
|
5,615
|
$
|
17,921
|
||||
|
Accrued compensation
|
33,781
|
49,178
|
||||||
|
Accrued liabilities
|
118,968
|
140,101
|
||||||
|
Income taxes payable
|
31,070
|
6,249
|
||||||
|
Current portion of deferred contract revenue
|
204,824
|
90,577
|
||||||
|
Other current liabilities
|
9,952
|
7,535
|
||||||
|
Total current liabilities
|
404,210
|
311,561
|
||||||
|
Long-term deferred contract revenue
|
249,272
|
287,768
|
||||||
|
1.75 percent convertible senior notes, net
|
561,609
|
—
|
||||||
|
0 percent convertible senior notes, net
|
624,594
|
622,242
|
||||||
|
0.125 percent convertible senior notes, net
|
44,287
|
544,504
|
||||||
|
Liability related to sale of future royalties, net
|
512,700
|
—
|
||||||
|
Long-term lease liabilities
|
173,038
|
178,941
|
||||||
|
Long-term obligations
|
48,801
|
15,973
|
||||||
|
Total liabilities
|
2,618,511
|
1,960,989
|
||||||
|
Stockholders’ equity:
|
||||||||
|
Common stock, $0.001 par value; 300,000,000 shares authorized, 143,393,493 and 142,057,736
shares issued and outstanding at September 30, 2023 (unaudited) and December 31, 2022, respectively
|
143
|
142
|
||||||
|
Additional paid-in capital
|
2,148,002
|
2,059,850
|
||||||
|
Accumulated other comprehensive loss
|
(46,037
|
)
|
(57,480
|
)
|
||||
|
Accumulated deficit
|
(1,786,648
|
)
|
(1,429,625
|
)
|
||||
|
Total stockholders’ equity
|
315,460
|
572,887
|
||||||
|
Total liabilities and stockholders’ equity
|
$
|
2,933,971
|
$
|
2,533,876
|
||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Revenue:
|
||||||||||||||||
|
Commercial revenue:
|
||||||||||||||||
|
SPINRAZA royalties
|
$
|
67,253
|
$
|
61,647
|
$
|
178,511
|
$
|
175,092
|
||||||||
|
Other commercial revenue
|
16,828
|
10,763
|
51,235
|
47,787
|
||||||||||||
|
Total commercial revenue
|
84,081
|
72,410
|
229,746
|
222,879
|
||||||||||||
|
Research and development revenue:
|
||||||||||||||||
|
Collaborative agreement revenue
|
44,167
|
69,250
|
173,513
|
157,282
|
||||||||||||
|
Eplontersen joint development revenue
|
15,959
|
18,107
|
59,883
|
55,317
|
||||||||||||
|
Total research and development revenue
|
60,126
|
87,357
|
233,396
|
212,599
|
||||||||||||
|
Total revenue
|
144,207
|
159,767
|
463,142
|
435,478
|
||||||||||||
|
Expenses:
|
||||||||||||||||
|
Cost of sales
|
2,191
|
1,515
|
6,071
|
10,430
|
||||||||||||
|
Research, development and patent
|
215,330
|
182,990
|
643,070
|
524,875
|
||||||||||||
|
Selling, general and administrative
|
69,951
|
34,416
|
161,608
|
102,345
|
||||||||||||
|
Total operating expenses
|
287,472
|
218,921
|
810,749
|
637,650
|
||||||||||||
|
Loss from operations
|
(143,265
|
)
|
(59,154
|
)
|
(347,607
|
)
|
(202,172
|
)
|
||||||||
|
Other income (expense):
|
||||||||||||||||
|
Investment income
|
23,935
|
7,524
|
63,355
|
13,447
|
||||||||||||
|
Interest expense
|
(4,203
|
)
|
(2,139
|
)
|
(8,102
|
)
|
(6,391
|
)
|
||||||||
|
Interest expense related to sale of future royalties
|
(17,779
|
)
|
—
|
(50,948
|
)
|
—
|
||||||||||
|
Gain (loss) on investments
|
(1,943
|
)
|
2,347
|
(1,753
|
)
|
(10,616
|
)
|
|||||||||
|
Other income (expense)
|
2,447
|
4,713
|
13,857
|
(7,923
|
)
|
|||||||||||
|
Loss before income tax expense
|
(140,808
|
)
|
(46,709
|
)
|
(331,198
|
)
|
(213,655
|
)
|
||||||||
|
Income tax expense
|
(6,602
|
)
|
(283
|
)
|
(25,825
|
)
|
(3,637
|
)
|
||||||||
|
Net loss
|
$
|
(147,410
|
)
|
$
|
(46,992
|
)
|
$
|
(357,023
|
)
|
$
|
(217,292
|
)
|
||||
|
Basic and diluted net loss per share
|
$
|
(1.03
|
)
|
$
|
(0.33
|
)
|
$
|
(2.50
|
)
|
$
|
(1.53
|
)
|
||||
|
Shares used in computing basic and diluted net loss per share
|
143,317
|
141,950
|
143,052
|
141,782
|
||||||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Net loss
|
$
|
(147,410
|
)
|
$
|
(46,992
|
)
|
$
|
(357,023
|
)
|
$
|
(217,292
|
)
|
||||
|
Unrealized gains (losses) on debt securities, net of tax
|
5,029
|
(8,734
|
)
|
11,421
|
(29,508
|
)
|
||||||||||
|
Currency translation adjustment
|
(153
|
)
|
(399
|
)
|
22
|
(964
|
)
|
|||||||||
|
Comprehensive loss
|
$
|
(142,534
|
)
|
$
|
(56,125
|
)
|
$
|
(345,580
|
)
|
$
|
(247,764
|
)
|
||||
|
Common Stock
|
Additional
|
Accumulated Other
|
Accumulated
|
Total
Stockholders’
|
||||||||||||||||||||
|
Description
|
Shares
|
Amount
|
Paid in Capital
|
Comprehensive Loss
|
Deficit
|
Equity
|
||||||||||||||||||
|
Balance at June 30, 2022
|
141,831
|
$
|
142
|
$
|
2,008,794
|
$
|
(54,007
|
)
|
$
|
(1,330,203
|
)
|
$
|
624,726
|
|||||||||||
|
Net loss
|
—
|
—
|
—
|
—
|
(46,992
|
)
|
(46,992
|
)
|
||||||||||||||||
|
Change in unrealized losses, net of tax
|
—
|
—
|
—
|
(8,734
|
)
|
—
|
(8,734
|
)
|
||||||||||||||||
|
Foreign currency translation
|
—
|
—
|
—
|
(399
|
)
|
—
|
(399
|
)
|
||||||||||||||||
|
Issuance of common stock in connection with employee stock plans
|
203
|
—
|
2,567
|
—
|
—
|
2,567
|
||||||||||||||||||
|
Stock-based compensation expense
|
—
|
—
|
23,837
|
—
|
—
|
23,837
|
||||||||||||||||||
|
Payments of tax withholdings related to vesting of employee stock awards and exercise of employee
stock options
|
(17
|
)
|
—
|
(644
|
)
|
—
|
—
|
(644
|
)
|
|||||||||||||||
|
Balance at September 30, 2022
|
142,017
|
$
|
142
|
$
|
2,034,554
|
$
|
(63,140
|
)
|
$
|
(1,377,195
|
)
|
$
|
594,361
|
|||||||||||
|
Balance at June 30, 2023
|
143,167
|
$
|
143
|
$
|
2,118,309
|
$
|
(50,913
|
)
|
$
|
(1,639,238
|
)
|
$
|
428,301
|
|||||||||||
|
Net loss
|
—
|
—
|
—
|
—
|
(147,410
|
)
|
(147,410
|
)
|
||||||||||||||||
|
Change in unrealized losses, net of tax
|
—
|
—
|
—
|
5,029
|
—
|
5,029
|
||||||||||||||||||
|
Foreign currency translation
|
—
|
—
|
—
|
(153
|
)
|
—
|
(153
|
)
|
||||||||||||||||
|
Issuance of common stock in connection with employee stock plans
|
226
|
—
|
3,729
|
—
|
—
|
3,729
|
||||||||||||||||||
|
Stock-based compensation expense
|
—
|
—
|
25,964
|
—
|
—
|
25,964
|
||||||||||||||||||
|
Balance at September 30, 2023
|
143,393
|
$
|
143
|
$
|
2,148,002
|
$
|
(46,037
|
)
|
$
|
(1,786,648
|
)
|
$
|
315,460
|
|||||||||||
|
Common Stock
|
Additional
|
Accumulated Other
|
Accumulated
|
Total
Stockholders’
|
||||||||||||||||||||
|
Description
|
Shares
|
Amount
|
Paid in Capital
|
Comprehensive Loss
|
Deficit
|
Equity
|
||||||||||||||||||
|
Balance at December 31, 2021
|
141,210
|
$
|
141
|
$
|
1,964,167
|
$
|
(32,668
|
)
|
$
|
(1,159,903
|
)
|
$
|
771,737
|
|||||||||||
|
Net loss
|
—
|
—
|
—
|
—
|
(217,292
|
)
|
(217,292
|
)
|
||||||||||||||||
|
Change in unrealized losses, net of tax
|
—
|
—
|
—
|
(29,508
|
)
|
—
|
(29,508
|
)
|
||||||||||||||||
|
Foreign currency translation
|
—
|
—
|
—
|
(964
|
)
|
—
|
(964
|
)
|
||||||||||||||||
|
Issuance of common stock in connection with employee stock plans
|
1,138
|
1
|
6,029
|
—
|
—
|
6,030
|
||||||||||||||||||
|
Stock-based compensation expense
|
—
|
—
|
74,575
|
—
|
—
|
74,575
|
||||||||||||||||||
|
Payments of tax withholdings related to vesting of employee stock awards and exercise of employee
stock options
|
(331
|
)
|
—
|
(10,217
|
)
|
—
|
—
|
(10,217
|
)
|
|||||||||||||||
|
Balance at September 30, 2022
|
142,017
|
$
|
142
|
$
|
2,034,554
|
$
|
(63,140
|
)
|
$
|
(1,377,195
|
)
|
$
|
594,361
|
|||||||||||
|
Balance at December 31, 2022
|
142,058
|
$
|
142
|
$
|
2,059,850
|
$
|
(57,480
|
)
|
$
|
(1,429,625
|
)
|
$
|
572,887
|
|||||||||||
|
Net loss
|
—
|
—
|
—
|
—
|
(357,023
|
)
|
(357,023
|
)
|
||||||||||||||||
|
Change in unrealized gains, net of tax
|
—
|
—
|
—
|
11,421
|
—
|
11,421
|
||||||||||||||||||
|
Foreign currency translation
|
—
|
—
|
—
|
22
|
—
|
22
|
||||||||||||||||||
|
Issuance of common stock in connection with employee stock plans
|
1,335
|
1
|
8,679
|
—
|
—
|
8,680
|
||||||||||||||||||
|
Stock-based compensation expense
|
—
|
—
|
79,473
|
—
|
—
|
79,473
|
||||||||||||||||||
|
Balance at September 30, 2023
|
143,393
|
$
|
143
|
$
|
2,148,002
|
$
|
(46,037
|
)
|
$
|
(1,786,648
|
)
|
$
|
315,460
|
|||||||||||
|
Nine Months Ended
September 30,
|
||||||||
|
2023
|
2022
|
|||||||
|
Operating activities:
|
||||||||
|
Net loss
|
$
|
(357,023
|
)
|
$
|
(217,292
|
)
|
||
|
Adjustments to reconcile net loss to net cash used in operating activities:
|
||||||||
|
Depreciation
|
7,748
|
11,301
|
||||||
|
Amortization of right-of-use operating lease assets
|
7,234
|
1,970
|
||||||
|
Amortization of other assets
|
1,904
|
1,805
|
||||||
|
Amortization of premium (discount) on investments, net
|
(20,396
|
)
|
9,072
|
|||||
|
Amortization of debt issuance costs
|
4,666
|
4,035
|
||||||
|
Non-cash royalty revenue related to sale of royalties
|
(27,814
|
)
|
—
|
|||||
|
Non-cash interest related to sale of future royalties
|
50,541
|
—
|
||||||
|
Stock-based compensation expense
|
79,473
|
74,575
|
||||||
|
Loss on investments
|
1,429
|
228
|
||||||
|
Gain on early retirement of debt
|
(13,389
|
)
|
—
|
|||||
|
Non-cash losses related to disposal of property, plant and equipment
|
14,646
|
528
|
||||||
|
Non-cash losses related to other assets
|
849
|
1,155
|
||||||
|
Changes in operating assets and liabilities:
|
||||||||
|
Contracts receivable
|
(116,814
|
)
|
55,251
|
|||||
|
Inventories
|
(3,601
|
)
|
4,161
|
|||||
|
Other current and long-term assets
|
(18,325
|
)
|
(988
|
)
|
||||
|
Income taxes payable
|
24,821
|
(20
|
)
|
|||||
|
Accounts payable
|
(12,462
|
)
|
5,607
|
|||||
|
Accrued compensation
|
(15,397
|
)
|
(8,400
|
)
|
||||
|
Accrued liabilities and other current liabilities
|
(24,219
|
)
|
38,263
|
|||||
|
Deferred contract revenue
|
75,751
|
(55,426
|
)
|
|||||
|
Net cash used in operating activities
|
(340,378
|
)
|
(74,175
|
)
|
||||
|
Investing activities:
|
||||||||
|
Purchases of short-term investments
|
(1,353,100
|
)
|
(1,223,791
|
)
|
||||
|
Proceeds from sale of short-term investments
|
1,193,724
|
764,101
|
||||||
|
Purchases of property, plant and equipment
|
(24,624
|
)
|
(11,582
|
)
|
||||
|
Acquisition of licenses and other assets, net
|
(3,414
|
)
|
(3,511
|
)
|
||||
|
Net cash used in investing activities
|
(187,414
|
)
|
(474,783
|
)
|
||||
|
Financing activities:
|
||||||||
|
Proceeds from equity, net
|
8,680
|
6,030
|
||||||
|
Payments of tax withholdings related to vesting of employee stock awards and exercise of
employee stock options
|
—
|
(10,217
|
)
|
|||||
|
Proceeds from issuance of 1.75 percent convertible senior notes
|
575,000
|
—
|
||||||
|
1.75 percent convertible senior notes issuance costs
|
(14,175
|
)
|
—
|
|||||
|
Repurchase of $504.4 million principal amount of 0.125 percent convertible senior notes
|
(487,943
|
)
|
—
|
|||||
|
Proceeds from sale of future royalties
|
500,000
|
—
|
||||||
|
Payments of transaction costs related to sale of future royalties
|
(10,434
|
)
|
—
|
|||||
|
Proceeds from real estate transaction
|
32,352
|
—
|
||||||
|
Principal payments on mortgage debt
|
(122
|
)
|
(89
|
)
|
||||
|
Net cash provided by (used in) financing activities
|
603,358
|
(4,276
|
)
|
|||||
|
Effects of exchange rates on cash
|
22
|
(964
|
)
|
|||||
|
Net increase (decrease) in cash and cash equivalents
|
75,588
|
(554,198
|
)
|
|||||
|
Cash and cash equivalents at beginning of period
|
276,472
|
869,191
|
||||||
|
Cash and cash equivalents at end of period
|
$
|
352,060
|
$
|
314,993
|
||||
|
Supplemental disclosures of cash flow information:
|
||||||||
|
Interest paid
|
$
|
952
|
$
|
2,204
|
||||
|
Income taxes paid
|
$
|
714
|
$
|
2
|
||||
|
Supplemental disclosures of non-cash investing and financing activities:
|
||||||||
|
Amounts accrued for capital and patent expenditures
|
$
|
341
|
$
|
3,032
|
||||
|
Right-of-use assets obtained in exchange for lease liabilities
|
$
|
—
|
$
|
657
|
||||
|
September 30, 2023
|
December 31, 2022
|
|||||||
|
Raw materials:
|
||||||||
|
Raw materials - clinical
|
$
|
18,427
|
$
|
17,061
|
||||
|
Raw materials - commercial
|
4,380
|
2,699
|
||||||
|
Total raw materials
|
22,807
|
19,760
|
||||||
|
Work in process
|
2,651
|
2,109
|
||||||
|
Finished goods
|
176
|
164
|
||||||
|
Total inventory
|
$
|
25,634
|
$
|
22,033
|
||||
|
September 30, 2023
|
December 31, 2022
|
|||||||
|
Clinical development expenses
|
$
|
89,217
|
$
|
116,460
|
||||
|
In-licensing expenses
|
7,560
|
7,945
|
||||||
|
Commercial expenses
|
8,575
|
3,498
|
||||||
|
Other miscellaneous expenses
|
13,616
|
12,198
|
||||||
|
Total accrued liabilities
|
$
|
118,968
|
$
|
140,101
|
||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Revenue:
|
||||||||||||||||
|
Commercial revenue:
|
||||||||||||||||
|
SPINRAZA royalties
|
$
|
67,253
|
$
|
61,647
|
$
|
178,511
|
$
|
175,092
|
||||||||
|
Other commercial revenue:
|
||||||||||||||||
|
TEGSEDI and WAYLIVRA revenue, net
|
8,286
|
5,920
|
25,420
|
22,467
|
||||||||||||
|
Licensing and other royalty revenue
|
8,542
|
4,843
|
25,815
|
25,320
|
||||||||||||
|
Total other commercial revenue
|
16,828
|
10,763
|
51,235
|
47,787
|
||||||||||||
|
Total commercial revenue
|
84,081
|
72,410
|
229,746
|
222,879
|
||||||||||||
|
Research and development revenue:
|
||||||||||||||||
|
Collaborative agreement revenue
|
44,167
|
69,250
|
173,513
|
157,282
|
||||||||||||
|
Eplontersen joint development revenue
|
15,959
|
18,107
|
59,883
|
55,317
|
||||||||||||
|
Total research and development revenue
|
60,126
|
87,357
|
233,396
|
212,599
|
||||||||||||
|
Total revenue
|
$
|
144,207
|
$
|
159,767
|
$
|
463,142
|
$
|
435,478
|
||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Revenue from our relationship with AstraZeneca
|
$
|
15,959
|
$
|
18,107
|
$
|
99,885
|
$
|
55,718
|
||||||||
|
Percentage of total revenue
|
11
|
%
|
11
|
%
|
22
|
%
|
13
|
%
|
||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Revenue from our relationship with Biogen
|
$
|
94,695
|
$
|
88,959
|
$
|
262,599
|
$
|
259,713
|
||||||||
|
Percentage of total revenue
|
66
|
%
|
56
|
%
|
57
|
%
|
60
|
%
|
||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Revenue from our relationship with GSK
|
$
|
—
|
$
|
—
|
$
|
15,000
|
$
|
—
|
||||||||
|
Percentage of total revenue
|
0
|
%
|
0
|
%
|
3
|
%
|
0
|
%
|
||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Revenue from our relationship with Novartis
|
$
|
1,908
|
$
|
—
|
$
|
2,023
|
$
|
—
|
||||||||
|
Percentage of total revenue
|
1
|
%
|
0
|
%
|
0
|
%
|
0
|
%
|
||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
R&D revenue
|
$
|
8,858
|
$
|
41,504
|
$
|
16,979
|
$
|
65,360
|
||||||||
|
Percentage of total revenue
|
6
|
%
|
26
|
%
|
4
|
%
|
15
|
%
|
||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
TEGSEDI revenue from our distribution agreement with Sobi in North America
|
$
|
794
|
$
|
1,008
|
$
|
2,189
|
$
|
3,443
|
||||||||
|
Percentage of total revenue
|
1
|
%
|
1
|
%
|
2
|
%
|
1
|
%
|
||||||||
| ● |
1.75 percent convertible senior notes, or 1.75% Notes;
|
| ● |
0 percent convertible senior notes, or 0% Notes;
|
| ● |
Note hedges related to the 0% Notes;
|
| ● |
0.125 percent convertible senior notes, or
0.125% Notes;
|
| ● |
Note hedges related to the 0.125% Notes;
|
| ● |
Dilutive stock options;
|
| ● |
Unvested restricted stock units, or RSUs;
|
| ● |
Unvested performance restricted stock units, or PRSUs; and
|
| ● |
Employee Stock Purchase Plan, or ESPP.
|
|
One year or less
|
72
|
%
|
||
|
After one year but within two years
|
20
|
%
|
||
|
After two years but within three and a half years
|
8
|
%
|
||
|
Total
|
100
|
%
|
|
Amortized
|
Gross Unrealized
|
Estimated
|
||||||||||||||
|
September 30, 2023
|
Cost
|
Gains
|
Losses
|
Fair Value
|
||||||||||||
|
Available-for-sale debt securities:
|
||||||||||||||||
|
Corporate debt securities (1)
|
$
|
535,408
|
$
|
13
|
$
|
(4,492
|
)
|
$
|
530,929
|
|||||||
|
Debt securities issued by U.S. government agencies
|
267,503
|
—
|
(1,429
|
)
|
266,074
|
|||||||||||
|
Debt securities issued by the U.S. Treasury (1)
|
520,013
|
1
|
(3,027
|
)
|
516,987
|
|||||||||||
|
Debt securities issued by states of the U.S. and political subdivisions of the states
|
23,906
|
—
|
(242
|
)
|
23,664
|
|||||||||||
|
Total debt securities with a maturity of one year or less
|
1,346,830
|
14
|
(9,190
|
)
|
1,337,654
|
|||||||||||
|
Corporate debt securities
|
195,674
|
183
|
(3,160
|
)
|
192,697
|
|||||||||||
|
Debt securities issued by U.S. government agencies
|
56,365
|
—
|
(507
|
)
|
55,858
|
|||||||||||
|
Debt securities issued by the U.S. Treasury
|
297,501
|
—
|
(3,364
|
)
|
294,137
|
|||||||||||
|
Debt securities issued by states of the U.S. and political subdivisions of the states
|
4,164
|
53
|
(51
|
)
|
4,166
|
|||||||||||
|
Total debt securities with a maturity of more than one year
|
553,704
|
236
|
(7,082
|
)
|
546,858
|
|||||||||||
|
Total available-for-sale debt securities
|
$
|
1,900,534
|
$
|
250
|
$
|
(16,272
|
)
|
$
|
1,884,512
|
|||||||
|
Equity securities:
|
||||||||||||||||
|
Publicly traded equity securities included in other current assets (2)
|
$
|
11,897
|
$
|
(1,252
|
)
|
$
|
(4,083
|
)
|
$
|
6,562
|
||||||
|
Privately held equity securities included in deposits and other assets (3)
|
23,115
|
25,001
|
(5,125
|
)
|
42,991
|
|||||||||||
|
Total equity securities
|
35,012
|
23,749
|
(9,208
|
)
|
49,553
|
|||||||||||
|
Total available-for-sale debt and equity securities
|
$
|
1,935,546
|
$
|
23,999
|
$
|
(25,480
|
)
|
$
|
1,934,065
|
|||||||
|
Amortized
|
Gross Unrealized
|
Estimated
|
||||||||||||||
|
December 31, 2022
|
Cost
|
Gains
|
Losses
|
Fair Value
|
||||||||||||
|
Available-for-sale debt securities:
|
||||||||||||||||
|
Corporate debt securities (1)
|
$
|
513,790
|
$
|
23
|
$
|
(4,365
|
)
|
$
|
509,448
|
|||||||
|
Debt securities issued by U.S. government agencies
|
133,585
|
—
|
(1,829
|
)
|
131,756
|
|||||||||||
|
Debt securities issued by the U.S. Treasury (1)
|
512,655
|
23
|
(5,124
|
)
|
507,554
|
|||||||||||
|
Debt securities issued by states of the U.S. and political subdivisions of the states
|
57,484
|
18
|
(686
|
)
|
56,816
|
|||||||||||
|
Other municipal debt securities
|
6,008
|
—
|
(14
|
)
|
5,994
|
|||||||||||
|
Total debt securities with a maturity of one year or less
|
1,223,522
|
64
|
(12,018
|
)
|
1,211,568
|
|||||||||||
|
Corporate debt securities
|
227,631
|
14
|
(10,143
|
)
|
217,502
|
|||||||||||
|
Debt securities issued by U.S. government agencies
|
34,339
|
—
|
(1,040
|
)
|
33,299
|
|||||||||||
|
Debt securities issued by the U.S. Treasury
|
245,030
|
—
|
(4,109
|
)
|
240,921
|
|||||||||||
|
Debt securities issued by states of the U.S. and political subdivisions of the states
|
18,314
|
116
|
(329
|
)
|
18,101
|
|||||||||||
|
Total debt securities with a maturity of more than one year
|
525,314
|
130
|
(15,621
|
)
|
509,823
|
|||||||||||
|
Total available-for-sale debt securities
|
$
|
1,748,836
|
$
|
194
|
$
|
(27,639
|
)
|
$
|
1,721,391
|
|||||||
|
Equity securities:
|
||||||||||||||||
|
Publicly traded equity securities included in other current assets (2)
|
$
|
11,897
|
$
|
—
|
$
|
(1,358
|
)
|
$
|
10,539
|
|||||||
|
Privately held equity securities included in deposits and other assets (3)
|
23,115
|
17,257
|
—
|
40,372
|
||||||||||||
|
Total equity securities
|
35,012
|
17,257
|
(1,358
|
)
|
50,911
|
|||||||||||
|
Total available-for-sale debt and equity securities
|
$
|
1,783,848
|
$
|
17,451
|
$
|
(28,997
|
)
|
$
|
1,772,302
|
|||||||
| (1) |
Includes investments classified
as cash equivalents in our condensed consolidated balance sheets.
|
| (2) |
Our publicly traded equity
securities are included in other current assets. We recognize publicly traded equity securities at fair value. In the nine months ended September 30, 2023, we recognized a $4.0
million unrealized loss in our condensed consolidated statements of operations related to a decrease in the fair value of our investments in publicly traded companies.
|
| (3) |
Our privately held equity securities are included in deposits and other assets. We recognize our privately held equity securities at cost minus impairments, plus or minus changes
resulting from observable price changes in orderly transactions for the identical or similar investment of the same issuer, which are Level 3 inputs.
In the nine months ended September 30, 2023, we recorded a net gain of $2.6 million in our condensed consolidated statements of
operations related to changes in the fair value of our investments in privately held companies.
|
|
Less than 12 Months of
Temporary Impairment
|
More than 12 Months of
Temporary Impairment
|
Total Temporary
Impairment
|
||||||||||||||||||||||||||
|
Number of
Investments
|
Estimated
Fair Value
|
Unrealized
Losses
|
Estimated
Fair Value
|
Unrealized
Losses
|
Estimated
Fair Value
|
Unrealized
Losses
|
||||||||||||||||||||||
|
Corporate debt securities
|
370
|
$
|
469,095
|
$
|
(2,390
|
)
|
$
|
207,073
|
$
|
(5,262
|
)
|
$
|
676,168
|
$
|
(7,652
|
)
|
||||||||||||
|
Debt securities issued by U.S. government agencies
|
98
|
293,408
|
(1,242
|
)
|
24,531
|
(694
|
)
|
317,939
|
(1,936
|
)
|
||||||||||||||||||
|
Debt securities issued by the U.S. Treasury
|
68
|
557,489
|
(3,420
|
)
|
225,634
|
(2,971
|
)
|
783,123
|
(6,391
|
)
|
||||||||||||||||||
|
Debt securities issued by states of the U.S. and political subdivisions of the states
|
85
|
11,644
|
(73
|
)
|
12,973
|
(220
|
)
|
24,617
|
(293
|
)
|
||||||||||||||||||
|
Total temporarily impaired securities
|
621
|
$
|
1,331,636
|
$
|
(7,125
|
)
|
$
|
470,211
|
$
|
(9,147
|
)
|
$
|
1,801,847
|
$
|
(16,272
|
)
|
||||||||||||
|
At
September 30, 2023
|
Quoted Prices in
Active Markets
(Level 1)
|
Significant Other
Observable Inputs
(Level 2)
|
||||||||||
|
Cash equivalents (1)
|
$
|
269,255
|
$
|
269,255
|
$
|
—
|
||||||
|
Corporate debt securities (2)
|
723,626
|
—
|
723,626
|
|||||||||
|
Debt securities issued by U.S. government agencies (3)
|
321,932
|
—
|
321,932
|
|||||||||
|
Debt securities issued by the U.S. Treasury (2)
|
811,124
|
811,124
|
—
|
|||||||||
|
Debt securities issued by states of the U.S. and political subdivisions of the states (2)
|
27,830
|
—
|
27,830
|
|||||||||
|
Publicly traded equity securities included in other current assets (4)
|
6,562
|
6,562
|
—
|
|||||||||
|
Total
|
$
|
2,160,329
|
$
|
1,086,941
|
$
|
1,073,388
|
||||||
|
At
December 31, 2022
|
Quoted Prices in
Active Markets
(Level 1)
|
Significant Other
Observable Inputs
(Level 2)
|
||||||||||
|
Cash equivalents (1)
|
$
|
211,655
|
$
|
211,655
|
$
|
—
|
||||||
|
Corporate debt securities (5)
|
726,950
|
—
|
726,950
|
|||||||||
|
Debt securities issued by U.S. government agencies (2)
|
165,055
|
—
|
165,055
|
|||||||||
|
Debt securities issued by the U.S. Treasury (2)
|
748,475
|
748,475
|
—
|
|||||||||
|
Debt securities issued by states of the U.S. and political subdivisions of the states (2)
|
74,917
|
—
|
74,917
|
|||||||||
|
Other municipal debt securities (2)
|
5,994
|
—
|
5,994
|
|||||||||
|
Publicly traded equity securities included in other current assets (4)
|
10,539
|
10,539
|
—
|
|||||||||
|
Total
|
$
|
1,943,585
|
$
|
970,669
|
$
|
972,916
|
||||||
| (1) |
Included in cash and cash
equivalents in our condensed consolidated balance sheets.
|
| (2) |
Included in short-term
investments in our condensed consolidated balance sheets.
|
| (3) |
$1.0 million was included in cash and cash equivalents, with the difference included in short-term investments, in our condensed consolidated balance
sheets.
|
| (4) |
Included in other current assets
in our condensed consolidated balance sheets.
|
| (5) |
$11.0 million was included in cash and cash equivalents, with the difference included in short-term investments, in our condensed consolidated
balance sheets.
|
|
Nine Months Ended
September 30,
|
||||||||
|
2023
|
2022
|
|||||||
|
Risk-free interest rate
|
3.7
|
%
|
1.9
|
%
|
||||
|
Dividend yield
|
0.0
|
%
|
0.0
|
%
|
||||
|
Volatility
|
47.1
|
%
|
54.9
|
%
|
||||
|
Expected life
|
6.3 years
|
6.3 years
|
||||||
|
Nine Months Ended
September 30,
|
||||||||
|
2023
|
2022
|
|||||||
|
Risk-free interest rate
|
3.8
|
%
|
2.9
|
%
|
||||
|
Dividend yield
|
0.0
|
%
|
0.0
|
%
|
||||
|
Volatility
|
53.0
|
%
|
56.2
|
%
|
||||
|
Expected life
|
7.7 years
|
7.4 years
|
||||||
|
Nine Months Ended
September 30,
|
||||||||
|
2023
|
2022
|
|||||||
|
Risk-free interest rate
|
5.3
|
%
|
1.2
|
%
|
||||
|
Dividend yield
|
0.0
|
%
|
0.0
|
%
|
||||
|
Volatility
|
36.0
|
%
|
50.1
|
%
|
||||
|
Expected life
|
6 months
|
6 months
|
||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Cost of sales
|
$
|
118
|
$
|
163
|
$
|
355
|
$
|
376
|
||||||||
|
Research, development and patent expense
|
18,727
|
17,733
|
57,543
|
55,315
|
||||||||||||
|
Selling, general and administrative expense
|
7,119
|
5,941
|
21,575
|
18,884
|
||||||||||||
|
Total stock-based compensation expense
|
$
|
25,964
|
$
|
23,837
|
$
|
79,473
|
$
|
74,575
|
||||||||
|
Proceeds from sale of future royalties
|
$
|
500,000
|
||
|
Royalty payments to Royalty Pharma
|
(27,814
|
)
|
||
|
Interest expense related to sale of future royalties
|
50,541
|
|||
|
Liability related to sale of future royalties as of September 30, 2023
|
522,727
|
|||
|
Issuance costs related to sale of future royalties
|
(10,434
|
)
|
||
|
Amortization of issuance costs related to sale of future royalties as of September 30, 2023
|
407
|
|||
|
Net liability related to sale of future royalties as of September 30, 2023
|
$
|
512,700
|
|
1.75%
Notes
|
||||
|
Outstanding principal balance
|
$
|
575.0
|
||
|
Unamortized debt issuance costs
|
$
|
13.4
|
||
|
Maturity date
|
June 2028
|
|||
|
Interest rate
|
1.75 percent
|
|||
|
Effective interest rate
|
2.3 percent
|
|||
|
Conversion price per share
|
$
|
53.73
|
||
|
Total shares of common stock subject to conversion
|
10.7
|
|||
|
0% Notes
|
||||
|
Outstanding principal balance
|
$
|
632.5
|
||
|
Unamortized debt issuance costs
|
$
|
7.9
|
||
|
Maturity date
|
April 2026
|
|||
|
Interest rate
|
0 percent
|
|||
|
Effective interest rate
|
0.5 percent
|
|||
|
Conversion price per share
|
$
|
57.84
|
||
|
Effective conversion price per share with call spread
|
$
|
76.39
|
||
|
Total shares of common stock subject to conversion
|
10.9
|
|||
|
0.125%
Notes
|
||||
|
Outstanding principal balance
|
$
|
44.5
|
||
|
Unamortized debt issuance costs
|
$
|
0.2
|
||
|
Maturity date
|
December 2024
|
|||
|
Interest rate
|
0.125 percent
|
|||
|
Effective interest rate
|
0.5 percent
|
|||
|
Conversion price per share
|
$
|
83.28
|
||
|
Effective conversion price per share with call spread
|
$
|
123.38
|
||
|
Total shares of common stock subject to conversion
|
0.5
|
|||
| ● |
Eplontersen: our medicine in development for transthyretin amyloidosis, or
ATTR
|
| o |
We are currently conducting a broad Phase 3 development program for ATTRv-PN and ATTR cardiomyopathy, or ATTR-CM, and additional studies supporting our
ATTR development program
|
| ● |
The FDA accepted the NDA of eplontersen in the U.S. for ATTRv-PN with a PDUFA
date of December 22, 2023, and eplontersen is currently under regulatory review by the EMA and Health Canada for ATTRv-PN
|
| ● |
In November 2023, we presented new positive data showing continued benefit in secondary endpoints from the Phase 3 NEURO-TTRansform study in patients with ATTRv-PN at
EU-ATTR congress
|
| ● |
In October 2023, the EMA granted orphan drug designation to eplontersen for the treatment of ATTR in the EU
|
| ● |
In September 2023, The Journal of the American Medical Association
(JAMA) published positive results from the Phase 3 NEURO-TTRansform study in patients with ATTRv-PN showing eplontersen halted disease
progression and continuously improved quality of life at 35-, 66- and 85-week analyses
|
| ● |
In July 2023, we completed enrollment of the Phase 3 CARDIO-TTRansform study of eplontersen in patients with ATTR-CM
|
| ● |
Olezarsen: our medicine in development for FCS and severe hypertriglyceridemia, or SHTG
|
| o |
We are currently conducting a broad Phase 3 development program for olezarsen that includes the Phase 3 Balance study in patients with FCS, three Phase 3
studies supporting development for the treatment of SHTG (CORE, CORE2 and ESSENCE) and a Phase 2b supporting study
|
| ● |
In September 2023, we reported positive results from the Phase 3 Balance
study in patients with FCS showing statistically significant triglyceride lowering and a substantial reduction in acute pancreatitis events in addition to a favorable safety and tolerability profile
|
| ● |
In January 2023, the FDA granted fast track designation to olezarsen for the
treatment of patients with FCS
|
| ● |
Donidalorsen: our medicine in development for hereditary angioedema, or HAE
|
| o |
We are currently conducting the Phase 3 OASIS-HAE study in patients with HAE and the Phase 3 OASIS-Plus supportive study for HAE patients previously
treated with other prophylactic therapies
|
| ● |
In September 2023, the FDA granted orphan drug designation to donidalorsen for the treatment of HAE
|
| ● |
In June 2023, we completed enrollment of the Phase 3 OASIS-HAE study of donidalorsen in patients with hereditary angioedema; we remain on track for data in the first half
of 2024
|
| ● |
We reported positive data from the Phase 2 study and Phase 2 open-label extension, or OLE, study throughout 2022 and early 2023, including new topline two-year OLE data in June 2023
|
| ● |
Ulefnersen (ION363): our medicine in development for amyotrophic lateral sclerosis, or ALS, with mutations in the fused in sarcoma gene, or FUS
|
| o |
We are currently conducting a Phase 3 study of ulefnersen in juvenile and adult patients with FUS-ALS
|
| ● |
In August 2023, the FDA granted orphan drug designation to ulefnersen for the treatment of FUS-ALS
|
| ● |
QALSODY: our medicine to treat patients with SOD1-ALS that is approved in the U.S., under regulatory review in the EU and in development for presymptomatic patients
|
| o |
In April 2023, the FDA granted Biogen accelerated approval of QALSODY
for patients with SOD1-ALS
|
| o |
The EMA is currently reviewing QALSODY’s Marketing Authorization
Application, or MAA, in the EU
|
| ● |
Pelacarsen: our medicine in development to treat patients with elevated
lipoprotein(a), or Lp(a)-driven cardiovascular disease, or CVD
|
| o |
Novartis is developing pelacarsen, including conducting the ongoing Lp(a) HORIZON Phase 3 cardiovascular outcome study in patients with elevated
Lp(a)-driven CVD
|
| ● |
In July 2022, Novartis achieved full enrollment in the Lp(a) HORIZON study
|
| ● |
Bepirovirsen: our medicine in development for hepatitis B virus, or HBV
|
| o |
GSK is developing bepirovirsen, including conducting the ongoing B-Well Phase 3 program in patients with HBV
|
| ● |
In October 2023, GSK reported positive results from the Phase 2b B-Together study followed by pegylated interferon in patients with chronic HBV
|
| ● |
In June 2023, GSK presented durable response data from the Phase 2 B-Sure
long-term follow-up study of bepirovirsen in complete responder patients from the Phase 2b B-Clear study of patients with HBV
|
| ● |
IONIS-FB-LRx: our medicine in development for IgAN and geographic atrophy, or GA
|
| o |
In the second quarter of 2023, Roche advanced IONIS-FB-LRx into Phase 3
development in patients with IgAN
|
| o |
In October 2023, we reported positive interim data from the ongoing Phase 2
study of IONIS-FB-LRx in patients with IgAN
|
| o |
In June 2023, we completed enrollment in the Phase 2 GOLDEN study of IONIS-FB-LRx in patients with GA
|
| ● |
Zilganersen: our medicine in development for AxD
|
| o |
In September 2023, we advanced zilganersen into the Phase 3 portion of its ongoing study for patients with AxD
|
| ● |
Assessing the propriety of revenue recognition and associated deferred revenue; and
|
| ● |
Determining the appropriate cost estimates for unbilled preclinical studies and clinical development activities
|
|
Three Months Ended
|
Nine Months Ended
|
|||||||||||||||
|
September 30,
|
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Total revenue
|
$
|
144.2
|
$
|
159.8
|
$
|
463.1
|
$
|
435.5
|
||||||||
|
Total operating expenses
|
$
|
287.5
|
$
|
218.9
|
$
|
810.7
|
$
|
637.7
|
||||||||
|
Loss from operations
|
$
|
(143.3
|
)
|
$
|
(59.2
|
)
|
$
|
(347.6
|
)
|
$
|
(202.2
|
)
|
||||
|
Net loss
|
$
|
(147.4
|
)
|
$
|
(47.0
|
)
|
$
|
(357.0
|
)
|
$
|
(217.3
|
)
|
||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Revenue:
|
||||||||||||||||
|
Commercial revenue:
|
||||||||||||||||
|
SPINRAZA royalties
|
$
|
67.3
|
$
|
61.6
|
$
|
178.5
|
$
|
175.1
|
||||||||
|
Other commercial revenue:
|
||||||||||||||||
|
TEGSEDI and WAYLIVRA revenue, net
|
8.3
|
5.9
|
25.4
|
22.5
|
||||||||||||
|
Licensing and other royalty revenue
|
8.5
|
4.9
|
25.8
|
25.3
|
||||||||||||
|
Total other commercial revenue
|
16.8
|
10.8
|
51.2
|
47.8
|
||||||||||||
|
Total commercial revenue
|
84.1
|
72.4
|
229.7
|
222.9
|
||||||||||||
|
R&D revenue:
|
||||||||||||||||
|
Amortization from upfront payments
|
18.0
|
18.1
|
46.8
|
54.0
|
||||||||||||
|
Milestone payments
|
16.2
|
14.9
|
89.8
|
59.7
|
||||||||||||
|
License fees
|
4.6
|
35.0
|
24.6
|
37.0
|
||||||||||||
|
Other services
|
5.3
|
1.3
|
12.3
|
6.6
|
||||||||||||
|
Collaborative agreement revenue
|
44.1
|
69.3
|
173.5
|
157.3
|
||||||||||||
|
Eplontersen joint development revenue
|
16.0
|
18.1
|
59.9
|
55.3
|
||||||||||||
|
Total R&D revenue
|
60.1
|
87.4
|
233.4
|
212.6
|
||||||||||||
|
Total revenue
|
$
|
144.2
|
$
|
159.8
|
$
|
463.1
|
$
|
435.5
|
||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Eplontersen joint development revenue
|
$
|
16.0
|
$
|
18.1
|
$
|
59.9
|
$
|
55.3
|
||||||||
|
Research and development expenses related to Phase 3 development expenses for eplontersen
|
32.4
|
36.2
|
117.8
|
107.3
|
||||||||||||
|
Medical affairs expenses for eplontersen
|
1.1
|
0.5
|
2.9
|
1.4
|
||||||||||||
|
Commercialization expenses for eplontersen
|
4.5
|
0.8
|
8.3
|
1.5
|
||||||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Operating expenses, excluding non-cash compensation expense related to equity awards
|
$
|
261.6
|
$
|
195.1
|
$
|
731.3
|
$
|
563.1
|
||||||||
|
Non-cash compensation expense related to equity awards
|
25.9
|
23.8
|
79.4
|
74.6
|
||||||||||||
|
Total operating expenses
|
$
|
287.5
|
$
|
218.9
|
$
|
810.7
|
$
|
637.7
|
||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Cost of sales, excluding non-cash compensation expense related to equity awards
|
$
|
2.1
|
$
|
1.3
|
$
|
5.8
|
$
|
9.9
|
||||||||
|
Non-cash compensation expense related to equity awards
|
0.1
|
0.2
|
0.3
|
0.5
|
||||||||||||
|
Total cost of sales
|
$
|
2.2
|
$
|
1.5
|
$
|
6.1
|
$
|
10.4
|
||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Research, development and patent expenses, excluding non-cash compensation expense related to
equity awards
|
$
|
196.5
|
$
|
165.3
|
$
|
585.5
|
$
|
469.6
|
||||||||
|
Non-cash compensation expense related to equity awards
|
18.8
|
17.7
|
57.6
|
55.3
|
||||||||||||
|
Total research, development and patent expenses
|
$
|
215.3
|
$
|
183.0
|
$
|
643.1
|
$
|
524.9
|
||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Drug discovery expenses, excluding non-cash compensation expense related to equity awards
|
$
|
26.6
|
$
|
25.0
|
$
|
78.8
|
$
|
68.6
|
||||||||
|
Non-cash compensation expense related to equity awards
|
4.0
|
4.2
|
11.9
|
12.8
|
||||||||||||
|
Total drug discovery expenses
|
$
|
30.6
|
$
|
29.2
|
$
|
90.7
|
$
|
81.4
|
||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
TEGSEDI and WAYLIVRA
|
$
|
3.0
|
$
|
3.9
|
$
|
5.6
|
$
|
9.4
|
||||||||
|
Eplontersen
|
25.1
|
24.5
|
90.8
|
75.8
|
||||||||||||
|
Olezarsen
|
38.5
|
18.3
|
96.6
|
39.7
|
||||||||||||
|
Donidalorsen
|
6.9
|
5.5
|
19.4
|
9.1
|
||||||||||||
|
Ulefnersen
|
2.5
|
2.3
|
7.7
|
5.8
|
||||||||||||
|
Other development projects
|
28.2
|
31.6
|
81.4
|
91.7
|
||||||||||||
|
Development overhead expenses
|
28.3
|
22.5
|
83.8
|
63.3
|
||||||||||||
|
Total drug development, excluding non-cash compensation expense related to equity awards
|
132.5
|
108.6
|
385.3
|
294.8
|
||||||||||||
|
Non-cash compensation expense related to equity awards
|
8.6
|
7.6
|
25.7
|
15.6
|
||||||||||||
|
Total drug development expenses
|
$
|
141.1
|
$
|
116.2
|
$
|
411.0
|
$
|
310.4
|
||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Medical affairs expenses, excluding non-cash compensation expense related to equity awards
|
$
|
4.9
|
$
|
3.8
|
$
|
13.8
|
$
|
11.4
|
||||||||
|
Non-cash compensation expense related to equity awards
|
0.8
|
0.6
|
2.7
|
1.3
|
||||||||||||
|
Total medical affairs expenses
|
$
|
5.7
|
$
|
4.4
|
$
|
16.5
|
$
|
12.7
|
||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Manufacturing and development chemistry expenses, excluding non-cash compensation expense related
to equity awards
|
$
|
12.3
|
$
|
14.0
|
$
|
49.2
|
$
|
53.4
|
||||||||
|
Non-cash compensation expense related to equity awards
|
2.2
|
2.3
|
6.5
|
7.6
|
||||||||||||
|
Total manufacturing and development chemistry expenses
|
$
|
14.5
|
$
|
16.3
|
$
|
55.7
|
$
|
61.0
|
||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Personnel costs
|
$
|
6.2
|
$
|
5.3
|
$
|
19.4
|
$
|
15.4
|
||||||||
|
Occupancy
|
7.2
|
4.1
|
21.4
|
12.3
|
||||||||||||
|
Patent expenses
|
0.9
|
0.9
|
2.8
|
3.1
|
||||||||||||
|
Insurance
|
0.9
|
1.0
|
2.7
|
2.8
|
||||||||||||
|
Computer software and licenses
|
0.8
|
0.1
|
2.0
|
1.1
|
||||||||||||
|
Other
|
4.2
|
2.5
|
10.1
|
6.7
|
||||||||||||
|
Total R&D support expenses, excluding non-cash compensation expense related to equity awards
|
20.2
|
13.9
|
58.4
|
41.4
|
||||||||||||
|
Non-cash compensation expense related to equity awards
|
3.2
|
3.0
|
10.8
|
10.4
|
||||||||||||
|
Total R&D support expenses
|
$
|
23.4
|
$
|
16.9
|
$
|
69.2
|
$
|
51.8
|
||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Selling, general and administrative expenses, excluding non-cash compensation expense related to
equity awards
|
$
|
62.9
|
$
|
28.5
|
$
|
140.0
|
$
|
83.6
|
||||||||
|
Non-cash compensation expense related to equity awards
|
7.1
|
5.9
|
21.6
|
18.8
|
||||||||||||
|
Total selling, general and administrative expenses
|
$
|
70.0
|
$
|
34.4
|
$
|
161.6
|
$
|
102.4
|
||||||||
|
Three Months Ended September 30,
|
Nine Months Ended September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Investment income
|
$
|
23.9
|
$
|
7.5
|
$
|
63.4
|
$
|
13.4
|
||||||||
|
Three Months Ended
September 30,
|
Nine Months Ended
September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Convertible notes:
|
||||||||||||||||
|
Non-cash amortization of debt issuance costs
|
$
|
1.6
|
$
|
1.3
|
$
|
4.3
|
$
|
4.0
|
||||||||
|
Interest expense payable in cash
|
2.5
|
0.2
|
3.5
|
0.5
|
||||||||||||
|
Interest on mortgage for primary R&D and manufacturing facilities
|
0.1
|
0.6
|
0.3
|
1.9
|
||||||||||||
|
Total interest expense
|
$
|
4.2
|
$
|
2.1
|
$
|
8.1
|
$
|
6.4
|
||||||||
|
Three Months Ended September 30,
|
Nine Months Ended September 30,
|
|||||||||||||||
|
2023
|
2022
|
2023
|
2022
|
|||||||||||||
|
Gain (loss) on investments
|
$
|
(1.9
|
)
|
$
|
2.3
|
$
|
(1.8
|
)
|
$
|
(10.6
|
)
|
|||||
|
Payments Due by Period (in millions)
|
||||||||||||
|
(selected balances described below)
|
Total
|
Less than 1 year
|
More than 1 year
|
|||||||||
|
1.75% Notes (principal and interest payable)
|
$
|
625.4
|
$
|
10.1
|
$
|
615.3
|
||||||
|
0% Notes (principal payable)
|
632.5
|
—
|
632.5
|
|||||||||
|
0.125% Notes (principal and interest payable)
|
44.7
|
0.1
|
44.6
|
|||||||||
|
Operating leases
|
284.6
|
20.3
|
264.3
|
|||||||||
|
Building mortgage payments (principal and interest payable)
|
10.3
|
0.5
|
9.8
|
|||||||||
|
Other obligations (principal and interest payable)
|
0.8
|
0.1
|
0.7
|
|||||||||
|
Total
|
$
|
1,598.3
|
$
|
31.1
|
$
|
1,567.2
|
||||||
| ITEM 4. |
CONTROLS AND PROCEDURES
|
| ITEM 1. |
LEGAL PROCEEDINGS
|
| ITEM 1A. |
RISK FACTORS
|
| ● |
Our ability to generate substantial revenue from the sale of our medicines;
|
| ● |
Τhe availability of adequate coverage and payment rates for our
medicines;
|
| ● |
Our and our partners’ ability to compete effectively;
|
| ● |
Our ability to successfully manufacture our medicines;
|
| ● |
Our ability to successfully develop and obtain marketing approvals for our medicines;
|
| ● |
Our ability to secure and maintain effective corporate partnerships;
|
| ● |
Our ability to sustain cash flows and achieve consistent profitability;
|
| ● |
Our ability to protect our intellectual property;
|
| ● |
Our ability to maintain the effectiveness of our personnel;
|
| ● |
The impacts of the COVID-19 pandemic and ongoing wars between Russia/Ukraine and Israel/Hamas; and
|
| ● |
The other factors set forth below.
|
| ● |
receipt and scope of marketing authorizations;
|
| ● |
establishment and demonstration in the medical and patient community of the efficacy and safety of our medicines and their potential advantages over
competing products;
|
| ● |
cost and effectiveness of our medicines compared to other available therapies;
|
| ● |
patient convenience of the dosing regimen for our medicines; and
|
| ● |
reimbursement policies of government and third-party payers.
|
| ● |
priced lower than our medicines;
|
| ● |
reimbursed more favorably by government and other third-party payers than our medicines;
|
| ● |
safer than our medicines;
|
| ● |
more effective than our medicines; or
|
| ● |
more convenient to use than our medicines.
|
| ● |
Onasemnogene abeparvovec and risdiplam compete with SPINRAZA;
|
| ● |
Patisiran, tafamidis, tafamidis meglumine and vutrisiran compete with TEGSEDI and could compete with eplontersen;
|
| ● |
Acoramidis could compete with TEGSEDI and eplontersen;
|
| ● |
ARO-APOC3, lomitapide and pegozafermin could compete with WAYLIVRA and olezarsen;
|
| ● |
Lanadelumab-flyo, C1 esterase inhibitor, berotralstat, C1 esterase inhibitor subcutaneous, garadacimab, and NTLA-2002 could compete with donidalorsen;
|
| ● |
Olpasiran and SLN360 could compete with pelacarsen; and
|
| ● |
NI-204 could compete with QALSODY.
|
| ● |
in the U.S., TEGSEDI’s label contains a boxed warning for thrombocytopenia and glomerulonephritis;
|
| ● |
TEGSEDI requires periodic blood and urine monitoring; and
|
| ● |
in the U.S., TEGSEDI is available only through a REMS program.
|
| ● |
fund our development activities for SPINRAZA and QALSODY;
|
| ● |
seek and obtain regulatory approvals for SPINRAZA and QALSODY; and
|
| ● |
successfully commercialize SPINRAZA and QALSODY.
|
| ● |
In April 2021, we entered into a distribution agreement with Sobi to commercialize TEGSEDI in the U.S. and Canada. Effective October 24, 2023, such
agreement was terminated;
|
| ● |
In December 2020, we entered into a distribution agreement with Sobi to commercialize TEGSEDI and WAYLIVRA in Europe; and
|
| ● |
In August 2018, we granted PTC the exclusive right to commercialize TEGSEDI and WAYLIVRA in Latin America and certain Caribbean countries.
|
| ● |
such authorities may disagree with the design or implementation of our clinical studies;
|
| ● |
we or our partners may be unable to demonstrate to the satisfaction of the FDA or other regulatory authorities that a medicine is safe and effective for
any indication;
|
| ● |
such authorities may not accept clinical data from studies conducted at clinical facilities that have deficient clinical practices or that are in
countries where the standard of care is potentially different from the U.S.;
|
| ● |
we or our partners may be unable to demonstrate that our medicine’s clinical and other benefits outweigh its safety risks to support approval;
|
| ● |
such authorities may disagree with the interpretation of data from preclinical or clinical studies;
|
| ● |
such authorities may find deficiencies in the manufacturing processes or facilities of third-party manufacturers who manufacture clinical and commercial
supplies for our medicines, or may delay the inspection of such facilities due to restrictions related to the COVID-19 pandemic; and
|
| ● |
the approval policies or regulations of such authorities or their prior guidance to us or our partners during clinical development may significantly
change in a manner rendering our clinical data insufficient for approval.
|
| ● |
the clinical study may produce negative or inconclusive results;
|
| ● |
regulators may require that we hold, suspend or terminate clinical research for noncompliance with regulatory requirements;
|
| ● |
we, our partners, the FDA or foreign regulatory authorities could suspend or terminate a clinical study due to adverse side effects of a medicine on
subjects or lack of efficacy in the trial;
|
| ● |
we or our partners may decide, or regulators may require us, to conduct additional preclinical testing or clinical studies;
|
| ● |
enrollment in our clinical studies may be slower than we anticipate;
|
| ● |
we or our partners, including our independent clinical investigators, contract research organizations and other third-party service providers on which we
rely, may not identify, recruit and train suitable clinical investigators at a sufficient number of study sites or timely enroll a sufficient number of study subjects in the clinical study;
|
| ● |
the institutional review board for a prospective site might withhold or delay its approval for the study;
|
| ● |
people who enroll in the clinical study may later drop out due to adverse events, a perception they are not benefiting from participating in the study,
fatigue with the clinical study process or personal issues;
|
| ● |
a clinical study site may deviate from the protocol for the study;
|
| ● |
the cost of our clinical studies may be greater than we anticipate;
|
| ● |
our partners may decide not to exercise any existing options to license and conduct additional clinical studies for our medicines; and
|
| ● |
the supply or quality of our medicines or other materials necessary to conduct our clinical studies may be insufficient, inadequate or delayed.
|
| ● |
AstraZeneca for the joint development and funding of eplontersen;
|
| ● |
Novartis for development and funding of pelacarsen;
|
| ● |
GSK for development and funding of bepirovirsen; and
|
| ● |
Roche for development and funding of IONIS-FB-LRx.
|
| ● |
conduct clinical studies;
|
| ● |
seek and obtain marketing authorizations; and
|
| ● |
manufacture and commercialize our medicines.
|
| ● |
pursue alternative technologies or develop alternative products that may be competitive with the medicine that is part of the collaboration with us;
|
| ● |
pursue higher-priority programs or change the focus of its own development programs; or
|
| ● |
choose to devote fewer resources to our medicines than it does to its own medicines.
|
| ● |
successful commercialization of QALSODY, SPINRAZA, TEGSEDI and WAYLIVRA;
|
| ● |
the profile and launch timing of our medicines, including bepirovirsen, donidalorsen, eplontersen, IONIS-FB-LRx, olezarsen, pelacarsen,
ulefnersen and zilganersen;
|
| ● |
changes in existing collaborative relationships and our ability to establish and maintain additional collaborative arrangements;
|
| ● |
continued scientific progress in our research, drug discovery and development programs;
|
| ● |
the size of our programs and progress with preclinical and clinical studies;
|
| ● |
the time and costs involved in obtaining marketing authorizations;
|
| ● |
competing technological and market developments, including the introduction by others of new therapies that address our markets; and
|
| ● |
our manufacturing requirements and capacity to fulfill such requirements.
|
| ● |
compliance with differing or unexpected regulatory requirements for our medicines and foreign employees;
|
| ● |
complexities associated with managing multiple payer reimbursement regimes, government payers or patient self-pay systems;
|
| ● |
difficulties in staffing and managing foreign operations;
|
| ● |
in certain circumstances, increased dependence on the commercialization efforts and regulatory compliance of third-party distributors or strategic
partners;
|
| ● |
foreign government taxes, regulations and permit requirements;
|
| ● |
U.S. and foreign government tariffs, trade restrictions, price and exchange controls and other regulatory requirements;
|
| ● |
anti-corruption laws, including the Foreign Corrupt Practices Act, or the FCPA, and its equivalent in foreign jurisdictions;
|
| ● |
economic weakness, including inflation, natural disasters, war, events of terrorism, political instability or public health issues or pandemics, such as
the COVID-19 pandemic, in particular foreign countries or globally;
|
| ● |
fluctuations in currency exchange rates, which could result in increased operating expenses and reduced revenue, and other obligations related to doing
business in another country;
|
| ● |
compliance with tax, employment, privacy, immigration and labor laws, regulations and restrictions for employees living or traveling abroad;
|
| ● |
workforce uncertainty in countries where labor unrest is more common than in the U.S.; and
|
| ● |
changes in diplomatic and trade relationships.
|
| ● |
interruption of our research, development and manufacturing efforts;
|
| ● |
injury to our employees and others;
|
| ● |
environmental damage resulting in costly clean up; and
|
| ● |
liabilities under federal, state and local laws and regulations governing health and human safety, as well as the use, storage, handling and disposal of
these materials and resultant waste products.
|
| ITEM 2. |
UNREGISTERED SALES OF EQUITY SECURITIES AND USE OF PROCEEDS
|
| ITEM 3. |
DEFAULT UPON SENIOR SECURITIES
|
| ITEM 4. |
MINE SAFETY DISCLOSURES
|
| ITEM 5. |
OTHER INFORMATION
|
| * |
Contract, instruction or written plan intended to satisfy the affirmative defense conditions of Rule 10b5-1(c) under the Exchange Act.
|
| ** |
“Non-Rule 10b5-1 trading arrangement” as defined in item 408(c) of Regulation S-K under the Exchange Act.
|
|
Action
|
Date
|
Trading Arrangement
|
Total Shares to Be Sold
|
Expiration Date
|
||||||||
|
Rule 10b5-1*
|
Non-Rule 10b5-1**
|
|||||||||||
|
Brett Monia,
CEO and Board Member
|
Termination
|
July 12, 2023
|
X
|
30,023
|
Upon the execution of all instructions provided in the plan
|
|||||||
|
Brett Monia,
CEO and Board Member
|
Adoption
|
July 13, 2023
|
X
|
54,442
|
Upon the execution of all instructions provided in the plan
|
|||||||
|
Eric Swayze,
EVP, Research
|
Adoption
|
July 13, 2023
|
X
|
85,614
|
Upon the execution of all instructions provided in the plan
|
|||||||
|
Spencer Berthelsen,
Board Member
|
Adoption
|
September 7, 2023
|
X
|
16,000
|
Upon the execution of all instructions provided in the plan
|
|||||||
|
B. Lynne Parshall,
Board Member
|
Adoption
|
September 27, 2023
|
X
|
122,638
|
Upon the execution of all instructions provided in the plan
|
|||||||
| a. |
Exhibits
|
|
Exhibit Number
|
Description of Document
|
|
|
Collaboration and License Agreement by and between the Registrant and Novartis Pharma AG dated as of August 2, 2023. Portions of this exhibit have
been omitted because they are both (i) not material and (ii) the type that the Registrant treats as private or confidential.
|
||
|
Amended and Restated Lease Agreement between the
Registrant and Lots 21 & 22 Owner (DE) LLC dated as of August 21, 2023. Portions of this exhibit have been omitted because they are both (i) not material and (ii) the type that the Registrant treats as private or confidential.
|
||
|
Research, Development, and License Agreement by and among the Registrant, F. Hoffmann-La Roche Ltd., and Hoffmann-La Roche Inc. dated as of September
26, 2023. Portions of this exhibit have been omitted because they are both (i) not material and (ii) the type that the Registrant treats as private or confidential.
|
||
|
Certification by Chief Executive Officer pursuant to Rules 13a-14(a) and 15d-14(a) under the Securities Exchange Act of 1934, as
amended.
|
||
|
Certification by Chief Financial Officer pursuant to Rules 13a-14(a) and 15d-14(a) under the Securities Exchange Act of 1934, as
amended.
|
||
|
32.1*
|
Certification Pursuant to 18 U.S.C. Section 1350 as Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002.
|
|
|
101
|
The following financial statements from the Ionis Pharmaceuticals, Inc. Quarterly Report on Form 10-Q for the quarter ended September 30, 2023, formatted in Inline Extensible Business Reporting Language (iXBRL): (i) condensed consolidated balance sheets, (ii) condensed
consolidated statements of operations, (iii) condensed consolidated statements of comprehensive income (loss), (iv) condensed consolidated statements of stockholders’ equity, (v) condensed consolidated statements of cash flows and (vi) notes to
condensed consolidated financial statements (detail tagged).
|
|
|
104
|
Cover Page Interactive Data File (formatted in iXBRL and included in exhibit 101).
|
| * |
This certification is deemed not filed for purposes of Section 18 of the Securities Exchange Act of 1934, as amended, or otherwise subject to the
liability of that section, nor shall it be deemed incorporated by reference into any filing under the Securities Act of 1933, as amended, or the Securities Exchange Act of 1934, as amended.
|
|
Signatures
|
Title
|
Date
|
||
|
/s/ BRETT P. MONIA
|
Director and Chief Executive Officer
|
|||
|
Brett P. Monia, Ph.D.
|
(Principal executive officer)
|
November 2, 2023
|
||
|
/s/ ELIZABETH L. HOUGEN
|
Executive Vice President, Finance and Chief Financial Officer
|
|||
|
Elizabeth L. Hougen
|
(Principal financial and accounting officer)
|
November 2, 2023
|
|
|
3.1. |
Joint Steering Committee.
|
|
|
(a) |
review, discuss, and determine whether to designate any Carryover Candidate or Novartis Proposed Development Candidate as a Development Candidate, as described in Section 2.2.4 (Development Candidate Selection Process);
|
|
|
(b) |
oversee each Technology Transfer Plan, as described in Section 5.9.1 (Licensed Know-How – Generally) with respect to any such Development Candidate; and
|
|
|
(c) |
oversee each Manufacturing Technology Transfer Plan, as described in Section 5.9.2 (Ionis Manufacturing and Analytical Know-How) with respect to any such Development Candidate.
|
| (i) |
[***];
|
|
|
(ii) |
[***];
|
|
|
(iii) |
[***]; and
|
|
|
(iv) |
[***].
|
|
Table 1
|
|
|
Early Development Milestone Event
|
Milestone Event Payment
|
|
1. [***]
|
US$[***]
|
|
2. [***]
|
US$[***]
|
|
Table 2
|
|
|
Development Milestone Event
|
Milestone Event Payment
|
|
1. [***]
|
US$[***]
|
|
2. (a) [***] or (b) [***]
|
US$[***]
|
|
3. [***]
|
US$[***]
|
|
4. [***]
|
US$[***]
|
|
5. [***]
|
US$[***]
|
|
6. [***]
|
US$[***]
|
|
Table 3
|
|
|
Annual Worldwide Net Sales Levels
|
Milestone Payment
|
|
Exceeding US$[***] in Annual Net Sales for a Licensed Product
|
US$[***]
|
|
Exceeding US$[***] in Annual Net Sales for a Licensed Product
|
US$[***]
|
|
Exceeding US$[***] in Annual Net Sales for a Licensed Product
|
US$[***]
|
|
Exceeding US$[***] in Annual Net Sales for a Licensed Product
|
US$[***]
|
|
Exceeding US$[***] in Annual Net Sales for a Licensed Product
|
US$[***]
|
|
Table 4
|
||
|
Royalty
Tier
|
Annual Worldwide Net Sales of such Licensed Product
|
Royalty
Rate
|
|
1
|
For the portion of Annual Worldwide Net Sales ≤ US$[***]
|
[***]%
|
|
2
|
For the portion of Annual Worldwide Net Sales > US$[***] but ≤ US$[***]
|
[***]%
|
|
3
|
For the portion of Annual Worldwide Net Sales > US$[***] but ≤ US$[***]
|
[***]%
|
|
4
|
For the portion of Annual Worldwide Net Sales > US$[***] but ≤ US$[***]
|
[***]%
|
|
5
|
For the portion of Annual Worldwide Net Sales > US$[***] but ≤ US$[***]
|
[***]%
|
|
6
|
For the portion of Annual Worldwide Net Sales > US$[***]
|
[***]%
|
|
|
with a copy to: |
Novartis Pharma AG
|
|
NOVARTIS PHARMA AG
|
||
|
By:
|
/s/ Guillaume Vignon
|
|
|
Name:
|
Guillaume Vignon
|
|
|
Title:
|
Global Head BD&L Partnering
|
|
|
NOVARTIS PHARMA AG
|
||
|
By:
|
/s/ Mark Victor Rogers
|
|
|
Name:
|
Mark Victor Rogers
|
|
|
Title:
|
Global Head Pharma Transactions
|
|
IONIS PHARMACEUTICALS, INC.
|
||
|
By:
|
/s/ Brett Monia
|
|
|
Name:
|
Brett Monia
|
|
|
Title:
|
Chief Executive Officer
|
|
|
(a) |
was in the lawful knowledge and possession of the Receiving Party or its Affiliates prior to the time it was disclosed to the Receiving Party or its Affiliates, or was otherwise developed independently by the Receiving Party or its
Affiliates, in each case, as evidenced by written records kept in the ordinary course of business, or other documentary proof of the Receiving Party or its Affiliates;
|
|
|
(b) |
was generally available to the public or otherwise part of the public domain at the time of its disclosure to the Receiving Party or its Affiliates;
|
|
|
(c) |
became generally available to the public or otherwise part of the public domain after its disclosure and other than through any act or omission of the Receiving Party or its Affiliates in breach of this Agreement; or
|
|
|
(d) |
was disclosed to the Receiving Party or its Affiliates, other than under an obligation of confidentiality, by a Third Party who had no obligation to the Disclosing Party or its Affiliates not to disclose such information to others.
|
|
|
(i) |
normal trade and cash discounts;
|
|
|
(ii) |
amounts repaid or credited by reasons of defects, rejections, recalls or returns;
|
|
|
(iii) |
rebates and chargebacks to customers and other Third Parties (including Medicare, Medicaid, Managed Healthcare and similar types of rebates);
|
|
|
(iv) |
amounts provided or credited to customers through coupons and other discount programs;
|
|
|
(v) |
delayed ship order credits, discounts or payments related to the impact of price increases between purchase and shipping dates or retroactive price reductions;
|
|
|
(vi) |
fee for service payments to customers for any non-separable services (including compensation for maintaining agreed inventory levels and providing information); and
|
|
|
(vii) |
other reductions or specifically identifiable amounts deducted for reasons similar to those listed above in accordance with Novartis’ Accounting Standards.
|
|
|
(a) |
Net Sales only include the value charged or invoiced on the first arm’s length sale to a Third Party. Sales between or among Novartis and its Affiliates and Sublicensees shall be disregarded for purposes of calculating Net Sales;
|
|
|
(b) |
If a Licensed Product is delivered to the Third Party before being invoiced (or is not invoiced), Net Sales will be calculated at the time all the revenue recognition criteria under Novartis’ Accounting Standards are met;
|
|
|
(c) |
In the event that the Licensed Product is in finished dosage form containing the Licensed Compound in combination with one (1) or more other active ingredients (such active ingredients, “Other
Components,” and such Licensed Product, a “Combination Product”), the Net Sales will be calculated by multiplying the Net Sales of the Combination Product by the fraction,
A/(A+B) where A is the weighted (by sales volume) average sale price in the relevant country of the Licensed Product containing the Licensed Compound as the sole active ingredient in finished form, and B is the weighted average sale price (by
sales volume) in that country of the product(s) containing the Other Components as the sole active ingredient(s) in finished form. Regarding prices comprised in the weighted average price when sold separately referred to above, if these are
available for different dosages from the dosages of the Licensed Compound and the Other Components, then Novartis shall be entitled to make a proportional adjustment to such prices in calculating the royalty-bearing Net Sales of the
Combination Product. If the weighted average sale price cannot be determined for the Licensed Product or other product(s) containing the single Licensed Compound or Other Components, the calculation of Net Sales for Combination Products will
be agreed by the Parties based on the relative value contributed by each component (each Party’s agreement not to be unreasonably withheld or delayed).
|
|
|
(a) |
a mandatory patent listing process in such country, only Licensed Patents that are listed in such country’s patent listing will be considered “Orange Book Patents” (and therefore royalty-bearing) in
such country, irrespective of whether the foreign equivalent Patent Rights of such Licensed Patents are listed in another country;
|
|
|
(b) |
a voluntary patent listing process in such country, both (x) Licensed Patents that are listed in such country’s patent listing, and (y) Licensed Patents that are not listed in such country’s patent listing but are the foreign equivalent
Patent Rights of the Licensed Patents listed in the mandatory patent listing of another country, in each case will be considered “Orange Book Patents” (and therefore royalty-bearing) in such country;
and
|
|
|
(c) |
no patent listing process in such country, Licensed Patents that are the foreign equivalent of the Licensed Patents listed in the mandatory patent listing of another country, in each case will be considered “Orange Book Patents” (and therefore royalty-bearing) in such country, irrespective of whether the foreign equivalent Patent Rights of such Licensed Patents are listed in another country.
|
|
|
(a) |
With respect to termination of this Agreement in its entirety prior to designation of a Development Candidate, or this Agreement expires in accordance with Section 11.1.3, (i) any Licensed Compounds that are not Novartis Compounds
and (ii) any Novartis Compounds that satisfy the Development Candidate Criteria as of the date of such termination;
|
|
|
(b) |
with respect to termination of this Agreement in part with respect to a Licensed Product, (i) such Licensed Product and (ii) such Licensed Product’s Backup Compounds, if any; and
|
|
|
(c) |
with respect to termination of this Agreement in its entirety, (i) any Licensed Product that is being Developed or Commercialized by Novartis, its Affiliates or Sublicensees as of the effective date of termination of this Agreement and
(ii) such Licensed Product’s Backup Compounds, if any.
|
| 1. |
[***].
|
| 2. |
[***].
|
| 1. |
[***]
|
| 2. |
[***]
|
| 3. |
[***]
|
| 4. |
[***]
|
| 5. |
[***]
|
| 6. |
[***]
|
| 7. |
[***]
|
| 8. |
[***]
|
| 9. |
[***]
|
| 10. |
[***]
|
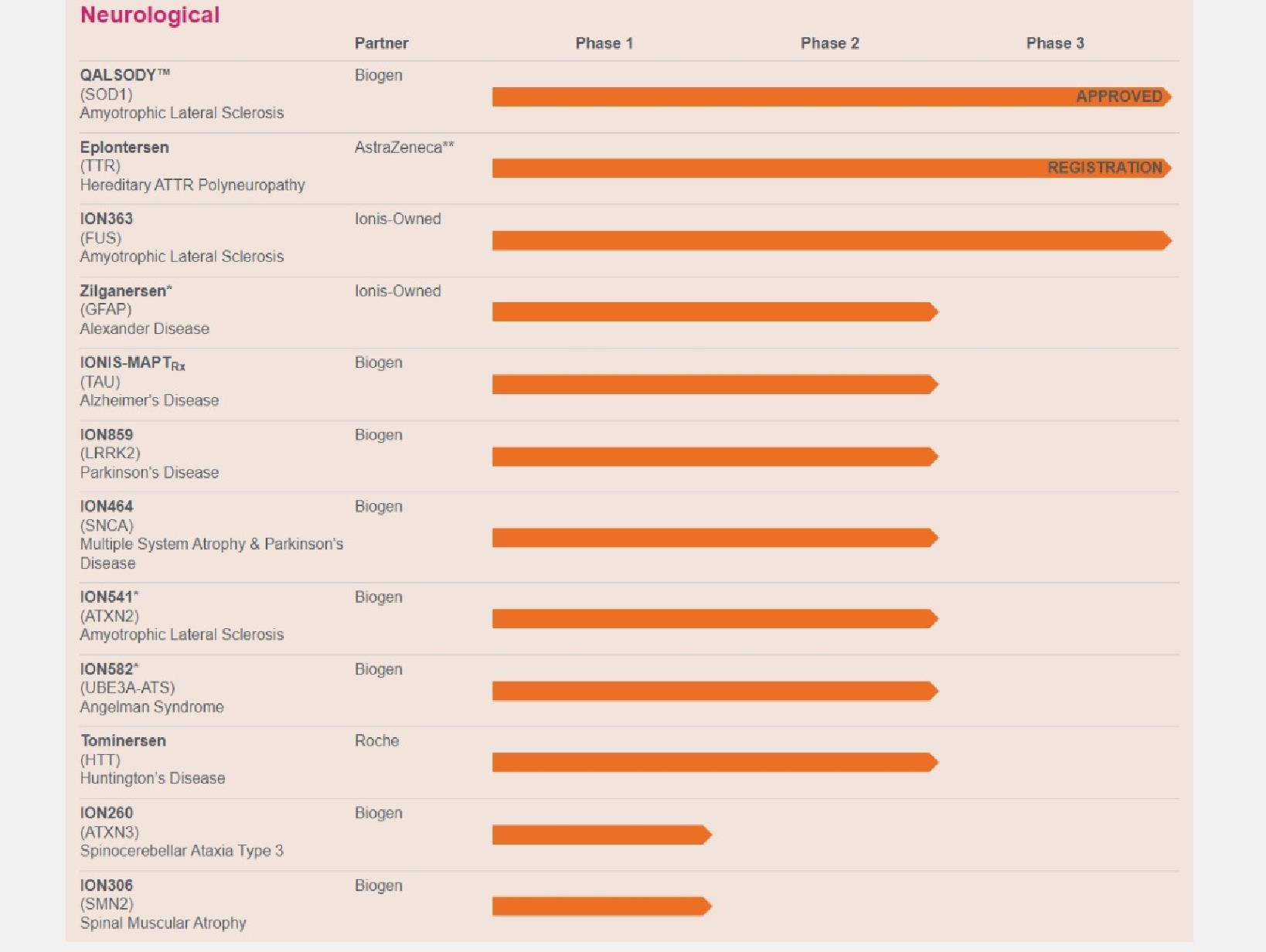

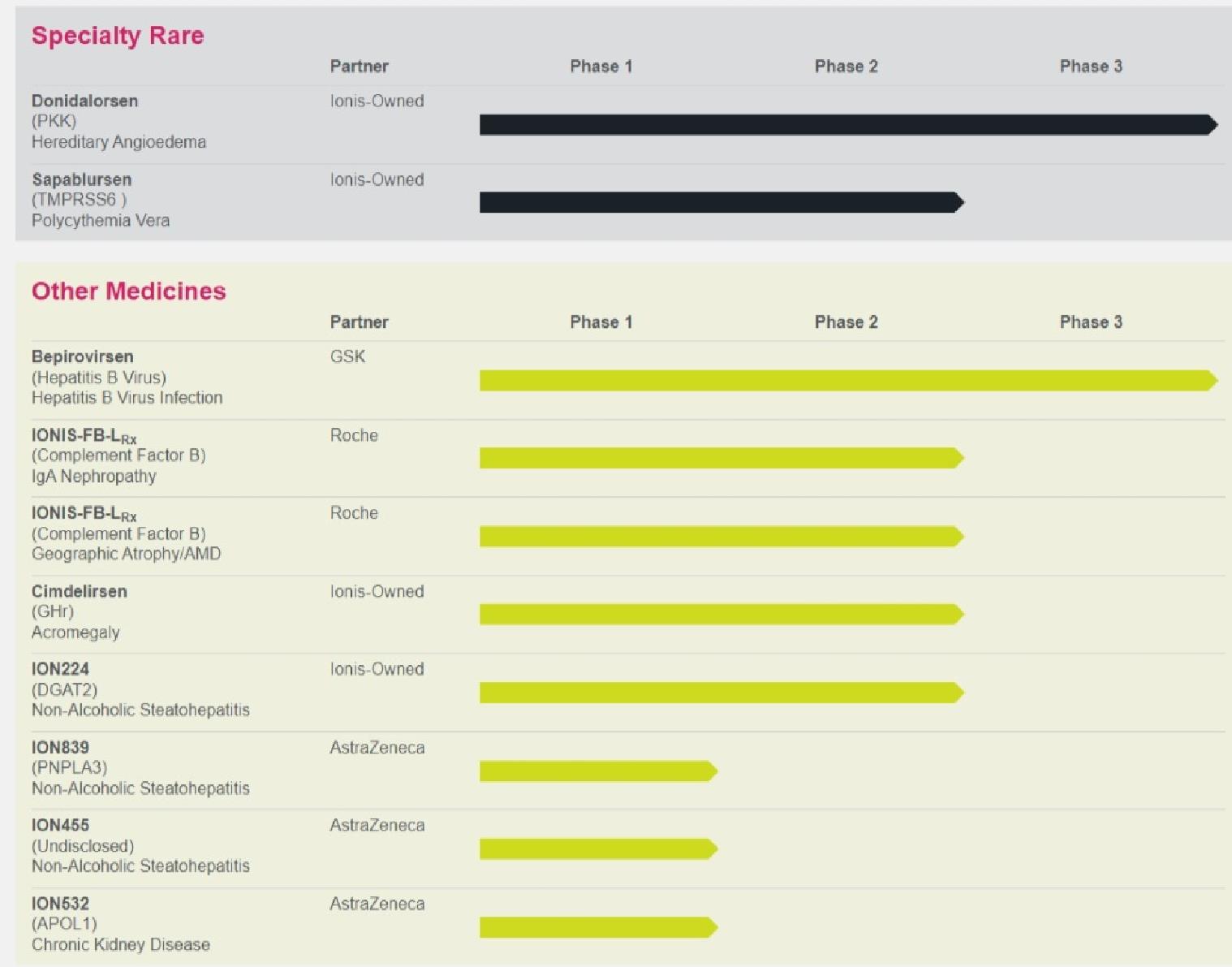
|
Ionis Docket Number
|
Country/Treaty
|
Application/
Patent
Number
|
Grant Date
|
Title
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
Ionis Docket Number
|
Country/Treaty
|
Application/
Patent
Number
|
Grant Date
|
Title
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
Ionis Docket Number
|
Country/Treaty
|
Application/
Patent
Number
|
Grant Date
|
Title
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
Ionis Docket Number
|
Country
|
Application/
Patent
Number
|
Grant Date
|
Title
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
| 1. |
[***]
|
| 2. |
[***]
|
| 3. |
[***]
|
| 4. |
[***]
|
| 5. |
[***]
|
| 6. |
[***]
|
| 7. |
[***]
|
| 8. |
[***]
|
| 9. |
[***]
|
| 10. |
[***]
|
|
Sender’s Logo
|
INVOICE
|
||
|
|
|||
|
Name
|
INVOICE Date: xx.xx.xxxx
|
||
|
Street
|
INVOICE No.: xxxx
|
||
|
Town, Country
|
|||
|
Phone and Fax Nr.
|
|||
|
Bill To:
|
|||
|
NOVARTIS PHARMA AG
|
|||
|
Zentraler Faktureneingang
Attn: Barbara Schultheiss
|
Purchase Order: xxxx
[Reference to Novartis Purchase Order]
|
||
|
PO Box
CH-4002 Basel
|
|||
|
Switzerland
|
|||
|
DESCRIPTION
[Please specify the service or event for which the invoice is due]
|
AMOUNT (xxx)
[specify the currency]
|
||
|
[Please reference the agreement and paragraph thereof describing the payment obligation]
[Please specify the service period, e.g. Q1 20xx]
|
000’000.00
|
||
|
VAT (if applicable)
|
000’000.00
|
||
|
Please remit by wire transfer within 60 days to:
[payment term as per agreement]
|
|||
|
Receiving Bank – (name, address and country).
|
|||
|
Swift Code -
|
|||
|
Bank account number -
IBAN (mandatory for Europe)
ABA Number -
|
|||
|
Have an electronic copy of this invoice please sent to:
• [***]
• [***]
• [***]
|
|||
|
TOTAL
|
000’000,00
|
||
|
If you have any questions concerning this invoice, contact ……… or e-mail to ………
[reference to invoice senders subject matter expert]
|
|||
|
Novartis Pharma AG: VAT –Reg. No. CHE-116.268.023 MWST
|
|||
Exhibit 1
|
TERMS OF LEASE
|
DESCRIPTION
|
|
|
|
|
“Commencement Date”:
|
The date that is the earlier of: (i) ten (10) months after the Delivery Date, as defined in the Work Letter, or (ii) the date Tenant begins operating in the Premises for business purposes.
|
|
|
|
|
“Premises”:
|
That certain Land described on Exhibit A attached hereto and the Building and other improvements to be constructed thereon, located on Lots 21 & 22, Whiptail Loop W in Carlsbad,
92010 California consisting of approximately 164,757 gross square feet, and the appurtenances thereto, as defined and further described in the Work Letter. The square footage of the Premises set forth above is deemed conclusive and shall not be
subject to remeasurement
|
|
|
|
|
“Companion Lease”:
|
That certain lease to be executed between Landlord or its affiliate and Tenant pursuant to the PSA for certain real property located at 2850, 2855 & 2859 Gazelle Court, Carlsbad, 92010
California (the “Companion Premises”) and the buildings consisting of approximately 246,699 square feet and other improvements located thereon and appurtenances thereto, as described therein.
|
|
|
|
|
“Term”: (Article 1)
|
The period commencing on the Commencement Date and ending on the Expiration Date.
|
|
|
|
|
“Expiration Date”
|
The date that is one hundred eighty (180) months after the Commencement Date, together with any Extension Period as to which an Extension Option is exercised under Section 1.4.
|
|
|
|
|
“Escalation”: (Article 2)
|
The percentage of increase, if any, shown by the Consumer Price Index for All Urban Consumers U.S. City Average, All Items (base years 1982-1984 = 100) (“Index”),
published by the United States Department of Labor, Bureau of Labor Statistics, for the month immediately preceding the Adjustment Date as compared with the Index for the month immediately preceding the Commencement Date (with respect to the
first Adjustment Date), or the month immediately preceding the prior Adjustment Date (for all subsequent Adjustment Dates), but the percentage of increase shall not be less than 2.5% nor greater than 5.5%.
|
| TERMS OF LEASE | DESCRIPTION |
|
|
|
|
“Option to Renew”: (Article 1)
|
Tenant shall have two (2), five year (5) options at ninety-five percent (95%) of Fair Market Rent.
|
|
|
|
|
“Base Rent”: (Article 2)
|
For the period beginning on the Commencement Date until the first Adjustment Date, a fixed annual amount (payable in monthly installments) determined by multiplying six and thirty-five
hundredths percent (6.35%) by the Construction Costs, as defined in the Work Letter, and thereafter the amount calculated pursuant to Section 2.1.3. A hypothetical example of the calculation of Base Rent is shown on Schedule 1, which shall be
for illustrative purposes only and shall not be deemed a representation of the actual amounts or categories to be used in the calculation of Base Rent.
|
|
|
|
|
“Net Lease”: (Article 2)
|
Landlord and Tenant acknowledge and agree that this is an “absolute net lease” and that Landlord shall receive the Base Rent during the Term, free from all charges, assessments, impositions,
expenses and deductions of any and every kind or nature whatsoever relating to the Premises. Landlord shall have no obligations relating to the repair, maintenance or operation of the Premises, or any part thereof. Tenant shall be solely
responsible for same.
|
|
|
|
|
“Purchase Option”: (Article 11)
|
Tenant shall have a right of first offer to purchase the entire Premises.
|
|
|
|
|
“Security Deposit/Letter of Credit”: (Article 2)
|
Tenant shall provide a Letter of Credit equal to the sum of (a) ten (10) months of the initial Base Rent, plus (b) $6,458,200, subject to adjustment as
provided in Section 2.5.1. The amount referenced in clause (b) above is referred to as the “Day 2 Improvements Security”.
|
|
|
|
|
“Permitted Use”: (Article 3)
|
Premises may be used solely for biotechnology and other life sciences uses, including research and development, laboratory, manufacturing, assembly, storage, warehousing, office and
administrative uses, all of which must be ancillary to biotechnology and other life sciences uses and, in each such case, to the extent Tenant remains in compliance with current zoning for the Premises and all Applicable Laws.
|
| TERMS OF LEASE | DESCRIPTION |
|
|
|
|
“Work Letter”: (Exhibit B)
|
The “Work Letter” attached hereto as Exhibit B.
|
|
“Address of Tenant”: (Article 10)
|
Ionis Pharmaceuticals, Inc.
2855 Gazelle Ct.
Attention: General Counsel
Email: [***]
With a copy to:
Cooley LLP
11951 Freedom Drive
Suite 1400
Reston, VA 20190
Attn: Michelle Garcia Schulman, Esq.
Email: [***]
|
| TERMS OF LEASE | DESCRIPTION |
|
|
|
|
“Address of Landlord”: (Article 10)
|
c/o Oxford Property Group
125 Summer Street, 12th Floor
Boston, MA 02110
United States
Attn: Kristin Binck, Esq.,
Vice President, Legal
Email: [***]
With a copy to:
c/o Oxford Property Group
101 Second St, Suite 300
San Francisco, CA 94105
Attn: Abby Mondani
and
DLA Piper LLP (US)
33 Arch Street, 26th floor
Boston, MA 02110
United States
Attn: John L. Sullivan, Esq.
Email: [***]
and all legal notices shall also be sent to:
[***]
|
|
ARTICLE 1 LEASE OF PREMISES; TERM
|
1
|
||
|
|
|
|
|
|
|
1.1
|
Lease
|
1
|
|
|
1.2
|
As Is; No Representations
|
1
|
|
|
1.3
|
Holdover
|
2
|
|
|
1.4
|
Extension Option
|
3 |
|
|
|
|
|
|
ARTICLE 2 RENT
|
4
|
||
|
|
|
||
|
|
2.1
|
Base Rent and Additional Rent
|
4
|
|
|
2.2
|
Expenses; Taxes; Insurance Expenses
|
6 |
|
|
2.3
|
Net Lease
|
8
|
|
|
2.4
|
Interest and Late Charge
|
9
|
|
|
2.5
|
Security Deposit
|
10 |
|
|
|
||
|
ARTICLE 3 USE, COMPLIANCE WITH LAWS, HAZARDOUS MATERIALS, DOGS
|
13 |
||
|
|
|
||
|
|
3.1
|
Use
|
13 |
|
|
3.2
|
Compliance with Applicable Laws
|
13
|
|
|
3.3
|
Hazardous Materials
|
14
|
|
|
3.4
|
Alterations
|
18
|
|
|
3.5
|
Jeopardy of Insurance
|
21
|
|
|
3.6
|
Dogs
|
22 |
|
|
3.7
|
LEED Standards
|
22
|
|
|
|
||
|
ARTICLE 4 REPAIR AND MAINTENANCE; SERVICES
|
22
|
||
|
|
|
||
|
|
4.1
|
Tenant Repair and Maintenance Obligations
|
22
|
|
|
4.2
|
Condition of Premises on Surrender
|
23 |
|
|
4.3
|
Services
|
24 |
|
|
|||
|
ARTICLE 5 ASSIGNMENT AND SUBLETTING
|
25 |
||
|
|
|
||
|
|
5.1
|
General Prohibition on Transfers
|
25 |
|
|
5.2
|
Other Permitted Transfers
|
27
|
|
|
5.3
|
Non-Transfers
|
27
|
|
|
5.4
|
Conditions to Effectiveness
|
28 |
|
|
5.5
|
Miscellaneous
|
28
|
|
|
|
||
|
ARTICLE 6 INDEMNIFICATION; RELEASE; INSURANCE
|
30 |
||
|
|
|
||
|
|
6.1
|
Indemnification and Release
|
30 |
|
|
6.2
|
Insurance
|
30
|
|
|
|
||
|
ARTICLE 7 THIRD PARTIES
|
33
|
||
|
|
|||
|
|
7.1
|
Subordination, Attornment and Non-Disturbance
|
33
|
|
|
7.2
|
Mortgagee’s Right to Cure
|
34
|
|
|
7.3
|
Sale of the Premises
|
34
|
|
|
7.4
|
Estoppel Certificates
|
34
|
|
|
7.5
|
Liens
|
35 |
|
|
|
||
|
ARTICLE 8 EVENTS OF DEFAULT & REMEDIES
|
35
|
||
|
|
|||
|
|
8.1
|
Events of Default
|
35
|
|
|
8.2
|
Remedies
|
37 |
|
|
8.3
|
Landlord Default
|
40
|
|
|
8.4
|
Non-Waiver
|
40
|
|
|
|
||
|
ARTICLE 9 CASUALTY & CONDEMNATION
|
41 |
||
|
|
|
||
|
|
9.1
|
Casualty
|
41 |
|
|
9.2
|
Waiver
|
42
|
|
|
9.3
|
Total Condemnation
|
43 |
|
|
9.4
|
Partial Condemnation
|
43
|
|
|
|
||
|
ARTICLE 10 MISCELLANEOUS
|
43
|
||
|
|
|
||
|
|
10.1
|
Notices
|
43
|
|
|
10.2
|
Certain Representations
|
44 |
|
|
10.3
|
Brokers
|
45
|
|
|
10.4
|
No Waivers
|
45 |
|
|
10.5
|
Future Development
|
45
|
|
|
10.6
|
Other Provisions
|
46
|
|
|
10.7
|
Interpretation
|
48
|
|
|
10.8
|
Tenant’s Signage
|
49
|
|
|
10.9
|
Choice of Law
|
49
|
|
|
10.10
|
CASp Inspection
|
50 |
|
|
10.11
|
Landlord Access
|
50
|
|
|
|
||
|
ARTICLE 11 TENANT’S RIGHT OF FIRST OFFER
|
51
|
||
|
|
|
||
|
|
11.1
|
Right of First Offer
|
51
|
|
|
11.2
|
Tenant’s Competitors
|
52
|
|
|
11.3
|
Transfers
|
52
|
|
|
11.4
|
Termination
|
53
|
|
|
11.5
|
Confidentiality
|
53
|
|
|
|
||
|
ARTICLE 12 LIMITATION OF LIABILITY
|
54
|
||
|
|
|
||
|
|
12.1
|
Landlord’s Liability
|
54
|
|
|
12.2
|
Assignment of Rents
|
54
|
|
Schedule 1
|
Example Showing Calculation of Base Rent
|
|
|
|
|
Exhibit A
|
Legal Description of the Premises
|
|
|
|
|
Exhibit B
|
Work Letter
|
|
|
|
|
Exhibit C
|
Form of Tenant Estoppel Certificate
|
|
|
|
|
Exhibit D
|
Form of Memorandum of Lease
|
|
|
|
|
Exhibit E
|
Form of SNDA
|
|
|
|
|
Exhibit F
|
Form of Non-Disclosure and Confidentiality Agreement
|
|
|
|
|
Exhibit G
|
Landlord Signage
|
|
|
|
|
Exhibit H
|
Commencement and Termination Date Agreement
|
|
|
|
|
Exhibit I
|
Tenant Construction Manual
|
|
|
|
|
Exhibit J
|
Tenant Standard Operating Procedures
|
|
LANDLORD:
|
|
|
|
|
|
LOTS 21 & 22 OWNER (DE) LLC, a Delaware
limited liability company
|
|
|
|
|
|
By: /s/ Tycho Suter
|
|
|
Name: Tycho Suter
|
|
|
Title: Vice President
|
|
|
By: /s/ Kristen Binck
|
|
|
Name: Kristen Binck
|
|
|
Title: Vice President
|
|
|
|
|
|
TENANT:
|
|
|
|
|
|
IONIS PHARMACEUTICALS, INC., a
Delaware corporation
|
|
|
|
|
|
By: /s/ Elizabeth L. Hougen
|
|
|
Name: Elizabeth L. Hougen
|
|
|
Title: CFO
|
|



|
CFM/SF (Supply)
|
CFM/SF (Exhaust)
|
Heating Loads (MBH)
|
Cooling Tons
|
|
|
Typical 40/60 Lab Office Ratio
|
1.52
|
0.93
|
5357
|
1040
|
|
Typical 50/50 Lab Office Ratio
|
1.6
|
1.15
|
5830
|
1163
|


|
“Tenant”
IONIS PHARMACEUTICALS, INC., a
Delaware corporation
|
||
|
|
By:
|
|
|
| Name: | |||
| Title: | |||
|
Date Signed:
|
|
LANDLORD:
LOTS 21 & 22 OWNER (DE) LLC, a
Delaware limited liability company
|
|
|
By:
|
|
|
| Name: | |||
| Title: | |||
|
STATE/COMMONWEALTH OF ____________)
CITY/COUNTY OF ____________)
|
|
|
Notary Public
|
|
|
TENANT:
IONIS PHARMACEUTICALS, INC., a
Delaware corporation
|
|
|
By:
|
|
|
| Name: | |||
| Title: | |||
|
A notary public or other officer
completing this
certificate verifies only the identity of the
individual who signed the document to
which this
certificate is attached, and not the
truthfulness,
accuracy, or validity of that document.
|
|
STATE OF CALIFORNIA
|
)
|
|
|
) §
|
||
|
County of
|
)
|
|
|
SUBORDINATION, NON-DISTURBANCE AND ATTORNMENT AGREEMENT
|
|
If to Tenant:
|
||
|
Primary Address:
|
||
|
|
||
|
|
||
|
|
||
|
Supplement Addresses:
|
||
|
|
||
|
|
||
|
|
||
|
If to Landlord:
|
||
|
Primary Address:
|
||
|
|
||
|
|
||
|
|
||
|
Supplement Addresses:
|
||
|
|
||
|
|
||
|
|
||
|
If to Agent:
|
||
|
Primary Address:
|
||
|
|
||
|
|
||
|
|
||
|
Supplement Addresses:
|
||
|
|
||
|
|
||
|
|
||
|
TENANT
|
||
|
|
||
|
By:
|
|
Name (Print)
|
, |
|
Title:
|
|
|
AGENT: | |
|
|
||
|
By:
|
|
Name (Print)
|
, |
|
Title:
|
|
|
LANDLORD
|
|
|
|
||
|
By:
|
|
Name (Print)
|
, |
|
Title:
|
|
If to Ionis:
|
Ionis Gazelle, LLC
|
||
|
Attention:
|
|||
|
Email:
|
|
with a copy to:
|
Cooley LLP
|
|
|
11951 Freedom Dr.
|
|
Suite 1400
|
|
|
Reston, VA 20190
|
|
|
Attn: Michelle Garcia Schulman, Esq.
|
|
Email: mschulman@cooley.com
|
|||
|
If to Prospective Purchaser:
|
|
||
|
|
|||
|
|
|||
|
|
|||
|
IONIS GAZELLE, LLC
|
[PROSPECTIVE PURCHASER]
|
|||
|
By:
|
By:
|
|||
|
Name:
|
Name:
|
|||
|
Title:
|
Title:
|


|
LANDLORD:
|
|
|
Lots 21 & 22 Owner (DE) LLC
|
|
|
|
By:
|
|
|
| Name: | |||
| Title: | |||
|
TENANT:
|
|
|
Ionis Pharmaceuticals, Inc.
|
|
|
By:
|
|
|
| Name: | |||
| Title: | |||
| 1.01 |
Introduction
|
| 1.02 |
Tenant Coordination
|
| 1.03 |
Tenant Design and Working Drawings
|
|
|
- |
Preliminary space plan
|
|
|
- |
Building Permit submission
|
|
|
- |
Issued for Construction Submission
|
|
|
- |
As-built drawings as further outlined in the Lease
|
|
|
a) |
Location of all major fixed elements within the leased premises dimensionally related to grid lines and demising partitions.
|
|
|
b) |
Location and layout of rooms of unusual loading concentrations such as cranes, racking areas and calculations of unusual loadings which may result in floor damage.
|
|
|
c) |
Location of power, telephone, data and communications outlets. Room names and uses.
|
|
|
d) |
Floor materials and finishes throughout the premises.
|
|
|
e) |
Location of exit lights.
|
| 1.04 |
Certificates and Approvals
|
|
|
1. |
Insurance. Landlord requires evidence of insurance for contractors and subcontractors as required by the Lease.
|
|
|
2. |
Permits Tenant’s design and construction work must comply with all applicable by-laws. The Tenant must obtain all necessary permits and approvals or construction work in the leased premises
from the appropriate government authorities. Permits and approval that are required to commence construction must be obtained before construction begins within the leased premises. A copy of all permits must be delivered to the Landlord.
The Tenant must correct immediately any work, which does not meet with the approval of the building inspector, even though the Tenants drawings may have been reviewed previously by the appropriate government authorities and the Landlord.
|
|
|
3. |
Applicable Hazardous Materials Law. All contractors, sub-trades and suppliers shall abide by applicable laws governing use of Hazardous Materials when working in the Building.
|
|
|
4. |
Workers Compensation To the extent applicable, Tenant contractor shall furnish written evidence of good standing with the North Carolina Industrial Commission and that all employees engaged
in the work are covered in accordance with the statutory requirements of authorities having jurisdiction.
|
|
|
5. |
Occupational Health and Safety The Tenant acknowledges that it is solely responsible for the health and safety of all its employees and workers, as well as for the continuing safe conditions
in the Premises. The Tenant shall comply with and shall require all of its employees and workers to comply with the provisions of all Laws respecting Occupational Health and Safety, the Environment, Worker’s Compensation and the safe
condition of the Premises.
|
| 1.05 |
Appointment of Contractors
|
| 1.06 |
Reserved
|
| 1.07 |
Commencement of Construction
|
|
|
a) |
Submitted acceptable evidence of insurance coverage to the Landlord as set out in these Rules and Regulations.
|
|
|
b) |
Posted all required permits and safety signage on site, where applicable.
|
|
|
c) |
For work subject to Landlord’s consent, made available on the leased premises one (1) set of prints, of the Tenant Design Working Drawings and Specifications for the duration of the construction period for reference by the Landlord’s
Tenant Coordinator.
|
|
|
d) |
For work subject to Landlord’s consent, submitted a schedule showing the timetable for the progress and completion of the Tenant’s work and a list of all trades requiring access to the premises including the trades address and telephone
number.
|
| 1.08 |
Completion of Tenant’s Construction
|
|
|
2.01 |
Inspection of Tenant Work in Progress
|
|
|
2.02 |
Security Control
|
|
|
2.03 |
Public Safety
|
|
|
2.04 |
Emergency Contact
|
|
|
2.05 |
Temporary Services
|
|
|
2.06 |
Work Areas
|
|
|
2.07 |
Waste Removal
|
|
|
2.08 |
Drilling or Cutting
|
|
|
2.09 |
Welding
|
|
|
2.10 |
Hot Work Permits
|
|
|
2.11 |
Wiring and Conduit
|
|
|
2.12 |
Smoking
|
|
|
2.13 |
PENALTIES FOR FALSE ALARMS
|
|
|
◾ |
Drawings, Specifications and Scope of Work as outlined within this document.
|
|
|
◾ |
Insurance Certificate provided on Landlord’s standard form.
|
|
|
◾ |
Building Permit or copy of Building Permit application.
|
|
|
◾ |
List of Contractors and Trades to be used including contact names and phone numbers.
|
|
|
◾ |
Detailed Construction Schedule.
|
|
|
◾ |
Emergency Contact Numbers for all contractors and supervisors responsible for project.
|
|
|
◾ |
Fire Alarm and Life Safety Verification.
|
|
|
◾ |
Architect’s Certificate of Completion.
|
|
|
◾ |
Final Electrical and Mechanical engineers sign-off stating work is completed substantially in accordance with design drawings and specifications.
|
|
|
◾ |
Copies of all permits and certificates related to work.
|
|
|
◾ |
As-built Mechanical Drawings with the associated CAD disk ver. 14 or later. Disks are to be labeled as follows:
|
|
|
• |
Indicate “As-Builts” – Mechanical
|
|
|
• |
Name of Contractor
|
|
|
• |
Project Name, Floor and Address
|
|
|
• |
Project Date (Month-Year)
|
|
|
• |
Name of Company Prepared By
|
|
|
◾ |
As and to the extent required by the Lease, As-built Electrical Drawings with the associated CAD disk ver. 14 or later. Disks are to be labeled as follows:
|
|
|
• |
Indicate “As-Builts” – Electrical
|
|
|
• |
Name of Contractor
|
|
|
• |
Project Name, Floor and Address
|
|
|
• |
Project Date (Month-Year)
|
|
|
• |
Name of Company Prepared By
|
|
|
1. |
Workers Compensation: Statutory Coverage in accordance with the laws of your
state.
|
|
|
2. |
Employers Liability: Limits of not less than $1,000,000 each accident/occurrence,
$1,000,000 each employee/disease, $1,000,000 disease/policy limit.
|
|
|
3. |
General Liability: Please see the chart below for General Liability per
Occurrence/ General Aggregate.
|
|
|
4. |
Automobile Liability: Bodily injury and property damage in an amount not less
than $2,000,000 combined single limit covering all owned, non-owned, hired or leased vehicles.
|
|
|
5. |
Excess / Umbrella Liability: $1,000,000 in excess of the above primary Employer’s
Liability, General Liability, and Automobile Liability.
|
|
|
6. |
Property: Property Insurance will cover the physical loss, including theft, or
damage to equipment, machinery, supplies or tools owned, leased, hired or borrowed by contractor, utilized or operated by contractor while performing contracted services. The valuation basis shall be “replacement cost”.
|
|
|
7. |
Professional Liability (Errors and Omissions): If the nature of the work involves
a professional liability exposure (e.g. design/build), contractor shall maintain professional liability (errors and omissions) coverage at a minimum limit of $2,000,000 or $5,000,000, depending on project size, for each claim.
|
|
|
8. |
Contractors Pollution - Asbestos Legal Liability: If the nature of the services
performed involves pollutants or any other materials which would affect soil, water or structures, then the contractor shall maintain contractors pollution – asbestos legal liability coverage for a limit of not less than $1,000,000 each
occurrence - $2,000,000 policy aggregate, including errors and omissions. However, see attached requirements for higher limits for asbestos abatement and hazardous material removal contractors.
|
|
|
1. |
For insurance requirements for crane lifts or any special contract work; contact for the property team for specific coverage and language.
|
|
|
2. |
Commercial General Liability Coverage Required (millions, per occurrence and aggregate) is the sum of the basic coverage + excess umbrella.
|
|
|
• |
Commercial General Liability Coverage Required = 5MM. This requirement is met by:
|
|
|
• |
General Liability each Occurrence 3MM + Excess umbrella 2MM = 5MM
|
|
|
• |
General Liability General Aggregate 3MM + Excess umbrella 2MM = 5MM
|
|
Type of Service
|
General Liability
Each Occurrence /
General Aggregate
|
Employers’ Liability
Each Accident / Disease
– Each Employee /
Disease – Policy Limit
|
Automobile
Liability
Combined
Single Limit
|
Excess / Umbrella
Liability Each
Occurrence / Aggregate
|
|
Alarm Systems Service and Repair
|
1MM / 2MM
|
1MM
|
1MM
|
2MM
|
|
Appliance Repair &
Maintenance
|
1MM / 1MM
|
1MM
|
1MM
|
1MM
|
|
Architectural *
|
3MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Asbestos Abatement and Hazardous Material Removal
****
|
5MM / 5MM
|
1MM
|
1MM
|
10MM
|
|
Audio-Visual Equipment
|
1MM / 1MM
|
1MM
|
1MM
|
1MM
|
|
Backflow Testing
|
1MM/1MM
|
1MM
|
1MM
|
1MM
|
|
Cabling
|
1MM / 1MM
|
1MM
|
1MM
|
2MM
|
|
Carpet/Floor Finishes
|
1MM / 2MM
|
1MM
|
1MM
|
2MM
|
|
Crane/Rigging
|
5MM / 5MM
|
1MM
|
1MM
|
10MM
|
|
Custom Fabrication &
Installation Millwork
|
1MM / 1MM
|
1MM
|
1MM
|
2MM
|
|
Doors & Locks
|
1MM / 1MM
|
1MM
|
1MM
|
1MM
|
|
Electrical Maintenance
|
3MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Elevator/Escalator Service & Maintenance
|
5MM / 5MM
|
1MM
|
1MM
|
10MM
|
|
Elevator interior installation
|
2MM / 3MM
|
1MM
|
1MM
|
5MM
|
|
Engineering Consulting
Service*
|
3MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Fire Extinguishing in
Restaurants
|
1MM / 1MM
|
1MM
|
1MM
|
2MM
|
|
Fitness Equipment Maintenance
|
1MM / 2MM
|
1MM
|
1MM
|
2MM
|
|
Garbage Removal & Disposal, incl. dumpster maintained on premises
|
1MM / 2MM
|
1MM
|
1MM
|
2MM
|
|
General Contractors
|
3MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Generator Maintenance
|
1MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Glass Repair & Maintenance
|
1MM / 2MM
|
1MM
|
1MM
|
2MM
|
|
Glass Repair & Maintenance elevated
|
5MM / 5MM
|
1MM
|
1MM
|
10MM
|
| Type of Service |
General Liability
Each Occurrence /
General Aggregate
|
Employers’ Liability
Each Accident / Disease
– Each Employee /
Disease – Policy Limit
|
Automobile
Liability
Combined
Single Limit
|
Excess / Umbrella
Liability Each
Occurrence / Aggregate
|
|
Graffiti Removal
|
1MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Handyman
|
1MM / 1MM
|
1MM
|
1MM
|
2MM
|
|
Heating, Ventilation & Air Conditioning Service/install
|
3MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Insulation/Fiberglass
|
1MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Interior Design Consulting*
|
1MM / 2MM
|
1MM
|
1MM
|
2MM
|
|
Life Safety/Fire Equipment
|
3MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Life Safety/Monitoring
|
3MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Lighting re-lamping (interior)
|
1MM / 2MM
|
1MM
|
1MM
|
2MM
|
|
Elevated Lighting Maintenance
|
5MM /5MM
|
1MM
|
1MM
|
5MM
|
|
Moves/Relocations/ reconfiguration
|
3MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Office Equipment Service
|
1MM / 2MM
|
1MM
|
1MM
|
2MM
|
|
Overhead and Revolving Door
|
1MM / 2MM
|
1MM
|
1MM
|
2MM
|
|
Painting
|
1MM / 2MM
|
1MM
|
1MM
|
2MM
|
|
Parking Surface
Maintenance/sweeping
|
2MM / 2MM
|
1MM
|
1MM
|
2MM
|
|
Paving and Striping
|
2MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Plumbing
|
3MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Power washing (non-elevated)
|
1MM / 2MM
|
1MM
|
1MM
|
2MM
|
|
Power washing (elevated)
|
5MM /5MM
|
1MM
|
1MM
|
5MM
|
|
Pump Maintenance
|
3MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Roofing
|
5MM /5MM
|
1MM
|
1MM
|
10MM
|
|
Signage non elevated
|
2MM / 2MM
|
1MM
|
1MM
|
2MM
|
|
Signage Elevated
|
5MM / 5MM
|
1MM
|
1MM
|
10MM
|
|
Sprinkler System Service and Repair
|
3MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Stonework/Marble/wood/ metal cleaners and refinishers Repair & Maintenance
|
1MM / 2MM
|
1MM
|
1MM
|
2MM
|
| Type of Service |
General Liability
Each Occurrence /
General Aggregate
|
Employers’ Liability
Each Accident / Disease
– Each Employee /
Disease – Policy Limit
|
Automobile
Liability
Combined
Single Limit
|
Excess / Umbrella
Liability Each
Occurrence / Aggregate
|
|
Telecommunications and TV Equip. Master Wiring and Antennas (non-elevated)
|
3MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Telecommunications and TV Equip. Master Wiring and Antennas (elevated or roof)
|
5MM / 5MM
|
1MM
|
1MM
|
5MM
|
|
UPS/SEP Equipment Maintenance
|
3MM / 3MM
|
1MM
|
1MM
|
2MM
|
|
Walk Off Mat Cleaning
|
1MM / 1MM
|
1MM
|
1MM
|
2MM
|
|
Water Treatment
|
1MM / 2MM
|
1MM
|
1MM
|
2MM
|
|
Window coverings (non- elevated)
|
1MM / 1MM
|
1MM
|
1MM
|
2MM
|
|
Window Washing and Swing Station Equipment Services
|
5MM / 5MM
|
1MM
|
1MM
|
10MM
|
|
|
o |
Architects = 5MM
|
|
|
o |
Engineers = 5MM
|
|
|
o |
Interior Design = 2MM (or call Risk Management if limited scope)
|
|
|
o |
Any deviation from requested amount should be cleared through Oxford Properties Risk Management.
|
|
|
o |
Errors & Omissions is not based on spend, but rather the scope detail (access liability), such as no structural issues and what amount of damage/cost could be sustained if done incorrectly.
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
| 2.1. |
The intent of this collaboration is for the Parties to conduct research programs directed to each of the gene targets [***] and [***]
(each a “Target”). The research programs will be (i) an Ionis [***] program
initially centered around the research of Oligonucleotides designed to bind to the RNA that encodes [***] with the goal of designating an Ionis Development Candidate for Huntington’s Disease and (ii) an Ionis [***] program centered around
the research of Oligonucleotides designed to bind to the RNA that encodes [***] with the goal of designating an Ionis Development Candidate for Alzheimer’s Disease, and in each case ((i) and (ii)), Ionis will deliver for each Program a Handoff Data Package to Roche. On a Program-by-Program basis, (a) Ionis will perform and fund the activities allocated to it under the applicable R&D Plan from the
Effective Date until the date of Handoff for such Program (the “Handoff Period”); (b) after Handoff has taken place, Roche will be responsible for all further development, manufacturing, regulatory and commercial activities related to the Products for such Program (including drafting the IND for
such Products), unless agreed otherwise in a written agreement signed by each Party’s authorized representative; and (c) the Parties, through a joint manufacturing committee, will discuss and agree upon a manufacturing strategy for each
Program through [***] for such Program.
|
|
Confidential
|
Execution Version
|
| 2.2. |
In addition, the Parties intend to enter into an agreement to conduct [***]. Upon completion of [***]. Any [***], which will incorporate
the terms and principles set forth in SCHEDULE 2.2 but in any event will not [***].
|
| 2.3. |
The purpose of this ARTICLE 2 is to provide a high-level overview of the roles, responsibilities, rights and obligations of each
Party under this Agreement with regard to the R&D Plans and Products, and therefore this ARTICLE 2 is qualified in its entirety by the more detailed provisions of this Agreement set forth below.
|
| 3.1. |
Programs.
|
|
|
3.1.1. |
R&D Plans. The R&D Plans for each Target are attached hereto as APPENDIX 2. Each such R&D Plan sets forth the research and
preclinical Development activities that Ionis (with certain contributions by Roche) will conduct within the given timelines to designate at least one Ionis Development Candidate and deliver one Handoff Data Package.
|
|
|
3.1.2. |
Conducting the R&D Plans. For each Target, during the Handoff Period, Ionis and Roche will
use Commercially Reasonable Efforts to conduct the research and Development activities allocated to such Party according to the applicable R&D Plan with the goal of delivering at least one Compound that fulfills the Success Criteria
for such Target.
|
|
|
3.1.3. |
Ionis Development Candidate Designation; IND-Enabling Toxicology Study Costs.
|
|
|
(a) |
Unless otherwise mutually agreed by the Parties, Ionis’ Research Management Committee (“RMC”) [***].
For a given Compound that Ionis’ RMC proposes as a potential Ionis Development Candidate, Ionis will provide to Roche the applicable Development Candidate Data Package, which Development Candidate Data Package will identify the proposed
Compound and all Related Compounds. Within [***] after Roche’s receipt of a Development Candidate Data Package pursuant to this Section 3.1.3(a), Roche will notify Ionis in writing of
any omissions or deficiencies that Roche reasonably believes in good faith cause the Development Candidate Data Package to be incomplete with respect to the applicable proposed Ionis Development Candidate or Related Compound(s) described
therein (a “Development Candidate Data Package Deficiency Notice”). Within [***] after Ionis’ receipt of a Development Candidate Data
Package Deficiency Notice, Ionis will resubmit a complete Development Candidate Data Package to Roche. Within [***] after Roche’s receipt of the applicable complete Development Candidate Data
Package, the JSC will convene to discuss and recommend the proposed Compound (or, alternatively, a Related Compound) as an Ionis Development Candidate. Should the JSC recommend the designation of the Compound (or a Related Compound) as an
Ionis Development Candidate, then within [***] after such recommendation, Roche shall confirm designation of such Compound as an Ionis Development Candidate.
|
|
Confidential
|
Execution Version
|
|
|
(b) |
If the JSC recommends such designation and Roche confirms such designation, then subject to Section 3.1.6 (Roche Performing IND- Enabling Toxicology Studies for
First Ionis Development Candidate), Ionis shall be responsible to perform IND-Enabling Toxicology Studies with respect to such Ionis Development Candidate. The cost for such studies, including CMC cost, shall be handled, as follows: (i)
if Ionis performs the IND-Enabling Toxicology Studies, then [***] will pay for the cost of such IND-Enabling Toxicology Studies for such Ionis Development Candidate (subject to Section 7.2 ([***])), and [***] will pay [***] for
the Costs of the IND-Enabling Toxicology Studies for any subsequent Ionis Development Candidate, if applicable, for the same Target, and (ii) if Roche performs the IND-Enabling Toxicology Studies, then [***] will pay all costs for such
studies.
|
|
|
(c) |
If (i) the JSC does not recommend such designation (and does not, as an alternative, recommend designation of a Related Compound), or
(ii) the JSC recommends such designation (or, alternatively, the designation of a Related Compound) but Roche does not confirm the designation of such Compound (or Related Compound, as applicable) as an Ionis Development Candidate within
[***], then, unless the Compound is [***] and designated by Ionis as a potential Follow-On Ionis Development Candidate under Section 3.6.2 (Follow-On Development Candidates [***]), Ionis shall have the right to terminate the
Agreement for such Program in accordance with Section 3.10 (Failure to Identify or Designate an Ionis Development Candidate).
|
|
|
3.1.4. |
Handoff Data Package. Once available to Ionis, Ionis will promptly deliver to Roche the
pharmacology, toxicology, histology and pharmacokinetic data generated from the IND-Enabling Toxicology Studies and any other studies it is responsible for under the R&D Plans and will provide Roche with Ionis’ portion of the
applicable Handoff Data Package (i.e., the data for which Ionis is responsible). Within [***] after receipt of such information, Roche will notify Ionis of any omissions or deficiencies that
Roche believes in good faith cause Ionis’ portion of the Handoff Data Package to be incomplete with respect to the applicable Ionis Development Candidate. Within [***] after Ionis’ receipt of
such notice from Roche, Ionis will resubmit its complete portion of the Handoff Data Package to Roche. Roche will complement the Handoff Data Package with data that Roche is responsible for, and the JSC will then review the Handoff Data
Package and assess if the Handoff Data Package Criteria are met, subject to Section 3.1.7 (Failure to Achieve Handoff Data Package Criteria; Disputes over Achievement of Handoff Data Package Criteria). For clarity, [***]. If the Parties mutually agree to [***], then the Parties will memorialize such agreement in a writing signed by each Party’s authorized representative.
|
|
Confidential
|
Execution Version
|
|
|
3.1.5. |
Handoff. On an Ionis Development Candidate-by-Ionis Development Candidate basis, once the
Handoff Data Package Criteria for the applicable Ionis Development Candidate is deemed to have been achieved as set forth in Section 3.1.4 (Handoff Data Package) or Section 3.1.6 (Roche Performing IND- Enabling
Toxicology Studies for First Ionis Development Candidate), as applicable (the “Handoff”), the responsibility for further Development of such Ionis Development Candidate
(including drafting the IND for such Ionis Development Candidate) transitions from Ionis to Roche, and Ionis will have no further responsibilities under the applicable R&D Plan.
|
|
|
3.1.6. |
Roche Performing IND-Enabling Toxicology Studies for First Ionis Development Candidate.
No later than [***] prior to the JSC’s expected designation of an Ionis Development Candidate for a given Target under Section 3.1.3 (Ionis Development Candidate Designation;
IND-Enabling Toxicology Study Costs), Roche may, at its discretion, decide to perform IND-Enabling Toxicology Studies for such Ionis Development Candidate and prepare the related data package to be included within the Handoff Data Package
instead of Ionis. In such instance, the milestone payment set forth in Section 7.2 ([***]) will not apply and [***] shall be responsible
for the costs of such IND-Enabling Toxicology Studies. Once available to Roche, Roche will consolidate the respective pharmacology, toxicology, histology and pharmacokinetic data generated from the IND-Enabling Toxicology Studies together
with other data each Party may be responsible for and present the completed Handoff Data Package at the JSC. The JSC will then convene to discuss such data package and confirm whether the Handoff Data Package Criteria have been met. If
the JSC does not agree as to whether the Handoff Data Package Criteria have been met, then such matter shall be resolved in accordance with Section 3.1.7 (Failure to Achieve Handoff Data Package Criteria; Disputes over Achievement
of Handoff Data Package Criteria). For clarity, [***]. If the Parties mutually agree to [***], then the Parties will memorialize such agreement in a writing signed by each Party’s authorized representative.
|
|
|
3.1.7. |
Failure to Achieve Handoff Data Package Criteria; Disputes over Achievement of Handoff Data Package Criteria.
|
|
|
(a) |
If the Parties agree that the Handoff Data Package Criteria for a given Ionis Development Candidate are not met, then for a period of [***] beginning [***], as applicable, the Parties shall reasonably
cooperate to identify suitable Related Compound(s) and Roche may select one of these Related Compounds and present it to the JSC to decide whether to designate such Related Compound as a replacement of the initial Ionis Development
Candidate. If the JSC designates such Related Compound as an Ionis Development Candidate, then, at [***] but subject to Section 3.1.7(b), either Roche or Ionis will perform the IND-Enabling Toxicology Studies for such Ionis
Development Candidate, and [***] will pay for the costs of such studies.
|
|
Confidential
|
Execution Version
|
|
|
(b) |
If the Parties disagree whether the Handoff Data Package Criteria have been met with respect to a given Ionis Development Candidate and Roche wishes to select a Related Compound to replace the initial Ionis
Development Candidate, then for a period of [***] beginning [***], Roche may so designate such Related Compound as a replacement Ionis Development Candidate, and Roche will perform the IND-Enabling Toxicology Studies for such Ionis
Development Candidate at [***] expense.
|
|
|
3.1.8. |
Development of Additional Compounds.
|
|
|
(a) |
Related Compounds. On a Target-by Target basis, at any time after [***] under Section
3.1.3(a), provided that [***], Roche may initiate a Development program on a Related Compound [***] Roche will promptly provide written notice to Ionis following [***], which notice will
identify the Related Compound that is the subject of such Development program. Roche will be solely responsible for all subsequent Development and Commercialization of such Related Compound.
|
|
|
(b) |
Compounds that Are Not Related Compounds. At any time after the Effective Date, provided that [***], if Roche wishes to [***] then Roche will notify Ionis in writing of the [***]. Within [***] after such notice, the JSC (or the Parties, if the JSC has been dissolved) will meet to
discuss whether [***]. Following such meeting, Roche may [***], unless [***]. Following such meeting [***], Roche will be responsible for all subsequent Development and Commercialization of such Compound.
|
|
|
3.1.9. |
Performance Milestones During the Handoff Period. For each Target, during the Handoff Period:
|
|
|
(a) |
Ionis will use Commercially Reasonable Efforts to present to the JSC (i) at least [***] for [***] that Ionis in good faith believes meets the Success Criteria within [***] after
the Effective Date and (ii) at least [***] for [***] that Ionis in good faith believes meets the Success Criteria within [***] after the Effective Date;
|
|
|
(b) |
if Ionis is conducting the IND-Enabling Toxicology Studies for a given Ionis Development Candidate, then Ionis will use Commercially Reasonable Efforts to complete the IND-Enabling Toxicology Studies for such
Ionis Development Candidate within [***] after the designation of such Ionis Development Candidate; and
|
|
|
(c) |
if Roche is conducting the IND-Enabling Toxicology Studies for a given Ionis Development Candidate, then Roche will use Commercially Reasonable Efforts to complete the IND-Enabling Toxicology Studies for such
Ionis Development Candidate within [***] after the designation of such Ionis Development Candidate.
|
|
Confidential
|
Execution Version
|
| 3.2. |
Disclosure of Results. Ionis and Roche will provide reports and analyses at each JSC meeting,
and more frequently [***], detailing the current status of each R&D Plan.
|
| 3.3. |
Governance.
|
|
|
3.3.1. |
Joint Steering Committee.
|
|
|
(a) |
Establishment; Composition; Working Groups; Dissolution. Within [***] after the Effective Date,
the Parties will establish a joint steering committee (“JSC”) to govern the activities under the R&D Plans (i.e. until Handoff). The Parties may utilize either a single JSC
or may choose to have a JSC for each Target. The JSC will consist of [***] qualified representatives appointed by Ionis, and [***] qualified representatives appointed by Roche. Each Party will designate one of its representatives to act
as the co-chairperson of the JSC. The co- chairpersons will be responsible for overseeing the activities of the JSC. The JSC will determine the JSC operating procedures at its first meeting, which will be codified in the written minutes
of the first JSC meeting. The JSC will determine the frequency of meetings, location of meetings, and responsibilities for agendas and minutes. The JSC may hold meetings in person or by audio or video conference as determined by the JSC.
Alliance Managers are invited to attend JSC meetings as participating non-members. In addition, upon prior approval of the other Party, each Party may invite its employees or consultants to attend JSC meetings, including any subject
matter expert(s) with valuable knowledge of [***], [***], Huntington’s Disease, or Alzheimer’s Disease. The co-chairpersons will be responsible for ensuring that activities occur as set forth in this Agreement, including ensuring that JSC
meetings occur, JSC recommendations are properly reflected in the minutes, and any dispute is given prompt attention and resolved in accordance with Section 3.3.1(c) (Decision-Making), Section 8.2.3 (Inventorship) and Section
13.1 (Dispute Resolution), as applicable. The JSC will form one or more subcommittees and working groups as it determines in order to discuss progress under the R&D Plans, [***], and to carry out its other activities under this
Agreement. Any such subcommittees or working groups may act as an informal forum for the exchange of data arising from the performance of the R&D Plans, and will dissolve when the JSC dissolves. Reasonably in advance ([***]) of any
meeting of [***], Ionis will share with any such working group [***]. On a Target-by-Target basis, the JSC will dissolve once all Ionis Development Candidates have either [***] or been terminated in accordance with Section 11.2
(Termination).
|
|
|
(b) |
Responsibilities. The JSC will perform the following functions:
|
|
|
(i) |
Oversee the Parties’ activities under the respective R&D Plans;
|
|
|
(ii) |
Oversee subcommittees of the JSC;
|
|
Confidential
|
Execution Version
|
|
|
(iii) |
Approve amendments to the R&D Plans;
|
|
|
(iv) |
Approve amendments to the Success Criteria;
|
|
|
(v) |
Determine whether a Development Candidate Data Package is complete;
|
|
|
(vi) |
Determine whether to recommend designating a Compound or, if applicable, Related Compound as an Ionis Development Candidate following review of the applicable Development Candidate Data Package;
|
|
|
(vii) |
Determine whether the Handoff Data Package Criteria have been satisfied;
|
|
|
(viii) |
Approve the Technology Transfer Plan and amendments following Handoff;
|
|
|
(ix) |
Record recommendations and decisions of the JSC in the JSC’s meeting minutes; and
|
|
|
(x) |
Such other review and advisory responsibilities assigned to the JSC pursuant to this Agreement.
|
|
|
(c) |
Decision-Making.
|
|
|
(i) |
Committee Decision-Making. Decisions by the JSC will be made by unanimous consent with each
Party’s representatives having, collectively, one vote. At any given meeting of the JSC, a quorum will be deemed reached if a voting representative of each Party is present or participating in such meeting. No action taken at any meeting
of the JSC will be effective unless there is a quorum at such meeting. Unless otherwise specified in this Agreement, no action will be taken with respect to a matter for which the JSC has not reached unanimous consensus.
|
|
|
(ii) |
Final Decision-Making Authority.
|
|
|
(1) |
If the JSC cannot unanimously agree on a matter to be decided by it within [***], then the matter will be referred to the Executives for resolution as set forth in Section 13.1.1 (Escalation), without
application of Section 13.1.2 (Binding Arbitration).
|
|
|
(2) |
Except as set forth otherwise in this Agreement, if the Executives cannot reach agreement on a matter referred by the JSC within [***], then: (A) each Party will have final decision-making authority with
respect to the conduct of its activities under each R&D Plan (provided that such conduct is in accordance with the applicable R&D Plan); (B) Roche will have final decision- making authority
with respect to (1) [***], (2) [***], (3) [***], (x) [***], (y) [***], and (z) [***], (4) [***], (5) [***], and (6) [***].
|
|
Confidential
|
Execution Version
|
|
|
(3) |
Additional Costs Due to [***]. For a given Program, if [***] or [***], then such increased Costs
shall be shared as follows: [***] shall bear such Costs [***]; and [***] shall bear such Costs [***].
|
|
|
(4) |
Notwithstanding anything to the contrary in this Section 3.3.1(c)(ii) (Final Decision-Making Authority), a Party will not have final decision-making authority with respect to [***] of a Product
if the other Party has a reasonable good faith belief that [***].
|
|
|
(5) |
Except as otherwise expressly stated in this Agreement, the JSC will have no decision-making authority and will act as a forum for sharing information about the activities conducted by the Parties hereunder
and as an advisory body, in each case only on the matters described in, and to the extent set forth in, this Agreement.
|
|
|
(iii) |
Decision-Making After Handoff. After Handoff has occurred for a given Ionis Development
Candidate, the JSC has no authority to make decisions with respect to such candidate.
|
|
|
3.3.2. |
Joint Manufacturing Committee.
|
|
|
(a) |
Establishment; Composition; Dissolution. Within [***] after the creation of the JSC, the Parties
will establish a joint manufacturing committee (“JMC”) to govern the manufacturing of API to support the activities under the R&D Plans (i.e. until Handoff). The JMC will be
deemed a subcommittee of the JSC, as described above in Section 3.3.1(a) (Establishment; Composition; Working Groups; Dissolution), and will have the responsibilities set forth in Section 3.3.2(b) (JMC
Responsibilities). The JMC will consist of [***] qualified representatives appointed by Ionis, and [***] qualified representatives appointed by Roche. Alliance Managers are invited to attend JMC meetings as participating non-members. In
addition, upon prior approval of the other Party, each Party may invite its employees or consultants to attend JMC meetings, including any subject matter expert(s) with valuable knowledge of [***], [***], Huntington’s Disease, or
Alzheimer’s Disease. The JMC will dissolve upon the earlier of (i) dissolution of the JSC, (ii) termination of this Agreement, and (iii) the Parties’ mutual agreement to dissolve the JMC.
|
|
|
(b) |
JMC Responsibilities. The JMC will perform the
following functions:
|
|
|
(i) |
develop, discuss and approve the manufacturing strategy and plan for manufacturing API and Finished Drug Product for each Program through [***] for such Program, which plan will be part of the R&D Plan
for the applicable Program;
|
|
Confidential
|
Execution Version
|
|
|
(ii) |
establish and revise the Handoff Data Package Criteria related to CMC matters;
|
|
|
(iii) |
determine whether the Handoff Data Package Criteria related to CMC matters for each Ionis Development Candidate have been satisfied;
|
|
|
(iv) |
establish the manufacturing part of the Technology Transfer Plan;
|
|
|
(v) |
perform such other functions as determined by the JSC.
|
|
|
(c) |
JMC Decision-Making.
|
|
|
(i) |
Committee Decision-Making. Decisions by the JMC will be made by unanimous consent with each
Party’s representatives having, collectively, one vote. At any given meeting of the JMC, a quorum will be deemed reached if a voting representative of each Party is present or participating in such meeting. No action taken at any meeting
of the JMC will be effective unless there is a quorum at such meeting. Unless otherwise specified in this Agreement, no action will be taken with respect to a matter for which the JMC has not reached unanimous consensus.
|
|
|
(ii) |
Escalation to JSC. If the JMC cannot unanimously agree on a matter to be decided by it within
[***], then the matter will be referred to the JSC for resolution in accordance with Section 3.3.1(c)(ii) (Final Decision-Making Authority).
|
|
|
3.3.3. |
Alliance Managers. Each Party will appoint a representative to act as its alliance manager
(each, an “Alliance Manager”) for facilitating communication and collaboration between the Parties. Each Alliance Manager will be responsible for (a) facilitating resolution of
potential and pending issues and potential disputes to enable the JSC to reach consensus and avert escalation of such issues or potential disputes, (b) supporting the co-chairpersons with organization of JSC meetings, information
exchange, meeting minutes, as necessary, and (c) preparing status and progress reports on the above as determined necessary by the JSC.
|
| 3.4. |
Manufacturing and Supply. Unless otherwise expressly agreed to by the JMC:
|
|
|
3.4.1. |
Supplies for Activities under R&D Plans.
|
|
|
(a) |
Supplies for Activities During Handoff Period. Subject to Section 3.4.1(c)
(Technology Transfer During Handoff Period), on a Program- by-Program basis, during the Handoff Period for such Program, at [***], Ionis will supply cGMP [***] for the [***] Program, and [***] API, or a combination of both [***] API for
the [***] Program, in each case for use in IND- Enabling Toxicology Studies for the first Ionis Development Candidate generated under the applicable R&D Plan and, at [***] cost, cGMP material for use in the [***] for such Ionis
Development Candidate. Ionis’ supply obligation hereunder will be at least [***], but no more than [***] of cGMP API per Ionis Development Candidate.
|
|
Confidential
|
Execution Version
|
|
|
(b) |
Supplies for Activities After Handoff. After Handoff, Ionis will deliver to Roche, if Roche
desires, any inventory of cGMP API, radiolabeled material, cGMP Finished Drug Product, packaged clinical trial material, and cGMP packaged trial material containing the Ionis Development Candidate then in Ionis’ possession, and Roche will
pay Ionis for such materials at Ionis’ Fully Absorbed Cost of Goods, as set forth in SCHEDULE 3.4.1(b).
|
|
|
(c) |
Technology Transfer During Handoff Period. For any Compound (except [***] as an Ionis
Development Candidate that is designed to bind to the RNA that encodes [***]), Roche may, at its discretion, no later than [***] before the anticipated designation of an Ionis Development Candidate, request a technology transfer under Section
3.5 (Technology Transfer Implementation) and assume responsibility to manufacture and supply API for the IND-Enabling Toxicology Studies for such Ionis Development Candidate (and for all future Clinical Studies of such Ionis
Development Candidate).
|
| 3.5. |
Technology Transfer Implementation. After Handoff for a given Ionis Development Candidate (or
earlier if agreed between the Parties), the Parties will establish a technology transfer plan (“Technology Transfer Plan”) under which Ionis will provide reports and information
generated by or on behalf of Ionis in performing its activities under the applicable R&D Plan that are necessary for Roche to draft the IND, and the Parties will conduct the transfer activities with respect to the applicable Ionis
Development Candidate in accordance with such Technology Transfer Plan.
|
|
|
3.5.1. |
Licensed Know-How – Generally. As part of the activities to be performed under the Technology
Transfer Plan, Ionis will deliver to Roche copies of Licensed Know-How with respect to the applicable Ionis Development Candidate (other than the Ionis Manufacturing and Analytical Know-How) in the Field in Ionis’ possession not
previously provided hereunder, for use solely in accordance with the license granted under Section 5.1.2 (Development and Commercialization License Grant to Roche) to Roche for such Ionis Development Candidate, together with all
regulatory documentation (including drafts) related to such Ionis Development Candidate. To assist with the transfer of such Licensed Know-How, Ionis will make its personnel reasonably available to Roche during normal business hours to
transfer such Licensed Know-How.
|
|
|
3.5.2. |
Ionis Manufacturing and Analytical Know-How. As part of the activities to be performed under the
Technology Transfer Plan, Ionis will deliver, at Roche’s election, to one of either (a) [***], or (b) [***] solely to Manufacture API on Roche’s behalf, copies of the Ionis Manufacturing and Analytical Know-How relating to the applicable
Ionis Development Candidate in Ionis’ possession not previously provided hereunder, which is necessary for the exercise by Roche, its Affiliates or a Third Party of the Manufacturing rights granted under Section 5.1.2 (Development
and Commercialization License Grant to Roche).
|
|
|
3.5.3. |
Technology Transfer Costs. Ionis will provide up to [***] of technology transfer activities
under this Section 3.5 (Technology Transfer Implementation) at [***]. Thereafter, if reasonably requested by Roche, Ionis will provide reasonable transfer support, and [***].
|
|
Confidential
|
Execution Version
|
| 3.6. |
Follow-On Ionis Development Candidates.
|
|
|
3.6.1. |
Follow-On Development Candidates [***]. On a Program- by-Program basis, for a period starting
from [***] and ending on the date that is [***], if Ionis’ RMC designates as a development candidate ready to start IND-Enabling Toxicology Studies one or more [***] discovered by or on behalf of Ionis that are designed to bind to the RNA
that encodes the applicable Target for such Program and that satisfy the Success Criteria and that are not Compounds, then Ionis will notify Roche in writing and will provide Roche with the data package presented to Ionis’ RMC in
connection with the approval by Ionis’ RMC of such [***]. Roche will then have [***] from delivery of such data package to provide written notice to Ionis electing to deem such [***] as a Follow-On Ionis Development Candidate and
thereafter such Follow-On Ionis Development Candidate and [***] shall be considered Compounds, and Roche shall continue activities under this Agreement with respect to such Follow-On Ionis Development Candidate. If Roche provides such
timely written notice, then Roche will be responsible for the conduct and costs of any further research, Development, Manufacturing, and Commercialization activities for such Follow-On Ionis
Development Candidate, and all terms of this Agreement will apply to such Follow-On Ionis Development Candidate, including, but not limited to, Section 7.3 (One-time Milestone Payments for Achievement of Milestone Events), Section
7.4 (Royalties), and Section 7.5 (Royalty Payments and Reports). If [***], then [***].
|
|
|
3.6.2. |
Follow-On Development Candidates [***]. Subject to Section 3.6.3 ([***]), on a
Program-by-Program basis, for a period starting from [***] and ending on the date that is [***], if Ionis’ RMC designates as a development candidate ready to start IND-Enabling Toxicology Studies one or more [***] discovered by Ionis that
are designed to bind to the RNA that encodes the applicable Target for such Program and that satisfy the Success Criteria and that are not Compounds, then Ionis will notify Roche in writing and will provide Roche with the data package
presented to Ionis’ RMC in connection with the approval by Ionis’ RMC of such [***]. Roche will then have [***] from delivery of such data package to provide written notice to Ionis electing to deem such [***] as a Follow-On Ionis
Development Candidate and thereafter continue activities under this Agreement with respect to such Follow-On Ionis Development Candidate. If Roche provides such timely written notice, then such Follow-On Ionis Development Candidate and
[***] shall be considered Compounds, and Roche will be responsible for the conduct and costs of any further research, Development, Manufacturing, and Commercialization activities for such Follow-On
Ionis Development Candidate, and all terms of this Agreement will apply to such Follow-On Ionis Development Candidate, including, but not limited to, Section 7.3 (One-time Milestone Payments for Achievement of Milestone Events),
Section 7.4 (Royalties), and Section 7.5 (Royalty Payments and Reports). If [***], then [***].
|
|
|
3.6.3. |
Roche [***]. On a [***] basis, at any time after [***] until the [***], if [***], then [***].
Upon [***] receipt of [***], [***] will [***] with respect to [***].
|
|
Confidential
|
Execution Version
|
|
|
3.6.4. |
Agreement by the Parties to Discover and Develop Additional Ionis Development Candidates.
At any time after the expiration of the period ending [***] after [***], the Parties may mutually agree to collaborate on the research, discovery and development of an Oligonucleotide designed to bind to a Target that is not a Related
Compound or a Follow-On Ionis Development Candidate. Any such future collaboration is subject to the Parties’ good faith negotiation and execution of a new agreement or an amendment of this Agreement.
|
| 3.7. |
Subcontracting. Each Party may engage Third Party subcontractors to perform certain of its
obligations under this Agreement. Any subcontractor engaged to perform a Party’s obligations under this Agreement will meet the qualifications typically required by such Party for the performance of work similar in scope and complexity
and will execute such Party’s standard nondisclosure agreement. Any Party engaging a subcontractor hereunder will remain responsible for such activities.
|
| 3.8. |
Materials Transfer. To facilitate the activities under each R&D Plan, either Party may
provide certain materials for use by the other Party. All such materials will be used by the receiving Party in accordance with terms of this Agreement solely for purposes of exercising its rights and performing its obligations under this
Agreement, and the receiving Party will not transfer such materials to any Third Party except with the written consent of the supplying Party. Except as expressly set forth herein, THE MATERIALS ARE PROVIDED “AS IS” AND WITHOUT ANY
REPRESENTATION OR WARRANTY, EXPRESS OR IMPLIED, INCLUDING ANY IMPLIED WARRANTY OF MERCHANTABILITY OR OF FITNESS FOR ANY PARTICULAR PURPOSE OR ANY WARRANTY THAT THE USE OF THE MATERIALS WILL NOT INFRINGE OR VIOLATE ANY PATENT OR OTHER
PROPRIETARY RIGHTS OF ANY THIRD PARTY.
|
| 3.9. |
Applicable Laws. Each Party will perform its activities pursuant to this Agreement in compliance
with good laboratory and clinical practices and, except where stated otherwise, cGMP, in each case as applicable under the laws and regulations of the country and the state and local government wherein such activities are conducted.
|
| 3.10. |
Failure to Identify or Designate an Ionis Development Candidate. On a Program- by-Program basis:
|
|
|
3.10.1. |
if, despite Ionis’ Commercially Reasonable Efforts, Ionis has not proposed any Compound for potential
designation as an Ionis Development Candidate within (a) [***] after [***] for the [***] Program, or (b) [***] after [***] for the [***] Program, then, notwithstanding any provision to the contrary in this Agreement, (i) work under the
applicable R&D Plan will stop; (ii) the Parties will no longer have an obligation to perform any activities under this ARTICLE 3 with respect to the applicable Program; and (iii) either Party may terminate the applicable
Program, as further specified in Section 11.2.6(a); and
|
|
Confidential
|
Execution Version
|
|
|
3.10.2. |
if the JSC does not recommend the designation of a proposed Compound (and does not, as an alternative, recommend designation of a Related Compound) within the timeline set forth in Section 3.1.3(a),
or Roche, following the JSC’s recommendation to designate a Compound (or Related Compound, as applicable) as an Ionis Development Candidate, does not confirm such recommendation [***] in accordance with Section 3.1.3 (Ionis
Development Candidate Designation; IND-Enabling Toxicology Study Costs), then, except as set forth in Section 3.1.3(c), and notwithstanding any other provision to the contrary in this Agreement, (a) work under the applicable R&D
Plan will stop; (b) the Parties will no longer have an obligation to perform any activities under this ARTICLE 3 with respect to the applicable Program; and (c) Ionis may terminate the applicable Program, as further specified in Section
11.2.6(b).
|
| 4.1. |
Exclusivity.
|
|
|
4.1.1. |
[***] Exclusivity Covenants Before Handoff. On a Target-by-Target basis, except in the
performance of its obligations or exercise of its rights under this Agreement, and except as set forth in Section 4.1.3 (Limitations and Exceptions to Exclusivity Covenants), [***] will work [***] (including the grant of any
license to any Third Party) with respect to the [***] that is designed to bind to the RNA that encodes the applicable Target in the Field from the Effective Date until the earlier of (a) the date of Handoff with respect to the first Ionis
Development Candidate directed to such Target and (b) termination of this Agreement with respect to such Target.
|
|
|
4.1.2. |
[***] Exclusivity Covenants After Handoff. On a Target-by-Target basis, except in the
performance of its obligations or exercise of its rights under this Agreement, and except as set forth in Section 4.1.3 (Limitations and Exceptions to Exclusivity Covenants), [***] will not work on its own or with any of its
Affiliates or any Third Party (including the grant of any license to any Third Party) with respect to the [***] that is designed to bind to the RNA that encodes the applicable Target in the Field from the date of Handoff with respect to
the first Ionis Development Candidate directed to such Target until the earlier of (a) the [***] and (b) termination of this Agreement with respect to such Target; provided, however, that if (i)
[***], then [***] exclusivity obligations hereunder will end on the [***], and (ii) provided that [***], then [***] exclusivity obligations hereunder will end on the [***].
|
|
|
4.1.3. |
Limitations and Exceptions to Exclusivity Covenants. Notwithstanding anything to the
contrary in Section 4.1.1 ([***] Exclusivity Covenants Before Handoff) and Section 4.1.2 ([***] Exclusivity Covenants After Handoff), the Parties and their Affiliates may perform the following activities:
|
|
|
(a) |
all activities permitted or contemplated under this Agreement; and
|
|
|
(b) |
with regard to Ionis and its Affiliates: (i) any activities pursuant to the Prior Agreements as in effect on the Effective Date; and (ii) the granting of, or performance of obligations or exercise of rights
under, Permitted Licenses.
|
|
Confidential
|
Execution Version
|
| 4.2. |
Effect of Exclusivity on Indications. Ionis and Roche are subject to certain exclusivity
covenants under Section 4.1.1 ([***] Exclusivity Covenants Before Handoff) and Section 4.1.2 ([***] Exclusivity Covenants After Handoff); however, the Parties acknowledge and agree
that each Party (on its own or with a Third Party or an Affiliate) may pursue products for the same indication as a Product so long as such product does not breach Section 4.1 (Exclusivity).
|
| 5.1. |
License Grants; Sublicense Rights.
|
|
|
5.1.1. |
Research License Grant to Roche. Subject to the terms of this Agreement, Ionis hereby grants to
Roche a worldwide, non-exclusive, royalty-free, sublicensable (in accordance with Section 5.1.4 (Sublicense Rights) below) license under the Licensed Technology to perform the Roche R&D Activities during the Handoff Period; provided that, (a) for clarity, such license shall not include a license to conduct any clinical Development activities with respect to, or Commercialize, any Compounds or Products, and (b) such
license includes a license under the Ionis Manufacturing and Analytical Patents and Ionis Manufacturing and Analytical Know-How to Manufacture API only if Roche elects to assume responsibility for Manufacturing and supplying such API
under Section 3.4.1(c) (Technology Transfer During Handoff Period).
|
|
|
5.1.2. |
Development and Commercialization License Grant to Roche. Subject to the terms of this
Agreement, Ionis hereby grants to Roche a worldwide, exclusive, royalty-bearing, sublicensable (in accordance with Section 5.1.4 (Sublicense Rights) below) license under the Licensed Technology to research, Develop, Manufacture,
have Manufactured (in accordance with Section 5.1.4 (Sublicense Rights) below) and Commercialize Compounds and Products in the Field; provided that [***].
|
|
|
5.1.3. |
Cross-Licenses under Collaboration Intellectual Property.
|
|
|
(a) |
Enabling License to Roche from Ionis. Subject to the terms of this
Agreement (including [***]), Ionis hereby grants to Roche a worldwide, fully paid-up, royalty-free, non-exclusive, sublicensable license under any Ionis Collaboration Technology to research, develop,
manufacture, have manufactured and commercialize products that are not Products in the Field; and
|
|
|
(b) |
Enabling License to Ionis from Roche. Subject to the terms of this Agreement (including [***]),
Roche hereby grants to Ionis a worldwide, fully paid-up, royalty-free, non-exclusive, sublicensable license under any Roche Collaboration Technology, to research, develop, manufacture, have manufactured and commercialize products that are
not Products in the Field.
|
|
Confidential
|
Execution Version
|
|
|
5.1.4. |
Sublicense Rights.
|
|
|
(a) |
Subject to the terms of this Agreement, Roche will have the right to grant sublicenses under any license granted under Section 5.1.1 (Research License Grant to Roche), Section 5.1.2
(Development and Commercialization License Grant to Roche) and Section 5.1.3(a) (Enabling License to Roche from Ionis):
|
|
|
(i) |
under the Ionis Core Technology Patents, Ionis Product-Specific Patents, Ionis Collaboration Technology and Ionis Know-How to an Affiliate of Roche or a Third Party; and
|
|
|
(ii) |
under the Ionis Manufacturing and Analytical Patents and Ionis Manufacturing and Analytical Know-How solely to (y) [***] or (z) [***].
|
|
|
(b) |
Requests to Grant Sublicenses to CMOs. If Roche provides Ionis with a written request that Ionis
grant a license under the Ionis Manufacturing and Analytical Patents and Ionis Manufacturing and Analytical Know- How to a CMO designated by Roche that is not a Licensed CMO, solely for such CMO to Manufacture Products for Roche, its
Affiliate or Sublicensee in a manufacturing facility owned or operated by such CMO, Ionis will [***].
|
|
|
(c) |
Enforcing Sublicenses. Each sublicense by Roche under this Agreement will be subject to, and
consistent with, the terms of this Agreement. Roche shall be responsible to ensure compliance by its Sublicensees with the terms and conditions of this Agreement. If Ionis reasonably believes a Roche Sublicensee may be violating the terms
of this Agreement, then, within [***] after [***], Roche will provide Ionis a full and complete copy of the sublicense Roche entered with such Sublicensee.
|
|
|
5.1.5. |
No Implied Licenses. All rights in and to Licensed Technology not expressly licensed to Roche
under this Agreement are hereby retained by Ionis or its Affiliates. All rights in and to Roche Technology not expressly licensed or assigned to Ionis under this Agreement, are hereby retained by Roche or its Affiliates. Except as
expressly provided in this Agreement, no Party will be deemed by estoppel or implication to have granted the other Party any license or other right with respect to any intellectual property.
|
|
|
5.1.6. |
License Conditions; Limitations. Any license granted under Section 5.1.2 (Development
and Commercialization License Grant to Roche) and the sublicense rights under Section 5.1.4 (Sublicense Rights) are subject to and limited by (a) the Permitted Licenses, and (b) the Prior Agreements, in each case [***].
|
|
|
5.1.7. |
Trademarks for Products. Roche shall be the owner and solely responsible for all trademarks,
trade dress, logos, slogans, designs, copyrights and domain names used on or in connection with Products licensed under Section 5.1.2 (Development and Commercialization License Grant to Roche).
|
|
Confidential
|
Execution Version
|
| 6.1. |
Roche Development, Manufacturing & Commercialization
|
|
|
6.1.1. |
Responsibility. On an Ionis Development Candidate-by-Ionis Development Candidate basis, after
Handoff for an Ionis Development Candidate, subject to the terms of this Agreement, Roche is solely responsible for all Development, Manufacturing and Commercialization activities, and for all costs and expenses associated therewith, with
respect to the Development, Manufacture and Commercialization of Products containing such Ionis Development Candidate.
|
|
|
6.1.2. |
Clinical Development Plan. After the Handoff for a given Ionis Development Candidate Roche will
provide Ionis with a written [***] plan [***] of the [***] of the applicable Product through [***]. Such plan will include key [***] events for the applicable Product [***] for the achievement of such events [***] for such Product. Roche
will provide to Ionis [***].
|
|
|
6.1.3. |
[***]. On an Ionis Development Candidate-by-Ionis Development Candidate basis, after Handoff for
an Ionis Development Candidate, [***] Roche will provide to Ionis the [***] for such Ionis Development Candidate, and subsequent updated versions of [***], [***]. The sharing of the [***] contemplated hereunder [***], and such [***]
shall be Roche’s Confidential Information.
|
|
|
6.1.4. |
Regulatory Communications and Submissions. As between the Parties, Roche will be responsible for
(a) determining the regulatory plans and strategies for the Products, (b) making all regulatory filings with respect to the Products, and (c) obtaining and maintaining Approvals in the name of Roche or its Affiliates or Sublicensees. Up
to and including [***], Roche will provide Ionis with any Key Regulatory Submissions to any Regulatory Authority in each Major Market for such Product before providing such submission to the applicable Regulatory Authority for Ionis to
provide any comments on the contents thereof. Ionis will have [***] from the date of receipt of such submission to provide comments with respect thereto. Roche will [***] by Ionis. Following [***], Roche [***] also [***]. For
clarification, [***].
|
|
|
6.1.5. |
Class Generic Claims. If Roche intends to make any claims in a Product label or regulatory
filing that are class generic to Oligonucleotides or Ionis’ chemistry platform(s), then Roche will provide such claims and regulatory filings to Ionis in advance and will [***] any proposals and comments made by Ionis.
|
| 6.2. |
Diligence. On a Target-by-Target basis, after Handoff for an Ionis Development Candidate, Roche
will use Commercially Reasonable Efforts to further Develop and Commercialize at least one Product with respect to the Target for such Ionis Development Candidate in the Field in the Territory. Without limiting the foregoing, within [***]
after [***], Roche will [***]; provided, however, [***].
|
|
Confidential
|
Execution Version
|
| 6.3. |
IND; Global Safety Database.
|
|
|
6.3.1. |
IND. Roche will be the holder of the IND for each Ionis Development Candidate and will assume
responsibility for the global safety database related to such Ionis Development Candidate. After Handoff, Roche will be solely responsible for reporting to Regulatory Authorities in accordance with the Applicable Law for expeditable
adverse events and for periodic safety reporting relating to the safety of such Ionis Development Candidate and all subsequent Ionis Development Candidates with respect to the same Target.
|
|
|
6.3.2. |
Ionis’ Antisense Safety Database.
|
|
|
(a) |
Ionis maintains an internal database that includes information regarding the tolerability of its drug compounds, individually and as a class, including information discovered during pre-clinical and clinical
development (the “Ionis Internal Oligonucleotide Safety Database”). In an effort to maximize understanding of the safety profile and pharmacokinetics of Ionis compounds, after
Handoff, Roche will cooperate in connection with populating the Ionis Internal Oligonucleotide Safety Database. Roche and Ionis will [***].
|
|
|
(b) |
Ionis utilizes the information in the Ionis Internal Oligonucleotide Safety Database to conduct analyses to keep Ionis and its partners informed regarding class generic properties of Oligonucleotides,
including with respect to safety. As such, if and when Ionis identifies safety or other related issues that may be relevant to a Product (including any potential class-related toxicity), Ionis will promptly inform Roche of such issues and,
if requested, provide the data supporting Ionis’ conclusions.
|
| 7.1. |
Upfront Fee. Within [***] days following the Effective Date and receipt by Roche of an invoice
from Ionis, Roche will pay Ionis an upfront fee equal to sixty million dollars ($60,000,000). All dollar amounts in this Agreement are in U.S. dollars.
|
| 7.2. |
[***] IND-Enabling Toxicology Studies [***]. For the first Ionis
Development Candidate for a given Target, [***] will decide which Party will conduct IND-Enabling Toxicology Studies in accordance with Section 3.1.6 (Roche Performing IND-Enabling Toxicology Studies for First Ionis Development
Candidate), and will either (a) [***], or (b) [***]. In case of (a) above, [***].
|
| 7.3. |
One-time Milestone Payments for Achievement of Milestone Events.
|
|
|
7.3.1. |
Development Milestones for [***]. Subject to Section 7.3.1(a) ([***]) and Section 7.3.1(b)
([***]), Roche will pay Ionis the milestone payments set forth in TABLE 1 below the first time a milestone event listed in TABLE 1 is first achieved by a Product directed to [***], regardless
of how many times thereafter such Milestone Event is achieved by a subsequent Product to such Target.
|
|
Confidential
|
Execution Version
|
|
Milestone Event
|
Milestone Payment for [***] Product
|
|
|
[***]
|
$[***]
|
|
|
[***]
|
$[***]
|
|
|
[***]
|
$[***]
|
|
|
[***]
|
$[***]
|
|
|
[***]
|
$[***]
|
|
|
[***]
|
$[***]
|
|
|
Total [***] Milestones
|
$[***]
|
|
|
(a) |
[***]. The Milestone Event payment for “[***]” in TABLE 1 will be [***],
subject to the following [***]:
|
|
|
(i) |
If achievement of the “[***]” Milestone Event is [***] and [***] achieves the “[***]” Milestone Event, then [***].
|
|
|
(ii) |
Following achievement of the “[***]” Milestone Event with a Product [***] and payment of the corresponding milestone payment, if [***], then [***].
|
|
|
(b) |
[***]. If (i) following [***] with respect to a Product directed to [***], [***], (ii) [***],
(iii) [***], and (iv) [***], then [***]. The [***].
|
|
|
7.3.2. |
Development Milestones for [***].
|
|
|
(a) |
Subject to Section 7.3.2(b) ([***]), Roche will pay Ionis the milestone payments set forth in TABLE 2 below the first time a Milestone Event listed in TABLE 2
is first achieved by a Product directed to [***], regardless of how many times thereafter such Milestone Event is achieved by a subsequent Product to such Target.
|
|
Milestone Event
|
Milestone Payment for [***] Product
|
|
|
[***]
|
$[***]
|
|
|
[***]
|
$[***]
|
|
|
[***]
|
$[***]
|
|
|
[***]
|
$[***]
|
|
|
[***]
|
$[***]
|
|
|
[***]
|
$[***]
|
|
|
Total [***] Milestones
|
$[***]
|
|
Confidential
|
Execution Version
|
|
|
(b) |
[***]. If (i) [***], (ii) [***], (iii) [***], and (iv) [***], then [***].
|
|
|
7.3.3. |
Milestone Payments for First Achievement of Sales Milestone Event by [***] Product. Roche will
pay Ionis the applicable one-time milestone payments set forth in TABLE 3 below after the first achievement of the listed events by a first Product directed against [***], regardless of how many times thereafter
another Product directed against [***] achieves such event, by or on behalf of Roche or its Affiliates or Sublicensees. For clarity, notwithstanding any provision to the contrary in this Agreement, [***]. Net Sales will be [***], as
applicable.
|
|
Table 3
|
|
|
Sales Milestone
|
Sales Milestone Payment
|
|
≥ $[***] in aggregate worldwide Annual Net Sales of [***] Product
|
$[***]
|
|
|
≥ $[***] in aggregate worldwide Annual Net Sales of [***] Product
|
$[***]
|
|
|
≥ $[***] in aggregate worldwide Annual Net Sales of [***] Product
|
$[***]
|
|
|
Total Sales Milestone Payments
|
$[***]
|
|
|
7.3.4. |
Limitations on Milestone Payments; Exceptions; Notice.
|
|
|
(a) |
Each milestone payment set forth in TABLE 1 in Section 7.3.1 (Development Milestones for [***]) and in TABLE 2 in Section 7.3.2 (Development
Milestones for [***]) and in TABLE 3 in Section 7.3.3 (Milestone Payments for First Achievement of Sales Milestone Event by [***] Product) above will be paid only once upon the first achievement of the
Milestone Event regardless of how many times such Milestone Event is achieved by the same or another Product directed to the same Target.
|
|
|
(b) |
If a particular Development Milestone Event (i.e., [***]) in TABLE 1 in Section 7.3.1 (Development Milestones for [***]) or in TABLE 2 in Section
7.3.2 (Development Milestones for [***]) is not achieved because Development activities transpired such that achievement of such earlier Milestone Event was unnecessary or did not otherwise occur, then upon achievement of a later
Milestone Event the Milestone Event payment applicable to such earlier Milestone Event will also be due. For example, if Roche proceeds directly to “[***]” without achieving the “[***],” then upon achieving the “[***]” Milestone Event, both
the “[***]” and “[***]” Milestone Event payments are due to Ionis.
|
|
Confidential
|
Execution Version
|
|
|
(c) |
Each time a Milestone Event is achieved under this Section 7.3 (One- time Milestone Payments for Achievement of Milestone Events), Roche will send to Ionis a written notice thereof promptly (but no
later than [***]) following the date of achievement of such Milestone Event and such payment will be due within [***] of the date such Milestone Event was achieved and receipt of an invoice by Roche from Ionis.
|
| 7.4. |
Royalties.
|
|
|
7.4.1. |
Royalty Rates. As partial consideration for the rights granted to Roche hereunder, subject to
the provisions of this Section 7.4.1 (Royalty Rates) and Section 7.4.3 (Royalty Adjustments), Roche will pay to Ionis royalties on worldwide Annual Net Sales of Products sold by Roche, its Affiliates or Sublicensees, on a
country-by-country and Product-by-Product basis during the Royalty Term, in each case in the amounts (on an incremental basis) as follows:
|
|
Table 4
|
|||||
|
Royalty Tier
|
Annual Worldwide Net Sales
|
Royalty Rate for Product
Comprising an Ionis
Development Candidate
|
|||
|
1
|
For the portion of Annual worldwide Net Sales < $[***]
|
[***]%
|
|||
|
2
|
For the portion of Annual worldwide Net Sales ≥ $[***] but < $[***]
|
[***]%
|
|||
|
3
|
For the portion of Annual worldwide Net Sales ≥ $[***] but < $[***]
|
[***]%
|
|||
|
4
|
For the portion of Annual worldwide Net Sales ≥ $[***]
|
[***]%
|
|||
|
|
(a) |
For purposes of TABLE 4, worldwide Annual Net Sales for a particular Product will be calculated by summing the aggregate Net Sales for that Product for all countries in the world for
such Product. For example, if worldwide Net Sales of a Product for a given Calendar Year are $[***], then royalties owed to Ionis on such Net Sales of such Product for that Calendar Year shall equal [***] dollars ($[***]), calculated as
follows:
[([***]) + ([***])] = $[***] royalty payment
|
|
|
7.4.2. |
Royalty Term. Roche’s obligation to pay Ionis royalties with respect to a Product will continue
on a country-by-country and Product-by-Product basis from the date of First Commercial Sale of such Product in such country until the latest of (a) the date of expiration of the last Valid Claim [***], as applicable ([***]) that are
licensed by Ionis to Roche under this Agreement that Covers the Product in the country in which such Product is made, used or sold, (b) the date of [***], and (c) the [***] anniversary after the First Commercial Sale of such Product in
such country (the “Royalty Term”). Notwithstanding anything herein to the contrary, [***].
|
|
Confidential
|
Execution Version
|
|
|
7.4.3. |
Royalty Adjustments. For the purpose of calculating royalties owed for a Product, the royalty
rates set forth in Section 7.4.1 (Royalty Rates) shall be subject to the following adjustments, as applicable:
|
|
|
(a) |
Net Sales Adjustment for [***] Product.
|
|
|
(i) |
If Roche or its Affiliates or Sublicensees intend to [***], then the Parties shall meet approximately [***] after [***] to negotiate in good faith and agree to an appropriate adjustment to Net Sales to
reflect [***]. If, after such good faith negotiations not to exceed [***], the Parties cannot agree to an appropriate adjustment, the dispute shall be initially referred to the Executives in accordance with Section 13.1.1
(Escalation), without application of Section 13.1.2 (Binding Arbitration).
|
|
|
(ii) |
If the Executives are unable to agree on the [***] within [***] of such referral (a “[***]”), then such [***] Dispute shall be determined by arbitration pursuant to the terms set forth in SCHEDULE
7.4.3(a)(ii).
|
|
|
(b) |
Adjustment of [***] for a [***] Product. On a [***] Product-by-[***] Product basis, for purposes
of determining the [***] for a [***] Product, [***] shall be the [***] of such [***] Product in the relevant country.
|
|
|
(c) |
No Valid Claim. Subject to Section 7.4.3(g) (Maximum Deduction), for a given Product,
(i) if in a given country within the Territory there is no Valid Claim [***] that Covers the [***] Product, then the royalty rates listed in TABLE 4 in Section 7.4.1 (Royalty Rates) applicable to such
Product will be reduced by [***] %; (ii) if [***], then [***]; and (iii) if both (i) and (ii) of this Section 7.4.3(c) (No Valid Claim) apply, then [***].
|
|
|
(d) |
Generic Product. Subject to Section 7.4.3(g) (Maximum Deduction), on a
country-by-country and Product-by-Product basis, if at any time during the Royalty Term a Generic Product is sold in such country and the aggregate Net Sales of such Product in such country in any Calendar Quarter thereafter decline by
more than [***]% of the level of the Net Sales of such Product achieved in any of the [***] full Calendar Quarters immediately prior to the Calendar Quarter in which the Generic Product was first sold, then the royalty rates listed in TABLE
4 in Section 7.4.1 (Royalty Rates) applicable to such Product in such country will be [***] reduced by [***] ([***]%).
|
|
|
(e) |
[***] Adjustment. Subject to Section 7.4.3(g) (Maximum Deduction), if at any time during
the Royalty Term (i) [***], (ii) [***], and (iii) [***], then the royalty rates listed in TABLE 4 in Section 7.4.1 (Royalty Rates) applicable to such [***] Product will be [***] ([***]%) in the [***] solely
for [***]. For any subsequent Calendar Quarter in which the [***].
|
|
Confidential
|
Execution Version
|
|
|
(f) |
Third Party Payments.
|
|
|
(i) |
Additional Ionis Core Intellectual Property.
|
|
|
(1) |
Roche will promptly provide Ionis written notice of any [***] (“Additional Ionis Core IP”) that Roche believes it has identified, and Ionis will
have the first right, but not the obligation, to negotiate with, and obtain a license from, the Third Party Controlling such Additional Ionis Core IP. For clarity, Additional Ionis Core IP does not include any Patent Rights claiming (or
intellectual property related to) [***]. If Ionis obtains such a Third Party license, then Ionis will include such Additional Ionis Core IP in the license granted to Roche under Section 5.1.2 (Development and Commercialization
License Grant to Roche), and any financial obligations under such Third Party agreement will be paid [***].
|
|
|
(2) |
If, however, Ionis elects not to obtain such a license to such Third Party intellectual property, then Ionis will so notify Roche, and Roche may obtain such a Third Party license and, subject to Section
7.4.3(g) (Maximum Deduction), Roche may offset an amount equal to [***]% of any [***] paid by Roche under such Third Party license against any [***] due to Ionis under Section [***] of this Agreement in such country for
[***].
|
|
|
(3) |
If Ionis does not agree with Roche that a license to such [***], then Ionis will send written notice to such effect to Roche, and the Parties will engage a mutually agreed independent Third Party intellectual
property lawyer, not regularly engaged or employed by either Party within the [***] prior to such written notice, with expertise in the patenting of Oligonucleotides and appropriate professional credentials in the relevant jurisdiction, to
determine the question of whether such Third Party intellectual property is Additional Ionis Core IP. The determination of the Third Party expert engaged under the preceding sentence will be binding on the Parties solely for purposes of
determining whether Roche is permitted to apply the offset under Section 7.4.3(f)(i)(2) above. The costs of any Third Party expert engaged under this Section 7.4.3(f)(i)(3) will be paid by the Party against whom the Third
Party lawyer makes his or her determination.
|
|
Confidential
|
Execution Version
|
|
|
(ii) |
Other Necessary Third Party Intellectual Property. Roche shall be responsible for and pay or
have paid any consideration owed to any Third Party in relation to any other Third Party intellectual property rights necessary to make, use or sell Products. Subject to Section 7.4.3(g) (Maximum Deduction), Roche shall have the
right to deduct [***]% of such consideration actually paid to a Third Party with respect to a Product from royalty payments otherwise due and payable by Roche to Ionis for such Product under Section 7.4 (Royalties). Any such
deduction shall be permitted on a Product-by-Product and country-by-country basis.
|
|
|
(g) |
Maximum Deduction. Except as set forth in this Section 7.4.3(g) (Maximum Deduction), in
no event shall the aggregate royalty reductions set forth in Section 7.4.3 (Royalty Adjustments) reduce the royalties payable to Ionis for Net Sales of a Product in the applicable country in any given period to an amount that is
less than [***]% of the royalty rates listed in TABLE 4 within Section 7.4.1 (Royalty Rates). Notwithstanding the foregoing, [***]. Roche may carry forward any amounts not utilized as a result of the maximum
deduction cap in this Section 7.4.3(g) (Maximum Deduction) and offset against future royalties payable to Ionis for the applicable Product any amounts that, but for this Section 7.4.3(g) (Maximum Deduction), Roche would
have been entitled to deduct from any royalty payments to Ionis until any amounts not utilized are fully deducted.
|
| 7.5. |
Royalty Payments and Reports.
|
|
|
7.5.1. |
Commencement. Beginning with the Calendar Quarter in which the First Commercial Sale, [***] is
made and for each Calendar Quarter thereafter, Roche will make royalty payments to Ionis under this Agreement within [***] days following the end of each such Calendar Quarter.
|
|
|
7.5.2. |
Royalty Reporting.
|
|
|
(a) |
Each royalty payment will be accompanied by a report, summarizing in writing for the relevant Calendar Quarter on a Product-by-Product basis the following information:
|
|
|
(i) |
Sales in Swiss Francs on a country-by-country basis;
|
|
|
(ii) |
Net Sales in Swiss Francs on a country-by-country basis;
|
|
|
(iii) |
Total worldwide Net Sales in Swiss Francs;
|
|
|
(iv) |
Exchange rate used for the conversion of Net Sales from Swiss Francs to U.S. dollars pursuant to Section 7.6 (Mode of Payment);
|
|
|
(v) |
Royalty rate pursuant to Section 7.4.1 (Royalty Rates) and/or Section 7.4.3 (Royalty Adjustments), as applicable; and
|
|
|
(vi) |
Total royalty payable in U.S. dollars.
|
|
|
(b) |
In addition, Roche will include in each report under this Section 7.5.2 (Royalty Reporting) information regarding any Net Sales of Products sold for named patient, compassionate use or other similar
sales and any consideration received from any Compulsory Sublicensees.
|
|
Confidential
|
Execution Version
|
|
|
(c) |
On a country-by-country basis, after first Approval, if no royalties or other payments from Product sales are payable in respect of a given Calendar Quarter, Roche will submit a written royalty report to
Ionis so indicating together with an explanation as to why no such royalties are payable. In addition, beginning with the Calendar Quarter in which the First Commercial Sale for a Product (or any named patient sale, compassionate use sale
or other similar sales of a Product) is made and for each Calendar Quarter thereafter, within [***] following the end of each such Calendar Quarter, Roche will provide Ionis a preliminary non-binding report estimating the total (A) Sales
and Net Sales for Products projected for such Calendar Quarter, and (B) if available, the amount of any consideration payable to Roche under sublicenses with Compulsory Sublicensees.
|
| 7.6. |
Mode of Payment. All payments under this Agreement will be (i) payable in full in U.S. dollars,
regardless of the country(ies) in which sales are made, (ii) made by wire transfer of immediately available funds to an account designated by Ionis in writing, and (iii) irrevocable and non-refundable. Any corrections to calculations of
royalty payments previously paid shall be adjusted to the next royalty payment due. When calculating the Sales of a Product that occur in currencies other than U.S. dollars, Roche will convert the amount of such sales into Swiss Francs
and then into U.S. dollars using Roche’s then current internal foreign currency translation actually used on a consistent basis in preparing its audited financial statements (currently YTD average rate as reported by Reuters).
|
| 7.7. |
Records Retention. Commencing with the First Commercial Sale [***] of a Product, Roche will keep
complete and accurate records pertaining to the sale of Products for a period of [***] after the year in which such sales occurred, and in sufficient detail to permit Ionis to confirm the accuracy of the Net Sales or royalties paid by
Roche hereunder.
|
| 7.8. |
Audits. During the Agreement Term and for a period of [***] thereafter, at the request and
expense of Ionis, Roche will permit an independent certified public accountant of internationally recognized standing appointed by Ionis, at reasonable times and upon at least [***] written notice, but in no case more than [***] per
Calendar Year, to examine such records as may be necessary for the purpose of verifying the accrual of any milestone payments, the calculation and reporting of Net Sales, and the correctness of any milestone or royalty payment made under
this Agreement for any full Calendar Quarter within the preceding [***]. No Calendar Year can be audited more than once. Any and all records of Roche examined by such independent certified public accountant will be deemed Roche’s
Confidential Information. The independent certified public accountant shall share all draft reports with Roche before the draft audit report is shared with Ionis and before the final document is issued. Upon completion of the audit, the
accounting firm will provide both Roche and Ionis with a written report disclosing whether the calculation of Sales, Net Sales and royalty payments made by Roche are correct and the specific details concerning any discrepancies (“Audit Report”). If any Audit Report shows that Roche’s payments under this Agreement were less than the milestone or royalty amount that should have been paid, then Roche will make
all payments required to be made by paying Ionis the difference between such amounts to eliminate any discrepancy revealed by said inspection with the next payment due, with interest calculated in accordance with Section 7.10
(Interest). If any Audit Report shows that Roche’s payments under this Agreement were greater than the amount that should have been paid, then [***] equal to the difference between the amounts paid by Roche and the amounts that should
have been paid. Ionis will pay all fees charged by such accountant pursuant to the audit, except that, if the audit determines that any additional amounts payable by Roche for an audited period
exceed [***] percent ([***]%) of the amount actually paid for such audited period, then, in addition to paying Ionis any unpaid amounts discovered in such audit, and interest calculated in accordance with Section 7.10 (Interest),
Roche will pay the fees and expenses charged by such accountant. Roche shall also [***].
|
|
Confidential
|
Execution Version
|
| 7.9. |
Taxes.
|
|
|
7.9.1. |
Taxes on Income. Each Party will be solely responsible for the payment of all taxes imposed on
its share of income arising directly or indirectly from the activities of the Parties under this Agreement.
|
|
|
7.9.2. |
Withholding Tax. To the extent the paying Party is required to deduct and withhold taxes on any
payment, the paying Party will pay the amounts of such taxes to the proper governmental authority for the account of the receiving Party and remit the net amount to the receiving Party in a timely manner. The paying Party will promptly
furnish the receiving Party with proof of payment of such taxes. If documentation is necessary in order to secure an exemption from, or a reduction in, any withholding taxes, the Parties will provide such documentation to the extent they
are able to do so. In accordance with the procedures set forth in Section 10.3 (Procedure), the receiving Party will also indemnify the paying Party for any tax, interest or penalties imposed on the paying Party if the paying
Party improperly reduces or eliminates withholding tax based upon representations made by the receiving Party.
|
|
|
7.9.3. |
Tax Cooperation. At least [***] prior to the date a given payment is due under this Agreement,
the non-paying Party will provide the paying Party with any and all tax forms that may be reasonably necessary in order for the paying Party to lawfully not withhold tax or to withhold tax at a reduced rate with respect to such payment
under an applicable bilateral income tax treaty. Following the paying Party’s timely receipt of such tax forms from the non- paying Party, the paying Party will not withhold tax or will withhold tax at a reduced rate under an applicable
bilateral income tax treaty, if appropriate under the Applicable Laws. The non-paying Party will provide any such tax forms to the paying Party upon request and in advance of the due date. Each Party will provide the other with reasonable
assistance to enable the recovery, as permitted by Applicable Law, of withholding taxes resulting from payments made under this Agreement, such recovery to be for the benefit of the Party who would have been entitled to receive the money
but for the application of withholding tax under this Section 7.9 (Taxes). The provisions of this Section 7.9 (Taxes) are to be read in conjunction with the provisions of Section 13.4 (Assignment and Successors)
below.
|
|
Confidential
|
Execution Version
|
| 7.10. |
Interest. Any undisputed payments to be made hereunder that are not paid on or before the date
such payments are due under this Agreement will bear interest at a rate per annum equal to the lesser of (i) [***]% above the six-month CME Term Secured Overnight Financing Rate (USD SOFR) from the date on which such payment would have
been first due until the date of payment, or (ii) the maximum rate permissible under Applicable Law.
|
| 8.1. |
Ownership.
|
|
|
8.1.1. |
Ionis Technology and Roche Technology. As between the Parties, Ionis will own and retain all of
its rights, title and interest in and to the Licensed Know- How and Licensed Patents and Roche will own and retain all of its rights, title and interest in and to the Roche Know-How and Roche Patents, subject to any assignments, rights or
licenses expressly granted by one Party to the other Party under this Agreement.
|
|
|
8.1.2. |
Agreement Technology. Each Party will promptly disclose to the other Party in writing, and will
cause its Affiliates to so disclose, the discovery, development, or creation of any invention made solely or jointly by such Party in connection with the performance of obligations under this Agreement. Except as expressly provided in
this Agreement, neither Party will have any obligation to account to the other for profits with respect to, or to obtain any consent of the other Party to license or exploit, Joint Collaboration Technology by reason of joint ownership
thereof, and each Party hereby waives any right it may have under the laws of any jurisdiction to require any such consent or accounting.
|
| 8.2. |
Joint Patent Committee.
|
|
|
8.2.1. |
Establishment. The Parties will establish a “Joint Patent Committee” or “JPC.” The JPC will serve as the primary contact and forum for discussion between the Parties with respect to intellectual property
matters arising under this Agreement and will cooperate with respect to the activities set forth in this ARTICLE 8. The JPC determines the invention classification for each invention arising under this Agreement. The
classifications are (i) Ionis Product-Specific Patents, (ii) Roche Collaboration Patents, (iii) Ionis Collaboration Patents, and (iv) Joint Collaboration Patents (collectively, the “Program
Patents”). The JPC will endeavor to separate the claims within such Patent Rights into separate and distinct patent applications corresponding with the categories described in this Section 8.2.1 (Establishment) to the
extent possible without diminishing the patentability of the inventions.
|
|
|
8.2.2. |
Scope. The JPC will discuss strategies with regard to (a) prosecution and maintenance, defense
and enforcement of Program Patents licensed to Roche under Section 5.1.2 (Development and Commercialization License Grant to Roche) in connection with a Product; (b) defense against allegations of infringement of Third Party
Patent Rights; and (c) licenses to Third Party Patent Rights or Know-How (including whether to obtain any licenses under any such Third-Party Patent Rights or Know-How, and whether there are any known Third Party Obligations applicable to
a particular Product), in each case ((a)- (c)) to the extent such matter would be reasonably likely to have a material impact on the Agreement or the licenses granted hereunder, which strategies will be considered in good faith by the
Party entitled to prosecute, enforce and defend such Patent Rights hereunder, but will not be binding on such Party.
|
|
Confidential
|
Execution Version
|
|
|
8.2.3. |
Inventorship. The JPC will be responsible for the determination of inventorship. The
determination of inventorship will be in accordance with United States patent laws and therefore will determine if the invention is solely or jointly owned by the relevant Party or Parties. In case of a dispute in the JPC (or otherwise
between Ionis and Roche) over inventorship or classification, if the JPC cannot resolve such dispute, even after seeking the JSC’s input, such dispute will be resolved by an independent patent counsel not engaged or regularly employed in
the past two years by either Party and reasonably acceptable to both Parties. The decision of such independent patent counsel will be binding on the Parties. Expenses of such patent counsel will be shared equally by the Parties.
|
|
|
8.2.4. |
Composition. The JPC will comprise an equal number of members from each Party. The JPC will meet
as often as agreed by them (and at least semi- Annually), to discuss matters arising out of the activities set forth in this ARTICLE 8. The JPC will determine by unanimous consent the JPC operating procedures at its first meeting.
To the extent reasonably requested by either Party, the JPC will solicit the involvement of more senior members of their respective legal departments with respect to critical issues. Each Party’s representatives on the JPC will consider
comments and suggestions made by the other in good faith. If either Party deems it reasonably advisable, the Parties will enter into a mutually agreeable common interest agreement covering the matters contemplated by this Agreement.
|
| 8.3. |
Prosecution and Maintenance of Patents.
|
|
|
8.3.1. |
Patent Filings. The Party responsible for Prosecution and Maintenance of any Patent Rights as
set forth in Section 8.3.2 (Ionis Patents and Roche Patents) and Section 8.3.3 (Joint Collaboration Patents) will endeavor to obtain patent protection for a Product as it Prosecutes and Maintains its other patents
Covering products in development, using counsel of its own choice but reasonably acceptable to the other Party, in such countries as the responsible Party sees fit.
|
|
|
8.3.2. |
Ionis Patents and Roche Patents.
|
|
|
(a) |
Ionis Patents.
|
|
|
(i) |
Ionis Core Technology Patents, Ionis Collaboration Patents, and Ionis Manufacturing and Analytical Patents. Ionis will at all times control and be responsible for all aspects of Prosecution and Maintenance of (A) the Ionis Core Technology Patents, (B) the Ionis Collaboration Patents, and (C) the Ionis Manufacturing
and Analytical Patents.
|
|
Confidential
|
Execution Version
|
|
|
(ii) |
Ionis Product-Specific Patents. On an Ionis Development Candidate-by-Ionis Development Candidate
basis, before Handoff with respect to such Ionis Development Candidate, Ionis will control and be responsible for all aspects of the Prosecution and Maintenance of all Ionis Product-Specific Patents and will use commercially reasonable
efforts to Prosecute and Maintain such Patent Rights. After Handoff and subject to Section 8.3.4 (Other Matters Pertaining to Prosecution and Maintenance of Patents), at Roche’s expense, Roche will control and be responsible for
all aspects of the Prosecution and Maintenance of all Ionis Product-Specific Patents and will either (i) use commercially reasonable efforts to Prosecute and Maintain such Patent Rights or (ii) offer to assign Roche’s entire right, title
and interest in such Patent Rights to Ionis, in which case following any such assignment all licenses granted in this Agreement by Ionis to Roche under such Patent Rights shall become non-exclusive and the exclusivity covenants under Section
4.1 (Exclusivity) will no longer apply to such Patent Rights. Such Ionis Product-Specific Patents will continue to be royalty-bearing under Section 7.4 (Royalties).
|
|
|
(b) |
Roche Patents. Roche will at all times control and be responsible for all aspects of the
Prosecution and Maintenance of all Roche Patents.
|
|
|
8.3.3. |
Joint Collaboration Patents. Roche will control and be responsible for all aspects of the
Prosecution and Maintenance of Joint Collaboration Patents and will (i) use commercially reasonable efforts to Prosecute and Maintain such Patent Rights, or (ii) offer to assign Roche’s entire right, title and interest in such Joint
Collaboration Patents to Ionis, in which case following any such assignment all licenses granted in this Agreement by Ionis to Roche under such Patent Rights shall become non-exclusive and the exclusivity covenants under Section 4.1
(Exclusivity) will no longer apply to such Patent Rights.
|
|
|
8.3.4. |
Other Matters Pertaining to Prosecution and Maintenance of Patents.
|
|
|
(a) |
Each Party will keep the other Party informed through the JPC as to material developments with respect to the Prosecution and Maintenance of the Program Patents for which such Party has responsibility for
Prosecution and Maintenance pursuant to Section 8.3.2 (Ionis Patents and Roche Patents), Section 8.3.3 (Joint Collaboration Patents) or this Section 8.3.4 (Other Matters Pertaining to Prosecution and Maintenance of
Patents), including by providing copies of material data as it arises, any office actions or office action responses or other correspondence that such Party provides to or receives from any patent office, including notice of all
interferences, reissues, re-examinations, oppositions or requests for patent term extensions, and all patent-related filings, and by providing the other Party the timely opportunity to have reasonable input into the strategic aspects of
such Prosecution and Maintenance.
|
|
Confidential
|
Execution Version
|
|
|
(b) |
If Roche elects (i) not to file and prosecute patent applications for the Program Patents for which Roche has responsibility for Prosecution and Maintenance pursuant to Section 8.3.2 (Ionis Patents
and Roche Patents) (“Roche-Prosecuted Patents”) in a particular country, (ii) not to continue the prosecution (including any interferences, oppositions, reissue proceedings,
re-examinations, and patent term extensions, adjustments, and restorations) or maintenance of any Roche-Prosecuted Patent in a particular country, or (iii) not to file and prosecute patent applications for the Roche-Prosecuted Patent in a
particular country following a written request from Ionis to file and prosecute in such country, then Roche will so notify Ionis promptly in writing of its intention in good time to enable Ionis to meet any deadlines by which an action must
be taken to establish or preserve any such Patent Right in such country; and Ionis will have the right, but not the obligation, to file, prosecute, maintain, enforce, or otherwise pursue such Roche- Prosecuted Patent in the applicable
country at its own expense with counsel of its own choice. In such case, Roche will cooperate with Ionis to file for, or continue to Prosecute and Maintain or enforce, or otherwise pursue such Roche-Prosecuted Patent in such country in
Ionis’ own name and Roche will keep a non-exclusive license under such Roche- Prosecuted Patents, and the exclusivity covenants under Section 4.1 (Exclusivity) will no longer apply to such Patent Rights. Notwithstanding anything to
the contrary in this Agreement, if Ionis assumes responsibility for the Prosecution and Maintenance of any such Roche-Prosecuted Patent under this Section 8.3.4(b), then Ionis will have no obligation to notify Roche if Ionis intends
to abandon such Roche-Prosecuted Patent. The analogous situation shall apply mutatis mutandis with regard to Patent Rights (excluding Ionis Core Technology Patents and Ionis Manufacturing and
Analytical Patents) for which Ionis has responsibility for Prosecution and Maintenance pursuant to Section 8.3.2 (Ionis Patents and Roche Patents).
|
|
|
(c) |
The Parties, through the JPC, will cooperate in good faith to determine if and when any divisional or continuation applications will be filed with respect to any Program Patents, and where a divisional or
continuation patent application filing would be practical and reasonable, then such a divisional or continuation filing will be made.
|
|
|
(d) |
If the Party responsible for Prosecution and Maintenance pursuant to Section 8.3.3 (Joint Collaboration Patents) intends to abandon such Joint Collaboration Patent, without first filing a continuation
or substitution, then such Party will notify the other Party of such intention at least [***] days before such Joint Collaboration Patent will become abandoned, and such other Party will have the right, but not the obligation, to assume
responsibility for the Prosecution and Maintenance thereof at its own expense (subject to Section 8.3.1 (Patent Filings)) with counsel of its own choice, in which case the abandoning Party will, and will cause its Affiliates to,
assign to the other Party (or, if such assignment is not possible, grant a fully-paid exclusive license in) all of their rights, title and interest in and to such Joint Collaboration Patents. If a Party assumes responsibility for the
Prosecution and Maintenance of any such Joint Collaboration Patents under this Section 8.3.4(d), then such Party will have no obligation to notify the other Party of any intention of such Party to abandon such Joint
Collaboration Patents.
|
|
Confidential
|
Execution Version
|
|
|
(e) |
In addition, the Parties will consult, through the JPC, and take into consideration the comments of the other Party for all matters relating to interferences, reissues, re-examinations and oppositions with
respect to those Patent Rights in which such other Party (i) has an ownership interest, (ii) has received a license thereunder in accordance with this Agreement, or (iii) may in the future, in accordance with this Agreement, obtain a
license or sublicense thereunder.
|
| 8.4. |
Patent Costs.
|
|
|
8.4.1. |
Joint Collaboration Patents. Unless the Parties agree otherwise, Ionis and Roche will share
equally the Patent Costs associated with the Prosecution and Maintenance of Joint Collaboration Patents according to Section 8.3.3 (Joint Collaboration Patents); provided that, either
Party may decline to pay its share of costs for filing, prosecuting and maintaining any Joint Collaboration Patents in a particular country or particular countries, in which case the declining Party will, and will cause its Affiliates to,
assign to the other Party (or, if such assignment is not possible, grant a fully-paid exclusive license in) all of their rights, titles and interests in and to such Joint Collaboration Patents.
|
|
|
8.4.2. |
Ionis Patents and Roche Patents. Except as set forth in and Section 8.3.4 (Other Matters
Pertaining to Prosecution and Maintenance of Patents), each Party will be responsible for all Patent Costs incurred by such Party prior to and after the Effective Date in all countries in the Prosecution and Maintenance of Patent Rights
for which such Party is responsible under Section 8.3.2 (Ionis Patents and Roche Patents); provided, however, that on an Ionis Development Candidate-by-Ionis Development Candidate basis
after Handoff, Roche will be solely responsible for Patent Costs arising from the Prosecution and Maintenance of the Ionis Product-Specific Patents Covering such Ionis Development Candidate; provided
that, Roche may decline to pay for filing, prosecuting and maintaining any Ionis Product-Specific Patents in a particular country or particular countries, in which case all licenses granted in this Agreement by Ionis to Roche
under such Patent Rights shall become non- exclusive and the exclusivity covenants under Section 4.1 (Exclusivity) will no longer apply to such Patent Rights.
|
|
Confidential
|
Execution Version
|
| 8.5. |
Defense of Claims Brought by Third Parties.
|
|
|
8.5.1. |
Third-Party Claims Regarding Products. If a Third Party initiates a Proceeding claiming a Patent
Right owned by or licensed to such Third Party is infringed by the Development, Manufacture or Commercialization of a Product, (a) Ionis will
have the first right, but not the obligation, to defend against any such Proceeding initiated prior to Handoff at its sole cost and expense and (b) Roche will have the first right, but not the obligation, to defend against any such
Proceeding initiated after Handoff at its sole cost and expense. If the Party having the first right to defend against such Proceeding (the “Lead Party”) elects to defend against
such Proceeding, then the Lead Party will have the sole right to direct the defense and to elect whether to settle such claim (but only with the prior written consent of the other Party, not to be unreasonably withheld, conditioned or
delayed). The other Party will reasonably assist the Lead Party in defending such Proceeding and cooperate in any such litigation at the request and expense of the Lead Party. The Lead Party will provide the other Party with prompt
written notice of the commencement of any such Proceeding that is of the type described in this Section 8.5 (Defense of Claims Brought by Third Parties), and the Lead Party will keep the other Party apprised of the progress of
such Proceeding. If the Lead Party elects not to defend against a Proceeding, then the Lead Party will so notify the other Party in writing within [***] after the Lead Party first receives written notice of the initiation of such
Proceeding, and the other Party (the “Step-In Party”) will have the right, but not the obligation, to defend against such Proceeding at its sole cost and expense and thereafter
the Step-In Party will have the sole right to direct the defense thereof, including the right to settle such claim. In any event, the Party not defending such Proceeding will reasonably assist the other Party and cooperate in any such
litigation at the request and expense of the Party defending such Proceeding. Each Party may at its own expense and with its own counsel join any defense initiated or directed by the other Party under this Section 8.5 (Defense of
Claims Brought by Third Parties). Each Party will provide the other Party with prompt written notice of the commencement of any such Proceeding under this Section 8.5 (Defense of Claims Brought by Third Parties), and such Party
will promptly furnish the other Party with a copy of each communication relating to the alleged infringement that is received by such Party.
|
|
|
8.5.2. |
Third Party Claims Regarding Discontinued Products. If a Third Party initiates a Proceeding
claiming that any Patent Right or Know-How owned by or licensed to such Third Party is infringed by the Development, Manufacture or Commercialization of a Discontinued Product, Ionis will have the first right, but not the obligation, to
defend against and settle such Proceeding at its sole cost and expense. Roche will reasonably assist Ionis in defending such Proceeding and cooperate in any such litigation at the request and expense of Ionis. Each Party may at its own
expense and with its own counsel join any defense directed by the other Party. Ionis will provide Roche with prompt written notice of the commencement of any such Proceeding, or of any allegation of infringement of which Ionis becomes
aware and that is of the type described in this Section 8.5.2 (Third Party Claims Regarding Discontinued Products), and Ionis will promptly furnish Roche with a copy of each communication relating to the alleged infringement
received by Ionis.
|
|
|
8.5.3. |
Interplay Between Enforcement of IP and Defense of Third Party Claims. Notwithstanding the
provisions of Section 8.5.1 (Third Party Claims Regarding Products) and Section 8.5.2 (Third Party Claims Regarding Discontinued Products), to the extent that a Party’s defense against a Third Party claim of infringement
under this Section 8.5 (Defense of Claims Brought by Third Parties) involves (a) the enforcement of the other Party’s Know-How or Patent Rights, or (b) the defense of an invalidity claim with respect to such other Party’s Know-How
or Patent Rights, then, in each case, the general concepts of Section 8.6 (Enforcement of Patents Against Competitive Infringement) will apply to the enforcement of such other Party’s Know-How or Patent Rights or the defense of
such invalidity claim (i.e., each Party has the right to enforce its own intellectual property, except that the relevant Commercializing Party will have the initial right, to the extent provided in Section 8.6 (Enforcement
of Patents Against Competitive Infringement), to enforce such Know-How or Patent Rights or defend such invalidity claim, and the other Party will have a step-in right, to the extent provided in Section 8.6 (Enforcement of Patents
Against Competitive Infringement), to enforce such Know-How or Patent Rights or defend such invalidity claim).
|
|
Confidential
|
Execution Version
|
| 8.6. |
Enforcement of Patents Against Competitive Infringement.
|
|
|
8.6.1. |
Duty to Notify of Competitive Infringement. If either Party learns of an infringement,
unauthorized use, misappropriation or threatened infringement by a Third Party to which such Party does not owe any obligation of confidentiality with respect to any Ionis Product-Specific Patents by reason of the development,
manufacture, use or commercialization of a product directed against the RNA that encodes a Target in the Field (“Competitive Infringement”), such Party will promptly notify the
other Party in writing and will provide such other Party with available evidence of such Competitive Infringement; provided, however, that for cases of Competitive Infringement under Section
8.6.6 (35 USC § 271(e)(2) Infringement) below, the notifying Party will provide such written notice within ten (10) Business Days.
|
|
|
8.6.2. |
Enforcement. For any Competitive Infringement with respect to a Product that Roche is Developing
or Commercializing, Roche will have the first right, but not the obligation, to institute, prosecute, and control a Proceeding with respect thereto by counsel of its own choice at its own expense, and Ionis will have the right, at its own
expense, to be represented in that action by counsel of its own choice. If Roche fails to initiate a Proceeding within a period of [***] after receipt of written notice of such Competitive Infringement (subject to a [***] extension to
conclude negotiations, if Roche has commenced good faith negotiations with an alleged infringer for elimination of such Competitive Infringement within such [***] period), then Ionis will have the right to initiate and control a
Proceeding with respect to such Competitive Infringement by counsel of its own choice, and Roche will have the right to be represented in any such action by counsel of its own choice at its own expense. Notwithstanding the foregoing,
Ionis will at all times have the sole right to institute, prosecute, and control any Proceeding under this Section 8.6.2 (Enforcement) to the extent involving any Ionis Core Technology Patents, Ionis Manufacturing and Analytical
Patents, or Ionis Collaboration Patents.
|
|
|
8.6.3. |
Joinder.
|
|
|
(a) |
If a Party initiates a Proceeding in accordance with this Section 8.6 (Enforcement of Patents Against Competitive Infringement), the other Party agrees to be joined as a party plaintiff where
necessary and to give the first Party reasonable assistance and authority to file and prosecute the Proceeding. Subject to Section 8.6.4 (Share of Recoveries), the costs and expenses of each Party incurred pursuant to this Section
8.6.3(a) will be borne by the Party initiating such Proceeding.
|
|
|
(b) |
If one Party initiates a Proceeding in accordance with this Section 8.6 (Enforcement of Patents Against Competitive Infringement), the other Party may join such Proceeding as a party plaintiff where necessary for such other Party
to seek lost profits with respect to such infringement.
|
|
Confidential
|
Execution Version
|
|
|
8.6.4. |
Share of Recoveries. Any damages or other monetary awards recovered with respect to a Proceeding
brought pursuant to this Section 8.6 (Enforcement of Patents Against Competitive Infringement) will be shared as follows:
|
|
|
(a) |
the amount of such recovery will first be applied to the Parties’ reasonable out-of-pocket costs incurred in connection with such Proceeding (which amounts will be allocated pro
rata if insufficient to cover the totality of such expenses); then
|
|
|
(b) |
any remaining proceeds constituting direct or actual damages for acts of infringement occurring prior to Handoff will be (i) [***]; or (ii) [***]; then
|
|
|
(c) |
any remaining proceeds constituting direct or actual damages for acts of infringement occurring after Handoff will be [***], and [***]; then
|
|
|
(d) |
any remaining proceeds constituting punitive or treble damages will be allocated between the Parties as follows: the Party initiating the Proceeding will receive and retain [***] of such proceeds and the
other Party will receive and retain [***] of such proceeds.
|
|
|
8.6.5. |
Settlement. Notwithstanding anything to the contrary under this ARTICLE 8, neither Party
may enter a settlement, consent judgment or other voluntary final disposition of a suit under this ARTICLE 8 that disclaims, limits the scope of, admits the invalidity or unenforceability of, or grants a license, covenant not to
sue or similar immunity under a Patent Right Controlled by the other Party without first obtaining the written consent of the Party that Controls the relevant Patent Right.
|
|
|
8.6.6. |
35 USC § 271(e)(2) Infringement. Notwithstanding anything to the contrary in this Section
8.6 (Enforcement of Patents Against Competitive Infringement), solely with respect to Licensed Patents, for a Competitive Infringement under 35 USC § 271(e)(2), the time period set forth in Section 8.6.2 (Enforcement) during
which a Party will have the initial right to bring a Proceeding will be shortened to a total of twenty-five (25) days, so that, to the extent the other Party has the right, pursuant to such Section to initiate a Proceeding if the first
Party does not initiate a Proceeding, such other Party will have such right if the first Party does not initiate a Proceeding within twenty-five (25) days after such first Party’s receipt of written notice of such Competitive
Infringement.
|
| 8.7. |
Other Infringement.
|
|
|
8.7.1. |
Joint Collaboration Patents. With respect to the infringement of a Joint Collaboration Patent
that is not a Competitive Infringement, the Parties will cooperate in good faith to bring suit together against such infringing party or the Parties may decide to permit one Party to solely bring suit. Any damages or other monetary awards
recovered with respect to a Proceeding brought pursuant to this Section 8.7.1 (Joint Collaboration Patents) will be shared as follows: (a) the amount of such recovery will first be applied to the Parties’ reasonable out- of-pocket
costs incurred in connection with such Proceeding (which amounts will be allocated pro rata if insufficient to cover the totality of such expenses); (b) any remaining proceeds constituting direct
damages will be [***]; and (c) any remaining proceeds constituting punitive or treble damages will be allocated as follows: (i) if the Parties jointly initiate a Proceeding pursuant to this Section 8.7.1 (Joint Collaboration
Patents), [***]; and (ii) if only one Party initiates the Proceeding pursuant to this Section 8.7.1 (Joint Collaboration Patents), such Party will receive [***] of such proceeds and the other Party will receive [***] of such
proceeds.
|
|
Confidential
|
Execution Version
|
|
|
8.7.2. |
Patents Solely Owned by Ionis. Ionis will retain all rights to pursue an infringement of any
Patent Right solely owned by Ionis which is other than a Competitive Infringement and Ionis will retain all recoveries with respect thereto.
|
|
|
8.7.3. |
Patents Solely Owned by Roche. Roche will retain all rights to pursue an infringement of any
Patent Right solely owned by Roche which is other than a Competitive Infringement and Roche will retain all recoveries with respect thereto.
|
| 8.8. |
Patent Listing.
|
|
|
8.8.1. |
Roche’s Obligations. Roche will promptly, accurately and completely list, with the applicable
Regulatory Authorities during the Agreement Term, all applicable Patent Rights that Cover a Product. Prior to such listings, the Parties will meet, through the JPC, to evaluate and identify all applicable Patent Rights, and Roche will
have the right to review, where reasonable, original records relating to any invention for which Patent Rights are being considered by the JPC for any such listing. Notwithstanding the preceding sentence, Roche will retain final
decision-making authority as to the listing of all applicable Patent Rights for a Product that are not Ionis Core Technology Patents, Ionis Collaboration Patents, or Ionis Manufacturing and Analytical Patents, regardless of which Party
owns such Patent Rights.
|
|
|
8.8.2. |
Ionis’ Obligations. Ionis will promptly, accurately and completely list, with the applicable Regulatory Authorities
during the Agreement Term, all applicable Patent Rights that Cover a Discontinued Product. Prior to such listings, the Parties will meet, through the JPC, to evaluate and identify all applicable Patent Rights, and Ionis will have the
right to review, where reasonable, original records relating to any invention for which Patent Rights are being considered by the JPC for any such listing. Notwithstanding the preceding sentence, Ionis will retain final decision-making
authority as to the listing of all applicable Patent Rights for such Discontinued Products, as applicable, regardless of which Party owns such Patent Rights.
|
| 8.9. |
Joint Research Agreement under the Leahy-Smith America Invents Act. If a Party intends to invoke
its rights under 35 U.S.C. § 102(e) of the Leahy-Smith America Invents Act, it will notify the other Party and neither Party will make an election under such provision when exercising its rights under this ARTICLE 8 without the
prior written consent of the other Party (such consent not to be unreasonably withheld, conditioned or delayed), and the Parties will use reasonable efforts to cooperate and coordinate their activities with respect to any submissions,
filings or other activities in support thereof. The Parties acknowledge and agree that this Agreement is a “joint research agreement” as defined in 35 U.S.C. § 100(h).
|
|
Confidential
|
Execution Version
|
| 8.10. |
Obligations to Third Parties. Notwithstanding any of the foregoing, each Party’s rights and
obligations with respect to Licensed Technology under this ARTICLE 8 will be subject to the Third Party rights and obligations (i) for which Roche is responsible under Section 7.4.3(f) (Third Party Payments) and (ii) under
Prior Agreements; provided, however, that, to the extent that Ionis has a non-transferable right to prosecute, maintain or enforce any Patent Rights licensed to Roche hereunder, and this Agreement
purports to grant any such rights to Roche, Ionis will act in such regard with respect to such Patent Rights at Roche’s direction.
|
| 8.11. |
Additional Right and Exceptions. Notwithstanding any provision of this ARTICLE 8,
but subject to Section 8.5.3 (Interplay Between Enforcement of IP and Defense of Third Party Claims), Ionis retains the sole right to Prosecute and Maintain Ionis Core Technology Patents, Ionis Collaboration Patents, and Ionis
Manufacturing and Analytical Patents during the Agreement Term and to control any enforcement of Ionis Core Technology Patents, Ionis Collaboration Patents, and Ionis Manufacturing and Analytical Patents, and will take the lead on such
enforcement solely to the extent that the scope or validity of any Patent Rights Controlled by Ionis and Covering the Ionis Core Technology Patents, Ionis Collaboration Patents, or Ionis Manufacturing and Analytical Patents is at risk. If
Ionis determines, in Ionis’ sole discretion, to not enforce any Ionis Core Technology Patents, Ionis Collaboration Patents, or Ionis Manufacturing and Analytical Patents and does not permit Roche to so enforce such Patent Rights, then the
Parties will mutually agree on an appropriate adjustment (if any) of the future consideration payable by Roche under this Agreement to reflect any adverse impact Ionis’ failure to enforce such Patent Rights has on Products.
|
| 8.12. |
Patent Term Extension. The Parties will cooperate with each other in gaining patent term
extension wherever applicable to a Product, including European supplementary protection certificates and pediatric exclusivity. After Handoff, Roche will determine which patents will be extended and what extensions will be sought.
|
| 9.1. |
Representations and Warranties of Both Parties. Each Party hereby represents and warrants and,
where indicated, covenants, to the other Party, as of the Effective Date, that:
|
|
|
9.1.1. |
such Party is duly organized, validly existing and in good standing under the laws of the jurisdiction of its incorporation or organization and has full corporate power and authority to enter into this
Agreement and to carry out the provisions hereof;
|
|
|
9.1.2. |
such Party has taken all necessary action on its part to authorize the execution and delivery of this Agreement and the performance of its obligations hereunder;
|
|
|
9.1.3. |
this Agreement has been duly executed and delivered on behalf of such Party, and constitutes a legal, valid and binding obligation, enforceable against it in accordance with the terms hereof;
|
|
Confidential
|
Execution Version
|
|
|
9.1.4. |
the execution, delivery and performance of this Agreement by such Party will not constitute a default under or conflict with any agreement, instrument or understanding, oral or written, to which it is a party
or by which it is bound, or to the best of its knowledge and belief violate any law or regulation of any court, governmental body or administrative or other agency having jurisdiction over such Party;
|
|
|
9.1.5. |
to the best of its knowledge and belief, no government authorization, consent, approval, license, exemption of or filing or registration with any court or governmental department, commission, board, bureau,
agency or instrumentality, domestic or foreign, under any applicable laws, rules or regulations currently in effect, is or will be necessary for, or in connection with, the transaction contemplated by this Agreement or any other agreement
or instrument executed in connection herewith, or for the performance by it of its obligations under this Agreement and such other agreements; and
|
|
|
9.1.6. |
it has not employed (and, to the best of its knowledge and belief, has not used a contractor or consultant that has employed) and in the future will not employ (or, to the best of its knowledge, use any
contractor or consultant that employs, provided that such Party may reasonably rely on a representation made by such contractor or consultant) any Person debarred by the FDA (or subject to a
similar sanction of EMA or foreign equivalent), or any Person which is the subject of an FDA debarment investigation or proceeding (or similar proceeding of EMA or foreign equivalent), in the conduct of the Pre-Clinical Studies or Clinical
Studies of a Product and its activities under the R&D Plans.
|
| 9.2. |
Representations and Warranties of Ionis. Ionis hereby represents and warrants to Roche, as of
the Effective Date, and solely with respect to the Oligonucleotide within an Ionis Development Candidate that:
|
|
|
9.2.1. |
To the best of its knowledge and belief, there are no additional licenses (beyond those that would be granted to Roche under Section 5.1.2 (Development and Commercialization License Grant to Roche)
upon the Handoff for a Product arising under the R&D Plans) under any intellectual property owned or Controlled by Ionis or its Affiliates as of the Effective Date that would be required in order for Roche to further Develop and
Commercialize a Product arising under the R&D Plan existing on the Effective Date.
|
|
|
9.2.2. |
The Licensed Technology existing as of the Effective Date constitutes all of the Patent Rights and Know-How Controlled by Ionis as of the Effective Date that are necessary to Develop, Manufacture or
Commercialize Compounds contemplated under the R&D Plan existing on the Effective Date in the Field. Ionis has not previously assigned, transferred, conveyed or otherwise encumbered its right, title and interest in the Licensed
Technology in a manner that conflicts with any rights granted to Roche hereunder.
|
|
|
9.2.3. |
There are no claims, judgments or settlements against or owed by Ionis or its Affiliates or pending against Ionis or, to the best of Ionis’ knowledge, threatened against Ionis, in each case relating to the
Licensed Technology that could impact activities under this Agreement. To the best of Ionis’ knowledge, there are no claims, judgments or settlements against or owed by any Third Party that is party to a Prior Agreement, or pending or
threatened claims or litigation against any Third Party that is party to a Prior Agreement, in each case relating to the Licensed Technology that would impact activities under this Agreement.
|
|
Confidential
|
Execution Version
|
|
|
9.2.4. |
SCHEDULE 9.2.4(a), SCHEDULE 9.2.4(b) and SCHEDULE 9.2.4(c) set forth true, correct and
complete lists of all Ionis Core Technology Patents, Ionis Manufacturing and Analytical Patents, and Ionis Product-Specific Patents that, as of the Effective Date, are expected to apply to the Compounds contemplated under the R&D Plan
as of the Effective Date, respectively, and indicates whether each such Patent Right is owned by Ionis or licensed by Ionis from a Third Party and if so, identifies the licensor or sublicensor from which the Patent Right is licensed.
Ionis Controls such Patent Rights existing as of the Effective Date and is entitled to grant all rights and licenses (or sublicenses, as the case may be) under such Patent Rights it purports to grant to Roche under this Agreement.
|
|
|
9.2.5. |
(a) There is no fact or circumstance known by Ionis that would cause Ionis to reasonably conclude that any Licensed Patent is invalid or un-enforceable, (b) there is no fact or circumstance known by Ionis
that would cause Ionis to reasonably conclude the inventorship of each Licensed Patent is not properly identified on each patent, (c) all official fees, maintenance fees and annuities for the Licensed Patents have been paid and all
administrative procedures with governmental agencies have been completed, (d) none of the Ionis Product- Specific Patents that would be licensed by Ionis to Roche upon Handoff under this Agreement are currently involved in any interference,
reissue, re- examination, cancellation or opposition proceeding and neither Ionis, nor any of its Affiliates, has received any written notice from any person, or has knowledge, of such actual or threatened Proceeding, and (e) to the best of
Ionis’ knowledge and belief, Roche’s practice of the inventions claimed in the Ionis Product-Specific Patents in the performance of the Roche R&D Activities contemplated as of the Effective Date will not [***].
|
|
|
9.2.6. |
SCHEDULE 9.2.6 is a complete and accurate list of all agreements that create Third Party Obligations that affect the rights granted by
Ionis to Roche under this Agreement with respect to Ionis Development Candidates contemplated by the R&D Plans on the Effective Date.
|
|
|
9.2.7. |
Ionis has all rights necessary to grant the Handoff and licenses contained in this Agreement, and has the ability to work exclusively with Roche as set forth in this Agreement, including the covenants granted
in Section 4.1 (Exclusivity).
|
| 9.3. |
Ionis Covenants. Ionis hereby covenants to Roche that, except as expressly permitted under this
Agreement:
|
|
|
9.3.1. |
Ionis will promptly amend SCHEDULE 9.2.4(a), SCHEDULE 9.2.4(b) and SCHEDULE 9.2.4(c) and submit such amended Schedules to Roche if Ionis
becomes aware that any Ionis Core Technology Patents, Ionis Manufacturing and Analytical Patents or Ionis Product-Specific Patents are not properly identified on the respective Schedules;
|
|
Confidential
|
Execution Version
|
|
|
9.3.2. |
during the Agreement Term, Ionis will maintain and not breach any agreements with Third Parties entered into after the Effective Date (“New Third Party
Licenses”) that provide a grant of rights from such Third Party to Ionis that are Controlled by Ionis and are licensed or may become subject to a license from Ionis to Roche for a Product under this Agreement;
|
|
|
9.3.3. |
Ionis will promptly notify Roche of any material breach by Ionis or a Third Party of any New Third Party License, and in the event of a breach by Ionis, will permit Roche to cure such breach on Ionis’ behalf
upon Roche’s request;
|
|
|
9.3.4. |
Ionis will not amend, modify or terminate any New Third Party License in a manner that would adversely affect Roche’s rights hereunder without first obtaining Roche’s written consent, which [***];
|
|
|
9.3.5. |
Ionis will not enter into any new agreement or other obligation with any Third Party, or amend an existing agreement with a Third Party, in each case that restricts, limits or encumbers the rights granted to
Roche under this Agreement;
|
|
|
9.3.6. |
Ionis will cause its Affiliates to comply with the terms of Section 4.1 (Exclusivity);
|
|
|
9.3.7. |
All employees and contractors of Ionis performing research and Development activities hereunder on behalf of Ionis will be obligated to assign all right, title and interest in and to any inventions (or grant
a license to Ionis prior to or upon Handoff to obtain such a license) developed by them, whether or not patentable, to Ionis or such Affiliate, respectively, as the sole owner thereof; and
|
|
|
9.3.8. |
If, after the Effective Date, Ionis becomes the owner or otherwise acquires Control of any formulation or delivery devices that would be necessary or useful in order for Roche to further Develop, Manufacture
or Commercialize a Product, and Roche has executed Handoff and the license granted to Roche under this Agreement is in effect, Ionis will make such technology available to Roche on commercially reasonable terms.
|
| 9.4. |
Representations and Warranties of Roche. Roche hereby represents and warrants to Ionis, as of
the Effective Date, that all employees and contractors of Roche performing research and Development activities hereunder on behalf of Roche will be obligated to assign all right, title and interest in and to any inventions (or grant a
license to Roche prior to or upon Handoff to obtain such a license) developed by them, whether or not patentable, to Roche or such Affiliate, respectively, as the sole owner thereof.
|
| 9.5. |
DISCLAIMER. EXCEPT AS OTHERWISE EXPRESSLY SET FORTH IN THIS AGREEMENT, NEITHER PARTY NOR ITS AFFILIATES MAKES ANY REPRESENTATION OR
EXTENDS ANY WARRANTY OF ANY KIND, EITHER EXPRESS OR IMPLIED, INCLUDING ANY WARRANTY OF MERCHANTABILITY OR FITNESS FOR A PARTICULAR PURPOSE. ROCHE AND IONIS UNDERSTAND THAT PRODUCTS ARE THE SUBJECT OF ONGOING RESEARCH AND DEVELOPMENT AND
THAT NEITHER PARTY CAN ASSURE THE SAFETY, USEFULNESS OR COMMERCIAL OR TECHNICAL VIABILITY OF THE PRODUCTS.
|
|
Confidential
|
Execution Version
|
| 10.1. |
Indemnification by Roche. Roche will indemnify, defend and hold harmless Ionis and its
Affiliates, and its or their respective directors, officers, employees and agents, from and against any and all liabilities, damages, losses, costs and expenses including the reasonable fees of attorneys (collectively “Losses”) arising out of or resulting from any and all Third Party suits, claims, actions, proceedings or demands (“Claims”)
based upon:
|
|
|
10.1.1. |
the gross negligence or willful misconduct of Roche, its Affiliates or Sublicensees and its or their respective directors, officers, employees and agents, in connection with Roche’s performance of its
obligations or exercise of its rights under this Agreement;
|
|
|
10.1.2. |
any breach of any representation or warranty or express covenant made by Roche under ARTICLE 9 or any other provision under this Agreement;
|
|
|
10.1.3. |
the Development or Manufacturing activities that are conducted by or on behalf of Roche or its Affiliates or Sublicensees (which will exclude any Development or Manufacturing activities that are conducted by
or on behalf of Ionis pursuant to this Agreement); or
|
|
|
10.1.4. |
the Commercialization of a Product by or on behalf of Roche or its Affiliates or Sublicensees;
|
| 10.2. |
Indemnification by Ionis. Ionis will indemnify, defend and hold harmless Roche and its
Affiliates, and its or their respective directors, officers, employees and agents, from and against any and all Losses arising out of or resulting from any and all Claims based upon:
|
|
|
10.2.1. |
the gross negligence or willful misconduct of Ionis, its Affiliates or Sublicensees or its or their respective directors, officers, employees and agents, in connection with Ionis’ performance of its
obligations or exercise of its rights under this Agreement;
|
|
|
10.2.2. |
any breach of any representation or warranty or express covenant made by Ionis under ARTICLE 9 or any other provision under this Agreement;
|
|
|
10.2.3. |
any Development or Manufacturing activities that are conducted by or on behalf of Ionis or its Affiliates or Sublicensees (which will exclude any Development or Manufacturing activities that are conducted by
or on behalf of Roche pursuant to this Agreement); or
|
|
|
10.2.4. |
any Development, Manufacturing or Commercialization activities that are conducted by or on behalf of Ionis or its Affiliates or Sublicensees with respect to a Discontinued Product;
|
|
Confidential
|
Execution Version
|
| 10.3. |
Procedure. If a Person entitled to indemnification under Section 10.1 (Indemnification
by Roche) or Section 10.2 (Indemnification by Ionis) (an “Indemnitee”) seeks such indemnification, such Indemnitee will (i) inform the indemnifying Party in writing of a
Claim as soon as reasonably practicable after such Indemnitee receives notice of such Claim, (ii) permit the indemnifying Party to assume direction and control of the defense of the Claim (including the sole right to settle such Claim at
the sole discretion of the indemnifying Party, provided that such settlement or compromise does not admit any fault or negligence on the part of the Indemnitee, or impose any obligation on, or
otherwise materially adversely affect, the Indemnitee or other Party), (iii) cooperate as reasonably requested (at the expense of the indemnifying Party) in the defense of the Claim, and (iv) undertake reasonable steps to mitigate any
Losses with respect to the Claim. The provisions of Section 8.5 (Defense of Claims Brought by Third Parties) will govern the procedures for responding to a Claim of infringement described therein. Notwithstanding anything in this
Agreement to the contrary, the indemnifying Party will have no liability under Section 10.1 (Indemnification by Roche) or Section 10.2 (Indemnification by Ionis), as the case may be, for Claims settled or compromised by
the Indemnitee without the indemnifying Party’s prior written consent.
|
| 10.4. |
Insurance.
|
|
|
10.4.1. |
Ionis’ Insurance Obligations. Ionis hereby represents and warrants to Roche that it will
maintain, at its cost, reasonable insurance against liability and other risks associated with its activities contemplated by this Agreement, including but not limited to its indemnification obligations herein, in such amounts and on such
terms as are customary for prudent practices for biotech companies of similar size and with similar resources in the pharmaceutical industry for the activities to be conducted by it under this Agreement taking into account the scope of
Development of Products. Ionis will furnish to Roche evidence of any insurance required under this Section 10.4.1 (Ionis’ Insurance Obligations), upon request.
|
|
|
10.4.2. |
Roche’s Insurance Obligations. Roche hereby represents and warrants to Ionis that it will
maintain, at its cost, reasonable insurance – which may be through self-insurance – against liability and other risks associated with its activities contemplated by this Agreement (including product liability), including but not limited
to its indemnification obligations herein, in such amounts and on such terms as are customary for prudent practices for large companies in the pharmaceutical industry for the activities to be conducted by Roche under this Agreement. Roche
will maintain such insurance throughout the Agreement Term and for [***] thereafter, and will furnish to Ionis evidence of such insurance, upon request.
|
|
Confidential
|
Execution Version
|
|
|
10.5. |
LIMITATION OF CONSEQUENTIAL DAMAGES. EXCEPT FOR (a) CLAIMS OF
A THIRD PARTY THAT ARE SUBJECT TO INDEMNIFICATION UNDER THIS ARTICLE 10 (INDEMNIFICATION; INSURANCE), (b) CLAIMS ARISING OUT OF A PARTY’S WILLFUL MISCONDUCT OF THIS AGREEMENT, (c) A
PARTY’S BREACH OF ARTICLE 4 (EXCLUSIVITY COVENANTS), OR A BREACH OF SECTION 11.4.2 (LICENSE TERMINATION) BY ROCHE OR ITS AFFILIATES OR (d) CLAIMS ARISING OUT OF A PARTY’S BREACH OF ITS CONFIDENTIALITY OBLIGATIONS UNDER
THIS AGREEMENT, NEITHER PARTY NOR ANY OF ITS AFFILIATES WILL BE LIABLE TO THE OTHER PARTY TO THIS AGREEMENT OR ITS AFFILIATES FOR ANY INCIDENTAL, CONSEQUENTIAL, SPECIAL, PUNITIVE OR OTHER INDIRECT DAMAGES, WHETHER LIABILITY IS ASSERTED
IN CONTRACT, TORT (INCLUDING NEGLIGENCE AND STRICT PRODUCT LIABILITY), INDEMNITY OR CONTRIBUTION, AND IRRESPECTIVE OF WHETHER THAT PARTY OR ANY REPRESENTATIVE OF THAT PARTY HAS BEEN ADVISED OF, OR OTHERWISE MIGHT HAVE ANTICIPATED THE
POSSIBILITY OF, ANY SUCH LOSS OR DAMAGE.
|
| 11.1. |
Agreement Term; Expiration. This Agreement is effective as of the Effective Date and, unless earlier terminated
pursuant to the other provisions of this ARTICLE 11, will continue in full force and effect until this Agreement expires as follows:
|
|
|
11.1.1. |
on a country-by-country and Product-by-Product basis, on the date of expiration of all payment obligations by the Commercializing Party under this Agreement with respect to such Product (or such Discontinued
Product) in such country; and
|
|
|
11.1.2. |
in its entirety upon the expiration of all payment obligations under this Agreement with respect to the last Product (or last Discontinued Product) in all countries pursuant to Section 11.1.1.
|
| 11.2. |
Termination.
|
|
|
11.2.1. |
Roche’s Termination for Convenience. Roche may terminate this Agreement for convenience as a
whole or on a Program-by-Program basis, Target-by- Target basis, Product-by-Product basis or country-by-country basis by providing ninety (90) days written notice to Ionis of such termination.
|
|
Confidential
|
Execution Version
|
|
|
11.2.2. |
Termination for Material Breach.
|
|
|
(a) |
Roche’s Right to Terminate. If Roche believes that Ionis is in material breach of this Agreement
(other than with respect to a failure to use Commercially Reasonable Efforts under Section 3.1.2 (Conducting the R&D Plans) or Section 3.1.9 (Performance Milestones During the Handoff Period), which are governed by Section
11.2.3 (Remedies for Failure to Use Commercially Reasonable Efforts) below), then Roche may deliver written notice of such material breach to Ionis. If the breach is curable, Ionis will have [***] to cure such breach. If Ionis fails
to cure such breach within the [***] period, or if the breach is not subject to cure, Roche may terminate this Agreement in whole or on a Program-by-Program basis, Target-by-Target basis, Product-by- Product basis or country-by-country
basis, as applicable, by providing written notice to Ionis.
|
|
|
(b) |
Ionis’ Right to Terminate. If Ionis believes that Roche is in material breach of this Agreement
(other than with respect to a failure to use Commercially Reasonable Efforts under Section 3.1.2 (Conducting the R&D Plans), Section 3.1.9 (Performance Milestones During the Handoff Period), or Section 6.2
(Diligence), which is governed by Section 11.2.3 (Remedies for Failure to Use Commercially Reasonable Efforts) below), then Ionis may deliver written notice of such material breach to Roche. If the breach is curable, Roche will
have [***] days to cure such breach (except to the extent such breach involves the failure to make a payment when due, which breach must be cured within [***] days following such notice). If Roche fails to cure such breach within the
[***] or [***] period, as applicable, or if the breach is not subject to cure, Ionis may terminate this Agreement by providing written notice to Roche in whole or on a Program-by-Program basis, Target-by-Target basis, Product-by-Product
basis or country-by- country basis, as applicable, by proving written notice to Roche.
|
|
|
11.2.3. |
Remedies for Failure to Use Commercially Reasonable Efforts.
|
|
|
(a) |
If Ionis, in Roche’s reasonable determination, fails to use Commercially Reasonable Efforts in the activities contemplated in Section 3.1.2 (Conducting the R&D Plans) or Section 3.1.9
(Performance Milestones During the Handoff Period), then Roche will notify Ionis and, within [***] thereafter, Ionis and Roche will meet and confer to discuss and resolve the matter in good faith, and attempt to devise a mutually agreeable
plan to address any outstanding issues related to Ionis’ use of Commercially Reasonable Efforts in Section 3.1.2 (Conducting the R&D Plans) or Section 3.1.9 (Performance Milestones During the Handoff Period), as
applicable. Following such a meeting, if Ionis fails to use Commercially Reasonable Efforts as agreed, Roche will have the right to terminate this Agreement in whole or on a Program-by-Program basis, Target-by-Target basis,
Product-by-Product basis or country-by-country basis, as applicable.
|
|
Confidential
|
Execution Version
|
|
|
(b) |
If Roche, in Ionis’ reasonable determination, fails to use Commercially Reasonable Efforts in the activities contemplated in Section 3.1.2 (Conducting the R&D Plans), Section 3.1.9
(Performance Milestones During the Handoff Period), or Section 6.2 (Diligence), then Ionis will notify Roche and, within [***] thereafter, [***]. Following [***], if Roche fails to use Commercially Reasonable Efforts as agreed and
subject to Section 11.2.4 (Disputes Regarding Material Breach), Ionis will have the right to terminate this Agreement in whole or on a Program-by-Program basis, Target-by-Target basis, Product-by-Product basis or country-by-country
basis, as applicable.
|
|
|
11.2.4. |
Disputes Regarding Material Breach. Notwithstanding the foregoing, if the Breaching Party in Section
11.2.2(a) (Roche’s Right to Terminate) or Section 11.2.2(b) (Ionis’ Right to Terminate) disputes in good faith the existence or materiality of, or failure to cure any such breach which is not a payment breach, and
provides notice to the Non-Breaching Party of such dispute within such [***] or [***] period, as applicable, then the Non-Breaching Party will not have the right to terminate this Agreement in accordance with Section 11.2.2(a)
(Roche’s Right to Terminate) or Section 11.2.2(b) (Ionis’ Right to Terminate), unless and until it has been determined in accordance with Section 13.1 (Dispute Resolution) that this Agreement was materially breached by the
Breaching Party and the Breaching Party fails to cure such breach within [***] days following such determination. It is understood and acknowledged that during the pendency of such dispute, all the terms of this Agreement will remain in
effect and the Parties will continue to perform all of their respective obligations hereunder, including satisfying any payment obligations.
|
|
|
11.2.5. |
Termination for Insolvency.
|
|
|
(a) |
Either Party may terminate this Agreement if, at any time, the other Party files in any court or agency pursuant to any statute or regulation of any state or country a petition in bankruptcy or insolvency or
for reorganization or for an arrangement or for the appointment of a receiver or trustee of the Party or of substantially all of its assets; or if the other Party proposes a written agreement of composition or extension of substantially all
of its debts; or if the other Party is served with an involuntary petition against it, filed in any insolvency proceeding, and such petition is not dismissed within ninety (90) days after the filing thereof; or if the other Party is a party
to any dissolution or liquidation; or if the other Party makes an assignment of substantially all of its assets for the benefit of creditors.
|
|
|
(b) |
All rights and licenses granted under or pursuant to any section of this Agreement are and will otherwise be deemed to be for purposes of Section 365(n) of Title 11, United States Code (the “Bankruptcy Code”) licenses of rights to “intellectual property” as defined in Section 101(35A) of the Bankruptcy Code. The Parties will retain and may fully exercise all of their
respective rights and elections under the Bankruptcy Code. Upon the bankruptcy of any Party, the non-bankrupt Party will further be entitled to a complete duplicate of, or complete access to, any such intellectual property, and such, if not
already in its possession, will be promptly delivered to the non-bankrupt Party, unless the bankrupt Party elects in writing to continue, and continues, to perform all of its obligations under this Agreement.
|
|
Confidential
|
Execution Version
|
|
|
11.2.6. |
Termination if Ionis Development Candidate Not Identified or Designated.
|
|
|
(a) |
On a Target-by-Target basis, if, [***], then either Party will have the right to terminate the Agreement with regard to the applicable Program by providing [***] days’ written notice to the other Party.
|
|
|
(b) |
If the JSC does not recommend a Compound for designation as an Ionis Development Candidate (and
does not, as an alternative, recommend designation of a Related Compound) within [***] after Roche’s receipt of the applicable Development Candidate Data Package, or Roche does not confirm the designation of a Compound (or Related
Compound, as applicable) as an Ionis Development Candidate within [***] after the date of the JSC’s recommendation (or a longer period as the Parties may mutually agree in writing), in each case in accordance with Section 3.1.3
(Ionis Development Candidate Designation; IND- Enabling Toxicology Study Costs), then Ionis will have the right to terminate the Agreement with regard to the applicable Program by providing [***] days’ written notice to Roche.
|
|
|
(c) |
If this Agreement is terminated under this Section 11.2.6(a) (Termination if Ionis Development Candidate Not Identified or Designated) with respect to a Program, then Section 11.4 (General Consequences of Termination of
the Agreement) will apply solely with respect to the applicable Program. Nothing in this Section 11.2.6 (Termination if Ionis Development Candidate Not Identified or Designated) will terminate or otherwise affect the provisions of
another Program, which shall remain in full force and effect.
|
|
|
(d) |
If this Agreement is terminated under this Section 11.2.6 (Termination if Ionis Development Candidate Not Identified or Designated) with respect to a Program, then [***].
|
|
|
11.2.7. |
Termination for Cessation of Development. If, (a) prior
to [***], Roche has suspended Development activities for a period of [***] but (i) has not [***], or (ii) has not [***], or (b) Section 3.1.7 (Failure of IND-Enabling Toxicology Study to Achieve Handoff Data Package Criteria; Disputes over Achievement of Handoff Data Package Criteria) becomes applicable under this
Agreement, within the [***] period no Related Compound has been designated as a replacement Ionis Development Candidate, and Roche is not otherwise [***], then in each case ((a) or (b))
Ionis shall have the right to terminate this Agreement in accordance with Section 11.2.2(b) (Ionis’ Right to Terminate). Ionis’ rights under this Section 11.2.7 (Termination for Cessation of Development) are without prejudice to Roche’s obligation to use Commercially Reasonable Efforts as set out in Section 6.2 (Diligence).
|
|
Confidential
|
Execution Version
|
| 11.3. |
Patent Challenge. If, during the Agreement Term, solely with respect to rights to the Ionis
Product-Specific Patents and the Ionis Core Technology Patents claiming technology incorporated in a Product, in each case that are included in a license granted to Roche under Section 5.1.2 (Development and Commercialization
License Grant to Roche), Roche, its Affiliates or Sublicensees, in any country, (a) commence or otherwise voluntarily determine to participate in (other than as may be necessary or reasonably required to [***]) any action or proceeding,
challenging or denying the enforceability or validity of any claim within an issued patent or patent application within such Ionis Product-Specific Patents or Ionis Core Technology Patents, or (b) direct, support or actively assist any
other Person (other than as may be necessary or reasonably required to [***]) in bringing or prosecuting any action or proceeding challenging or denying the validity of any claim within an issued patent or patent application within such
Ionis Product-Specific Patents or Ionis Core Technology Patents, then unless, within [***] after written notice from Ionis, Roche [***], Ionis may, to the extent permitted under Applicable Law, terminate this Agreement and the provisions
of Section 11.4 (General Consequences of Termination of the Agreement) and Section 11.5 (Special Consequences of Termination of the Agreement) will apply, provided, however, [***].
|
| 11.4. |
General Consequences of Termination of the Agreement. If any
portion of this Agreement or the whole Agreement is terminated by a Party in accordance with this ARTICLE 11 at any time and for any reason, the following terms will apply to any such termination, either in whole or on a
Program-by-Program basis, Target-by-Target basis, Product-by-Product basis or country-by-country basis:
|
|
|
11.4.1. |
Return of Information and Materials. The Parties will return (or destroy, as directed by the
other Party) all data, files, records and other materials containing or comprising the other Party’s Confidential Information that are the subject of such termination to the extent separable from data, files, records and other materials
(a) required for the continuation of the Program(s), Target, Product(s) or countries which have not been terminated or (b) that constitute the Confidential Information of the returning Party. Notwithstanding the foregoing, the Parties
will be permitted to retain one copy of such data, files, records, and other materials for archival and legal compliance purposes.
|
|
|
11.4.2. |
License Termination. Upon termination but not expiration of the Agreement, except for the
licenses granted under Section 5.1.3 (Cross-Licenses under Collaboration Intellectual Property) and Section 11.2.6(d), any licenses granted by Ionis to Roche under this Agreement will terminate and Roche, its Affiliates
and Sublicensees will cease selling all Products that are the subject of such termination, unless Ionis elects to have Roche continue to sell the applicable Product(s) as part of the Transition Activities under Section 11.5.9
(Transition Activities).
|
|
|
11.4.3. |
Exclusivity Covenants. On a Target-by-Target basis, neither Party will have any further
obligations under Section 4.1 (Exclusivity) of this Agreement.
|
|
|
11.4.4. |
Accrued Rights. Termination or expiration of this Agreement for any reason will be without
prejudice to any rights or financial compensation that will have accrued to the benefit of a Party prior to such termination or expiration. Such termination or expiration will not relieve a Party from obligations that are expressly
indicated to survive the termination or expiration of this Agreement. For purposes of clarification, milestone payments under ARTICLE 7 accrue as of the date the applicable Milestone Event is achieved even if the payment is not
due at that time.
|
|
Confidential
|
Execution Version
|
|
|
11.4.5. |
Survival. The following provisions of this Agreement will survive the expiration or termination
of this Agreement: Section 3.10 (Failure to Identify or Designate an Ionis Development Candidate) (but only if this Agreement is terminated under Section 11.2.6 (Termination if Ionis Development Candidate Not
Identified or Designated)), Section 5.1.3 (Cross-Licenses under Collaboration Intellectual Property), Section 5.1.5 (No Implied Licenses), Section 7.2 ([***]) through Section 7.6 (Mode of Payment) (in each
case solely with respect to any financial compensation that has accrued prior to expiry or termination), Section 7.7 (Records Retention), Section 7.8 (Audits), Section 7.9 (Taxes), Section 7.10 (Interest),
Section 8.1 (Ownership), Section 8.5.2 (Third Party Claims Regarding Discontinued Products), Section 8.5.3 (Interplay Between Enforcement of IP and Defense of Third Party Claims), Section 8.6.5 (Settlement),
Section 9.5 (Disclaimer), ARTICLE 10 (Indemnification; Insurance), Section 11.1 (Agreement Term; Expiration), Section 11.2.5 (Termination for Insolvency), Section 11.2.6 (Termination if Ionis
Development Candidate Not Identified or Designated), Section 11.4 (General Consequences of Termination of the Agreement), Section 11.5 (Special Consequences of Termination of the Agreement), Section 11.6 (Reverse
Royalty Payments to Roche for Discontinued Products), Section 11.7 (Sublicensees), ARTICLE 12 (Confidentiality), ARTICLE 13 (Miscellaneous) and
|
|
|
11.4.6. |
APPENDIX 1 (Definitions) (to the extent definitions are embodied
in the foregoing listed Articles and Sections).
|
| 11.5. |
Special Consequences of Termination of the Agreement. If Roche terminates the Agreement under Section
11.2.1 (Roche’s Termination for Convenience), or Ionis terminates this Agreement under Section 11.2.2(b) (Ionis’ Right to Terminate), Section 11.2.3 (Remedies for Failure to Use Commercially Reasonable
Efforts), or Section 11.2.7 (Termination for Cessation of Development), then in addition to the terms set forth in Section 11.4 (General Consequences of Termination of the Agreement), the following additional terms
will also apply:
|
|
|
11.5.1. |
License to Ionis for Discontinued Products Comprising Ionis Development Candidates. Roche
will and hereby does grant to Ionis a sublicensable, royalty- bearing, worldwide, exclusive license or sublicense, as the case may be, under all Roche Technology (excluding Companion Diagnostic IP) Controlled by Roche as of the date of
such reversion that Covers Discontinued Products comprising an Ionis Development Candidate solely as necessary to Develop, make, have made, use, sell, offer for sale, have sold, import and otherwise Commercialize such Discontinued
Products in the Field;
|
|
|
11.5.2. |
License to Ionis for Companion Diagnostic Products. On request by Ionis, Roche will make
available to Ionis, on commercially reasonable terms, any diagnostic products and/or services to select patients who will use Products (each, a “Companion Diagnostic Product”)
and any Patent Rights and Know- How Covering such Companion Diagnostic Products (such intellectual property, “Companion Diagnostic IP”) Controlled by Roche as of the date of such
reversion that is necessary to Develop or Commercialize such Companion Diagnostic Products;
|
|
Confidential
|
Execution Version
|
|
|
11.5.3. |
Know-How Transfer. Within [***] following the date of termination, Roche will transfer to Ionis
for use with respect to the Development and Commercialization of Discontinued Products, copies of any Roche Collaboration Know-How data, results, and regulatory information, and to the extent solely related to and necessary for the
Development or Commercialization of such Discontinued Products, [***] in the possession of Roche as of the date of such reversion that relate to such Discontinued Products, and any other information or material specified in Section
3.5 (Technology Transfer Implementation);
|
|
|
11.5.4. |
Regulatory Materials. Within [***] following the date of the termination, Roche will assign, and
hereby does assign, to Ionis all of Roche’s right, title and interest in and to all regulatory materials for the Discontinued Product, including any IND, orphan drug designation and marketing authorizations that relate to the applicable
Discontinued Product;
|
|
|
11.5.5. |
Trademarks. Roche will license to Ionis any trademarks that are specific to Discontinued
Products solely for use with such Discontinued Products; provided, however, that in no event will Roche have any obligation to license to Ionis any trademarks used by Roche both in connection with
a Product and in connection with the sale of any other product or service, including any Roche- or Roche-formative marks, company logos, or trademarks of its Affiliates or Sublicensees;
|
|
|
11.5.6. |
Prosecution and Maintenance of Patents for Discontinued Products. Ionis will control and be
responsible at its sole cost for all aspects of the Prosecution and Maintenance of all Joint Collaboration Patents for Discontinued Products that claim Ionis Development Candidates and Ionis Product-Specific Patents for Discontinued
Products, and Roche will provide Ionis with (and will instruct its counsel to provide Ionis with) all of the information and records in Roche’s and its counsel’s possession related to the Prosecution and Maintenance of such Joint
Collaboration Patents for Discontinued Products that claim Ionis Development Candidates and Ionis Product-Specific Patents for Discontinued Products.
|
|
|
11.5.7. |
Stocks of API and Finished Drug Product. Ionis will have the right to purchase from Roche any or
all of the inventory of API and/or Finished Drug Product for such Discontinued Product held by Roche as of the effective date of termination (that are not committed to be supplied to any Third Party or Sublicensee, in the ordinary course
of business, as of the effective date of termination), if any, [***]. Ionis will notify Roche within [***] after the effective date of termination whether Ionis elects to exercise such right;
|
|
Confidential
|
Execution Version
|
|
|
11.5.8. |
Manufacturing Technology Transfer. If Roche or Roche’s CMO is manufacturing API and/or Finished
Drug Product as of the termination triggering this provision, then Ionis may request Roche to conduct (or cause to be conducted by Roche’s CMO) a technology transfer to Ionis (or Ionis’ designated Third Party supplier) of any technology,
information and data reasonably related to Roche’s or such CMO’s manufacturing and supply of API and/or Finished Drug Product for such Discontinued Product, and if so requested, Roche will conduct (or cause to be conducted by Roche’s CMO)
such a technology transfer, and Ionis [***], and Roche will (or will cause Roche’s CMO to) continue to (i) provide reasonable support and cooperation with Ionis’ regulatory filings and interactions with Regulatory Authorities related to
Roche’s or such CMO’s API and/or Finished Drug Product manufacturing (including any required inspections), and (ii) supply (or cause to be supplied by Roche’s CMO) API and/or Finished Drug Product to Ionis, [***] to enable Ionis to
identify and contract with a suitable Third Party API and/or Finished Drug Product manufacturer; and
|
|
|
11.5.9. |
Transition Activities. For a period of up to [***] following the effective date of termination:
|
|
|
(a) |
The Parties wish to provide a mechanism to ensure that, assuming the Discontinued Product is available to patients as of the reversion date, patients who were being treated with the Discontinued Product prior
to such termination or who desire access to the Discontinued Product can continue to have access to such Discontinued Product while the regulatory and commercial responsibilities for the Discontinued Product are transitioned from Roche to
Ionis. As such, Ionis may request Roche to perform transition activities that are necessary or useful to (1) transition Roche’s Commercialization activities (if any) to Ionis to minimize disruption to sales, (2) provide patients with
continued access to the applicable Discontinued Products (if applicable), (3) enable Ionis (or Ionis’ designee) to assume and execute the responsibilities under all Approvals and ongoing Clinical Studies for the applicable Discontinued
Product, and (4) ensure long-term continuity of supply for the Discontinued Product (collectively, the “Transition Activities”), including, if applicable, the categories of
services and deliverables listed on SCHEDULE 11.5.9, but no longer than [***] following the effective date of termination. If applicable, Roche will [***] SCHEDULE 11.5.9; provided Roche and Ionis may mutually agree in writing to [***].
|
|
|
(b) |
Ionis may elect to have Roche perform the applicable Transition Activities by providing written notice to Roche no later than [***] following the effective date of the termination. If Ionis requests
Transition Activities, without limiting the provisions of Section 11.5 (Special Consequences of Termination of the Agreement), the Parties will mutually agree upon a transition plan for Roche to perform the applicable Transition
Activities including delivery and transition dates. In addition, the Parties will establish a transition committee consisting of at least each Party’s Alliance Managers, a representative from each Party’s chemistry, manufacturing and
controls (“CMC”) group who was responsible for the Discontinued Product prior to the termination, and up to two (2) additional representatives from each Party who are from other
relevant functional groups to facilitate a smooth transition. While Roche is providing applicable Transition Activities, Roche and Ionis will [***].
|
|
|
(c) |
Ionis will [***] to perform the Transition Activities. In addition, Ionis will [***] to perform the Transition Activities. Ionis will own [***].
|
|
Confidential
|
Execution Version
|
| 11.6. |
Reverse Royalty Payments to Roche for Discontinued Products.
|
|
|
11.6.1. |
If either party terminates the Agreement with respect to a Product after payment of the applicable milestone amount for the “[***]” Milestone Event set forth in TABLE 1 of Section
7.3.1 (Development Milestones for [***]) or TABLE 2 of Section 7.3.2 (Development Milestones for [***]) but before the payment of the applicable milestone amount for the “[***]” Milestone Event set forth
in TABLE 1 of Section 7.3.1 (Development Milestones for [***]) or TABLE 2 of Section 7.3.2 (Development Milestones for [***]) with respect to the Discontinued Product that is the
subject of such termination, and Ionis or any of its Affiliates or Sublicensees Commercializes such Discontinued Product, then Ionis will pay Roche a reversion royalty of [***] of Annual worldwide Net Sales of such Discontinued Product
[***].
|
|
|
11.6.2. |
If either Party terminates this Agreement with respect to a Product after payment of the applicable milestone amount for the “[***]” Milestone Event set forth in TABLE 1 of Section
7.3.1 (Development Milestones for [***]) or TABLE 2 of Section 7.3.2 (Development Milestones for [***]) but before the payment of the first applicable milestone amount for the “[***]” Milestone Event set
forth in TABLE 1 of Section 7.3.1 (Development Milestones for [***]) or TABLE 2 of Section 7.3.2 (Development Milestones for [***]) with respect to the Product that is the
subject of such termination, and Ionis or any of its Affiliates or Sublicensees Commercializes such Discontinued Product, then Ionis will pay Roche a reversion royalty of [***] of Annual worldwide Net Sales of such Discontinued Product
[***].
|
|
|
11.6.3. |
If either Party terminates this Agreement with respect to a Product after payment of the first applicable milestone amount for the “[***]” Milestone Event set forth in TABLE 1 of Section
7.3.1 (Development Milestones for [***]) or TABLE 2 of Section 7.3.2 (Development Milestones for [***]) with respect to the Discontinued Product that is the subject of such termination, and Ionis or any
of its Affiliates or Sublicensees Commercializes such Discontinued Product, then Ionis will pay Roche a reversion royalty of [***] of Annual worldwide Net Sales of such Discontinued Product [***].
|
|
|
11.6.4. |
Ionis will pay Roche reversion royalties under this Section 11.6 (Reverse Royalty Payments to Roche for Discontinued Products) in accordance with the provisions governing payment of royalties from
Roche to Ionis in Section 7.4 (Royalties) (mutatis mutandis).
|
|
|
11.6.5. |
Limitations on Grant-Backs.
|
|
|
(a) |
All transfers and licenses from Roche to Ionis (or other obligations of Roche) under Section 11.6 (Reverse Royalty Payments to Roche for Discontinued Products) are solely with respect to Product(s)
[***]. Should at the effective date of termination (i) [***], or (ii) [***], then [***]. For clarity, Roche [***].
|
|
Confidential
|
Execution Version
|
|
|
(b) |
In connection with research studies, Clinical Studies or other activities associated with the Development and Commercialization of Products, Roche may have collected (i) personally identifiable information
about individual human subjects or (ii) human biological samples (collectively, “PII/Samples”). Legal and contractual restrictions may apply to such PII/Samples. Roche shall
have no obligation to transfer such PII/Samples unless necessary for the continued development of the Product, in which case Roche shall not be obliged to transfer any PII/Samples that Roche in good faith believes would be prohibited or
would subject Roche to potential liability by reason of Applicable Law, contractual restrictions or insufficient patient consent. If Roche transfers any such PII/Samples, the Parties will enter into the relevant agreements under applicable
data privacy laws (such as a data transfer agreement) when required in accordance with Section 12.1 (Confidentiality; Exceptions). Upon the transfer of such PII/Samples by Roche, Ionis shall use such PII/Samples for the sole purpose
of Developing and Commercializing the Product, and Ionis shall be responsible for the correct and lawful use of the PII/Sample in compliance with the applicable data protection laws, the informed consent forms and privacy notices
(including but not limited to potential re-consenting of the patients at Ionis’ costs if the legal basis for the processing of the patients’ data was their explicit consent).
|
| 11.7. |
Sublicensees. If this Agreement terminates for any reason, any Sublicensee granted a sublicense
by Roche to Develop or Commercialize Products will, from the effective date of such termination, automatically become a direct licensee of Ionis with respect to the rights sublicensed to the Sublicensee by Roche; so long as (i) such Sublicensee is not in breach of its sublicense agreement, (ii) such Sublicensee agrees in writing to comply with all of the terms of this Agreement to the extent applicable to the rights originally
sublicensed to it by Roche, and (iii) such Sublicensee agrees to pay directly to Ionis such Sublicensee’s payments under this Agreement to the extent applicable to the rights sublicensed to it by Roche. Roche agrees that it will confirm
clause (i) of the foregoing in writing at the request and for the benefit of Ionis and if requested, the Sublicensee.
|
|
Confidential
|
Execution Version
|
| 12.1. |
Confidentiality; Exceptions. Except to the extent expressly authorized by this Agreement or
otherwise agreed in writing, the Parties agree that, during the Agreement Term and for five years thereafter, the receiving Party (the “Receiving Party”) and its Affiliates will
keep confidential and will not publish or otherwise disclose or use for any purpose other than as provided for in this Agreement any confidential or proprietary information or materials, patentable or otherwise, in any form (written,
oral, photographic, electronic, magnetic, or otherwise) which is disclosed to it by the other Party (the “Disclosing Party”) or its Affiliates or otherwise received or accessed
by a Receiving Party in the course of performing its obligations or exercising its rights under this Agreement, including trade secrets, Know-How, inventions or discoveries, proprietary information, formulae, processes, techniques and
information relating to the past, present and future marketing, financial, and research and development activities of any product or potential product or useful technology of the Disclosing Party or its Affiliates and the pricing thereof
(collectively, “Confidential Information”). Without limiting the foregoing, Roche agrees that it will not disclose or use Ionis’ Confidential Information or the Licensed Know-How
for any purpose other than as provided in this Agreement. Roche has procedures in place to protect such information and will ensure it implements such procedures appropriately to prevent Ionis’ Confidential Information and the Licensed
Know-How from being disclosed or used for any purpose other than as provided in this Agreement. In addition to the confidentiality obligations set forth in this Agreement, in the event that, during
the implementation of the Agreement, one of the Parties processes any information that is protected by applicable data privacy laws, including without limitation, the Health Insurance Portability & Accountability Act of 1996 (HIPAA)
in the US, the Swiss Data Protection Act, the European Union General Data Protection Regulation 2016/679 (GDPR), the Personal Information Protection and Electronic Documents Canada (PIPEDA) in Canada, and any other applicable data
protection laws, the respective Party agrees to fully comply with such laws, as they may be applicable to the Party. If and as required by applicable data privacy laws, including but not limited to the EU GDPR, Swiss or United Kingdom
laws, the Parties shall execute additional data protection documentation, such as the Standard Contractual Clauses issued by the European Commission to cover any cross-border transfers of personal data. If a Party to this Agreement
becomes subject to an investigation by a data protection authority or any other competent authority in relation to the processing of personal data under this Agreement, then the Party affected will inform the other Party without any
undue delay unless the Party is not permitted to provide such information. The Parties will inform each other without any undue delay and provide each other with reasonable support in case of a Data Subject Request (as such term is
defined under the applicable data privacy law(s)) or in case of a Data Breach (as such term is defined under the applicable data privacy law(s)) that would potentially affect the other Party to this Agreement. If changes to the
applicable data privacy laws affect the compliance of data processing activities under this Agreement, then the Parties will negotiate in good faith adjustments to the data privacy language in this Agreement, or additional data privacy
documentation if and as required by applicable data privacy laws. The Parties warrant that they will implement and maintain adequate technical and organizational measures to ensure the integrity and security of personal data processed
under this Agreement.
|
| 12.2. |
Prior Confidentiality Agreement. The Non-Disclosure Agreement executed by Ionis and Roche on
November 11, 2021 (including any and all amendments thereto) (the “CDA”) will govern disclosures of Information (as defined in the CDA) between the Parties prior to the Effective
Date. All Confidential Information exchanged between the Parties on or after the Effective Date under this Agreement will be subject to the terms of this ARTICLE 12.
|
|
Confidential
|
Execution Version
|
| 12.3. |
Authorized Disclosure. Except as expressly provided otherwise in this Agreement, a Receiving
Party or its Affiliates may use and disclose to Third Parties Confidential Information of the Disclosing Party as follows: (i) solely in connection with the performance of its obligations or exercise of rights granted or reserved in this
Agreement under confidentiality provisions no less restrictive than those in this Agreement, provided, a Receiving Party may disclose Confidential Information to a governmental entity or agency
without requiring such entity or agency to enter into a confidentiality agreement; (ii) to the extent reasonably necessary to file or prosecute patent, copyright and trademark applications (subject to Section 12.4 (Press Release;
Publications; Disclosure of Agreement) below), complying with applicable governmental regulations, obtaining Approvals, conducting Pre-Clinical Studies or Clinical Studies, marketing a Product, or as otherwise required by Applicable Law,
regulation, rule or legal process (including the rules of the SEC and any stock exchange); provided, however, that if a Receiving Party or any of its Affiliates is required by law or regulation to
make any such disclosure of a Disclosing Party’s Confidential Information it will, except where impracticable for necessary disclosures, give reasonable advance notice to the Disclosing Party of such disclosure requirement and will use
its reasonable efforts to secure confidential treatment of such Confidential Information required to be disclosed; (iii) in communication with actual or potential lenders, investors, merger partners, acquirers, consultants, or
professional advisors on a need-to-know basis, in each case under confidentiality provisions no less restrictive than those of this Agreement; (iv) to the extent such disclosure is required to comply with existing expressly stated
contractual obligations owed to such Party’s or its Affiliates’ licensor with respect to any intellectual property licensed to the other Party under this Agreement; or (v) as mutually agreed to in writing by the Parties. Notwithstanding
the foregoing, if either Party concludes based on the reasonable opinion of counsel that a copy of this Agreement must be filed with the United States Securities and Exchange Commission or similar regulatory agency in a country other than
the United States, such Party will, within a reasonable time prior to any such filing (and to the extent possible at least [***] prior to any such filing), provide the other Party with a copy of this Agreement showing any provisions
hereof as to which such Party proposes to request confidential treatment, and the Parties shall coordinate with each other and will use good faith efforts to mutually agree on the redaction of certain provisions of this Agreement
(together with all exhibits and schedules) before filing such copy of this Agreement, provided that notwithstanding the foregoing, the filing Party shall retain final decision-making authority over the redactions to be made in its filed
copy of this Agreement.
|
| 12.4. |
Press Release; Publications; Disclosure of Agreement.
|
|
|
12.4.1. |
Public Announcements – Generally. Either Party may issue a press release announcing the
execution of this Agreement and disclosing selected key terms, subject to prior agreement of the Parties on the final draft of press release to be issued. Except to the extent required to comply with Applicable Law, regulation, rule or
legal process or as otherwise permitted in accordance with this Section 12.4 (Press Release; Publications; Disclosure of Agreement), each Party agrees not to issue any other press release or other public statement disclosing other
information relating to this Agreement or the terms of this Agreement or the transactions contemplated hereby without the prior written consent of the other Party, which consent will not be unreasonably withheld or delayed.
|
|
|
12.4.2. |
Use of Name. Except as set forth in Section 12.4.8 (Acknowledgement; Commercial
Materials), neither Party will use the other Party’s name in a press release or other publication without first obtaining the prior written consent of the Party to be named.
|
|
|
12.4.3. |
Notice of Significant Events. Each Party will notify (no later than [***] after the information
or results are obtained) the other Party of any significant event related to a Product (including any data, serious adverse event or regulatory advice or approval) so that the Parties may analyze the need to or desirability of publicly
disclosing or reporting such event. Notwithstanding Section 12.4.1 (Public Announcements – Generally) above, any press release or other similar public communication by either Party related to a Product’s efficacy or safety data
and/or results, will be submitted to the other Party for review and approval at least [***] in advance of such proposed public disclosure, which approval will not be unreasonably withheld or delayed.
|
|
Confidential
|
Execution Version
|
|
|
12.4.4. |
Press Releases. Roche will have the sole right, consistent with its practice with its other
compounds and products, to issue press releases, publish, present or otherwise disclose the progress and results regarding the Products to the public; provided, that with respect to any proposed
press release or other similar public communication by Roche disclosing regulatory discussions, the efficacy or safety data or results related to the Products or Roche’s sales projections, (i) Roche will submit such proposed communication
to Ionis for review at least [***] in advance of such proposed public disclosure, (ii) Ionis will have the right to review and recommend changes to such communication, and (iii) Roche will in good faith consider any changes that are
timely recommended by Ionis.
|
|
|
12.4.5. |
Scientific or Clinical Presentations. Regarding any proposed scientific publications related to
results from any Clinical Studies of a Product, the Parties agree to use Commercially Reasonable Efforts to control public scientific disclosures of such results to prevent any adverse effect of any premature public disclosure of such
results. The Parties will establish a procedure for publication review and each Party will first submit to the other Party through the JPC an early draft of all such publications or presentations, at least [***] prior to submission for
publication including to facilitate the publication of any summaries of Clinical Studies data and results as required on the clinical trial registry of each respective Party. Each Party will review such proposed publication to avoid the
unauthorized disclosure of a Party’s Confidential Information and to preserve the patentability of inventions arising from an R&D Plan. If, during such [***] period, the other Party informs such Party that its proposed publication
contains Confidential Information of the other Party, then such Party will delete such Confidential Information from its proposed publication. In addition, if during such [***] period, the other Party informs such Party that its proposed
publication discloses non-public inventions made by either Party in the course of the Development under this Agreement, or the public disclosure of such proposed publication may have a material adverse effect on any Patent Rights or
Know-How solely owned or Controlled by such other Party, then such Party will either (i) delay such proposed publication for up to [***] from the date of such Party’s objection, to permit the timely first filing of patent application(s),
or (ii) remove the identified disclosures prior to publication.
|
|
|
12.4.6. |
SEC Filings. Each Party will give the other Party a reasonable opportunity to review all
material filings with the SEC describing the terms of this Agreement prior to submission of such filings, and will give due consideration to any reasonable comments by the non-filing Party relating to such filing.
|
|
|
12.4.7. |
Subsequent Disclosure. Notwithstanding the foregoing, to the extent information regarding this
Agreement or a Product has already been publicly disclosed, either Party (or its Affiliates) may subsequently disclose the same information to the public without the consent of the other Party.
|
|
|
12.4.8. |
Acknowledgment; Commercial Materials. Each Party will acknowledge in any press release, public
presentation, publication or commercial marketing materials regarding the collaboration or a Product, the other Party’s role in discovering and Developing a Product or Discontinued Product, as applicable, that the Product is under license
from Ionis and otherwise acknowledge the contributions from the other Party, and each Party’s stock ticker symbol (e.g., Ionis: Nasdaq: IONS; Roche: SIX: RO, ROG; OTCQX: RHHBY). Ionis may include the Products (and identify Roche as
its partner for the Product) in Ionis’ drug pipeline. In addition, subject to Applicable Law, the words “Discovered by Ionis” will be included in Product communications, provided that Roche will
have final decision-making authority regarding the applicability of any legal and regulatory requirements for such acknowledgement.
|
|
Confidential
|
Execution Version
|
| 13.1. |
Dispute Resolution.
|
|
|
13.1.1. |
Escalation. If any dispute occurs under this Agreement (other than a dispute regarding the
construction, validity or enforcement of either Party’s Patent Rights, which disputes will be resolved pursuant to Section 13.2 (Governing Law; Jurisdiction; Venue; Service of Process)), then either Party may request in writing
that the dispute be referred for resolution to the Head of Roche Pharma Partnering and the Chief Executive Officer of Ionis (the “Executives”). Within [***] after such a request,
the Executives will meet in person at a mutually acceptable time and location or by means of telephone or video conference to negotiate a settlement of the dispute. Each Party’s JSC representatives may participate in such meeting if
desired. If the Executives fail to resolve the dispute within such [***] period, then the dispute will be referred to binding arbitration under Section 13.1.2 (Binding Arbitration).
|
|
|
13.1.2. |
Binding Arbitration. If a dispute subject to Section 13.1.1 (Escalation) is not resolved
pursuant to Section 13.1.1 (Escalation), except as otherwise set forth in this Agreement such dispute will be resolved through binding arbitration in accordance with this Section 13.1.2 (Binding Arbitration) and under the
Commercial Arbitration Rules of the [***] then in effect, including application of the “Expedited Procedures” (sections E-1, et al) of the Commercial Arbitration Rules of the [***]. The proceedings
and decisions of the arbitrator will be confidential, final and binding on the Parties, and judgment upon the award of such arbitrators may be entered in any court having jurisdiction thereof. The arbitration will take place in Boston,
Massachusetts USA and will be conducted by three (3) arbitrators. Each of Roche and Ionis shall appoint one (1) arbitrator within thirty (30) days after the notice that initiated the arbitration. These two (2) arbitrators shall in turn
appoint a third arbitrator who will be reasonably acceptable to the Parties and who will be appointed in accordance with [***] rules. Each arbitrator chosen hereunder will have educational training and industry experience sufficient to
demonstrate a reasonable level of scientific, financial, medical and industry knowledge relevant to the particular dispute.
|
| 13.2. |
Governing Law; Jurisdiction; Venue; Service of Process.
|
|
|
13.2.1. |
This Agreement and any dispute will be governed by and construed and enforced in accordance with the laws of [***], without reference to conflicts of laws principles.
|
|
|
13.2.2. |
Each Party hereby agrees that service of process: (a) made in any manner permitted by [***] law, or (b) made by overnight express courier service (signature required), prepaid, at its address specified
pursuant to Section 13.7 (Notices), will constitute good and valid service of process in any such action and (c) waives and agrees not to assert (by way of motion, as a defense, or otherwise) in any such action any claim that
service of process made in accordance with clause (a) or (b) does not constitute good and valid service of process.
|
|
Confidential
|
Execution Version
|
| 13.3. |
Remedies. Notwithstanding anything to the contrary in this Agreement, each Party will be
entitled to seek, in addition to any other right or remedy it may have, at law or in equity, a temporary restraining order or a preliminary injunction, without the posting of any bond or other security, enjoining or restraining the other
Party from any violation or threatened violation of this Agreement, and the Parties agree that in the event of a threatened or actual material breach of this Agreement injunctive relief would be appropriate. Neither Party may recover any
Losses relating to any matter arising under one provision of this Agreement to the extent that such Party has already recovered Losses with respect to such matter pursuant to other provisions of this Agreement (including recoveries under
Section 10.1 (Indemnification by Roche) or Section 10.2 (Indemnification by Ionis)). Except for the offsets and credits explicitly set forth in Section 7.4.3(f)(i) (Additional Ionis Core Intellectual Property) and Section
7.8 (Audits), neither Party will have the right to set-off any amount it is owed or believes it is owed against payments due or payable to the other Party under this Agreement.
|
| 13.4. |
Assignment and Successors. Neither this Agreement nor any obligation of a Party hereunder may be
assigned by either Party without the consent of the other, except that each Party may assign this Agreement and the rights, obligations and interests of such Party, in whole or in part, without the other Party’s consent, to any of its
Affiliates, to any purchaser of all or substantially all of its assets or all or substantially all of its assets to which this Agreement relates or to any successor corporation resulting from any merger, consolidation, share exchange or
other similar transaction; provided, if a Party transfers or assigns this Agreement to [***], then such transferring Party (or such Affiliate) (“Transferring Party”), will [***] any payment that the Transferring Party is obligated to pay to the non-transferring Party (“Non-Transferring Party”) under ARTICLE
7 for the taxes withheld such that the Non-Transferring Party receives [***]. In addition, Ionis may assign or transfer its rights to receive payments under this Agreement (but no liabilities), without Roche’s consent, to an
Affiliate or to a Third Party in connection with a payment factoring transaction. Any purported assignment or transfer made in contravention of this Section 13.4 (Assignment and Successors) will be null and void. To the extent
the Non-Transferring Party utilizes [***], the Non- Transferring Party will [***] to the Transferring Party [***]. To assist the Transferring Party in determining when [***], beginning with the first Annual tax return for the year in
which the Transferring Party [***], and each year thereafter (including, for clarity, all years in which the Non-Transferring Party utilizes [***], the Non-Transferring Party will provide the Transferring Party with the Non-Transferring
Party’s’ Annual tax returns (federal and state) and, in years in which the Non-Transferring Party utilizes [***], supporting documentation for such [***].
|
| 13.5. |
Change of Control. If Ionis undergoes a Change of Control, then Roche shall have the right at
any time after Handoff to disband the JSC and make unilateral decisions with respect to the R&D Plans, and the Development and Commercialization of Products with no obligation to seek input from Ionis or its successor, if applicable.
|
|
Confidential
|
Execution Version
|
| 13.6. |
Force Majeure. No Party will be held responsible to the other Party nor be deemed to be in
default under, or in breach of any provision of, this Agreement for failure or delay in performing any obligation of this Agreement when such failure or delay is due to force majeure, and without the fault or negligence of the Party so
failing or delaying. For purposes of this Agreement, force majeure means a cause beyond the reasonable control of a Party, which may include acts of God; acts, regulations, or laws of any government; war; terrorism; civil commotion; fire,
flood, earthquake, tornado, tsunami, explosion or storm; pandemic; epidemic and failure of public utilities or common carriers. In such event the Party so failing or delaying will immediately notify the other Party of such inability and
of the period for which such inability is expected to continue. The Party giving such notice will be excused from such of its obligations under this Agreement as it is thereby disabled from performing for so long as it is so disabled for
up to a maximum of ninety (90) days, after which time the Parties will negotiate in good faith any modifications of the terms of this Agreement that may be necessary to arrive at an equitable solution, unless the Party giving such notice
has set out a reasonable timeframe and plan to resolve the effects of such force majeure and executes such plan within such timeframe. To the extent possible, each Party will use reasonable efforts to minimize the duration of any force
majeure.
|
| 13.7. |
Notices. Any notice or request required or permitted to be given under or in connection with
this Agreement will be deemed to have been sufficiently given if in writing and personally delivered or sent by certified mail (return receipt requested), facsimile transmission (receipt verified), or overnight express courier service
(signature required), prepaid, to the Party for which such notice is intended, at the address set forth for such Party below:
|
| If to Ionis, addressed to: |
Ionis Pharmaceuticals, Inc.
2855 Gazelle Court
Carlsbad, CA 92010
Attention: Chief Executive Officer
|
|
|
with a copy to: |
Ionis Pharmaceuticals, Inc.
2855 Gazelle Court
Carlsbad, CA 92010
Attention: General Counsel
Email: [***]
|
|
|
If to Roche, addressed to: |
F. Hoffmann-La Roche Ltd
Grenzacherstrasse 124
4070 Basel, Switzerland
Attention: Corporate Legal Department
Fax: [***]
|
|
|
If to Roche, addressed to: | Hoffmann-La Roche Inc. 150 Clove Road, Suite 8
Little Falls, New Jersey 07424
Attention: Corporate Secretary
Fax: [***]
|
|
|
with a copy to: |
F. Hoffmann-La Roche Ltd
Grenzacherstrasse 124
4070 Basel, Switzerland
Attention: Alliance Manager
Fax: [***]
|
|
Confidential
|
Execution Version
|
| 13.8. |
Invoices. All invoices that are required or permitted hereunder shall be in writing and sent by
Ionis to Roche at the following address or any other address that Roche may later provide:
F. Hoffmann-La Roche AG
Kreditorenbuchhaltung
Grenzacherstrasse 124
CH - 4070 Basel
Upon Ionis’ request, Roche’s Alliance Manager will provide Ionis’ Alliance Manager with any additional information reasonably requested by Ionis to facilitate the prompt delivery of
invoices to Roche, including an email address for sending invoices.
|
| 13.9. |
Export Clause. Each Party acknowledges that the laws and regulations of the United States
restrict the export and re-export of commodities and technical data of United States origin. Each Party agrees that it will not export or re-export restricted commodities or the technical data of the other Party in any form without the
appropriate United States and foreign government licenses.
|
| 13.10. |
Waiver. Neither Party may waive or release any of its rights or interests in this Agreement
except in writing. The failure of either Party to assert a right hereunder or to insist upon compliance with any term or condition of this Agreement will not constitute a waiver of that right or excuse a similar subsequent failure to
perform any such term or condition. No waiver by either Party of any condition or term in any one or more instances will be construed as a continuing waiver or subsequent waiver of such condition or term or of another condition or term.
|
| 13.11. |
Severability. If any provision hereof should be held invalid, illegal or unenforceable in any
jurisdiction, the Parties will negotiate in good faith a valid, legal and enforceable substitute provision that most nearly reflects the original intent of the Parties and all other provisions hereof will remain in full force and effect
in such jurisdiction and will be liberally construed in order to carry out the intentions of the Parties hereto as nearly as may be possible. Such invalidity, illegality or unenforceability will not affect the validity, legality or
enforceability of such provision in any other jurisdiction.
|
| 13.12. |
Entire Agreement. This Agreement, together with the Schedules and Appendices hereto, sets forth
all the covenants, promises, agreements, warranties, representations, conditions and understandings between the Parties regarding the subject matter hereof and supersedes and terminates all prior agreements and understanding between the
Parties pertaining to the subject matter hereof. There are no covenants, promises, agreements, warranties, representations, conditions or understandings, either oral or written, between the Parties regarding the subject matter hereof
other than as set forth in this Agreement and the Schedules and Appendices hereto. No subsequent alteration, amendment, change or addition to this Agreement will be binding upon the Parties hereto unless reduced to writing and signed by
the respective authorized representatives of the Parties.
|
|
Confidential
|
Execution Version
|
| 13.13. |
Independent Contractors. Nothing herein will be construed to create any relationship of employer
and employee, agent and principal, partnership or joint venture between the Parties. Each Party is an independent contractor. Neither Party will assume, either directly or indirectly, any liability of or for the other Party. Neither Party
will have the authority to bind or obligate the other Party and neither Party will represent that it has such authority.
|
| 13.14. |
Interpretation. Except as otherwise explicitly specified to the contrary, (a) references to a
section, exhibit or schedule means a section of, or schedule or exhibit to this Agreement, unless another agreement is specified, (b) the word “including” (in its various forms) means “including without limitation,” (c) the words “will”
and “shall” have the same meaning, (d) references to a particular statute or regulation include all rules and regulations thereunder and any predecessor or successor statute, rules or regulation, in each case as amended or otherwise
modified from time to time, (e) words in the singular or plural form include the plural and singular form, respectively, (f) references to a particular Person include such Person’s successors and assigns to the extent not prohibited by
this Agreement, (g) unless otherwise specified, “$” is in reference to United States dollars, and (h) the headings contained in this Agreement, and in any Appendix or Schedule to this Agreement are for convenience only and will not in any
way affect the construction of or be taken into consideration in interpreting this Agreement.
|
| 13.15. |
Further Actions. Each Party will execute, acknowledge and deliver such further instruments, and
do all such other acts, as may be necessary or appropriate in order to carry out the expressly stated purposes and the clear intent of this Agreement.
|
| 13.16. |
Construction of Agreement. The terms and provisions of this Agreement represent the results of
negotiations between the Parties and their representatives, each of which has been represented by counsel of its own choosing, and neither of which has acted under duress or compulsion, whether legal, economic or otherwise. Accordingly,
the terms and provisions of this Agreement will be interpreted and construed in accordance with their usual and customary meanings, and each of the Parties hereto hereby waives the application in connection with the interpretation and
construction of this Agreement of any rule of law to the effect that ambiguous or conflicting terms or provisions contained in this Agreement will be interpreted or construed against the Party whose attorney prepared the executed draft or
any earlier draft of this Agreement.
|
| 13.17. |
Supremacy. In the event of any express conflict or inconsistency between this Agreement and any
Schedule or Appendix hereto, the terms of this Agreement will apply. The Parties understand and agree that the Schedules and Appendices hereto are not intended to be the final and complete embodiment of any terms or provisions of this
Agreement, and are to be updated from time to time during the Agreement Term, as appropriate and in accordance with the provisions of this Agreement.
|
|
Confidential
|
Execution Version
|
| 13.18. |
Counterparts. This Agreement may be executed in one or more counterparts, each of which shall
be deemed an original and all of which taken together shall be deemed to constitute one and the same agreement. The Parties agree that execution of this Agreement by e-Signatures or by exchanging executed signature pages in .pdf format
shall have the same legal force and effect as the exchange of original signatures. As used in this Section, “e-Signature” shall mean a signature that consists of one or more letters, characters, numbers or other symbols in digital form
incorporated in, attached to or associated with the electronic document, that (a) is unique to the person executing the signature; (b) the technology or process used to make the signature is under the sole control of the person making the
signature; (c) the technology or process can be used to identify the person using the technology or process; and (d) the electronic signature can be linked with an electronic document in such a way that it can be used to determine whether
the electronic document has been changed since the electronic signature was incorporated in, attached to or associated with the electronic document.
|
|
Confidential
|
Execution Version
|
|
F. Hoffmann-La Roche Ltd
|
||
|
By:
|
/s/ James Sabry | |
|
Name: James Sabry
|
||
|
Title: Global Head – Roche Pharma Partnering
Date: September 26, 2023
|
||
|
By:
|
/s/ Stephanie Harloff | |
|
Name: Stephanie Harloff
|
||
|
Title: Legal Counsel – Legal Pharma Partnering
|
||
| Date: September 26, 2023 | ||
|
Confidential
|
Execution Version
|
|
Hoffmann-La Roche Inc.
|
||
|
|
|
|
| By: | /s/ Gerald Bohm |
|
| Name: Gerald Bohm | ||
| Title: Principal Counsel Associate General Counsel | ||
| Date: September 26, 2023 | ||
|
Confidential
|
Execution Version
|
|
Ionis Pharmaceuticals, Inc.
|
||
|
|
|
|
| By: | /s/ Brett Monia |
|
| Name: Brett Monia | ||
| Title: Chief Executive Officer | ||
| Date: September 26, 2023 | ||
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
|
(a) |
was in the lawful knowledge and possession of the Receiving Party or its Affiliates prior to the time it was disclosed to, or learned by, the Receiving Party or its Affiliates, or was otherwise developed
independently by the Receiving Party or its Affiliates, as evidenced by written records kept in the ordinary course of business, or other documentary proof of actual use by the Receiving Party or its Affiliates;
|
|
|
(b) |
was generally available to the public or otherwise part of the public domain at the time of its disclosure to the Receiving Party or its Affiliates;
|
|
|
(c) |
became generally available to the public or otherwise part of the public domain after its disclosure and other than through any act or omission of the Receiving Party or its Affiliates in breach of this
Agreement; or
|
|
|
(d) |
was disclosed to the Receiving Party or its Affiliates, other than under an obligation of confidentiality, by a Third Party who had no obligation to the Disclosing Party or its Affiliates not to disclose such
information to others.
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
|
(i) |
to which Roche has granted a sublicense or license under any Licensed Technology or Roche Technology, as the case may be, in accordance with the terms of this Agreement;
|
|
|
(ii) |
that is not an Affiliate of Roche; and
|
|
|
(iii) |
that is consolidated within Roche’s externally published audited financial statements,
|
|
|
(iv) |
excluding Chugai unless Roche explicitly includes Chugai as an Affiliate pursuant to the “Affiliate” definition.
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
|
(i) |
the amount stated in Roche sales line of its externally published audited financial statements with respect to such Product for such period (excluding sales to any Sublicensee that are used for research or
Development or re-sold by such Sublicensee as sales under item (ii) below). This amount reflects the gross invoice price at which such Product was sold or otherwise disposed of (other than for use as clinical supplies or free samples) by
Roche/Genentech, its Affiliates and Group Sublicensees to Third Parties (excluding sales to any Sublicensee that are used for research or Development or re-sold by such Sublicensee as sales under item (ii) below) in such period reduced by
gross-to- net deductions, if not previously deducted from such invoiced amount, taken in accordance with the then currently used International Financial Reporting Standards (IFRS).
|
|
|
(a) |
credits, reserves or allowances granted for (w) damaged, outdated, returned, rejected, withdrawn or recalled Product, (x) wastage replacement and short-shipments, (y) billing errors and (z) indigent patient and similar programs (e.g.,
price capitation);
|
|
|
(b) |
governmental price reductions and government mandated rebates;
|
|
|
(c) |
chargebacks, including those granted to wholesalers, buying groups and retailers;
|
|
|
(d) |
customer rebates, including cash sales incentives for prompt payment, cash and volume discounts; and
|
|
|
(e) |
taxes, duties and any other governmental charges or levies imposed upon or measured by the import, export, use, manufacture or sale of a Product (excluding income or franchise taxes).
|
|
Confidential
|
Execution Version
|
|
|
(ii) |
Sublicensee (excluding Compulsory Sublicensee) sales amounts reported to Roche and its Affiliates in accordance with Sublicensee contractual terms and their then currently used accounting standards. For the purpose of clarity, any
Sublicensee sales as reported to Roche in accordance with Compulsory Sublicense agreements will be excluded from the Sales amount.
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
|
1. |
[***].
|
|
|
2. |
[***].
|
|
|
3. |
[***].
|
|
|
4. |
[***].
|
|
|
5. |
[***].
|
|
|
6. |
[***].
|
|
|
7. |
[***].
|
|
|
8. |
[***].
|
|
|
9. |
[***].
|
|
|
10. |
[***].
|
|
|
11. |
[***].
|
|
|
12. |
[***].
|
|
|
13. |
[***].
|
|
|
14. |
[***].
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
Confidential
|
Execution Version
|
|
Ionis Docket Number
|
Country/Treaty
|
Application/
Patent
Number
|
Grant Date
|
Title
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
Confidential
|
Execution Version
|
|
Ionis Docket Number
|
Country/Treaty
|
Application/
Patent
Number
|
Grant Date
|
Title
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
Confidential
|
Execution Version
|
|
Ionis Docket Number
|
Country/Treaty
|
Application/
Patent
Number
|
Grant Date
|
Title
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
Confidential
|
Execution Version
|
|
Ionis Docket Number
|
Country/Treaty
|
Application/
Patent
Number
|
Grant Date
|
Title
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
Confidential
|
Execution Version
|
|
Ionis Docket Number
|
Country/Treaty
|
Application/
Patent
Number
|
Grant Date
|
Title
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
Confidential
|
Execution Version
|
|
Ionis Docket Number
|
Country/Treaty
|
Application/
Patent
Number
|
Grant Date
|
Title
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
Confidential
|
Execution Version
|
|
Ionis Docket Number
|
Country
|
Application/
Patent
Number
|
Grant Date
|
Title
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
Confidential
|
Execution Version
|
|
Ionis Docket Number
|
Country
|
Application/
Patent
Number
|
Grant Date
|
Title
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
Confidential
|
Execution Version
|
|
Ionis Docket Number
|
Country
|
Application/
Patent
Number
|
Grant Date
|
Title
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
|
[***]
|
[***]
|
[***]
|
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
|
Confidential
|
Execution Version
|
|
Ionis File No.
|
Country
|
Status
|
Application
No.
|
Filing
Date
|
Title
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
[***]
|
|
Confidential
|
Execution Version
|
|
|
1. |
[***].
|
|
Confidential
|
Execution Version
|
| 1. | I have reviewed this Quarterly Report on Form 10-Q of Ionis Pharmaceuticals, Inc.; |
| 2. | Based on my knowledge, this quarterly report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this quarterly report; |
| 3. | Based on my knowledge, the condensed consolidated financial statements, and other financial information included in this quarterly report, fairly present in all material respects the financial condition, condensed consolidated results of operations and condensed consolidated cash flows of the registrant as of, and for, the periods presented in this quarterly report; |
| 4. | The registrant’s other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have: |
| a) | Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared; |
| b) | Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles; |
| c) | Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and |
| d) | Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and |
| 5. | The registrant’s other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of registrant’s board of directors (or persons performing the equivalent functions): |
| a) | All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information; and |
| b) | Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal control over financial reporting. |
Dated: November 2, 2023 |
|
/s/ BRETT P. MONIA |
|
Brett P. Monia, Ph.D. |
|
Chief Executive Officer |
| 1. | I have reviewed this Quarterly Report on Form 10-Q of Ionis Pharmaceuticals, Inc.; |
| 2. | Based on my knowledge, this quarterly report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this quarterly report; |
| 3. | Based on my knowledge, the condensed consolidated financial statements, and other financial information included in this quarterly report, fairly present in all material respects the financial condition, condensed consolidated results of operations and condensed consolidated cash flows of the registrant as of, and for, the periods presented in this quarterly report; |
| 4. | The registrant’s other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have: |
| a) | Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared; |
| b) | Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles; |
| c) | Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and |
| d) | Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and |
| 5. | The registrant’s other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of registrant’s board of directors (or persons performing the equivalent functions): |
| a) | All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information; and |
| b) | Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal control over financial reporting. |
Dated: November 2, 2023 |
|
/s/ ELIZABETH L. HOUGEN |
|
Elizabeth L. Hougen |
|
Chief Financial Officer |
| 1. | The Company’s Quarterly Report on Form 10-Q for the period ended September 30, 2023, to which this Certification is attached as Exhibit 32.1 (the “Periodic Report”), fully complies with the requirements of Section 13(a) or Section 15(d) of the Securities Exchange Act of 1934, as amended; and |
| 2. | The information contained in the Periodic Report fairly presents, in all material respects, the financial condition of the Company at the end of the period covered by the Periodic Report and the results of operations of the Company for the period covered by the Periodic Report. |
Dated: November 2, 2023 |
|||
/s/ BRETT P. MONIA |
/s/ ELIZABETH L. HOUGEN |
||
Brett P. Monia, Ph.D. |
Elizabeth L. Hougen |
||
Chief Executive Officer |
Chief Financial Officer |